Abstract
Phosphatidylinositol 3-kinase (PI3K) δ is a lipid kinase primarily found in leukocytes, which regulates important cell functions. AMG2519493 was a PI3K δ-specific inhibitor in development for treatment of various inflammatory diseases. AMG2519493-related changes in the male and/or female reproductive organs were observed in the 1- and 3-month oral repeat dose toxicology studies in the rat and cynomolgus monkey. Hemorrhagic corpora lutea cysts and increased incidence of corpora lutea cysts without hemorrhage were observed in the ovaries at supra pharmacological doses in the rat. A decrease in seminiferous germ cells in the testis, indicative of spermatogenesis maturation arrest, was observed in both the rat and cynomolgus monkey. Although the characteristics were comparable, the drug systemic exposures associated with the testicular changes were very different between the 2 species. In the rat, the testicular change was only observed at supra pharmacologic exposure. Isotype assessment of PI3K signaling in rat spermatogonia in vitro indicated a role for PI3K β, but not δ, in the c Kit/PI3K/protein kinase B signaling pathway. Therefore, changes in both the ovary and testis of the rat were considered due to off target effect as they only occurred at suprapharmacologic exposure. In contrast, the testicular changes in the cynomolgus monkey (decrease in seminiferous germ cells) occurred at very low doses associated with PI3K δ-specific inhibition, indicating that the PI3K δ isoform may be important in spermatogenesis maturation in the cynomolgus monkey. Our results suggest species-related differences in PI3K isoform-specific control on reproductive organs.
Introduction
The phosphatidylinositol 3 kinase (PI3K) family is divided into 4 different classes, I to IV. Class I molecules are further divided between IA and IB. PI3K δ is 1 of 3 class IA PI3Ks which are heterodimeric lipid kinases that function in a key proximal role of multiple cell surface transduction receptors to regulate intracellular signaling events. 1 In contrast to the other class IA isoforms (PI3K α and β) which are ubiquitously expressed, PI3K δ has a much more restricted expression pattern, primarily found in leukocytes where it regulates important cell functions such as growth, differentiation, cell death/survival, adhesion, motility, and migration. 2 Phosphatidylinositol 3-kinase δ is required for B cell receptor–mediated activation and differentiation leading to immunoglobulin secretion, and PI3K δ inhibition leads to a selective depletion of antigen-activated B cells. In addition to its critical role in B cell activation, PI3K δ inhibition partially impairs other leukocyte functions, 3 including cytokine/chemokine production by T cells, natural killer cells, basophils, mast cells, and neutrophils.
The PI3K δ gene is highly conserved across animal species. Rodent models have been used extensively to investigate the role of PI3K δ in inflammation. Mice expressing a catalytically inactive form of PI3K δ by genetic targeting are healthy and fertile, and have a leukocyte predominant phenotype. 4 These PI3K δ-deficient mice lack antigen-specific B cell functions and have partial reductions in the function of T cells, neutrophils, monocytes/macrophages, and mast cells. Blockade of PI3K δ has demonstrated efficacy in multiple inflammatory disease animal models.3,5 The role of PI3K signaling in immune cells of the intestinal mucosa has been extensively reviewed. 6 Evidence suggests that PI3K γ 6 and PI3K δ7-9 are important isoforms in intestinal inflammation and that deregulation of the PI3K pathway can result in inflammatory bowel disease and its associated cancers.
In contrast to the extensive information on the roles of PI3K δ on the immune system, information on PI3K δ involvement in the reproductive system in both males and females is limited. 10 Okkenhaug et al reported that their mice with a catalytically inactive form of PI3K δ were fertile. 4 In contrast, Li et al reported that their PI3K δ-null female mice were subfertile exhibiting fewer growing follicles and more atretic antral follicles in the ovary; female null mice also responded poorly to exogenous gonadotropins as compared with control mice. 11 In the male reproductive system, PI3K is reported to have a role in several functions, including self-renewal, survival and differentiation of spermatogonial stem cells (SSC), sperm motility, Sertoli cell glucose transport and proliferation, and Leydig cell steroidogenesis.12-16 These reports were primarily based on mouse or rat models with an only exception of the study using human spermatozoa. 16 Ciraolo et al reported a pivotal PI3K β isoform-specific role for spermatogenesis in mice. 14 However, to date, no PI3K δ-specific role has been reported for the male reproductive system.
AMG2519493, a highly selective and potent small molecule inhibitor of PI3K δ, was developed for the treatment of inflammatory diseases. In vitro, AMG2519493 inhibits PI3K δ with a high selectivity against all other PI3K class I isoforms (>400-fold δ selectivity over β, α, or γ based on recombinant human PI3K δ and other class I PI3K isoforms). The nonclinical safety profile of AMG2519493 was evaluated in repeat dose toxicology studies up to 3 months, in the Sprague Dawley (SD) rat and the cynomolgus monkey. Here, we report AMG2519493-related changes observed in the testes and ovaries including their characteristics, time course, relation with the drug exposure, and putative mechanisms of action.
Materials and Methods
Test and Control Article
Test articles, including AMG2519493 and 15 additional PI3K inhibitors with different PI3K isoform specificity, were synthesized at Amgen Inc. In the toxicology studies, the vehicle control article was 0.5% (wt/vol) hydroxypropyl methylcellulose/1% (wt/vol) Pluronic F68 in reverse osmosis water, and pH adjusted to 2.0 ± 0.1 with dilute hydrochloric acid. AMG2519493 was suspended with the vehicle control for the dosing formulation with appropriate concentrations for the toxicology studies. Stability of the test article was confirmed for the duration of the toxicology studies, and all dose formulations were homogeneous and met concentration verification criteria. For in vitro functional assessment in rodent spermatogonia, AMG2519493 and other PI3K inhibitors were dissolved in dimethyl sulfoxide.
In Vivo Repeat-Dose Toxicology Studies
All animals were housed at Association for Assessment and Accreditation of Laboratory Animal Care international accredited facilities, and all research protocols were approved by their respective institutional animal care and use committees. All repeat-dose toxicology studies were performed in accordance with Good Laboratory Practices (US Code of Federal Regulations, Title 21, Part 58: Good Laboratory Practice for Nonclinical Laboratory Studies).
Sprague Dawley (Crl:CD) rats were group-housed by sex (up to 3 animals/sex/cage) in polycarbonate cages with bedding. Animals had ad libitum access to water and pelleted feed, except for an overnight food fast prior to blood collection at scheduled necropsy. All animals were maintained on a 12:12 hour light:dark cycle in rooms with appropriate temperature and humidity controls.
Cynomolgus monkeys (Macaca fascicularis) of Chinese origin were group-housed (up to 4 animals/sex/cage) in stainless steel cages and offered Certified Primate Diet #2055C (Harlan Laboratories, Inc) 1 to 2 times daily and water ad libitum. Environmental controls for the animal room were set to maintain 18 °C to 26 °C, a relative humidity of 30% to 70%, 10 or greater air changes/hour, and 12:12 hour light:dark cycle. Animals were given fruits, vegetables, or additional supplements and various forms of environmental enrichment. Both rats and cynomolgus monkeys euthanized for unscheduled or scheduled necropsies were anesthetized with sodium pentobarbital, exsanguinated, and necropsied.
Rat Toxicology Study Designs
In the rat 1-month repeat-dose toxicology study, AMG2519493 was administered daily by oral gavage at dose levels of 0 (vehicle control), 3, 10, or 100 mg/kg for 1 month (main toxicology groups: n = 10-15/sex/group; toxicokinetic satellite groups: n = 4/sex/group; 7 weeks old at initiation of dosing). Scheduled necropsies were conducted at the end of the 28-day dosing phase (n = 10/sex/group) and following a 28-day recovery phase (n = 5/sex in the vehicle and 100 mg/kg groups).
In the rat 3-month repeat-dose toxicology study, AMG2519493 was administered daily by oral gavage at dose levels of 0 (vehicle control), 3, 10, or 100 mg/kg (main toxicology groups: n = 20-25/sex/group; toxicokinetic satellite groups: n = 4/sex/group, 8-weeks-old at initiation of dosing). Scheduled necropsies were conducted on study day 29 (n = 10/sex/group), at the end of the 91-day dosing phase (n = 9-10/sex/group), and following a 2-month recovery phase (n = 5/sex in vehicle and 100 mg/kg groups).
Cynomolgus Monkey Toxicology Study Designs
In the cynomolgus monkey 1-month repeat-dose toxicology study, AMG2519493 was administered daily to sexually mature animals by oral gavage at dose levels of 0 (vehicle control), 0.1, 1, 8, or 200 mg/kg (n = 3-5/sex/group, 5.6- to 8.5-years-old at initiation of dosing). The 8 mg/kg dose level was anticipated to result in PI3K δ-specific inhibition, without inhibition of other PI3K isoforms. The lowest dose level (0.1 mg/kg) was expected to be subtherapeutic. Scheduled necropsies were conducted at the end of the 28-day dosing phase (n = 2-3/sex/group), and following a 1-month recovery (n = 2/sex in vehicle or 200 mg/kg group).
In the cynomolgus monkey 3-month repeat-dose toxicology study, AMG2519493 was administered daily to animals by oral gavage at dose levels of 0 (vehicle control), 1, 8, or 100 mg/kg (n = 4-6/sex/group, 4-8 years old at initiation of dosing). The intended duration of dosing was 3 months with a 2-month recovery phase; however, the dosing was discontinued in the 100 mg/kg group on study day 49 due to unexpected nontolerability (see below).
Toxicology Study Parameters
For both the rat and monkey studies, end points included clinical observations, body weight, qualitative food consumption, toxicokinetics, ophthalmic evaluation, clinical pathology, organ weights, macroscopic observation, and light microscopic evaluation of a full tissue list. Only the AMG2519493-related changes in the reproductive tract are described here.
In Vitro Functional Assessment of PI3K β and δ in Rodent Spermatogonia
To understand the role of PI3K β and δ on rodent spermatogenesis, phosphorylation of protein kinase B (AKT) was evaluated as a surrogate of PI3K activity in isolated rodent spermatogonia stimulated with stem cell factor (SCF) and incubated with PI3K inhibitors with different β and δ isoform selectivity (AMG2519493 and 15 other PI3K inhibitors).
Spermatogonia were isolated from 10- to 12-day-old SD rats and 8- to 9-day-old CD-1 mice as previously described. 17 Immediately following isolation, the spermatogonia were incubated with PI3K inhibitors with varying specificity for PI3K δ and β for 60 minutes at 34 °C, followed by stimulation with SCF (100 ng/mL, #455 MC/CF, R&D Systems) for 7 minutes. Spermatogonia were then fixed with 3.7% paraformaldehyde (Thermo Fisher Scientific) for 10 minutes and washed with 2% fetal bovine serum (#110099-141; Thermo Fisher Scientific) in Dulbecco phosphate-buffered saline (without Ca2+/Mg2+; #14190-144; Thermo Fisher Scientific). After washing, spermatogonia were permeabilized and stored overnight in 90% methanol (Thermo Fisher Scientific) at 20 °C. Spermatogonia were then washed as above with 2% fetal bovine serum in Dulbecco phosphate-buffered saline (without Ca2+/Mg2+) and stained with a rabbit anti-phospho AKT (Ser473) monoclonal antibody (#4058 L; Cell Signaling Technology) and an Alexa Fluor 488-conjugated mouse anti-GATA4 monoclonal antibody (#560330; BD Biosciences). The phosphor AKT (Ser473) antibody was detected with an Alexa Fluor 647-conjugated goat anti-rabbit secondary antibody (#A21245; Invitrogen). Flow cytometry was then performed on a BD LSRII flow cytometer (BD Biosciences). Fluorescent mean values of phosphor AKT gated on GATA4 negative cells (spermatogonia population) were calculated using BD DIVA software v6.1.1 (BD Biosciences).
The half maximal inhibitory concentration values (IC50) of SCF-mediated phosphorylation of AKT in spermatogonia were determined for the PI3K inhibitors from transformed normalized data using nonlinear regression on a variable slope in GraphPad Prism software (version 4.01, GraphPad Software Inc., San Diego, CA, USA). The PI3K β IC50 and the PI3K δ IC50 for the PI3K inhibitors were previously determined in a PI3K β counter screen assay in MDA MB 468 cells 18 and an anti-IgM-induced AKT phosphorylation (Ser473) assay in mouse B cells, 19 respectively. A correlation analysis between the IC50 for PI3K β or δ against the IC50 for AKT phosphorylation in the spermatogonia assay was performed for R2 coefficient of determination to establish which PI3K isoform is primarily involved in the SCF/c Kit/PI3K/AKT signaling pathway in spermatogonia in the rat.
Results
In-Life Results in the Toxicology Studies
In the rat toxicology studies, there were no AMG2519493-related clinical signs. In the cynomolgus monkey toxicology studies, gastrointestinal (GI) toxicity characterized by inflammation in the large intestine secondary to AMG2519493-related immunomodulation was observed as a dose-limiting toxicity. 9 In the 3-month cynomolgus monkey study, dosing in animals at the high dose of 100 mg/kg was discontinued on study day 49 due to clinical signs of GI toxicity.
AMG2519493 systemic exposure increased with ascending dose with no significant accumulation after repeat dosing in both rats and cynomolgus monkeys (Supplemental Tables 1 and 2). In the 1-month cynomolgus monkey study, flow cytometry analysis of peripheral blood lymphocytes demonstrated a dose-dependent decrease in B cells, consistent with the intended pharmacological effects of PI3K δ inhibition. 9
Ovarian Changes in the Rat
Cystic corpora lutea (CL), with and without hemorrhage, were observed in the rat. Both types of cystic changes were considered AMG2519493-related (Figure 1 and Supplemental Table 3). Hemorrhagic cystic CL were observed at a low incidence (10%-20%) at the high-dose level in both studies. In contrast, cystic CL without hemorrhage were observed at all dose levels including control in the 3-month study. At the end of the 3-month administration, the incidence of cystic CL was the same between control and 10 mg/kg and slightly higher at only 100 mg/kg compared to the control. Therefore, hemorrhagic cystic CL and increased incidence in cystic CL (without hemorrhage) observed only at the high dose of 100 mg/kg were considered AMG2519493-related changes; both changes did not progress in terms of incidence or severity between 1- and 3-month durations.
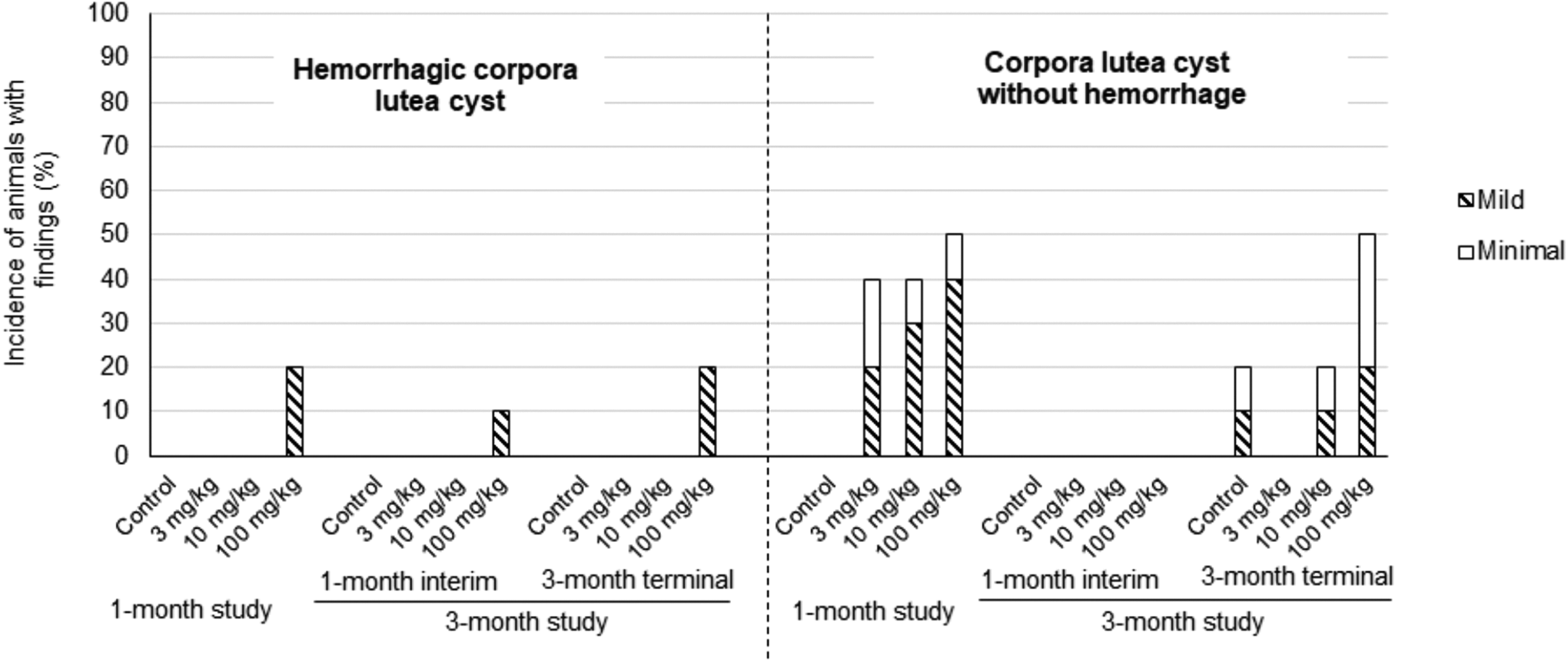
Incidence and severity of cystic changes in the ovaries in 1- and 3-month rat studies AMG2519493 was administered to the rat once daily by oral gavage at dose levels of 0 (vehicle control), 3, 10, or 100 mg/kg for 1 month or 3 months.
Hemorrhagic cystic CL were distinguished from cystic CL by the presence of red blood cells filling the cystic space (Figure 2) and by the occasional correlation with red to black discoloration of the ovary at necropsy. Among the affected animals, the cystic changes in the ovary were unilateral or bilateral, focal (minimal grade) to multifocal (mild to slight grade), and within CL of normal size from the most recent ovulation. The absence of cystic changes in older CL suggests that cystic CL were transient and did not accumulate following repeated estrous cycles. In all animals administered AMG2519493, the ovary, vagina, and uterus were in appropriate morphologic synchrony, and there was no apparent change in the number of follicles or CLs in the ovary, which indicates a normal functioning hypothalamic-pituitary-ovarian hormonal axis. After the recovery phases, no cystic changes were observed in the rat indicating full recovery. No changes in the ovary were observed in the AMG2519493 cynomolgus monkey toxicology studies.
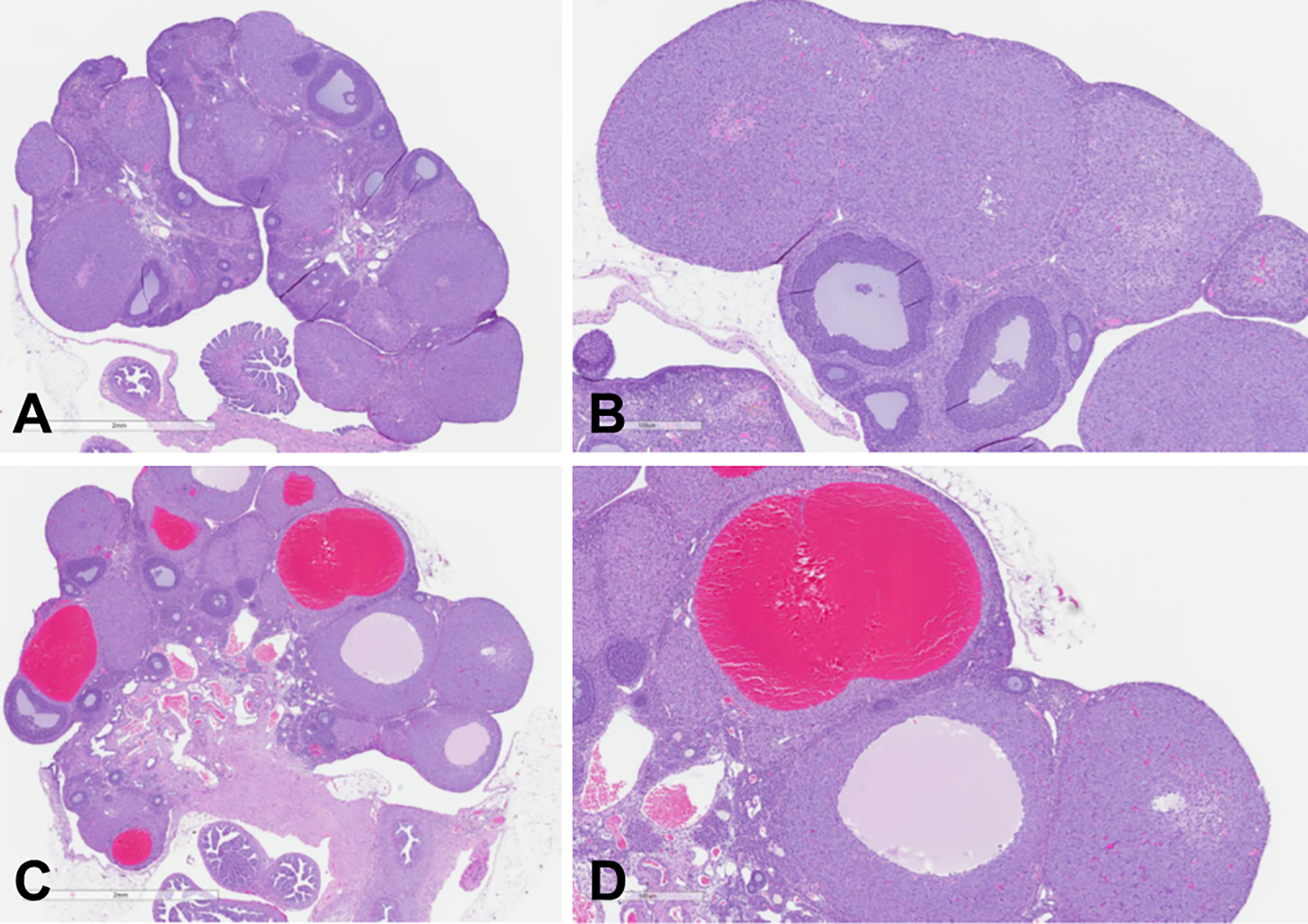
Representative low (original objective ×2, panel A and C) and high (original objective ×5, panel B and D) magnification photomicrographs of normal control rat ovary (A and B) and an ovary from a rat administered 100 mg/kg AMG2519493 (C and D) in the 3-month toxicology study. Images demonstrate multiple AMG2519493-related cystic corpora lutea, some of which are filled with blood. Staining: hematoxylin and eosin.
Testicular Changes in the Rat and Cynomolgus Monkey
Testicular changes characterized by decreased testes weight and decreased seminiferous germ cells were observed in both the rat and cynomolgus monkey (Supplemental Tables 4 and 5). Although the pattern of the testicular changes was similar between the rat and monkey, the monkey was more sensitive. In the rat, decrease in testes weight was observed in all AMG2519493-dosed groups in the 3-month study (Figure 3); the magnitude of the decrease was approximately 80% of controls. Light microscopically, decreased germ cells were generally observed only at the high dose of 100 mg/kg (Figure 4 and Supplemental Table 4). No testicular change was observed in recovery animals, indicating reversibility.
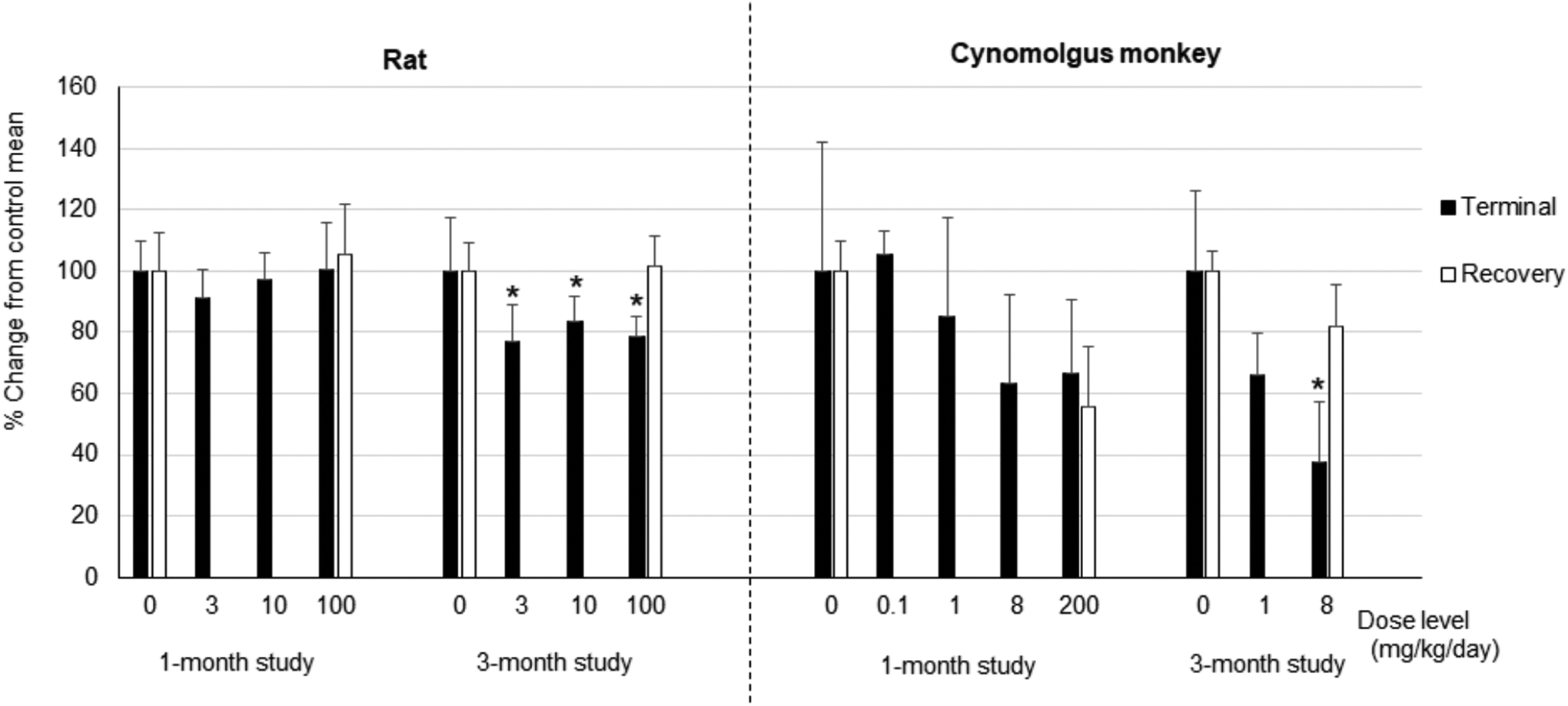
Testes weight in the rat and cynomolgus monkey AMG2519493 was administered daily by oral gavage to the rat and cynomolgus monkey for 1 or 3 months. The animals were euthanized at the end of the administration period or recovery period, and testes weight (absolute weight and weight relative to body weight) was measured. The graph represents percentage change from the time-matched control mean value of the testes weight relative to body weight. Error bar = SD. Rat study: n = 9 or 10 at terminal necropsy, n = 5 at recovery necropsy; cynomolgus monkey study: n = 3 at terminal necropsy (n = 2 at 200 mg/kg) and n = 2 at recovery necropsy in the 1-month study; n = 4 at terminal necropsy (n = 2 at 8 mg/kg) and n = 2 at recovery necropsy in the 3-month study. Recovery period: 1-month studies: 1-month recovery and 3-month studies: 2-month recovery. *P < .05.
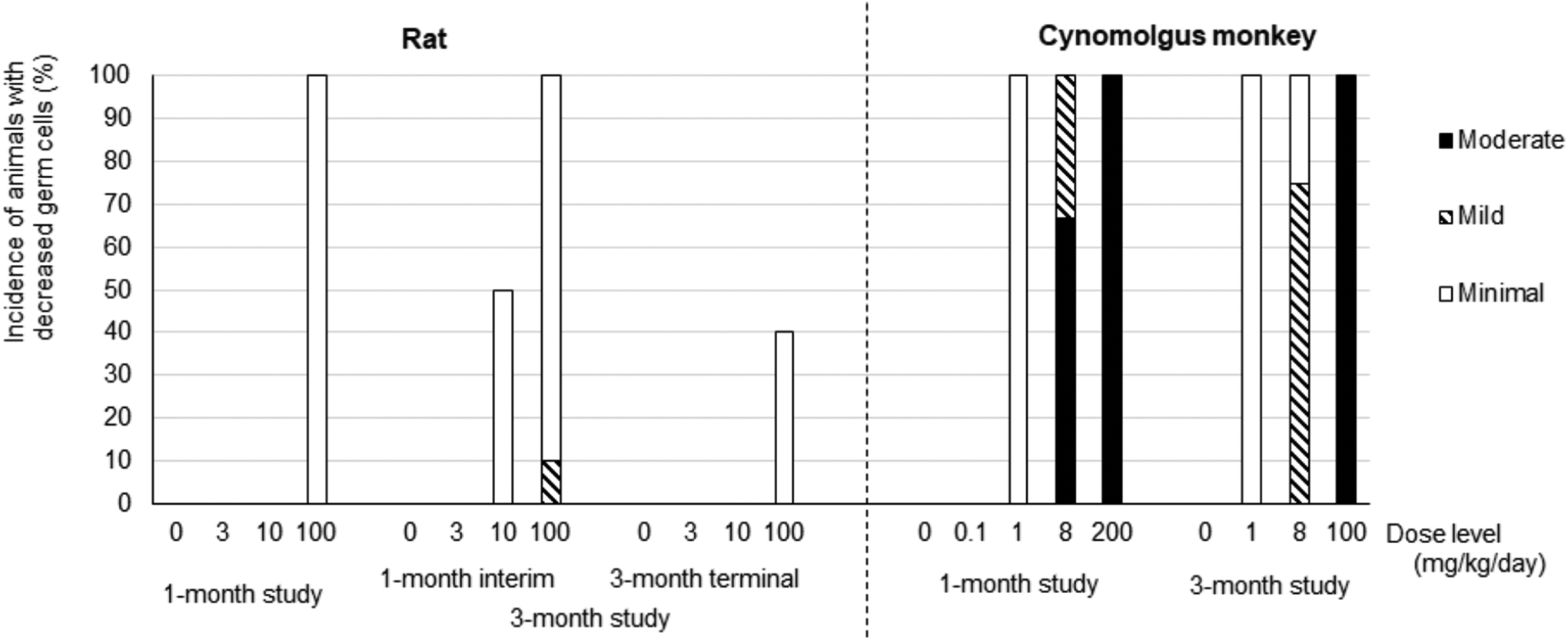
Incidence of animals with decreased germ cells in the testicular seminiferous tubules AMG2519493 was administered daily by oral gavage to the rat and cynomolgus monkey for 1 or 3 months. The animals were euthanized at the end of the administration period or recovery period. The dosing in cynomolgus monkeys at 100 mg/kg in the 3-month study was discontinued on study day 49, followed by a 2-month recovery phase. The graph represents % incidence of animals with decreased germ cells. The decreased germ cells were classified based on the severity described in Figure 5 into minimal, mild, and moderate. Rat study: n = 9 or 10; cynomolgus monkey study: N = 3 (n = 2 at 200 mg/kg) in the 1-month study; n = 4 in the 3-month study.
In the cynomolgus monkey, a dose-dependent decrease in testes weight was observed (Figure 3). At 8 mg/kg in the 3-month study, the testes weight was approximately 40% of controls. Decreased testes weight was still observed after the 1-month recovery in the 1-month study; however, the testes weight at 100 mg/kg recovered to approximately 80% of the time-matched control mean after the 2-month recovery period in the 3-month study (Supplemental Tables 5). Light microscopically, decreased germ cells were observed at ≥1 mg/kg, and no testicular changes were observed at the subtherapeutic dose of 0.1 mg/kg (Figure 4 and Supplemental Table 5). Consistent with the change in testes weight, the magnitude of decreased germ cells was also dose-dependent from minimal to moderate; decreased germ cells were still observed after a 1-month recovery in the 1-month study but were fully reversible after a 2-month recovery in the 3-month study.
Despite significant differences in sensitivity, the light microscopic characteristics of the testicular changes were similar between the rat and cynomolgus monkey (Figure 5 and Supplemental Tables 4 and 5). A decrease in germ cells is consistent with maturation arrest of spermatogenesis occurring at the level of an early germ cell progenitor, such as spermatogonia. 20 The primary target cell population was considered late stage spermatogonia based on the morphology in the animals with the findings at a minimal degree of severity (Figure 5B). In the monkey at higher dose levels, testicular changes also involved spermatocytes, spermatids, and sperm (Figure 5D). There was no light microscopic evidence indicative of tissue injury in the other components of the testes, including seminiferous tubule basement membrane, Sertoli cells, interstitial cells, rete testis, or vasculature. There were no AMG2519493-related changes in other male reproductive organs except for decreased weight of the epididymis that correlated light microscopically to luminal cellular debris and aspermia/hypospermia (Supplemental Tables 4 and 5).
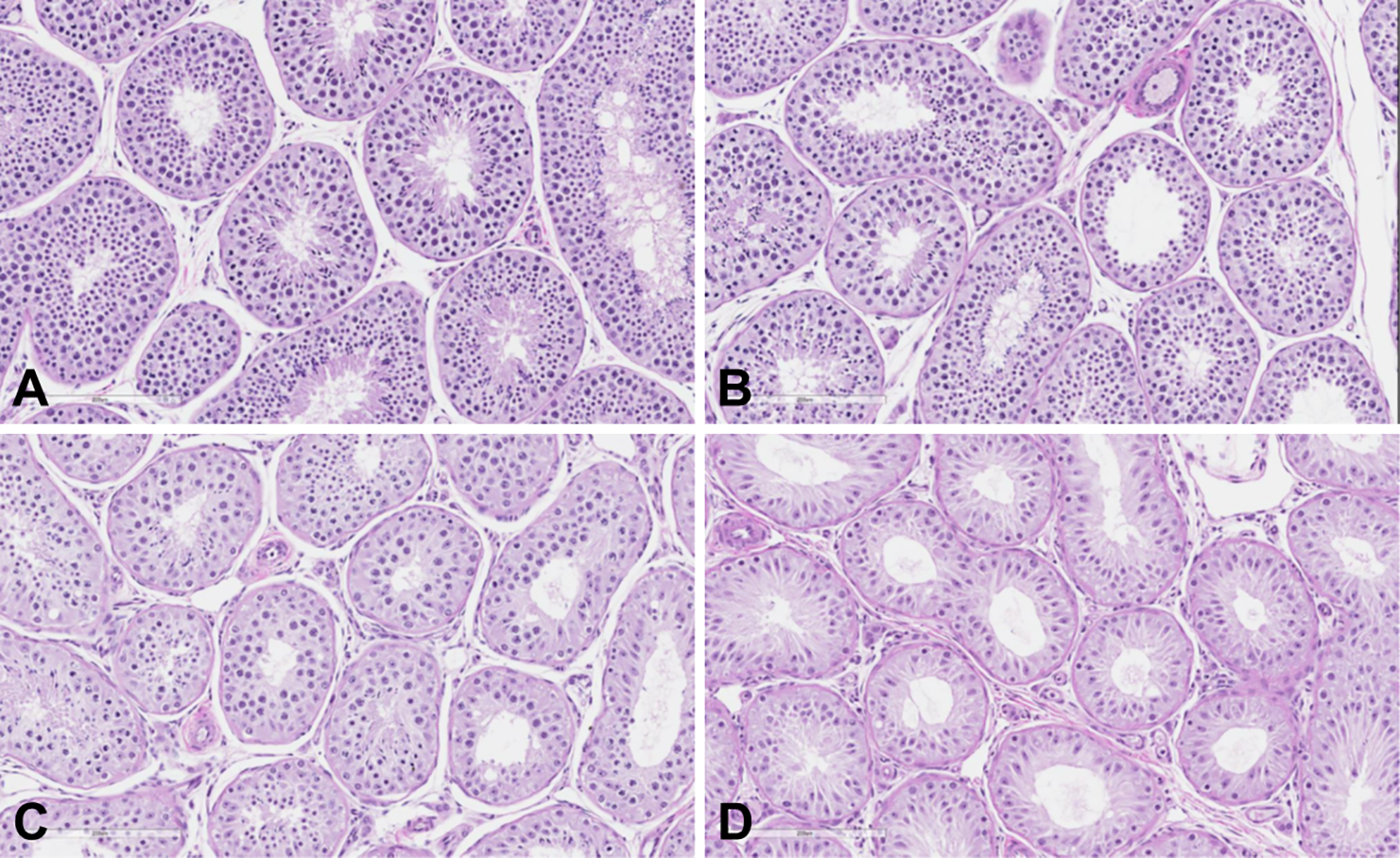
Representative photomicrographs of normal control cynomolgus monkey testis (A) and testes from cynomolgus monkeys administered AMG2519493 in the 3-month toxicology study (B-D). Images demonstrate a dose-dependent decrease in germs cells: minimal decrease at 1 mg/kg (B), mild decrease at 8 mg/kg (C), and moderate decrease at 100 mg/kg (D). Staining: hematoxylin and eosin, original objective ×20.
Interspecies AMG2519493 Systemic Exposure Comparison
AMG2519493 systemic exposures (plasma drug concentration) associated with the testicular changes were compared between the rat and cynomolgus monkey and with human in vitro IC50 for PI3K δ and β (Figure 6). In the rat, the testicular change was associated only at the supra pharmacologic dose at which the selectivity of AMG2519493 for PI3K δ over PI3K β was diminished, and not only the PI3K δ but also β isoforms were also presumably inhibited. In contrast, in the cynomolgus monkey, the testicular change was observed at a very low therapeutic dose (≥1 mg/kg), corresponding ostensibly with δ-selective exposures. No testicular changes were observed at the subtherapeutic dose (0.1 mg/kg), at which plasma AMG2519493 concentration was higher than δ IC50 only at limited time points (1 and 2 hours postdose, only).
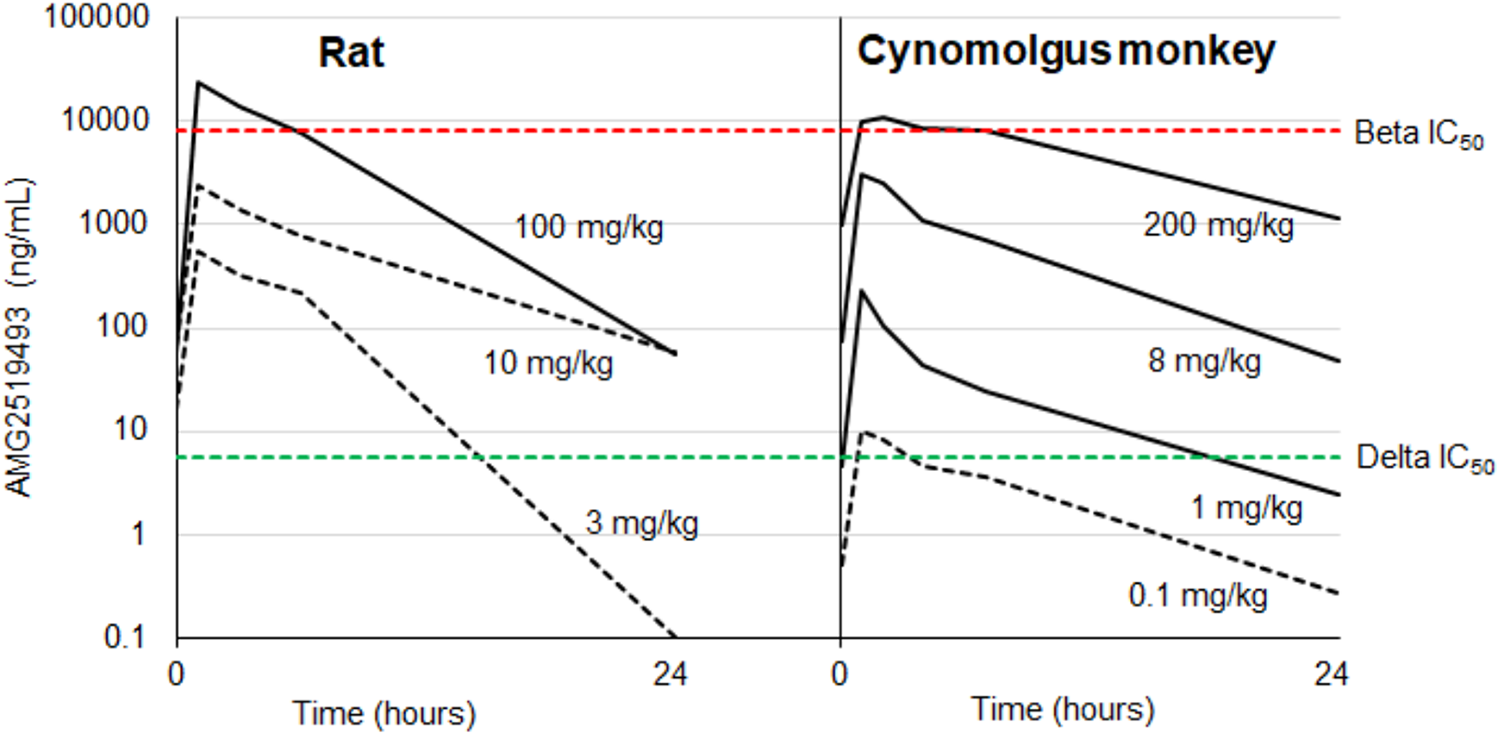
Plasma AMG2519493 concentration time course in the 1-month studies in the rat and cynomolgus monkey AMG2519493 was administered daily by oral gavage to the rat and cynomolgus monkey at dose levels of 0 (vehicle control), 3, 10, or 100 mg/kg and 0 (vehicle control), 0.1, 1, 8, or 200 mg/kg for 1 month, respectively. Time course of plasma AMG2519493 concentrations (ng/mL) after the last dose (dot line: testicular change not present; solid line: testicular change present) is compared between the rat and cynomolgus monkey and with human in vitro IC50 for PI3K δ (green dotted line) and β (red dotted line).
In Vitro Functional Assessment of PI3K Isoforms (β and δ) in Rodent Spermatogonia
In order to further evaluate PI3K isoform-specific effects on the testis, PI3K signaling was evaluated in isolated rat spermatogonia in vitro with PI3K inhibitors of various potencies on β and δ isoforms. Stem cell factor is a Sertoli cell–produced paracrine factor that binds to c-Kit expressed in differentiating spermatogonia. 21 C-Kit stimulation leads to activation of the downstream PI3K/AKT signaling pathway, to promote spermatogenesis. 22 Protein kinase B phosphorylation (pAKT) was therefore used as a surrogate of PI3K activity, in response to SCF.
Concentration response inhibition curves for PI3K isoform-specific assays (β or δ) and the pAKT rat spermatogonia assay were overlaid for AMG2519493. AMG2519493 was approximately 1,000 fold more potent in the δ-specific assay (IC50: 0.0022 µM) compared to the β-specific assay (IC50: 2.09 µM; Figure 7). AMG2519493 had low potency for inhibition of pAKT in rat spermatogonia (IC50: 1.15 µM). AMG2519493 was also examined in mouse spermatogonia with a similar pAKT IC50 value (1.1 µM) seen compared to the rat (data not shown).
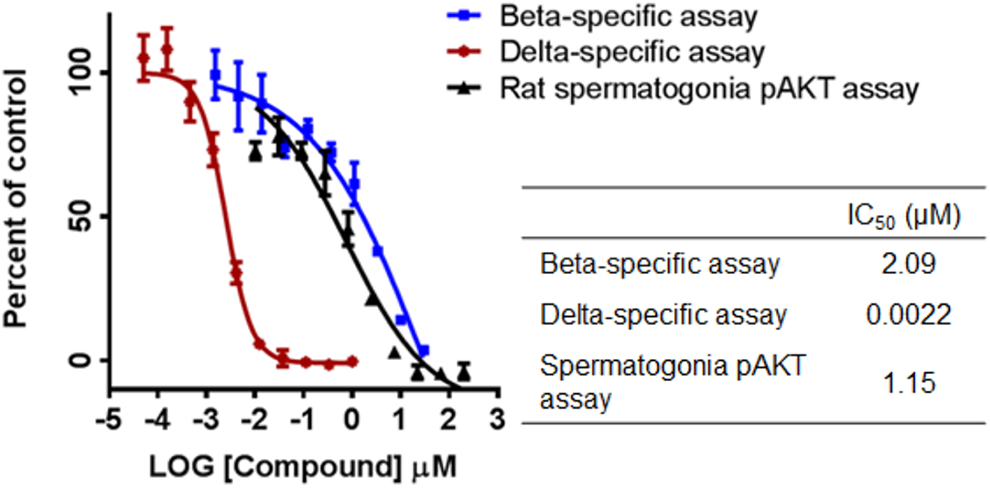
Concentration response curves in PI3K β-specific, PI3K δ-specific, and rat spermatogonia pAKT assays for AMG2519493. pAKT indicates protein kinase B phosphorylation. PI3K, phosphatidylinositol 3-kinase.
The results from a correlation analysis between the IC50 for PI3K β or δ-specific assays and the IC50 for pAKT in rat spermatogonia for 16 PI3K inhibitors including AMG2519493 and 15 other PI3K inhibitors with different PI3K isoform specificities are presented in Figure 8. A significant correlation (R2 = 0.7217) between the rat spermatogonia pAKT IC50 and the PI3K β-specific assay IC50 was observed. In contrast, no correlation was observed between the rat spermatogonia pAKT IC50 and the PI3K δ-specific assay IC50. Taken together, the data indicate a role for the β isoform in SCF/c-Kit/PI3K/AKT signaling in rat spermatogonia.
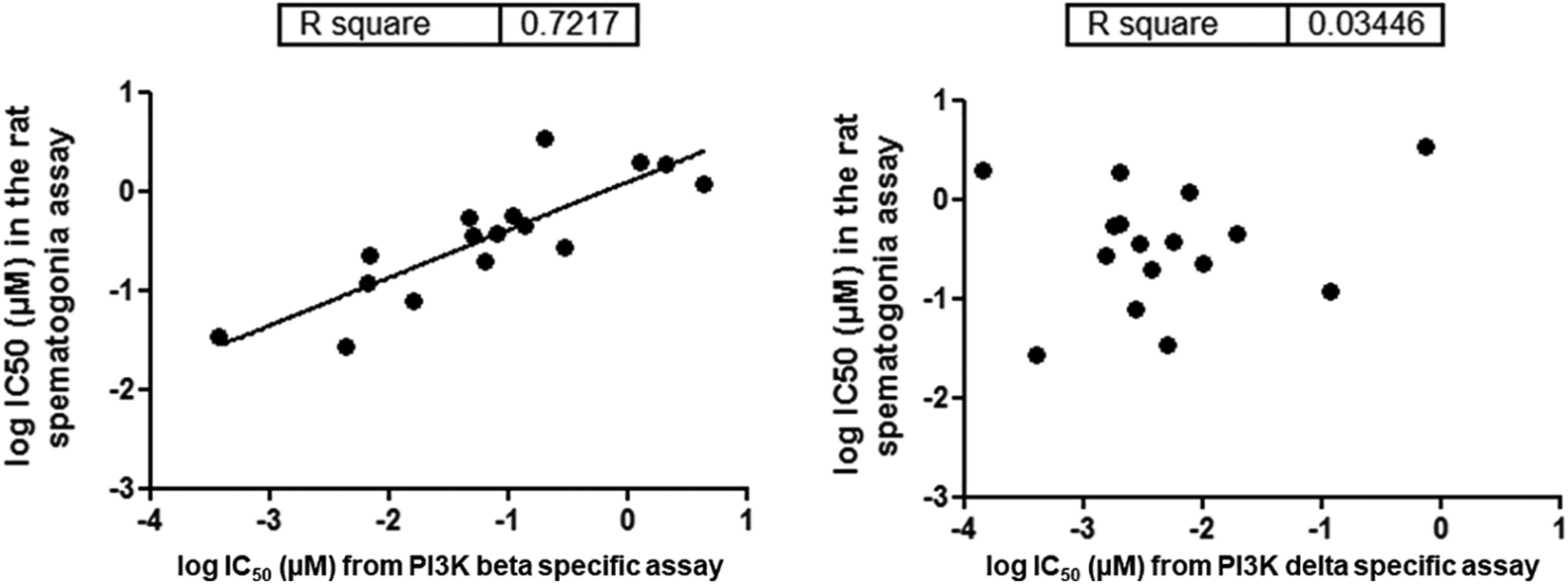
Correlation analysis between the IC50 for PI3K β-specific assay versus the IC50 of the rat spermatogonia pAKT assay (left) and between the IC50 for the PI3K δ-specific assay versus the IC50 of the rat spermatogonia pAKT assay (right) Each dot represents a different compound with different PI3K isoform specificity. A significant correlation (R2 = 0.7217) between the rat spermatogonia pAKT IC50 and the PI3K β-specific assay IC50 was observed (left). In contrast, no correlation was observed between the rat spermatogonia pAKT IC50 and the PI3K δ-specific assay IC50 (right). pAKT indicates protein kinase B phosphorylation; PI3K, phosphatidylinositol 3-kinase.
Discussion
Light microscopic changes in the ovary (rat) and testis (rat and cynomolgus monkey) were observed in the AMG2519493 toxicology studies. Interestingly, the ovary and testes changes in the rat were observed only at the highest dose evaluated corresponding with exposures far greater than the δ isoform-specific exposure, suggesting an off-target effect. In contrast, the testicular changes in the cynomolgus monkey was observed at very low doses, suggesting an on-target effect. The metabolite profiles of AMG2519493 is similar across species (rat-cynomolgus monkey-human) based on an in vitro assessment using hepatocytes (data not shown). It is unlikely, therefore, that a species-specific metabolite contributed to the difference in sensitivity to the ovarian or testicular changes between the rat and cynomolgus monkey. In contrast to PI3K δ primarily expressed only in leukocytes, PI3K α and β have broad tissue distribution.1,23 Therefore, the ovarian or testicular changes observed in the rat at only suprapharmacologic exposure may be attributed to nonspecific inhibition of other PI3K isoforms.
In the rat ovary, mild bilateral hemorrhagic CL cysts and/or an increased incidence of minimal to mild CL cysts without hemorrhage were observed at the highest dose administered (100 mg/kg). The finding of hemorrhagic cystic CL was not observed at the end of the recovery period; therefore, it was considered reversible. Observation of an increased incidence of cystic CL (without hemorrhage) was not considered adverse based on the following characteristics: similarity to the spontaneous change 24 ; transient presentation; lack of progression; and absence of any other light microscopic changes in the ovaries. In addition, there were no changes in the estrus cycle or other reproductive parameters in a female rat fertility study including no macro- or microscopic changes up to the highest dose of 100 mg/kg tested (data not shown), indicating intact and functioning hypothalamic-pituitary-ovarian hormonal axis. 25 There were no light microscopic changes in the ovary from the cynomolgus monkey studies up to the same administration duration and similar exposure levels to the rat.
While the pathogenesis of these rodent ovarian changes is unknown, the changes are not consistent with the described primordial follicle loss associated with suppression of PI3K activity in the ovary.26,27 The phosphatase and tensin homolog (PTEN)/PI3K/AKT/forkhead box O3 (FOXO3) signaling pathway plays an important role in the regulation of dormancy and initial follicular activation in ovary. 28 Hypoactivation or negative regulation of the PTEN/PI3K/AKT/FOXO3 pathway may cause retardation of follicle activation and excessive primordial follicle atresia. On the contrary, inhibition of PTEN and activation of PI3K may cause activation of dormant follicles since PTEN is a negative regulator of the PI3K/AKT signaling pathway. Although there are few reports on PI3K isoforms involved in ovarian follicle development, Yan et al recently reported that cell division cycle 42 governs the process of primordial follicle activation by binding to PI3K β (p110 β) and regulating the expression levels of PTEN in oocytes. 29 In contrast to PI3K α or β having broad tissue distribution, PI3K δ has been previously reported to be selectively expressed in leukocytes.1,30 However, Li et al recently reported that PI3K δ was not only highly expressed in immune organs but also highly expressed in mouse oocytes and granulosa cells (GC). 11 Pik3cd-null mice were viable and had normal body size. However, the females were subfertile, exhibited fewer growing follicles and more atretic antral follicles in the ovary, and responded poorly to exogenous gonadotropins compared with wild type animals. Ovary transplantation showed that Pik3cd-null ovaries responded poorly to follicle-stimulating hormone (FSH) stimulation in vitro; this confirmed that the follicle growth defect was intrinsically ovarian. In addition, estradiol (E2)-stimulated follicle growth and GC proliferation in preantral follicles was impaired in Pik3cd-null ovaries. FSH and E2 substantially activated the PI3K/Akt pathway in GCs of control mice but not in those of Pik3cd-null mice. Administration of a PI3K δ-specific inhibitor, SW30, to mice on postnatal day 21 strongly inhibited the formation of antral follicles and ovulation. On the other hand, primordial follicle activation and oocyte meiotic maturation were not affected by Pik3cd knockout, indicating PI3K δ is a key isoform of the PI3K pathway for follicle growth in ovarian GCs; however, it is not required for primordial follicle activation and oocyte development. Such ovarian phenotypes of Pik3cd-null mice, including small ovary size and poor response to gonadotropins, returned to normal with increasing age. mRNA levels of PIk3ca in the ovary were significantly increased in Pik3cd-null mice, and the increase in PIk3ca mRNA was much more significant in adult ovaries at 12 weeks of age compared to 3 weeks of age, suggesting that other PI3K catalytic isoforms such as PI3K α may play compensatory roles in ovarian GCs and oocytes with increasing age. Our studies with AMG2519493 used young adult rats (7- to 8-weeks-old at initiation of dosing) and adult monkeys (4- to 8-years-old). These adult animals may be mature enough to compensate for the effect of PI3K δ-specific inhibition and maintain normal ovarian functions. Therefore, the ovarian findings observed in the rat with AMG2519493 only at suprapharmacologic exposure were considered due to nonspecific inhibition of other PI3K isoforms. There are no reports on ovarian findings in the toxicology studies with another PI3K δ-specific inhibitor, idelalisib, in the rat (1- to 6-month repeat-dose studies, 7 weeks of age at study initiation) or dog (1- to 9-month repeat-dose studies, 5-8 months of age at study initiation).31,32 To understand the potential effects of PI3K δ-specific inhibition on the ovary in children or adolescents, further studies using juvenile animals may be warranted.11,33
The histopathologic characteristics of the AMG2519493-induced testicular changes (ie, primary target is considered late stage spermatogonia) are consistent with spermatogenesis maturation arrest via inhibition of the SCF/c Kit/PI3K/AKT signaling pathway.14,20,34 C-Kit was not expressed on the undifferentiated SSC; however, once SSC starts differentiation, c-Kit is expressed on differentiating spermatogonia. 35 Spermatogenesis maturation arrest has been reported with the c-Kit inhibitor, imatinib, which impaired spermatogonial differentiation, but did not affect SSC renewal. 34 In mice, mutations of the c-Kit gene (with its PI3K docking site mutated) notably inhibit spermatogenesis occurring at the level of the spermatogonia. 36 Since PI3K signaling is known to be downstream of c-Kit pathway activation,22,37 the testicular change may be attributed to AMG2519493-related inhibition of c-Kit receptor-mediated PI3K signaling. In rodents, Ciraolo et al reported that PI3K β isoform is primarily involved in c-Kit-mediated spermatogenesis. Phosphatidylinositol 3-kinase β deficient male mice are subfertile, while female mice are fully fertile. 14 In contrast, PI3K δ-deficient male mice show normal fertility.4,11 In the AMG2519493 rat toxicology studies, testicular changes were observed only at supra pharmacologic dose of AMG2519493, at which the selectivity of AMG2519493 for δ over β are diminished and the PI3K β isoform would also be inhibited (AMG2519493 δ selectivity over β: ×1,500). Our in vitro study using rat spermatogonia also demonstrated that pAKT inhibition in spermatogonia was clearly dependent on inhibition of PI3K β isoform but not PI3K δ. Thus, rat testicular changes may be attributed to AMG2519493 off-target inhibition of PI3K β at high doses.
It is unknown which PI3K isoforms are involved in SCF/c Kit/PI3K/AKT pathway-mediated spermatogenesis in nonrodents. Given the testicular changes observed at low and presumably δ-specific exposures in the monkey, it is plausible that PI3K δ may be directly involved in spermatogenesis in the monkey and potentially humans. The in vitro spermatogonia assay used in this study required a collection of rat and mouse spermatogonia from multiple infant animals, and similar experiments cannot be simply applied to monkeys or humans for humane reasons; therefore, there are no existing data. Notably in human mast cells, PI3K δ is a primary isoform for the Kit-mediated pathway.38,39 Based on the available data, it appears likely that the PI3K isoform involved in c-Kit signaling may differ between the rodent and monkey (and human) testis. Testicular effects of another PI3K δ-specific inhibitor, idelalisib, was reported in the rat and dog toxicology studies, and testicular effects were observed in the rat at relatively high doses. In contrast, testicular effects were observed in the dog at relatively low doses in support of our observations.31,32
Phosphatidylinositol 3-kinase is involved in several different pathways for other testicular functions (Table 1). Based on the light microscopic evaluation (ie, SSCs and Sertoli cells not adversely affected by AMG2519493, and no other tissue injury on other testicular compartments observed), it is unlikely that PI3K δ inhibition by AMG2519493 affects those functional pathways other than the SCF/c-Kit/PI3K/AKT pathway-mediated spermatogenesis differentiation (maturation). Therefore, other non-δ PI3K isoforms may be dominant for these other testicular functions.
Phosphatidylinositol 3-Kinase Roles in Testicular Function and Potential Involvement of AMG2519493.

Abbreviations: GDNF, glial cell line–derived neurotrophic factor; PI3K, phosphatidylinositol 3-kinase; PKB, protein kinase B; SCF, stem cell factor; SSCs, spermatogonial stem cell; RXFP1, relaxin family peptide receptor 1.
In conclusion, oral repeat-dose toxicology studies with a PI3K δ-specific inhibitor, AMG2519493, in the rat and cynomolgus monkey identified the ovary (rat only) and testis (both species) as key toxicology target organs. In the rat, hemorrhagic CL cysts and increased incidence of CL cysts without hemorrhage in the ovary and decreased seminiferous germ cells in the testis were observed only at the supra pharmacologic exposure, suggestive of off-target effects. In contrast, decreased germ cells in the cynomolgus monkey seminiferous tubules were observed at a very low dose associated with PI3K δ-specific inhibition, suggestive of on-target effects. The characteristics of the testicular changes were consistent with spermatogenesis maturation arrest. Significantly different sensitivity to the testicular effects between the 2 rat and cynomolgus monkey suggests that different PI3K isotypes may be involved in spermatogenesis maturation (ie, β in rodents and δ in monkey and potentially human).
Supplemental Material
Supplemental Material, sj-docx-1-ijt-10.1177_10915818211008175 - Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey
Supplemental Material, sj-docx-1-ijt-10.1177_10915818211008175 for Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey by Katsu Ishida, Jonathan A. Werner, Marc Lafleur, John Wisler, Sharon Wannberg, Jackson Kalanzi, Jeanine L. Bussiere and Thomas M. Monticello in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-docx-2-ijt-10.1177_10915818211008175 - Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey
Supplemental Material, sj-docx-2-ijt-10.1177_10915818211008175 for Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey by Katsu Ishida, Jonathan A. Werner, Marc Lafleur, John Wisler, Sharon Wannberg, Jackson Kalanzi, Jeanine L. Bussiere and Thomas M. Monticello in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-docx-3-ijt-10.1177_10915818211008175 - Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey
Supplemental Material, sj-docx-3-ijt-10.1177_10915818211008175 for Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey by Katsu Ishida, Jonathan A. Werner, Marc Lafleur, John Wisler, Sharon Wannberg, Jackson Kalanzi, Jeanine L. Bussiere and Thomas M. Monticello in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-docx-4-ijt-10.1177_10915818211008175 - Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey
Supplemental Material, sj-docx-4-ijt-10.1177_10915818211008175 for Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey by Katsu Ishida, Jonathan A. Werner, Marc Lafleur, John Wisler, Sharon Wannberg, Jackson Kalanzi, Jeanine L. Bussiere and Thomas M. Monticello in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-docx-5-ijt-10.1177_10915818211008175 - Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey
Supplemental Material, sj-docx-5-ijt-10.1177_10915818211008175 for Phosphatidylinositol 3-Kinase δ-Specific Inhibitor-Induced Changes in the Ovary and Testis in the Sprague Dawley Rat and Cynomolgus Monkey by Katsu Ishida, Jonathan A. Werner, Marc Lafleur, John Wisler, Sharon Wannberg, Jackson Kalanzi, Jeanine L. Bussiere and Thomas M. Monticello in International Journal of Toxicology
Footnotes
Acknowledgments
The authors thank the following toxicology study personnel: Jay Albretsen (study director, 1 month rat study), Mingyi Trimble (study director, 1-month cynomolgus monkey study), Shana Dalton (study director, 3-month rat and cynomolgus monkey studies).
Author Contributions
Katsu Ishida contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Jonathan A. Werner contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Marc Lafleur contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; John Wisler contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Sharon Wannberg contributed to conception and design, contributed to analysis, and critically revised manuscript; Jackson Kalanzi contributed to analysis and interpretation and critically revised manuscript; Jeanine L. Bussiere contributed to conception and design, contributed to analysis and interpretation and critically revised manuscript; Thomas M. Monticello contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are or were employees of and own stock in Amgen Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Amgen funded this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
