Abstract
Biological oxidative responses are involved in the toxicity of multiwall carbon nanotubes (MWCNTs), which may cause asbestos-like pathogenicity. Superoxide dismutase 2 (SOD-2) has been proposed as a biomarker of early responses to mesothelioma-inducing fibers. This study was conducted to investigate the alteration of SOD-2 expression in the human mesothelial cell lines Met-5A after exposure to nontoxic doses of MWCNTs and the potential signaling pathway. The parameters measured included the viability, morphological change, superoxide formation, extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation, and messenger RNA (mRNA)/protein levels of SOD-2. Our results showed that MWCNTs upregulated SOD-2 expression at both mRNA and protein level. Coincidently, both superoxide formation and ERK1/2 phosphorylation were observed in Met-5A cells exposed to MWCNTs and were diminished by pretreatment with the reactive oxidative species (ROS) scavenger, N-acetyl-
Keywords
Introduction
Carbon nanotubes (CNTs), with a unique combination of stiffness, strength, and tenacity, typically have intrinsic mechanical and transport properties and show high performance of thermal and electrical conductivity (see http://www.nanocyl.com/en/CNT-Expertise-Centre/Carbon-Nanotubes). Because of these physical characteristics, CNTs can be applied in electronics, polymer composites, thermoplastics, coatings, and adhesives. 1 The wide use of CNTs may lead to potential health problems for humans, as occupational inhalation exposure, dermal exposure, and consumer exposure to CNTs are likely, although workers generally have higher exposure than the general population. 1,2 Indeed, some animal studies have revealed that fiber pathogenicity paradigm is applicable to CNTs-induced toxicity, and some adverse health effects caused by CNTs include macrophage infiltration, type II cell hyperplasia, fibrosis with alveolar wall thickening, development of an extensive collagen network developed within granulomas, and asbestos-like pathogenicity. 3 –5 These studies suggest that because of the physical resemblance to asbestos in terms of high aspect ratio, CNTs may elicit significant pulmonary injury.
The translocation of CNTs within the lung tissue may cause damage to various target cells, including alveolar macrophages, epithelial cells, and pleural mesothelial cells. Previous studies have shown that CNTs, distributed into the lung through the airway, may penetrate alveolar macrophage cells as well as alveolar epithelium and visceral pleura. Carbon nanotubes-loaded macrophages can translocate into lung-associated lymph nodes, 6 whereas the nonphagocytosed CNTs may directly pierce into subpleural tissues, visceral pleura, and pleural space. 5 Recent studies have further demonstrated that thin and rigid CNTs could penetrate plasma and nuclear membranes of mesothelial cells without active cellular processes, such as endocytosis or phagocytosis. 7 The interactions between CNTs and target cells within the lung tissue (eg, mesothelial cells) may initiate adverse health effects, such as reduced cell viability, disrupted plasma and nuclear membranes, DNA damage with oxidative effects in vitro, and fibrotic inflammation and mesothelioma in vivo. 7 –11 Previous studies have indicated that the different profile of superoxide dismutase 2 (SOD-2) expression and its activity, as well as biologically relevant trace elements (eg, manganese, copper, and zinc), may be potential disease-related markers of mesothelioma and considered as a biomarker of early responses to mesothelioma-inducing fibers. 12,13 The same genetic signature of asbestos- and CNTs-induced mesothelial cell injury leading to mesothelioma has been proposed, 7 and SOD-2 may be valuable for predicting fiber pathogenicity. 13 Nevertheless, the possible molecular pathway mediating SOD-2 expression (eg, oxidative stress 13 and extracellular signal-regulated kinase (ERK) 1/2 14,15 ) has not been well described in mesothelial cells insulted by CNTs, which possess a needle-like morphology comparable to asbestos. Therefore, we first investigated SOD-2 expression in human pleural mesothelial cells exposed to multiwall carbon nanotubes (MWCNTs) and then examined whether the reactive oxidative species (ROS) generation and ERK1/2 phosphorylation are associated with SOD-2 expression. Our data indicated that SOD-2 was expressed in response to ROS-ERK1/2 activation induced by MWCNTs.
Materials and Methods
Carbon Nanotubes and Their Characterization
Multiwall carbon nanotubes were purchased from Sigma-Aldrich (St Louis, Missouri). Their main characteristics have been previously described and summarized. 16 Briefly, their dimensions (diameter × length) were 110 to 170 nm × 5 to 9 μm, the purity was more than 90% with metal contaminants less than 0.1%, and the specific surface area was 130 m2/g. In the present study, MWCNTs were dispersed in the cell medium, RPMI 1640, supplemented with 10% fetal bovine serum (FBS). The MWCNTs’ microstructure in the solution was needle-like fiber with some occasional agglomerates as shown in the transmission electron microscopy (TEM) image (Figure 1). In addition, the estimated diameters of MWCNTs ranged from 120 to 280 nm, and the estimated length ranged from 2 to 10 µm.
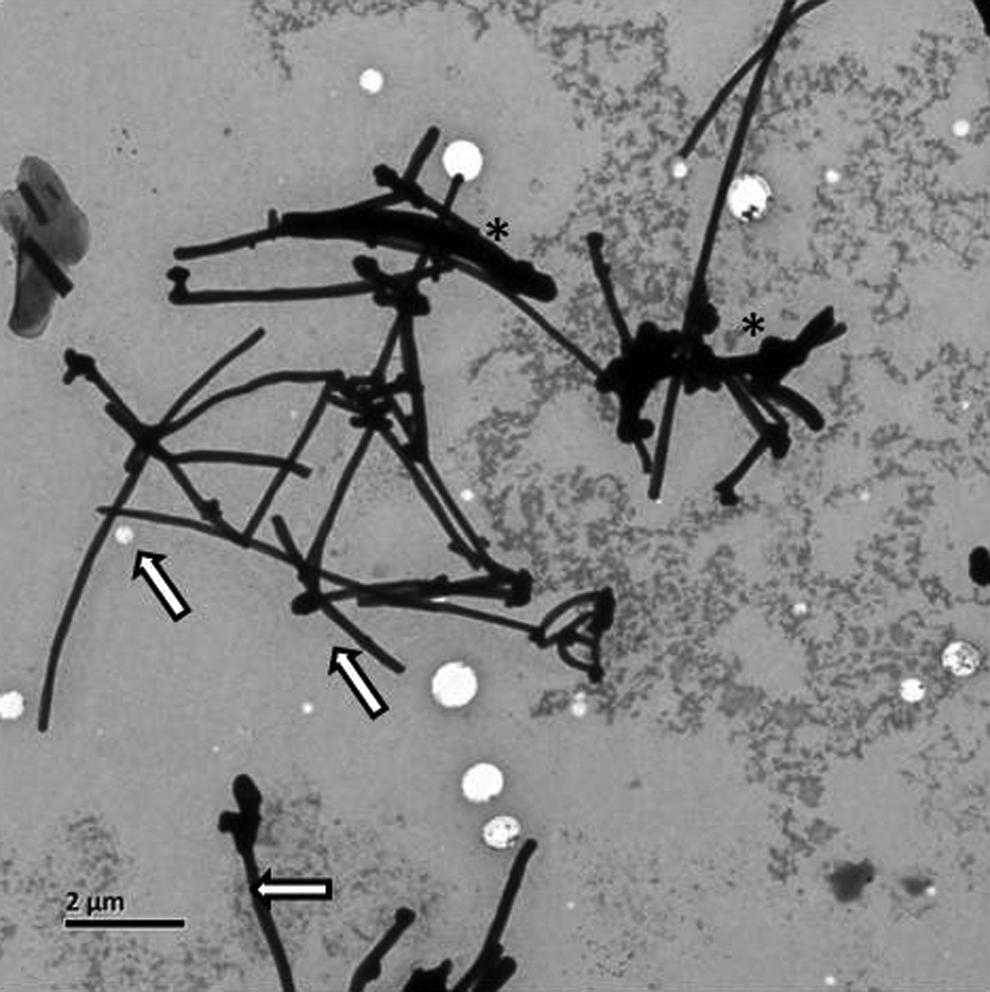
Representative transmission electron microscopy (TEM) image of multiwall carbon nanotube (MWCNT) powder suspension in RPMI 1640 with 10% fetal bovine serum (FBS). Magnification ×5000. Arrows indicate the single MWCNT; asterisks, bundles of MWCNT.
Chemicals and Reagents
Dihydroethidium (DHE) and U0126 were obtained from Beyotime (Jiangsu, China), N-acetyl-
Cell Culture
The immortalized human mesothelial cell line Met-5A was obtained from the American Type Culture Collection (ATCC) and maintained at subculture in RPMI 1640 supplemented with 10% FBS at 37°C and 5% CO2.
Cytotoxicity of MWCNTs
The cytotoxicity of MWCNTs in Met-5A cells was determined by Cell Titer-Glo luminescent cell viability assay (Promega, Madison, Wisconsin) or lactate dehydrogenase (LDH) cytotoxicity assay kit (Beyotime) according to the manufacturer’s instruction. Cell Titer-Glo luminescent cell viability assay is a method for determining the number of viable cells in culture based on quantification of the adenosine triphosphate present, an indicator of metabolically active cells. Briefly, Met-5A cells were treated with different concentrations (0, 10, 20, 40, and 80 µg/mL) of MWCNTs in a total volume of 100 µL/well for 24 hours, and then 100 µL Cell Titer-Glo reagent was added into each well, and luminescence was recorded with SpectraMax M5 multimode microplate reader (Molecular Devices, Sunnyvale, California). Another method, LDH release assay, is a colorimetric method for determining cellular membrane damage. Briefly, Met-5A cells were exposed to different doses (ranging from 0 to 80 μg/mL) of MWCNTs for 24 hours, and the supernatant was moved to a 96-well plate and 60 μL of the LDH test reagent were added to each well. Following a 30-minute incubation period, the absorbance of each well at a wavelength of 490 nm was measured using SpectraMax M5 multimode microplate reader (Molecular Devices). The cell viability was expressed as the percentage of the control which was without treatment.
Transmission Electron Microscopy of MWCNTs Interaction With Met-5A Cells
Met-5A cells exposed to MWCNTs were analyzed by TEM as described previously with minor modification. 17 Briefly, cells were exposed to 0, 20, 40, or 80 μg/mL MWCNTs for 24 hours and then trypsinized, fixed in suspension of 2.5% paraformaldehyde, postfixed in 1% osmic acid, and embedded in Epon (SPI Suppliers, West Chester, Pennsylvania). Ultrafine (60-70 nm thick) slices were cut, stained with uranyl acetate and lead citrate, and examined under JEOL 1400 TEM (JEOL Ltd., Tokyo, Japan).
Measurement of ROS generation
DHE, a fluorescent probe for detecting superoxide anions, was used to determine intracellular ROS generation. The intensity of DHE fluorescence in Met-5A cells exposed to 0, 10, 20, or 40 μg/mL MWCNTs for 24 hours was measured using SpectraMax M5 multimode microplate reader (Molecular Devices) at 535 nm excitation (λex) and 610 nm emission (λem). Untreated cells were used as control. Results, in arbitrary fluorescence units (AFUs), are expressed according to the ratio [(AFU of treated cells)/(AFU of control cells)] × 100. To examine the effect of antioxidants on ROS generation in Met-5A cells after exposure to MWCNTs, Met-5A cells were pretreated with NAC (2.5 mmol/L) for 1 hour and then exposed to 40 μg/mL of MWCNTs for 24 hours. Fluorescence values were measured as described earlier.
Protein Extraction and Western Blot
Met-5Acells were lysed with Radio Immunoprecipitation Assay (RIPA) lysis buffer (Beyotime) supplemented with Phenylmethanesulfonyl fluoride (PMSF) (Beyotime) and phosphatase inhibitor complex (Sangon Biotech, China) on ice for 40 minutes. The supernatant was collected after centrifugation at 10 000×g and 4°C for 15 minutes. The protein concentration in the supernatant was measured by the BCA protein assay (Bio-Rad, Hercules, California). Fifteen micrograms of proteins from each sample were resolved on 12% sodium dodecyl sulfate–polyacrylamide gel and were electrotransferred into Immun-Blot polyvinylidene fluoride membranes. The membranes were incubated with antibodies against GAPDH, SOD-2, phospho-ERK1/2, or total ERK1/2, respectively, at 4°C overnight with gentle shaking. The blot was washed 3 times with 1 × Phosphate Buffer Saline with Tween-20 (PBST), incubated with HRP-conjugated secondary antibodies for 1 hour and exhaustively washed 3 times with 1 × PBST. Immunoreactive bands were detected by chemifluorescence using ECL Western Blotting Detection Reagents (Amersham Biosciences, Piscataway, New Jersey) and visualized by using Fluo Chem FC (Alpha Innotech, San Leandro, California).
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
Total RNA was extracted from Met-5A cells using Ultrapure RNA kit (CWbiotech, China) according to the manufacturer’s instruction. One microgram of total RNA was reverse transcribed into complementary DNA (cDNA) using TaKaRa RNA PCR kit (AMV) Ver.3.0 (TaKaRa, Dalian, China) in a total volume of 20 µL. A total of 2 µL of cDNA, 0.4 µL of 10 µmol/L each primer, and 10µL SYBR Premix Ex Taq (Tli RNaseH Plus; 2×; TaKaRa) were used in each polymerase chain reaction (PCR) at a final volume of 20 µL. The PCR was performed on a Light Cycler 480 System Real Time PCR (Roche Diagnosis, Shanghai, China). The primers for human GAPDH were 5′-ACG GAT TTG GTC GTA TTG GG-3′ and 5′-CTC GCT CCT GGA AGA TGG TG-3′, for human SOD-2 were 5′-GAC ACT TAC AAA TTG CTG CTT GTC C-3′ and 5′-CTT TCA GTT ACA TTC TCC CAG TTG-3′, messenger RNA (mRNA) expression was normalized against GAPDH.
Statistical Analysis
Values were presented as mean ± standard deviation. The data were analyzed by 1-way analysis of variance using SPSS Statistics 17.0. A value of P < 0.05 was considered statistically significant.
Results
Cytotoxicity and Morphological Change
Cell viability in response to the indicated doses of MWCNTs, as determined by the LDH assay and the Cell Titer-Glo luminescent cell viability assay, is shown in Figure 2. Multiwall carbon nanotubes at concentrations of 40 µg/mL and below did not have a significant cytotoxic effect on Met-5A cells compared with the untreated cells. In contrast, 80 µg/mL MWCNTs remarkably reduced the cell viability compared with the untreated cells as determined by both assays. The results showed that the 24-hour treatment of Met-5A cells with MWCNTs at up to 40 µg/mL was not toxic.
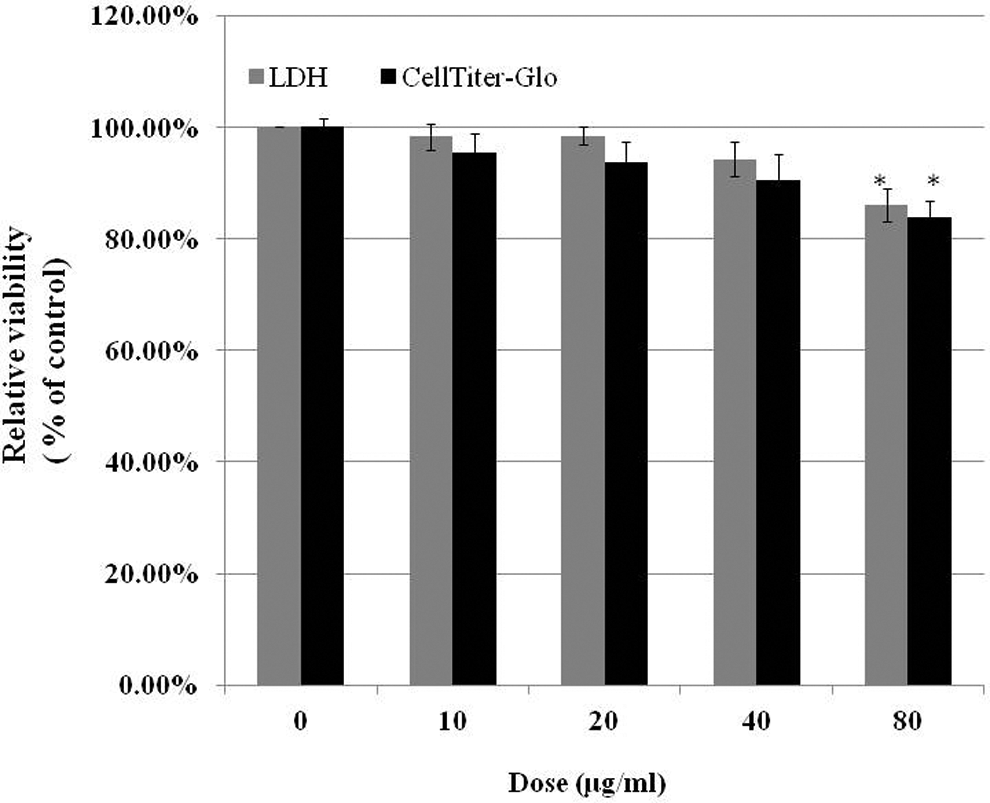
Cell viability assessed by lactate dehydrogenase (LDH) assay and Cell Titer-Glo luminescent cell viability assay. Met-5A cells were treated with different doses of multiwall carbon nanotubes (MWCNTs) for 24 hours, and cytotoxicity was determined by the indicated assay. Met-5A cells without treatment were used as control. Values are mean ± standard error (SE) of 5 experiments. *Significant difference compared with the control group, P < 0.05.
To investigate the interaction between MWCNTs and Met-5A cells, TEM was used to examine the cell morphology. In Figure 3, untreated cell had a well-preserved cytoplasm and intact nucleus membrane with nucleolus. Neither significant morphological changes nor internalization of MWCNTs were observed in cells exposed to 20 or 40 µg/mL MWCNTs. However, lysosome-like organelles were clearly seen in cells exposed to 80 µg/mL MWCNTs, and some MWCNTs were occasionally present in lysosome-like organelles. Thus, MWCNTs were not present in Met-5A cells exposed to nontoxic doses of MWCNTs (eg, 40 µg/mL and below).
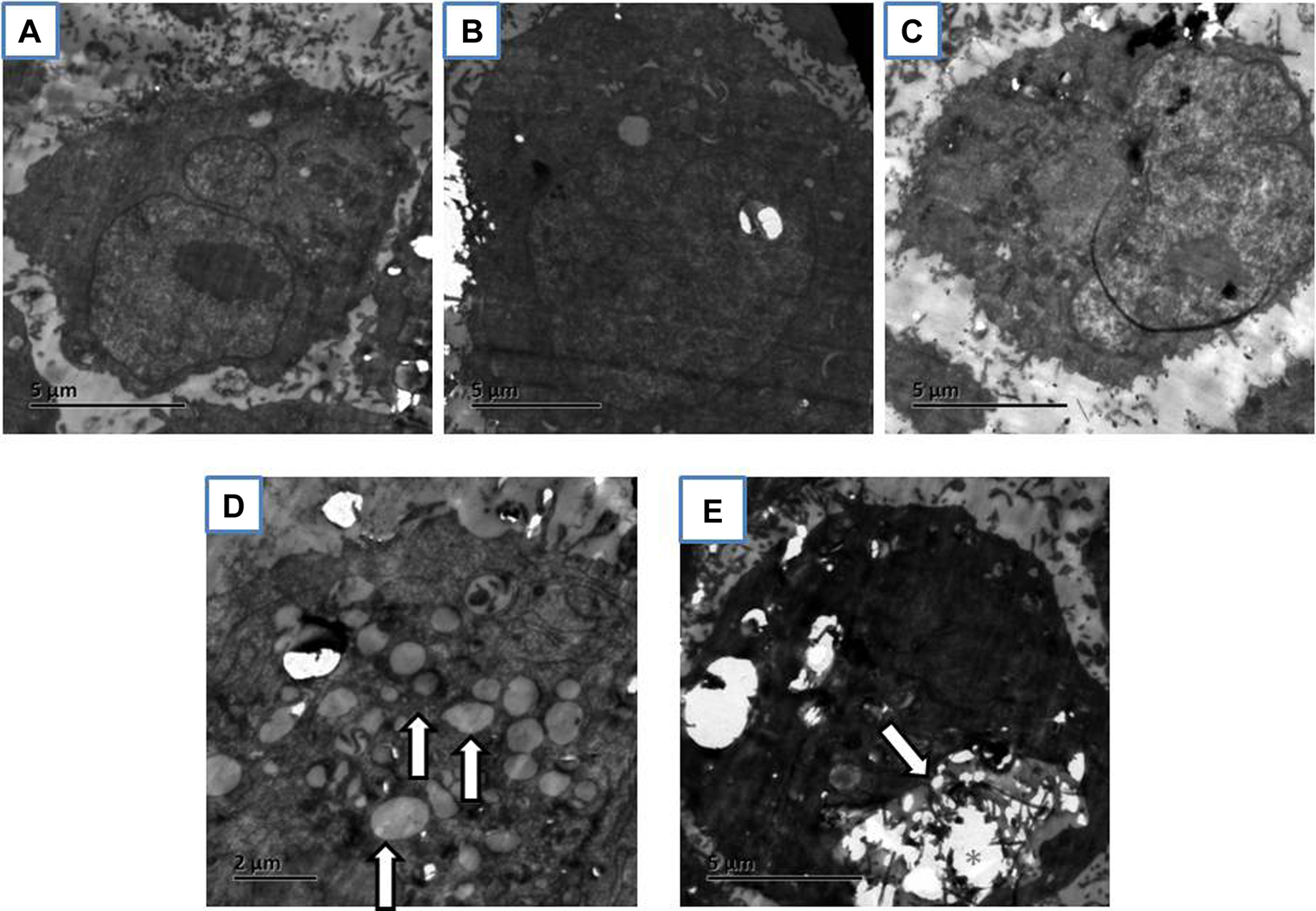
Representative of transmission electron microscopy (TEM) images of Met-5A exposed for 24 hours to 0 µg/mL multiwall carbon nanotubes (MWCNTs; A), 20 µg/mL MWCNTs (B), 40 µg/mL MWCNTs (C), or 80 µg/mL MWCNTs (D and E). Lysosome-like organelles are indicated by arrows. Clusters of MWCNTs are indicated by asterisks.
Reactive Oxidative Species Generation
The ROS generation in Met-5A cells was detected by the DHE fluorescent probe. The intensity of DHE fluorescence, as determined by a microplate reader, demonstrated that MWCNTs significantly augmented the intracellular ROS level in a dose-dependent manner, and an approximately 1.7-fold increase in ROS production was detected in 40 µg/mL MWCNTs-treated cells after 24-hour treatment. In contrast, pretreatment of the cells with the ROS inhibitor NAC prior to exposure to 40 µg/mL MWCNTs significantly reduced the ROS level (Figure 4).
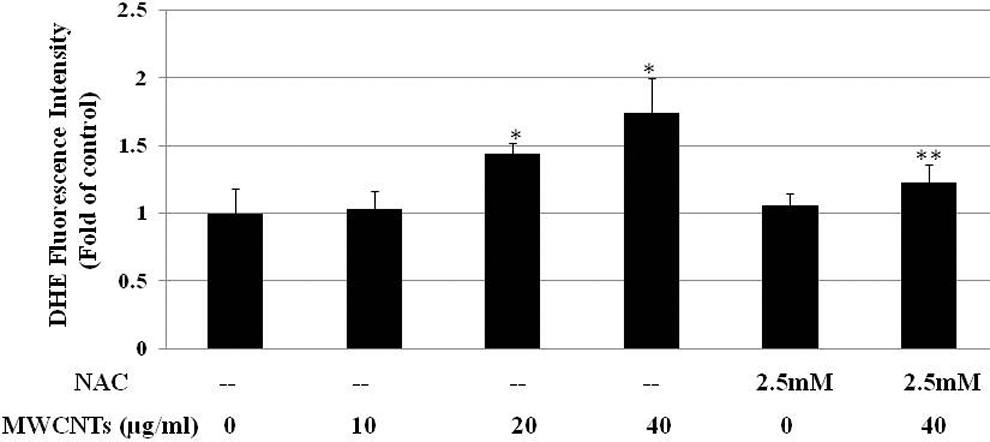
Multiwall carbon nanotubes (MWCNTs) stimulate intracellular reactive oxidative species (ROS) in Met-5A cells. Dihydroethidium fluorescence intensity determined by a microplate reader and plotted as fold of ROS change compared with untreated cells. *Significant difference compared with control, P < 0.05. **Significant difference compared with the 40 μg/mL MWCNTs-treated group, P < 0.05.
Extracellular Signal-Regulated Kinase 1/2 Activation Mediated by ROS
There was a dose-dependent increase in ERK1/2 phosphorylation after Met-5A cells were treated with the indicated concentrations of MWCNTs for 24 hours (Figure 5A). To further explore the relationship between ROS generation and ERK1/2 activation, cells were pretreated with the ROS inhibitor NAC (2.5 mmol/L) for 1 hour prior to exposure to 40 µg/mL MWCNTs. Figure 5B shows that NAC significantly decreased the MWCNTs-induced ERK1/2 phosphorylation, indicating the ERK1/2 activation is associated with ROS generation.
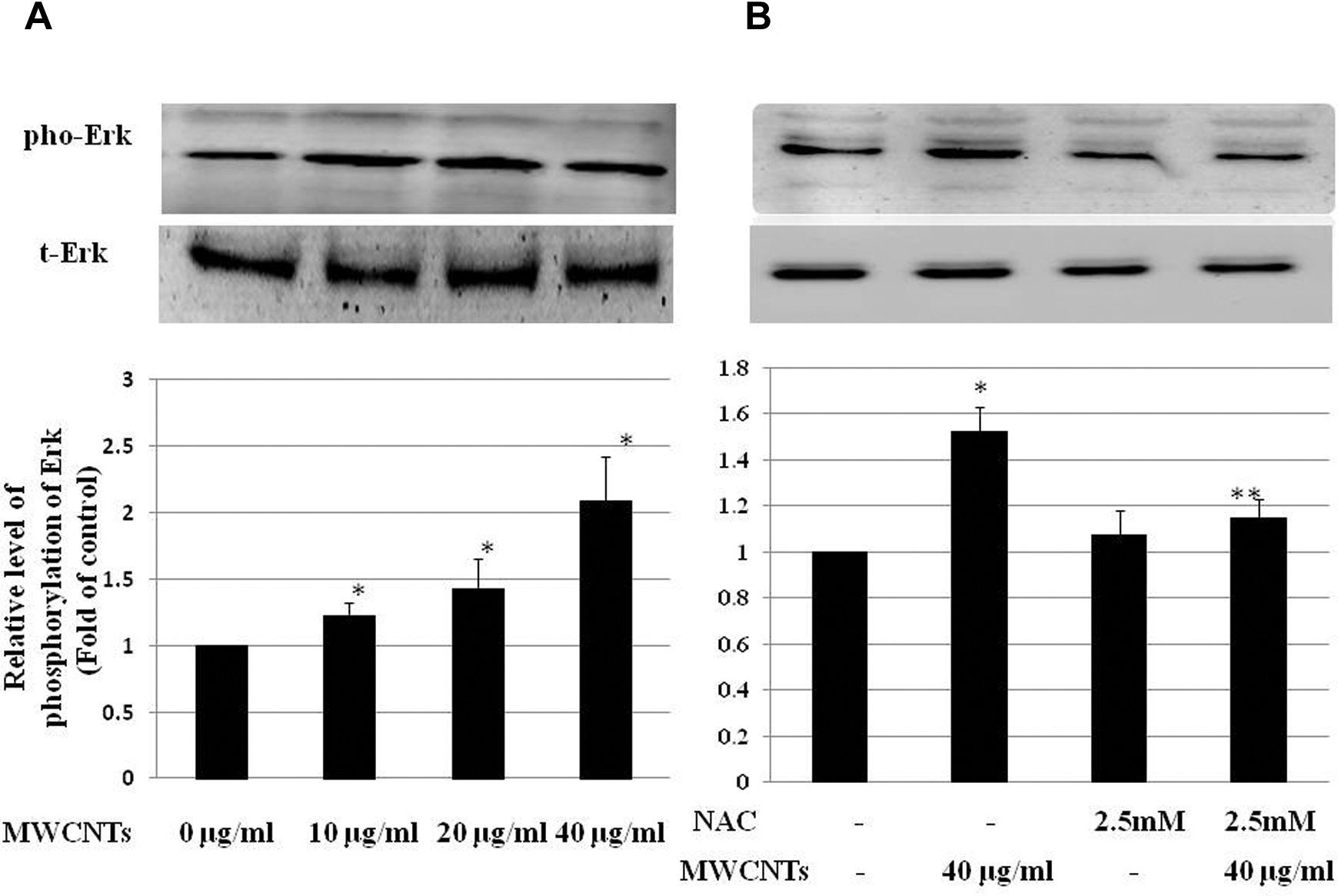
Multiwall carbon nanotubes (MWCNTs) induce extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation via reactive oxidative species (ROS) in Met-5A cells. A, Met-5A cells were treated with 0 to 40 µg/mL MWCNTs for 24 hours. Untreated cells were used as control. B, Met-5A cells were pretreated with 2.5 mmol/L N-acetyl-
Expression of SOD-2
The effect of MWCNTs on SOD-2 mRNA and protein expression in Met-5A cells was determined by real-time PCR and by Western blot, respectively. Figure 6A shows there was an increase in the mRNA level after exposure to 0 to 40 µg/mL MWCNTs for 24 hours, in a dose-dependent manner. Consistent with the transcriptional regulation, there was an increase in the SOD-2 protein level in Met-5A cells exposed to 20 or 40 µg/mL MWCNTs as illustrated in Figure 6B.
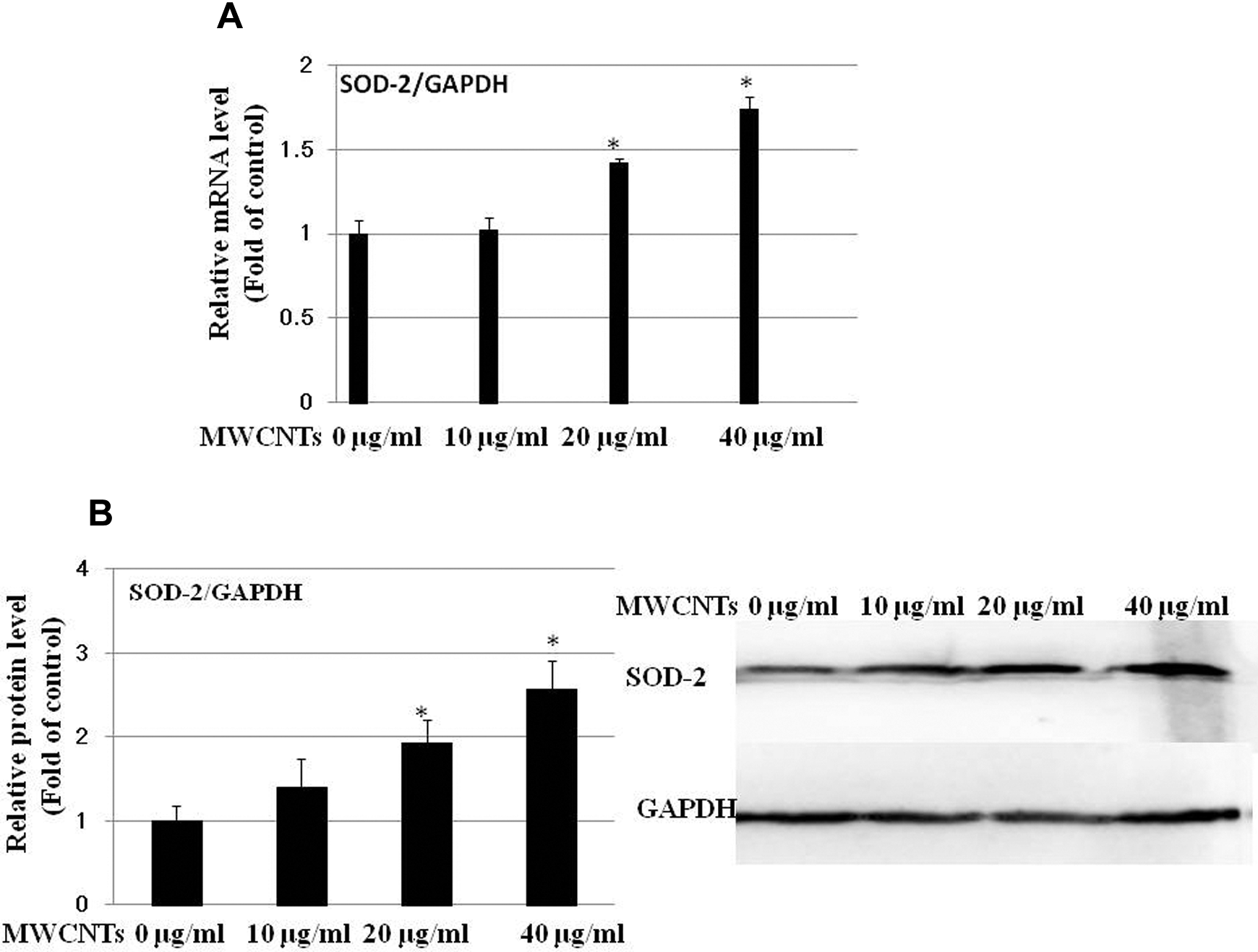
Multiwall carbon nanotubes (MWCNTs) upregulate superoxide dismutase 2 (SOD-2) expression at both messenger RNA (mRNA) and protein level. Met-5A cells were treated with 0 to 40 µg/mL of MWCNTs for 24 hours. Untreated cells were used as control. A, Normalized SOD-2 mRNA levels are an average of at least 2 independent experiments ± standard error (SE) of real-time PCR (RT-PCR). B, Left: normalized band densitometry values are an average of 3 independent experiments ± SE of Western blot results. B, Right: representative Western blot experiment was shown. *Significant difference as compared with control, P < 0.05.
Induction of SOD-2 Expression is Associated With ROS-ERK1/2
To further investigate whether ROS generation and ERK1/2 activation are involved in the induction of SOD-2 in Met-5A cells after exposure to MWCNTs, cells were pretreated for 1 hour with the ROS inhibitor, NAC (2.5 mmol/L), or the MEK1/2 inhibitor, U0126 (20 µmol/L), prior to exposure to 40 µg/mL MWCNTs. The results showed that pretreatment with 2.5 mmol/L NAC (Figure 7) or 20 µmol/L U0126 (Figure 8) significantly decreased the upregulation of MWCNTs-induced SOD-2.
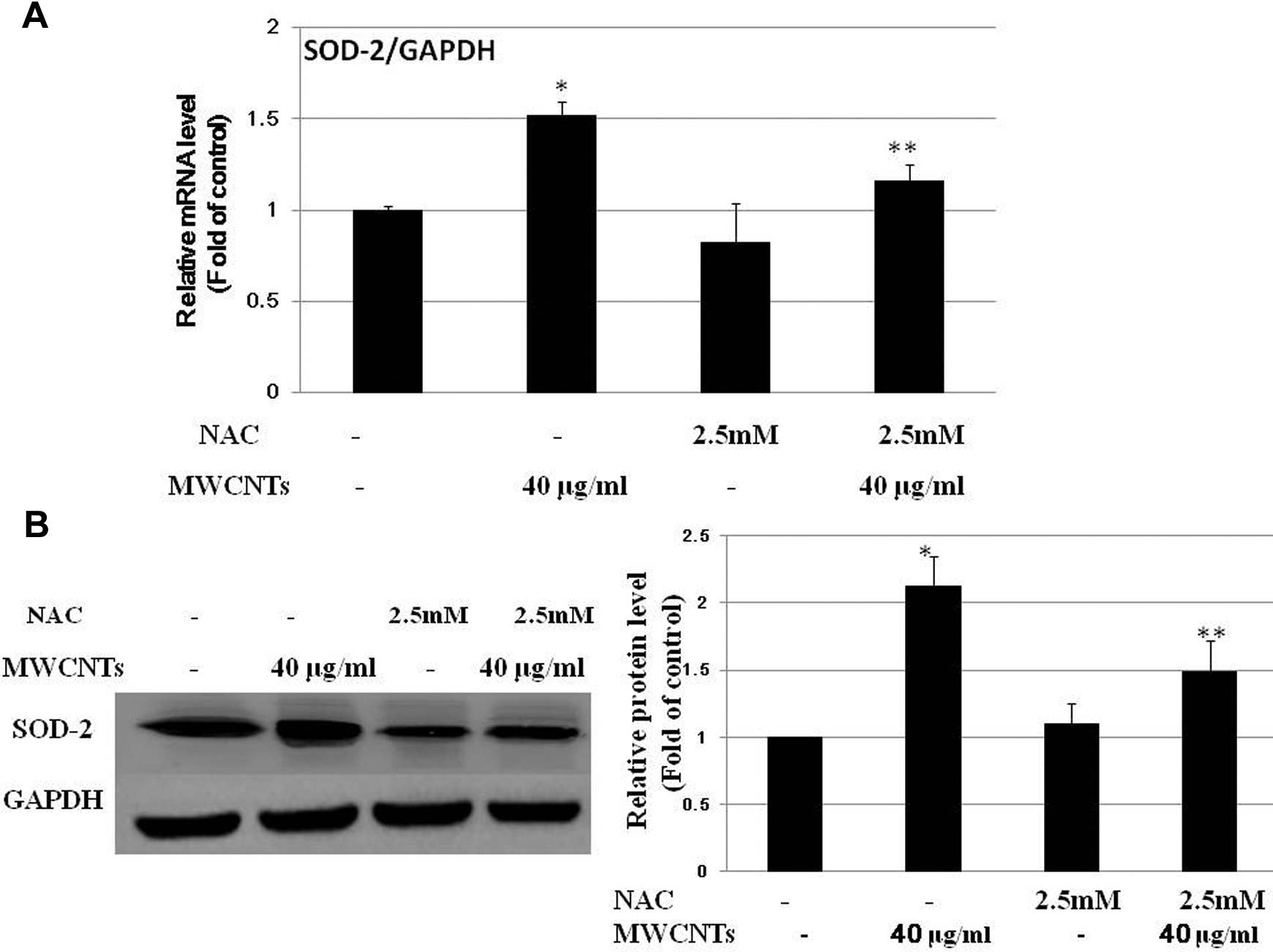
Effects of N-acetyl-
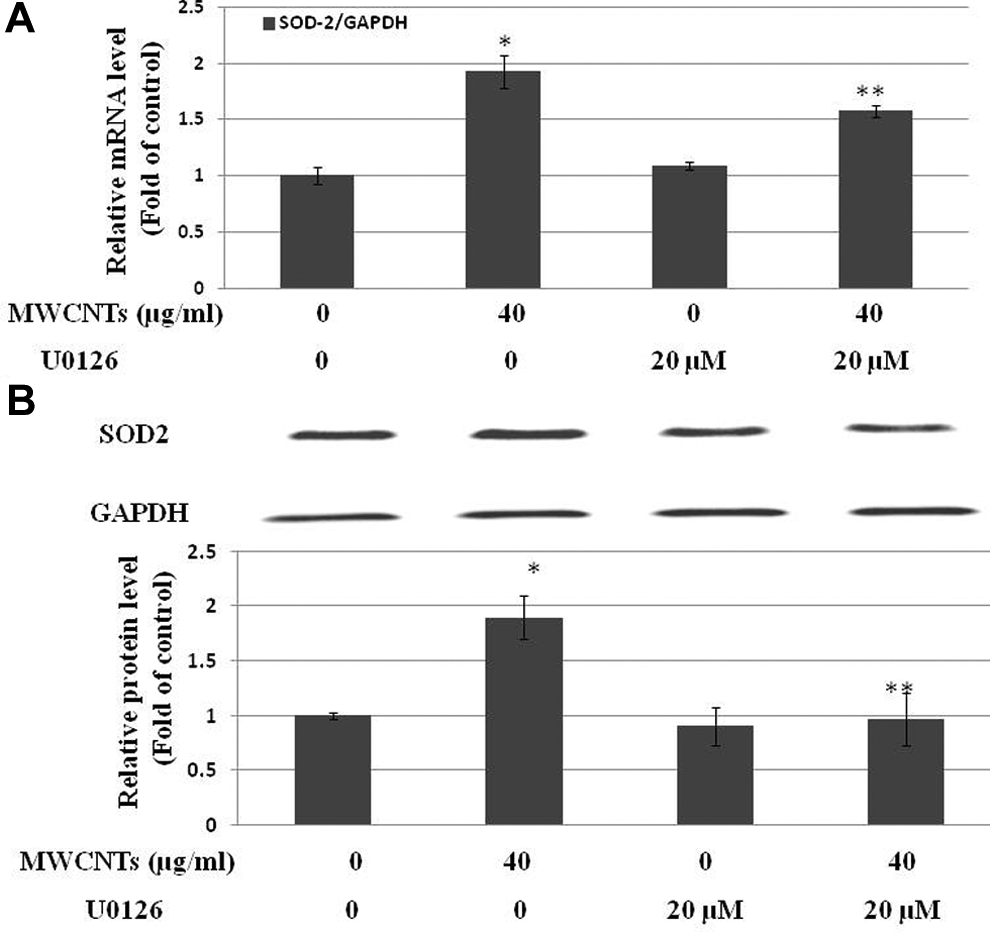
Effect of U0126 on multiwall carbon nanotubes (MWCNTs)-induced superoxide dismutase 2 (SOD-2) upregulation. Met-5A cells were pretreated with the 20 µmol/L U0126 for 1 hour prior to exposure to 40 µg/mL MWCNTs for 24 hours. Untreated cells were used as control. A, Normalized SOD-2 messenger RNA (mRNA) level are an average of at least 2 independent experiments ± standard error (SE) of real-time PCR (RT-PCR). B, Representative Western blot experiment was shown and normalized band densitometry readings averaged from 3 independent experiments ± SE of Western blot results were given. *Significant difference compared with control, P < 0.05. **Significant difference compared with the 40 μg/mL MWCNTs-treated group, P < 0.05.
Discussion
Given that CNTs’ unique physicochemical properties and biopersistence are comparable to asbestos, great concerns regarding CNTs-induced adverse health effects have emerged. Previous studies have shown that inhaled CNTs were not only translocated within the pulmonary but also transported into extrapulmonary organs, such as parietal pleura, the respiratory musculature, liver, kidney, heart, and brain. 5,6,18 Because of the resemblance between CNTs and asbestos, it is reasonable that CNTs induce initiation of inflammation and pleural pathology. 19 In the pleural cavity, mesothelial cells are a possible target of retained CNTs, because of CNTs’ binding affinity to cellular membranes, 20 therefore, we investigated the biological responses to certain CNTs (eg, MWCNTs) in pleural mesothelial cells by studying their cytotoxic effects, and their effects on ROS generation, ERK1/2 activation, and SOD-2 gene expression.
Our results showed that exposure to 80 µg/mL MWCNTs caused significant cytotoxic effects on Met-5A cells as determined by both the LDH assay and the Cell Titer-Glo luminescent cell viability assay, indicating that high concentrations of MWCNTs induced cellular membrane permeability and disturbance of mitochondrial metabolism. However, MWCNTs at concentrations 40 µg/mL and below showed no significant toxic effects on Met-5A cells. The cytotoxicity of MWCNTs depends on many parameters, such as diameter, length, rigidity, shape, surface properties, and residue trace metal elements. 7,21 –23 Although the MWCNTs used in the present study were characterized by higher surface/mass ratio (130 m2/g vs 60 m2/g), longer length (5-9 μm vs 1-2μm), larger diameter (110-170 nm vs 10-30 nm), and smaller amounts of metal contaminants (<0.1 wt% Fe vs <0.5 wt% Ni), compared with the MWCNTs used in a study by Lindberg et al, 11 the toxic effects were comparable to their results, which demonstrated that a >50% reduction in viable Met-5A cells was seen from a concentration of 152 µg/mL. The low cytotoxicity of MWCNTs in our experiments may be attributed to their stiffness and low potential to penetrate into cells, 11,16,24 because no internalization of MWCNTs was observed in Met-5A cells exposed to 20 or 40 µg/mL MWCNTs in our experiments.
It has been shown that oxidative damage is predictive of potential toxicity of various nanomaterials including CNTs.
25,26
Previous studies have shown that the formation of unique free radicals other than
Superoxide dismutase 2, an intracellular antioxidant enzyme, counteracts against ROS-mediated toxicity by catalyzing the dismutation of superoxides into hydrogen peroxide and oxygen. Transfection of the SOD-2 gene into hamster tracheal epithelial cells reduced asbestos-induced cytotoxicity, an oxidant-dependent process. 29 Previous studies have shown that alterations in the amount and/or activity of SOD-2 were influenced by fiber in different types of pulmonary cells. Asbestos and Libby six-mix elevated SOD-2 gene expression in human pleural mesothelial cells and in tracheal epithelial cells, 13,30,31 hence, early increases in SOD-2 expression, protein levels, and activity in human mesothelial cells after exposure to malignant mesothelioma-inducing fibers may be valuable for predicting fiber pathogenicity. 13 In agreement with these studies, our results revealed that MWCNTs caused a dose-dependent increase in SOD-2 expression at both mRNA and protein level in Met-5A cells. In contrast, another group has shown that there was no significant change in SOD-2 gene expression in Met-5A cells after exposure to asbestos or MWCNTs. 17 One possible explanation for this discordance is that nonoxidant mechanism may play a role in MWCNTs and asbestos-elicited biological responses as suggested by Tabet et al, 17 whereas the MWCNTs used in the present study are potent to induce ROS generation.
Oxygen free radicals and oxidative stress play a role in regulating SOD-2 expression, 32 –34 and an intense, continuous labeling of SOD-2 transcripts in pleura mesothelial cells from lung suffering from oxidative stress were observed. 35 Here, ROS was involved in the induction of SOD-2 in Met-5A cells exposed to nontoxic concentration of MWCNTs. Our results demonstrated that pretreatment with the ROS scavenger NAC significantly decreased MWCNTs-induced upregulation of SOD-2 expression at both the mRNA and protein level. The induction of SOD-2 via the NF-κB pathway may be a protection mechanism of cells against oxidative stress. 36 –38
Upstream kinases (eg, ERK Mitogen-activated Protein Kinase (MAPK)) responsible for activation of transcription factors (eg, NF-κB) were stimulated in normal and malignant human mesothelial cells insulted with single-wall CNTs. 39 The present results showed increased ERK1/2 phosphorylation in human mesothelial Met-5A cells treated with MWCNTs, and the increased ERK1/2 phosphorylation was diminished by the ROS scavenger NAC, indicating that the MWCNTs-activated ERK1/2 signaling is associated with ROS formation. Previous studies have demonstrated that ERK1/2 signaling cascades participate in many biological responses, such as remodeling of the lung during the development of pulmonary fibrosis, 40 phenotypic responses elicited by oxidant stress in rat pleural mesothelial cells, 41 and regulation of the antioxidant gene SOD-2. 14,15,42 To further investigate whether ROS-ERK1/2 activation is associated with SOD-2 induction, the level of SOD-2 expression was compared between Met-5A cells exposed to MWCNTs and Met-5A cells exposed to both MWCNTs and ERK1/2 activation inhibitor U0126. Our results demonstrated that pretreatment Met-5A cells with the MEK activation inhibitor, U0126, significantly inhibited the MWCNTs-induced SOD-2 upregulation. Based on these results, it is evident that ERK1/2 MAPK plays a role in the ROS-mediated SOD-2 induction in Met-5A cells insulted by MWCNTs.
In summary, our results demonstrated that exposure to MWCNTs enhanced SOD-2 expression in human pleural mesothelial Met-5A cells. The regulation of SOD-2 induction was mediated by ROS formation and ERK1/2 activation. These results provide further understanding of the redox-sensitive responses to low toxicity MWCNTs. The regulatory pathway of SOD-2 induction and its biological effects on pleural mesothelium insulted by materials characterized by high aspect ratio remains to be elucidated.
Footnotes
Author Contributions
Min Yu contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Riping Chen contributed to acquisition and analysis and critically revised the manuscript. Zhenyu Jia contributed to analysis and interpretation and critically revised the manuscript. Junqiang Chen contributed to acquisition and interpretation and critically revised the manuscript. Jianlin Lou contributed to conception, contributed to interpretation, and critically revised the manuscript. Shichuan Tang contributed to conception, contributed to acquisition and interpretation, and critically revised the manuscript. Xing Zhang contributed to conception, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Key Project of Zhejiang Province Medicine Plan (11ZC02), the Innovation Projects of the Beijing Academy of Science and Technology (PXM2014-178304-000001-00130138), the Science and Technology Project for Key Technology of Major Accident Prevention of State Administration of Work Safety (Beijing-0003-2014AQ), and Young start-up funding from Zhejiang Academy of Medical Sciences (PT1131E). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
