Abstract
Our recent retrospective analysis of data, collected from 29 Tg.rasH2 mouse carcinogenicity studies, determined how successful the strategy of choosing the high dose for the 26-week studies was based on the estimated maximum tolerated dose (EMTD) derived from earlier 28-day dose range finding (DRF) studies conducted in CByB6F1 mice. Our analysis demonstrated that the high doses applied at EMTD in the 26-week Tg.rasH2 studies failed to detect carcinogenic effects. To investigate why the dose selection process failed in the 26-week carcinogenicity studies, the initial body weights, terminal body weights, body weight gains, food consumption, and mortality from the first 4 weeks of 26-week studies with Tg.rasH2 mice were compared with 28-day DRF studies conducted with CByB6F1 mice. Both the 26-week and the earlier respective 28-day studies were conducted with the exact same vehicle, test article, and similar dose levels. The analysis of our results further emphasizes that the EMTD and subsequent lower doses, determined on the basis of the 28-day studies in CByB6F1 mice, may not be an accurate strategy for selecting appropriate dose levels for the 26-week carcinogenicity studies in Tg.rasH2 mice. Based on the analysis presented in this article, we propose that the Tg.rasH2 mice and not the CByB6F1 mice should be used in future DRF studies. The Tg.rasH2 mice demonstrate more toxicity than the CByB6F1 mice, possibly because of their smaller size compared to CByB6F1 mice. Also, the Tg.rasH2 males appear to be more sensitive than the female Tg.rasH2 mice.
Keywords
Introduction
The 2-year rodent carcinogenicity assays involving conventional rats and mice have been conducted for over 3 decades. As an alternative to the 2-year mouse carcinogenicity bioassays, 26-week short-term carcinogenicity bioassays were approved using transgenic mouse strains, including Tg.rasH2. 1 The Tg.rasH2 model, which can be used for both genotoxic and non-genotoxic compounds, has gained popularity and its use has increased over the years. Over the last decade, 26-week transgenic Tg.rasH2 mouse studies have replaced more than 75% of all mouse carcinogenicity studies. 2 –4 The Tg.rasH2 model predicts neoplastic findings relevant to human cancer risk assessment, produces fewer nonbiologically significant neoplastic outcomes, and is thus often is preferable to a 2-year mouse study. 5
The purpose of the 26-week studies conducted in Tg.rasH2 mouse is to evaluate the carcinogenic potential of a test article. Usually, 5- and 28-day dose range finding (DRF) studies are conducted in CByB6F1 mice, the wild type littermates of Tg.rasH2 mice, to establish the doses used in the 26-week Tg.rasH2 carcinogenicity study. The CByB6F1 mouse has the same genetic background as the Tg.rasH2 mouse except for the omission of the transgene (Tg) element. For this reason, the nontransgenic wild type littermates CByB6F1 mice are used in earlier DRF studies. Another reason that the CByB6F1 mice are used in earlier DRF studies is that the Tg.rasH2 mice cost about double than the CByB6F1 mice. 6 The purpose of the 5-day study is to estimate the doses for the 28-day study, and the purpose of the 28-day study is to estimate the doses for the 26-week study. Following a 28-day study, the dose selection and particularly the estimated maximum tolerated dose (EMTD) is determined based on criteria similar to the ones used in the conventional 2-year mouse studies. Parameters taken into account include, but are not limited to, mortality, body weight gains (BWGs), clinical findings, clinical pathology findings, and gross and histopathology findings. A draft protocol for the 26-week study is then submitted to the Carcinogenicity Assessment Committee (CAC) at the Food and Drug Administration (FDA) for the approval of the dose levels and the study design.
Based on our recent publication on the evaluation of dose selection process of our set of 26-week carcinogenicity studies, 7 we concluded that the EMTD in the high dose of both sexes was overestimated for several reasons including:
– the drop in % BWG was greater than 10% in the high-dose groups of both sexes,
– the decrease in % BWG in the high-dose group of both sexes was not due to decreased food consumption (FC). There was an increase in FC in all test article–treated groups of both sexes when compared to controls, and
– the % mortality in the high-dose groups of both sexes was considerably higher than their control counterparts, and the mortality was not due to carcinogenic effects of the test article.
In addition, tumors as the cause of death (COD) decreased in the test article–treated dose groups of both sexes compared to the control groups, and this decrease was highest in the high-dose groups of both sexes. The incidence of undetermined COD was highest in the high-dose groups of both sexes and lowest in the controls of both sexes. Thus, the high-dose groups in both sexes did not serve their purpose in these studies, which is to assess the carcinogenic potential of the test article. Based on our analysis, we made several recommendations as clarified in our article. 7
In this article, we further emphasize the fact that the EMTD, determined on the basis of the 28-day studies conducted in CByB6F1 mice, may not be accurate in predicting appropriate dose levels for the 26-week carcinogenicity studies in Tg.rasH2 mice and modifications should be made in the dose selection process of the 26-week carcinogenicity studies. To come to this conclusion, we evaluated additional data that included comparison of initial body weight (IBW), terminal body weight (TBW), BWG, FC, and mortality in the first 4 weeks of Tg.rasH2 mice used in the 26-week carcinogenicity studies and CByB6F1 wild type mice used in the prior 28-day DRF studies with exact same vehicle, test article, and similar dose levels.
Methods and Materials
Tg.rasH2 and CByB6F1 mice used in these studies were obtained from Taconic Biosciences, Germantown, NY. The primary criterion for selection of studies being evaluated was that both the 26-week carcinogenicity studies and prior 28-day DRF studies must have been conducted at our facility, which qualified 24 studies for the analysis. Another important criterion was that the vehicle (formulation) and the test article–treated dose levels must be similar in these studies. If, in any of the studies, an individual dose group was not similar in the 26-week and 28-day studies, that study was eliminated from the analysis. Based on the above criteria, the qualified studies along with qualified dose groups are tabulated below:

This table indicates that while most of the vehicle, low, and mid doses used in 28-day DRF studies conducted in CByB6F1 mice were maintained until termination in the 26-week studies, approximately 66% of the high doses (EMTD) in these 28-day DRF studies were eliminated due to early mortality and/or severe toxicity. Of these 24 studies, only 33% of the high doses originally selected in the 28-day DRF were maintained until termination in the subsequent 26-week study and thus included in this analysis. In the remaining 8 high-dose groups used in the subsequent 26-week studies, a carcinogenicity effect was not detected. Therefore, in our previous article, 7 we stated that the high dose should be eliminated altogether and the mid and low doses that showed dose responsive carcinogenicity should be maintained and the studies should be conducted with only 2 dose groups.
In the 28-day DRF and 26-week Tg.rasH2 studies there were 4 dose groups, designated as vehicle, low, mid, and high. In each group/sex of DRF studies, there were 10 CByB6F1 mice, whereas in the Tg.rasH2 studies, there were 25 Tg.rasH2 mice for each group/sex. Individual 28-day DRF or 26-week carcinogenicity studies followed the same study design. For the 28-day DRF studies, IBW were collected on day 1 and mice were weighed weekly thereafter for the next consecutive 4 weeks. After 4 weeks, the body weights were collected just prior to necropsy as TBW. Food consumption was recorded weekly for individual animals from day 1 until the termination of the study. For the 26-week carcinogenicity studies, the IBW were collected on day 1 and mice were weighed weekly thereafter for the next consecutive 13 weeks. After 13 weeks, the body weights were collected biweekly until the termination of the study at 26 weeks. FC was recorded weekly for individual animals from day 1 until the termination of the study at 26 weeks.
For the purpose of this article, the parameters compared included IBW, TBW, BWG, FC, and mortality collected for 28-day DRF studies conducted in CByB6F1 mice and the same data collected for the first 28 days of the 26-week carcinogenicity studies conducted in Tg.rasH2 mice. A
Results
Tables 1 and 2 present the comparative analysis between 28-day DRF studies conducted in CByB6F1 mice and up to 28 days of data collected from the 26-week carcinogenicity studies conducted in Tg.rasH2 mice for vehicle, low, mid, and high doses for each sex, respectively. These tables include the number of studies, and the mean +/− standard deviation (SD) for IBW, TBW, BWG, FC, and mortality for each of the males dose groups (VM, LM, MM, and HM) and females (VF, LF, MF, and HF). Also shown in the table are percentage differences for IBW, TBW, BWG, FC, and mortality in Tg.rasH2 mice when compared with the CByB6F1 mice. Important findings of this comparative analysis between the CByB6F1 and Tg.rasH2 mice for the vehicle, low, mid, and high doses are summarized below.
Male Mice—Comparative Analysis of Data Between 28-day DRF Studies Conducted in CByB6F1 Mice and up to 28 Days of Data Collected from the 26-Week Carcinogenicity Studies.
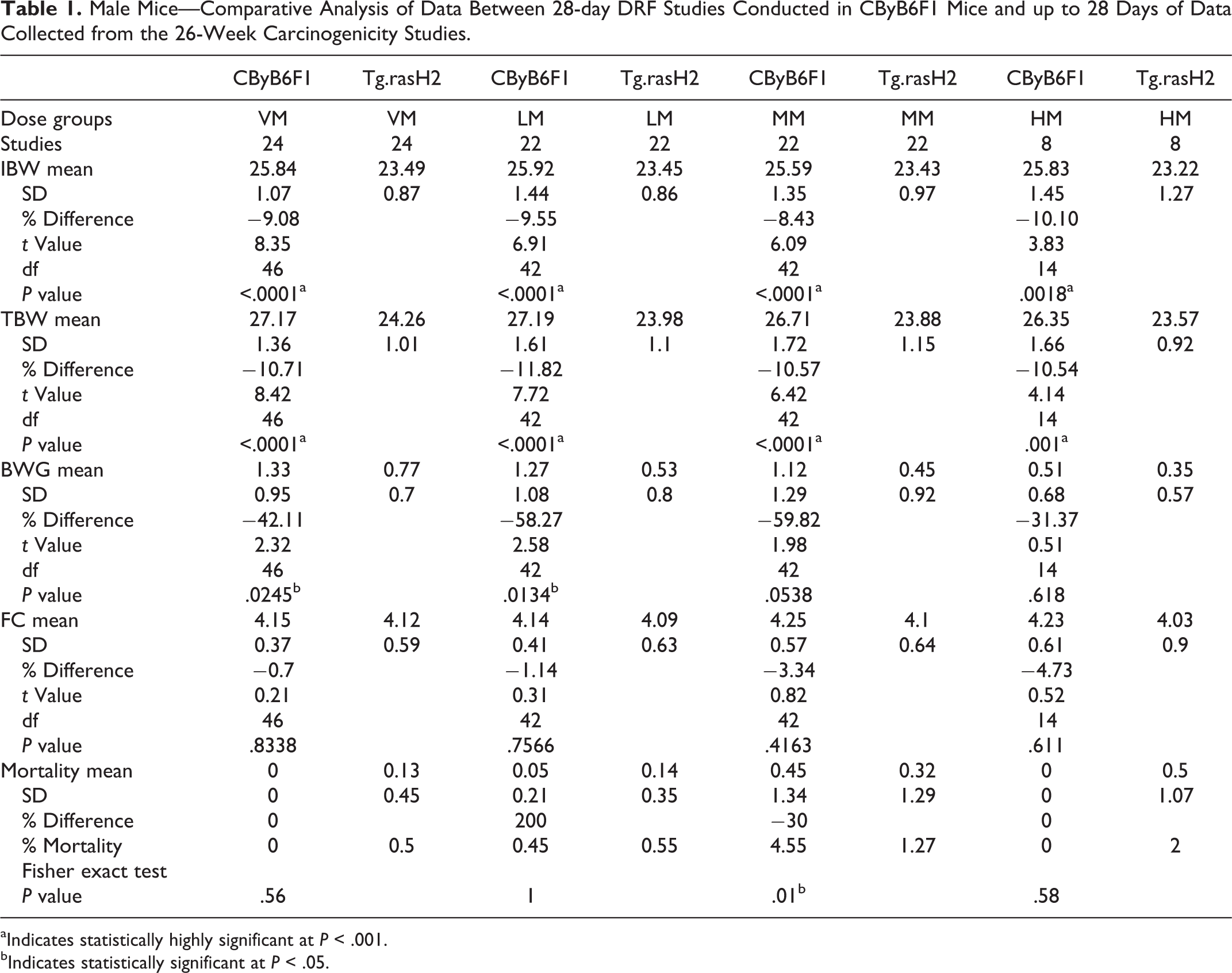
aIndicates statistically highly significant at
bIndicates statistically significant at
Female Mice—Comparative Analysis of Data Between 28-Day DRF Studies Conducted in CByB6F1 Mice and up to 28 Days of Data Collected from the 26-Week Carcinogenicity Studies.
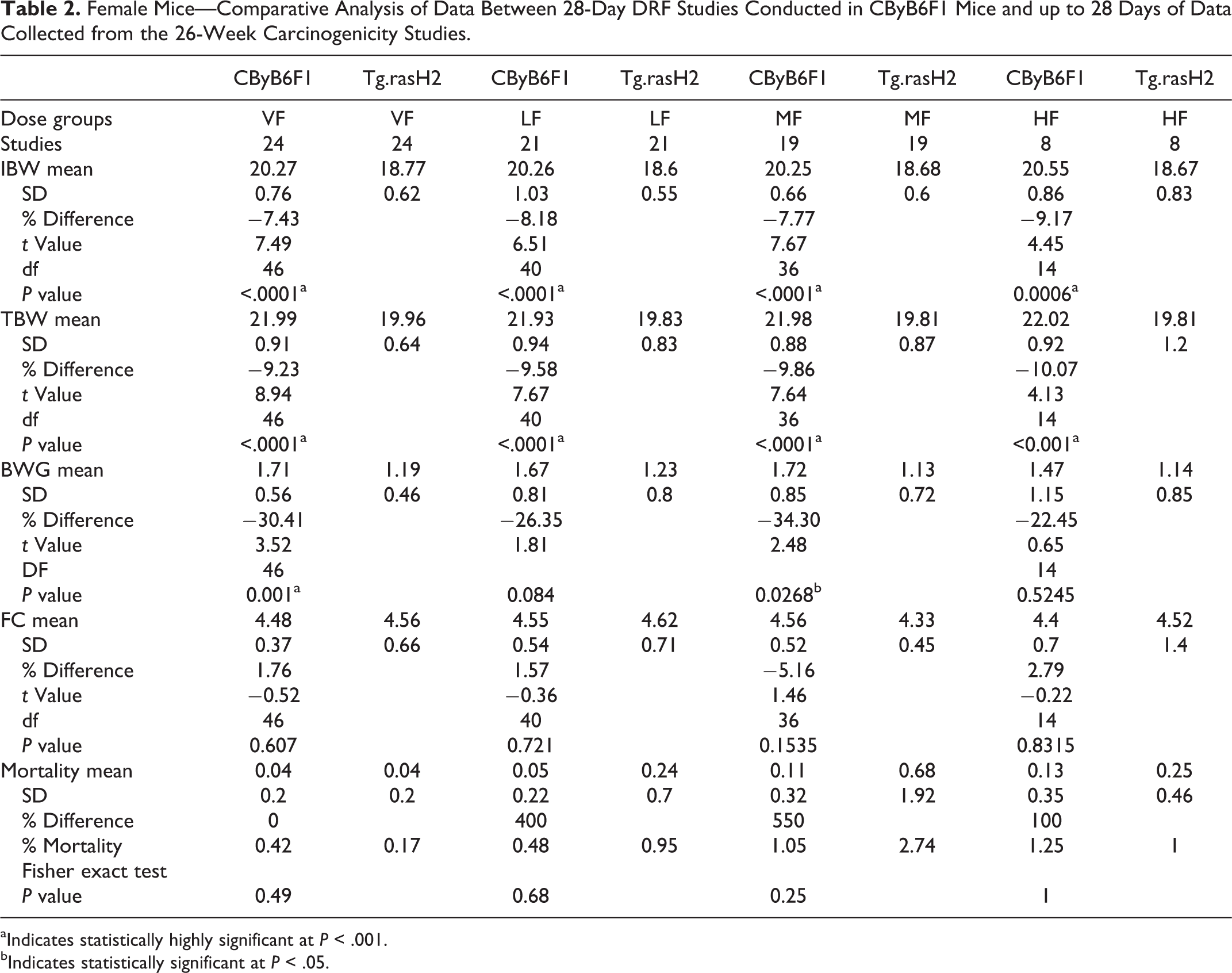
aIndicates statistically highly significant at
bIndicates statistically significant at
The IBW and TBW of Tg.rasH2 mice were about 10% lower than the CByB6F1 mice. The striking differences were that the BWG% was lower in Tg.rasH2 mice when compared to CByB6F1 mice. In male Tg.rasH2 mice, these differences were lower by 42.11%, 58.27%, 59.82%, and 31.37%, for VM, LM, MM, and HM, respectively (Figure 1). In the female Tg.rasH2 mice for the VF, LF, MF, and HF, the BWG% differences were lower than the CByB6F1 mice by 30.41%, 26.35%, 34.30%, and 22.45%, respectively (Figure 2). It is important to note that even in VM and VF, the BWG% was much lower in Tg.rasH2 mice than the CByB6F1 mice, more so in males than the females. There were no noteworthy changes in FC and mortality.
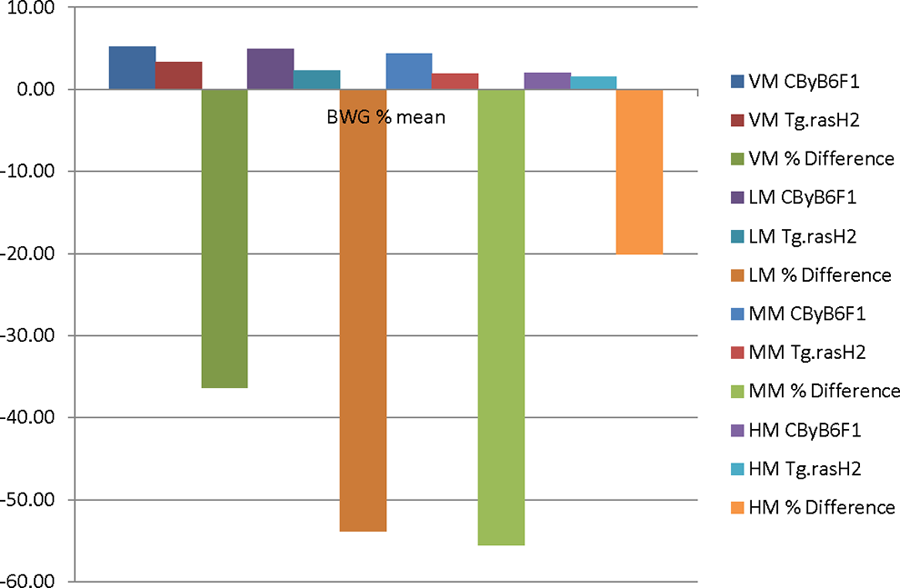
Bar diagram illustrating the BWG % comparison between CByB6F1 and Tg.rasH2 male mice. BWG indicates body weight gain.
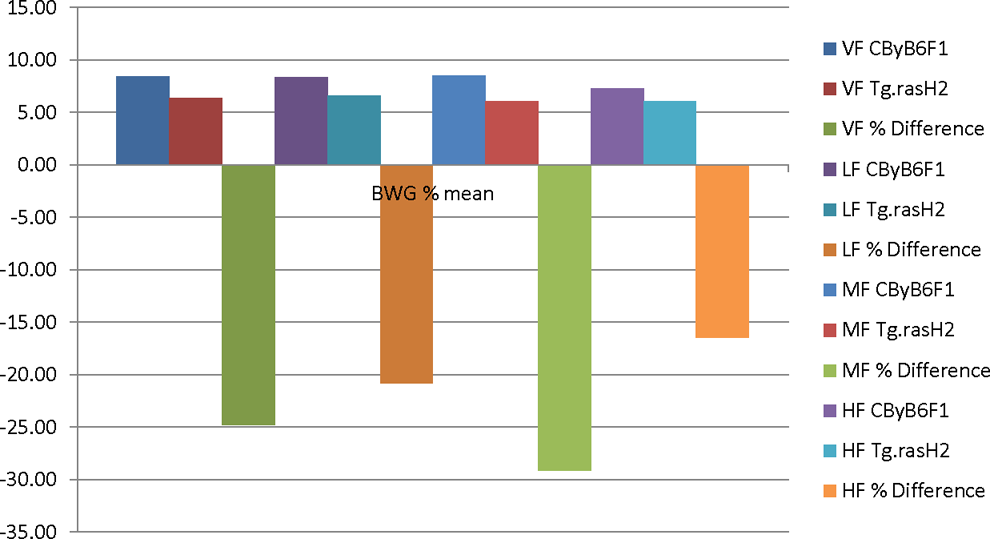
Bar diagram illustrating the BWG % comparison between CByB6F1 and Tg.rasH2 female mice. BWG indicates body weight gain.
Discussion and Conclusions
The results of our retrospective analysis showed that the IBW, as well as TBW, of the Tg.rasH2 mice were approximately 10% lower than the wild type CByB6F1 counterparts, which is expected and similar to what has been described before, as Tg.rasH2 mice are generally 10% to 15% smaller in IBW at the start of the experiment. 9 –11
The most important outcome of our analysis is that for the exact same vehicle, test article and similar doses, the BWG% of the Tg.rasH2 male mice were lower than the CByB6F1 mice by 42.11%, 58.27%, 59.82%, and 31.37%, in VM, LM, MM, and HM dose groups, respectively. Similarly, in the female Tg.rasH2 mice for the vehicle, low, mid, and high doses, the BWG% differences were lower by 30.41%, 26.35%, 34.30%, and 22.45%, respectively. It should be emphasized here that the BWG% differences for males and females in all 4 dose groups were lower by more than 10% in Tg.rasH2 mice when compared to the CByB6F1 mice. It is also important to note that the BWG% differences in the HM and HF dose groups were lowest compared to other dose groups, most likely because fewer number of studies (8 studies) were available for comparison. Given the fact that for all studies for which the analysis was performed, not only the environmental, husbandry practices, diet, and water provided were similar, but the vehicle and test articles provided were identical with similar doses. Therefore, we feel that these differences in the BWG% for the Tg.rasH2 mice are excessive and clearly greater than what otherwise would have been caused by simply lower IBW. Our analysis also shows that the CByB6F1 mice are prone to gain more weight compared to Tg.rasH2 mice. This higher BWG% in CByB6F1 mice used in DRF studies to estimate the MTD for the Tg.rasH2 mouse carcinogenicity studies plays a prominent role in not accurately estimating the MTD for the carcinogenicity studies.
The BWG% is compared between 28 days of the DRF studies in CByB6F1 mice with the first 28 days of the 26-week Tg.rasH2 studies, as the DRF studies are terminated at 28 days. We assume that these BWG% differences will continue at least to the same extent if not more for the remaining period of the 26-week studies. Overall, our findings also show that the male mice were more susceptible than the female mice as the BWG% differences were higher in the male mice than the females, which is in line with our previously published findings. 7 Also in males, the BWG% differences were lower in Tg.rasH2 mice than the CByB6F1 mice in a dose responsive manner, at least for groups VM, LM, and MM. However, in the female mice, such dose responsiveness was not evident.
The data from the 28-day DRF studies conducted in CByB6F1 mice are used in determining the doses for the 26-week Tg.rasH2 studies, particularly the EMTD. While the CByB6F1 mice are well accepted for these DRF studies and have been used in these studies at least for the last 10 years, we did not find any data published with similar comparison between the CByB6F1 and Tg.rasH2 mice. The definition of MTD that is well accepted defines MTD as the highest dose of a test article without excessive toxicity and is estimated from earlier DRF studies and then applied to the carcinogenicity study. This MTD is such that when given for the duration of the carcinogenicity study, it should be high enough to elicit minimal signs of toxicity but should not cause >10% decrease in the body weights compared to concurrent controls, and should not shorten the animal’s normal longevity, or unduly compromise normal well-being of the animal except for the effects of carcinogenicity. 12 We recently evaluated the dose selection process for the 26-week Tg.rasH2 studies and recommended that the current high-dose group set at MTD in 26-week Tg.rasH2 studies should be completely eliminated because it did not contribute to the overall interpretation of the outcome of the studies. 7 We also determined that the males are more sensitive than females to doses at MTD in 26-week Tg.rasH2 studies and therefore proposed that the maximum dose applied in Tg.rasH2 studies should be ½ of the EMTD and the next dose should be at ¼ of EMTD in males. Similarly, maximum dose in females should be about approximately 2/3 of the EMTD, as derived from the 28-day DRF studies conducted in CByB6F1 mice 7 and the next should be 1/3 of the EMTD. Thus, these carcinogenicity studies should be conducted with 1 vehicle and 2 test article doses because the currently recommended highest dose group does not serve any purpose in the study since it fails to detect a potential carcinogenic effect. This recommendation will reduce the number of animals in the carcinogenicity studies by 50 per study. The findings presented in this article further confirm our previous findings, 7 and we propose that the differences in the BWG% between the Tg.rasH2 and CByB6F1 mice must be taken into account while determining the doses for the 26-week Tg.rasH2 studies. Thus, based on our analysis, we now recommend that Tg.rasH2 mice should be used in 28-day DRF studies instead of CByB6F1 mice, which will help in predicting the EMTD for the 26-week studies more accurately, because both DRF and 26-week studies will be conducted in the same mouse strain. We also believe that the EMTD, as derived currently by comparing the findings of DRF studies performed in CByB6F1 mice, are not accurate because the EMTD is overestimated when applied to 26-week Tg.rasH2 mice in carcinogenicity studies, due to differences in the mice strains and weights.
The Tg.rasH2 mice demonstrate more toxicity, but fewer spontaneous tumors, than the CByB6F1 mice, possibly because of their smaller size compared to CByB6F1 mice. Also, the Tg.rasH2 males appear to be more sensitive than the female Tg.rasH2 mice. Thus, in conclusion, we recommend that both 28-day DRF and 26-week carcinogenicity studies should be conducted in Tg.rasH2 and that the EMTD derived from the Tg.rasH2 DRF studies should be taken at its face value. Based on our analyses, conducting 28-day DRF studies in Tg.rasH2 instead of CByB6F1 mice could result in potentially more, but not excessive, toxicity, fewer spontaneous tumors, greater sensitivity (particularly in the males), and <10% decrease in BW or BWG compared to the control, leading to a more accurate selection of EMTD for the 26-week carcinogenicity studies.
On the other hand, if the 28-day DRF studies are to be conducted in CByB6F1 mice as a cost savings due to the higher cost of the Tg.rasH2 mice, 6 then the EMTD derived from 28-day DRF studies conducted in CByB6F1 mice should be modified as recommended in our previous publication 7 and the highest dose group should be eliminated in the 26-week carcinogenicity studies and these studies should be conducted with 1 vehicle and 2 test article–treated dose groups/sex/study. Performing the DRF studies in CByB6F1 mice will certainly keep the cost of the studies down; however, our analysis also showed that only 33% of the high doses in the CByB6F1 DRF studies qualified as the high doses in the 26-week carcinogenicity studies with Tg.rasH2 mice. Even in these 33% qualified studies with the same high doses, a carcinogenicity effect was not detected.
Footnotes
Author Contribution
M. Paranjpe contributed to conception and design, acquisition, analysis, and interpretation and drafted the manuscript. J. Belich contributed to design and analysis and drafted and critically revised the manuscript. T. Vidmar contributed to design and analysis and critically revised the manuscript. R. Elbekai contributed to conception and interpretation and drafted and critically revised the manuscript. M. McKeon contributed to conception and interpretation and drafted and critically revised the manuscript. C. Brown contributed to conception and interpretation and drafted and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
