Abstract
Objective
Sulfur mustard (SM) is an important chemical warfare agent. The mechanisms underlying SM toxicity have not been completely elucidated. However, oxidative stress and the subsequent damage to macromolecules have been considered ascrucial steps in SM toxicity. In this study, a rat model of SM-induced acute pulmonary injury was established using an equal toxicity dose (1LD50). This study employed two methods to directly compare oxidative stress indices in serum enzymes and the epithelial cells of the alveolar septa.
Methods
Male Sprague-Dawley rats were randomly divided into intraperitoneal SM, intraperitoneal propylene glycol control, tracheal SM, tracheal propylene glycol control, and control groups. SM-induced serum enzyme levels and protein expression in the epithelial cells of the alveolar septa were measured using enzyme-linked immunosorbent assay and immunohistochemistry.
Results
Serum levels of superoxide dismutase, catalase, and glutathione peroxidase were upregulated in the intraperitoneal SM group compared with those in the tracheal SM group. Positive expression ratios of CuZn-superoxide dismutase, Mn-superoxide dismutase, paraoxonase-1, and apolipoprotein-1 proteins in the epithelial cells of the alveolar septa in the intraperitoneal SM group were elevated compared with those in the tracheal SM group.
Conclusion
Under SM (1LD50) exposure, there were significantly higher serum enzyme levels and protein expressions in the epithelial cells of the alveolar septa of rats injected with SM intraperitoneally compared with SM administered by intratracheal instillation. The results demonstrated that the differences in oxidative stress indices at the molecular level in SM-induced pulmonary injury were dependent on the route of exposure.

Introduction
Sulfur mustard (SM) is an alkylating and vesicating agent that has been used for one century.1,2 In its pure form, SM is a viscous and colorless fluid. SM enters the body through the eyes, skin, respiratory tract, and gastrointestinal tract, causing acute and delayed effects on different body organs.3,4 Different cellular mechanisms and molecular pathways are responsible for damage to body tissues, such as DNA damage, oxidative stress, apoptosis, and inflammation.5,6 The lungs are one of the major target organs of SM-associated injury. Mechanisms underlying SM-induced pulmonary injuries have not been completely elucidated, and no effective treatments are currently available, leading to high morbidity and mortality rates. 7 It has been demonstrated that SM can induce pulmonary oxidative stress reactions by depleting cells of intracellular antioxidants, including glutathione, and increasing lipid peroxidation, thioredoxin, and the mitochondrial membrane potential. This results in the increased production of reactive oxygen species (ROS) and loss of antioxidant enzyme activity, such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSHPx), thioredoxin reductase (TrxR), and glutathione S-transferase (GST).8,9 Animal experiments have confirmed that SM-induced pulmonary injuries extend beyond the respiratory tract and can manifest through multiple pathways.10–13 In addition, SM is a dose- and time-dependent toxic agent. Therefore, a positive correlation between dose and time and the degree of SM-induced pulmonary injury has been reported.14,15 Mechanisms underlying SM-induced inflammation and oxidative stress are interdependent and mutually reinforcing, playing a notable role in the development of pulmonary injury.16,17 In the present study, preliminary experiments were conducted to calculate the equivalent toxic dose (1LD50) using Horn’s method. A rat model of SM-induced acute pulmonary injury was established via two administration routes. This study compared differences in serum enzyme levels and protein expression in alveolar septa, resulting from the different SM administration methods. Furthermore, this study investigated the molecular mechanisms underlying SM-induced acute pulmonary injury to provide a theoretical basis for targeted treatment.
Materials and methods
Reagents and instruments
Reagents were sourced from the following suppliers. SM (99% pure) was provided by the Institute of Pharmacology and Toxicology, Academy of Military Medical Sciences (Beijing, China). The 1, 2-propylene glycol solution was attained from Tianjin Zhiyuan Chemical Co., Ltd (Tianjun, China). SOD, CAT, and GSH-Px ELISA kits were provided by Hangzhou Lianke Biotech Co., Ltd (Hangzhou, China). Goat serum was supplied by Beijing Zhongshan Jinqiao Biotech Co., Ltd (Beijing, China). Immunohistochemical kits for CuZn-superoxide dismutase (CuZn-SOD), Mn-superoxide dismutase (Mn-SOD), paraoxonase-1 (PON-1), and apolipoprotein-1 (ApoA1), as well as secondary antibodies (biotin-labeled goat anti-rabbit IgG) and tertiary antibodies (horseradish peroxidase-labeled streptavidin), were provided by Beijing Bioss Biotech Co., Ltd (Beijing, China). Finally, the instruments used in this study included an automatic biochemical immune analyzer (COBAS 8000 type; Roche, Basel, Switzerland), a cold light source (AXEL300 type; Olympus, Tuttlingen, Germany), a light microscope (BX51 type; Olympus, Tokyo, Japan), a EMKA small animal non-invasive pulmonary function monitoring system (EMKA Technologies, Paris, French), and Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, MD, USA).
Experimental animals and grouping
In this experiment, 236 Sprague-Dawley rats (male, 280-300 g, 15 weeks, specific pathogen-free) were purchased from the Experimental Animal Center of the Chinese PLA Military Academy of Medical Sciences (Beijing China; Certification No. 0015902). Subsequently, 136 rats were randomly divided into groups as follows: intraperitoneal SM group, n = 32; intraperitoneal propylene glycol control group, n = 32; tracheal SM group, n = 32; tracheal propylene glycol control group, n = 32; and control group, n = 8. SM (96% pure) was diluted in propylene glycol for later use. The animal model was established through intratracheal instillation. A subcutaneous dose of atropine (0.05 mg/kg) was administered, and 30 min later, ketamine hydrochloride (100 mg/kg) was utilized for anesthesia. SM [0.1 mL per rat (0.98 LD50 = 2 mg/kg)] was instilled intothe tracheas of rats in the SM group, while propylene glycol (0.1 mL per rat) was instilled into the tracheas of those in the propylene glycol control group. The animal model was established via intraperitoneal injection as follows: Rats were anesthetized as described previously. Next, SM [0.1 mL per rat (0.96 LD50 = 8 mg/kg)] was administered intraperitoneally, while propylene glycol (0.1 mL per rat) was administered to the intraperitoneal propylene glycol control group. The control group received no treatment.
Measurement of serum oxidative stress indices
Intracardiac puncture and collection of blood samples (2 mL per rat) from the SM and propylene glycol groups were performed at 6, 24, 48, and 72 h. Samples were placed ina 37°C water bath for 1 h, stored at 4°C overnight, and centrifuged at 2000× g for 20 min. The supernatant was thereafter decanted into a sterile vial and stored at −80°C until further use. Serum levels of SOD, CAT, and GSH-Px were measured using an automatic biochemical immune analyzer.
Immunohistochemistry immunolabeling
Immunohistochemical staining (using the streptavidin-peroxidase method) was employed to measure the expression levels of CuZn-SOD, Mn-SOD, PON-1, and ApoA1. Tissue samples were embedded into paraffin, sectioned, and treated by conventional xylene dewaxing, followed by antigen retrieval. Rabbit anti-rat monoclonal antibodies against CuZn-SOD, Mn-SOD, PON-1, and ApoA1 (20 μL/section) were then added. Following incubation with primary antibodies (1:800, 1:800, 1:400, 1:400, and 1:400, respectively), sections were thereafter incubated with secondary (biotin-labeled goat antirabbit IgG) and tertiary (horseradish enzyme-labeled streptavidin) antibodies. Finally, 3, 3′-diaminobenzidine staining was employed. Sections were counterstained with hematoxylin, and the slides were mounted with conventional resin. For negative controls, phosphate-buffered saline (PBS) was utilized rather than the primary antibodies. Known positive sections served as positive controls.
Microscopic image analysis
Image-Pro Plus 6.0 Pathological Cell Image Analysis system was used to analyze immunohistochemical staining of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins from each group. All measurement parameters were manually set, and the abundance of both positive and strongly positive cells was quantified. A field of view was selected for each interval at high magnification (×400). At least five high-power fields were examined per slice, and the ratio of positive cells in the alveolar septum was calculated as follows: (number of positive cells/total number of cells in five high-power fields) × 100%. Mean values were also calculated.
Statistical analysis
Statistical analysis was carried out using SPSS 22.0 software (IBM Corp., Armonk, NY, USA). All values were expressed as mean ± standard deviation. Two-way ANOVA with multiple comparisons were performed to determine differences between groups, where applicable. Dunnetts’ post-test was performed to determine differences between groups over time and corrected for multiple comparisons. For analysis of oxidative stress indices, two-way ANOVA with Student-Newman-Keuls multiple comparisons test corrected for multiple comparisons were performed to determine differences between these groups, and a p value of <0.05 was considered significant.
Results
The results (LD50) of the preliminary experiment in rats after SM exposure
SM (LD50) was injected via intraperitoneal administration (Horn’s method, mg/kg, %, n = 10).

Note: LD50 = 8.31 mg/kg, 95% confidence 7.76-8.71 mg/kg.
SM (LD50) was instilled via intratracheal administration (Horn’s method, mg/kg, %, n = 10).

Note: LD50 = 2.04 mg/kg, 95% confidence 1.862 - 2.29 mg/kg.
The respiratory clinical distress score criteria in rats with SM (LD50)-induced pulmonary injury via intraperitoneal injection or intratracheal instillation.

The breathing and the pulmonary function parameters in rats with SM (LD50)-induced acute pulmonary injury via intratracheal instillations (x ± s, n = 8).

Note: RF: respiratory frequency (breaths/min); SaO2: oxygen saturation (%); TV: Tidal volume (ml); MV: Minute ventilation (L/min); PIF: Peak inspiratory volume (ml/s); PEF: Peak exspiratory volume (ml/s); Penh: enhanced pause; EF50: Mid-expiratory flow (ml/s).
aCompared with n, P < 0.05.
bCompared with n and a, P < 0.05.
cCompared with n, a, and b, P < 0.05.
dCompared with n, a, b, c, P < 0.05.
The breathing and the pulmonary function parameters in rats with SM (LD50)-induced pulmonary injury via intraperitoneal injections (x ± s, n = 8).
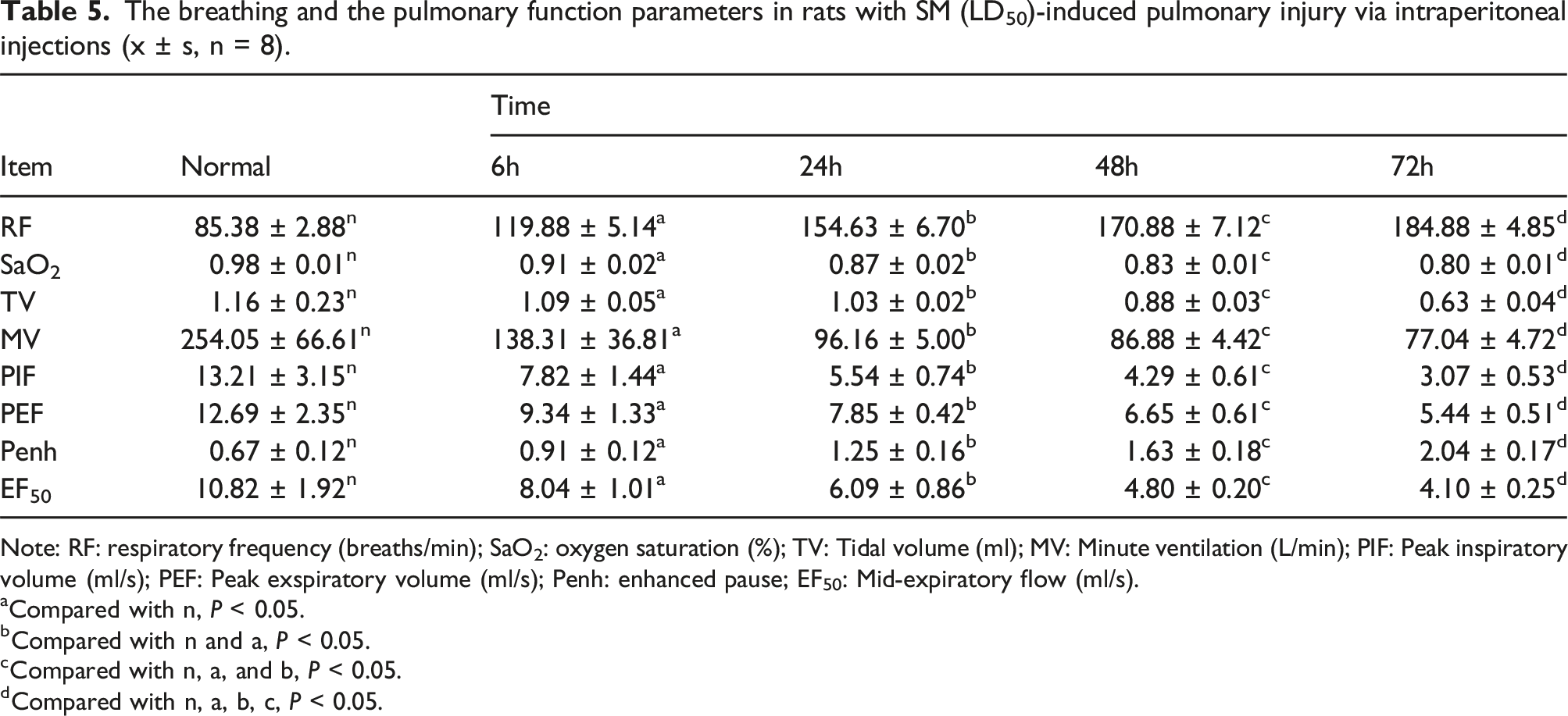
Note: RF: respiratory frequency (breaths/min); SaO2: oxygen saturation (%); TV: Tidal volume (ml); MV: Minute ventilation (L/min); PIF: Peak inspiratory volume (ml/s); PEF: Peak exspiratory volume (ml/s); Penh: enhanced pause; EF50: Mid-expiratory flow (ml/s).
aCompared with n, P < 0.05.
bCompared with n and a, P < 0.05.
cCompared with n, a, and b, P < 0.05.
dCompared with n, a, b, c, P < 0.05.
Changes in serum oxidative stress indices in rats with SM (1LD50)-induced acute pulmonary injuries
Serum levels of SOD, CAT, and GSH-Px in both the intraperitoneal and tracheal SM groups peaked at 24 h, and they were subsequently gradually reduced. Comparison of serum SOD, CAT, and GSH-Px levels among the five groups by repeated measures ANOVA at different times points revealed that serum levels of SOD, CAT, and GSH-Px in the intraperitoneal and tracheal SM groups at each time point were significantly elevated. Moreover, compared with the other four groups, serum levels of SOD, CAT, and GSH-Px were significantly upregulated in the intraperitoneal SM group. In the intraperitoneal and tracheal SM groups, there was a trend of decreasing serum levels of SOD, CAT, and GSH-Px over time which reached statistical significance (Figures 1–3). Serum SOD levels in rats with SM (1LD50)-induced acute pulmonary injury at different time points. Rats of the tracheal SM group were injected intratracheally with diluted SM (1LD50 = 2 mg/kg), the rats of intraperitoneal SM group were injected intraperitoneally with diluted SM (1LD50 = 8 mg/kg). x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group. Serum CAT levels in rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group. Serum GSH-Px levels in rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group.


Expression of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury
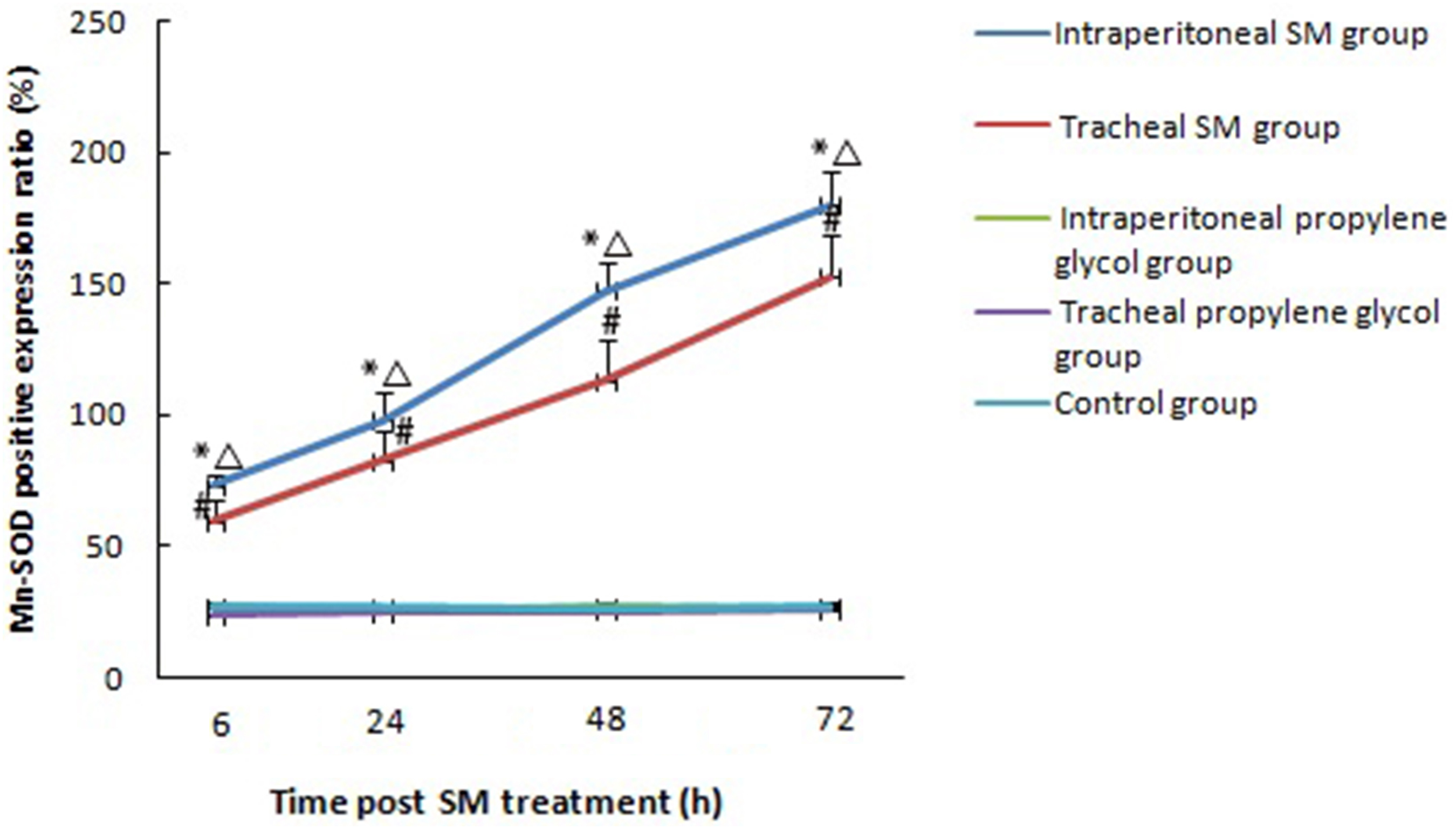
In the intraperitoneal and tracheal SM groups, the expression levels of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the epithelial cells of the alveolar septa were diffusely distributed at 6 h. They were found to aggregate into clusters at 24 h. This aggregation was also evident at 48 and at 72 h. In contrast, in intraperitoneal and tracheal propylene glycol control, and control groups, the expression levels of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins were scattered (Figures 4–7). Positive expression ratios of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the epithelial cells of the alveolar septa were compared among the five groups by repeated measures ANOVA at different time points. It was found that positive expression ratios of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the epithelial cells of the alveolar septa in the intraperitoneal and tracheal SM groups at each time point were significantly elevated. Moreover, compared with the other four groups, positive expression ratios of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the epithelial cells of the alveolar septa significantly increased in the intraperitoneal SM group. In the intraperitoneal and tracheal SM groups, there were upward trends of positive expression ratios of CuZn-SOD, Mn-SOD, PON1, and ApoA1 proteins in the alveolar septa over time, reaching statistical significance (Figures 8–11). Expression of CuZn-SOD proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. (A-D) show the positive expression (arrow) at 6, 24, 48, 72 hours intraperitoneal SM group; (F-I) show the positive expression (arrow) at 6, 24, 48, 72 hours in tracheal SM group; (E) and (J) show the negative expression in control group (CuZn-SOD immunolabeling, × 400, magnification, Bars = 20 μm). Expression of Mn-SOD proteins in the the epithelial cells of alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. (A-D) show the positive expression (arrow) at 6, 24, 48, 72 hours intraperitoneal SM group; (F-I) show the positive expression (arrow) at 6, 24, 48, 72 hours in tracheal SM group; (E) and (J) show the negative expression in control group (Mn-SOD immunolabeling, × 400, magnification, Bars = 20 μm). Expression of PON-1 proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury via intraperitoneal injections and intratracheal instillation at different time points. See Figure 1 for the rat treatment. (A-D) show the positive expression (arrow) at 6, 24, 48, 72 hours intraperitoneal SM group; (F-I) show the positive expression (arrow) at 6, 24, 48, 72 hours in tracheal SM group; (E) and (J) show the negative expression in control group (PON-1 immunolabeling, × 400, magnification, Bars = 20 μm). Expression of ApoA1 proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. (A-D) show the positive expression (arrow) at 6, 24, 48, 72 hours intraperitoneal SM group; (F-I) show the positive expression (arrow) at 6, 24, 48, 72 hours in tracheal SM group; (E) and (J) show the negative expression in control group (ApoA1 immunolabeling,, × 400, magnification, Bars = 20 μm). Positive expression ratios of CuZn-SOD proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group. Positive expression ratios of Mn-SOD proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group. Positive expression ratios of PON-1 proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group. Positive expression ratios of ApoA1 proteins in the epithelial cells of the alveolar septa of rats with SM (1LD50)-induced acute pulmonary injury at different time points. See Figure 1 for the rat treatment. x ± s, n = 8. *P < 0.05, compared with intraperitoneal propylene glycol control group. #P < 0.05, compared with tracheal propylene glycol control group. ΔP < 0.05, compared with tracheal SM group. Intraperitoneal and tracheal propylene glycol control group compared with corresponding control group.






Discussion
SM is a lipophilic and damaged cell alkylating agent. SM depletes glutathione (GSH) in the blood, liver, and lungs, increases the levels of oxidized glutathione (GSSG), and decreases the activity of antioxidant enzymes. A decrease in GSH content leads to the accumulation of naturally-produced ROS in cells, inducing lipids, proteins, and nucleic acid damage.19,20 ROS may alter remodeling of apoptosis, the extracellular matrix, the mitochondrial respiration system, maintenance of surfactant, cell proliferation, the anti-protease screen, effective alveolar repair responses, and immune modulation in the lungs. Following an increase in intracellular ROS levels, mitochondrial damage and dysfunction may lead to apoptosis. 16 The balance between oxidation and antioxidant processes is essential for maintaining normal cellular physiological metabolism. Otherwise, the pathological conditions of imbalance can be transformed. Cellular antioxidant systems include antioxidant enzymes (SOD, CAT, GSH-Px, and GST) and antioxidants (GSH, and vitamins E, A, and C). SOD catalyzes dismutation of the superoxide anion into H2O2, and CAT and GSHPx independently degrade H2O2 to water, inhibiting the formation of OH radical. 21 The present study indicated that SM induced a gradual increase in serum levels of SOD, CAT, and GSHPx, reaching a peak at 24 h, and then exhibited a downward trend. Under SM (1LD50) conditions, serum zymogram changes were more remarkable in the intraperitoneal SM group compared with the tracheal SM group. The results demonstrated that SM caused a transient increase in serum antioxidant enzyme levels through both administration routes, indicating that SM can be absorbed into the bloodstream via either route and induce a systemic oxidative stress response. A short-term increase in serum levels of SOD, CAT, and GSH-Px represents the body’s compensatory response to an imbalance between oxidation and antioxidants. This compensatory response is transient. If the antioxidant enzymes and antioxidants become depleted, it can lead to a toxic reaction from SM exposure. The results of the present study are consistent with Jafari et al.’s findings. 21 The findings suggested that the elevated enzyme levels may result in protection against SM-induced acute pulmonary injury by enhancing the production of superoxide anions to counteract oxidative stress in the alveoli and bloodstream. It was reported that the levels of antioxidant enzymes and antioxidants were significantly reduced in patients with SM-induced chronic pulmonary injuries, reflecting a severe imbalance between ROS production and the cellular antioxidant defense system.17,22 Therefore, supplementing with exogenous antioxidant enzymes and antioxidants at the appropriate time may be a crucial step in preventing the development of SM-induced pulmonary injuries.23,24
It has been demonstrated that SM-induced oxidative stress responses, including cytochrome C release and ROS accumulation, occur through ROS and reactive nitrogen species signaling pathways, resulting in cellular damage. 25 PON-1 is a multi-functional antioxidant enzyme that hydrolyzes a variety of organic compounds, including organophosphates, aryl esters, lactones, and specific oxidized lipids. 26 SOD forms a family containing three isoforms: cytosolic (CuZn), mitochondrial (Mn), and extracellular (CuZn) enzymes. Mn-SOD is moderately expressed in respiratory epithelium, alveolar type II epithelial cells, alveolar macrophages, and interstitial fibroblasts in hypoxia-exposed rats. In the airway epithelium, CuZn-SOD is highly expressed in ciliated epithelial cells.27,28
The present study also demonstrated that SM increased the expression levels of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in alveolar septa. Positive expression ratios of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 proteins in the intraperitoneal SM group significantly increased, positively correlating with exposure time. The results suggested that pulmonary oxidative stress responses were linearly enhanced during the acute phase of pulmonary injury, aligning with the pattern of SM-induced pulmonary injury, where damage progressively worsens over time. This was different from the transient elevation of serum enzyme spectrum (an excessive increase), which indicated that the oxidative stress response of SM to the target organ was present under continuous conditions. • Our results also revealed that oxidative stress is known to play a central role in SM-induced pulmonary toxicity.
29
SM triggers molecular and cellular pathways linked to oxidative stress resulting in cytotoxicity and loss of pulmonary structure and function. Tumor necrosis factor-α (TNF-α) has emerged as an important mediator of mustard-induced oxidative stress.30,31 This is due to TNF-α-induced activation of reactive nitrogen species and ROS generating enzymes such as inducible nitric oxide synthase and nicotinamide adenine dinucleotide phosphate oxidase in inflammatory cells, and upregulation of p38 mitogen-activated protein kinase and c-Jun N-terminal kinase, signaling pathways important in mitochondrial dysfunction and ROS production.32,33 TNF-α also contributes to a pro-oxidant environment by depleting intracellular glutathione.34,35 In response to oxidative stress, cells upregulate antioxidant enzymes in order to eliminate ROS and maintain cellular redox homeostasis.
36
Consistent with SM-induced oxidative stress, CuZn-SOD, Mn-SOD, PON-1, and ApoA1 protein expressions were increased in alveolar septa and levels of SOD, CAT, and GSH-Px were elevated in serum.
Studies have demonstrated that, in the acute phase of SM-induced pulmonary injury, the expression levels of CuZn-SOD, Mn-SOD, PON-1, and ApoA1 protein markers in bronchoalveolar lavage fluid and lung tissue were elevated, with corresponding increases in both mRNA expression and protein content.27,28,37 During the chronic period, however, the expression and content of the markers were significantly reduced.38,39 These results suggest that elevation in antioxidant markers during the acute phase of SM-induced pulmonary injuries indicates the body’s antioxidant ability, playing a notable role in maintaining normal physiological metabolism. 28 PON-1 and ApoA1 proteins in the chronic phase may be associated with SM-induced chronic obstructive pulmonary disease or bronchiolitis. 38
It was speculated that the oxidative stress mechanisms underlying SM-induced pulmonary injuries may be related to intracellular depletion of GSH and thioredoxin, alterations in the mitochondrial membrane, decreased activity of mitochondrial electron transfer chain, and accumulation of intracellular ROS. This is accompanied by the reduced levels of antioxidant enzymes, disruption of oxidation-related channel regulation, and an imbalance between oxidation and antioxidants in the internal environment. These processes damage cellular membrane lipids, proteins, and DNA, eventually leading to pulmonary edema, pulmonary surfactant injury, emphysema, and the condition known as “SM lung”.40,41 Under SM (1LD50) exposure, rats injected intraperitoneally with SM exhibited significantly higher serum inflammatory factor levels and protein expression in the alveolar septa compared with those administered SM via intratracheal instillation. The results suggest that differences in oxidative stress indices at the molecular level in SM (1LD50)-induced pulmonary injury are dependent on the route of exposure. Nevertheless, the limitations of the present study included the potential variability in SM absorption and distribution via different administration routes, the small sample size, and the lack of long-term follow-up.
Conclusions
Methods for developing pulmonary injury models have been successfully proposed to assess acute SM-induced pulmonary injury in rats. Under SM (1LD50) exposure, there were significantly higher serum oxidative stress biomarkers and protein expressions in the alveolar septa of rats injected intraperitoneally with SM compared with rats administered SM by intratracheal instillation. To investigate pulmonary injury, SM is delivered to the lungs either directly or indirectly through an animal model established using intraperitoneal injection and intratracheal instillation. Initial findings of oxidative stress indices in serum and pathology were presented in this study, with a more detailed characterization of the model to be reported separately. The model was utilized to explore the mechanisms and pathogenesis of SM-induced pulmonary injury and to evaluate the efficacy of potential treatments.
Footnotes
Acknowledgments
We thank Institute of Toxicology and Pharmacology in Academy of Military Medical Sciences.
Author contribution
Lu, Liu and Xiaoxuan, Hu: Writing – review, editing & original draft. Na, Zhang: Methodology, Data curation. Xiaoji, Zhu and Tao, Liu: Validation. Formal analysis, Conceptualization, Project administration. Yuxu, Zhong: Supervision, Conceptualization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Military Program on the great subject of Logistic Project of China (AWS17J008).
