Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) reviewed the safety of alkyl polyethylene glycol (PEG) sulfosuccinates, which function in cosmetics mostly as surfactants/cleansing agents. Although these ingredients may cause ocular and skin irritation, dermal penetration is unlikely because of the substantial polarity and molecular size of these ingredients. The Panel considered the negative oral carcinogenicity and reproductive and developmental toxicity data on chemically related laureths (PEG lauryl ethers) and negative repeated dose toxicity and skin sensitization data on disodium laureth sulfosuccinate supported the safety of these alkyl PEG sulfosuccinates in cosmetic products, but. The CIR Expert Panel concluded that the alkyl PEG sulfosuccinates are safe in the present practices of use and concentration when formulated to be nonirritating.
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook, 1 these 18 mostly function as surfactants-cleansing agents (Table 1). The ingredients included in this report are
Definitions, Functions, and Structures of the Sulfosuccinate Ingredients. 1

Abbreviation: CAS, Chemical Abstracts Service.
disodium laureth sulfosuccinate, disodium laureth-6 sulfosuccinate, disodium laureth-9 sulfosuccinate, disodium laureth-12 sulfosuccinate, disodium deceth-5 sulfosuccinate, disodium deceth-6 sulfosuccinate, magnesium laureth-3 sulfosuccinate, disodium C12-14 pareth-1 sulfosuccinate, disodium C12-14 pareth-2 sulfosuccinate, disodium C12-15 pareth sulfosuccinate, disodium coceth-3 sulfosuccinate, disodium laneth-5 sulfosuccinate, disodium C12-14 sec-pareth-3 sulfosuccinate, disodium C12-14 sec-pareth-5 sulfosuccinate, disodium C12-14 sec-pareth-7 sulfosuccinate, disodium C12-14 sec-pareth-9 sulfosuccinate, disodium C12-14 sec-pareth-12 sulfosuccinate, and disodium oleth-3 sulfosuccinate
Data from previous safety assessments on alkyl polyethylene glycol (PEG) ethers, 2 sodium laureth sulfate and related salts of sulfated ethoxylated alcohols, 3 and dioctyl sodium sulfosuccinate (diethylhexyl sodium sulfosuccinate, current International Nomenclature of Cosmetic Ingredient [INCI] name) 4 are included in this review. Data on laureths (PEG lauryl ethers) are included to fill gaps in the data for carcinogenicity and reproductive and developmental toxicity. These data are relevant because the first-level metabolites of alkyl PEG sulfosuccinates would likely include the corresponding alkyl PEG ethers; for example, magnesium laureth-3 sulfosuccinate may be metabolized to laureth-3 (PEG-3 lauryl ether) and sulfosuccinic acid. Alkyl PEG ethers (other than laureths), sodium laureth sulfate, and diethylhexyl sodium sulfosuccinate from the previous safety assessments are included because of structural similarities to the alkyl PEG sulfosuccinates included in this safety assessment.
Chemistry
Definition and Structure
The definitions, structures, and functions of the anionic surfactants reviewed in this safety assessment are included in Table 1. 1 The ingredients in this review share a sulfo-substituted, succinic acid core. Accordingly, the salts of these materials are sulfosuccinates. These ingredients are also the salts of alkyl PEG, monoesters of sulfosuccinic acid, and are collectively referred to as alkyl PEG sulfosuccinates, even though none of the INCI names includes PEG. 5 For example, disodium laureth sulfosuccinate consists of a 12-carbon alkyl chain (lauryl), connected to the sulfosuccinate core via a PEG chain, wherein the average number of ethoxy repeat units (n) is between 1 and 4 (ie, laureth-1 through laureth-4; Figure 1).
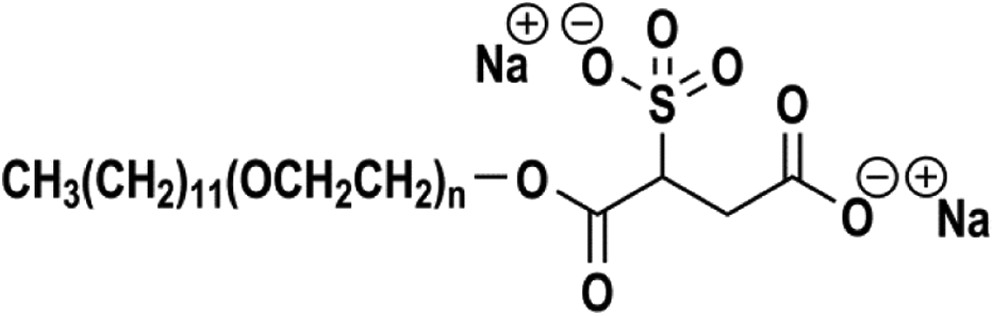
Disodium laureth sulfosuccinate.
Chemical and Physical Properties
Sulfosuccinate monoesters contain a hydrophobic end that consists of a fatty alcohol chain. 5 The chain length and degree of saturation of the fatty acid may vary this hydrophobicity. The level of hydrophobicity imparted by the fatty alcohol is also affected by the different degrees of ethoxylation of the PEG chain. For instance, monoesters based on linear fatty alcohols are only partially soluble in water. Those based on fatty alcohol ethoxylates have greater water solubility. Water solubility is also increased when the structure contains branched chains. Solubility in less polar solvents, such as isopropanol and 1,2-propylene glycol, is considered more difficult to achieve. Due to the ester linkage, these sulfosuccinate ingredients are sensitive to hydrolysis, especially under acidic conditions. Properties of sulfosuccinate ingredients (trade name materials included) are found in Table 2. Tables 3 and 4 contain specifications/actual composition data for disodium laureth sulfosuccinate trade name mixtures tested at various concentrations of disodium laureth sulfosuccinate in studies summarized later in the Toxicology section of this report.
Properties of Sulfosuccinate Ingredients.
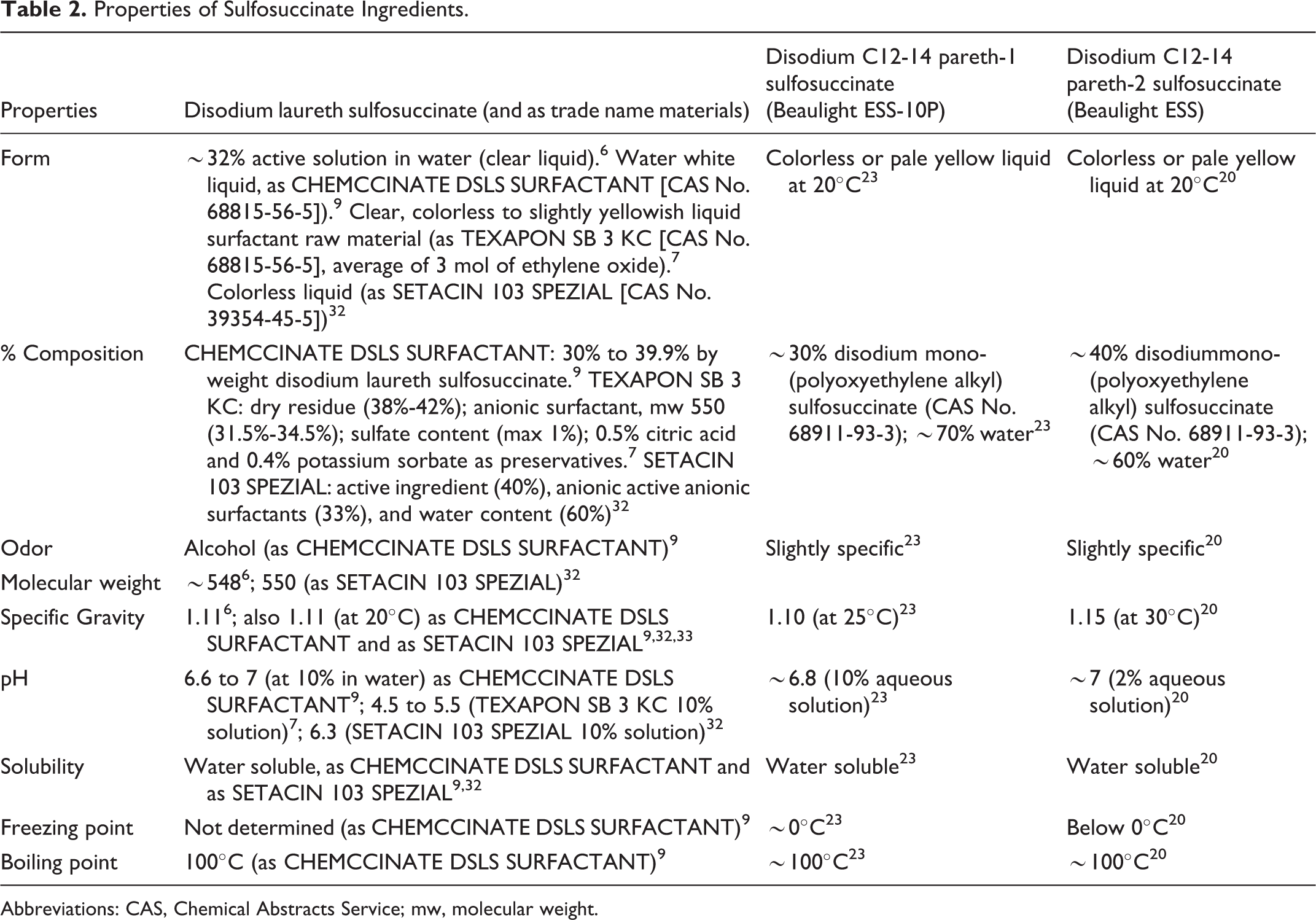
Abbreviations: CAS, Chemical Abstracts Service; mw, molecular weight.
Specifications for a Disodium Laureth Sulfosuccinate (16.8% Active)/Sodium Lauryl Sulfoacetate (7.2% Active) Trade Name Mixture 34 .

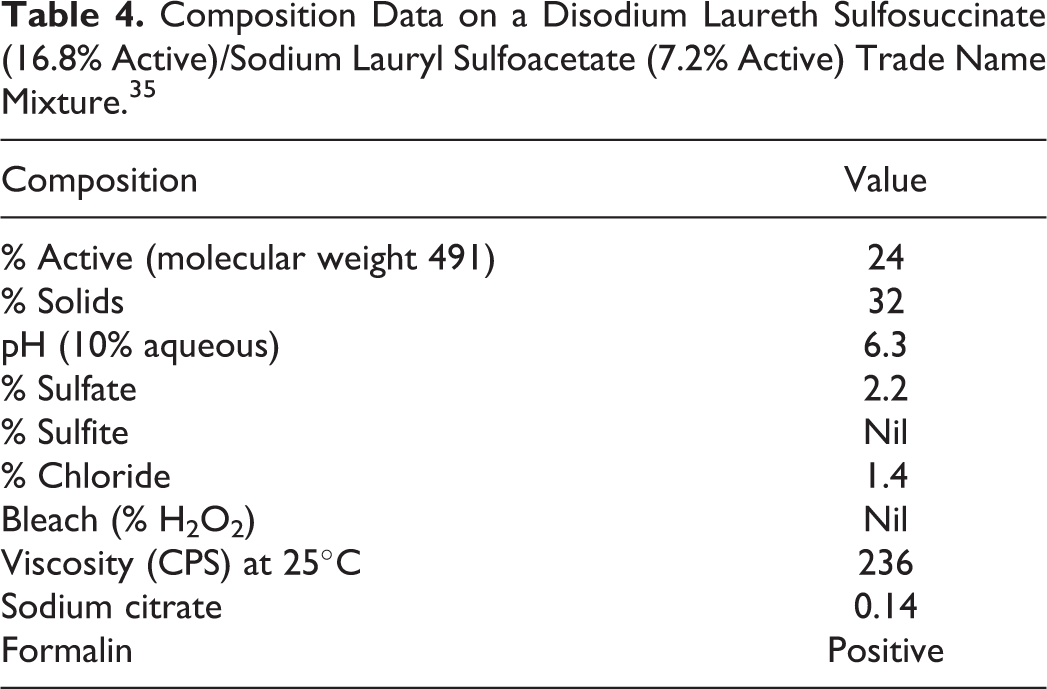
Composition Data on a Disodium Laureth Sulfosuccinate (16.8% Active)/Sodium Lauryl Sulfoacetate (7.2% Active) Trade Name Mixture. 35
Methods of Manufacture
The synthesis of these ingredients occurs according to a 2-step procedure. 5 In the first step, maleic anhydride is reacted with an ethoxylated fatty alcohol. The second step involves sulfonation of the resulting maleic ester. For example, in the production of disodium laureth sulfosuccinate, a monoester is formed by reacting the ethoxylated alcohol (eg, laureth-2) with maleic anhydride. 6 The monoester is then reacted with sodium bisulfite to form the sulfosuccinate. 7,8
Impurities
According to one report, disodium laureth sulfosuccinate contained the following impurities/by-products: residual sodium sulfite (<0.1%), residual sodium sulfate (1%-3%), residual laureth-2 (1%-5%), and 1,4-dioxane by-product (<10 ppm). 6 Another source indicates that a disodium laureth sulfosuccinate trade name material (active ingredient [40%]; anionic active anionic surfactants [33%]) contains: <1 ppm 1,4-dioxane and <1 ppm ethylene oxide (if presence of either is technically unavoidable in good manufacturing practice); <1 ppm residual monomers; and heavy metals, Pb (<10 ppm), Ni (<10 ppm), Cd (<1 ppm), As (<1 ppm), Sb (<1 ppm), and Hg (<0.2 ppm). 8 A third source indicates that disodium laureth sulfosuccinate contains formaldehyde at a maximum level of 0.056% and 1,4-dioxane at a maximum level of 0.001%. 9
Use
Cosmetic
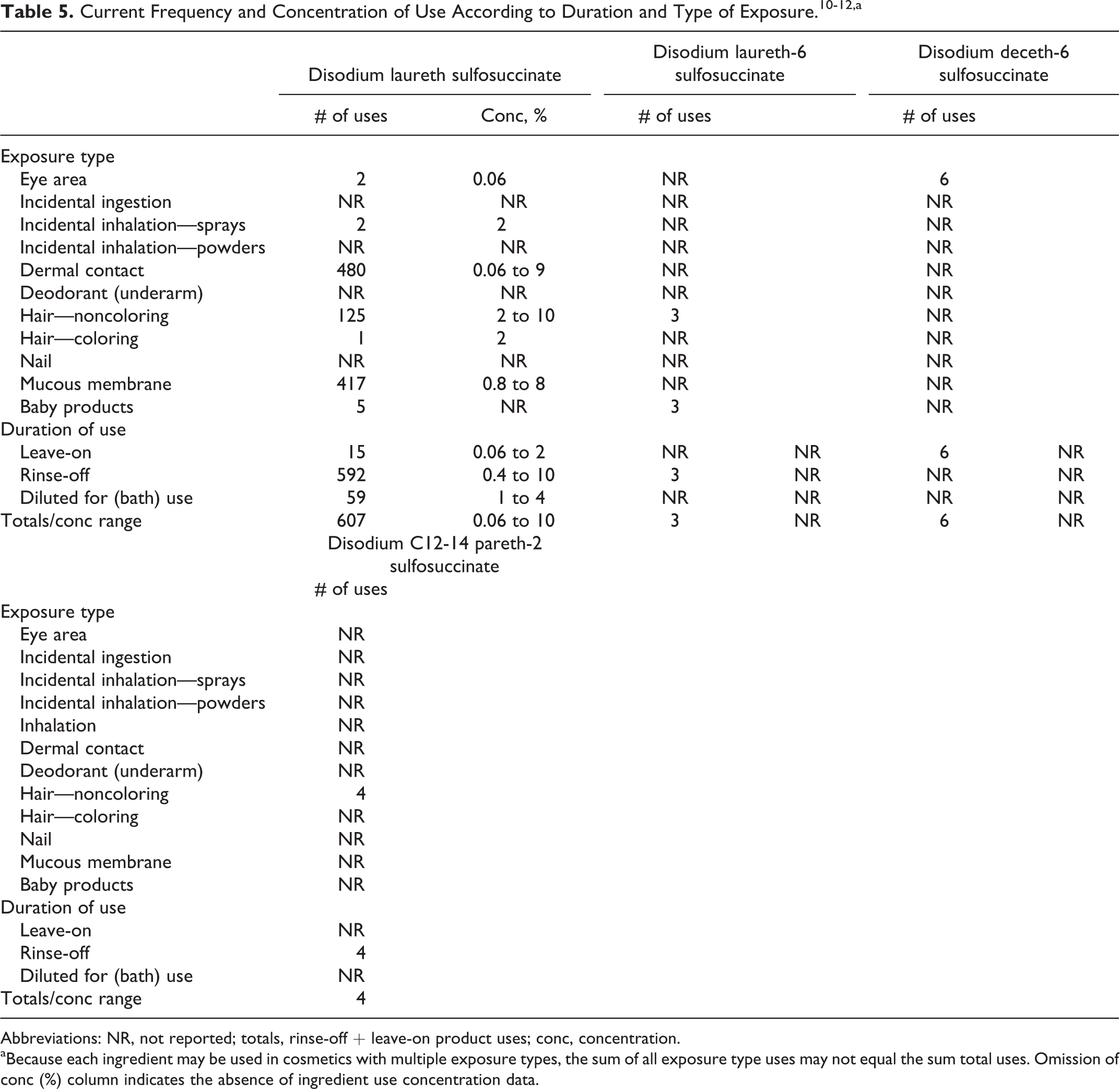
According to information supplied to the Food and Drug Administration by industry as part of the Voluntary Cosmetic Registration Program (VCRP), disodium laureth sulfosuccinate, disodium laureth-6 sulfosuccinate, disodium deceth-6 sulfosuccinate, and disodium C12-14 pareth-2 sulfosuccinate are being used in personal care products. 10 The use data are summarized in Table 5. A survey of ingredient use concentrations that was conducted by the Personal Care Products Council (Council) in 2011 (Table 5) provided use concentrations for disodium laureth sulfosuccinate (0.06%-10%). 11 The highest use concentration of this ingredient was in shampoos (noncoloring). There was no use concentration data reported for magnesium laureth-3 sulfosuccinate in a 2011 Council survey. 12 No uses of the remaining ingredients reviewed in this safety assessment were reported in the VCRP database or in the Council survey.
Abbreviations: NR, not reported; totals, rinse-off + leave-on product uses; conc, concentration.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses. Omission of conc (%) column indicates the absence of ingredient use concentration data.
Cosmetic products containing the ingredients reported as being used may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Disodium laureth sulfosuccinate is used in hair color sprays at a maximum reported concentration of 2% and could be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 13,14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, able to enter the lungs) to any appreciable amount. 15,16
Noncosmetic
Reportedly, sulfosuccinates are used to improve the wetting and spreading characteristics of water-soluble pesticide sprays and are found in liquid fertilizer, insecticides, fungicides, and herbicides. 5
Toxicokinetics
Studies on the toxicokinetics of sulfosuccinate ingredients in this safety assessment, following oral, dermal, or inhalation exposure, were not found in the published literature.
Toxicological Studies
Acute Oral Toxicity
Disodium laureth sulfosuccinate
According to a material safety data sheet (MSDS) on disodium laureth sulfosuccinate (30%-39.9% by weight), the acute oral median lethal dose (LD50) is > 10 000 mg/kg (rats), based on data on components or similar materials. Additional details were not provided. 9
In an acute oral toxicity test involving rats, the LD50 for a disodium laureth sulfosuccinate trade name material (active ingredient [40%] and active anionic surfactants [33%]) was >3000 mg/kg. 8 Additional study details were not provided.
Groups of 5 male and 5 female NMRI mice (∼6-8 weeks old) were used to evaluate the genotoxicity of disodium laureth sulfosuccinate in a micronucleus test. 17 Test animals received a single oral dose of the test substance (dose = 2000 mg/kg body weight) in distilled water and were monitored for signs of acute toxicity, up to 48 hours postdosing. None of the animals died, and there was no evidence of clinical signs.
The acute oral toxicity of disodium laureth sulfosuccinate (trade name material, 40% active anionic surfactants) was evaluated using fasted, young adult male albino rats (number not stated). 18 After a single dose of the test substance (dose volume = 10 mL/kg) was administered by gavage, no adverse effects were observed. An LD50 of >2000 mg/kg was reported.
The acute oral toxicity of a trade name mixture containing 24% active anionic surfactants was evaluated using 10 Sprague-Dawley rats (5 males and 5 females; ages not stated). 19 Data on the composition of this material are included in Table 4. The total active anionic surfactants (disodium laureth sulfosuccinate plus sodium lauryl sulfoacetate) in this mixture was 24%. Of this 24%, 70% (16.8% of the total mixture) was disodium laureth sulfosuccinate and 30% (7.2% of the total mixture) was sodium lauryl sulfoacetate. Each rat received a 5000-mg/kg oral dose (gavage) of the test substance, and the animals were observed for 15 days. Because the concentration of active anionic surfactants in the test material was 16.8%, each rat received an 840-mg/kg dose of disodium laureth sulfosuccinate (0.168 × 5000 = 840 mg/kg). All of the female rats died within 24 hours of dose administration, but all male rats survived to the end of the study. The following signs were observed in all rats: decreased activity, diarrhea, labored breathing, and an unsteady gait. Gross necropsy findings in the female rats included distended/gas-filled stomach, red stomach mucosa and mucosal erosion of the stomach (with rugae absent), red intestinal mucosa, and red substance in the intestines. The trade name material containing 16.8% disodium laureth sulfosuccinate was classified as a toxic substance.
Disodium C12-14 pareth-2 sulfosuccinate
Similarly, the acute oral LD50 of disodium C12-14 pareth-2 sulfosuccinate (∼40% disodium mono-(polyoxyethylene alkyl) sulfosuccinate in trade name material) is 3490 mg/kg (rats). 20 Additional details were not provided.
Acute Dermal Toxicity
Disodium laureth sulfosuccinate
Disodium laureth sulfosuccinate (30%-39.9% by weight) has an acute dermal LD50 of >2000 mg/kg in rabbits, based on data on components or similar materials. Additional details were not provided. 9
Repeated Dose Oral Toxicity
Disodium laureth sulfosuccinate
The repeated dose oral toxicity of a disodium laureth sulfosuccinate trade name material (active ingredient [40%] and active anionic surfactants [33%]) was evaluated using groups of Sprague-Dawley Crl: CD (SD) BR rats. 21 The control (deionized water) and 1000 mg/kg/d dose groups each contained 10 males and 10 females, and the 62.5 and 250 mg/kg dose groups each contained 5 males and 5 females. The test material was administered (dose volume = 10 mL/kg) 7 days/wk for 4 consecutive weeks. At the end of the dosing period, some of the animals were observed during a 4-week recovery period and the remaining animals were killed for pathological investigations.
There were no treatment-related deaths or changes, based on ophthalmological examinations and evaluations for clinical signs, body weight changes, or food consumption. In the highest dose group (1000 mg/kg/d), treatment-related increases in alkaline phosphatase (moderate) and in serum glutamic pyruvic transaminase (SGPT) (slight) were observed in both sexes, and there was a trend toward increased protein in the urine in males of this group. These changes were not observed at the end of the recovery period. Mild reversible changes in the liver (increased weight and hypertrophy) were also observed in the highest dose group. The no observed efffect level (NOEL) was considered to be 250 mg/kg/d. 21
In another study, groups of Sprague-Dawley CD, SPF-quality rats (10 males and 10 females/group) received oral doses of disodium laureth sulfosuccinate (trade name material, composition not stated) daily for 28 days. 18 Three groups received doses of 100, 300, and 1000 mg/kg bw/d, and the fourth group served as the untreated control group. Additionally, a recovery group consisting of 5 males and 5 females was used to determine the reversibility of possible test substance-related findings. None of the animals died and, compared to the control group, there was no deviation in body weight development in any of the 3 dose groups.
The following test substance-related findings, all in the highest dose group, were reported: stimulation of propulsion (swallowing and peristalsis) and salivation, alterations in hematological parameters and in clinical chemistry (ie, alterations in alanine aminotransferase values), significantly increased liver weight, and ulceration and edema of the forestomach mucosa (local irritation effects) at macroscopic examination. Effects on the forestomach mucosa were not observed after a 34-day recovery period. Other findings included small cellular infiltrations (no further details) in 100 and 300 mg/kg dose groups. In this study, the systemic no observed adverse effect level (NOAEL) was 300 mg/kg body weight . Additionally, a no-observed adverse effect concentration of <1% for local compatibility was deduced. It was stated that the labeling of this disodium laureth sulfosuccinate trade name material for possible toxic effects after chronic exposure is not necessary. 18
Ocular Irritation
Disodium laureth sulfosuccinate
The ocular irritation potential of diluted disodium laureth sulfosuccinate (25% active anionic surfactants in a trade name material) was evaluated according to Organization for Economic Cooperation and Development (OECD) Guideline No. 405 using 4 rabbits of the Kleinrusse Chbb: HM strain. 18 The test substance (0.1 mL) was instilled once into 1 eye of each animal, and untreated eyes served as controls. Mean ocular irritation scores (24, 48, and 72 hours) were as follows: 0.9 (cornea), 2.5 (conjunctival erythema), 1.1 (conjunctival edema), and 0 (iris). At the end of the observation period, slight conjunctival erythema (score = 1) and slight corneal opacity (score = 1) persisted in 1 rabbit. It was concluded that “the test material has to be classified and labeled to pose a risk of serious damage to the eyes.”
According to an MSDS on disodium laureth sulfosuccinate (30%-39.9% by weight), this chemical substance is considered a moderate to strong eye irritant based on data on components or similar materials. 9 Additional details were not provided.
The ocular irritation potential of a trade name mixture containing 10% active anionic surfactants was evaluated using 6 New Zealand white, young adult rabbits 22 Data on the composition of this material are included in Table 4. The total active anionic surfactants (disodium laureth sulfosuccinate plus sodium lauryl sulfoacetate) in this mixture is 24%. Of this 24%, 70% (16.8% of the total mixture) is disodium laureth sulfosuccinate and 30% (7.2% of the total mixture) is sodium lauryl sulfoacetate. Because the trade mixture evaluated contained 10% active anionic surfactants, the concentration of disodium laureth sulfosuccinate tested was 1.68% (0.10 × 0.168 = 1.68%). The undiluted test substance (0.1 mL) was instilled into the right eye of each rabbit. Contralateral eyes served as controls. Ocular irritation reactions were scored according to the method of Draize at 24-, 48-, and 72-hour postinstillation. A positive ocular response was observed in all rabbits tested, and the trade name mixture was classified as a primary ocular irritant.
Disodium C12-14 pareth-1 sulfosuccinate
According to an MSDS on disodium C12-14 pareth-1 sulfosuccinate (∼30% disodium mono-(polyoxyethylene alkyl) sulfosuccinate in trade name material), a 3% solution in physiological saline of a similar chemical is a minimal ocular irritant (unrinsed eyes) and a nonirritant (rinsed eyes) in guinea pigs. 23 Additional details were not provided.
Disodium C12-14 pareth-2 sulfosuccinate
Similarly, information in an MSDS on disodium C12-14 pareth-2 sulfosuccinate (∼40% disodium mono-(polyoxyethylene alkyl) sulfosuccinate in trade name material) indicates that a 3% solution of this material in physiological saline is a minimal ocular irritant (unrinsed eyes) and that a 3% solution in physiological saline of a similar chemical is a nonirritant (rinsed eyes) in guinea pigs. 20 Additional details were not provided.
Irritation and Sensitization
Disodium laureth sulfosuccinate
Nonhuman studies
A disodium laureth sulfosuccinate trade name material (active ingredient [40%] and active anionic surfactants [33%]) was evaluated in skin irritation tests involving rabbits. 8 At a concentration of 3% active anionic surfactants (effective concentration = 1.2% active ingredient; procedure not stated), the test substance was nonirritating. In the Duhring chamber test, the test substance was nonirritating at a concentration of 2% active anionic surfactants (effective concentration = 0.40 × 0.02 = 0.8% active ingredient). Details relating to either test procedure were not provided. 18 The same test material did not induce mucous membrane irritation in rabbits when tested at a concentration of 3% active anionic surfactants (effective concentration = 0.40 × 0.03 = 1.2% active ingredient). 8 Details relating to the test procedure were not provided.
The skin sensitization potential of the same disodium laureth sulfosuccinate trade name material (active ingredient [40%] and active anionic surfactants [33%]) was evaluated in the maximization test using 2 groups of 20 guinea pigs of the Pirbright White strain. 8 One of the groups served as the control group. Test concentrations administered during induction included 10% disodium laureth sulfosuccinate (4% active anionic surfactants; effective concentration = 1.6% active ingredient) in physiological saline (intradermal induction) and 5% (2% active anionic surfactants; effective concentration = 0.8% active ingredient) in physiological saline (dermal induction). The challenge concentration was 2% active anionic surfactants (effective concentration = 0.8% active ingredient), and reactions were scored at 24 and 48 hours postexposure. None of the animals had signs of primary skin irritation or sensitization, and the test substance was considered nonsensitizing.
The skin irritation potential of a disodium laureth sulfosuccinate trade name material (25% active anionic surfactants) was evaluated using 5 albino rabbits according to OECD guideline No. 404. 18 The test substance was applied to 1 flank (shave dorsal skin) of each animal. An untreated area served as the control. The contact time under occlusive conditions was 4 hours. Skin reactions were described as weak, with scores of 0.2 for erythema and 0 for edema at 24, 48, and 72 hours.
A skin sensitization study on the same disodium laureth sulfosuccinate trade name material (25% active anionic surfactants), performed according to the Magnusson-Kligman method, involved guinea pigs of the Pirbright Hoe: DHPK strain. 18 The number of animals tested was not stated. Based on the results of a preliminary study on a 10% dilution of the test substance in deionized water, a 5% dilution of the test substance (effective concentration = 1.6% active anionic surfactants) was applied during induction (intracutaneous and epicutaneous; 48 hours occlusive conditions) and during the challenge (epicutaneous) phase. Following both induction procedures, the animals were challenged with the test substance under occlusive conditions for 24 hours. Challenge application did not result in any signs of adverse dermal reactions.
Ten guinea pigs per sex were initially injected with 10% disodium laureth sulfosuccinate in water (0.05 mL), followed by injection with 10% disodium laureth sulfosuccinate in Freund’s Complete Adjuvant (FCA, 0.05 mL), and, then, FCA (0.05 mL). 24 After a 7-day nontreatment period, the animals were challenged dermally with 5% disodium laureth sulfosuccinate in water and then rechallenged with 5% disodium laureth sulfosuccinate in water. Control animals were treated with water only. Skin sensitization was not observed in any of the animals tested with disodium laureth sulfosuccinate or water.
According to an MSDS on disodium laureth sulfosuccinate (30%-39.9% by weight), this chemical substance is considered a severe skin irritant, based on data on components or similar materials. Additional details were not provided. 9
Disodium C12-14 pareth-1 sulfosuccinate
According to an MSDS on a disodium C12-14 pareth-1 sulfosuccinate trade name material (∼30% disodium mono-(polyoxyethylene alkyl) sulfosuccinate), a 10% aqueous solution of a similar chemical is neither a primary nor cumulative skin irritant in guinea pigs. 23 Additional details were not provided.
Disodium C12-14 pareth-2 sulfosuccinate
Information in an MSDS on a disodium C12-14 pareth-2 sulfosuccinate trade name material (∼40% disodium mono-(polyoxyethylene alkyl) sulfosuccinate) indicates that a 10% aqueous solution of this chemical is neither a primary nor a cumulative skin irritant in guinea pigs. 20 Additional details were not provided.
Human Studies
The skin irritation potential of disodium laureth sulfosuccinate was evaluated using 12 healthy participants (between ages of 22 and 64 years). 25 The test substance was diluted in a citrate buffer (final pH = 6 ± 0.5). Finn chambers (12 mm) containing 10% disodium laureth sulfosuccinate (50 µL) were applied to the left volar forearm and removed after 48 hours. Citrate buffer (10 mmol/L) served as the control. Sites were examined 1 hour after patch removal on day 1 and after 24 hours on day 2. Transepidermal water loss, cutaneous blood flow, and skin capacitance were also measured. Erythema (mild irritation) was observed on day 1. Application of the citrate buffer control also resulted in erythema. Compared to the control, transepidermal water loss was significantly elevated in day 2 after test substance application. The test substance did not induce a significant increase in cutaneous blood flow but caused a decrease in skin capacitance.
Patch tests were performed to evaluate the role of preexisting dermatitis in the response to irritants. 26 The study involved 40 healthy participants and 480 patients with the following types of skin disease: active atopic dermatitis (n = 40), psoriasis (n = 57), eczema (n = 124), urticaria (n = 79), and pruritus (n = 40). The 6 groups (males and females; mean age range: 18-55) were patch tested with 5% and 10% aqueous solutions of disodium laureth sulfosuccinate (volume = 17 µL). Patch tests were applied on both sides of the upper back for 48 hours using Al-test on Fixomul. Reactions were scored 1 hour after removal of the strips. For patients with urticaria, 2 additional strips were applied to both sides of the back and then removed 30 minutes later. There were no positive reactions to either test concentration of disodium laureth sulfosuccinate in healthy participants or patients with preexisting dermatitis.
The skin irritation potential of a trade name mixture containing 24% active anionic surfactants was evaluated in a 14-day cumulative irritation test using 28 patients (ages not stated). 27 Disodium laureth sulfosuccinate comprised 70% of the active anionic surfactants; thus, the effective concentration of disodium laureth sulfosuccinate in the trade name mixture was 16.8%. Sodium lauryl sulfoacetate, also present in the mixture, was diluted (1% dilution) to a test concentration of 0.168% disodium laureth sulfosuccinate in the trade mixture. The test concentration (100 µL) was applied repeatedly to estimate the mean number of days of continuous exposure that would produce a clinical irritation grade of 2. The positive control (1% SLS) was similarly applied. The mean number of days of continuous exposure to the 0.168% disodium laureth sulfosuccinate trade mixture that produced a clinical irritation grade of 2 was 9.86 days (3.93 days for positive control). A cumulative irritation score of 163 (based on N = 10) was reported for this trade mixture (score of 346 for positive control), classifying it as possibly a mild irritant during normal use. The positive control was classified as an experimental irritant.
The skin irritation and sensitization potential of a mixture containing 10% active anionic surfactants was evaluated in a repeated insult patch test using 51 participant (males and females; 19-65 years old). 28 Data on the composition of this material are included in Table 3. The total active anionic surfactants (disodium laureth sulfosuccinate plus sodium lauryl sulfoacetate in this mixture) is 24%, 70% (16.8% of the total mixture) of which is disodium laureth sulfosuccinate and 30% (7.2% of the total mixture) is sodium lauryl sulfoacetate. The concentration of disodium laureth sulfosuccinate tested in the 10% active anionic surfactants was 1.68% (0.1 × 16.8% = 1.68%). During induction, an occlusive patch containing the test material (0.2 mL) was applied (24 hours) repeatedly to the back of each participant for a total of 9 applications. After a 10- to 21-day nontreatment period, a challenge patch was applied to a new test site and reactions were scored at 24 and 48 hours postapplication. Barely perceptible erythema (1 participant) and barely perceptible to moderate cumulative irritant reactions (3 participants) were observed during induction. Reactions were not observed during the challenge phase. It was concluded that the trade name mixture containing 1.68% disodium laureth sulfosuccinate did not induce clinically significant irritation or any evidence of allergic contact dermatitis.
Disodium pareths sulfosuccinates
The following skin irritation study is included because the Chemical Abstracts Service number 68115-56-5 for the chemical tested, poly(oxy-1,2-ethanediyl), alpha-(3-carboxy-1-oxosulfopropyl)-omega-hydroxy-, C10-C16 alkyl ethers, disodium salts, is generic for any disodium C10-16 alkyl laureth sulfosuccinate. It contains disodium sulfosuccinate and other components (eg, sodium lauryl sulfoacetate) that differ from disodium laureth sulfosuccinate only by modest variations in chain length. The test article is also identified as a 15% trade name material. In this study, 6 New Zealand white rabbits each received a single dermal application of the 15% trade name material (0.5 mL) at two 2-cm2 sites, abraded and intact, on opposite sides. Each site was covered with an occlusive patch (2 cm2) for 24 hours, and reactions were scored at 24 and 72 hours. The test substance was not classified as a dermal irritant under the conditions of this test (primary irritation index = 3.15). 29
Comedogenicity
The comedogenicity of a trade name mixture containing 24% active anionic surfactants was evaluated using 3 young adult New Zealand white rabbits (1 male and 2 females; ages not stated). 30 Data on the composition of this material are included in Table 3. The total active anionic surfactants (disodium laureth sulfosuccinate plus sodium lauryl sulfoacetate) in this mixture is 24%. Of this 24%, 70% (16.8% of the total mixture) is disodium laureth sulfosuccinate and 30% (7.2% of the total mixture) is sodium lauryl sulfoacetate. The test substance (undiluted; volume not stated) was applied to the left ear of each rabbit for 3 consecutive weeks (5 consecutive days/wk). Untreated right ears served as controls. At the end of the dosing period, control and treated ears were excised and subjected to microscopic examination for comedone formation. Neither follicular hyperkeratosis nor comedone formation was observed during week 1. However, hyperkeratosis and dry, flaky skin were observed on all treated ears from week 2 to the end of the study. Reactions were not observed on untreated ears throughout the study. Acanthotic thickening (hyperplasia) of the epithelium, mild hyperkeratosis, and mild acute inflammatory infiltrates in the dermal layers were observed at microscopic examination of treated ears. However, there was no evidence of comedone formation. Based on microscopic findings, the trade name mixture containing 16.8% disodium laureth sulfosuccinate received a comedogenic score of 1, defined as an increase in visible hyperkeratosis without comedone formation.
Reproductive and Developmental Toxicity
In a previous Cosmetic Ingredient Review (CIR) final safety assessment on alkyl PEG ethers, studies on the reproductive and teratogenic toxicity of compounds analogous to laureth 9 were evaluated. 2 The oral NOAEL in maternal rabbits was >50 mg/kg bw/d for C12AE6 (alcohol ethoxylate [AE] with 12 carbon atoms in alkyl chain; average number of ethylene oxide units = 6) administered on days 2 to 16 of gestation, based on ataxia and slight decrease in body weight observed at 100 and 200 mg/kg bw/d, but there were no effects on reproductive parameters even at the highest dose rate tested (ie, 200 mg/kg bw/d). 1 In a 2-generation feed study to evaluate the reproductive toxicity of C14-15AE7 in male and female CD rats, no compound-related effects were observed in fertility, gestation, and viability indices in the parental animals or in the maternal and fetal parameters examined on days 13 and 21 of gestation. The reported NOAEL for reproductive and developmental parameters was >0.5% in the diet (equivalent to 250 mg/kg bw/d), which was the greatest concentration tested in this study. In a similar reproductive toxicity study evaluating C12AE6 in rats, the NOAEL for reproductive parameters was >250 mg/kg bw/d (ie, the greatest dose rate tested). The NOAEL reported for maternal and developmental toxicity was 50 mg/kg bw/d, based on embryolethality and reduced body weight gains observed at 250 mg/kg bw/d. 31
Genotoxicity
Disodium Laureth Sulfosuccinate
A disodium laureth sulfosuccinate trade name material (active ingredient [40%] and active anionic surfactants [33%]) was not mutagenic in the Ames test. 8 Details relating to the test procedure were not included. The mutagenicity of another disodium laureth sulfosuccinate trade name material (32% active anionic surfactants) was evaluated using the following Salmonella typhimurium strains with and without metabolic activation 18 : TA 100, TA 1535, TA 1537, TA 1538, and TA 98. The test substance was evaluated at concentrations ranging from 0.32 µL to 200 µL/plate. A bacteriotoxic effect was observed at a dose of 200 µL/plate. The test material was not mutagenic, with or without metabolic activation, over the range of concentrations tested.
The genotoxicity of a trade name material identified as disodium laureth sulfosuccinate was evaluated in the micronucleus test using groups of 5 male and 5 female NMRI mice (∼6-8 weeks old). 17 Test animals received a single oral dose of the test substance (dose = 2000 mg/kg body weight) in distilled water. Results relating to acute oral toxicity are included in that section of the safety assessment. Cyclophosphamide and distilled water served as positive and negative controls, respectively. Two thousand polychromatic erythrocytes (PCE’s) per mouse were analyzed for the presence of micronuclei in bone marrow smears. For an investigation of bone marrow toxicity, the proportion of PCEs among total erythrocytes was evaluated on the basis of ∼200 erythrocytes. The frequency of micronucleated PCEs in the vehicle control group was within the physiological range, whereas the positive control was genotoxic. Disodium laureth sulfosuccinate did not induce an increase in the number of micronucleated PCEs in any of the test groups. There was also no statistically significant difference in the proportion of PCEs among total erythrocytes when compared to the vehicle control. Disodium laureth sulfosuccinate was not genotoxic.
Carcinogenicity
In a previous safety assessment on alkyl PEG ethers, 2 the carcinogenic potential of compounds analogous to laureth 9 was evaluated in a 2-year rat feed study. There was no evidence of a carcinogenic effect for 1% C14-15AE7 and 1% C12-13AE6.5. 31
Laureths
Summary
The safety of the following ingredients in cosmetics is reviewed in this safety assessment: disodium laureth sulfosuccinate, disodium laureth-6 sulfosuccinate, disodium laureth-9 sulfosuccinate, disodium laureth-12 sulfosuccinate, disodium deceth-5 sulfosuccinate, disodium deceth-6 sulfosuccinate, magnesium laureth-3 sulfosuccinate, disodium C12-14 pareth-1 sulfosuccinate, disodium C12-14 pareth-2 sulfosuccinate, disodium C12-15 pareth sulfosuccinate, disodium coceth-3 sulfosuccinate, disodium laneth-5 sulfosuccinate, disodium C12-14 sec-pareth-3 sulfosuccinate, disodium C12-14 sec-pareth-5 sulfosuccinate, disodium C12-14 sec-pareth-7 sulfosuccinate, disodium C12-14 sec-pareth-9 sulfosuccinate, disodium C12-14 sec-pareth-12 sulfosuccinate, and disodium oleth-3 sulfosuccinate.
Data reported to the FDA’s VCRP in 2011 and the results of a 2011 Council survey indicated use of the following ingredients in cosmetics, mostly in rinse-off products: disodium laureth sulfosuccinate (0.06% [eyeliner] to 10% [noncoloring shampoos]), disodium laureth-6 sulfosuccinate, disodium deceth-6 sulfosuccinate, and disodium C12-14 pareth-2 sulfosuccinate. A method for the production of disodium laureth sulfosuccinate involves ethoxylation of a fatty alcohol, esterification with maleic acid anhydride, addition of sodium sulfite, and neutralization with sodium hydroxide. Impurities present in disodium laureth sulfosuccinate include 1,4-dioxane, ethylene oxide, and formaldehyde. According to an MSDS on disodium laureth sulfosuccinate, this chemical contains formaldehyde at a maximum level of 0.056% and 1,4-dioxane at a maximum level of 0.001%.
Studies were not found in the published literature on the toxicokinetics of sulfosuccinates, following oral, dermal, or inhalation exposure. However, certain predictions may be made based on their chemical and physical properties. Due to the ester linkage, these sulfosuccinate ingredients are sensitive to hydrolysis, especially under acidic conditions. Accordingly, if these ingredients have the ability to penetrate the skin, then first-level metabolites would likely include the corresponding alkyl PEG ethers (eg, magnesium laureth-3 sulfosuccinate may be metabolized to laureth-3 and sulfosuccinic acid).
In 2 acute oral toxicity studies on disodium laureth sulfosuccinate trade name materials involving rats, LD50s of >10 000 mg/kg and > 2000 mg/kg were reported. However, all of the female rats that received a single 840 mg/kg oral dose of a trade name material containing 16.8% disodium laureth sulfosuccinate died within 24 hours of dosing, whereas all male rats survived to the end of the study. An acute oral LD50 of 3490 mg/kg (rats) was reported for disodium C12-14 pareth-2 sulfosuccinate. Reportedly, the acute dermal LD50 for 1 disodium laureth sulfosuccinate trade name material is >2000 mg/kg (rabbits), based on data on components or similar materials. In two 28-day oral toxicity studies (rats) on disodium laureth sulfosuccinate trade name materials, an NOEL of 250 mg/kg/d and a systemic NOAEL of 300 mg/kg/d were reported. Increased liver weight was observed in both studies.
A disodium laureth sulfosuccinate trade name material (25% active anionic surfactants) was classified as posing a risk of serious ocular damage in rabbits. Furthermore, a trade name mixture containing 1.68% active disodium laureth sulfosuccinate was classified as a primary ocular irritant in rabbits. Disodium C12-14 pareth-2 sulfosuccinate (∼40% disodium mono-(polyoxyethylene alkyl) sulfosuccinate in trade name material) was minimally irritating to the eyes of guinea pigs when tested as a 3% solution in physiological saline.
Disodium laureth sulfosuccinate (10% in citrate buffer) was classified as a mild skin irritant in healthy participants. In patients with dermatitis and healthy participants in another study, disodium laureth sulfosuccinate (5% and 10% aqueous solutions) did not induce skin irritation. The results of a human cumulative skin irritation study on a trade name mixture containing 0.168% disodium laureth sulfosuccinate predicted that this material would be a mild skin irritant during normal use. A trade name mixture containing 1.68% disodium laureth sulfosuccinate did not induce clinically significant irritation or any evidence of allergic contact dermatitis in normal human participants. A disodium C10-16 alkyl laureth sulfosuccinate, similar to disodium laureth sulfosuccinate, and a disodium laureth sulfosuccinate trade name material (effective test concentrations = 0.8% and 1.2% active ingredient) did not induce skin irritation in rabbits. Skin sensitization was not observed at an effective challenge concentration of 0.8% active ingredient in a separate study (guinea pigs) involving the same trade name material. At a higher concentration (25% active anionic surfactants), a different disodium laureth sulfosuccinate trade name material also did not induce skin irritation, and sensitization was not observed in guinea pigs when the material was diluted to an effective concentration of 1.6% active anionic surfactants. A 10% aqueous solution of a disodium C12-14 pareth-2 sulfosuccinate trade name material (∼40% disodium mono-(polyoxyethylene alkyl) sulfosuccinate) induced neither primary nor cumulative irritation in guinea pigs.
A trade name mixture containing 16.8% disodium laureth sulfosuccinate induced an increase in visible hyperkeratosis, without comedone formation, in rabbits.
Oral reproductive and development toxicity studies on C12AE6 or C14-15E7 in rabbits and rats reported maternal and developmental NOAELs as low as 50 mg/kg bw/d. In contrast, the NOAELs for reproductive toxicity were >200 to 250 mg/kg bw/d in these studies.
Ames test and micronucleus test results for disodium laureth sulfosuccinate trade name materials were negative. Compounds that are analogous to laureth 9 were not carcinogenic in feeding studies in which rats were given up to 1% in the diet for 2 years.
Discussion
The mammalian genotoxicity data received on disodium laureth sulfosuccinate were negative, and no study data were received on dermal absorption and inhalation toxicity. In the absence of inhalation and dermal absorption data, the Panel reasoned that skin penetration of these alkyl PEG sulfosuccinates would be unlikely because of their substantial polarity and molecular sizes. In addition, the high acute LD50s reported in oral animal studies suggests that the absorption of these substances through the skin at relevant doses has little potential to cause systemic effects.
The Panel did acknowledge studies that report of statistically significant increases in liver weights in animals that received repeated oral doses of disodium laureth sulfosuccinate, but given the absence of any other findings indicative of liver toxicity, such findings were not considered to be relevant. The Panel noted that sulfosuccinates have the potential for causing ocular/skin irritation but not sensitization. Therefore, products containing these ingredients should be formulated to be nonirritating.
Because disodium laureth sulfosuccinate can be used at maximum reported concentration of 2% in cosmetics that may be sprayed (hair color sprays), the Panel discussed the issue of incidental inhalation exposure. In the absence of sufficient inhalation data, the Panel considered data characterizing the potential for alkyl PEG sulfosuccinates to cause systemic toxicity, genotoxicity, ocular or dermal irritation, or sensitization, and the potential for laureths to cause reproductive and developmental toxicity or carcinogenicity. The Panel noted that 95% to 99% of the droplets/particles produced in cosmetic aerosols would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
The Panel also addressed the potential for ethylene oxide and 1,4-dioxane impurities in alkyl PEG sulfosuccinates. Due to the volatility of ethylene oxide, it would be unexpected to find any appreciable quantity of the chemical residing as an impurity in these ingredients. The available data indicate that current methods of manufacture do not result in significant levels of ethylene oxide. The available data have demonstrated contaminant levels of 1,4-dioxane to be less than 10 ppm in these ingredients, again supporting that current methods of manufacture do not result in significant levels of 1,4-dioxane. Because of the toxicity of ethylene oxide and 1,4-dioxane, the Panel stressed that the cosmetics industry should continue to use the necessary procedures to remove these impurities from the alkyl PEG sulfosuccinates before blending them into cosmetic formulations.
According to an MSDS on disodium laureth sulfosuccinate, this chemical may contain formaldehyde at a maximum level of 0.056%. This level is less than the 0.076% formaldehyde limit established by the Panel in its final safety assessment on this ingredient and is well below the threshold for any toxicological concerns relating to this chemical. Furthermore, the effective formaldehyde concentration yielded by disodium laureth sulfosuccinate in formulation would be even lower, considering that this ingredient is being used at concentrations up to 10% in rinse-off products and at concentrations up to 2% in leave-on products. At the maximum use concentration of 10%, the formaldehyde concentration would be no more than 0.006%.
Conclusion
The CIR Expert Panel concluded that the following cosmetic ingredients are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonirritating: disodium laureth sulfosuccinate, disodium laureth-6 sulfosuccinate, disodium laureth-9 sulfosuccinate*, disodium laureth-12 sulfosuccinate*, Article disodium deceth-6 sulfosuccinate, magnesium laureth-3 sulfosuccinate*, disodium C12-14 pareth-1 sulfosuccinate*, disodium C12-14 pareth-2 sulfosuccinate, disodium C12-15 pareth sulfosuccinate*, disodium coceth-3 sulfosuccinate*, disodium laneth-5 sulfosuccinate*, disodium C12-14 sec-pareth-3 sulfosuccinate*, disodium C12-14 sec-pareth-5 sulfosuccinate*, disodium C12-14 sec-pareth-7 sulfosuccinate*, disodium C12-14 sec-pareth-9 sulfosuccinate*, disodium C12-14 sec-pareth-12 sulfosuccinate*, and disodium oleth-3 sulfosuccinate*
Were ingredients in this group not in current use to be in the future (indicated by *), the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Author Contribution
W. Johnson contributed to conception and design; acquisition, analysis, and interpretation; and drafted the article. B. Heldreth contributed to conception and design; acquisition, analysis, and interpretation; drafted the article, and critically revised the article. L. J. Gill, W. F. Bergfeld, D. V. Belsito, A. Hill, C. D. Klaassen, D. C. Liebler, R. C. Shank, T. J. Slaga, P. W. Snyder, and J. G. Marks contributed to conception and design, analysis and interpretation, and critically revised the article. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.