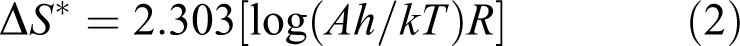Abstract
In the present work, multiwalled carbon nanotube (MWCNT) chemically modified with (3-mercaptopropyl) silanetriolate is efficiently used for the solid-phase extraction of Cu2+, Ag+, Cd2+, Pb2+, Zn2+ and Mn2+ ions prior to their flame atomic absorption spectrometric determination. The influences of the various analytical parameters, including pH, amounts of solid phase, sample volume and eluent conditions and so on, on the recoveries of target analytes were investigated and optimized by one at a time optimization method. The influences of alkaline, alkaline earth and some transition metals on the adsorption and elution of the analytes were also examined. The detection limits for all understudied metal ions were between 1.4 and 2.8 ng mL−1 (3Sb, n = 10). The evaluation of the thermodynamic parameters such as enthalpy (positive value), Gibbs free energy (negative value) in addition to high value of entropy shows the endothermic and spontaneous nature of sorption process. Following the optimization of variables, the adsorption process follows the intraparticle kinetic model with R 2 of 0.98 and the Langmuir isotherm with high correlation coefficient (R 2 > 0.95). The procedure was applied for the analytes determination in the food samples with satisfactory results (recoveries >95% and relative standard deviation’s (RSD) lower than 4%).
Introduction
Trace metal analysis is a great and important task for analytical chemists. To prevent the interference effect of matrix and to determine the low levels of trace metal ions in the water samples by flame atomic absorption spectrometry (FAAS) performing an efficient preconcentration step is usually required in order to bring the concentration of the analyte within the dynamic measuring range of analytical tools. The separation enrichment techniques generally improve the sensitivity and selectivity of the trace metal analysis. 1 –4 The enrichment of heavy metal ions in off-line or online performance has been reported and reviewed. 5 –10 Among various separation and/or preconcentration procedures solid-phase extraction (SPE) is widely applied due to its high enrichment factor and low consumption of reagent in simple and low-cost pathway. The selection of efficient support has greater importance in this procedure to obtain full recovery and high enrichment factor. 11 –13 The compatible selection of support and modifier leads to overcome the disadvantages of SPE like poor sensitivity to provide low detection limit and avoid matrix interference in the analysis of the real samples.
Carbon nanotubes (CNTs) have come under intense multidisciplinary study because of their unique physical and chemical properties, comprising one atom thick sheet of graphite rolled up into a seamless cylinder with a diameter of the order of a nanometer and length-to-diameter ratio exceeding 1000. The name of CNTs is based on their nanometer diameter, while they can be up to several millimeters in length. CNTs represent a very interesting carbon material that offers all the advantages of nanocarbons (extremely high surface area, adsorption sites, reactive surface sites, conductivity, etc.). Nowadays, nanomaterials such as CNT, 14 nano zirconium dioxide (ZrO2), 15 titanium dioxide (TiO2) 16,17 and silicon dioxide (SiO2) 18 have been successfully applied in SPE. 19 –21
Treated or oxidized-multiwalled CNTs (MWCNTs) as most general SPE sorbent also have quite poor selectivity for metal ions. 20 –24 It is well known that MWCNTs can introduce oxygen-containing functional groups like –COOH and -OH on their surface site after oxidation process. 14,23 –26 Hence, it is expected that MWCNTs modified with organic compounds would be more selective than untreated and oxidized-MWCNTs for the SPE of metal ions. 27 –33
To improve the durability and life time of solid phase and increase the selectivity of SPE procedure on CNTs, their chemical modification with a new chelating agent is required to replace the hydroxyl and carboxylic groups with new reactive sites.
In the present work, Chemically modified MWCNTs have been used as solid-phase materials for the preconcentration and separation of Cu2+, Ag+, Cd2+, Pb2+, Zn2+ and Mn2+ ions. The proposed sorbent was characterized with Fourier transform infrared spectrum and scanning electron microscopic analysis that showed homogenous surface of sorbent. The analytical parameters for quantitative recoveries of analytes were investigated.
Experimental
Reagents and solutions
MWCNT with the highest purity available was purchased from Merck (Darmstadt, Germany) and was used as received without any purification. Acids and bases’ (with the highest purity available from Merck) solution was prepared in double-distilled deionized water. Palladium chloride and nitrate salts of cadmium (Cd), mercury, copper (Cu), zinc (Zn), Mangenese (Mn), calcium (Ca), strontium, barium, silver (Ag), sodium (Na) and potassium (all from Merck) were of the highest purity available and used without any further purification.
Preparation of MWCNT-MPSTO
Oxidization, activation and purification of MWCNTs were carried out according to our previous publication 34,35 . In order to activate, MWCNTs was suspended in 10% (v/v) hydrochloric acid (HCl) while stirring for 2 h. The treated MWCNTs were filtered and dried at 80°C for 5 h. Then for the oxidation of MWCNTs, 1 g of activated MWCNTs was added into 250 mL of 32.5% (w/v) nitric acid (HNO3) solution and stirred for 36 h at room temperature. The mixture was filtered and thoroughly rinsed with distilled deionized water until it attains neutral pH and dried at 80°C for 2 h. Oxidation of MWCNTS creates some functional groups such as hydroxyl and phenolic groups and carboxylic acid groups on MWCNT structure surface.
Then, oxidized MWCNT was chemically modified to the MWCNT supported (3-mercaptopropyl) silanetriolate (MWCNT-MPSTO) including thiol groups by refluxing 0.1 g of oxidized MWCNTs with 1.8 mL of 3-(trimethoxysilyl) propane-1-thiol (TMSPT) in dry dichloromethane (20 mL) for 24 h and then the solid was filtered and dried at room temperature. The schematic diagram of preparation of this new sorbent is presented in Figure 1.

Schematic diagram for the synthesis of MWCNT-MPSTO. [MWCNT-MPSTO: multiwalled carbon nanotube supported (3-mercaptopropyl) silanetriolate].
A short glass column with an inner diameter of 0.5 cm and a length of 50 cm, equipped with porous frits was filled up to a height of about 1 cm with a suspension of 0.1 g of MWCNT-MPSTO. The sorbent was preconditioned by the blank solution prior to each use. Following the application, column was rinsed with water and stored for the next experiment.
Instruments
A Shimadzu 680A atomic absorption spectrometry with air–acetylene flame and hallow cathode lamps and deuterium background corrector were used for the analysis of Cu, Ag, Cd, Zn, Pd and Mn. All the instrumental settings were those recommended in the manufacturer’s manual book. The pH was determined with a Metrohm 691 pH/ion meter with a combined glass–calomel electrode.
Preconcentration procedure
An off-line column procedure was applied for the preconcentration. A 250-mL of sample solution (100 mL) containing 50 mg of target metal ions in the pH value in the range of 3.0–8.0 (adjusted by the addition of NaOH and/or HCl solution) was prepared. The solutions were passed through the column containing 0.1 g of modified MWCNT at a flow rate of 2.5 mL min−1 with gravity, to effect the deposition of analyte. The uptake of metal ions was then eluted with 5 mL of 4.0 mol L−1 HNO3 with a 1.5-mL min−1 flow rate. The concentrations of the metal ions in the eluent were determined by FAAS.
Analysis of the real samples
An amount of 20 g of real samples (rosemary, spinach and thymus; purchased from Shiraz, Iran) were heated in the silica crucible for 3 h on a hot plate and the overcooked material was transferred into a furnace for overnight heating at 650°C. The residue (5 g) was cooled, treated with 10.0 mL of concentrated HNO3 and 3 mL of 30% (w/w) hydrogen peroxide and again kept in a furnace for 2 h at the same temperature so that no organic compound traces are left. The final residue was treated with 3 mL of concentrated HCl and 2–4 mL of 70% (w/w) perchloric acid and was evaporated to fumes, so that all the metals change to their respective ions. The solid residue was dissolved in water, filtered and by keeping the pH at 6.0 made up to 250 mL by the addition of KOH and/or HNO3. The dissolved solution was suitably diluted and metal concentrations were determined by FAAS after the application to preconcentration procedure given above.
Results and discussion
It can be seen from the field emission scanning electron microscopic images of MWCNTs, oxidized MWCNTs and chemically modified MWCNTs(MWCNTs-MPSTO (Figure 2) that the surface morphology of MWCNTs is homogeneous and relatively smooth, while the morphologies of oxidized and the chemically modified MWCNTs are seemed to be without any aggregation and approximately uniform in size distribution. For obtaining maximum accurate and precise signal for the evaluation of analyte ion (Pd, Cu, Ag, Cd, Zn and Mn) contents, the influence of parameters such as pH of sample, amount of solid phase, type, volume and concentration , type and volume of eluent and flow rate on metal ions recoveries must be optimized.

SEM of MWCNTs(a), Oxidized MWCNT(b) and Chemically modified MWCNTs(c)[SEM: scanning electron microscopy; MWCNT: multiwalled carbon nanotube]
Thermal analysis of adsorbent
The adsorbent was subjected to thermogravimetric analysis (Figure 3) from room temperature to 1000 C. As shown, the adsorbent is stable till 270 C indicating the lack of water as hydrated molecules. Then its decomposition is initiated in one step from 270 to 925°C so that the final residue is 5.57% that may be related to SiO2. Therefore, the percentage of silicon (Si) in adsorbent is suggested to be 2.59% based on SiO2 residual content. This suggested amount of Si is approximately in agreement with 30% (w/w) support of TMSPT on MWCNTs. The thermodynamic activation parameters of decomposition process of adsorbent including activation energy (E*), enthalpy (ΔH*), entropy (ΔS*) and Gibbs free energy change in the decomposition (ΔG*) can be estimated graphically by employing the Coats–Redfern relation (equation (1))
36
–39
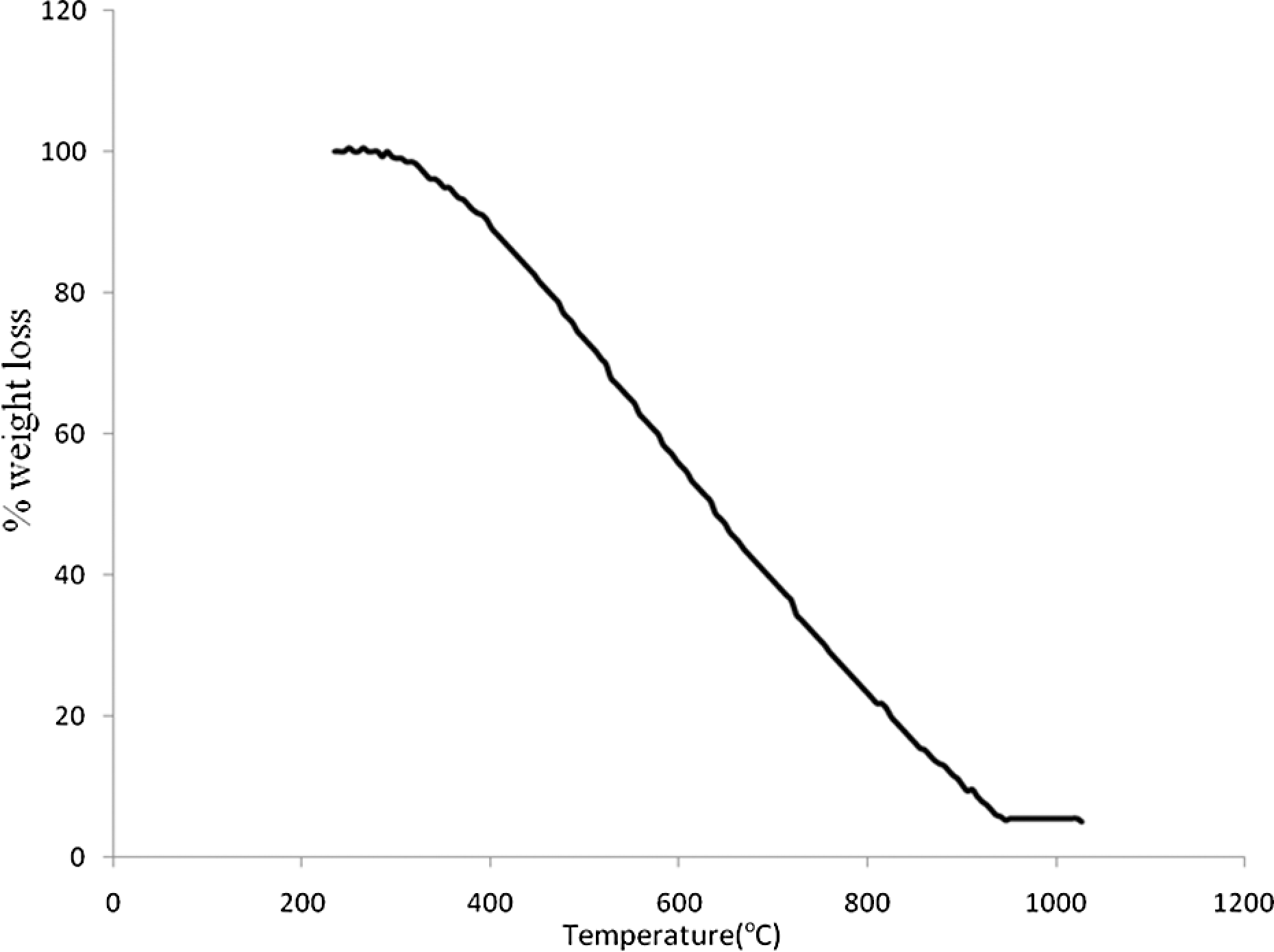
Thermogravimetric plot of adsorbent.
where W
f is the mass loss at the completion of the reaction step, W is the mass loss up to temperature T; R is the gas constant, E* is the activation energy in kilojoules per mole, θ is the heating rate (5°C min−1). A graph of the left-hand side of equation (1) against 1/T gives a slope and intercept equal to E* and A (Arrhenius constant), respectively. The entropy of activation (ΔS*), enthalpy of activation (ΔH*) and the free energy change of activation (ΔG*) are calculated using the following equations
The data including the activation energies of decomposition E*, ΔS*, ΔH* and ΔG* are shown in Table 1.
Thermodynamic parameters of the thermal decomposition of solid phase

Effect of pH
The pH of the sample, by influencing the present state of analytes in solution as ionic or molecular form, plays an important role in the SPE procedure and significantly influences the extraction efficiency of the target analytes. The oxidation of carbon active with HNO3 and its chemical modification with tri methoxysilylpropanethiol leads to the surface modification with thiol groups (–SH). The sorbent surface charge depends on the pH of the surrounding electrolyte and the pH of zero charge (PZC) at which the surface has zero net charge. The pH higher than pHPZC, the negative charge on the surface of the chemically modified MWCNTs provides electrostatic interactions that are favorable for the retention of cationic species. The pHPZC of the proposed modified MWCNT sorbent was determined to be 6.
The effect of pH on the sorption of metal ions by this new sorbent was investigated by conducting a set of similar SPE procedure by passing 250 mL of the sample solution containing 50 µg of all the understudied metal ions in the pH range of 2.0–8.0. Then, the retained metal ions efficiently were eluted using 5 mL of 4.0 mol L−1 of HNO3 and respective results are presented in Figure 4. As it can be seen, sorption of tested analytes increased by increasing the pH till 6.0. The low recovery in the acidic region can be attributed to the competition between hydrogen and metal ions for binding reactive sites. In this study, no correlation was found between the radii of the hydrated metal ion and its sorption.

Effect of pH on metal ions recovery.
Effect of the amount of chemically modified MWCNT
In the subsequent work, it is necessary to investigate the influence of the amount of MPSTO and solid phase on metal ions recoveries. In this regard, a set of similar experiments was undertaken at a fixed value of all variables and various amounts of solid phase and MPSTO in the range of 0.2–1.2–0.12 g and 0.5–2.5 g and results are presented in Figures 5,6. It was found that by addition of amount of solid phase and MPSTO until 0.8 and 1.8 mL, respectively, recoveries were increased and at higher value a decrease in recoveries was seen. At a higher amount of solid phase probably eluting solution is insufficient for efficient elution of target ions. At a lower amount, the extent of complexation and retention of metal ions on the chemically modified MWCNT are low and hence the recoveries were decreased.
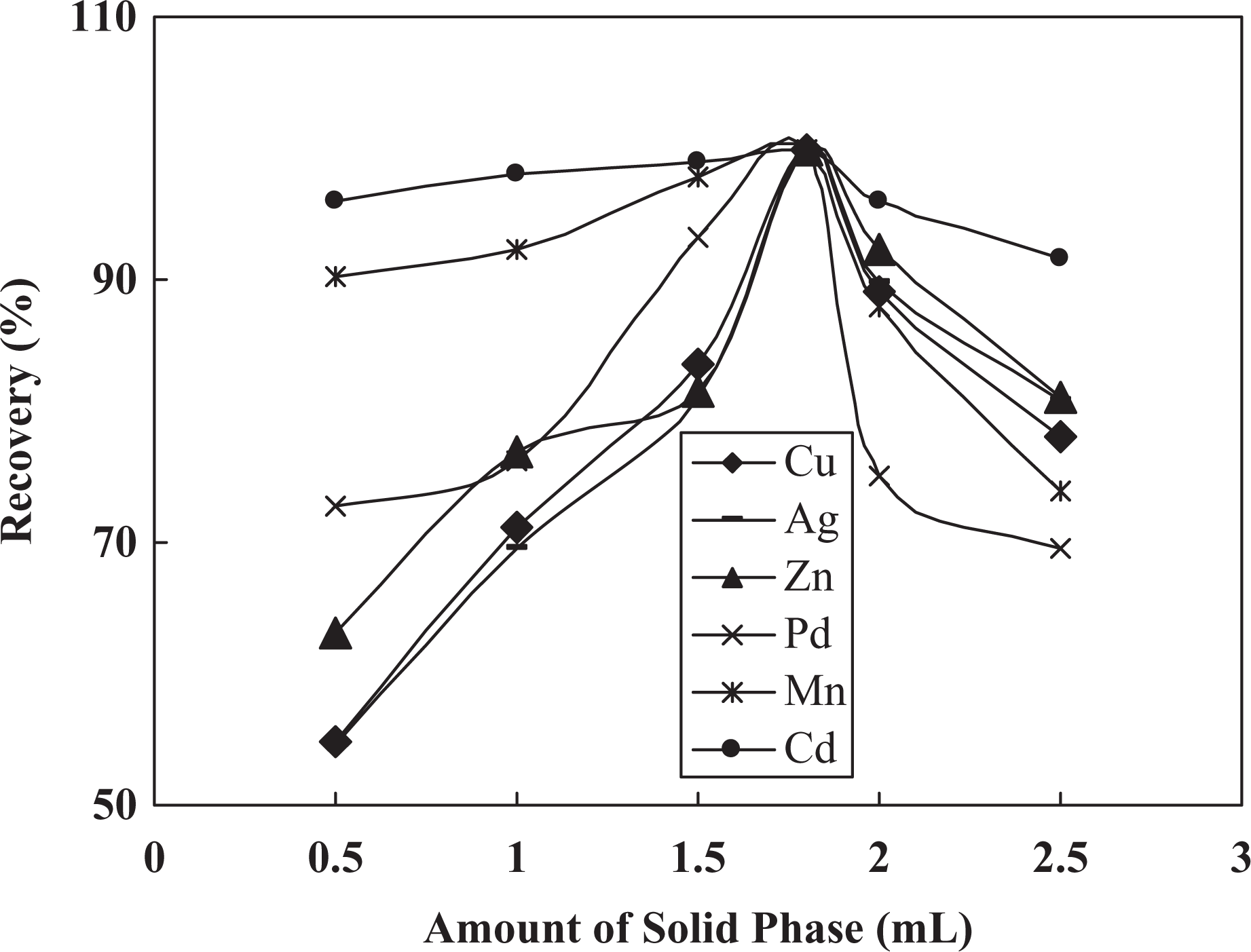
Effect of amount of MPSTO on metal ions recoveries.
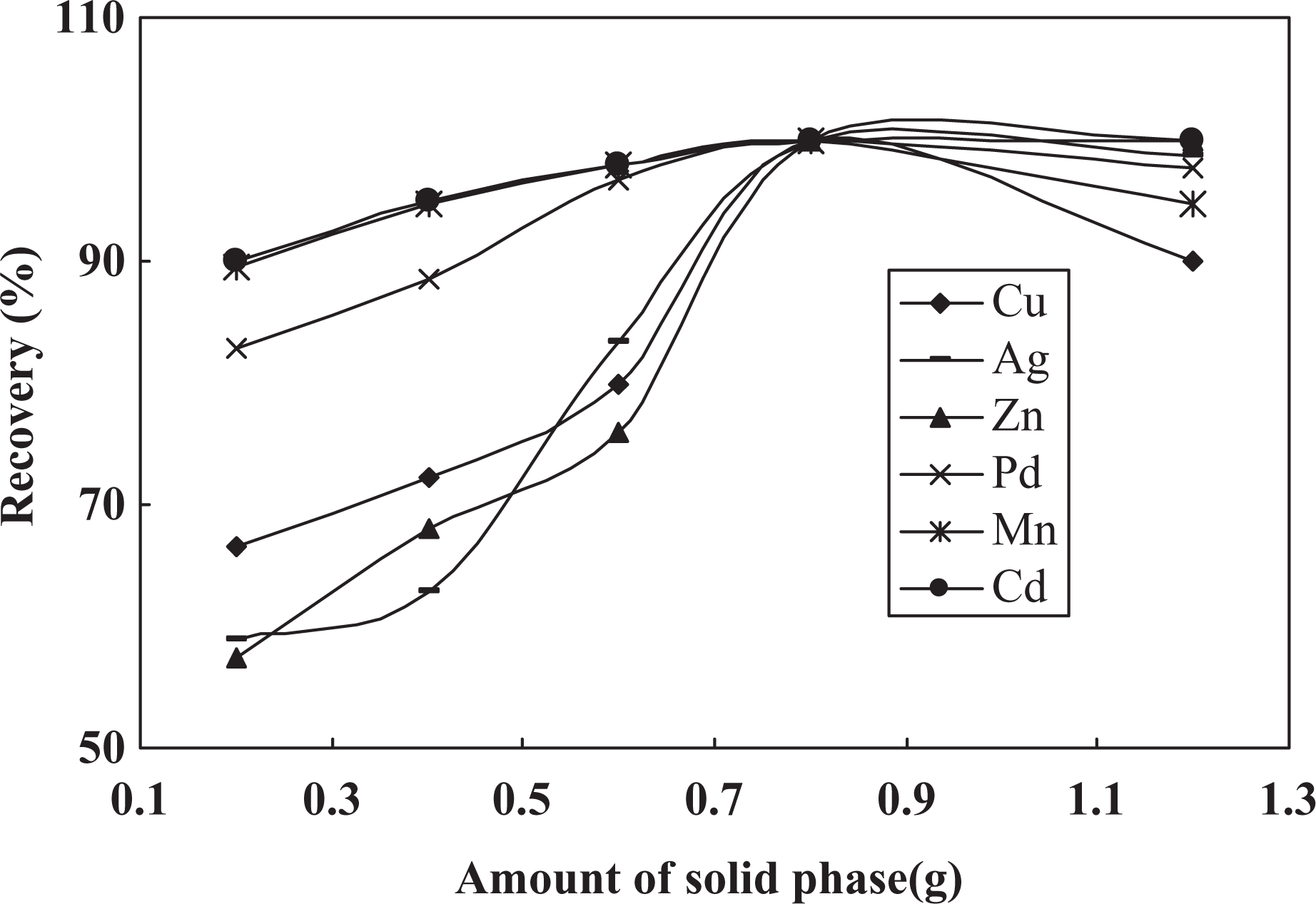
Effect of amount of solid phase on metal ions recoveries.
Effect of type and amount of elution solution
The other important factor that affects the preconcentration procedure is the type, volume and concentration of the eluent used for the stripping of metal ions from the sorbent. Optimization of the elution conditions was performed in order to obtain the maximum recoveries with the minimal concentration and volume of the eluent. The different concentrations of HNO3, HCl and sulfuric acid were tested to remove the bound metal ions from the sorbent and the respective results are presented in Table 2. As can be seen, 5 mL of 4 mol L−1 HNO3 solution was found to be satisfactory. (recoveries >95%) for all understudy metal ions.
Condition of eluent on metal ion recovery

Cu: copper; Ag: silver; Cd: cadmium; Zn: zinc; Pd: palladium; Mn: manganese; HCl: hydrochloric acid; H2SO4: sulfuric acid; CH3COOH: acetic acid; H3PO4: phosphoric acid; HNO3: nitric acid.
Effect of flow rates on metal ion recoveries
The retention of an element on an adsorbent depends on the flow rate of the sample solution. Therefore, the effect of the sample flow rate on the recoveries of the metal ions was investigated under the optimum conditions (pH, eluent type, etc.). The sample solution was passed through the column with the flow rates adjusted in a range of 1.0–6.0 mL min−1 by gravity. Carrying out the SPE procedure based on the flow rate of the sample 0.5-2.5 g and solution of <2.5 mL min−1 and elution solution flow rate of <1 mL min−1 leads to obtain quantitative and reproducible recoveries of metal ions.
Sample volume and enrichment factor
To obtain reliable and reproducible analytical results and high concentration factor, it is very important to get satisfactory recoveries for all the target analytes in a large volume of sample solutions as possible. The possibility of up taking low concentrations of analyte from large volumes of solution, the effect of sample volume (range of 250–2000 mL) on the retention of metal ions was investigated and seen that the quantitative sorption (≥95%) of investigated metal ions was obtained at 1500 mL and at a greater volume percent of adsorption decreased. The enrichment factor for simultaneous preconcentration and extraction is 300 (the ratio of the maximum sample volume (1500 mL) to the eluent volume (5.0 mL)).
Effect of interfering ions
In order to examine the effect of the various tested metal ions in the optimum conditions, SPE of these metal ions in the presence of some selected metal ions has been carried out, which admits us to have an intensive selectivity study and evaluate the possible interference of some metal ions in the process of selective SPE of understudied metal ions. Interferences that may be concomitant with these ions were investigated. The effects of Ca, magnesium and other alkali and alkaline earth ions are presented in Table 3. As can be seen, these ions interfering up to 1000-fold are negligible and this high selectivity enables the method for accurate and precise determination of analyte ions in real samples.
Effects of the matrix ions on the recoveries of the metal ions (N = 3)

Na: sodium; Hg2+: mercuric ion; K: potassium; Cl: chlorine; SO4: sulfate; CH3COO−: acetate; Fe: iron; Mg2+: magnesium ion; HCO3 −: bicarbonate; PO4 3−: phosphate; NaCl: sodiunm chloride; KCl: potassium chloride; (NH4)2SO4: ammonium sulfate; CH3COO Na: sodium acetate; MgCl2: magnesium sulfate; NaHCO3: sodium bicarbonate; Na3PO4: sodium phosphate.
Analytical features
The analytical features of the proposed method such as precision, linear range of calibration curve and limit of detection were also examined. The calibration curves were obtained using 100 mL of solutions at pH 6 at the optimum condition (Table 4). The results are shown in Table 3, which indicate a wide linear range and applicability of method for trace metal enrichment. The adsorption capacity (as the largest amount of analytes can be sorbed on fixed amount of sorbent) was adopted from the procedure recommended by contacting 10 mL of understudy metal ion solutions with the concentrations (50–300 mg mL−1) at an appropriate pH (Table 4). The good performance of the method shows its applicability for trace metal enrichment with high recoveries and low RDS.
Specification of the present method at optimum conditions for each element

Cu: copper; Ag: silver; Cd: cadmium; Zn: zinc; Pd: palladium; Mn: manganese; RSD: relative standard deviation.
Application
The procedure was applied for the determination of understudy metal ions in different samples including natural food samples, and the results are presented in Table 5. The recovery of spiked samples is satisfactorily reasonable and was confirmed using the standard addition method. The high recoveries and low RDS indicate the capability of the system for the determination of analytes in different samples.
Recovery of analytes from samples by using proposed method (N = 3)

The values for samples are expressed in microgram per gram. Cu: copper; Ag: silver; Cd: cadmium; Zn: zinc; Pd: palladium; Mn: manganese; RSD: relative standard deviation.
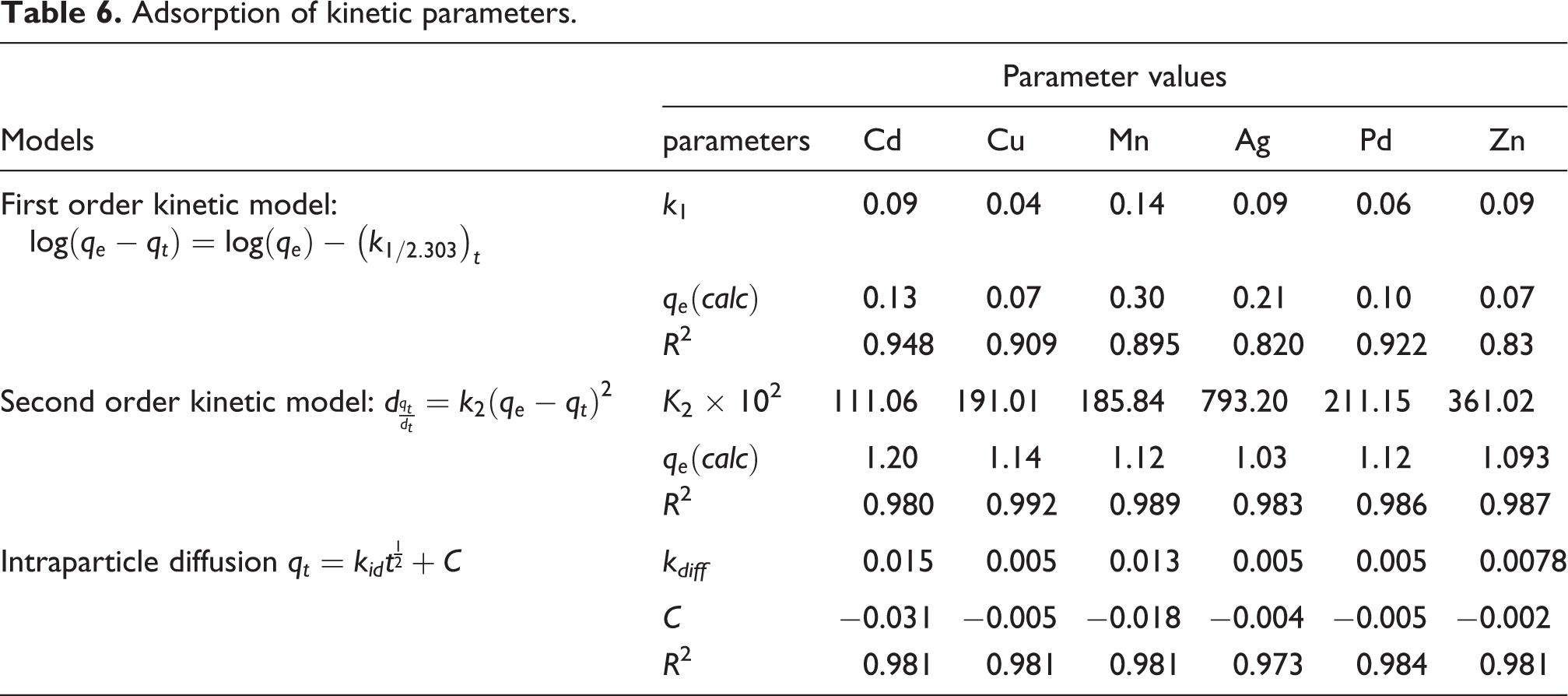
Adsorption kinetics
In this regard, the respective experimental data correspond to change in removal percentage and the absorbance of solution was fitted using different kinetic models including pseudo first order, pseudo second order, Elovich and intraparticle diffusion models, and the properties of each model are presented in Table 6.
Adsorption of kinetic parameters
Adsorption isotherm
Adsorption isotherms are mathematical models that describe the distribution of the adsorbate species among liquid and sorbent, based on a set of assumptions that are mainly related to the heterogeneity/homogeneity of adsorbents, the type of coverage and possibility of interaction between the adsorbate species. The Langmuir model assumes that there is no interaction between the adsorbate molecules and the adsorption is localized in a monolayer. The Freundlich isotherm model is an empirical relationship describing the adsorption of solutes from a liquid to a solid surface, and assumes that different sites with several adsorption energies are involved. The slopes of the linearized Tempkin, Freundlich and Langmuir plots were used to calculate the adsorption constants tabulated in Table 7 that higher correlation coefficients show the Langmuir model fits the adsorption data better than the other model. As it can be seen, high correlation coefficients of langmuir models and its high adsorption capacity show suitability of this new sorbent for simultaneous enrichment and determination of analytes
Isotherm parameters

Adsorption thermodynamics
The thermodynamic parameters, the values of enthalpy (
The parameters can be calculated from the binding, K°, obtained from the Langmuir’s equation, using the following equations

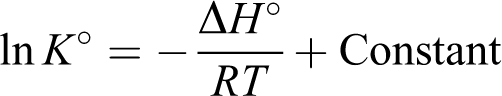
The values of 
where R (8.3145 J mol−1 K−1) is the ideal gas constant, and T (K) is the temperature. Relevant data calculated from above equations are tabulated in Tables 8 and 9. The adsorption of understudy metal ions increases with the rise in temperature and the value of
Thermodynamic parameters

Thermodynamic parameters

Cu: copper; Ag: silver; Cd: cadmium; Zn: zinc; Pd: palladium; Mn: manganese.
Conclusion
The results presented in this article demonstrate the usability of chemically modified MWCNT for the preconcentration of trace metal ions. The proposed procedure provides a simple, sensitive, precise, reliable and accurate technique for the preconcentration and determination of analytes. The recoveries of analytes studied were nearly quantitative (>95%) with RDS lower than 4%. The recoveries for these elements were very satisfactory, and it is evident for the reliability of the proposed method for the analysis of food samples. At optimum values of all variables, the sorption process follows the Langmuir isotherm model with high adsorption capacity. The mechanism of metal ions’ sorption on the proposed sorbent follows the pseudo second order kinetic model with an involvement of interparticle diffusion model.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.