Abstract
Almega PL is an eicosapentaenoic acid-rich ω-3 oil that is isolated from Nannochloropsis oculata algae and developed as a dietary supplement. The safety of the algal oil was evaluated in 14- and 90-day studies in Sprague-Dawley rats by oral gavage at dose levels of 0, 250, 500, and 2500 mg/kg/d and 0, 200, 400, and 2000 mg/kg/d, respectively. No mortalities occurred and no signs of toxicity were observed during the studies. No treatment-related effects were seen for body weight, food consumption, ophthalmology, neurological effects, urinalysis, clinical pathology, gross pathology, organ weights, or histopathology. Although statistically significant effects were noted for some end points, none were considered to be of toxicological significance. The no observed adverse effect level for Almega PL was 2000 mg/kg/d. Additionally, Almega PL was not mutagenic in Salmonella typhimurium or Escherichia coli, did not induce chromosome aberrations in Chinese hamster ovary cells, and did not induce genotoxic effects in vivo in rat bone marrow erythrocytes.
Keywords
Introduction
Nannochloropsis is a small green microalga genus that is well known due to its nutritional value and ability to produce valuable lipophilic and lipophobic materials. 1 In the aquaculture industry, Nannochloropsis is extensively used as feedstock in growing small zooplanktons such as rotifers and in fish hatcheries 2 and for producing green water. 3 In addition, the biochemical composition of Nannochloropsis makes it a valuable food source for animals and humans. 1
Nannochloropsis oculata is 1 of the 6 species contained in the genus Nannochloropsis and was originally isolated off the coast of Scotland. Species of the genus Nannochloropsis have been shown to contain high concentrations of eicosapentaenoic acid (EPA) while containing no docosahexaneoic acid (DHA). 4,5 Nannochloropsis oculata is a rich source of EPA, and the oil extracted from the green microalgae is one of the few sources of EPA that does not also contain DHA. 4 Recent studies comparing EPA and DHA have suggested that not only is the EPA component primarily responsible for reducing inflammation 6 and depression 7 but that any DHA component acts to block the activity of EPA. 8 Therefore, the oil extracted from N oculata presents a means of maximizing the available levels and desired beneficial aspects of EPA as evidenced in humans. 4
Almega PL is an EPA-rich ω-3 oil isolated from N oculata. It is being developed by Qualitas Health Ltd, (Jerusalem, Israel) as a dietary supplement ingredient with a suggested daily intake of 300 to 1250 mg (2.0-20.8 mg/kg/d for a 60-kg person), thus providing a maximum of 312.5 mg/d ω-3 fatty acids. 9 To evaluate the potential toxicity of Almega PL, a battery of Good Laboratory Practice (GLP)-compliant in vivo and in vitro assays were performed. In rat models, a 14-day maximum-tolerated dose (MTD) study and a 90-day repeated dose subchronic toxicity study were performed. Clinical observations, clinical chemistry, hematology, urinalysis, functional observational batteries (FOBs), and gross and microscopic pathology were conducted to determine whether or not repeated oral exposure of Almega PL resulted in any signs of toxicity. Additionally, to evaluate the genotoxic potential of Almega PL, a bacterial reverse mutation assay, an in vitro mammalian chromosome aberration test in Chinese Hamster (Cricetulus griseus) ovary (CHO) cells, and an in vivo micronucleus assay in rats were performed.
Materials and Methods
Test Item
Almega PL (Lots 130202a and 130610; Qualitas Health Ltd) is a nonpurified, dark green oily paste obtained from N oculata. The source biomass, N oculata, is cultured in raceway-type pools and harvested daily followed by extraction of the oil from the biomass, resulting in Almega PL. Almega PL consists of approximately 55% total fatty acids, of which 25% is EPA. Almega PL also contains chlorophyll (approximately 3%) and 15% polar lipids (phospholipids and glycolipids).
The test item was analyzed for the presence of organochlorine and organophosphate pesticides. No pesticides were found. An analysis for dioxin, dioxin-like polychlorinated biphenyls (PCBs), and nondioxin-like PCBs indicated that the levels of these substances are below the maximum levels for marine oils as listed by the World Health Organization and when adjusted for fat content. Toxin analyses conducted on Almega PL indicated that no natural product toxins were detected above detection limits including microcystins/nodularin, anatoxin-a, cylindrospermopsin, paralytic shellfish toxin/saxitoxins, okadaic acid, and brevetoxins. 10
Chemicals and Materials
Extra virgin olive oil used as the vehicle for the in vivo studies was supplied by Qualitas Health Ltd except for in the micronucleus assay where it was supplied by Pharmaseed Ltd (Ness Ziona, Israel). 2-Nitrofulorene and 2-aminoanthracene were obtained from Sigma-Aldrich (Munich, Germany). 9-Aminoacridine was obtained from Novel Organic Synthesis (Andhra Pradesh, India). 4-Nitroquinoline-1-oxide, sodium azide, ethyl methanesulfonate, cyclophosphamide monohydrate, and colchicine were obtained from Sigma-Aldrich (St Louis, Missouri). Dimethyl sulfoxide (DMSO) was obtained from RFCL Ltd (Faridabad, India). Oxoid nutrient broth No. 2 was obtained from Oxoid Ltd (Basingstoke, Hampshire, England). Fetal bovine serum was obtained from Sera Laboratories (West Sussex, United Kingdom). Methanol was obtained from Merck Specialties Private Limited (Mumbai, India). Potassium phosphate monobasic, sodium phosphate dibasic, and acridine orange hydrochloride hydrate were obtained from Sigma-Aldrich (Buchs, Switzerland). DPX (Distyrene, Plasticizer, Xylene) mountant was obtained from SD Fine Chemicals Limited (Mumbai, India).
Animals and Organisms
For the 14-day MTD study performed at Pharmaseed Ltd, 24 (12 male and 12 female) 8-week-old Sprague-Dawley SD rats obtained from Harlan (Israel) were housed in polyethylene cages (3/cage). The acclimatization period was 3 days prior to testing. Initial mean group body weights were 237 g for males and 181 g for females. The minimum and maximum initial weight in the groups were within a range of ±20% of the groups mean weight. Room temperature was maintained at 20°C to 24°C with a relative humidity of 30% to 70%, a minimum of 15 air changes/h and a 12-hour light and 12-hour dark cycle. Autoclaved and acidified drinking water (pH between 2.5 and 3.5) and commercial rodent diet (Teklad Certified Global 18% Protein Diet cat # 2018SC) were provided ad libitum.
For the 90-day repeated dose, subchronic toxicity assay performed at Advinus Therapeutics, Ltd (Bangalore, India), male and female Sprague-Dawley rats raised in house by Advinus Therapeutics were used. The acclimatization period was 5 days prior to testing. The rats were 6 to 7 weeks old at the beginning of the assay. Initial mean group body weights ranged from 145 to 148 g for the male rats and 127 to 130 g for the female rats. At the beginning of treatment, the weight variation of the test animals did not exceed ±20% of the mean body weight in each group and sex. Rats were housed in groups of 2 per sex per cage in solid floor standard polysulfone cages with stainless top grills for providing pelleted food and drinking water in polycarbonate bottles with stainless steel sipper tubes in rooms with 12 hours light cycles. Steam sterilized clean corn cob was used as bedding and changed along with the cage once during the acclimatization period and twice a week during the treatment period. Room temperature was maintained at 19°C to 24°C with a relative humidity of 58% to 68%, with a minimum of 12 air changes/h. Filtered deep-bore well water and Teklad Global 14% protein rodent maintenance diet (Harlan Laboratories, An Venray, the Netherlands) were provided ad libitum.
The Salmonella typhimurium and Escherichia coli test strains for the bacterial reverse mutation assays performed at Advinus Therapeutics Ltd were obtained, respectively, from Health Protection Agency National Collection of Type Cultures (London, Great Britain) and The National Collection of Industrial and Marine Bacteria Ltd (Scotland, United Kingdom). The CHO cell line CHO-51, (ATCC CCL-61, Lot 4765275) for the chromosome aberration assay performed at Advinus Therapeutics Ltd were obtained from the American Type Culture Collection (Manassas, Virginia).
For the in vivo erythrocyte micronucleus test performed at Advinus Therapeutics Ltd, male and female Sprague-Dawley rats (in-house random bred) were acclimated for 5 days before dosing. The rats were at least 10 weeks old (range, 10-11 weeks) at the beginning of the assay. Initial body weights ranged from 250 to 253 g for the male rats and 188 to 190 g for the female rats. All rats were visually inspected prior to the start of treatment. Rats were individually housed during acclimatization and treatment in standard polysulfone cages with stainless steel top grills in rooms with 12 hours light cycles. Steam sterilized corn cob was used as bedding which along with cages was changed at least once a week. Room temperature was maintained at 20°C to 23°C with a relative humidity of 65% to 67% and 12 to 15 high-efficiency particulate air-filtered air changes/h. Deep bore-well water (passed through activated charcoal filter and exposed to ultraviolet [UV] rays) and Teklad Certified (2014C) Global 14% Protein Rodent Maintenance pellets were provided ad libitum.
Guidelines
Because the 14-day rat study was a dose-range finding (DRF) study and used to determine the MTD of Almega PL as well as the doses for the subsequent subchronic (90 day) study, it was not performed in full compliance with GLP guidelines. The subchronic toxicity study in rats was performed in accordance with the OECD Guideline for Testing of Chemicals, (408): Repeated Dose 90-Day Oral Toxicity Study in Rodents 11 and under GLP in accordance with OECD 12 Principles of GLP for the Testing of Chemicals. The bacterial reverse mutation (Ames) assays were performed in accordance with the OECD 13 Guideline 471 for testing of chemicals: Bacterial Reverse Mutation Test and under GLP in accordance with OECD 14 Principles of GLP. The chromosome aberration assay was performed in accordance with the OECD 15 Guideline 473 for testing of chemicals: In Vitro Mammalian Chromosome Aberration Test and under GLP in accordance with OECD 14 Principles of GLP. The rat micronucleus assay was performed in accordance with the OECD 16 Guideline 474 for testing of chemicals: Mammalian Erythrocyte Micronucleus Test and under GLP in accordance with OECD 12 Principles of GLP.
Experimental Design
Maximum-tolerated dose oral toxicity study
Groups of 3 male and 3 female SD rats each were administered a single dose of 0, 5% (250 mg/kg), 10% (500 mg/kg), or 50% (2500 mg/kg) Almega PL by oral gavage on day 1. The administrations were performed using 16-gauge gavage needles. The vehicle was warm extra virgin olive oil, and the dose volume was 5 mL/kg. Following the treatment period, all rats were monitored for 14 days and euthanized on day 15.
Rats were observed twice daily for signs of morbidity and mortality and once daily for clinical signs until study termination. Body weights were recorded prior to the initial dosing, twice weekly during the study, and at study necropsy. On day 15, rats were euthanized by carbon dioxide asphyxiation. After killing the animals, the organs and tissues identified in Table 1 were removed and examined for gross pathological anomalies.
List of Evaluated Organs.
Subchronic oral toxicity study
Groups of 10 male and 10 female Sprague-Dawley rats each were administered 0, 250, 500, or 2500 mg/kg Almega PL in extra virgin olive oil by oral gavage once daily for 90 days. Doses were administered using disposable plastic syringes attached with a metal feeding cannula. All doses were administered at a constant dose volume of 5 mL/kg. Following the treatment period, all animals were euthanized on day 91.
Rats were observed twice daily for signs of morbidity and mortality and once daily for clinical signs until study termination. Detailed clinical examinations were performed prior to the item administration on day 1 and at weekly intervals (±2 days) thereafter. Body weights were recorded on day 1 prior to the test item administration and weekly thereafter except for week 13 when the body weights were recorded on day 5 of the week. Fasting body weights were recorded prior to necropsy on day 91. Food consumption was measured once per week on the same days that body weight measurements were made. At the end of the study, rats were fasted overnight (water allowed), anesthetized with isoflurane, and exsanguinated.
Ophthalmological examination
Prior to the start of treatment (on day 5 of acclimatization), ophthalmological examinations were performed for all animals with an ophthalmoscope. Mydriasis was induced before examination using a solution of 1% tropicamide. The eyes were examined in subdued light. Subdued light was maintained in the animal room for the remainder of the day. These procedures were repeated at the end of the treatment period for all animals.
Functional observational battery tests
To assess the behavioral and neurological status of each rat, FOB tests were performed for all of the group rats on day 90 of the treatment period. The FOB tests included home cage observations for posture and for presence or absence of abnormal vocalizations and convulsion; observations during removal from rat from home cage and handling; open field observations; and functional tests including sensory evaluation, landing hindlimbs foot splay, grip performance, and motor activity (Table 2).
Functional Observational Battery (FOB) Tests.

Urinalysis
Before killing, urine was collected from all rats in urine collection tubes. Each rat was placed in specially fabricated cages overnight (water allowed) and the next morning the urine was collected for analysis. Urinalysis parameters examined in the collected samples included color, clarity, bilirubin, erythrocytes, glucose, ketone bodies, leukocytes, nitrite, proteins, pH, specific gravity, urobilinogen, and volume. Urine was also subjected to microscopic examination for sediments such as crystals, epithelial cells, and casts.
Clinical pathology
At the end of the treatment period, rats were fasted overnight (water allowed) after which blood was collected from the retro-orbital sinus plexus with fine capillary tubes under isoflurane anesthesia. An aliquot of blood was collected in tubes containing 3.2% sodium citrate solution for determination of coagulation parameters, and the remaining blood was collected into K2EDTA and lithium heparainized tubes for hematology and clinical chemistry examinations (Table 3).
Clinical Pathology Investigations.

Abbreviations: DLC, differential leukocyte count; HCT, hematocrit; HGB, hemoglobin; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MPV, mean platelet volume; Plat, platelets; RBC, red blood corpuscles; Retic, reticulocytes; WBC, white blood corpuscles; PT, prothrombin time; APTT, activated partial thromboplastin time; A/G, albumin/globulin ratio (calculated values); ALT, alanine aminotransferase; Alb, albumin; ALP, alkaline phosphatase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Ca, calcium; Cl, chloride; Creat, creatine; GGT, γ-glutamyl transpeptidase; Glob, globulin (calculated values); Glu, glucose; Pi, inorganic phosphorous; K, potassium; Na, sodium; TBil, total bilirubin; TChol, total cholesterol; TPro, total plasma protein; Trig, triglycerides; urea, urea (calculated values).
aUrea values are not reported separately as both BUN and urea values are the same when expressed in mmol/L.
Pathology
All animals were fasted overnight prior to terminal killing on day 91. At the scheduled termination, all survivors were euthanized by exsanguination under isoflurane anesthesia. The organs identified in Table 4 were removed, weighed, and examined for gross and microscopic pathological anomalies.
Tissue Collection and Organ Weighing.
aOrgans weighed.
Bacterial reverse mutation assays
The mutagenic potential of Almega PL was evaluated using the bacterial reverse mutation assay (Ames test) using standard plate incorporation (experiment I) and preincubation (experiment II) methods. The ability of Almega PL to induce mutagenicity in S typhimurium tester strains TA98, TA100, TA1535, and TA1537 and E coli strain WP2uvrA was assessed in the presence and absence of rat microsomal fraction S9 mix. Based on the observations of preliminary experiments to evaluate toxicity, 5000 μg/plate was selected as the highest dose for all test strains and conditions. A stock solution of 50 000 μg/mL test item in DMSO was prepared by mixing 500 mg Almega PL in DMSO and bringing the volume to 10 mL in a volumetric flask with DMSO. For experiment I, 4 lower dose levels (50, 158, 500, and 1581 μg/plate) were prepared by serially diluting the stock solution with DMSO. For experiment II, 4 lower dose levels (100, 266, 707, and 1880 μg/plate) were prepared by serially diluting the stock solution with DMSO. The positive controls in the absence of S9 mix were 2-nitrofluorene for S typhimurium TA98, sodium azide for S typhimurium TA100 and TA1535, 9-aminoacridine for S typhimurium TA1537, and 4-nitroquinoline-1-oxide for E coli WP2uvrA. The positive control for all bacterial strains in the presence of S9 mix was 2-aminoanthracene. The vehicle control for all strains in the presence or absence of S9 mix was DMSO (100 μL).
Each of the tester strains from the master plates were inoculated into tubes containing Oxoid Nutrient broth No. 2 and then incubated at 37°C ± 1°C for approximately 17 hours. For experiment I, the bacterial suspensions were exposed to the test item, vehicle, and the positive controls in the presence or absence of the exogenous metabolic activation system. These bacterial suspensions were then mixed with overlay agar and plated immediately onto minimal medium, viz, his− for S typhimurium and trp− for E coli, respectively. The plates were then incubated at 37°C ± 1°C for 67 hours. Revertant colonies were counted manually, and the plates were examined for bacterial background lawn. For experiment II, the test constituents were mixed with the bacteria inside tubes, incubated in an incubator shaker for 30 minutes at 37°C ± 1°C, mixed with overlay agar, and plated immediately onto minimal medium his− for S typhimurium and trp− for E coli, respectively. The plates were then incubated at 37°C ± 1°C for 67 hours. Revertant colonies were counted manually, and the plates were examined for bacterial background lawn.
The tests were considered positive for mutagenicity if the increase in mean revertants at the peak of the dose response was equal to or greater than twice the mean vehicle control value for TA98, TA100, or WP2uvrA or equal to or greater than 3 times the mean vehicle control value for TA1535 or TA1537. Data were not analyzed statistically.
Chromosome aberration assay
The potential of Almega PL to induce structural chromosome aberrations in cultured mammalian cells was evaluated using CHO cells. The maximum concentrations of Almega PL tested were selected based on preliminary range finding test in which concentrations between 17.5 and 35 μg/mL with 4-hour exposure (presence of S9) and 35 and 70 μg/mL with 4-hour exposure (absence of S9) caused cytotoxicity of at least 50% cell growth inhibition compared to the vehicle control. Similarly, cytotoxicity was seen at concentrations between 4.4 and 8.45 μg/mL with 21-hour exposure in the absence of metabolic activation. A stock solution of 10 000 μg/mL Almega PL was prepared by mixing 250 mg Almega PL in DMSO and bringing the volume to 25 mL with DMSO in a volumetric flask. Almega PL was exposed to CHO cells in duplicate at concentrations of 0 (DMSO negative control), 1.9, 6, and 19 μg/mL and at 0 (DMSO negative control), 4, 13, and 40 μg/mL of the medium in the presence and absence of metabolic activation, respectively, with 4-hour exposure. Additionally, Almega PL was exposed to CHO cells in duplicate at concentrations of 0 (DMSO negative control), 1.75, 3.5, and 7 μg/mL of the medium in the absence of metabolic activation, with 21-hour exposure. Ethyl methanesulfonate (600 μg/mL) was used as the positive control in the absence of S9 mix, and cyclophosphamide monohydrate (55 μg/mL) was used as the positive control in the presence of S9 mix. Following exposure of the cell cultures to the test items for 4 or 21 hours, they were treated with Colchicine to arrest the cells in a metaphase-like stage of mitosis (c-metaphase). Cells were then harvested and chromosome preparations were made. Preparations were stained, and metaphases were analyzed under microscope for chromosomal aberrations. Concurrent cytotoxicity for all treated and control cultures was also assessed.
In vivo micronucleus assay
The micronucleus assay was performed in order to determine the potential of Almega PL to cause genotoxicity in the form of micronuclei containing lagging chromosome fragments (clastogenicity) or whole chromosomes (aneugenicity). Prior to the main experiment, a DRF study was performed to determine the MTD of Almega PL and to select the doses for the main experiment. Due to a lack of observed toxicity in the DRF study, the MTD was set at 2000 mg/kg/d, the highest dose tested. For the main study, groups of 5 male and 5 female Sprague-Dawley rats each were administered 0, 500, 1000, or 2000 mg/kg/d Almega PL in extra virgin olive oil by oral gavage, 2 times, at an interval of 24 hours. Five male and five female rats served as positive controls and received 15 mg/kg cyclophosphamide monohydrate in sterile water (10 mL/kg) as a single intraperitoneal injection. The vehicle control and Almega PL-treated rats were observed twice daily for clinical signs and mortality on days 1 and 2 and once on day 3. Positive control rats were observed twice for clinical signs and mortality on day 1 and once on day 2. Body weights of rats were recorded on days 1, 2, and 3, except for the body weights of positive control animals which were recorded on days 1 and 2. All rats were anesthetized with isoflurane and killed 23 to 24 hours following the second treatment of Almega PL. After killing, 1 femur was removed from either side of the body after clearing the musculature. Femur heads were trimmed to expose the marrow canals, femur bone marrow was flushed, collected in centrifuge tubes, and smears were prepared and stained. Prepared slides were scored (minimum of 2000 polychromatic erythrocytes [PCE] from each rat) for the incidence of micronuclei in immature erythrocytes or PCEs and the proportion of immature erythrocytes among total erythrocytes (number of PCE divided by number of total red blood corpuscle [RBC]) was determined for each rat by counting at least 200 to 256 erythrocytes.
Statistical Analyses
Maximum-tolerated dose oral toxicity study
For the MTD oral toxicity study, group means and standard deviations were calculated for male and female body weights. Statistical analysis was performed by 2-way analysis of variance (ANOVA) followed by Bonferroni post hoc analysis.
Subchronic oral toxicity study
For all quantitative variables like functional observation, parameters (neuromuscular observations, motor activity, body weight, and body temperature) were tested for normality and homogeneity of variances within the group before performing a 1-factor ANOVA modeling by treatment groups. When the data were found to be heterogeneous, a suitable transformation was performed. In the case of nonparametric data, the Kruskal-Wallis test was used when the variances were significantly different. Comparison of means between treatment groups and control group was done using Dunnett test when the overall treatment “F” test was found significant. The data pertaining to male and female rats were evaluated separately. All analyses and comparisons were evaluated at the 5% (P = 0.05) level.
Chromosome aberration assay
Statistical analysis of the experimental data was carried out using validated SYSTAT Statistical package ver. 12.0. Data were analyzed for proportions of aberrant metaphases in each sample excluding gaps as aberrations were analyzed. Pooled data from each test concentration and the positive control were compared with the vehicle control using Fisher exact test. All analysis and comparisons were evaluated at 5% (P = 0.05) level.
In vivo micronucleus assay
The statistical analysis of the experimental data was carried out using validated copies of the SYSTAT Statistical package ver. 12.0. All quantitative variables like change in net body weight were tested for normality (Shapiro-Wilk test) and homogeneity (Levene test) of within group variance before performing ANOVA. For percentages, data were normalized using square root transformation before ANOVA. Comparison of means between the control and the treatment groups was done using Dunnett test where “F” test was found to be significant in ANOVA. All analyses and comparisons were evaluated at 5% (P < 0.05) level.
Results
Maximum-Tolerated Dose Oral Toxicity Study
No mortalities occurred during the study. No abnormal clinical signs were observed in any of the animals. Body weight increases were observed in all groups over time as shown in Figure 1. However, when compared to the control group, no statistically significant differences between groups were identified. No gross pathological findings were observed in any animals at study termination. Under the conditions of this study, it is concluded that Almega PL is safe and tolerated in rats at doses up to 2500 mg/kg, the highest dose tested in this study.
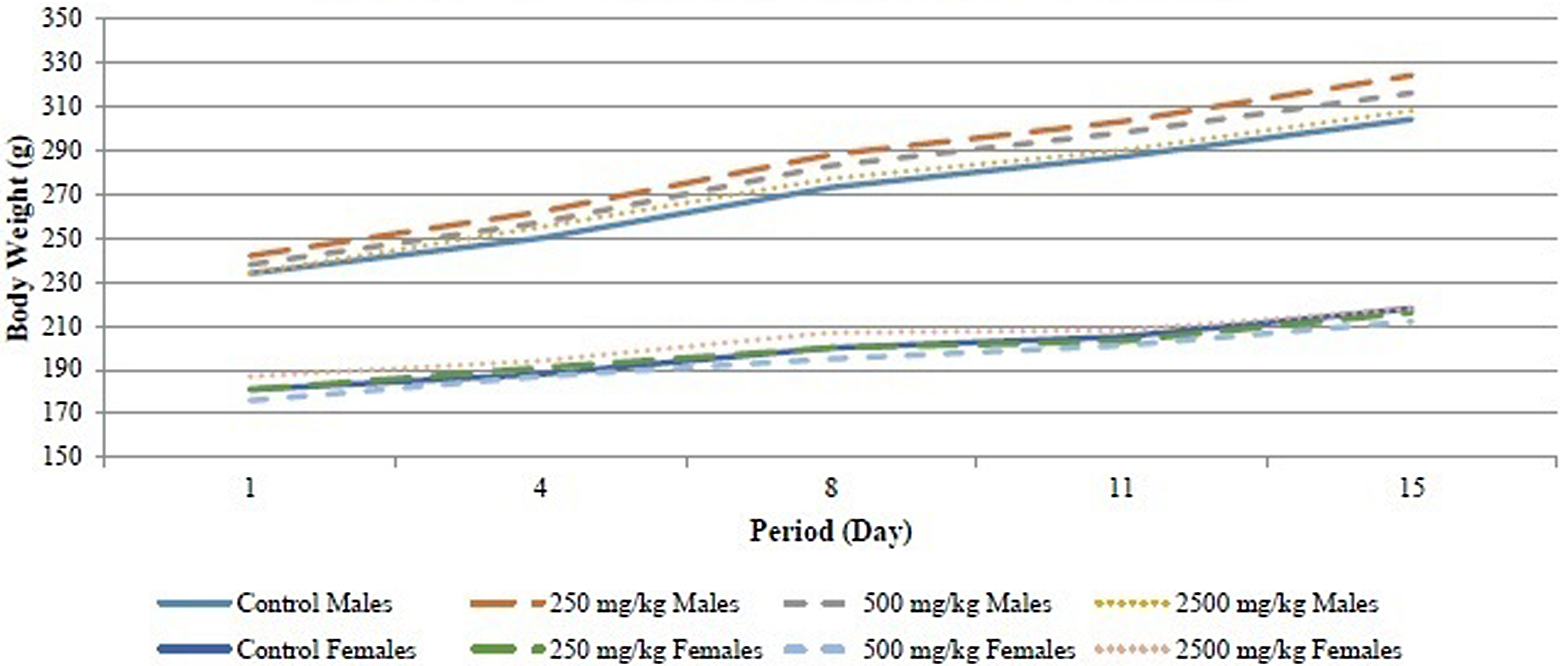
Mean body weights of male and female rats during the 14-day study.
Subchronic Oral Toxicity Study
No mortalities or treatment-related clinical signs were observed at any of the doses tested. Dark-colored feces were observed in all rats of the high-dose group. The dark-colored feces were attributed to the high chlorophyll content of Almega PL and considered a treatment-related nonadverse effect. No statistically significant changes were observed in food consumption for any treated group as compared to the control group. No treatment-related changes were observed in body weights at any of the tested doses in either sex during the entire treatment period (Figure 2). The results of ophthalmological examinations were normal for all of the study animals.
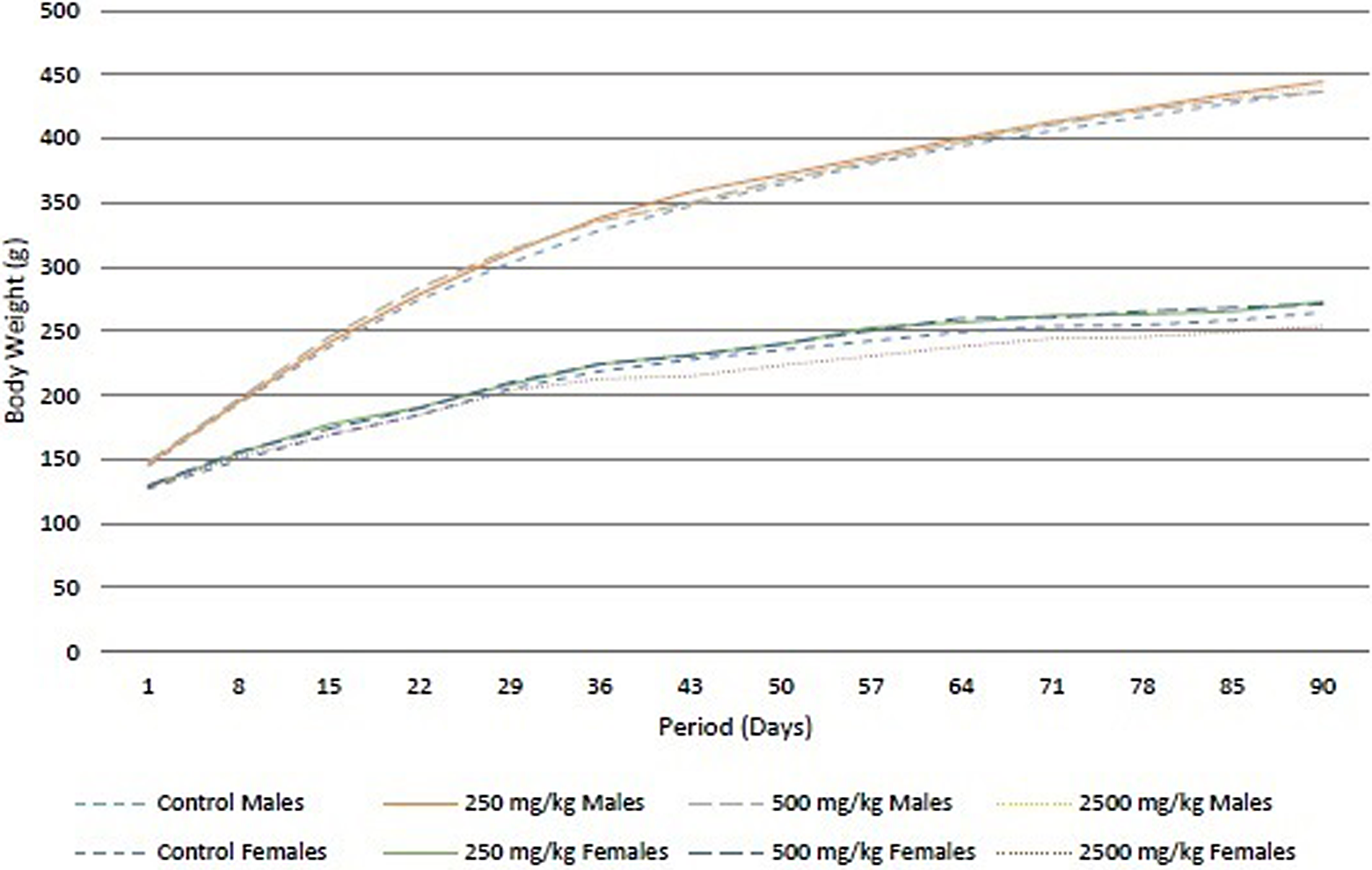
Mean body weights of male and female rats during the 90-day study.
Neurological examinations including functional tests were conducted on both male and female rats and were generally negative. Statistically significant differences were identified for forelimbs grip strength in mid-dose males and low- and high-dose females, hindlimbs foot splay in mid- and high-dose females, body temperature in high-dose males and low- and high-dose females, and motor activity in low- and mid-dose females (Table 5). However, these changes were considered to be incidental as either the changes were of minimal magnitude or not dose dependent.
Selected Results From the Functional Observation Battery.a
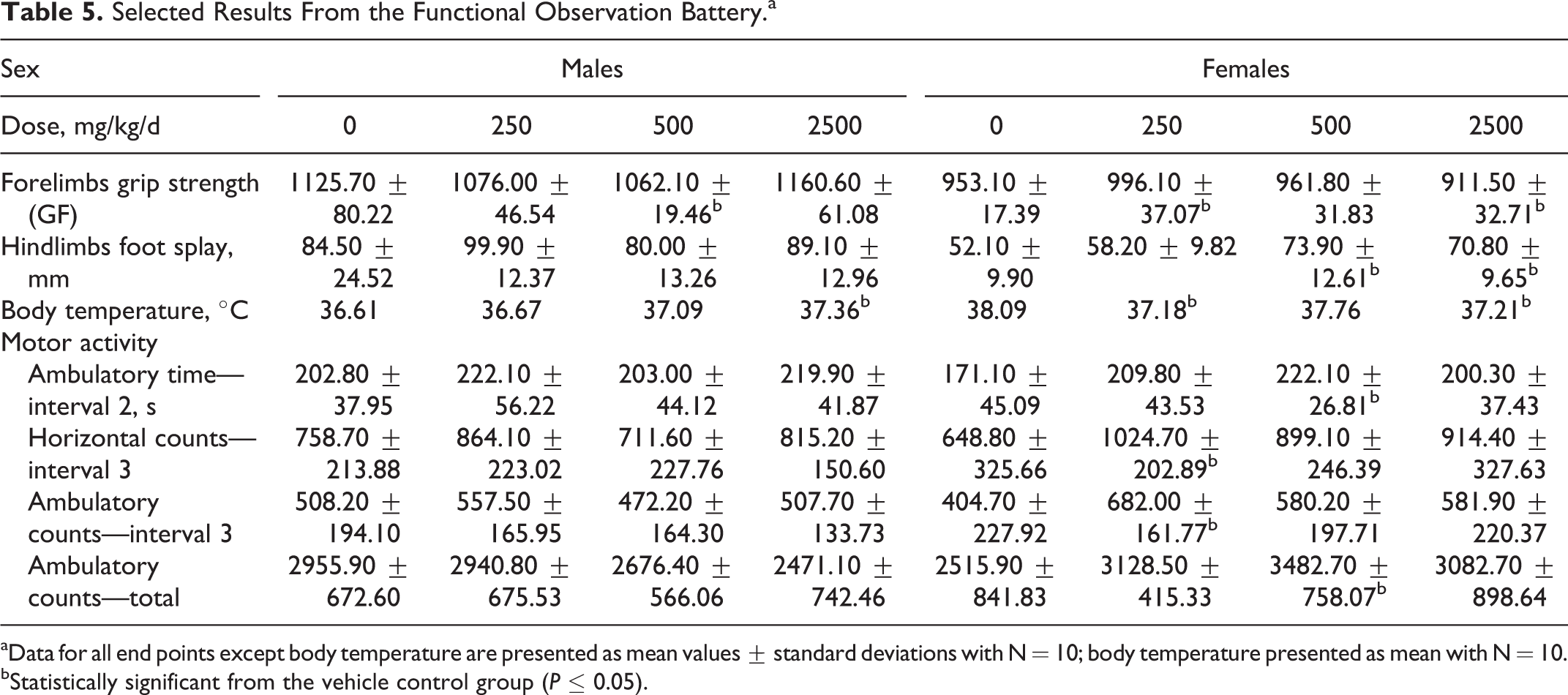
aData for all end points except body temperature are presented as mean values ± standard deviations with N = 10; body temperature presented as mean with N = 10.
bStatistically significant from the vehicle control group (P ≤ 0.05).
Data from the hematology, coagulation, and clinical chemistry analyses performed on day 91 are presented in Tables 6 and 7 for males and females, respectively. Statistically significant changes in hematology parameters were seen in high-dose males including hematocrit (HCT), mean corpuscular hemoglobin concentration (MCHC), and mean platelet volume (MPV); MPV was also statistically different in mid-dose males. In female rats, statistically significant hematological changes including RBC, HCT, mean corpuscular hemoglobin, MCHC, and MPV were seen in the mid- and high-dose groups. These changes were considered incidental due to the lack of a dose–response relationship and/or the changes being within the physiological range. For coagulation parameters, a decrease in activated partial thromboplastin time was seen in mid-dose males, and an increase in prothrombin time was seen in high-dose females. These values were considered incidental due to the lack of a dose response and the minor magnitude of the changes. Clinical chemistry values including blood urea nitrogen (BUN), alanine aminotransferase (ALT), total cholesterol (TChol), calcium (Ca), and potassium (K) were statistically different in the high-dose males when compared to controls. The BUN, aspartate aminotransferase, ALT, γ-glutamyl transpeptidase, TChol, total plasma protein, albumin, and albumin/globulin ratio were statistically different in high-dose females when compared to controls. Aspartate aminotransferase was also statistically different in low- and mid-dose females. The minimal increases in ALT activity (39% for males and 28% for females) and TChol levels (21% for males and 16% for females) seen at the highest dose (2500 mg/kg/d) were considered to be a test item-related, nonadverse effect due to the severity of change being limited and below the threshold of concern. In addition, the increased ALT activity was not accompanied by correlating histologic changes in the liver. All other statistically significant changes in clinical chemistry were of a low magnitude and within the physiological range and therefore considered toxicologically insignificant. No treatment-related changes were seen in any of the urine parameters analyzed in terminally killed animals.
Summary of Hematology, Coagulation, and Clinical Chemistry Values on Day 91—Males.a
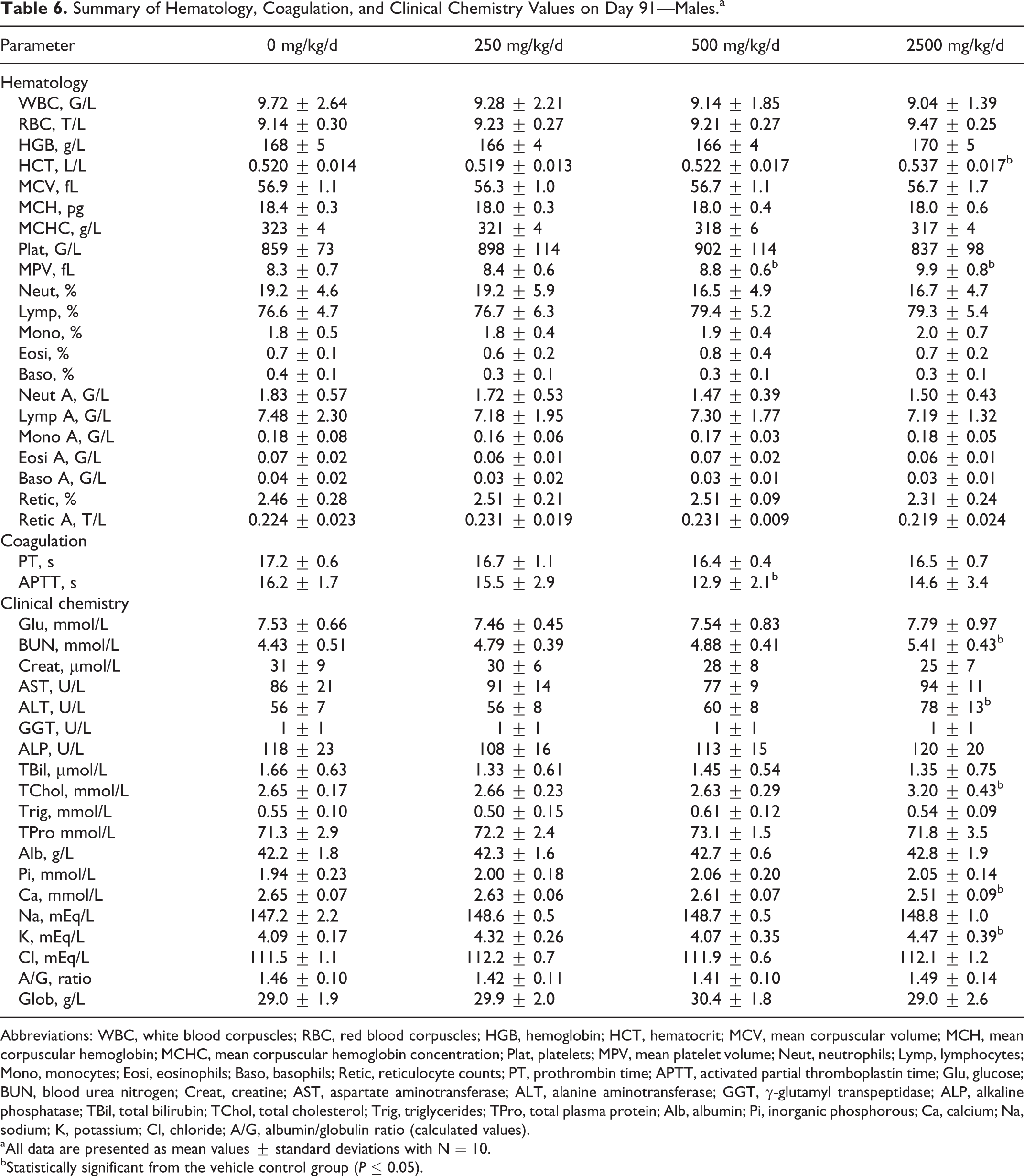
Abbreviations: WBC, white blood corpuscles; RBC, red blood corpuscles; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; Plat, platelets; MPV, mean platelet volume; Neut, neutrophils; Lymp, lymphocytes; Mono, monocytes; Eosi, eosinophils; Baso, basophils; Retic, reticulocyte counts; PT, prothrombin time; APTT, activated partial thromboplastin time; Glu, glucose; BUN, blood urea nitrogen; Creat, creatine; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, γ-glutamyl transpeptidase; ALP, alkaline phosphatase; TBil, total bilirubin; TChol, total cholesterol; Trig, triglycerides; TPro, total plasma protein; Alb, albumin; Pi, inorganic phosphorous; Ca, calcium; Na, sodium; K, potassium; Cl, chloride; A/G, albumin/globulin ratio (calculated values).
aAll data are presented as mean values ± standard deviations with N = 10.
bStatistically significant from the vehicle control group (P ≤ 0.05).
Summary of Hematology, Coagulation, and Clinical Chemistry Values on Day 91—Females.a
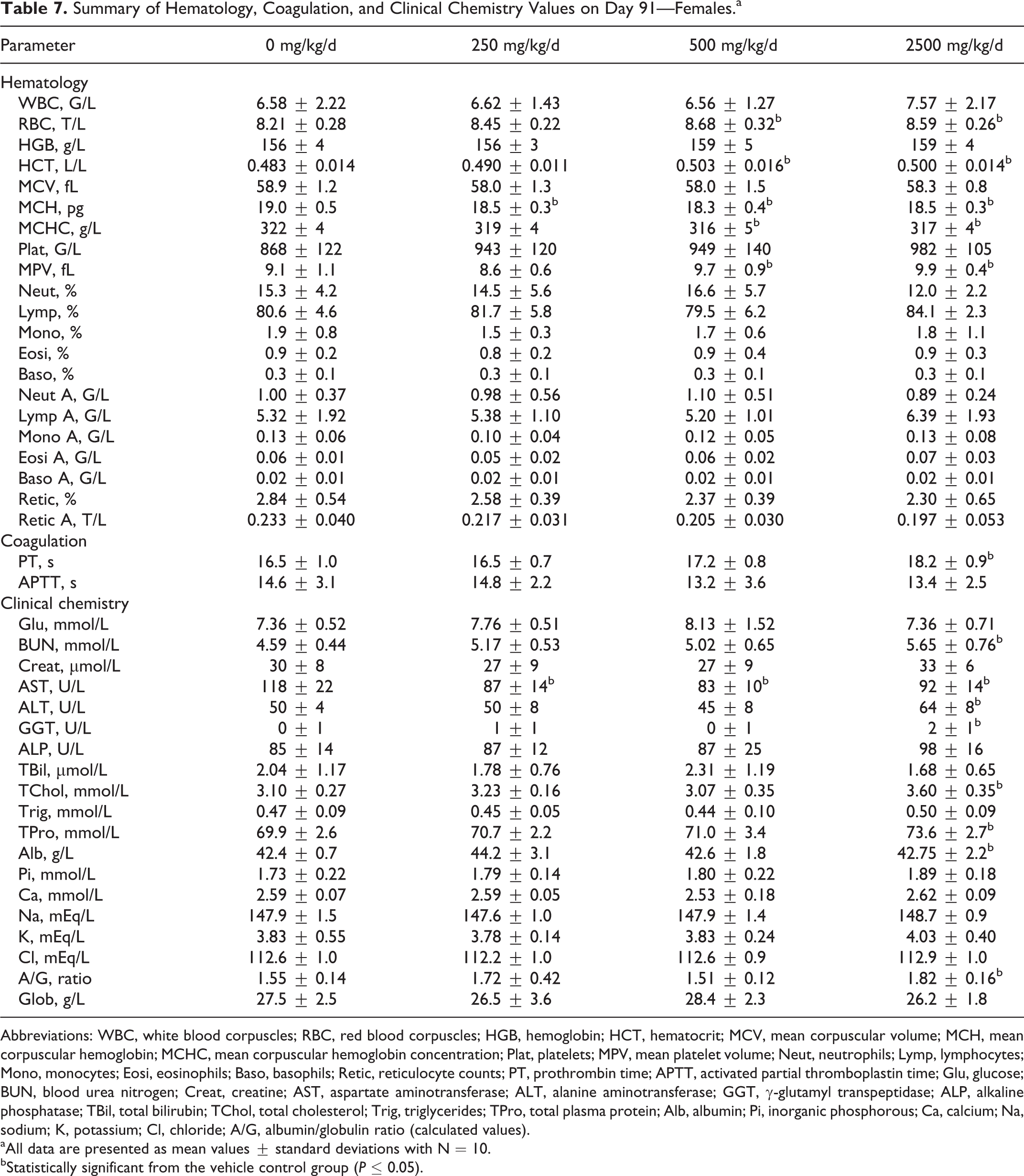
Abbreviations: WBC, white blood corpuscles; RBC, red blood corpuscles; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; Plat, platelets; MPV, mean platelet volume; Neut, neutrophils; Lymp, lymphocytes; Mono, monocytes; Eosi, eosinophils; Baso, basophils; Retic, reticulocyte counts; PT, prothrombin time; APTT, activated partial thromboplastin time; Glu, glucose; BUN, blood urea nitrogen; Creat, creatine; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, γ-glutamyl transpeptidase; ALP, alkaline phosphatase; TBil, total bilirubin; TChol, total cholesterol; Trig, triglycerides; TPro, total plasma protein; Alb, albumin; Pi, inorganic phosphorous; Ca, calcium; Na, sodium; K, potassium; Cl, chloride; A/G, albumin/globulin ratio (calculated values).
aAll data are presented as mean values ± standard deviations with N = 10.
bStatistically significant from the vehicle control group (P ≤ 0.05).
When compared to the control groups, no significant changes attributed to treatment were seen in organ weights or organ to body weight ratios for male or female animals at any dose level evaluated (Tables 8 and 9). Statistically significant changes were limited to increased thyroid weights (absolute and relative) in low- and high-dose males, increased adrenals weight in mid- (absolute) and high-dose (relative) females, and increased relative liver weights in high-dose females. These were considered incidental due to the absence of clinical signs and histopathological changes.
Summary of Terminal Fasting Body Weights and Absolute Organ Weights on Day 91.a
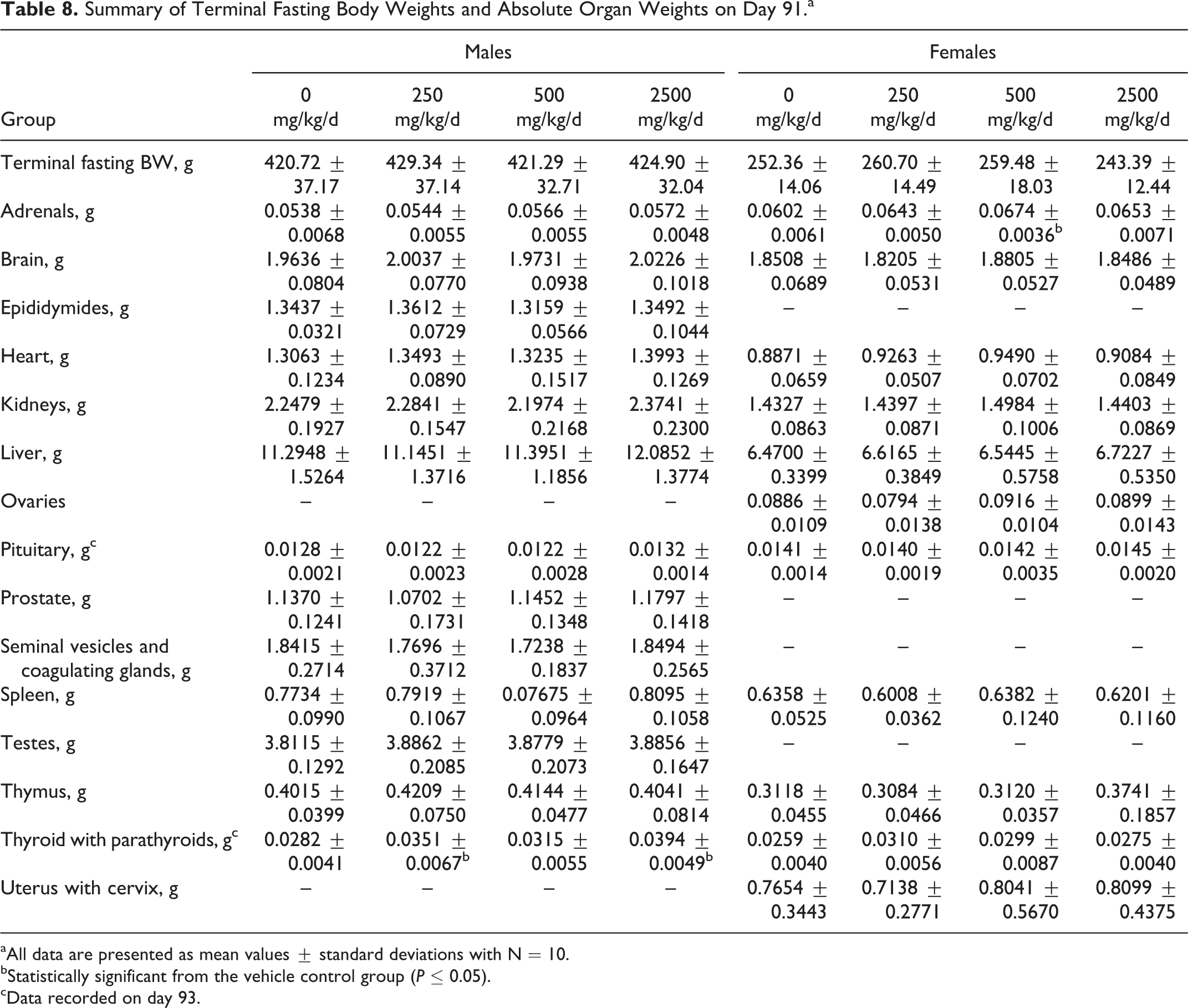
aAll data are presented as mean values ± standard deviations with N = 10.
bStatistically significant from the vehicle control group (P ≤ 0.05).
cData recorded on day 93.
Summary of Organ to Body Weight Ratios on Day 91.a
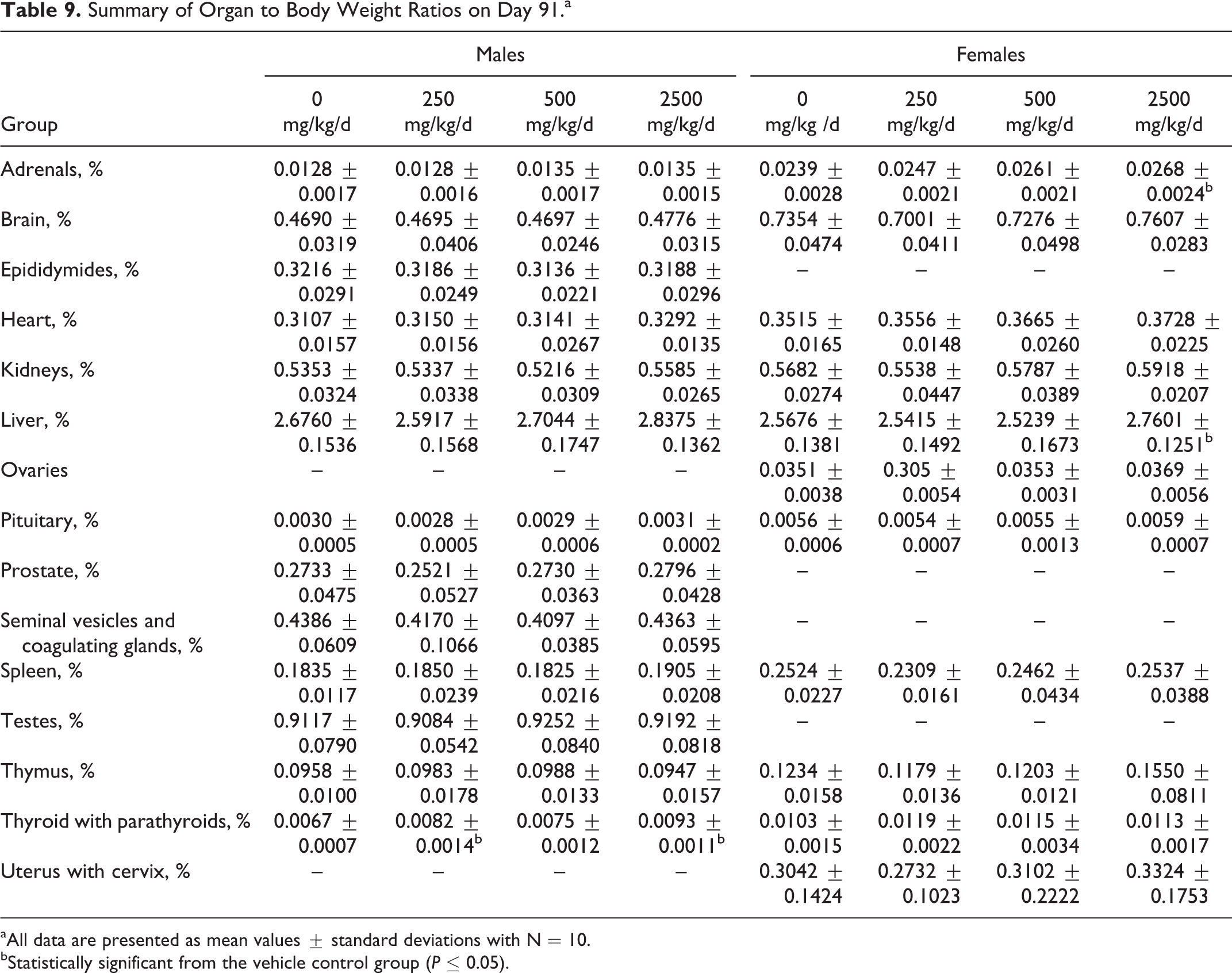
aAll data are presented as mean values ± standard deviations with N = 10.
bStatistically significant from the vehicle control group (P ≤ 0.05).
Compared to their respective control groups, no test item-related gross pathological lesions were evident in male or female rats at terminal necropsy (day 91). No incidental findings were seen in males. Incidental findings were found in females as dilated uterus noted across the tested groups (2 per test group including controls). The dilated uterus findings were confirmed microscopically (mild to severe dilation across all treatment groups including controls) and was not considered to be treatment related. All other microscopic findings (inflammatory foci in esophageal adnexal tissue, proteinaceous material in kidney tubules, harderian gland alterations, hepatocyte vacuolation, mesenteric lymph node erythrophagocytosis, mandibular lymph node plasmacytosis and/or erythrophagocytosis, unilateral ovarian cysts, prostate mononuclear cell infiltration, and rectum nematode) occurred at low frequencies in the control and high-dose groups and were not considered treatment related.
Under the conditions of this study, the “no observed adverse effect level” (NOAEL) for Almega PL would be 2500 mg/kg/d based on a lack of treatment-related effects at 2500 mg/kg/d, the highest dose evaluated in both male and female rats. However, because dose formulation analyses revealed consistently lower (approximately −20%) concentrations of the test item from the corresponding theoretical doses, the highest intended dose of 2500 mg/kg/d would result in an actual dose of 2000 mg/kg/d. Therefore, the NOAEL for the subchronic oral gavage administration of Almega PL in rats is determined as 2000 mg/kg/d.
Bacterial Reverse Mutation Assays
Genotypic characterization of tester bacteria indicated that the assays were valid because (1) all S typhimurium strains demonstrated the requirement of histidine amino acid for their growth and E coli strain WP2uvrA demonstrated the requirement of tryptophan amino acid for its growth, (2) TA98, TA100, and WP2uvrA demonstrated their typical resistance to ampicillin, (3) the presence of characteristic mutations like the rfa mutation was demonstrated by all S typhimurium strains by their sensitivity to crystal violet, (4) The uvrA mutation in the E coli strain and the uvrB mutation in the S typhimurium strains were demonstrated by their sensitivity to UV light, (5) all of the tester strains produced spontaneous revertant colonies which were within the frequency ranges of historical control data, and (6) the numbers of revertant colonies in the positive control groups were greater than 3 times the number of revertant colonies in the corresponding negative controls.
The summarized results for experiments I and II are presented in Tables 10 and 11, respectively. Slight precipitation of Almega PL was visually observable in all test strains both with and without S9 mix at concentrations of 1581 μg/plate and higher in experiment I and 1880 μg/plate and higher in experiment II. However, this slight precipitation did not interfere with the scoring of the bacterial background lawn intensity. No toxicity was observed up to the highest concentration of 5000 μg/plate for experiment I or II, as the intensity of the bacterial background lawn as well as the mean number of revertant colonies were comparable to the DMSO control. For both experiment I and II, no positive mutagenic responses were observed up to the top dose of 5000 μg/plate in any of the tester strains both in the presence and absence of metabolic activation. It is concluded that all criteria for the bacterial reverse mutation assays are valid and that under the conditions of this assay, Almega PL was not mutagenic at any of the doses tested.
Summary of Results of Initial Mutation Assay.
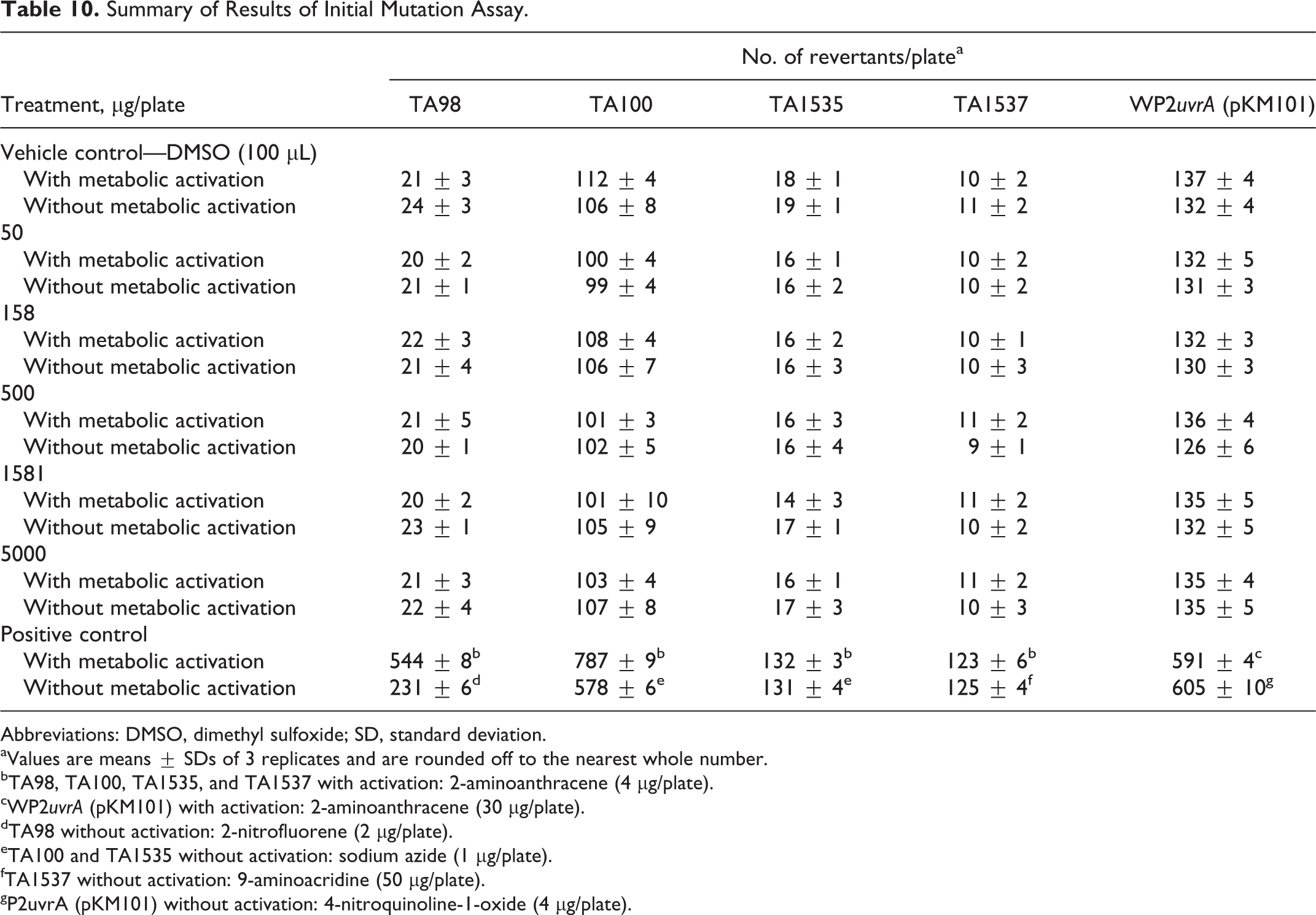
Abbreviations: DMSO, dimethyl sulfoxide; SD, standard deviation.
aValues are means ± SDs of 3 replicates and are rounded off to the nearest whole number.
bTA98, TA100, TA1535, and TA1537 with activation: 2-aminoanthracene (4 µg/plate).
cWP2uvrA (pKM101) with activation: 2-aminoanthracene (30 µg/plate).
dTA98 without activation: 2-nitrofluorene (2 µg/plate).
eTA100 and TA1535 without activation: sodium azide (1 µg/plate).
fTA1537 without activation: 9-aminoacridine (50 µg/plate).
gP2uvrA (pKM101) without activation: 4-nitroquinoline-1-oxide (4 µg/plate).
Summary of Results of Confirmatory Mutation Assay.
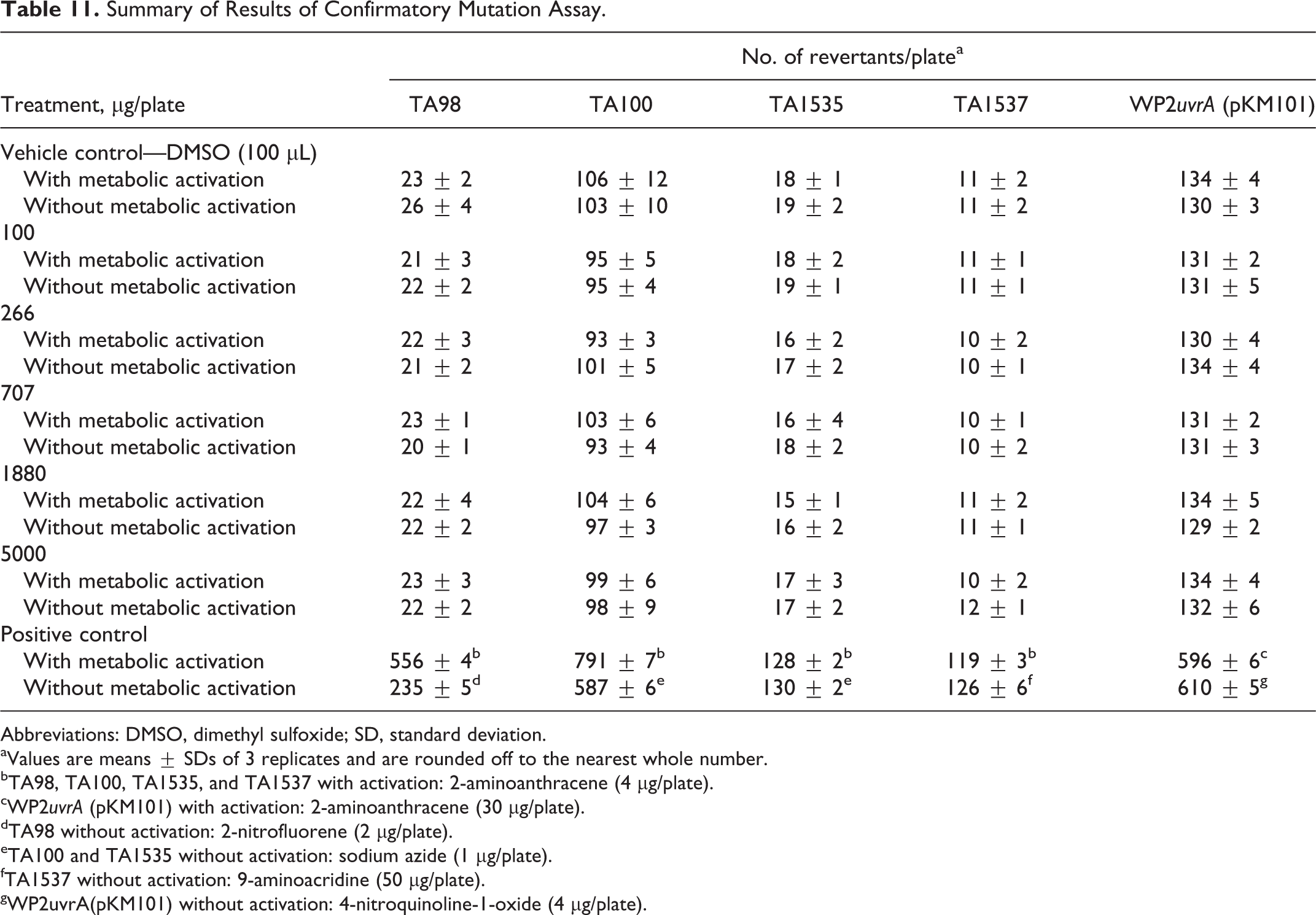
Abbreviations: DMSO, dimethyl sulfoxide; SD, standard deviation.
aValues are means ± SDs of 3 replicates and are rounded off to the nearest whole number.
bTA98, TA100, TA1535, and TA1537 with activation: 2-aminoanthracene (4 µg/plate).
cWP2uvrA (pKM101) with activation: 2-aminoanthracene (30 µg/plate).
dTA98 without activation: 2-nitrofluorene (2 µg/plate).
eTA100 and TA1535 without activation: sodium azide (1 µg/plate).
fTA1537 without activation: 9-aminoacridine (50 µg/plate).
gWP2uvrA(pKM101) without activation: 4-nitroquinoline-1-oxide (4 µg/plate).
Chromosome Aberration Assay
The results for the chromosome aberration assay are presented in Table 12. After 4 hours of exposure and in the presence of metabolic activation, cytotoxicity was seen at 19 μg/mL, the highest concentration tested. Similarly, cytotoxicity was seen at the highest doses tested in the absence of metabolic activation following 4 hours of exposure (40 μg/mL) and 21 hours of exposure (7 μg/mL). For all doses and times, and in the absence or presence of metabolic activation, no positive incidence of chromosomal aberrations was observed following treatment with Almega PL. The in vitro mammalian chromosome aberration assay is considered valid because (1) the incidence of aberrations in the vehicle control cultures is in the range of in-house historical control data and (2) the positive control substances produced a significant increase in the incidence of aberrations compared to the respective vehicle control.
Summary Results of Chromosome Aberration Assay With and Without Metabolic Activation.a
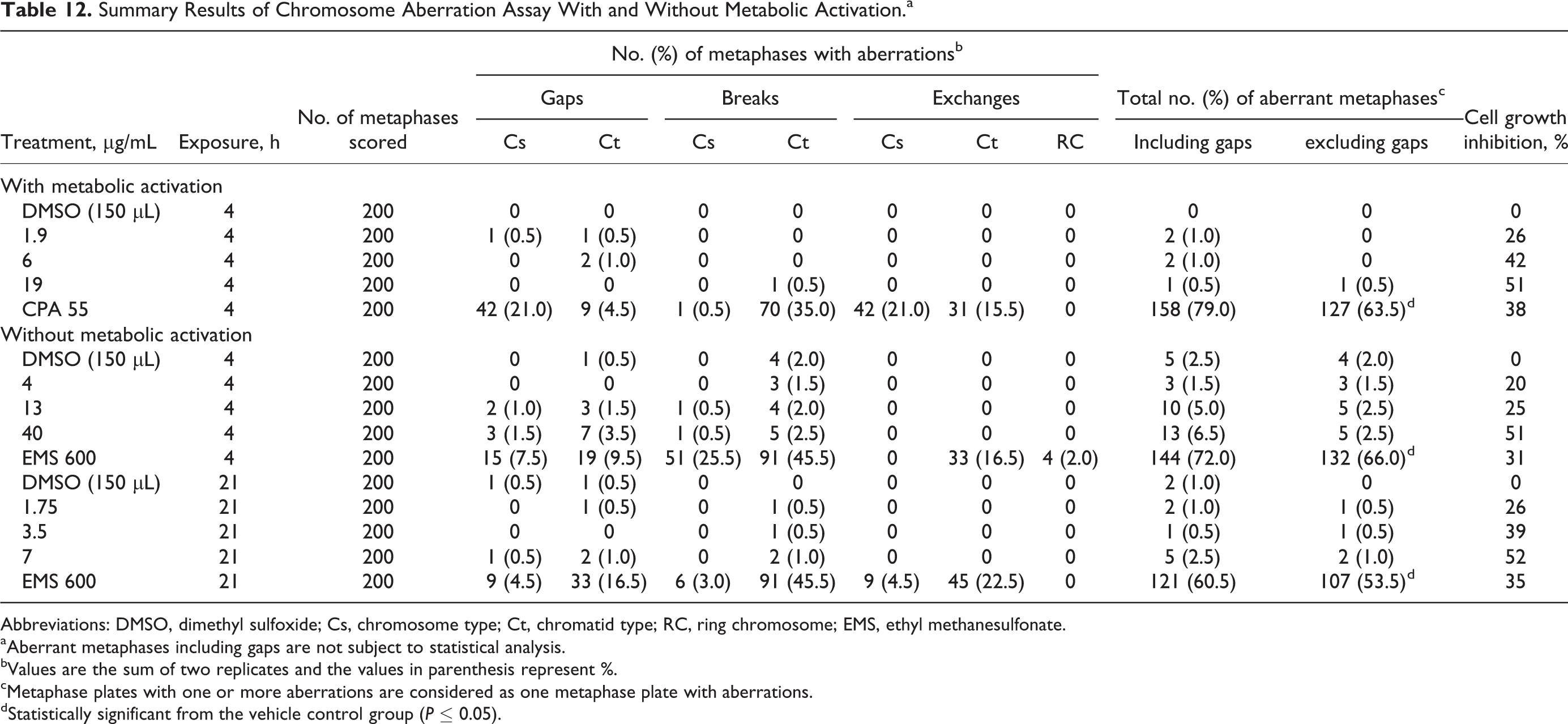
Abbreviations: DMSO, dimethyl sulfoxide; Cs, chromosome type; Ct, chromatid type; RC, ring chromosome; EMS, ethyl methanesulfonate.
aAberrant metaphases including gaps are not subject to statistical analysis.
bValues are the sum of two replicates and the values in parenthesis represent %.
cMetaphase plates with one or more aberrations are considered as one metaphase plate with aberrations.
dStatistically significant from the vehicle control group (P ≤ 0.05).
In Vivo Micronucleus Assay
No mortality was seen in any of the tested animals, and body weight gains were comparable to vehicle controls. No treatment-related clinical signs were observed in any dose groups except for dark-colored feces seen at 1000 and 2000 mg/kg/d on days 2 and 3. The dark-colored feces were attributed to the high chlorophyll content of Almega PL and considered a treatment-related nonadverse effect. Data for the total RBC ratio and micronucleus incidence are summarized in Table 13. No significant increases in micronucleated PCE were observed between mice treated with Almega PL and the vehicle control. A significant decrease in PCE:total RBC ratio was seen in males at 500 and 2000 mg/kg/d and for combined sex at 500 and 1000 mg/kg/d. However, the values were comparable to historical data ranges and therefore determined to be nonbiologically relevant. The test was considered valid because (1) the incidence of micronucleated PCE in the vehicle control group was within the historical control range, (2) the positive control animals demonstrated a significant increase in micronucleated PCE compared to controls, and (3) at the end of the study, the weight variation in animals was minimal and did not exceed ±20% of the mean weight of each sex.
Summary of PCE:Total RBC Ratio and Micronucleus Incidence.
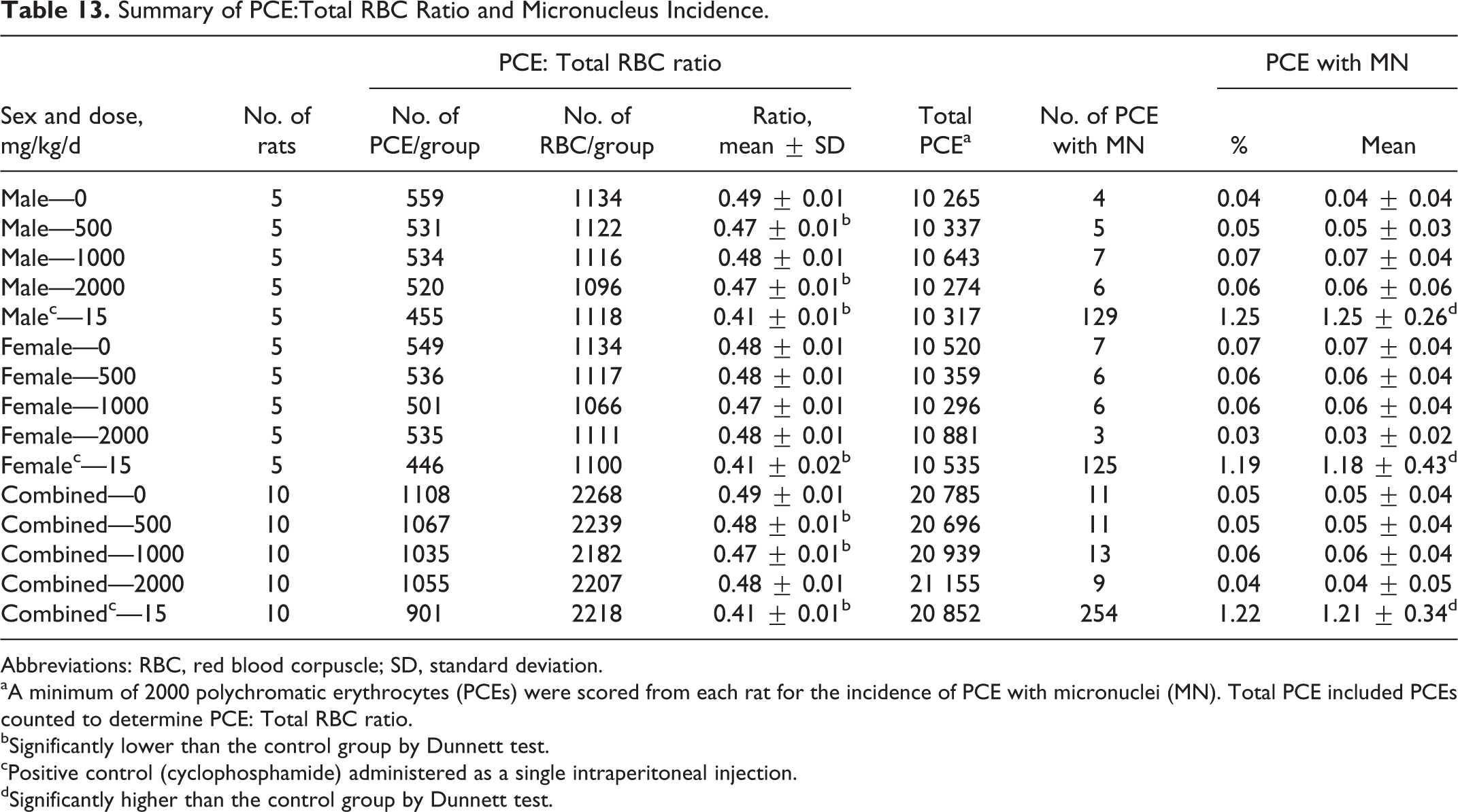
Abbreviations: RBC, red blood corpuscle; SD, standard deviation.
aA minimum of 2000 polychromatic erythrocytes (PCEs) were scored from each rat for the incidence of PCE with micronuclei (MN). Total PCE included PCEs counted to determine PCE: Total RBC ratio.
bSignificantly lower than the control group by Dunnett test.
cPositive control (cyclophosphamide) administered as a single intraperitoneal injection.
dSignificantly higher than the control group by Dunnett test.
Discussion
Omega-3 fatty acids possess significant health benefits for brain function and development as well as cardiovascular conditions. 17,18 Currently most of these fatty acids isolated for human consumption are sourced from fish. However, due to a limited amount of global fish stocks, alternative sources of fatty acids are needed. Potential substitutes include oil extracts from plants, krill, microalgae, and microalgae-like protists.
Of the potential substitutes, algae may be one of the more promising alternatives. Due to their small size, algae are easy to grow on a large scale. Algae do not require freshwater or arable land and are capable of producing significant amounts of fatty acids even under adverse conditions. 18,19 In the past, the potential for using microalgal oils on a larger scale has been limited by cost, extraction, and purification methods. 17 At present, various strains of algae are being used for the production of fatty acids for human consumption. For example, Schizochytrium sp and/or Crypthecodinium cohnii are included in infant formulas, food additives, cosmetics, and pharmaceutical products. 17
Nannochloropsis oculata cultures have been used as a source of live food for rotifers in marine fish hatcheries and as crustacean feed for over 30 years. 20 No data were located in the published literature indicating a history of consumption of N oculata-derived oil products by humans, either in conventional food or dietary supplements. Currently, Almega PL is not regulated by the US Food and Drug Administration and has not been determined Generally Recognized As Safe (GRAS) as a food ingredient. However, EPA has been evaluated and approved in GRAS notifications and food additive petitions for use as an ω-3 fatty acid. In addition, the FDA has stated that dietary supplements should not recommend or suggest in their labeling a daily intake exceeding 2 g EPA and DHA. 9
Previously, the acute and subchronic oral toxicities of N oculata biomass were evaluated in male and female Sprague-Dawley rats. 1 The oral median lethal dose of N oculata was greater than 12 g/kg and the subchronic toxicity was 6 g/kg/d. However, this study focused only on the potential hepatotoxicity and nephrotoxicity of N oculata. Furthermore, the study was limited to the evaluation of N oculata biomass and did not evaluate the potential toxicity of oil extracted from the biomass. Therefore, the studies reported herein provide new and relevant data on the safety of N oculata (as Almega PL) oil extract.
The oral administration of Almega PL at concentrations of up to 2000 mg/kg/d was well-tolerated by both male and female rats in the 14-day MTD study and the GLP-compliant 90-day subchronic toxicity study. Almega PL had no adverse effects on health or growth as measured by survival, appearance, behavior, food consumption, body weight, or body weight gain. No treatment-related effects were seen for ophthalmology, neurological examinations, hematology, coagulation, clinical chemistry, gross pathology, absolute and relative organ weights, or histopathology in animals of any dose group. Although statistical significance was noted for several end points in the 90-day subchronic toxicity study, these were not considered to be biologically relevant or attributable to Almega PL because these effects were (1) incidental or sporadic in nature, (2) within the range of historical control data for rats of this age and strain, and/or (3) occurred in the absence of corroborative clinical or histopathological changes.
The battery of GLP-compliant in vitro and in vivo genotoxicity studies provide overall support that Almega PL is not genotoxic. In 2 independent bacterial reverse mutation assays, Almega PL was negative for mutagenicity at levels up to 5000 μg/plate using S typhimurium strains TA98, TA100, TA1535, and TA1537 and E coli strain WP2uvrA, both with and without metabolic activation. Almega PL was not clastogenic as evaluated by the in vitro chromosome aberration assay in the CHO cell line at concentrations of 1.9, 6, or 19 μg/mL and 4, 13, or 40 μg/mL with or without metabolic activation, respectively, for 4 hours exposure as well as concentrations of 1.75, 3.5, or 7 μg/mL without metabolic activation for 21 hours of exposure. When evaluated in the in vivo erythrocyte micronucleus assay, Almega PL was not clastogenic or aneugenic. No treatment-related structural and/or numerical chromosomal damage was seen in immature erythrocytes in rats administered Almega PL at doses of 500, 1000, or 2000 mg/kg/d.
The available human toxicity data for Almega PL are limited to a randomized, crossover study conducted in 10 male patients (aged 18-45 years) which compared the appearance of fatty acids in the blood plasma of healthy humans after consumption of a high fat meal followed by either Almega PL or a commercially available krill oil supplement (NKO; Neptune Technologies & Bioresources Inc, Quebec, Canada). 4 Both oils were encapsulated in gelatin soft capsules manufactured by Catalent (Eberbach, Germany). The capsules provided approximately 1.5 g EPA and no DHA for Almega PL or 1.02 g EPA and 0.54 g DHA for krill oil. Both oils were well tolerated. Plasma samples collected before and 0.5 to 10 hours after capsule administration indicated time-dependent increases in EPA for both oils with the concentration of EPA being higher with algal oil than with krill oil at multiple time points.
Described here is the nonclinical safety assessment (and limited clinical assessment) of the algal oil Almega PL, which is being developed as a dietary supplement in the United States, providing a vegetarian and sustainable source of ω-3 fatty acids in the diet, primarily EPA. Based on the results of the GLP-compliant nonclinical studies, the NOAEL for the algal oil is 2000 mg/kg/d, which will provide a reasonable margin of safety (96-fold) over the suggested maximum oral intake of 1250 mg/d Almega PL (20.8 mg/kg/d for a 70-kg individual).
Footnotes
Acknowledgments
This work was supported by Qualitas Health Ltd.
Author Contributions
M. Kagan contributed to conception and design, acquisition, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. D. Sullivan contributed to analysis and interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. S. Gad contributed to analysis and interpretation, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. C. Ballou contributed to interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael L. Kagan is an employee of Qualitas Health Ltd.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
