Abstract
Probiotics are living microorganisms that, when given in sufficient quantities, boost the host’s health. Probiotics have been used more often in recent years as dietary supplement to prevent illnesses, including diarrhea, obesity, and constipation. The probiotic strain Bacillus subtilis BSP110 is being developed as a dietary supplement. In this work, we assessed the in vivo safety of the probiotic strain Bacillus subtilis BSP110 in rats as per OECD-423 and 407 guidelines. Female rats were given a single oral (gavage) dosage of 2000 mg/kg (4 × 1011 CFU/g) to assess the maximum tolerated dose (MTD). This was followed by a 28-day sub-acute toxicity study in which Bacillus subtilis BSP110 was given orally (gavage) once daily to male and female rats at dose levels of 250, 500, and 1000 mg/kg/day. No effects of Bacillus subtilis BSP110 were noted at any of the dosage levels given over the course of 28 days. Between the control and test groups, there was no discernible difference in body weight, clinical symptoms, urinalysis, hematological tests, clinical biochemistry, gross pathology, or histopathological evaluation. Additionally, in vitro cytotoxicity was evaluated using the MTT assay with the VERO cell line, and Bacillus subtilis BSP110 was found to be non-cytotoxic at 4 × 1011 CFU/kg. The single dose MTD was ≥2000 mg/kg, and the NOAEL was 1000 mg/kg/day.
Introduction
Probiotics are non-pathogenic microorganisms that, when given in sufficient amounts, may aid the host’s health. 1 Probiotic-based fermented foods have become popular again since the early 2000s, mostly because of health claims. 2 In addition to disorders like chronic diarrhea, allergies, dermatitis, and bacterial and viral infections, all of which are strongly linked to an unhealthy lifestyle, fermented foods have also been linked to the prevention of obesity, lactose intolerance, irritable bowel syndrome, and gastroenteritis.3-5 There are, however, both documented and anticipated side effects from taking probiotics. Bacillus strains such as Bacillus cereus have been reported to cause outbreaks.6-8 Concise and practical suggestions on how to move toward the safe and efficient use of probiotics are necessary in light of the introduction of new probiotic strains and products, as well as the expansion of probiotic usage into populations at risk. 9 Probiotic safety is a topic addressed in various regulatory guidance documents and initiatives, including the FAO/WHO, EFSA (European Food Safety Authority), IDF (International Dairy Federation) bulletins, US FDA (US Food and Drug Administration), Health Canada probiotic monograph, and IPA (International Probiotics Association) guidelines. 10 These documents focus on ensuring the safety of probiotics, safe use in food, approaches to generally recognized as safe (GRAS) food, and dietary supplements. 10 The Indian Council of Medical Research and Department of Biotechnology (ICMR DBT) and International Scientific Association for Probiotics and Prebiotics (ISAPP) have set guidelines for evaluating probiotic strains in food. 11 These include genus, species, strain identification, in vitro tests, in vivo safety studies in animal models, and in vivo efficiency studies. Researchers have studied the safety assessment of various probiotic strains using in vitro and in vivo approaches.11-13
Despite being a fascinating category of probiotic bacteria, the Bacillus species have not gotten much attention. This group of microbes has been consistently investigated and has shown significant potential as probiotics. Even though Bacillus has advantages, it appears necessary to assess the safety of possible probiotic strains. Bacillus cereus and Bacillus anthracis have been linked to foodborne diseases and toxin production, although Bacillus subtilis and other species are generally thought to be harmless. 14 In this study, single dose and 28-day toxicity studies were used to evaluate the potential toxic effects. Morbidity, mortality, change in hematological parameters, serum and urine biochemistry, and histopathological evaluation of rats once daily exposure to the Bacillus subtilis BSP110 for 28 consecutive days were evaluated as per OECD-407 guideline. 15
Material and Methods
Materials
The Bacillus subtilis BSP110 (MTCC 25471) test organism is a probiotic being developed as a dietary supplement. This investigation employed spray-dried B. subtilis BSP110 powder, which was produced at Pellucid Lifesciences PVT Ltd’s fermentation facility and had a concentration of 4 × 1011 CFU/g. Several medical diagnostic kits were purchased from Lab Care Diagnostics in India, including those for glucose, cholesterol, triglycerides, total protein, albumin, alkaline phosphatase (ALP), bilirubin, creatinine, chloride, potassium, sodium, serum glutamic pyruvic transaminase (SGPT), serum glutamic oxaloacetic transaminase (SGOT), and albumin.
Experimental Animals
Wistar rats, weighing between 200 and 300 g each, were selected as the appropriate surrogate based on anatomical and similar microbiota to humans. 10 The rats were kept in the Institute of Pharmacy’s animal house at Nirma University. The animals were housed on a 12-hour light/dark cycle with a temperature of 22 ± 5°C and a humidity of 55 ± 5%. For 7 days, rats were acclimated to their surroundings. Following the guidelines of the Committee for Control and Supervision of Experiments on Animals (CCSEA), Ministry of Fisheries, Animal Husbandry and Dairying, Government of India, New Delhi, the study protocol was approved by the Institute of Pharmacy’s IAEC (Institutional Animal Ethics Committee) under protocol number IP/PCOL/FAC/35/2023/27. The cages were replaced 3 times a week, and corn cobs were used as bedding. The males and females were kept apart, housed in groups, and provided with water and an ad libitum supply of a conventional rodent pellet diet (Keval Sales Corporation, Baroda, India).
Single-Dose (Acute) Toxicity Study
To assess the possible toxic effects of Bacillus subtilis BSP110, a single-dose oral toxicity study followed by a 14-day observation period was carried out on female rats in accordance with the OECD-423 guideline. Twelve female Wistar rats weighing between 200 and 300 g were randomly assigned to one of the two groups: a vehicle control group or a test group (n = 6) for the single-dose toxicity test. The rats were fasted overnight before the probiotic was given. Using a graduated syringe, B. subtilis BSP110 was given orally (by gavage) to the test group at a dosage of 2000 mg/kg, having a concentration of 4 × 1011 CFU/g (suspension of probiotic having a concentration of 250 mg/mL was prepared in distilled water and administered (8 mL/kg) according to the animal’s body weight). Distilled water was provided to the vehicle control group. On the day of dosing, clinical observations were made at various intervals of time (0.25, 0.5, 1, 2, 3, 4, and 8 h post-dosing). From Day 2 until the study’s conclusion (Day 15), the animals were observed twice a day for clinical signs and death. Changes in the animals’ hair coat, skin, eyes, mucous membranes, respiratory system, circulation, behavior patterns, central and autonomic nervous systems, and spontaneous motor activity were evaluated. Particular symptoms including sleep, convulsions, tremors, coma, lethargy, diarrhea, and salivation were the main emphasis. On Days 0, 8, and 15, the animals’ body weights were recorded. The animals were euthanized on Day 15 and underwent a selected necropsy. All macroscopic abnormalities were noted at the conclusion of the research period.
28-Day Repeat Dose Oral Toxicity Study
In accordance with the OECD 407 guideline, a 28-day oral toxicity study in rats was designed and carried out to determine the potential toxicity and no-observed-adverse-effect level (NOAEL) of Bacillus subtilis BSP110. This was achieved by giving Wistar rats oral (by gavage) B. subtilis BSP110 once daily for a period of 28 consecutive days.
Six groups of 10 Wistar rats (5 males and 5 females, 8 to 10 weeks old) each were randomly selected from a total of 60 Wistar rats weighing between 200 and 300 g. Groups 1 through 6 were assigned labels. Rats were given different dosages of a test probiotic as part of the investigation. Groups 2 through 4 were administered varying amounts of the test substance, which is 250, 500, and 1000 mg/kg/day, respectively (1 mL/kg, 2 mL/kg, and 4 mL/kg for low, medium, and high doses, respectively), whereas Group 1 was given distilled water, the vehicle control. 1000 mg/kg/day of B. subtilis BSP110 and distilled water were given to Groups 5 and 6, respectively. Following a 28-day dosing period, the recovery groups (Groups 5 and 6) were monitored for an extra 2 weeks to see if any toxic consequences were evident. This was to ascertain if the occurrence of any toxic effects was reversible, delayed, or chronic. The rats constantly had access to water and a regular pellet meal.
Clinical Observations and Body Weights
Rats were examined at the same time every day, with an emphasis on when the expected effects would peak following dosage administration. For 2 weeks, the rats were observed every day for indications of disease or death. Once a week, observations were made of the skin, eyes, fur, mucous membranes, frequency of secretions and excretions, and autonomic activity, which included lacrimation, piloerection, pupil size, and irregular breathing patterns. Stereotypes like excessive grooming or repetitive circling, tonic or clonic movements, odd behaviors like self-mutation or reverse walking, and adjustments to posture, stride, and responsiveness in response to handling were also seen. On Days 7, 14, 21, and 28 of therapy, body weight was taken once every 7 days.
Hematological Examinations
Following the research, after the last dose administration, 3 animals from each group were fasted overnight and were anesthetized, and K2-EDTA tubes were used to draw blood through retro-orbital plexus for hematological analysis. The following parameters were measured during this examination: White blood cell (WBC) count, lymphocyte percentage (Lym %), monocyte percentage (Mon %), granulocyte percentage (GR %), red blood cell (RBC) count, hemoglobin (Hb), platelet count, hematocrit (HCT), mean corpuscular volume (MCV), mean cell hemoglobin (MCH), mean cell corpuscular hemoglobin concentration (MCHC), platelet distribution width (PDW), mean platelet volume (MPV), red cell distribution width (RDW), activating partial thromboplastin clotting time (APTT), and prothrombin time (PT). The Nihon Kohden Celltac Alpha Hemato analyzers were used to test the hematological parameters.
Clinical Biochemistry
The rats did not receive food for the night after the research was over. The serum was obtained for biochemical examination at the end of the study; blood was obtained via the retro-orbital method. As directed by the manufacturer, tests were performed on the separated serum to measure levels of several parameters, including glucose (GLULSR 500), triglycerides (TGLSLR 25), cholesterol (CHOLSLR 125), SGOT (GOTSLR 125), SGPT (GPTSLR 125), total protein (TP 200), bilirubin (TDBIL 100), albumin (ALB 200), alkaline phosphatase (ALPSLR 50), urea (LURE 200), creatinine (CRE 200), sodium (SODM 50), chloride (CHLOM 50), and potassium (KM 50).
Urine Analysis
Urine was collected at the end of the study by putting animals in metabolic cages. Specific gravity, pH, total protein, glucose, creatinine, and albumin levels were analyzed using a semi-automated biochemical analyzer.
Gross Necropsy, Organ Weights, and Histopathology
Three animals (both male and female) from Groups 1 and 4 were sacrificed on Day 29, while Groups 5 and 6 were sacrificed on Day 43. Selected organs (kidneys, liver, heart, lungs, and colon) were dissected for gross necroscopy and examined for macroscopic morphology. Following the fixation of the tissue slices with 10% formalin, the samples were dehydrated using alcohol. The dehydrated samples were stained with hematoxylin and eosin after embedding in paraffin wax. After that, the H&E slides were examined using an Olympus CX21 I LED research microscope equipped with a Magcam DC3 lens for taking pictures using Image View software.
In Vitro Cell Viability Assay
The Vero cell line was procured from the National Centre for Cell Science (NCCS), Pune, India. A normal monkey epithelial Vero cell line was used to evaluate the cell viability. 16 The cells were grown in DMEM media with 10% FBS and 1% antibiotics. After the cells reached 80 to 90% confluency, they were detached and harvested using trypsin EDTA. 10 × 103 cells were seeded in each well of a 96-well plate. The plate was incubated in a 37°C incubator for 24 h. The cells were added with different concentrations of Bacillus subtilis BSP110, except for the control group. After 24 h of incubation, the cell growth media were then removed and washed with 200 μL phosphate-buffered saline. MTT was added to every well and incubated for 4 h at 37°C. For each well, 50 μL of dimethyl sulfoxide (DMSO) was added. The formazan absorbance was read using an ELISA plate reader with a wavelength of 550 nm.
Statistical Analysis
Body weight, hematological parameters, serum, and urine parameters were analyzed, and the results were expressed using the mean and standard deviation. The treatment (low, mid, and high dosage) groups were compared to the control group, and the recovery high-dose group was compared to the recovery normal control group using the One-way ANOVA and Two-way ANOVA. If the P-value was less than .05, statistical significance was determined. For every statistical analysis, GraphPad Prism software version 5.0 was used. Mean ± SD is used to represent the data.
Results
Effect of Bacillus subtilis BSP110 After a Single Dose
There were no abnormal clinical signs following a single administration of Bacillus subtilis BSP110 at a dose of 2000 mg/kg and a concentration of 4 × 1011 CFU/g. The body weight of the rats was measured on Days 1, 8, and 15. In the control group, the mean body weight was 221.33 ± 9.07 g on Day 1, 225.83 ± 8.33 g on Day 8, and 231.17 ± 6.94 g on Day 15. In the B. subtilis BSP110 group, the mean body weight was 221.00 ± 10.08 g on Day 1, 222.67 ± 10.16 g on Day 8, and 228.33 ± 9.27 g on Day 15. According to these findings, body weight has been steadily rising (Figure 1). From the day of dosage until the study’s conclusion, no anomalies in the rats’ clinical symptoms were noticed. There were no obvious pathological symptoms associated with the therapy. Effect of B. subtilis BSP110 on body weight of rats during acute toxicity studies. Statistical analysis was carried out using Two-way analysis of variance (ANOVA) followed by Bonferroni post-tests to compare multiple means ± SD.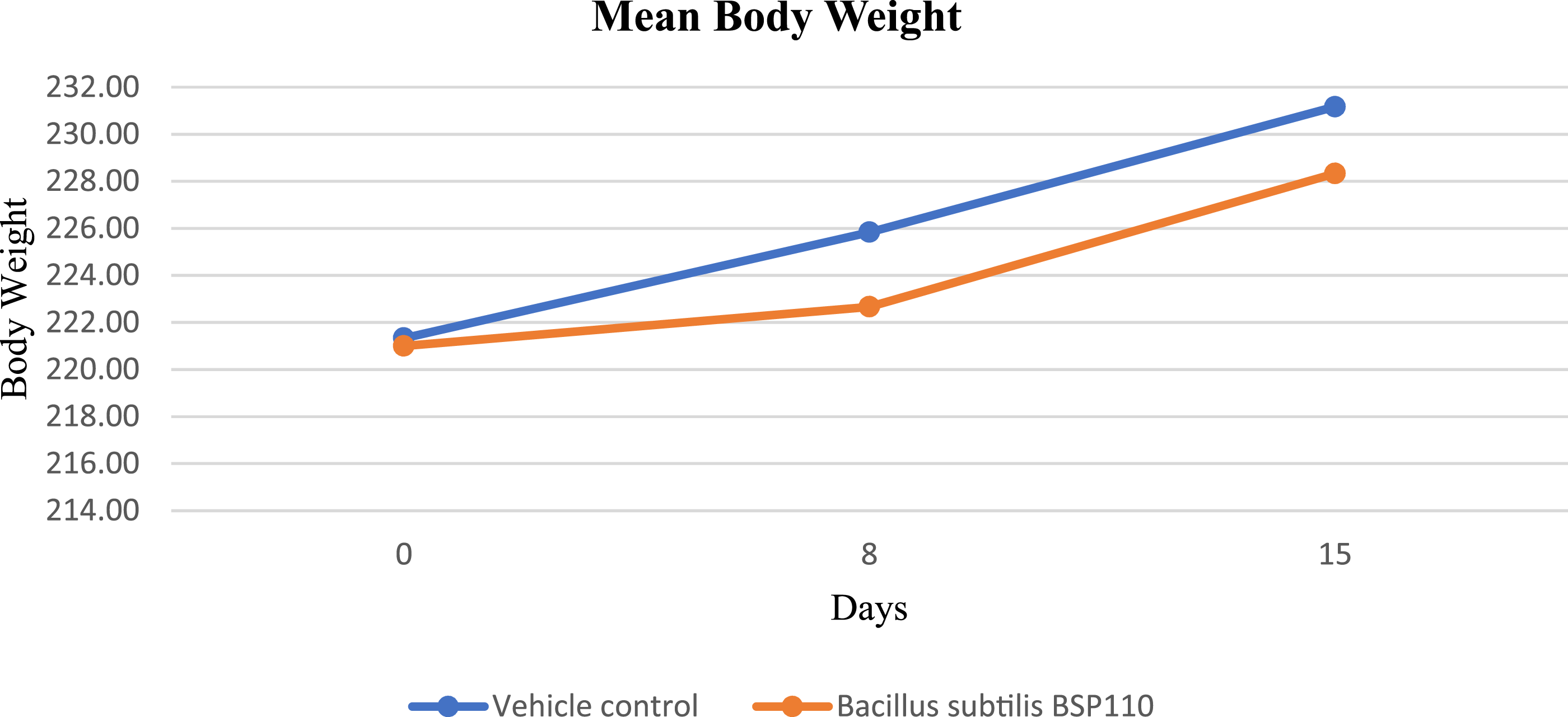
Effect of Bacillus subtilis BSP110 on 28-Day Oral Toxicity
After once daily oral (gavage) administration of Bacillus subtilis BSP110 for 28 days at a dose of up to 1000 mg/kg/day, no toxic effects were reported. No mortality was observed during the study or in the recovery groups.
Clinical Observations
Physical characteristics such as fur color, skin, eyes, lacrimation, mucosal membrane, piloerection, pupil, and breathing pattern did not alter. Following the administration of B. subtilis BSP110, no changes in handling behavior, posture, or gait were seen. There have also been no reports of other behavioral consequences, including self-mutilation, repeated circling, walking backwards, stereotyped behavior, and excessive grooming.
Body Weight
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Body Weight of Control (NC), Treated, and Recovery (R) Group Male Rats (n = 5/Group) During the 28-Day Repeated Dose Oral Toxicity Study.
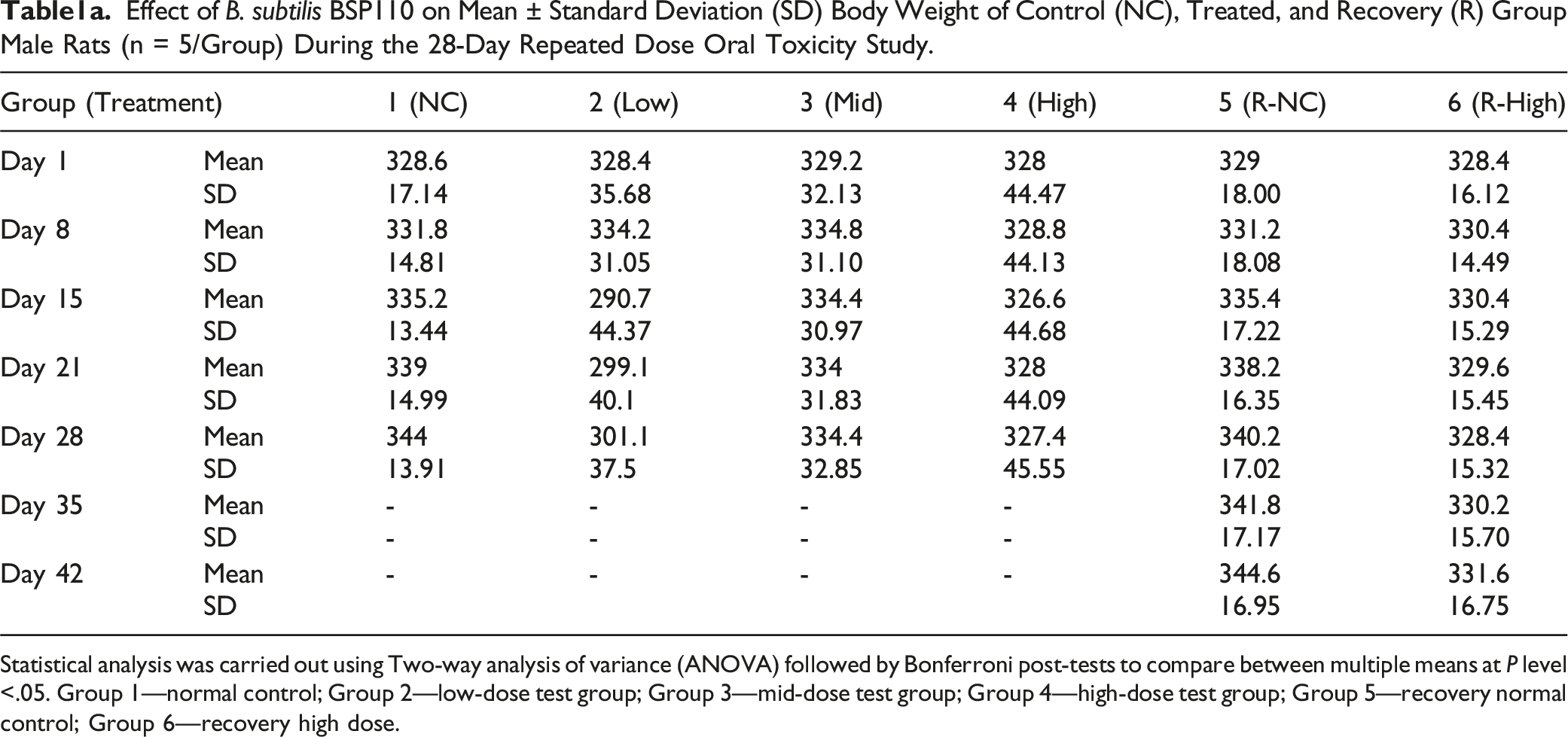
Statistical analysis was carried out using Two-way analysis of variance (ANOVA) followed by Bonferroni post-tests to compare between multiple means at P level <.05. Group 1—normal control; Group 2—low-dose test group; Group 3—mid-dose test group; Group 4—high-dose test group; Group 5—recovery normal control; Group 6—recovery high dose.
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Body Weight of Control (NC), Treated, and Recovery (R) Group Female Rats (n = 5/Group) During the 28-Day Repeated Dose Oral Toxicity Study.
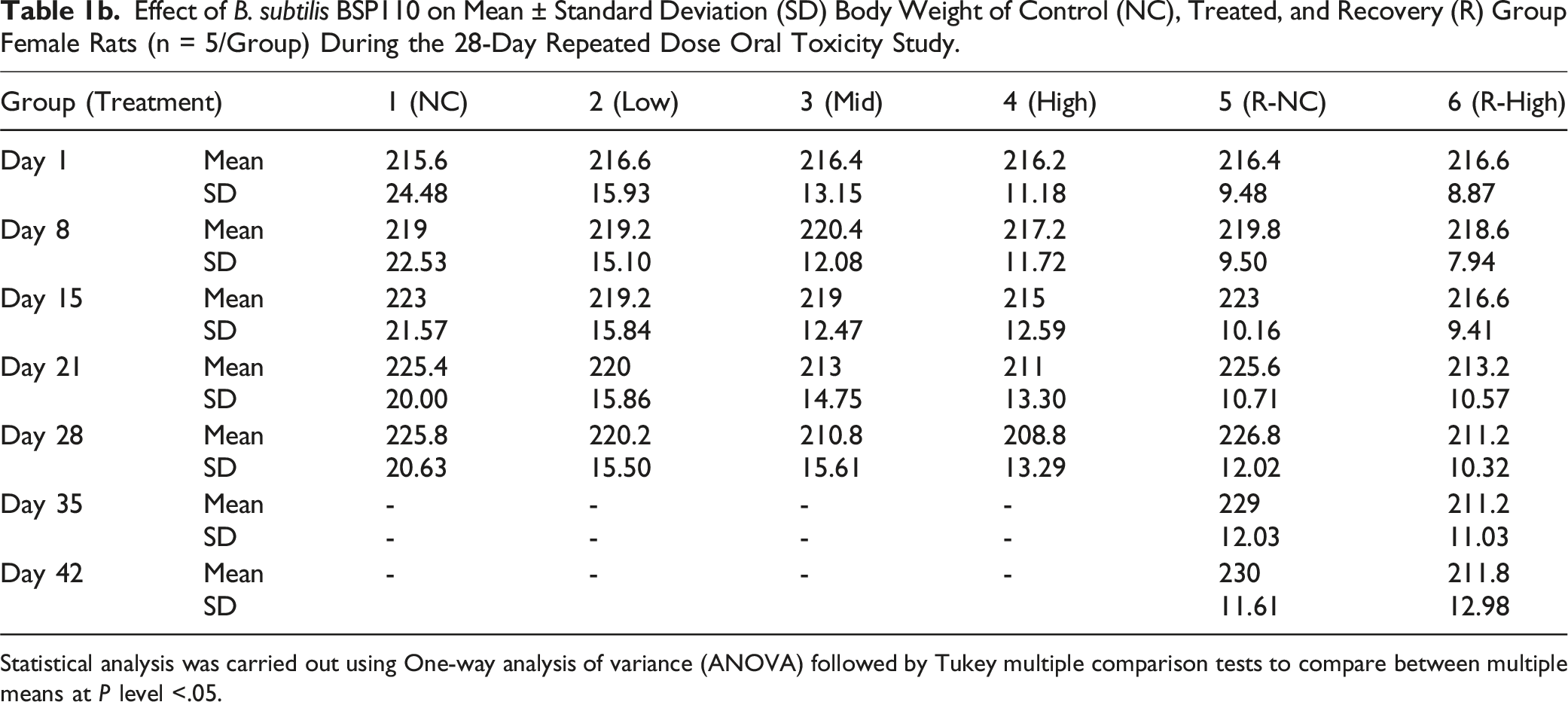
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Hematological Examination
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Hematological Parameters of Control (NC), Treated, and Recovery (R) Group Male Rats (n = 3/Group) During 28-Day Repeat Dose Oral Toxicity Study.
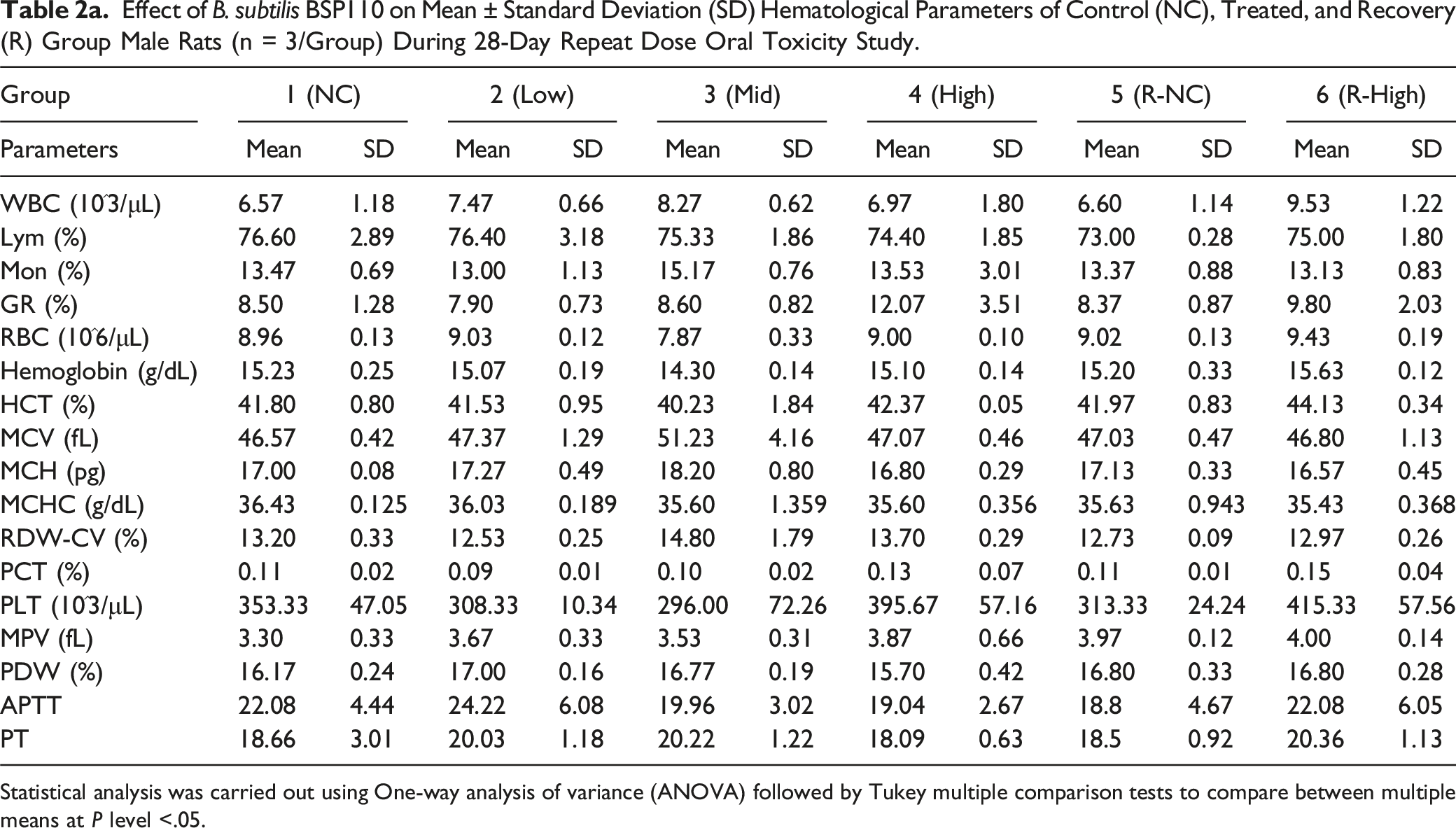
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Hematological Parameters of Control (NC), Treated, and Recovery (R) Group Female Rats (n = 3/Group) During 28-Day Repeat Dose Oral Toxicity Study.
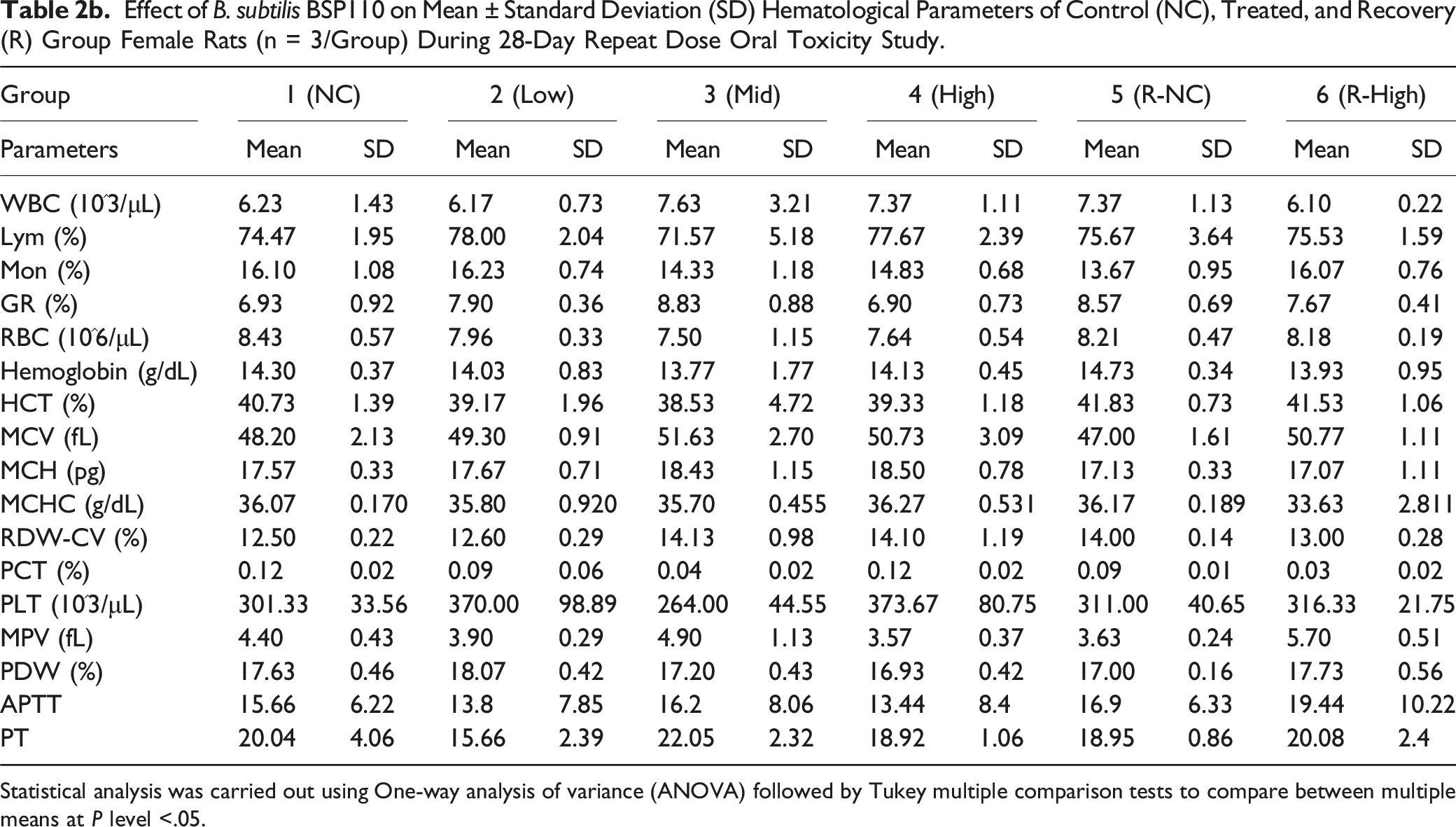
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Biochemical Examination
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Biochemical Parameters of Control (NC), Treated, and Recovery (R) Group Male Rats (n = 5/Group) During 28-Day Repeat Dose Oral Toxicity Study.
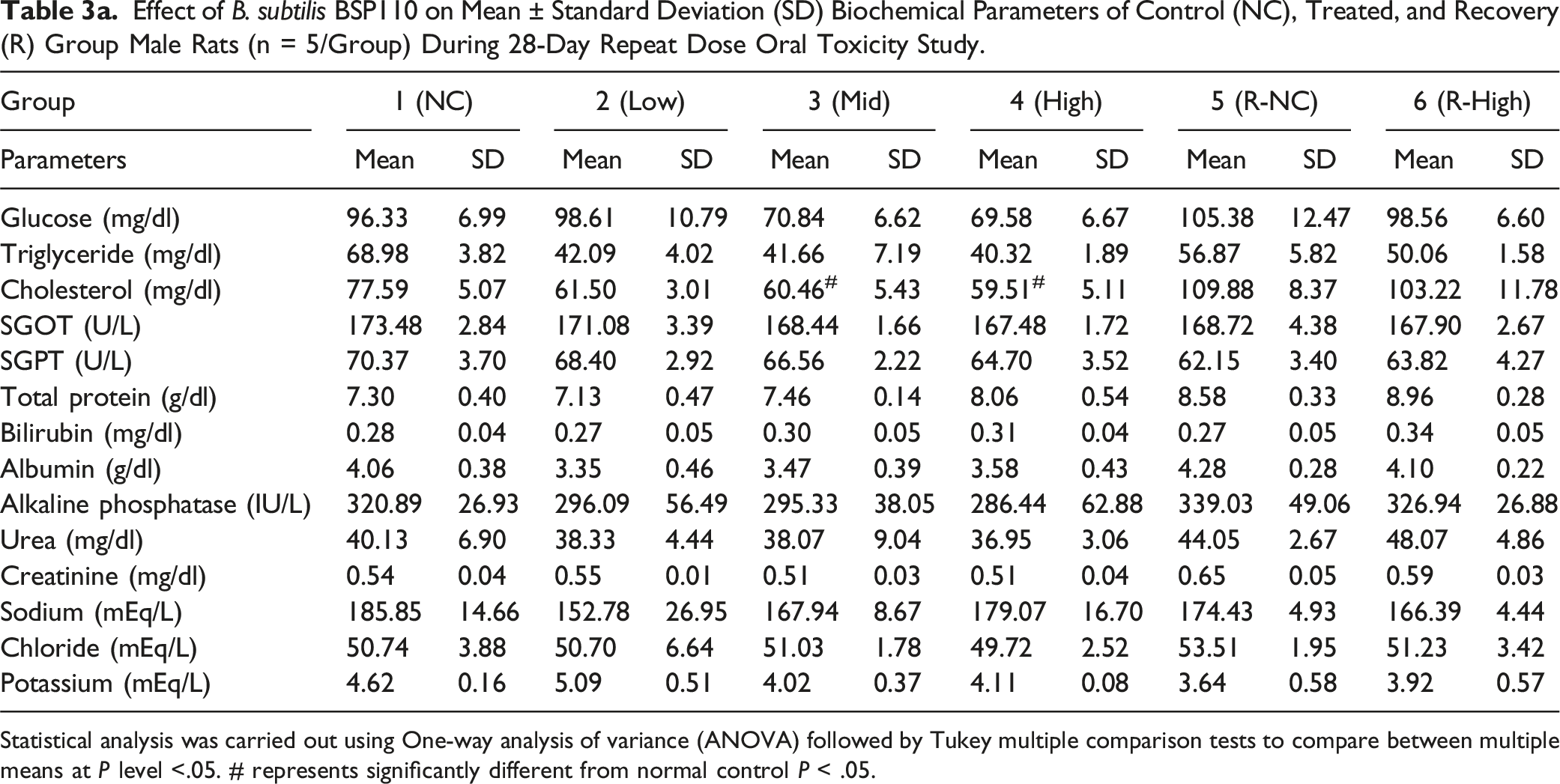
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05. # represents significantly different from normal control P < .05.
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Biochemical Parameters of Control (NC), Treated, and Recovery (R) Group Female Rats (n = 5/Group) During 28-Day Repeat Dose Oral Toxicity Study.
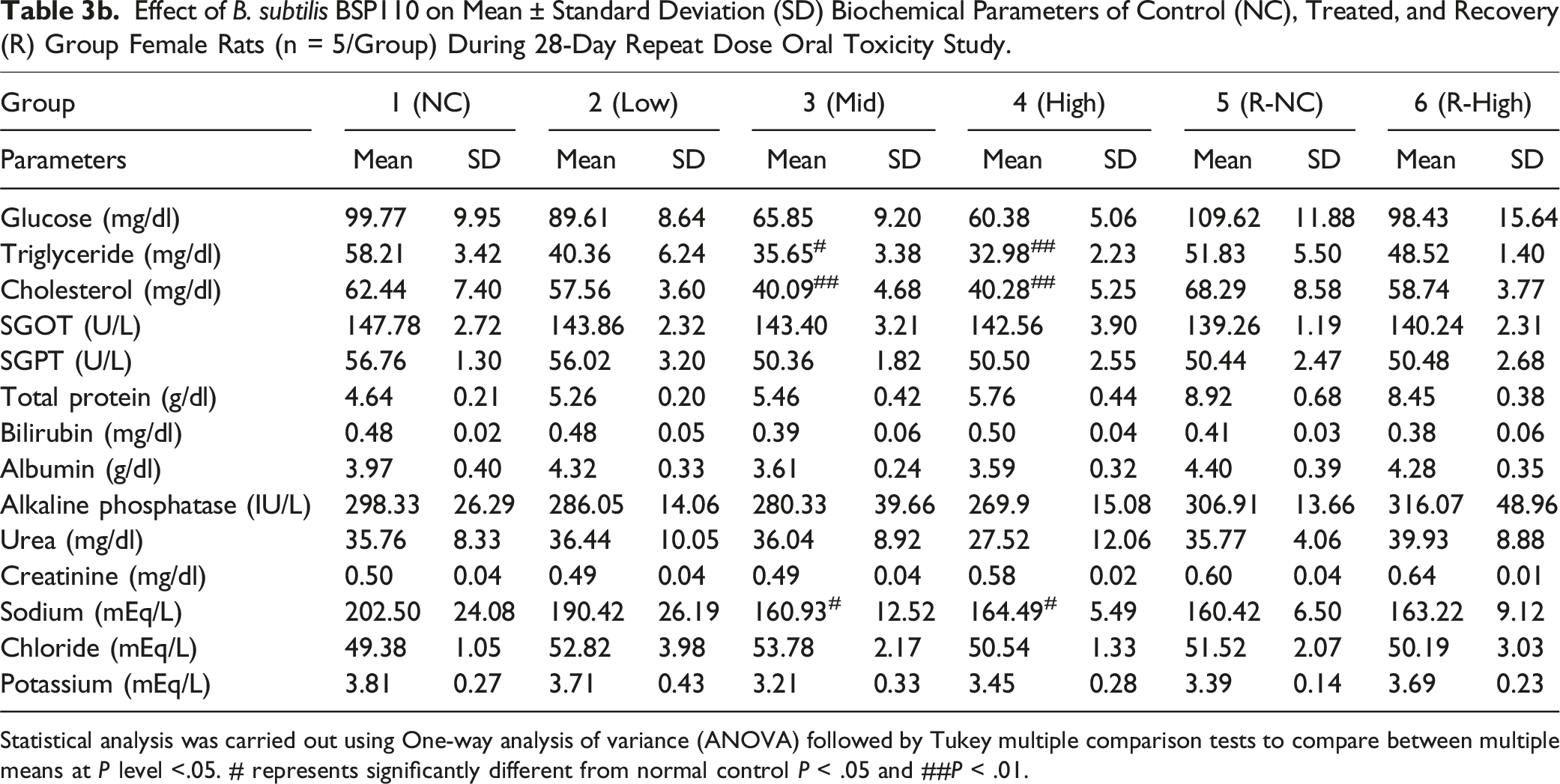
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05. # represents significantly different from normal control P < .05 and ##P < .01.
Urine Analysis
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Urine Analysis of Control (NC), Treated, and Recovery (R) Group Male Rats (n = 5/Group) During 28-Day Repeat Dose Oral Toxicity Study.
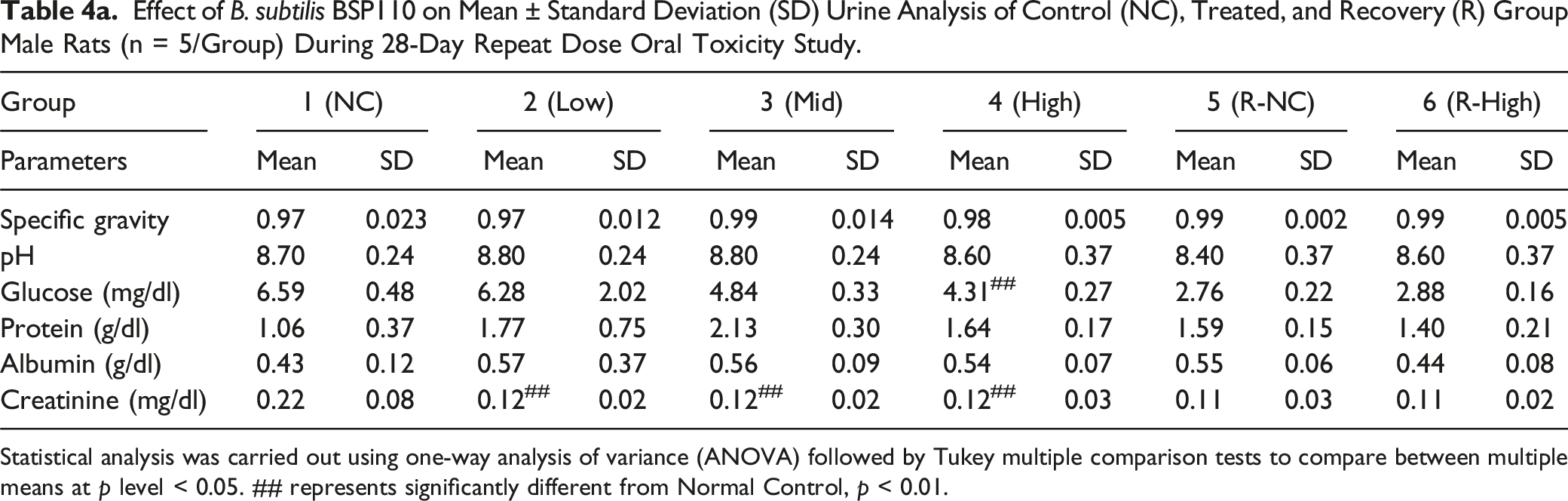
Statistical analysis was carried out using one-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at p level < 0.05. ## represents significantly different from Normal Control, p < 0.01.
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Urine Analysis of Control (NC), Treated, and Recovery (R) Group Female Rats (n = 5/Group) During 28-Day Repeat Dose Oral Toxicity Study.
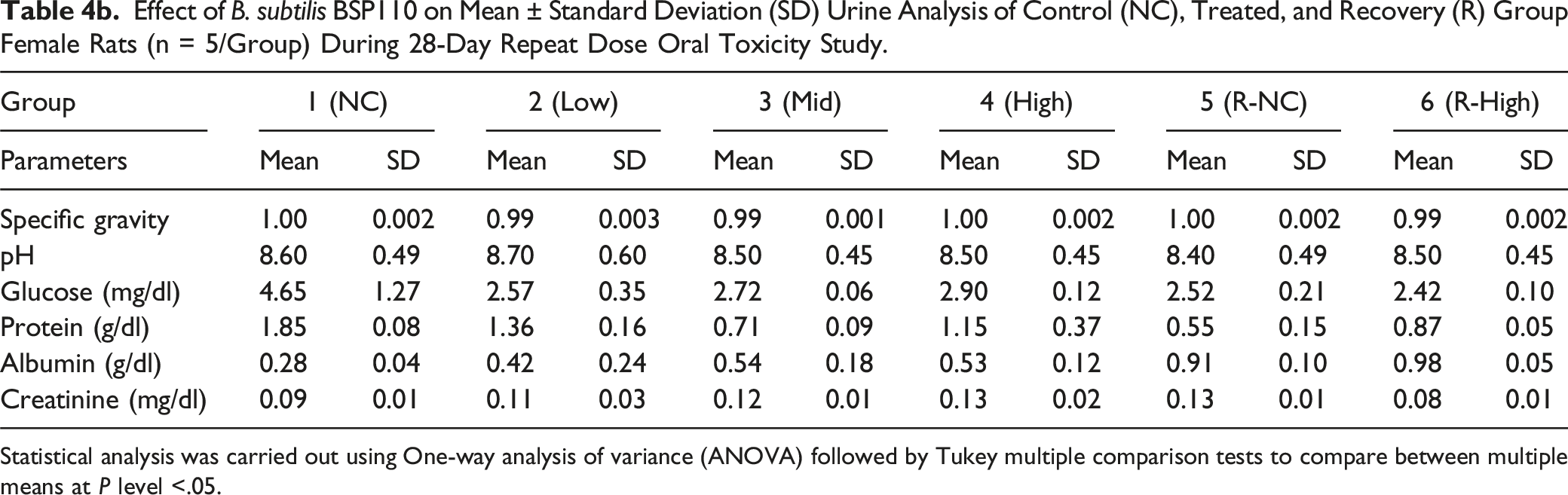
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Gross Necropsy Study
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Relative Organ Weight of Control (NC), Treated, and Recovery (R) Group Male Rats (n = 3/Group) During 28-Day Repeat Dose Oral Toxicity Study.
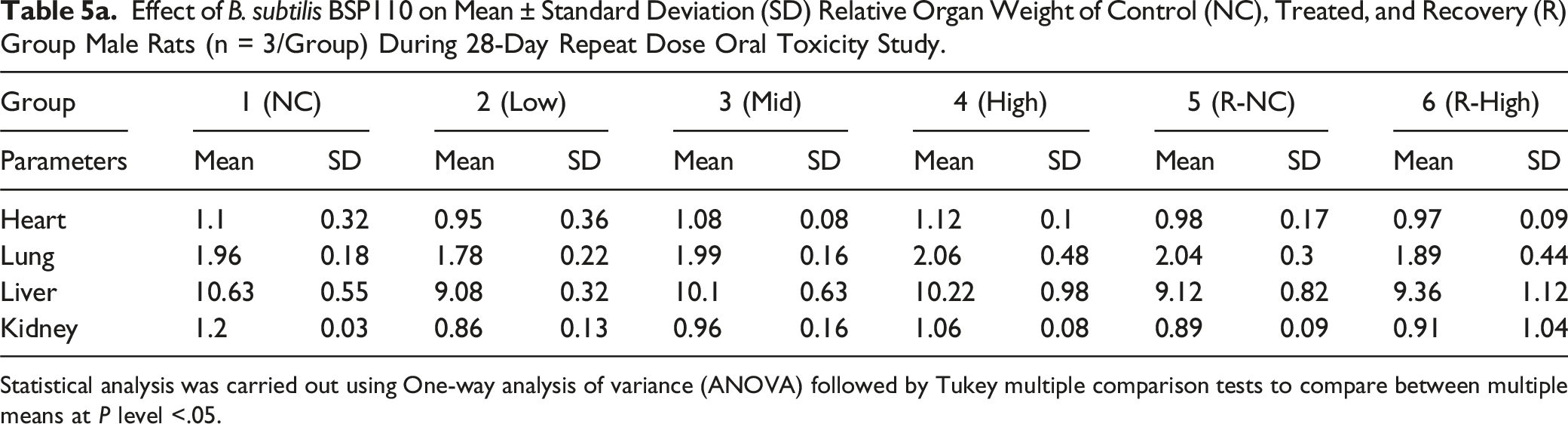
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Effect of B. subtilis BSP110 on Mean ± Standard Deviation (SD) Relative Organ Weight of Control (NC), Treated, and Recovery (R) Group Female Rats (n = 3/Group) During 28-Day Repeat Dose Oral Toxicity Study.
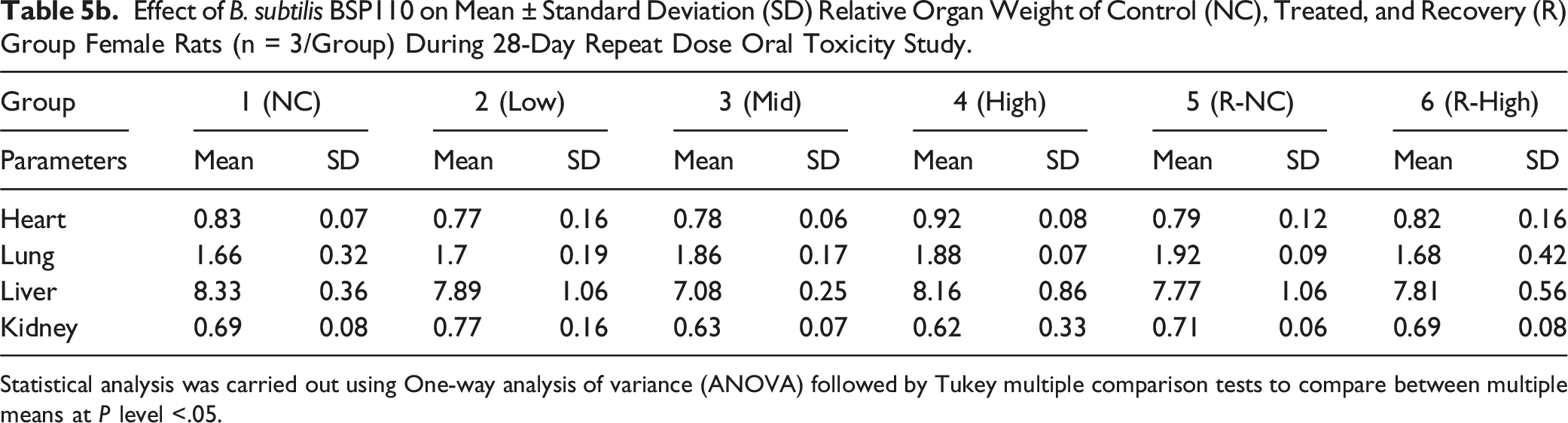
Statistical analysis was carried out using One-way analysis of variance (ANOVA) followed by Tukey multiple comparison tests to compare between multiple means at P level <.05.
Histopathological Examination
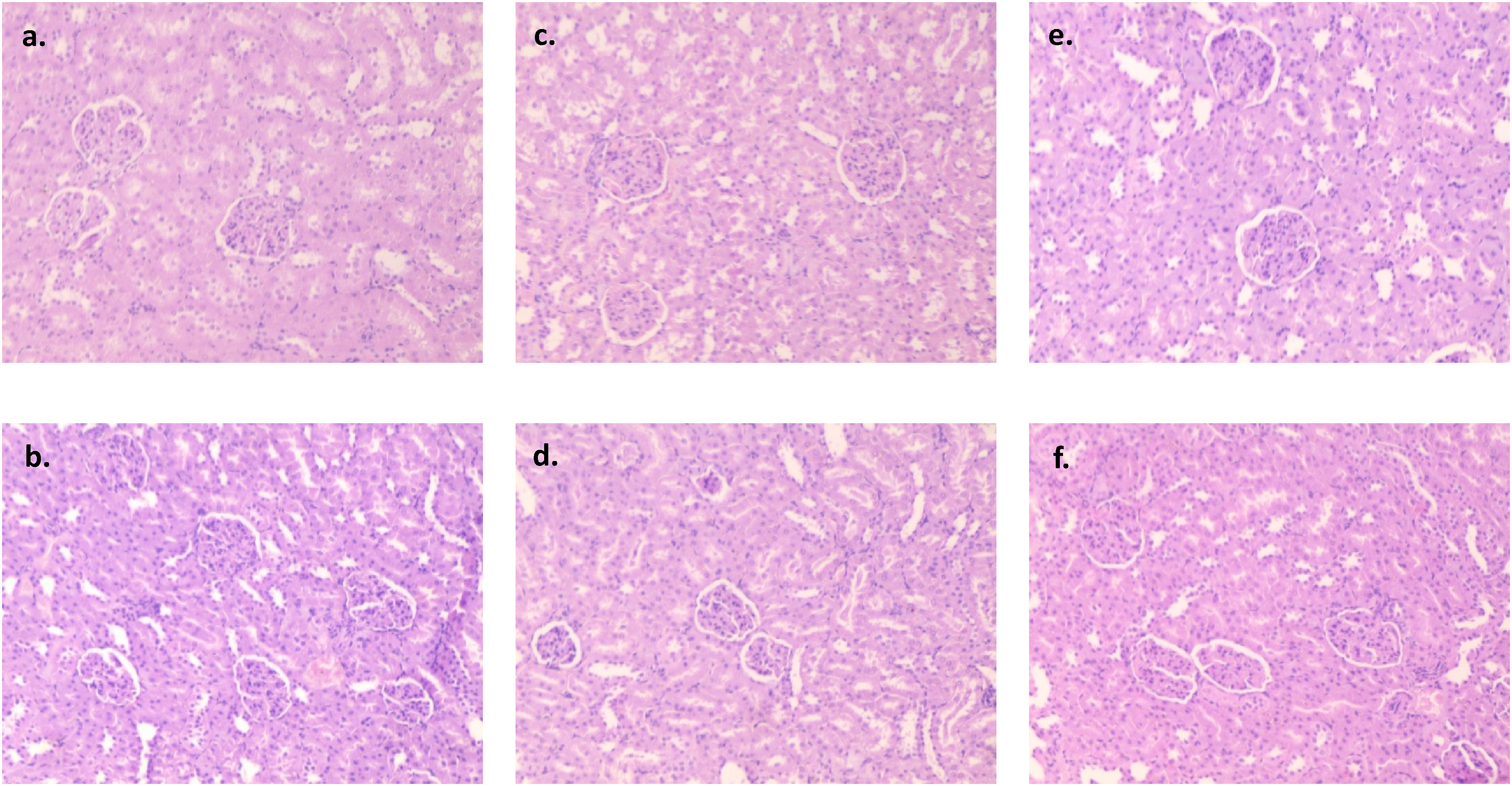
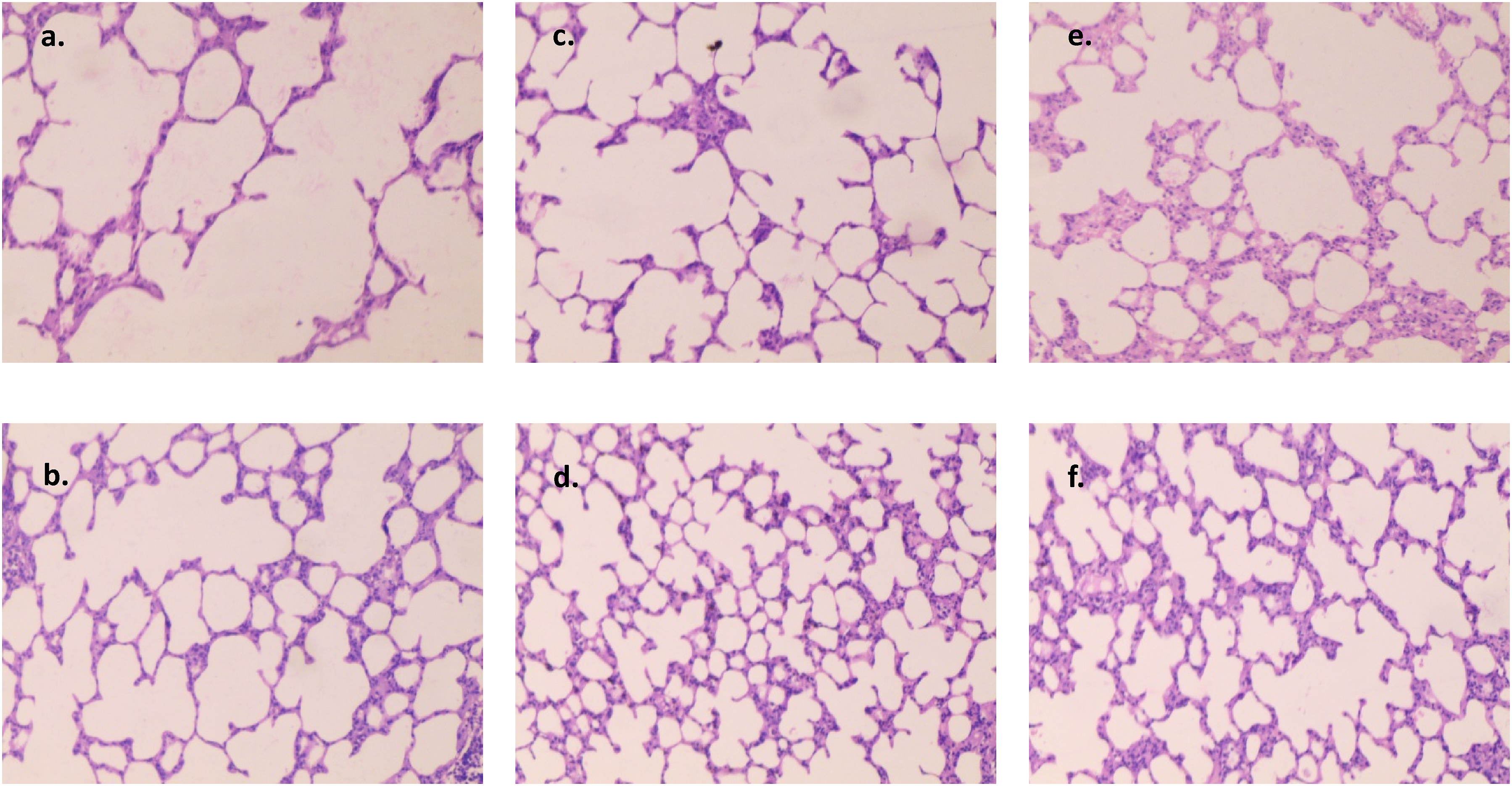
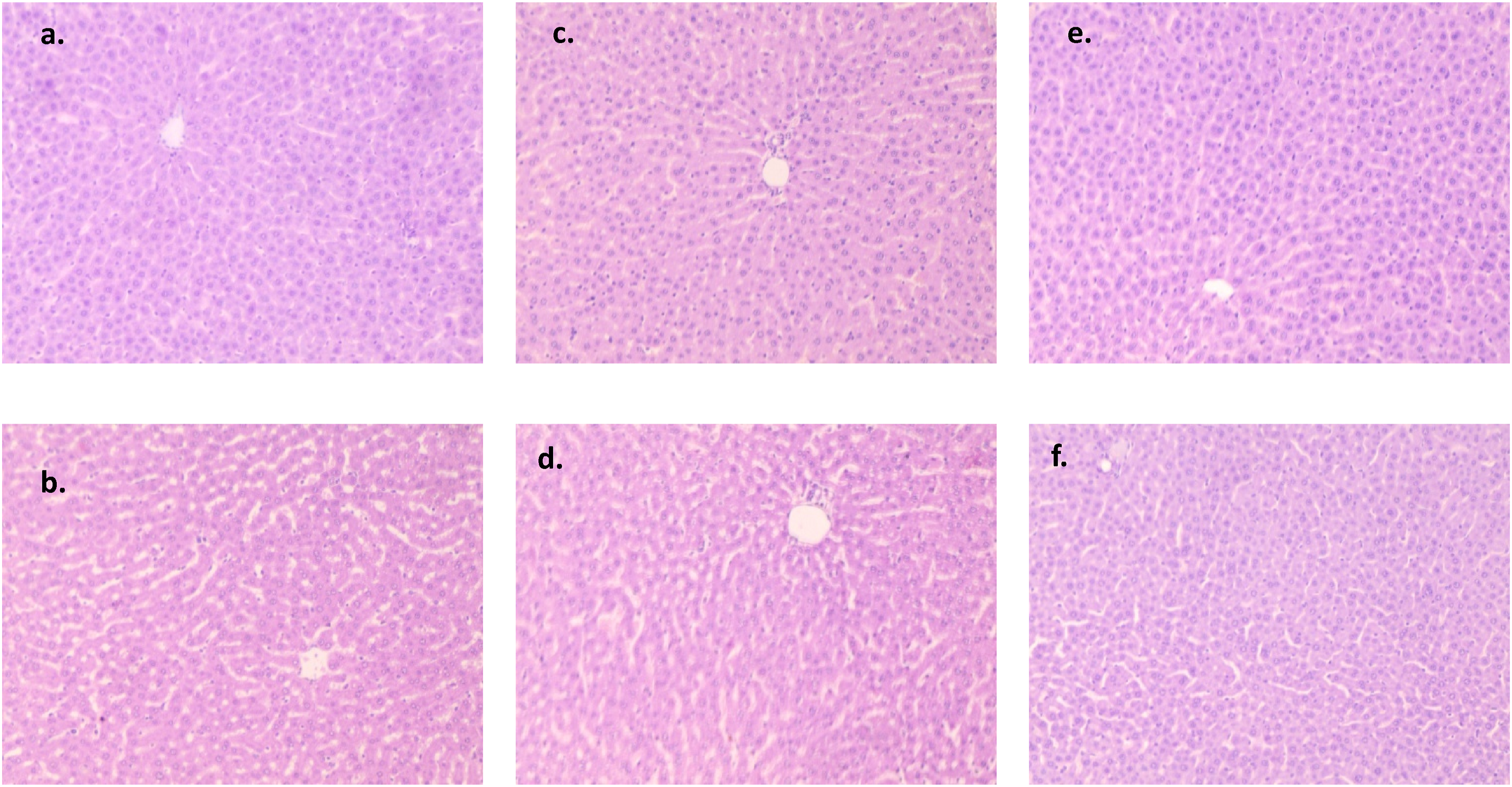
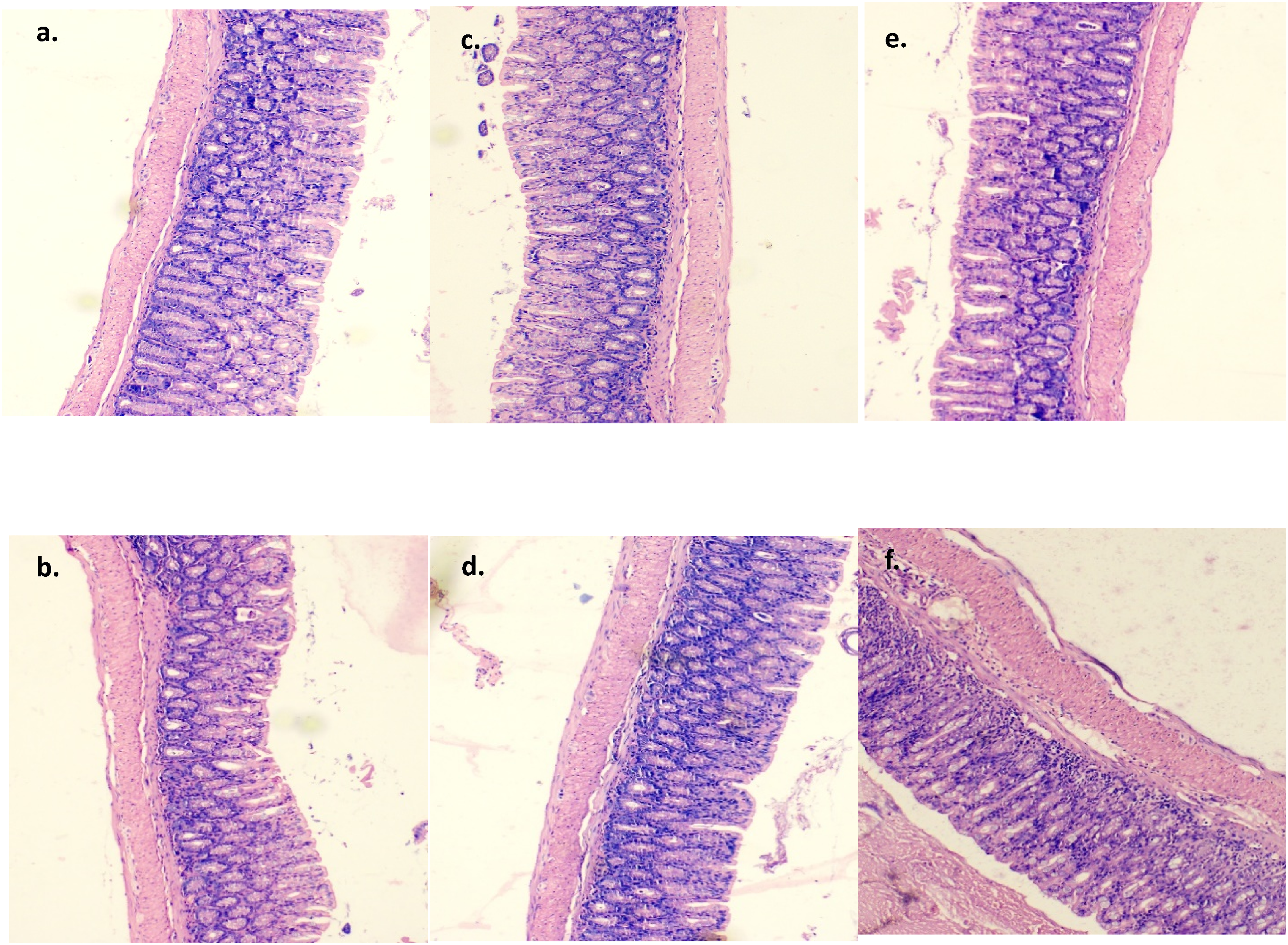
Three samples of heart, lung, liver, kidney, and colon tissues from the normal, treatment, and recovery groups were examined under a microscope and one representative image is shown from each group. No group’s heart tissue showed any indications of cardiac degeneration or necrosis upon evaluation (Figure 2). The cardiac myocyte structure was also determined to be normal and free of anomalies, with neither vacuolation nor mononuclear cell infiltration present. Thin glomerular capillary loops and normal mesangial and endothelial cells were seen in the kidney sections (Figure 3), indicating normal glomerular filtration, whereas there were no discernible alterations in the lung sections between the normal, treatment, and recovery groups (Figure 4). There were no indications of inflammation, nodule development, inflammatory cell infiltration, or lipid buildup in the liver sections, which had a typical hexagonal hepatocyte shape (Figure 5). Finally, the colon showed intact mucosa, no sign of ulceration, and no inflammation (Figure 6). Effect of B. subtilis BSP110 on histology of heart section of control group Male [a], Female [b], high dose group Male [c], Female [d], High dose recovery group Male [e], Female [f], shows no evidence of any abnormality. Effect of B. subtilis BSP110 on histology of kidney section of control group Male [a], Female [b], high dose group Male [c], Female [d], High dose recovery group Male [e], Female [f], shows no evidence of any abnormality. Effect of B. subtilis BSP110 on histology of lungs section of control group Male [a], Female [b], high dose group Male [c], Female [d], High Dose recovery group Male [e], Female [f], shows no evidence of any abnormality. Effect of B. subtilis BSP110 on histology of liver section of control group Male [a], Female [b], high dose group Male [c], Female [d], High dose recovery group Male [e], Female [f], shows no evidence of any abnormality. Effect of B. subtilis BSP110 on histology of colon section of control group Male [a], Female [b], high dose group Male [c], Female [d], High dose recovery group Male [e], Female [f], shows no evidence of any abnormality.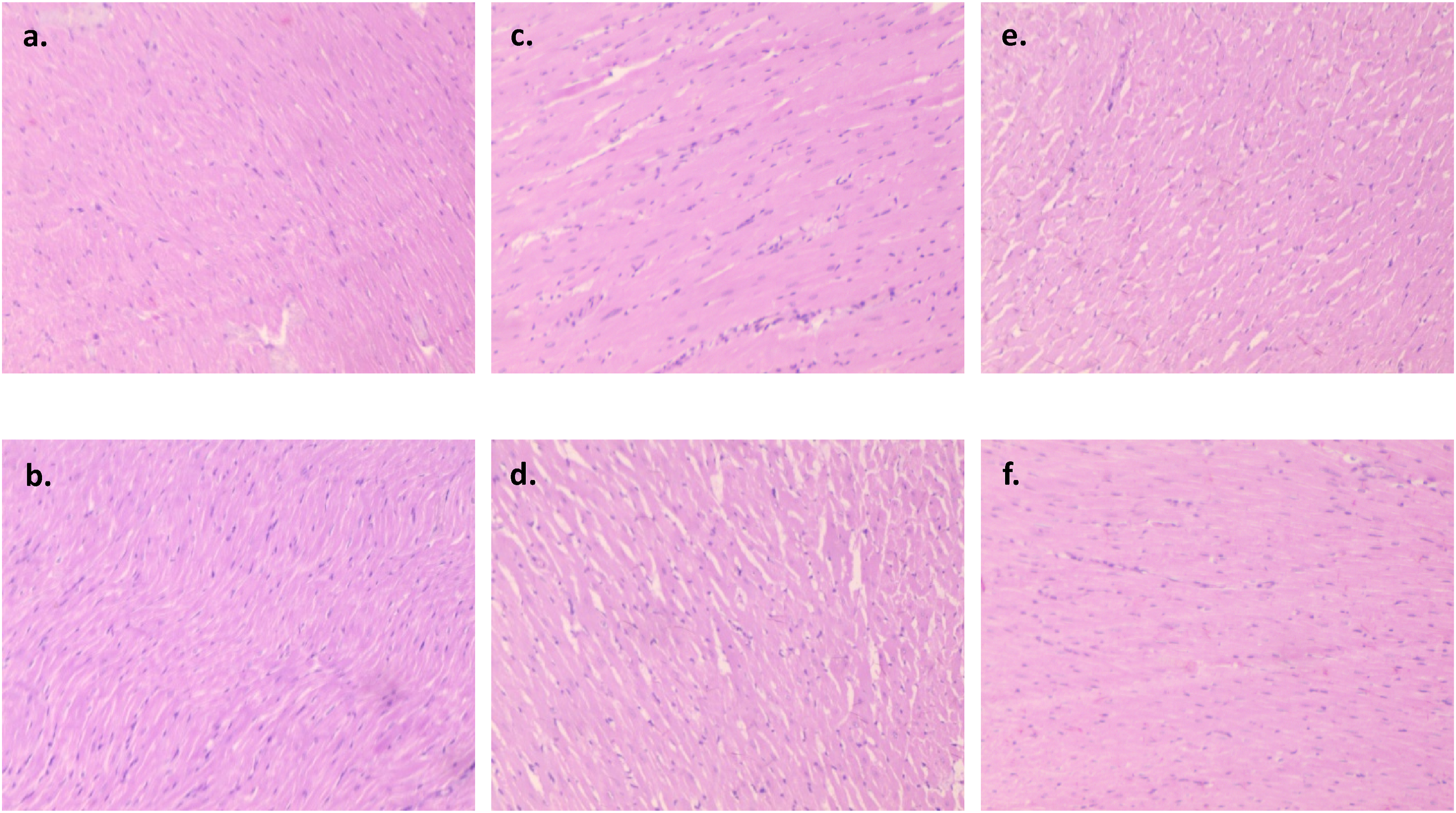
In Vitro Cell Viability
In-Vitro Cell Viability Assay of Bacillus subtilis BSP110 on Vero Cell Line.

Discussion
In the age of functional foods, probiotics, whether in the form of food items or supplements, have become the most popular component. Probiotics have long been a crucial element and a lucrative target due to their possible health advantages. 1 Despite having numerous health advantages, several probiotics have been reported to show adverse effects.17,18 Conducting acute and sub-acute toxicity studies is essential to addressing these issues, along with assessing other safety factors.9,19 The purpose of this preliminary study was to evaluate the possible acute and sub-acute toxicity linked to oral administration of the Bacillus subtilis BSP110 probiotic strain.
Acute and sub-acute toxicity tests were required to ascertain the NOAEL and toxic effects to evaluate the safety of the probiotic strain Bacillus subtilis BSP110. Thus, in accordance with the OECD recommendations, acute and sub-acute toxicity experiments were carried out in rats. 20
An acute toxicity study is the main test for safety evaluation that is advised in a number of safety assessment studies. 21 A single oral administration of the probiotic strain B. subtilis BSP110 at a dosage of 2000 mg/kg, equivalent to a concentration of 4 × 1011 CFU/g in female rats, did not result in any aberrant clinical symptoms, morbidity, or death in an acute toxicity investigation. Clinical indicators like appearance, respiration, motor activity, tremors, convulsions, corneal reflexes, lacrimation, mydriasis, salivation, diarrhea, and other discharges were all normal in the current investigation. The acute toxicity study’s findings suggest that MTD of B. subtilis BSP110 is up to 2000 mg/kg.
Studies on sub-acute toxicity have been proposed as a crucial test for evaluating safety. 22 To assess the NOAEL and adverse effects of repeated exposure to B. subtilis BSP110, a preliminary repeated dose sub-acute toxicity study was conducted. To examine the post-recovery effects of the test probiotic, Wistar rats of both sexes were given varying dosages of B. subtilis BSP110 (250 mg/kg/day, 500 mg/kg/day, and 1000 mg/kg/day, having a concentration of 4 × 1011 CFU/g) once daily by oral gavage for 28 days. The recovery groups were then kept for further 14 days. Animals were weighed once a week, and when the treatment groups were compared to the normal control group, no discernible difference in body weight was found. This suggests that the probiotic strain B. subtilis BSP110 does not alter the test group rats’ natural nutritional digestion.
Changes in physical appearance and behavioral characteristics are frequently used to detect toxicity. However, neither the test participants’ physical characteristics nor their behavioral patterns changed significantly after being exposed to different dosages of Bacillus subtilis BSP110.
We assessed the levels of alkaline phosphatase, urea, total protein, bilirubin, albumin, triglycerides, cholesterol, SGOT, SGPT, sodium, chloride, potassium, and triglycerides to identify any changes in organ function brought on by the harmful effects of administering B. subtilis BSP110. The biochemical markers showed no discernible alterations. This demonstrates that B. subtilis BSP110 does not affect organ function or nutrient metabolism. Parameters, including pH, specific gravity, glucose, protein, creatinine, and albumin levels, did not significantly alter, according to the urine analysis findings. These findings also show that Bacillus subtilis BSP110 has no harmful effects and preserves the body’s natural metabolic balance.
One of the most important factors in determining a drug’s toxicity is hematological alteration. WBC, neutrophil, lymphocyte, monocyte, eosinophil, basophile, RBC, hemoglobin, HCT, MCV, MCH, MCHC, RDW-CV, RDW-SD, PLT, MPV, PDW, APTT, and PT were among the biochemical markers that were assessed in this study. In the test groups (Groups 1, 2, 3, and 4), all hematological parameters were determined to be within normal limits and comparable to the animals in the control group. None of these parameters showed any unusual changes.
The heart, lungs, liver, kidneys, and colon are targeted organs for harmful and poisonous chemicals and substances. These organs are thought to be very helpful in toxicity research because they are connected to either detoxification or excretion processes, or because they provide vital bodily activities. 23 Changes in the weight of these essential organs due to toxicity are frequently followed by similar histological abnormalities. All treatment groups were examined for changes in important organ weights. Gross organ weight and absolute organ weight were unaffected by treatment. Furthermore, B. subtilis BSP110 therapy did not affect the macroscopic anatomy of the liver, kidney, heart, or lungs. Additionally, hematological and biochemical assays are supported by histopathological investigations. Following the administration of B. subtilis BSP110, histopathology of the heart, lungs, liver, and kidney revealed no interstitial inflammation in the kidney and lung tissue, normal hepatocellular architecture without hepatocyte degradation, and no change in the histology of the cardiac tissue.
In vitro cytotoxicity assessment using cell lines gives a preliminary idea of the general safety of the probiotic. Only live cells can create formazan products during this reaction, which makes the MTT assay a very sensitive assay for assessing cellular respiration, cell viability, and cytotoxicity. 24 Cytotoxicity of Bacillus subtilis BSP110 was assessed using the Vero cell line, and B. subtilis BSP110 was found to be non-cytotoxic and, in fact, showed cell proliferative effect which needs to be evaluated further in depth.
In this sub-acute toxicity study, the changes observed in hematological, biochemical, and urine parameters were reversible and could not be linked to significant toxicity. The absence of a clear dose-response relationship and the return to baseline levels in recovery groups suggest that the treatment did not cause serious toxic effects. Furthermore, no morphological changes were seen in the histological examination, indicating that Bacillus subtilis BSP110 does not affect the normal functioning of body organs. These histopathological findings substantiate the biochemical results obtained. Similar studies performed by various groups of researchers on similar or different species of probiotics corroborate with results obtained in this study.
Conclusion
Acute toxicity results show that Bacillus subtilis BSP110 does not result in mortality or adverse effects. The sub-acute toxicity study’s findings showed no appreciable alterations in histology, clinical pathology, clinical symptoms, or body weight. The findings of acute and sub-acute toxicity studies showed that B. subtilis BSP110 does not show adverse effects in rats. In sub-acute toxicity experiments, B. subtilis BSP110 was given at a concentration of 4 × 1011 CFU/g at a dosage of 1000 mg/kg (the maximum dose). A 28-day repeated dosage sub-acute toxicity study yielded a No Observed Adverse Effect Level (NOAEL) of 1000 mg/kg/day, representing 4 × 1011 CFU/kg. For a human weighing an average of 70 kg, this corresponded to 2.8 × 1013 CFU. The proposed human dose is 2 × 109 CFU per serving, which provides a safety factor of 17.5 × 102.
Footnotes
Author Contributions
SSS, FM, and VG: Writing—review and editing, writing—original draft, conceptualization, and project administration. SSS and SP: Validation, visualization, data curation, and methodology. SP and PP: Supervision, investigation, and formal Analysis. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
The animal study protocol was reviewed and approved by the Institutional Animal Ethics Committee (IAEC) of the Institute of Pharmacy under protocol number IP/PCOL/FAC/35/2023/27. All procedures were conducted in accordance with the guidelines of the Committee for the Control and Supervision of Experiments on Animals (CCSEA), Ministry of Fisheries, Animal Husbandry and Dairying, Government of India, New Delhi.
Data Availability Statement
All data and probiotic strains used in this study are available from corresponding authors with reasonable request.
