Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of 24 Vitis vinifera (grape)-derived ingredients and found them safe in the present practices of use and concentration in cosmetics. These ingredients function in cosmetics mostly as skin-conditioning agents, but some function as antioxidants, flavoring agents, and/or colorants. The Panel reviewed the available animal and clinical data to determine the safety of these ingredients. Additionally, some constituents of grapes have been assessed previously for safety as cosmetic ingredients by the Panel, and others are compounds that have been discussed in previous Panel safety assessments.
Introduction
As given in the Code for Federal Regulations (21CFR101, subpart C), grapes are among the 20 most frequently consumed raw fruits and are subject to regulation by the Food and Drug Administration (FDA) as foods.
This report assesses the safety of the following 24 Vitis vinifera (grape)-derived ingredients for use in cosmetic formulations:
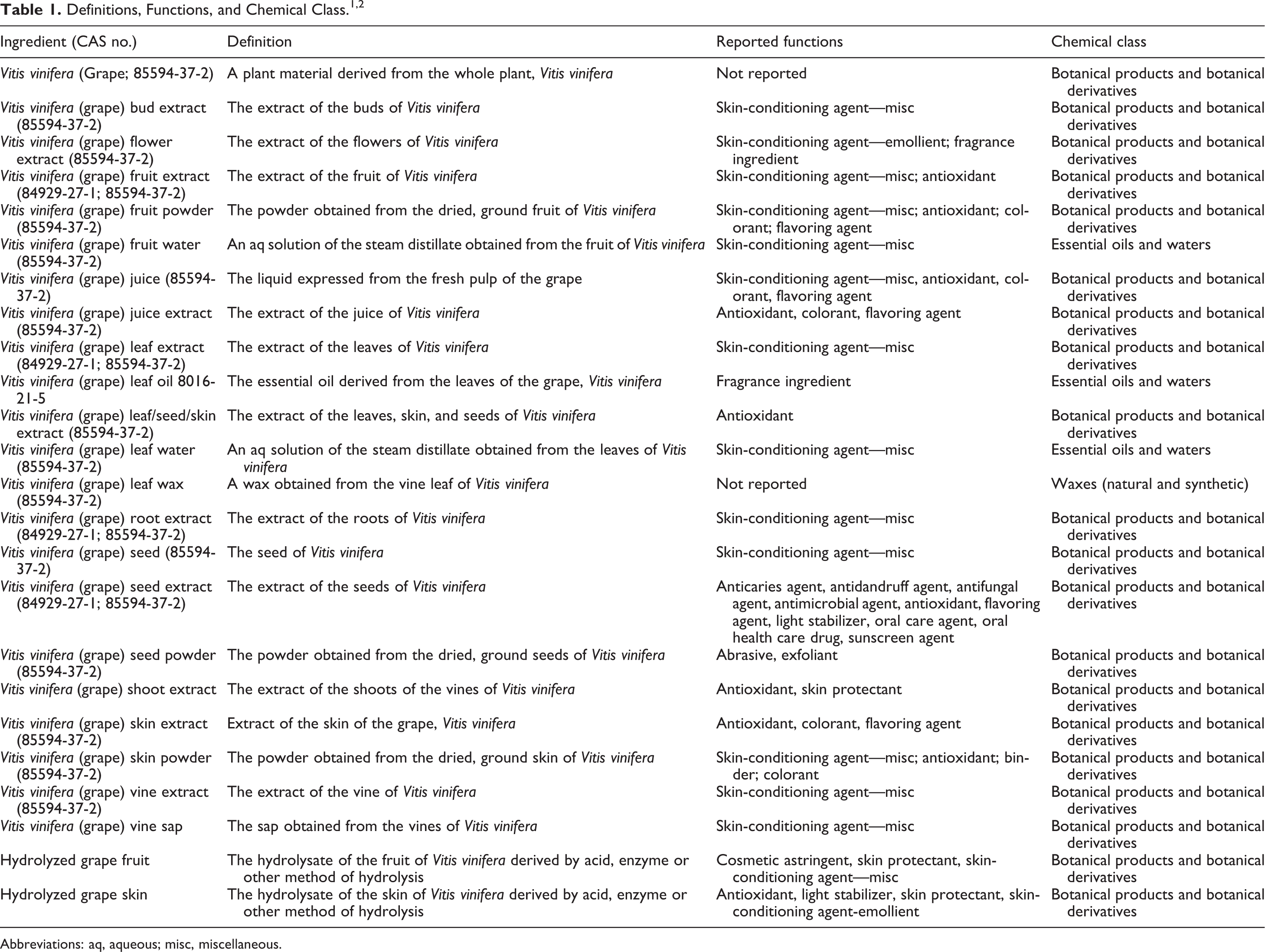
vitis vinifera (grape);
vitis vinifera (grape) bud extract;
vitis vinifera (grape) flower extract;
vitis vinifera (grape) fruit extract;
vitis vinifera (grape) fruit powder;
vitis vinifera (grape) fruit water;
vitis vinifera (grape) juice;
vitis vinifera (grape) juice extract;
vitis vinifera (grape) leaf extract;
vitis vinifera (grape) leaf oil;
vitis vinifera (grape) leaf/seed/skin extract;
vitis vinifera (grape) leaf water;
vitis vinifera (grape) leaf wax;
vitis vinifera (grape) root extract;
vitis vinifera (grape) seed;
vitis vinifera (grape) seed extract;
vitis vinifera (grape) seed powder;
vitis vinifera (grape) shoot extract;
vitis vinifera (grape) skin extract;
vitis vinifera (grape) skin powder;
vitis vinifera (grape) vine extract;
vitis vinifera (grape) vine sap; Hydrolyzed grape fruit; Hydrolyzed grape skin.
These ingredients are reported to have many functions in cosmetics, most frequently as skin-conditioning agents. 1,2 Some of these ingredients are reported to function as antioxidants, flavoring agents, and/or colorants (Table 1).
Abbreviations: aq, aqueous; misc, miscellaneous.
The safety of Vitis vinifera (grape) seed oil and hydrogenated grapeseed oil was reviewed previously by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) in the Safety Assessment of Plant-Derived Fatty Acid Oils as Used in Cosmetics, at which time the Panel concluded that these ingredients are safe as used in cosmetics. 3 These 2 ingredients are not included in this safety assessment.
The detailed chemical composition of vitis vinifera (grape) is given later in this assessment. As shown in Table 2, some of the constituents of grape, such as ascorbic acid, biotin, malic acid, and so on, are cosmetic ingredients for which a Panel safety assessment is available; others are compounds that have been discussed in previous Panel safety assessments. 4 –19
Conclusions of CIR Safety Assessments on Ingredients That Are Constituents of Vitis vinifera (Grape).

Abbreviation: CIR, Cosmetic Ingredient Review.
Although many studies conducted using vitis vinifera (grape)-derived ingredients address health claims, antioxidant activity, and so on, this safety assessment only includes studies that relate directly to the safety of the cosmetic use of these ingredients.
In many of the published studies, it is not known how the substance being tested is compared to the cosmetic-grade ingredient. Therefore, if it is not known whether the ingredient being discussed is a cosmetic ingredient, the test substance will be identified as “grape…” (eg, grape seed extract); if it is known that the substance is a cosmetic ingredient, the terminology “Vitis vinifera (grape)…” (eg, vitis vinifera [grape] seed extract) will be used.
Chemistry
Definition
The definitions of the vitis vinifera (grape)-derived ingredients are provided in Table 1. Vitis vinifera is also known as wine grape, European grape, 20 and grapevine. 21
Chemical and Physical Properties
Chemical and physical property data are provided in Table 3. 22 –29
Chemical and Physical Properties.

Abbreviation: aq, aqueous.
Composition
Grapes contain fruit acids, and the unripe fruit contains 34 ppm oxalic acid. 20,26 Grape seeds contain 6% to 20% oil. Phenols are the third most abundant constituent in grapes; carbohydrates and fruit acids are the most and second most abundant, respectively. 30 The total extractable phenolics in grapes are present at ≤10% in the pulp, 60% to 70% in the seeds, and 28% to 35% in the skin.
The amount of a constituent present in the plant varies with the region in which it is grown. 26 For example, fruit of grapes from Africa and Asia contained 50.0 μg β-carotene equivalents per 100 g of fruit while elsewhere trace β-carotene equivalent were present in the fruit. The cultivar, climate condition, and degree of maturation also affect the composition, as does whether the grapes are red or white. 30
It has also been shown that the amount of a constituent present in an extract is dependent on the medium used during extraction and the variety of vitis vinifera (grape) used. 31 For example, a red grape methanolic extract, red grape water extract, white grape methanolic extract, and white grape water extract each contained 0.22, 0.04, 0.01, and 0.02 mg/g trans-resveratrol, respectively; 0.9, 0.35, 2.25, and 4.09 mg/g (+)-catechin, respectively; 1.1, 0.32, 1.08, and 2.10 mg/g (-)-epicatechin, respectively; and 0, 0.13, 0.04, and 0.03 mg/g quercetin, respectively.
Melatonin (N-acetyl-5-methoxytryptamine) is present in grapes. 21 Depending on variety and location, levels of melatonin in grape skin have ranged from 0.005 to 1.2 ng/g. The stage of growth also affects the amount present. Studies have indicated that melatonin may also be present in the flesh and seeds of grapes.
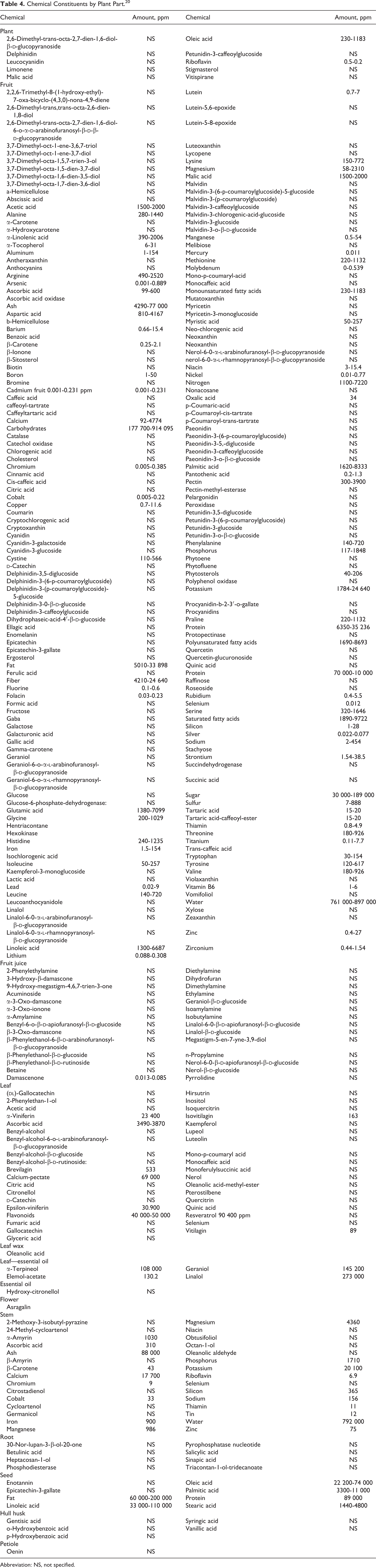
A detailed list of chemical constituents by plant part is presented in Table 4, 20 and a more focused listing of constituents of vitis vinifera is provided in Table 5. 30,33,34 As stated earlier, Table 2 provides the conclusions from CIR safety assessments that exist for some of the constituents of grape, and Table 6 includes information on the toxicity of some constituents. 32,35 –47
Chemical Constituents by Plant Part. 20
Abbreviation: NS, not specified.
Additional Constituent Data.

Toxicity Information on Some Components of Vitis vinifera (Grape).

Abbreviations: DMSO, dimethyl sulfoxide; IARC, International Agency for Research on Cancer; ip, intraperitoneal; LD50, median lethal dose; NOAEL, no-observed adverse effect level; SCE, sister chromatid exchange; UV, ultraviolet; wt, weight.
Vitis vinifera (grape) fruit extract
Fruit acids, sugars, minerals, pectin, tannins, proteins, anthocyanins, waxes, flavonoids, xanthophylls, carotene, vitamins, polysaccharides, aromatic substances, and procyanidins are part of the composition of vitis vinifera (grape) fruit extract. 22
Vitis vinifera (grape) juice
A commercial brand grape juice contained 4.4 mg/L quercetin and 6.2 mg/L myricetin. 48
Vitis vinifera (grape) leaf extract
Potassium and calcium bitartrate, calcium malate, fruit acids, sugar, flavonoids, and tannins are part of the composition of vitis vinifera (grape) leaf extract. 23
Vitis vinifera (grape) seed extract
The main constituents of grape seeds are reported to be phenolic compounds. Those phenolic compounds from standardized grape seed extracts are reported to be 92% to 95% oligomeric proanthocyanidins. 49 Proanthocyanidin structures vary depending upon the source of the flavanol(s) building blocks (monomer units), the degree of oligomerization (how many flavanol repeat units), and the presence of modifications (such as esterification) of the 3-hydroxyl group. 50 The most prominent grape seed extract proanthocyanidin is depicted in Figure 1. 49 Catechin, epicatechin, and taxifolin are the primary flavanols present in grape seeds and comprise the majority of the remaining phenols in grape seed extracts (Figure 2). Heating of oligomeric proanthocyanidins, under acidic conditions, leads to the release of anthocyanins, and in turn, flavanols. Accordingly, the length of oligomeric proanthocyanidins and the concentration of flavanols in grape seed extracts are highly dependent on the extraction techniques used.
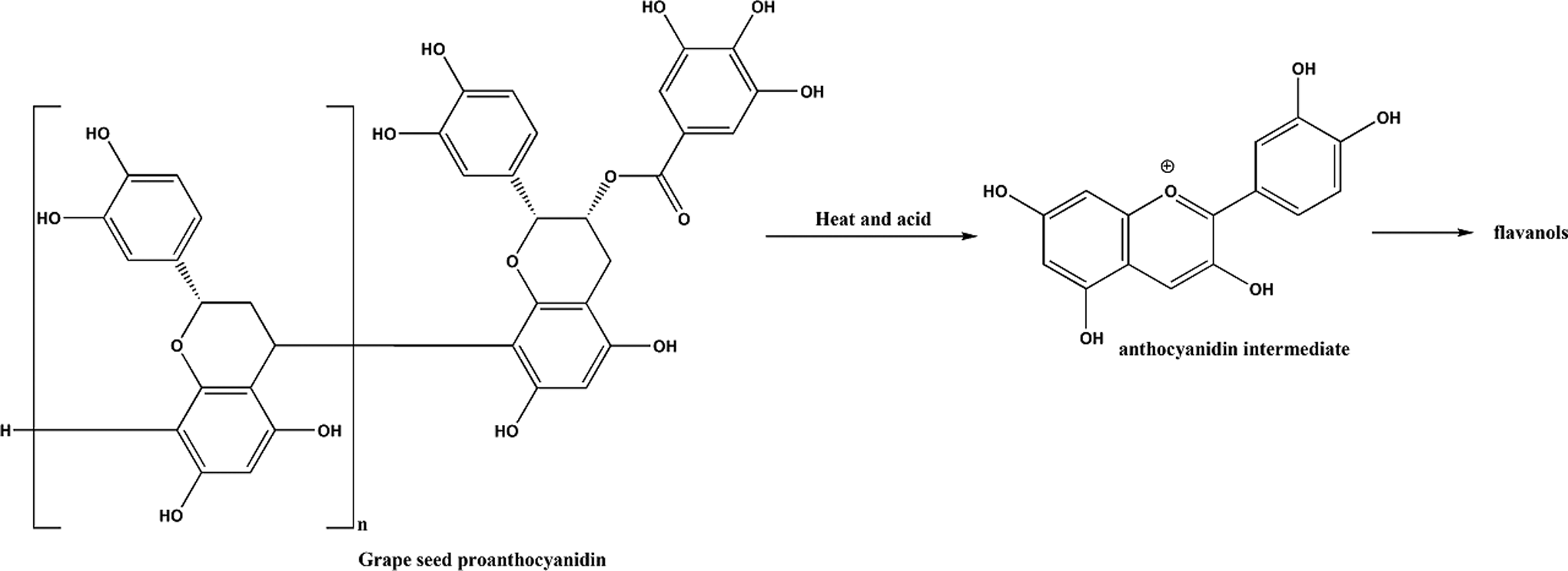
Grape seed acid proanthocyanidin.
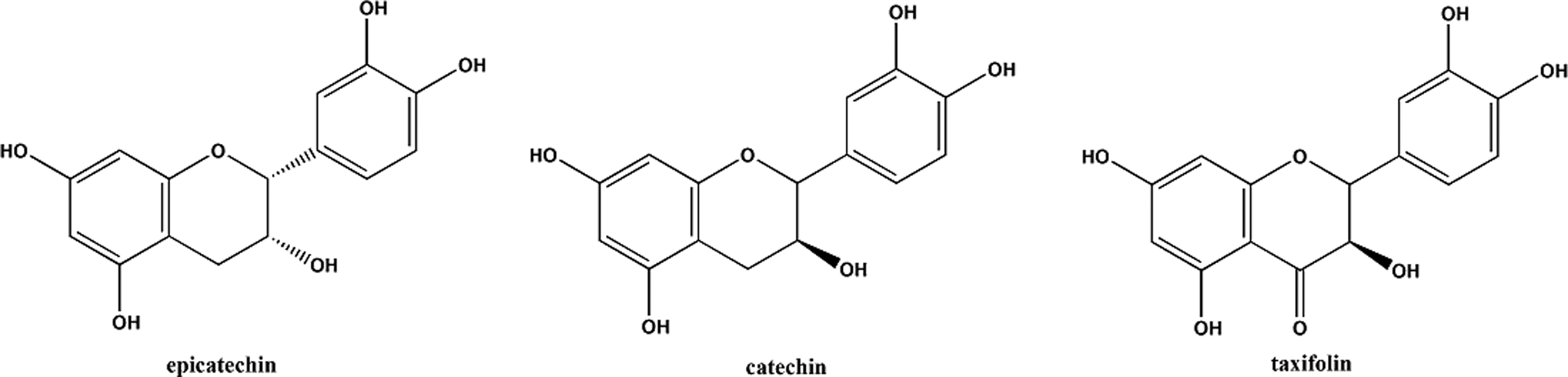
Primary flavanols in grape seeds.
Grape seed oligomeric proanthocyanidins (United States Pharmacopeia [USP] grade for dietary supplements) contain no more than 10 ppm heavy metals, no more than 19.0% catechin and epicatechin on the anhydrous basis, no more than 8.0% water, and no more than 2% water-insoluble fraction. 51
Vitis vinifera (grape) seed extract, as the trade name ActiVin, contains 54% dimeric, 13% trimeric, and 7% tetrameric oligomeric proanthocyanidins and a small amount of catechin derivatives, flavonoids, and other oligomeric proanthocyanidins. 52
Vitis vinifera (grape) skin extract
Grape skin extract (enocianina) is an approved food color additive exempt from batch certification. The FDA describes the color additive as containing the common components of grape juice: anthocyanins, tartaric acid, tannins, sugars, and minerals (21CFR73.170). A small amount of residual sulfur dioxide may be present following aqueous (aq) extraction in the presence of sulfur dioxide. The grape anthocyanins are usually either monoglycerides or diglycosides. 46 The Food Chemicals Codex states the primary color components of grape skin extract are anthocyanins, such as the glucosides of malvidin, peonidin, petunidin, delphinidin, or cyanidin. Food-grade grape skin extract is to contain no more than 1 mg/kg arsenic and no more than 5 mg/kg lead.
Preparation/Extraction
Vitis vinifera (grape) fruit extract
A product information sheet submitted by industry on a mixture that contains vitis vinifera (grape) fruit extract states that the solvent of extraction is glycerin. 22 The resulting composition of the mixture is 75% to 100% glycerin, 50% to 75% vitis vinifera (grape) fruit extract, and 10% to 25% water, and the ratio of extract to botanical is 2:1. Potassium sorbate and sodium benzoate, 0.3% each, are used as preservatives. The extract is filtered clear after preparation.
Vitis vinifera (grape) leaf extract
A product information sheet submitted by industry on a mixture that contains vitis vinifera (grape) leaf extract states that the solvent of extraction for this product is also glycerin. 23 The resulting composition of the mixture is 75% to 100% glycerin, 10% to 25% water, and 5% to 10% vitis vinifera (grape) leaf extract. As mentioned earlier, potassium sorbate and sodium benzoate, 0.3% each, are used as preservatives, and the extract is filtered clear after preparation.
Another source reported the extraction of grape leaves with a propylene glycol solution. 34 The composition of this extract was not provided.
Vitis vinifera (grape) seed extract
One manufacturer reported that vitis vinifera (grape) seed extract is prepared as a concentrated extract by separating the seeds from the fruit, cleaning and comminuting the seeds, extracting with alcohol, and then filtering the extract. 24 The filtrate is concentrated by distillation and then spray-dried. The ratio of fresh plant material to extract is 133:1.
The USP-grade grape seed oligomeric proanthocyanidins (dietary supplement) is a fraction of an extract of ripe vitis vinifera seeds. 51 The extract is prepared using alcohol, methanol, acetone, ethyl acetate, water, or mixtures of these solvents. The extract is then further enriched in oligomeric proanthocyanidins by fractionation with ethyl acetate or by other means.
Vitis vinifera (grape) skin extract
Grape skin extract (enocianina), the FDA-approved color additive, is prepared by the aq extraction (steeping) of the fresh deseeded marc remaining after grapes have been pressed to produce grape juice or wine (21CFR73.170). During the steeping process, sulfur dioxide is added and most of the extracted sugars are fermented to alcohol. The extract is concentrated by vacuum evaporation, during which practically all of the alcohol is removed.
Use
Cosmetic
The vitis vinifera (grape)-derived ingredients included in this safety assessment are reported to have many possible functions in cosmetic formulations. Vitis vinifera (grape) seed extract is reported to function as an anticaries agent, antidandruff agent, antifungal agent, antimicrobial agent, antioxidant, flavoring agent, light stabilizer, oral care agent, oral health care drug, and sunscreen agent. 53 Many of the other vitis vinifera (grape) ingredients are reported to function as skin-conditioning agents, and a few are reported to function as antioxidants. Five ingredients—the seed extract, the fruit powder, the juice, the juice extract, and the skin extract—are reported to function as flavoring agents and 4 of those 5 (all except the seed extract), as well as the skin powder, are reported to function as colorants. The International Cosmetic Ingredient Dictionary and Handbook does not list the functions for vitis vinifera (Grape) and vitis vinifera (grape) leaf wax. A list of all the reported functions for each ingredient is provided (Table 1).
The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2012 indicate that vitis vinifera (grape) seed extract is used in 495 cosmetic formulations, vitis vinifera (grape) fruit extract is used in 238 cosmetic formulations, and vitis vinifera (grape) leaf extract is used in 80 cosmetic formulations. 54 The other in-use vitis vinifera (grape)-derived ingredients are used in less than 15 formulations, and no uses were reported for 11 other vitis vinifera (grape)-derived ingredients.
The vitis vinifera (grape)-derived ingredients are used at relatively low concentrations in cosmetic formulations. Vitis vinifera (grape) leaf extract is included at up to 3% in leave-on formulations (perfumes); vitis vinifera (grape) fruit extract and vitis vinifera (grape) juice are included at up to 2% in rinse-off skin cleansing products and paste masks and mud packs, respectively. 55 All others are used at <1% in formulation. Although no reported uses were received in the VCRP for vitis vinifera (grape) shoot extract, use concentration data were provided in the industry survey. Thus, it should be presumed that vitis vinifera (grape) shoot extract is used in at least 2 cosmetic formulations.
Frequency and concentration of use data categorized by exposure and duration of use are provided in Table 7, and the ingredients for which no uses are reported are listed in Table 8.
Frequency and Concentration of Use According to Duration and Type of Exposure.
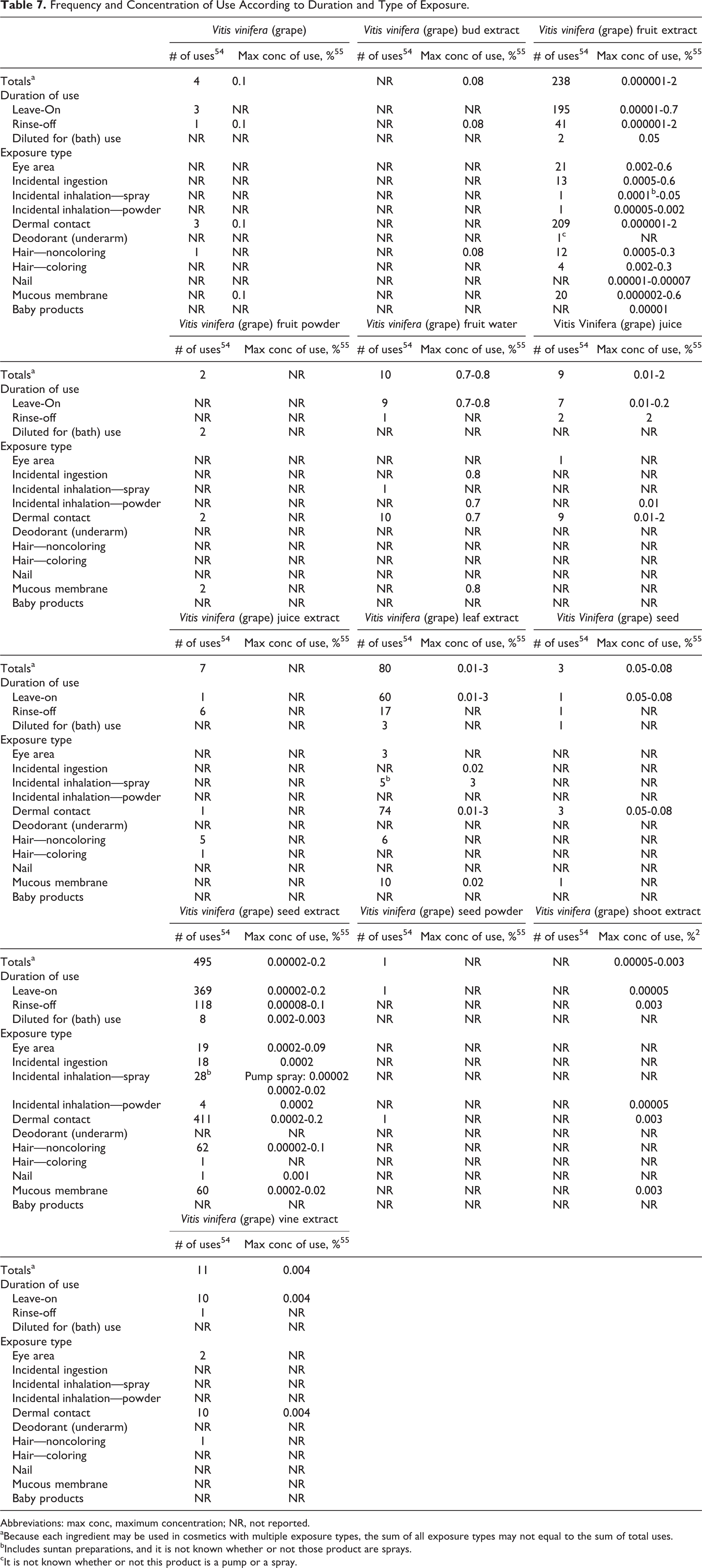




Abbreviations: max conc, maximum concentration; NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal to the sum of total uses.
bIncludes suntan preparations, and it is not known whether or not those product are sprays.
cIt is not known whether or not this product is a pump or a spray.
Ingredient Not Reported to be Used.

Various products containing vitis vinifera (grape)-derived ingredients may be applied to the eye area or mucous membranes or could be incidentally ingested. Additionally, vitis vinifera (grape) fruit extract, vitis vinifera (grape) fruit water, vitis vinifera (grape) juice, vitis vinifera (grape) leaf extract, and vitis vinifera (grape) seed extract are used in cosmetic products that could possibly be inhaled; concentrations of use for ingredients used in products that could be inhaled range from 0.00002% vitis vinifera (grape) seed extract in pump hairsprays to 3% vitis vinifera (grape) leaf extract in perfumes. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 56 –59 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) in any appreciable amount. 56,59
All vitis vinifera (grape)-derived ingredients named in this safety assessment, with the exception of hydrolyzed grape skin, are listed in the European Union inventory of cosmetic ingredients. 60
Noncosmetic
As given in the Code for Federal Regulations (21CFR101, subpart C), grapes are among the 20 most frequently consumed raw fruit and are subject to regulation by the Food and Drug Administration (FDA) as foods.
Vitis vinifera (grape) seed extract
Grape seed extracts are used as nutritional supplements. 49
Vitis vinifera (grape) skin extract
Grape skin extract (enocianina) is a food color additive exempt from batch certification that can be used for coloring only still and carbonated drinks and ades, beverage bases, and, with restrictions, alcoholic bases (21CFR73.170). According to the evaluation of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), the acceptable daily intake (ADI) of grape skin extract is 0 to 2.5 mg/kg bw. 61
Toxicokinetics
It has been reported that most phenolic compounds in grapes are readily metabolized by the gut flora, producing metabolites that potentially can be absorbed into the bloodstream by passive diffusion or active transport systems. 62 A number of factors may play a role in the bioavailability of polyphenols, but maximum plasma values are generally reached between 5 minutes and 2 hours after administration. Oligomeric procyanidins and other higher molecular-weight phenols are not appreciably absorbed, but they can release monomer and dimer units and epicatechin that can be absorbed.
Toxicological Studies
Single Dose (Acute) Toxicity
Dermal
Vitis vinifera (grape) seed extract
The acute dermal toxicity of vitis vinifera (grape) seed extract (trade name ActiVin; a water-ethanol extract) was evaluated in 5 male and 5 female albino rats. 52 A single dose of 2 g/kg moistened with 0.3 mL deionized water was applied to the clipped intact dorsal skin of each animal for 24 hours, and the dose covered approximately 5% to 6% of the total body surface. The test site was covered with a gauze bandage that was secured with tape, and collars were placed on the animals to avoid ingestion. The animals were observed for 14 days. None of the animals died during the study, and there were no test material-related clinical findings, body weight changes, or findings at necropsy. Very slight to slight erythema and desquamation was observed in all animals; these dermal responses subsided in all but 3 animals by day 12. One male rat had edema from days 6 to 9. The dermal median lethal dose (LD50) of vitis vinifera (grape) seed extract in albino rats was >2 g/kg; this dose was also the no-observed effect level (NOEL) for systemic toxicity in this dermal study.
Oral
Vitis vinifera (grape) seed extract
Five male and five female albino rats were given a single dose of 5 g/kg vitis vinifera (grape) seed extract (trade name ActiVin) by gavage. 52 The animals were observed for 14 days. One female died on day 1 of the study. Matting and test material around the mouth, hypoactivity, and ocular discharge were noted for some animals; all animals appeared normal by day 3. The oral LD50 of vitis vinifera (grape) seed extract in albino rats was >5 g/kg.
The acute oral toxicity of a grape seed extract (extracted in water and ethanol) containing 89.3% proanthocyanidins was determined using groups of 5 male and 5 female F344/DuCrj rats. 63 The extract was dissolved in purified water, and the animals were dosed by gavage with 0, 2, or 4 g/kg of the extract at a rate of 10 mL/kg bw. None of the animals died, and the LD50 of the grape seed extract was >4 g/kg.
Vitis vinifera (grape) seed/(grape) skin extract
The acute oral toxicity of a mixed grape seed and grape skin extract (extracted in ethanol) containing 76% total polyphenols was determined in a litmus test using female Wistar rats. 62 Three rats were given a single oral dose by gavage of 5 g/kg in saline at a rate of 10 mL/kg. Three negative control rats were dosed with saline only. There were no signs of toxicity for up to 14 days after dosing, and no gross lesions were observed at necropsy. The LD50 of the mixed grape seed/skin extract was >5 g/kg.
Repeated Dose Toxicity
Dietary repeated dose toxicity studies are presented in Table 9. 52,63 –67
Repeated Dose Toxicity Studies.
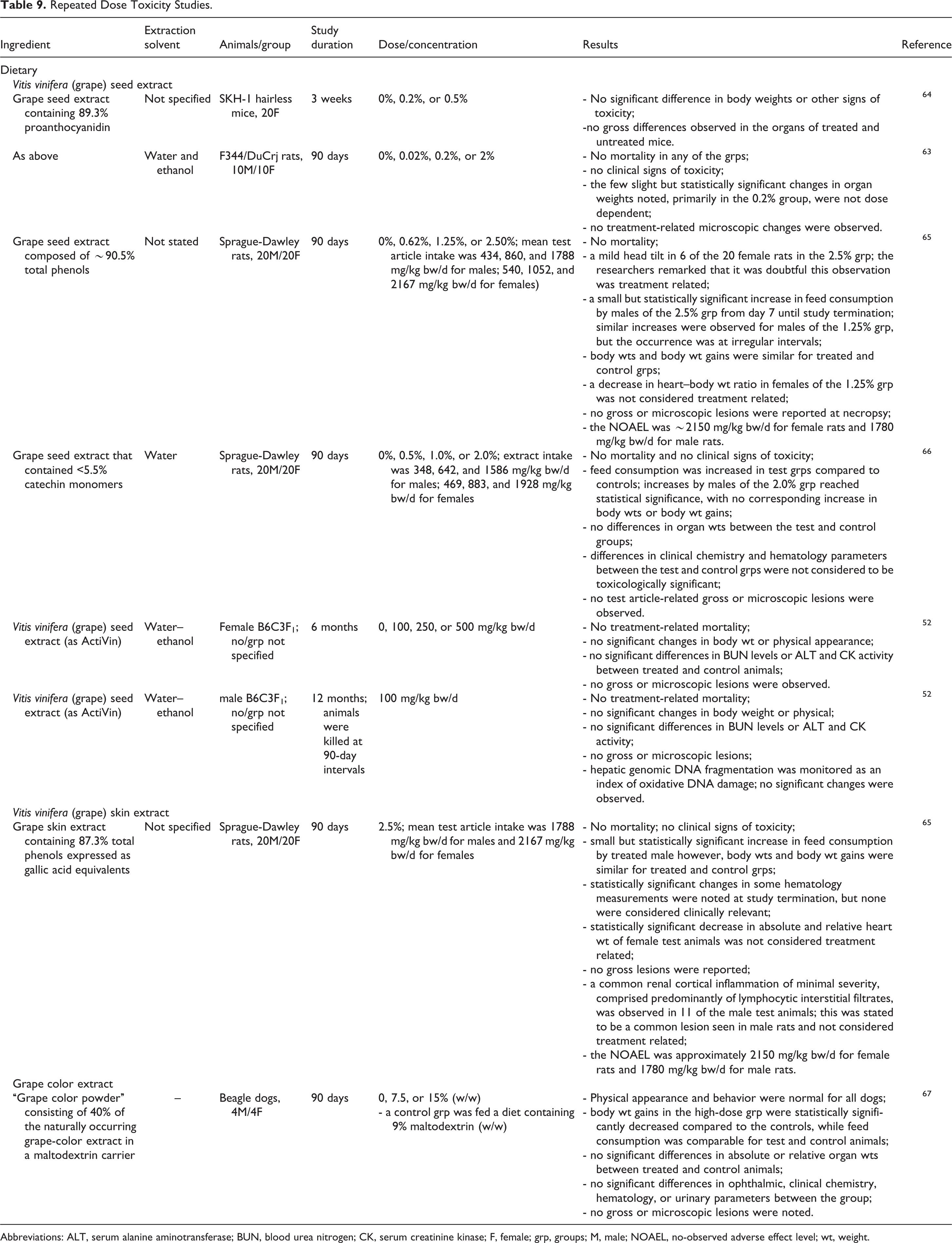
Abbreviations: ALT, serum alanine aminotransferase; BUN, blood urea nitrogen; CK, serum creatinine kinase; F, female; grp, groups; M, male; NOAEL, no-observed adverse effect level; wt, weight.
In a 3-week study in which female SKH-1 hairless mice were fed a diet containing 0%, 0.2%, or 0.5% grape seed extract containing 89.3% proanthocyanidins for 3 weeks, no treatment-related signs of toxicity were reported. 64 In 90-day dietary repeated dose studies in rats, the no-observed adverse effect levels (NOAELs) of grape seed extract and grape skin extract were approximately 2150 and 1780 mg/kg bw/d for male and female rats, respectively. 65 No toxic effects were observed in female B6C3F1 mice after 6 months of dietary administration of up to 500 mg/kg bw/d vitis vinifera (grape) seed extract or in male rats fed 100 mg/kg bw/d vitis vinifera (grape) seed extract for 12 months. 52 Dietary administration of 7.5% or 15% of a grape color extract to Beagle dogs for 90 days resulted in a statistically significant decrease in body weight gains in the high-dose group; however, feed consumption was comparable, leading the researchers to suggest that the decrease in body weight gain was due to the lower calorific value per gram of feed supplemented with grape color extract. No other significant changes were observed. 67
Effect on Ultraviolet-induced skin pigmentation
Vitis vinifera (grape) seed extract
The lightening effect of the oral administration of a grape seed extract (extracted in water and ethanol) containing 89.3% proanthocyanidins on ultraviolet (UV)-induced pigmentation of guinea pig skin was examined. 68 The extract did not contain resveratrol or other phenolic compounds, such as anthocyanidins and flavonols. Using a PEN-RAY lamp (UV containing UVA and UVB, peak at 366 nm), 2 areas on the backs of male and female brownish guinea pigs were irradiated 2×/week for 3 weeks with 0.9 J/cm2 UV. One week after the final UV exposure, groups of 5 irradiated animals were fed a diet containing 1% of the grape seed extract or a standard diet for 8 weeks. The lightening effect was determined every 2 weeks by measuring the L*-value (lightness) and the melanin index at the 2 irradiated sites and an unexposed site. The L*-value was measured with a reflectance spectrophotometer, and the melanin index was calculated using these data. After 8 weeks of dosing, blood samples were taken from each animal, and the animals were then killed. Skin samples were taken from UV-irradiated and a nontreated sites and evaluated for 3,4-dihydroxyphenylalanine (DOPA)-positive melanocytes and markers of oxidative DNA damage.
There were no differences in body weights between the groups. The UV-induced skin pigmentation was reduced in the group fed grape seed extract, as indicated by the increase in L*-value and the decrease in melanin index in UV-induced pigmented skin throughout the study as compared to control values; these differences were not statistically significant. These parameters were similar for both groups in unirradiated skin. The number of DOPA-positive melanocytes in the grape seed extract group was decreased compared to the control group. The number of melanin 8-hydroxy-2′-deoxyguanosine-positive cells, melanin-Ki-67-positive cells, and melanin proliferating cell nuclear antigen-positive cells in irradiated skin also decreased in the grape skin extract group compared to controls; the decrease observed with melanin-Ki-67-positive cells was statistically significant.
Reproductive and Developmental Toxicity
Published reproductive and developmental toxicity data were not found for vitis vinifera (grape)-derived ingredients. A reproduction study on grape color extract is described subsequently. Information on estrogenic activity of some of the constituents of vitis vinifera is provided in Table 6.
Grape Color Extract
A 2-generation reproductive study on grape color extract was performed using Sprague-Dawley rats. 69 The Code of Federal Regulations (21CFR73.169) states that the color additive grape color extract is an aq solution of anthocyanin grape pigments made from Concord grapes (Vitis labrusca) or a dehydrated water soluble powder prepared from the aq solution. The aq solution is prepared by extracting the pigments from precipitated lees produced during the storage of Concord grape juice. It contains the common components of grape juice, namely anthocyanins, tartrates, malates, sugars, and minerals, and so on, but not in the same proportion as found in grape juice. The dehydrated water soluble powder is prepared by spray drying the aq solution containing added maltodextrin. Groups of 25 male and 25 female rats (F0 generation) were fed diets containing 0%, 7.5%, or 15% (w/w) grape color powder or a diet containing 9% by wt maltodextrin for 3 weeks; after 3 weeks, the rats were mated within their respective groups. Female F0 rats, which were allowed to deliver, were fed the test diets throughout mating, gestation, and lactation. Each litter (the F1 generation) was culled to 10 pups (5 males and 5 females if possible) on day 4. On day 21 of lactation, 2 F1 males and 2 F1 females were selected for a subsequent 13-week study followed by a reproduction study. The F0 parents and the remaining offspring were killed.
The selected F1 animals were fed the same dietary levels of grape color extract as their parents. After 13 weeks of dosing, the rats were mated within their respective groups. The F1 rats were also allowed to deliver and were fed the test diets throughout mating, gestation, and lactation. The F2 generation litters were culled as described previously. On day 21 of lactation, all F1 parents and F2 pups were killed.
All animals, except 1 F1 male of the maltodextrin group, survived until scheduled termination. Dietary administration of up to 15% grape color powder had no effect on reproductive parameters or fertility. In the F1 animals fed the test diets for 13 weeks prior to dosing, the group mean body weight gain was statistically significantly decreased in the high-dose females. Body weights of the F1 and F2 pups of both test groups were statistically significantly decreased compared to controls at day 21 of lactation, and, compared to controls, the body weights of F0 pups of the high-dose group were statistically significantly decreased on day 4, while the body weights of F1 pups of both test groups were statistically significantly decreased at birth. The researchers suggested because there were no significant differences in food-conversion data between groups, the decrease in body-weight gain was due to the lower calorific value per gram of food of the diet supplemented with grape color powder compared with the control diet.
Statistically significant differences in several clinical chemistry parameters were observed between groups after 6 weeks of dosing; the values, which were within the normal physiological range for Sprague-Dawley rats, were comparable at the end of 13 weeks of dosing. At necropsy, absolute and relative liver weights were decreased in males and females of both test groups; absolute adrenal gland weights were decreased in males of both test groups and high-dose females; and relative thyroid gland weights were decreased in males of both test groups; the researchers stated that it was unlikely these changes were related to feeding of the test article because there were no corresponding effects on clinical chemistry values or microscopic observations. No microscopic lesions were reported in any of the neonate groups.
Genotoxicity
Genotoxicity testing on grape-derived extracts is summarized in Table 10. 31,62,63,70 –76 In vitro, mixed results were reported in the genotoxicity of vitis vinifera (grape)-derived ingredients, but in vivo, mostly negative results were obtained (Table 10). Fractions of raw grapes demonstrated potent mutagenic activity in an Ames test, 70 and water and ethanol extracts of red and white grapes enhanced mitomycin-C (MMC)-induced sister chromatid exchanges (SCEs) in an SCE assay in human lymphocytes, but there was no effect on SCEs without MMC. 31 Grape juice was also mutagenic in vitro, as demonstrated in the Ames test. 71,72 However, grape seed extract was not mutagenic in vitro in an Ames test or chromosomal aberration assay 63 nor in vivo in the mouse micronucleus test. 63,76 A mouse micronucleus test with grape skin extract was negative. 76 In vitro, grape seed/grape skin extract was weakly mutagenic in an Ames test but not genotoxic in a chromosomal aberration assay, and the mixed extract demonstrated a statistically significant increase in micronuclei after 48 hours, but not after 72 hours. 62 (Table 6 includes information on the genotoxic potential of some of the constituents of vitis vinifera.)
Genotoxicity Studies.
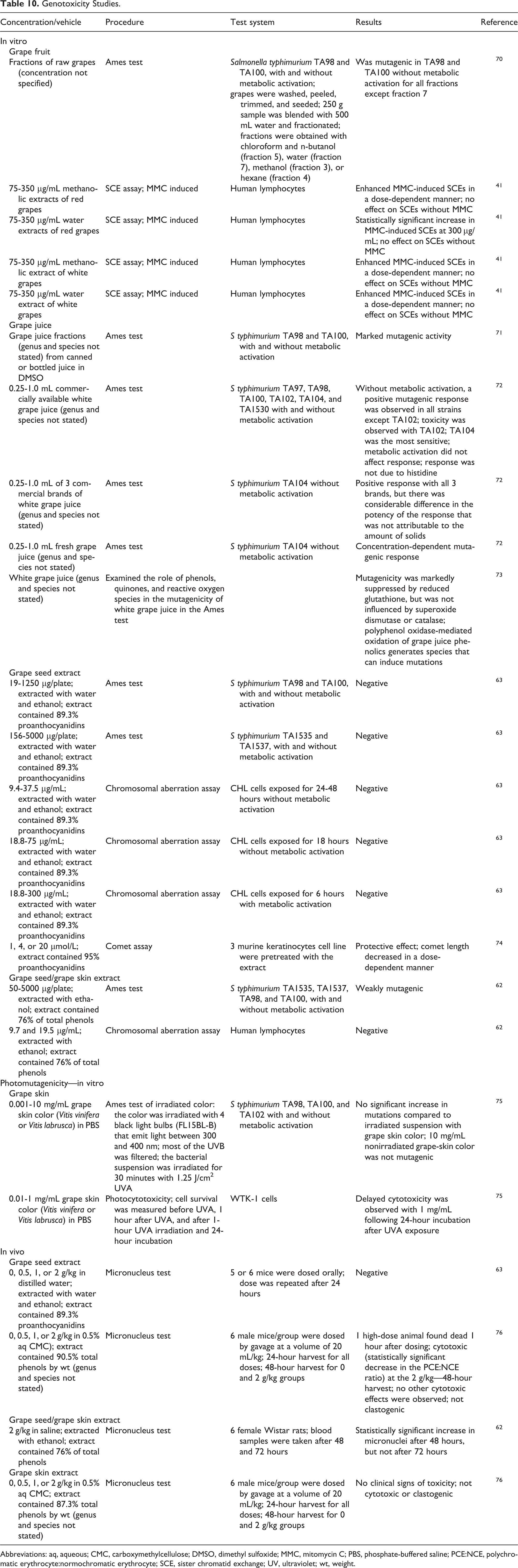
Abbreviations: aq, aqueous; CMC, carboxymethylcellulose; DMSO, dimethyl sulfoxide; MMC, mitomycin C; PBS, phosphate-buffered saline; PCE:NCE, polychromatic erythrocyte:normochromatic erythrocyte; SCE, sister chromatid exchange; UV, ultraviolet; wt, weight.
Carcinogenicity
Oral
Vitis vinifera (grape) seed extract
In a photocarcinogenicity study (described later in this report in Table 10), a group of 20 SKH-1 hairless mice were fed a diet containing 1% grape seed extract that contained 89.3% proanthocyanidins for 30 weeks to determine whether dietary grape seed extract alone had any effect on skin tumor formation. 64 No skin tumors formed.
Tumor Promotion
The effect on tumor promotion by vitis vinifera has been assessed in many studies; some of these studies are summarized in Table 11. 64,74,77 –83 Seed polyphenols and extracts in particular were shown to inhibit 7,12-dimethylbenz[a]anthracene (DMBA)-initiated and 12-O-tetradecanoylphorbol-13-acetate (TPA)-promoted tumors in mouse skin; both dermal application and dietary administration had significant inhibitory activity. 74,79,80,82,84 Dietary grape seed extract also inhibited UV-initiated, UV-promoted, or UV-initiated and promoted skin tumors in hairless mice, 64 and it inhibited the formation of azoxymethane (AOM)-induced aberrant crypt foci (ACF) in the intestines of rats. 83 Some of the studies summarized in Table 11 examined the effect of applying DMBA to mice and then later either treating the animals topically or in the diet with grape seed extract without TPA. 79,81,84 Mice did not develop tumors when dosed dermally or orally with grape seed extract after initiation with DMBA.
Effect on Tumor Promotion.
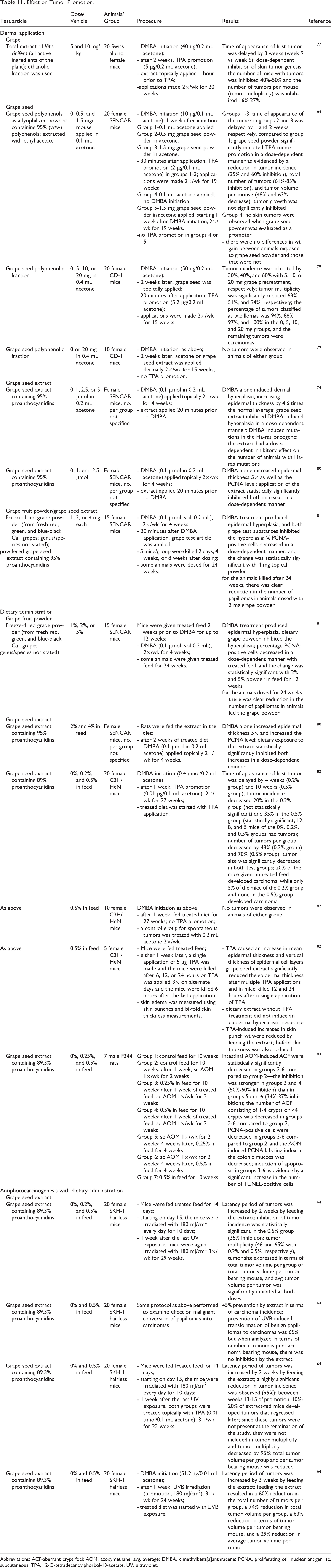
Abbreviations: ACF-aberrant crypt foci; AOM, azoxymethane; avg, average; DMBA, dimethylbenz[a]anthracene; PCNA, proliferating cell nuclear antigen; sc, subcutaneous; TPA, 12-O-tetradecanoylphorbol-13-acetate; UV, ultraviolet.
Irritation and Sensitization
Skin Irritation/Sensitization
Dermal irritation and sensitization data are presented in Table 12. 52,85 –99 In in vitro predictive model testing, a product containing 3% vitis vinifera (grape) fruit extract was a nonirritant in a dermal irritection test in human skin, 85 a product containing 10% vitis vinifera (grape) fruit extract was non-/minimally irritating in an Epiderm MTT viability assay, 86 and hydrolyzed grape skin was nonirritating in an MTT assay. 87 In a single-dose study in NZW rabbits, vitis vinifera (grape) seed extract applied neat was classified as moderately irritating 52 ; in a human 2-week use study, a formulation containing 0.15% vitis vinifera (grape) seed extract was not an irritant. 88 In an in vitro assay of prosensitizing potential, hydrolyzed grape skin did not increase the expression of the investigated markers and did not show any stimulating potential of the immune cellular response mediated by monocytes/macrophages. 89 In clinical testing, products containing up to 10% vitis vinifera (grape) fruit extract, 90 –93 a formulation containing 0.1% vitis vinifera (grape) juice, 94 cosmetic formulations containing 0.5% vitis vinifera (grape) juice extract, and vitis vinifera (grape) seed extract tested at a maximum concentration of 1% in a raw material 95 –99 were not irritants or sensitizers in human repeated insult patch testing (HRIPTs).
Dermal Irritation and Sensitization.
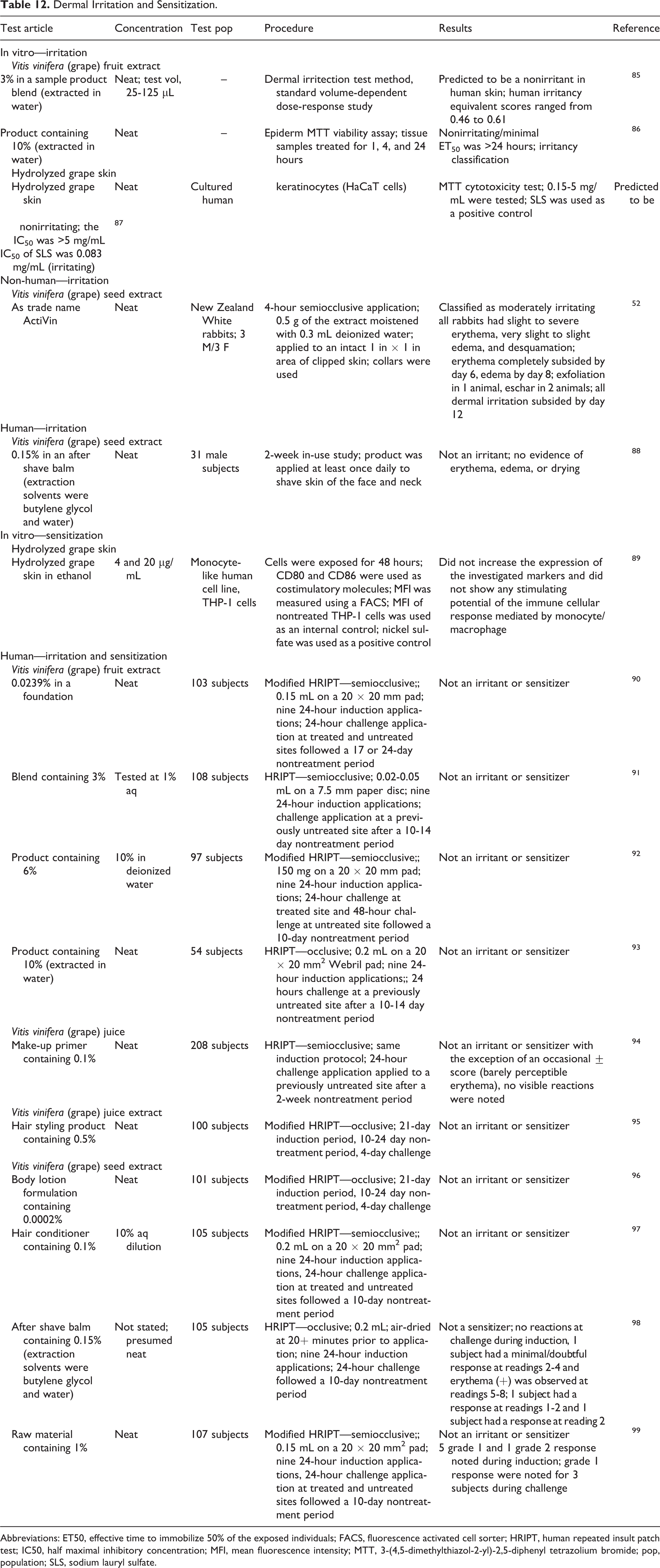
Abbreviations: ET50, effective time to immobilize 50% of the exposed individuals; FACS, fluorescence activated cell sorter; HRIPT, human repeated insult patch test; IC50, half maximal inhibitory concentration; MFI, mean fluorescence intensity; MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide; pop, population; SLS, sodium lauryl sulfate.
Occupational exposure
A skin prick-to-prick test was performed on vineyard workers to assess the prevalence of sensitization to grapes with occupational exposure. 100 Three groups of vineyard workers, 120/group, were tested: harvesters (group A), workers in grape selection (group B), and workers operating destemming/crushing/pressing machines (group C); a group of 120 office employees (group D) was used as a negative control group. For the test, the needle was inserted into a cleaned grape and then inserted into the skin. Normal saline was used as a negative control. Eight harvesters in group A (6.7%) and 5 grape selection workers in group B (4.2%) had positive prick-to-prick tests to grapes; an additional 15 workers in group A and 9 workers in group B had weak positive reactions that were considered negative in this study. None of the workers in the other 2 groups had positive reactions. (Workers in groups A and B had greater exposure to grapes than did workers in groups C or D.) The reported sensitization to grapes was asymptomatic; none of the employees tested had any reported history or symptoms upon exposure.
Case report
A female grape farmer presented with an eczematous dermatitis of the hand. 101 The genus and species of grape were not stated. Patch testing with a crushed bud that had not been exposed to gibberellin (a vegetable hormone she applied to the grapes), an ethanol extract of a bud, a crushed leaf, an ethanol extract of a leaf, and with gibberellin was performed using Finn chambers, as was patch testing with standard allergens and several photoallergens. The only positive reactions were to the crushed and ethanol-extracted bud preparations. Irradiation with 0.7 J/cm2 UVA and 15 mJ/cm2 UVB light increased the erythema and edema. The minimal response dose of UVA was >1.4 J/cm2, and the minimal erythema dose of UVB was 45 mJ/cm2. In similar testing of 22 farmers, a weak positive reaction to the bud and/or leaf was observed in 6 subjects. The reactions did not increase with UV irradiation and subsided within 96 hours.
Ocular Irritation
In vitro
Vitis vinifera (grape) fruit extract
In an EpiOcular assay, a product containing 3% vitis vinifera (grape) fruit extract was predicted to be a minimal ocular irritant. 102 The ocular irritation potential of a single sample of a blend containing 3% vitis vinifera (grape) fruit extract, extracted in water, was evaluated in a standard volume-dependent dose–response study using the ocular irritection test method. The irritection Draize equivalent scores ranged from 4.5 to 6.4/80 for neat samples of the product tested at volumes ranging from 25 to 125 μL.
The irritancy classification for a product containing 10% vitis vinifera (grape) fruit extract was nonirritating/minimal. 103 An EpiOcular MTT viability assay was performed to determine the ocular irritation potential of a product containing 10% vitis vinifera (grape) fruit extract that was extracted with water. The tissue samples were treated with neat test article for 16, 64, and 256 minutes. The effective time to immobilize 50% of the exposed individuals was >256 minutes.
Vitis vinifera (grape) seed extract
A product containing 0.15% vitis vinifera (grape) seed extract was classified as a mild ocular irritant during in vitro testing. 104 A bovine corneal opacity and permeability assay (BCOP) was performed with undiluted samples of an after shave lotion containing 0.15% vitis vinifera (grape) seed extract; the extract was prepared with the extraction solvents butylene glycol and water. Sterile deionized water served as the negative control and ethanol as the positive control. The in vitro score for the test article was 1.0. (Test materials with in vitro scores of 0 to 25 are classified as mild irritants.) The positive control had an in vitro score of 43.2; test materials with in vitro scores of 25.1 to 55 are classified as moderate irritants.
Hydrolyzed grape skin
Hydrolyzed grape skin was predicted to be nonirritating to eyes in a cytotoxicity assay evaluating ocular irritation potential. 105 A neutral red uptake (NRU) assay using fibroblast cultures was performed with 0.15 to 5 mg/mL hydrolyzed grape skin. Sodium lauryl sulfate (SLS) was used as a positive control. The half maximal inhibitory concentration (IC50) value (ie, the concentration of test compound that induces a 50% decrease in cell growth/survival) for hydrolyzed grape skin was >5 mg/mL. The IC50 value for the positive control was 0.063 mg/mL.
Nonhuman
Vitis vinifera (grape) seed extract
The ocular irritation potential of vitis vinifera (grape) seed extract (trade name ActiVin) was evaluated in 6 female NZW rabbits. 52 The test article, 85 mg, was instilled into the conjunctival sac of the right eye, the eyelid was held closed for 1 second, and the eye was not rinsed. The contralateral eye served as an untreated control. The eyes were scored for irritation using the Draize method at 1, 24, 48, and 72 hours and 4, 7, and 14 days after instillation of the test article. Conjunctival irritation was observed in all animals, 4 animals had iridal reactions, and 3 had corneal reactions. The irritation was reversible and completely subsided by day 14. The maximum average score (MAS) at 24 hours for vitis vinifera (grape) seed extract was 16.7/110.
Summary
This report addresses the safety of 24 vitis vinifera (grape)-derived ingredients as used in cosmetics. These ingredients are reported to have many functions in cosmetics, but the most frequently reported function is as a skin-conditioning agent. According to VCRP data obtained from the FDA, vitis vinifera (grape) seed extract is used in 495 cosmetic formulations, vitis vinifera (grape) fruit extract is used in 238 cosmetic formulations, and vitis vinifera (grape) leaf extract is reported to be used in 80 cosmetic formulations; 9 other vitis vinifera-derived ingredients are reported to be in use, and they are used in less than 15 formulations. These ingredients are used at relatively low concentrations in cosmetic formulations. For example, vitis vinifera (grape) leaf extract is included at up to 3% in leave-on formulations (perfumes) and vitis vinifera (grape) fruit extract and vitis vinifera (grape) juice are included at up to 2% in rinse-off skin cleansing products. All others are used at <1% in formulation.
Fruit acids and trans-resveratrol are constituents of vitis vinifera, and polyphenols are found in all parts of the plant. The main constituents of grape seeds are reported to be phenolic compounds, and standardized grape seed extracts are reported to contain 92% to 95% oligomeric proanthocyanidins. Grape skin extract contains anthocyanins, tartaric acid, tannins, sugars, and minerals. The oral LD50 values of grape seed extract and grape skin extract in rats were >4 to 5 and >5 g/kg, respectively, and the dermal LD50 (and NOEL for systemic toxicity) in albino rats was >2 g/kg.
In a 3-week dietary study in which female SKH-1 hairless mice were fed a diet containing 0%, 0.2%, or 0.5% grape seed extract containing 89.3% proanthocyanidins for 3 weeks, no signs of toxicity were reported. In 90-day dietary repeated dose studies in rats, the NOAELs of grape seed extract and grape skin extract were approximately 2150 and 1780 mg/kg bw/d for male and female rats, respectively. No toxic effects were observed in female B6C3F1 mice after 6 months of dietary administration of up to 500 mg/kg bw/d vitis vinifera (grape) seed extract or in male rats fed 100 mg/kg bw/d vitis vinifera (grape) seed extract for 12 months. Dietary administration of 7.5% or 15% of a grape color extract to Beagle dogs for 90 days resulted in a statistically significant decrease in body weight gains in the high-dose group; however, feed consumption was comparable, leading the researchers to suggest that the decrease in body weight gain was due to the lower calorific value per gram of feed supplemented with grape color extract. No other significant changes were observed. Grape seed extract reduced UV-induced skin pigmentation in guinea pigs, but the difference was not statistically significant when compared to controls that did not receive grape skin extract.
A 2-generation reproductive study in which 7.5% or 15% grape color extract was fed in the diet was performed using Sprague-Dawley rats. The only statistically significant effects observed were decreases in the body weights of F1 and F2 pups of both test groups and in body weights of F1 animals fed the test article for 30 days prior to mating; because there were no significant differences in food-conversion data between groups, the researchers suggested the decrease in body weight gain was due to the lower calorific value per gram of food of the diet supplemented with grape color powder compared with the control diet. Liver, adrenal gland, and thyroid gland weights in F1 animals fed the test article for 30 days prior to mating were statistically significantly decreased; these changes were not attributed to the test article because there were no corresponding effects on clinical chemistry values or microscopic observations.
In vitro, mixed results were reported in the genotoxicity of vitis vinifera (grape)-derived ingredients but in vivo, mostly negative results were obtained. Fractions of raw grapes demonstrated potent mutagenic activity in an Ames test, and water and ethanol extracts of red and white grapes enhanced MMC-induced SCEs in an SCE assay in human lymphocytes, but there was no effect on SCEs without MMC. Grape juice was also mutagenic in vitro, as demonstrated in the Ames test. However, grape seed extract was not mutagenic in vitro in an Ames test or chromosomal aberration assay nor in vivo in the mouse micronucleus test. 63,76 A mouse micronucleus test with grape skin extract was negative. In vitro, grape seed/grape skin extract was weakly mutagenic in an Ames test but not genotoxic in chromosomal aberration assay, and the mixed extract demonstrated a statistically significant increase in micronuclei after 48 hours but not after 72 hours.
Vitis vinifera, the seed extract in particular, was shown to inhibit DMBA-initiated and TPA-promoted tumors in mouse skin; both dermal application and dietary administration had significant inhibitory activity. Dietary grape seed extract also inhibited UV-initiated, UV-promoted, or UV-initiated and promoted skin tumors in hairless mice. The formation of AOM-induced ACF in the intestines of rats was also inhibited by dietary grape seed extract. Dietary administration of 1% grape seed extract for 30 weeks did not produce skin tumors in mice, and grape seed extract and grape seed powder were not tumor promoters when applied dermally to mice following initiation with DMBA.
In in vitro predictive model testing, a product containing 3% vitis vinifera (grape) fruit extract was a nonirritant in a dermal irritection test in human skin, a product containing 10% vitis vinifera (grape) fruit extract was non-/minimally irritating in an Epiderm MTT viability assay, and hydrolyzed grape skin was nonirritating in an MTT assay. In a single-dose study in NZW rabbits, vitis vinifera (grape) seed extract applied neat was classified as moderately irritating; in a human 2-week use study, a formulation containing 0.15% vitis vinifera (grape) seed extract was not an irritant. In an in vitro assay of prosensitizing potential, hydrolyzed grape skin did not increase the expression of the investigated markers and did not show any stimulating potential of the immune cellular response mediated by monocytes/macrophages. In clinical testing, products containing up to 10% vitis vinifera (grape) fruit extract, a formulation containing 0.1% vitis vinifera (grape) juice, cosmetic formulations containing 0.5% vitis vinifera (grape) juice extract, and vitis vinifera (grape) seed extract tested at a maximum concentration of 1% in a raw material were not irritant or sensitizers in HRIPTs. Some asymptomatic sensitization reactions were seen in an occupational setting in vineyard workers who had substantial exposure to grapes. One case study was found that reported positive reactions to grape bud preparations.
Products containing 3% and 10% vitis vinifera (grape) fruit extract were predicted to be minimal ocular irritants in in vitro testing. In a study using rabbits, the MAS at 24 hours for vitis vinifera (grape) seed extract was 16.7/110. A product containing 0.15% vitis vinifera (grape) seed extract was classified as a mild ocular irritant during a BCOP assay, and hydrolyzed grape skin was predicting to be nonirritating to eyes in an NRU study.
Discussion
The Panel recognizes that there are data gaps for some of these vitis vinifera-derived ingredients. However, the overall information available on the types of products in which these ingredients are used and at what concentration indicate a pattern of use, which was considered by the Panel in assessing safety. Additionally, the Panel noted that vitis vinifera (grape) seed oil has previously been found safe.
Most of the irritation and sensitization testing performed on the vitis vinifera-derived ingredients included in this report demonstrated that these ingredients are not dermal irritants or sensitizers, with the exception of one 4-hours semiocclusive study of vitis vinifera (grape) seed extract that reported moderate irritation using rabbits when the test substance was applied neat. Additionally, in clinical testing with vitis vinifera (grape) seed extract at a maximum concentration of 1% in a raw material was not an irritant or sensitizer; the grape seed extract is reported to be used at a maximum leave-on concentration of 0.2%. Also, because all the other irritation and sensitization tests were negative, including a human study using up to 10% vitis vinifera (grape) fruit extract in a product, the Panel was of the opinion that the 1 study was an outlier and that the weight of evidence supports the view that these ingredients are not irritants or sensitizers.
The Panel discussed the findings of mutagenic activity of grape and grape juice in some of the bacterial mutagenicity tests. The Panel is aware that there is a history of positive Ames tests with some foods, including grape. Although positive results for mutagenicity occur in bacterial assays, based on the expertise of the Panel and information provided by the European Organization of Cosmetic Ingredients Industries and Services (UNITIS), the constituents of foods in grapes, for example, flavonoids, do not appear to be genotoxic to mammals in vivo. Additionally, vitis vinifera-derived extracts have demonstrated an inhibition of tumor promotion. Therefore, the mutagenic effects in bacterial systems were not considered relevant to the safety of these cosmetic ingredients.
The vitis vinifera plant parts contain some constituents, such as ascorbic acid, biotin, and malic acid, which are cosmetic ingredients for which separate Panel safety assessments are available. Others constituents are compounds that have been discussed in previous CIR assessments. For example, vitis vinifera, and therefore derived extracts, contains a variety of phytochemicals. The Panel has discussed in previous safety assessments that although some of the phytochemicals present in grapes could exert significant biological effects, the low levels in conjunction with the currently reported exposure routes and low use concentrations preclude significant effects. Also, although no dermal absorption data were available for constituents of vitis vinifera, extensive data are available showing that these phytosterol constituents at the potential exposure levels are not estrogenic, are not reproductive toxicants, are not genotoxic, and are not carcinogenic. The Panel also noted that 1 particular constituent that could be of concern, that is, quercetin, can be present at low levels in some components of vitis vinifera. However, again, because the vitis vinifera-derived ingredients are used at very low concentrations in cosmetics, and because the concentrations of quercetin in the plant parts are low, the presence of quercetin was below the level of toxicological concern.
The leaf extract, which is used at up to 3% in perfumes, is a highly colored component, and the Panel discussed the possibility that the leaf extract could be photoactive. The dermatologists on the Panel remarked that phototoxicity issues have not been reported in vineyard workers, and the Panel relied on this clinical experience to alleviate the concern of possible phototoxic effects of vitis vinifera (grape) leaf extract.
The Panel discussed the issue of incidental inhalation exposure to vitis vinifera (grape)-derived ingredients from products that may be aerosolized. There were no inhalation toxicity data available. Vitis vinifera (grape) seed extract is reportedly used at a concentration of 0.00002% in pump hairsprays and vitis vinifera (grape) leaf extract is reportedly used at a concentration of 3% in perfumes. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered the data available to characterize the potential for vitis vinifera (grape)-derived ingredients to cause systemic toxicity, irritation, sensitization, or other effects. They noted that vitis vinifera (grape)-derived ingredients did not produce systemic toxicity in oral single-dose or long-term (up to 12 months) repeated dose studies; grape color extract was not a reproductive or developmental toxicant; vitis vinifera (the seed extract in particular) inhibits the promotion of tumors; and the vitis vinifera (grape)-derived ingredients do not appear to be irritants or sensitizers. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products are available at http://www.cir-safety.org/cir-findings.
Finally, the Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
Conclusion
The CIR Expert Panel concluded the vitis vinifera (grape)-derived ingredients listed subsequently are safe in the present practices of use and concentration in cosmetics.
vitis vinifera (grape);
vitis vinifera (grape) bud extract;
vitis vinifera (grape) flower extract;*
vitis vinifera (grape) fruit extract;
vitis vinifera (grape) fruit powder;
vitis vinifera (grape) fruit water;
vitis vinifera (grape) juice;
vitis vinifera (grape) juice extract;
vitis vinifera (grape) leaf extract;
vitis vinifera (grape) leaf oil;*
vitis vinifera (grape) leaf/seed/skin extract;*
vitis vinifera (grape) leaf water;*
vitis vinifera (grape) leaf wax;*
vitis vinifera (grape) root extract;*
vitis vinifera (grape) seed;
vitis vinifera (grape) seed extract;
vitis vinifera (grape) seed powder;
vitis vinifera (grape) shoot extract;
vitis vinifera (grape) skin extract;*
vitis vinifera (grape) skin powder;*
vitis vinifera (grape) vine extract;
vitis vinifera (grape) vine sap;* Hydrolyzed grape fruit;* Hydrolyzed grape skin.*
Were ingredients in this group not in current use (as indicated by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Contribution
Fiume contributed to conception, design acquisition, analysis, and interpretation, and drafted the article. L. Gill contributed to conception, design, analysis, and interpretation, critically revised the article, and gave final approval. W. Bergfeld, D. Belsito, R. Hill, C. Klaasen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception, design acquisition, analysis, and interpretation, critically revised the article, and gave final approval.
Authors’ Note
Unpublished sources cited in this report are available from the Lillian J. Gill, Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.