Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 6 dialkyl malate compounds used in cosmetics. These ingredients function mostly as skin-conditioning agents—emollients. The Panel reviewed relevant animal and human data related to the ingredients along with a previous safety assessment of malic acid. The similar structure, properties, functions, and uses of these ingredients enabled grouping them and using the available toxicological data to assess the safety of the entire group. The Panel concluded that these dialkyl maleate compounds are safe in the present practices of use and concentration as given in this safety assessment.
Keywords
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook (INCI), 1 these 6 dialkyl malates mostly function as skin-conditioning agents—emollients (Table 1). The ingredients included in this report are:
The CAS Numbers, Definitions, and Functions of the Ingredients in This Safety Assessment.a

Abbreviation: CIR, Cosmetic Ingredient Review.
a The First Definition is provided by the International Cosmetic Ingredient Dictionary and Handbook; the Definition in Italics was Developed by the CIR Staff. 54
diisostearyl malate,
dibutyloctyl malate,
di-C12-13 alkyl malate,
diethylhexyl malate,
diisoamyl malate, and
dioctyldodecyl malate
The similar chemical structures, physicochemical properties, and functions and concentrations used in cosmetics enable grouping these ingredients and reading across the available toxicological data to support the safety assessment of the entire group. Dibutyl malate is not a cosmetic ingredient, however, it is a dialkyl malate and data regarding this chemical were considered relevant to the entire group.
Because these ingredients are diesters, esterases in the skin may metabolize them to the monoester and/or possibly to the free acid as well as the corresponding alcohol. For example, diisostearyl malate may result in isostearyl malate (the monoester), malic acid, and isostearyl alcohol, which may penetrate into the dermis. Summary data for malic acid are provided where appropriate in the main document and the toxicity data on the corresponding alcohols are included in Appendix A.
Chemistry
Definition, Structure, and Manufacture
Dialkyl malate ingredients have at their core succinic acid (a 4-carbon, alkyl diacid), that is, monohydroxy substituted. Malic acid (a dicarboxylic acid) is a monohydroxy succinic acid. Because of this diacid structure, these ingredients may be esterified with an alkyl group at each end of the molecule. For instance, diisostearyl malate is monohydroxy-substituted succinic acid, which is esterified at each end with a branched, 18 carbon alkyl chain (ie, isostearyl chain; Figures 1 and 2).

Diisostearyl malate.
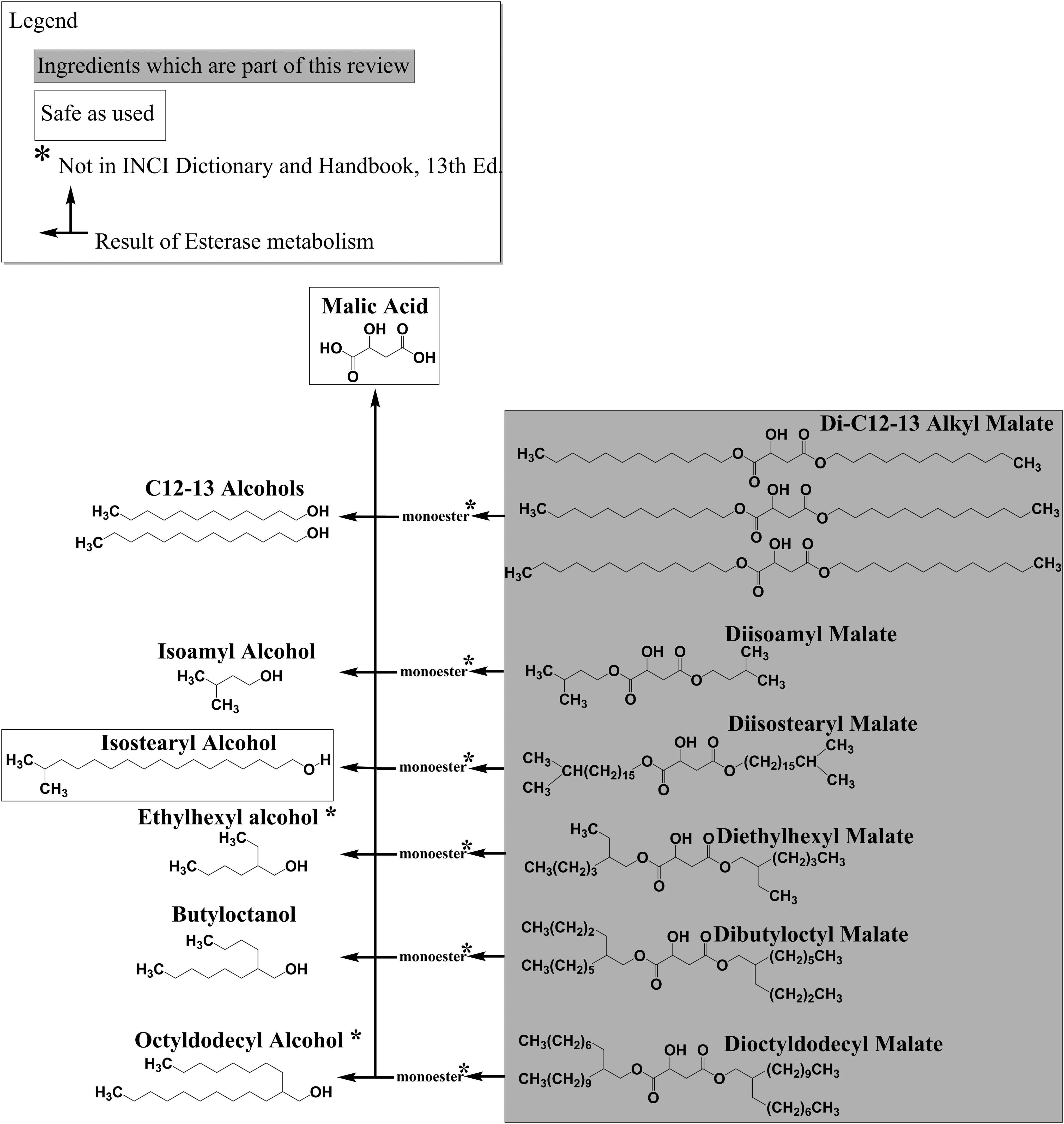
Map of the ester ingredients in this assessment and possible associated esterase metabolites.
All of these ingredients have at least 1 stereocenter denoted by
Dialkyl esters
The dialkyl esters of malic acid can be manufactured from malic acid by traditional esterification techniques, with the appropriate alcohol, and with or without acid or metal catalyst (Fischer esterification). 1 For example, diethylhexyl malate can be manufactured from malic acid and ethylhexanol with a titanium catalyst. 2 It is likely that all of the diacid is consumed in the reaction and no malic acid is present in the final product.
Malic acid
Malic acid (monohydroxysuccinic acid), a white crystalline material, has one stereocenter, at the carbon bearing the hydroxyl group.
3
The
A mixture of maleic, fumaric, and rac-malic (or ±malic) acids heated with water in a closed space will cause the maleic acid to be consumed and the resulting solution to reach an equilibrium between fumaric acid and
In 2001, the Cosmetic Ingredient Review Expert Panel (Panel) reviewed and concluded that the dicaboxylic acid, malic acid, and its sodium salt (sodium malate) are safe for use as pH adjusters in cosmetic formulations. 7 The Panel determined that the data are insufficient to determine the safety of these ingredients for any other functions. The data from the malic acid and sodium malate safety assessment are summarized in this report.
Chemical and Physical Properties
Chemical and physical property data are provided in Table 2. Diethylhexyl malate was reported to be 99% pure, with the impurities being unreacted raw materials. 8 It is soluble in oil, ethanol, and silicone and insoluble in propylene glycol, water, and dimethicone. 8
Physical and Chemical Properties of Alkyl Malates.

Use
Cosmetic
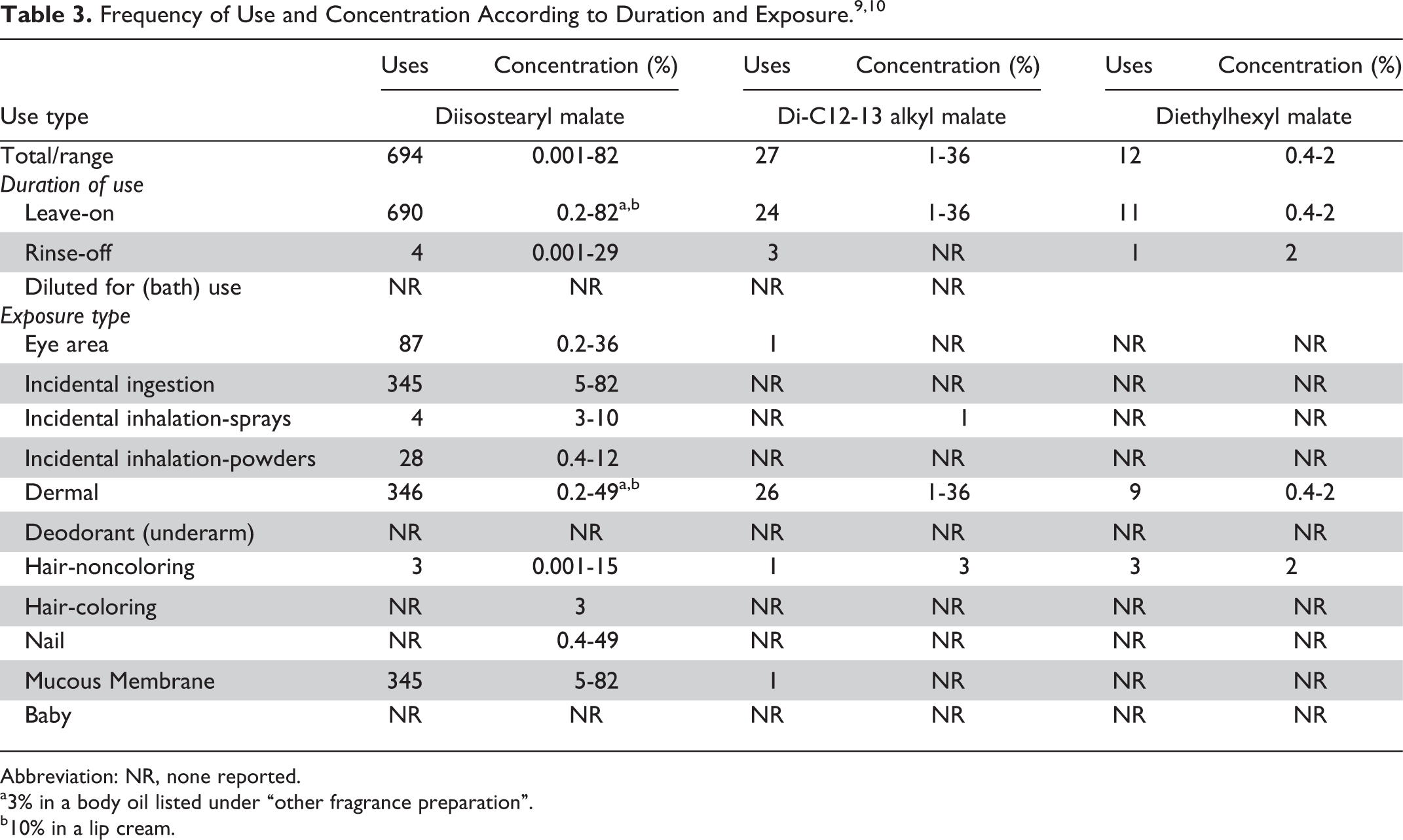
Data on ingredient usage are provided to the US Food and Drug Administration Voluntary Cosmetic Registration Program. A survey was conducted by the Personal Care Products Council regarding the maximum use concentrations for ingredients in this group (Table 3). 9,10
Abbreviation: NR, none reported.
a3% in a body oil listed under “other fragrance preparation”.
b10% in a lip cream.
Diisostearyl malate was reported to be used in 690 leave-on products. Maximum concentration of use was reported to be up to 82% in leave-on products and 29% in rinse-off products. Di-C12-13 alkyl malate was reported to be used in 27 products and at a reported maximum use concentration of up to 36%. Diethylhexyl malate was reported to be used in 12 leave-on products at a maximum use concentration of up to 2% in leave-on and 2% in rinse-off products. There were no reported uses of dibutyloctyl malate, diisoamyl malate, and dioctyldodecyl malate.
Diisostearyl malate is reported to be used at concentrations of up to 10% in cosmetic products that may include sprays of which airborne particles could possibly be inhaled. The size distribution of 95% to 99% of the particles released from cosmetic sprays has had aerodynamic equivalent diameters >10 µm. 11 -14 Therefore, particles incidentally inhaled from cosmetic sprays would likely be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 11,13
Noncosmetic
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
No absorption, distribution, metabolism, or excretion studies on the dialkyl malates in this report were found or submitted to Cosmetic Ingredient Review.
Oral and intraperitoneal
Malic Acid
Malic acid plays a part in carbohydrate metabolism and is a precursor of oxalacetic and pyruvic acids.
16
Most of the radioactivity from 2.5 mg/kg U 14C-
Toxicological Studies
Acute Toxicity
Dermal
Di-C12-13 alkyl malate
The acute dermal median lethal dose (LD50) of di-C12-13 alkyl malate for male and female Wistar rats (n = 5) was >2000 mg/kg (2 mL/kg; 100%). 19 There were no clinical signs during the 7-day observation period and no pathology at necropsy.
Oral—nonhuman
Di-C12-13 alkyl malate
The acute oral LD50 of di-C12-13 alkyl malate for Wistar rats (n = 5/sex) was >5000 mg/kg in sesame seed oil. 20 There were no mortalities. Intense fur erection and ante- and postmortem mycosis was observed in one foreleg.
Diethylhexyl malate
The reported LD50 for diethylhexyl malate is >5g/kg for albino rats (strain not provided). 21
Malic acid and sodium malate
Malic acid was administered as a 25% aqueous solution. Signs of toxicity included ataxia, prostration, convulsions, retraction of the abdomen, respiratory distress, cyanosis, and death. The oral LD50s of malic acid for albino CD-1 outbred mice (n = 5/sex), albino Wistar rats (n = 5/sex), and Dutch-Belted rabbits (n = 5/sex) were approximately 2.66, 3.5, and 3 g/kg, respectively. 22 -25
The oral LDLO of malic acid for rabbits was 5 g/kg.
26
The oral “lethal dose” of
Dibutyl malate
The oral LD50 of dibutyl malate was reported to be 3730 mg/kg for rats. 28
Other dose administration
Malic Acid
The acute LD50 of malic acid administered intravenously (iv) was 2.4 g/kg for rabbits, and the ip LD50 values for mice and rats were 50 to 100 and 100 to 200 mg/kg, respectively. 27
In a comparative study of the
The ip administration of 2 g/kg
Repeated Dose Toxicity
Dermal
Di-C12-13 alkyl malate
Di-C12-13 alkyl malate (10 mL/kg; 1000 mg/kg/d) was dermally applied under occlusion daily to the shaved dorsal area of male and female New Zealand white rabbits (n = 5/sex) for 28 days. 31 No clinical signs were observed during treatment and the 2-week observation period. Necropsies, hematology, and clinical chemistry (including liver function) were unremarkable.
Oral—nonhuman
Malic acid and sodium malate
In a chronic oral study of 3 extracts of malic acid (500, 5000, and 50 000ppm; 0.05%, 0.5%, and 5.0%), Charles River rats (n = 30/sex) had no compound-related lesions (types not provided) after feeding the test substance for 104 weeks. 32 No significant changes or lesions were observed when dogs were fed malic acid (500, 5000, and 50 000 ppm) for 104 weeks. 33
Dibutyl malate
Dibutyl malate (300 mg/kg) produced renal tubular lesions and increased liver and kidney weights in a repeated oral dose and reproductive toxicity/developmental toxicity screening assay using rats. The no observed effects level (NOEL) was 95 mg/kg. 28
Reproductive and Developmental Toxicity
No reproductive or developmental studies were discovered for the ingredients in this safety assessment, however, data from developmental toxicity studies were available for malic acid and dibutyl malate.
Malic Acid
Malic acid did not cause developmental toxicity in albino CD-1 outbred mice (n = 25) up of to 266 mg/kg (days 6-15 of gestation), rats (n = 25-29) of up to 350 mg/kg (for 10 days during gestation), or Dutch-belted rabbits (n = 15-23) of up to 300 mg/kg (days 6-18 of gestation). 34 -36 In a multigenerational oral study of malic acid, there were no reproductive or developmental effects to albino rats for up to 10 000 ppm of feed for the P1, P2, F1, and F2 generations. 37
Malic acid (10.00 mg/egg in water) was injected into the air sac or yolk of white leghorn chicken eggs (n = 20) at the 0 or 96 hours of incubation. 38 There were no developmental effects observed when the chicks were examined after hatching.
Dibutyl Malate
In a repeated dose and reproductive toxicity/developmental toxicity screening assay for dibutyl malate (300 mg/kg), there were no adverse reproductive effects reported in rats. 28
Genotoxicity
Diisostearyl Malate
The potential of diisostearyl maleate to induce gene mutation was studied in Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537, using reverse mutation assay. 39 The assay was performed with and without metabolic activation at concentrations of 312.5, 625, 1250, 2500, and 5000 μg/plate. The test substance was not mutagenic under these test conditions. The results of the positive controls were as expected.
Di-C12-13 Alkyl Malate
In an Ames test, di-C12-13 alkyl malate (1, 10, 100, 1000, and 10 000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, TA1537, and TA1538) with or without metabolic activation. 40 The results of the positive controls were as expected.
In a micronucleus assay using C57BL mice (n = 5/sex), di-C12-13 alkyl malate (250 mg/mL; 12 500 mg/kg in sesame seed oil) administered ip was not mutagentic. 41 Erythrocytes harvested from bone marrow were examined at 24, 48, and 72 hours.
Carcinogenicity
There were no carcinogenicity studies discovered for the ingredients in this safety assessment.
Malic Acid
In a chronic oral study, feeding malic acid (500, 5000, and 50 000 ppm; 0.05%, 0.5%, and 5.0%) to Charles River rats (n = 30/sex) for 104 weeks resulted in no compound-related lesions (types not provided). 32 No significant changes or lesions were observed when dogs were fed malic acid (500, 5000, and 50 000 ppm) for 104 weeks. 33
Irritation and Sensitization
Irritation
Dermal—nonhuman
Di-C12-13 alkyl malate
Di-C12-13 alkyl malate (10 mL/kg) was dermally applied under occlusion daily to the shaved dorsal area of male and female New Zealand white rabbits (n = 5) for 28 days. 31 No erythema or edema was observed.
Di-C12-13 alkyl malate (500 mg in 0.5 mL sesame seed oil) was not a dermal irritant when administered to the shaved skin (20 cm2) of New Zealand white rabbits (n = 6) for 4 hours. 42 No erythema or edema were observed at 1, 24, 48, and 72 hours.
Diethylhexyl malate
Diethylhexyl malate (100%; 0.5 mL) was applied to intact and abraded skin of New Zealand white rabbits (n = 6) under occlusion. The test sites were observed at 24 and 72 hours. The primary irritation index (PII) was 1.18; diethylhexyl malate was determined not to be a primary irritant to rabbits. 43
A primary dermal irritation test of diethylhexyl malate (100%) under occlusion for 24 hours was conducted using New Zealand white rabbits (n = 6). 44 The PII was 3.53. The authors concluded that diethylhexyl malate was not a dermal irritant to rabbits.
Malic acid
Malic acid (500 mg/24 hours) was moderately irritating to rabbit skin and was a strong irritant to guinea pigs. 25
Dermal—human
Di-C12-13 alkyl malate
In a human patch test (n = 38), di-C12-13 alkyl malate (100%; 0.5 mL) was not irritating when administered to a 1 cm2 area under occlusion for 48 hours. 45 No erythema or edema was observed at 15 minutes and 24 hours.
Malic acid
In a test determining the subjective skin irritation potential (n = 10), the average irritation scores over a 15-minute period were 39.4, 37.1, and 23.1 for malic acid (1 mol/L in ethanol [SD40], ethoxydiglycol, and butylene glycol) at pH 3, 5, and 7, respectively. 46
In 2 human repeated insult patch tests (HRIPTs) of products containing malic acid (0.022725% and 0.00375%), these products were predicted to be non- to moderate irritants (Table 4). 47
Dermal Irritation, Sensitization, and Ocular Irritation Studies of Products Containing Malic Acid. 47
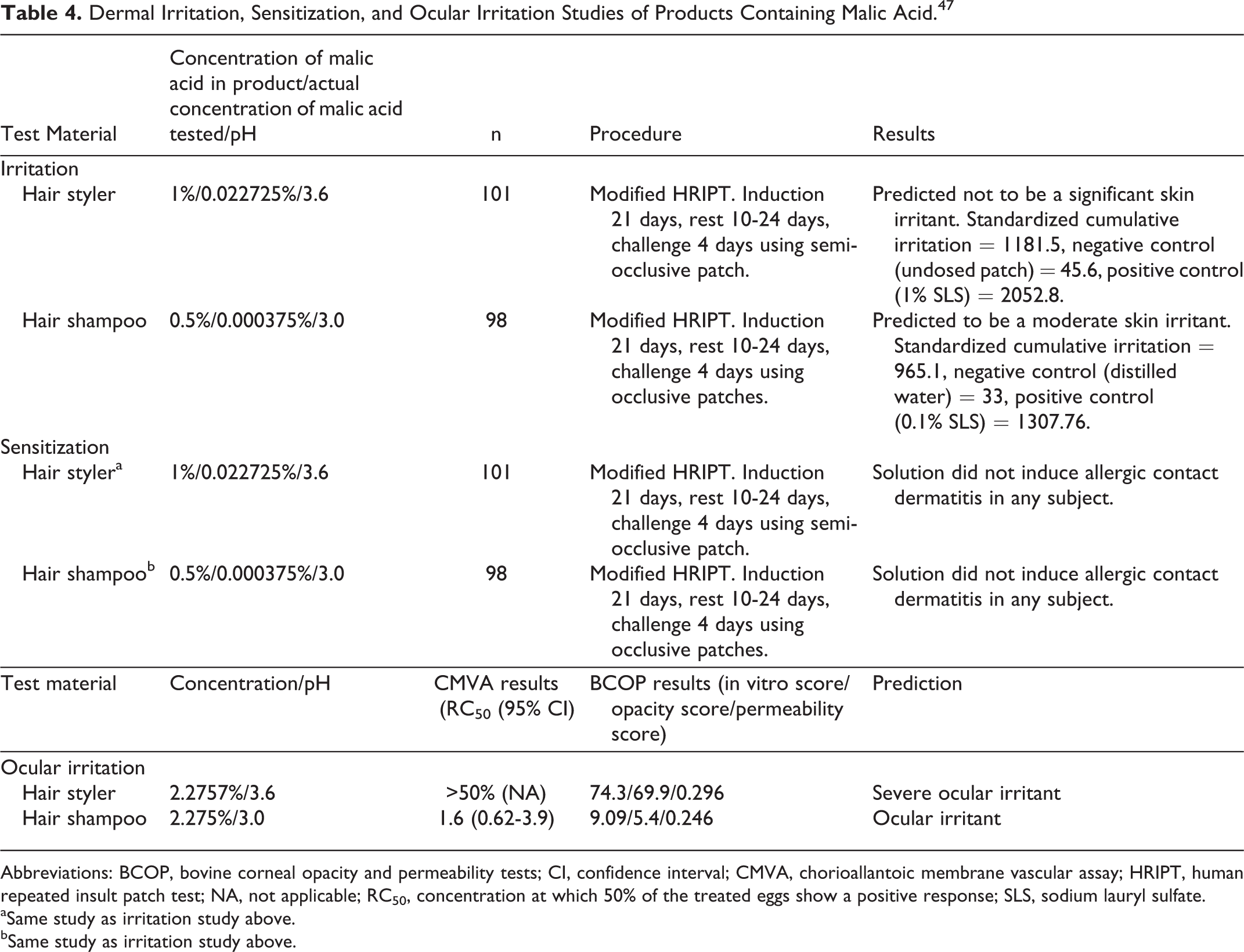
Abbreviations: BCOP, bovine corneal opacity and permeability tests; CI, confidence interval; CMVA, chorioallantoic membrane vascular assay; HRIPT, human repeated insult patch test; NA, not applicable; RC50, concentration at which 50% of the treated eggs show a positive response; SLS, sodium lauryl sulfate.
aSame study as irritation study above.
bSame study as irritation study above.
Ocular
Di-C12-13 alkyl malate
Di-C12-13 alkyl malate (100%; 0.1 mL) was not irritating to the eyes of New Zealand white rabbits (n = 6). 48 In a chorioallantoic membrane (CAM) assay, di-C12-13 alkyl malate (200 μL) was not predicted to be an ocular irritant. 49
Diethylhexyl malate
Diethylhexyl malate (100%; 0.1 mL) was administered into 1 eye of New Zealand white rabbits (n = 6). 50 The eyes were unwashed for 24 hours. At 24 and 72 hours and 4 and 7 days, the Draize score was 0. At 48 hours, the score was 0.3. The authors determined that diethylhexyl malate was not an ocular irritant to rabbits.
Diethylhexyl malate (100%; 0.1 mL) was administered into 1 eye of New Zealand white rabbits (unwashed, n = 6; washed after 4 seconds, n = 3). 44 At 24 hours, the Draize score for the unwashed eyes was 2.0 and 0.7 for the washed eyes. At 48 and 72 hours, the Draize score was 0. The authors determined that diethylhexyl malate was practically nonirritating to rabbits.
Malic acid
Malic acid caused severe ocular irritation in rabbit eyes. 25 In CAM vascular assay and bovine corneal opacity and permeability tests of products containing malic acid (2.2725%), these products were predicted to be ocular irritants (Table 4). 47
Sensitization
Dermal—nonhuman
Di-C12-13 alkyl malate
In a guinea pig sensitization assay (Buehler test) using female Hartley guinea pigs (n = 10), epicutaneous administration of di-C12-13 alkyl malate (100%) for three 6-hour exposures 7 days apart under occlusion was not sensitizing. 51
Diethylhexyl malate
In a guinea pig sensitization test (Buehler test; n = 12) of diethylhexyl malate (100%), there was slight erythema on 2 sites after the sixth dose. 21 There was no sensitization observed at challenge.
Dermal—human
Diisostearyl malate
An HRIPT (n = 51) of diisostearyl malate (100%; 0.2 mL, 0.2 g) was performed. 52 No adverse effects were observed during induction or challenge.
Malic acid
In predictive testing using patients with atopic dermatitis, 18 of 34 patients reacted to a diet high in malic and citric acids and 6 reacted to a diet high in malic acid. 53 In an in vitro study assessing the effect of malic acid on cell renewal on human skin, an 18%, 10%, and 5% increase was observed at pH 3, 5, and 7, respectively. In 2 HRIPTs of products containing malic acid (0.022725% and 0.00375%), sensitization was not induced (Table 4). 47
Summary
Dialkyl malates are cosmetic ingredients that have a core of succinic acid (a 4-carbon, alkyl diacid) that is mono substituted. All of these ingredients have at least 1 stereocenter, and the stereoisomers are
Dialkyl malates function mostly as skin-conditioning agents—emollients. Malic acid can be produced by a process that uses maleic acid (a related but structurally different dicarboxylic acid), but the reaction products are generally purified until the amounts of fumaric and residual maleic acid are 7.5 and <500 ppm, respectively. It was considered likely that all of the diacid is consumed in the reaction, and no malic acid is present in the final product. Because these ingredients are diesters, however, esterases in the skin may metabolize them to the monoester or possibly to the free acid and corresponding alcohol.
The total number of reported uses of diisostearyl malate was 574 (572 uses in leave-on products) and was reported to be used at 0.001% to 82%. Di-C12-13 alkyl malate was reported to be used in 29 products and was reported to be used at 1% to 36% in leave-on products. Diethylhexyl malate was reported to be used in 10 products and was reported to be used at 0.4% to 2% in leave-on products and 2% in rinse-off products. There were no reported uses for dibutyloctyl malate, diisoamyl malate, and dioctyldodecyl malate.
Radiolabeled malic acid orally and ip administered to rats was excreted mostly as carbon dioxide.
The acute dermal LD50 of di-C12-13 alkyl malate for rats was >2000 mg/kg. The acute oral LD50 of di-C12-13 alkyl malate for rats was > 5000 mg/kg. The reported LD50 for diethylhexyl malate was > 5g/kg for rats.
The oral LD50 of malic acid for mice was 2.66 g/kg, 3.5 g/kg for rats, and 3 g/kg for rabbits. The oral lethal dose of malic acid was 5 g/kg in rabbits. The oral lethal dose of sodium malate was 1 g/kg in dogs. The iv LD50 of malic acid was 2.4 g/kg for rabbits, and the ip LD50 values for mice and rats were 50 to 100 and 100 to 200 mg/kg, respectively. The ip administration to rats of 1 g/kg
There were no clinical signs observed during treatment and the 2-week observation period when 10 mL/kg di-C12-13 alkyl malate was dermally applied to rabbits for 28 days. In a chronic oral study, feeding malic acid up to 50 000 ppm to rats for 104 weeks resulted in some changes in body weight gains and feed consumption, but compound-related lesions were not observed. No significant changes or lesions were observed when dogs were fed malic acid in a chronic 104-week study.
Malic acid did not cause developmental toxicity in mice up to 266 mg/kg, rats up to 350 mg/kg, or rabbits up to 300 mg/kg. In a multigenerational oral study of malic acid, there were no reproductive or developmental effects in rats up to 10 000 ppm in the P1, P2, F1, and F2 generations. Malic acid at 10.00 mg in water/egg injected into the air sac or yolk of chicken eggs at the 0 or 96 hours of incubation caused no developmental effects in the chicks.
In a reverse mutation assay using S typhimurium, diisostearyl malate was not mutagenic with or without metabolic activation up to 5000 μg/plate. In an Ames test, di-C12-13 alkyl malate was not mutagenic to S typhimurium with or without metabolic activation up to 10 000 μg/plate.
In a micronucleus assay using C57BL mice (n = 5/sex), di-C12-13 alkyl malate (250 mg/mL; 12 500 mg/kg in sesame seed oil) administered ip was not mutagenic.
Di-C12-13 alkyl malate at 100% was not a dermal irritant in rabbits treated daily for 28 days. Malic acid (500 mg/24 h) was moderately irritating to rabbit skin and was a strong irritant to guinea pigs. In a test determining the subjective skin irritation potential, the average irritation scores over a 15-minute period were 39.4, 37.1, and 23.1 for malic acid at pH 3, 5, and 7, respectively.
In a human patch test, di-C12-13 alkyl malate was not irritating at 100%. Di-C12-13 alkyl malate was not irritating to the eyes of rabbits at 100%. In a CAM assay, di-C12-13 alkyl malate was not predicted to be an ocular irritant. Malic acid caused severe ocular irritation in rabbit eyes at 500 mg.
Di-C12-13 alkyl malate and diethylhexyl malate were not sensitizing to guinea pigs at 100%. No adverse effects were observed during induction or challenge in an HRIPT of diisostearyl malate at 100%.
In predictive testing using patients with atopic dermatitis, 18 of 34 patients reacted to a diet high in malic and citric acids, and 6 reacted to a diet high in malic acid. In assessing the effect of malic acid on cell renewal, an 18%, 10%, and 5% increase was observed at pH 3, 5, and 7, respectively. In 2 HRIPTs of products containing malic acid up to 0.022725%, sensitization was not induced.
Discussion
The similar chemical structures, physicochemical properties, functions, and concentrations in cosmetics allowed grouping these ingredients together and for extending the available toxicological data to support the safety of the entire group.
The Panel noted that the previous conclusion of the malic acid/sodium malate safety assessment stated that there were insufficient data to reach a safety conclusion on the use of these ingredients as anything but pH adjusters, and there was a need for dermal sensitization studies. An HRIPT of diisostearyl malate at 100% did not result in irritation during induction or sensitization at challenge, and guinea pig sensitization tests of diethylhexyl malate and di-C12-13 alkyl malate, both at 100%, also did not produce sensitization in these animals. The Panel concluded that even though malic acid/sodium malate may be irritating, these results support the view that the dialkyl malates are not irritating or sensitizing.
There was concern expressed that the possible metabolite octyldodecanol is a penetration enhancer and dermal irritant. However, the potential concentration of the alcohol that would be generated in skin would be well below the level of concern.
The Panel discussed the issue of incidental inhalation exposure from cosmetic sprays and loose powders (ie, perfumes, face powders, body and hand sprays, and foot powders and sprays). These ingredients are reportedly used at concentrations up to 10% in cosmetic products that may be aerosolized. While there was a lack of inhalation toxicity data for the dialkyl malates in this safety assessment, the Panel noted that 95% to 99% of particles/droplets would not be respirable to any appreciable amount. Furthermore, these ingredients are not likely to cause any direct toxic effects in the upper respiratory tract, based on the properties of the dialkyl malates and on data that show that these ingredients are not irritants. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for dialkyl malates to cause systemic toxicity, irritation, or sensitization. They noted the lack of systemic toxicity at high doses in acute and subchronic oral exposure studies, no irritation or sensitization in multiple tests of dermal and ocular exposure, and the absence of genotoxicity in 2 Ames assays and a micronucleus assay. A detailed discussion of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following dialkyl malates are safe for use in cosmetics in the present practices of use and concentration in this safety assessment: diisostearyl malate, dibutyloctyl malate*, di-C12-13 alkyl malate, diethylhexyl malate, diisoamyl malate*, and dioctyldodecyl malate*
*Not reported in current use. Were ingredients in this group to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Appendix A: Possible Esterase Metabolite Summary Data Of Alcohols
Author Contributions
Becker contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Gill contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Bergfeld contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Belsito contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Hill contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Klaassen contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Liebler contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Marks contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Shank contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Slaga contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Snyder contributed to conception and design; contributed to analysis and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Former CIR Director F. Alan Anderson contributed to conception and design, analysis, and interpretation and critically revised the manuscript.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.