Abstract
Sperm analysis is one of the end points in reproductive toxicology studies. Different methods for quantitative sperm analysis have been described. For qualitative morphological sperm analysis, either such techniques or smears of sperm and histological sperm staging are in use. Any of these methods provides morphological results on a light microscopy level. Laser scanning microscopy is a technique using a focused laser for scanning an object. The Olympus 3D Laser Scanning Microscope LEXT OLS4000 with optional possibilities of differential interference contrast provides a microscopic method for visualizing microasperities, which are far beyond the resolving power of a typical light or laser microscope. This technique was applied to sperm of mice, rats, rabbits, and cynomolgus monkeys at magnifications up to ×17 090. The obtained images are comparable to those of a scanning electron microscope under relatively low-power magnifications. Measurements on sperm parameters were taken by an integrated image analysis software tool. Abnormalities were easily detectable.
Keywords
Introduction
Different methods for quantitative sperm analysis (total count of motile [live] and nonmotile [dead] sperm, motility percentage, motility grade profile, pH, white blood cell count, agglutination and if necessary, vitality, and fructose count) have been described. Those include computer-assisted sperm analysis, 1 –4 the less expensive sperm quality analyzer which measures sperm count and motility by the optical method, 5,6 and biochemical method (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide method) which measures color changes in the tetrazolium reaction to mitochondrial reductase by absorption spectrometry reflecting the overall numerical and functional power of sperm activity. 7 Further attempts were published measuring toxicity to the sperm. 8
For qualitative morphological sperm analysis, either the above-mentioned techniques or simply smears and histological evaluation of sperm by staging are in use. Any of these methods provides morphological results on a light microscopy level. For mechanistic considerations, however, ultrastructural evaluation may be more useful. In such case, scanning or electron microscopy is applied. Therefore, an attempt was made to use laser scanning microscopy (LSM) for the evaluation of sperm. The images obtained, similar to scanning electron micrographs and the related image analysis capabilities, with highest measuring accuracy, are deemed to be useful tools for the interpretation of induced sperm injury. The measurements on sperm parameters were taken by an integrated image analysis software tool. Abnormalities were easily detectable.
Sperm Sampling
Sperm was sampled from different laboratory animal species. At BSL BIOSERVICE Laboratories GmbH, Planegg, Germany, sperm was sampled from the vas deferens of 5 control Crl: WI(Han) rats and 5 control BALB/c mice (age approximately 10-12 weeks).
In addition, at BSL BIOSERVICE Laboratories GmbH, Planegg, Germany, sperm samples were collected by means of an artificial vagina (AV; COLLAP system with temperature maintenance, IMV Technologies, L’Aigle, France) from 5 naive New Zealand rabbits. Similar techniques have been described elsewhere. 9 –11 The AV was assembled by inserting a cone and a fitting collection tube and adjusting the temperature to 50°C to 55°C. To increase the animals’ libido, a teaser doe was presented to the bucks. The AV was held tight in the collector’s hand with a preserved fur from a skinned female rabbit covering his forearm, which was laid flat onto the ground of the buck’s cage to allow the buck to mount.
At Vivotecnia Research S.L., Madrid, Spain, sperm samples were collected from from 3 naive cynomolgus monkeys (Macaca fascicularis) that were fasted overnight but allowed access to water ad libitum by electroejaculation using an electrostimulator (PTE Model 304) coupled to a rectal probe (0.7 cm diameter) as described elsewhere. 12 –14
The sperm, if not sampled as ejaculate from living animal, was sampled on necropsy from the vas deferens by gently squeezing out the sperm of the vas deferens using blunt forceps, if necessary. With respect to animal welfare, all studies were performed in facilities accredited by AAALAC (BSL Bioservice Laboratories GmbH) or FELASA (Vivotecnia Research S.L.).
Smear Preparation
Sperm samples were placed into 2.5% glutaraldehyde solution (in 0.1 mol/L sodium cacodylate buffer) for 24 hours prior to evaluation. After delivery, the samples were rinsed with distilled water followed by 5 minutes staining with nondiluted modified May-Grünwald (Merck; 1:1), 0.5% eosin in water, or in 1% toluidine blue solution, VWR International GmbH, Switzerland. Thereafter, the samples were 3× rinsed with distilled water. Between each rinsing, they were carefully centrifuged at 300g for 1 minute. Smears were prepared and air dried for 1 hour at room temperature. The smears were not coverslipped but underwent immediate evaluation by the Olympus 3D Laser Scanning Microscope LEXT OLS4000 (Olympus Schweiz AG, Switzerland).
The best results for light and laser scanning microscopy were obtained using 1% toluidine blue solution in a vial with a glutaraldehyde-fixed sperm pellet washed by distilled water at a dilution of 1:1 for 5 minutes followed by 3× rinsing with distilled water. A droplet of the diluted semen was placed on a glass slide and the slide was slowly air dried. Under light microscopy, sperm structures were easily recognized. Those structures were easily visible, especially in species with a distinct droplet (ejaculate sperm) on the tail (mouse, rat). The same procedures were applied to sperm pellets with modified May-Grünwald (Merck; 1:1) that stained dark and many crystals remained on dried smears or with 0.5% eosin solution. The latter caused shadows by fluorescence seen in light and laser microscopy, and droplets were not clearly visible except when investigating under an ocular ×100 and oil immersion.
Measured Parameters
Measurements were taken from normal sperm only: 120 sperms from cynomolgus monkeys and 100 sperms each from rabbits, rats, and mice. The measured parameters included the lengths, widths, circumference, area of the head, and the lengths and widths of the tail which are taken from rabbit, monkey, and mice sperms. In addition, the distance from the tail insertion at the head until the mid of the cytoplasmic droplet was also measured in mice and rats. In rats, the measured parameters included the head lengths from tip of anterior acrosomal segment up to the distal end of postacrosomal segment, the widths at the equatorial segment of acrosome, the circumference and area of the head, the tail lengths and widths as well as the distance of the tail insertion at the head until mid of the cytoplasmic droplet. Sperm dimensions were measured applying the Olympus LEXT software (provided by Olympus and included within the microscope system) to original images obtained at a magnification of ×2136, whereby the pictures were afterward enlarged by an additional software supplying a 400% zoomed image for detailed parameter measurements. The values were entered and calculated within an EXCEL spreadsheet. Descriptive statistical parameters included mean, standard deviations, minimum, and maximum of all measured parameters.
The results obtained by measurements of sperm dimensions by LSM were within the range of previously published data that were obtained, for example, by calibrated projection microscope. 15
Evaluation of Cynomolgus Sperm by LSM
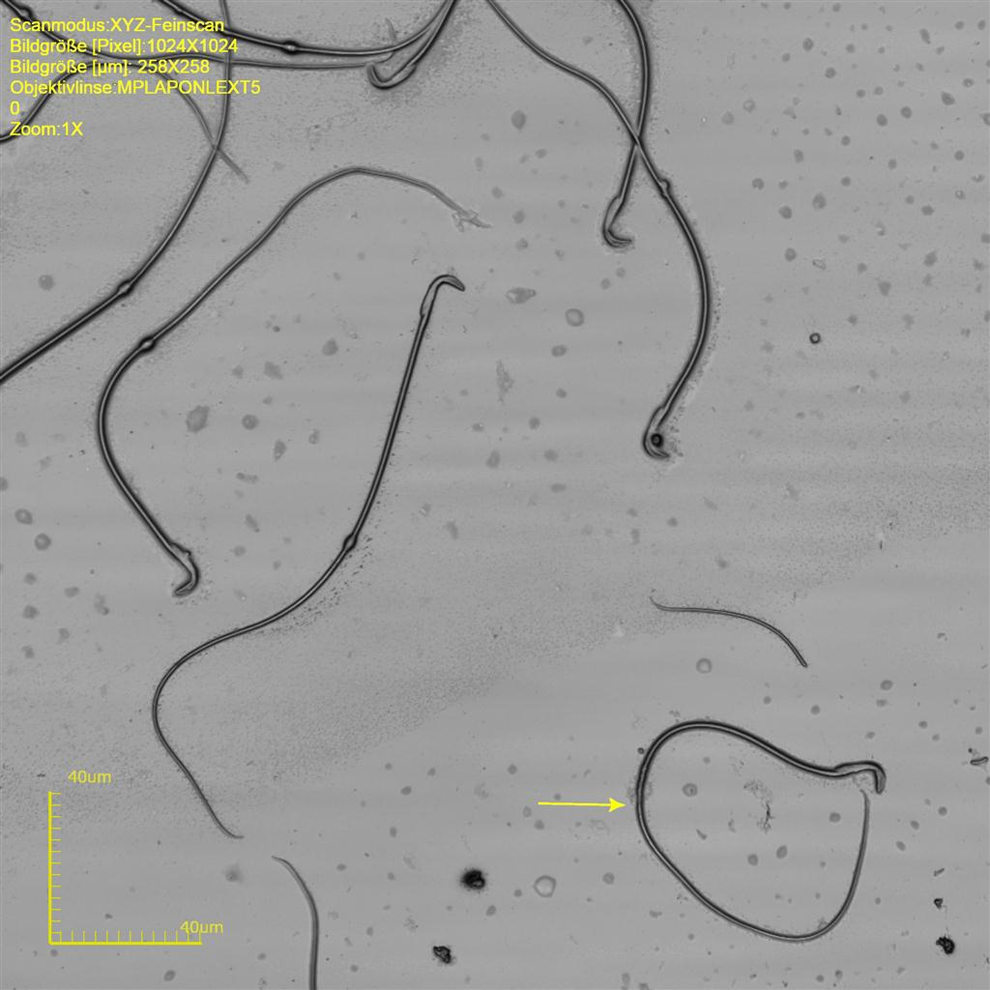
The sperm head of cynomolgus monkeys is a flat oval structure (ovoid shape) at an approximate width of 3.2 to 4.3 µm and an approximate length of 4.6 to 6.0 µm (approximate circumference 14.2-18.3 µm and an area of 13.8-20.4 µm2) that forms an angle to the neck of approximate 120°. The tail with an approximate length of 71.0 to 76.3 µm and a width at the midst of 0.5 to 0.8 µm makes around 93.0% of the total sperm length (Table 1 and Figure 1).
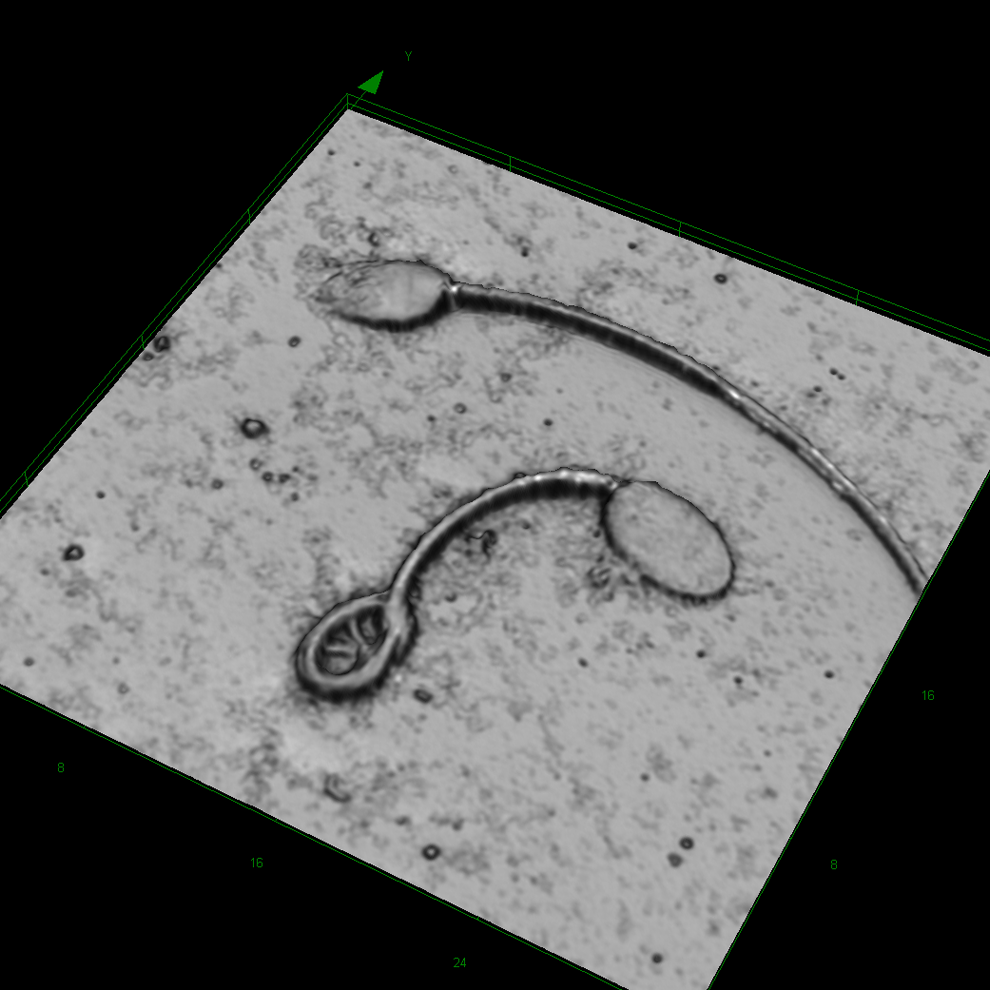
Cynomolgus sperm at original magnification ×8545. Upper sperm cell is normal. Lower sperm cell with coiled tail.
Descriptive Statistics on Measured Dimensions From Cynomolgus Sperm.a
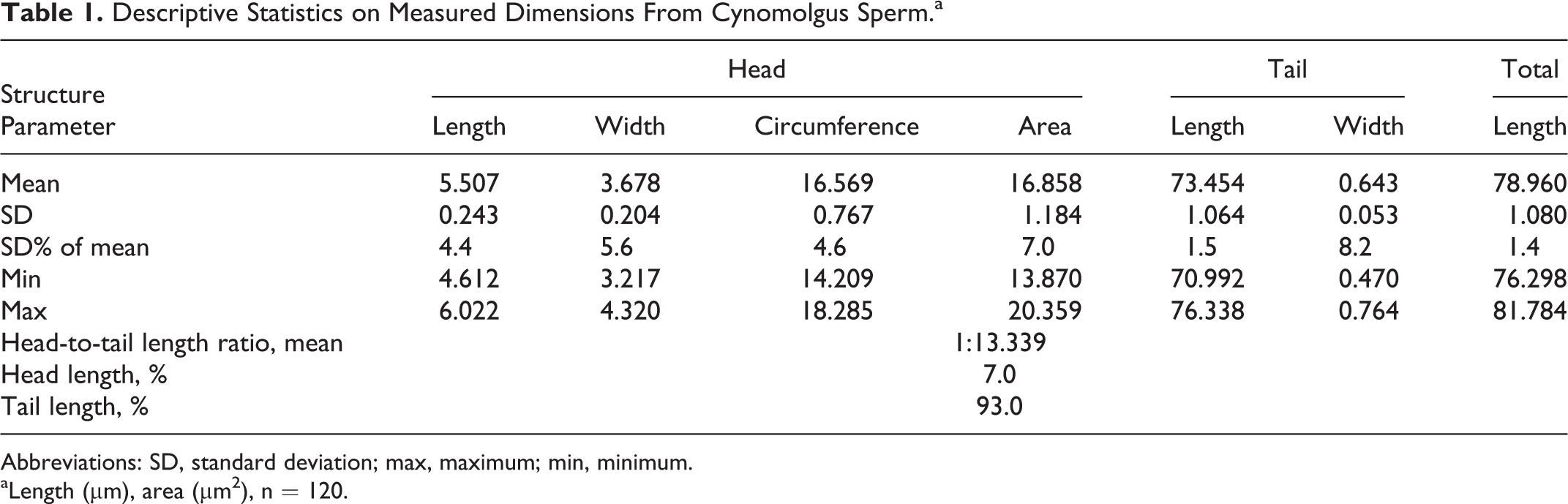
Abbreviations: SD, standard deviation; max, maximum; min, minimum.
aLength (µm), area (µm2), n = 120.
In the present samples, collected by electroejaculation, more than 90% of the cynomolgus sperm in an ejaculate did not bear a cytoplasmic droplet. In contrast, it is published that during development, sperm collected from the epididymal caput have the cytoplasmic droplet at the neck region, whereby the droplet is predominant in over 97% of the sperm. In more distal regions of the epididymides, the droplet is observed more at the midpiece. However, in the epididymal cauda, only approximately 30% of the sperm cells bear a droplet between the distal midpiece and the proximal region of the flagellum. 16
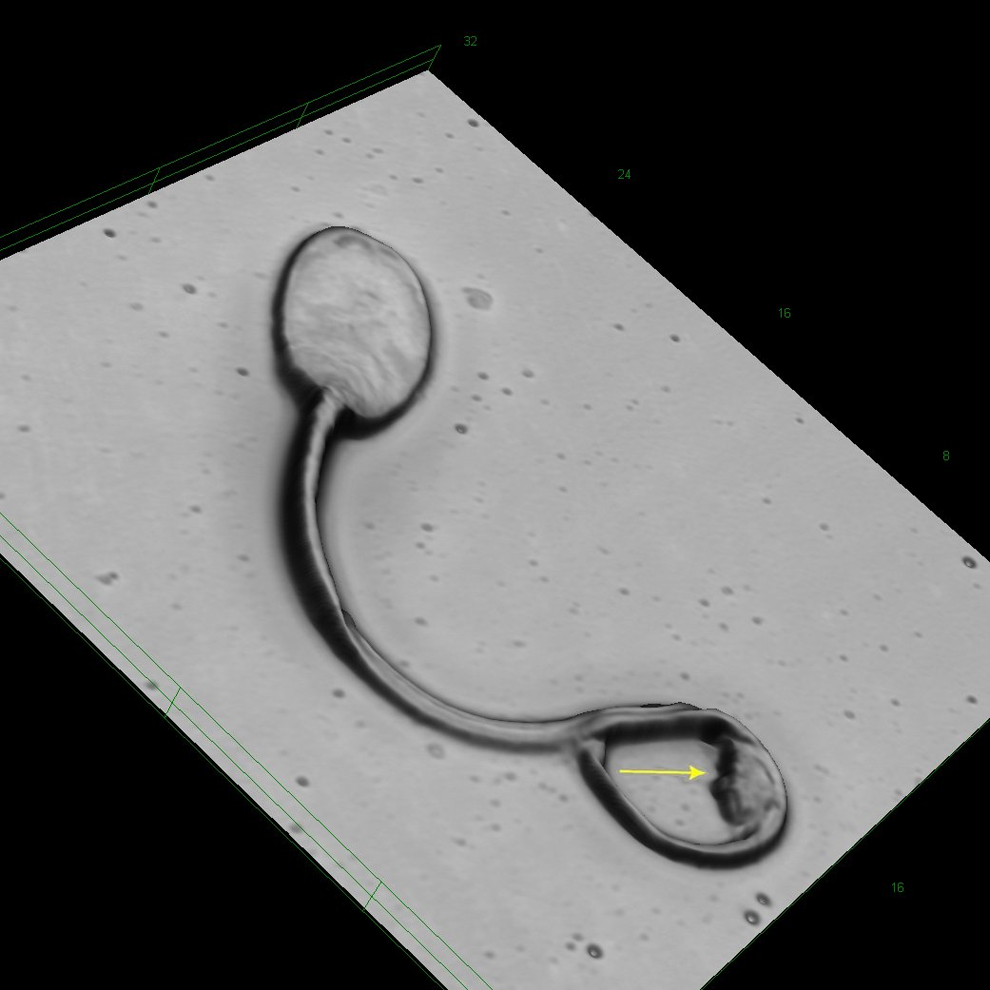
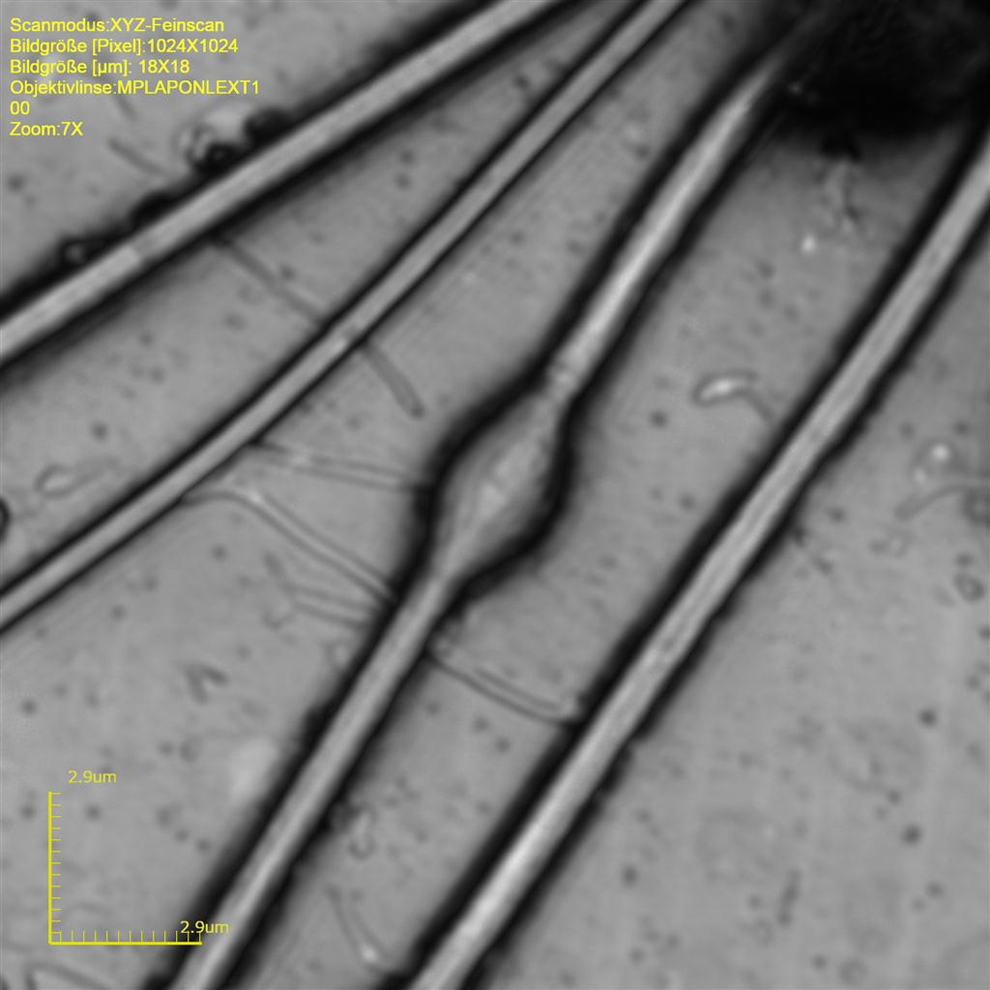
In most cases, droplet bearing sperm was abnormal, that is, such sperm showed kinks at the middle or end position of the midpiece (Figures 2 and 3). Occasionally, there was a kink at the middle part of the midpiece that was not related to a cytoplasmic droplet. In such cases, however, there was a structure similar to a plasmatic remnant, likely equivalent to the cytoplasmic droplet (Figures 5). Coiled tails were a common finding in the cynomolgus sperm (Figures 1, 3 and 4). The coiled tails were noted in up to 40% of the control sperm samples on eosin-stained smears. However, using LSM, there were only up to 5% real coiled tails. Similar to the kink associated with cytoplasmic droplets, the real coiled tails were associated with a structure that presumably represents cytoplasmic remnants (Figure 1). Other coiled tails were due to placement of the tail in coils that was reproduciblely changing the pH value during fixation.
Cynomolgus sperm at original magnification ×17 090 (Image Slope). Kink at the end of midpiece associated with cytoplasmic droplet.
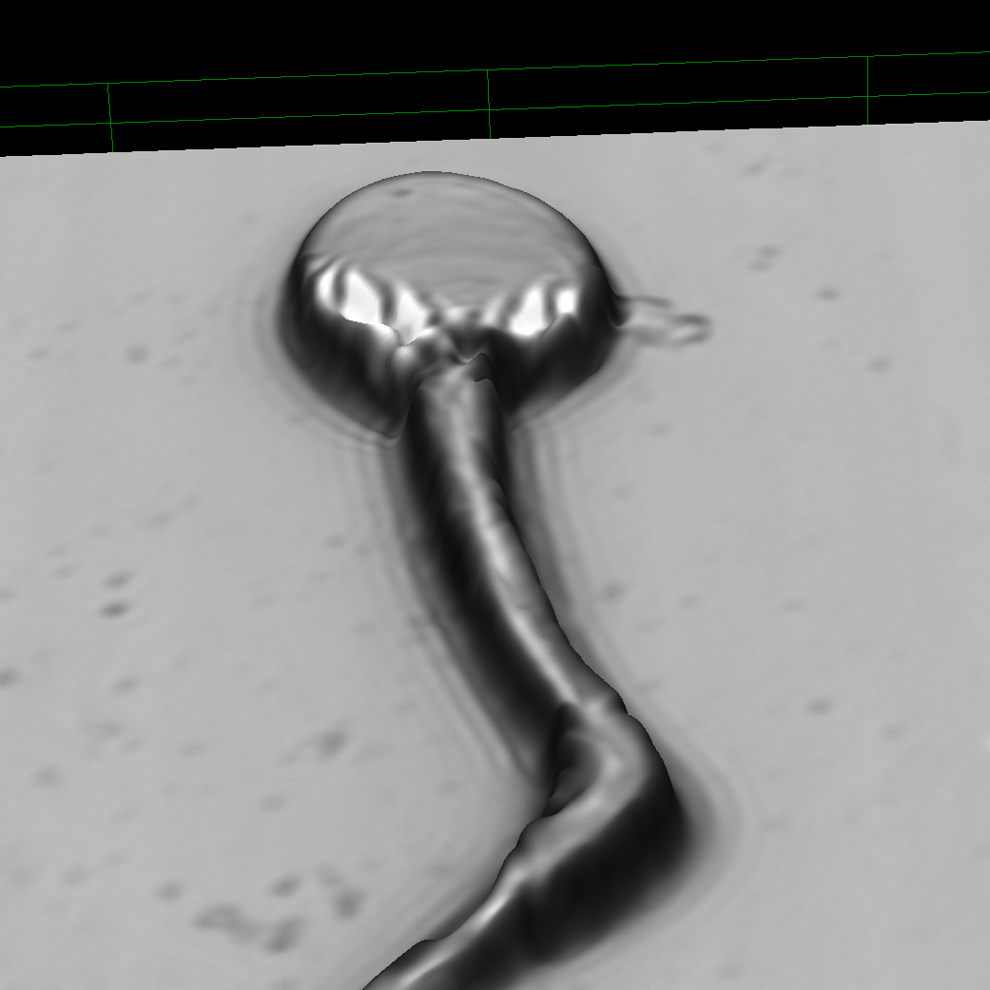
Cynomolgus sperm at original magnification ×14 954. Kink at the end of midpiece associated with cytoplasmic droplet.

Cynomolgus sperm at original magnification ×4273. Kink and coiled tail.
Cynomolgus sperm at original magnification ×17 090. Kink at midpiece with a structure that may be considered a cytoplasmic remnant similar to droplets (arrow).
The term “cytoplasmic droplet” is proposed to describe normal droplets associated with functional sperm produced by normal spermatogenesis and “excess residual cytoplasm” that is suggested to describe abnormal droplets associated with dysfunctional sperm that are products of faulty spermatogenesis. 17
Evaluation of Rabbit Sperm by LSM
The rabbit sperm head is similar in shape to cynomolgus sperm but is of spatulate shape, that is, the dorsal surface is slightly concave. However, the head is larger and more variable in rabbits, that is, there is an approximate width of 4.1 to 5.9 µm and an approximate length of 7.4 to 10.0 µm (approximate circumference 21.9-29.7 µm and an area of 24.2-46.6 µm2) that also forms an angle to the neck of approximate 120°. The tail with an approximate length of 45.5 to 49.0 µm and a width at the midst of 0.5 to 1.0 µm makes around 84.9% of the total sperm length (Table 2 and Figure 6).
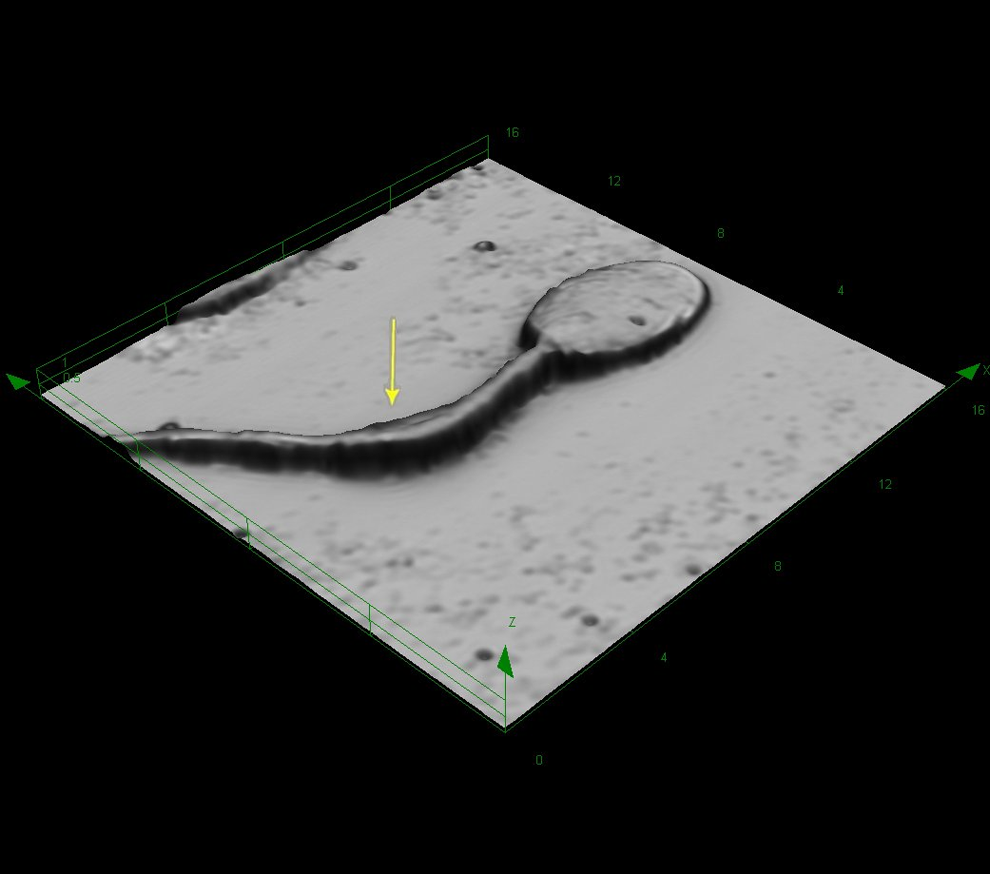
Rabbit sperm at original magnification ×8545. Coiled tail associated with a structure that may be considered a cytoplasmic remnant (arrow).
Descriptive Statistics on Measured Dimensions From Rabbit Sperm.a
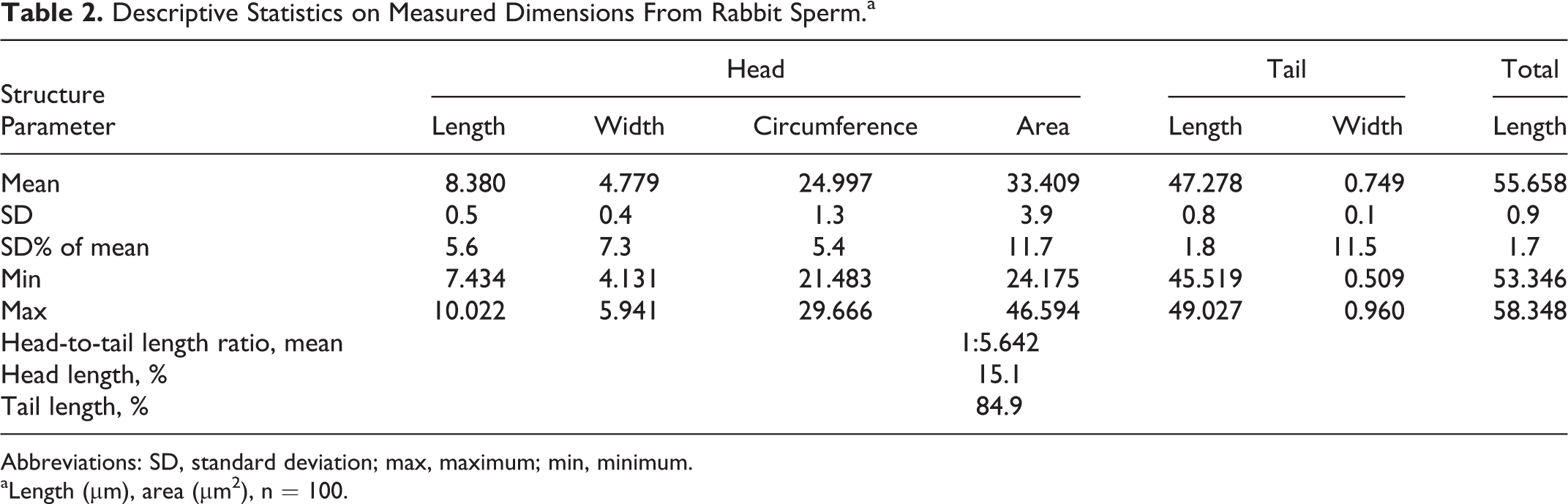
Abbreviations: SD, standard deviation; max, maximum; min, minimum.
aLength (µm), area (µm2), n = 100.
In rabbits, a cytoplasmic droplet rarely recorded in ejaculated sperm. It is known that the cytoplasmic droplet is visible on sperms sampled from the epididymides that is considered an indicator for maturation. 18 Coiled tails were rare findings, but similar to the finding observed in monkey, the coiled tails were associated with a structure that presumably represents cytoplasmic remnants (Figure 6). Data on the incidence of head, midpiece, and tail abnormalities, as well as of multiple defects in a single spermatozoa, and the position of the cytoplasmic droplet along the sperm midpiece were evaluated in samples from the testis, different regions of the epididymis, and the vas deferens are published elsewhere. 18,19 The incidence of spermatozoa with an abnormal midpiece and those with multiple defects decreased significantly passing from the testis to the proximal caput of the epididymis indicating a mechanism for the disposal of defective spermatozoa.
Evaluation of Rat Sperm by LSM
The rat sperm head differs from the monkey sperm head (ovoid shape) and rabbit sperm (spatulate shape). The rat acrosome is of a falciform type that is characterized by an angular shape. 20 The head can be divided into 3 different parts, the anterior acrosomal segment followed by the equatorial segment and the postacrosomal segment (Figure 7). The morphologically different regions are related to functional cytoskeletal changes prior to fertilization. 21
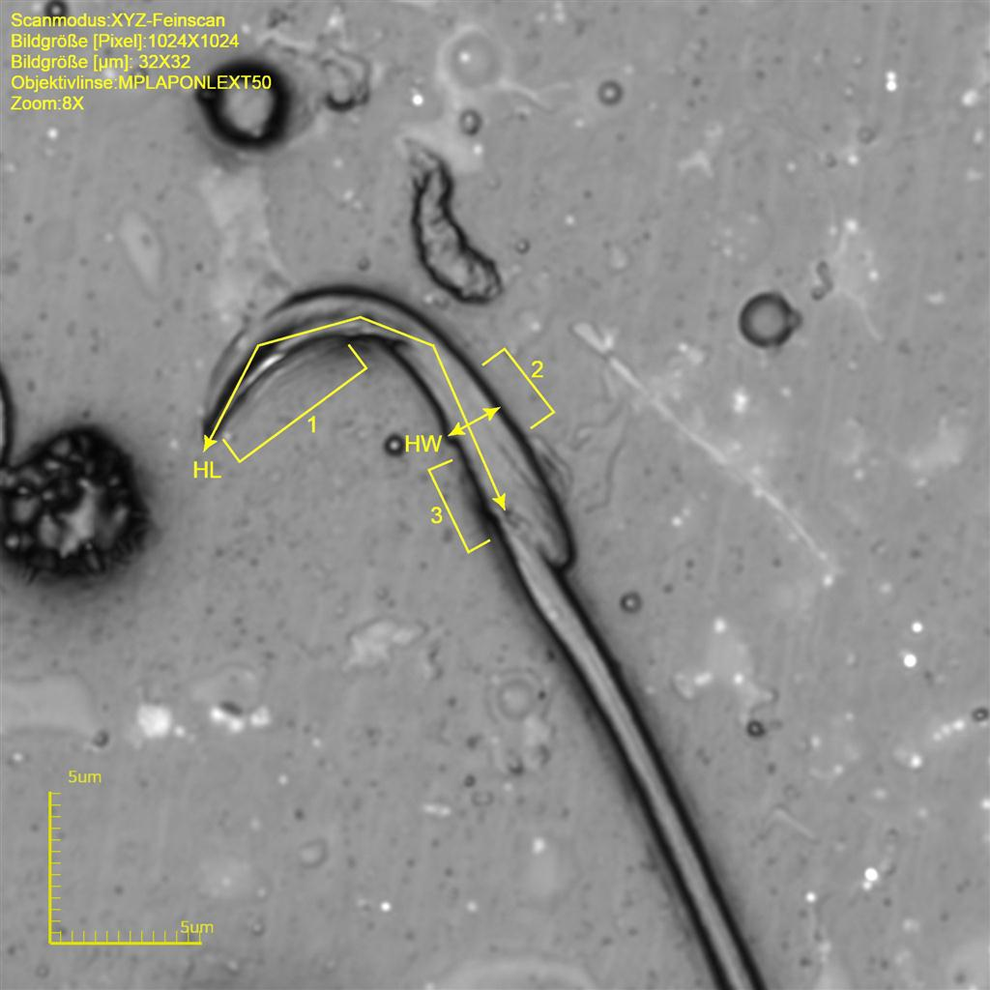
Rat sperm at original magnification ×8576. Note different morphological acrosome regions (1 = anterior acrosomal segment, 2 = equatorial segment, and 3 = postacrosomal segment). Measured length dimensions are indicated by lines and arrows; HL indicates head lengths; HW, width at the equatorial segment.
The angle of approximate 90° is formed by the anterior acrosomal segment. The approximate width at the equatorial segment is 1.9 to 2.4 µm and the approximate length is 12.2 to 16.9 µm (approximate circumference 28.0 to 44.1 µm and an area of 24.1 to 35.0 µm2). The tail of rat sperm is very long, with an approximate length of 171.0 to 178.8 µm and a width at the midst of 0.8 to 1.2 µm makes around 92% of the total sperm length (Table 3 and Figure 7).
Descriptive Statistics on Measured Dimensions From Rat Sperm.a
Abbreviations: SD, standard deviation; max, maximum; min, minimum.
aLength (µm), area (µm2), n = 100.
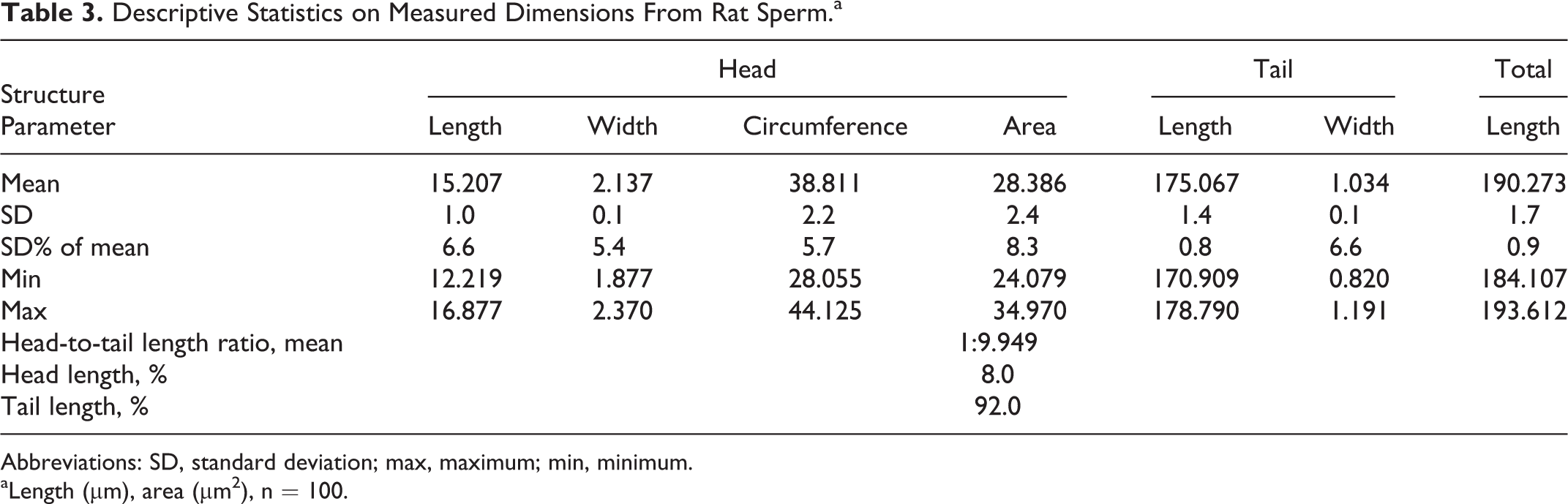
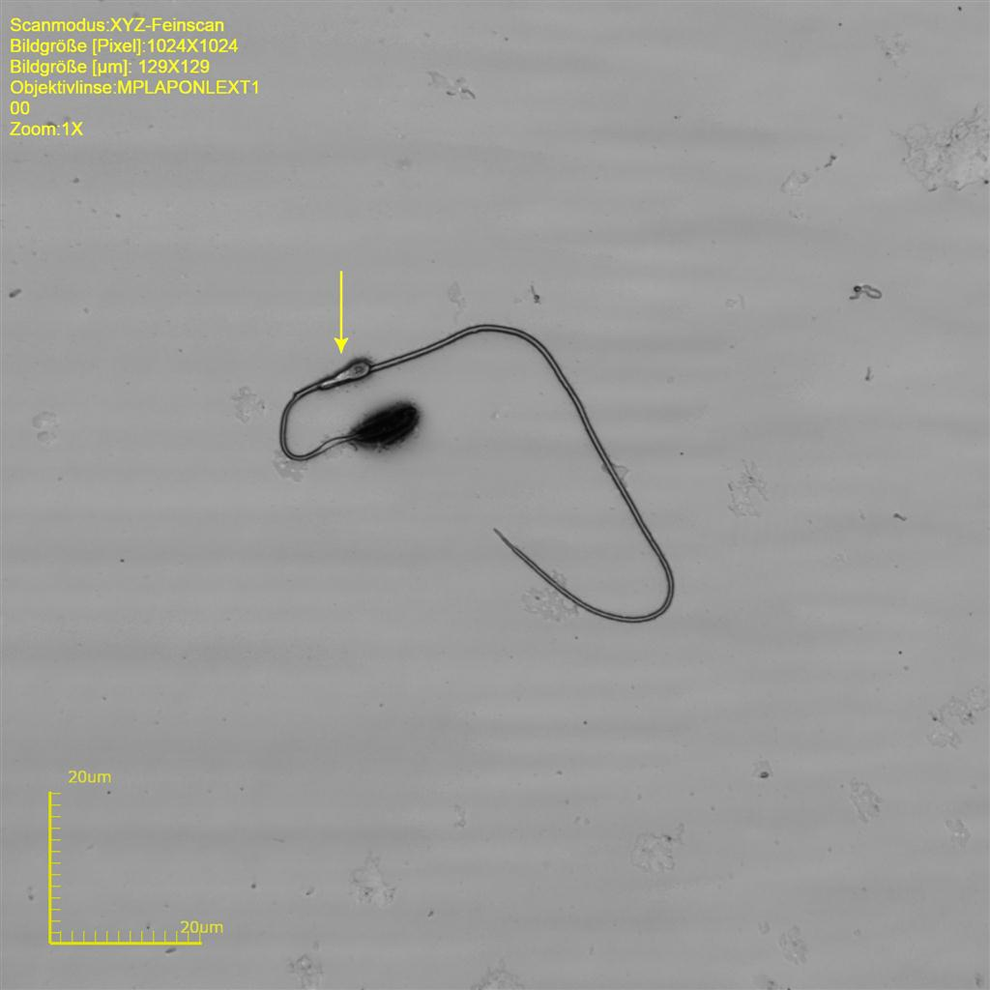
Almost every sperm sampled from the vas deferens shows a cytoplasmic droplet at a mean distance of 63.4 µm distal to the head (Figure 8). The droplets are considered to be an indicator for sperm motility and normal spermiogenesis. The authors assume that abnormal droplet formation, for example, lack of droplet or ectopic droplet, is indicative of defective spermiogenesis. 17,22 Abnormalities in naive animals do not appear often, but missing droplet (Figure 9), pyknotic sperm heads (Figure 10), plasma membrane defects on tail (Figures 11 and 12), curvature abnormalities (Figure 13), and kinks in the region of the cytoplasmic droplet (Figure 14) were observed. Single heads (Figure 15) and tails without head are common in smears and are considered to be preparation artifacts rather than abnormalities in most cases.
Rat sperm at original magnification ×14954. Cytoplasmic droplet at tail.
Rat sperm at original magnification ×1072. Note 1 sperm without droplet (arrow).
Rat sperm at original magnification ×2136. Pyknotic head and kink tail (arrow).
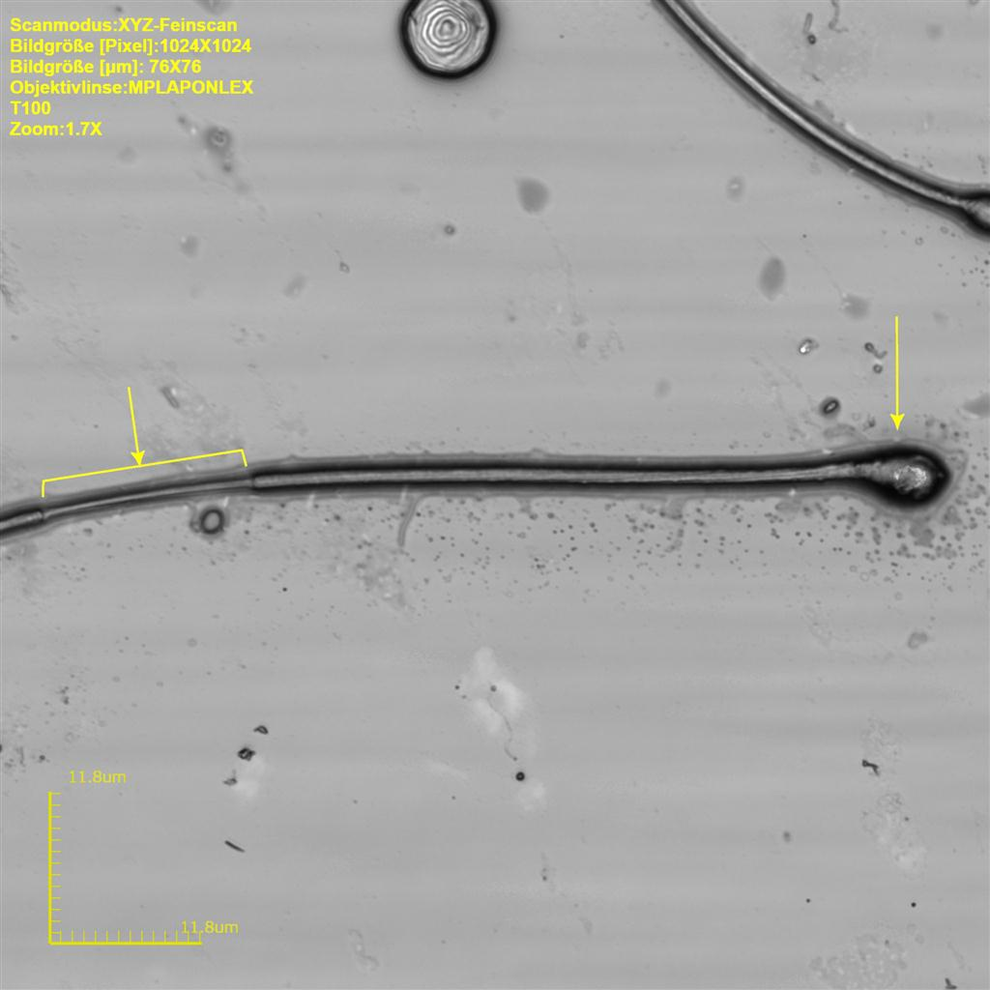
Rat sperm at original magnification ×3632. Reduced plasma membrane in region of droplet insertion (square bracket) and pyknotic sperm head (arrow).

Rat sperm at original magnification ×12 818. Higher magnification of Figure 11.
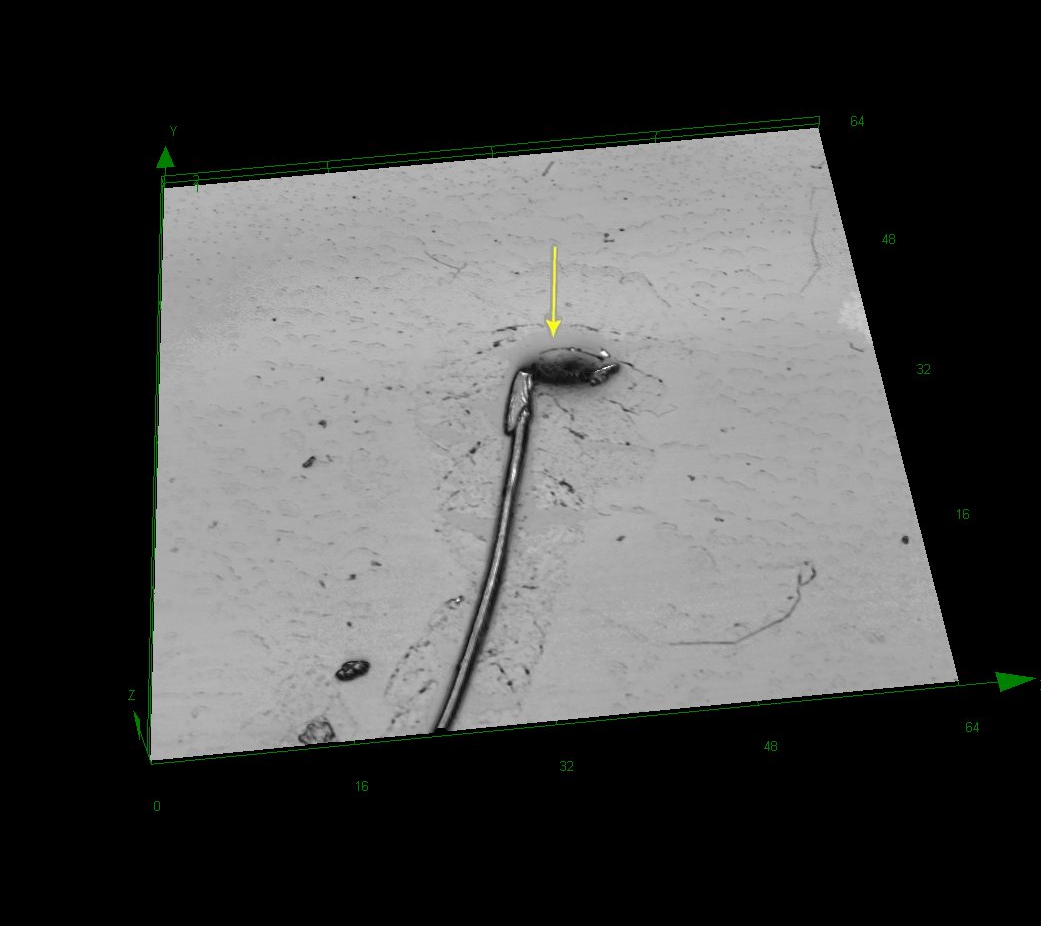
Rat sperm at original magnification ×4273. Curvature abnormality and broken head (arrow).
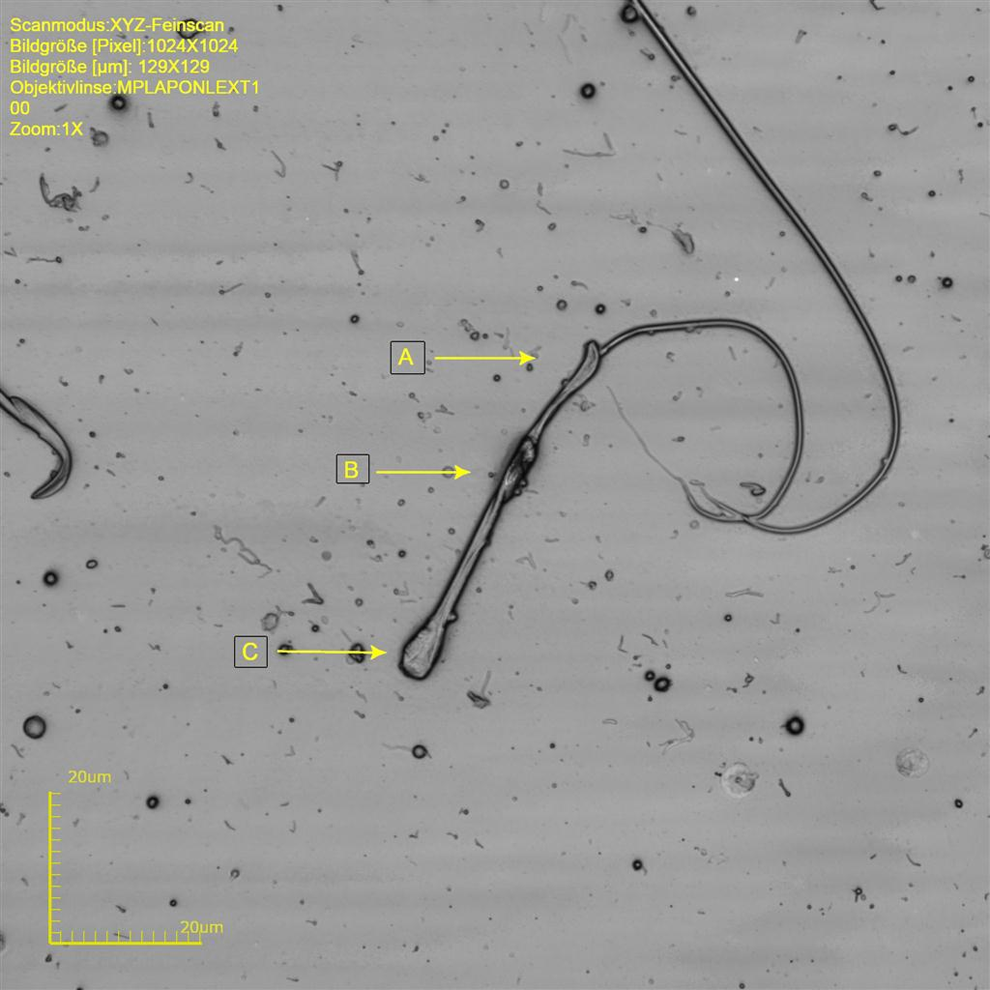
Rat sperm at original magnification ×8545. C, Kink tail at position of cytoplasmic droplet. A, Sperm head with abnormal curvature. B, Cytoplasmatic remnant.
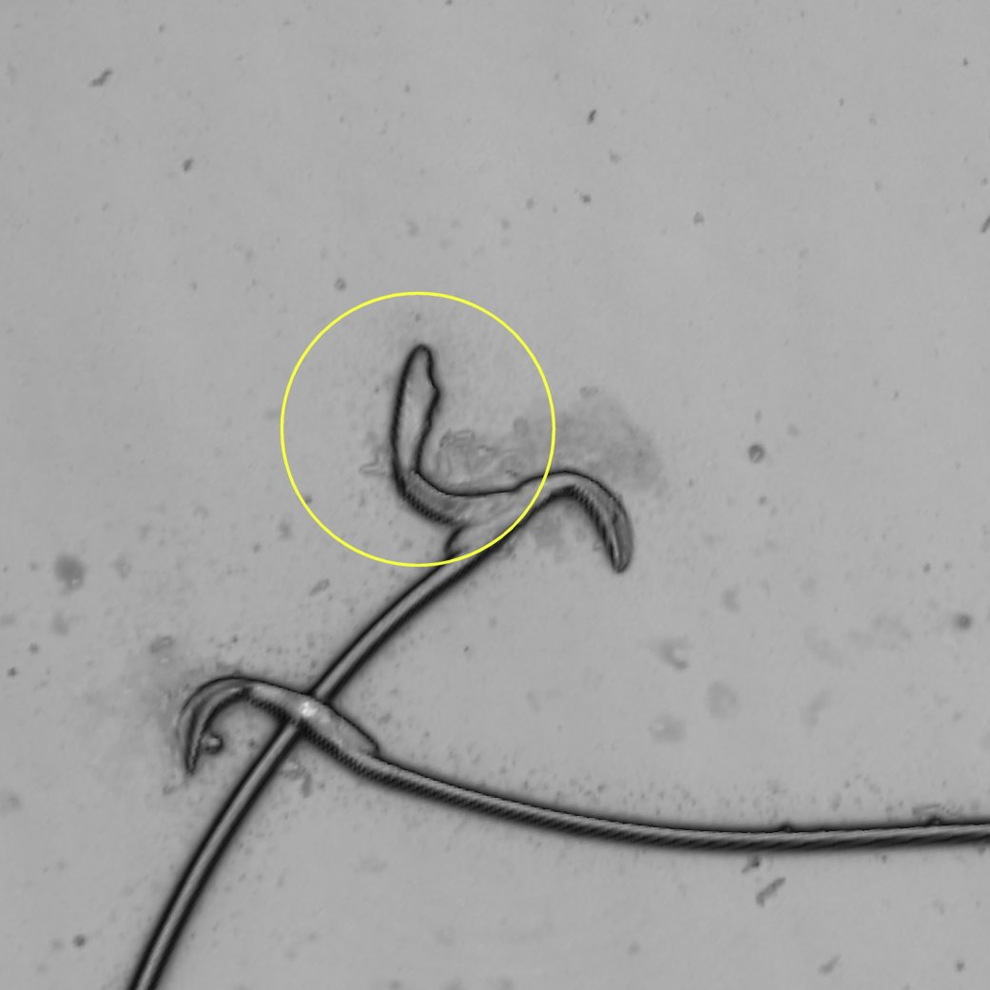
Rat sperm at original magnification ×2136. Single sperm head attached to intact sperm mimicking double-headed sperm. Artifact (circle).
Evaluation of Mouse Sperm by LSM
The mouse sperm head is relatively short and plump, however resembles rat sperm head (falciforme shape). Again, the sperm head can be divided into the anterior acrosomal segment followed by the equatorial segment and the postacrosomal segment (Figure 16).
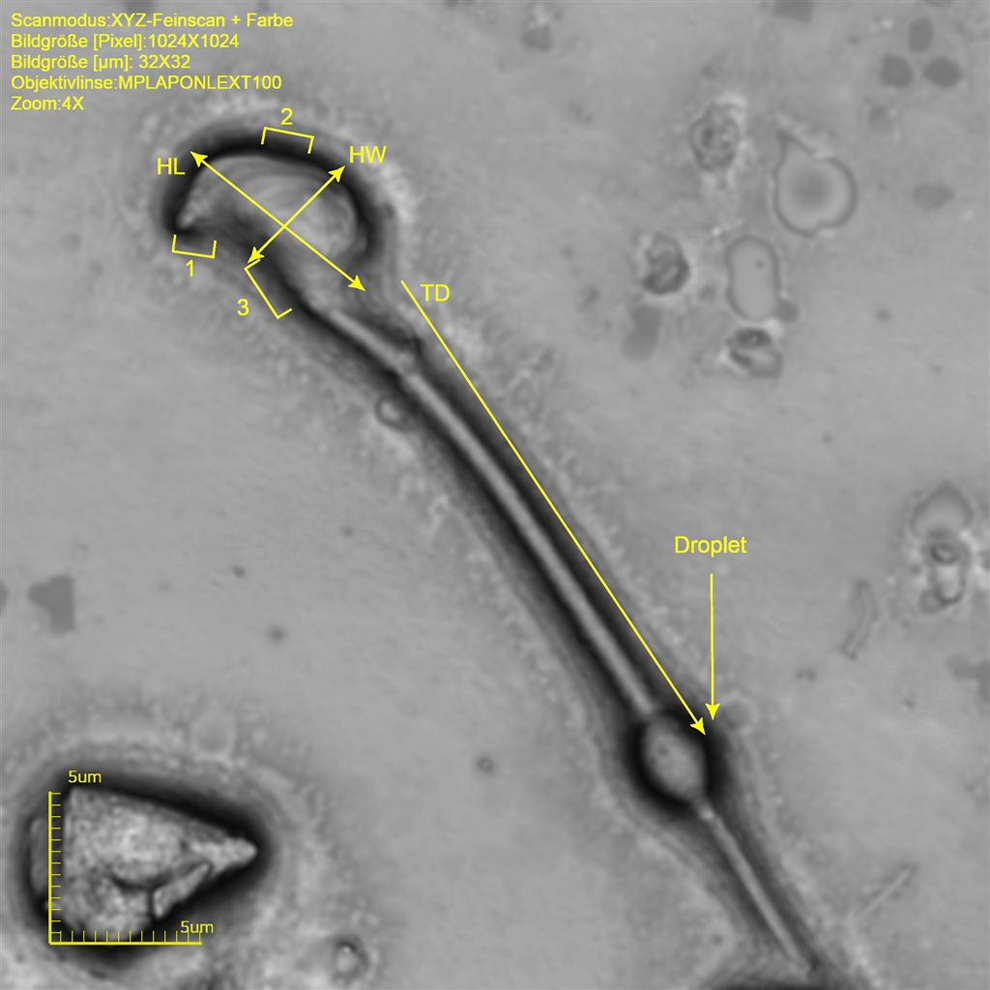
Mouse sperm at original magnification ×8536. Note different morphological acrosome regions (1 = anterior acrosomal segment, 2 = equatorial segment, and 3 = postacrosomal segment). Measured length dimensions are indicated. HL indicates head lengths; HW, width at the equatorial segment, TD, distance of tail insertion to head until mid of droplet.
Also in mouse sperm, the approximate angle of the anterior acrosomal segment is 90°. The approximate width at the equatorial segment is 3.7 to 4.4 µm and an approximate length is 6.4 to 8.6 µm (approximate circumference 21.1-29.4 µm and an area of 19.5-38.0 µm2). The tail of mouse sperm is not as long when compared to rat sperm, with an approximate length of 114.0 to 121.9 m and a width at the midst of 0.9 to 1.2 µm. The sperm tail makes around 94% of the total sperm length (Table 4 and Figure 16). As in rats, almost every sperm sampled from the vas deferens shows a cytoplasmic droplet at a mean distance of 21.1 µm distal to the head (Figure 16).
Descriptive Statistics on Measured Dimensions From Mouse Sperm.a
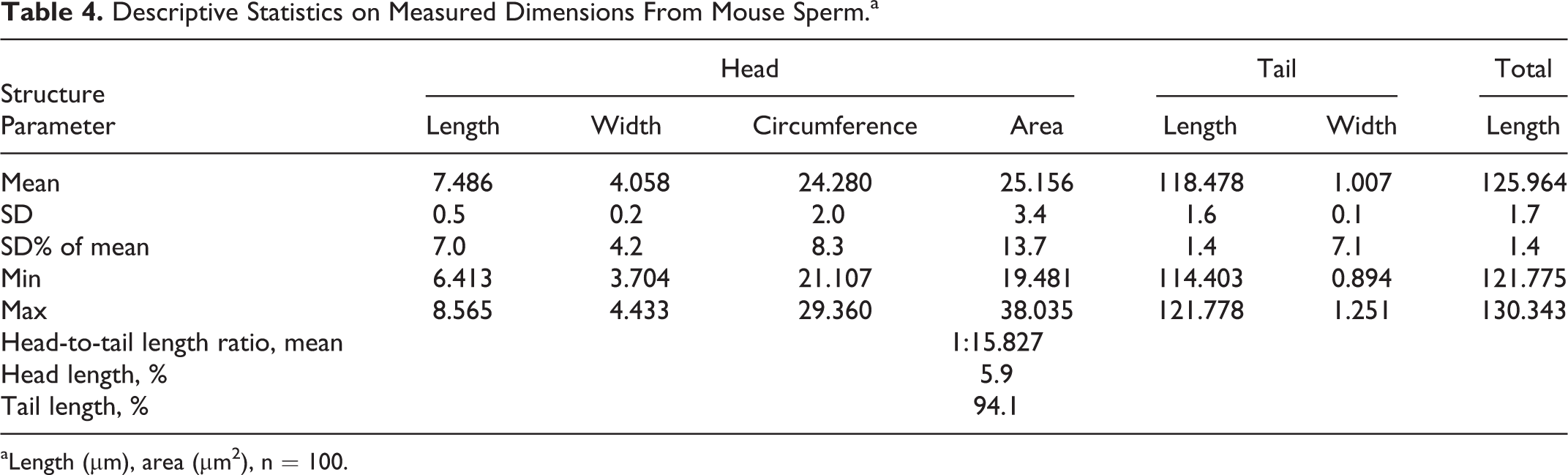
aLength (µm), area (µm2), n = 100.
Conclusions
Laser scanning microscopy is a technique using a focused laser for scanning an object. The technique provides live images that are comparable to those of a scanning electron microscope under relatively low-power magnifications. This technology was not applied to tissues or cells before. The reason is simply that a scanning laser is always reflected by wet and/or reflecting surfaces and hence, imaging is hindered or impossible. The focused laser beam did not damage any of the prepared samples. However, using a fixative for electron microscopy evaluation and handling dried slides of cell smears without coverslipping, the application of this technology was deemed to be possible.
Sperm of rats, mice, rabbits, and cynomolgus monkeys was evaluated at magnifications up to ×17 090. The application of this technology provided results within minutes once a properly prepared slide underwent the evaluation process, even at magnifications that are usually only achieved by electron microscopy. Usually, light microscopy is applied to the evaluation of sperm smears, that is, information on abnormalities is lost since magnifications of maximally ×1200 are applicable by light microscopy. For higher magnifications, the expensive methods, for example, electron microscopy, must be applied. Furthermore, morphological details on stained sperm smears, for example, cytoplasmic droplets, are not visible at low magnifications (without using oil immersion) and at least objectives of ×100. In contrast, LSM provides the possibility of evaluations at both light and electron microscopy levels.
The measurements on sperm parameters were taken by an integrated image analysis software. Length and width of the head, as well as the total length in the sperm of all species evaluated for this presentation, are consistent with published data. 15 This supports further the high accuracy of the presented technology. Abnormalities were easily detectable. Therefore, the technology used is deemed to be a useful tool for qualitative sperm analysis and the interpretation of induced sperm injury.
Footnotes
Author Contribution
The studies were designed by Weber K, Fendl D, Ordonez P, Takawale P, König A, Ribero J, and Riedel W. The data aquisition was performed by Weber K, Waletzky A, Paepke S. Ordonez P, Fendl D, Hein F, Ronano I, and Quirici R.The manuscript was drafted by Weber K and Waltzky A, and a critical review was performed by Okazaki Y, Querici R, Romano I and Hardisty J.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
