Abstract
Prostate cancer is a common disease in older men. Rodent models have demonstrated that an early and later-life exposure to estrogen can lead to cancerous lesions and implicated hormonal dysregulation as an avenue for developing future prostate neoplasia. This study utilizes a human fetal prostate xenograft model to study the role of estrogen in the progression of human disease. Histopathological lesions were assessed in 7-, 30-, 90-, 200-, and 400-day human prostate xenografts. Gene expression for cell cycle, tumor suppressors, and apoptosis-related genes (ie, CDKN1A, CASP9, ESR2, PTEN, and TP53) was performed for 200-day estrogen-treated xenografts. Glandular hyperplasia was observed in xenografts given both an initial and secondary exposure to estradiol in both 200- and 400-day xenografts. Persistent estrogenic effects were verified using immunohistochemical markers for cytokeratin 10, p63, and estrogen receptor α. This model provides data on the histopathological state of the human prostate following estrogenic treatment, which can be utilized in understanding the complicated pathology associated with prostatic disease and early and later-life estrogenic exposures.
Introduction
Prostate adenocarcinoma (PCa) is the second leading cause of cancer-related deaths in the United States and was responsible for the largest percentage (28%) of new noncutaneous cancer in 2013. 1 Age, race, and family history 2 are well-known risk factors for PCa development, and new evidence supports the idea that dietary factors, 3 such as increased consumption of soy products and red meat, may play an important role in disease progression. 4,5 Although PCa predominantly occurs in older men, recent evidence from rodent studies suggests that exposure to estrogenic chemicals during development may be responsible for earlier disease onset. 6 –8
Rodent studies have been instrumental in demonstrating that exposure to exogenous estrogens during a critical time in development, also known as developmental estrogenization, can lead to an increase in prostate pathology. 9 A model, termed “1-hit” versus “2-hit,” explored this concept by exposing neonatal rats to estradiol and bisphenol A during prostate organogenesis to assess the resulting histological and molecular changes in the prostate 8,10 ; the rats were then allowed to develop without further exposure (1 hit) or exposed to estrogen a second time later in life (two hit). This model has been informative and has revealed that a brief early exposure to estrogenic chemicals, in conjunction with a subsequent later exposure, can lead to an increase in the incidence of prostatic neoplasia. Although the mechanisms are unknown, these results indicate that the early exposure to estrogen can play a role in “priming” this hormonally sensitive organ and produce molecular changes that may be responsible for both the earlier onset and the faster progression of prostatic disease. 10
This concept of hormonal priming of prostate tissue has been extensively explored in animal models 11 ; however, little data are available on the relationship between estrogen exposure during early development and disease progression in humans. Human prostate development differs from rodent prostate development in many ways, as it begins at approximately 11 weeks of gestation arising from the urogenital sinus. 12 During this differentiation process, the prostate forms ducts and glands responsible for prostatic secretions and a surrounding smooth muscle stroma. 13 –15 Cross-talk between the epithelial and the stromal tissues directs the maturation process and is critical for normal prostate development. 13,16,17 This delicate process is halted soon after birth, 18 and this timing is quite different from rodent prostate development in which most development and maturation occurs after birth. 10 In addition to these differences in timing of development and maturation, there are also significant structural differences; specifically, the human prostate is arbitrarily divided into 3 zones (peripheral, central, and transitional), 15 while the rodent prostate is made of 4 lobes (ventral, lateral, dorsal, and anterior). 19 This raises an important question in regard to species comparability 20 and emphasizes the need to study the effects of estrogen in human tissue.
The present study examines the histopathological effects of developmental estrogenization in a human fetal prostate xenograft model and provides new insight regarding the human response to estrogens during this critical period of development and the role it plays in prostate disease progression.
Materials and Methods
Brown University entered into an institutional review board (IRB) Intra-Agency Agreement (#10-15) naming Women and Infants Hospital as the IRB of record for this study (project #: 09-09077; protocol title: Children’s Environmental Health Project). Informed and written consent was obtained from donors. Brown University Institutional Animal Care and Use Committee (IACUC) has reviewed and approved all experimental protocols for this project, and all animals were euthanized by an overdose of isoflurane (Baxter Healthcare Corporation, Deerfield, Illinois) followed by cervical dislocation.
Chemicals
Estradiol benzoate (β estradiol 3-benzoate, CAS#: 50-50-0), testosterone (CAS#: 58-22-0), and corn oil (CAS#: 8001-30-7) were purchased from Sigma Aldrich (St Louis, Missouri).
Silastic Capsule Preparation
Tubing (inner diameter: 1.47 mm, outer diameter: 1.96 mm) for silastic capsules was purchased from Dow Corning (Cat. No: 508-006, Midland, Michigan). Silastic tubing was cut into 2-cm pieces and filled with either estradiol benzoate (E8515, Sigma) or testosterone (T1500, Sigma). Acetoxy silicone (A-564, Factor II Inc., Lakeside, Arizona), a medical adhesive, was used to seal both ends of the tube. Tubes were allowed to dry for at least an hour prior to its use. 21
Animals
T-cell deficient adult male Nude rats (Crl: NIH-Foxn1 rnu, strain code 316, aged 8-9 weeks old) were obtained from Charles River Laboratories (Wilmington, Massachusetts). Rats were provided with free access to water and fed Purina Rodent Chow 5001 (Farmer’s Exchange, Framingham, Massachusetts). The animals were kept in a temperature and humidity controlled room along with a continuous 12-hour light–dark cycle following national regulations. The Brown University IACUC approved all experimental protocols for the project entitled “Formative Center for the Evaluation of Environmental Impacts on Fetal Development.”
Human Prostate Tissue Acquisition
Human fetal prostate tissue (second trimester, gestational age 17-23.7 weeks) acquisition is in accordance with the Women and Infants Hospital (WIH, Providence, Rhode Island) IRB and was acquired after spontaneous pregnancy loss, in collaboration with WIH. This period was chosen for tissue acquisition because it is an important period of organogenesis in human fetal prostate. 12 De Paepe et al provides a detailed description of tissue procurement. 22 Briefly, written and informed consent is obtained from donors, and a board-certified pathologist evaluated the condition of the human tissue prior to implantation. Anonymous identifiers were used for each human sample.
Xenotransplantation Surgery
In accordance with IRB, surgeries were performed at Brown University’s Xenotransplantation Core Facility. We have previously described the procedure in detail in Saffarini et al. 23 Briefly, human prostate tissue was kept in ice-cold transport media (Leibovitz L-15, penicillin, streptomycin, and gentamycin) until surgery. Rats were placed under isoflurane anesthesia (Baxter Healthcare Corporation), and a small peritoneal incision was made to expose the kidney. A piece of human fetal prostate (1-3 mm) was inserted under the kidney capsule, and the incision was sutured. Adverse effects resulting from surgery were monitored daily for 2 weeks following xenotransplantation.
Xenograft Dosing Paradigm
The exposure model used in this study resembles the 1 hit versus 2-hit model. 24,25 Rat hosts were given a subcutaneous injection of corn oil (control) or 250 µg/kg of β estradiol 3-benzoate (treatment) in corn oil following xenotransplantation, and then every other day, resulting in a total of 3 injections (one hit). This dose was chosen to determine an effect on human fetal prostate tissue while preventing toxicity within the rodent host. Prior studies that developed the 1-hit versus 2-hit model used 25 µg of estradiol benzoate as the 1-hit dose and found that this exposure was relevant to a prior exposure of diethylstilbestrol that was given to pregnant women. Additionally, the estradiol benzoate was measured and found to be highly bound to α-fetoprotein in the serum of neonates. 26 Xenografts that evaluated early estrogenic exposures were collected at 7, 30, or 90 days post xenotransplantation (Figure 1A). For xenografts that evaluated early and late estrogen exposures, a subset of animals was given a 1-cm slow-releasing silastic capsule packed with approximately 10 mg β estradiol 3-benzoate (not dissolved in corn oil), and two 2-cm silastic capsules containing 25 mg of testosterone (not dissolved in corn oil; total 50 mg testosterone). This dose of estradiol led to the development of prostatic intraepithelial neoplasia (PIN) in previous rat models, 8 while testosterone was given in conjunction to prevent prostatic involution resulting from estradiol treatment. 24 Empty silastic capsules were given to control animals, and all control and treatment capsules were replaced after 8 weeks. A subset of rat hosts was collected on day 200, while some were allowed a recovery period of 200 days and then subsequently collected on day 400 (Figure 1B). Due to the limited amount of human prostate tissue obtained, secondary treatment groups evaluating estrogen alone or testosterone alone could not be included in this investigation. Tissue was separated among the treatment groups, but the small amount of original tissue was a limiting factor. The number of hosts per treatment group are as follows: 7-day corn oil (2), 7-day estradiol (1), 30-day corn oil (2), 30-day estradiol (1), 90-day corn oil (2), 90-day estradiol (1), 200-day corn oil/empty capsules (4), 200-day corn oil/estradiol and testosterone (3), 200-day estradiol/empty capsules (2), 200-day estradiol/estradiol and testosterone (4), 400-day corn oil/empty (1), 400-day corn oil/estradiol and testosterone (2), 400-day estradiol/empty capsules (2), and 400-day estradiol/estradiol and testosterone (2).
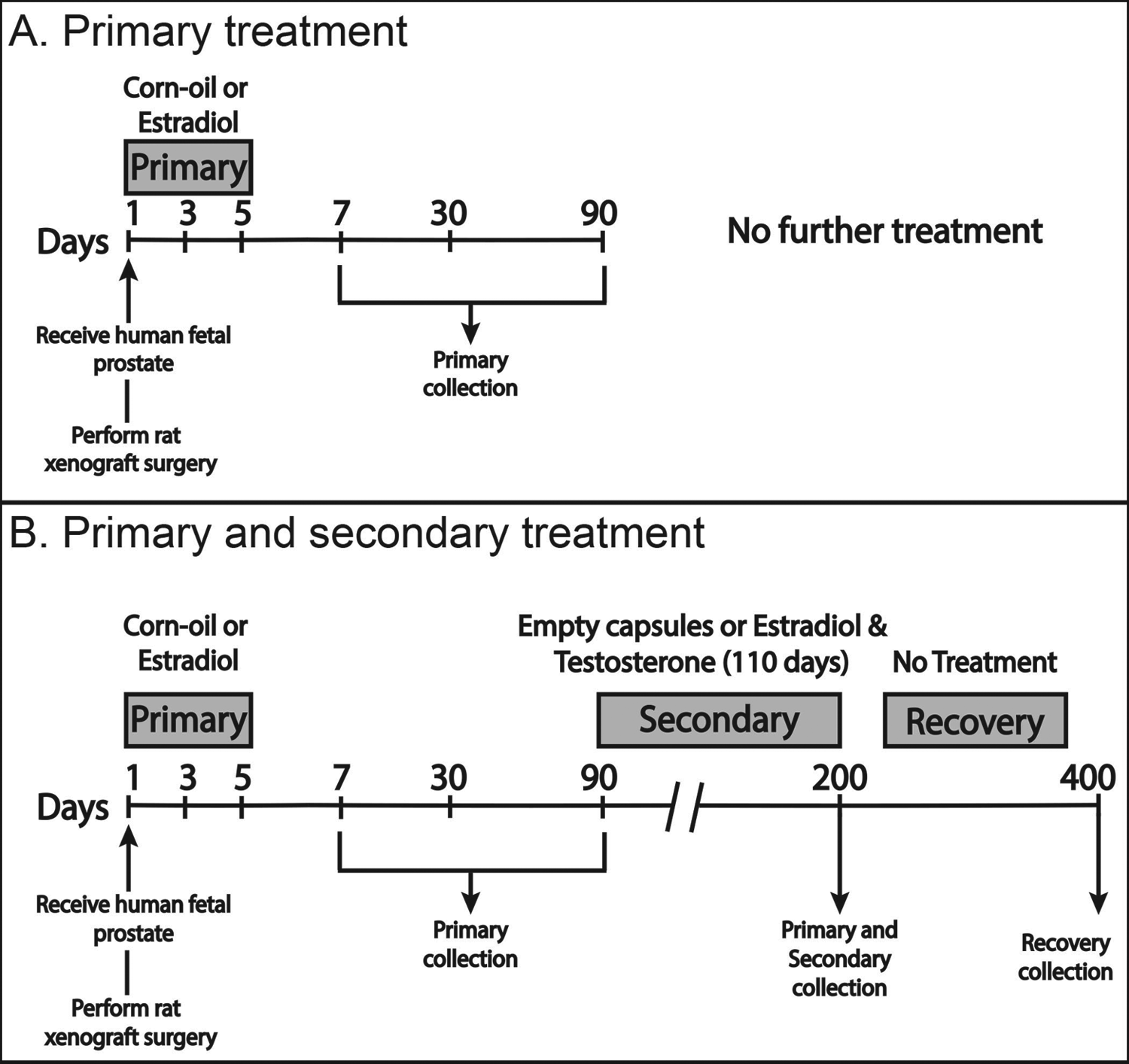
Dosing paradigm used in this study to examine the early- and later life effects following estrogen exposure on human prostate xenografts. A, Human fetal prostates from spontaneous pregnancy losses were obtained and implanted under the renal subcapsular space of an immunodeficient rat host (day 1). Following xenograft surgery, rat hosts were given a subcutaneous injection of either corn oil (control) or 250 µg/kg of β estradiol 3 benzoate (treatment) on days 1, 3, and 5. To explore the initial effects of estrogen, a subset of xenografts were collected on days 7, 30, and 90 (primary collection). B, An additional subset of rat hosts was administered silastic capsules on day 90, containing either empty capsules (control) or β estradiol 3-benzoate and testosterone capsules (secondary treatment), for a total of 110 days. To examine the effects of paired early- and later-life estrogen exposure, some rat hosts were collected on day 200 (primary and secondary collection), while a subset of animals had treatment removed and were subsequently collected on day 400 (recovery group).
Xenograft Collection and Histological Processing
Prostate implants were collected at 7, 30, 90, 200, and 400 days post xenotransplantation and divided into 3 different pieces. One piece was placed in 10% neutral-buffered formalin for paraffin embedding, while another was put into Tissue-Tek O.C.T. compound (Electron Microscopy Sciences, Hatfield, Pennsylvania) for frozen sectioning. The remaining piece was placed in the −80°C freezer for later RNA extraction. Paraffin blocks were sectioned at 5 µm and stained with hematoxylin and eosin and various immunohistochemical markers. Aperio Scanscope CS microscope (Aperio Technologies, Vista, California) was utilized for slide scanning and ImageScope software (Aperio Technologies) for visualization.
RNA Isolation
RNA was isolated, along with the additional DNase step, using the RNeasy Micro Kit (Qiagen, Valencia, California) according to manufacturer’s instructions and stored in the −80°C freezer until further use.
Immunohistochemistry
Xenograft tissue slices were stained with immunohistochemistry markers for cytokeratin 10 (Abcam, Cambridge, Massachusetts, ab76318, 1 µg/mL), p63 (Santa Cruz Biotechnology, Santa Cruz, California, sc-8431, 0.5 µg/mL), and estrogen receptor-α (Dako Cytomation, Carpinteria, California, M7047, 1 µg/mL) to examine changes in the microenvironment of the epithelial and stromal compartments. Paraffin tissue sections were deparaffinized and rehydrated with xylene and graded ethanols. A 10 mmol/L citrate buffer solution (pH 6) heated in a vegetable steamer was used for antigen retrieval. Sections were blocked for avidin and biotin (SP-2001, Vector Laboratories, Burlingame, California). Antibodies were optimized for concentration, and the appropriate secondary antibody was added. Staining was visualized using an avidin–biotin complex (PK-6100, Vector Laboratories) and peroxidase substrate 3,3’-diaminobenzidine (SK-4100, Vector Laboratories). Appropriate positive and negative controls were incorporated for each staining experiment.
Quantitative Polymerase Chain Reaction
Optimized primer assays for human CASP9 (Cat. PPH00353B), CDKN1A (Cat. PPH00211E), ESR2 (Cat. PPH00992C), PTEN (Cat. PPH00327F), and TP53 (Cat. PPH00213F), as well as 3 housekeeping genes RPLP1 (Cat. PPH17813G), 18S rRNA (Cat. PPH05666E), and GAPDH (Cat. PPH00150F), were obtained from Qiagen. The RT 2 First Strand Kit (Cat. 330401, Qiagen) was used to synthesize complementary DNA, and SYBR Green/ROX master mix (Cat. 330520, Qiagen) was prepared according to manufacturer’s instructions. The original piece of human tissue was considered as the statistical unit for analysis for both 200-day control/control (n = 3) and 200-day estrogen/estrogen (n = 3) treatment groups. Prepared samples were placed onto 96-well plates (Cat. 4346906, Life Technologies, Grand Island, New York) and run on a ViiA 7 Real-Time PCR System (Life Technologies) using recommended cycling conditions from the manufacturer.
Statistical Analysis
Figures were generated using Adobe Illustrator (Adobe Systems, San Jose, California). Raw quantitative polymerase chain reaction data imported from the ViiA 7 software program (Life Technologies) was normalized against the geometric mean of the housekeeping genes (ie RPLP1, 18S rRNA, and GAPDH). Data from the PCR arrays were analyzed using the ΔΔCt method, comparing the control against the estrogen-treated values, according to manufacturer’s instructions (Qiagen). Significance was determined by a Student t test (P value < .05).
Results
This study evaluated the histopathological effects of estrogen on human prostate tissue comparing the 1-hit and 2-hit models; the former used early-life estrogen exposure alone and the latter paired early-life and later-life estrogen exposures to examine prostate development and carcinogenesis (Figure 1).
Treatment With Estradiol Produces Early Ductal and Stromal Changes in Prostate Xenografts
Histopathological analysis revealed ductal and stromal differences in estradiol-treated (Figures 2B, 2D, and 2F) compared to control samples (Figures 2A, 2C, and 2E) in 7-, 30-, and 90-day prostate xenografts. The 7-day control (Figure 2A) and estrogen-treated xenografts (Figure 2B) presented with primordial small glands that were surrounded by a highly cellular undifferentiated mesenchyme. Squamous metaplasia was also found in the 7-day xenografts.
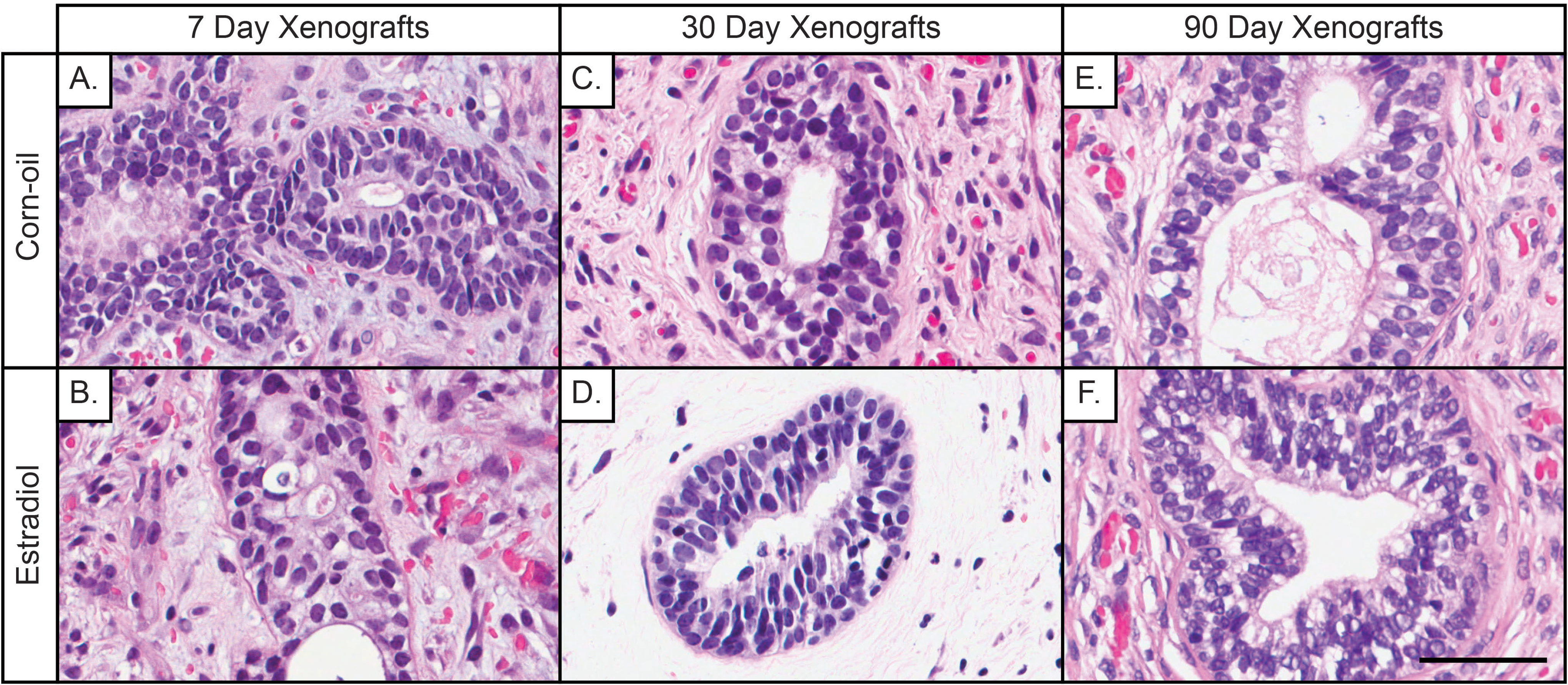
Histology of 7-, 30-, and 90-day human prostate xenografts that received an early exposure to estradiol. A-B, At day 7, both control (corn oil) and estrogen-treated prostate xenografts have primordial small ducts with basal cell hyperplasia. A, The stroma in the corn oil group is composed of a busy cellular mesenchyme, representative of normal fetal prostate stroma, while the (B) treated group shows better stromal maturation with immature smooth muscle cells. C, At 30 days postimplantation, the corn oil xenografts present with immature ducts, but a better-developed stroma, while (D) treated xenografts show similar epithelial features as the controls but an immature edematous and hypocellular stroma. E and F, Both corn oil and estradiol-treated xenografts appear morphologically similar at 90 days, demonstrating mature ducts with proper secretory function and stromal maturation similar to the adult prostate. Prior to implantation, the gestational age of the human fetal prostate was 23.7 weeks. (Hematoxylin and Eosin staining; scale bar = 50 μm).
At 30 days postimplantation, untreated xenografts displayed basal cell hyperplasia, focal glandular maturation with capability of producing normal prostatic secretions. These glands were surrounded by blood vessels and newly developed smooth muscle cells (Figure 2C). In comparison, 30-day estradiol-treated xenografts presented with small primordial glands surrounded by an immature stromal mesenchyme. The treated stroma appeared markedly different than the control stroma with little differentiation in smooth muscle and fibroblast cells and marked edema and hypocellularity in some cases (Figure 2D). In the 90-day xenografts, both control and estradiol-treated presented with a normal adult prostate appearance containing large ducts with well-developed basal and luminal cell layers and a mature stroma with well-developed smooth muscle and connective tissue (Figures 2E and 2F).
Initial and Secondary Treatments With Estradiol (Estradiol/Estradiol and Testosterone) Lead to Hyperplasia in 200-Day Human Xenografts
To examine the effect of both early and later estradiol exposures, rat hosts were exposed to initial (corn oil vs estradiol) and secondary (empty capsules vs. estradiol and testosterone capsules) treatments on day 90 for a total of 110 days, and the xenografts were subsequently collected at day 200 (Figure 3). Control xenografts, which received no initial or secondary treatments (corn oil/empty capsules; C/C), appeared as phenotypically normal adult prostate tissue, containing normal tortuous glands capable of secretion and a mature stroma (Figure 3A). Xenografts that received an initial dose of estradiol alone with no subsequent later life exposure (estradiol/control; E/C) appeared phenotypically similar to the C/C implants. An early exposure to estradiol without a subsequent later exposure did not alter long-term prostate morphology, and these xenografts presented with well-developed glands and a mature stroma at 200 days (Figure 3B), while xenografts that were given only the later estradiol (corn oil/estradiol and testosterone; C/E) contained numerous atrophic ducts. Ductal tissue was primarily affected by the treatment, while the stromal compartment was histologically normal (Figure 3C). The prostate implants that received the double estrogenic exposure (estradiol/estradiol and testosterone; E/E) displayed hyperplastic and enlarged glands at 200 days postimplantation, and the stromal compartment had mature fibroconnective and smooth muscle tissues (Figure 3D) without any evidence of disrupted growth.
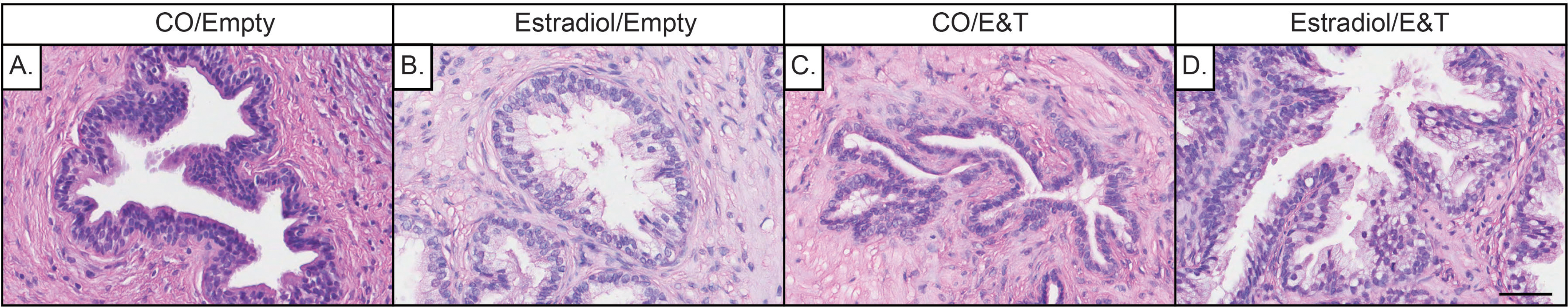
Histology of human prostate xenografts at 200 days postimplantation that was given initial (corn oil or estradiol) and secondary (empty or estradiol and testosterone silastic capsules) treatments to evaluate the effects of early- and later-life exposures to estradiol. Controls for the secondary treatment were empty silastic capsules. A, Control human prostate xenografts (corn-oil/empty) present with normal, phenotypically adult prostatic ducts encompassed by a cellular stroma. B, Human prostate xenografts given only an early dose of estradiol with no subsequent exposure (estradiol/empty) appear similar to control xenografts, with adult glands and a mature stromal environment. C, Xenografts given only a later dose of estradiol (corn oil/estrogen and testosterone) present with atrophic ducts incapable of secretory function. D, Xenografts that received both early and later doses of estradiol (estradiol/estradiol and testosterone) demonstrate significant glandular hyperplasia, with no apparent changes to the stroma. A-D, Gestational age of human prostate before implantation was 21 weeks, and treatment conditions are depicted as primary/secondary exposure. CO indicates corn oil; E&T, estradiol and testosterone. (Hematoxylin and Eosin staining; scale bar = 50 μm).
Xenografts Allowed to Recover for 200 Days after Estrogen Exposure Reveal Persistent Treatment-Related Effects
A subset of E/E-treated xenografts was allowed a recovery period of 200 days, beginning on day 200 post implantation and ending on day 400 (Figure 4). The recovery xenografts demonstrated persistent estrogen-related effects that manifested as ductal hyperplasia. The 400-day human E/E-treated prostate xenografts did not develop any further histopathological indicators of precancerous or cancerous lesions (e.g., PIN lesions) beyond persistent hyperplasia.
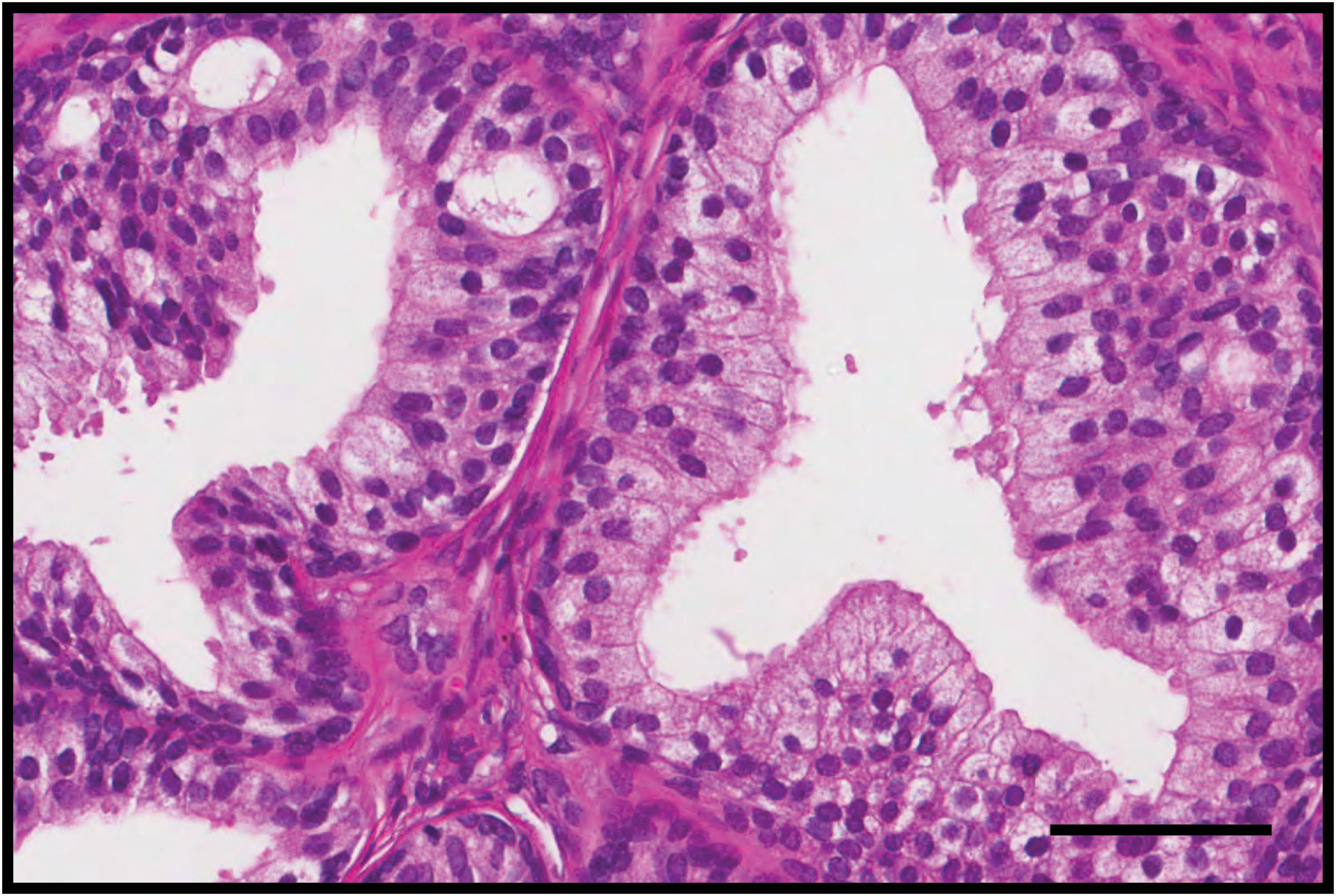
Histology of a human prostate xenograft at 400 days postimplantation that was exposed to both early and later doses of estradiol (E/E&T) followed by a recovery period of 200 days. Estrogen-related treatment effects persist in the epithelial compartment after a 200-day recovery period as demonstrated by the presence of ductal hyperplasia. Gestational age of human prostate before implantation was 22 weeks. E&T indicates estradiol and testosterone. (Hematoxylin and Eosin staining; scale bar = 100 μm).
To further characterize the persistent estrogenic effects seen in 200- and 400-day E/E-treated prostate xenografts, the tissue was stained using immunohistochemical markers for cytokeratin 10 (CK10), p63, and estrogen receptor-α (ERα; Figure 5). CK10 showed strong staining in the epithelial compartment of 200-day human xenografts (Figure 5A), which persisted to day 400 (Figure 5D). In addition, basal cell proliferation was assessed using p63. The 200-day xenografts appeared normal, with cells in the ductal tissue displaying p63 staining in a continuous fashion along the basal lamina (Figure 5B), while ducts in the 400-day xenografts exhibited basal cell hyperplasia (Figure 5E). The expression of ERα was located in both the epithelial and the stromal compartments in 200-day xenografts (Figure 5C) but was primarily found in the epithelial compartment of 400-day xenografts (Figure 5F).
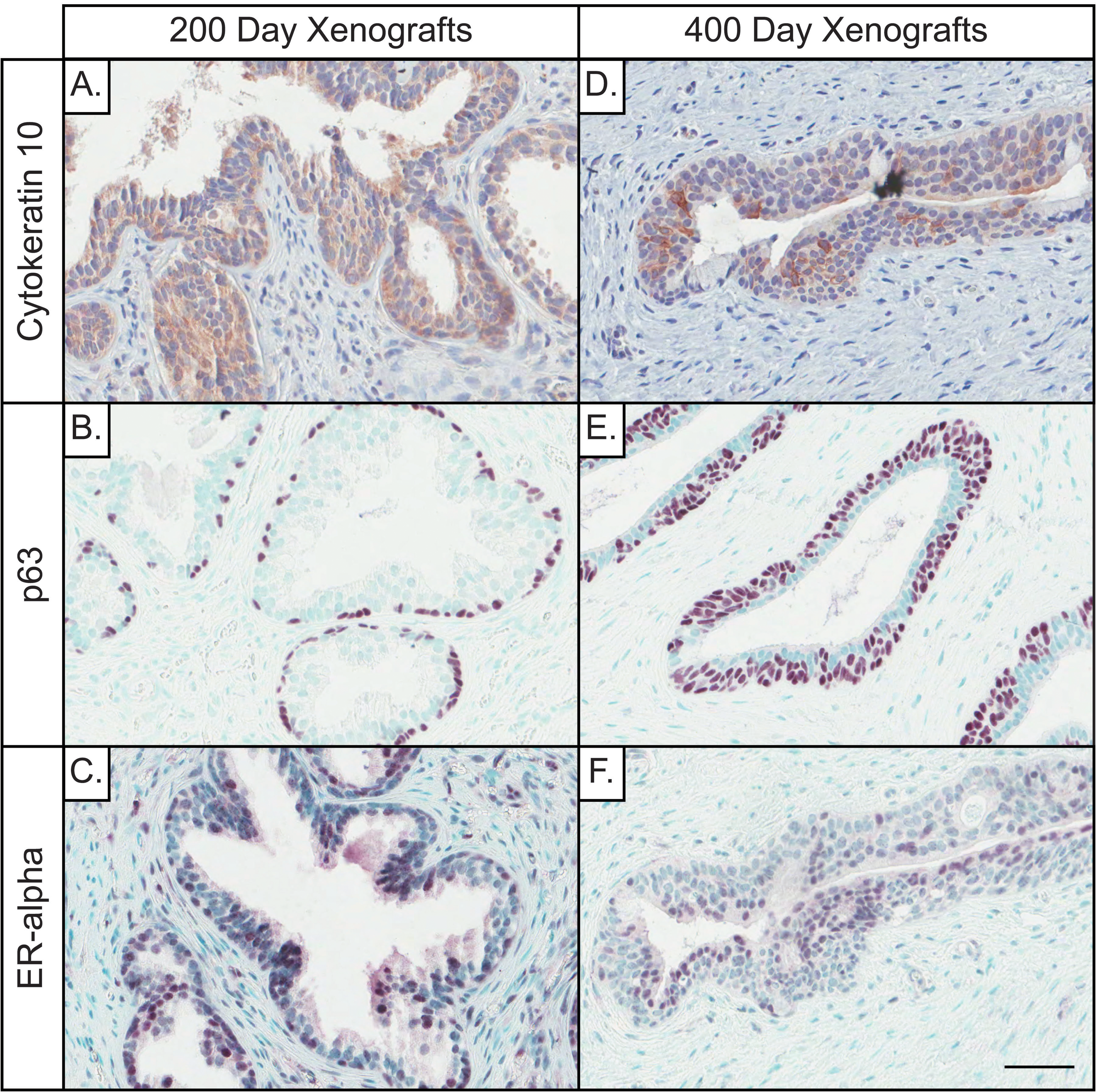
Immunohistochemical staining of cytokeratin 10 (squamous metaplasia), p63 (basal cell), and estrogen receptor-α (hormone receptor) markers in 200- and 400-day E/E human prostate xenografts at 200 and 400 days postimplantation. A and D, The epithelia show a similar pattern of CK10 staining at 200 and 400 days postimplantation, suggesting that squamous metaplasia is a persistent consequence of estrogen exposure. B, At 200 days postimplantation, xenografts display a mature phenotype with epithelial hyperplasia and 1 layer of basal cells. E, After 200 days of recovery, epithelial hyperplasia is still present, along with some areas of significant basal cell hyperplasia. C, ERα shows strong epithelial and stromal staining after 200 days, (F) while there is residual epithelial but less stromal staining at 400 days. Gestational age of human prostate before implantation is 17 weeks (A-C), and 22 weeks (D-F). Counterstain is Hematoxylin (A and D), and methyl green (B, C, E, and F). (Scale bar = 50 μm).
Expression of genes important to cell cycle progression, apoptosis, and tumor suppression in prostate cancer was measured in 200-day C/C- and E/E-treated xenografts (n = 3). These genes were studied based upon a previous analysis in which they were significantly altered following estrogenic treatment of human fetal prostate that was implanted into athymic nude mouse hosts. These genes included cyclin-dependent kinase inhibitor 1A (CDKN1A), caspase 9 (CASP9), estrogen receptor β (ESR2), phosphatase and tensin homolog (PTEN), and tumor protein 53 (TP53). Interestingly, in our rat hosts, no significant changes were found in any of the aforementioned gene expression data between the 200-day C/C and E/E xenografts (Supplemental Figure 1).
Discussion
It is not surprising that research is increasingly focused on early-life events as a basis for adult disease. Exposure to exogenous chemicals during fetal and early postnatal development can affect normal growth processes during this important hormone-sensitive period, altering cellular differentiation and interfering with critical molecular mechanisms. 27,28 Animal studies are commonly used to investigate the consequences of exposure to endocrine disrupting chemicals in the male reproductive tract, 29,30 and while these studies provide important insight into the cellular pathways that may be affected, they may not accurately replicate the processes that occur in human body. This study uses a human fetal prostate xenograft model, which avoids the problems associated with species extrapolation, to investigate the pathology resulting from early- and later life exposures to estrogen.
An Initial Exposure to Estrogen Affects Stromal Growth in Human Xenografts
Proper development of the prostate depends on cross-talk between epithelial and stromal compartments. 31 At 7 days postimplantation, both control and treated xenografts displayed squamous metaplasia; however, this is not considered abnormal during the fetal period, as the fetal prostate is normally exposed to high levels of maternal estrogens 32 that produce squamous metaplasia. 33,34 In conjunction with squamous metaplasia, ducts were surrounded by a highly cellular mesenchyme, indicating that cells are not undergoing apoptosis following xenotransplantation (Figures 2A and 2B). This indicates that the prostate is capable of acclimatization under the renal subcapsular space quickly following implantation.
At 30 days, the epithelial cells of the control xenografts were beginning to develop, although basal cell hyperplasia remained prominent. Relative to the 7-day xenografts, the 30-day untreated samples displayed a more organized stromal compartment, with premature smooth muscle and connective tissue present (Figure 2C). However, the 30-day estrogen-treated samples contained ducts that were largely underdeveloped and primordial in appearance (Figure 2D). The most striking difference in the estrogen-treated xenografts was the lack of cellularity and advanced development in the stromal compartment. Interactions between the epithelial and stromal compartments are critical for proper development, with both compartments regulating growth and hormonal balance through paracrine signaling. 35 The lack of a proper stroma indicates that developmental estrogen exposure may primarily target the stromal compartment or may interfere with the paracrine signals secreted from the epithelial compartment which aid in stromal growth. Although there are obvious histopathological effects at 30 days, these effects disappeared by 90 days, and both control and treated xenografts appeared histologically similar (Figures 2E and 2F). This suggested that the prostate was able to recover from the estrogen-induced damage, and the mechanisms of this recovery warrant further investigation. Since the cross-talk between the epithelial and the stromal compartments are critical for prostate maturation, it is important to isolate these cell compartments (ie, epithelial cells from stromal cells) to investigate these changes associated with the recovery response seen in the 90-day xenografts. This can easily be done using the technique of laser capture microdissection and will be employed in our future studies.
Initial and Secondary Exposures to Estrogen/Estrogen and Testosterone Produce Hyperplasia in Prostate Xenografts
Paired early- and later-life estrogen treatments Estrogen/Estrogen and Testosterone (E/E&T) were used to explore the long-term 200 days histopathological effects of estrogenic exposure. These E/E&T xenografts have marked histopathological changes including glandular hyperplasia (Figure 3). The response seen in these human xenografts differs from what has been previously demonstrated in rat studies. High-grade PIN lesions, which are hypothesized to be precursors to prostate carcinoma, 2 developed in rat prostates in response to paired initial and secondary exposures to high-dose estradiol, 10,25 while our human xenograft model showed hyperplasia with no further indication of prostatic neoplasia or carcinogenesis (eg, PIN). The striking difference between the rat and the human prostate models highlights the need to study human tissues in greater depth and to investigate whether rodents are more susceptible to estrogenic effects in the context of their use as model organisms. This also leads us to question the life span of our rodent host and whether it is adequate enough to demonstrate the prostatic responses that we are investigating following treatment. Our xenograft model might be well suited to investigate early exposures over an acute period but may need to be modified if we want to investigate and compare it to human disease, which has quite a long latency to develop into cancer.
A subset of the 200-day E/E&T human xenografts was allowed to recover for an additional 200 days, to be collected on day 400, to investigate persistent estrogenic effects. Ductal hyperplasia remained prominent in these samples, demonstrating that double-hit estrogen effects are long lasting in the human prostate (Figure 4). However, the extent to which this occurs is unclear because the life span of the rat host limits the duration of xenograft growth and development to the 400 days used in this experiment. Again this brings up a limitation of this model, as 400 days is only a minor fraction of the time required for human prostate cancer development and therefore may not be long enough to observe important and potentially carcinogenic effects in our human xenotransplants. Future experiments will employ the method of serial surgeries to maintain the life span of the xenograft tissue and allow it to grow for an extended period of time. This process would involve removing the xenograft after 400 days and reimplanting it in a new young rodent host. The hormonal environment of the host is also a concern, as hormones play a vital role in prostatic growth. Our previous publication has demonstrated that the human xenografts display accelerated maturation, as shown by the expression of the adult smooth muscle marker caldesmon in 30-day xenografts. 36 It may also be necessary to use a castrated model that would allow controlling the hormones administered to our hosts as well as the xenograft exposure. Despite this limitation, however, the use of human tissue to evaluate the developmental origins of PCa is novel and useful.
The 200-day E/E&T Human Xenografts Yield no Significant Changes in Gene Expression
We have used a similar xenotransplant in a murine host to investigate the expression of 40 genes that are important in stromal and epithelial development and prostate carcinogenesis (unpublished data). Results from this human into mouse model revealed significant changes in expression of CDKN1A, CASP9, ESR2, PTEN, and TP53, which are involved in the cell cycle, 37 apoptosis, 38 and tumor suppression 39,40 following a similar dosing paradigm (one hit versus two hit). These five genes were analyzed in this rat host study, but no significant changes were observed between C/C and E/E xenografts at the 200-day time point (Supplemental Figure 1). There is a possibility that we did not observe any changes due to the low number of technical replicates (n = 3) and that more animals are needed for future evaluation. There was also variability within the treatment groups, which could be responsible for the nonsignificant changes. It was particularly interesting though that no changes were found, as it addressed that there are differences in the response of our rodent hosts (ie, rat versus mouse) to estrogen treatment. Our earlier study evaluating these gene expression changes were also performed in whole tissue using a small number of xenografts (n = 3-5), which provide evidence that the low technical replicates may not be the primary issue. Although this does beg the question of whether the dose given to the rat hosts was adequate enough to produce a similar response as the one seen in the mouse hosts, or whether it is the hosts’ hormonal environment in addition to our treatment that led to these expression changes. Additionally, mice were employed in our previous studies because they were able to handle the treatment better with a limited immunological reaction. On the other hand, our immunodeficient rat hosts (ie, T-cell null, but had B cells) had an immune response that made it difficult to determine what was caused from the normal aging process, and what was caused from rejection of the graft or estrogen treatment. These factors are critical in understanding the pathology of human prostatic disease using a xenograft model, and issues such as dosage of estrogen, immune competence of our hosts, and using castrated hosts (for better hormonal control) will be addressed.
Paired Estrogen Exposures (E/E&T) Produce Persistent Estrogenic Effects in Human Xenografts
To further demonstrate that estrogen exposure has long-lasting effects on the human prostate xenografts, we stained 200- and 400-day xenografts for CK10, p63, and ERα (Figure 5). CK10, a marker of squamous metaplasia, 41 was present in 200 days and persisted in the ductal compartment at 400 days postimplantation. This finding verifies an estrogen-related resilient cellular change in the ductal tissue by forming more resilient squamous epithelium. Basal cell hyperplasia, indicated by p63 staining, 42 was also seen in ducts at 400 days. The increase in basal cells may be a compensatory mechanism for ductal tissue recovery from estrogen-induced damage or a consequence of the aging process in the human prostate tissue. Estrogen receptor α was seen throughout the prostate at 200 days but was localized largely to the epithelial cells by day 400. Estrogen receptor α is thought to function as a promoter of prostate carcinogenesis, 43 although this is still under investigation. The spatial transition in ERα expression observed between 200 and 400 days may be important, and the use of human tissue is instrumental in further understanding the role of ERα in the human PCa development.
Conclusion
Here we provide evidence for the hypothesis that early exposure to estrogenic chemicals can render the prostate vulnerable to subsequent exposures and may shift normal developmental processes and produce persistent hyperplasia that could affect subsequent prostate growth. This human fetal prostate xenograft model of early-life hormonal effects could serve as a screening tool for potential endocrine disrupting chemicals, thereby providing mechanistic insight that may lead to new treatments for men having prostate disease.
Footnotes
Acknowledgments
The authors would like to sincerely thank the donors who contributed to this study. We would like to acknowledge Paula Weston and Melinda Golde for their great work in processing human fetal prostate tissue sections.
Author Contribution
C.M. Saffarini contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. E.V. McDonnell-Clark contributed to acquisition, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Amin contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. K. Boekelheide contributed to conception and design, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr Boekelheide is an occasional expert consultant for chemical and pharmaceutical companies, and he owns stock in CytoSolv, an early-stage biotechnology company developing a wound-healing therapeutic based on growth factors. These activities are unrelated to the current work but are mentioned in the spirit of full disclosure.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by U.S. Environmental Protection Agency RD-83459401, National Institute of Environmental Health Sciences 5P20ES018169-02, and Training grant in Environmental Pathology T32-ES007272.
