Abstract
Thiodiglycol (TDG) is the main product of sulfur mustard hydrolysis and is an environmental contaminant. Subacute and subchronic oral toxicity studies with TDG were conducted in Sprague-Dawley rats. Neat TDG was administered by gavage at doses of 157, 313, 625, 1250, 2500, 5000, and 9999 mg/kg/d, 5 days per week, for 14 days. In the 14-day study, decreased body weight and food consumption were observed at 5000 mg/kg/d. In the 90-day study, rats received neat TDG at doses of 50, 500, or 5000 mg/kg/d for 5 days per week. A fourth group served as a sham control. Individual body weight and food consumption were measured weekly. At termination of the experiment, urine, blood, and tissue samples were collected. Rats displayed significant decreased body weight with no effect on food consumption following administration of TDG at 5000 mg/kg/d. Both male and female rats showed significant increased kidney weights at 5000 mg/kg/d. The organ to body weight ratios increased significantly for liver, kidneys, testes, and brain in males and adrenals in females for 5000 mg/kg/d. At all doses of TDG, hematological and clinical parameters and tissue histopathology remained unaltered. The no observed adverse effect level (NOAEL) for oral subchronic toxicity was 500 mg/kg/d. Benchmark dose (BMD) was derived from the decreased gain in body weight that was seen in male rats. A BMD based on a 10% decrease in body weight was 1704 mg/kg/d, and the lower confidence limit on the dose BMD, the BMDL, was 372 mg/kg/d.
Introduction
Sulfur mustard (HD) undergoes hydrolysis resulting in various products such as thiodiglycol (TDG), 1,4-oxathiane, 1,4-dithiane, 2-vinylthionethanol, and mustard chlorohydrin in biological or environmental matrices. 1,2 Thiodiglycol, the main hydrolysis product of HD, has been detected in ground and surface waters near a disposal site for chemical warfare agents. 3,4 Stockpiles of HD have been disposed of using a hydrolysis process that converts it into a biodegradable compound TDG. 5,6 Thiodiglycol is a precursor in the production of HD and is also considered a “Schedule 2” compound (dual-use chemical with low to moderate commercial use and high-risk precursors) within the terms of the Chemical Weapon Convention Treaty. 7,8 Thiodiglycol was also used as the starting material in preparation of HD. 9 Toxicity data on TDG are limited to only a few reports. The acute oral median lethal dose (LD50) value reported for male Wistar rats was 6610 mg/kg. In guinea pigs of mixed sexes, it is reported as 3960 mg/kg. 10 A subcutaneous LD50 of 4 g/kg for rats and mice and an intravenous LD50 of 3 g/kg for rabbits were reported. 11 In rabbits, the dermal LD50 was 20 mL TDG/kg/bw. 12 Thiodiglycol caused mild irritation to skin and moderate irritation to the eyes of rabbits. 12,13 It was also found not to be a sensitizer in guinea pigs. 14 Exposure to aerosolized TDG at 5.4 mg/L for 4 hours did not present an acute inhalation hazard to rats. 15 In a developmental study using rats, maternal toxicity was observed in dams receiving 3870 mg/kg during gestation; however, no adverse developmental effects were observed at 1290 mg/kg/d in rats. 16 There are no reports of long-term repeated dose toxicity information for TDG in mammals. Therefore, 14-day and 90-day mammalian toxicity studies were performed to obtain data needed to determine a no observed adverse effect level (NOAEL) for the derivation of a point of departure (POD) from which to characterize risk to humans and the environment.
Materials and Methods
Chemical
Thiodiglycol (2,2′-thiodiethanol, CAS # 111-48-8, 99% pure, lot number 057 01 EQ) was purchased from Aldrich Chemical Company, Inc, Milwaukee, Wisconsin. The TDG used was a clear, pale yellow liquid with a specific gravity of 1.221 at 25°C. Neat compound was used for the oral dosing studies.
Animals
Male and female Sprague-Dawley rats were purchased from the Charles River Laboratories, Raleigh, North Carolina. The animals were 4 weeks of age upon arrival at this facility and approximately 10 weeks old at the start of the TDG oral dosing. All rats were maintained at a set temperature of 65°F to 78°F, relative humidity of 40% to 70%, and 12-hour light/dark cycle. A certified pesticide-free rodent chow and drinking quality water were made available ad libitum. Rats were housed individually in stainless steel hanging wire cages. They were uniquely identified by number using cage cards and ear tags. The details were described in the report. 17 Animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals. 18 These studies were conducted in accordance with US EPA Health Effects Test Guidelines in compliance with Good Laboratory Practices. 19
Approximate Lethal Dose
Acute toxicity was estimated using an acute approximate lethal dose (ALD) method. A single bolus of TDG was orally administered to 8 rats of each sex using 16-gauge, 2-in stainless steel gavage needles. The doses selected were 579, 869, 1304, 1956, 2933, 4400, 6600, or 9900 mg/kg. Following the administration of test compound, the rats were visually observed daily and body weights were recorded 7 and 14 days postdosing. On day 14, all survivors were euthanized for gross necropsy.
Fourteen-Day Range-Finding Study
The 14-day range-finding study was conducted to determine the sublethal threshold from repeated oral exposures to ascertain appropriate treatment levels for the subsequent subchronic toxicity study. In all, 48 male and 48 female Sprague Dawley rats were randomly distributed using LAB CAT randomization program (Innovative Programming Associates, Princeton, New Jersey) into 8 treatment groups of 6 males and 6 females each. Dosage levels were set at 0 (control), 157, 313, 625, 1250, 2500, 5000 and 9999 mg/kg/d. Controls were given tap water at volumes equivalent to those receiving 9999 mg/kg/d. Rats were administered neat TDG via oral gavage using stainless steel needles 5 days per week, excluding weekends, for 2 weeks. Body weights and feeder weights were recorded on days −3, −1, 0, 1, 3, 7, and 14 days. Food consumption was also recorded on a weekly basis. Animals were observed daily for abnormal signs. Water consumption was not monitored during this study. Rats that died during the course of study were submitted for necropsy. All in-life data were recorded using the LABCAT Body Weights program.
Following the 14-day study period, the surviving rats were euthanized using carbon dioxide. Blood was collected via intracardiac puncture after CO2 euthanasia. Clinical chemistry and hematology values were obtained from blood samples. The brain, liver, kidneys, spleen, adrenals, and testes/ovaries were removed and weighed to obtain absolute organ weights, organ to body weight ratios, and organ to brain weight ratios. Gross necropsies were performed on all terminal animals. The following parameters were analyzed and comparisons to controls were made: body weights, food consumption, absolute organ weights, and organ to body weight ratios.
Hematology analysis included hematocrit, hemoglobin, red blood cell count, red blood cell distribution width, mean cell hemoglobin, mean cell volume, mean cell hemoglobin concentration, white blood cell (WBC) count, WBC differential (monocyte, lymphocyte, neutrophil, eosinophil, and basophil ratios), platelets, and mean platelet volume. Clinical chemistry analysis included plasma glucose, blood urea nitrogen, cholesterol, alkaline phosphatase, total protein, triglycerides, serum alanine aminotransferase, serum aspartate aminotransferase, creatinine, creatinine kinase, lactic dehydrogenase, calcium, and total bilirubin concentrations.
90-Day Subchronic Study
A 90-day oral toxicity study was conducted in male and female rats. Forty male and 40 female Sprague-Dawley rats between 7 and 8 weeks old were used for this study. Following a quarantine/acclimatization period of 1 week, animals were randomly distributed using the LABCAT randomization program into 4 groups consisting of 10 male and 10 female rats each. Dosage levels of TDG were set at 50, 500, and 5000 mg/kg/d. The fourth group was a control where animals were treated in a like manner as those in the test compound groups except that they were sham gavaged using an empty syringe fitted with a 16 GA × 2 in feeding needle. Neat TDG was administered on a 5-day per week schedule (excluding weekends) for 91 to 92 calendar days.
Body and feeder weights for each rat were recorded on test days −3, −1, 0 (first day of compound administration), 1, 3, 7, and weekly thereafter. Doses were adjusted weekly to reflect the change in individual body weights. Animals were observed daily for any clinical toxic signs. All data were recorded using a PC in the LABCAT Body Weights program. Water consumption was not monitored during this study.
Prior to the commencement of the dosing period and 7 days before termination, ophthalmic examinations were performed by an attending veterinarian on all control and 5000 mg/kg/d rats. Urine samples for routine and microscopic urinalyses were collected by placing rats in metabolism cages toward the end of the in-life portion of the study.
Necropsy
Following the study period, the surviving rats were euthanized using carbon dioxide. Just prior to death, blood samples were collected by intracardiac puncture from euthanized rats. Clinical chemistry and hematology values were determined from collected samples. Each rat was then submitted for complete necropsy. Brain, liver, kidneys, spleen, adrenals, and testes/ovaries were removed and weighed for comparison among groups. These weights were also used for calculations of organ to body weight and organ to brain weight ratios. The following parameters, by test group, were considered when test results were analyzed and comparisons to controls were made: body weights, food consumption, absolute organ weights, organ to body weight ratios, and organ to brain weight ratios.
The tissues harvested for histopathological evaluation were brain, pituitary, thyroid w/ parathyroid, thymus, lungs, trachea, heart, bone marrow, salivary gland, liver, spleen, kidney, adrenal, pancreas, gonad, uterus, aorta, esophagus, stomach, duodenum, jejunum, ileum, cecum, colon, urinary bladder, lymph node, peripheral nerve, thigh musculature, eye, spinal cord (2 or 3 levels), and exorbital lachrymal gland.
Hematology and Clinical Chemistry
The hematology parameters measured or calculated included hematocrit, hemoglobin, red blood cell count, red blood cell distribution width, mean cell hemoglobin, mean cell volume, mean cell hemoglobin concentration, WBC count, differential white count (monocyte, lymphocyte, neutrophil, eosinophil, and basophil ratios), platelets, and mean platelet volume. The clinical chemistry parameters measured included plasma glucose, blood urea nitrogen, cholesterol, alkaline phosphatase, total protein, triglycerides, serum alanine aminotransferase, serum aspartate aminotransferase, creatinine, creatinine kinase, lactic dehydrogenase, and calcium total bilirubin concentrations.
Urinalysis parameters measured included volume, color, appearance, pH, glucose, bilirubin, urobilinogen, ketone, blood, protein, nitrite, leukocytes, and microscopic granular casts (Siemens Healthcare Diagnostics Multistix Reagent Strips,Tarrytown, NY). Specific gravity was measured with a refractometer (American Optical Corp., Buffalo, New York).
Statistical Analysis
Food consumption, body weights, weight gains, organ to brain weight ratios, and organ to body weight ratios were statistically compared using a one-way analysis of variance (ANOVA) and when significance was observed, the data were further analyzed using Dunnett post hoc test. Clinical chemistry, hematology, and urinalysis were statistically compared using a one-way ANOVA and Bonferroni post hoc test. Where a normality test failed, an ANOVA on ranks was performed. Statistical significance was defined at the P < .05 level. 20
Determination of Benchmark Dose
Benchmark dose (BMD) is an alternative approach to the NOAEL/LOAEL approach in the development of a POD that has been used for a number of years in dose–response assessment. The BMD method gained favor with the risk assessment community because it incorporates and conveys more information than the NOAEL/LOAEL method. The dose–response assessment is a 2-step process, one defining POD and other is extrapolating from POD for relevance to environmental exposure.
The BMD and its 95% lower confidence limit for 10% effect levels (BMDL10) were determined according to the United States Environmental Protection Agency’s BMD approach. The dose–response relationship was based on the decrease in body weight gain data. This finding represented the most sensitive dose-responsive adverse event in the 90-day study. The BMD analysis used Benchmark Dose Software (BMDS, version 2.4) to calculate the POD using the standard suite of models for continuous data. 21 The decrease in body weight gain was plotted against the dose administered.
Results
Approximate Lethal Dose
Acute oral administration of neat TDG produced no toxic effects or deaths in male and female rats at oral dosages as high as 9900 mg/kg. The only effects observed occurred in a single male rat given 9900 mg/kg where slight lethargy starting 1-hour posttreatment was observed, but it recovered within 4 hours. No other toxic effects were noted.
Fourteen-Day Oral Toxicity Study
In the 14-day study, rats in the high-dose group (9999 mg/kg/d) experienced lethality in 4 of 6 males and 5 of 6 females. All deaths occurred within 1 to 3 days following the commencement of exposure. In all cases, mortality was preceded by lethargy of increasing severity.
Male and female rats dosed with TDG at 5000 and 9999 mg/kg/d consumed less feed when compared to controls beginning on the first day. There was a distinct, but not statistically significant, dose-related trend toward higher absolute kidney weights for those males receiving 1250, 2500, and 5000 mg/kg/d. For females, kidney–body and kidney–brain weight ratios were higher than controls but not significantly different. Absolute kidney weights in females were higher than controls in the 5000 mg/kg/d groups. There were no significant changes in clinical chemistries or hematology values from blood collected at the termination of the study (data not shown). 17
Based upon the results of this study, 5000 mg/kg/d dose was selected as the highest dosage level used for the 90-day subchronic oral study.
90-Day Oral Toxicity Study
Ophthalmic examinations were performed prior to the scheduled start of the 90-day study and within a week of the scheduled necropsies. All observations were within normal limits. Twelve rats randomly chosen for this study, but housed in the same room, were screened for serology, bacteriology, pathology, and parasitology at the conclusion of the 90-day study (6 males and 6 females). The results from this testing showed the animals to be in good health.
During course of the study, death and unscheduled sacrifices occurred in both sexes which appeared to be not compound related; necropsy of animals’ revealed urogenital complications in 1 male and dosing error as noted from perforated esophagus in 2 female and in control rats (2 males and 1 female).
There were no consistent signs of toxicity noted in the course of daily observations during the in-life study period. At times, several rats of both sexes displayed irritated behavior during the dosing procedure, but no dose-related pattern was observed.
The results of food consumption presented show that there were no significant differences in food consumption between TDG-treated groups and control group at all dose level except during the beginning of study in the rats from 5000 mg/kg/d group (day 3 for males and day 1 for females). This may be due to early adjustments for the food consumption (Tables 1 and 2).
Mean Food Consumption (g) of Male Rats Dosed Orally With Thiodiglycol During 90 Days, 5 d/Week.a
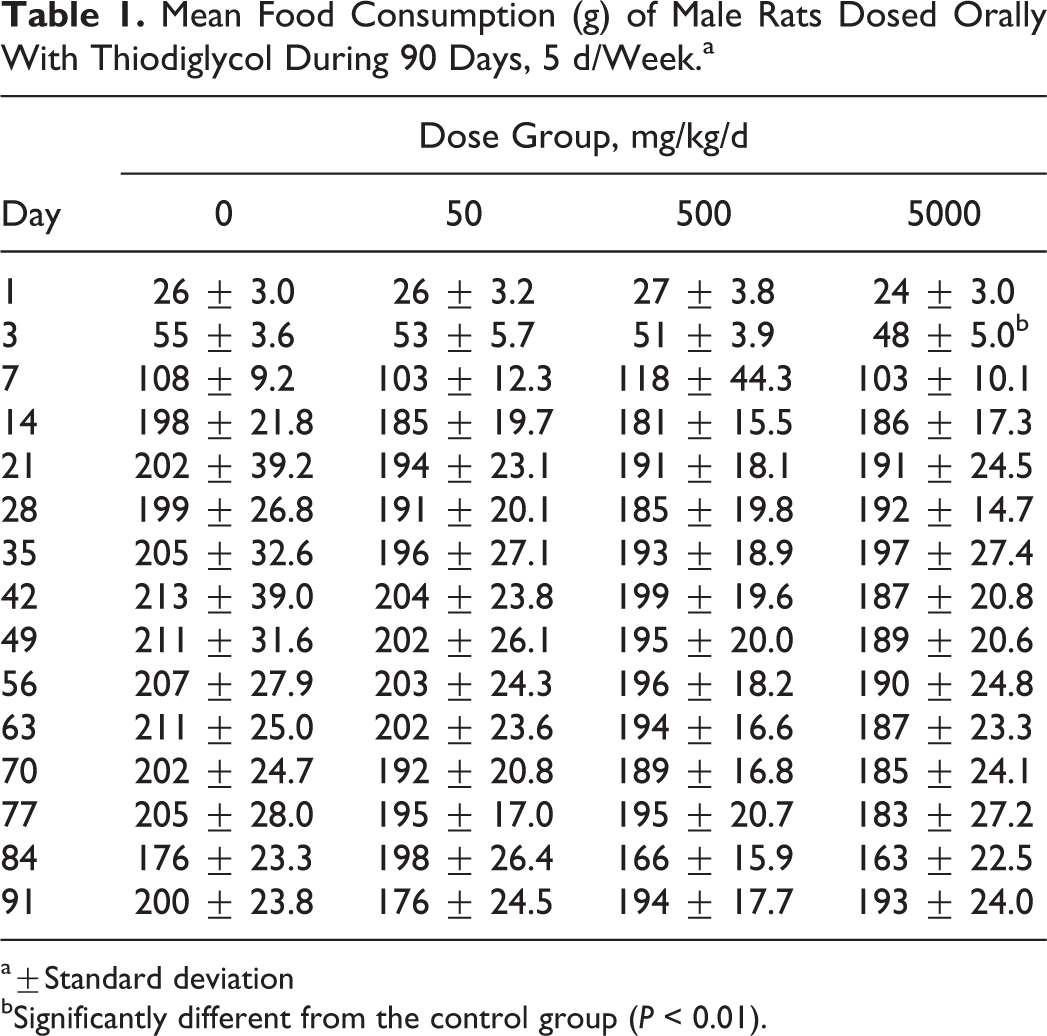
a±Standard deviation
bSignificantly different from the control group (P < 0.01).
Mean Food Consumption (g) of Female Rats Dosed Orally With Thiodiglycol During 90 Days 5 d/Week.a
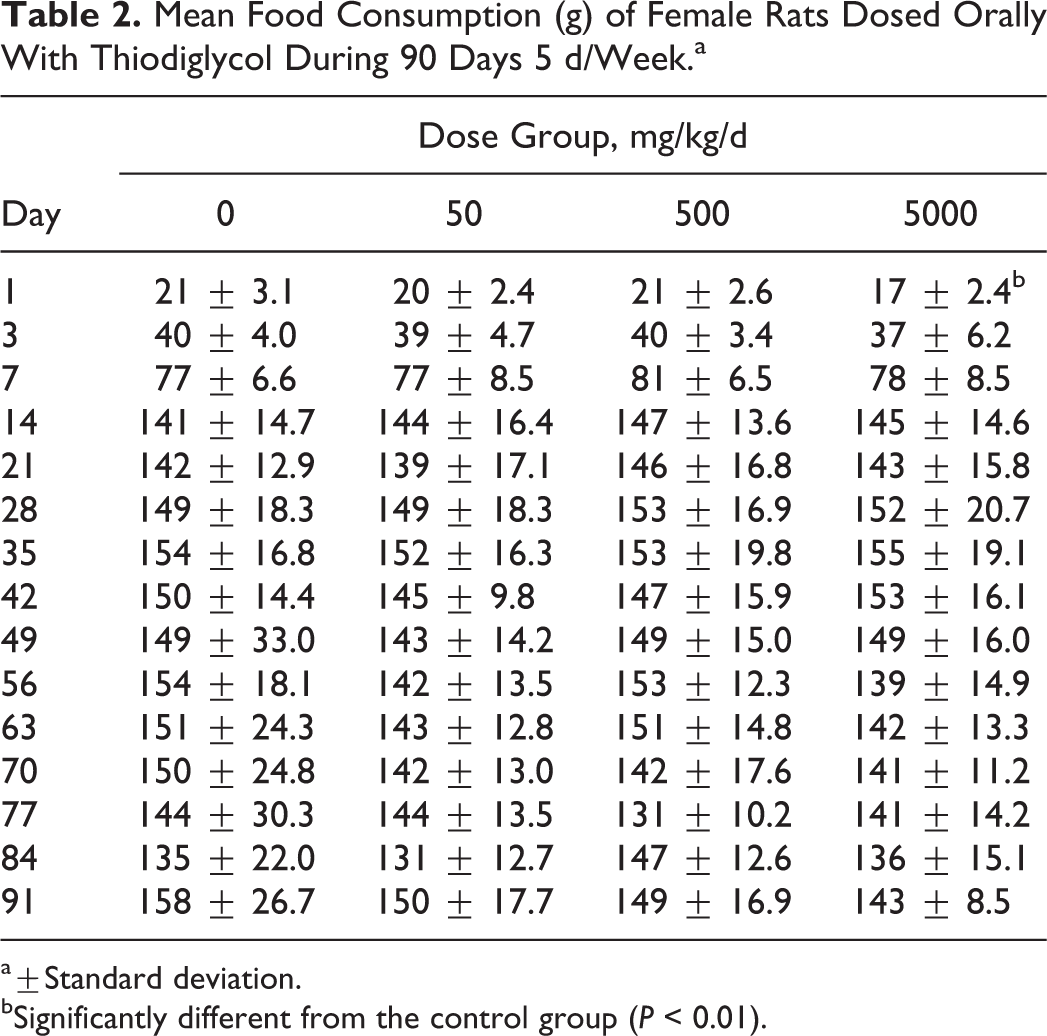
a±Standard deviation.
bSignificantly different from the control group (P < 0.01).
Administering TDG at 5000 mg/kg/d affected absolute body weights and body weight gains in both male and female rats. Mean body weights were significantly lower in males from this group than controls from week 9 through 13. Correspondingly, females administered 5000 mg/kg/d of TDG also resulted in lower mean body weights toward the end of the 90-day study and significantly lower total body weight gains. The results of mean body weights are presented in Tables 3 and 4.
Mean Body Weights (g) for Male Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
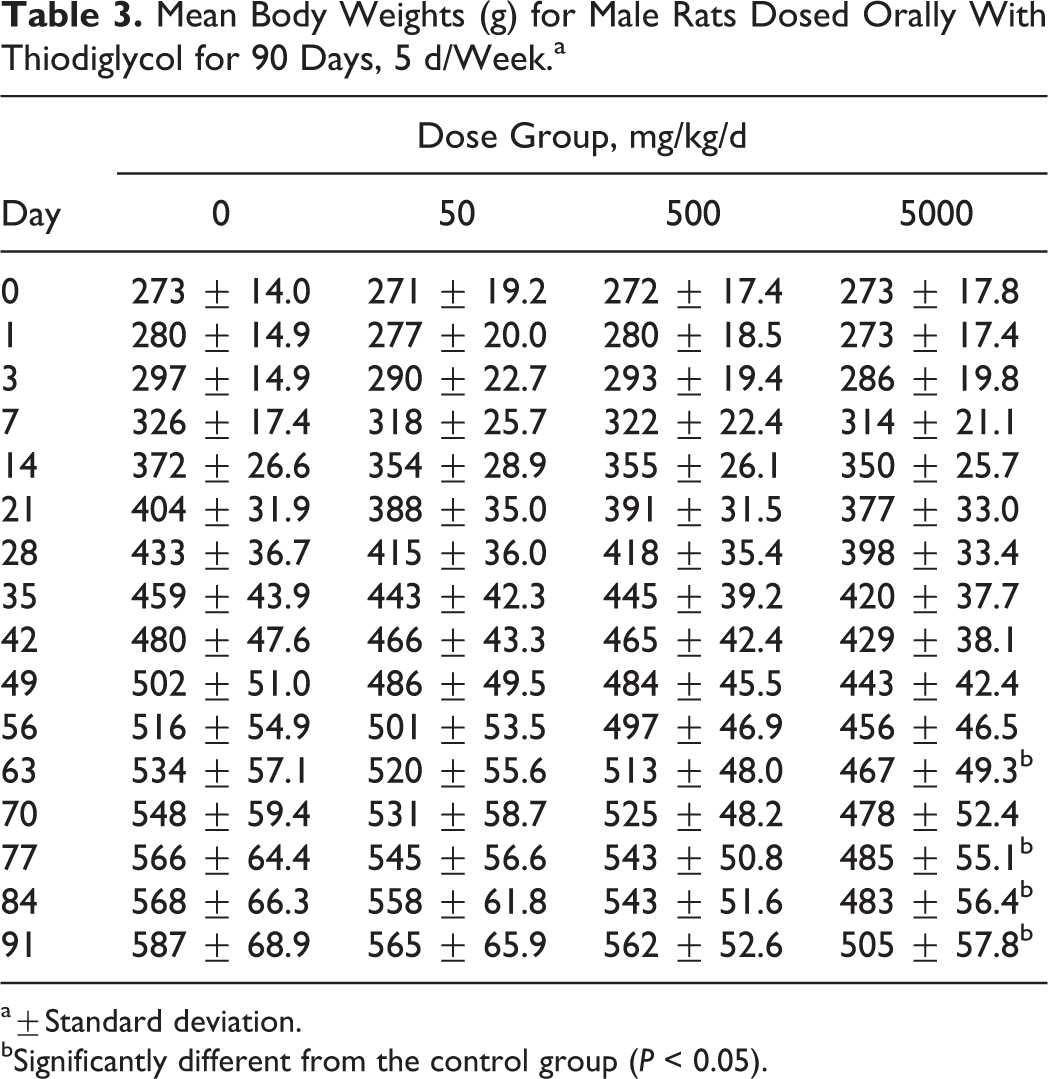
a±Standard deviation.
bSignificantly different from the control group (P < 0.05).
Mean Body Weights (g) for Female Rats Dosed Orally With Thiodiglycol During 90 Days, 5 d/Week.a
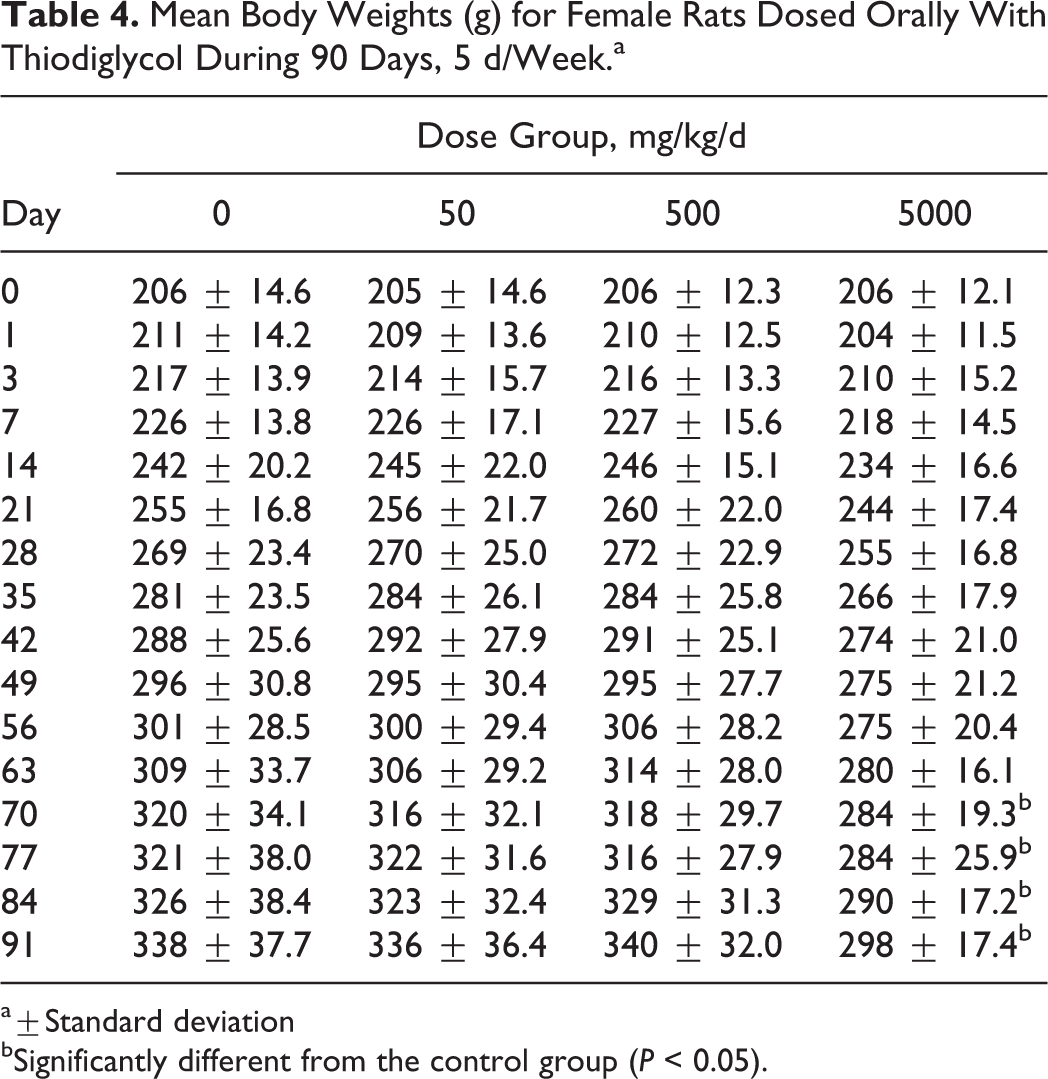
a±Standard deviation
bSignificantly different from the control group (P < 0.05).
All hematological and blood chemistry parameters for TDG-treated rats were comparable to values obtained for control groups. There were no significant changes in any of the parameters studied. The results of these parameters are presented in Tables 5 to 8.
Hematology Results of Male Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
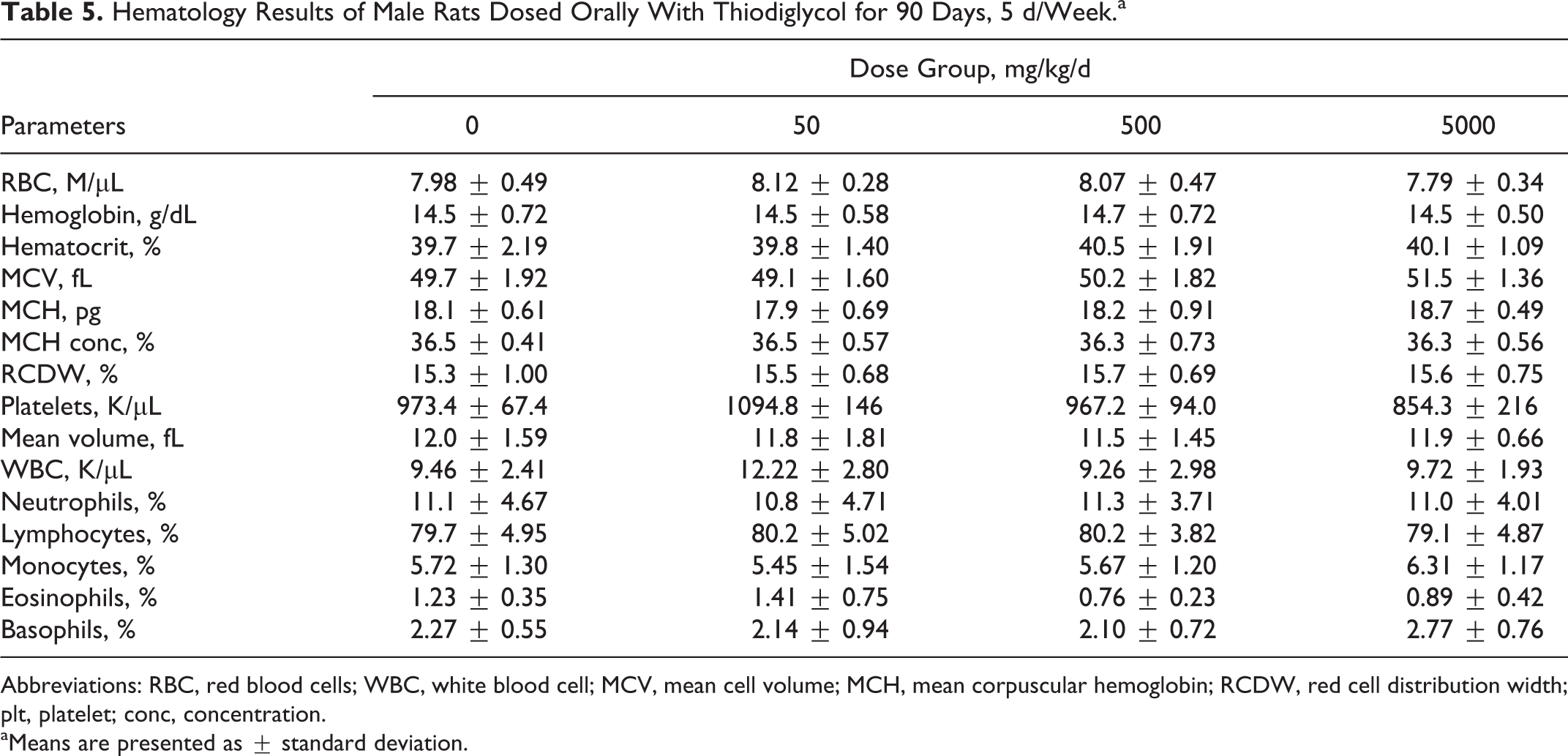
Abbreviations: RBC, red blood cells; WBC, white blood cell; MCV, mean cell volume; MCH, mean corpuscular hemoglobin; RCDW, red cell distribution width; plt, platelet; conc, concentration.
aMeans are presented as ± standard deviation.
Hematology Results of Female Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
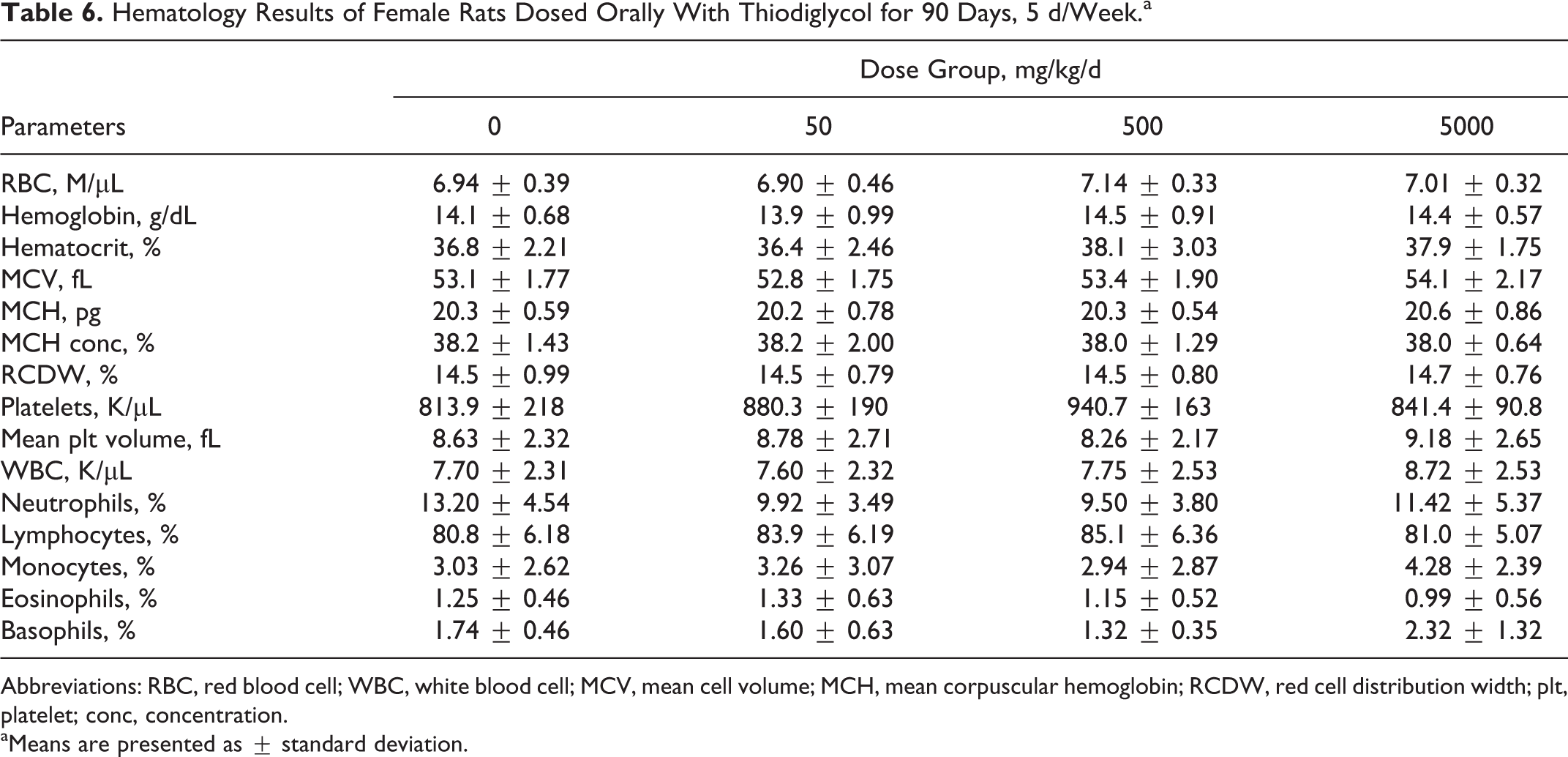
Abbreviations: RBC, red blood cell; WBC, white blood cell; MCV, mean cell volume; MCH, mean corpuscular hemoglobin; RCDW, red cell distribution width; plt, platelet; conc, concentration.
aMeans are presented as ± standard deviation.
Clinical Chemistry Values of Male Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
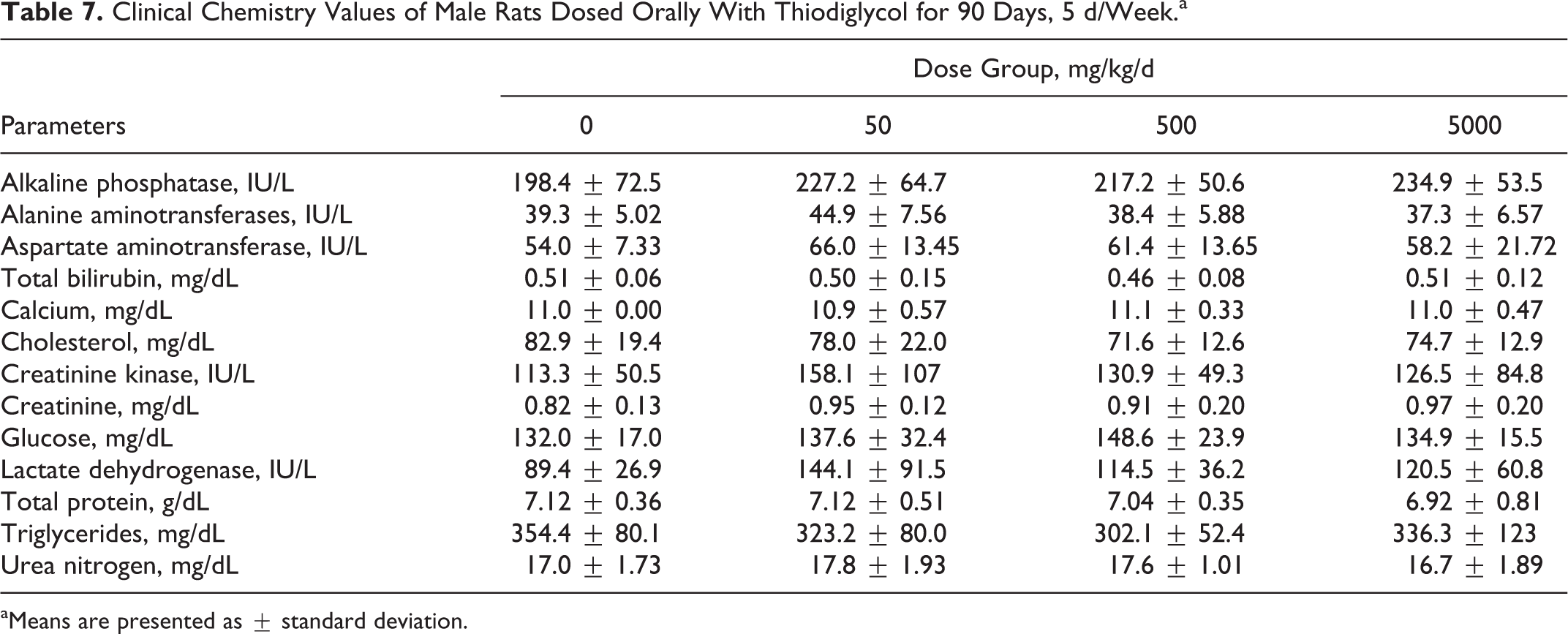
aMeans are presented as ± standard deviation.
Clinical Chemistry Values From Female Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
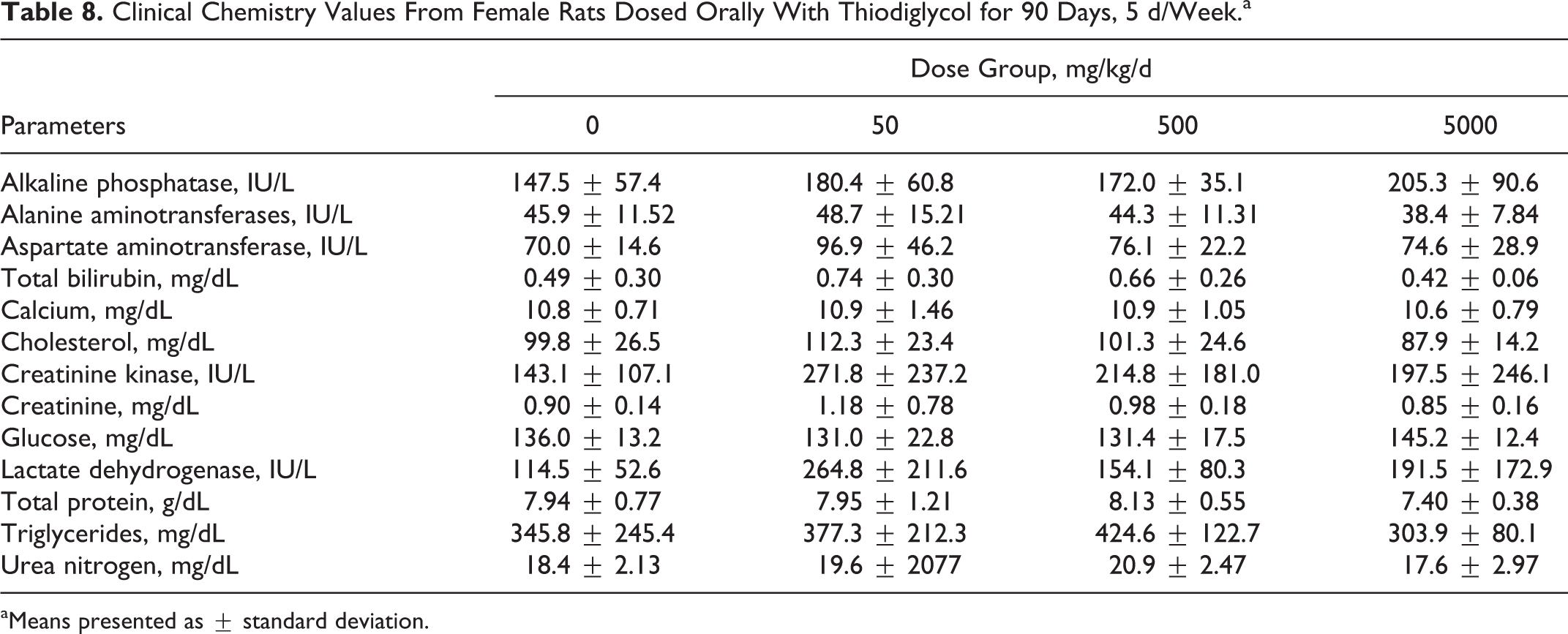
aMeans presented as ± standard deviation.
Absolute mean kidney weights and kidney to body weight ratios were significantly higher in both sexes than in the control animals (Table 9). Liver, kidney, testes, and brain weights to body weight ratios were significantly higher than controls at 5000 mg/kg/d in male rats. In the high-dose females, the adrenal and kidney to body weight ratios were also significantly higher than controls (Table 10).
Mean Body Weight and Absolute Organ Weights (g) From Male and Female Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
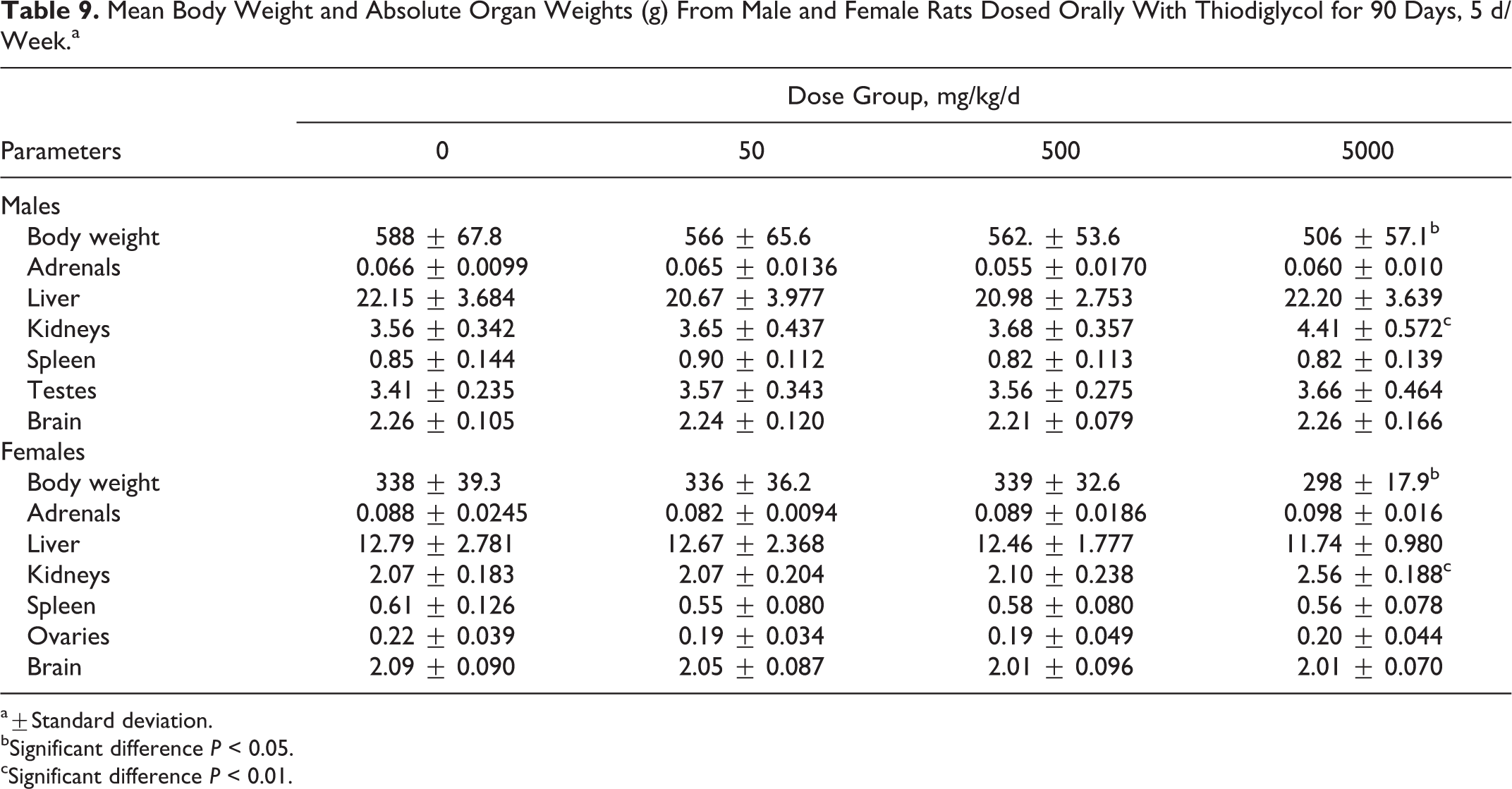
a±Standard deviation.
bSignificant difference P < 0.05.
cSignificant difference P < 0.01.
Organ Weights (Percentage of Body Mass) in Male and Female Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
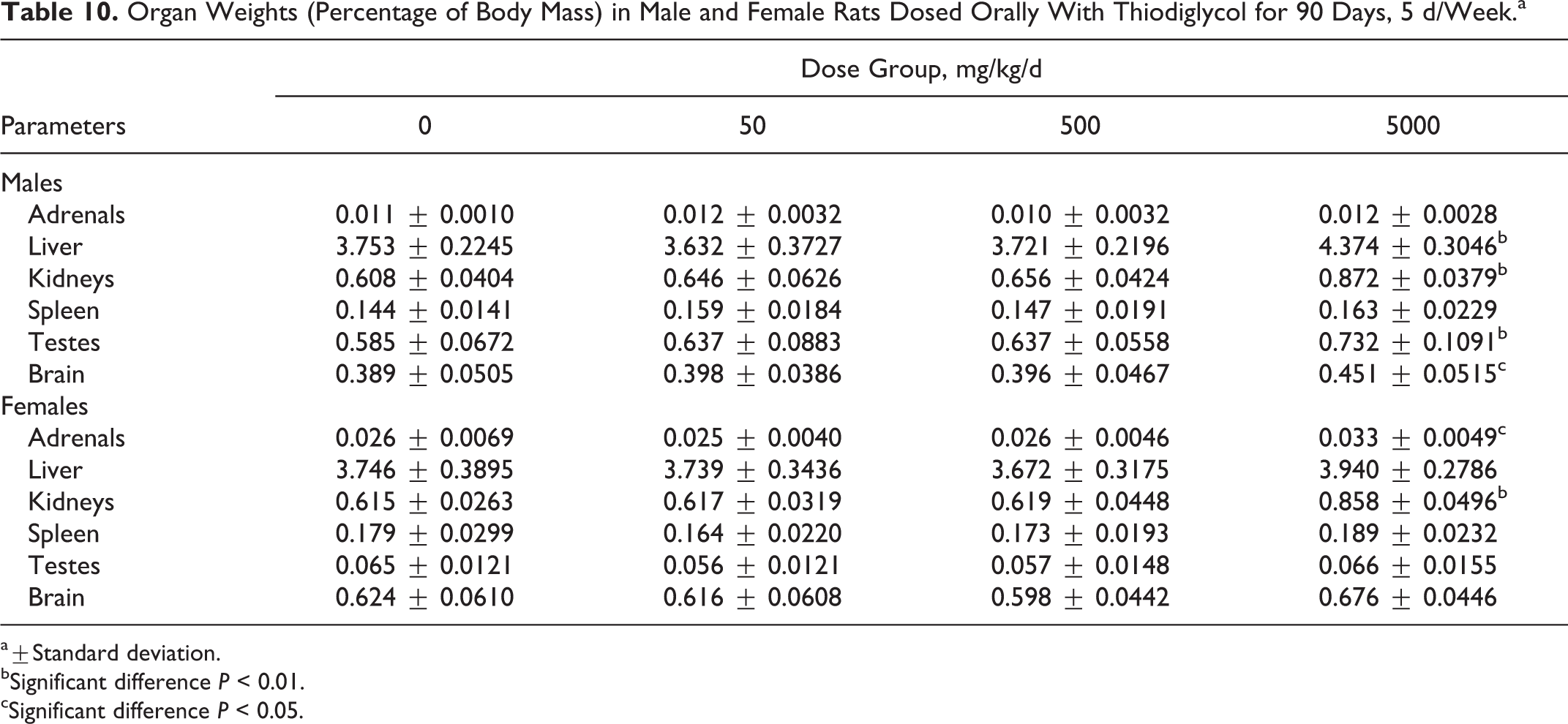
a±Standard deviation.
bSignificant difference P < 0.01.
cSignificant difference P < 0.05.
There were no consistent gross pathological changes in rats among any of the test groups or controls. Histopathological examination of harvested tissues did not reveal any test compound-related anomalies (Pathology Contract report By Dr George A. Parker, Biotechnics, Sterling, Virginia). 17
Analysis of the urine collected from each test animal revealed several significant changes. The volume of urine produced by rats receiving TDG at 5000 mg/kg/d was significantly higher than that of the controls. Urine pH was significantly lower in males (5000 mg/kg/d) and females receiving 5000 and 500 mg/kg/day. Specific gravity of the urine was also significantly higher in males receiving 5000 mg/kg/d than controls. Granular casts were found only in microscopic examination of urine from female rats receiving 5000 mg/kg/d. The urinalyses results are presented in Table 11.
Results of Urinalyses From Male and Female Rats Dosed Orally With Thiodiglycol for 90 Days, 5 d/Week.a
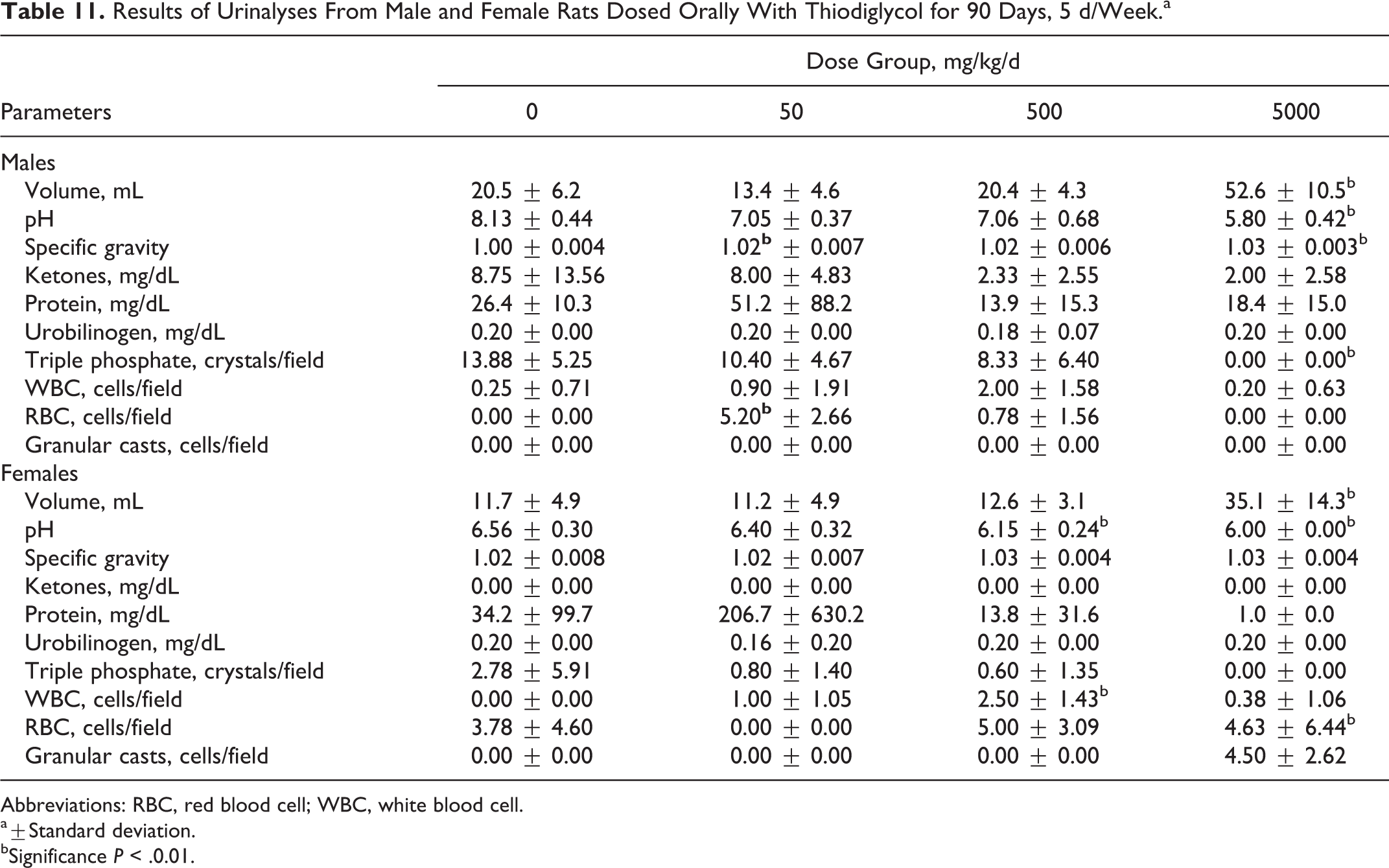
Abbreviations: RBC, red blood cell; WBC, white blood cell.
a±Standard deviation.
bSignificance P < .0.01.
Benchmark dose analysis was performed on male and female body weight data. The BMD run on the female body weights did not meet the criteria for a valid estimate. The male body weight data run in the session files showed an adequate fit from several of the models. An inspection of the Bench mark response (BMR) plots from these models revealed that most estimates missed the control mean. We reran these models with a specified background level set to the control mean. The Hill and Exponential 4 and 5 models gave an adequate visual fit to the data, but the levels were still more than 3-fold different from the other runs. Based on this analysis, we chose the most conservative BMDL estimate of 372 mg/kg/day from the Hill model with specified parameters. The BMD was 1704 mg/kg/d. (figure 1)
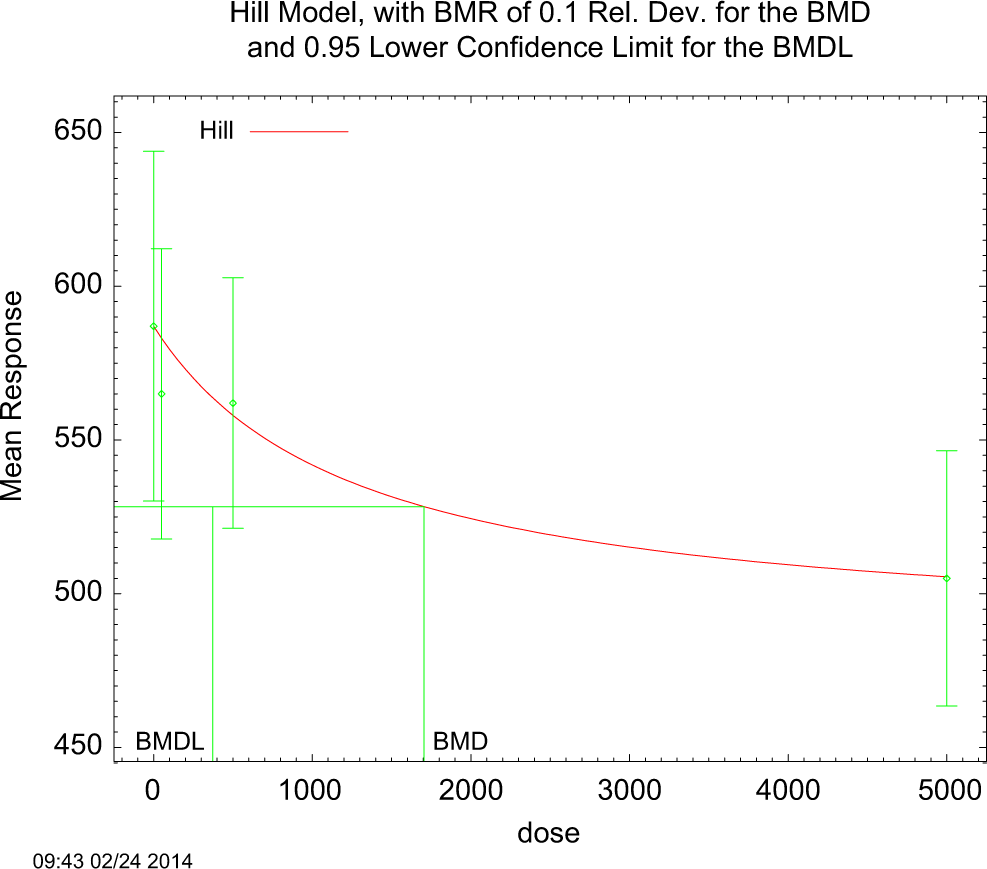
The experimental data and the BMDs output with 95% confidence interval for Hill model of continuous data for the relationship between ingested dose (mg/kg/d) and the body weight. The benchmark dose (BMD) and lower confidence limit on the dose used as the BMD (BMDL) values represent estimates for a 10% decrease in body weight as result of treatment with thiodiglycol (TDG).
Discussion
There is considerable information on the toxicology and pharmacology of chemical warfare agent HD. It is highly toxic to animals and humans. 22 Thiodiglycol is the major hydrolysis product of HD and has been detected as a contaminant of soil and water at certain army installations where HD production, storage, or disposal activities were executed. 2 It is considerably less toxic than HD. The acute oral LD50 value reported for male Wistar rats was 6610 mg/kg and for guinea pigs of mixed sexes were 3960 mg/kg. 10 In mammals, TDG is found as thiodiglycol sulfoxide in urine of rats, a primary metabolite of sulfur mustard. 23,24 Thiodiglycol has been found as urinary metabolite in humans either accidentally or as occupational exposure to sulfur mustard. 25 It was also detected in biological samples collected from victims of alleged attacks with mustard gas. 26,27 Sulfur mustard is reported as mutagenic and carcinogenic in animals. The genotoxicity studies of TDG resulted in negative responses in in vitro Ames test and mouse lymphoma. It was also negative in the in vivo mouse micronucleus assay but was positive in in vitro chromosomal aberrations in Chinese hamster ovary cells. 28,29,30,31 The HD showed an increase in the expression of urokinase in 3T3 fibroblasts at 100 µmol/L, while its degradation product TDG at 100 µmol/L did not influence the expression of urokinase (plasminogen activator) which is associated with inflammatory reaction, cell migration, tissue destruction, and remodeling. 32 Recently, the fate and toxicity of chemical warfare agents and degradation products were described. 3 However, the mammalian toxicity data on TDG are limited to only a few acute studies. Therefore, we collected subchronic toxicity data to be used in developing standards for health and environmental risk.
Our acute toxicity data on neat TDG in both sexes of rats show an ALD of about 9900 mg/kg. The 14-day repeated dosing data showed that oral bolus exposures of 5000 and 9999 mg/kg/d reduced body mass and resulted in certain organ weight abnormalities but did not affect hematological or clinical chemistry parameters.
In the 90-day study, rats dosed at 50, 500, and 5000 mg/kg/d for 5 days per week showed no significant effects in food consumption or hematological and clinical chemistry parameters at all dose levels tested. However, in the high-dose males and females, there was a significant decrease in body weights and a significant increase in absolute kidney weights. The data on blood glutathione antioxidant and hepatic mixed functions oxidase in rats subjected to TDG administration obtained from this subacute and subchronic toxicity (50, 500, and 5000 mg/kg/d) study showed no significant variation in antioxidant systems at any of the dose groups. But, an increase in pentoxyresorufin O-dealkylation and a significant decrease in cytochrome b5 in the high-dose (5000 mg/kg/d) males were observed; there was no change in these parameters in female rats. 33,34 Sulfur mustard, the parent compound of TDG at sublethal doses decreases enzyme antioxidant activities (superoxide dismutase, catalase, and glutathione peroxidase) in rat erythrocytes, leukocytes, spleen, and brain after topical application in rats. 35 Sulfur mustard also causes oxidative stress and depletion of antioxidants in muscle, liver, and kidney in rats. 36 Thiodiglycol during subchronic oral exposures in the present study showed no effects on hematology or other clinical parameters. Thiodiglycol is metabolized by oxidative processes in rats following intraperitoneal administration. 37 Metabolites from oral TDG exposure in rats that have been identified include thiodiglycol sulfoxide (>90%) as the primary metabolite, and smaller amounts of thiodiglycol sulfones, S-(2-hydroxyethylsulfinyl), acetic acid, and its sulfide analogue S-(2-hydroxyethylthio) acetic acid.
Our subchronic study data demonstrated that male and female rats dosed orally with TDG at 50, 500, and 5000 mg/kg/d for 5 days/week showed effects only at 5000 mg/kg/d, which were manifested by a decrease in body mass and an increase in kidney mass. The results of urine analysis showed that both sexes excreted more volume with TDG treatments; treated animals also showed low urine pH and this may have been due to excreted acid metabolites. 37 Histopathological evaluation showed no histological changes in the kidney. The increase in kidney weights in both sexes may have been due to the high volumes of urine excretion or possibly induction of enzymes as seen in the livers of TDG-treated rats. 34 In the absence of histological changes, it was considered not an adverse effect. The decreased body weight gains in both sexes were the most sensitive adverse effect. Based on this result, we estimated the NOAEL to be 500 mg/kg/d, 5 days per week oral exposure. Based on the BMD analysis, the BMDL estimate of 372 mg/kg/d from Hill model is slightly lower than the NOAEL.
Footnotes
Acknowledgments
We thank Dr Wilfred McCain and Maj. Sang H. Lee for critical review of this manuscript and Ms Allison Jackovitz for the help in preparation of tables.
Authors’ Note
This work was presented in part at the 38th Annual Meeting of the Society of Toxicology, March 14-18, 1999. New Orleans, LA. Toxicologist 1999; 48:1-S. P. 318.
Authors’ Contribution
Angerhofer A. Richard contributed to conception and design, acquisition, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Michie W. Mark contributed to conception, acquisition, and interpretation. Leach J. Glenn contributed to conception and design, analysis and interpretation, drafted manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Johnson Mark contributed to design, analysis and interpretation, drafted manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Gunda Reddy contributed to conception, analysis and interpretation, drafted manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and /or publication of this article: US Army Corps of Engineers’ Environmental Technology Program.
