Abstract
The present study was conducted to examine the chronic effects of potassium octatitanate fibers (trade name TISMO; chemical formula K2O·6TiO2) on the mouse lung and thoracic cavity. This method of infusion was employed to examine the direct effects of the fibers to the pleura. In the present study, 52- and 65-week experiments were employed to examine the long-term chronic effects after infusion of fiber-shaped TISMO into the thoracic cavities of A/J mice. Following this infusion, TISMO fibers were observed in the alveoli, indicating penetration through the visceral pleura. The additional histopathological detection of TISMO fibers in the liver, spleen, kidneys, ovary, heart, bone marrow, and brain of TISMO-infused mice indicated migration of the fibers out from the thoracic cavity. Atypical mesothelial cells with severe pleural proliferation were observed, but malignant mesotheliomas were not detected. This study demonstrated that intrathoracic infusion of TISMO fiber did not cause malignant mesothelioma but did cause severe chronic inflammation and proliferation of pleural mesothelial cells.
Introduction
The incidence of pleural malignant mesothelioma is on the increase and is anticipated to rise further in future decades as a result of widespread exposure to asbestos, particularly in less regulated countries. 1,2 An appropriate animal model of malignant mesothelioma is necessary for understanding the fundamental mechanisms of tumorigenesis and the development of new approaches for therapy. There have been reports of the induction of peritoneal mesothelioma by chemicals or fibers in conventional rats, and this model has been widely used for animal investigations of malignant mesothelioma. 3 –6 However, pleural malignant mesothelioma is more common than the peritoneal form in humans. 7 Therefore, a bioassay model featuring pleural lesions in otherwise normal animals would be preferable. But, there is only limited information on pleural mesothelioma induced in experimental wild-type animals using direct infusion of particles into the thoracic cavity.
In a previous study, we examined the influence of physical pulmonary collapse in mice by performing a thoracotomy followed by polymer gel infusion directly into the left cavity of the thorax. 8 In this model, we observed pronounced mesothelial cell reactions on the left lung surfaces and parietal pleura. In an associated experiment, 9 thoracotomy was followed by infusion of each of 3 kinds of test particles directly into the thoracic cavity of A/J mice. Fiber-shaped particles of potassium octatitanate (trade name TISMO; chemical formula K2O·nTiO2) and granule shaped, micro and nanosized particles of titanium dioxide (TiO2) were employed (administered at 1.5 mg [in 0.2 mL saline]/mouse). The experiment was terminated after 21 weeks to assess pleural reactions. The TISMO fibers are morphologically similar to asbestos and are often used as a friction material for cars and in precision filters. Although no reaction was observed with micro and nanosized particles of TiO2, the TISMO fibers induced a severe reaction with the accumulation of iron derived from endogenous sources. The results indicated that the risk of mesothelial cell reactions could not depend solely on particle size but rather also on shape. Whether induction of malignant mesothelioma might result in the long term could not be determined because of the relatively short experimental period.
In the present experiment, we assessed the development of pleural malignant mesothelioma in this model using either of 2 study intervals, one extending for 52 weeks (experiment 1) and another extending as long as possible (over 52 weeks; experiment 2). In experiment 1, coadministration of iron (III) nitrate enneahydrate dissolved in nitrilotriacetic acid disodium salt (Fe-NTA) and TISMO was examined because iron as a component of some types of asbestos or as a consequence of asbestos-induced pathology is a major candidate as an agent contributing to mechanisms underlying carcinogenesis. 10 Experiment 2 was performed not only to examine induction of malignant mesotheliomas with a longer experimental period but also to assess the possibility of dissemination of TISMO fibers to organs outside the lungs, including the brain.
Materials and Methods
Chemicals
Potassium octatitanate fibers (trade name TISMO-D; chemical formula K2O·6TiO2) were supplied by Otsuka Chemical Co. Ltd (Osaka, Japan) as fibers with length (mean dimension) <50 µm and width <2 µm. We had confirmed these dimensions in previous work using scanning electron microscope (SEM) imaging of the TISMO fibers.
9
The length and width of TISMO fibers on the previous SEM image, counted 21 fibers, were 16.6 ± 10.5 µm (average ± standard deviation) and 0.7 ± 0.9 μm, respectively. For administration, the fibers were suspended in saline (isotonic sodium chloride solution; Otsuka Pharmaceutical Factory, Inc, Tokushima, Japan). The picture of the slide with test solution, 3 mg of TISMO particles suspended in 0.2 mL saline, is shown in Figure 1. Some of the TISMO fibers are aggregated, on the other hand, separated single fibers are also observed. Fe-NTA was formulated using nitrilotriacetic acid disodium salt (lot no. M9H6941) purchased from Nacalai Tesque, Inc (Kyoto, Japan), iron (III) nitrate enneahydrate (lot no. CDM1504) purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan), and sodium carbonate, anhydrous (Na2CO3; lot no. M5K8672) purchased from Nakarai Chemicals Ltd (Kyoto, Japan).
The slide with test solution, 3 mg of TISMO particles suspended in 0.2 mL saline. Some of the TISMO fibers are aggregated, on the other hand, separated single fibers are also observed. Scale bar is 100 μm.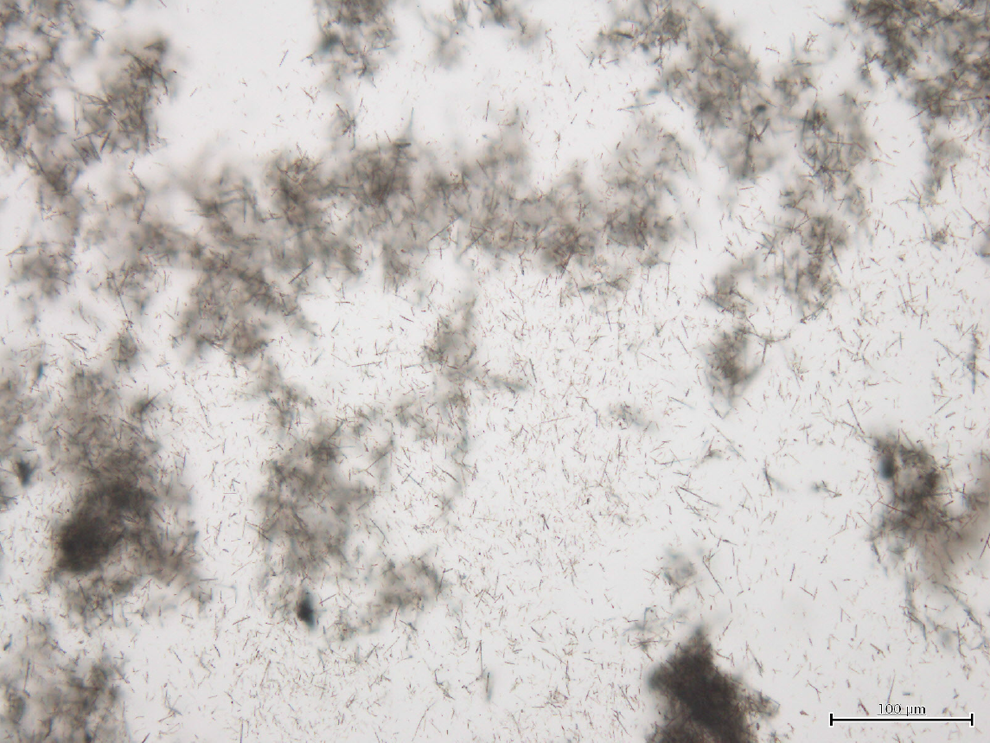
Animals
Female A/J mice (5 weeks of age at arrival) were purchased from Japan SLC, Inc (Shizuoka, Japan) and maintained in the Division of Animal Experiments, Life Science Research Center, Kagawa University, according to the Institutional Regulations for Animal Experiments. The regulations included the best considerations on animal welfare and good practice of animal handling contributing to the replacement, refinement, and reduction of animal testing (3Rs). The protocols used for these experiments were approved by the Animal Care and Use Committee of Kagawa University. The animals were housed in polycarbonate cages on bedding consisting of recycled paper chips (EchoChip, CL-4163, CLEA Japan, Inc, Tokyo, Japan). Mice were provided with free access to drinking water and a basal diet, Oriental MF (Oriental Yeast Co., Ltd, Tokyo, Japan), and were maintained under controlled conditions of humidity (60 ± 10%), lighting (12-hour light/dark cycle), and temperature (24 ± 2°C). Animals were acclimated for 2 weeks prior to the start of the experiments as described here.
Experimental Design and Tissue Preparation
Experiment 1
A total of 80 A/J mice at 7 weeks of age were divided into 5 groups of 20, 20, 20, 15, and 5 mice each (groups 1-5, respectively). Group 5 is the untreated control group. At the beginning of the experiment, all mice of groups 1 to 4 underwent a left thoracotomy, groups 1 and 2 being administrated 3 mg of TISMO particles suspended in 0.2 mL saline/mouse into the left thoracic cavity, and groups 1 and 3 being administrated 0.3 mL of Fe-NTA. The test solution of Fe-NTA was prepared as follows: iron (III) nitrate enneahydrate and nitrilotriacetic acid disodium salt were made up to 300 and 600 mmol/L, respectively, with deionized water and mixed together to give a 1:2 Fe-NTA solution. Finally, the Fe-NTA solution was neutralized to pH 7.4 with Na2CO3. Surgery for administration of TISMO: For anesthesia, each mouse was given an intraperitoneal injection of 0.2 mL of 10-fold diluted pentobarbital sodium (0.06-0.1 mL /10 g body weight; Somunopentyl, Kyoritsu Seiyaku Co., Tokyo, Japan). Under deep anesthesia, a skin incision (about 7 mm long) was performed on the left axilla. After confirmation of the location of the thoracic wall, thoracotomy was completed with an incision (approximately 5 mm long) between the ribs. The left lung was observed directly through the opened hole, and atelectasis was confirmed. After infusion of the test solutions into the left thoracic cavity, the skin was clipped together to close the thorax. A total of 12 mice (2 of group 1, 5 of group 2, 2 of group 3, and 3 of group 4) died just after the administration; these mortalities were presumed to be the result of surgery and/or anesthesia. The experiment was terminated at 52 weeks post-surgery, when all surviving animals were sacrificed under deep anesthesia. 9
At necropsy, the lungs, liver, and kidneys were removed after blood sampling via abdominal aorta. Removed organs were weighed, but the lungs could not be weighed successfully due to severe adhesions. After clotting of the blood samples, the serum was removed and submitted for the determination of ferrous (Fe) ion concentrations. Segments of the liver, including the right lateral and caudate lobes, were also frozen and submitted for determination of Fe ion concentrations. The examinations for Fe concentrations of blood serum and frozen liver from groups 1, 2, and 4 were assayed at SRL Inc (Tokyo, Japan). Other organs, including the remaining liver tissues, were fixed and processed for histopathological evaluation of hematoxylin and eosin-stained sections. Specifically, the organs were immersed in 10% neutral buffered formalin for 1 week, and 2 longitudinal slices from the left lobe of the lung and 1 orthogonally oriented hilum slice from each of the right lobes of the lung were processed for embedding in paraffin. When lung nodules were observed by gross inspection, additional slices were processed to allow histopathological assessment. Lung lesions were categorized as bronchioloalveolar adenoma (adenoma) and bronchiolo-alveolar adenocarcinoma (adenocarcinoma), in accordance with the established criteria provided in International Harmonization of Nomenclature and Diagnostic Criteria (INHAND). 11 The diagnosis of mesothelial lesions also followed the INHAND. Sections were also prepared for staining by the special Berlin blue and elastica-van Gieson (E.V.G.) methods to detect Fe3+ accumulation and elastic lamina of visceral pleura in the lung, respectively.
In addition, immunostaining of calretinin was performed to identify mesothelial cells in the lung. 12 The lungs were immunostained by the avidin–biotin complex (ABC) method; all processes from deparaffinization to counterstaining with hematoxylin were performed automatically using the Ventana Discovery staining system (Ventana Medical Systems, Arizona). The antimouse calretinin monoclonal antibody (clone 5A5), purchased from Novocastra Laboratories Ltd (Newcastle upon Tyne, United Kingdom), was employed at 1:100 dilution.
For observation by transmission electron microscopy (TEM), lung, liver, and kidney tissues from group 2 were deparaffinized in xylene, rehydrated in graded ethanol/water solutions, postfixed in 2.5% phosphate-buffered glutaraldehyde and osmium tetroxide, dehydrated in graded ethanol/water solutions and propylene oxide, and embedded in epoxy resin. Thick sections of 1 μm were taken from each block and stained with 0.5% toluidine blue. Ultrathin sections were cut, placed on uncoated or coated grids, and observed with a JEM-1400 electron microscope (JEOL, Tokyo, Japan).
Experiment 2
A total of 40 A/J mice at 7 weeks of age were divided into 2 groups of 20 mice each (groups 1 and 2). At the beginning of the experiment, each mouse of group 1 underwent a left thoracotomy and administration of 3 mg of TISMO fibers suspended in 0.2 mL saline. Anesthesia, surgery, and agent administration were performed for group 1 using the same technique as in experiment 1, and group 2 was the untreated control group. The experiment was terminated after 65 weeks (457 days) due to decreasing survival of the mice of group 1, and all surviving animals were sacrificed under deep anesthesia.
At necropsy, the lungs, liver, spleen, kidneys, uterus, ovary, heart, bone marrow from the sternum, and brain were removed. The weights of the organs, except for lungs demonstrating adhesion, were measured. Tissues were immersed in 10% neutral buffered formalin for 1 week and processed for histopathological examination as in experiment 1. To detect the fibers, polarized light microscopy was used for screening purposes with a BX51 microscope equipped with the Olympus Polarizer for Transmitted Light (U-P110), OLYMPUS Corp (Tokyo, Japan).
Statistical Analysis
Data for body and organ weights and Fe concentrations were analyzed by the Tukey-Kramer test (multicomparison test). The Fisher exact probability test was used for histopathological data to analyze increases in the incidence.
Results
Experiment 1
Body Weights (g) and Relative Organ Weights (%).
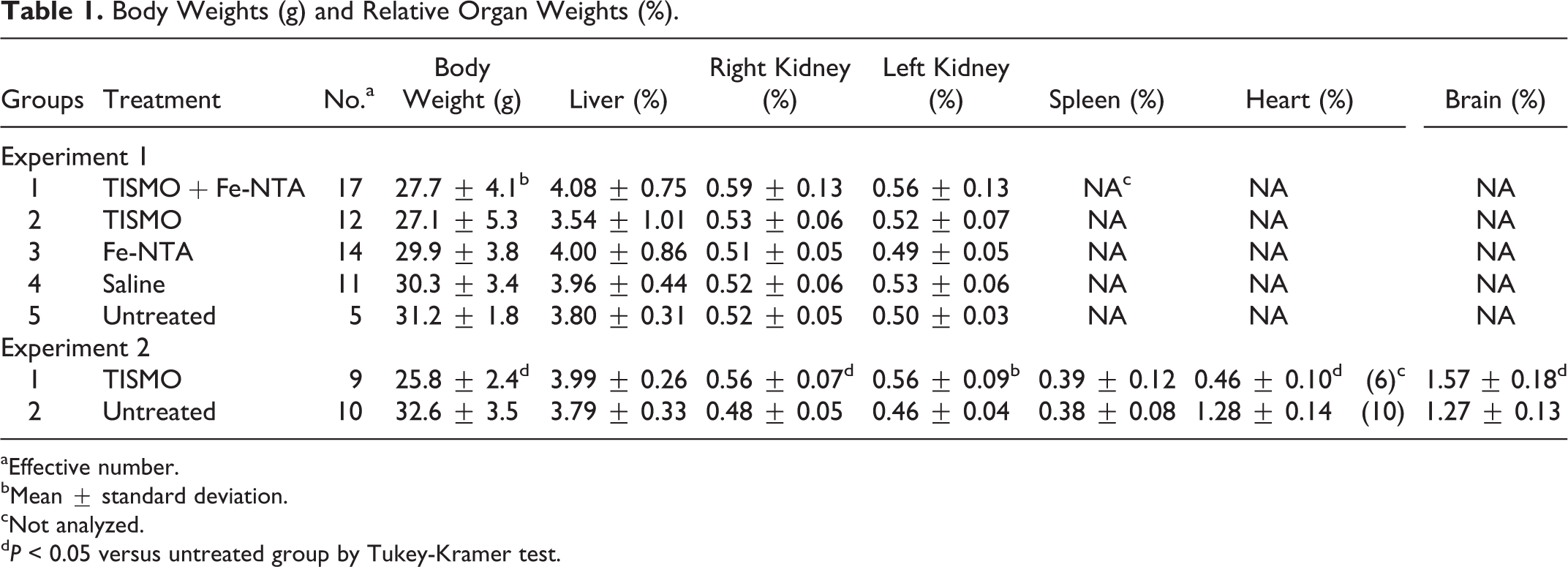
aEffective number.
bMean ± standard deviation.
cNot analyzed.
d P < 0.05 versus untreated group by Tukey-Kramer test.
Concentrations of Fe at 52 Weeks (Experiment 1).a
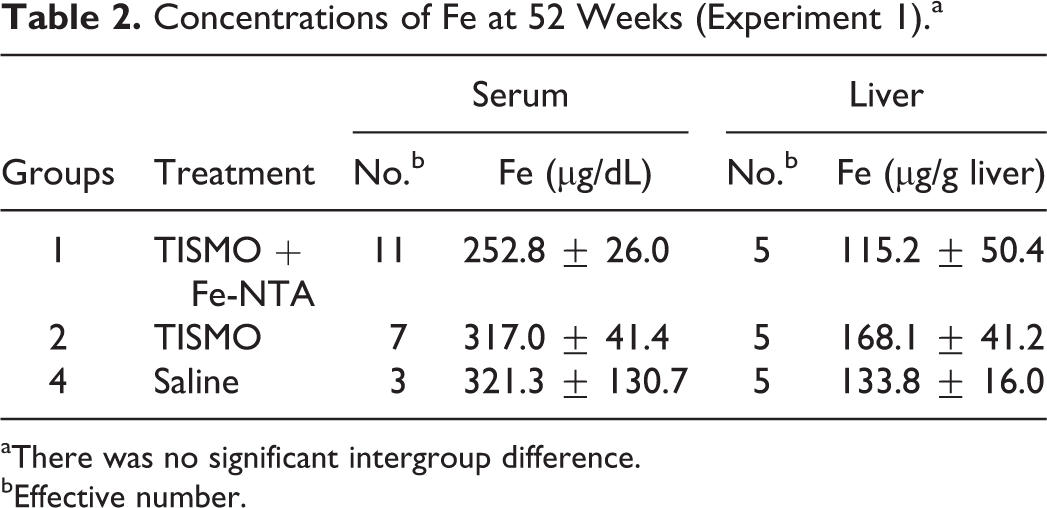
aThere was no significant intergroup difference.
bEffective number.
Grossly at autopsy, some of the mice in groups 1 to 4 demonstrated whitish lung nodules. In all the mice of the TISMO-treated groups 1 and 2, the infused TISMO fibers formed discrete masses in the cavity of the chest, with severe adhesions attaching the left lung (especially the left lobe) to the thorax. The right lung showed slight or no adhesion to the surrounding tissue and adequate expandability. The macroscopic findings did not seem to be affected by Fe-NTA.
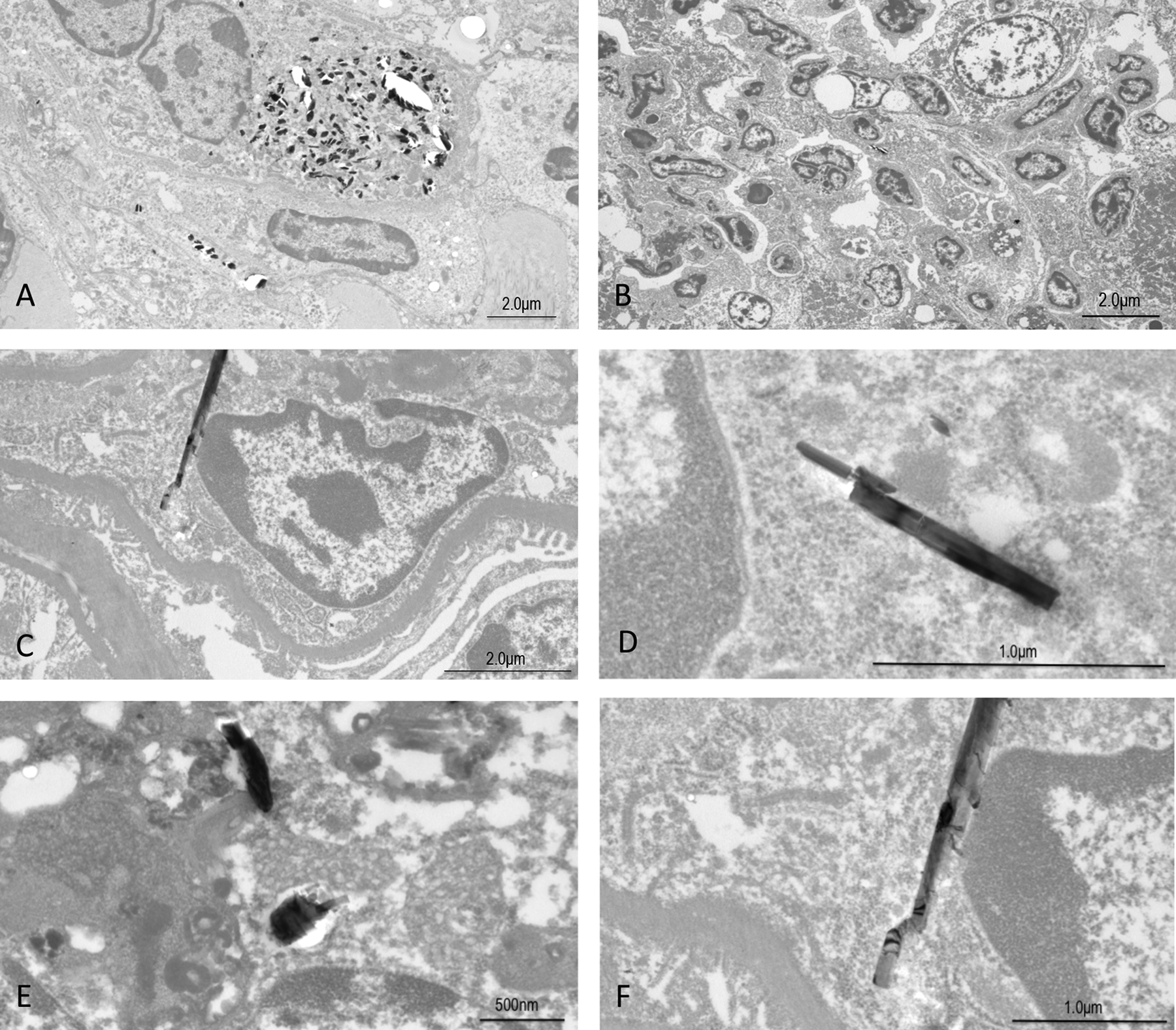
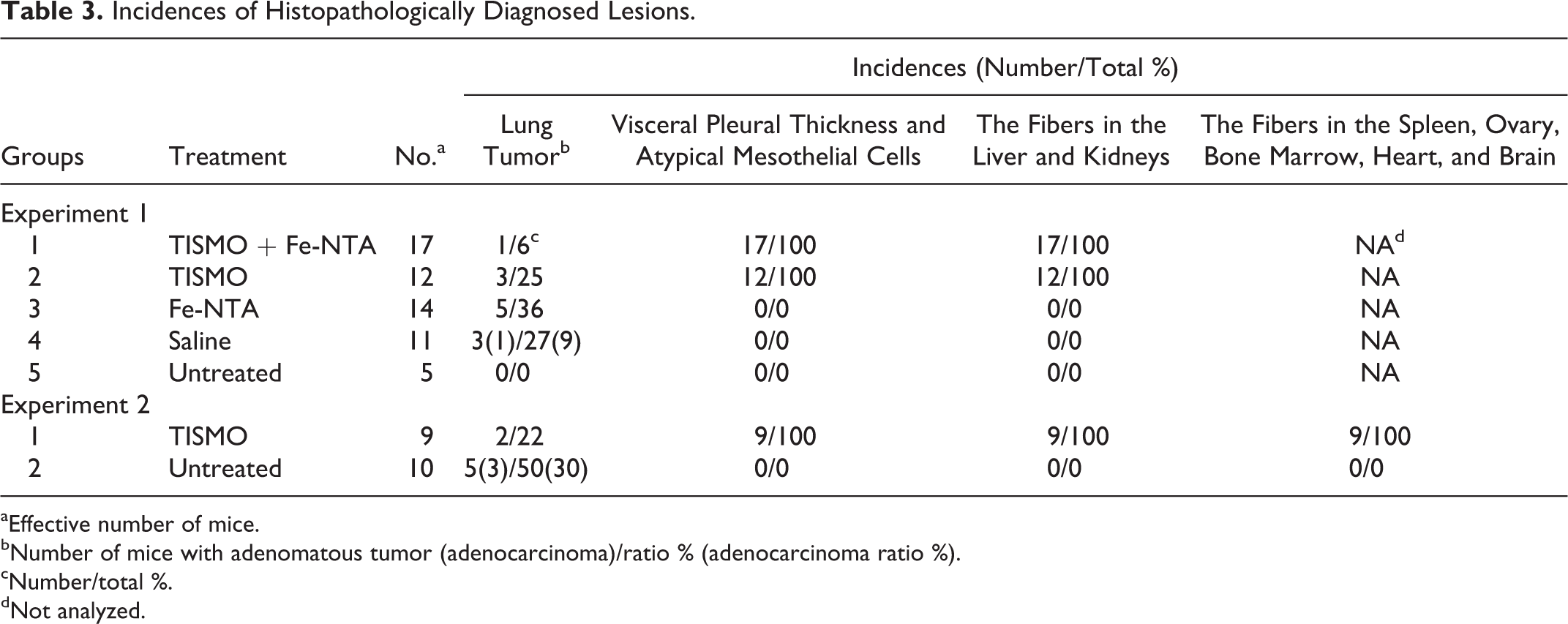
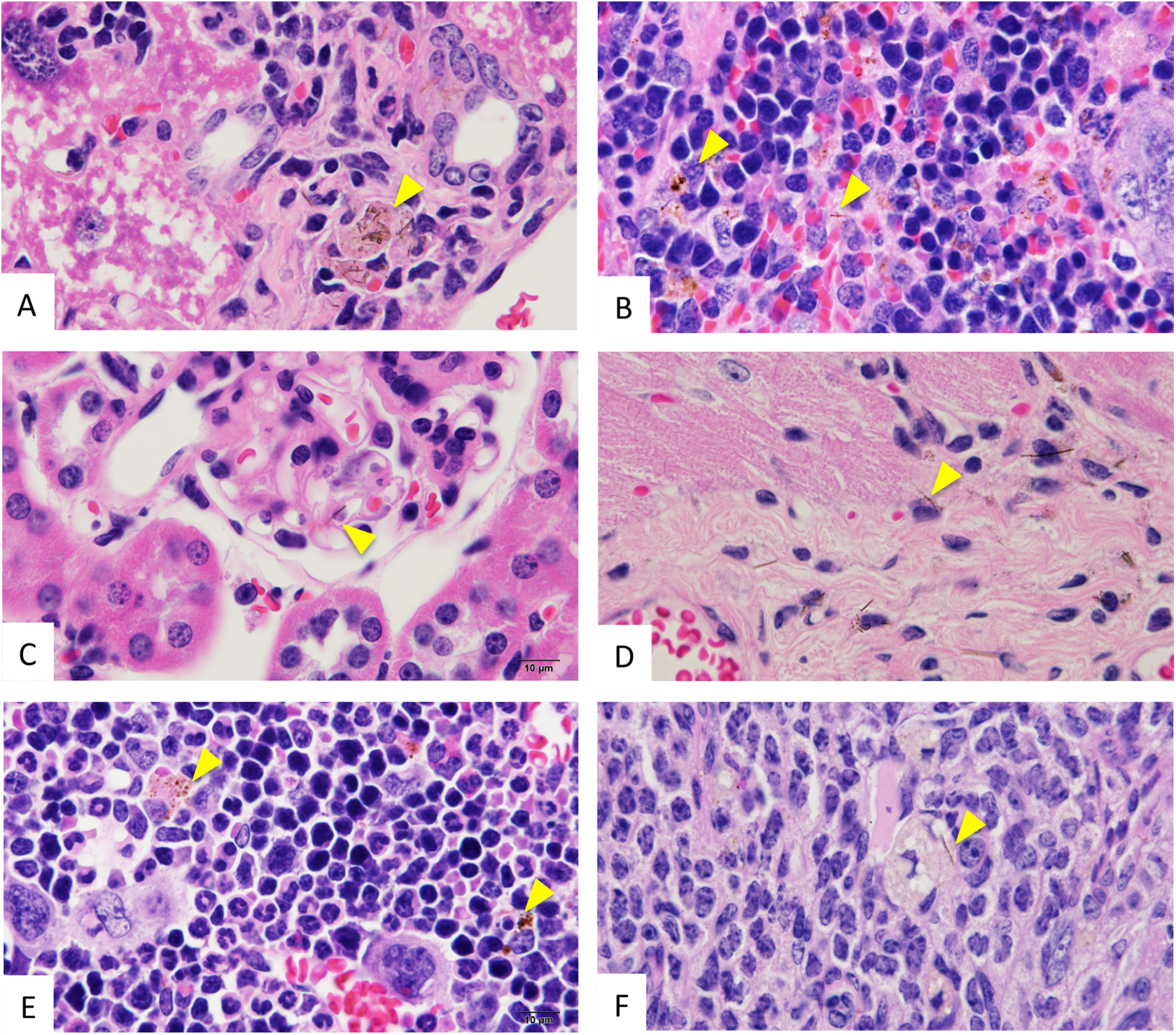
Histopathologically, the macroscopic lung nodules were confirmed to be adenomas or adenocarcinomas, but there were no significant intergroup differences in the incidence or multiplicity of these nodules. The incidences of histopathological findings are summarized in Table 3. Pleural thickening, characterized by chronic inflammation and prominent fibrosis, of the lungs and infolding of the pleura into the lungs were observed histopathologically in all mice of the TISMO-treated groups (groups 1 and 2). Several TISMO fibers, in either aggregated or isolated form, were observed in the alveoli, indicating penetration of the fibers through the pleura (Figure 2A). Positive spots of Berlin blue staining and calretinin-positive cells were also observed in pleural thickening lesions with TISMO fibers (Figure 2B and D). Grossly and histopathologically, there were no differences in the lungs of animals with or without Fe-NTA administration, whether comparing between the TISMO-treated groups (groups 1 and 2) or between the TISMO-untreated groups (groups 3 and 4). In TISMO-treated groups, groups 1 and 2, atypical mesothelial cells were observed diffusely in the thickened pleura with TISMO fibers of the lungs and positive for calretinin immunostaining with severe pleural mesothelial cell proliferation, but malignant mesotheliomas could not be confirmed (Figure 2C and D). Inflammatory reactions with prominent fibrosis were limited to the pleura; although the fibers were also detected (histopathologically) in the liver and kidneys, the hepatic and renal tissues did not exhibit inflammation (Figure 2E and F). In the liver, lymphocytic infiltration was observed around the fibers. In the TEM images of group 2, the fibers were detected not only in the lung but also in the liver and kidney (Figure 3). The size and shape of the fibers in the TEM images were the same (before and after injection), allowing assignment of these particles as TISMO fibers.
Histopathological findings for TISMO-treated group 2 at week 52 (experiment 1). A, The lung of group 2 with Elastica-van Gieson staining (40×); (B) the lung of group 2 with Berlin Blue staining (100×); (C) the lung of group 2 with hematoxylin and eosin (H&E) staining (400×); (D) the lung of group 2 with immunohistochemical staining of calretinin (100×); (E) the liver of group 2 with H&E staining (200×); and (F) the kidney of group 2 with H&E staining (200×). Pleural thickening and intrusion of the pleura were observed (A). Positive spots of Berlin blue staining and calretinin-positive cells were also observed in pleural thickening with TISMO fibers (B and D). Atypical mesothelial cells (yellow arrow) with positive for immunostaining of calretinin were observed with severe pleural proliferation (C and D). Fibers were detected in the liver and kidneys histopathologically, despite infusion only into the thoracic cavity (E and F, yellow arrows). Lymphocytic infiltration was observed around the fibers in the liver. Transmission electron microscopy (TEM) images of TISMO-treated group 2 at week 52 (experiment 1). A and D, Lung; (B and E) liver; and (C and F) kidney. The shape of the fiber in every lung (D), liver (E), and kidney (F) was the same so that they were all concluded to be TISMO fibers. Incidences of Histopathologically Diagnosed Lesions.
aEffective number of mice.
bNumber of mice with adenomatous tumor (adenocarcinoma)/ratio % (adenocarcinoma ratio %).
cNumber/total %.
dNot analyzed.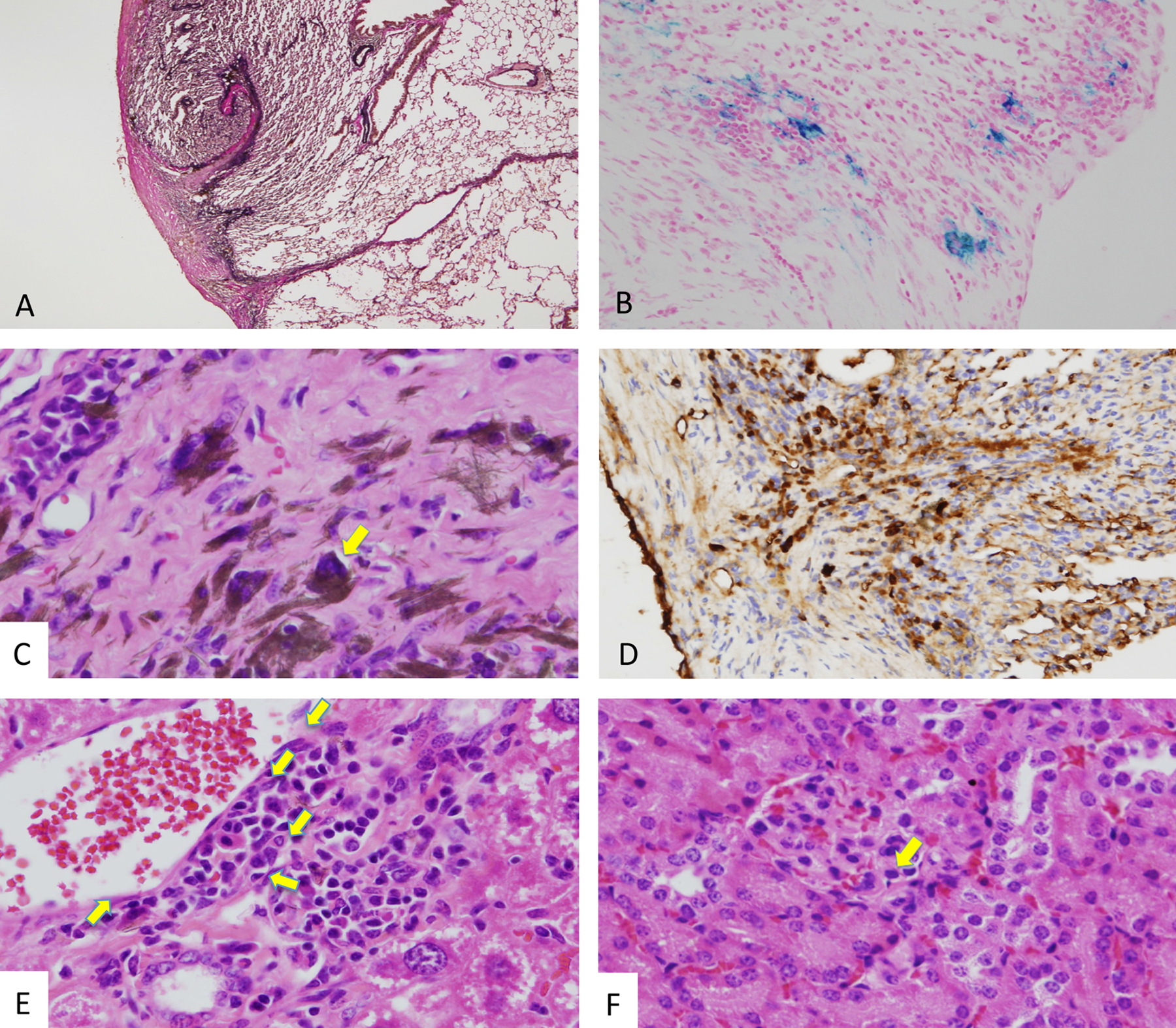
Experiment 2
From day 220, increasing mortality was observed in the mice of group 1 (TISMO treated; Figure 4). Therefore, the experiment was terminated on day 457, and the lung, liver, spleen, kidneys, ovaries, uterus, heart, brain, and bone marrow were examined. The surviving animals in both groups 1 and 2 demonstrated no remarkable change in their general condition during the experimental period. Final body and organ weights are shown in Table 1. The average body weight of the TISMO-treated (group 1) animals was significantly decreased compared with that of the untreated (group 2) animals. The relative weights (body weight normalized) of the kidneys, heart, and brain in group 1 were significantly increased compared with those in group 2. Lung weights could not be obtained due to the severe adhesion to adjacent tissues.
Number of surviving mice (experiment 2). From day 220, the mortality of the TISMO-treated group was increased. Because of the decrease in the effective number, the experiment was terminated on day 457 (week 65).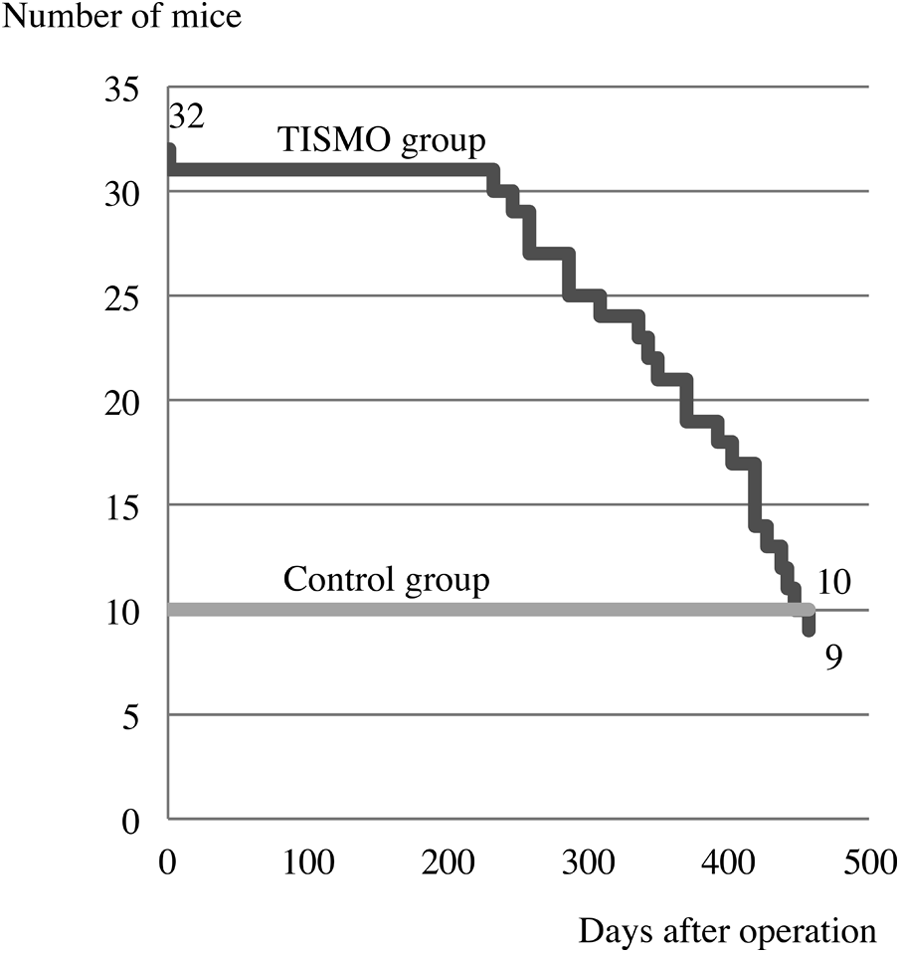
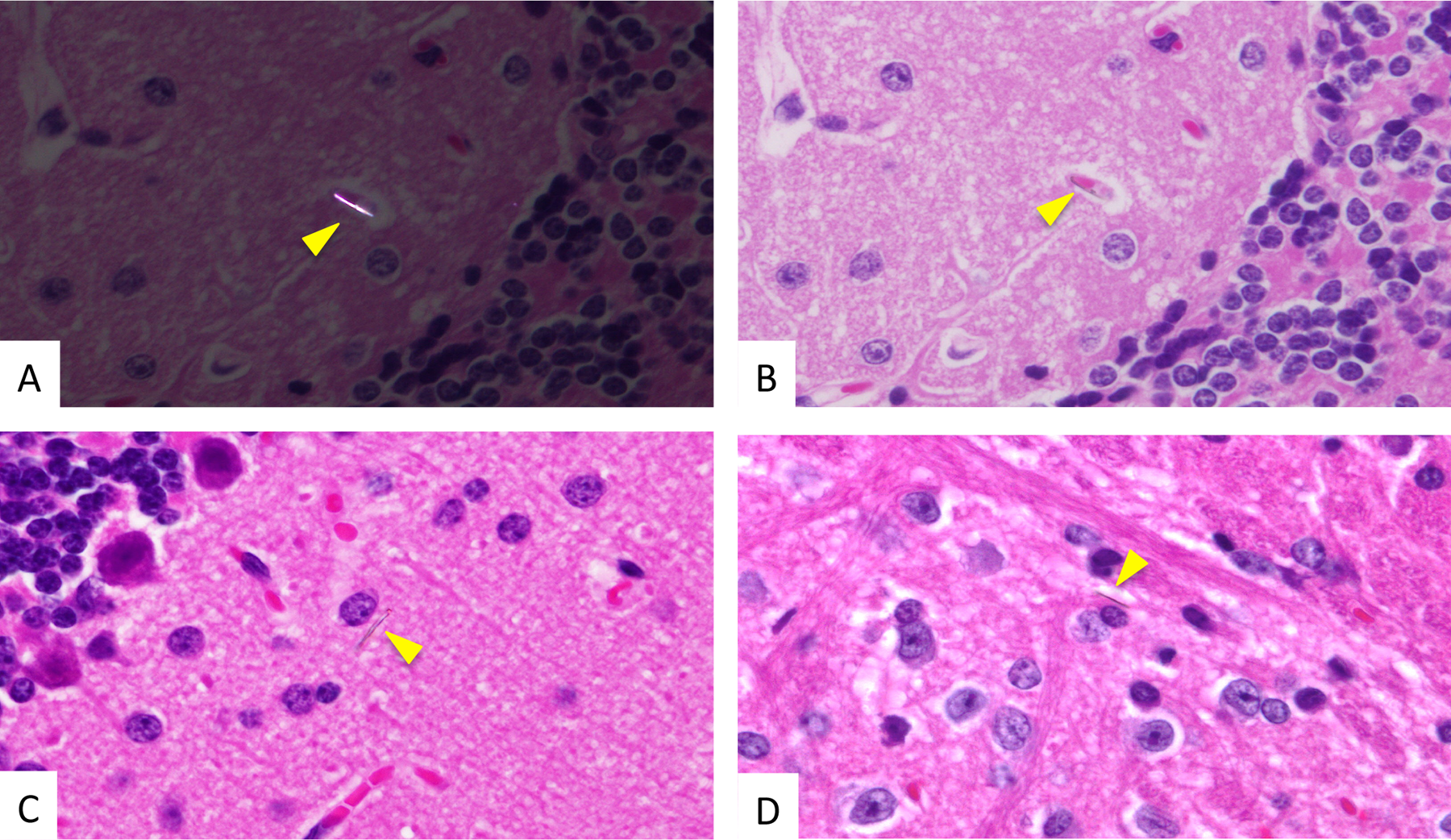
Grossly at necropsy, as in experiment 1, the lung whitish nodules were observed in some mice of groups 1 and 2. The infused TISMO fibers were associated with discrete masses and severe adhesions around the left lobe of the lungs in all of the TISMO-treated (group 1) mice. Histopathologically, the macroscopic lung nodules were confirmed to be adenomas or adenocarcinomas, but significant intergroup differences in rates were not detected. The incidences of histopathological findings are summarized in Table 3. In TISMO-treated group 1, pleural thickening and atypical cells were observed in the lung, but there were no findings indicative of malignancy, like invasion or metastasis (Figure 5). Although TISMO fibers were administered solely into the thoracic cavity, fibers were detected in several organs, including liver, kidneys, spleen, heart, ovaries, bone marrow (Figure 6), and brain (Figure 7). In the liver and spleen, the fibers were found within the foci of lymph cell accumulation. Fibers without infiltration of inflammatory cells were observed in the glomeruli of kidneys. In the heart, the fibers were observed in the pericardium close to the cardiac muscle. The fibers were also observed in myeloid tissues of the bone marrow and in the stroma of the ovaries. In the brain, the fibers were apparent in the microvascular network of the brain cortex (Figure 7). Some fibers were also located in the parenchyma of the brain cortex, outside the blood vessels. Such dissemination of TISMO fibers to remote organs from the left thorax was observed in all mice treated with TISMO fibers (Table 3).
Histopathological findings of lung for TISMO-treated group 1 at week 65 (experiment 2). A, The lung of group 1 with hematoxylin and eosin (H&E) staining (20×) and (B) the lung of group 1 with H&E staining (400×). In the lung, reactive pleural thickening (A) and atypical cells (B) were observed, but there were no findings indicating malignancy, like invasion or metastasis. Histopathological findings of liver, spleen, kidney, heart, bone marrow, and ovary for TISMO-treated group 1 at week 65 (experiment 2). A, The liver with hematoxylin and eosin (H&E) staining (400×); (B), the spleen with H&E staining (400×); (C) the kidney with H&E staining (400×); (D) the heart with H&E staining (400×); (E) the bone marrow with H&E staining (400×); and (F), the ovary with H&E staining (400×). In the liver (A) and spleen (B), fibers were observed in the foci of lymph cell accumulation. All yellow arrowheads in A-F indicate fibers. Fibers without infiltration of inflammatory cells were observed in the glomeruli of the kidneys (C). In the heart, fibers were observed in the pericardium close to cardiac muscle (D). Fibers were also observed in myeloid tissues of bone marrow (E) and stroma of the ovaries (F). Histopathological findings for the brain in TISMO-treated group 1 at week 65 (experiment 2). A-D, Hematoxylin and eosin (H&E) staining (1000×). A, Polarized light microscopy. A and B show the same location in the brain. Yellow arrowheads indicate fibers. Note the microvascular location of a fiber in the brain cortex (B). Some fibers were located in the parenchyma of the brain cortex outside of blood vessels (C and D).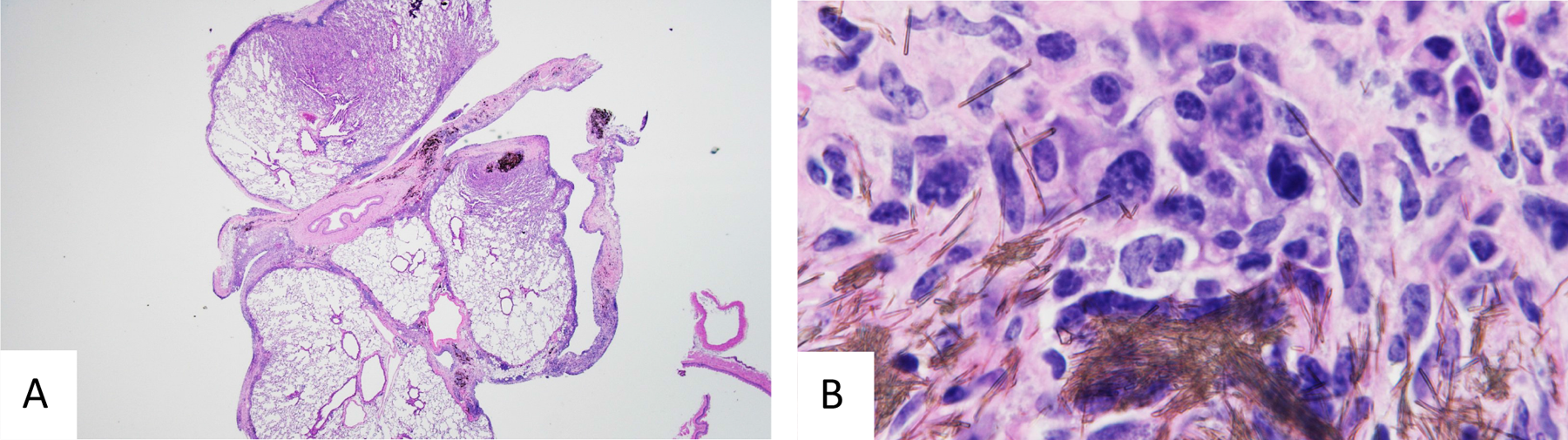
Discussion
The present study showed that the long-term exposure to TISMO fibers (initially infused into the thorax) resulted in lung damage. Although the fibers disseminated to many sites throughout the body, these fibers did not appear to significantly induce any inflammation or tumor development.
In experiment 1, 9 mice died before the scheduled sacrifice (excluding deaths in the perioperative period). In experiment 2, the mortality of the TISMO-treated mice increased from day 220, so this experiment was terminated on day 457 in order to ensure maintenance of sufficient experimental numbers. The commencement of increase in deaths in experiments 1 and 2 was almost the same at approximately day 220 (postsurgery/infusion). Although the exact cause of their death is unknown, it is probably due to chronic toxicity of TISMO fibers or respiratory dysfunction by severe adhesion of the lung. Although there were no significant intergroup differences in body weights in experiment 1, values for TISMO-treated groups 1 and 2 showed a tendency for decrease. In experiment 2, the body weights of the TISMO-treated (group 1) mice were significantly decreased compared with the untreated (group 2) mice. These presumably reflected toxicity of TISMO fibers for respiratory system in the thoracic cavity. The increase of relative weights of the kidneys, heart, and brain of group 1 in experiment 2 could be explained by body weight loss.
Fibrous particles greater than 1 µm in length with a length-to-width ratio greater than 10:1 have been reported to present mesothelial risk. 13 Asbestos, inducing malignant mesotheliomas, meets this criterion, as does the TISMO used in our experiments, with dimensions mostly shorter than 50 µm in length and less than 2 µm in width. In our previous experiments with TISMO fibers, administered by intratracheal instillation to F344 rats at 4 mg/ rat, only mild inflammatory reactions were observed in the lungs after 28 days. 14,15 Other reports of TISMO administration by subchronic and chronic inhalation in rats also featured only limited inflammation in the lung. 16,17 In the present experiments, 1 A/J mouse exhibited granulation with the presence of some fibers in the lung alveoli, even though infusion was targeted outside the lung and into the thoracic cavity. Thus, fibers can readily penetrate through the pleura. Administration by infusion into the thoracic cavity is not physiologically relevant, so the method used in the present experiments represents an unusual route of exposure. However, our model is expected to be useful for examining the direct effects of TISMO fibers on the pleura during limited experimental period. This model may also be useful for screening of materials with hazard potential for the pleura.
Histopathologically, the number of lung tumors demonstrated no significant intergroup differences in experiment 1 or experiment 2, contrary to the expectation that mesothelial reaction or inflammation induced by TISMO would promote lung tumor development. The A/J mouse is sensitive to lung carcinogens and has been reported to develop spontaneous lung tumors even in the absence of any chemical treatment. 18 In addition, A/J mice are known to be one of the most susceptible lineages for 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK)-induced carcinogenesis. 19,20 The NNK is a tobacco-specific N-nitrosamine that is considered to play important roles in tobacco-related human lung cancer. 21,22 In the A/J mouse, it is also well known that females are generally more sensitive than males to chemical lung carcinogenesis. 23,24 Therefore, female A/J mice were employed in the present experiments.
Malignant mesotheliomas are reported to show frequent genetic inactivation of tumor suppressor genes p16 (INK4a)/p14 (ARF) and neurofibromatosis type 2 (NF2) and epigenetic inactivation of RASSF1A. However, no frequent mutations of well-known oncogenes (such as those encoding K-RAS and PIK3CA) have been identified. 25 Meanwhile, activating mutations of the Kras gene at codon 12 were detected in neoplastic lesions induced by NNK in A/J mice. 26 This difference of the mechanisms to induce lung tumors or mesotheliomas may be a possible reason for the lack of modifying effects on neoplastic development demonstrated by TISMO in the present experiments.
From the Berlin blue staining in the present experiment 1, iron accumulation (Fe3+) was observed around TSMO fibers in the thickened pleura of the lung, almost colocating with immunostaining for calretinin, indicative of mesothelioma cells. Iron, as a component of asbestos or as a consequence of asbestos-induced pathology, is a major candidate for underlying carcinogenic mechanisms, because iron overload is recognized to be carcinogenic. 10 Iron works as a catalyst for the Fenton reaction (Fe2+ + H2O2 > Fe3+ + OH·+ OH−), thus promoting the generation of the undesirable hydroxyl radical (OH·). 27 DNA damage and apoptosis resulting from iron-derived reactive oxygen species are regarded as important for malignant mesothelioma development. 28 –31 Respiratory exposure to asbestos is associated with mesotheliomas in humans, and electron spin resonance analyses have shown that crocidolite and amosite contain high amounts of iron. 32 While the chemical formula of TISMO used in the present experiments is K2O·nTiO2 without any Fe component, intraperitoneal or intrascrotal application of multiwall carbon nanotube (MWCNT) also without any Fe component has been reported to induce mesotheliomas. 33,34 Furthermore, 2 different mineral dusts, iron oxide with high iron content and calcium tungstate with very low iron content (0.01%), are associated with accumulation of iron and ferritin by alveolar macrophages. This result indicates that alveolar macrophages accumulate iron and ferritin in response to both iron loading of the lungs with iron oxide exposure and lung inflammation induced by calcium tungstate exposure. 35 Therefore, an iron component in a hazardous material is clearly not an absolute necessity. Iron is typically in the Fe3+ form in vivo and would need to be reduced prior to any Fenton activity; lung-lining fluid contains antioxidants, such as glutathione and ascorbic acid, which could reduce Fe3+ to Fe2+. 36 In the present experiment 1, the concentrations of Fe in the blood and liver tissues did not demonstrate any differences between TISMO-treated and untreated groups and Fe-NTA-treated or untreated groups. The speculation for this result is that TISMO fiber does not contain any Fe component, and Fe accumulation around TISMO fibers could be derived from endogenous Fe in vivo.
Pleural thickenings featuring the fibers were observed in all groups treated with TISMO in the present experiments 1 and 2. The mesothelial cells in the thickened pleura showed slight atypia but a diagnosis of malignancy could not be given because of the absence of invasion or metastasis and the absence of multilayering. Thus, the observed mesothelial proliferation with atypia was considered a reactive change. The mortality rate due to mesotheliomas has been reported to increase from about 130 days after intraperitoneal injection of MWCNT or crocidolite in p53-heterozygous mice. 34 Our failure to observe mesotheliomas, even at 457 days, may reflect our use of wild-type animals, which have inherently lower baseline rates of mesothelioma induction. Our previous 21-week experiment demonstrated no unequivocal strain differences in the degree of the pleural thickening in response to TISMO fibers, but there was clear strain variation in iron and lymphocyte accumulation in the pleura and the liver. 37 This in vivo information likely will be important when considering the development of mesothelioma in animal models and in extrapolation for human risk assessment. In order to obtain more detailed information on the relevance to malignancy, a long-term experiment using different strains is now in progress in our laboratory.
Although TISMO fibers were administered into the thoracic cavity, the fibers subsequently were detected histopathologically in the liver and kidneys in experiment 1, and in several organs, including liver, kidneys, spleen, heart, ovaries, bone marrow, and brain in experiment 2. The route to these other organs from the thoracic cavity is suspected to be via blood vessels because the fibers were detected in glomeruli in the kidney, in the vessels and around the vessels in the liver and brain, and in the bone marrow and spleen. But it remains unclear whether the vessels provide the only route to remote organs. Generally, a proportion of all particles that are deposited in the peripheral lung are reported to transit through the pleura and normally exit through the stomata in the parietal pleura to the underlying lymphatic system and thereby to the mediastinal lymph nodes. 38,39 Infact, fiber deposition was confirmed in the mediastinal lymph nodes in the present experiments, our results also indicating the possibility of hematogenous dissemination of the fibers in addition to lymphogenous. The presence of long fibers into the pleural space is expected to lead to persistent inflammation and eventually fibrosis at the parietal mesothelium, in contrast, short fibers are expected to cause no or modest resolving inflammation, with no parietal pleural pathological features. 40 Therefore, the apropriate size of fiber would appear to be necessary for dissemination and induction of toxicity.
In the brain, the fibers were observed in the microvascular system of the cortex and in the parenchyma outside the blood vessels in the present experiment 2. The presence of the fibers was not noted on the first histopathological screening, and the particles were detected only by using polarized light microscopy to detect the luminescence of TISMO fibers. In forming a functional blood–brain barrier, cerebral capillaries feature tightly apposed endothelial cells with highly selective bidirectional transport properties, anchored to extracellular matrices. 41,42 In the present study, the fibers are suspected to have reached the microvascular system with subsequent mechanical penetration of the blood vessels in the brain. However, there was no apparent inflammatory cell reaction or histiocytic cell infiltration; remarkable neurologic symptoms were not evident during the in-life period. While the exact toxicological hazard of the fibers in the brain is unknown, these histopathological findings may be important for particle toxicity.
In conclusion, this study demonstrated that intrathoracic infusion of TISMO fiber did not cause malignant mesothelioma but severe chronic inflammation and proliferation of pleural mesothelial cells. Asbestos-like fibers may spread from the thoracic cavity to other sites of the body, including the brain. This observation should be incorporated into future hazard risk assessments that employ this particle type.
Footnotes
Acknowledgments
We thank Dr Malcolm A. Moore for his help in critical reading of the scientific English in this manuscript.
Author Contribution
Masanao Yokohira contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript; Nozomi Hashimoto contributed to conception and design; drafted the manuscript; and critically revised the manuscript; Tashitaka Nakagawa contributed to analysis and critically revised the manuscript; Yuko Nakano contributed to acquisition and analysis and critically revised the manuscript; Keiko Yamakawa contributed to acquisition and analysis and critically revised the manuscript; Sosuke Kishi contributed to acquisition and analysis and critically revised the manuscript; Shohei Kanie contributed to acquisition and analysis and critically revised the manuscript; Fumiko Ninomiya contributed to acquisition and analysis and critically revised the manuscript; Kousuke Saoo contributed to acquisition and interpretation and critically revised the manuscript; Katsumi Imaida contributed to conception and design, contributed to acquisition and analysis, drafted the manuscript and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
