Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of talc for use in cosmetics. The safety of talc has been the subject of much debate through the years, partly because the relationship between talc and asbestos is commonly misunderstood. Industry specifications state that cosmetic-grade talc must contain no detectable fibrous, asbestos minerals. Therefore, the large amount of available animal and clinical data the Panel relied on in assessing the safety of talc only included those studies on talc that did not contain asbestos. The Panel concluded that talc is safe for use in cosmetics in the present practices of use and concentration (some cosmetic products are entirely composed of talc). Talc should not be applied to the skin when the epidermal barrier is missing or significantly disrupted.
Introduction
This assessment presents information relevant to the safety of talc as used in cosmetic formulations. Reported functions of talc in cosmetics include abrasive, absorbent, anticaking agent, bulking agent, opacifying agent, skin protectant, and slip modifier. 1 The noncosmetic issue of the prohibition of the use of talc in medical examination gloves 2 will not be addressed in this safety assessment.
In 1976, specifications for cosmetic talc requiring that no detectable fibrous, asbestos mineral be present were developed. 3 Therefore, this report will only address the safety of talc that does not contain asbestos. Because the specification was developed in 1976, that year was used in determining what data are more likely relevant to the safety of cosmetic talc; therefore some studies performed prior to 1976 may not be relevant to talc as currently used in cosmetics, and they might not be included in this assessment.
Reviews and responses specific to the National Toxicology Program (NTP) study are included in the section on Carcinogenicity. The following are conclusions from various workshops and review articles on talc: In 1978, the Public Citizen Health Research Group contacted the US Food and Drug Administration (FDA) with a letter stating their concern that talc is possibly carcinogenic and that the FDA should eliminate the use of talc in drugs and cosmetics even if the results are not conclusive.
4
The FDA responded that it was studying talc and believed that any risk from talc was related to contamination by asbestos.
5
In 1983, the FDA received a citizen’s petition requesting that cosmetic talc be labeled with an asbestos warning statement, information on asbestos particle size, and the proportion of impurities in the product.
6
The FDA denied this request, stating that “there is no basis at this time for the agency to conclude that this is a health hazard attributable to asbestos in cosmetic talc. Without evidence of such a hazard, the agency concludes there is no need to require a warning label on cosmetic talc.” In 1992, the Environmental Protection Agency (EPA) issued a “Health Assessment Document for Talc.”
7
The review concluded that talc is not carcinogenic following inhalation exposure or intraperitoneal (ip), intrapleural, or intrabursal administration to rats, hamsters, and mice. However, these studies were not considered fully adequate to evaluate the carcinogenic potential of talc. The review noted that evidence from 2 studies suggests that talc may be an effective cocarcinogen when administered intratracheally with benzo[a]pyrene (B[a]P) to hamsters.
8,9
The Cosmetic Ingredient Review (CIR) Expert Panel determined that the results of these studies were not relevant to the cosmetic use of talc and that the study was not well-designed to study talc. In 1993, the NTP issued a report, “Toxicology and Carcinogenesis Studies of Talc (CAS No. 14807-96-6) in F344/N Rats and B6C3F1 Mice (Inhalation Studies),” that concluded there was some evidence of carcinogenic activity in male F344 rats, clear evidence of carcinogenic activity in female F344/N rats, and no evidence of carcinogenic activity in male or female B6C3F1 mice exposed to aerosols of 6 or 18 mg/m3 nonasbestiform cosmetic-grade talc in a lifetime study.
10
(This study and responses to the report will be described in detail later in this report). In 1994, a public workshop titled “Talc: Consumer Uses and Health Perspectives” was organized under joint sponsorship of the FDA; the Cosmetics, Toiletry, and Fragrance Association (CTFA, now known as the Personal Care Products Council [the Council]); and the International Society of Regulatory Toxicology and Pharmacology (ISRTP).
11,12
The purpose of the workshop was to provide a forum for an updated discussion of the origins, manufacture, characterization, toxicology, and epidemiology of talc and related products. The principal focus was the then-latest toxicological and epidemiological studies as they related to the safe uses of talc in cosmetic products. The characteristics of cosmetic-grade talc, the history of talc use, and quality-control measures for talc were discussed, as was an appraisal of the NTP inhalation study on talc. The regulatory history of talc was also reviewed. The workshop concluded that the NTP bioassay results could not be considered a relevant predictor of human risk, and in regard to proposed association of talc exposure and ovarian cancer, the workshop Panel found that the epidemiological data were conflicting and remain equivocal. In 1994, the Cancer Prevention Coalition (CPC) submitted a citizen petition to the FDA seeking labeling on all cosmetic talc products.
13
The requested labeling was a warning that talcum powder causes cancer in laboratory animals; frequent talc application in the female genital area increases the risk of ovarian cancer. This petition was denied.
14
In 2000, talc was nominated for review in the NTP 10th Report on Carcinogens because the NTP bioassay reported clear evidence of carcinogenic activity of talc (nonasbestiform) based on increased incidences of alveolar/bronchiolar adenomas and carcinomas of the lung in female rats and because published epidemiology studies suggested that talc exposure was associated with lung cancer in pottery workers and ovarian neoplasms in women. (65 FR 17891)
15
However, the NTP deferred consideration of listing talc (cosmetic and occupational exposure; both asbestiform and nonasbestiform) as a carcinogen because of considerable confusion over the mineral nature and consequences of exposure to talc (70 FR 60548),
16
and in 2005, talc was withdrawn from review.
17
In 2008, the CPC again submitted a petition to FDA seeking labeling on all cosmetic talc products.
14
The requested labeling was a warning that frequent application of talcum powder in the female genital area substantially increases the risk of ovarian cancer. It does not appear that FDA has responded to this petition. In 2010, the International Agency for Research on Cancer (IARC) Working Group published that there is limited evidence in experimental animals for the carcinogenicity of talc not containing asbestos or asbestiform fibers.
18
The Working Group reviewed studies in which talcs of different grades were tested for carcinogenicity in mice by inhalation exposure or intrathoracic, ip, or subcutaneous (sc) injection; in rats by inhalation exposure or intrathoracic or ip injection, oral administration, or intrapleural or ovarian implantation; and in hamsters by inhalation exposure or intratracheal injection.
For humans, the determination of the IARC Working Group was that perineal use of talc-based body powder is possibly carcinogenic to humans (Group 2B), and that inhaled talc not containing asbestos or asbestiform fibers is not classifiable as to its carcinogenicity (Group 3). 18 In evaluating the carcinogenicity of talc in humans, the working group reviewed cohort studies of talc miners and millers; cohort and case-controlled studies examining the association of cosmetic talc use and the risk of ovarian cancer in humans; and the animal data and evidence regarding the potential mechanisms through which talc might cause cancer in humans. The working group found there is inadequate evidence in humans for the carcinogenicity of inhaled talc not containing asbestos or asbestiform fibers, and there is limited evidence in humans for the carcinogenicity of perineal use of talc-based body powder.
Many occupational exposure studies are available that describe the effects reported in talc workers. Although the occupational exposure to talc is not at all similar to the cosmetic exposure to talc, these reports are summarized in this safety assessment to provide a total overview of available information. Occupational studies in which talc was known to contain asbestos are not included.
Mineralogy and Chemistry
Definition and Structure
The term talc has 2 meanings: (1) as a mineral, the talc corresponding to the chemical formula of hydrous magnesium silicate and (2) commercially, as a product that can be used industrially in pharmaceuticals and cosmetics. 19 The mineral talc has the formula Mg3Si4O10(OH)2 and 20 a theoretical chemical composition, expressed as oxides, of 31.7% by weight (wt) magnesium oxide (MgO), 63.5% silicon dioxide (SiO2), and 4.8% water (H2O). 21 As a cosmetic ingredient, talc (CAS No. 14807-96-6) is defined as a powdered native hydrous magnesium silicate, sometimes containing a small portion of aluminum silicate. 1
Talc belongs to the silicate subclass phyllosilicates 22 and is a sheet silicate. The structural unit consists of 3 sheets, that is, octahedrally coordinated magnesium hydroxide groups (brucite layer) sandwiched between 2 layers of tetrahedrally linked silica layers. 23,24 The apical oxygen atom positions of the tetrahedral layers are shared with one of the oxygen atom positions of the octahedral layer. 25 The composite sheets repeat every 9.4 Å. Stacks of the triple-sheet crystalline units are held together by van der Waals forces 26 (Figure 27 1).
Small amounts of aluminum and iron(III) can substitute for silicon in tetrahedral sites. 21 Trace amounts of nickel and small to moderate amounts of iron(II), iron(III), aluminum, and/or manganese can substitute for magnesium in octahedral sites. Such substitutions are bound within the crystal lattice and therefore do not exert any biological action. The replacement of hydroxyl groups by fluorine may also occur.
The relationship between talc and asbestos is commonly misunderstood. 26 The presumption that asbestos and talc are commonly associated, or comined, is incorrect. Talc and asbestos (or even asbestiform materials; asbestiform refers to a crystallization product of a mineral in which the crystals are thin, hair-like fibers with enhanced strength, flexibility, and durability 28 ) form under different geological conditions and are separated into adjacent, but disparate, strata. Accordingly, by utilizing proper mining methodologies, asbestos contamination is avoided. Moreover, the absence of asbestos in talc is routinely confirmed in ore samples through a battery of analytical techniques.
Physical and Chemical Properties
The mineral talc has a predominantly plate-like structure, with adjacent layers very weakly bonded by Van der Waals forces. 21 This allows talc to be easily sheared along the plane, giving it its natural slippery feel as well as its softness. Talc is the softest mineral, with a hardness of 1 Mohs (scale of 1-10).
The physical form of talc rock is related to the source and geological conditions that exist during formation of the deposit. 21 The platelet size of talc determines its lamellarity, which, in turn, is related to the genesis of talc deposits. Highly lamellar talc (informally classified as macrocrystalline talc) has large individual platelets, whereas microcrystalline talc has small, randomly oriented platelets. The size of an individual talc platelet can vary from 1 µm to over 100 µm, depending on the formation of the deposit. 29
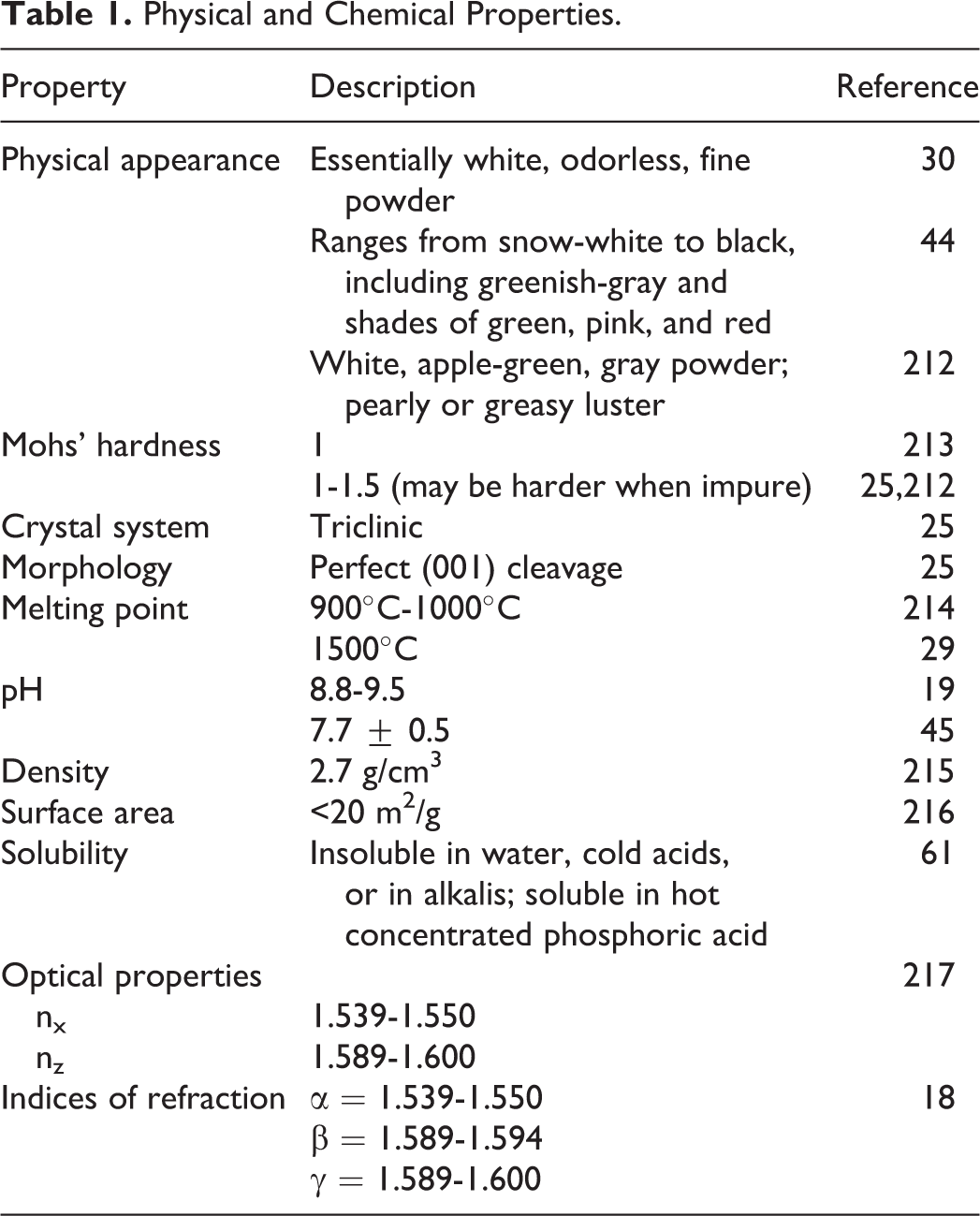
The particle size of talc powder depends on the process used to make the powder. 21 Typical cosmetic talcs have average particle sizes ranging between 4 and 15 μm when measured by sedimentation methods, with only minor fractions consisting of particles considered respirable. Another source recites that the “fineness” of talc used, characterized as 200, 325, or 400 mesh (ie, particle size distribution that allows 95% to 99% of the product to pass through a 200, 325, or 400 mesh, respectively [74, 44, or 37 µm, respectively], when wetted out with alcohol and dispersed in water) depends on the use in cosmetics. 26 For example, 200-mesh talc is preferred for body powders, while 400-mesh talc might be used for pressed powders. The cosmetic ingredient specifications for talc state that in a screen test, 100% passes through 100 mesh, 98% minimum passes through 200 mesh, and finer grades are as specified by the buyer. 30 Physical and chemical properties of talc are summarized in Table 1.
Physical and Chemical Properties.
Analytical Methods
According to CTFA test method J 4-1, the absence of asbestiform amphibole minerals in cosmetic talc is determined using the generally accepted method of X-ray diffraction and optical microscopy with dispersion staining. 31 Other methods for the detection of fibrous amphibole, such as transmission electron microscopy (TEM) with selected area diffraction and electron microprobe, were considered but were not adopted by the cosmetics industry trade association when the testing methods were first published because of the drawbacks associated with those methods, that is, the amount of the material examined is small; the expertise required; and the expense of the equipment. However, electron microscopy, including TEM and scanning electron microscopy, are now routinely used as supplemental and complementary methods of X-ray diffraction and optical microscopy. 32 Infrared spectroscopy, which permits detection at a 0.1% (w/w) minimum detection level, also can be used to identify asbestos in talc. 21
Free crystalline silica (quartz) in talc can be detected using differential thermal analysis, which permits detection at a 0.5 to 1.0% (w/w) minimum detectable level (CTFA test method J 5-1) 33 or by X-ray diffraction (CTFA test method J 6-1). 34
In early studies, the analytical methods used to identify the asbestos in talc were not performed and/or interpreted correctly. Misidentification of asbestos in talc can result from misinterpretation of the data obtained when performing an analytical procedure. 35
Constituents/Impurities
Associated minerals found in commercial talc products vary from deposit to deposit depending on the conditions of formation of the deposit. 21 The most common minerals associated with talc are chlorite, magnesite, dolomite, calcite, mica, quartz, and fluorapatite. Amphiboles and serpentine are associated with certain specific talc deposits. These deposits are rare and historically were used for low-grade industrial applications due to the impurities present.
In 1976, the CTFA issued purity standards for talc. 12 Cosmetic talc consists of a minimum of 90% hydrated magnesium silicate, with remainder consisting of naturally associated minerals such as calcite, chlorite, dolomite, kaolin, and magnesite; it contains no detectable fibrous, asbestos minerals. 30 Additional specifications for cosmetic talc include 6.0% maximum (max) acid-soluble substances; 6.0% max loss on ignition; 3 ppm max arsenic (as As); 20 ppm lead (as Pb); 0.1% max water-soluble substances; no detectable fibrous amphibole (asbestiform tremolite, etc); free crystalline silica (quartz) as specified by the buyer; in a screen test, 100% through 100 mesh, 98% through 200 mesh, and finer grades as specified by the buyer.
As a color additive for drugs, talc sometimes contains a small proportion of aluminum silicate (21CFR73.1550). It is required to meet the specifications for talc listed in the United States Pharmacopeia (USP), and it must also contain not more than 20 ppm lead (as Pb) and not more than 3 ppm arsenic (as As). The following are the acceptance criteria for USP-grade talc: 17.0% to 19.5% magnesium; not more than 0.1% water-soluble substances with neutral pH; no more than 0.25% iron; not more than 10 ppm lead; not more than 0.9% calcium; not more than 2.0% aluminum; and a demonstration of an absence of asbestos. 20 Talc intended for topical application is to have a total aerobic microbial count of not more than 100 cfu/g and a total combined molds and yeasts count of not more than 50 cfu/g; talc intended for oral administration is to have a total aerobic microbial count of not more than 1000 cfu/g and a total combined molds and yeasts count of not more than 100 cfu/g. The acceptance criteria for food-grade talc are not more than 3 mg/kg arsenic and not more than 5 mg/kg lead, and the talc must be derived from deposits that are not associated with asbestos. 36
Batches of cosmetic talc have been analyzed for asbestos and/or asbestiform minerals throughout the years. Analyses performed in the 1970s that indicated asbestos might be present in talc 37 -40 may have used methodology that was unreliable or inaccurate. In the most recent study, which was completed by the FDA in 2012, 9 cosmetic talc suppliers were asked for samples of their talc, 4 complied with the request. 41 The FDA also selected 34 talc-containing retail products. As requested by the FDA, a contract laboratory analyzed the raw material and retail products using polarized light microscopy and TEM, finding no asbestos fibers or structures in any of the samples. The FDA stated that the results were limited, however, because of the limited response by the suppliers and by the number of products tested.
Separate correspondence received by the CIR from the talc industry addressed the issue of the limited response noted earlier from the suppliers of talc. 32 Representatives of the talc industry stated that although not all suppliers of talc (including distributors) contacted by the FDA participated, the study can be considered representative of the US cosmetic talc market as the majority of US cosmetic products were represented.
Sample certificates of analysis were made available from the talc industry. 42,43 One certificate demonstrated that the absence of asbestos was determined using CTFA J 4-1 and USP test methods, 42 and the other stated that the talc products produced by this company do not contain detectable regulated asbestiform minerals. 43
Production
Talc is obtained from naturally occurring rock ore. 30 Talc commonly forms by hydrothermal alteration of rocks rich in magnesium and iron (ultramafic rocks) and by low-grade thermal metamorphism of siliceous dolomites. 25 Soapstone refers to impure, massive talc rock; 19 pure talc was once called steatite. 44 Talc is typically mined in open-pit operations, 26 and cosmetic talcs are mined in Italy, France, Norway, India, Spain, China, Egypt, Japan, and the United States. 45
Crude talc ore can be sorted (beneficiated) to improve the purity of commercial products by either dry or wet processing. 26 In either case, the talc ore is crushed and ground to a fineness suitable for specific end-uses. A dilute talc/slurry water is conditioned for flotation by the addition of a frothing agent (often a low-molecular-weight alcohol), and the slurry is then processed through a series of cells through which air is pumped. This processing causes bubbles to form, and as the bubbles rise to the surface, the talc particles attach to the bubbles due to their organophilic nature; the nontalc impurities are hydrophilic and do not tend to attach to the bubbles. The float (or froth) is then collected. The process is repeated until the desired purity levels are obtained. The talc particles can be further processed by magnetic separation or acid washing to remove iron-bearing minerals, soluble salts, and metals. The talc is then filtered, washed, and dried. Cosmetic talc is typically sterilized by heat treatment. 21
Use
Cosmetic
Talc is reported to have the following functions in cosmetics: abrasive, absorbent, anticaking agent, bulking agent, opacifying agent, skin protectant, and slip modifier. 1 The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA 46 in 2013 and data received in response to a survey of the maximum reported use concentration by category conducted by the Council 47 in 2009 indicate that talc is used in 3469 cosmetic formulations at concentrations up to 100%; it is used in almost every category of cosmetic product. In 2012, the Council completed a survey to assess the frequency and the use concentration of talc in spray products and the highest reported concentration used in spray products was 35% in a makeup base (aerosol). 48 Frequency and concentration of use data are provided in Table 2.
Frequency and Concentration of Use—Complete Table in FDA Format and Summary Information by Exposure Type.
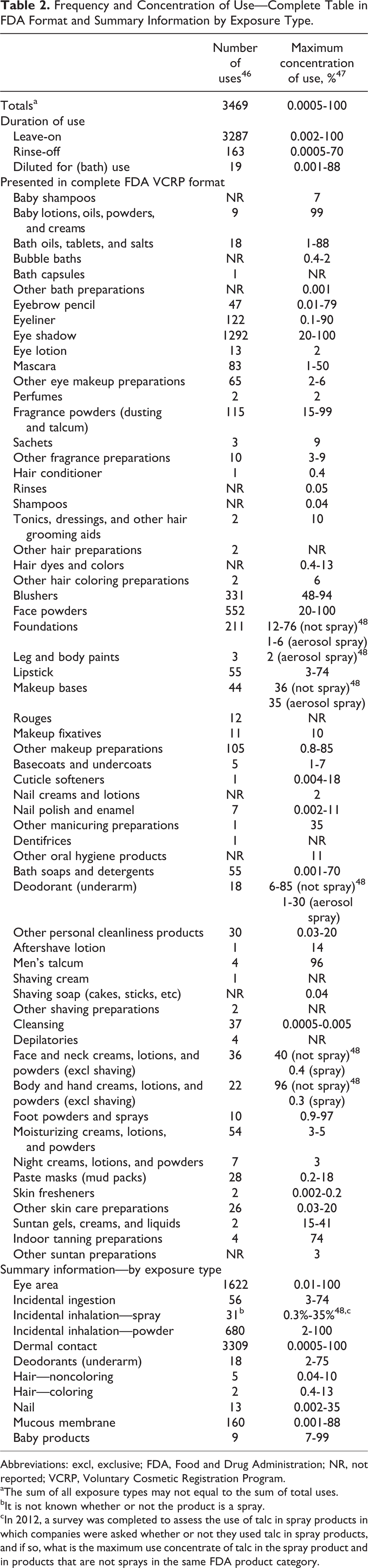
Abbreviations: excl, exclusive; FDA, Food and Drug Administration; NR, not reported; VCRP, Voluntary Cosmetic Registration Program.
aThe sum of all exposure types may not equal to the sum of total uses.
bIt is not known whether or not the product is a spray.
cIn 2012, a survey was completed to assess the use of talc in spray products in which companies were asked whether or not they used talc in spray products, and if so, what is the maximum use concentrate of talc in the spray product and in products that are not sprays in the same FDA product category.
Products containing talc may be applied to baby skin, used in products that could be incidentally ingested, or used near the eye area or mucous membranes. Additionally, talc is used in cosmetic sprays and powders; for example, talc is reported to be used in face powders at 100%, baby powders at 99%, 47 aerosol makeup bases at up to 35%, and in aerosol deodorants at up to 30%. 48 (Talc is not used in extremely high concentrations in spray or aerosol products because talc clogs the nozzle. 49 ) These products could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 50 -53 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 50,52 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 50 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Studies on exposure during the use of cosmetic talc are summarized in Table 3. 54 -56 Many of the researchers noted that there was a wide variation in times and methods for talc use, often by the same volunteer during different applications. Reported application times ranged from 17 to 31 seconds.
Exposure During Cosmetic Talc Use.
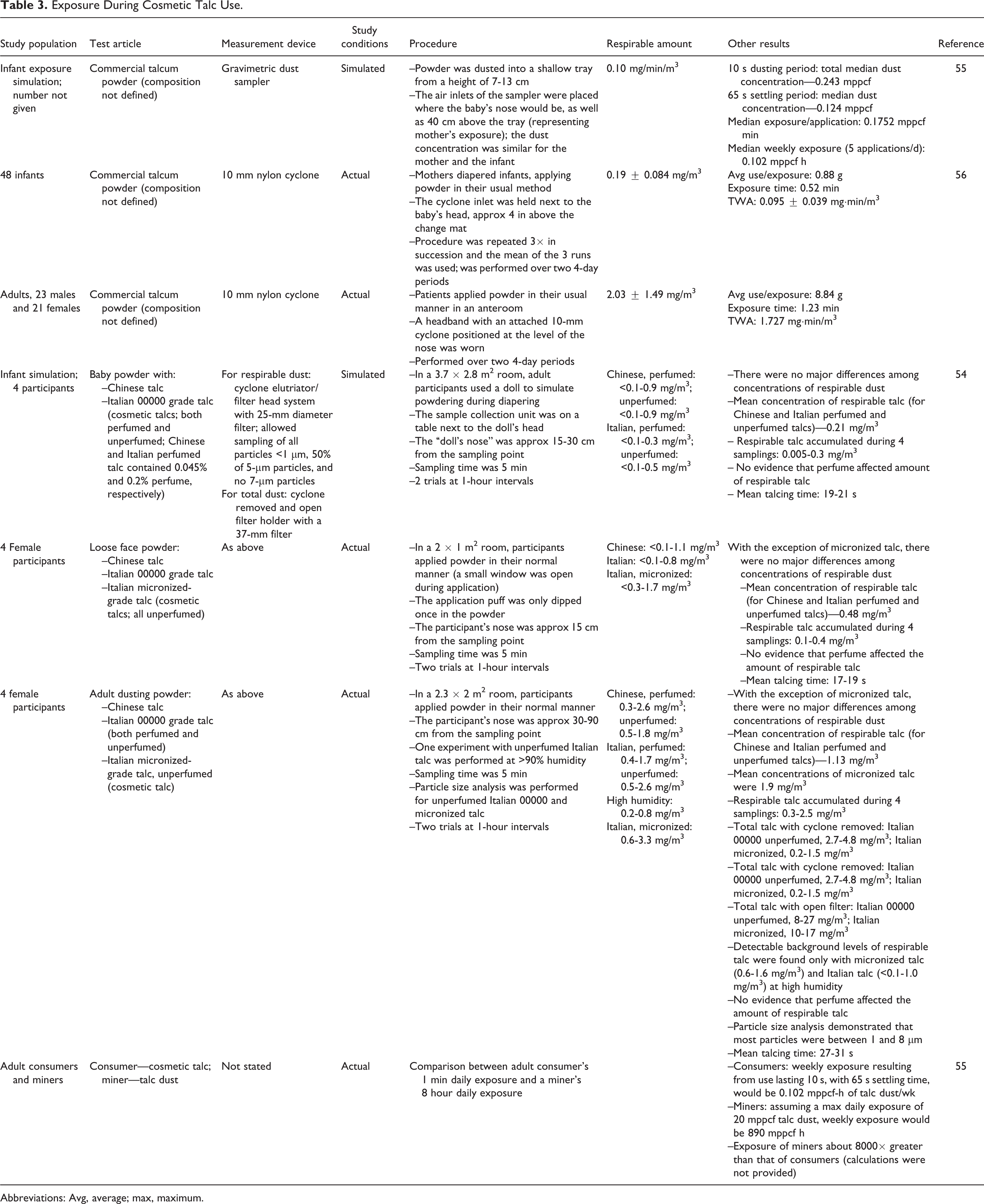
Abbreviations: Avg, average; max, maximum.
The particle size of talc raw material varies widely by product type and by manufacturer but has “no practical significance with regard to human exposure since encapsulation by the other ingredients in the product matrices” (such as a lipstick or deodorant stick) “renders the talc constituents essentially nonrespirable”. 26 Semi-solid matrix formulations (typically pressed powders such as blushes, eye shadows, pressed finishing powders, and base powders) incorporate binder systems. Fine talc, with a larger than average particle size (200 mesh), is often preferred for use in blushes, eye shadows, and finishing powders. Loose-talc–based formulations, such as loose finishing makeup powders, baby powders, body powders, and foot powders, do not include a binder system. The majority of cosmetic talcs in loose-matrix powders contain talc particles that are of a larger diameter than those used in other cosmetic applications; for loose powders, a 200 mesh is normally used, and in these loose powders, substantial agglomeration occurs due to electrostatic and crystalline charges on the talc powders.
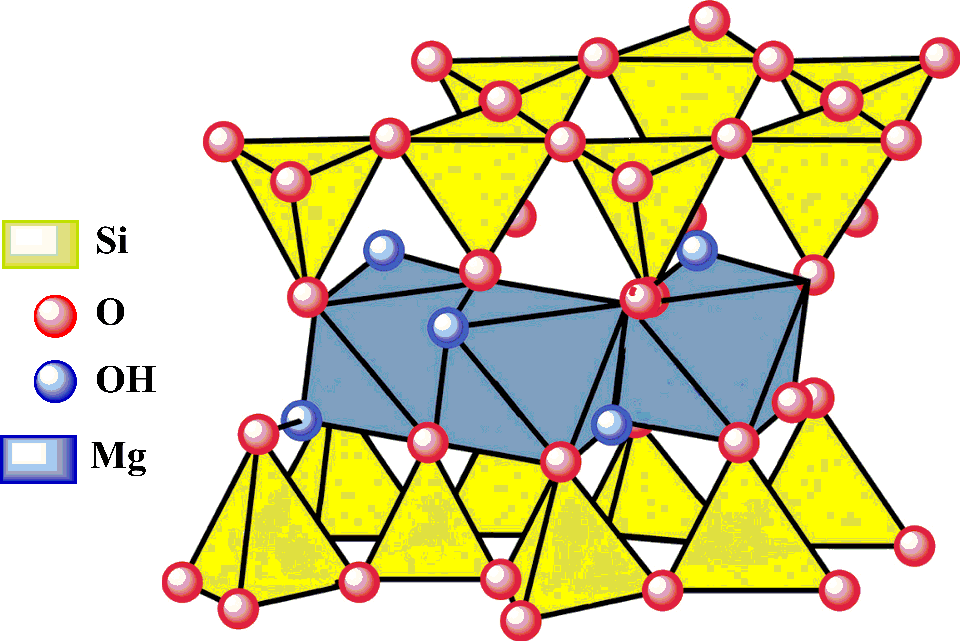
Schematic structure of talc. 27
While some researchers state that the inclusion of a fragrance oil may act as a minimal binder system causing further agglomeration, 26 another researcher found that there was no evidence that the presence of perfume in adult or baby dusting powders containing Italian 00000 grade talc or Chinese talc influenced the level of respirable talc dust. 54
In the European Union, the use of talc in powdery products intended to be used for children under 3 years is restricted by the requirement of labeling that warns to keep powder away from children’s nose and mouth. In Canada, the inner and outer label of preparations in powder form intended for infants and children shall carry cautionary statements to the effect: “Keep out of reach of children,” “Keep powder away from child’s face to avoid inhalation which can cause breathing problems.” 57
Noncosmetic
Sterile talc is approved as a sclerosing agent. 58 Sterile talc powder is indicated for administering intrapleurally via chest tube to decrease the recurrence of malignant pleural effusions in symptomatic patients. Talc is not allowed for use on the surface of medical gloves. 59
Talc is used as a color additive in drugs and is exempt from certification and it may be safely used in amounts consistent with good manufacturing practice to color drugs (21CFR73.1550). In foods, talc is used as an anticaking agent, coating agent, lubricating and release agent, surface-finishing agent, and texturizing agent. 36 Talc is generally recognized as a safe substance migrating from cotton and cotton fabrics used in dry food packaging (21CFR182.70) and as a substance migrating to food from paper and paperboard products (21CFR182.90). It is approved as an indirect food additive as a colorant (21CFR 176.170; 21CFR178.3297). According to the World Health Organization, the acceptable daily intake for talc (as magnesium silicate) is “not specified.” 60
The FDA determined that data are inadequate to establish general recognition of the safety of talc as an active ingredient (astringent) in over-the-counter (OTC) drug products (21CFR310.545(e)(18)(ii)).
Talc is used as a dusting powder, alone or with starch or boric acid, for medicinal and toilet preparations. 61 It is used as an excipient and filler for pills and tablets, for dusting tablet molds, and for clarifying liquids by filtration. Talc is also used as a pigment in paints, varnishes, and rubber; as a filler for paper, rubber, and soap; in fireproof and cold-water paints for wood, metal, and stone; for lubricating molds and machinery; as glove and shoe powder; and as an electric and heat insulator. Talc is used in the leather industry, in the roofing and ceramic tile industry, as a carrier for insecticides and herbicides, 55 and it is used in plastics. 27
Toxicokinetics
Inhalation
Nonhuman
To determine the deposition, distribution, and clearance of talc, 44 female Syrian golden hamsters received a single 2-hour nose-only exposure to a neutron-activated talc aerosol and subgroups of 4 animals were then killed at 11 different intervals from 15 minutes to 132 days after exposure. 62 The talc tested was a commercial baby powder. (Chemical characterization data were not provided). Nine unexposed control animals were used, of which 4 were killed on the day the test animals were exposed and 5 were killed on the final day of the study. The aerosol exposure system had 7 tiers of exposure ports, and the talc aerosol was passed through a cyclone elutriator to remove particles that were larger than ∼10 µm in diameter; the activity median aerodynamic diameter was 6.4 to 6.9 µm. The mean aerosol concentration was 40 and 75 µg/: at the 15 to 30 and 60 to 90 min sampling periods, respectively. In the presentation of the results, the γ-ray counts from the controls were expressed as µg talc equivalent, and the γ-ray counts of the exposed animals were not corrected for control values.
Variations among animals killed at the same time were attributed to variations in aerosol concentration at different tiers. The mean pulmonary talc content in the lungs of test animals at various time intervals was 33.08 (15 minutes after exposure), 24.08 (100 minutes), 42.70 (4 hours), 18.75 (21 hours), 21.30 (2 days), 21.03 (after 4 days), 13.85 (after 8 days), and 8.95 µg (after 18 days); the mean for the day 0 control animals was 1.78 µg. The biological half-life of the talc deposited in the lungs was 7 to 10 days. At the time of termination of the final group, that is, 132 days, there was no statistically significant difference in the talc burden of the lungs of test (3.70 µg) and control (2.30 µg) animals. The amount of talc in the liver, kidneys, and lungs was also determined; the only statistically significant differences compared to controls in any of these organs were found in the liver; there was a decrease at 4 hours compared to day 0 controls, an increase at day 36 compared to both days 0 and 132 controls, and an increase on day 68 compared to day 132 controls. Analysis of the data using the Kruskal-Wallis test showed that there were no significant differences among the mean talc burden values for the liver, kidneys, and ovaries, including the control values, and that there was no significant trend, indicating there was no translocation of talc to these tissues. As noted, no translocation from the respiratory tract to other tissues was found in this study, and the clearance of talc from the lungs was complete within 4 months after exposure.
Oral
Nonhuman
Six female Syrian golden hamsters (outbred Ela: ENG strain) were dosed by gavage with 1 mL neutron-activated talc suspended in physiological saline containing 0.6% (w/w) 1% methyl cellulose (concentration not specified), and the animals were killed 24 hours after dosing. 63 The talc used was a commercial baby powder. (Chemical characterization data and particle size were not provided). Four hamsters were dosed similarly with a nonirradiated talc solution. The neutron-activated talc was exposed to an integrated neutron flux of 7 × 10 16 n/cm2 30 days prior to dosing. The skinned carcass, gastrointestinal (GI) tract, lungs, liver, kidneys, and excreta were analyzed for 60 Co and 46 Sc by γ-ray spectrometry, and the γ-ray counts were compared with those of 4 hamsters that were not dosed with talc.
The γ-ray counts of the tissue and excreta of the dosed animals were equivalent to a total of 2.94 mg talc. Based on γ-ray counts, 74.5% of the neutron-activated talc was recovered in the feces and 23.5% was recovered in the GI tract, while 1.91% was recovered in the skinned carcass, 0.09% in the urine, 0.04% in the kidneys, and 0.02% in the liver. The amount found in the urine of the hamsters given irradiated talc was statistically significantly increased compared to the controls. No talc was recovered in the lungs.
The absorption, distribution, and excretion of orally administered talc were determined in mice, rats, and guinea pigs. 64 (Chemical characterization data were not provided). With all species, [ 3 H]talc was administered as a suspension in aqueous (aq) glycerol jelly solution (10 mg/mL; 1 µCi/mL). Four LACA female mice were given a single oral dose of 40 mg/kg body weight (bw) [ 3 H]talc. Two mice were killed at 6 hours and 2 at 24 hours after dosing. In the mice killed 6 hours after dosing, 95% and 96% of the radioactivity was recovered in the large intestines and feces, 9% and 7% was recovered in the small intestines and stomach, and 0.7% and 0% in the urine of each mouse. In the 2 mice killed at 24 hours after dosing, 99% and 101% of the radioactivity wasrecovered in the large intestines and feces, 4% and 6% was recovered in the small intestines and stomach, and 1.3% and 1.5% in the urine of each mouse. Less than 0.005% of the radioactivity was found in the carcass of any of the mice.
Three male Wistar albino rats were given a single oral dose and 3 rats were given 6 daily oral doses by gavage of 50 mg/kg bw [ 3 H]talc. After the last dose, urine and feces were collected every 24 hours for 4 days and on day 10 and then the rats were killed. Within 24 hours after administration of the single dose, approximately 75% of the radioactivity was recovered in the feces and only 1% was recovered in the urine. After 96 hours, a total of 95.8% of the dose was excreted in the feces and 1.7% in the urine, with a total excretion of 97.5% of the dose. No radioactivity was recovered in the liver or kidneys 10 days after a single dose of talc. On day 10 in the rats given 6 daily doses of [ 3 H]talc, there was no radioactivity found in the feces or livers, and there was a trace of radioactivity (<0.02%) in the kidneys of these rats.
Three female Dunkin Hartley guinea pigs were administered a single oral dose of 25 mg/kg bw [ 3 H]talc, and urine and feces were collected as described previously; all animals were killed on day 10. Talc was excreted more slowly in the guinea pig than in the rat. Within 24 hours after dosing, 31% of the radioactivity was recovered in the feces, and 0.2% was recovered in the urine. At 24 to 48 and 48 to 72 hours after dosing, 39% and 19% of the radioactivity, respectively, was recovered in the feces, with <0.01% of the dose being recovered in the urine at each of these time periods. Within 96 hours of dosing, a total of 94.4% of the radioactivity was recovered in the feces and 0.2% was recovered in the urine, with a total of 94.6% of the dose being excreted over 96 hours.
Intrapleural
Nonhuman
Wistar rats were used to determine the systemic distribution of talc following intrapleural administration. 65 Groups of 20 rats (sex not specified) were administered 10 or 20 mg talc in 1 mL of saline as a slurry into the pleural cavity. (Chemical characterization data were not provided). Ten animals of each group were killed 24 hours after instillation, and the remaining 10 animals were killed 48 hours after instillation. The lungs, chest wall, liver, kidneys, spleen, heart, and brain of each animal were removed for examination. There were no gross lesions in the examined tissues. Microscopic examination revealed that the chest wall had the most common lesions, and these lesions were represented by an early pneumoconiosis characterized by stellate interstitial collections of dust-laden macrophages containing pale yellow particles associated with inflammatory infiltrate of lymphocytes with mild fibroblastic proliferation. Polarized light used to locate birefringent particles revealed “large numbers of irregular, strongly birefringence platy, acicular, and ‘Maltese Cross’ crystals that varied in length from 5.7 to 70 µm” in the chest wall. The deposition index of talc crystals was greater in the chest wall and the lungs after administration of 10 mg (3.90 in the chest and 3.18 in the lungs) than 20 mg talc (3.58 in the chest and 2.50 in the lungs); this difference was statistically significant. (It is not stated whether these values were from the 24-hour group, 48-hour group, or an average of the 2). Pneumoconiosis reactions were not observed in the other organs; however, talc crystals were present inside the microvessels of these organs. The researchers suggested talc was absorbed rapidly through the pleura, reaching the systemic circulation with deposition in other organs within 24 hours after administration, and that the distribution was not dose related.
Toxicological Studies
Single Dose Toxicity
Oral
The median lethal dose (LD50) of talc in rats was determined to be 920 mg/kg bw. 66 Ten male rats were dosed by gavage with 5000 mg/kg bw talc suspended in 0.85% saline; all 10 rats died within 24 hours. Groups of 5 rats were then intubated with 50, 100, 500, 1000, 2000, or 3000 mg/kg bw talc in saline. All 5 animals dosed with 3000 mg/kg bw, 4 dosed with 2000 mg/kg bw, 3 with 1000 mg/kg bw, and 1 with 500 mg/kg bw talc died. (Chemical characterization data were not provided).
In another single-dose study in rats, the LD50 was >5000 mg/kg bw. 66 All the animals survived dosing with 5000 mg/kg bw talc in 0.85% saline.
The oral LD50 of 18.3% talc in saline was >5000 mg/kg bw. 66 A single oral dose of 5000 mg/kg bw of talc prepared as an 18.3% (w/v) suspension in saline was administered to 10 male rats. All animals survived, and there were no signs of toxicity.
Inhalation
Eight mice were placed in a box with baby powder that was circulated with compressed air. 67 (Details regarding the composition of the baby powder, the amount of baby powder, or the size of the box were not provided). Two mice were removed from the box at 30-minute intervals, that is, after 30, 60, 90, or 120 minutes. The mice removed after 30 and 60 minutes recovered completely; symptoms that were observed were not specified. The mice removed after 90 minutes died in 5 to 6 hours; the mice removed at 120 minutes died immediately upon removal. The mice that died were necropsied, and the mucous membrane of the airway was found covered with baby powder. Microscopically, hemorrhage, edema, and desquamation of bronchial epithelium admixed with baby powder were observed.
Intrabursal
Groups of 10 anesthetized female Sprague-Dawley rats (10-15 weeks of age) were given a single bilateral intrabursal injection of 100 µL of 100 mg/mL talc in phosphate-buffered saline (PBS) into the bursa around the ovaries, and groups of 3 age-matched, sham-operated, and sham-treated rats were used as controls. 68 Asbestos-free Italian 00000 talc, composed of platy crystals ranging in size from 0.3 to 14 µm, was used. The animals were killed 1, 3, 6, 12, or 18 months after dosing. There was no effect on the production of physiological concentrations of steroid hormones. Gross examination was made for all animals, and microscopic examination was performed 12 months after dosing. One or both ovaries of rats dosed with talc were cystic in appearance at all time periods; no gross changes were seen in the ovaries of the control animals; the cystic structures were not derived from the ovaries but were due to distention of the bursal sac. Focal areas of papillary change were seen in the surface epithelium of 4 injected ovaries but not in any of the controls. There was no correlation between the presence of foreign body granulomas and the presence of the papillary changes. No evidence of cellular lesions or of mitotic activity was seen in the nonpapillary areas of the surface epithelium of injected ovaries, and neoplasia was not observed. Foreign body granulomas, without surrounding inflammation, were seen in the cortical area of 5 of the injected ovaries, with similar lesions in the supracapsular fat in the connective tissue matrix of the capsule. Talc was observed in the granulomas.
Intraperitoneal
The induction of fibrosis following an ip injection of 50 mg/kg bw nonfibrous talc in physiological saline was evaluated in 6 male and 6 female Wistar rats. 69 A granulomatous reaction in which foreign-body giant cells containing refractile materials was observed in the rats at 1 month after dosing, and this lesion was still observed at 3 months but there was no fibrosis.
Groups of 5 female Wistar rats were used to evaluate the toxicity of talc following a single ip injection of 0.02, 0.1, or 0.5 g in 5 mL normal saline. 70 Although the talc was described as irregular crystalline plates, it was also stated that it could vary from all plates to all fibers. The talc was composed of 49% to 56% silicon dioxide, 20% to 22% magnesium oxide, and 6% to 8% calcium oxide; the particle size ranged from 10 to 120 µm, with a mode of 20 µm. The control group was administered saline only. The animals were killed 7 days after dosing. There were no adhesions in the control group, but adhesions were observed, mainly in the upper abdomen, in the test animals; 3 animals of the 0.5 g group had mild/intermediate adhesions and 4 animals in the 0.5 g group had 4 intermediate adhesions. Talc particles could be seen in the adhesions. The parietal peritoneal mesothelium was examined microscopically using the Hautchen technique, and clusters of foci of inflammatory cells were observed scattered on the surface of the peritoneum. Again, talc particles were seen in the center of each focus of inflammatory cells. Powder deposits adherent to the viscera or omentum without adhesions were reported in 3 animals dosed with 0.02 g talc and in all animals dosed with 0.1 or 0.5 g talc; ascites did not occur in any of these animals.
Cellular effects
Cellular effects in various systems are described in Table 4. There were no remarkable results found in studies examining the cellular effect of talc, such as cytotoxicity assays, assays examining the effect of talc on cell viability, or studies on the induction of apoptosis (among others). 69,71 -78
Cellular Effects.
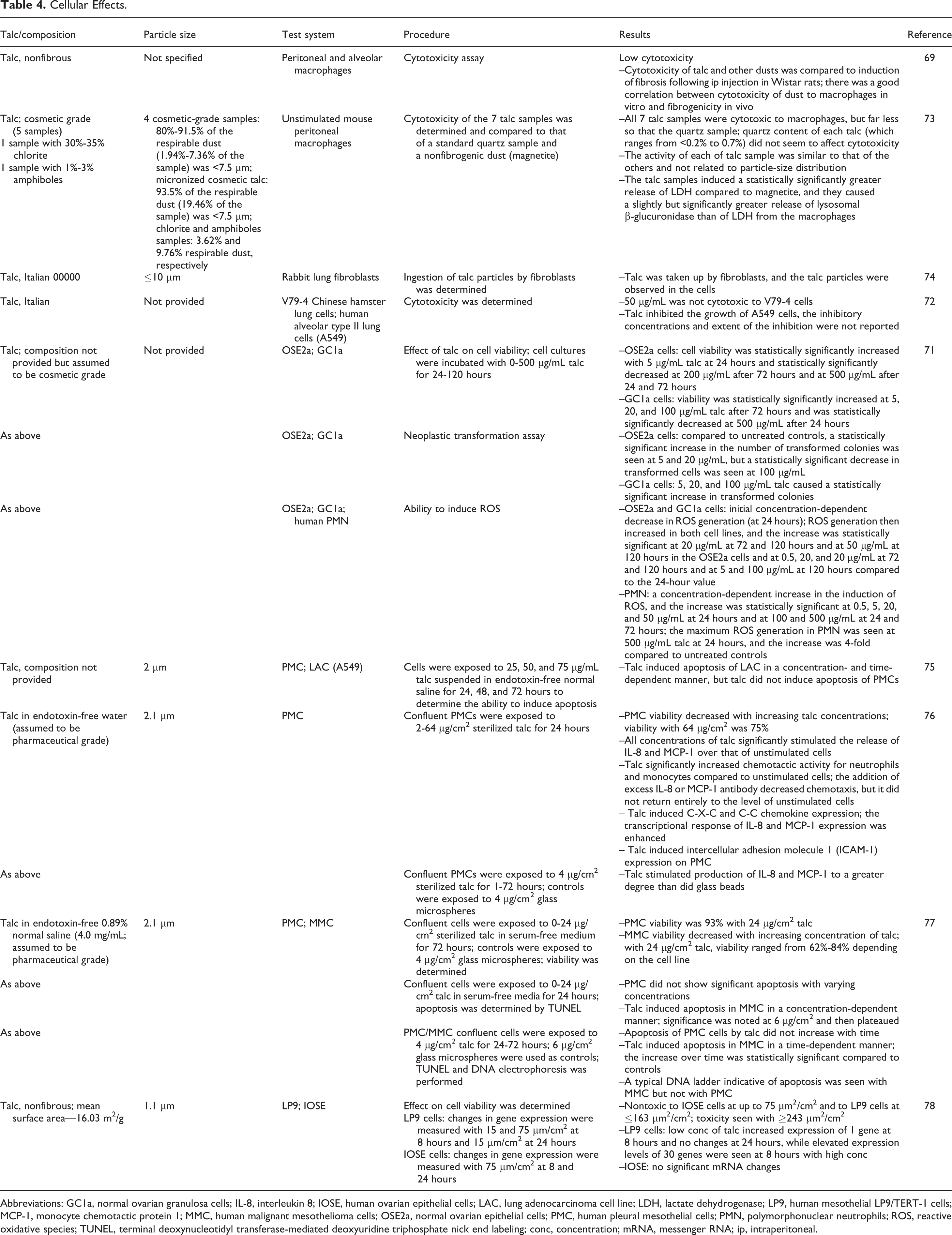
Abbreviations: GC1a, normal ovarian granulosa cells; IL-8, interleukin 8; IOSE, human ovarian epithelial cells; LAC, lung adenocarcinoma cell line; LDH, lactate dehydrogenase; LP9, human mesothelial LP9/TERT-1 cells; MCP-1, monocyte chemotactic protein 1; MMC, human malignant mesothelioma cells; OSE2a, normal ovarian epithelial cells; PMC, human pleural mesothelial cells; PMN, polymorphonuclear neutrophils; ROS, reactive oxidative species; TUNEL, terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling; conc, concentration; mRNA, messenger RNA; ip, intraperitoneal.
Repeated Dose Toxicity
Repeated dose animal toxicity studies are summarized in Table 5. Dermal application of talc to shaved rabbit skin for 6 weeks resulted in dryness of the skin and skin erosion. 79 Oral administration to rats for 5 days produced minimal toxicity 66 ; no toxicologically significant effects were noted in a 5-month study in which rats were fed a diet containing 100 mg/d Italian talc. 80 In inhalation studies, exposure of mice and rats for 4 weeks (25 µm particle size) resulted in macrophages in the alveolar space, with more found in the mice than in the rats. 10,81 In rats exposed for 3, 6, or 12 months, minimal to slight fibrosis resulted. 80 In hamsters, exposure by inhalation to baby powder (95% talc; 4.9-6.0 µmol/L) for 30 days did not result in clinical toxicity. 82 Intrapleural administration of talc (25 µm) to rats did not result in mesotheliomas; granulomas at the injection site were common. 80 Infections occurred, but no neoplastic or perineal changes, when talc was instilled intravaginally or perineally in rats. 83 Upon intravenous (iv) injection of talc (<5 µm) once weekly for 3 weeks in guinea pigs, talc was found in the lungs and the liver throughout the study. 84
Repeated Dose Toxicity Studies.
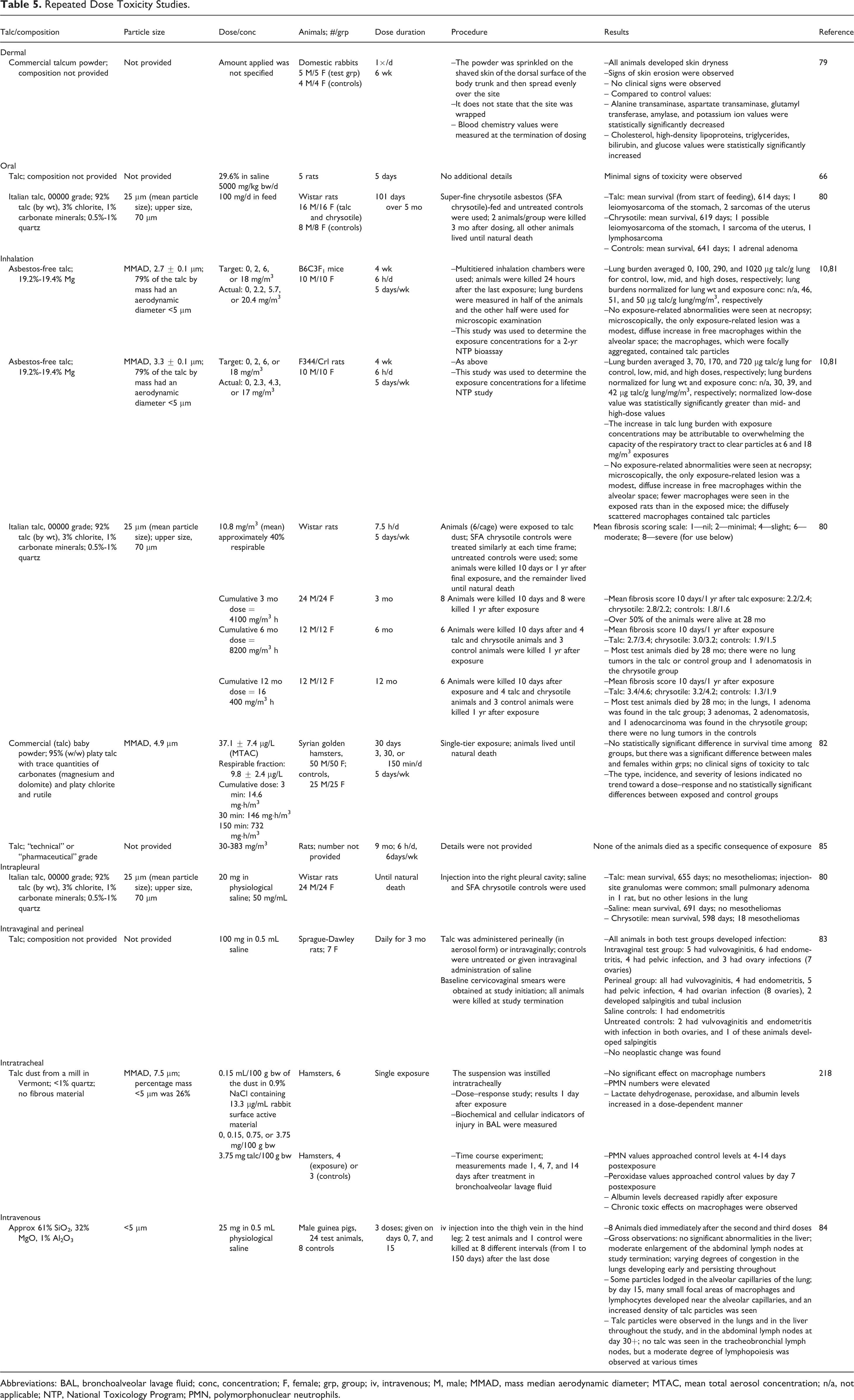
Abbreviations: BAL, bronchoalveolar lavage fluid; conc, concentration; F, female; grp, group; iv, intravenous; M, male; MMAD, mass median aerodynamic diameter; MTAC, mean total aerosol concentration; n/a, not applicable; NTP, National Toxicology Program; PMN, polymorphonuclear neutrophils.
Ocular Irritation
Two unpublished ocular irritation studies were briefly summarized in the International Uniform Chemical Information Database data set on talc. 85 Talc was not irritating to the eyes of rabbits in 1 study and was slightly irritating to the eyes of rabbits in the other study. No details were provided.
A case study was reported in which a woman presented with a foreign body sensation and inflammation of the conjunctiva of both eyes. 86 Following a biopsy and electron microscopy and electron diffraction analysis of the sample, a diagnosis of foreign body granuloma secondary to talc was made. It was postulated that the talc originated from surgical gloves used during a surgery performed decades earlier.
Granuloma Formation in the Skin
Application of talc on wounds can give rise to scab formation, possible infection, and foreign body granulomas in the dermis. 87 In 1 case study, talc powder applied to postvaricella lesions resulted in granulomas. In another case study, hundreds of granulomas of the skin developed in a patient that had open, draining furuncles and who had liberally applied talc daily. 88
Occupational Exposure
Talc has a threshold limit value (TLV; respirable fraction) of 2 mg/m3 as a 10-hour time-weighted average (TWA). 89 The National Institute for Occupational Safety and Health states the immediately dangerous-to-life-or-health concentration is 1000 mg/m3. The Occupational Health and Safety Administration mineral dust limit for talc is 20 million of particles per cubic foot (mppcf) of air, if containing less than 1% quartz; if ≥1% quartz is present, then the quartz limit is used (250/[%SiO2 + 5] mppcf) (29CFR1910.1000 Table Z-3).
Human pulmonary effects of chronic occupational inhalation of talc include diffuse interstitial fibrosis and progressive massive fibrosis (often called complicated pneumoconiosis). 90 Depending on the composition and contaminants of talc, 3 forms of talc-related pulmonary effects have been described: pure talcosis, produced by exposure to talc that is free of silica and asbestiform minerals; talco-asbestosis, produced by the inhalation of talc with asbestiform fibers; and talcosilicosis, produced by exposure to talc associated with silica and other nonasbestiform fibers. 91 A fourth talc-related disease, stemming from iv administration of talc, is not related to occupational exposure but instead is usually associated with abuse of oral medications. Each form has a distinctly different radiographic appearance. The radiographic abnormalities associated with pure talcosis consist of small nodules that are usually seen in the lower pulmonary fields. Reticulations may occur but this is less common. Pure talcosis results in pulmonary function test results that are consistent with restrictive pulmonary disease.
Effects of occupational exposure
Studies examining the pulmonary effects of occupational exposure to talc by talc miners and millers and by workers in industries that use talc are summarized in Table 6. Statistically significantly elevated standardized mortality ratios (SMRs) for silicosis and silico-tuberculosis were observed in an early study of talc miners and millers in the Italian Piedmont region. 92 The miners were employed for at least 1 year and the millers for at least 2 years in their respective occupations. Talc in this region reportedly contained no fibrous material, except for tremolite microinclusions. This study also found statistically significantly reduced SMRs for malignant neoplasms, including lung, bronchial, and tracheal cancers. Updates of this study reported similar results, including statistically significant increases in mortality, which were attributable primarily to nonmalignant respiratory diseases among the miners, no increases in SMRs for cancer, including lung cancer, and no mesothelioma cases. 93,94
Pulmonary Effects of Occupational Exposure.a

Abbreviations: CI, confidence interval; CV, cardiovascular; exp, expected; FEF, forced expiratory flow; FEV, forced expiratory volume; FVC, forced vital capacity; GI, gastrointestinal; GSD, geometric standard deviation; MMEF, maximum midexpiratory flow; NS, nonstatistically significant; obs, observed; ONMRD, other nonmalignant respiratory disease; OR, odds ratio; PMN, polymorphonuclear cells; pop, population; RR, relative risk; SD, standard deviation; SIR, standardized incidence ratio; SMR, standardized mortality ratio; TB, tuberculosis; TCO, transfer factor for carbon monoxide; VC, vital capacity
aBolded text was used to highlight statistically significant increases. Italicized text was used to highlight statistically significant decreases
A cohort study of talc miners and millers employed for at least 1 year found no statistically significant SMRs for all causes, all cancers, or diseases of the circulatory system or respiratory tract. 95 These workers were exposed to talc and magnesite-containing trace amounts of quartz, tremolite, and anthophylite. There were no cases with lung cancer or mesothelioma even among the workers in the highest exposure category.
The results of several other epidemiological studies were likely confounded by the presence of up to 3% silica or 6% actinolite in the talc, exposures to high concentrations of silica with or without exposures to fibrous talc or tremolite, or concurrent exposures to radon daughters. 96 -102
A meta-analysis of studies of miners and millers who worked with nonasbestiform talc reported summary SMRs for lung cancer of 0.92 (95% confidence interval [CI]: 0.67-1.25) for millers in 5 countries exposed to high levels of talc without exposure to other occupational carcinogens, and 1.2 (95% CI: 0.86-1.63) for miners in 3 countries exposed to high levels of talc as well as to silica or radon and radon daughters. 28 The corresponding SMRs for death from all causes were 0.95 for the millers and 1.10 for the miners.
Studies examining radiological, lung function, and clinical (eg, wheezing, coughing, bronchitis) parameters in talc miners and millers and rubber workers found some statistically significant decreases in lung function. 97,103 -107
Case Report
A 70-year-old nonsmoking female was determined to have intense endobronchitis and airway stricture following inhalation of large amounts of cosmetic talc. 108 The patient frequently poured a “small pile of talcum powder” into her hand and applied it to her face. Bronchoscopy showed diffuse, severe endobronchitis that extended throughout both main stem bronchi. Chest radiography and computed tomography imaging showed complete collapse of the right upper and middle lobes of the lung; the right lung was normal with the exception of scattered areas of mild bronchial wall thickening, bronchial plugging, and a few nonspecific nodules. Bronchial biopsies showed edema, chronic inflammation, and fibrosis, and there were confluent foreign-body granulomata that contained birefringent crystalline material. Spectral analysis confirmed the crystals were the same composition as the talc used by the patient.
A case of chronic pulmonary granulomatous reaction was reported in a woman who applied “nonpowdering talc” to her face for 20 years, followed by use of talcum powder 2 to 3 times a day during a 10-year period, usually in an unventilated room. 109 The patient had smoked for 20 years. The amount of powder used per year was described as 2 boxes, but the amount per box was not stated. Chest X-rays showed fine diffuse opacities, and anterolateral thoracotomy showed a diffuse nodular consistency. A heavy intraalveolar and interstitial granulomatous inflammation was found at biopsy, and numerous birefringent particles were found inside the giant cells. The foreign body material contained in the granulomas was characteristic of talc. After 2 years of follow-up, a biopsy of an enlarged lymph node showed granulomatous inflammation. It was the opinion of the investigators that this was a case of not true talc pneumoconiosis but chronic sarcoidosis and coincidental talc deposition in the lung.
Pulmonary talcosis was reported in several cases of misuse of talcum powder in which the patients dusted their entire body with large amounts of powder at least once a day, 110,111 including one in which an individual also dusted the bed sheets every day, 112 and in a case in which the powder was purposefully inhaled. 113 A woman who excessively used talc for herself and her children died from rapidly progressive disease and pulmonary hypertension. Cases of accidental inhalation of large amounts of talc by infants and children have been reported, and consequences have ranged from complete recovery to death. 67,114 -118 Specifics of these cases are not included because the results are not from normal, intended use of the product. Also not included in this safety assessment are reports of adverse effects due to injection of talc with iv drug abuse.
Reproductive and Developmental Toxicity
Oral
Orally administered talc was not a developmental toxicant in mice, rats, hamsters, 119 or rabbits. 120 Chemical characterization of the talc was not provided in any of these studies.
Groups of 20 to 22 gravid albino CD-1 mice and groups of 20 to 24 gravid Wistar rats were dosed by gavage with 0, 16, 74, 350, or 1600 mg/kg bw talc as an anhydrous corn oil suspension on days 6 to 15 of gestation. 119 Aspirin was used as a positive control in both species. The mice were killed on day 17 and the rats on day 20 of gestation and the number of implantation sites, resorptions sites, and live and dead fetuses, and the live pup body weights were recorded. In both mice and rats, the administration of up to 1600 mg/kg bw talc in corn oil had no effect on reproductive or developmental parameters and had no effect on maternal or fetal survival.
In hamsters, groups of 20 to 23 gravid female golden hamsters were dosed by gavage with 0, 12, 56, 260, or 1200 mg/kg bw talc as an anhydrous corn oil suspension on days 6 to 10 of gestation. 119 The animals were killed on day 14 of gestation and examined as described previously. The administration of up to 1200 mg/kg bw talc in corn oil had no reproductive or developmental effects and had no effect on maternal or fetal survival.
Groups of 12 to 15 gravid Dutch-belted female rabbits were dosed orally with 9, 42, 195, or 900 mg/kg bw talc in corn oil on days 6 to 18 of gestation. 120 Eight gravid negative controls were given only vehicle and 9 gravid positive controls were dosed with 2.5 mg/kg bw of 6-aminonicotinamide on day 9 of gestation. The dams were killed on day 29 of gestation. A total of 1/8, 4/15, 2/12, 5/15, and 2/13 dams of the negative control, 9, 42, 195, and 900 mg/kg bw dose groups, respectively, died or aborted before day 29 of gestation, and the number of live litters for these groups was 6/7, 10/11, 8/10, 10/10, and 7/11, respectively. The researchers concluded that administration of up to 900 mg/kg bw talc on days 6 to 18 of gestation “had no discernible effect on nidation or on maternal or fetal survival.” The researchers also stated the number of abnormalities did not differ between test and control animals.
In a dominant-lethal study, groups of 10 male rats were dosed by gavage with a single dose or once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc. 66 Saline was used as the negative control and 0.1 µg/mL triethyl melamine (ip) was the positive control. (The results of the reproductive portion of the study are presented here; the genotoxicity results are presented in that section of the safety assessment). Each treated rat was mated with 2 previously unmated females, and 2 weeks after mating, the female rats were killed and the effects on fertility and preimplantation loss were determined. In the single-dose study, significant dose-related decreases in average corpora lutea and preimplantation losses were reported in the test groups at weeks 4 and 5. In the repeated dose study, significant increases in average implantations and corpora lutea were reported in the test groups at week 6, as were significant differences in the proportions of females with 1+ or 2+ dead implants. However, the results observed at the highest dose did not vary significantly from the negative control, and no dose–response or time-trend patterns were indicated.
Genotoxicity
In Vitro
Talc was not genotoxic in an unscheduled DNA synthesis (UDS) assay or a sister chromatid exchange (SCE) assay in rat pleural mesothelial cells (RPMCs). 121,122 Three samples of European talc (French, Italian, and Spanish talc) were tested. The samples, which contained 90% to 95% talc with chlorite and dolomite, were asbestos free, and the mean particle size of the samples ranged from 2.6 µm (Spanish and French talc) to 4.0 µm (Italian talc). In the UDS assay, the cells were treated with 0, 10, 20, or 50 µg/cm2 of each sample of talc for 24 hours. A negative reference particle control, anatase, and 2 positive controls reference particles, Rhodesian chrysotile and crocidolite, were used, and the mean particle sizes of the 3 talc samples were 0.7, 3.2, and 3.1 µm, respectively. The particles were dispersed in culture medium at a concentration of 560 µg/mL by sonication. None of the talc samples enhanced UDS. The negative and positive particles yielded the expected results.
In the SCE assay, RPMCs were treated with 0, 2, 5, 10, and 15 µg/cm2 of each talc sample for 48 hours. Two negative reference particle controls, anatase and attapulgite, and the 2 positive control reference particles named previously were used, as were the chemical controls mitomycin C in water and K2CrO4 in culture medium. Talc did not cause a statistically significant increase in SCEs and was not clastogenic. The negative particle controls and chemical controls gave expected results; chrysotile and crocidolite statistically significantly increased SCEs in 2/4 and 3/8 experiments, respectively.
In Vitro/In Vivo
Talc was not genotoxic in a host-mediated assay or cytogenetic assay. (Chemical characterization data were not provided in either assay). In the host-mediated assay, male ICR mice served as the host and groups of 10 animals were dosed by gavage with a single dose or once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc. 66 Salmonella typhimurium TA1530 and G46 and Saccharomyces cerevisiae D3 were the indicator organisms. Saline was the negative control and 100 mg/kg bw dimethyl nitrosamine and intramuscular (im) administration of 350 mg/kg bw ethyl methane sulfonate were the positive controls. For comparison, a microdrop of solution, 0.01 to 0.25 mL, of talc was evaluated in an Ames test using S typhimurium TA1530 and G46 and S cerevisiae D3. Talc caused no significant increase in mutant or recombinant frequencies in the host-mediated assay, and it was not mutagenic in the Ames test.
Groups of 15 male albino rats were given a single dose by gavage and groups of 5 rats were dosed once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc in the cytogenetics assay. 66 Saline was used as the negative control and 0.3 mg/kg bw triethyl melamine (ip) was used as the positive control. The concentrations used during the in vitro aspect of the study were 2, 20, and 200 µg/mL in human embryonic lung culture (WI-38) cells. Talc produced no significant aberrations during the in vivo or in vitro phase and was not genotoxic.
In Vivo
Talc was not genotoxic in a rat dominant lethal assay. 66 (Chemical characterization data were not provided). Groups of 10 male rats were dosed by gavage with a single dose or once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc. Saline was used as the negative control and 0.1 µg/mL triethyl melamine (ip) was used as the positive control. There were no dose–response or time-trend patterns; talc did not induce dominant lethal mutations in this assay.
Carcinogenicity
In 2010, the IARC Working Group published the monograph stating that there is limited evidence in experimental animals for the carcinogenicity of talc not containing asbestos or asbestiform fibers. 18 The Working Group reviewed studies in which talc of different grades was tested for carcinogenicity in mice by inhalation exposure or intrathoracic, ip, or sc injection, in rats by inhalation exposure or intrathoracic or ip injection, oral administration, or intrapleural or ovarian implantation, and in hamsters by inhalation exposure or intratracheal injection.
For humans, the evaluation of the IARC Working Group was that perineal use of talc-based body powder is possibly carcinogenic to humans (Group 2B), and that inhaled talc not containing asbestos or asbestiform fibers is not classifiable as to its carcinogenicity (Group 3). 18 In evaluating the carcinogenicity of talc in humans, the Working Group reviewed cohort studies of talc miners and millers, cohort and case-controlled studies examining the association of cosmetic talc use and the risk of ovarian cancer in humans, and the animal data and evidence regarding the potential mechanisms through which talc might cause cancer in humans. The Working Group found there is inadequate evidence in humans for the carcinogenicity of inhaled talc not containing asbestos or asbestiform fibers, and there is limited evidence in humans for the carcinogenicity of perineal use of talc-based body powder.
The references cited by the IARC in their review were obtained by the CIR and are cited as appropriate in this safety assessment.
Inhalation
Exposure of hamsters to talc via inhalation did not produce carcinogenic effects. 82 Groups of 50 male and 50 female Syrian golden hamsters were exposed for 30 or 150 min/d, 5 days/wk, to 27.4 ± 3.4 µg/L mean total aerosol concentration commercial baby powder (95%, w/w platy talc with trace quantities of carbonates and platy chlorite and rutile) until natural death, or, for a maximum of 300 days. A group of 25 male and 25 female hamsters served as the control group. A single-tier exposure was used. There was no statistically significant difference in survival time among groups, but there was a significant difference between males and females within all groups. No clinical signs of toxicity to talc were observed. The type, incidence, and severity of lesions indicated no trend toward a dose–response and no statistically significant differences between exposed and control groups. The incidence of focal alveolar cell hyperplasia (25% in treated groups and 10% in controls) appeared to be affected by treatment, but a 2-way weighted analysis showed no significant association.
A bioassay using mice and rats was performed by the NTP to determine the carcinogenic potential of nonasbestiform, cosmetic-grade talc following exposure by inhalation. 10 There was no evidence of carcinogenic activity in male or female B6C3F1 mice, some evidence of carcinogenic activity in male F344/rats, and clear evidence of carcinogenic activity in female F344/N rats. The talc used was asbestos free and virtually silica-free microtalc; scanning electron microprobe analysis of one lot of talc indicated that 1/1466 particles examined were silica, 136/1466 particles tremolite, and 1241/1466 particles were talc. More than 75% of the particles were in the 1.0 to 3.0 µm range. This study is discussed in greater detail subsequently
A 2-year study was performed in mice; groups of 50 male and 50 female B6C3F1 mice (7 weeks old) were exposed to target concentrations of 0, 6, or 18 mg/m3 talc for 6 h/d, 5 days/wk, for 103 to 104 weeks. The concentrations were selected based on the results of a 4-week inhalation study in B6C3F1 mice, and that study is presented in Table 5. These exposure concentrations provided a dose equivalent of 0, 2, or 6 mg/kg bw/d for male mice, respectively, and 0, 1.3, or 3.9 mg/kg bw/d for female mice, respectively. The mass median aerodynamic diameter (MMAD) was 3.3 ± 1.9 µm in the 6 mg/m3 chamber and 3.6 ± 2.0 µm in the 18 mg/m3 chamber. Groups of 40 male and 40 female mice were similarly exposed and killed at 6, 12, and 18 months for interim microscopic evaluations. Some problems were experienced in maintaining control of the chamber concentrations, and there was a 12-week period beginning at week 70 during which the chamber concentrations were substantially lower than the target concentrations. Mean body weights were similar for test and control animals, and there were no clinical findings attributable to talc exposure.
Compared to the 6-month value, the lung talc burden (normalized to control lung wt) was statistically significantly increased at 24 months in 6 mg/m3 males, at 12 and 24 months in 18 mg/m3 males, at 18 and 24 months in 6 mg/m3 females, and at 12, 18, and 24 months in 18 mg/m3 females. When lung talc burdens were normalized to exposure concentration, a statistically significant difference was observed between the 6 and 18 mg/m3 males at 12 and 24 months but not at 6 and 18 months. The mouse lung talc burdens are provided in Table 7.
Lung Talc Burden in Mice.10,a
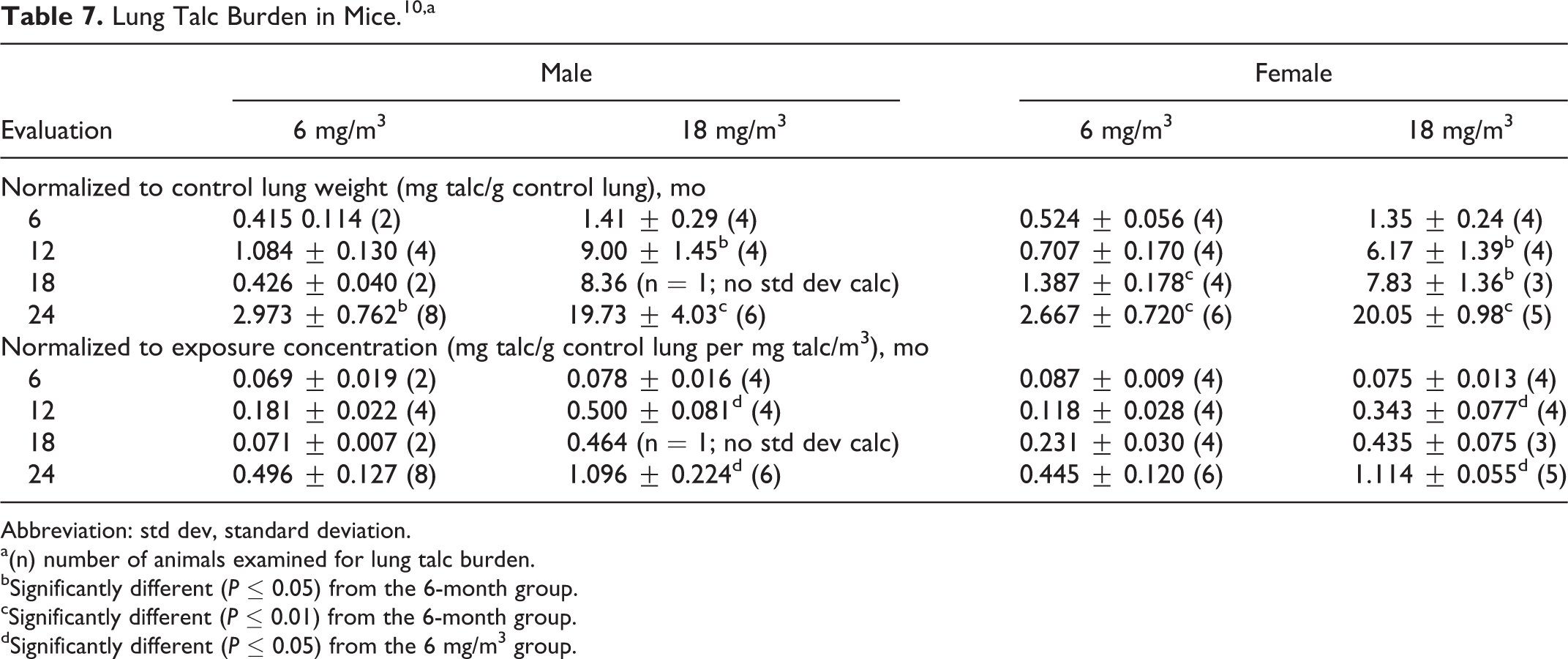
Abbreviation: std dev, standard deviation.
a(n) number of animals examined for lung talc burden.
bSignificantly different (P ≤ 0.05) from the 6-month group.
cSignificantly different (P ≤ 0.01) from the 6-month group.
dSignificantly different (P ≤ 0.05) from the 6 mg/m3 group.
Changes in enzymatic activities in bronchoalveolar lavage fluid were noted mostly in the 18 mg/m3 males and females; measured enzymatic activity was increased in the high-dose animals at 18 and 24 months. A statistically significant increase in β-glucuronidase activity was seen at 12 months in the high-dose animals, and at 24 months, the activity was increased in all test groups. Lavage fluid polymorphonuclear cells were statistically significantly increased in males and females of the 18 mg/m3 group at all times except at 12 months; statistically significant increases were observed in some 6 mg/m3 interim groups. The population of bronchoalveolar lavage fluid macrophages was significantly decreased in the female test groups at 24 months. The phagocytic activity of the macrophages recovered from the lavage fluid at 12, 18, and 24 months was statistically significantly decreased by exposure to 18 mg/m3 talc. At 24 months, there was no effect on the viability of the macrophages. Lung tissue collagen and proteinase activity were significantly increased in exposed male and female mice. At 24 months, collagen and lung fluid collagenous peptides were statistically significantly increased in the 18 mg/m3 group, and most proteinase activity was increased as well.
Chronic active inflammation without alveolar epithelium hyperplasia, squamous metaplasia, or interstitial fibrosis was reported in exposed mice. An accumulation of macrophages was observed in the lungs, and talc-containing macrophages were found in the bronchial lymph nodes. The incidence of pulmonary neoplasms was similar for test and control animals. In the upper respiratory tract, cytoplasmic eosinophilic droplets in the nasal mucosal epithelium occurred and were concentration dependent. There was no evidence of carcinogenic activity in male or female B6C3F1 mice exposed to talc.
A lifetime study was performed in rats; groups of 50 male and 50 female F344/N rats (6-7 weeks old) were exposed to the same dosing regimen and target concentrations of talc as mice until mortality reached 80% in any exposure group, that is, males were exposed for 113 weeks and females for 122 weeks. (The concentrations selected were based on the results of a 4-week inhalation study in F344/N rats and that study is described in Table 5). The MMAD was 2.7 ± 1.9 µm in the 6 mg/m3 chamber and 3.2 ± 1.9 µm in the 18 mg/m3 chamber. As with the mice, there was difficulty in maintaining the chamber concentrations for the rats; there was a 7-week period beginning at week 11 during which time the concentration for the 18 mg/m3 group varied from 30 to 40 mg/m3 and there was a 12-week period beginning at week 70 during which the chamber concentrations were substantially lower than the target concentrations for both groups. Groups of 22 male and 22 female rats were exposed similarly and killed at 6, 11, 18, and 24 months for interim evaluations. Survival was similar for test and control animals. Body weights of the low-dose animals were similar to controls and final body weights of the high-dose animals were slightly (14%) lower than controls. Compared to controls, the absolute and relative lung weights in high-dose males were statistically significantly increased at 6, 11, 18 months, and at study termination; in high-dose females at 11, 18, 24 months, and at study termination; and in low-dose females at 18 months and study termination.
A concentration-related impairment of respiratory function was observed in exposed male and female rats, and the severity increased with increasing duration of exposure. In the 6 and 18 mg/m3 males and in the 6 mg/m3 females, the lung talc burden (normalized to control lung wt) was statistically significantly increased at 11, 18, and 24 months compared to the 6-month value. In the 18 mg/m3 females, the 18- and 24-month values were statistically significantly increased compared to the 6-month values. When lung talc burdens were normalized to exposure concentration, a statistically significant difference was observed between the 6 and 18 mg/m3 males at 6 and 11 months but not at 18 and 24 months. At 24 months, the lung talc burden (normalized to exposure concentration) was higher in the 6 mg/m3 males than in the 18 mg/m3 males. In the females, the only statistically significantly difference between the low- and high-dose groups was at 6 months. The interim rat lung talc burdens are provided in Table 8.
Lung Talc Burden in Rats.10,a
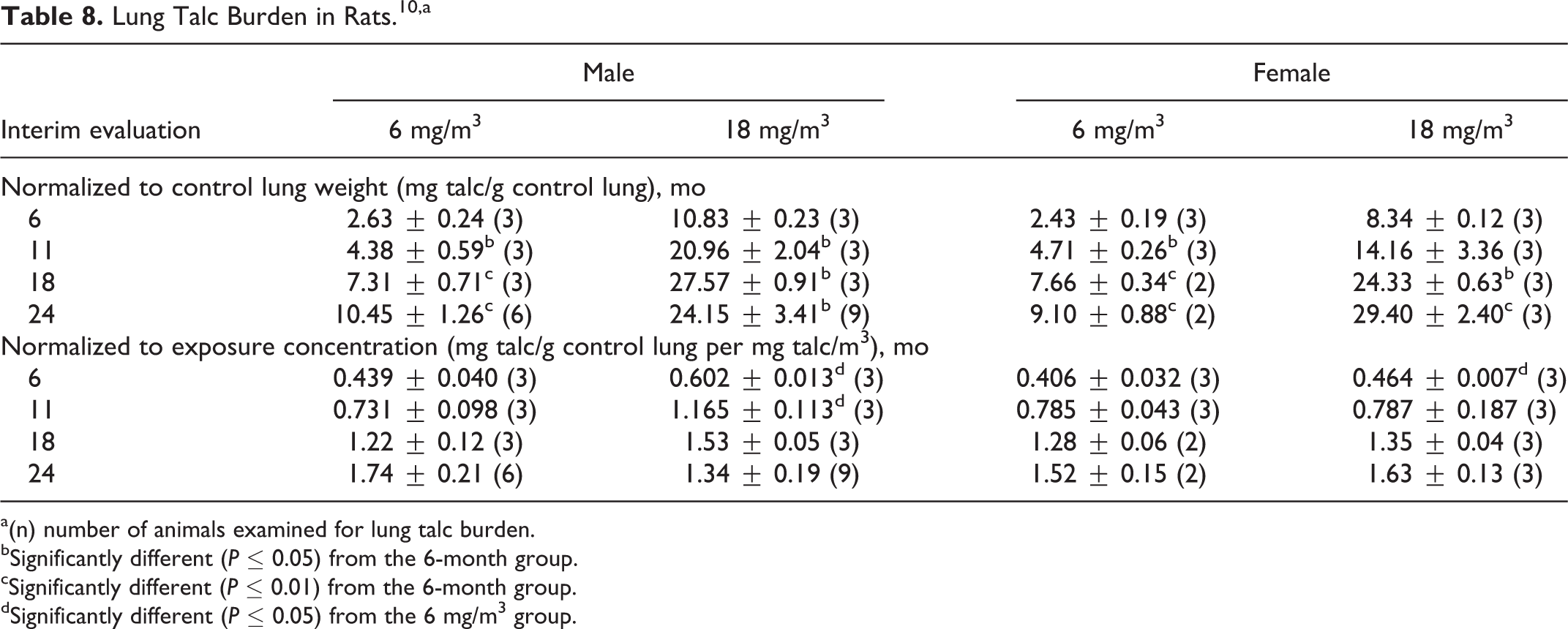
a(n) number of animals examined for lung talc burden.
bSignificantly different (P ≤ 0.05) from the 6-month group.
cSignificantly different (P ≤ 0.01) from the 6-month group.
dSignificantly different (P ≤ 0.05) from the 6 mg/m3 group.
Pulmonary function was impaired (ie, restricted) in a concentration-related manner, increasing in severity with exposure duration. After 24 months of exposure, changes in enzymatic activities in bronchoalveolar lavage fluid were noted compared to controls; statistically significant increases in β-glucuronidase were seen in all test animals. Also, lavage fluid polymorphonuclear cells were statistically significantly increased and macrophage cells were statistically significantly decreased in all test animals; a statistically significant increase in lymphocyte cell populations was reported in all test group females. The viability and phagocytic activity of the macrophages recovered from the lavage fluid were not affected by exposure to talc. Lung tissue collagen and proteinase activity were significantly increased in exposed male and female rats.
Granulomatous inflammation occurred in the lungs of most test animals, and severity increased with duration and concentration. Hyperplasia of the alveolar epithelium and focal interstitial fibrosis were statistically significantly increased at study termination; squamous metaplasia of the alveolar epithelium and squamous cysts were significantly increased in the 18 mg/m3 females only. Talc-containing macrophages were reported in the peribronchial lymphoid tissue of the lung and in the bronchial and mediastinal lymph nodes. In the full study, the incidences of pulmonary neoplasms in male rats of the test group were similar to controls. However, in female rats of the 18 mg/m3 group, the incidences of alveolar/bronchiolar adenoma, carcinoma, and adenoma/carcinoma (combined) were statistically significantly greater than controls; 1 squamous cell carcinoma was reported in this group. In the upper respiratory tract, hyperplasia of the respiratory epithelium of the nasal mucosa was observed in male test animals and accumulation of cytoplasmic eosinophilic droplets in the nasal mucosal epithelium was observed in males and female test animals; the incidence of these lesions was concentration dependent. Benign, malignant, or complex (combined) adrenal medulla pheochromocytomas occurred with a significant positive trend in male and female rats, and the incidences in the 18 mg/m3 group were statistically significantly increased compared to controls. The incidence of adrenal medulla hyperplasia was statistically significantly decreased in exposed males, but not exposed females, compared to controls. It was concluded that there was some evidence of carcinogenic activity of talc in male F344/rats based on an increased incidence of benign or malignant pheochromocytomas of the adrenal gland and clear evidence of carcinogenic activity of talc in female F344/N rats based on increased incidences of alveolar/bronchiolar adenomas and carcinomas of the lung and benign or malignant pheochromocytomas of the adrenal gland.
Responses to/reviews of the NTP inhalation bioassay
One member of the NTP Board of Scientific Counselors, Technical Reports Review Subcommittee, voted against the NTP conclusions on the carcinogenic potential of nonfibrous talc in rats. 123 This board member asserted that talc-induced lung tumors occurred only in the group of animals that experienced the most chronic toxicity and inflammation, and that the lung toxicity data were presented as an empirical observation rather than related to the risk assessment implications of the bioassay. Additionally, it was the opinion of the board member that the evaluation of the pheochromocytomas was inadequate because the spontaneous incidence of this tumor in rats was not sufficiently addressed and that the incidence of pheochromocytomas was not treatment related.
At a talc workshop that was cosponsored by the FDA, CTFA, and ISRTP, a unanimous consensus was reached regarding the NTP talc bioassay. 11 It was the opinion of the Panel at the workshop that “because of the extreme doses and the unrealistic particle sizes of the talc that was used, because of the negative results in mice and male rats, because of the lack of tumor excess at the low doses, and because of the clear biochemical and cytological markers of excessive toxicity in the female rats, the positive talc bioassay results in female F344/N rats were the likely experimental artifacts and nonspecific generic response of a dust overload of the lungs and not a reflection of a direct activity of talc. Given the gross differences of rodent and human lungs, the lung clearance capabilities of humans, and the possible conditions of customary human exposures, the NTP bioassay results in F344/N female rats cannot be considered as relevant predictors of human risk.”
A critical appraisal of the NTP study discussed test concentration selection and the effect of lung particle overload. 124 The appraisal noted that a 4-week study, rather than a subchronic study, was used to determine the test concentrations used in the bioassay; additionally, only 2 test concentrations were used and exposure at these concentrations impaired lung clearance in the 4-week study. The appraisal cited a recommendation that, instead, the long-term bioassay should be performed using 3 concentrations and that only the highest concentration tested should show interference with lung defense mechanisms, and the 2 lower concentrations should not interfere with clearance and particle accumulation. It was the opinion of this appraisal that lung particle clearance in both rats and mice was impaired, resulting in altered accumulation kinetics, with long-term exposure at concentrations of 6 and 18 mg/m3. Therefore, the maximum tolerated dose (MTD) was exceeded at both exposure concentrations, and because the MTD was exceeded, “classification of such particles with respect to human pulmonary carcinogenicity should be considered carefully.” Finally, the appraisal stated that the NTP conclusion of clear carcinogenicity in female rats should be qualified by a statement indicating that the lung tumors that occurred were mostly likely produced secondary to particle overload and related chronic toxicity.
The human exposure to respirable talc particles during normal product use (values obtained from studies by Russell et al 56 and/or Aylott et al 54 ) was compared to the exposure of rats and mice in the NTP study. 26 According to these researchers, based upon the determinations reported in the literature, human exposure to respirable talc particles during normal product use is approximately 2000 to 20 000 times lower than that used for rats and mice in the NTP study.
The International Life Sciences Institute convened the Workshop on Relevance of the Rat Lung Response to Particle Overload for Human Risk Assessment. 125 The workshop addressed studies reporting lung tumors in rats resulting from chronic inhalation of poorly soluble, nonfibrous particles (PSPs) that are of low acute toxicity and not directly genotoxic, including nonasbestiform talc. The workshop noted that PSP-induced tumors in rats are associated with the following sequence of responses: particle accumulation, chronic active inflammation, epithelial cell hyperplasia, and metaplasia; the chronic active inflammation is associated with the emergence of neoplastic cells. It was stated that, although for direct-acting mutagens the rat appears to be a good qualitative predictor of the human lung cancer, for PSPs it appears to be more sensitive than humans and other rodent species at doses and exposure intervals that result in particle overload in the rat lung. However, because it is not known whether high lung burdens of PSPs can lead to lung cancer in humans via mechanisms similar to those in rats, “it was the consensus view of the workshop that there are insufficient data at present to conclude that the PSP-induced tumor response in the rat model is not relevant for human hazard identification. In other words, in the absence of mechanistic data to the contrary, it must be assumed that the rat model of tumorigenicity can identify potential carcinogenic hazards to humans.”
Another comment paper discussed the use of micronized talc in the NTP study, which resulted in a significantly reduced particle size compared to cosmetic talc, that is, 2.7 to 3.2 µm instead of 6.0 to 6.9 µm. 126 The commenter stated that the use of micronized talc significantly affected the bronchopulmonary deposition and clearance characteristics of the inhaled aerosol; the micronized talc particles were deposited deeper in the lung where clearance depended on alveolar macrophages, whereas cosmetic talc particles would have deposited in the ciliated portion of the respiratory tract. The commenter also remarked on the difficulty in controlling aerosol concentrations and that the 7-week period in which the rats were exposed to twice the intended aerosol concentration most likely aggravated an existing overload condition.
Parenteral
Intrapleural
Talc did not induce pleural tumors in rats following intrapleural injection. 121 A group of 35 Sprague-Dawley rats were given an intrapleural injection of 20 mg talc (mean size 2.6 ± 2.3 µm; no other chemical characteristics provided), and control groups were given an intrapleural injection of saline (40 rats) or no injection (38 rats). The animals were killed when moribund. No pleural tumors were observed in the test or control group. As a comparison, the researchers examined the effect of Canadian chrysotile (90% of the fibers were <8 µm in length) in 39 rats and found that 25.6% of the rats developed mesothelioma.
Intratracheal
Groups of 24 male and 24 female Syrian golden hamsters were dosed weekly with intratracheal instillations of 0 or 3 mg talc in 0.2 mL saline for 18 weeks. 8 The chemical composition of talc was 61% to 63% silicon dioxide, 32% to 34% magnesium dioxide, and 0.85% to 1.06% other dusts; the particle size distribution was 93% <25 µm, 86% <16 µm, 54% <10 µm, 26% <5 µm, and 2% <1 µm. An untreated control group was also included. The animals were allowed to live until natural death or until killed when moribund. Animals given talc had a shorter lifespan (46 weeks) when compared to the saline controls (55 weeks). The talc-treated animals showed signs of minor respiratory disorders during treatment, and at necropsy, microscopic examination revealed pulmonary congestion and interstitial fibrosis, but no detectable dust deposits, granulomas, or mesothelial proliferations. There were 3 tumor-bearing animals; no tumors were in the respiratory tract, although 3 benign lung lesions (mucoepidermoid lesions) were reported. Two forestomach papillomas, 1 thyroid adenoma, and 1 adrenal adenoma were also found.
In a lifetime study, groups of 48 Syrian golden hamsters were dosed once weekly with intratracheal instillations of 3 mg talc. 9 The talc was defined as USP grade and contained 64% to 66% SiO2, 34% to 36% MgO2, and <1% other dusts. Dust-laden macrophages and an accumulation of interstitial cells were observed in the talc-treated animals. A proliferation of fibrillar material, primarily elastic fibers, and multinucleated giant cells with foreign material were observed in the alveolar and interstitial spaces, and occasional accumulation of proteinaceous exudate was seen in the alveoli. No increase in collagen fibers or granulomas was observed. The severity of premalignant lesions was evaluated in the tracheobronchial and alveolar zone of the animals. No dysplasia was observed with talc in either zone. Slight metaplasia and moderate epithelial destruction were observed in the tracheobronchial zone. Moderate hyperplasia was observed in the alveolar zone. The number of lesions induced by talc was not given.
Both intratracheal studies also examined the effects of administering 3 mg talc + 3 mg B[a]P in 0.2 mL saline to hamsters for the 18-week period 8 or for a lifetime. 9 Although the researchers reported that results of the study indicated that talc + B[a]P had a co-carcinogenic effect, the Expert Panel noted that appropriate controls were not used.
Intraperitoneal
Forty 6-week-old Swiss albino mice were given an ip injection of 20 mg of ultraviolet (UV)-sterilized commercial talc (composition not stated) in 1 mL saline, and the animals were observed until there were obvious signs of a tumor or spontaneous death. 127 Fifty-five control animals were injected with 1 mL physiological saline. Animals that died before 9 months elapsed were not included. Twenty-four treated mice were included in the results. Three (12.5%) developed mesothelioma, and no lymphomas were reported. Forty-six the control animals were included in the results; 3 mesotheliomas and 1 lymphoma developed (8.7% total tumors).
Forty Wistar rats were given weekly ip injections of 25 mg talc suspended in 2 mL saline weekly for 4 weeks, and the animals were allowed to live until natural death. 128 It is stated that the talc was composed of magnesium silicate but no other components are given, and the particle size was not known. Eighty control animals were injected with saline only. Few tumors developed in the test animals, and; the tumor rate was 2.5%. The time to first tumor was 587 days. No tumors were reported in the control animals.
Ovarian Cancer Risk
Particulate migration in the genital tract
Migration of particles through the female genital tract has been examined as a possible explanation of the presence of talc in the ovaries. However, at the “Talc: Consumer Uses and Health Perspectives” workshop, it was stated that “available histologic and physiologic studies provide no basis to conclude that talc can migrate to the ovaries from the perineal region.” 11 Because of the discussion on whether or not translocation is a viable theory in general, several studies on the transport of particulate matter other than talc are briefly summarized subsequently, and mixed results were found. Studies specifically relating to talc migration then follow.
Nonhuman
No translocation of bone black (ie, carbon black) from the vagina to the oviducts was found in monkeys. 129 Cynomolgus monkeys were restrained so that their pelvis was elevated, and 0.3 mL of a suspension of 4% bone black in 30% dextran was placed in the vaginal posterior fornix of 4 monkeys and 0.3 mL of a suspension of 4% bone black in physiological saline containing carboxymethyl cellulose (CMC) was placed in the vaginal posterior fornix of 1 monkey. Ten units of oxytocin were administered by im injection at the same time. The monkeys were released after 20 minutes. One hour after deposition of the bone black, 2 monkeys that received suspensions in dextran and the one that received the saline with CMC suspension were anesthetized and the reproductive tract of each animal was removed; the oviducts were flushed. The remaining 2 monkeys were processed in the same manner 72 hours after deposition. The test samples, the solutions without bone black (negative controls), and samples with a suspension of bone black (positive control) were filtered with Millipore membrane filters (0.45 µm). Particles resembling bone black were found on filters used for oviduct flushing solutions as well as the solution blanks; the numbers ranged from very few to occasional on all filters and no distinct differences in numbers or shape of these particles were apparent. The new filter blank that was examined immediately upon removal was the only sample on which no bone black particles were found. The researchers stated that these results suggest that there was no translocation of bone black from the vagina to the oviducts.
Twenty-six New Zealand white rabbits were used to examine whether starch particles migrate from the vagina to the peritoneal cavity. 130 Anesthetized rabbits were divided into an untreated control group, a group given 50 mg of a glove lubricant powder intravaginally, and a group given 50 mg of the lubricant powder and Chlamydia trachomatis (an inclusion former). Ovulation was then induced in all groups. After 1 to 4, 17, and 25 days, the rabbits were anesthetized and the peritoneal cavity was rinsed; the lavage fluids were analyzed for starch particles. Small numbers of starch particles were found on all slides. Retrograde migration was found after 3 days. The number of small particles between the treated and control groups was not statistically significantly different. Large starch particles were statistically more numerous in the 2 test groups compared to the controls.
Human
Sterile carbon particles were suspended in 30% dextran and 3 to 4 mL of the suspension was deposited into the posterior fornix of 3 women placed in the lithotomy position (ie, head tilted downward at a 15° angle from horizontal) that were undergoing abdominal surgery; 1 mL (10 U) of oxytocin given simultaneously via im injection. 131 During surgery, 20 to 34 minutes after deposition of the particles, the Fallopian tubes were sutured 1 cm lateral to the uterus, excised, and then flushed with saline. Carbon particles were found in the rinsate from 2 of the 3 patients. In a study using India ink, it was found that India ink (0.2 mL) that was injected into the uterine cavity 15 minutes to 24 hours prior to abdominal surgery was transferred to the Fallopian tubes in 27 of 50 women in the proliferative phase and in 23 of 35 women in the secretory phase of the menstrual cycle. 132 Injection of ink into the cervical canal often resulted in immediate back flow into the vagina; the ink reached the Fallopian tubes in 17 of 56 women. However, when the ink was placed into the vagina, the ink was transferred to the Fallopian tubes in only 1 of 18 women in the proliferative phase in 12 to 24 hours. The ink was found to pass from the vagina to the uterus in 2 of 37 women; one of these woman where the ink was transferred had a lacerated cervix. (In this study, some of the women had received an im injection of 2 units of oxytocin at the same time the ink was administered, but it did not appear to affect the results, and the women were placed in the Trendelenburg position after the abdomen had been opened.)
In a study using a radionuclide procedure, the migration of a particulate radioactive tracer from the vagina to the peritoneal cavity and ovaries was determined in 24 women scheduled to undergo gynecological surgery. 133 The day prior to surgery, the women were placed in a supine position, and less than 3 mL of 10 to 15 mCi [99mTc]-labeled human albumin microspheres with a size range of 30 to 50 µm were deposited in the posterior fornix. Each patient remained in a supine position for 24 hours. The radionuclide material remained in place for 21 women, and in 16 of these women, “sufficiently high radioactivity levels” were determined as evidence of migration to the uterus or the Fallopian tubes and ovaries. In 14 of the 21 patients, radioactivity was measured in adnexa separately from the uterus. Nine of the 14 patients had “marked” radioactivity in the tubes and ovaries; the 5 patients that did not had severe tubal occlusions. Another group of researchers stated that this finding may be misleading because only 1 radioactive label was used. 129
The migration of starch particles from powdered gloves was examined in groups of female patients who were undergoing abdominal surgery. 134 A group of 17 females was examined with powdered gloves 1 day prior to surgery and a group of 12 females was examined with powdered gloves 4 days prior to surgery. Corresponding control groups of 15 and 14 females, respectively, were examined with powder-free gloves. Peritoneal fluid was collected during surgery. The number of starch particles found in patients examined with powered gloves 1 day prior to surgery was statistically significantly increased for both small and large particles at all locations of the genital tract and for large particles in the peritoneal fluid. No particles were found in 2 patients examined with powdered gloves and a few particles were found in 3 patients examined with powder-free gloves 1 day prior to surgery. In patients examined with powdered gloves 4 days prior to surgery, there were statistically significantly more small and large starch particles in the cervix and uterus, but not in the Fallopian tubes or peritoneal fluid, compared to patients examined with powder-free gloves.
A catheter was used to apply 1 to 2 mL of 10 ± 2 MBq-TC-99m-labeled macroaggregates of human serum albumin, 5 to 20 Hm in size, into the posterior vaginal fornix of 1000 women with primary or secondary infertility in the follicular phase of the menstrual cycle; 15 women were examined during the early to midluteal phase. 135 The women were in a supine position, and hysterosalpingoscintigraphy (HSS) scans (a method to evaluate the transport function of uterus and Fallopian tubes) were obtained immediately and at various intervals for 4 hours after application. Labeled particles were detected in the uterus at the time of the first HSS scan of every woman examined, and women in both the follicular and luteal phases were examined. In women in the follicular phase, radioactivity entered the Fallopian tubes on both in 15% of the patients and on one side in 64% of the patients; significant radioactivity entered the pelvis of 6% of the patients. Radioactivity was not found to migrate to the Fallopian tubes of the remaining women that were in the follicular phase or in any of the women examined during the luteal phase.
Talc migration in the genital tract
Nonhuman
Particles of talc were identified in the ovaries of rats that received intrauterine instillations of talc. 136 In a pilot study, one group of 4 female exbreeder Sprague-Dawley rats received 1 intrauterine instillation of 100 mg/mL talc in 250 µL PBS; these rats were killed 5 days after dosing. A second group of 4 rats received intrauterine instillations of the talc suspension on days 0, 6, and 15; 2 animals were killed on day 20. (Spectral analysis reported a 3:1 ratio of silicon to magnesium; it is not stated whether the talc was nonfibrous). The remaining 2 animals were dosed again on days 22 and 30 and killed on day 49. The ovaries of each animal were analyzed by an ashing procedure.
Two groups of 12 female exbreeder Sprague-Dawley rats were then dosed intravaginally with 250 µL of the same talc suspension or PBS only, and 2 animals per group were killed 24 hours, 48 hours, or 4 days after dosing. Their ovaries were removed and analyzed as mentioned earlier. Particles of talc were found in the ovaries of the 2 rats of the talc group that were killed after 4 days but not in those killed at 24 or 48 hours or in the PBS-treated animals.
Radioactivity was not found in the ovaries of rabbits dosed intravaginally with talc. 64 Three female large white rabbits received a single intravaginal instillation of 0.5 mL of [ 3 H]talc administered as a suspension in aq glycerol jelly solution (10 mg/mL; 1 µCi/mL) and 3 were given 6 daily doses of the talc suspension. (Chemical characterization data were not provided). In the single-dose rabbits, urine was collected every 24 hours for 3 days; the animals were then killed, the urogenital tract was dissected out, and the total radioactivity was determined in the urine, ovaries, Fallopian/uterine tubes and cervix, the bladder, and the vagina. In the urogenital tract 72 hours after dosing, radioactivity (0.004% of the dose) was only detected at the site of administration. (The limit of detection was 0.25 µg). Total recovery was not quantitated.
In the multiple-dose group, the rabbits were killed 72 hours after the final dose; radioactivity was determined as described for the single-dose animals. In the urogenital tract at 72 hours after the final dose, 0.035% of the radioactivity was found at the site of administration and 0.006% was found associated with the cervix and Fallopian/uterine tubes. No radioactivity was found in the ovaries.
Talc was not found to translocate from the vaginas of female cynomolgus moneys to the ovaries. 129 A pilot study was first performed with 2 female cynomolgus monkeys. Talc samples were exposed to a calculated neutron fluency of 1.2 × 10 17 n/cm2, and 125 mg neutron-activated talc suspended in 0.3 mL deionized water containing 1% CMC was placed into the vaginal posterior fornix of each monkey. (Deposition was similar to that of bone black, described previously). Three days after talc deposition, the animals were anesthetized and peritoneal lavage was performed; when the peritoneal cavity was opened to collect the fluid, the lavage was repeated through the abdominal incision. Peritoneal lavage was also performed on a control animal. Radionuclide activity was determined with 46 Sc, 59 Fe, and 60 Co. There was no measurable translocation of activated talc from the site of deposition to the uterine cavity, oviducts, ovaries, or peritoneal cavity. (The vagina and the cervix were analyzed together). It appeared that detectable amounts of 60 Co were found in a portion of the oviducts of each test animal, but this was not supported by 46 Sc or 59 Fe data. Approximately 0.3 and 2.3 mg talc were found in the vaginas of the 2 test monkeys 3 days after instillation.
In the definitive study, 6 monkeys were dosed with a neutron-activated purified blend of cosmetic talc for 30 consecutive workdays. 137 The animals were restrained and dosed as defined in the pilot study; additionally, 10 units of oxytocin were administered by im injection once weekly. 46 Sc, 59 Fe, 60 Co, and 61 Cr were used as tracers. The peritoneal lavage was performed as above 2 days after the last talc deposition. Measurable quantities of talc were observed in the vagina + cervix sample, and the quantities ranged from 0.006 to 117 mg talc. (The researchers theorized that the wide variations were most likely due to different menstrual cycle phases). No measurable levels of talc (>∼0.5 µg) were present in the samples from the peritoneal lavage fluid, ovaries, oviducts, or body of the uterus.
Human
Talc particles were found in approximately 75% (10 of 13) of the ovarian tumors and 50% (12 of 21) of the cervical tumors during an extraction–replication technique used to examine tissue from patients with ovarian or cervical cancer. 138 (The technique involved replicating embedded tissue using a polyvinyl alcohol solution, tape stripping the replicated tissue, and using an AEI-6B electron microscope to examine the replicas.) The particles found in the ovarian tumors were located deep within the tumor tissue and were not universally dispersed; some of the particles were 1000 Å but most ranged from 1000 Å to 2 µm. The particles found in the cervical tumor tissue were generally larger than those in the ovarian tumors; some crystals were as large as 5 µm. Additionally, many particles of talc were found concentrated in the deeper layers of a primary carcinoma of the endometrium; however, talc was not found in a secondary tumor in the ovary of the same patient. Talc particles were also found in 5 of 12 normal ovarian tissue samples removed from patients with breast cancer. (Chemical characterization data were not provided for the talc that was found; the researchers noted that no asbestos fibers were found in any of the tissues studied.)
In 100 consecutive cases of women operated on for pelvic disease at Johns Hopkins Hospital, a total of 175 normal ovaries were removed and examined for particulate matter. 139 Seventy-two cases were classified as having laminated calcifications referred to as psammoma bodies. Six examples of crystalline foreign bodies were found and examined by scanning electron microscopy, and computer-assisted microscopic X-ray analysis was used to determine the elemental composition of the foreign bodies in 4 cases. The particles were composed primarily of magnesium and silicon; the researchers stated that in industrial North America, the most common compounds containing magnesium and silicate are talc and asbestos. Nine percent of the patients appeared to have magnesium silicate granulomas in their normal ovaries, and an additional 9% contained very similar histologic entities.
The ovaries of 24 women with benign ovarian neoplasms who were undergoing surgery at Columbia Presbyterian Medical Center between 1992 and 1993 were examined for the presence of talc using both light and electron microscopy. 140 Twelve women reported talc application directly to the perineum or underwear, and 12 women were age-matched controls. The mean number of lifetime exposures for women reporting talc use was 14 820, with a range of 4784 to 39 312 lifetime exposures. The ovaries of 2 stillborn fetuses were analyzed as negative controls; no talc was found in these ovaries. Sections of normal ovary from the 12 women who reported the talc use were analyzed. A linear relationship between ovarian talc particle burden and exposure was not found. Neither light nor electron microscopy values correlated with perineal talc usage. Electron microscopy counts were 0 for about half of the patients exposed to talc as well as half of the controls; talc was observed with light microscopy in all patients exposed to talc and 11 of 12 controls. There was a negative correlation between the values obtained by light microscopy and electron microscopy. The mean electron microscopic particle count was higher in those exposed to talc and the mean light microscopic particle count was higher in the women who did not report talc use. In 1 patient for which both ovaries were analyzed, both talc counts varied greatly between the right and left sides (0 vs 1 669 000 particles/g wet tissue wt by electron microscopy and 556 vs 6 particles by light microscopy, respectively). Asbestos was detected in the ovaries of 4 talc-exposed patients and 5 of the control patients.
The pelvic lymph nodes of a woman with stage III ovarian papillary serous carcinoma, with metastatic serous carcinoma in 2 of the 6 right external iliac and obturator nodes, were examined using polarized light microscopy and scanning electron microscopy and X-ray spectroscopy. 141 The patient applied talc daily for 30 years to the perineum and also applied it to underwear and sanitary napkins. She had 3 term deliveries, followed by a tubal ligation and she did not smoke, use oral contraceptives, or, with the exception of 6 months of progesterone therapy, use postmenopausal hormone therapy. Birefringence was seen using polarized light; 3 of the 4 nodes that did not contain metastases displayed polarizing material. Examination of lymph nodes by combined scanning electron microscopy and X-ray spectroscopy revealed plate-like particulates in the 5 to 10 µm range within the lymph nodes; the energy dispersive X-ray spectroscopy showed a magnesium and silicate signature that was compatible with talc. Nodes from 12 other patients were examined; this case was strongest for birefringence. (Electron microscopy or X-ray spectroscopy had not been performed).
Epidemiological studies
Numerous epidemiological studies have been performed examining the risk of ovarian cancer following talc exposure. 142 -174 These studies are summarized in Table 9. There is a large amount of information presented in these studies, and a variety of parameters were examined. Table 10 is a summary of the relative risk (RR) for ovarian cancer presented in case–control studies; this table only includes those studies that indicated “ever” use of talc in the perineal area, independent of the manner of use. 143 -146,148,149,152 -155,157,159,161 -163,165 -167,170,173,174
Epidemiological Studies Evaluating Talc Exposure and Ovarian and Endometrial Cancer Risk.a

Abbreviations: BMI, body mass index; CI, confidence interval; CLE, cumulative lifetime exposure; HOPE, Hormone and Ovarian Cancer Prediction; HRT, hormone replacement therapy; IRR, incidence rate ratios; NECC, New England Case Control; NSAID, nonsteroidal anti-inflammatory drug; OC, oral contraceptive; OR, odds ratio; PMH, postmenopausal hormone; pop, population; RR, relative risk.
aBolded text was used to highlight statistically significant increases. Italicized text was used to highlight statistically significant decreases.
Summary of Case–Control Studies Evaluating Ovarian Cancer Risk for “Ever” Use of Talc in the Perineal Area.
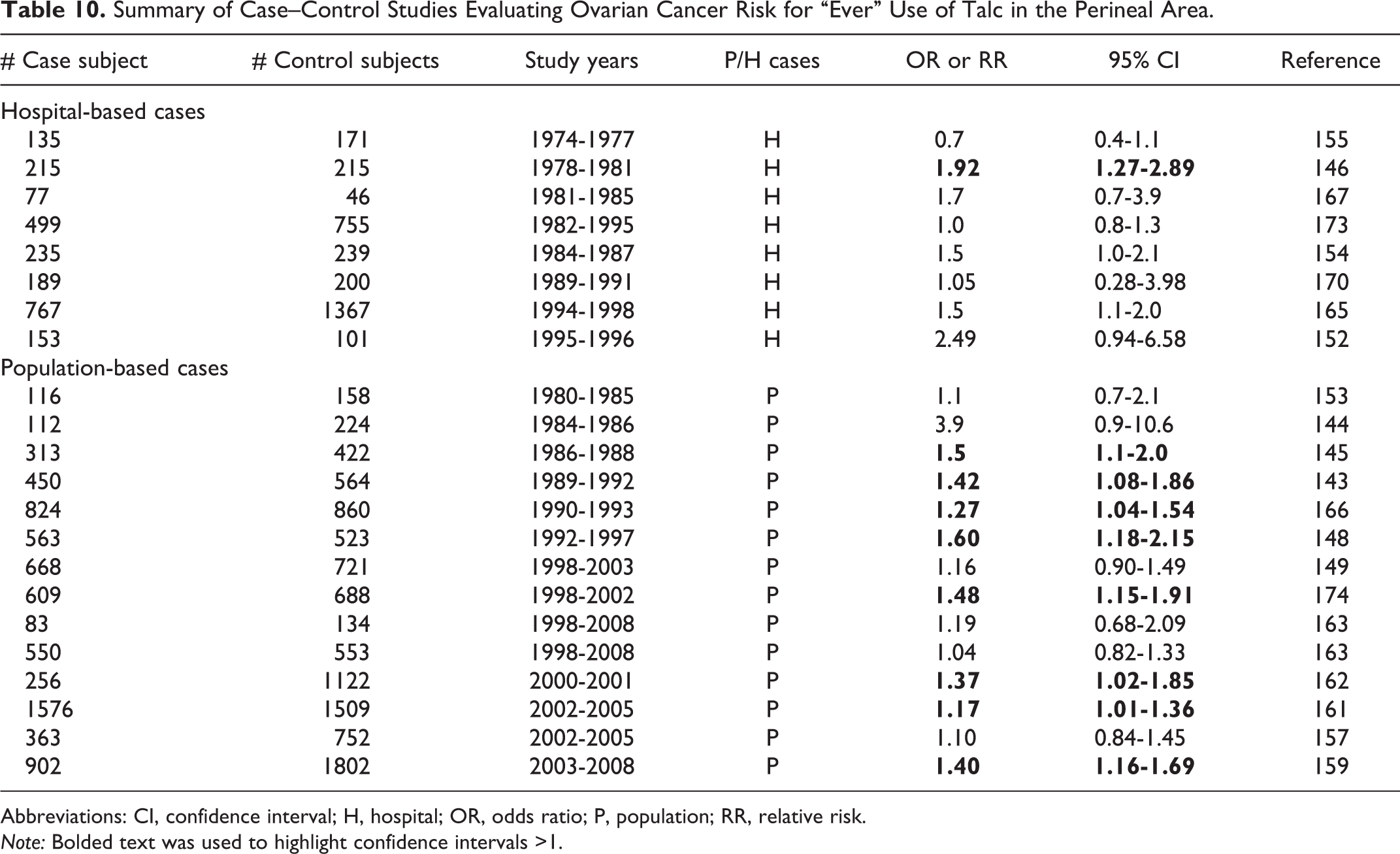
Abbreviations: CI, confidence interval; H, hospital; OR, odds ratio; P, population; RR, relative risk. Note: Bolded text was used to highlight confidence intervals >1.
Analysis of Ovarian Cancer Risk in the Epidemiological Studies
Concerns about cosmetic talc are based on reports suggesting that talc may migrate from the perineum to the ovaries and epidemiological studies suggesting a weak but fairly consistent association between perineal dusting and ovarian cancers. 12
The possibility that using cosmetic talc powder can cause ovarian cancer was suggested when talc particles were found in or on human ovarian tissues. 131 -133,138,175,176 The translocation of talc particles from the perineum to the ovaries would require that these particles pass from the perineum through the vagina and cervical canal, move across the uterus and against the ciliary motion of the Fallopian tubes, cross the peritoneal space between the fimbriae and ovaries, escape phagocytosis in the peritoneal space, and attach to the surface of the ovaries to accumulate in the ovaries. 177,178
However, there is evidence that talc particles found in ovaries are attributable to sample contamination, rather than to particle translocation. 12,179 This view is supported by studies finding talc in 100% of women with no known talc exposure, for example, as well as in 85% of women reporting frequent perineal talc applications. 140
Further, many translocation studies have been criticized for using particles with only a single radionuclide 133 because the radiolabel leaches from such particles, leading to the untenable assumption that the leached radioactive marker represents translocated particles. 12,62,137,179 -185 In a study conducted to help address this issue, 46 Sc, 60 Co, 59 Fe, and 51 Cr served as tracers in 125 mg neutron-activated talc deposited intravaginally 30 times over 45 days to ensure exposure through at least 1 menstrual cycle in cynomolgus monkeys. 12,137,179,182 The tracers were not detected in the uterus, Fallopian tubes, ovaries, or peritoneal lavage fluid 2 days after the 30th talc application.
The migration of many different types of materials from the vagina through the cervix has been demonstrated in patients in a supine or in the Trendelenburg position or with a lacerated or a dilated cervix. In addition, retrograde menstrual flow is a well-known phenomenon that could help explain the movement of particles to the ovaries in some cases. However, the findings of at least 1 study 132 has been interpreted as demonstrating the formidable barrier that the cervix presents to the translocation of particles from the vagina to the ovaries. 182,186
Many women may have been exposed to talc in infancy. 140 Infants are typically placed in a supine position and their legs separated during diapering, which could facilitate the passage of talc into the vagina. This may help explain the ubiquitous presence of talc in ovarian tissue. However, it has not been determined whether the hymen blocks exposure to the infant genital tract, or otherwise to what extent, if any, talc can enter the genital tract during diapering. 22
Several epidemiological studies suggested that medical procedures that would be expected to prevent the translocation of talc to the ovaries, such as tubal ligation or hysterectomy, reduce the RRs estimated for talc use. 148,154,172,187 However, in one of these studies, women who were exposed to talc for 1 to 9 years before tubal ligation or hysterectomy appeared to have an increased risk of ovarian cancer but not in women who had talc exposure for 10 or more years before their surgery. 172 Other studies found no difference in RR between women who had tubal ligation or hysterectomy and women who did not have these procedures. 143,173 One study reported inverse exposure-effect trends with duration of talc exposure after adjusting for tubal ligation. 165 Thus, the literature provides no clear, convincing support for the hypothesis that procedures that would preclude the passage of talc particles from the perineum to the ovaries reduce the risk of ovarian cancer.
The use of talc-dusted condoms or diaphragms, which would clearly result in exposure close to the cervical opening, was not associated with an increased RR estimate for ovarian cancer. 146,148,167 A meta-analysis found no association between talc-dusted diaphragm use and ovarian cancer risk. Overall, these studies do not support the hypothesis that talc can migrate from the perineum or the vagina to the ovaries.
Numerous case–control studies have reported small increases in RR estimates of all ovarian cancers combined in women using cosmetic talc products compared to women with minimal or no exposure, including population-based and hospital-based case–control studies (Tables 9 and 10; Figure 2). Other investigations found no statistically significant increase in risk estimates for ovarian cancer (all subtypes combined), including many case–control studies and 1 prospective cohort study. 151 Presumably the patients in all of these studies used products that contained cosmetic grade talc but information on fibrous content is generally lacking.
Odds ratio and confidence intervals in case–control studies evaluating ovarian cancer risk for “ever” use of talc in the perineal area. References143-146,148,149,152-155,157,159,161-163,165-167,170,173,174.
Some studies found statistically significant associations between talc use and invasive cancer 143,148,151 while another study reported an association only between talc use and tumors of low malignant potential. 154 Some studies found no statistically significant associations with all subtypes of ovarian cancer considered together but reported statistically significant associations only with specific subtypes of ovarian cancer or endometrioid tumors. 145,148,151,154
Among the epidemiological investigations reporting statistically significant associations, the RR estimates ranged between 1.0 and 2.0 and were barely statistically significant (Tables 9 and 10; Figure 2). For such low estimates, epidemiological methods generally cannot distinguish causality from even minor confounding risk factors or biases. 188 -191 Age, race, low parity, infertility, and a family history of ovarian, endometrial or breast cancer are among the most likely risk factors in the etiology of epithelial ovarian cancer. 12,192 Others have suggested that the effects of cancer treatment, smoking, and consuming coffee regularly could explain the small increases in the RR estimates reported for ovarian cancer in women using cosmetic talc products perineally. 172,193,194 Many physiological, sociological, and exposure factors have been linked to ovarian cancer, a number of them with a stronger association than the hygienic use of cosmetic talc, but causality has not been established for any of them. 182 The etiology of the majority of ovarian cancer cases is still unknown.
Prospective cohort studies do not suffer from recall bias because the exposures are recorded before the cancers were diagnosed. The single cohort study available found no statistically significantly association between perineal talc use and all ovarian cancer subtypes combined but did report such an association with invasive serous ovarian cancer (RR = 1.4; 95% CI: 1.02-1.91) 151 The odds ratios for serous ovarian cancer were also elevated in several case–control studies. 143,148,154,173 All of the odds ratio estimates reported in these studies were less than 1.7.
Talc exposure probably varies over time as women age and their reasons for deciding to use talc change. Talc use might be sporadic, seasonal, or change with circumstances (eg, sexual activity and parity). However, no studies have characterized either the feminine hygiene habits involving the use of cosmetic talc products in the general population or the latency of purported talc-induced ovarian cancer to enable resolving these issues. 190 Moreover, the epidemiological studies used questionnaires that did not focus specifically on the patients’ use of talc or talcum powders, as distinct from nontalc powders or sprays of known (eg, corn–starch based) or unknown compositions. 177 It is not clear that all of the patients understood the distinction between talc or talcum powders and talc-free powders when answering the questions. These factors contribute substantially to the uncertainties associated with the risk estimates of the prospective study as well as the case–control studies.
An early meta-analysis found a statistically significant adjusted pooled odds ratio of 1.27 (95% CI: 1.09-1.48) for ovarian cancer in women who ever used talc in the perineal or abdominal region compared to women who never used talc 195 However, the authors cautioned that this result does not provide the basis for inferring causality because many of the studies had substantial design limitations.
A more recent meta-analysis yielded a statistically significant overall summary RR of 1.33 (95% CI: 1.16-1.45). 194,196 However, sensitivity analyses revealed clear differences in outcome based on study design. Population-based case–control studies yielded a statistically significant increase in the risk of ovarian cancer for hygienic use of talc, but hospital-based case–control studies showed no statistically significant difference. There were no differences in the frequency of talc use in the respective control groups. The authors suggested that the difference in outcomes may be attributable to a bias, such as a “treatment effect” among the cases (ie, side effects from treatments for ovarian cancer may make talc use more likely in the patients than in the controls).
A still more recent meta-analysis reported a statistically significant overall summary RR of 1.35 (95% CI: 1.26-1.46). 194,197 However, a statistical test for data heterogeneity indicated substantial inconsistencies among the pooled studies and an invalid pooled summary RR estimate. Thus, the outcome provided no support for a causal association between perineal talc use and ovarian cancer. 188,198
Most of the epidemiological studies found no trend of increasing ovarian cancer risk with increasing exposure duration or frequency or cumulative exposure, despite a 5-fold difference between the lowest and the highest exposure groups (Table 10). 199 Several of these studies reported an apparent inverse trend. 142,143,148,151,196,200 In 1 study, suggestions of an exposure-effect relationship were obtained only after excluding exposures during pregnancy, during oral contraceptive use, and after sterilization. 148 Overall, however, the results of the epidemiological studies are not consistent with known mechanisms of carcinogenesis, which would be expected to yield positive exposure-effect trends. The inverse trends, in particular, are not compatible with a causal relationship between perineal talc exposure and ovarian cancer. 193,196
No plausible biological mechanism has been identified to explain how exposure to talc containing no asbestos or other asbestiform fibers could cause ovarian cancer. If perineal talc use can cause ovarian cancer in a dose-dependent manner, then there should be also be associations between such talc use and both cervical and uterine cancers, where talc exposure would be expected to be greater than ovarian exposure. No such associations have been reported.
Thirty or more years ago, cosmetic talc was thought to contain substantial amounts of asbestos fibers, 23,201 which would clearly represent a carcinogenic risk. However, FDA and IARC found that this contention could not be substantiated. 18,38,201 -204 Further, stringent quality criteria have been in place for cosmetic talc since 1976. 205 Meeting these criteria requires the elimination of detectable asbestos and other asbestiform fibers. Thus, the increased ovarian cancer risks associated with cosmetic talc use reported in some of the more recent epidemiological studies have generally not been attributed to contamination with asbestiform fibers.
However, the potential carcinogenicity of talc has been attributed by some authors to the chemical similarity of talc to asbestos. Both substances are magnesium silicates, but they share no other characteristics in common. 12,22,205 The aspect ratio of the fibrils is generally considered to be critical for the carcinogenicity of asbestos. In contrast, talc consists of 3-layer silica–brucite–silica sheets stacked together to form small platy packets with highly insoluble, hydrophobic surfaces. Cosmetic talc does not contain fibrils.
Alternatively, some researchers have suggested that talc in the ovaries could cause cancer, indirectly, through a talc-induced inflammatory response, analogous to the action of asbestos fibers in the lungs. 206 However, pelvic inflammatory diseases, such as endometriosis, peritonitis, and tubo-ovarian abscess formation, have not been found to be associated with increased risks of ovarian cancer. In addition, anti-inflammatory drug use did not reduce ovarian cancer risk estimates in several studies. 161,207
Most recently, 1 group proposed that elevated expression of anti-MUC1 antibodies induced by perineal talc in the peritoneal lymph nodes might explain the reported associations between talc exposure and ovarian cancer. 149 However, the application of talc powder to other parts of the body appears to induce anti-MUC1 antibody expression as well, and elevated anti-MUC1 antibody levels generally have not been associated with increased risks of ovarian cancer. Thus, this proposal remains highly speculative.
Talc is commonly used clinically as the active agent for pleurodesis. This procedure involves introducing a talc slurry directly into the pleural space to induce fibrogenesis. No increase in the incidences of lung or pleural cancers has been found in multiple clinical studies involving hundreds of patients followed for decades after pleurodesis. 193,208,209
The results of these clinical studies are consistent with epidemiological investigations reporting no statistically significant increase in mortality from lung cancer or mesothelioma in workers occupationally exposed to “pure” talc. 92,95,97 As stated by 1 author, “the likelihood that talc could selectively induce ovarian cancer and not lung cancer at exposure concentrations orders of magnitude lower than that experienced in occupational settings, argue against its toxicity.” 22 Others have noted the absence of reports suggesting that talc inhalation is associated with either lung cancers or mesothelioma in consumers 12 .
Accordingly, animal cancer bioassays using rodents exposed to high concentrations of talc in air indicate that talc is not a primary carcinogen. The NTP life-time inhalation carcinogenesis bioassay found no ovarian lesions in female mice or rats and no malignant respiratory-tract lesions in male rats or male or female mice. 10 Further, the lung cancers found in female rats can be plausibly attributed to chronic pulmonary particle overload, rather than to the possible carcinogenicity of talc. 124,210 The use of micronized talc in the NTP study probably contributed to the pulmonary overloading. This interpretation is supported by the results of an earlier lifetime inhalation study in hamsters. The animals were exposed to a talc baby powder aerosol at rates that exceeded those measured in infant-dusting simulations (mg h/m3) by 30- to 1700-fold. 12,82 The exposures had no effect on the type, incidence, or degree of histopathological findings in the lungs or other tissues examined, or on body weight, survival, or any other parameter evaluated, compared with the sham-exposed controls.
Further, the injection of talc into ovarian bursa of rats in 1 study (100 µL/ovary of 100 mg 0.4 to 14 µm platy talc crystals/mL buffered saline) induced no cancers. 68
In summary, critical issues that call into question the validity of the statistically significant associations reported in some of the epidemiological studies include: absence of persuasive evidence that talc can migrate from the perineum to the ovaries; lack of consistent statistically significant positive associations across studies; uniformly small RR estimates in studies reporting positive associations; failure to rule out plausible alternative explanations of the statistically significant results, including biases, confounding risk factors, and exposure misclassifications; absence of statistically significant associations between ovarian cancer and using talc-dusted diaphragms or condoms; overall lack of positive exposure-effect relationships; inverse trends for both duration of use and frequency of use in some studies; absence of a plausible biologic mechanism; and lack of credible, defensible evidence of carcinogenicity from the results of epidemiological studies of occupational exposures and animal bioassays.
Irritation and Sensitization
Sensitization
Nonhuman
Talc was not a sensitizer in female Hartley guinea pigs. 211 Female Hartley guinea pigs (number not stated) received an intradermal injection of 10 mg sterile talc in an emulsion of 0.5 mL sterile saline and 0.5 mL Freund complete adjuvant; 6 guinea pigs were dosed in the same manner with 10 mg starch glove powder. (Chemical characterization data were not provided; the talc was British Pharmacopeia grade). Eleven control animals were injected with the emulsion only. Skin tests were then performed at various intervals by challenging all animals with suspensions of starch glove powder in one ear and talc in the other. Slight cutaneous thickening was observed in all control animals 24 hours after challenge with both suspensions, and the responses were similar to both talc and the starch. The response to challenge with talc in the talc test group was similar to that seen in the controls. Animals in the starch group had a statistically significantly greater response to the starch challenge compared to the controls.
Summary
Talc is a sheet silicate that belongs to the silicate subclass phyllosilicates. In its purest form, it is a mineral that corresponds to the chemical formula of hydrous magnesium silicate; commercially, it contains varying amounts of other minerals naturally found in the ore. Only talc containing no detectable fibrous, asbestos minerals is used in cosmetics, and cosmetic talc must consist of a minimum of 90% hydrated magnesium silicate, with the remainder consisting of naturally associated minerals such as calcite, chlorite, dolomite, kaolin, and magnesite.
In 2013, FDA VCRP data indicated that talc was used in 3469 cosmetic formulations and, according to concentration of use data received in response to a Council survey, talc is used at up to 100% in cosmetic formulations. Talc is used in almost every category of cosmetic product and it is used in products that may be applied to baby skin, products that could be incidentally ingested, products used near the eye area or mucous membranes, and in products that are sprayed. The particle size of talc raw material varies widely by product type and by manufacturer, although typical cosmetic talcs are reported to have average particle sizes ranging between 4 and 15 µm when measured by sedimentation method.
Talc has many commercial uses and it has pharmaceutical use. It is used as a color additive in drugs and is exempt from certification. Sterile talc is approved as a sclerosing agent. Talc is not allowed for use on the surface of medical gloves. It is used in the production of foods, and it is approved as an indirect food additive as a color.
Syrian golden hamsters received a single 2-hour nose-only exposure to talc tested as a commercial baby powder (chemical characteristics unknown), with a median aerodynamic diameter of 6.4 to 6.9 µm. The biological half-life of the talc deposited in the lungs was 7 to 10 days. No translocation from the respiratory tract to other tissues was found in this study, and the clearance of talc from the lungs was complete within 4 months after exposure. Following oral administration of [ 3 H]talc to mice, rats, and guinea pigs, most of the radioactivity was excreted in the feces. Wistar rats were used to determine the systemic distribution of talc following intrapleural administration; the study suggested that talc is absorbed very rapidly through the pleura, reaching the systemic circulation with deposition in other organs within 24 hours of administration, and that the distribution is not dose related.
The acute oral LD50 of rats was 920 mg/kg bw in one study and >5000 mg/kg bw in another. In a study in which mice were placed in a box with circulated baby powder, the mice removed after 30 or 60 minutes recovered completely and the mice removed after 90 or 120 minutes died; the chemical composition, amount of powder, and size of the box were not specified. In rats dosed with a single bilateral injection of 100 mg/mL talc into the ovarian bursa and killed 1 to 18 months after dosing, one or both ovaries of rats dosed with talc were cystic in appearance at all time periods; the cystic structures were attributed to distention of the bursal sac. Foreign body granulomas, without surrounding inflammation, were seen in the cortical area of 5 of the injected ovaries, and talc was observed in the granulomas. In rats, a granulomatous reaction in which foreign-body giant cells containing refractile materials was observed without fibrosis at 1 and 3 months after a single ip injection of 50 mg/kg bw nonfibrous talc. In rats dosed with a single ip injection of 0.02, 0.1, or 0.5 g talc in 5 mL normal saline, clusters of foci of inflammatory cells were observed scattered on the surface of the peritoneum, and talc particles were seen in the center of each focus.
There were no remarkable results found in studies examining the cellular effect of talc, such as cytotoxicity assays, assays examining the effect of talc on cell viability, or studies on the induction of apoptosis (among others).
Dermal application of talc to shaved rabbit skin for 6 weeks resulted in dryness of the skin and skin erosion. Oral administration to rats for 5 days produced minimal toxicity. In inhalation studies, exposure of mice and rats for 4 weeks (25 µm particle size) resulted in macrophages in the alveolar space, with more found in the mice than in the rats. In rats exposed for 3, 6, or 12 months, minimal to slight fibrosis resulted. In hamsters, exposure to baby powder (95% talc; 4.9-6.0 µmol/L) did not result in clinical toxicity, and no trends were observed. Intrapleural administration of talc (25 µm) to rats did not result in mesotheliomas; granulomas at the injection site were common. Infections occurred, but no neoplastic or perineal changes, when talc was instilled intravaginally or perineally in rats. Upon iv injection of talc (<5 µm) once weekly for 3 weeks, talc was found in the lungs and in the liver throughout the study.
Talc is non- or slightly irritating to rabbit eyes. In a female patient who presented with a foreign body sensation and inflammation of the conjunctiva of both eyes, a diagnosis of foreign body granuloma secondary to talc was made. Application of talc to wounded skin can give rise to scab formation, possible infection, and foreign body granulomas in the dermis.
Talc has a TLV (respirable fraction) of 2 mg/m3 as a TWA. Human pulmonary effects of talc include diffuse interstitial fibrosis and progressive massive fibrosis (often called complicated pneumoconiosis). In occupational exposure studies, statistically significantly elevated SMRs for silicosis and silico-tuberculosis were observed in an early study of talc miners and millers in the Italian Piedmont region exposed to talc that contained no fibrous material except for tremolite micro-inclusions; SMRs were statistically significantly reduced for malignant neoplasms, including lung, bronchial, and tracheal cancers. A follow-up of this group found statistically significant increases in mortality, which were attributed primarily to nonmalignant respiratory diseases among the miners. A cohort study of talc miners and millers exposed to talc and magnesite containing trace amounts of quartz, tremolite, and anthophylite found no statistically significant SMRs for all causes, all cancers, or diseases of the circulatory system or respiratory tract. The results of several other epidemiological studies were likely confounded by the presence of up to 3% silica or 6% actinolite in the talc, exposures to high concentrations of silica with or without exposures to fibrous talc (tremolite), or concurrent exposures to radon daughters. A meta-analysis of studies of miners and millers who worked with nonasbestiform talc reported summary SMRs for lung cancer of 0.92 (95% CI: 0.67-1.25) for millers in 5 countries exposed to high levels of talc without exposure to other occupational carcinogens, and 1.2 (95% CI: 0.86-1.63) for miners in 3 countries exposed to high levels of talc as well as to silica or radon and radon daughters. Studies examining radiological, lung-functional, and clinical parameters in talc miners and millers and rubber workers found some statistically significant changes.
In exposure-during-cosmetic use studies, the researchers noted that there was a wide variation in talcing times and methods, often by the same volunteer during different applications. Reported talcing times ranged from 17 to 31 seconds. Endobronchitis and airway stricture were reported in one case in which a patient applied large amounts of talc powder to her face. In another case, a chronic pulmonary granulomatous reaction was reported in a patient who applied “nonpowdering talc” to her face for 20 years, followed by use of talcum powder 2 to 3 times a day for a 10-year period.
Talc administered orally as a suspension in corn oil was not a developmental toxicant in mice (16-1600 mg/kg bw on days 6-15 of gestation), rats (16-1600 mg/kg bw on days 6-15 of gestation), hamsters (12-1200 mg/kg bw on days 6-10 of gestation), or rabbits (9-900 mg/kg bw on days 6-18 of gestation). No dose–response or time-trend pattern was observed in rats that received a single oral dose or once daily dose for 5 days of 30 to 5000 mg/kg bw talc.
In vitro, talc was not genotoxic in an UDS assay (10, 20, or 50 µg/cm2) or an SCE assay (2, 5, 10, and 15 µg/cm2) in RPMCs. Talc was not genotoxic in a host-mediated assay in mice dosed by gavage with a single dose or once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc or cytogenetic assay in rats dosed by gavage once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc. Talc was also not genotoxic in a dominant lethal assay in which rats were dosed by gavage with a single dose or once daily for 5 days with 30, 300, 3000, or 5000 mg/kg bw talc.
In a lifetime inhalation study, a carcinogenic effect was not observed upon exposure of hamsters to a commercial baby powder containing 95% platy talc for 30 or 150 min/d, 5 days/wk. A bioassay using mice and rats was performed by the NTP to determine the carcinogenic potential of nonasbestiform, cosmetic-grade micro-talc following exposure by inhalation, and it was concluded there was no evidence of carcinogenic activity in male or female B6C3F1 mice, some evidence of carcinogenic activity in male F344/rats, and clear evidence of carcinogenic activity in female F344/N rats. The mice were exposed to 6 mg/m3 (MMAD 3.3 ± 1.9 µm) or 18 mg/m3 (MMAD 3.6 ± 2.0 µm) talc for 6 h/d, 5 days/wk, for 103 to 104 weeks. The rats were exposed to 6 mg/m3 (MMAD 2.7 ± 1.9 µm) or 18 mg/m3 (MMAD 3.2 ± 1.9 µm) talc for 6 h/d, 5 days/wk, for 113 weeks (males) or 122 weeks (females). Concerns have been raised about this study, including concerns that micronized talc having a significantly smaller particle size distribution than cosmetic talc was used, aerosol concentrations were not properly controlled, proper procedures for dose selection were not followed resulting in the MTD being exceeded at both concentrations tested, and particle overload in the lungs was most likely the cause of the adverse effects reported.
Talc did not induce pleural tumors in rats following intrapleural injection of 20 mg talc (mean size 2.6 ± 2.3 µm). Few tumors developed in rats given weekly ip injections of 25 mg talc suspended in 2 mL saline weekly for 4 weeks. In mice given an ip injection of 20 mg of UV-sterilized commercial talc in 1 mL saline, 12.5% of the animals developed mesothelioma. The researchers also examined the effects of administering 3 mg talc + 3 mg B[a]P in 0.2 mL saline to hamsters in both studies, concluding that talc + B[a]P had a cocarcinogenic effect; however the Panel noted that appropriate controls were not used.
Results of studies examining particulate migration in the genital tract have been mixed. In one study using monkeys, there was no translocation of bone black from the vagina to the oviducts. However in a human study, researchers concluded that there was evidence of migration of carbon particles to the uterus or the Fallopian tubes and ovaries, although other researchers stated that this finding is misleading because only 1 radioactive label was used. In a study in rabbits, the number of large starch particles in peritoneal cavity rinsate was greater in test groups that were exposed intravaginally to glove lubricant (ie, starch) than in controls. In humans, it appeared that starch particles migrated to the cervix and uterus.
In studies specific to talc migration, mixed results have also been reported. In rats, talc was found in the ovaries of rats dosed intrauterinally with talc; in rats exposed with a single intravaginal dose, talc was found in the ovaries 4 days after dosing, but not 24 or 48 hours after dosing. Talc was not found in the ovaries of rabbits given 6 daily intravaginal doses, and there was no translocation of talc from the vaginas of monkeys to the ovaries, oviducts, or the body of the uterus. In humans, talc particles were found in 10 of 13 ovarian tumors and 12 of 21 cervical tumors; the particles found in the ovarian tumors were generally smaller than those in the cervical tumors, that is, 1000 Å to 2 µm versus up to 5 µm, respectively. In women with benign ovarian neoplasms, half of whom applied talc to the perineum or underwear, there was no linear relationship between ovarian talc powder burden and exposure. Electron microscopy counts were 0 for about half of the patients exposed to talc as well as half of the controls; talc was observed with light microscopy in all patients exposed to talc and 11 of 12 controls.
Numerous epidemiological studies have been performed examining the risk of ovarian cancer following talc exposure. Among the epidemiological investigations reporting statistically significant associations, the RR estimates ranged between 1.0 and 2.0 and were barely statistically significant. Many physiological, sociological, and exposure factors have been linked to ovarian cancer, a number of them with a stronger association than that of hygienic use of cosmetic talc, but causality has not been established for any of them. Most of the epidemiological studies found no trend of increasing ovarian cancer risk with increasing exposure duration or frequency or cumulative exposure, despite a 5-fold difference between the lowest and the highest exposure groups. Several of these studies reported an apparent inverse trend. The results of several epidemiological studies suggested that medical procedures expected to prevent the translocation of talc to the ovaries, such as tubal ligation or hysterectomy, reduce the RR estimates associated with talc use. Other studies found no difference in RR between women who had tubal ligation or hysterectomy and women who did not have these procedures. One study reported inverse exposure-effect trends with duration of talc exposure after adjusting for tubal ligation. The use of talc-dusted condoms or diaphragms (including diaphragms known to have been stored in talc powder), which would clearly result in exposure close to the cervical opening, was not associated with an increased estimate of RR of ovarian cancer. Talc was not a sensitizer in female Hartley guinea pigs.
Discussion
The safety of talc has been the subject of much debate through the years, partly because the relationship between talc and asbestos is commonly misunderstood. Often in early studies, some of the analytical methods used to identify asbestos in talc were not performed and/or interpreted correctly, leading to incorrect conclusions that high levels of asbestos were present in talc. In 1976, the CTFA issued stringent purity standards for talc used in cosmetics, including specifications that talc must contain no detectable fibrous, asbestos mineral; generally accepted methods for the determination of asbestiform amphibole minerals in cosmetic talc were also identified by the CTFA. Therefore, the CIR Expert Panel evaluated the safety of only talc that does not contain detectable fibrous, asbestos minerals.
During its deliberations, the Panel discussed a 2012 FDA study, in which talc samples and talc-containing products were analyzed for the presence of asbestos. Of the 9 companies contacted, 4 supplied data to the FDA. No asbestos was detected in any of the talc samples or the talc-containing products. The Panel requested clarification of the analytical methods used to confirm lack of significant asbestiform amphibole content. In response to this request, the Panel was advised that talc is certified to be asbestos free, and their mines are monitored for asbestos concentrations. The Panel received documentation from industry of the analytical methods used to confirm the purity of talc, particularly with respect to contamination by asbestos, quartz, and other inorganics. The analysis protocol, which is standardized in CFTA Method J 4-1, employs X-ray diffraction and polarizing light microscopy to detect asbestos fibers at levels below 0.05%.
As evidenced in this safety assessment, numerous studies have been performed to investigate whether or not a causative relationship exists between the cosmetic use of talc in the perineal area and ovarian cancer. The Panel reviewed these studies thoroughly and determined that they do not support a causal link. The Panel stated that causation would depend on the migration of talc from the perineum to the ovaries. There is no conclusive explanation for the presence of talc in the ovaries reported in some studies. However, the Panel agreed that there is no known physiological mechanism by which talc can plausibly migrate from the perineum to the ovaries. Further, the Panel noted that if typical perineal applications of talc increased the risk of ovarian cancer, then it would be expected to increase the risks of uterine and, especially, cervical cancer as well; the absence of reports of associations between perineal talc use and either uterine or cervical cancer indicates that perineal talc application does not cause ovarian cancer. Additional support for this conclusion comes from, for example, studies demonstrating that the use of talc-dusted condoms or diaphragms, which would clearly result in exposure close to the cervical opening, was generally not associated with increased RR estimates for ovarian cancer.
Studies have also examined whether the inhalation of cosmetic-grade talc is associated with respiratory tract cancers. Although an inhalation study performed by the NTP using nonasbestiform, cosmetic-grade talc concluded that there was some evidence of carcinogenic activity in male rats and clear evidence in female rats, the Panel stated that these results were attributable to an artifactual effect caused by particle overload in the lungs of the rats. The talc that was used in this study, that is, micronized talc at high, saturating concentrations, had particle size distributions much smaller than those of cosmetic-grade talc. The Panel concluded that the use of talc at concentrations up to 35% in spray products, as reported for aerosol makeup bases, or even at 100% in powders, as reported for face powders, would not overwhelm pulmonary clearance mechanisms and would, therefore, not cause pulmonary overload or adverse respiratory effects attributable to cosmetic talc use.
One group of researchers looked at the effect of intratracheal administration of talc plus B[a]P in hamsters, concluding that talc may be co-carcinogenic when administered with B[a]P. The Expert Panel noted the potential for co-carcinogenicity but determined that the results of these studies were not attributable to a specific effect of talc, appropriate controls were not used, including control animals exposed to B[a]P alone and, thus, the results were not relevant for assessing the safety of the cosmetic use of talc.
Finally, the Panel warned that talc should not to be used on skin where the epidermal barrier is removed or on skin that has greater than first degree burns. Case reports were available in which granulomas formed if talc was applied to skin when the epidermal barrier was absent.
Conclusion
The CIR Expert Panel concluded that talc is safe in the present practices of use and concentration described in this safety assessment.
Footnotes
Author Contribution
M. Fiume contributed to conception and design; acquisition, analysis, and interpretation; and drafted the article. I. Boyer contributed to conception and design; acquisition, analysis, and interpretation; drafted the article, and critically revised the article. L. Gill, W. Bergfeld, D. Belsito, C. Klaassen, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, analysis and interpretation, and critically revised the article. R. Hill and D. Liebler contributed to conception and design, analysis and interpretation, and critically revised the article. Former CIR Director F. Alan Anderson contributed to conception and design, analysis, and interpretation and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
