Abstract
The subchronic toxicity of sodium tungstate dihydrate aqueous solution in male and female Sprague-Dawley rats was evaluated by daily oral gavage of 0, 10, 75, 125, or 200 mg/kg/d for 90 days. Measured parameters included food consumption, body weight measurements, hematology, clinical chemistry, and histopathological changes. There was a significant decrease in food consumption and body weight gain in males at 200 mg/kg/d from days 77 to 90; however, there was no effect in food consumption and body weights in females. There were no changes in the hematological and clinical parameters studied. Histopathological changes were seen in kidney of male and female and epididymis of male rats. Histopathological changes were observed in the kidneys of male and female rats dosed at 125 or 200 mg/k/d consisting of mild to severe cortical tubule basophilia in 2 high-dose groups. Histological changes in epididymides included intraluminal hypospermia with cell debris in the 200 mg/kg/d dosed male rats. Histopathological changes were observed in the glandular stomach including inflammation and metaplasia in the high-dose groups (125 or 200 mg/kg/d) of both sexes of rats. Based on histopathology effects seen in the kidneys, the lowest observable adverse effect level was 125 mg/kg/d and the no observable adverse effect level was 75 mg/kg/d in both sexes of rats for oral subchronic toxicity.
Keywords
Introduction
Tungsten is a naturally occurring element that can be used as a pure metal or in alloys and is being used in many industrial applications. Heavy alloys are composites of tungsten with other metals acting as binders and were used as armor penetrators during World War II. The US Army recently proposed the use of tungsten metal as a replacement for lead in small caliber ammunition. Tungsten–nylon composites were manufactured into 5.56-mm small arms ammunition (M855) and used for training on small arms training ranges. Approximately 100 installations Army-wide have used 1 or more of these tungsten–nylon rounds since 1999. Production ceased in 2003 when problems were reported from several installations concerning the ballistic properties of the bullets. Since that time, due to increases in small caliber requirements to support mobilization training and the Global War on Terrorism, the Army determined that the transition to lead-free rounds was cost prohibitive. 1 –3 In other military applications, heavy metal tungsten alloy-based materials have been recently introduced as replacement for depleted uranium in armor-penetrating munitions. 3 –5 It has been reported that tungsten was detected in ground water and biological sample. 6 Tungsten was also suggested as a possible casual factor in a cluster of cases with childhood leukemia near in Fallon, Nevada, but a detailed investigations of this issue found no direct link between tungsten and incidence of leukemia. 6 –9
There is considerable information on the toxicity, environmental fate and health effects of tungsten metal and soluble tungsten salts. 3,10 –12 The acute toxicity of tungsten and selected compounds has been studied in various laboratory animals. 13 The acute oral median lethal dose (LD50) value of sodium tungstate was 1928.4 mg/kg and the intravenous LD50 was 61.9 mg/kg in rats, whereas in mice, the acute oral LD50 of sodium tungstate was 1904.1 mg/kg and the intravenous LD50 was 107.1 mg/kg. 14 The acute oral toxicity of tungsten metal powder, 15 and ammonium paratungstate, 16 evaluated in rats (both sexes) showed LD50 values were greater than 2000 mg/kg with the exception of sodium tungstate dehydrate which was 1453 mg/kg in combined sexes. 17
There are several studies describing the subchronic and chronic effects of sodium tungstate exposures from drinking water. Cohen et al 18 reported that there were no effects on body weight or liver weight of DC male rats exposed to sodium tungstate (100 ppm) in drinking water for 3 weeks. Similarly, there were no noted effects in body weight or histopathology of Sprague-Dawley (SD) rats exposed to sodium tungstate in drinking water (200 ppm) for 20 weeks. 19 Rats exposed to sodium tungstate in drinking water at 5 ppm for a life time showed slight enhancement of growth and slight shortening of survival. 20 Sodium tungstate at 50 mg/kg/d in water was administered orally to Wistar male rats for 60 days to assess reproductive effects; there were no severe histological effects in testis and epididymis and no significant changes in plasma testosterone or other biochemical parameters studied. There were no adverse effects on body weight, and other vital organs remained unchanged. 21 Rats dosed orally with sodium tungstate at 0.005 to 5 mg/kg/d for 7 months showed slight destruction of terminal villi in the small intestine at 0.5 mg/kg/d. Similar histological effects were also reported in rabbits at 5 mg/kg/d after 8 months of oral dosing with sodium tungstate. 22
The study reported here was intended to better understand and document the oral toxicity of tungstate. We studied the toxicity of sodium tungstate in rats by daily oral gavage with various dose levels of aqueous solutions of sodium tungstate for 90 days to determine target organ toxicity and to identify a no-observable-adverse-effect-level (NOAEL). Based on the NOAEL, the Bench Mark Dose Lower confidence limit (BMDL) values can be derived. This information can be used to identify effect levels, define target organs, support regulatory actions, and provide risk assessment information.
Materials and Methods
Test Substance
Sodium tungstate dihydrate (Tungstic acid sodium salt dihydrate, Na2WO42H2O, CAS # 10213-10-2, Batch # 12330JO) 99% pure was purchased from Sigma-Aldrich, Inc, (St Louis, Missouri). The purity of the test compound and dosing solutions were tested at our Aberdeen Test Center and found to be consistent for the purity and stability during testing. (Figure 1)
Sodium tungstate.
Animals
Male and female 5-week-old SD rats were purchased from Charles River laboratories, Raleigh, North Carolina, and they were held for 1 week in quarantine prior to initiation of treatments. At the start of testing, rats weighed between 199 and 230 g. Test animals were identified by individual cage cards and microchip implants and were individually housed in polycarbonate cages. Bedding (certified Sani-Chip; Harlan Sani-Chip, P. J. Murphy Forest Products Corporation, Montville, New Jersey) was placed in the bottom of each cage and replaced twice weekly. Drinking quality water and a certified laboratory diet (Harlan Teklad, 8728C Certified Rodent Diet; Teklad Certified Rat Diet, Teklad, Madison, Wisconsin) were available ad libitum. Animal rooms were maintained at 64°F to 79°F, with relative humidity of 30% to 70% and a 12-hour light–dark cycle.
Experimental Methods
These studies were conducted according to the Guide for the Care and Use of Laboratory Animals. 23 The studies reported herein were performed in animal facilities fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. A 90-day oral toxicity study was conducted in rats according to the procedure described in the Environmental Protection Agency (EPA) Health Effects Testing Guidelines (40 CFR, Part 798.2650) in compliance with Good Laboratory Practice. 24
Following 1-week quarantine/acclimatization period, 50 male and 50 female SD rats were randomly distributed using the LABCAT Randomization Program (Innovative Programming Associates, Princeton, New Jersey) into 4 treatment groups and 1 control group (10 animals of each sex per group). Dosage levels were set at 0 (control), 10, 75, 125, and 200 mg/kg/d. Control animals were dosed with deionized (DI) water (1 mL/kg). The dose levels were selected on the basis of our previous studies where the highest dose used was 200 mg/kg/d in a subchronic toxicity study. 25 Sodium tungstate dihydrate was solubilized with DI water to produce 4 dosing solutions of 200, 125, 75, and 10 mg Na2WO4/mL. Tungstate concentrations of the dosing solutions were verified by the Aberdeen Test Center and found to be consistent for purity and stability during the study period.
Four rats of each sex were housed in the same animal room as study animals and used for health monitoring purposes. Two rats of each sex were shipped to Charles River Laboratories for health monitoring after the midpoint of the study and at the conclusion of the 90-day study to assess the general health of the purchased animals. Serology, bacteriology, and parasitology testing were performed by Bio Reliance Invitrogen Bioservices, Rockville, Maryland, and the sentinel rats were found to be free of viral antibodies, pathogenic bacteria, and parasites.
Test chemical solutions were administered daily (7 days per week) for 90 days. A 16 GA × 2-in stainless steel gavage needle (Popper and Sons, New Hyde Park, New York) was used to facilitate oral dosing. After dosing the rats, they were returned to their individual cages. Food and water were provided ad libitum.
A clinical examination was made for each animal prior to initiation of treatment and once weekly during treatment. Observations included but were not limited to changes in skin and fur, eyes, mucous membranes, occurrence of secretions and excretions, and autonomic activity (eg, lacrimation, piloerection, pupil size, and unusual respiratory pattern). Changes in gait, posture, and response to handling as well as the presence of clonic or tonic movements, stereotypes (eg, excessive grooming, repetitive circling), or bizarre behavior (eg, self-mutilation, walking backward) were recorded according to testing guidelines. 24
Body and feeder weights were recorded on days −3, −1, 0 (first day of dosing), 7, and weekly thereafter. Doses were adjusted weekly to reflect the change in individual body weights. Animals were observed daily for any toxic signs. All data were recorded using the LABCAT In-Life Program. Water consumption was not monitored during this study.
Ophthalmic examinations were performed on all control and treated animals prior to the scheduled start of the study and within a week of the scheduled 90-day necropsies. Urinalysis was also performed on 8 of 10 animals from all dose groups (including negative control) within 2 weeks of the final (90-day) necropsies.
Necropsy
Following the 90-day study period, the rats were anesthetized with a 1.5-mL dose of a mixture of ketamine (100 mg/mL), xylazine (20 mg/mL), and acetylpromazine (10 mg/mL) by intraperitoneal injection. Blood was collected by cardiac puncture for hematology and clinical analysis, and the rats were euthanized using carbon dioxide. Each rat was then submitted for complete necropsy. The brain, heart, liver, kidneys, spleen, adrenals, thymus, epididymis/uterus, and testes/ovaries were removed and weighed for absolute organ weights. The tissues harvested for histopathological evaluation included the brain, pituitary, thyroid with parathyroid gland, thymus, lungs, trachea, heart, bone marrow, salivary gland, liver, spleen, kidney, adrenal gland, pancreas, testis, ovaries, uterus, aorta, esophagus, stomach, duodenum, jejunum, ileum, caecum, colon, urinary bladder, salivary lymph node peripheral nerve (siatic), thigh musculature (vastus lateralis), eye, spinal cord (3 levels), and exorbital lachrymal gland. Histopathological evaluations were performed for this 90-day study (Dr. George A. Parker, DVM, Hillsborough, North Carolina) and the Quality Systems Office of the US Army audited critical phases of these studies. 26,27
Hematology and Clinical Chemistry
Hematology parameters were analyzed using Abbott Cell-Dyn 3700 Hematology Analyzer. The parameters include white blood cell count, neutrophils, lymphocytes, monocytes, eosinophils (EOSs), basophils, red blood cell count, hemoglobin, hematocrit, mean cell volume, mean cell hemoglobin, mean cell hemoglobin concentration, red blood cell distribution width, platelets, and mean platelet volume.
Clinical chemistry parameters were measured using a VetTest 8008 Chemistry Analyzer and VetLyte sodium (Na), potassium (K), chloride (Cl) Analyzer (IDEXX Laboratories, Glen Burnie, MD). The clinical chemistry analytes included alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, blood urea nitrogen, calcium, cholesterol, creatinine kinase, creatinine, glucose (GLU; nonfasting), lactate dehydrogenase, total bilirubin, total protein, triglycerides, Na, K, and Cl.
Urinalysis was conducted by measuring volume, color, appearance, pH, specific gravity, GLU, bilirubin, urobilinogen, ketone, blood, protein, nitrite, and leukocytes (Siemens Healthcare Diagnostics Multistix Reagent Strips, Tarrytown, New York).
Data and Statistical analysis
Food consumption, body weights, and absolute organ weights were compared among dosage groups and controls using a 1-way analysis of variance (ANOVA). When significance was observed, the data were further analyzed using a Dunnett test to compare the doses to the control group. Statistical significance was defined at the P ≤ .05 level and analysis was performed using Sigma-Stat (Sigma-Stat, Jandel Scientific, Corte Madera, California). Clinical chemistry, hematology, and urinalysis data were analyzed with Sigma-Stat using a 1-way ANOVA and Bonferroni post hoc test to compare dosage groups to the control group. Where a normality test failed after log transforming the data, an ANOVA on ranks was performed.
Results
General
There were no unscheduled deaths (a single male rat in 200 mg/kg/d dose group was moribund and was euthanized on day 79; a few tissues from this rat were submitted for histopathological examination and death was determined to be not compound related), no evidence of overt toxicity and no treatment-related clinical signs were seen in any dose levels. Ophthalmic examinations prior to study initiation and within a week of the scheduled necropsies revealed no abnormalities.
Food Consumption and Body Weight
The food consumption data from male and female rats are presented in Tables 1 and 2. The results show that there were significant decreases in food consumption in male rats at 200 mg/kg/d from weeks of 4 to 13, while there was no change in food consumption in female rats during the 90-day study.
Feed Consumption (Grams) of Male Rats Dosed Orally With Sodium Tungstate for 90 Days.a
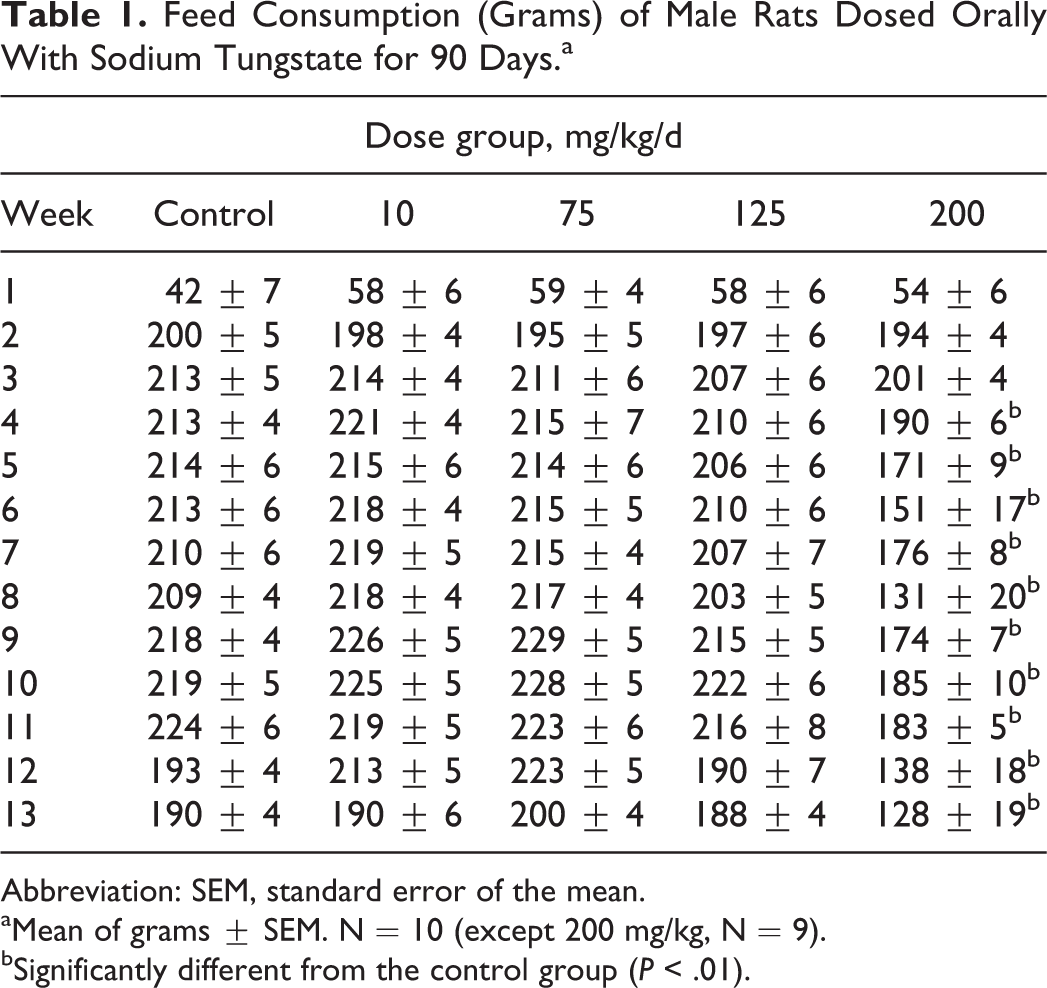
Abbreviation: SEM, standard error of the mean.
aMean of grams ± SEM. N = 10 (except 200 mg/kg, N = 9).
bSignificantly different from the control group (P < .01).
Feed Consumption (g) of Female Rats Dosed Orally With Sodium Tungstate for 90 Days.a
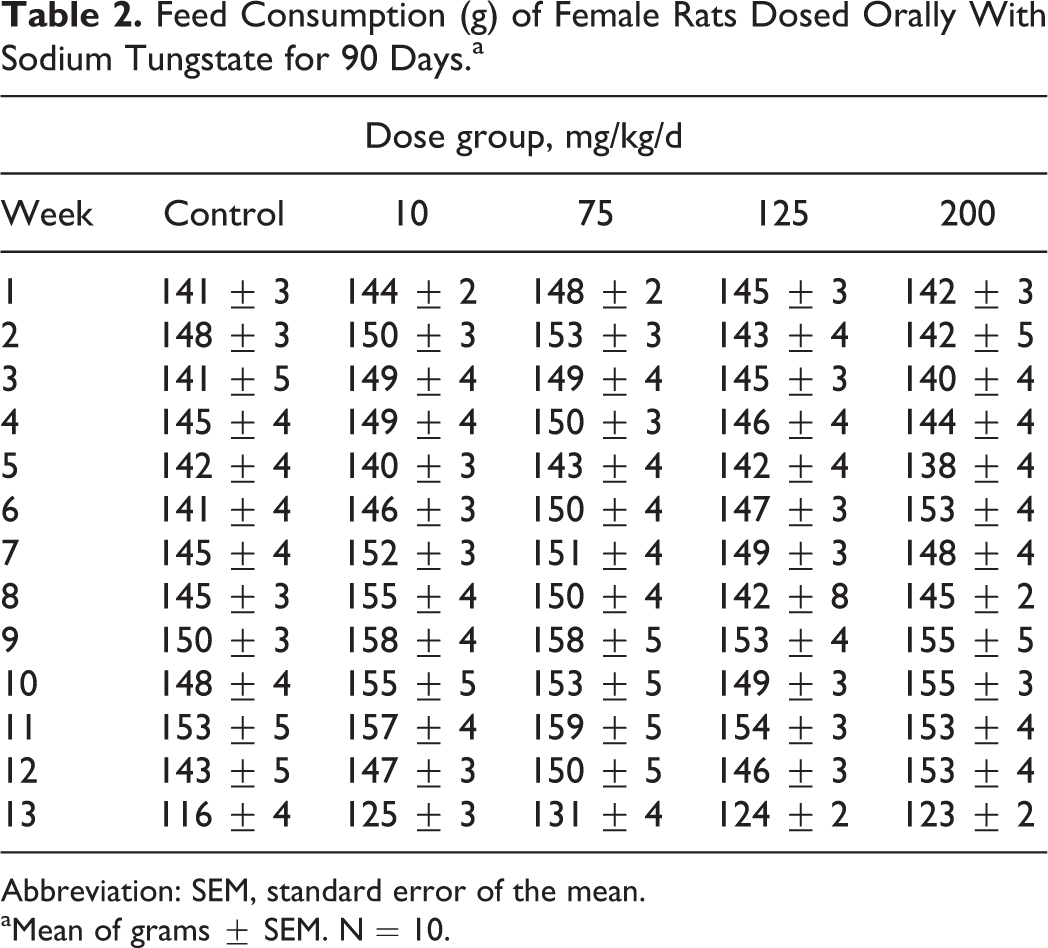
Abbreviation: SEM, standard error of the mean.
aMean of grams ± SEM. N = 10.
Male and female rat body weight measurements obtained during the subchronic study are presented in Figures 2 and 3. The data show that there was a significantly lower body weight gain in male rats during the final weeks (70-90 days) in the 200 mg/kg/d group; however, there were no body weight changes in female rats in any dose groups throughout study period when compared to control rats.
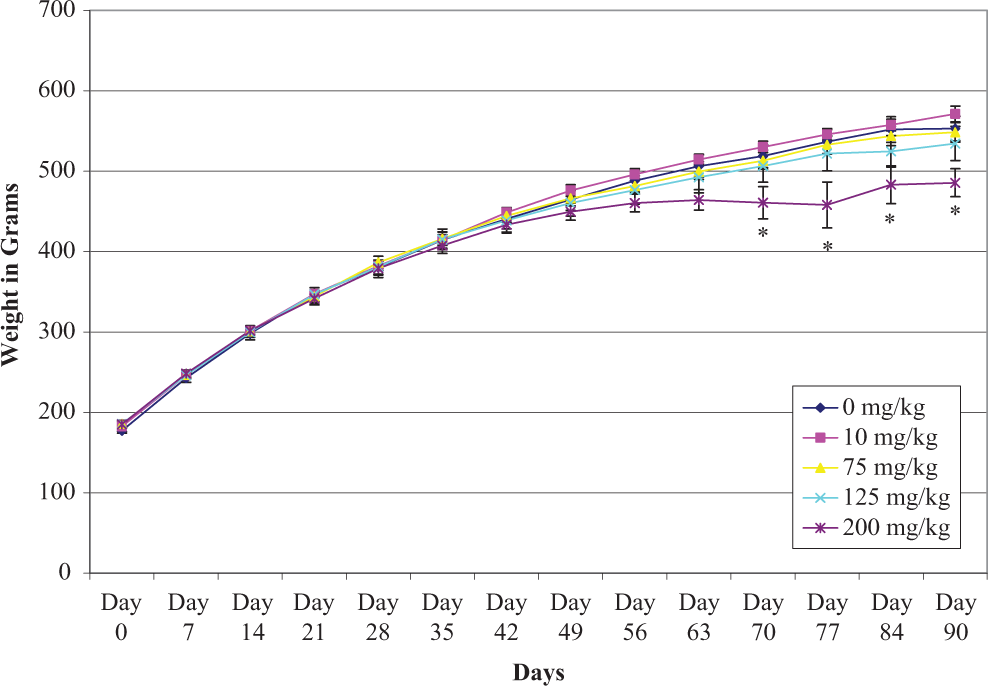
Mean ± standard error of the mean (SEM) individual body weights of male rats given sodium tungstate by gavage daily for 90 days.
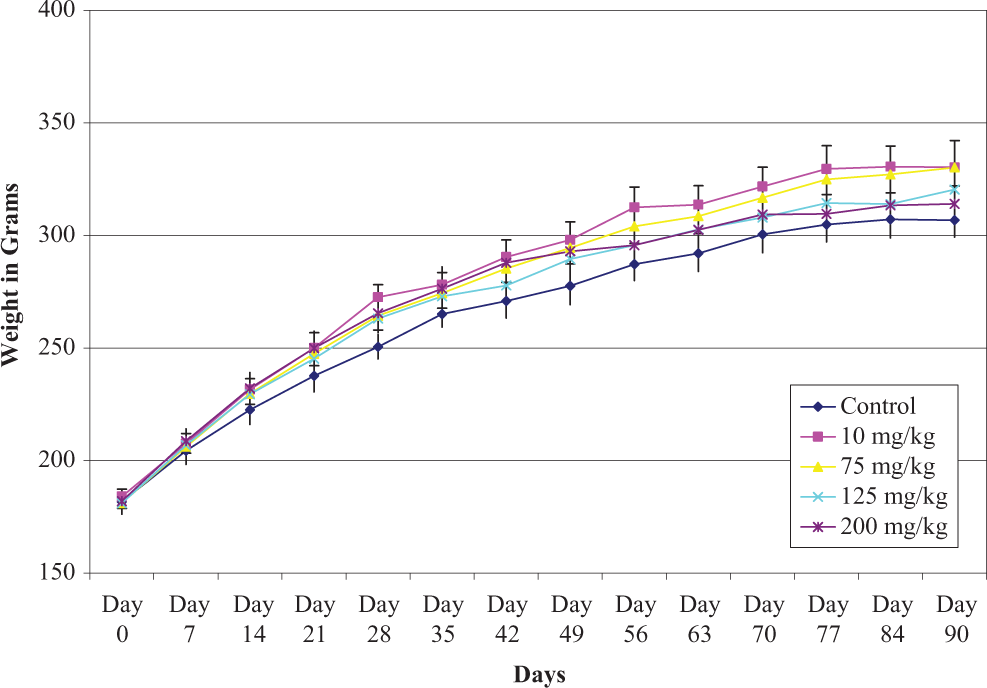
Mean ± standard error of the mean (SEM) individual body weights of female rats given sodium tungstate by gavage daily for 90 days.
Hematology and Clinical chemistry
Male and female rats showed no significant differences in any hematological parameters at any dose levels of sodium tungstate (supplementary Tables 1 and 2). The results of clinical chemistry parameters studied in rats showed no significant changes in any dose levels of sodium tungstate in rats (supplementary Tables 3 and 4). The parameters studied showed some changes in levels that were not dose related and insignificant and considered within normal range limits when compared to controls. All other parameters were found to be similar to control rats.
Urinalysis
Examination of urine samples taken approximately 1 week prior to necropsy revealed no significant changes in volume, specific gravity, or pH. No distinct dose-related trends were observed in GLU, bilirubin, ketone, blood, protein, urobilinogen, nitrite, or leukocytes (data not presented). 28
Body Weights and Organ Weights
The body weights, absolute heart, liver, and thymus weights were significantly lower in male rats dosed at 200 mg/kg/d compared to control rat, but there were no effects on body weights and organ weights of female rats (Tables 3 and 4).
Absolute Body Weights and Organ Weights (g) of Male Rats Dosed Orally with Sodium Tungstate for 90 Days.a
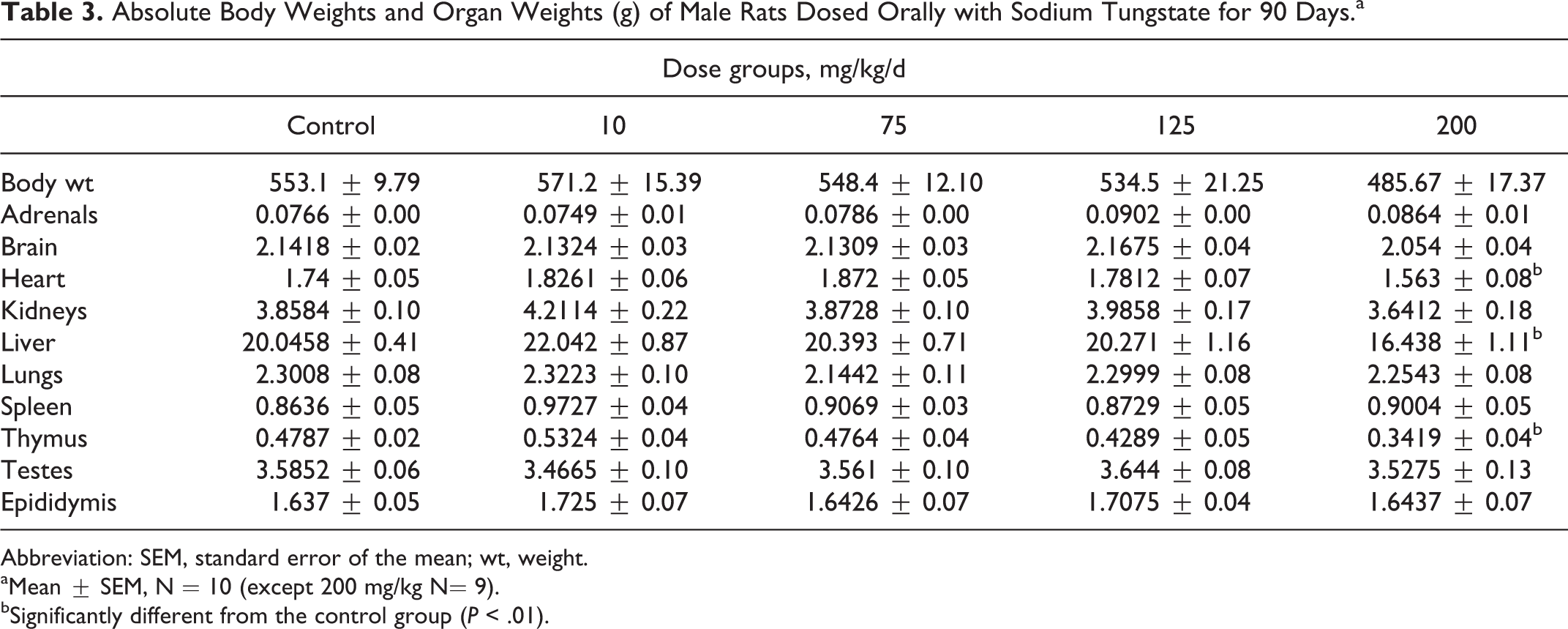
Abbreviation: SEM, standard error of the mean; wt, weight.
aMean ± SEM, N = 10 (except 200 mg/kg N= 9).
bSignificantly different from the control group (P < .01).
Absolute Body Weights and Organ Weights (g) of Female Rats Dosed Orally with Sodium Tungstate for 90 Days.a
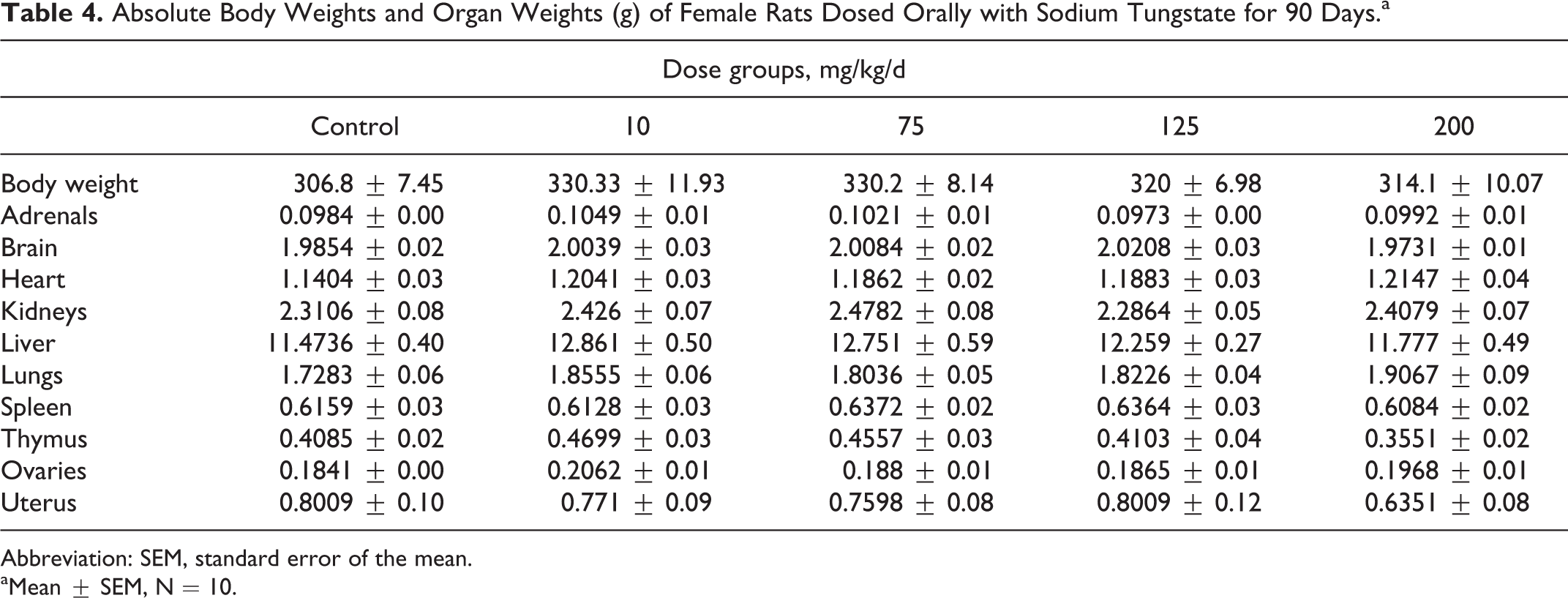
Abbreviation: SEM, standard error of the mean.
aMean ± SEM, N = 10.
Histopathology
Histopathology examination revealed effects on the urogenital system of the sodium tungstate-treated rats. Changes included mild to severe basophilia of renal cortical tubules in 1 of 9 and 10 of 10 males and 1 of 10 and 8 of 10 females in 2 high-dose groups (125 and 200 mg/kg/d), respectively. The lesion was commonly disseminated throughout the entire cortex bilaterally but was occasionally distributed multifocal. The affected tubules exhibited abundant pale basophilic cytoplasm and closely packed typically basally located nuclei with moderate anisokaryosis and scattered karyomegaly. In contrast, basophilic tubular profiles bearing thickened basement membranes are generally a spontaneous development in rats interpreted as early sign of chronic progressive nephropathy (CPN), which begins in susceptible strains as early as 2 to 4 months of age, at that time is generally minimal and scattered about cortex. Males are more commonly affected than females. However, the renal tubular basophilia in this study was graded mild to severe in the rats and was not associated with the thickened basement membranes. It is far more extensive than seen in background lesion, therefore the lesion was not considered to be early CNP. Basophilic tubules were occasionally lined with low numbers of cytomegalic cells (Figures 4 and 5). The figures illustrate the extent of the lesion in high-dose rats compared to an unaffected vehicle control.
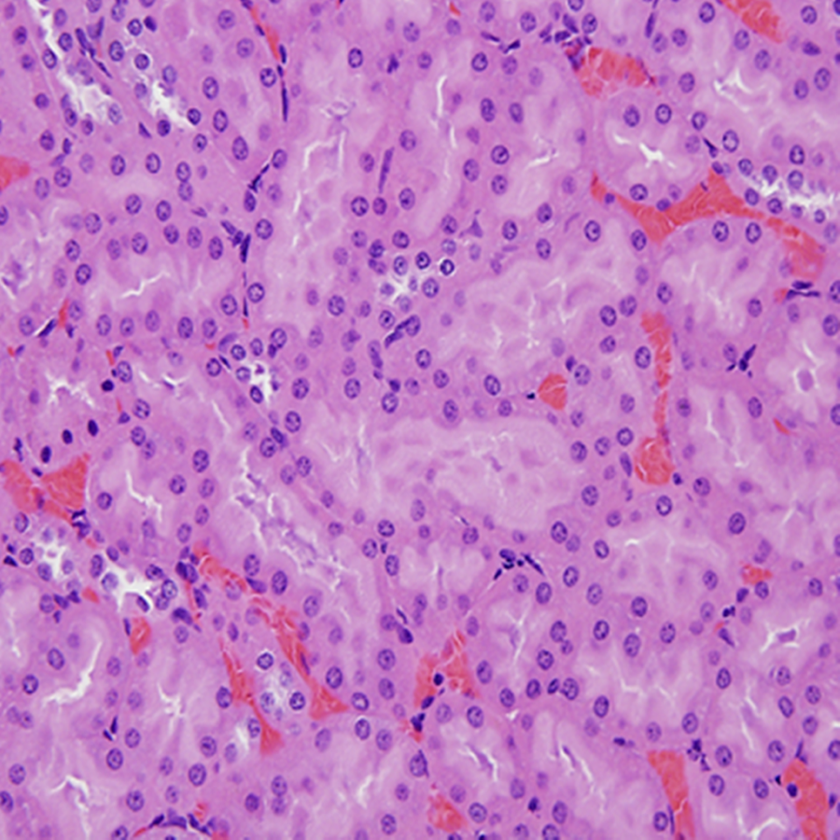
Photomicrograph of kidney section (×400) from a control male rats showing normal tubular development.
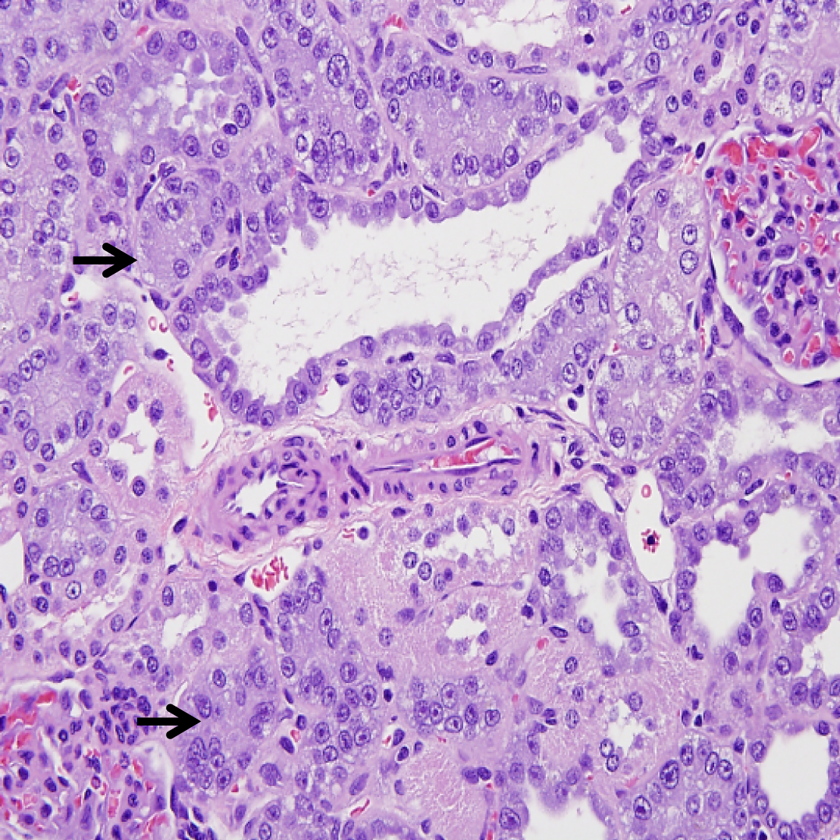
Photomicrograph of kidney section (×400) from male rats exposed to 200 mg/kg/d demonstrating (arrows) renal tubular damage.
Histopathological analysis of epididymides of rats dosed with sodium tungstate showed considerable effects in the high-dose group (200 mg/kg/d; Figures 6 and 7). Intraluminal cellular debris with and without hypospermia was noted in the epididymides of 3 of 10 males in the 200 mg/kg/d dose group. The lesion was not observed in the 10, 75, and 125 mg/kg/d dose groups.
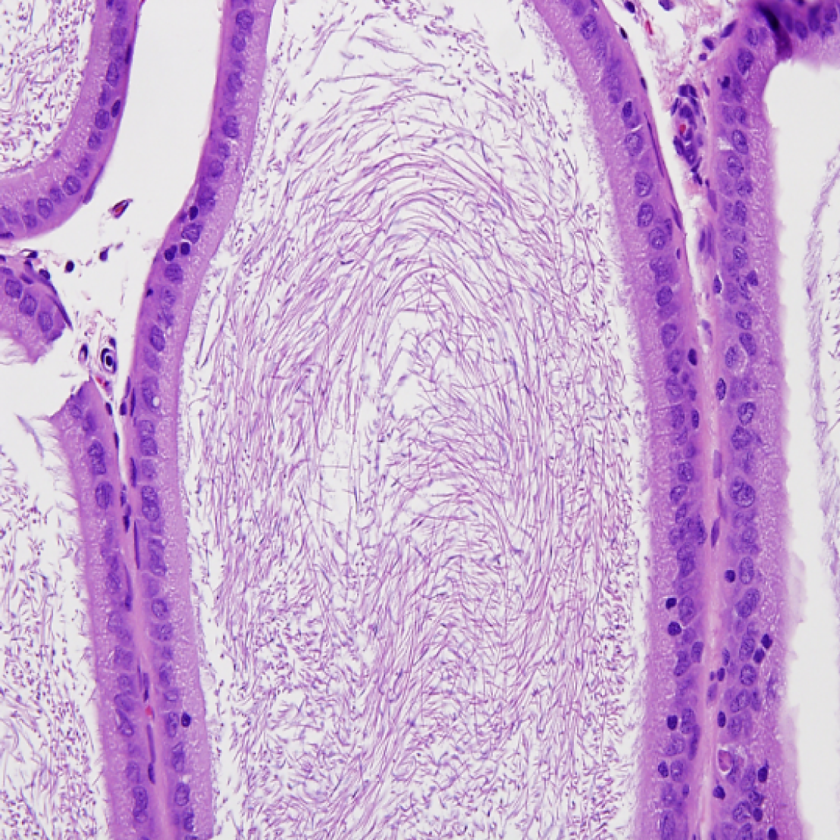
Photomicrograph of epididymis from a control male rats (×400).
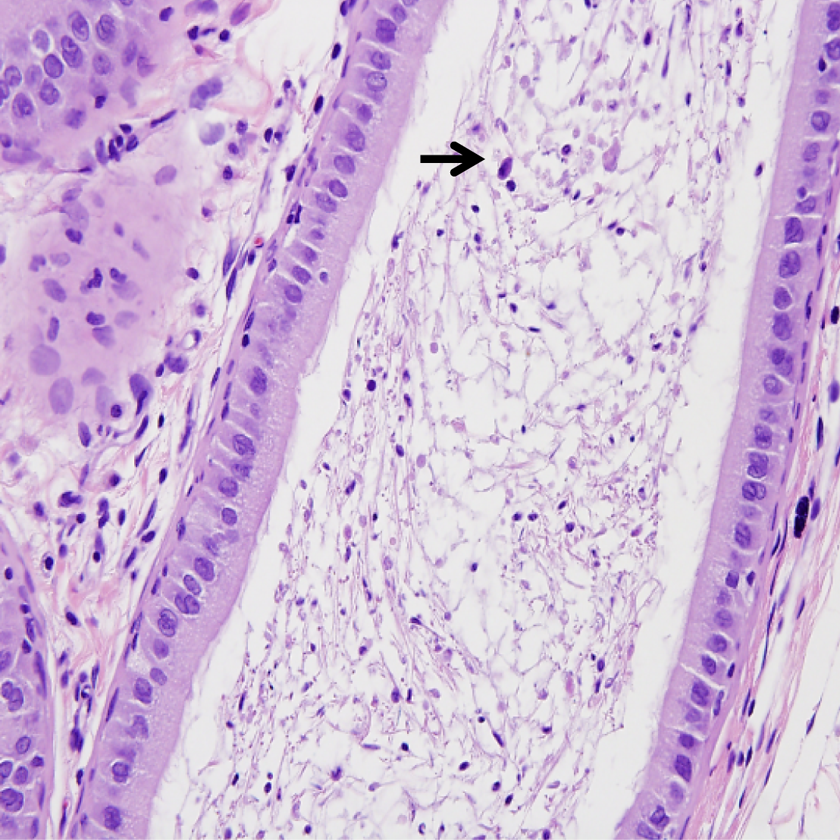
Photomicrograph of epididymis (×400) from male rats exposed 200 mg/kg/d indicating (arrows) cellular debris within the lumen with hypospermia.
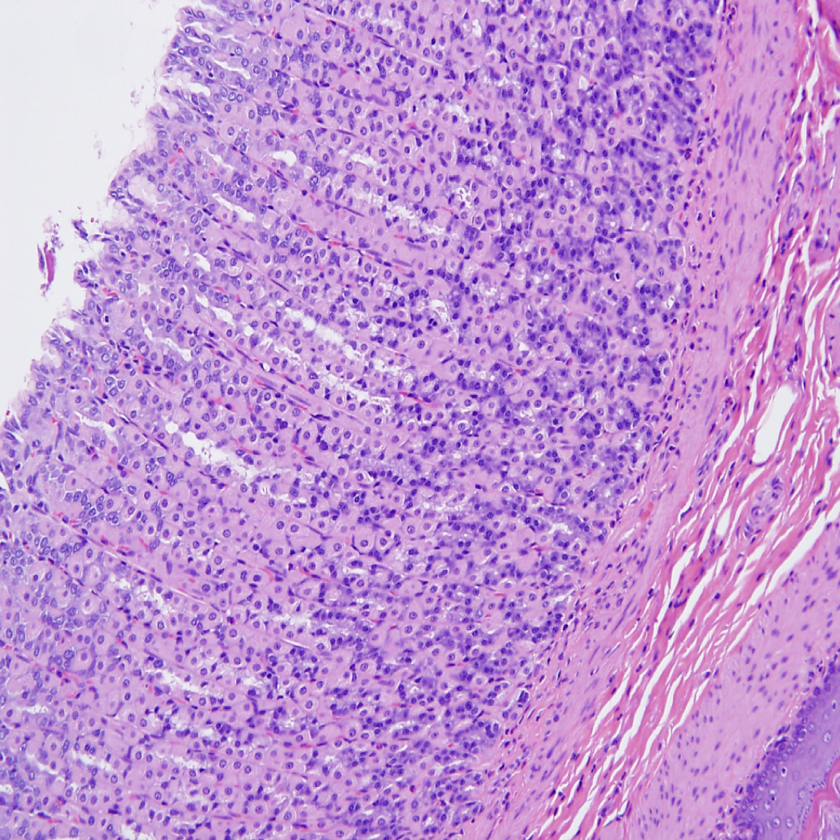
Histologic changes were also noted in the glandular stomach of males and females in high dosage groups. The changes included subacute inflammation consisting primarily of EOSs admixed with fewer mononuclear cells observed throughout the submucosa of 5 of 9, 4 of 10 males, and 8 of 10, 9 of 10 females in, 125 and 200 mg/kg/d dosage groups, respectively. Goblet cell metaplasia was also observed in the mucosa of the glandular stomach 8 of 9, 8 of 10 males and 8 of 10, 10 of 10 females of 125 and 200 mg/kg/d dosage groups, respectively (Figures 8 and 9). The gastric histologic findings in the lower dosed group were negative when compared to 2 high-dose groups.
Photomicrograph of stomach of control male rats (×200).
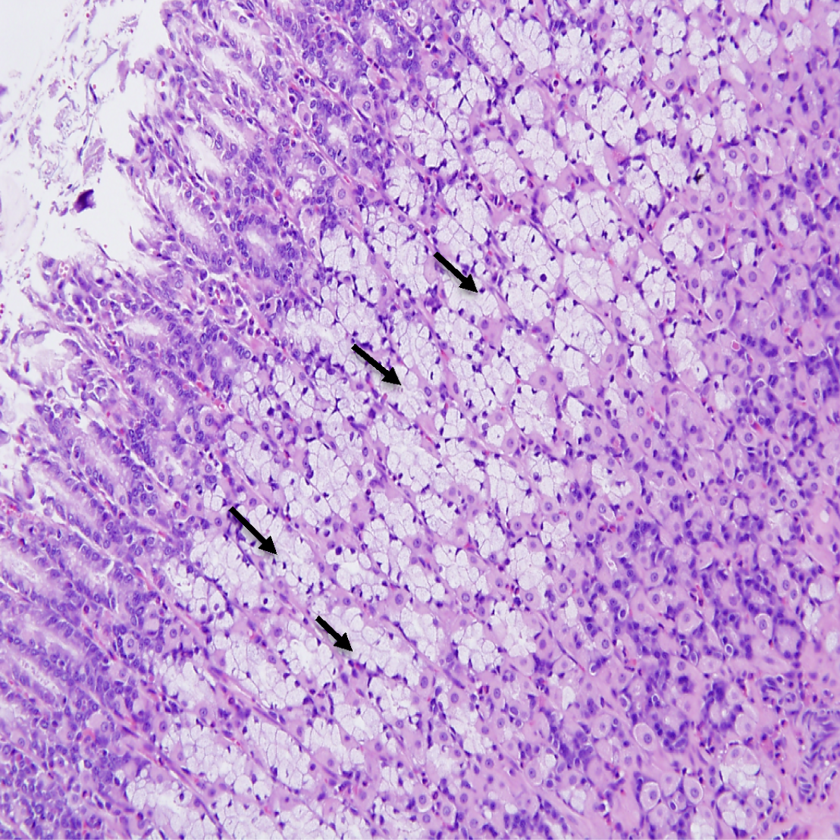
Photomicrograph of stomach (×200) from male rats exposed to 200 mg/kg/d demonstrating (arrows) Goblet cell metaplasia throughout the mucosa of the glandular stomach.
Discussion
There is considerable information on the toxicity of tungsten, but most of the early studies were focused on its interactions with other metals and the subsequent carcinogenicity and nutritional effects. There were also several studies that looked at its occurrence, fate, and transport in the environment, 2,10 –12 but there is no complete study to address the potential toxic effects of repeated oral exposure to tungsten. Therefore, we conducted a 90-day toxicity study in rats gavaged orally with sodium tungstate in water. A preliminary range-finding study in rats (with doses of 2, 10, 20, 200 mg/kg/d) helped to select the highest dose level (200 mg/kg/d) and dose range for the main study (data not presented). Our results showed that rats dosed with sodium tungstate in water for 90 consecutive days had no abnormal clinical signs at any of the dose levels (10, 75, 125, and 200 mg/kg/d). These results are similar to studies published previously, in subchronic exposure in drinking water to rats at doses of 100 or 200 ppm sodium tungstate for 2 to 8 weeks. 18,19 In our present study, the high-dose male group exhibited a significant dose-related decrease in food consumption from week 4 and a lower body weight gain from week 10 to 13 in male rats dosed at 200 mg/kg/d. No significant dose-related effects in food consumption or body weights were observed in female rats. The lower body weight gain in males may be due to a combination of decreased food consumption and sodium tungstate. Animals given 200 mg/kg/d displayed significantly lower absolute body weights and also displayed significantly lower liver, heart, and thymus weights compared to control rats. No effects in body weights or organ weights were noted in female rats. In addition, we found no significant changes in hematological and clinical parameters studied in either sex of rats. An increase in transaminase activities, creatinine, and reactive oxygen species was reported in a 14-day oral dose of 238 mg/kg/d and with intraperitoneal administration at 41 mg/kg/d of sodium tungstate. These changes may be due to effects on kidney and oxidative stress. 29
The histopathological evaluation indicated an increase in renal tubular basophila in the 175 and 200 mg/kg/d dose level which were due to tungsten. Basophilic epithelial cells are one of the most frequently encountered manifestations of induced nephron injury, particularly in repeat dose toxicity studies. 30 When associated with a thickened basement membrane, it is associated with CPN, a spontaneous background lesion of susceptible strains of laboratory rats such as Fischer 344 and SD. 31 –33 In this study, however, the tubules were not generally associated with thickened basement membrane, were more extensive, and the severity had a dose-dependent effect, indicating the effect was test article related.
Most metals that are absorbed through the gastrointestinal tract are excreted through renal clearance. The fate of oral sodium tungstate (50, 100, or 200 mg/kg/d) for 7 or 28 days and single intravenous (9 mg/kg) of sodium tungstate in rats showed the absorption of tungsten was rapid (1-3 hours). The plasma clearance and elimination half-life averaged 2.8 mL/min/kg and 3.04 hours in males and 3 mL/min/kg and 2.74 hours in females. 34 It has been reported that a single intravenous administration of sodium tungstate of 1 mg/kg in rats and mice or repeated oral exposure of sodium tungstate by gavage (10 mg/kg/d) or in drinking water (560 mg/L) to rats and mice for 14 days showed accumulation of tungsten in plasma and tissue such as intestine, kidneys, and femur of mice. 35 –37 Mice treated with 50 mg/kg/d sodium tungsten orally for 28 days showed accumulation in bone, kidney, liver, and brain of mice. 21 Renal effects with heavy metals were also reported with chemicals such as cadmium, uranium, and bismuth. 38 –40
We also noted histopathological changes in the gastric mucosa of dosed rats, namely, more prominent goblet cells than the gastric mucosa of control rats. These changes were observed in 2 high-dose groups (125 and 200 mg/kg/d). Sodium tungstate dosed orally at 5 mg/kg/d to rats for 7 months showed slight destruction of terminal villi with necrosis alterations in the small intestine. 22 Histopathological effects were reported in rabbit’s intestine at 5 mg/kg/d of sodium tungstate dosed orally for 8 months. 22 Rats exposed to sodium tungstate 100 or 200 ppm in drinking water for 30 weeks to evaluate the effects in esophagus and fore stomach on carcinogenicity and interactions with molybdenum and found no histological changes and no effects in influencing carcinogenicity. 19 In this study, we gavaged rats with sodium tungstate in water. The daily bolus dose in the stomach may produce irritation in the stomach of rats when compared to exposure in drinking water where no histopathological changes were noted in rats and mice. 19
Luminal cellular debris was observed in the epididymis of 3 rats from the 200 mg/kg/d group. Epididymal changes of this type are commonly encountered as rats reach sexual maturity and are presumed to represent degenerative cells that were released from the testis. However, rats in the present study should have reached sexual maturity before the time of necropsy. The epididymis of one had luminal cell debris that was limited to the tail region, suggesting some transient event that resulted in release of degenerative cells from the testis or epididymis. There were no identifiable testicular lesions that would explain the presence of degenerated cells in the lumen of the epididymis. It was reported that Wistar rats dosed orally with sodium tungstate at 50 mg/kg/d for 60 days showed no histopathological changes in testis and cauda epididymis. 21 The hypospermia and luminal cellular debris in the rat epididymis of high-dose (200 mg/kg/d) male rats is due to sodium tungstate. Male reproductive toxicity has been demonstrated in association with the exposure to certain metals such as cadmium, sodium arsenate and hexavalent chromium. These compounds injured seminiferous tubules and caused a reduction in spermatozoa. 41
We have evaluated sodium tungstate effects in rats dosed orally for 90 days and found renal effects that we used to develop an NOAEL, which could be used to develop an oral reference dose for risk assessment. Heavy metals such as cadmium and uranium are reported as nephrotoxicants. The NOAEL of cadmium was estimated to be 0.005 mg/kg/d, and this represents a dose not expected to produce proteinuria in humans. 38 Uranium is a classical nephrotoxin to rats, dogs, and rabbits. Rabbits showed greater sensitivity to uranium toxicity. Based on renal damage, the 2 lower doses (2.8 and 14 mg/kg/d) showed moderate and at high dose (71 mg/kg/d) severe renal effects in rabbits, and a lowest observable adverse effect level (LOAEL)of 2.8 mg/kg/d was reported. 39 These data suggest that cadmium and uranium are more toxic than the sodium tungstate that we reported here. Sodium tungstate administered orally to male and female SD rats by gavage for 90 consecutive days induced a number of statistically significant alterations at 125 and 200 mg/kg/d to male and female SD rats via oral gavage for 90 consecutive days. Based on our evaluation of the data, the LOAEL for renal changes for the subchronic oral toxicity of sodium tungstate in male and female SD rats was 125 mg/kg/d and the NOAEL was 75 mg/kg/d. Recently, Schell and Pardus 42 calculated a benchmark dose (BMD) for sodium tungstate using US EPA Benchmark dose software of our data of histological changes in kidneys of male and female (combined) animals. They calculated a BMDL10 of 102 mg/kg/d for sodium tungstate. This BMDL10 is in close agreement with the BMDL of 93 mg/kg/d of sodium tungstate derived from the data of reproductive toxicity study 43 by same authors. 42 Based on these data, we calculated BMDL10 of 98 and 91 mg/kg/d for males and females for sodium tungstate, respectively.
Footnotes
Author Contributions
Dr. W.C McCain contributed to conception and design, contributed to acquisition, analysis, and interpretation; drafted manuscript and critically revised manuscript. Mr. Lee Crouse contributed to conception and design, contributed to acquisition and analysis; drafted manuscript, and critically revised manuscript. Mr Matt. Bazar contributed to conception and design, contributed to acquisition, analysis, and interpretation. Dr. L.E Rossell contributed to design, contributed to acquisition and analysis, and critically revised manuscript. Dr. G.J. Leach contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. Mr. J. Middleton contributed to conception and design, and contributed to interpretation. Dr. Gunda Reddy contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript and critically revised manuscript. All authors gave final approval and all authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
We would like to thank LTC Erica E. Carroll, DVM, PhD, DACVP chief, Toxicologic Pathology Division, for critical review of manuscript and Martha Thompson, Mike Quinn, Patricia Beal, and Teresa Hanna for their technical support. We would also like to thank Mr Frank Hanzl, Program Manager (ARDEC), for his encouragement and support.
Authors’ Note
This work was presented in part at the 47th Annual Meeting of the Society of Toxicology, March 16-20, 2008, Seattle, WA, Toxicologist 102, 1-S, p 85.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from Army Research Development and Engineering Center (ARDEC), Picatinny, NJ.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
