Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 16 galactomannans as used in cosmetics. These ingredients are legume polysaccharides that function mostly as hair/skin-conditioning agents and viscosity-increasing agents in cosmetic products. Their substantial molecular sizes suggest that skin penetration of these ingredients would be unlikely. The Panel concluded that these galactomannans are safe in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook, these 16 galactomannans function mostly as hair/skin-conditioning agents and viscosity-increasing agents in cosmetic products.
1
The ingredients included in this report are as follows:
Cyamopsis tetragonoloba (guar) gum; hydroxypropyl guar; C18-22 hydroxyalkyl hydroxypropyl guar; guar hydroxypropyltrimonium chloride; hydroxypropyl guar hydroxypropyltrimonium chloride; carboxymethyl hydroxypropyl guar; hydrolyzed guar;
Ceratonia siliqua gum; locust bean hydroxypropyltrimonium chloride; hydrolyzed Ceratonia siliqua gum extract;
Caesalpinia spinosa gum;
Caesalpinia spinosa hydroxypropyltrimonium chloride; hydrolyzed Caesalpinia spinosa gum;
Trigonella foenum-graecum hydroxypropyltrimonium chloride; cassia gum; and cassia hydroxypropyltrimonium chloride.
Gum guar, oxidized 2-hydroxypropyl 3-hydroxy-3-(trimethylammonio) propyl ether, chloride—also known as cationic guar, is not a cosmetic ingredient. Because of the similarity of this ingredient to guar hydroxypropyltrimonium chloride, acute oral toxicity data on cationic guar were considered. Similarly, acute oral toxicity data on carboxymethyl guar (not a cosmetic ingredient) are being considered in the absence of acute oral toxicity data on carboxymethyl hydroxypropyl guar. Data on Trigonella foenum-graecum seed powder/paste are included because the gum derived from T foenum-graecum is part of the chemical structure of T foenum-graecum hydroxypropyltrimonium chloride.
Chemistry
Definition and Structure
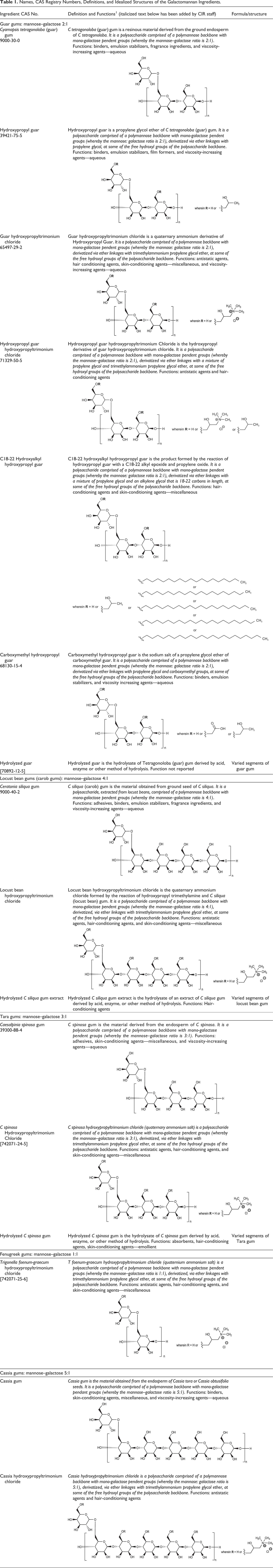
Definitions of the ingredients reviewed in this safety assessment along with their chemical structures are given in Table 1. 1
Names, CAS Registry Numbers, Definitions, and Idealized Structures of the Galactomannan Ingredients.
Properties
Seed-bearing plants deposit energy-containing reserves to support the growth of the embryo within the seed. Seed reserves can be of protein, lipid, or polysaccharide character. Although the most ubiquitous reserve polysaccharide is starch, the reserve polysaccharides, or gums, of the plant family Leguminoseae consist of polysaccharides described as having a polymannose backbone with galactose pendent groups. They are commonly called galactomannans because of the mannose/galactose chemical makeup of these legume polysaccharides (see Figure 1). Unlike starch, which is stored in amyloplasts in the cytoplasm, nonstarch reserve polysaccharides, such as galactomannans, are deposited in the cell walls of the seeds.
The 5 primary galactomannan gums are guar gum from the seeds of Cyamopsis tetragonoloba, locust bean gum from the seeds of Ceratonia siliqua (carob tree), tara gum from seeds of Caesalpinia spinosa, fenugreek gum from the seeds of T foenum-graecum, and cassia gum from seeds of Cassia obtusifolia or tora. Although all seed galactomannans possess the same basic structure of a β(1→4) linked polymannose backbone with α(1→6) linked galactose pendent residues, the ratio of galactose to mannose is species specific and is roughly a constant average within each species. In other words, guar gums average a mannose—galactose ratio of 2:1, locust bean gums are 4:1, tara gums are 3:1, fenugreek gums are 1:1, and cassia gums are 5:1, roughly. The distribution of galactose pendent groups along the polymannose backbone, in each group, is not uniform but more closely resembles a random block copolymer. In other words, within 1 polysaccharide chain, there are sections that are highly substituted with galactose pendent groups, and there are sections that are more sparsely substituted with galactose pendent groups. Yet, the overall average ratio is maintained within the gum of a species (Figure 2).
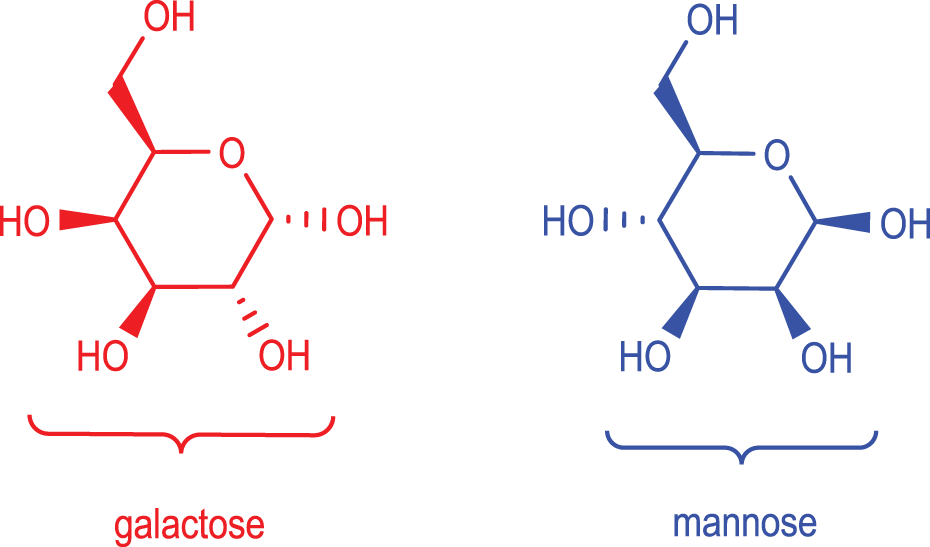
Galactose and mannose, the building blocks of the galactomannans.
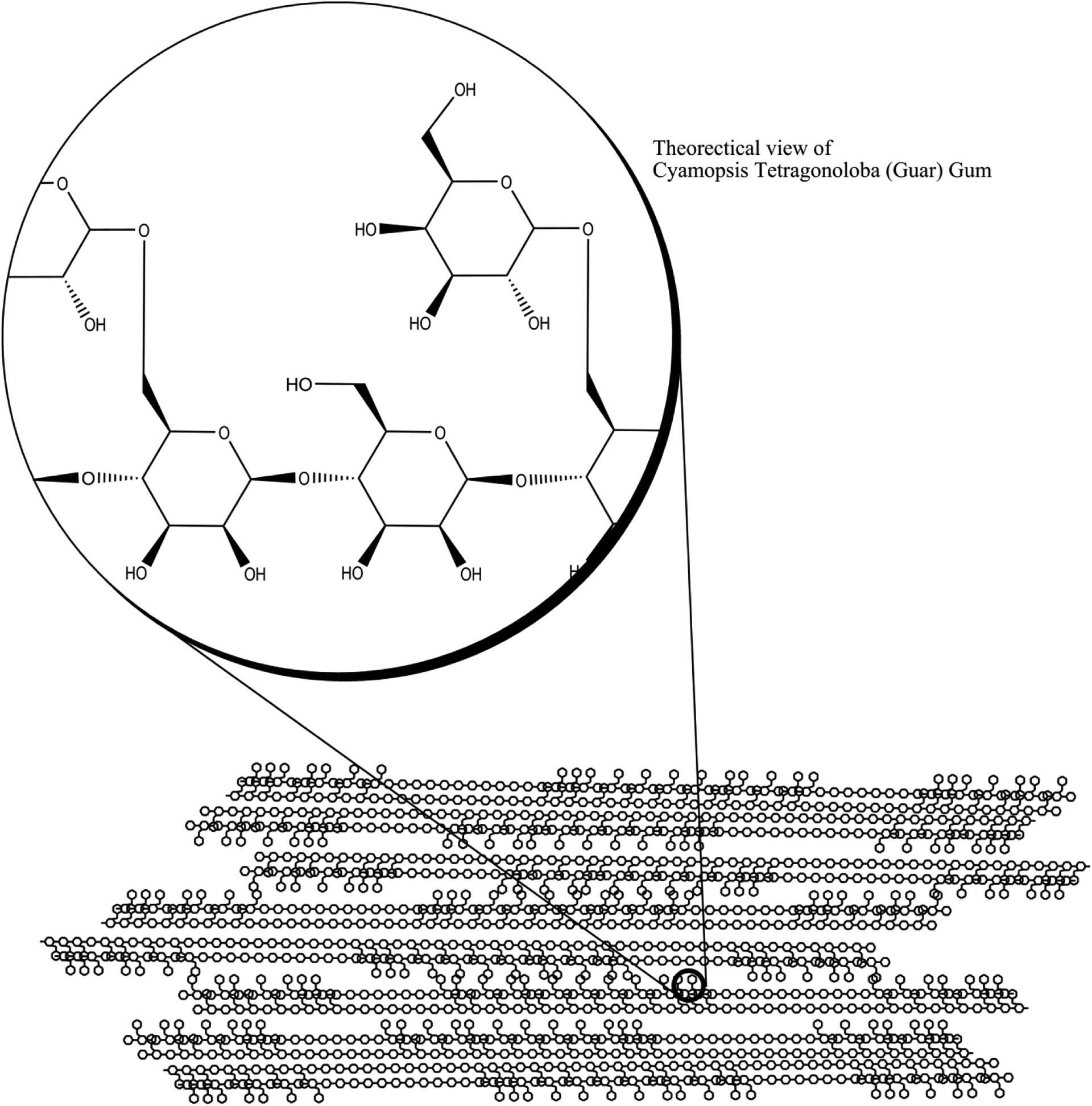
Packing and structure of Cyamposis tetragonoloba (guar) gum.
Although the ratio of mannose to galactose is 2:1, in guar gums, and the above-exploded theoretical structure depicts a galactose pendent group on every other mannose, the true structure is an assortment of pendent placements. These natural gums are typically of a molecular weight greater than 50 000 g/mole and often exceed 2 000 000 g/mole.
In addition to these underivatized, natural gums are those ingredients that have been etherified at the C6 position of galactose and/or mannose with various substituents (Figure 3). The most likely point of derivatization is at the C6 positions of a galactose pendant groups and the polymannose backbone, although all of the free hydroxyl groups are potentially susceptible substrates for condensation. Reported degrees of substitution are related as saturated (ie, equal to 1.0) when there is 1 stoichiometric equivalent of substituent per 1 potentially free C6 hydroxyl group (Figure 3). The average degree of substitution, for these types of derivatized gums, is reportedly 0.7 or less. Accordingly, these ingredients have less than 1 substituent (eg, hydroxypropyltrimonium chloride) per mannose or galactosylmannose. For example, a hydroxypropyl trimonium derivative of guar gum (guar hydroxypropyltrimonium chloride) would be substituted at some available C6 hydroxyls but might not be at all (Figure 3).
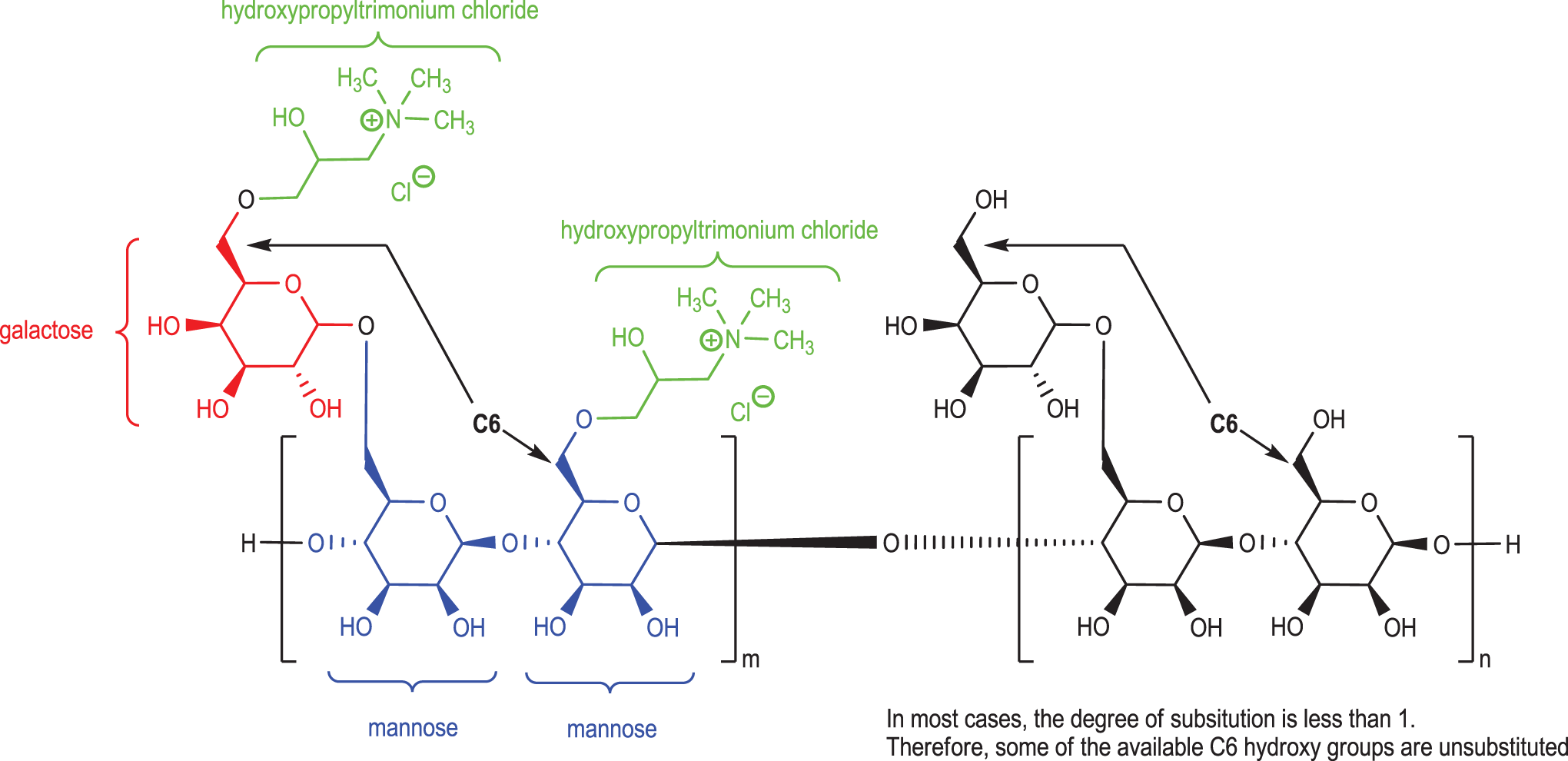
Idealized structure of guar hydroxypropyltrimonium chloride. 2
Those gums listed in this report as “hydrolyzed” are really partially hydrolyzed polysaccharides (eg, hydrolyzed guar gum = guar oligomers). For example, hydrolyzed guar is a mixture of guar gum fragments that have been broken down at the 1→6 and 1→4 linkages (via acidic, enzymatic, or other methods of hydrolysis) into shorter, lower molecular weight oligosaccharides.
According to the Food Chemicals Codex, both C tetragonoloba (guar) gum and C siliqua gum occur as a white to yellow-white powder. 3 Each gum is dispersible in either hot or cold water, forming a sol (pH between 5.4 and 7.0) that may be converted to a gel by addition of small amounts of sodium borate.
Method of Manufacture
Production of natural gums consists of various techniques for the milling of seeds, followed by simple purification steps such as dissolving in hot water, filtering, and precipitating with isopropanol. Typical production of the derivatized gums in this report involves the reaction of the natural gum with the appropriate epoxide. Additional information relating to the production of C tetragonoloba (guar) gum, hydroxypropyl guar, carboxymethyl hydroxypropyl guar, C siliqua gum, Caesalpinia spinosa gum, and cassia gum is included subsequently.
Cyamopsis tetragonoloba (guar) gum
Cyamopsis tetragonoloba (guar) gum is a gum obtained from the ground endosperms of Cyamopsis tetragonolobus (Linné) Taub. 4
Hydroxypropyl guar
Hydroxypropyl Guar gum can be prepared by alkaline etherification of guar gum with propylene oxide. 5
Guar hydroxypropyltrimonium chloride
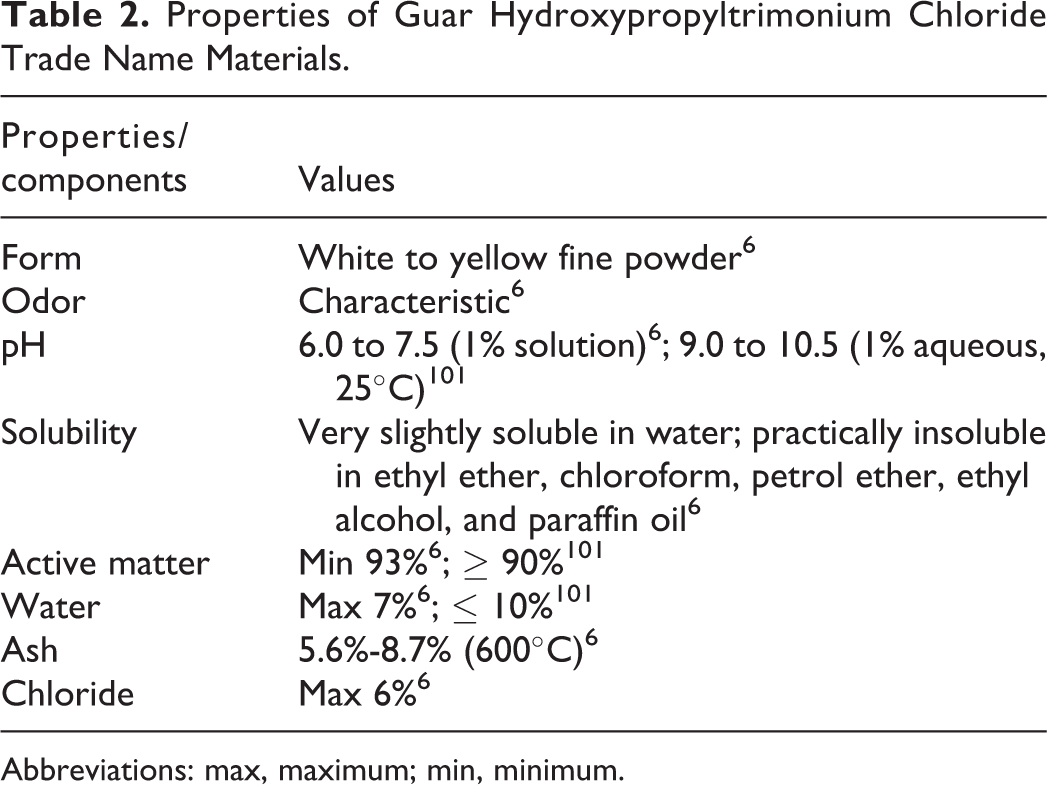
One method of production of a guar hydroxypropyltrimonium chloride trade name material involves the conversion of guar with 3-chloro-2 hydroxypropyl trimethyl ammonium chloride. 6 Data on the properties/composition of this trade name material are found in Table 2.
Properties of Guar Hydroxypropyltrimonium Chloride Trade Name Materials.
Abbreviations: max, maximum; min, minimum.
Carboxymethyl hydroxypropyl guar
Carboxymethylation and hydroxypropylation of C tetragonoloba (guar) gum are carried out simultaneously using monochloroacetic acid and propylene oxide in the presence of a hydrophilic solvent (ie, 2-propanol) with an alkaline pH. 7 These simultaneous reactions result in the formation of carboxymethyl hydroxypropyl guar gum.
Ceratonia siliqua gum
Ceratonia siliqua gum is a gum obtained from the ground endosperms of C siliqua Linné Taub. (Family: Leguminosae). 4
Caesalpinia spinosa gum
Commercial production of C spinosa gum involves the incomplete separation of the endosperm from the germ and the husk. Thus, commercial products may contain small amounts of husk as well as varying amounts of protein and fat from unseparated germ. Consequently, commercially available C spinosa gum may contain larger percentages of ash and acid insoluble than are present in the hand-dissected endosperm. 8
Cassia gum
Cassia gum is derived from Cassia tora or C obtusifolia. 1 The production method for cassia gum includes cleaning of the source material, dehusking, and degerming by thermal and mechanical treatment. 9 These steps are followed by milling and screening of the endosperm, which is further purified by extraction with isopropanol. Semirefined cassia gum is produced in a similar manner, with the exception of an additional isopropanol step to significantly reduce the level of anthraquinones in the latter.
Composition/Impurities
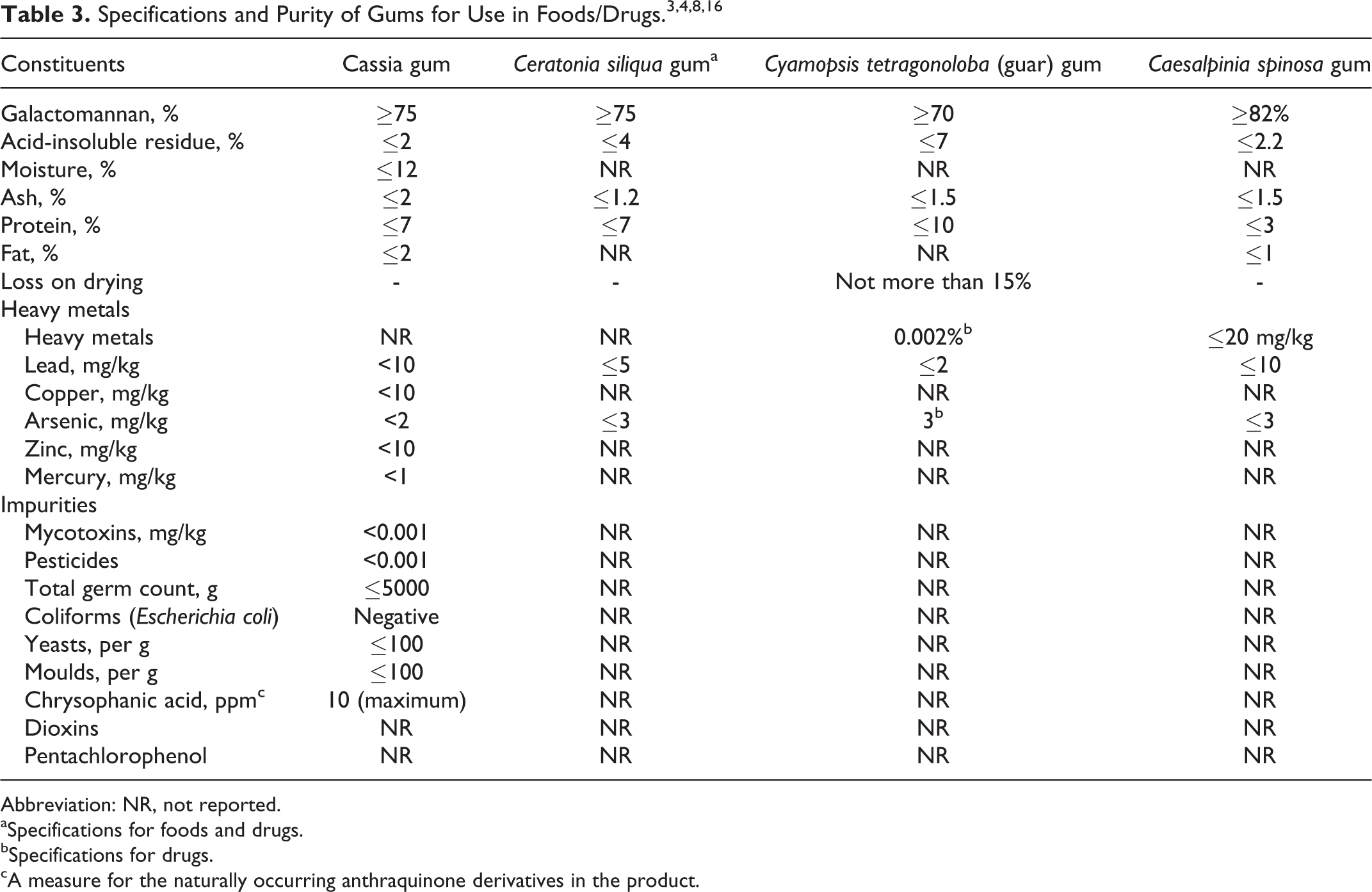
Specifications on the following gums relating to their use in foods/drugs in the United States are included in Table 3: C tetragonoloba (guar) gum, C siliqua gum, C spinosa gum, and cassia gum.
Abbreviation: NR, not reported.
aSpecifications for foods and drugs.
bSpecifications for drugs.
cA measure for the naturally occurring anthraquinone derivatives in the product.
Cyamopsis tetragonoloba (guar) gum
Cyamopsis tetragonoloba (guar) gum typically consists of the following: galactomannan (80%), water (12%), protein (5%), acid-insoluble matter (2%), and fat (1%). 10 Available data on 11 bulk commercial samples of this gum indicate that relative proportions of the most abundant amino acids, glycine, glutamic acid, aspartic acid, serine, and alanine vary considerably. Proportions of histidine, isoleucine, phenylalanine, threonine, tyrosine, and valine remain remarkably constant. 11
Commercial samples of C tetragonoloba (guar) gum, purified and unpurified, were analyzed in this study. 12 The 4 methods of purification included (1) treatment with proteolytic enzyme (porcine pancreatin); (2) successive steps of dissolution, centrifugation, and precipitation with acetone and ethanol; (3) Fehling solution used as precipitation agent; and (4) application of the second method, followed by the third one. In all samples (purified and unpurified), mannose and galactose were the major constituents, and glucose and arabinose (2 monosaccharide contaminants) were also present. Uronic acid content ranged from 3.28% to 4.17%. The unpurified sample had a protein content of 3.6%. All purification methods reduced the protein and mono/oligo/polysaccharide contaminants. Method 4 resulted in total elimination of protein. However, use of Fehling solution in methods 3 and 4 contaminated the gum with small amounts of Cu(II), 0.079% and 0.044%, respectively. Methods 2 and 4 resulted in purer (ie, small amount of protein) C tetragonoloba (guar) gum.
The following main components in the lipid fraction of C tetragonoloba (guar) gum were identified using gas-liquid chromatography GLC-mass spectrometry (MS): palmitate (hexadecanoate), oleate (9-octadecenaoate), and linoleate (9,12-octadecadienoate). 13
Using MS, very low fluoroacetate concentrations (0.07-1.42 µg/g, 10 samples) were detected in C tetragonoloba (guar) gum used as a raw material for a guar gum powder. A pharmaceutical formulation of guar gum contained 0.08 ppm fluoroacetate. 14
The noncatalytic hydrolysis of C tetragonoloba (guar) gum under hydrothermal conditions (temperature range: 180°C-240°C) produced oligosaccharides with degrees of polymerization of up to ∼20, monosaccharides containing mannose and galactose, and 5-hydroxymethyl-2-furaldehyde. 15
In 2007, the Rapid Alert System for Food and Feed received a notification from Switzerland concerning a finding of serious contamination of C tetragonoloba (guar) gum, originating from India, with dioxins and pentachlorophenol (PCP). 16 The levels of these contaminants in certain batches of C tetragonoloba (guar) gum were approximately 1000 times the level of what can be considered as normal background contamination. According to a European Commission decision in 2008, all consignments of C tetragonoloba (guar) gum or products containing C tetragonoloba (guar) gum at significant amounts originating in or consigned from India and imported into the Community intended for human or animal consumption shall be accompanied by an analytical report, endorsed by the competent authority from the country where the laboratory that has performed the analysis is located. With the 2008 decision in effect, it was determined that there had been no improvement in the control system and no significant reduction in the associated risks. Because the 2008 decision resulted in no improvement in the control system and no significant reduction in the associated risks, in 2010 the European Commission added a requirement for official sampling, analysis, and certification by competent authorities of India of all consignments of C tetragonoloba (guar) gum intended for export to the European Union.
Guar hydroxypropyltrimonium chloride
When a guar hydroxypropyltrimonium chloride trade name material is heated to 600°C, 5.6% to 8.7% of the material remains as ash. 6 This information and additional data on the properties/composition of this trade name material are found in Table 2.
Ceratonia siliqua gum
Ceratonia siliqua gum has been known to contain tannins and trypsin inhibitors. 17
Cassia gum
Cassia gum (C tora or C obtusifolia) is composed of at least 75% high molecular mass (∼200 000-300 000) polysaccharide, which consists primarily of a linear chain of 1,4-β-
Use
Cosmetic
The ingredients reviewed in this safety assessment function mostly as hair-/skin-conditioning agents and viscosity-increasing agents in cosmetic products. These and additional functions are included in Table 1.
According to the information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2011, the following 9 ingredients were being used in cosmetic products: C tetragonoloba (guar) gum, hydroxypropyl guar, guar hydroxypropyltrimonium chloride, hydroxypropyl guar hydroxypropyltrimonium chloride, hydrolyzed guar, C siliqua gum, C spinosa gum, hydrolyzed C spinosa gum, and cassia hydroxypropyltrimonium chloride. 19 These data are summarized in Table 4. Results from a survey of ingredient use concentrations conducted by the Personal Care Products Council (also included in Table 4) in 2011 indicate that locust bean hydroxypropyltrimonium chloride was also being used in cosmetics. 20 Additionally, results from this survey indicate that galactomannans were being used at concentrations of up to 93% (hydroxypropyl guar, in a hair straightener leave-on product) in cosmetic products. Additionally, guar hydroxypropyltrimonium chloride was being used in foot powders and sprays at a maximum concentration of 0.05%. Together, the VCRP data and Council survey results suggest that the remaining 6 ingredients included in this safety assessment are not being used in cosmetic products.
Current Frequency of Use According to Duration and Type of Exposure.19,20,a
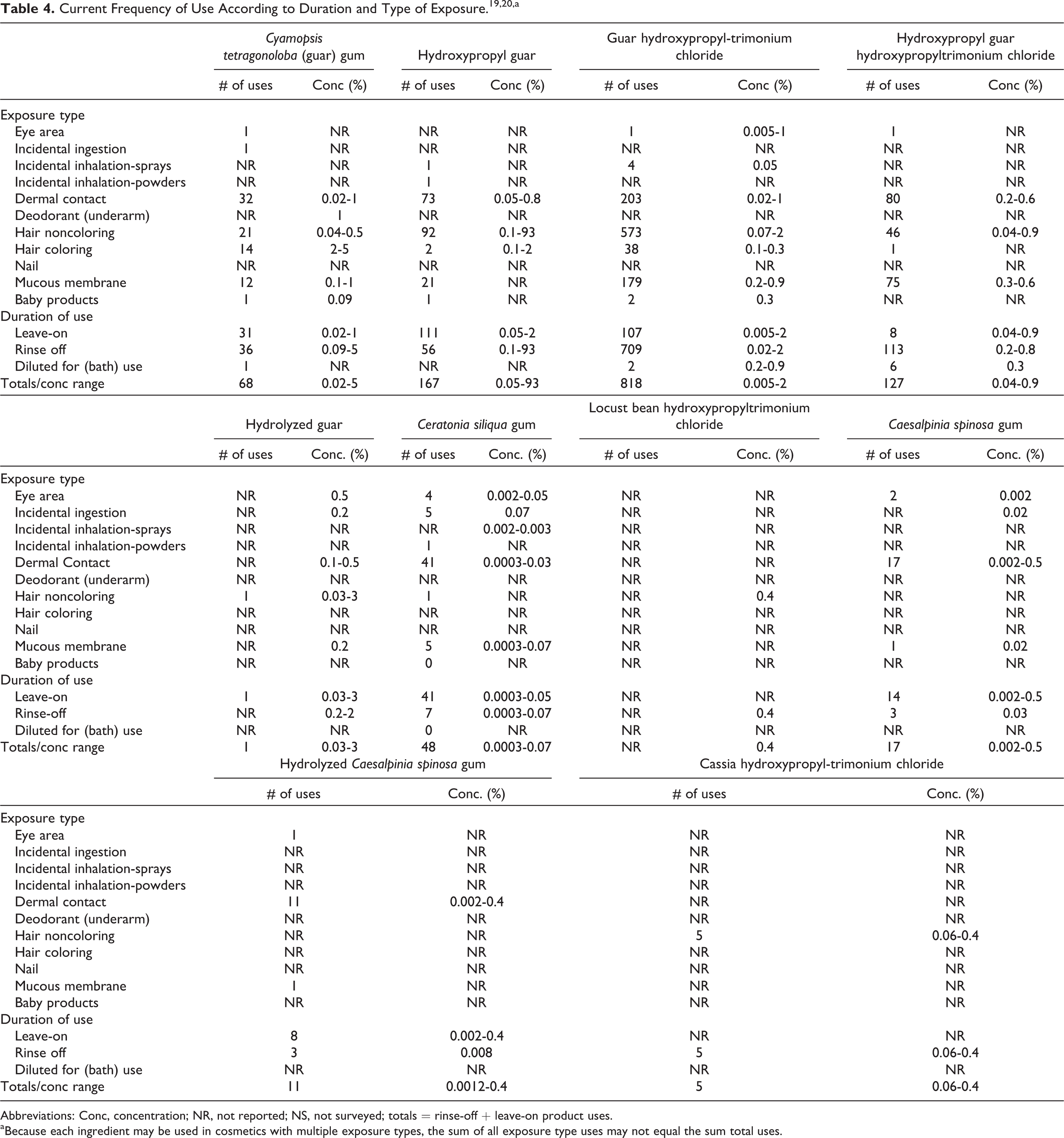
Abbreviations: Conc, concentration; NR, not reported; NS, not surveyed; totals = rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Cosmetic products containing the ingredients reported as being used may be applied to the skin and hair, or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Hydroxypropyl guar and guar hydroxypropyltrimonium chloride are used in hair sprays and possibly could be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 21,22 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions of the respiratory tract and would not be respirable (ie, able to enter the lungs) to any appreciable amount. 23,24
Noncosmetic
The following ingredients reviewed in this safety assessment are approved for use as direct food additives by the FDA: C tetragonoloba (guar) gum (21 CFR 133.124) 25 ; C siliqua gum (12CFR 182.20) 26 ; and C tetragonoloba (guar) gum and C siliqua gum (21 CFR 133.178; 133.179; 150.141; 150.161). 27,28,29,30 Additionally, C tetragonoloba (guar) gum and C siliqua gum are direct food additives that are considered generally recognized as safe (21 CFR 184.1339; 184.1343). 31,32 Hydroxypropyl guar is approved for use as an indirect food additive by FDA (21 CFR 176.170). 33 An evaluation of the health aspects of C siliqua gum (aka carob bean gum) as a food, prepared for FDA, is available. 17
Cyamopsis tetragonoloba (guar) gum
Noncosmetic uses of C tetragonoloba (guar) gum include 34 in paper sizing as a protective colloid, stabilizer, thickening and film-forming agent for cheese, salad dressings, ice cream, and soups; as a binding and disintegrating agent in tablet formulations; in pharmaceutical jelly formulations; in suspensions, emulsions, lotions, creams, and toothpastes; in the mining industry as a flocculant and a filtering agent; and in water treatment as a coagulant aid. It also functions as an emulsifier. 3
The distribution of Cal-Ban 3000 diet tablets and capsules was halted by the FDA because of the potential to cause esophageal, gastric, and intestinal obstruction. 35 Cyamopsis tetragonoloba (guar) gum was listed as the main ingredient of Cal-Ban 3000. This gum is a complex sugar that swells when it becomes wet and can create a sense of fullness when ingested.
Cyamopsis tetragonoloba (guar) gum has been approved for use as a food ingredient by the Joint Food and Agriculture Organization of the United Nations (FAO)/World Health Organization (WHO) Expert Committee on Food Additives, and this committee assigned a “not specified” value for the acceptable daily intake (ADI) by man. 36 This means that the use of C tetragonoloba (guar) gum as a food substance does not represent a human health hazard, and, therefore, the establishment of an ADI in mg/kg body weight was not deemed necessary.
Hydroxypropyl guar
Hydroxypropyl guar is used as a gelling polymer in an artificial tear solution. It is believed to mimic the mucin layer of the tear film. 37
Ceratonia siliqua gum
Ceratonia siliqua gum has been approved for use as a food ingredient by the Joint FAO/WHO Expert Committee on Food Additives (JECFA), and this committee assigned a not specified value for the ADI by man. 36 This means that the use of C siliqua gum as a food substance does not represent a human health hazard, and, therefore, the establishment of an ADI in mg/kg body weight was not deemed necessary. C siliqua gum functions as a stabilizer and thickener in food. 3
Caesalpinia spinosa gum
Caesalpinia spinosa gum has also been approved for use as a food ingredient by the JECFA, and this committee assigned a not specified value for the ADI by man. 38 This means that the use of Cyamopsis tetragonoloba (guar) gum as a food substance does not represent a human health hazard, and, therefore, the establishment of an ADI in mg/kg body weight was not deemed necessary.
Cassia gum
Noncosmetic uses of cassia gum include thickener; emulsifier; foam stabilizer; and moisture retention agent and/or texturizing agent in processed cheese, frozen dairy desserts and mixes, meat products, and poultry products. After considering data supporting the low oral toxicity and negative genotoxicity of cassia gum, the JECFA allocated an average daily intake “not specified” for cassia gum, when used in the applications specified and in accordance with Good Manufacturing Practice. 9
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
The only available data for these ingredients that relate to absorption, distribution, metabolism, and excretion were found in dietary studies.
Cyamopsis tetragonoloba (guar) gum
When a basal diet containing C tetragonoloba (guar) gum (dose = 100 g/kg) was fed to 5 rats, the galactomannan of the gum was digested almost quantitatively. Thus, approximately 1% of the mannose and 4% of the galactose were excreted in the feces. 39 The minor components (ie, arabinose, glucose, and uronic acids) were also fermented almost completely.
Caesalpinia spinosa gum
Two groups of 20 Wistar rats were fed a semisynthetic diet containing C spinosa gum (73.9% galactomannan) at galactomannan dietary concentrations of 2% and 5%, respectively, for 11 days. 40 At the end of this feeding period, 12 rats from each dietary group were selected (based on body weight gain, etc) for an experimental feeding period of 10 days. Digestibility was defined as the portion of ingested material that was not excreted in the stool. Digestibility of galactomannan was 97% and 98.1% for the 2% and 5% diets, respectively. In another study, rats were fed a normal diet containing C spinosa gum. 41 The composition and dietary concentration of the gum as well as the feeding periods were not stated. Stools were analyzed for mannose obtained by hydrolysis. It was concluded that all of the mannose added to the feed in the form of galactomannan was excreted in the feces.
Ceratonia siliqua gum
Groups of Purdue rats (5 males and 5 females per group; ages not stated) were fed 1% C siliqua gum in the diet for 18 hours. 42 Of the mannose fed as 1% C siliqua gum, 85% to 100% was excreted in the feces over a 30-hour period. It was noted that some decrease in galactomannan chain length may have occurred. This decrease was believed to have been due to the action of the microflora because mammals are not known to have the mannosidase enzyme. The liberation of galactose units was not determined.
Cassia gum
The JECFA noted that specific absorption, distribution, metabolism, and excretion data were not available on cassia gum at the time of evaluation of this food additive. 9 However, based on data on related galactomannans, the Committee concluded that cassia gum will be largely excreted unchanged, although fermentation by gut microflora may occur to some extent. It was noted that if hydrolysis of cassia gum occurs, the resulting oligosaccharides or monosaccharides would be expected to be absorbed and metabolized in normal biochemical pathways.
Toxicology
Acute Oral Toxicity
Cyamopsis tetragonoloba (guar) gum
The acute oral toxicity of C tetragonoloba (guar) gum was evaluated using 10 F344 rats (5 males and 5 females; ages not stated) and 10 B6C3F1 mice (5 males and 5 females). 43 Each animal received a single dose (0.42 g/kg) of the gum (in water) by gavage. None of the animals died, and no test substance-related effects were observed. In another acute oral toxicity study, 18 rats (strain not stated) received C tetragonoloba (guar) gum (in cocoa butter) at a dietary concentration of 30% for 48 hours. No adverse effects were observed. 44
Guar hydroxypropyltrimonium chloride
In an acute oral toxicity study on guar hydroxypropyltrimonium chloride (40% w/v in corn oil), 4 groups of 10 rats (5 males and 5 females) received doses of 7.1, 10.0, 14.2, and 20.0 g/kg, respectively, by oral intubation. 45 Dosing was followed by a 14-day observation period, and gross necropsy was performed on all animals that died spontaneously. Clinical signs observed included ataxia, tremors, nasal and oral discharge, urinary and fecal staining, abdominal griping, soft stool, decreased motor activity, labored breathing, and piloerection. The acute oral median lethal dose (LD50) was 12.5 g/kg (95% confidence limits of 10.22-14.78 g/kg).
The acute oral toxicity of a guar hydroxypropyltrimonium chloride trade name material (composition data in Table 2) was evaluated using Bor: WISW (SPF) Cpb rats (5 males and 5 females, ∼10 weeks old). 46 A single oral dose of the test substance (in arachidis oil, 2000 mg/kg) was administered to each animal by gavage. Dosing was followed by a 14-day observation period. Animals that survived were killed and subjected to postmortem examination. None of the animals died, and there were no macroscopic findings at necropsy. An LD50 of >2000 mg/kg was reported.
Gum guar, oxidized 2-hydroxypropyl 3-hydroxy-3-(trimethylammonio)propyl ether, chloride (aka cationic guar and guar hydroxypropyltrimonium chloride)
The acute oral toxicity of guar hydroxypropyltrimonium chloride (40% w/v solution in corn oil) was evaluated using 3 groups of 10 Sprague-Dawley albino rats (5 males and 5 females/group) receiving doses of 10.0, 14.2, and 20.0 g/kg, respectively. 47 Dosing was followed by a 14-day observation period, and gross necropsy was performed on animals that died spontaneously. In-life observation included ataxia; nasal, oral, and ocular discharge; urinary and fecal staining; abdominal griping; decreased motor activity; irritability; and piloerection. Convulsions, tremors, hyopothermia, and prostration were observed only at the highest dose (20.0 g/kg), which yielded 90% mortality. The acute oral LD50 was 15 g/kg (95% confidence limits of 13-17 g/kg).
Hydroxypropyl guar hydroxypropyltrimonium chloride
In an acute oral toxicity study on hydroxypropyl guar hydroxypropyltrimonium chloride (40% w/v solution in corn oil), 4 groups of 10 Sprague-Dawley albino rats (5 males, 5 females/group) received doses of 7.1,10.0, 14.2, and 20.0 g/kg, respectively, by oral intubation. 48 Dosing was followed by a 14-day observation period, and gross necropsy was performed on all animals. Clinical signs observed included ataxia; fine and coarse tremors; nasal, oral, and ocular discharge; decreased respiratory rate and motor activity; abdominal griping; piloerection; irritability; and generally poor condition. The acute oral LD50 was 12.0 g/kg (95% confidence limits of 9.2-14.8 g/kg).
Carboxymethyl guar
The acute oral toxicity of carboxymethyl guar (40% w/v solution in corn oil) was evaluated using 5 groups of 10 Sprague-Dawley albino rats (5 males and 5 females/group) receiving doses of 5.0, 7.1, 10.0, 14.2, and 20.0 g/kg, respectively, by oral intubation. 49 Dosing was followed by a 14-day observation period, and gross necropsy was performed on all animals that died spontaneously. Clinical signs observed included ataxia; tremors; nasal, oral, and ocular discharge; labored breathing; piloerection; increased/decreased activity; hypothermia; generally poor condition; urinary and fecal staining; and irritability. The acute oral LD50 was 17.8 g/kg (95% confidence limits of 13.1-22.5 g/kg).
Hydrolyzed guar
An acute oral toxicity study on partially hydrolyzed guar was performed using groups of 16 (8 males and 8 females per group) 4-week-old Jcl: ICR mice and Jcl: SD rats. 50 Partially hydrolyzed guar was administered by gavage at a concentration of 30% in distilled water (dose = 6000 mg/kg body weight; dose volume = 20 mL/kg) to 1 group per species. The control group was dosed orally with distilled water. Dosing was followed by a 14-day observation period, after which all animals were killed and examined macroscopically. Soft feces were reported for male and female mice, but no abnormal signs were reported for rats. There were no test substance-related effects on body weight (rats and mice), food consumption (rats), or necropsy findings (mice and rats). None of the animals died, and the LD50 was >6000 mg/kg in both the species.
Ceratonia siliqua gum
The acute oral toxicity of C siliqua gum (in corn oil; dose = 10 g/kg body weight) was evaluated using 5 male Sprague-Dawley rats. 51 None of the animals died, and transient depression was the only sign observed in animals tested. In other studies involving rats (number and strain not stated), acute oral LD50s of 5000 mg/kg body weight and 13 100 mg/kg body weight were reported. Details relating to the test protocol and study results were not included. 52
In an acute oral toxicity studies involving mice, rabbits, and hamsters (number and strain not stated for each), the following LD50s were reported: 13 100 mg/kg body weight (mice), 9100 mg/kg (rabbits), and 10 300 mg/kg (hamsters). In each of the 3 studies, details relating to the test protocol and study results were not included. 52
Cassia gum
The acute oral toxicity of semirefined cassia gum was evaluated using 5 male Wistar-Han-Schering rats. 9,53 The animals received 2 oral doses of 5000 mg/kg body weight at a 2-hour interval. An oral LD50 value of >5000 mg/kg body weight was reported. In a similar test, 10 male and 10 female KM mice received 4 oral doses of cassia gum (gavage, 10 000 mg/kg body weight) over a 24-hour period. 54 The acute oral LD50 was >10 000 mg/kg body weight.
Repeated Dose Toxicity—Oral Studies
Cyamopsis tetragonoloba (guar) gum
In a short-term oral toxicity study, 18 rats (strain not stated) received C tetragonoloba (guar) gum (in cocoa butter) at a dietary concentration of 30% for 48 hours. 55 There was no evidence of adverse effects.
Five male Sprague-Dawley rats received oral doses of C tetragonoloba (guar) gum (in corn oil; dose = 5 g/kg) daily for 5 days. 56 No unusual or adverse effects were observed. The feeding of 27% C tetragonoloba (guar) gum to rats for 7 days resulted in the death of 7 to 10 animals. The deaths were probably due to intestinal blockage. 55
Groups of rats (strain not stated) were fed C tetragonoloba (guar) gum in the diet at concentrations of 0%, 1%, 2%, and 5%, respectively, for 90 days. 57 Dosing did not affect general behavior, appearance, or survival. Growth was described as relatively low in males fed the 2% and 5% C tetragonoloba (guar) gum diets. No effect was observed on the following: hematology, urinalysis, serum enzyme activities, or blood glucose levels. However, blood urea nitrogen values were slightly increased in males of the 5% dietary group. The relative weight of the thyroid was increased only in males of the 2% and 5% dietary groups. The results of gross and histopathological examinations did not reveal any changes that were attributable to gum ingestion.
The following levels of C tetragonoloba (guar) gum were fed to 6 groups of 130 male and female Osborne-Mendel rats (∼4-week-old; 65 males and 65 females/group) for 91 days: 0%, 1.0%, 2.0%, 4.0%, 7.5%, and 15.0% in diet. 58 A significant decrease in body weight was observed for female rats of all dietary groups and in males of the 7.5% and 15% dietary groups. There were no deaths were reported. Hematocrit values for males were less than control values in all dietary groups; however, this decrease was of borderline significance. Male hemoglobin levels and erythrocyte and leucocyte counts were all within control ranges. In females, hemoglobin levels and erythrocyte counts were significantly decreased only in the 4% dietary group. Compared to control males, liver weights in all dietary groups were significantly decreased. Kidney weights were significantly decreased in 7.5% and 15.0% dietary groups and were also decreased (borderline significance) in the 4.0% dietary group. Except for the bone marrow of male rats in the 15.0% dietary group, no tissue examined had consistent histopathological alterations attributed to ingestion of C tetragonoloba (guar) gum. Although there was a suggestion of regressive changes in the bone marrow (moderate bone marrow cellularity) at this dietary level, this finding was within normal limits. The results for several rats fed 15.0% C tetragonoloba (guar) gum were at the lower end of the normal range and suggested a subtle or borderline effect.
A long-term toxicity study on C tetragonoloba (guar) gum was performed using 2 groups of rats (15 males, 15 females/group; ages and strain not stated). One of the groups was fed 5% C tetragonoloba (guar) gum in the diet, and the other group (control) was fed diet only. Seven males and 8 females in each group survived and were monitored for 24 months. Of these, 1 test animal died after 12, 18, 19, and 22 months, and the last survivor was killed after 24 months. Three control animals survived to 24 months. All animals appeared in good health and had similar body weights.
Cyamopsis tetragonoloba (guar) gum (1 g in diet) was fed daily to 2 monkeys (ages and strain not stated). 44 After 16 months of feeding, one of the monkeys died. The other monkey was killed at 24 months. Well-being, growth, and hematology (red blood cells, white blood cells, hemoglobin, and urea) were considered normal. The results of gross and histopathological examinations did not indicate any abnormalities in any of the following organs/tissues: liver, kidney, spleen, gut, and bone marrow.
Cyamopsis tetragonoloba (guar) gum and hydrolyzed guar
Diets containing 5% C tetragonoloba (guar) gum and 5% hydrolyzed guar (partially hydrolyzed) were fed to 2 groups of 5 Sprague-Dawley rats (8 months old), respectively, for 3 weeks. 59 Another group of rats was fed a diet containing 5% cellulose. A significant decrease in food intake and weight gain as well as a significant increase in liver weight were reported after feeding with 5% C tetragonoloba (guar) gum. The serum immunoglobulin (Ig) G (IgG) level of rats fed 5% C tetragonoloba (guar) gum was significantly lower when compared to that of rats fed 5% cellulose. Also, the productivity of IgA, IgG, and IgM in mesenteric lymph node (MLN) lymphocytes was significantly higher in rats fed 5% C tetragonoloba (guar) gum than in rats fed 5% cellulose. The effect of 5% C tetragonoloba (guar) gum on productivity of IgA, IgG, and IgM in spleen cells was not as marked. The epididymal adipose tissue weight in rats fed 5% hydrolyzed guar was significantly higher than that reported for rats fed 5% cellulose. The results of this study suggest that the enhancement of immune function by C tetragonoloba (guar) gum is expressed mainly in the gut immune system.
Hydrolyzed guar
In a 28-day oral feeding study, 2 groups of 10 rats (5 males and 5 females/group) were fed partially hydrolyzed C tetragonolobus (guar) gum in the diet (500 and 2,500 mg/kg doses, respectively) daily. 60 Body weights and food consumption were measured, and gross and microscopic pathology were evaluated. No adverse effects were observed at either administered dose.
A repeated dose oral toxicity study was performed using 7-week-old male and female Jcl: SD rats. The 2 groups of 20 rats per sex were control (diet without partially hydrolyzed guar) and 5.0% partially hydrolyzed guar dietary groups, respectively. 50 The remaining 2 groups of 10 rats per sex received dietary concentrations of 0.2% and 1.0%. All groups were fed daily for 13 weeks. Ten rats per sex per group were then randomly selected from control and 5.0% dietary groups and maintained on the control diet for an additional 4 weeks (recovery period). Recovery animals were used to investigate the reversibility of possible partially hydrolyzed guar toxicity. All surviving animals (recovery animals included) were killed and subjected to macroscopic and microscopic examination. There were no deaths during dosing or recovery periods or test substance-related effects on body weight, or food/water consumption. Additionally, there were no test substance-related changes relative to the following: ophthalmoscopic examination, urinalysis, hematological examination, blood biochemical examination, necropsy, organ weights, or histopathological examination.
Cyamopsis tetragonoloba (guar) and C siliqua gum
A precooked mixture of C tetragonoloba (guar) and C siliqua gum (proportions not stated) was fed to groups of 5 female Beagle dogs (5 males and 5 females per group) for 30 weeks. 61 The gum was fed at dietary concentrations of 0%, 1%, 5%, and 10%. Hypermotility was observed at the highest dietary concentration. There was no evidence of adverse hematological, urinary, gross/histopathological, or ophthalmological findings.
Ceratonia siliqua gum
Three groups of 8 rats (ages and strain not stated) were fed a stock diet, a stock diet with 1% cholesterol, and a stock diet with 1% cholesterol and 10% C siliqua gum, respectively, for 28 days. 62 There were no significant differences in weight gain between the 3 groups, and no adverse effects were reported. A soybean–corn meal diet containing 2% C siliqua gum was fed to groups of newly weaned Sprague-Dawley rats (10 rats per group, ages not stated) for 36 days. 63 There were no significant effects on growth.
Ceratonia siliqua gum was fed, in the diet, to groups of rats (strain not stated) at concentrations of 0%, 1%, 2%, and 5%, respectively, daily for 90 days. 57 Except for increased blood glucose in the 5% dietary group, there were no treatment-related differences between the test and the control groups regarding the following parameters: general behavior, survival, growth, food intake, hematology, blood biochemistry, and urinalysis. Neither gross nor microscopic examination results indicated any pathological changes that were due to ingestion of the gum. Five male Sprague-Dawley rats (ages not stated) received oral doses of C siliqua gum (in corn oil; dose = 5 g/kg) daily for 5 days. 51 No unusual or adverse effects were observed.
In a study investigating the effect of various gums on nitrogen balance and dry matter digestibility, a group of 12 weanling Sprague-Dawley rats was fed C siliqua gum at a concentration of 10% in a casein–saccharose–corn starch diet. 87 Following a 3-day adaptation period, feed remnants, urine, and feces were collected during an 8-day balance period. Trypsin inhibitory activity of C siliqua gum in the diet was also measured. Only slight enzyme inhibition was associated with this gum.
Caesalpinia spinosa gum
In a 90-day oral feeding study, 50 rats (strain and ages not stated) of each sex were fed diets containing 0%, 1%, 2%, or 5% C spinosa gum. 64 At the 5% dietary level, body weight gains were depressed in both sexes and, possibly, in males at the 2% dietary level. Compared to the other dietary groups, food intake in the 5% dietary group was decreased. There were no effects on hematology or urinalysis parameters in any of the groups tested. A statistically significant increase in blood urea nitrogen was noted in rats fed 5% C spinosa gum in the diet; however, blood levels were within the normal range of values for rats. There were no treatment-related differences in other clinical chemistry parameters between the control group and any group fed C spinosa gum in the diet. Increased male kidney–body weight ratios (5% group) and increased relative weights of the thyroids and testes (2% and 5% groups) were observed. Results of gross and microscopic examination of tissues, including those associated with increased relative organ weight, were not indicative of changes related to C spinosa gum in the diet.
Groups of 10 F344 rats and 10 B6C3F1 mice of each sex (ages of animals not stated) were fed diets containing 0%, 0.31%, 0.63%, 1.25%, 2.50%, or 5.0% C spinosa gum for 13 weeks. 65 The gum contained 86.2% galactomannan. The animals were killed at the end of the study, and microscopic examination was performed on control animals and those fed 5% C spinosa gum in the diet. None of the mice or rats in any of the groups died during the feeding period. Compared to controls, male rats experienced small decreases in body weight gain, while weight increases were reported for female rats. Opposite effects on body weight gain were reported for mice. Changes in body weight gain were not dose related. The only changes related to feeding with C spinosa gum included fewer mature spermatozoa in 4 of 10 male rats in the 5% dietary group.
Purebred Beagle dogs (3 per sex) were fed experimental diets containing 1% or 5% C spinosa gum for 90 days, and the control group was fed α-cellulose. 66 For male dogs fed the 5% diet, feed intake values were 14% below those reported for the control group. No behavioral changes were reported, and hematological, urinalysis, and clinical chemistry results were unremarkable. There were also no gross or microscopic findings that were related to feeding with diets containing C spinosa gum.
Cassia gum
Groups of 5 male and 5 female crl: CD (SD)BR Sprague-Dawley rats (5-6 weeks old) were fed semirefined cassia gum daily at the following dietary concentrations in a 28-day study: 0, 2500, 10 000, 25 000, or 50 000 mg/kg. 67,67 The control group was fed an untreated powdered diet. An additional group of rats received the test substance (in distilled water; dose = 1000 mg/kg body weight) by gavage twice daily. Histopathologic examination was performed only on the major organs of animals from the following groups: control, 50 000 mg/kg (in feed) group, and the group dosed (1000 mg/kg body weight) via gavage twice daily. There were no treatment-related deaths or clinical changes in any of the groups. Statistically significant reductions in body weight were noted in males (50 000 mg/kg group) and females (10 000 and 25 000 mg/kg groups); however, these changes were considered related to the viscous nature of cassia gum and toxicologically irrelevant.
The statistically significant hematological and clinical chemistry findings were not dose related or occurred in 1 sex only. These values were within the normal range for this species, however, historical control data were not provided. The only changes outside the historical control range that could have been treatment related were increased mean concentrations of glucose and triglyceride in the 10 000 and 25 000 mg/kg but not the 50 000 mg/kg groups. Statistically significant reductions in group mean absolute kidney weights were reported for the 10 000 and 50 000 mg/kg (in diet) groups and for the 1000 mg/kg dose (gavage) group. Additionally, a statistically significant increase in relative kidney weights was reported for the 50 000 mg/kg group. The minimal changes in absolute and relative organ weights were considered effects due to decreased body weights at termination. At necropsy or during microscopic examination of major organs (kidneys included), no treatment-related effects were observed. It was concluded that cassia gum did not elicit any apparent toxic changes that were attributable to dosing at dietary levels up to 50 000 mg/kg or at a dose (intragastric) of 1000 mg/kg/d. 67
Groups of Sprague-Dawley rats (10 males and 10 females per group) were fed cassia gum in the diet at doses of 0, 250, 500, and 1000 mg/kg body weight per day for 30 days. 54 There were no treatment-related effects on the following: mortality, body weight gain, food consumption, food utilization, hematological parameters, or various biochemical parameters (eg, albumin, cholesterol, and aspartate aminotransferase). Gross examination results were negative, and there were no treatment-related histopathologic changes or effects on weight in the following organs: liver, kidney, spleen, ovaries, and testes. There were also no histopathologic changes in the stomach or intestines. An no-observable-adverse-effect-level (NOAEL) of 1000 mg/kg body weight was reported for cassia gum in rats.
Semirefined cassia gum (in dog food) was administered to 2 groups of 4 male and 4 female Beagle dogs at dietary doses of 1 and 3.5 g/kg/d, respectively, for 90 days. 68 The control group received dog food without cassia gum; however, cassia gum was replaced by a substance with similar technological characteristics. A dose-related increase in water consumption was the only treatment-related effect noted but was not considered toxicologically significant. Hematological effects and effects on biochemical parameters and organ weight were not considered treatment related, and there were no treatment related necropsy or histopathologic findings. It was concluded that cassia gum in the diet did not induce any remarkable effects.
Two groups of 5 male and 5 female cats (species: Felix catus) of the European (mongrel) strain (9 months old) were fed semirefined cassia gum in the diet (pet feed) at concentrations of 0.5% and 2.5%, respectively, for 13 weeks. 69 The control group received a standard (pet feed) diet without the gum. Neither adverse effects nor treatment-related effects on the following were reported: mortality, behavior, clinical signs, body weight gain, food and water consumption, hematology, clinical biochemistry, organ weights, macroscopy, or microscopy. No lesion was found at necropsy that was indicative of either a local effect on the digestive tract or a general effect on other organs. At microscopic examination, the incidence of changes was similar in all groups, and no lesions indicative of a toxic effect of cassia gum were found.
Ocular Irritation
Cyamopsis tetragonoloba (guar) gum, C siliqua gum, C spinosa gum, and cassia gum
Cyamopsis tetragonoloba (guar) gum (0.1 g) was instilled into the right eyes of New Zealand white rabbits. 70 The eyes of 3 rabbits were rinsed after instillation, and the eyes of 6 were not rinsed. Ocular irritation was scored according to the Draize scale at 1-, 24-, 48-, and 72-hour postinstillation. Cyamopsis tetragonoloba (guar) gum induced minimal ocular irritation in rinsed and unrinsed eyes. Results for other gums (same procedure) were as follows: C siliqua gum (minimally irritating, rinsed, and unrinsed eyes) and cassia gum (nonirritating—rinsed eyes; minimally irritating—unrinsed eyes).
Skin Irritation and Sensitization
Cyamopsis tetragonoloba (guar) gum, C siliqua gum, C spinosa gum, and cassia gum
The skin irritation potential of C tetragonoloba (guar) gum was evaluated using New Zealand white rabbits (3 males and 3 females). 70 The gum (0.5 g/test site) was moistened with saline and applied to shaved, intact skin. Test sites were covered with a semiocclusive wrap for 4 hours. Reactions were scored according to the Draize scale at 4.5, 24, 48, and 72 hours. Cyamopsis tetragonoloba (guar) gum was nonirritating to the skin of rabbits. Results for other gums (same procedure) were as follows: C siliqua gum (minimally irritating; PII = 0.04/8.0), C spinosa gum (nonirritating), and cassia gum (nonirritating).
Hydroxypropyl guar
A leave-on hair styling product containing 2% hydroxypropyl guar was evaluated in a Draize repeated insult patch test involving 111 human participants (ages not stated). 71 During induction, the undiluted test substance (0.02-0.05 mL) was applied to the back under occlusive conditions (8 mm aluminum Finn chamber supported on occlusive tape or an equivalent) for 24 hours. Applications were made 3 times/week for 3 consecutive weeks. After a 2-week nontreatment period, challenge patches were applied to adjacent new sites on the back for 24 hours. Reactions were scored at 48 and 96 hours postapplication. There was no evidence of skin reactivity in any of the participants during the study.
Allergenicity
Cyamopsis tetragonoloba (guar) gum
The prevalence of occupational asthma and immunologic sensitization to C tetragonoloba (guar) gum was evaluated in 162 employees of a carpet-manufacturing plant, where this gum was used to adhere dye to the fiber. 72 Immunoglobulin E and IgG antibodies to C tetragonoloba (guar) gum were measured in 133 of the 162 participants who agreed to blood tests. Thirty-seven (23%) participants had a history of occupational asthma, and 59 (39%) had a history of occupational rhinitis. Skin prick tests with C tetragonoloba (guar) gum (1 mg/mL) were conducted. Immediate skin reactivity to C tetragonoloba (guar) gum was observed in 8 participants, and 11 participants had serum IgE antibodies to C tetragonoloba (guar) gum. In the second part of the study (161 participants), spirometry and assessment of bronchial responsiveness to methacholine were performed during a regular working day at the time of the workshift or in the following 3 to 4 hours. Five participants had a concentration of methacholine causing a 20% decrease in forced expiratory volume of < 16 mg/mL (significant bronchial hyperresponsiveness) and positive skin reactions to C tetragonoloba (guar) gum. It was concluded that the prevalence of IgE sensitization to guar gum was between 5% (8 of 162 participants, as assessed by skin tests) and 8.3% (11 of 133 participants, as assessed by measurement of serum IgE antibodies).
Case Reports
Cyamopsis tetragonoloba (guar) gum
Three male patients (27, 42, and 49 years) developed allergic rhinitis after exposure to C tetragonoloba (guar) gum. 73 Two of the participants developed rhinitis after 2 years of exposure to fine C tetragonoloba (guar) gum (insulator in rubber cables) powder when opening cables in a power cable factory. The allergenicity of C tetragonoloba (guar) gum in these participants was confirmed using scratch chamber, nasal provocation, radioallergosorbent (RAST) tests, and observations of nasal eosinophilia. A third participant also developed allergic rhinitis after 2 years of exposure to another C tetragonoloba (guar) gum product in a paper factory. A positive skin test and nasal provocation test confirmed the gum-induced allergenicity.
Symptoms of rhinitis and asthma were reported for 3 male atopic participants (29, 30, and 32 years) after exposure to C tetragonoloba (guar) gum on the job. 74 An immediate skin reaction to C tetragonoloba (guar) gum was observed in skin prick tests, and all 3 had high levels of serum IgE antibodies to the gum. When the participants were exposed for short intervals (≤ 4 minutes) to C tetragonoloba (guar) gum powder, isolated immediate bronchospastic reactions were observed in 2 participants , and a dual reaction was observed in the remaining participant.
An allergy prick test was performed on a 38-year-old male employee of a pet food processing company, where he frequently inhaled guar powder. 75 Within 20 minutes of the test, the patient reacted positively to C tetragonoloba (guar) gum. A 10-mm wheal with pseudopods and surrounding flare was observed initially. After 4 hours, the reaction developed into an erythematous swelling (2 cm diameter) that remained for 24 hours.
A 52-year-old male participant experienced generalized urticaria and anaphylactic shock after consuming a meal substitute that contained C tetragonoloba (guar) gum. 76 Skin and radioallergosorbent (CAP system) tests on this gum as well as C siliqua gum were performed. Details relating to test procedures were not provided. Results were positive for both gums, with evidence of an IgE-mediated mechanism.
Cyamopsis tetragonoloba (guar) gum and C siliqua gum
A 63-year-old male experienced a life-threatening immediate-type hypersensitivity reaction after mucosal application of a local anesthetic gel that contained C tetragonoloba (guar) gum. Severe contact urticaria and dyspnea were reported, and the patient collapsed. 77 A 1-fold positive prick test reaction to C tetragonoloba (guar) gum and a 2-fold positive prick test reaction to native guar beans were reported. Prick test results for a highly purified molecular grade C siliqua gum were negative. Negative IgE assay results for C tetragonoloba (guar) gum and C siliqua gum were reported. A possible explanation for the discrepancy between prick test and IgE assay results for C tetragonoloba (guar) gum could be the varying degree of allergen contamination in different guar products, remaining from the germ or hull of the Cyamopsis bean, that are not detected by commercial IgE assays that were probably established with highly purified C tetragonoloba (guar) gum.
A 48-year-old male complained of work-related rhinitis, irritated eyes, and asthma after exposure to C siliqua gum and C tetragonoloba (guar) gum on the job in a jam factory. 78 Skin prick tests on C siliqua gum and a blend of C tetragonoloba (guar) gum, C siliqua gum, and carrageenan were negative. Asthma attacks did not occur after the patient stopped handling the gums. When a single-blind provocation test (manipulation of carob bean flour for 15 minutes) was performed, cough, rhinitis, and sneezing developed. Both C tetragonoloba (guar) gum and C siliqua gum were evaluated in the RAST, and the results were positive.
A 59-year-old female developed lip edema a few minutes after ingesting a dessert. 79 She also complained of nasal hydrorrhea and sneezing while handling powder to prepare the dessert. Results of skin prick tests indicated a positive reaction to C siliqua gum in saline (11 mm wheal), and the test concentration was not stated. Skin prick test results for C siliqua gum at concentrations of 5, 15, and 25 mg/mL induced 6, 8, and 9 wheals, respectively. Positive skin prick tests were also reported for raw C siliqua gum (14 mm wheal) and boiled C siliqua gum (9 mm wheal). Control skin prick test results were negative in 10 nonatopic participants. The positive test results and high titers of serum-specific IgE to C siliqua gum supported an IgE-mediated mechanism. Skin prick test results for C tetragonoloba (guar) gum were negative.
Ceratonia siliqua gum
A 30-year-old male with allergic rhinitis developed asthma regularly after handling carob bean flour. 80 A prick test for C siliqua gum and an RAST were both positive (U RAST = 8.86, class 2).
Urticaria and vomiting were reported after feeding of an 8-month-old infant with a milk-based antiregurgitation formula containing C siliqua gum as a thickening agent. 81 Feeding with a milk-based antiregurgitation formula thickened with waxy rice starch was readily accepted.
Trigonella foenum-graecum seed powder
Cases of immediate allergy following exposure to T foenum-graecum seed powder have been reported. 82 A 36-year-old female with a history of allergy to chickpeas and mild asthma experienced sneezing, rhinorrhea, and excessive tearing after smelling T foenum-graecum seed powder. These signs were followed by persistent coughing, wheezing, and fainting. Scratch testing with 10 µL of the legume extract (contained ∼ 22-25 µg protein) revealed a severe (4+) reaction. According to another report, a 45-year-old female with a history of allergic rhinitis and asthma developed congestion and hoarseness shortly after applying T foenum-graecum seed paste to the scalp. These signs were followed by facial angioedema, wheezing, and numbness of the head. Scratch testing revealed a 3+ reaction to the legume extract.
Clinical Testing
Cyamopsis tetragonoloba (guar) gum
An experimental trial to evaluate the effect of C tetragonoloba (guar) gum on arterial blood pressure was performed using 40 moderately overweight men with mild hypertension. 83 Each participant received 7 g of the gum 3 times daily for 2 weeks. There were no changes in body weight or body composition; however, total cholesterol decreased during the feeding period. Blood pressure was found to decrease by 9.8% (systolic) and 9% (diastolic) after 2 weeks of feeding. Blood pressure readings returned to pretreatment levels after a 3-week washout period. It was concluded that C tetragonoloba (guar) gum exerts a lowering effect on high blood pressure in moderately obese men, even in the absence of any change in body weight.
A study was conducted to determine the efficacy of dietary fiber C tetragonoloba (guar) gum as a therapeutic option for reducing body weight, by performing a meta-analysis of randomized controlled trials. 84 Of the 34 experimental trials identified, only 11 provided data that were suitable for statistical pooling. Results of the meta-analysis indicated a nonsignificant difference in patients who received C tetragonoloba (guar) gum, when compared to patients who received a placebo (weighted mean difference −0.04 kg; 95% confidence interval [CI]: −2.2-2.1). Furthermore, these findings were corroborated by an analysis of 6 trials with similar methodologic features (weighted mean difference −0.3 kg; 95% CI: −4.0 to 3.5). The more frequently reported adverse events included abdominal pain, flatulence, diarrhea, and cramps. The meta-analysis suggested that C tetragonoloba (guar) gum is not efficacious for decreasing body weight.
Eight patients (5 men, 3 women; 49.6 ± 3.05 years) with noninsulin-dependent diabetes mellitus consumed at least 30 g of C tetragonoloba (guar) gum in the diet for at least 16 weeks. 85 Each participant was able to consume 4 granola-type bars (6.6 g guar gum/bar) per day. There were no changes in hematologic, hepatic, or renal function. Serologic screening results indicated no changes in lipid, protein, or mineral metabolism and no changes in electrolyte balance. It was concluded that consumption of 30 g C tetragonoloba (guar) gum per day for prolonged periods is without serious consequences. Five participants ingested C tetragonoloba (guar) gum (1 g in capsule) daily for 10 days. 86 No effects were apparent.
Reproductive and Developmental Toxicity
Oral Studies
Cyamopsis tetragonoloba (guar) gum
Groups of male and female Osborne-Mendel rats (4 weeks old; 34 to 40/group) were fed C tetragonoloba (guar) gum in the diet at concentrations of 0%, 1%, 3%, 4%, 7.5%, or 15% 13 weeks prior to mating, during mating, and throughout gestation. 88 Groups of female rats consumed 0, 0.7, 1.4, 2.7, 5.2, or 11.8 g/kg body weight/d, respectively, during gestation. The animals were killed on gestation day 20. None of the females died during the study. The ingestion of C tetragonoloba (guar) gum prior to mating had no effect on fertility. No effects on the number of corpora lutea or implantations were observed in the 1% to 7.5% C tetragonoloba (guar) gum dietary groups. When compared to the control group, slightly fewer corpora lutea and implantations were observed in the 15% dietary group; however, there was no effect on implantation efficiency. A slight reduction (not statistically significant) in the number of viable fetuses per litter was also noted in the 15% dietary group. Because the number of resorptions was not affected in this group, this reduction appeared to have been an effect of the decreased number of corpora lutea. It was concluded that C tetragonoloba (guar) gum in the diet had no effect on fetal development or sex distribution and was not teratogenic.
In another teratogenicity study on C tetragonoloba (guar) gum, groups of virgin adult female albino CD-1 outbred mice (ages not stated) were mated with young adult males, and observation of the vaginal sperm plug was considered day 0 of gestation. 89 The following groups of mated female mice received daily oral doses of C tetragonoloba (guar) gum (in corn oil; dose volume = 1 mL/kg body weight) on days 6 through 15 of gestation: 22 mice (8 mg/kg/d), 26 mice (37 mg/kg/d), 25 mice (170 mg/kg/d), and 29 mice (800 mg/kg/d). A sham-treated control group was also included, and aspirin served as the positive control. Doses up to 170 mg/kg/d had no clearly discernible effect on nidation or on maternal or fetal survival. Additionally, the number of abnormalities observed in either skeletal or soft tissues of test groups did not differ from the number that occurred spontaneously in sham-treated controls. In the 800 mg/kg/d dose group, a significant number of maternal deaths (6 of 29) were reported. Surviving dams appeared completely normal and the same was true of fetuses. No effects on the rate of nidation or survival of live pups in utero were noted. It was concluded that, under the conditions of this test, C tetragonoloba (guar) gum was not teratogenic in mice. The teratogenicity of C tetragonoloba (guar) gum in rats and hamsters was also evaluated in this study. Except for the administration of different doses and a shorter dosing period for hamsters, the test procedure was the same. Results are summarized subsequently.
Four groups of 24 mated female rats (ages not stated) received C tetragonoloba (guar) gum (in corn oil) at daily oral doses of 9, 42, 200, and 900 mg/kg/d, respectively. Doses up to 900 mg/kg/d had no clearly discernible effect on nidation or on maternal or fetal survival. The number of abnormalities observed in either skeletal or soft tissues of test groups did not differ from the number that occurred spontaneously in sham-treated controls. The following groups of mated female hamsters (ages not stated) received daily oral doses of C tetragonoloba (guar) gum (in corn oil): 22 hamsters (6 mg/kg/d), 22 hamsters (28 mg/kg/d), 25 hamsters (130 mg/kg/d), and 20 hamsters (600 mg/kg/d) on days 6 through 10 of gestation. Doses up to 600 mg/kg/d had no clearly discernible effect on nidation or on maternal or fetal survival. Furthermore, the number of abnormalities observed in either skeletal or soft tissues of test groups did not differ from the number that occurred spontaneously in sham-treated controls. 89
Ceratonia siliqua gum
A 3-generation reproduction study was performed using groups of Charles River albino rats of the CD strain (10 males and 10 females per group). 90 Two groups were fed chow diet containing 2% and 5% C siliqua gum, respectively. The control group was fed chow diet containing 5% cellulose. Parental animals in each generation were fed the test diet for 11 weeks prior to mating and through mating, gestation, and weaning. Two or 3 litters were raised per generation, and the second litter was used to produce the F3b generation. Histopathological examination of major organs and tissues was performed on 10 males and 10 females from each treatment group of the F3b generation. All of the other animals were subjected to gross necropsy only. Statistically significant decreases in premating body weight gain (F0 females fed 2% C siliqua gum) and final body weight (F0 females fed 5% C siliqua gum) were noted. Compared to controls, the following significant differences in organ weight ratios were reported for the F3b group fed 5% C siliqua gum: smaller spleen to body weight and liver to body weight ratios, smaller absolute liver weights, and larger brain to body weight ratio. These differences were due to the highly variable values for these parameters in young rats and because all of the animals may not have been at the same age when killed. These age differences may have also had an effect on organ weight ratios in young animals. It was concluded that C siliqua gum did not cause significant treatment-related effects on reproductive indices or gross microscopic pathology.
The teratogenicity of C siliqua gum (in anhydrous corn oil) was evaluated using the following 6 groups of pregnant adult female albino, CD-1 outbred mice: 13 mg/kg C siliqua gum (20 mice), 60 mg/kg (20 mice), 280 mg/kg (21 mice), 1300 mg/kg (21 mice), 0 mg/kg (20 mice, sham treated), and aspirin, 150 mg/kg (21 mice). 91 Doses were administered orally (intubation) on gestation days 6 through 15 (10 days). On day 17, fetuses were removed by cesarean section, and each was examined grossly for the presence of external congenital abnormalities. Detailed visceral examinations were performed on one-third of the fetuses of each litter, and the remaining two-thirds were examined for skeletal defects. There were no clearly discernible effects on nidation or on maternal or fetal survival at doses up to 280 mg/kg. The number of soft or skeletal tissue abnormalities in test groups did not differ from the number that occurred spontaneously in sham-treated controls. A significant number of maternal deaths (5 of 21 females) occurred in the 1300 mg/kg dose group. However, the surviving dams in this group appeared completely normal and delivered normal fetuses, and there were no effects on the rate of nidation or survival of live pups in utero. It was concluded that C siliqua gum was not teratogenic in mice at the doses administered in this study. The positive control was teratogenic.
In other experiments in the preceding study, the teratogenicity of C siliqua gum was evaluated in rats, hamsters, and rabbits according to a similar test procedure. The following 6 groups of adult female Wistar albino rats were used: 13 mg/kg C siliqua gum (23 rats), 60 mg/kg (21 rats), 280 mg/kg (24 rats), 1300 mg/kg (23 rats), 0 mg/kg (23 rats, sham treated), and aspirin (250 mg/kg, 21 rats). On day 20, the fetuses were delivered by cesarean section. There were no clearly discernible effects on nidation or on maternal or fetal survival at doses up to1300 mg/kg. The number of soft or skeletal tissue abnormalities in test groups did not differ from the number that occurred spontaneously in sham-treated controls. It was concluded that C siliqua gum was not teratogenic. In the experiment involving pregnant adult female, golden outbred hamsters, the following groups were used: 10 mg/kg C siliqua gum (21 hamsters), 45 mg/kg (20 hamsters), 220 mg/kg (24 hamsters), 1000 mg/kg (20 hamsters), 0 mg/kg (20 hamsters, sham treated), and aspirin, 250 mg/kg (20 hamsters). Doses were administered orally on gestation days 6 through 10 (5 days). Except for the dosing period, doses administered, and day of cesarean section (day 14), the protocol was identical to the one used for mice and rats. There were no clearly discernible effects on nidation or on maternal or fetal survival at doses up to1300 mg/kg. The number of soft or skeletal tissue abnormalities in test groups did not differ from the number that occurred spontaneously in sham-treated controls. It was concluded that C siliqua gum was not teratogenic in hamsters. 91
The teratogenicity of C siliqua gum (in anhydrous corn oil) was evaluated using the following 6 groups of pregnant adult Dutch-belted female rabbits: 9 mg/kg C siliqua gum (11 rabbits), 42 mg/kg (12 rabbits), 196 mg/kg (13 rabbits), 910 mg/kg (12 rabbits), 0 mg/kg (14 rabbits, sham treated), and 6-amino nicotinamide, 2.6 mg/kg (13 rabbits, on day 9). Doses were administered orally (intubation) on gestation days 6 through 18 (13 days) of gestation, and the fetuses were delivered by cesarean section on day 29. Except for the dosing period, doses administered, and day of cesarean section, the protocol was identical to the one used for mice and rats. Significant maternal toxicity was observed at doses of 910 mg/kg, and, other than hemorrhage of the intestinal mucosa (small intestine), there were no gross pathological findings at necropsy. The fetuses of the highest dose group were normal. All fetuses were examined grossly for the presence of external congenital abnormalities and subjected to examination for visceral abnormalities and skeletal defects. There were no discernible effects on nidation or on maternal or fetal survival at doses up to 910 mg/kg. The number of soft or skeletal tissue abnormalities in test groups did not differ from the number that occurred spontaneously in sham-treated controls. It was concluded that C siliqua gum was not teratogenic in rabbits. 91
Caesalpinia spinosa gum
A multigeneration reproduction study on C spinosa gum was performed using groups of Charles River CD albino rats (22 days old). 92 The gum was administered to male and female rats at a dietary level of 5% (50 000 ppm) through 3 successive generations. All matings involved 10 males and 20 females per group, and fertility was described as high. Litters were maintained until the end of lactation, at which time they were at least 21 days old. There were no consistent, statistically significant test substance-related adverse effects on any of the parameters evaluated, including mortality, food consumption, body weight gains, general health, and behavior. Similarly, regarding mating and reproductive performance indices, there were no consistent, statistically significant differences between control and gum-treated groups. These data suggest that C spinosa gum had no adverse effect on reproductive performance and in utero development. Data from those progeny selected as parental animals for subsequent generations (F1 and F2 parents) indicated that these animals had normal growth patterns and reproductive performance. Gross examination of the parental animals and offspring and microscopic examination of tissues from selected F3b progeny did not identify any abnormalities that were related to administration of the gum. It was concluded that C spinosa gum did not have an adverse effect on reproductive performance or development of progeny.
The teratogenicity and embryotoxicity of C spinosa gum were evaluated using groups of Wistar/HAN rats. Groups of 25 rats were fed pellet-sized diets containing 0, 1.25% (12 500 ppm), 2.5% (25 000 ppm), or 5% (50 000 ppm) of the gum from days 6 to 16 of gestation. All females were killed on day 21 postcoitum, and fetuses removed by cesarean section. None of the animals died, and there were no statistically significant differences in food consumption, body weight gain, general health, or behavior between dams that received control diet and those that received C spinosa gum in the diet. Necropsy results did not reveal any abnormalities that were related to gum administration. There was no evidence of test substance-related abnormalities after external, visceral, and skeletal examinations of fetuses were performed. Additionally, there were no differences in the sex ratios of fetuses or statistically significant differences in fetal body weights. It was concluded that C spinosa gum did not induce maternal toxicity, embryotoxicity, or teratogenicity. Based on the results from this study and the preceding study, the NOAEL was considered to be >50 000 ppm (5%) in the diet. 92
Cassia gum
In a 2-generation reproductive toxicity study (OECD Test Guideline 416), semirefined cassia gum was administered to groups of 25 female Ico: OFA.SD Sprague-Dawley rats (6 weeks old) at dietary concentrations of 0, 5000, 20 000, or 50 000 mg/kg. 93 An additional group was fed a diet containing purified semirefined cassia gum (which resulted from an additional isopropano extraction step) at a dose of 50 000 mg/kg. Parental animals were dosed for approximately 10 weeks prior to mating and during mating, gestation, and lactation. The only effects observed included a slightly reduced pregnancy rate (not observed in a subsequent second mating resulting in an F1b generation), and a slight, nonsignificant decrease in pup weights in the F1a and F2 generations. These effects were observed at the highest dose level (both groups), and therefore, 50 000 mg/kg feed (equal to 5280 mg/kg body weight per day) was considered the no-observed-effect level NOEL.
Groups of 12 pregnant female SD rats received oral doses of cassia gum (by gavage) at doses of 0, 250, 500, or 1000 mg/kg body weight per day on days 7 through 16 of gestation. 54 There were no treatment-related effects on maternal body weight, the number of resorptions or dead embryos, or the weight and length of fetuses. Also, abnormalities were not observed at skeletal or visceral examination of the fetuses. Therefore, no adverse effects were observed in dams or offspring at doses up to and including 1000 mg/kg body weight per day.
The developmental toxicity of semirefined cassia gum (in distilled water) was evaluated using groups of 28 pregnant Crl: CD (SD)BR Sprague-Dawley rats (8 to 12 weeks old). 94 The test substance was administered orally (by gavage) at doses of 0, 350, or 1000 mg/kg per day on gestation days 6 to 19 postcoitum, in accordance with OECD Test Guideline 414. Purified semirefined cassia gum (1000 mg/kg body weight) was administered to a fourth group of 29 pregnant rats according to the same procedure. The animals were killed and examined on day 20 postcoitum. There were no test substance-related effects on pregnancy incidence, implantations, postimplantation loss, or fetal defects at necropsy. In pregnant animals that received semirefined cassia gum or purified semirefined cassia gum at the 1000 mg/kg dose level, a statistically significant reduction in mean daily food consumption and mean body weight gain was noted. These effects were thought to have been related to the viscous nature of the gum and were not considered toxicologically relevant. A statistically significant increase in fetal weight after dosing with purified semirefined cassia gum was the only finding in offspring. There was no evidence of embryotoxicity or teratogenicity in any of the treatment groups, and the NOAEL was 1000 mg/kg body weight per day.
In another developmental toxicity study, groups of 20 pregnant New Zealand white rabbits (14-17 weeks old) were dosed with semirefined cassia gum or purified semirefined cassia gum, following the procedure (same doses for each) in the preceding study. 95 The rabbits were dosed on days 6 to 27 postcoitum and were killed on day 28 postcoitum. The following animals died or were killed moribund during the study: 4 controls, 1 and 4 rabbits dosed with semirefined cassia gum (350 and 1000 mg/kg body weight, respectively), and 2 rabbits dosed with purified semirefined cassia gum (1000 mg/kg body weight). All deaths were attributed to improper gavaging and/or were incidental. A nonsignificant reduction in mean daily food consumption was observed in the group dosed with 1000 mg/kg semirefined cassia gum. A slight, nonsignificant reduction in mean fetal weight was also observed in this group and, possibly, was a secondary effect to the reduced feed consumption. These effects may be related to the viscous nature of cassia gum and were not considered to be of toxicological relevance. No treatment-related adverse effects were observed relative to pregnancy incidence, implantations, postimplantation loss, or fetal defects upon necropsy. Cassia gum was neither teratogenic nor embryotoxic in rabbits, and an NOAEL of 1000 mg/kg body weight per day was reported.
In a 1-generation reproductive toxicity study (OECD Test Guideline 415), semirefined cassia gum was administered to 2 groups of cats (Felis catus) of the Ico: FecEur strain (10 males and 20 females per group) at dietary concentrations of 0.75% w/w and 2.5% w/w, respectively, for 83 to 85 weeks. 96 The control group was fed a standard diet without cassia gum. High mortality in the control group resulted in a high litter loss, which impaired appropriate comparison between treatment and control groups. Generally, no obvious effects on short-term toxicity parameters were observed in the parental generation and offspring. Reproductive performance was not affected, and the same was true for growth and development of the offspring. The following effects, possibly related to dosing, were observed only at the highest administered dose: slight decrease in food consumption during late gestation and a slight increase in absolute and relative ovarian weights in parental females + a significantly higher combined incidence of stillborns and neonatal deaths. In the absence of treatment-related hisotpathological alterations, the changes in ovarian weight were thought to have been of little biological relevance. Because of the unusually high mortality rate in the control group, the JECFA considered this study unsuitable for use in evaluating the reproductive toxicity of semirefined cassia gum. 9
Dermal Study
Cyamopsis tetragonoloba (guar) gum
The teratogenicity of C tetragonoloba (guar) gum (in deionized water) was evaluated using groups of 20 New Zealand white SPF rabbits (4 months old when received). 97 After a 2-month acclimation period, the test substance was administered, via nonocclusive dermal application (6 hours), to 3 groups at doses of 2, 10, and 50 mg/kg/d (dose volume = 2 mL/kg), respectively, on days 6 through 18 of gestation. Applications were made to dorsal skin (10 × 20 cm2). Deionized water was administered to the control group according to the same procedure. Three mortalities were reported, and 3 rabbits either aborted or produced litters prematurely. A dose-related increase in dermal irritation was noted in 10- and 50-mg/kg dose groups, with frequent observations of erythema, edema, and desquamation. Analyses of behavior observations, body weights, and food consumption were not indicative of test substance-related responses in any of the 3 dose groups. A moderate increase in mean postimplantation loss, accompanied by a decrease in viable fetuses, was noted in the highest dose group (50 mg/kg/d). The increase in early resorptions observed in this group was found to be statistically significant (P < 0.05). A slight increase in mean postimplantation loss was noted in the 10-mg/kg/d dose group; however, uncertainty over the significance of this observation was expressed. Increased postimplantation loss was not observed in the 2-mg/kg/d dose group. No other abnormalities were observed in any of the cesarean section parameters. Analyses of fetal morphological observations were not indicative of test substance-related responses in any of the 3 dose groups.
Genotoxicity
Genotoxicity data on galactomannans are summarized in Table 5. Most of the results for bacterial and mammalian assays are negative.
Genotoxicity of Galactomannans.

Carcinogenicity
Cyamopsis tetragonoloba (guar) gum, C siliqua gum, and C spinosa gum
The carcinogenicity of C tetragonoloba (guar) gum, C siliqua gum, and C spinosa gum was evaluated in a National Toxicology Program study using groups of F344 rats (50 males, 50 females/group; 4 weeks old) and groups of B6C3F1 mice (50 males, 50 females/group; 4 to 5 weeks old). 98 The respective groups received either gum in feed at concentrations of 25 000 and 50 000 ppm, respectively, daily for 103 consecutive weeks. Untreated control groups were also used. Animals that were moribund as well as those that survived to the end of the study were killed and necropsied. There were no significant differences in survival between any dose group (rats or mice) and respective control groups. A 10% reduction in body weight gain, compared to controls, was noted in the following dose groups: female mice (50 000 ppm C tetragonoloba (guar) gum), male mice (50 000 ppm C siliqua gum), and male and female mice (50 000 ppm C spinosa gum). In all groups, mean daily feed consumption for dosed rats and mice was ≥79% of respective control values. A variety of nonneoplastic degenerative and inflammatory lesions was observed in rats and mice of all dose groups. However, these lesions were not related to feeding with any of the gums. A number of significant changes in the incidences of primary tumors (P < 0.05, in both positive and negative directions) was reported for rats and mice. In all cases, differences in the tumor incidence observed in one sex were not observed in statistically significant proportions in the other sex of the same species.
A statistically significant increase in the incidence of pheochromocytomas of the adrenal gland was noted in female rats fed both dietary concentrations of C tetragonoloba (guar) gum (25 000 and 50 000 ppm). However, the combined incidence of female rats with pheochromocytoma or malignant pheochromocytoma of the adrenal gland was not significantly different from control values. Similarly, the incidence of pituitary adenomas in male rats fed C tetragonoloba (guar) gum and the incidence of alveolar/bronchiolar adenomas (in lung) in male mice fed C siliqua gum were not significantly different from control values, when combined with incidences of the corresponding carcinomas at those sites. Alveolar/bronchiolar adenomas of the lung in male mice fed 25 000 ppm C siliqua gum and pituitary adenomas in female mice fed the same dietary concentration were considered unrelated to feeding, in that the incidences of these tumors were not significantly increased in the respective 50 000 ppm C siliqua gum groups.
Additionally, the incidence of pituitary adenomas in female mice fed 25 000 ppm C siliqua gum was not significantly different from the historical control rate of this tumor in control male and female mice. The increased incidence of interstitial cell tumors of the testis of male rats was not related to dietary administration of C spinosa gum because of the high spontaneous rate of this tumor. Therefore, in each case in which a significant incidence (P < 0.05) of primary tumors was observed in F344 rats or B6C3F1 mice, a definite association between increased tumor incidence and gum administration could not be made. Decreased incidences of certain other tumor types were also reported for rats and mice fed these gums in the diet. However, when compared to historical control rates, the differences reported were not statistically significant. No histopathologic effects were associated with administration of either of the gums. It was concluded that, under the conditions of these bioassays, C tetragonoloba (guar) gum, C siliqua gum, and C spinosa gum were not carcinogenic in F344 rats or B6C3F1 mice. 98
Cassia gum
Carcinogenicity data on cassia gum were not available for review by the JECFA during its evaluation of the safety of cassia gum as a food additive in 2009. 9 However, given the negative results for cassia gum in genotoxicity assays and negative results reported in carcinogenicity assays on C siliqua gum and C spinosa gum, the Committee determined that a long-term toxicity and/or carcinogenicity study was not necessary for the safety evaluation of cassia gum.
Cocarcinogenicity
The effect of C tetragonoloba (guar) gum in the diet on 1,2-dimethylhydrazine (DMH) initiation of colon tumors was studied using groups of 30 male Sprague-Dawley rats (3 weeks old). 99 One group of rats was fed 5% C tetragonoloba (guar) gum in the diet during the entire initiation period: 4 weeks of acclimatization, 12-week period of DMH injections, and a 2-week postinjection period. During the last 10 weeks (promotion period), the rats were given standard rat pellets. The control group was fed the basic diet. All animals were killed randomly during the 13th week after the last DMH injection and necropsies performed. None of the animals died spontaneously during the study. Rats fed the C tetragonoloba (guar) gum + DMH had more tumors than those fed the basic diet; however, the difference was not statistically significant. It was concluded that C tetragonoloba (guar) gum did not significantly influence carcinogenesis in rats initiated with DMH.
Anticarcinogenicity
The effect of dietary supplementation with C tetragonoloba (guar) gum on 1,2-dimethylhydrazine (DMH)-induced carcinogenesis was studied using groups of 30 male Sprague-Dawley rats (5 weeks old). 100 Each of 60 rats received a basal, fiber-free diet (supplemented with 5% cellulose) and a weekly subcutaneous injection of DMH for 8 weeks (initiation phase), and the animals were then subdivided into 2 groups. One group of 30 rats was then maintained on a basal fiber-free diet supplemented with 10% C tetragonoloba (guar) gum for 24 weeks (promotional stage). The other group of 30 (control) was maintained on a basal diet (fiber free) for 24 weeks. The animals were killed 32 weeks after initiation of the study, and the tumor incidence, location, and frequency in the colon were determined. Compared to the control group, dietary fiber supplementation with 10% C tetragonoloba (guar) gum resulted in a 27% (P = 0.01) decreased incidence of the total colon adenocarcinomas and a 43% (P < 0.0005) decreased incidence of adenocarcinomas in the descending colon.
Summary
The safety of the following ingredients in cosmetics is reviewed in this safety assessment: C tetragonoloba (guar) gum, hydroxypropyl guar, C18-22 hydroxyalkyl hydroxypropyl guar, guar hydroxypropyltrimonium chloride, hydroxypropyl guar hydroxypropyltrimonium chloride, carboxymethyl hydroxypropyl guar, hydrolyzed guar, C siliqua gum, locust bean hydroxypropyltrimonium chloride, hydrolyzed C siliqua gum extract, C spinosa gum, C spinosa hydroxypropyltrimonium chloride, hydrolyzed C spinosa gum, T foenum-graecum hydroxypropyltrimonium chloride, cassia gum, and cassia hydroxypropyltrimonium chloride. Because of the mannose/galactose chemical makeup of these legume polysaccharides, they are commonly called galactomannans.
Data reported to the FDA’s VCRP in 2011 indicated the use of the following 9 ingredients in cosmetics: C tetragonoloba (guar) gum, hydroxypropyl guar, guar hydroxypropyltrimonium chloride, hydroxypropyl guar hydroxypropyltrimonium chloride, hydrolyzed guar, C siliqua gum, C spinosa gum, hydrolyzed C spinosa gum, and cassia hydroxypropyltrimonium chloride. Results from a survey of ingredient use concentrations conducted by the Personal Care Products Council in 2011 indicate that locust bean hydroxypropyltrimonium chloride was also being used in cosmetics. Additionally, results from this survey indicate that galactomannans were being used at concentrations up to 93% (hydroxypropyl guar in a leave-on hair straightener) in cosmetic products.
Production of natural gums consists of various techniques for the milling of seeds, followed by simple purification steps such as dissolving in hot water, filtering, and precipitating with isopropanol. Typical production of the derivatized gums in this report involves the reaction of the natural gum with the appropriate epoxide.
Impurities that have been detected in C tetragonoloba (guar) gum include heavy metal impurities and fluoroacetate. In 2007, the impurities dioxin and PCP were detected in certain batches of C tetragonoloba (guar) gum originating from India. Anthraquinone impurities have been detected in cassia gum (where C occidentalis is a natural contaminant) and tannins, and trypsin inhibitors have been detected in C siliqua gum. A guar hydroxypropyltrimonium chloride trade name material, when heated up to 600°C, contains ash (signifying the presence of inorganic salts) as an impurity at concentrations up to 8.7%.
In rats fed C tetragonoloba (guar) gum in a basal diet, the galactomannan was fermented quantitatively and approximately 1% of the mannose and 4% of the galactose were excreted in the feces. The feeding of rats with C siliqua gum in the diet resulted in 98% digestibility of galactomannan; all of the mannose added to the feed in the form of galactomannan was excreted in the feces. Similarly, 85% to 100% of the mannose fed to rats as C siliqua gum in the diet was excreted in the feces. Based on these study results, it is expected that cassia gum would be largely excreted unchanged and that fermentation by gut microflora may occur to some extent.
In acute oral toxicity studies, the feeding of rats with C tetragonoloba (guar) gum did not cause death or test substance-related adverse effects. The following acute oral LD50 values (rats) have been reported for related ingredients: guar hydroxypropyltrimonium chloride (LD50 = 12.5 g/kg), hydroxypropyl guar hydroxypropyltrimonium chloride (LD50 = 12.0 g/kg), carboxymethyl guar (LD50 = 17.8 g/kg), hydrolyzed guar (LD50 > 6 g/kg), cassia gum (LD50 > 5 g/kg), and C siliqua gum (LD50 = 5 g/kg). The LD50s for C siliqua gum in other species include 13.1 g/kg (mice), 9.1 g/kg (rabbits), and 10.3 g/kg (hamsters).
Most of the repeated dose oral toxicity studies (rats, mostly dietary feeding) involving the following ingredients indicated no test substance-related adverse effects: C tetragonoloba (guar) gum (up to 27%), hydrolyzed guar (up to 5%), C siliqua gum (up to 30%), C spinosa gum (up to 5%), and cassia gum (up to 50 000 mg/kg dietary concentration). Significant reductions in liver weights (at 1%-15% in diet) and kidney weights (at 4%, 7.5%, and 15% in diet) in rats were noted after feeding with C tetragonoloba (guar) gum, and 7 of 10 rats died (possibly due to intestinal blockage) after feeding with C siliqua gum. Increased male kidney–body weight ratios (at 5% in diet) and increased relative weights of the thyroid and testes (at 2% and 5% in diet) in rats were observed after feeding with C spinosa gum; however, the results of gross and microscopic examination of these organs were not indicative of test substance-related changes. Furthermore, feeding with Caesalpinia gum (at 5% in diet) resulted in fewer mature spermatozoa in 4 of 10 male rats. Statistically significant reductions in group mean absolute kidney weights in rats fed cassia gum at dietary concentrations of 10 000 mg/kg and 50 000 mg/kg and at a dose (intragastric) of 1000 mg/kg body weight were not considered treatment related, in that results of gross and microscopic examinations of the kidneys were negative. The same was true for the statistically significant increase in relative kidney weights associated with the 50 000 mg/kg dietary concentration.
Cyamopsis tetragonoloba (guar) gum, C siliqua gum, and cassia gum were minimally irritating when instilled (0.1 g) into the eyes of rabbits. In semiocclusive patch tests (0.5 g applied), C siliqua gum was minimally irritating and C tetragonoloba (guar) gum, C spinosa gum, and cassia gum were nonirritating to the skin of rabbits. Results for a hair styling product containing 2% hydroxylpropyl guar were negative for skin irritation and sensitization in a human repeated insult patch test RIPT. The allergenicity of C tetragonoloba (guar) gum and C siliqua gum following inhalation exposure and ingestion has been demonstrated in various case reports. Also, no effects were apparent after ingestion of C tetragonoloba (guar) gum in another case report. The allergenicity of T foenum-graecum seed powder following inhalation exposure has also been demonstrated in a case study. It was concluded that the prevalence of IgE sensitization to C tetragonoloba (guar) gum was between 5% and 8.3% in an occupational study.
Cyamopsis tetragonoloba (guar) gum was not teratogenic in mice (doses up to 800 mg/kg/d), rats (doses up to 11 800 mg/kg/d), or hamsters (doses up to 600 mg/kg/d) when administered orally, and there was no evidence of maternal toxicity. Ceratonia siliqua gum also was not teratogenic when administered orally to rats (doses up to 1300 mg/kg/d), mice (doses up to 1300 mg/kg/d), hamsters (doses up to 1000 mg/kg/d), and rabbits (doses up to 910 mg/kg/d). However, in one of the studies, significant maternal toxicity was induced by C siliqua gum (at 910 mg/kg/d) in rabbits of the highest dose group. Caesalpinia spinosa gum (up to 50 000 ppm/d) was not a reproductive toxicant or teratogen when administered orally to rats and also did not induce maternal toxicity.
Cassia gum was not teratogenic in rats (doses up to 6120 mg/kg/d), rabbits (doses up to 1000 mg/kg/d), or cats (doses up to 2950 mg/kg/d) when dosed orally. However, a statistically significant reduction in maternal body weight gain in rats was observed in the highest dose group (1000 mg/kg/d) in one study. This finding was thought to have been related to the viscous nature of the gum and was not considered toxicologically relevant. In another study on cassia gum (doses of 1000 mg/kg/d), nonsignificant reductions in feed consumption and fetal weight in rabbits were also thought to have been due to the viscous nature of the gum and were not considered toxicologically relevant. According to the JECFA, the usefulness of the study involving cats is questionable, given the high mortality rate in the control group.
In a dermal teratogenicity study on C tetragonoloba (guar) gum involving rabbits (doses up to 50 mg/kg/d), a statistically significant increase in early resorptions was observed in the highest dose group. However, analyses of fetal morphological observations were not indicative of test substance-related responses at any dose level. A dose-related increase in dermal irritation was noted in the 2 higher dose groups (10 and 50 mg/kg/d).
In bacterial and mammalian assays, C tetragonoloba (guar) gum, C siliqua gum, C spinosa gum, and cassia gum were not found to be genotoxic. In the Ames test, hydroxypropyl guar was mutagenic in 1 of 5 bacterial strains without metabolic activation and in 4 of 5 strains with metabolic activation. Ames test results were negative for hydrolyzed guar and guar hydroxypropyltrimonium chloride (trade name material) with and without metabolic activation. Cyamopsis tetragonoloba (guar) gum, C siliqua gum, and C spinosa gum (concentrations up to 50 000 ppm/d) were not carcinogenic in a dietary carcinogenicity study involving F344 rats and B6C3F1 mice.
Rats fed C tetragonoloba (guar) gum and 1,2-dimethylhydrazine had more tumors than those fed the basic diet, however, the difference was not statistically significant. The anticarcinogenic activity of C tetragonoloba (guar) gum has been demonstrated using rats.
Discussion
Data on toxicokinetics, short-term and repeated dose toxicity, reproductive and developmental toxicity, genotoxicity, carcinogenicity, and dermal irritation and sensitization are not available for all of the ingredients included in this safety assessment, and these data are available for several of the ingredients. Because these ingredients are legume polysaccharides, or derivatives thereof, used for similar purposes in cosmetics, the CIR Expert Panel (Panel) determined that the available data could be used to support the safety of the entire group. In the absence of dermal penetration data, the Panel noted that the galactomannans are large molecules and probably would not be absorbed systemically to any significant level.
The Panel is aware of the prevalence of IgE sensitization to C tetragonoloba (guar) gum reported in an occupational study but determined that the findings were not relevant to cosmetic use. The Panel concluded that this finding was likely due to (1) protein contamination of the gum, considering that allergenicity is generally not associated with carbohydrate moieties and (2) the high breathing zone exposures that are possible in an occupational setting but not relevant to the use of these ingredients in cosmetics.
Given the botanical sources of the galactomannans reviewed in this safety assessment, the Panel expressed concern regarding pesticide residues and heavy metals that may be present. For example, dioxin and PCP impurities have been detected in certain batches of C tetragonoloba (guar) gum. The absence of significant findings in repeated dose toxicity, teratogenicity, or carcinogenicity studies on this gum, however, suggested that the levels of impurities present were not toxic. There was also no evidence that any of the other galactomannans reviewed induced significant toxicity in these types of studies or in skin irritation/sensitization studies. The Panel stressed that the cosmetics industry should continue to use the necessary procedures to limit pesticide residues and heavy metals in the ingredient before blending into cosmetic formulations.
Because hydroxypropyl guar and guar hydroxypropyltrimonium chloride can be used in cosmetics that may be sprayed, the Panel discussed the issue of incidental inhalation exposure. Guar hydroxypropyltrimonium chloride has been used in foot powders and sprays at a maximum concentration of 0.05%. In the absence of inhalation data, the Panel considered data characterizing the potential for galactomannans to cause systemic toxicity, reproductive and developmental toxicity, carcinogenicity, and dermal irritation or sensitization. The Panel noted that 95% to 99% of the droplets/particles produced in cosmetic aerosols would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The Panel concluded that the following cosmetic ingredients are safe in the present practices of use and concentration described in this safety assessment:
Cyamopsis tetragonoloba (guar) gum; hydroxypropyl guar; C18-22 hydroxyalkyl hydroxypropyl guar*; guar hydroxypropyltrimonium chloride; hydroxypropyl guar hydroxypropyltrimonium chloride; carboxymethyl hydroxypropyl guar*; hydrolyzed guar;
Ceratonia siliqua gum; locust bean hydroxypropyltrimonium chloride; hydrolyzed Ceratonia siliqua gum extract*;
Caesalpinia spinosa gum;
Caesalpinia spinosa hydroxypropyltrimonium* Chloride; hydrolyzed Caesalpinia spinosa gum;
Trigonella foenum-graecum hydroxypropyltrimonium chloride*; cassia gum*; and cassia hydroxypropyltrimonium chloride
Were ingredients in this group not in current use to be used in the future (indicated by *), the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Author Contribution
W. Johnson contributed to conception and design, acquisition, analysis, and interpretation and drafted the manuscript. B. Heldreth contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, and critically revised manuscript. L. Gill, W. Bergfeld, R. Hill, C. Klaassen, J. Marks, R. Shank, T. Slaga and P. Snyder contributed to conception and design, analysis, and interpretation and critically revised the manuscript. D. Belsito contributed to conception and design, analysis, and interpretation and critically revised the manuscript. D. Liebler contributed to conception and design, analysis, and interpretation and critically revised manuscript. Former CIR Director F. Alan Anderson contributed to conception and design, analysis, and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.