Abstract
The toxicity of parenterally administered vitamin E isomers, delta-tocotrienol (DT3) and gamma-tocotrienol (GT3), was evaluated in male and female CD2F1 mice. In an acute toxicity study, a single dose of DT3 or GT3 was administered subcutaneously in a dose range of 200 to 800 mg/kg. A mild to moderately severe dermatitis was observed clinically and microscopically in animals at the injection site at doses above 200 mg/kg. The severity of the reaction was reduced when the drug concentration was lowered. Neither drug produced detectable toxic effects in any other tissue at the doses tested. Based on histopathological analysis for both DT3 and GT3, and macroscopic observations of inflammation at the injection site, a dose of 300 mg/kg was selected as the lowest toxic dose in a 30-day toxicity study performed in male mice. At this dose, a mild skin irritation occurred at the injection site that recovered completely by the end of the experimental period. At a dose of 300 mg/kg of DT3 or GT3, no adverse effects were observed in any tissues or organs.
Introduction
With the ever-present threat of a nuclear event, first responders must be prepared for and protect against radiological incidents. Many compounds have been screened as potential candidates. 1 -6 Tocopherols and tocotrienols belong to a group of vitamin E isomers that play a pivotal role as essential, fat-soluble nutrients and function as antioxidants in the human body. Eight compounds belong to this group of isomers which include alpha, beta, gamma, and delta tocopherols and tocotrienols. They all contain a chromanol ring with a 15-carbon tail that differs in its saturation between isoforms; the tocopherol tail is saturated while the tocotrienol tail is unsaturated. The unsaturated chain contains conjugated C-C double bonds that allow it to cross membranes with relative ease 7,8 and contribute to its high bioavailability compared to tocopherols. The strong antioxidant properties of tocotrienols, namely delta- and gamma-tocotrienol, also contribute to their function as tumor suppressors for a multitude of tumor cells including breast, 9 -13 prostate, 14,15 and colon. 16 Exposure to low to moderate linear energy transfer ionizing radiation induces many harmful cellular changes, including the formation of reactive oxygen species. 17 Therefore, natural antioxidants, such as Vitamin E, have been considered as potential radioprotectants due to their strong antioxidant capabilities, 18,19 ease of absorption, and minimal toxicity. 20
It has been demonstrated that delta-tocotrienol (DT3, Figure 1A) is an effective radioprotectant. 19,21 When administered 24 hours prior to radiation exposure, a single subcutaneous (sc) injection of DT3 (150 or 300 mg/kg) yielded a dose reduction factor (DRF) of 1.19 or 1.27, respectively, in male CD2F1 mouse model. 19 Additionally, when a single dose of DT3 (150 mg/kg) was injected in male CD2F1 mice 2 hours after radiation exposure, the DRF was 1.1. Pharmacokinetics of DT3 was evaluated previously in CD2F1 mice. 21 Authors determined the plasma concentration of a single dose of DT3 (300 mg/kg) administered sc in CD2F1 mice. The peak plasma concentration (Cmax) of DT3 was 195 mmol/L 1 hour after injection (Tmax). The plasma half-life (t1/2) was 1.8 hours, and DT3 was cleared from plasma within 12 hours after administration. 21
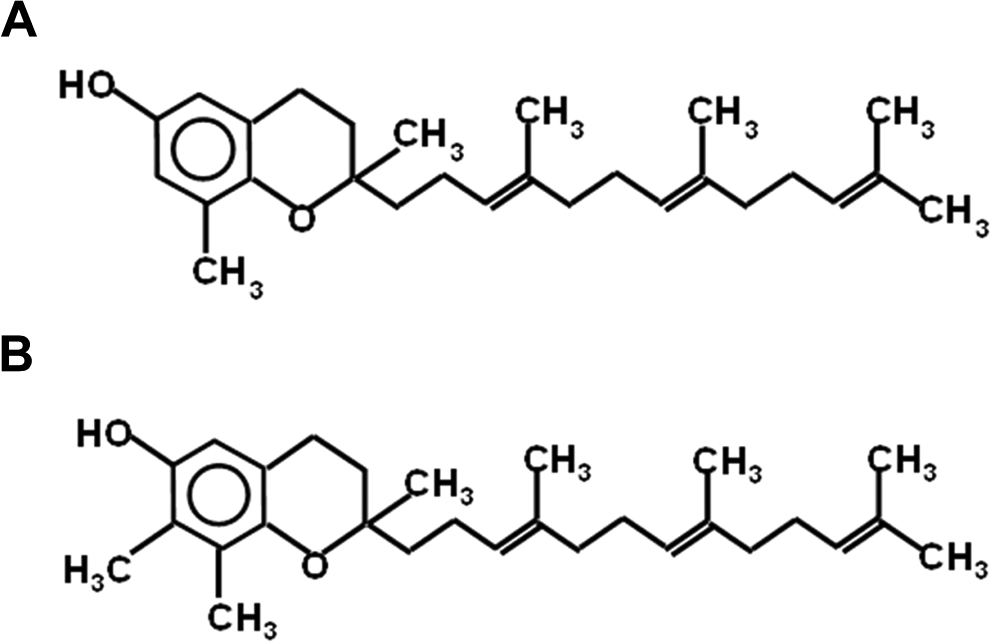
Chemical structure of (A) delta-tocotrienol (DT3) and (B) gamma-tocotrienol (GT3).
Gamma-tocotrienol (GT3, Figure 1B) also exhibits potent radioprotective activity following whole-body γ-radiation exposure in a mouse model. It was previously demonstrated that GT3 provided higher radioprotection than alpha-tocopherol. 18,22 We have shown that a single dose of 200 mg/kg of GT3 injected sc 24 hours before radiation exposure protected 100% of CD2F1 mice from radiation-induced hematopoietic syndrome, reduced neutropenia and thrombocytopenia, and protected bone marrow stem cells and progenitor cells. 18,23
Although DT3 and GT3 (a single dose administered sc 24 h before radiation) have been developed as promising radiation countermeasures in a mouse model, it has not been determined whether there are any adverse systemic effects at their effective doses, and the lowest toxic dose remains unknown. The purpose of this study was to first determine the lowest toxic doses of DT3 and GT3, using 14- and 30-day preliminary toxicity tests performed in both male and female CD2F1 mice injected sc with a single dose of either drug 24 and then further perform a more comprehensive acute toxicity study with the selected lowest toxic dose of DT3 and GT3 to investigate any effects on systemic toxicity.
Materials and Methods
Animals and Husbandry
Pathogen-free male and female CD2F1 mice (9-10-week-old) were purchased from Harlan Laboratories (Indianapolis, Indiana) and housed (8 per cage) in an air-conditioned facility at the Armed Forces Radiobiology Research Institute (AFRRI), which is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. 18 Pathogen-free mice were held in quarantine for 2 weeks and then housed separately by gender for the duration of the study. The rooms were maintained at a temperature of 21°C ± 2°C with a relative humidity of 50% ± 10%, ventilation frequency of 10 to 15 cycles/h, and a 12-hour light cycle. Mice were provided ad libitum access to certified rodent rations (Harlan Teklad Rodent Diet; Harlan Teklad, Wisconsin) and acidified water (with HCl, pH 2.5–3.0) to control opportunistic infections.
All animal procedures were performed in accordance with a protocol approved by the AFRRI’s Institutional Animal Care and Use Committee (IACUC). Research was conducted using the principles and procedures outlined in the National Research Council’s Guide for the Care and Use of Laboratory Animals. 25
Drug Formulation and Administration
Samples from both the drugs and the vehicle were sent to Charles Rivers Laboratories (Charleston, South Carolina) for endotoxin testing prior to initiating the experiments. All samples yielded an endotoxin maximum of less than 0.50 EU, which is acceptable for drug testing in mice according to IACUC standards.
Both DT3 and GT3 (Figure 1) were formulated in 5% Tween-80/95% saline (Yasoo Health Inc, Johnson City, Tennessee) and injected sc at 800, 400, or 200 mg/kg body weight in a volume of 0.1 to 0.2 mL. The vehicle was administered sc as equivalent dose volume of 5% Tween-80 (0.1-0.2 mL) in saline.
Dose Determination Study
Male and female CD2F1 mice were randomly assigned to groups by body weight (26 ± 2 g for males; 22 ± 2 g for females) with an ear punch for identification (n = 4/group). The experimental groups included saline control, vehicle control (VEH, 5% Tween-80), DT3 (800, 400, and 200 mg/kg), and GT3 (800, 400, or 200 mg/kg; Table 1). A single dose of either saline or vehicle or DT3 or GT3 was administered sc to groups of mice and animals and was monitored over 14- and 30-day end points. It was previously determined that the optimum doses for protection against radiation-induced lethality for DT3 and GT3 were 200 and 300 mg/kg, respectively. 18,19 We escalated the starting dose to 800 mg/kg based on previously published methods in which drug doses were escalated to several fold higher than the safe dose to assess their toxicity. 26,27 Animals from each group were euthanized at 14 or 30 days postinjection in order to perform histopathological analysis for drug-related toxicity. The doses of both drugs were then reduced sequentially to determine a dose with no overt acute toxicity. The criteria used to determine acute toxicity were weight loss, injection site reactions, altered behavior or activity level, or any physical abnormalities.
Experimental Overview: Dose, Duration, Number of Animals Per Group, and End Points Examined for Each Study.
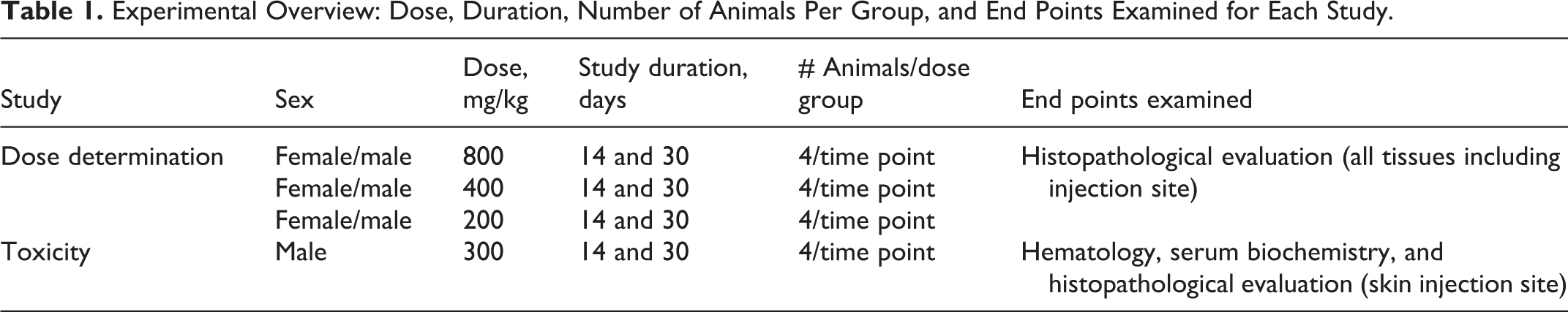
The first cohort of mice received a single sc injection of 800 mg/kg between the scapulae and was observed daily for symptoms related to acute toxicity. Although no systemic signs of toxicity were observed, focal hair loss with irritation was observed at the injection site by 5 days postinjection. Therefore, the next lower dose (400 mg/kg) was administered sc to the subsequent cohort. 24,26 Similar focal hair loss at the injection site of the animals in the 400 mg/kg dose group was observed within 5 days of receiving the drug. They were observed daily for the remainder of the study to determine whether the skin reaction would resolve. The next dose tested was 200 mg/kg. This dose did not result in a severe skin reaction at the injection site during the experimental period.
Gross clinical observation of the skin lesions were performed daily after the drug was administered (when the lesions occurred) and lasted for the duration of the study. Clinical signs began with erythema and hemorrhage, followed by alopecia, edema, erosion, deep excoriation, scaling, and dryness of the skin. Photographic images at the skin inflammation site (nape of the neck) were captured on days 14 and 30 from randomly selected animals (males and females) treated with 200 or 400 mg/kg of either DT3 or GT3 (figure not shown). Severity scoring of the inflammation was performed based on clinical observations in this study.
Gross Pathology and Histopathology
On day 14, a blinded histopathological analysis was performed on all major tissues for signs of toxicity. The tissues analyzed included adrenal glands, aorta (thoracic), bone and bone marrow (sternum), cerebrum, cerebellum, brain stem, pituitary gland, eyes, optic nerves, nasal cavity, larynx, trachea, lungs, heart, tongue, salivary gland, thymus, thyroid gland, stomach, esophagus, duodenum, pancreas, jejunum, ileum, cecum, colon, rectum, liver, gallbladder, spleen, kidneys, haired skin (ventral abdomen and injection site, lymph nodes (mesenteric and submandibular), mammary gland, skeletal muscle, sciatic nerve, and urinary bladder. 27 In addition, the testes, epididymides, seminal vesicles, and prostate glands were collected from male mice, whereas the ovaries, oviduct, uterus, and vagina were harvested from female mice. All tissue samples were fixed in 10% buffered formalin, embedded in paraffin, then sectioned (5-6 µm), and stained with hematoxylin and eosin for routine histopathological processing by a board-certified veterinary pathologist A severity grade was determined based on the board-certified veterinary pathologist’s semiquantitative assessment of the degree and type of inflammation, necrosis, extent of the lesion, ulceration and adjunct changes to the epidermis, loss or changes to the adnexae, and underlying rhabdomyocytes. The grading scheme consisted of ordinal categories as follows: 0 = within normal limits, 1 = minimal (minor changes that involve <10% of region of interest [ROI]), 2 = mild (noticeable change in the tissue involving 10%-20% of the ROI with a 10%-20% loss in tissue volume), 3 = moderate (a prominent feature that affects almost half of the tissue with a 20%-40% decrease in tissue volume), 4 = marked (the lesion is an overwhelming feature that affects most of the ROI with a 40%-100% decrease in tissue volume), and 5= severe. 28 A representative image for the upper limit (severe) of the injection site inflammation in the animals (treated with 800 mg/kg GT3) is shown in Figure 2. Images were captured with an Olympus DP70 camera (Olympus, Melville, New York) and imported into Adobe Photoshop (Version CS4, Adobe Systems, San Jose, California) for analysis.
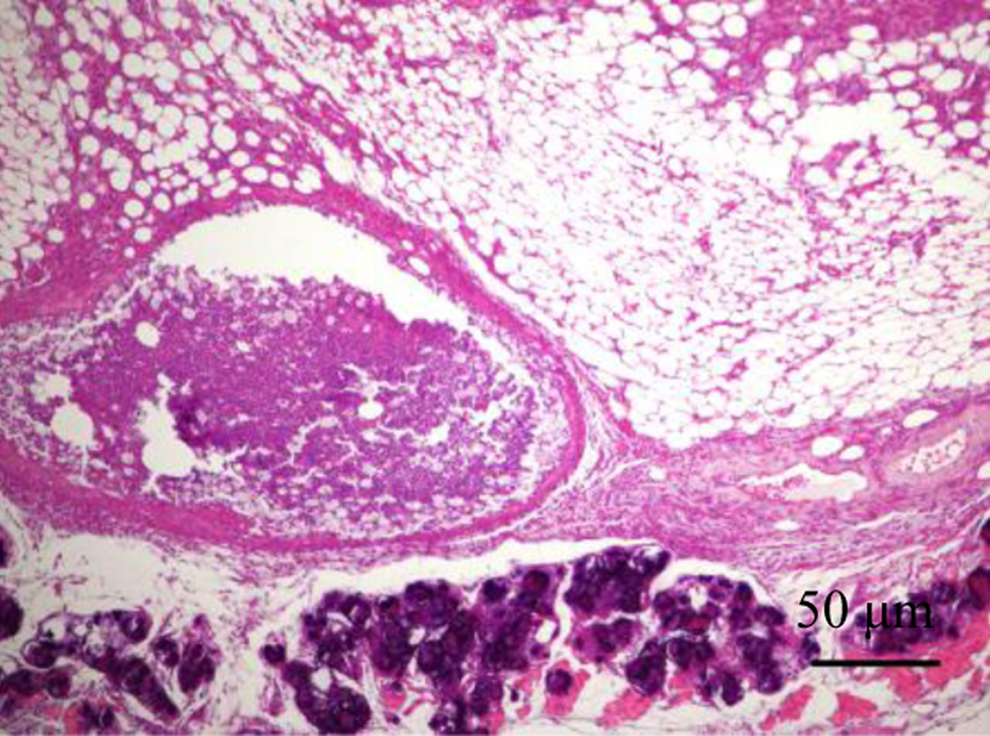
Hematoxylin and eosin-stained mouse skin section (800 mg/kg, GT3, day 14 postinjection) with severe inflammation at injection site: This photomicrograph is representative of the upper limits of the histopathological severity noted in this study. In this instance, there is a sterile abscess replacing the subcuticular tissues and the underlying rhabdomyocytes are atrophic, degenerate, lost, or are undergoing mineralization. Scale = 50 μm.
Acute Toxicity Study
General study design
Male CD2F1 mice were assigned to 1 of the 4 groups at the start of the experiment, saline, vehicle (VEH, 5% Tween-80 in saline), DT3 (300 mg/kg), and GT3 (300 mg/kg; Table 2). Mice were monitored for 30 days after the single sc injection of the respective drug for signs of delayed toxicity that included weight loss, injection site reactions, changes in behavior or activity level, or any physical abnormalities. At days 14 and 30 after drug administration, mice (n = 7-8 mice/group/time point) were humanely euthanized, and blood was collected for hematology and serum chemistry analyses while major tissues including injection site area were harvested for histopathological analyses. The grading system for skin inflammation scoring was the same as described in the previous experiment.
Skin Reaction Scoring: Drug, Dose, Sex, Percentage of Affected Per Group and Severity Score.
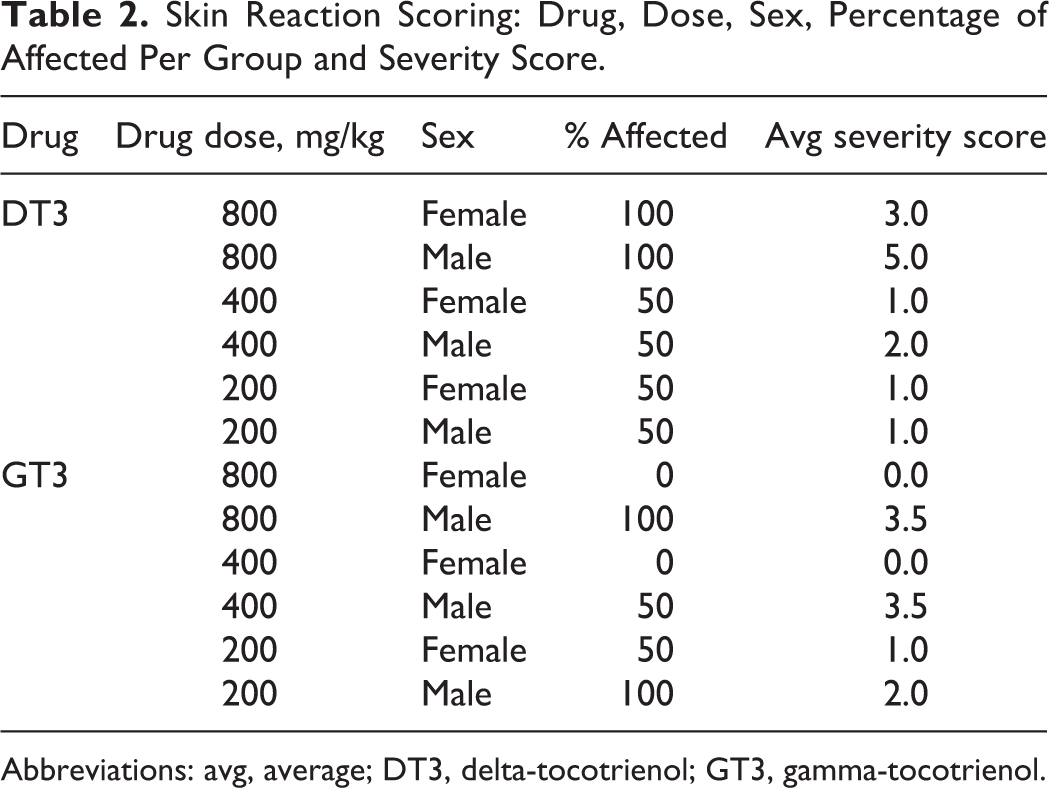
Abbreviations: avg, average; DT3, delta-tocotrienol; GT3, gamma-tocotrienol.
Hematology and serum chemistry
Blood (0.6-1.0 mL) was collected from the inferior vena cava as a terminal procedure at days 14 and 30 from deeply anesthetized animals (Isofluorane; Abbott Laboratories, Chicago, Illinois). For the hematological analysis, blood was transferred immediately into EDTA-containing blood collection tubes and mixed gently in a rotary shaker. Analysis included cell counts for red blood cells (RBCs), white blood cells (WBCs), platelets (PLTs), hemoglobin, hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), basophils, eosinophils, leukocytes, lymphocytes (LYMPHs), monocytes (MONOs), and neutrophils (NEUs) using an Advia 120-cell counter (Bayer Corporation, Tarrytown, New York). A serum chemistry analysis was performed on whole-blood samples that were collected in serum separator tubes (BD Microtainer; BD Biosciences, San Jose, California) that was allowed to clot for 30 minutes at room temperature. After centrifugation at 1500g for 10 minutes, serum was collected and stored at −80°C until assayed for alanine aminotransferase (ALT), albumin, alkaline phosphatase, aspartate aminotransferase (AST), blood urea nitrogen (BUN), and creatinine.
Statistical Analysis
Means ± standard error of the means (SEMs) were calculated for hematological and clinical chemistry data. One-way analysis of variance was performed for each time point with a Tukey test for post hoc analysis when appropriate (SPSS, Version 20, Somers, New York).
Results
Dose Determination Study
We performed a 14- and 30-day toxicity study (n = 4/group/time point) with 3 doses (800, 400, and 200 mg/kg) to determine the lowest toxic doses of DT3 and GT3. A single dose of either DT3 or GT3 or vehicle or saline was administered, and the animals were observed over either 14 or 30 days. No clinical signs or symptoms of toxicity or behavioral changes were observed until day 5 postinjection (DT3 or GT3, 800 mg/kg) when a skin reaction in the dorsum between the shoulder blades was observed at the injection site for both drugs in all mice. Gross clinical observation of the skin lesions included erythema and hemorrhage, followed by alopecia, edema, erosion, deep excoriation, scaling, and dryness. No topical ointment was used to reduce skin inflammation due to its potential interference in the evaluation of the toxicity of the drugs. After skin toxicity was observed at 800 mg/kg, the next lower dose of drug (DT3 or GT3, 400 mg/kg) was administered, and mice were monitored. An injection site reaction was observed in these animals as well. Therefore, the dose of drug (DT3 or GT3) was reduced to 200 mg/kg for the subsequent group of animals. Although severity increased with increasing dose, skin inflammation was greatly reduced by day 30 in mice injected with 400 and 200 mg/kg. No inflammation was observed in mice injected with either vehicle or saline.
A histopathological evaluation of all tissues was performed on day 14 to screen for any microscopic evidence that either drug exerted toxic effects. Animals treated with the 800 and 400 mg/kg of either DT3 or GT3 developed a severe necroulcerative and mineralizing dermatitis at the injection site that extended into the underlying subcuticular and muscular layers as shown in Figure 2. Occasionally, the inflammation separated and surrounded atrophic, degenerate, and necrotic rhabdomyocytes. In some cases, this resulted in sterile abscess formation with eventual epidermal rupture and the creation of draining tracts. Following the administration of the 800 mg/kg dose of DT3, all of the males exhibited a more severe injection site reaction than the females with an average severity score of 5.0 in males versus 3.0 in the females (Table 2). The same dose of GT3 resulted in a similar sex-specific reaction with an average severity score of 3.5 for the males and 0 for the females. At 400 mg/kg, DT3 yielded an average skin reaction that was scored at 2.0 for the males and 1.0 in females, while the similar dose of GT3 caused a reaction that was scored as an average of 3.5 for the males with no reaction in females (Table 2). The 200 mg/kg dose of either DT3 or GT3 did demonstrate necrotizing subcuticular dermatitis; however, the rhabdomyocytic loss was observed to be minimal to moderate (images not shown). The scores for this dose group (200 mg/kg) were 1.0 for both males and females following administration of DT3. Mice that received GT3 had average scores of 1.0 for the females and 3.0 for the males (Table 2). It should be noted that the collection of tissues can alter these scores markedly, as tissue adjacent to a point of ulceration might be ranked as mild or moderate while those directly harvested from the ulcer would be scored as severe. No bacteria were found in the abscesses that had not ulcerated, and special bacterial stains, including Brown and Brenn and Brown and Hopps, were used on the abscesses and adjacent tissue to assess the presence or absence of bacteria.
Background lesions were observed with relative frequency but were determined to be unassociated with the drug treatments in this study. These lesions included epicardial mineralization with an associated granulomatous epicarditis and rare hepatocellular glycogenosis. 29 Several mice (n = 3/group) in the higher drug dose group (800 mg/kg, DT3 and GT3) had enlarged spleens compared to the vehicle-treated control groups. The spleen is not only a site for direct and indirect toxicity but it is also one of the key organs responsible for extramedullary hematopoiesis. 30 However, there was no evidence of abnormal splenic pathology outside robust extramedullary hematopoiesis, suggesting the enlargement was related to the increased hematopoiesis. 18,23,31,32 No treatment-related abnormalities were detected in any other tissues based on histopathological assessment. Histopathological evaluation was not extended to day 30 because we did not observe any toxicity in any tissues on day 14. Based on microscopic and macroscopic evaluation of the injection site inflammation on day 14 and day 30, a dose of 300 mg/kg was extrapolated as the lowest toxic dose for both DT3 and GT3, and this dose was selected for a more comprehensive acute toxicity study.
Acute Toxicity Study With 300 mg/kg of DT3 and GT3
We performed a 14- and 30-day acute toxicity study with 300 mg/kg of DT3 and GT3 (the lowest toxic dose for both the drugs as extrapolated from the previous study). A single dose of either DT3 or GT3 (300 mg/kg) was administered sc and body weight change, histopathological analysis of tissues, hematological, and serum chemistry parameters were examined. No significant changes in body weight were observed over the 30-day experimental period (Table 3). At day 14 post-injection, a skin reaction developed excoriation subsequent to scratching with mild erythema and alopecia after the administration of each drug. Subcutaneous injection with DT3 and GT3 yielded a skin reaction in 71% of animals with an average severity score of 3.4 (n = 7) and 3.8 (n = 7), respectively. These reactions were resolved by day 30 for both drugs, as determined by both clinical observation and histopathological analysis. A significant reduction in necrotizing subcuticular dermatitis was observed on day 30 compared to day 14 with mild rhabdomyocytic loss in the drug-treated animals (Figure 3A-C). No injection site reaction or skin inflammation was observed either in saline- or in vehicle (5% Tween-80 in saline)-treated mice. Since we did not find any toxicity in the major tissues and organs including bone marrow sternum, liver, heart, kidney, and spleen from the drug (DT3 or GT3)-treated groups (200, 400, and 800 mg/kg), histopathological analysis of the other tissue samples except the skin inflammation site from the 300 mg/kg groups was not performed.
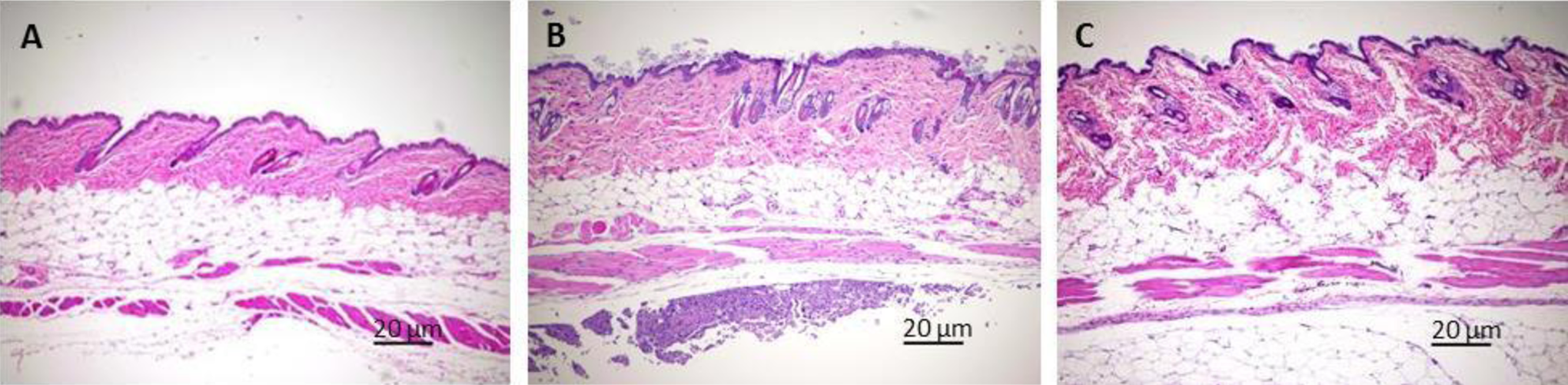
Representative microscopic image for the histopathological analysis of the skin inflammation site reaction that occurred in mice treated with 300 mg/kg GT3: a skin reaction developed excoriation subsequent to scratching with mild erythema and alopecia after the administration of each drug (DT3 or GT3), no inflammation was observed either in saline- or in vehicle (5% Tween-80 in saline)-treated mice. Skin histopathology of representative images of hematoxylin and eosin (H&E)-stained skin injection site inflammation in male CD2F1 mice is shown. Control (saline-treated mice on day 14) with normal skin (A). Severity of the inflammation is significantly reduced from 14 days (B) to 30 days (C) after administration the drug as demonstrated by the reduction in necrotizing subcuticular dermatitis on day 30 compared to day 14 with mild rhabdomyocytic loss in these group of animals. Scale = 20 μm.
Body Weights for Male CD2F1 Mice Following the Administration (sc) of Delta-Tocotrienol (DT3, 300 mg/kg) and Gamma-Tocotrienol (GT3, 300 mg/kg).a
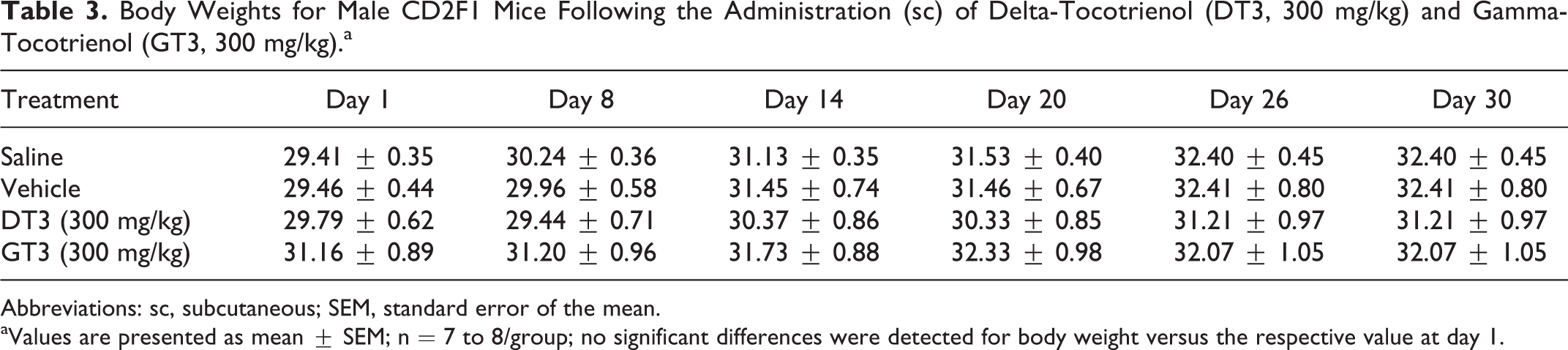
Abbreviations: sc, subcutaneous; SEM, standard error of the mean.
aValues are presented as mean ± SEM; n = 7 to 8/group; no significant differences were detected for body weight versus the respective value at day 1.
Table 4 summarizes results of the hematological analyses. On day 14, significant alterations to serum hematology following the administration of DT3 were detected. Values for WBC (+76%), PLT (+49%), LYMP (+37%), NEU (+252%), and MONO (+89%) increased, while MCV (−2%), MCH (−7%), and MCHC (−5%) decreased for DT3- versus the saline-treated group. By day 30, WBC had returned to normal levels. However, levels of PLT were elevated (+10%), while levels of MONO (−31%) and MCV (−2%) were depressed significantly in peripheral blood of the DT3- versus the saline-treated group. There was a similar response following the administration of GT3. On day 14, there was an increase in RBC (+5%), WBC (+52%), PLT (+23%), LYMP (+47%), NEU (+91%), and HCT (+5%) and while levels of MCHC (−4%) decreased for GT3- versus the saline-treated group. By day 30, RBC and WBC had returned normalized, but levels of PLT (+5%), MCHC (+3%), LYMPH (−12%), MONO (−44%), NEU (−23%), HCT (−6%), and MCV (−2%) were still perturbed in peripheral blood of GT3- versus the saline-treated group.
Hematological Parameters at Days 14 and 30 for Male CD2F1 Mice That Were Injected (sc) Once With Delta-Tocotrienol (DT3, 300 mg/kg) or Gamma-tocotrienol (GT3, 300 mg/g).a
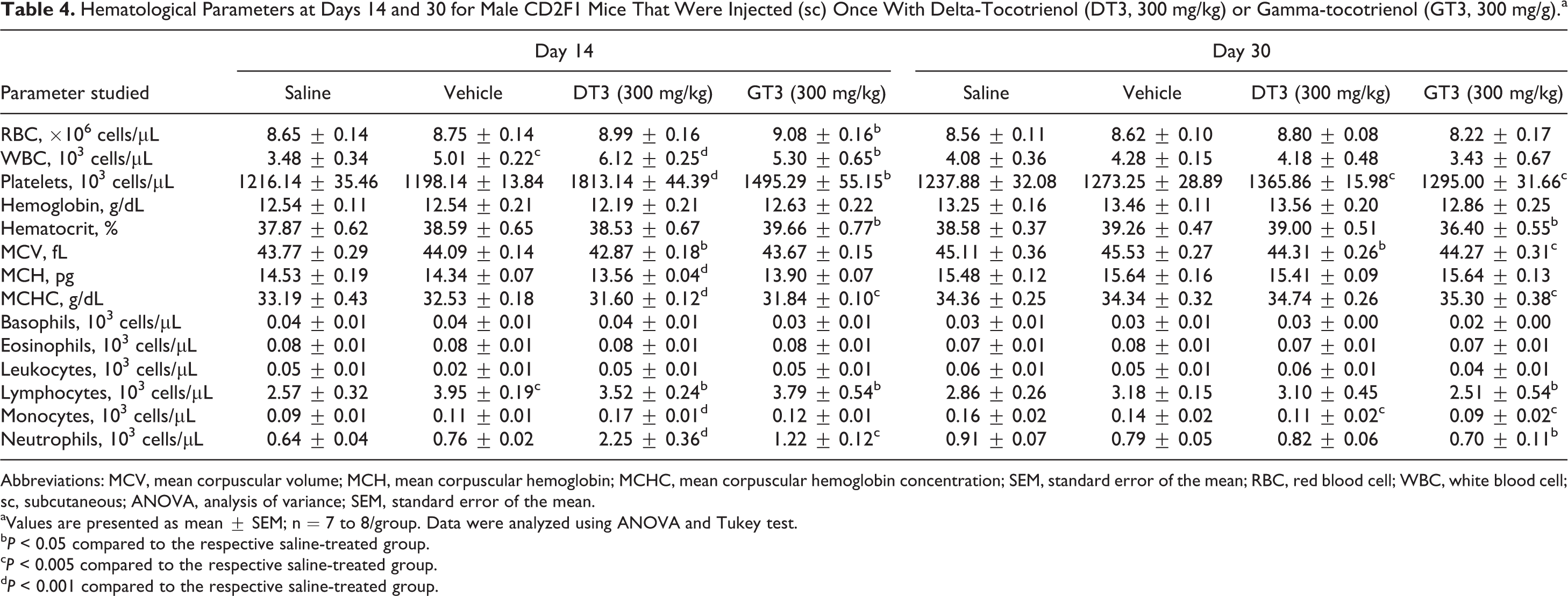
Abbreviations: MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; SEM, standard error of the mean; RBC, red blood cell; WBC, white blood cell; sc, subcutaneous; ANOVA, analysis of variance; SEM, standard error of the mean.
aValues are presented as mean ± SEM; n = 7 to 8/group. Data were analyzed using ANOVA and Tukey test.
b P < 0.05 compared to the respective saline-treated group.
c P < 0.005 compared to the respective saline-treated group.
d P < 0.001 compared to the respective saline-treated group.
Table 5 summarizes the serum biochemistry data. There was an increase at day 14 in ALT levels for both = VEH (+64%) and DT3 (+18%) versus the saline-treated group, but this effect was no longer detectable by day 30. At day 14, ALP levels were decreased for DT3-treated mice versus VEH (−12%); however, this effect was no longer present at day 30. There was a significant increase in ALT levels for the GT3-treated mice (+62%) versus the saline-treated group on day 14 that was detected through day 30 (+31%). There were no significant changes in serum levels of BUN and creatinine in the GT3- and DT3-treated groups compared to saline-treated or vehicle-treated groups. Serum levels of AST, which is a common marker for liver health, also remained unchanged in all groups.
Clinical Chemistry Results at Days 14 and 30 Posttreatment for Male CD2F1 Mice That Were Injected (sc) Once With Delta-Tocotrienol (DT3, 300 mg/kg) or Gamma-Tocotrienol (GT3, 300 mg/kg).a
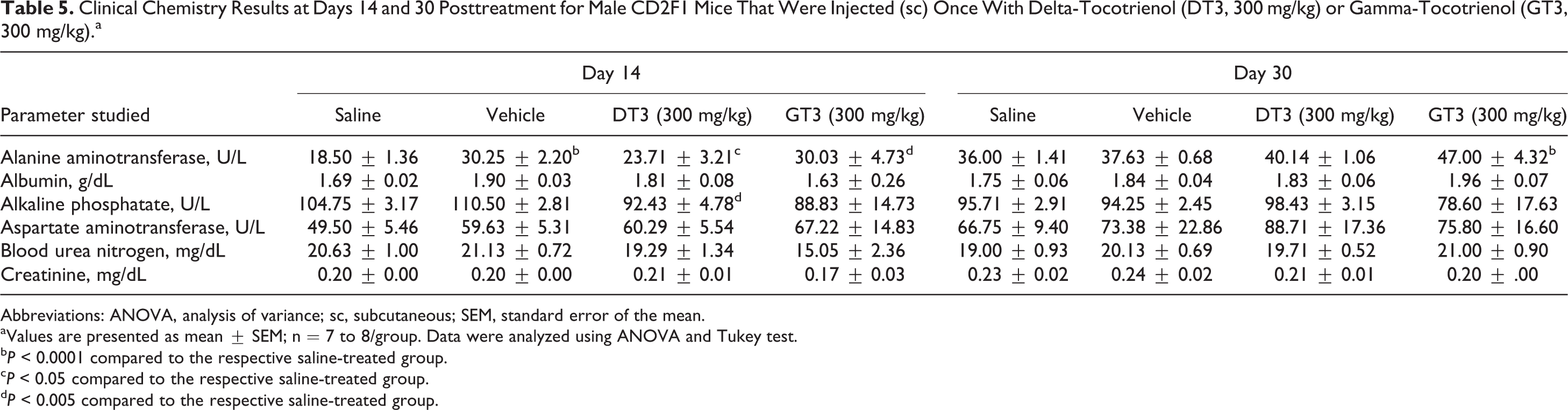
Abbreviations: ANOVA, analysis of variance; sc, subcutaneous; SEM, standard error of the mean.
aValues are presented as mean ± SEM; n = 7 to 8/group. Data were analyzed using ANOVA and Tukey test.
b P < 0.0001 compared to the respective saline-treated group.
c P < 0.05 compared to the respective saline-treated group.
d P < 0.005 compared to the respective saline-treated group.
Discussion
Although vitamin E isomers, DT3 and GT3, have been studied extensively in vitro and in vivo, 7,33,34 the acute toxicity has not been determined when the drug was administered sc. Here, we determined the lowest sc toxic dose of DT3 and GT3 in a 30-day acute toxicity study. Both drugs are under investigation as potential radiation countermeasures. In the present study, male and female CD2F1 mice were injected with a single 200 to 800 mg/kg dose (sc) of DT3 and GT3, and skin inflammation was examined over 30 days following previously established criteria. These criteria were based on the dermal response of acrylic acid in 3 strains of mice that were evaluated by microscopic histopathological observation. 35 In our preliminary preclinical toxicity study of the vitamin E isomers DT3 and GT3, no systemic toxicity was observed with any dose (200-800 mg/kg) of the drugs. Skin inflammation was observed starting at day 5 after drug administration that began with erythema and hemorrhage, followed by edema and erosion and deep excoriation in some cases. However, the severity of the clinical symptoms was reduced when a lower concentration of the drug was administered. Both male and female CD2F1 mice that were administered a single dose of 300 mg/kg of GT3 and DT3 demonstrated a full recovery of the mild skin inflammation over 30 days.
Serum levels of liver enzymes ALT, and AST are commonly assayed biomarkers indicative of hepatotoxicity. 36 Alanine aminotransferase is largely liver-specific enzyme, but AST is present in liver as well as skeletal muscle and erythrocytes. Neither DT3 nor GT3 produced detectable systemic toxicity in the organs, including the liver, from any treatment group or drug dose (200-800 mg/kg). Although relatively higher levels of ALT were detected in the vehicle group and both drug-treated groups (300 mg/kg) versus the saline-treated group on day 14, there were no histopathological changes in the liver of either vehicle- or drug-treated groups (200-800 mg/kg) compared to the saline control. It is interesting to note that ALT levels did not change in the drug-treated groups compared to vehicle control group on day 14. Similarly, the significantly higher ALT levels in the GT3 (300 mg/kg)-treated mice on day 30 compared to saline control were not related to hepatotoxicity as examined by a histopathological analysis of liver. Moreover, since no changes in serum levels of BUN and creatinine were detected, and there were no histopathological renal changes in the DT3- and GT3-treated mice compared to vehicle or saline controls, it indicates that renal health was preserved in the presence of the drugs. In a 13-week oral toxicity study in rats, a mixed tocopherol–tocotrienol preparation in diet resulted in higher ALT and BUN levels in blood with slight hepatocellular hypertrophy as observed by histopathological examination of liver. 33 Also, there was a significant decrease in PLT levels in the drug-treated animals after 13 weeks, an indication of the toxic effect of the drug as reported by the authors. 33 However, we did not observe any abnormality in the PLT levels in blood or histopathological analysis of any organs and tissues except injection site skin inflammation.
A semiquantitative histopathological evaluation of tissue effects was performed to provide an evaluation of potential toxic effects of the varying compounds as evidenced by the skin lesion that developed at the injection sites after drug administration. It was reported previously that animal studies either overpredicted or underpredicted the potential for human skin reaction with a particular drug 37 and therefore it is important to note that skin inflammation at the injection site should not necessarily be considered a definitive representation of the expected response of this drug in human clinical investigations. There were no microscopically evident toxic effects observed in any tissue outside the injection site lesions (200-800 mg/kg dose range). Therefore, the lowest toxic dose was determined to be 300 mg/kg based on the severity of the skin inflammation. Although a mild skin irritation was observed in several animals treated with 300 mg/kg within 5 days of drug administration, the lesion resolved completely by the completion of the 30-day experimental period.
Although the administration of these drugs resulted in a dose-dependent skin irritation at the injection site, the dose selected as the lowest sc toxic dose did not induce long-term inflammation. Additionally, there was no evidence of systemic toxicity with either of the drugs at any of the doses studied. These drugs have already been demonstrated to be potent radiation protective agents. 18,19,22,23,38 It is therefore important that further studies be conducted to improve the sc formulation of these tocotrienols to reduce injection site reactions.
Footnotes
Acknowledgments
The authors thank Dr Mark Whitnall and CAPT John Gilstad for their help in this study. The authors also thank Kevin Hieber and Dr David Bolduc for technical help.
Authors’ Note
The opinions contained herein are the private views of the authors and are not necessarily those of the Armed Forces Radiobiology Research Institute, the Uniformed Services of the University of the Health Sciences, or the Department of Defense.
Author Contributions
S. Ghosh contributed to conception and design and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. S. Swift contributed to analysis and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. R. Pessu contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. K. Chakraborty contributed to design, acquisition and analysis, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. V. Villa contributed to design, acquisition, analysis, and interpretation, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. E. Lombardini contributed to conception, acquisition, analysis, and interpretation, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by AFRRI Intramural research funding (RAA610).
