Abstract
Sulfur mustard (SM) is believed to be a major threat to civilian populations because of the persistent asymmetric threat by nonstate actors, such as terrorist groups, the ease of synthesis and handling, and the risk of theft from stockpiles. The purpose of this study was to establish mechanisms of acute tracheal injury in rats induced by SM using histopathologic, immunohistochemical, and biochemical parameters. Male rats (Sprague-Dawley) were anesthetized, intratracheally intubated, and exposed to 2 mg/kg of SM. Animals were euthanized 6-, 24-, 48-, and 72-hour postexposure, and intracavitary blood samples from the heart and tracheal tissues were collected. Exposure of rats to SM resulted in rapid tracheal injury, including tracheal epithelial cell shedding, focal ulceration, and abundant lymphocyte invasion of the submucosa. There was also evidence of a large number of apoptotic cells in the epithelium and submucosa, the serum levels of tumor necrosis factor α, interleukin 1β (IL) 1β, IL-6, and γ-glutamyl transferase peaked at 24 hours, and the serum levels of lactate dehydrogenase, glutathione peroxidase, and thiobarbituric acid reactive substance peaked at 6 hours. The SM exposure also resulted in a loss of the cellular membrane, leakage of cytoplasm, fuzzy mitochondrial cristae, medullary changes in ciliated and goblet cells, and the nuclear chromatin appeared marginated in basal cells and fibroblasts. The results in the propylene glycol group were the same as the control group. These data demonstrated the histologic changes, inflammatory reactions, apoptosis, oxidative stress, and DNA damage following SM (2 mg/kg)-induced acute tracheal injury; the severity of changes was time dependent.
Introduction
In 1917, sulfur mustard (SM) was first used by the German military. Sulfur mustard was named for its odor. 1 Sulfur mustard has continuous corrosive effects, thus SM is referred to as the “king of toxic agents.” 2 In many parts of the world, SM is now reemerging as a major threat not only to troops but also to civilians. 3 According to statistical data, the respiratory tract, eyes, and skin were the target organs most affected by SM in the Iran–Iraq War. 4 Sulfur mustard has acute and chronic toxicity to the respiratory system that is more serious than toxicity to any other organ systems. 5
The epithelial lesions are accompanied by underlying edema and inflammatory cell infiltration. 6 Sulfur mustard is an alkylating agent causing single- and double-strand breaks in the DNA, and SM also reacts with many proteins in human tissues. 7 Thus, SM leads to a disordered cell metabolism, causing apoptosis or necrosis. After cutaneous SM exposure, widespread cell death of basal keratinocytes results in the clinical symptoms of massive inflammation, skin blistering, and ulceration. 8 The toxicity of SM is attributed to the lipophilic nature of SM, which allows SM to rapidly penetrate target tissues and alkylate proteins, lipids, and nucleic acids, resulting in DNA damage and cytotoxicity. 9 Respiratory tract lesions are the main source of morbidity and mortality after SM exposure. Few studies have investigated the pathophysiology of respiratory tract lesions, in particular the mechanisms involved in the inflammatory processes. 10 Moreover, no effective specific treatment is available for the respiratory lesions induced by the SM-induced respiratory lesions. 11
Experimental animal exposure data suggest that rats and guinea pigs subjected to intratracheal administration of SM (0.3 mg/kg [5 hours and 14 days]) mainly exhibit pathological changes characterized in the trachea as focal ulcers and detachment of tracheal epithelial lining from the basement membrane. 6 Thus, following exposure of rodents or pigs to SM by inhalation or intratracheal instillation, inflammatory cells are observed in the upper and lower respiratory tracts, which persist for weeks to months. 12,13 Similarly, in humans, increased numbers of neutrophils and eosinophils are evident in the lung even a decade after exposure. 14 The lesions observed in an SM-intoxicated animal model are consistent with clinical human observations and are relevant to the study of airway epithelial damage induced by SM. Vitamin E and glucocorticoid effects reduce the acute inflammatory cell influx and suppress collagen formation in lung tissues for protection against chemical-induced lung injury. 15 The aim of this study was to establish the mechanisms of acute tracheal injury induced by SM using histopathological, immunohistochemical, and biochemical parameters in rats.
Materials and Methods
Animal Model
Ethical approval of the study protocol was obtained from the Animal Research Ethics Committee of Weifang Medical University (Weifang, China). Seventy-two Sprague-Dawley rats (males, 280-300 g, 15 weeks old, SPF; purchased from the Experimental Animal Center of the Chinese PLA Military Academy of Medical Sciences, Beijing, China) were randomized as follows: SM group, n = 32; propylene glycol group, n = 32; and control group, n = 8. Sulfur mustard (purity >90%) was diluted by propylene glycol for later use. Subtegumental atropine (0.05 mg/kg of body weight) was administered, and 30 minutes later ketamine hydrochloride (100 mg/kg of body weight) was used as an anesthetic. The rats were then fixed in the supine position on the platforms so that the cold light source illuminated the necks of the rats. The epiglottises were elevated to expose the glottises using a self-made “L” hook. A 16# trocar (shaped to a blunt tip without a stylet) was placed into the trachea, and then a fine plastic pipe was then inserted into the trocar to complete the intubation. Sulfur mustard (0.1 mL per rat [approximately 2 mg/kg of body weight]) was administered to the SM group, propylene glycol (0.1 mL per rat; Zhiyuan Corporation, Tianjin, China) was administered to the propylene glycol group, and the control group was not treated.
Histopathologic Assessment of Tracheal Injury
The rat tracheal tissues were obtained 6, 24, 48, and 72 hours after the intratracheal injection of SM or propylene glycol (8 rats per designated time). Intraperitoneal pentobarbital (30 mg/kg body weight) was used as an anesthetic before surgery, after which all of the rats were killed. Specimens were fixed in 10% formalin for 24 hours, dehydrated, embedded, and sliced (4 µm serial sections using a RM 2235 slicing machine; Leica, Solms, Germany). Each specimen was cut into 15 sections (5 sections per group). After hemotoxylin and eosin (H&E) staining (H&E kit; Labest Biotech Co, Ltd, Beijing, China), sections were observed under a light microscope (BX51; Olympus, Tokyo, Japan) for histomorphologic analyses. A total of 1 mm3 of fresh specimens from all of the groups was obtained 72 hours after the intratracheal injection. The specimens were fixed in 3% glutaraldehyde for 24 to 48 hours, dehydrated, embedded, and dried. Specimens were made into ultrathin (70 nm) slices (UC7 ultrathin slicing machine; Leica). Sections were cleansed with water and soaked in a saturated aqueous solution of uranyl acetate (Zhenpu Biotech Co, Ltd, Shanghai, China). The sections were then cleaned with double-distilled water and soaked in lead citrate (Tianyuan Co, Ltd, Yingkou, China) solution. The ultrastructure of tracheal cells was observed using a Hitachi H-7500 transmission electron microscope (Hitachi, Tokyo, Japan).
Immunohistochemical Staining (Streptavidin Peroxidase)
Each specimen was cut into 15 sections (5 sections per group) and placed into EDTA (pH 8.5; Deye Co, Ltd, Changzhou, China) for antigen retrieval and then blocked by H2O2 and goat serum, respectively. After discarding the goat serum and dropping the rabbit antirat caspase 3 and rabbit antirat caspase 9, the sections were incubated overnight at 4°C. Coloration was achieved with 3,3-diaminobenzidin, and hematoxylin was used to finish the staining. The negative control group was carried out with the same steps as the above-mentioned description, but the rabbit antirat caspase 3 and rabbit antirat caspase 9 were replaced with phosphate-buffered saline. The streptavidin peroxidase kits were purchased from Bayer Corporation (Leverkusen, Germany).
Measurement of Serum Levels of Inflammatory Factors and Enzymes
Intracavitary puncture of the heart and collection of blood samples (2 mL per rat) from the rats in the SM and propylene glycol groups were performed at 6, 24, 48, and 72 hours, respectively. The samples were placed in a 37°C water bath for 1 hour, stored at 4°C overnight, and centrifuged (2000 g/min) for 10 minutes. Then, the supernatant was decanted into a sterile vial and kept at −80°C. The serum levels of tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), IL-6, and thiobarbituric acid reactive substances (TBARSs) were measured using a Versa Max enzyme-labeled instrument (Molecular Devices Corporation, Sunnyvale), and lactate dehydrogenase (LDH), glutathione peroxidase (GP), and γ-glutamyl transferase (GGT) were measured using a COBAS 8000 fully automatic biochemical immune analyzer (COBAS 8000 type; Roche, Basel, Switzerland). Enzyme-linked immunosorbent assay kits were purchased from the Shenzhen Kerunda Biotech Co, Ltd (Shenzhen, China). All of the processes were performed strictly according to the instructions provided by the manufacturers.
Statistical Analyses
Data analyses were carried out using SPSS 17.0 software (SPSS, Inc, Chicago, Illinois). All values are expressed as the mean ± standard deviation. Study groups were compared with the control group using a t test. A P < 0.05 (2 sided) was considered significant.
Results
Rat Tracheal Histologic Changes and Inflammatory Cells Infiltration (H&E Staining)
Morphologic assessment of tracheal injury
Histological changes in the trachea, partially shed tracheal epithelial cells, focal ulceration, deranged cilia, damaged continuity of the epithelium, increased glands in the lamina propria mucosae, and incrassated submucosa as well as a few inflammatory cells invading the submucosa were observed after 6 hours (Figure 1A). The delaminated area of the epithelium and submucosa increased, and small blood vessels were congested and invaded by a small number of inflammatory cells after 24 hours (Figure 1B). A large quantity of inflammatory cells was noted after 48 hours (Figure 1C). The deciduous area of the tracheal epithelial cells converged down through the submucosa or the outer layer, and an enlarged area of ulcers was observed after 72 hours (Figure 1D). The results in the propylene glycol group were the same as the control group (Figure 1E and F).
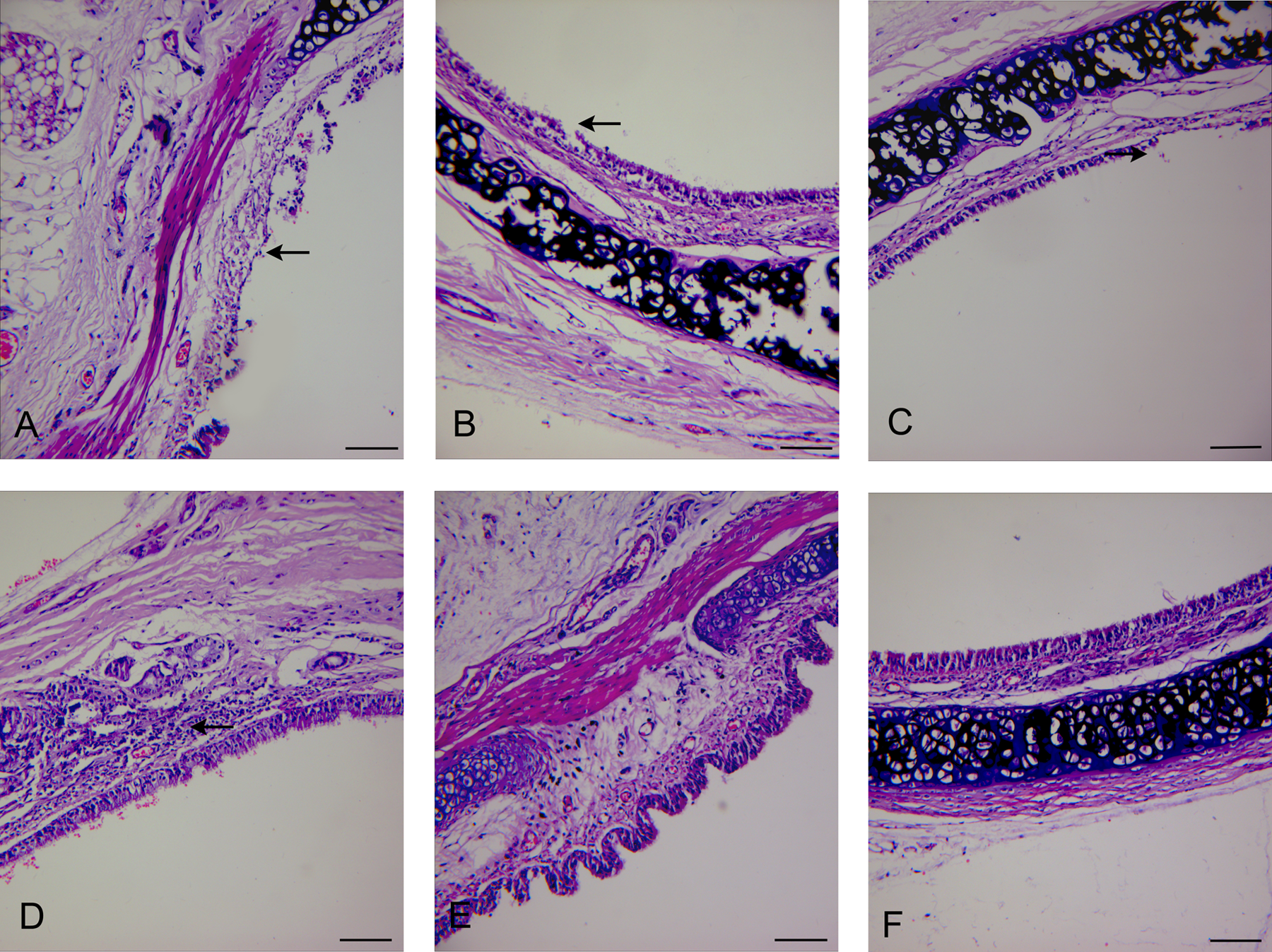
Light micrographs of tracheal sections stained with hematoxylin and eosin (H&E) showing epithelial lesions induced by sulfur mustard (SM) 6, 24, 48, and 72 hours in rats after intratracheal administration. Ulceration in tracheal membranous part at 6 hours (A, arrow), delaminated area of the epithelium and submucosa at 24 hours (B, arrow), focal ulceration in tracheal nonmembranous part at 48 hours (C, arrow), and thickening of the submucosa at 72 hours (D, arrow) can be seen. The propylene glycol group was the same with that in the control group (E and F; H&E staining, ×200 magnification; bars, 538.2 μm).
Distribution of various inflammatory cells in tracheal injury
In the SM group, the lymphocytes were scattered in the submucosa at 6 hours (Figure 2A). The lymphocytes were aggregated in clusters, the macrophagocytes were scattered, and the neutrophils were rare in the submucosa at 24, 48, and 72 hours (Figure 2B–D). In the propylene glycol and control groups, inflammatory cells of 3 types were rare (Figure 2E and F).
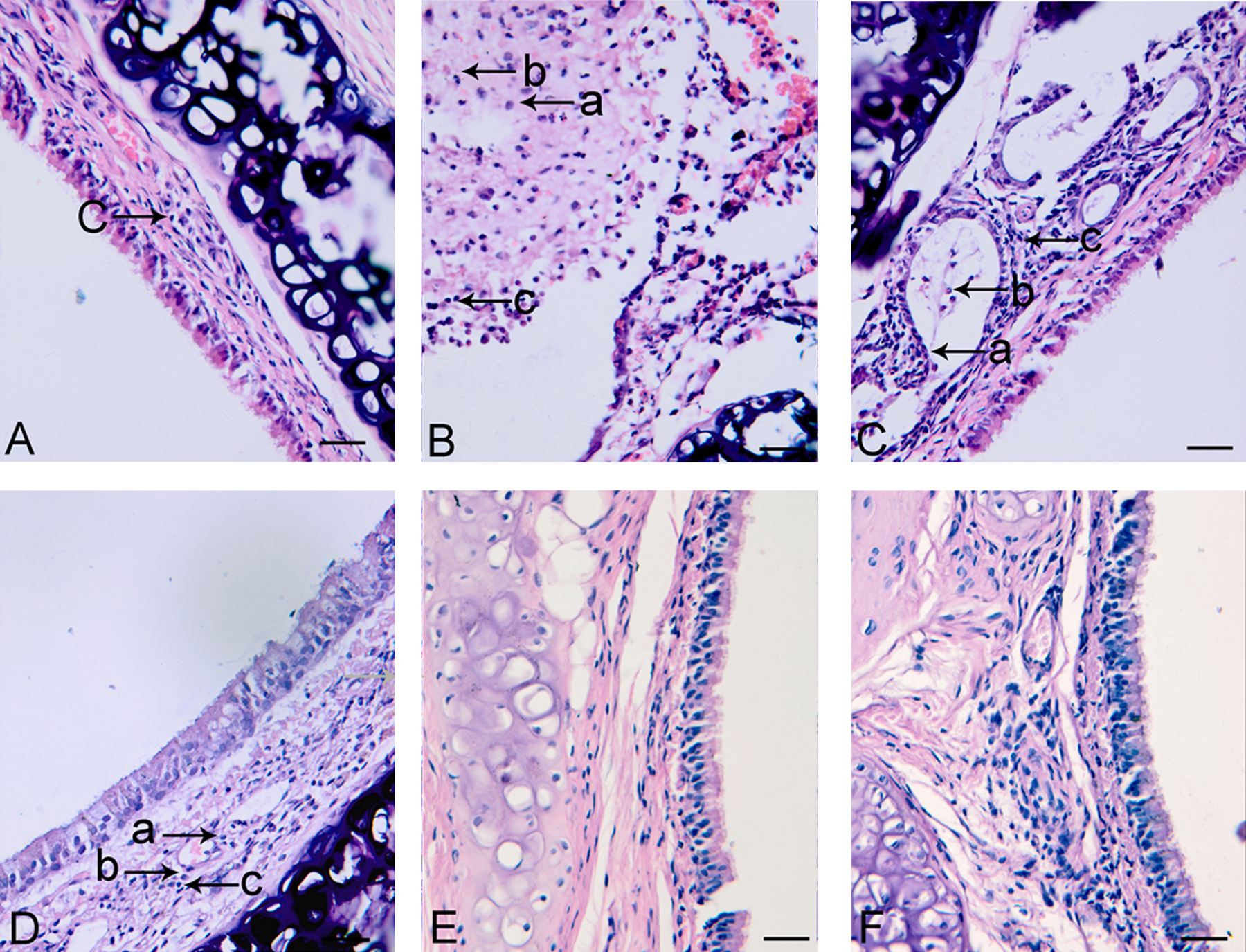
In the sulfur mustard (SM) group, (A) shows inflammatory cell infiltration in tracheal wall at 6 hours (c→ indicates lymphocyte, H&E staining, ×400 magnification). B, C, and D show inflammatory cell infiltration in tracheal wall at 24, 48, and 72 hours, respectively (a→ indicates neutrophil, b→ indicates macrophagocyte, and c→ indicates lymphocyte, H&E staining, ×400 magnification). The propylene glycol group was the same with that in the control group (E and F; H&E staining, ×400 magnification; bars, 269.1 μm). H&E indicates hemotoxylin and eosin.
Cytomorphologic changes
Figure 3A, B, C, I, J, and M is SM treated. In ciliated cells, loss of membranes, outflow of cytoplasm, exposure of the nucleus, lodged cilia and microvilli, disarranged basal bodies, fuzzy mitochondrial cristae, and medullary changes as well as margination of the nuclear chromatin were observed (Figure 3A–C). In goblet cells, loss of membranes, fuzzy mitochondrial cristae, swollen rough endoplasmic reticulum, and dilated intercellular spaces were seen (Figure 3A and C). In basal cells, swollen rough endoplasmic reticulum, fuzzy mitochondrial cristae, vacuolated mitochondria, irregular nuclear shapes, and margination of the nuclear chromatin were observed (Figure 3I and J). In fibroblasts, fuzzy mitochondrial cristae, vacuolated mitochondria, obscured karyotheca and karyopyknosis, and margination of the nuclear chromatin were observed (Figure 3M). The results in the propylene glycol group were the same as the control group (Figure 3D–H, K, L, N, and O).
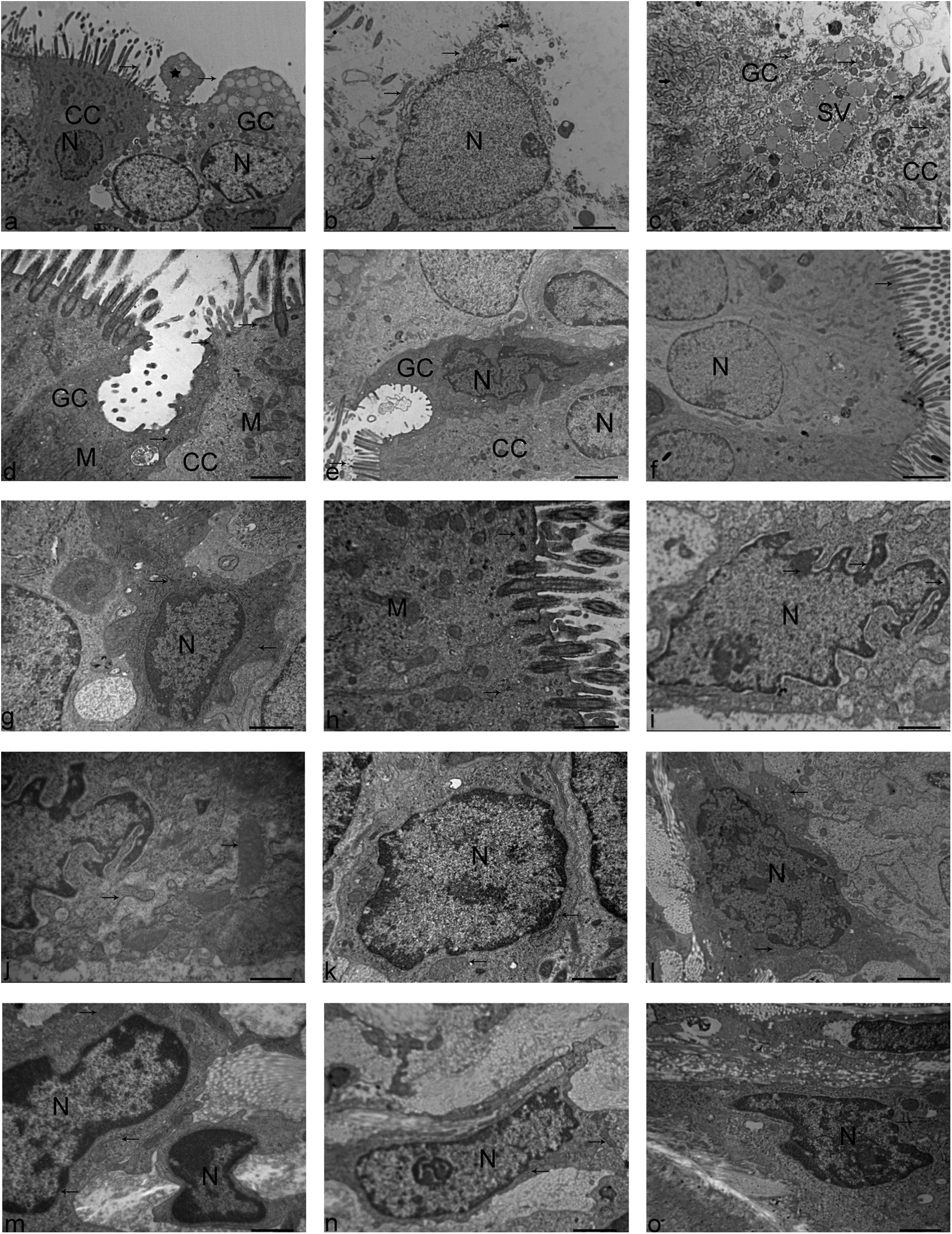
Electron micrograph shows SM-induced tracheal cellular injury after 72 hours. A, In the SM group, ciliated cells (CC), goblet cells (GC), nucleus (N), cilia (left arrow), and cellular membranes (right arrow) are observed. * shows cell debris detached from GC (×4000 magnification; bar, 6.99 µm). B, In the SM group, CC and nucleus (N) are observed. It is shown that the cytoplasm has extruded from the cell (upper arrow), cilia (middle arrow), and cell membrane (lower arrow). Basal bodies (solid arrows; ×10 000 magnification; bar, 2.5 µm). C, In the SM group, CC, GC, secretory vacuole (SV), loss of cellular membranes and the cytoplasm flowed out (left arrow), mitochondria (middle arrow, right arrow), rough endoplasmic reticulum (left solid arrow), and junctional complex (right solid arrow; ×10 000 magnification; bar, 2.5 µm) are observed. D, In the propylene glycol group, CC, GC, mitochondria (M), junctional complex (upper arrow), basal bodies (middle arrow), and rough endoplasmic reticulum (lower arrow; ×20 000 magnification; bar, 1.0 µm) are observed. E, In the propylene glycol group, CC, GC, and nucleus (N) are observed. Arrow shows cilia (×8000 magnification; bar, 3.49 µm). F, In the control group, CC and N (nucleus) are observed. Arrow shows cilia (×4000 magnification; bar, 4.37 µm). G, In the control group, GC, nucleus (N), rough endoplasmic reticulum (upper arrow), and mitochondria (lower arrow; ×20 000 magnification; bar, 1.0 µm) are observed. H, In the control group, CC, mitochondria (M), basal bodies (upper arrow), junctional complex (middle arrow), and rough endoplasmic reticulum (lower arrow; ×20 000 magnification; bar, 1.0 µm) are observed. I, In the SM group, basal cells, nucleus (N), and chromatin (arrow; ×10 000 magnification; bar, 1.35 µm) are observed. J, In the SM group, basal cells, rough endoplasmic reticulum (left arrow), and mitochondria (right arrow; ×20 000 magnification; bar, 1.0 µm) are observed. K, In the propylene glycol group, basal cells, nucleus (N), mitochondria (left arrow), rough endoplasmic reticulum (right arrow; ×20 000 magnification; bar, 1.0 µm) are observed. L, In the control group, basal cells, nucleus (N), mitochondria (upper arrow), and rough endoplasmic reticulum (lower arrow; ×20 000 magnification, bar, 1.0 µm) are observed. M, In the SM group, fibroblasts, nucleus (N), mitochondria (upper arrow), rough endoplasmic reticulum (middle arrow), and nuclear membrane (lower arrow; ×20 000 magnification; bar, 1.0 µm) are observed. N, In the propylene glycol group, nucleus (N), rough endoplasmic reticulum (left arrow), and mitochondria (right arrow; ×8000 magnification, bar, 1.88 µm) are observed. O, In the control group, nucleus (N) and endoplasmic reticulum (arrow; ×20 000 magnification; bar, 1.0 µm) are observed. SM indicates sulfur mustard.
Apoptosis Analysis (Immunohistochemical Staining)
In the SM group (caspase 3), apoptosis was scattered at 6 hours (Figure 4A), increased at 24 hours (Figure 4B), and clearly increased at 48 and 72 hours (Figure 4C and D) in the epithelium and submucosa. In contrast, in the propylene glycol and control groups, apoptosis was scattered (Figure 4E and F).
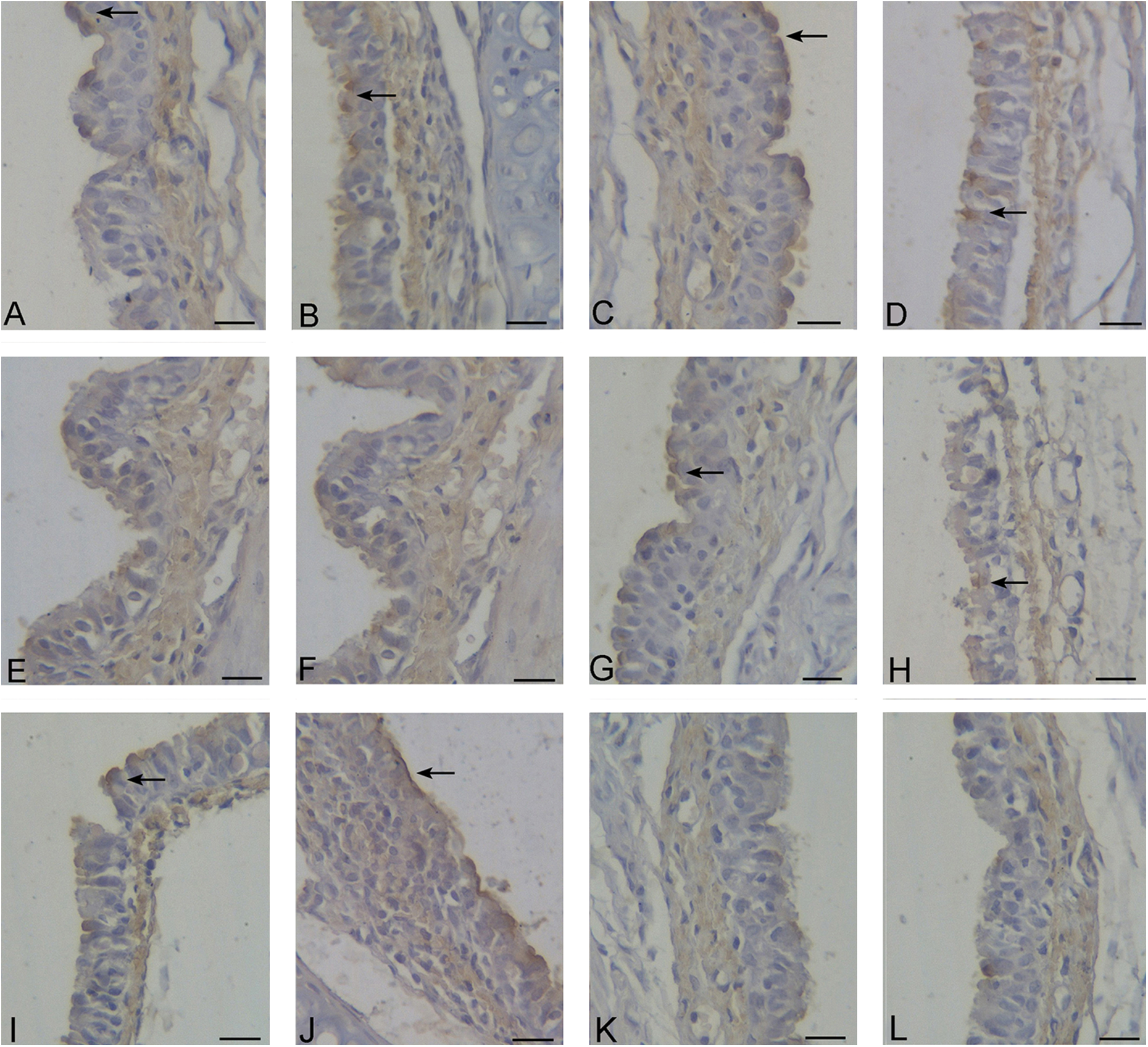
In the SM group, (A), (B), (C), and (D) show the apoptosis in tracheal wall at 6, 24, 48, and 72 hours, respectively (→ indicates positive, immunohistochemistry for caspase 3, ×400 magnification). The propylene glycol group was the same with that in the control group (E and F; immunohistochemistry for caspase 3, ×400 magnification). In the SM group, (G), (H), (I), and (J) show the apoptosis in tracheal wall at 6, 24, 48, and 72 hours, respectively (→ indicates positive, immunohistochemistry for caspase 9, ×400 magnification). The propylene glycol group was the same with that in the control group (K and L; immunohistochemistry for caspase 9, ×400 magnification; bars, 398.7 µm). SM indicates sulfur mustard.
In the SM group (caspase 9), apoptosis was scattered at 6 hours (Figure 4G), increased at 24 hours (Figure 4H), and clearly increased at 48 and 72 hours (Figure 4I and J) in the epithelium and submucosa. In contrast, in the propylene glycol and control groups, apoptosis was scattered (Figure 4K and L).
Quantitative Analysis of Serum Inflammatory Factors
In the SM group, the serum levels of TNF-α, IL-1β, and IL-6 peaked at 24 hours. Compared with the propylene glycol group, the difference was statistically significant (P < 0.05; Figures 5– 7).
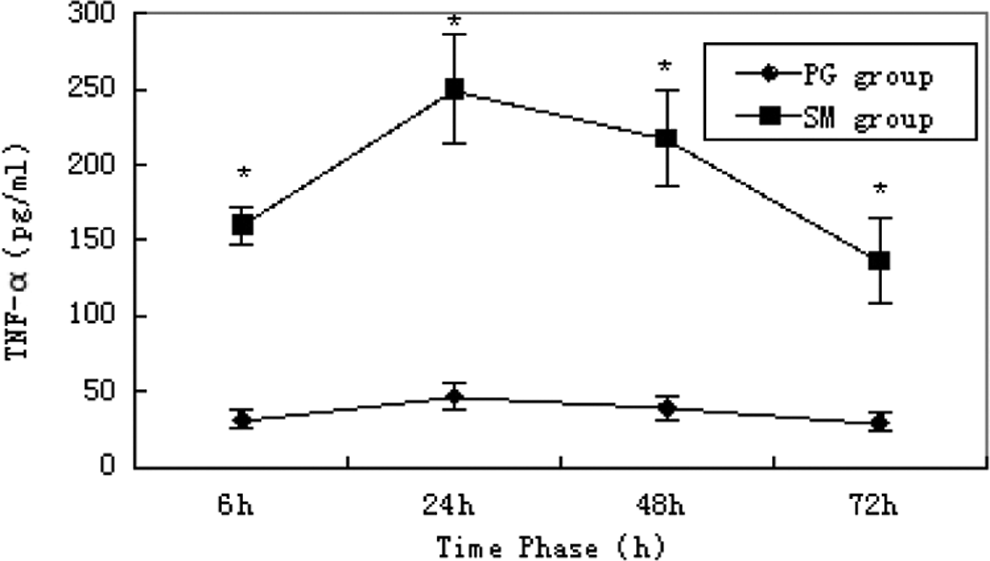
Serum level of tumor necrosis factor α (TNF-α). * indicates significant difference over control.
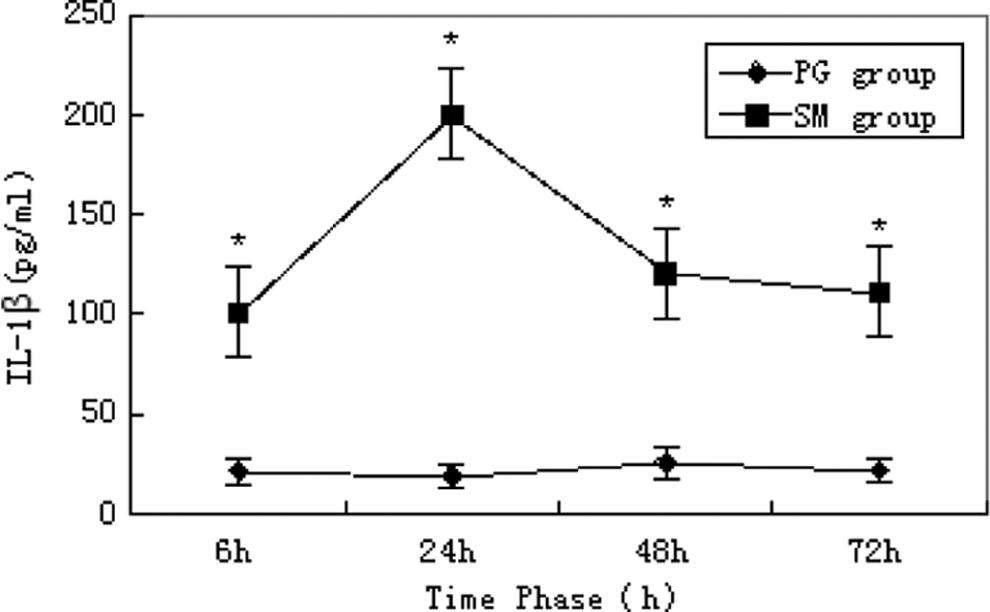
Serum level of interleukin 1β (IL-1β). * indicates significant difference over control.
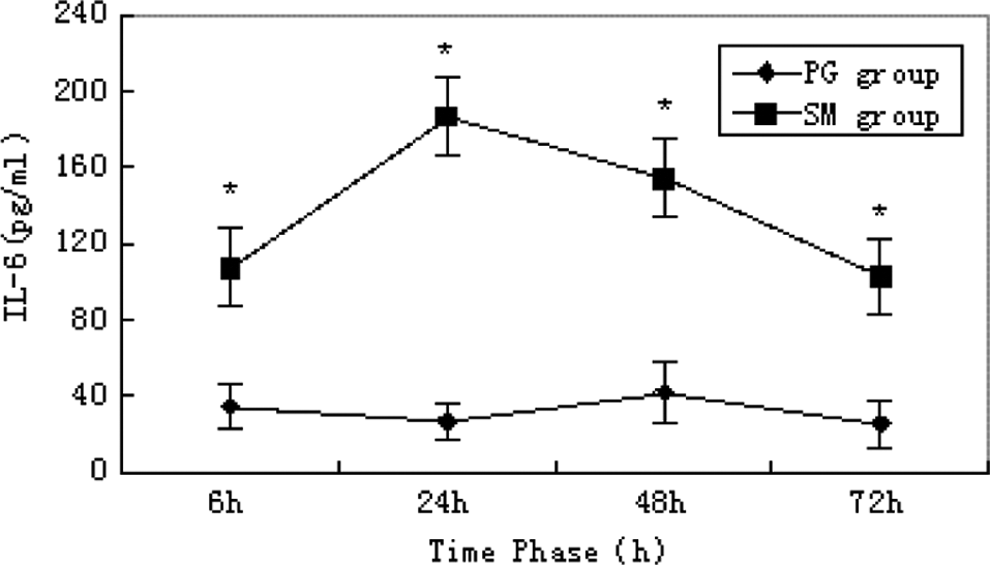
Serum level of interleukin 6 (IL-6). * indicates significant difference over control.
Quantitative Analysis of Serum Enzyme
In the SM group, the serum levels of LDH, GP, and TBARS peaked at 6 hours, and GGT peaked at 24 hours. Compared with the propylene glycol group, the difference was statistically significant (P < 0.05; Figures 8 –11).
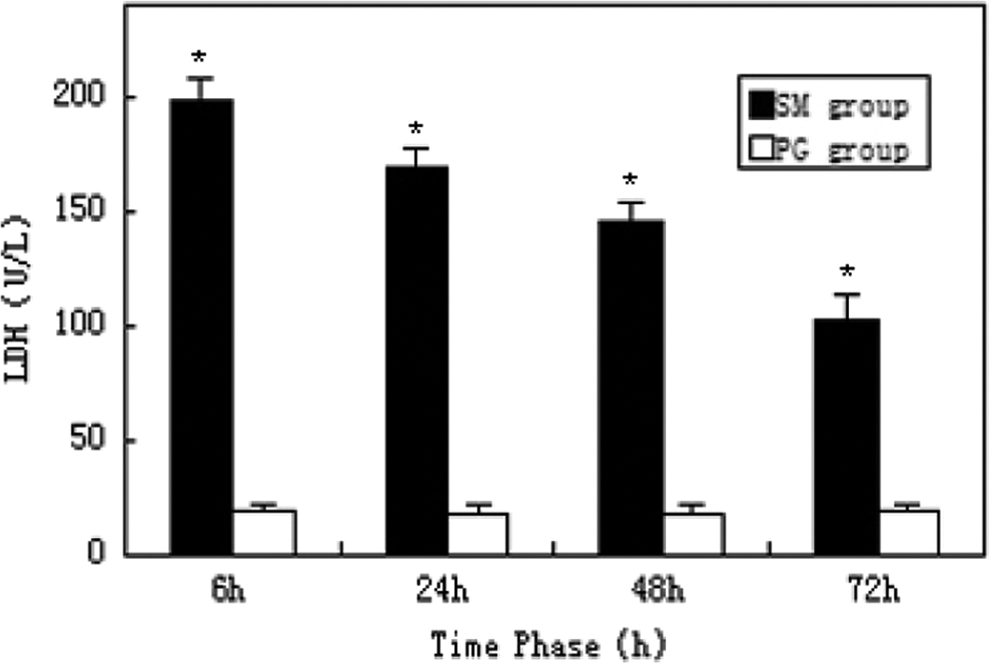
Serum level of lactate dehydrogenase (LDH). * indicates significant difference over control.
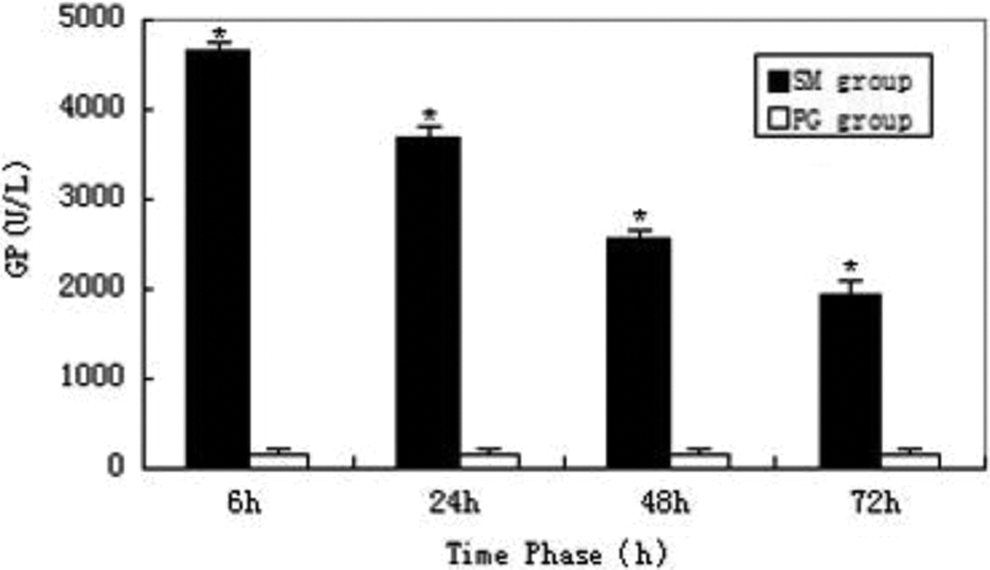
Serum level of glutathione peroxidase (GP). * indicates significant difference over control.
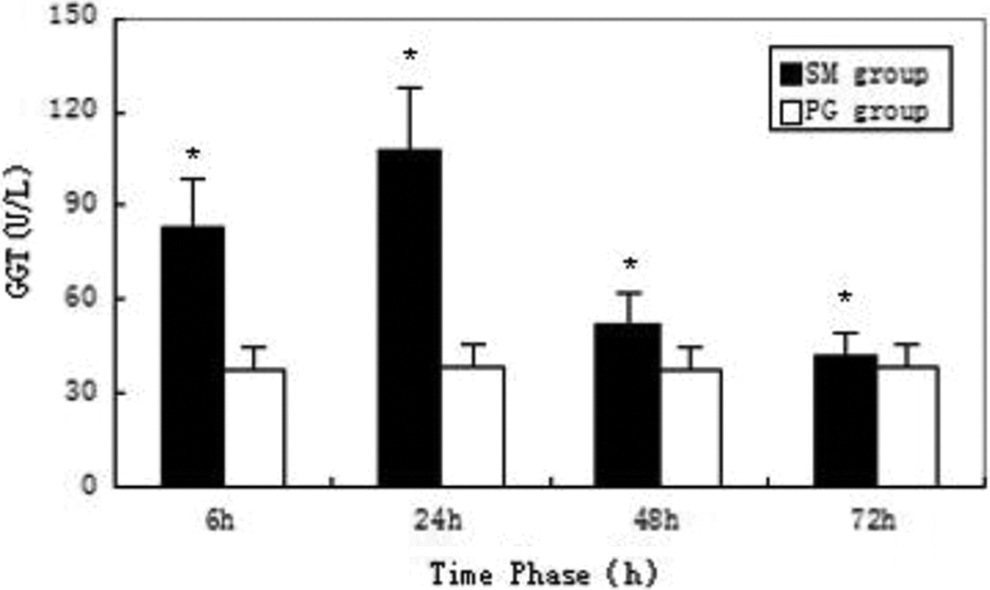
Serum level of γ-glutamyl transferase (GGT). * indicates significant difference over control.
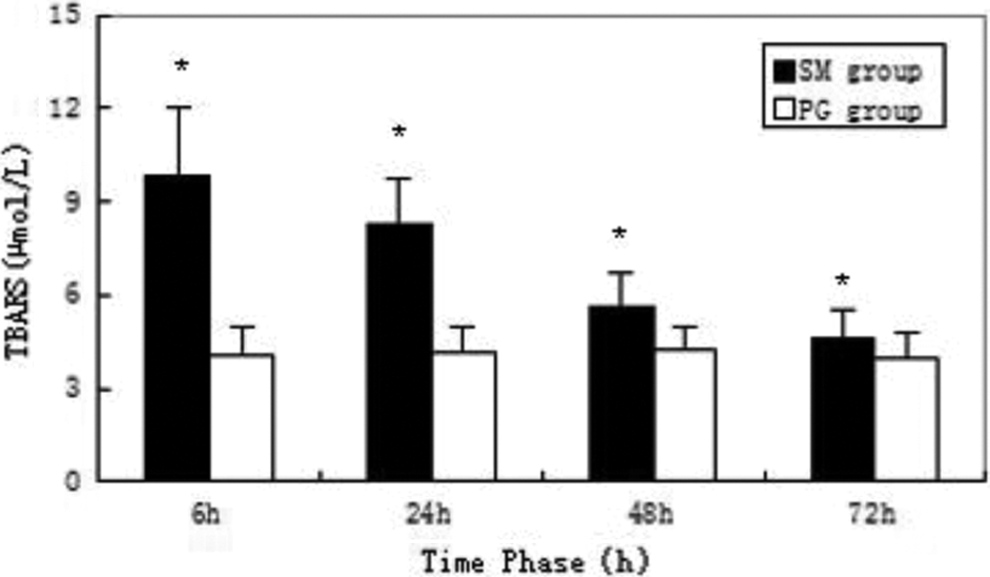
Serum level of thiobarbituric acid reactive substance (TBARS). * indicates significant difference over control.
Discussion
Clinical studies of combat casualties exposed via inhalation to the chemical warfare vesicant, SM, suggest that airway inflammation and the hypersecretion of mucus comprise the prominent features of SM-related injuries. 16,17 Collectively, these findings suggest that SM-related pulmonary lesions tend to be characterized by airway inflammation and the hypersecretion of mucus; however, the effects of SM on the production of airway epithelial mucin were not previously studied. 18 It was recently shown that exposure to nitrogen mustard (HN2), an SM analogue and simulant, induced cytotoxic and apoptotic changes in human bronchial epithelial cells in a dose- and time-dependent fashion. The HN2 challenge also induces secretion of chemokines and proinflammatory cytokines. 19 Sulfur mustard can be dispersed in a variety of ways, including part of an explosive device in a military situation. Unfortunately, exposure data assessments from previous battlefields or civilian population exposures are lacking. 20 Previous studies using SM instillation or vapor delivery have focused on the lung or used SM analogs, such as CEES or nitrogen mustard, to investigate lung injury. 21 –23 The present studies provide additional detail on the approach to the SM (2 mg/kg) intratracheal administration and provide a superior approach to characterization of a tracheal injury model involving high concentrations of SM. These injuries are similar to the pathology observed in humans, and this creates a platform for screening the efficacy of potential therapeutics.
Accumulating evidence suggests that mediators released from inflammatory cells contribute to lung injury induced by diverse pulmonary toxicants. 24 The role of inflammatory mediators in SM-induced pulmonary injury is unknown. Findings that anti-inflammatory steroids, such as betamethasone, can inhibit airway injury and hyperresponsiveness suggest that phagocytic leukocytes are involved in the pathogenic response. 25 This is supported by reports that inhaled corticosteroids improve pulmonary function in patients with chronic bronchiolitis as a result of SM inhalation. 26 In the present study, we characterized tracheal injury and inflammatory responses to SM with intratracheal injection in a rat model. Identification of the specific cytotoxic/proinflammatory mediators involved in SM-induced tracheal injury may help in the development of agents to mitigate the toxicity of this vesicant.
Exposure of rats to SM with intratracheal injection resulted in rapid tracheal toxicity. Thus, within 6 hours of exposure, histopathologic changes were noted in the upper airways and included detachment of the tracheal epithelial lining from the basement membrane, deposits of fibrin containing cellular debris in the airway lumen, and edema of the submucosal lining. Similar alterations in the airway epithelium have been described in the lungs of rodents exposed to SM. 27,28 Sulfur mustard with intratracheal injection also caused upper airway effects, including thickening of the tracheal wall and perivascular edema, suggesting potential alterations in the integrity of the tracheal epithelial lining. This was supported by our findings that serum inflammatory factor levels (TNF-α, IL-1β, and IL-6) increased significantly after exposure of rats to SM. These data are consistent with previous reports of SM-induced alveolar epithelial damage in rodents. 28,29 Structural alterations in the trachea were accompanied by inflammatory cell infiltration into the airways, as assessed histologically, and by increased serum TNF-α, IL-1β, and IL-6. Our results of H&E staining showed that the majority of these inflammatory cells were lymphocytes, thus supporting the idea that phagocytic leukocytes are important in the pathogenic response to inhaled SM. 30
Although both apoptosis and necrosis result in cell death, there are many differences regarding these 2 mechanisms and their effects on neighboring cells. During apoptosis, the cell shrinks, while producing apoptotic bodies that are taken up by neighboring cells via phagocytosis. Very little cytoplasmic material is released into the intercellular space, and no or limited inflammation is caused in neighboring tissues. In contrast, during necrosis, cells swell, burst, and release the cytoplasmic contents, thus triggering a massive inflammation in the surrounding area. 31 More recent work with SM on DNA has characterized genomic changes that show increases in markers of apoptosis, cell cycle regulation, and various other response genes. 32 We have identified that cellular membranes are lacking, cytoplasm has leaked out, mitochondrial cristae were vague, medullary changes existed in ciliated cells and goblet cells, and the nuclear chromatin appeared marginated in basal cells and fibroblasts. The results of immunohistochemistry indicated that apoptotic cells were significantly increased in the epithelium and submucosa at 48 hours. All of these results indicated that SM can induce delayed mitochondrial dysfunction, apoptosis, and the nuclear DNA damage. We are concerned with the quantification and discrimination of apoptosis and necrosis in order to investigate potential active pharmaceutical ingredients against SM-induced cytotoxicity.
Studies suggest that CEES produces mitochondrial dysfunction that is followed by an increase in mitochondrial reactive oxygen species (ROS) production and cellular oxidative stress.
11
Furthermore, exploring the role of oxidative stress in CEES-mediated injury and supplementation with antioxidants as a treatment has been a major area of interest. Studies have indicated that TNF-α is increased in lung macrophages of guinea pigs 1 hour after intratracheal instillation with CEES.
33
Other studies suggest a role for oxidative stress mediated by CEES that include changes in superoxide dismutase (SOD), catalase, glutathione (GSH) reductase, and GSH transferase activities, inhibition of inducible nitric oxide synthase, depletion of GSH, and increases in ROS.
34
–37
As a consequence, supplementation with catalase, resveratrol, N-acetyl-
This research suggests that the histomorphologic changes following SM intratracheal injection are similar to inhalation, except for the range; the former is the main source of local damage, while the latter is for extensive damage. 21 The mitochondrial damage is predominant in SM-induced cellular injury because of the cytotoxicity of SM, alkylation can induce DNA melting, and mitochondria contain DNA. 56 Some research has shown that mitochondria participate and play a key role in SM-induced apoptosis of epithelial cells. 57 Sulfur mustard will trigger endoplasmic reticulum stress under various conditions, such as glucose starvation, disturbance of intracellular calcium homeostasis, inhibition of protein glycosylation, or exposure to free radicals, and excessive endoplasmic reticulum stress induces cell death. 58 This study identified histologic changes, inflammatory reactions, apoptosis, oxidative stress, and DNA damage characteristic of SM (2 mg/kg)-induced acute tracheal injury, and the severity of changes is time dependent.
Footnotes
Author Contribution
Zhu contributed to conception and design, critically revised the article, and gave final approval. Xu contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, and critically revised the article. Meng contributed to design, acquisition and analysis, and critically revised the article. Chu contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Zhao contributed to acquisition and gave final approval. Lian contributed to conception. Wang contributed to acquisition and analysis. Guo contributed to acquisition and analysis. Zhang contributed to acquisition and analysis and critically revised the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Department of Pathology and Department of Electron Microscopy in Weifang Medical University.
