Abstract
Sustained drug delivery formulations are developed to reduce dose frequency while maintaining efficacy of intravitreal (ITV) administered therapeutics. Available safety data for components novel to the eye’s posterior segment may be limited, requiring preclinical assessments to identify potential toxicities. We evaluated the in vivo and in vitro safety of two solvents, acetyl triethyl citrate (ATEC) and benzyl benzoate (BB), as novel sustained delivery formulations for ITV administration. In vivo tolerability was assessed following ITV administration of ATEC and BB to rabbits and cynomolgus monkeys. In rabbits, ITV solvent administration resulted in moderate to severe retinal toxicity characterized by focal retinal necrosis and/or degeneration, sometimes accompanied by inflammation, with a clear association between the physical presence of the solvent and areas of retinal damage. In contrast, solvent administration in monkeys appeared well tolerated, producing no histologic abnormalities. Toxicity in primary human retinal pigment epithelial cells, characterized by cellular toxicity and mitochondrial injury, corroborated the retinal toxicity in rabbits. In conclusion, ITV solvent depots of ATEC or BB result in chemical and focal retinal toxicity in rabbits, but not monkeys. Additional investigation is needed to demonstrate a sufficient margin of safety prior to use of ATEC or BB in ITV drug products.
Introduction
Ocular drug developers are pursuing sustained drug delivery systems utilizing novel excipients and platforms to maintain therapeutic drug concentrations in the posterior segment for retinal disease treatments. 1 Intravitreal (ITV) tolerability and toxicity data are often limited or not available for components of novel formulations, which might relate to the limited success achieved within the field of long-acting delivery of therapeutics for retinal diseases. When available data are insufficient, individual components of candidate drug formulations require thorough safety evaluation via the most relevant route of administration, that is, ITV, in nonclinical studies to enable novel formulations and identify potential retinal toxicities. 2
Acetyl triethyl citrate (ATEC) and benzyl benzoate (BB) are low molecular weight plasticizers that aid in the dissolution of nonaqueous components of injectable sustained-release formulations. These organic, biocompatible solvents are novel in posterior segment use and nonclinical ITV data have not been published to date. 3,4 In vivo studies are routinely used to enable candidate drug product formulations containing novel excipients by identifying potential clinical toxicities and establishing safety limits where no existing ITV data are available. 2 Rabbit is a commonly utilized species in the initial step of identifying drug-induced ITV toxicities in vivo as the lowest order species, with similar globe size to cynomolgus monkeys and an adequate vitreous volume for ITV administration. 5,6 Furthermore, strain selection has an important role in proper toxicity evaluation. Although the use of albino rabbit strains, such as New Zealand White (NZW), is common, potential melanin-binding pharmaceuticals should be evaluated in pigmented strains, such as the Dutch-Belted (DB) rabbit, in order to adequately identify toxicity risks associated with potential drug accumulation and altered drug distribution in the eye. 7 Regardless of strain, the rabbit eye has several significant anatomical and physiological differences from the human eye, including vitreous viscosity and volume, retinal vasculature, and choroidal flow rate, which can challenge data translatability. 6,8 Translatability improves when utilizing monkeys, which are the most closely related nonclinical species to humans. Because of genetic similarity, monkeys are often the most pharmacologically relevant model to predict human toxicity, and monkeys possess similar anatomy and physiology to humans, with common ocular structures such as a fovea and a macula. 8,9
In the present work, we describe in vivo and in vitro studies designed to evaluate toxicity associated with ATEC and BB, as potential drug delivery depots used to extend the release of intraocular therapeutics following single ITV administration. Rabbits, both DB and NZW strains, and cynomolgus monkeys were utilized to assess safety and provide a weight of evidence to predict toxicity in humans. In vivo toxicity studies carry high financial costs, ethical concerns, and controlled throughput, which limit screening efforts of novel formulation components. 10,11 With less stringent limitations, assessments in vitro may provide a preliminary identification of potential safety liabilities. 12 Therefore, ATEC and BB were assessed in vitro using primary human retinal pigment epithelium (RPE) cells and human hepatocytes (HepG2) to further characterize the ITV toxicity profile and provide insight into potential mechanisms of toxicity.
Methods
Materials
Both ATEC (CAS No. 77-89-4) and BB (CAS No. 120-51-4) were obtained through ICON Biosciences for in vivo use, and were ordered from Sigma-Aldrich for in vitro use. All chemicals were of analytical grade and used as received without further purification.
Animal Studies
Tolerability of ATEC and BB was assessed in DB rabbits and cynomolgus monkeys. An additional study was conducted with ATEC in NZW rabbits only to assess the effects of vitreous placement as well as strain-specific differences on solvent tolerability (Table 1). All procedures and protocols were conducted in accordance with the standard operating procedures approved by the Institutional Animal Care and Use Committee of the testing facility where the study was conducted and adhered to the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research.
Summary of In Vivo Study Designs.a

Abbreviations: ATEC, acetyl triethyl citrate; BB, benzyl benzoate; DB, Dutch-Belted; IOP, intraocular pressure; OEs, ophthalmic examinations; ITV, intravitreal.
a On day 1, ATEC or BB was administered via ITV injection to rabbit and monkey eyes. Ophthalmic examinations were performed prior to dosing (for baseline reference) and immediately following dose administration. Intraocular pressure was measured roughly concurrently with OEs. Necropsy was performed at week 4.
All animals were kept under controlled conditions in individual cages, with food and water ad libitum. Female DB rabbits (∼9 months, 2.2-2.8 kg, Myrtle Rabbitry), male NZW rabbits (∼3 months, 2.3-2.8 kg, Charles River), and male cynomolgus monkeys (Macaca fascicularis, 2-3 years old, 2.3-3.9 kg, Covance Research Products, Inc) of Chinese origin were utilized for in vivo toxicity assessment. Sexes of each species were irrelevant to the tolerability assessment and chosen based on operational convenience. A topical anesthetic was applied to the ocular surface followed by aseptic preparation of the eye for dosing. Acetyl triethyl citrate and BB were each administered bilaterally to 3 DB rabbits and 3 monkeys (6 rabbits and 6 monkeys total). Dutch-belted rabbits and monkeys received 30- and 50 μL, respectively, via a single ITV injection to the inferior vitreous followed by a topical antibiotic. Additionally, ATEC was administered bilaterally to the inferior or superior vitreous in 3 NZW rabbits (6 rabbits total). New Zealand white rabbits received 50 µL via a single ITV injection, followed by a topical antibiotic. The day of solvent administration was designated as day 1, and all animals were observed for a 4-week observation period. Control data were obtained from same-study naive animals that received similar volumes of normal saline ITV or from internal historical control data that are not detailed here.
Clinical observations (CL), intraocular pressure (IOP) measurements, and ophthalmic examinations (OE) were performed in each study as outlined in Table 1. Clinical observations were performed regularly to ensure animal welfare and assess clinical changes related to the solvent. Intraocular pressure was measured with an applanation tonometer prior to solvent administration (for baseline reference) and at intervals roughly concurrent with OEs. Ophthalmic examinations were performed by a veterinary ophthalmologist and consisted of slit-lamp biomicroscopy examination of the adnexa, anterior segment, aqueous humor, and vitreous humor and indirect ophthalmoscope examination of the fundus. All eyes were examined prior to injection and at regular intervals during the observation period. Digital fundus photography was performed on DB rabbits and monkeys only (Table 1). A Topcon 50EX fundus camera was used to acquire digital fundus images to include the optic nerve centered, inferior to the optic nerve, inferior nasal to the optic nerve, and inferior temporal to the optic nerve. At week 4, 28 to 30 days after solvent administration, animals were euthanized in accordance with the standard operating procedures at each facility.
Histopathology
Immediately following euthanasia, eyes with bulbar conjunctivae and optic nerves were removed, marked at the 12 o’clock position for orientation and fixed in modified Davidson’s for 48 to 96 hours before transfer to 10% neutral-buffered formalin. Rabbit eyes were trimmed in a sagittal plane of section (superio–inferior), with a cut on either side of the optic disc to divide the eye into a central segment (containing the optic disc), temporal, and nasal calottes that were embedded in paraffin. Three sections, at least 100 microns apart, were collected from each block. Monkey eyes were sectioned horizontally, across the middle of the eye, with a cut on either side of the optic disc to capture both the macula and optic disc in a single section. The superior and inferior calottes were then bisected in the midline, along a sagittal plane, to produce nasal and temporal segments of both inferior and superior calottes, a total of 5 blocks. After processing and staining with hematoxylin and eosin, the slides were examined microscopically by a board-certified veterinary pathologist.
In Vitro Toxicity Assessments
Cell culture
Primary human RPEs (Lonza) were first expanded in RtEGM Retinal Pigment Epithelial Growth Medium BulletKit (Lonza) and frozen down in cryotubes using media supplemented with 10% dimethyl sulfoxide (DMSO). Before the start of the experiments, RPEs were plated in black-walled, collagen-coated 384 well plates (Corning) at 125,000 cells/cm2 using the same media for cell viability or 200,000 cells/cm2 for mitochondrial stress assays. Retinal pigment epithelium cells were allowed to mature for 3 days in culture (37 °C, 5% CO2) prior to initiation of the treatments. Human hepatocytes (ATCC HB8065), an established cell line to investigate drug-related toxicities, were used as a control to identify potential tissue-specific differences. Human hepatocytes were plated at 140,000 cells/cm2 in complete Dulbecco’s modified Eagle medium (10% serum, 1% penicillin/streptomycin, 1% L-glutamine) for cell viability assays or 235,000 cells/cm2 for mitochondrial stress assays, 24 hours prior to treatment. Human hepatocytes were incubated overnight (37 °C, 5% CO2) and dosed the following day.
Solvent and control compound dosing
Acetyl triethyl citrate and BB were each dosed in RPEs and HepG2s and treated for 48 hours at 0.0005, 0.005, 0.05, 0.5, 5, 50, and 500 mM. The dose range was chosen based on a calculated ITV concentration of 100 mM (based on a 2 mL volume of vitreous in a monkey eye). 5 Solvent dilutions were made in appropriate cell culture media. At 500 mM, BB was insoluble and readily precipitated out of solution, therefore BB was not tested at this concentration. Carbonilcyanide p-triflouromethoxyphenylhydrazone (FCCP; Sigma-Aldrich) was used as a positive control for mitochondrial stress, diluted in DMSO, and dosed at 50, 10, and 1 µM with a final culture DMSO concentration of 1%. Histidine buffer (10 mM histidine HCl, 10% α,α-trehalose dihydrate, 0.01% polysorbate 20, pH 5.5) and methionine (Sigma-Aldrich) were diluted in tissue culture grade water (Life Technologies) to 100, 10, and 1, and used as negative controls for cytotoxicity. Selection of negative controls was based on internal studies showing intraocular tolerability in nonclinical studies (data not shown). Technical replicates were used for all treatments and controls (n = 3).
IncuCyte live-cell imaging
Confluence of RPE cells was measured using a microscopy-based label-free, noninvasive assay by IncuCyte Live-Cell Imaging Systems (Essen Bioscience). Retinal pigment epithelial cells (1000 cells/well) were seeded overnight on a 96-well plate, treated, and placed in an XL-3 incubation chamber maintained at 37 °C, where the cells were photographed using a 10× objective. Images were collected every 8 hours over the course of 40 hours. Confluence was calculated using IncuCyte 2011 software, which provides real-time cellular confluence data based on segmentation of high-definition phase-contrast images. 13
Cytotoxicity assessment
We assessed overall cell viability as a function of ATP production using the standard CellTiter-Glo assay (Promega) following manufacturer protocol. Cell viability was determined based on the percentage of luminescent signal for each treatment, as compared to its specific vehicle control. 14 Luminescence was determined on an EnVision Multiplate Reader (PerkinElmer). Inhibition curves and IC50 estimates were generated by nonlinear regression of log-transformed inhibitor concentrations (4-point serial dilutions including vehicle; GraphPad Prism, GraphPad Software).
Mitochondrial stress
Oxygen consumption rate (OCR) was measured on a Seahorse XF96 Analyzer at 37 °C (Agilent) as described previously. 15 Retinal pigment epithelial cells were seeded in a Seahorse XF96 Analyzer plate. After 48 hours, cells were treated with 1 dose of ATEC or BB (5 mM); control cells were treated with histidine buffer. The following steps were all performed as per manufacturer recommendations, 2 hours after treatment. The treated plate was loaded into a Seahorse XF96 Analyzer. Three baseline OCR measurements were taken at 6-minute intervals, and then separate injections of 1.5 mM oligomycin, 1 mM FCCP, and 0.5 mM rotenone/antimycin-A were administered consecutively by the instrument at 20-minute intervals. Three OCR measurements were taken at 6-minute intervals following each administration of oligomycin, FCCP, and rotenone/antimycin-A, which are well known modulators of ATP synthase, mitochondrial membrane potential, and complexes I and III, respectively. 15
Results
In-Life Assessments
Clinical observations, IOP, OE, and fundus digital photography were conducted to assess tolerability for 4 weeks following ITV administration of ATEC and BB. No significant clinical or IOP changes related to ATEC or BB were noted in either rabbits or monkeys. In both rabbits and monkeys, OE immediately following ATEC administration identified the solvent as a transparent focal or spherical depot in the inferior or superior vitreous (depending on location of dosing) (Figure 1A and D).
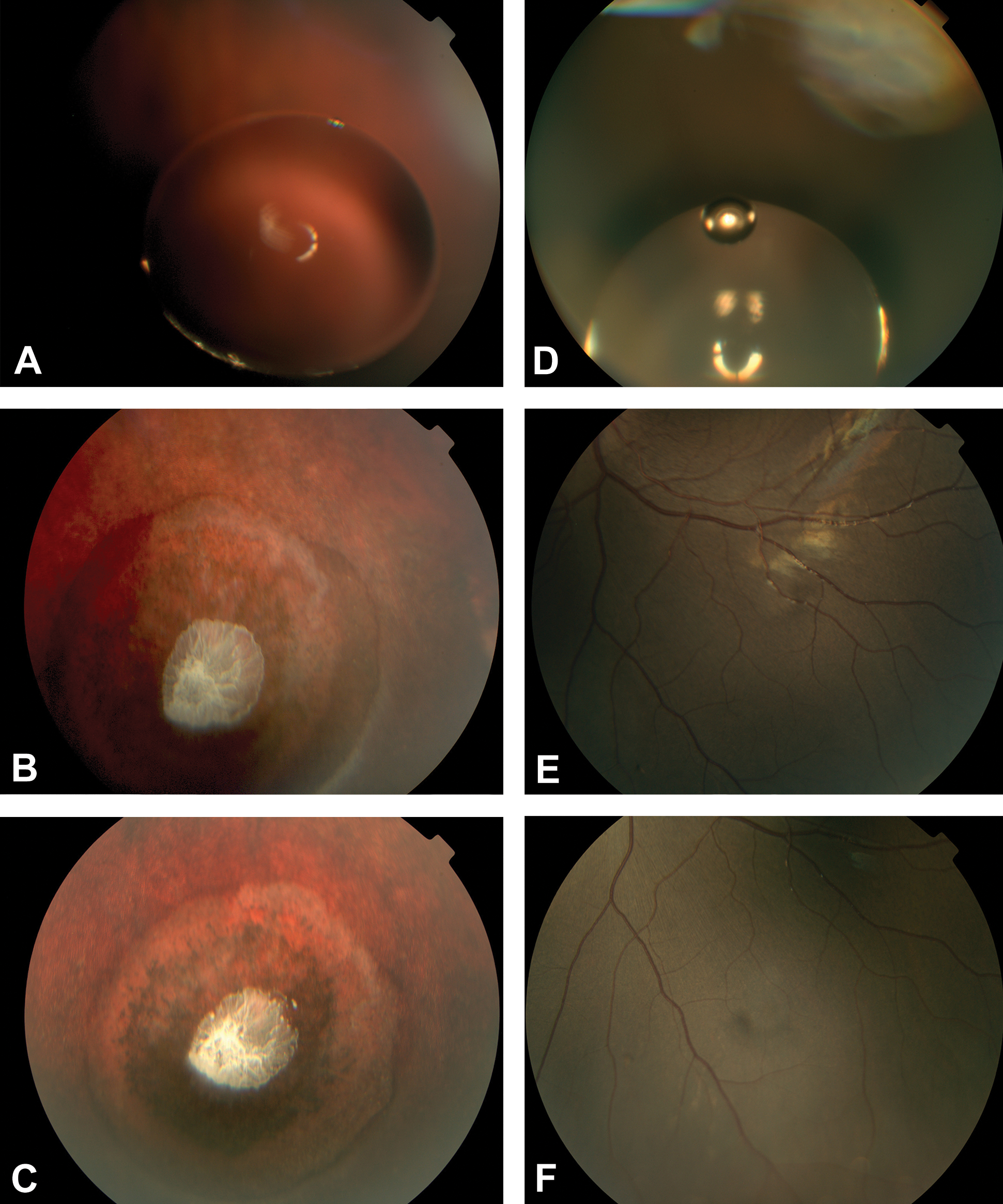
Fundus images following ITV administration of ATEC. In DB rabbits, the ATEC depot appeared as a transparent spherical mass on day 1 (A) and was no longer visible after day 5. At weeks 2 and 4 (B and C), a white deposit at the center of marked circular RPE clumping was present in the retina underlying the location of the depot noted on day 1. In monkeys, the ATEC depot appeared as one or more spherical masses on day 1 (D). As the study progressed, the depot was no longer within the photographic field of view and not captured in fundus photography (E and F). However, ATEC was still visible during OE for most eyes throughout the study with no remarkable findings at each interval. ATEC, acetyl triethyl citrate; DB, Dutch-Belted; OE, ophthalmic examination; ITV, intravitreal; RPE, retinal pigment epithelium.
Acetyl triethyl citrate elicited different responses in rabbits and monkeys. The DB and NZW rabbit strains responded similarly, with slightly greater severity and frequency of abnormal findings in DB rabbits. At week 1 OE, retinal whitening was seen immediately adjacent to the ATEC depot in all DB rabbit eyes, along with a mild inflammatory response characterized by trace to mild amounts of vitreous cells, while NZW rabbit eyes appeared normal. By week 2, OE and fundus images in DB rabbits could no longer visualize any ATEC, but retinal thinning and marked circular RPE clumping were present at the depot location noted on the day of dosing, with a white deposit at the center of each circle and retinal whitening at the periphery (Figure 1B). Furthermore, OE revealed trace aqueous flare, trace amounts of cells in the vitreous, and mild conjunctival hyperemia in these eyes. In NZW rabbits, mild to marked amounts of predominantly white vitreous cells were noted during OE, but retinal changes were not observed. The DB rabbits did not undergo a week 3 assessment, while NZW rabbits continued to show a mild to marked inflammatory response that was unchanged from week 2. At week 4, retinal thinning and RPE clumping remained visible in fundus images of all DB rabbit eyes (Figure 1C), with OEs revealing that trace to mild amounts of vitreous cells were present, and aqueous flare and conjunctival hyperemia were resolved. In NZW rabbits, a faint refractile material, presumed to be residual solvent, was visible in the anterior vitreous and on the vitreous floor with reduced amounts of vitreous cells present. No significant difference was observed between inferior versus superior vitreous placement of the ATEC depot.
In contrast, ATEC was visible in monkey eyes during OE throughout the study, starting as a depot of one or more spherical masses and subjectively decreasing in size as the study progressed. The ATEC depot was captured via fundus photography on day 1 (Figure 1D), but no longer within the field of view for any monkeys at subsequent time points (Figure 1E and F). Trace amounts of predominantly brown cells were observed in the vitreous from day 5 to week 4, which likely represented pigmented cells liberated during the injection procedure; no other findings were observed.
In DB rabbits, BB was visible at all time points, changing from a transparent depot, to gray to white and subjectively decreasing in size over the 4-week observation period (Figure 2A-C). At day 5 and week 1, all eyes had retinal whitening immediately adjacent to the BB depot, with mild to moderate conjunctival hyperemia. At week 2 OEs, all eyes had mild conjunctival hyperemia and anterior segment inflammation, characterized by trace to moderate aqueous flare and trace to mild amounts of cells in the anterior chamber. Furthermore, the pupil in one of 4 eyes incompletely dilated for examination. At week 4 OEs, anterior segment inflammation resolved, but moderate amounts of predominantly white cells were present in the vitreous of all 4 eyes along with retinal whitening immediately adjacent to the BB depot. At weeks 2 and 4, fundus photography revealed white clumps that often obscured the RPE, with evidence suggesting mild RPE clumping was present. In monkeys, BB appeared as a depot of one or more spherical masses throughout the study (Figure 2D-F). Trace to mild amounts of predominantly brown cells were present in the vitreous until week 1 and likely represented pigmented cells liberated during the injection procedure. At weeks 2 and 4, mild amounts of small, bright white refractile material were present in 1 eye; because of its refractile nature and varying particle size, this material likely represented residual solvent material and not inflammatory cells.
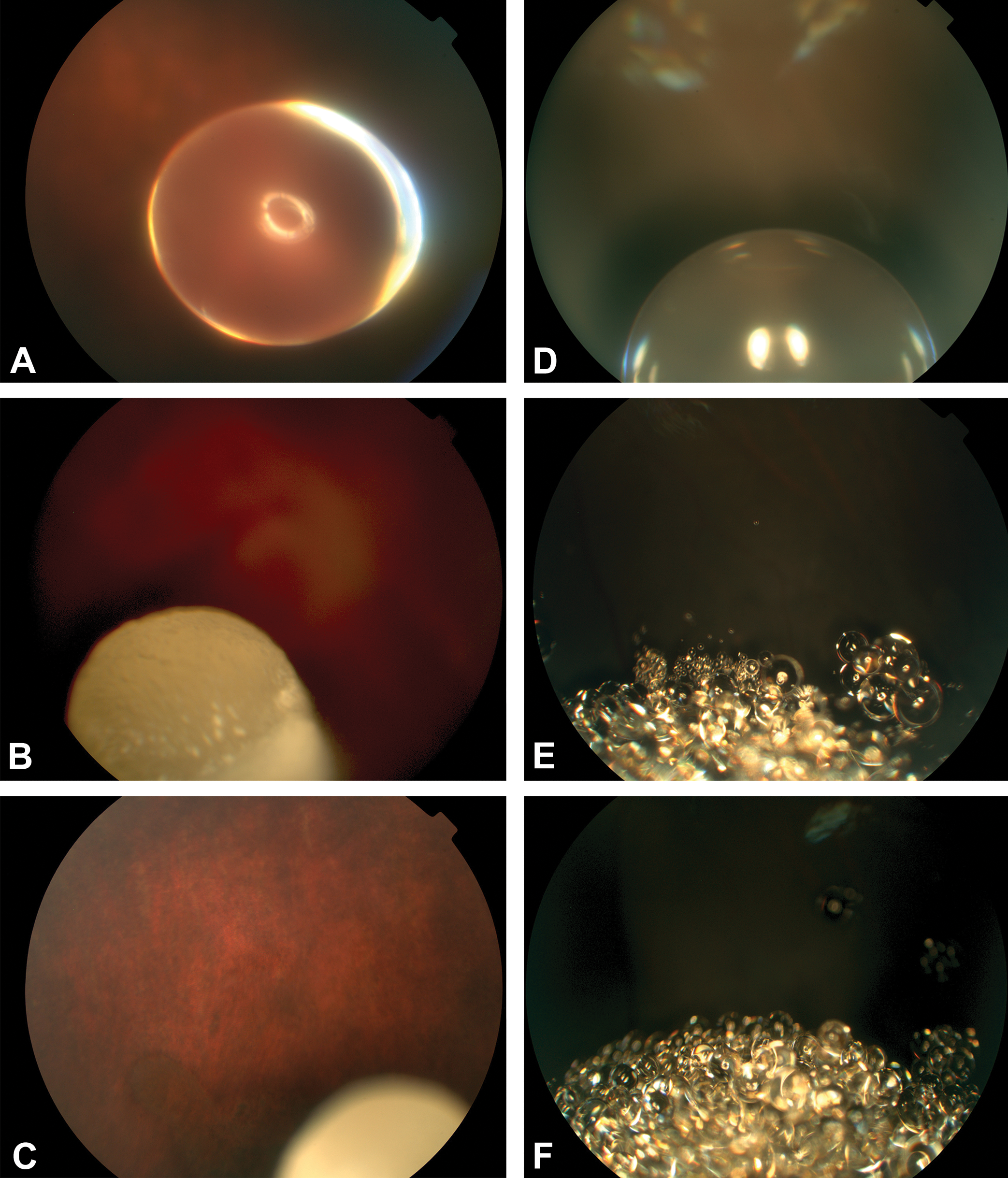
Fundus images following ITV administration of BB. In Dutch-Belted rabbits, the BB depot appeared as a transparent spherical mass on day 1 (A) and appeared increasingly opaque and subjectively smaller at weeks 2 and 4 (B and C). In monkeys, the BB depot appeared as one or more transparent spherical masses on day 1 (C). At weeks 2 and 4, the BB depot appeared as multiple smaller spherical masses localized to the inferior vitreous (E and F). BB indicates benzyl benzoate; ITV, intravitreal.
Histopathology
Intravitreal administration of ATEC resulted in focal to multifocal moderate to severe retinal degeneration in NZW and DB rabbits but not cynomolgus monkeys. There was no difference in the character or severity of lesions between NZW and DB rabbits. Retinal degeneration was characterized by disorganization and loss of retinal layers, with retinal folding. In some sections, foci of full thickness retinal necrosis were present, associated with hypertrophy and vacuolation of the remaining RPE and fibrosis. In some sections, ATEC was apparent as basophilic and/or refractile material associated with inflammatory debris, degenerate heterophils, and small numbers of multinucleated giant cells, consistent with a mild foreign body reaction (Figure 3C and D). Minimal degeneration in the optic nerve, considered secondary to the retinal changes, was also occasionally observed. Lesions progressed over the 4-week period and were considered irreversible.
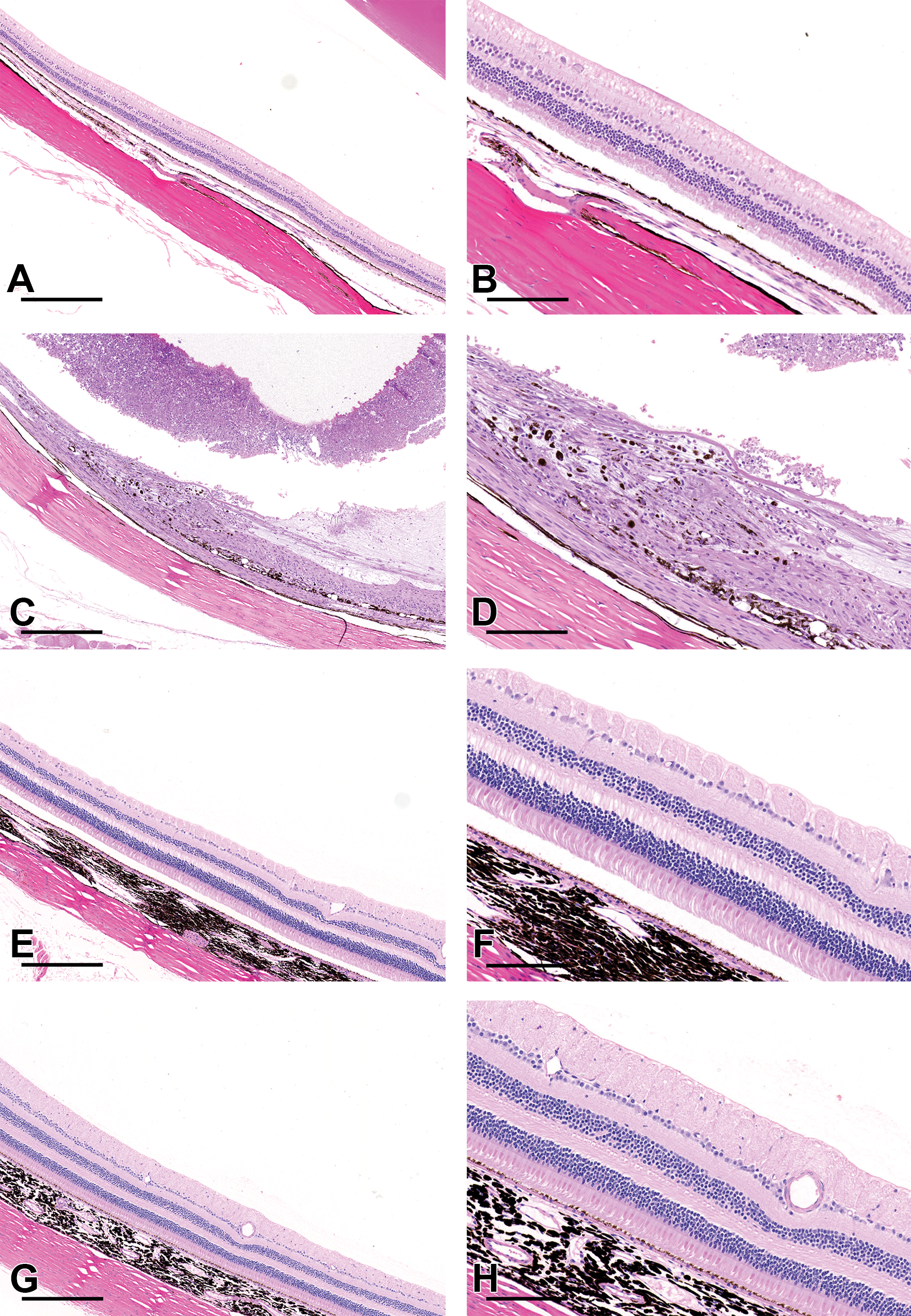
Effects of ITV ATEC in H&E stained tissues. Focal to multifocal degeneration, necrosis, and loss of the retina were observed in rabbits treated with ATEC, with hypertrophy of the RPE and retinal atrophy (C and D). Moderate amounts of inflammatory debris were observed in the vitreous (C). Eyes from cynomolgus monkeys treated with ATEC (G and H) were normal, with no significant difference from those treated with saline (E and F). A and B, Rabbit saline (control); C and D, rabbit ATEC; E and F, cynomolgus monkey saline (control); G and H, cynomolgus monkey ATEC (A, C, E, G scale bar 400 μm; B, D, F, and H scale bar 100 μm). ATEC indicates acetyl triethyl citrate; H&E, hemotoxylin and eosin; ITV, intravitreal.
Correlation with fundus photography indicated that lesions were limited to areas adjacent to the test article. No histological abnormalities were observed following ITV administration of ATEC to the eyes of cynomolgus monkeys (Figure 3G and H).
Species differences in the ITV tolerability of BB were also observed. In DB rabbits, there was mild to moderate focal retinal degeneration, with atrophy of the photoreceptor outer segments, disorganization of the inner and outer nuclear layers, and drop out of nuclei. In some regions, beneath solvent depots, proliferations of spindle cells covered the inner limiting membrane of the retina (epiretinal membranes). These findings were associated with foci of subretinal inflammation, hypertrophy, and vacuolation of the RPE and subretinal fibrosis. There was a moderate vitreous inflammation comprised predominantly of large numbers of heterophils admixed with inflammatory debris (Figure 4C and D). In addition, there was hypertrophy of the ciliary body epithelium. No histological abnormalities were attributed to ITV administration of BB to cynomolgus monkeys (Figure 4G and Figure 4H).
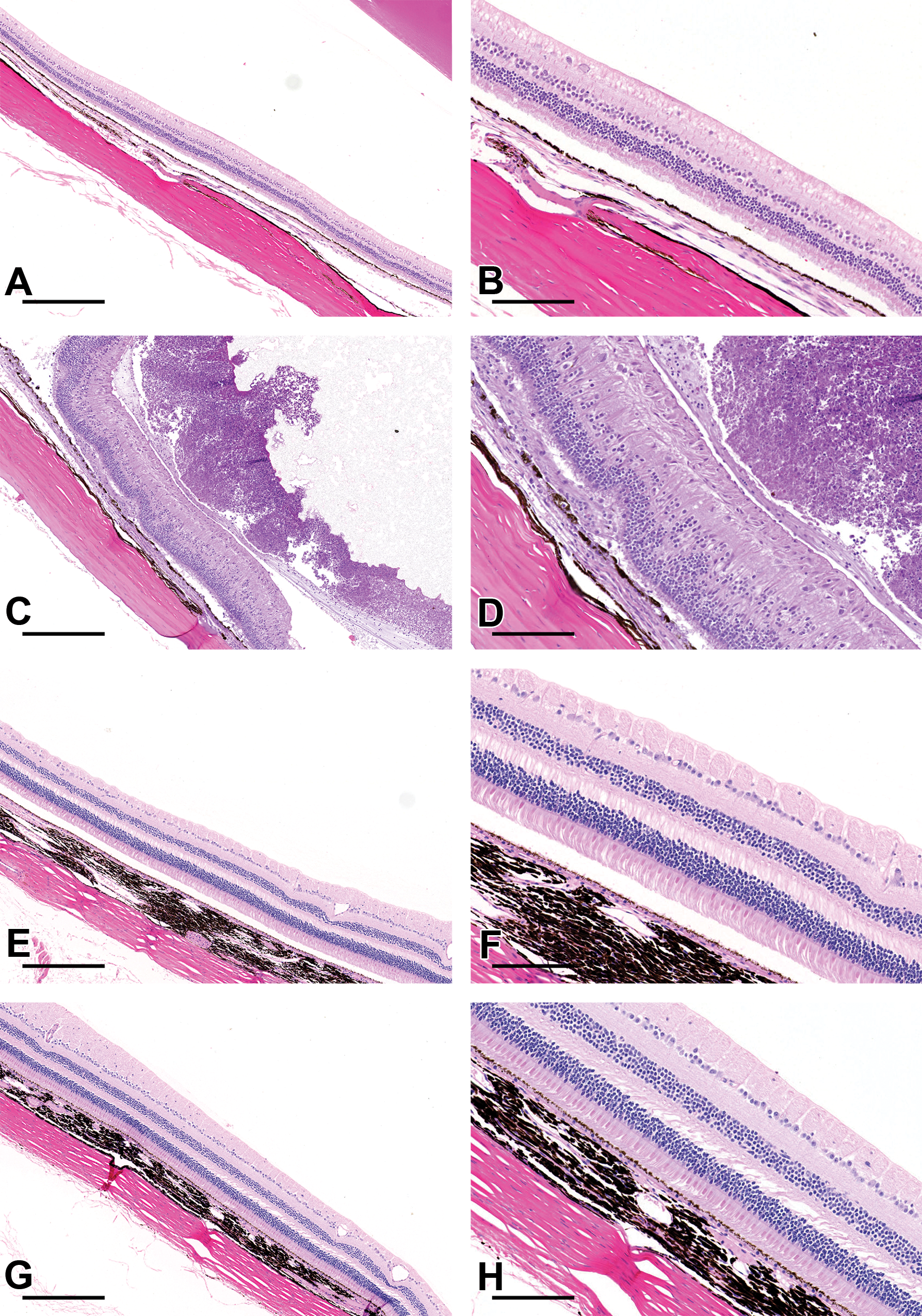
Effects of ITV BB in H&E stained tissues. Significant degenerative and inflammatory changes were observed focally to multifocally in the eyes of rabbits treated with BB (C and D) but not cynomolgus monkeys (E and F). Large numbers of degenerate heterophils admixed with inflammatory debris were observed in the vitreous of rabbits, overlying epiretinal membranes, areas of retinal degeneration, and subretinal fibrosis (C and D). Eyes from cynomolgus monkeys treated with BB (G and H) were normal, with no significant difference from those treated with saline (E and F). A and B, rabbit saline (control); C and D, rabbit BB; E and F, cynomolgus monkey saline (control); G and H, cynomolgus monkey BB (A, C, E, G scale bar 400 μm; B, D, F, H scale bar 100 μm). BB indicates benzyl benzoate; H&E, hemotoxylin and eosin; ITV, intravitreal.
In vitro assessments
To evaluate potential liabilities related to solvent-induced retinal toxicity in humans, cellular ATP was measured in primary human RPEs and HepG2s following treatment with BB and ATEC. Our results showed that at 48 hours postdose, both solvents induced toxicity in RPE and HepG2 cells, however RPEs were significantly more susceptible to the solvents tested (Figure 5). In RPEs, the IC50 for ATEC and BB was 0.0038 mM and 0.013 mM, respectively; in HepG2s, the IC50 for ATEC and BB was 0.4 mM and 31.8 mM, respectively. To understand the time-dependent kinetics of injury, ATP was measured at 2 hours postdose. After only 2 hours, both ATEC and BB were toxic to RPEs at 5 mM, the lowest concentration tested, reducing viability of RPEs by 40.2% and 25%, respectively (Figure 6). IncuCyte Live-Cell imaging was used to assess cell morphology every 8 hours for a period of 40 hours and the dose range tested was 0.005 mM and 500 mM. At 0.05 mM, signs of toxicity were seen with both solvents. Cells showed morphological changes, such as reduced size and aggregation, as well as apoptosis with both ATEC and BB as early as 8 hours following treatment (Figure 7), suggesting that even at very low doses, both solvents induced fast and robust toxicity to RPEs.
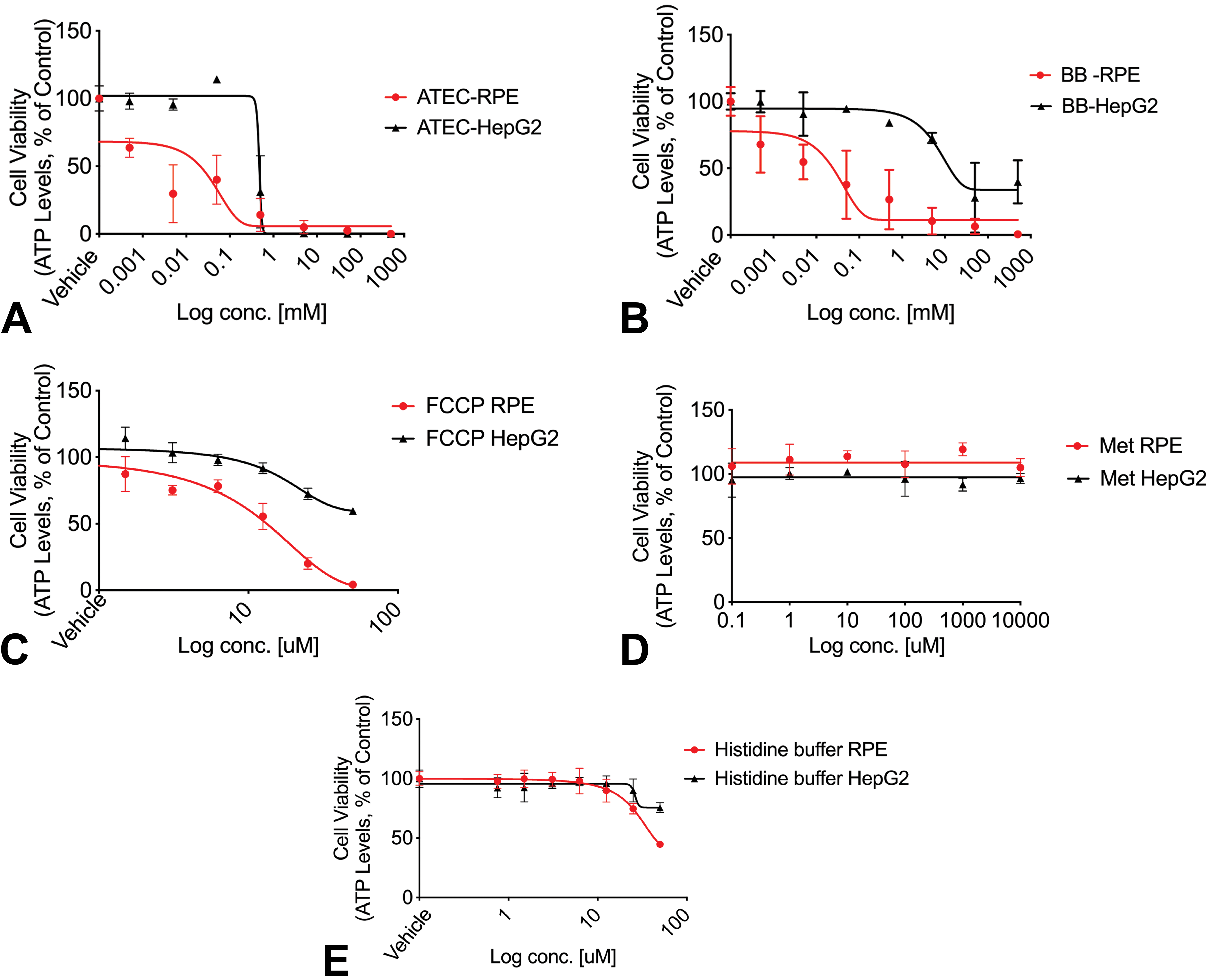
ATP levels in two cell lines following 48 hours incubation with solvent. Primary human RPE (red) and HEPG2 (black) cells were treated separately with ATEC, BB, FCCP (positive control), methionine, and histidine buffer (negative controls) for 48 hours. Graphs show mean ± SEM of triplicates. ATEC indicates acetyl triethyl citrate; BB, benzyl benzoate; FCCP, carbonilcyanide p-triflouromethoxyphenylhydrazone; HEPG2, human hepatocytes; RPE, retinal pigment epithelium; SEM, standard error of the mean.
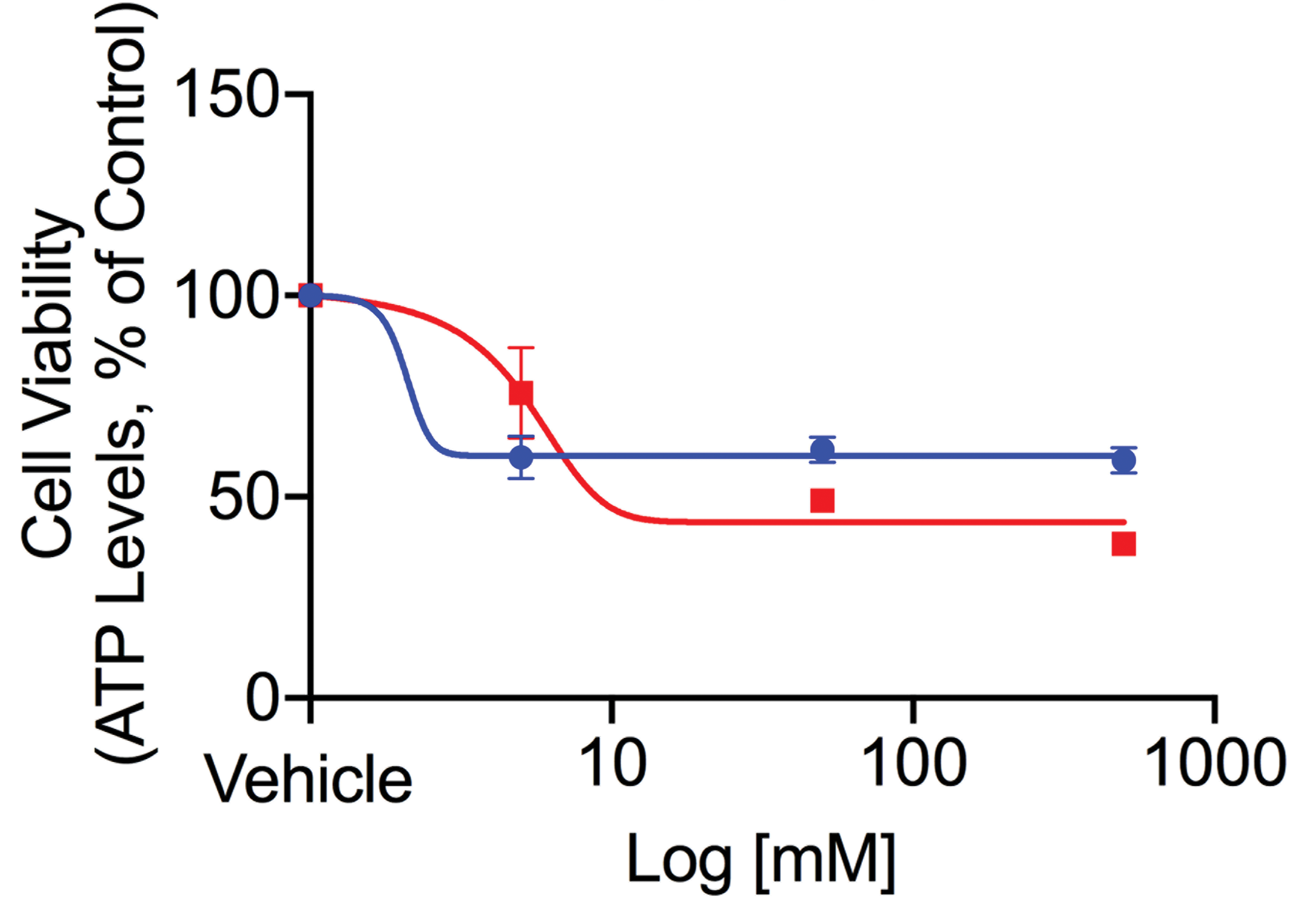
Acetyl triethyl citrate- and BB-induced RPE cytotoxicity after 2 hours incubation in vitro. RPE cytotoxicity measured at 2 hours post-treatment with ATEC (red) or BB (blue) at 5, 50, and 500 mM. Graphs show EC50 curve fitting using nonlinear regression and show mean ± SEM of triplicates. ATEC indicates acetyl triethyl citrate; BB, benzyl benzoate; RPE, retinal pigment epithelium; SEM, standard error of the mean.
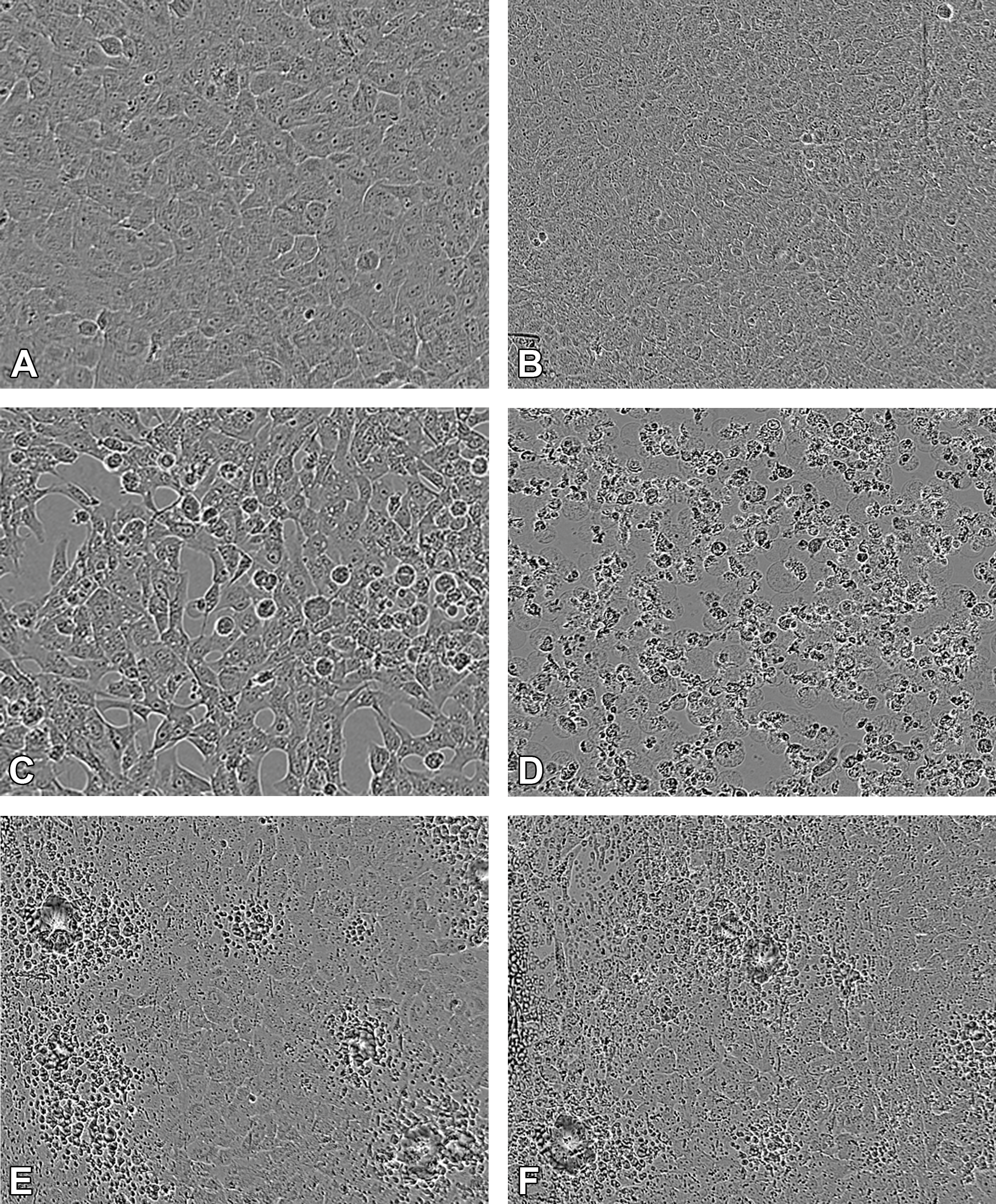
Bright field images of treated RPEs. Morphological changes evident at 8 and 40 hours following treatment with histidine buffer (control; A and B), ATEC (C and D), or BB (E and F) at original objective 10×. ATEC indicates acetyl triethyl citrate; BB, benzyl benzoate; RPEs, retinal pigment epithelium cells.
Given that mitochondria have a major role in ATP production, changes in mitochondrial respiration were evaluated in treated RPEs. Seahorse 96-well Extracellular Flux Analyzer was used to map the effects of the solvents to the OCR. A dose of 5 mM was chosen, and effects in the mitochondria were recorded after 2 hours. Our results showed that the decrease in cell viability observed in RPEs after treatment with BB was not due to mitochondrial injury. Although BB had no impact in OCR, ATEC induced a robust basal reduction in OCR immediately after treatment, suggesting that the cell death observed with ATEC was established prior and most likely independent of mitochondrial injury (Figure 8).
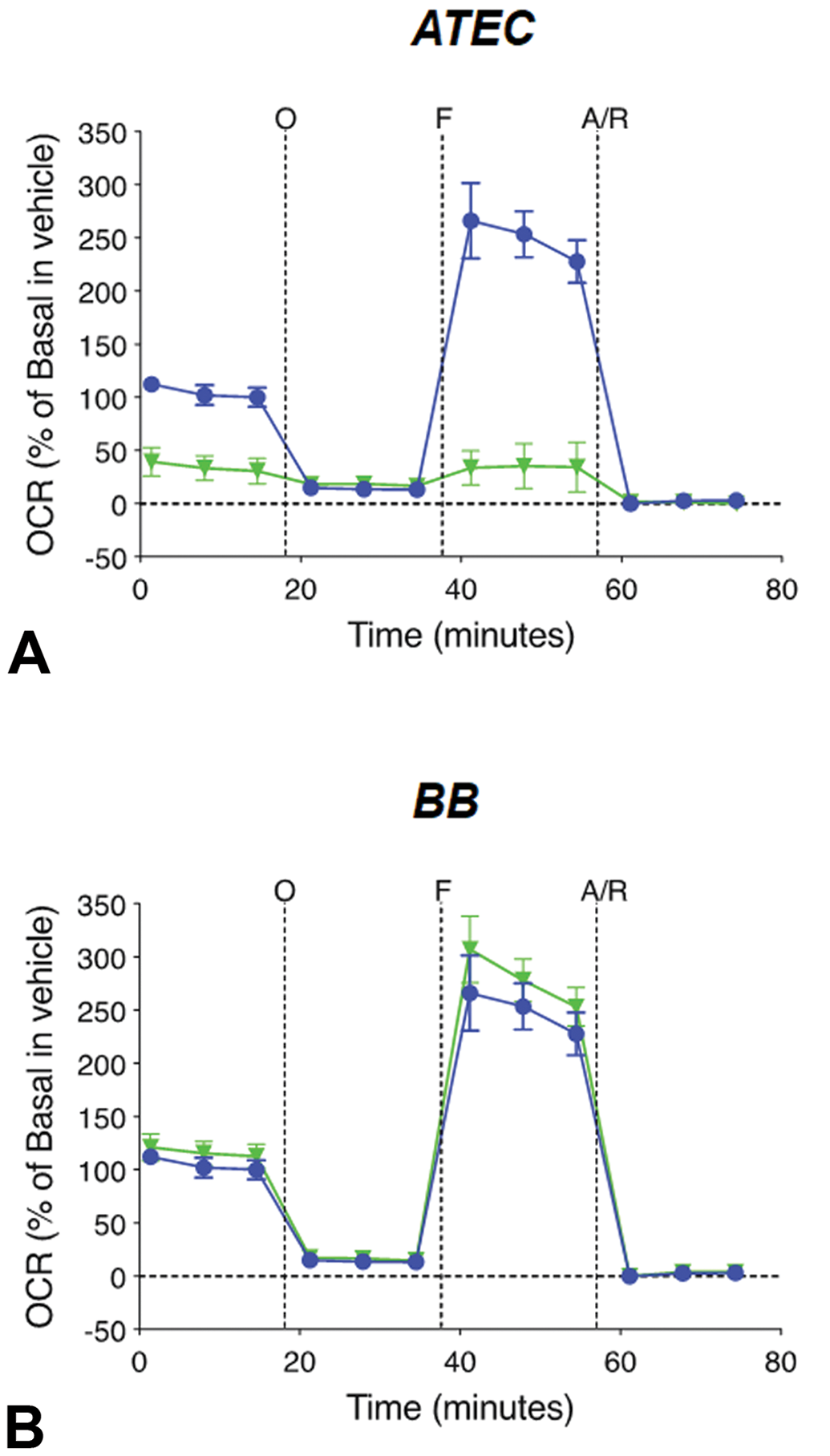
Profiles of mitochondrial stress tested using Seahorse XF96 Analyzer graphs show the oxygen consumption rate (OCR) of RPEs treated with ATEC or BB. Green lines represent solvent-treated cells and blue lines represent histidine buffer (control)-treated cells. Dotted vertical lines indicate injections of the specific stressors: oligomycin (O), FCCP (F), and rotenone/ antimycin A (A/R). Graphs show mean ± SEM of triplicates. ATEC indicates acetyl triethyl citrate; BB, benzyl benzoate; FCCP, carbonilcyanide p-triflouromethoxyphenylhydrazone; RPEs, retinal pigment epithelium cells; SEM, standard error of the mean.
Discussion
Toxicities after ITV injection are often observed with excipients that are well tolerated by other routes of administration because of the unique anatomy and physiology of the retina (ie, retina lacking regenerative capacity). This significantly limits the number of potential excipients and long-acting delivery formulations and necessitates testing of ocular tolerability of novel drug delivery systems considered for ITV administration. This work presents tolerability data of ATEC and BB following ITV administration in rabbits and monkeys. Moderate to severe, focal to multifocal retinal degeneration was observed histopathologically in DB and NZW rabbits following ITV administration of ATEC. Similar findings were observed in DB rabbits treated with BB; ATEC was not tested in DB rabbits. These findings correlated with the retinal whitening observed with indirect ophthalmoscopy and digital fundus imaging (Figures 1B, C and 2B, C). The location of solvent depots documented during in-life OEs and digital fundus photography were adjacent to the areas of retinal degeneration, suggesting a possible concentration-related and local mechanism of toxicity. As a potential component of melanin-binding drug product formulations, ATEC was assessed in both pigmented DB and nonpigmented NZW rabbits to explore any differences in tolerability, and there were no significant differences identified between the 2 strains when evaluated by OE and histopathology. Furthermore, in NZW rabbits, ATEC was administered inferiorly or superiorly to determine whether injection site and the subsequent depot location affected tolerability of the solvent. The site of retinal toxicity was similar from both inferior and superior administrations indicating that injection site did not mitigate ATEC toxicity. The findings suggest that after superior injection the depot settled, likely in a gravity-dependent manner to the inferior vitreous, and elicited focal toxicity at a location similar to rabbit eyes given inferior injection.
In contrast, ATEC and BB were well tolerated in cynomolgus monkeys and did not elicit the retinal toxicities seen in rabbits. In-life inflammation in monkeys was absent in most eyes or minimal if observed. Histopathology did not reveal any solvent-induced microscopic changes. Species differences in the biochemical and physical properties of the vitreous and the anatomy and physiology of the posterior chamber may have contributed to the way the solvent depot behaved, resulting in the species-specific toxicity profiles. 5 Monkeys have a less viscous vitreous than rabbits, which could allow movement of the solvent depot as well as relatively quicker dissolution of the solvent depot throughout the vitreous, decreasing the propensity for local, concentration-dependent effects. 16 In addition, the retinal vasculature differs significantly between the nonhuman primate and the rabbit. The cynomolgus monkey has a holangiotic retinal vasculature similar to humans, where blood vessels are located in the light-sensitive portion of the retina; rabbits have a merangiotic vasculature, where the vessels radiate horizontally from the optic nerve coincident with the myelinated nerve fibers, resulting in largely avascular retina except for the highly vascularized visual streak. 5,17 We hypothesize that the thicker vitreous and less perfused retina in rabbits prolonged exposure of the underlying nearby retina to a concentrated solvent depot. However, further experiments are needed to determine the role of specific physiological differences in the tolerability of solvents.
Both ATEC and BB were assessed in primary human RPEs to characterize the local toxicity effects of solvents in vitro, and to further investigate potential tissue-specific differences, the assays were also performed in HepG2s. The dose range tested (0.0005-500 mM) was based on the calculated solvent concentration of 100 mM in the monkey eye when a vitreous volume of 2 mL was considered. 5 Both solvents were cytotoxic to RPEs and HepG2s, however, RPEs were significantly more susceptible to ATEC when compared to HepG2s. This result suggests that to investigate ocular toxicity, tissue-specific cells should be used to avoid underestimation of the magnitude of injury. Moreover, both solvents showed rapid kinetics of toxicity, where morphological changes and decrease in viability of RPEs were observed as early as 2 to 8 hours posttreatment.
Oxygen consumption rate is measured before and after the addition of inhibitors targeting specific drivers of the electron transport chain, in order to investigate several parameters of mitochondrial respiration. Initially, baseline cellular OCR is measured, from which basal respiration can be derived. A decrease in OCR is expected following oligomycin administration, where the change in OCR represents the ATP-linked respiration rate. An increase in OCR is expected following FCCP administration, as the inner mitochondrial membrane becomes uncoupled. The subsequent introduction of protonophores reveals the maximal capacity of the electron transport chain. Addition of rotenone/antimycin-A completely inhibits the electron transport chain; therefore, a decrease in OCR from the maximum is expected, and from this the maximal respiration rate can be ascertained. 18 While our results show that mitochondrial stress was not the driver of cell death induced by BB, the data surrounding ATEC are less clear. A 45% reduction in basal cell respiration compared to control may be indicative of mitochondrial stress or it could be a result of other cellular stress pathways not covered in our studies, as overall cell viability is reduced to 80% under these conditions. Future experiments should consider lower doses and earlier time points to address these questions. Additional studies utilizing toxicants with well-characterized mechanisms of toxicity and artificial vitreous over the RPE cell monolayer may also shed light on the contribution of vitreous in the different toxicity profiles observed in rabbits and cynomolgus monkeys. Although more studies are required to understand the predictive power and translatability of the in vitro results, it is clear that ATEC and BB are perturbing the intracellular and/or extracellular microenvironment, impacting the ability of cells to restore their homeostasis. Importantly, our work highlights correlation between in vivo and in vitro findings as translatable between rabbit and in vitro human RPEs, thereby supporting an efficient screening in vitro paradigm for novel ITV solvent formulation excipients.
ITV administration of ATEC and BB produced moderate to severe, irreversible focal and multifocal retinal toxicities in rabbits characterized by disorganization and loss of retinal layers, and infrequent retinal folding, whilst appearing well tolerated in cynomolgus monkeys, suggesting a higher tolerance to solvent-induced retinal toxicity in cynomolgus monkeys. The reason for this difference was hypothesized to be due to the thicker vitreous and less perfused retina in rabbits prolonging exposure of the underlying retina to a localized and highly concentrated solvent depot. 17 ATEC and BB tested in vitro using human RPEs, demonstrated solvent-induced cytotoxicity at concentrations well below values used in vivo, with evidence suggesting different mechanisms of toxicity for each solvent. Therefore, we were unable to establish an acceptable therapeutic index for ATEC and BB as potential ITV formulation excipients. In conclusion, at the concentrations tested, ATEC and BB should not be considered for sustained drug delivery formulations delivered ITV. Further investigation is required to determine the feasibility of leveraging in vitro data to select 1) appropriate models for intravitreal tolerability assessments of solvents and 2) appropriate dose levels in nonclinical studies that would demonstrate tolerability with a sufficient margin of safety.
Footnotes
Acknowledgments
Thierry Nivaggioli, Karthik Rajagopal, Ann Daughtery, Aaron Fullerton, Will R. Proctor, and IVSg of Genentech for their scientific input and technical support. Evan Thackaberry, Florence Lorget, Cindy Farman, Covance Madison, OSOD, and ICON Biosciences for their contributions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
