Abstract
The safety of 6 modified terephthalate polymers as cosmetic ingredients was assessed. These ingredients mostly function as exfoliants, bulking agents, hair fixatives, and viscosity-increasing agents—nonaqueous. Polyethylene terephthalate (PET) is used in leave-on products up to 100% and in rinse-off products up to 2%. The Cosmetic Ingredient Review Expert Panel (Panel) considered that the PET used in cosmetics is chemically equivalent to that used in medical devices. The Panel determined that the Food and Drug Administration’s determination of safety of PET in several medical devices, which included human and animal safety data, can be used as the basis for the determination of safety of PET and related polymers used in cosmetics. Use studies of cosmetic eye products that contain PET demonstrated no ocular irritation or dermal sensitization. The Panel concluded that modified terephthalate polymers were safe as cosmetic ingredients in the practices of use and concentration described in this safety assessment.
Introduction
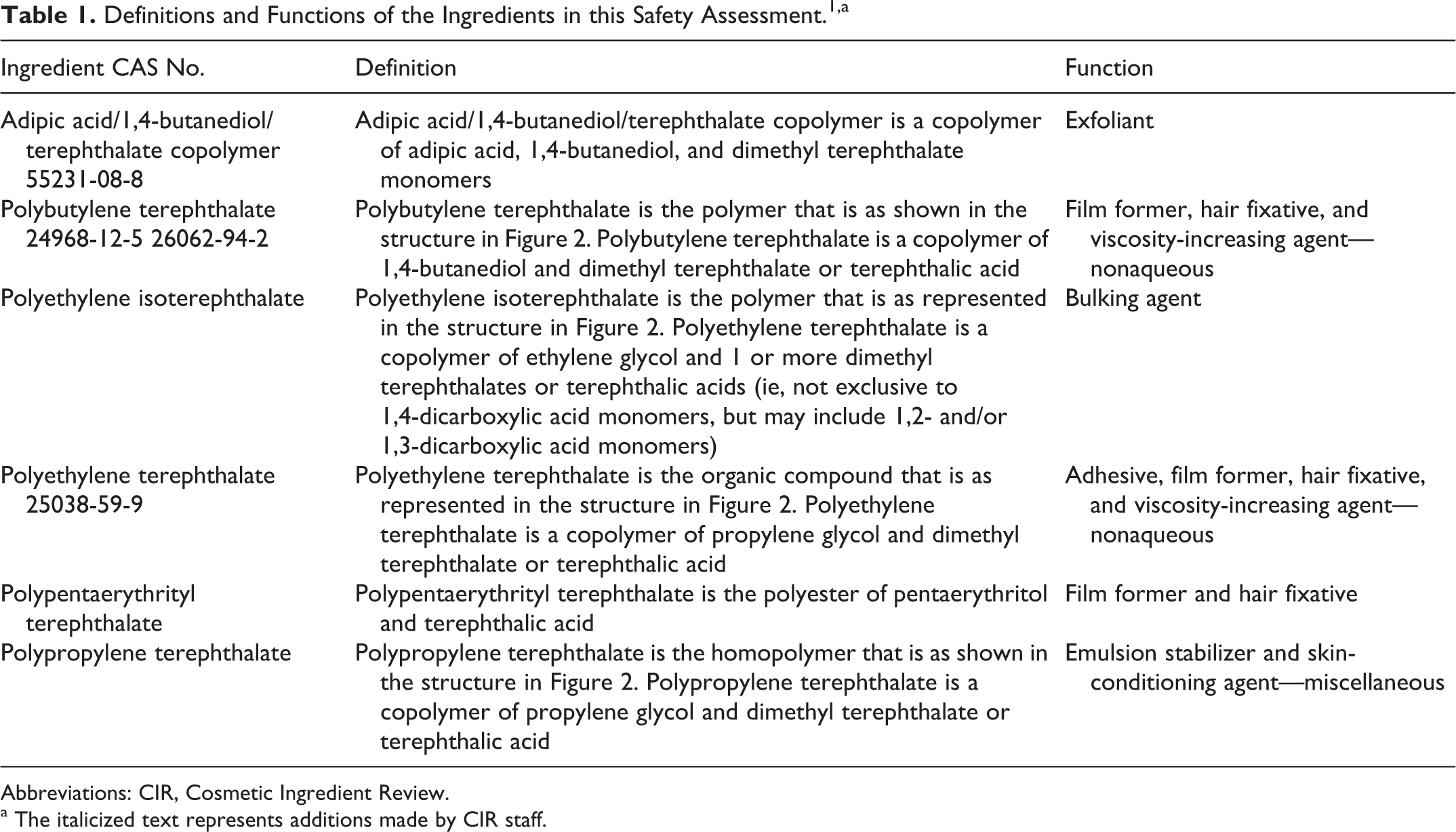
Definitions and Functions of the Ingredients in this Safety Assessment. 1 ,a
Abbreviations: CIR, Cosmetic Ingredient Review.
a The italicized text represents additions made by CIR staff.
Adipic acid/1,4-butanediol/terephthalate copolymer,
polybutylene terephthalate,
polyethylene isoterephthalate,
polyethylene terephthalate (PET),
polypentaerythrityl terephthalate,
polypropylene terephthalate.
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) considered that the modified terephthalate polymers produced for cosmetics are chemically equivalent to the polymers used in surgical sutures and other commercial medical devices made of terephthalate polymers. The safety information for those medical devices has been provided to the Food and Drug Administration (FDA) in medical device applications. The FDA found those data to be adequate and determined that terephthalate polymers are safe for use in devices when used in soft tissue approximation and/or ligation, including cardiovascular, ophthalmic, and neurological tissue. Systemic exposures of terephthalate polymers from devices used in these settings far exceed that expected for modified terephthalate polymers use in cosmetics.
The Panel considers that the assessment of modified terephthalate polymer safety as used in medical devices by the FDA provides the basis to establish the safety of these polymers in cosmetics because the PET is substantially equivalent as that used in approved medical devices and is used in a manner that presents less exposure risk. The Panel also contends that given the chemical similarity of the modified terephthalate polymer used in cosmetics to the same ingredient used in medical devices, data previously submitted to the FDA on that ingredient could be extrapolated to support the safety of adipic acid/1,4-butanediol/terephthalate copolymer, polybutylene terephthalate, polyethylene isoterephthalate, PET, polypentaerythrityl terephthalate, and polypropylene terephthalate.
One issue has been raised that appears unique to cosmetic uses of these polymers in glitter eye makeup. There is a concern that glitter made of modified terephthalate polymers may cause eye damage. The American Academy of Ophthalmology states that flakes of makeup that get into the eye and get into the tear film can increase eye irritation. 2 Glitter eye makeup that gets in the eye reportedly is a cause of corneal irritation and can cause eye redness. It was suggested that if particles get between the contact lens and corneal surface, they could scratch the cornea and may lead to corneal infection.
Chemistry
Overview of Chemistry and Manufacture
Modified terephthalate polymer ingredients are related to polyesters, wherein terephthalic acid (or an ester thereof) is a primary monomeric repeat unit. Terephthalic acid is an aromatic, p-dicarboxylic acid, which does not readily form a homopolymer, but easily copolymerizes with polyols (ie, multifunctional alcohols). The ingredients in this review are formed from diols (ie, 2 alcohol functional groups per molecule), with the exception of polypentaerythrityl terephthalate, which is prepared from a tetrol (ie, 4 alcohol moieties per molecule; pentaerythritol). Accordingly, with the exception of polypentaerythrityl terephthalate, these polymers are essentially linear. The polymerization reaction proceeds first through the esterification of terephthalic acid (or transesterification of a simple terephthalate ester, such as dimethyl terephthalate), with a diol (Figure 1). 3,4 This results in a new, difunctional monomeric unit with alcohols at both ends (eg, bis[hydroxyethyl]terephthalate). The exception, again, is polypentaerythrityl terephthalate, wherein the synthesis results in a mixture of tetrafunctional monomers. These new monomers then undergo condensation to produce the modified terephthalate polymer ingredients. Idealized structures of the modified terephthalate polymer ingredients in this safety assessment are provided in Figure 2.
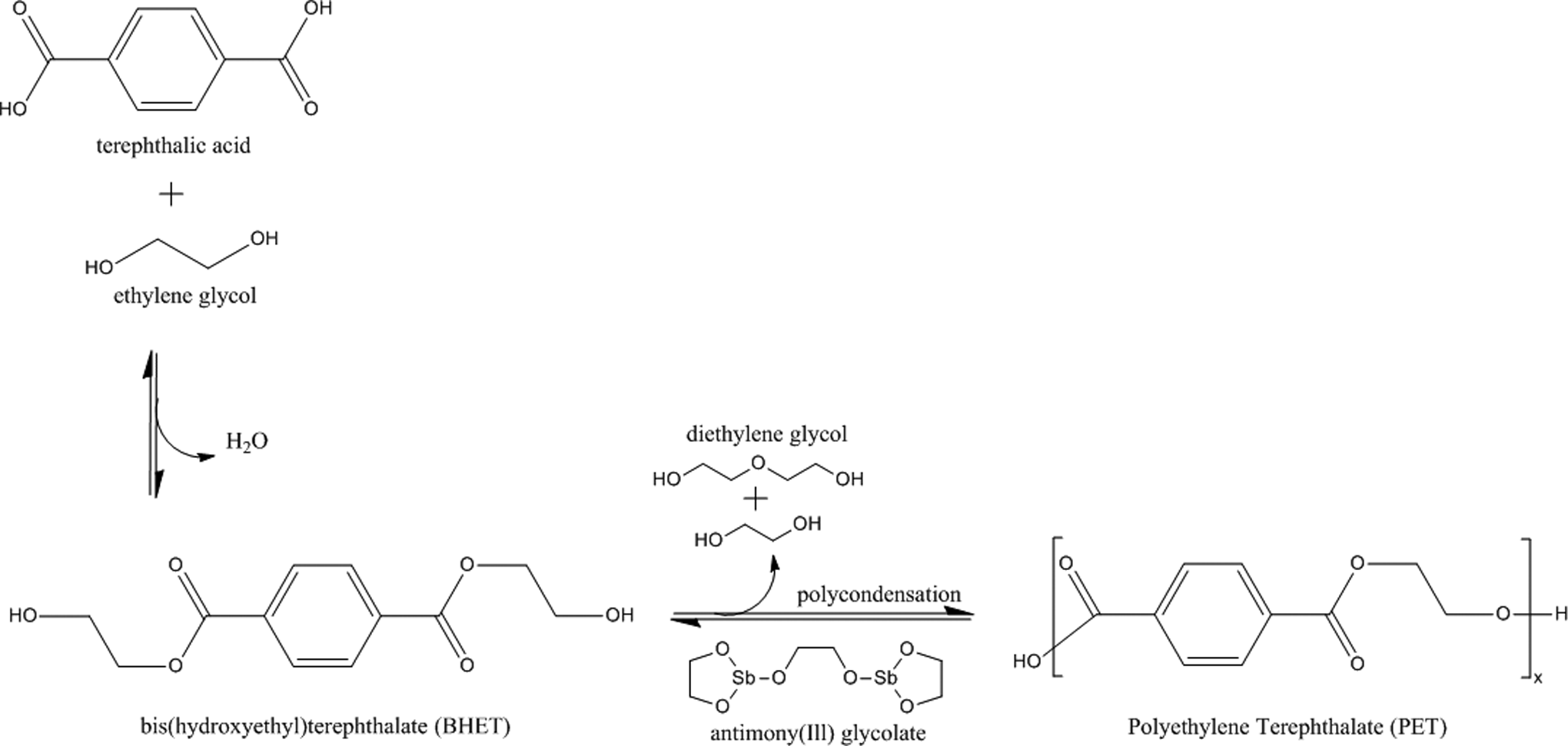
Most common manufacturing method for polyethylene terephthalate (PET).
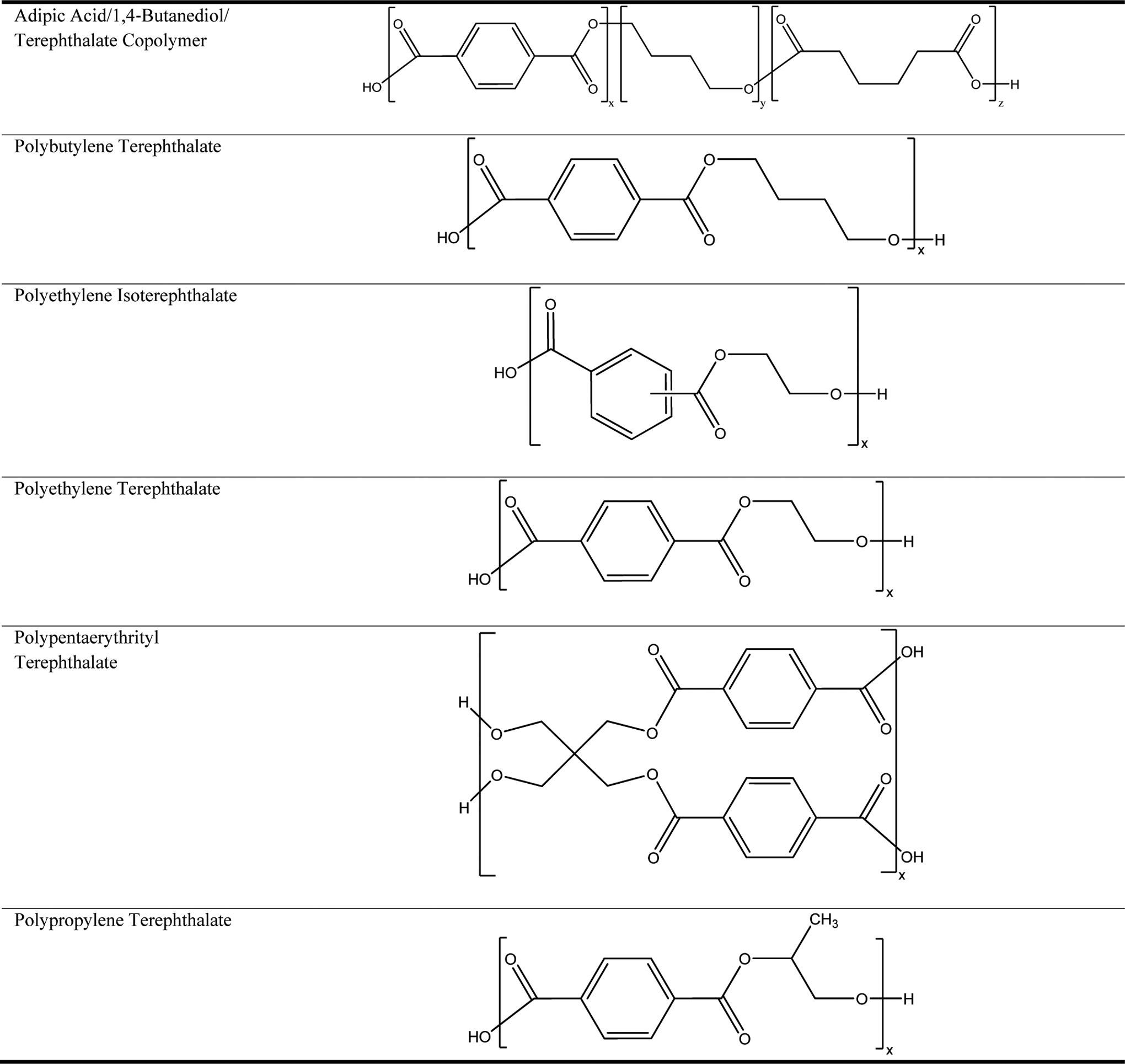
Idealized structures of the modified terephthalate polymer ingredients in this safety assessment. These idealized structures are merely generalized, 2-dimensional estimations of the true 3-dimensional frameworks that comprise these polymers. Although monomer units are in some instances drawn sequentially, by necessity, this by no means implies that these are block-type polymers. Instead, these structures are meant to represent only 1 example of the multitude of potentially produced connectivities found within these macromolecules.
When terephthalic acid is used as the starting material, water is released from the initial condensation reactions. 3,4 When a simple terephthalate ester is used (eg, dimethyl terephthalate), the corresponding alcohol (eg, methanol) is the byproduct. Early manufacturing methods of modified terephthalate polymers proceeded primarily from terephthalate esters, because the ester was easier to purify; however, since the mid-1960s, when significant progress was made in high-yielding, high-purity acid syntheses, terephthalic acid has become the primary starting material for these polymers, because of the lack of alcohol (eg, methanol) produced.
The condensation step, however, is essentially independent of whether an acid or ester has been used to synthesize the ester intermediate. 3,4 The condensation step proceeds via a metaloid oxide (eg, antimony[III] glycolate)-catalyzed transesterification and results in the release of some of the starting material alcohol and dimers thereof (some of which may be incorporated into the backbone of the polymer). Depending on processing methodologies (eg, product cooling rates), most of these ingredients can range from an amorphous glass to having a high degree of crystallinity. 5
It is common for manufacturers to market copolymers for purposes previously filled by homopolymer PET. Copolymer blends, such as polybutylene terephthalate/PET, have certain advantages over homopolymer PET with regard to mechanical properties and resistance to degradation. 6,7 In the United States, clear plastic bottles made with copolymers may still be legally marketed as PET (21CFR177.1630).
The size and shape of PET particles (as flakes) are defined by precision cutting PET sheets, and there is no typical size distribution. One reported particle size is 0.004 in 2 (0.1016 mm2). 8 The shapes of these particles may be hexagonal or square.
Physical and Chemical Properties
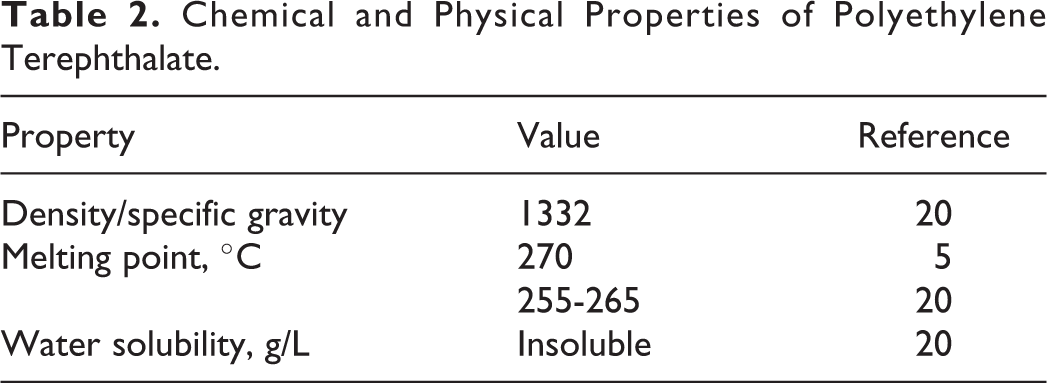
Limited physical and chemical properties for PET were discovered in the literature (Table 2). No physical and chemical properties were discovered for the rest of the ingredients in this safety assessment.
Chemical and Physical Properties of Polyethylene Terephthalate.
Impurities
It has been noted that not all PET materials are of the same chemical quality. 9 Therefore, it is important that the PET-related ingredients in this safety assessment are considered by the Panel to be analogous to those used in medical devices and that there are no concerns about impurities and estrogenic activity.
An analysis of the impurities of a sample of PET glitter showed the following results: arsenic, <0.05; antimony, 169.0; lead, 2.0; cadmium, <0.5; mercury, <0.1; nickel, <1; and chromium, <1 mg/kg. 10 The analysis of the migration of heavy metals showed that arsenic, antimony, lead, cadmium, mercury, chromium, barium, and selenium were below the levels of detection. Any possible free formaldehyde was below the limits of detection.
Diisononylphthalate, diethylhexylphthalate, dibutylphthalate (DBP), diisodecylphthalate, di-n-octylphthalate, butylbenzylphthalate (BBP), diisobutylphthalate (DiisoBP), dimethylphthalate (DMP), diethylphthalate (DEP), dibutylsebacate, di(2-ethylhexyl)adipate, and tributylacetylcitrate were below the levels of detection for this sample. 10 The same was found for nonylphenols, organotin compounds, organic solvents, primary aromatic amines, polycyclic aromatic hydrocarbons, and monomeric plasticizers. The color was fast in the presence of perspiration and saliva.
Modified terephthalate copolymers do not contain phthalates nor leach them with use. Polyethylene terephthalate is not considered an orthophthalate nor does PET require the use of phthalates or other softening additives. 11
Use
Cosmetic
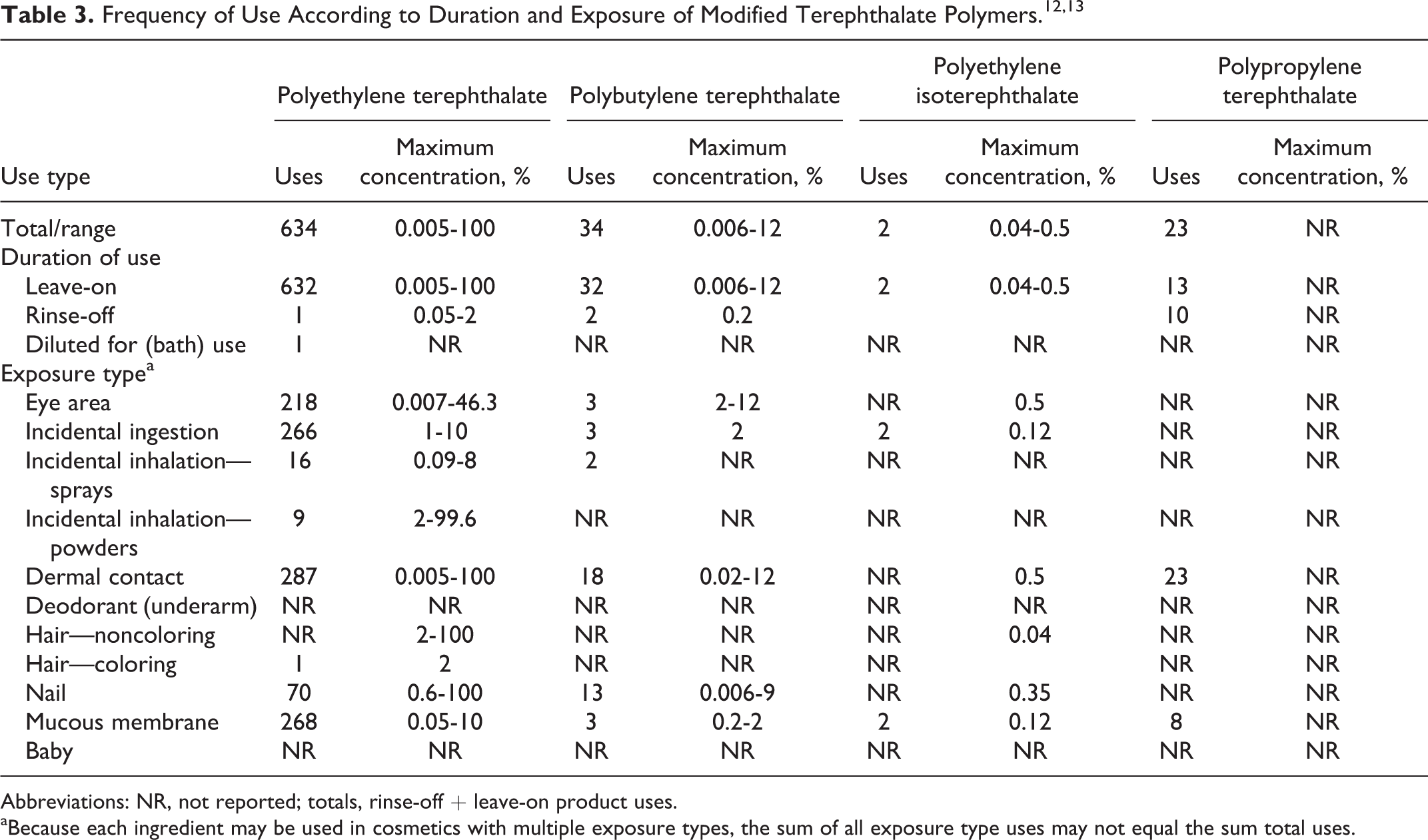
Data on ingredient usage are provided to the FDA Voluntary Cosmetic Registration Program (Table 3). 12 A survey has been conducted by the Personal Care Products Council (Council) of the maximum use concentrations for ingredients in this group. 13 This survey also included the physical forms (eg, flake and powder) of the ingredients being used.
Abbreviations: NR, not reported; totals, rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Polyethylene terephthalate was reported to be used in 632 leave-on products (266 lipsticks and 218 used in the eye area), 1 rinse-off product, and 1 diluted for bath. It is used in leave-on products up to 100% and in rinse-off products up to 2%. There were no concentrations of use reported for products used in the bath. Products used around the eye were reported to be used in flake form up to 46.3% (eye shadow), in powders (form not reported) up to 2%, and in make-up preparations up to 100% (face powders up to 99.6% and other makeup preparations up to 100%). Polyethylene terephthalate is used in body and hand sprays up to 0.3%. Polyethylene terephthalate is reported to be used in fiber form in mascara up to 0.05%. It is reported to be used as a gel in nail extenders up to 0.6% and in powder form up to 14%. It is also used in powder form in body and hand creams, lotions, and powders up to 0.005%. It was not reported what form of PET is used in powders (dusting and talcum), other fragrance preparations, bath soaps and detergents, other personal cleanliness products, and body and hand sprays.
Polybutylene terephthalate was reported to be used in 32 leave-on products up to 12% and 2 rinse-off products up to 0.2%. It was reported to be used in flake form in eye liner up to 12%, blushers up to 4%, and nail polish and enamel up to 9%. It was used in powder form in nail extenders up to 6%. The form of polybutylene terephthalate used in lipstick; other personal cleanliness products; and body and hand creams, lotions, and powders was not reported.
Polyethylene isoterephthalate was reported to be used in 2 lipsticks. The Council reported that it was used in flake form in eye shadow (up to 0.5%); tonics, dressings, and other hair grooming aids (up to 0.04%); lipstick (up to 0.12%); and nail polish and enamel (up to 0.35%).
Polypropylene terephthalate was reported to be used in 13 leave-on products and 10 rinse-off products (7 in bath soaps and detergents). There were no concentrations of use reported by the Council. There were no reported uses or concentrations of use for adipic acid/1,4-butanediol/terephthalate copolymer and polypentaerythrityl terephthalate.
Polyethylene terephthalate was reported to be used in body and hand sprays, powders, and fragrance preparations and could possibly be inhaled. Some cosmetic products contain flakes, fibers, or powders of modified terephthalate polymers. These ingredients are reportedly used at concentrations up to 99.6%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles less than 10 µm compared with pump sprays. Flakes used as glitter would be expected to have aerodynamic diameters substantially greater than 10 µm. The dimensions and size distributions of the fibers and powders used in cosmetics have not been reported. However, most droplets/particles incidentally inhaled from cosmetic aerosols would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 14 –19
Noncosmetic
Polyethylene terephthalate is used for high-impact resistant containers. 20 It is used for packaging of soda, mouthwash, pourable dressings, edible oils, and peanut butter. It is used for cereal box liners, soda bottles, boil-in-the-bag pouches, and microwave food trays. Modified PETs can be heated in a microwave or in a conventional oven at 180°C for 30 minutes.
Regulation
Food and Drug Administration regulations regarding the ingredients in this safety assessment are provided in Table 4. These regulations allow for contact with food substances.
Code of Federal Regulations Regarding the Polybutylene Terephthalate and PET.

Abbreviation: FDA, Food and Drug Administration.
Pet in Medical Devices
Food and Drug Administration considered the safety of PET when approving the following medical devices that include this material:
Polyethylene terephthalate in surgical sutures was approved by the FDA as a class II (special controls) medical device that requires premarket notification and adherence to standards (21CFR878.5000). Required testing includes acute and long-term (>30 day) biocompatibility testing for cytotoxicity, irritation or intracutaneous reactivity, sensitization, systemic toxicity, implantation effects, and hemocompatibility. 25 The sutures may be provided uncoated, coated, undyed, or dyed with appropriate approved colors. The FDA found the data on the safety of PET to be adequate and determined that medical devices containing PETs are safe and effective when used for soft tissue approximation and/or ligation, including cardiovascular, ophthalmic, and neurological tissue. 26
Esophageal and gastrointestinal dilators are class II medical devices (21CFR876.5365) that must adhere to the same standards listed previously for the surgical sutures. An esophageal dilator, used to dilate a stricture of the esophagus, may consist of a hollow cylindrical instrument (bougie), a weighted bougie with a mercury or metal olive-shaped weight that slides on a guide, such as a string or wire, or may consist of a bougie with a deflated balloon attached to a guide wire. The balloon is made of PET.
Patches, pledgets, and intracardiac devices (surgical mesh) are made of polypropylene, PET, or polytetrafluoroethylene. They are fabric devices placed in the heart to repair septal defects, for patch grafting, to repair tissue, and to buttress sutures (21CFR870.3470). These devices are also class II devices and adhere to the same standards listed previously for the surgical sutures.
Irritation and Sensitization
Ocular Irritation
In several use tests of eye shadows, gels, liners, and mascara containing PET (up to 46.272%), there were few reports of adverse effects, including in individuals wearing contact lenses (Table 5).
Use Tests of Cosmetic Products Containing Polyethylene Terephthalates.
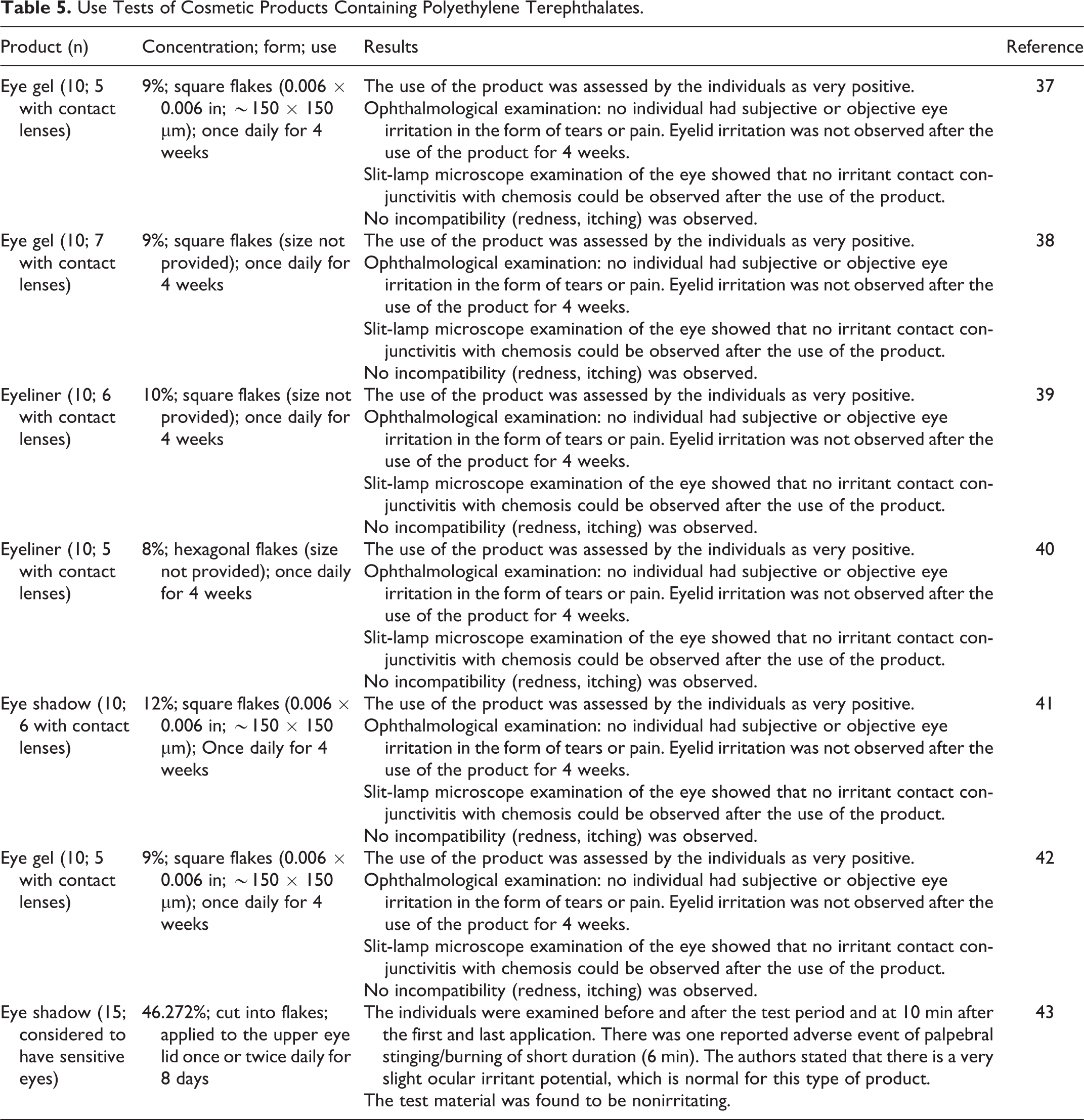
In a use test of 3 mascaras containing PET (4.2%) and polybutylene terephthalate (4.8%), they were found to not have the potential to be irritating or sensitizing. 27 The individuals (n = 29) applied the test material at least once per day for 4 weeks. The terephthalate polymers were described as solid particles cut from sheets of these materials.
Sensitization
In a human repeated insult patch test (HRIPT) of an eyeliner containing PET (1.5%), there were no signs of irritation or sensitization. 28 The test material (0.2 g) was administered to the backs of individuals (n = 107) under occlusion 3 times/wk for 3 weeks. After a 2-week rest, the test material was applied to a naive site.
An HRIPT (n = 30) of an eye shadow pencil containing PET (12%; square flakes) administered undiluted to healthy skin was negative for primary irritation and allergic hypersensitivity reactions at 24, 48, and 72 hours. 29
In several use tests of eye makeup formulations (Table 5), there was no irritation or sensitivity observed or reported by the individuals.
In a use test of a liquid eye liner containing PET (15% as glitter) lasting 4 weeks, the individuals (n = 20 women; 8 with very sensitive skin and 6 wearing contact lenses) did not report any adverse events during use, had no effects detected in a fissure lamp and dermatological examination, nor showed any signs of sensitization when administered a patch test at the end of the test period. 30 The individuals applied the test material daily and were examined before and after the test period. No changes in the area of the conjuctiva, cornea, eyelids, or tear ducts were observed. In the patch test of the liquid eye liner after the 4-week test period, the test substance was applied to the volar surface of the arm for 24 hours under occlusion then observed at removal and at 48 and 72 hours.
In a use test of 2 eye shadow formulations containing PET (15% as glitter) lasting 4 weeks, the individuals (n = 20 women, 6 with very sensitive skin and 5 wearing contact lenses) did not report any adverse events during use, had no effects detected in a fissure lamp and dermatological examination, nor showed any signs of sensitization when administered a patch test at the end of the test period. 31 The individuals applied the eye shadows, one to each eye lid, daily. They were examined before and after the test period and no changes to the area of the conjuctiva, cornea, eyelids, or tear ducts were observed. In a patch test of the eye shadows after the 4-week test period, the test substances were applied to the back for 24 hours under occlusion then observed at removal and at 48 and 72 hours.
Genotoxicity
In Vitro
Polyethylene terephthalate leachate
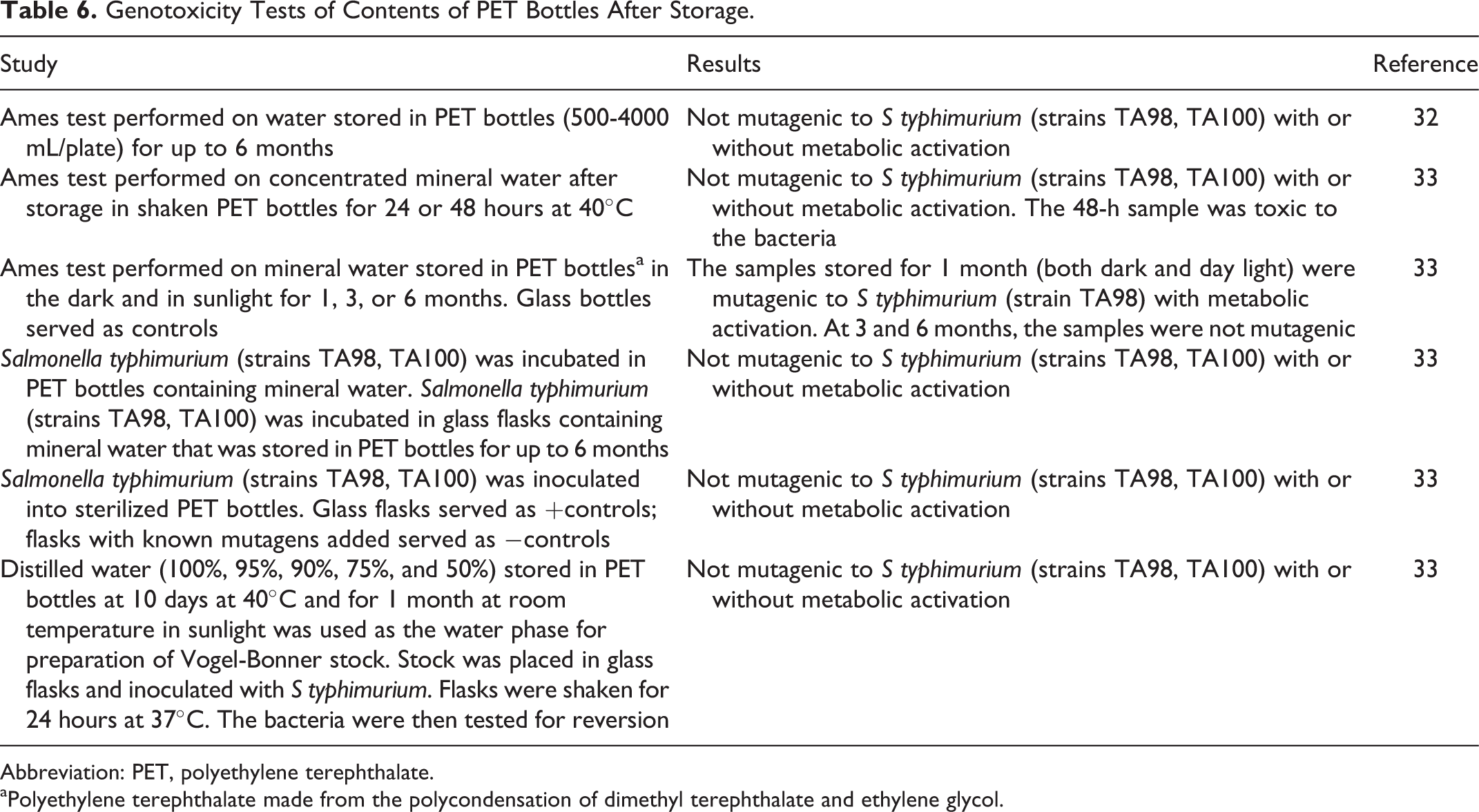
In several tests of water stored in PET for up to 6 months, the water was not mutagenic to Salmonella typhimurium (strains TA98 and TA100) with or without metabolic activation except for one test, where the water was mutagenic after storage for 1 month but not at 3 and 6 months (Table 6). 32,33
Genotoxicity Tests of Contents of PET Bottles After Storage.
Abbreviation: PET, polyethylene terephthalate.
aPolyethylene terephthalate made from the polycondensation of dimethyl terephthalate and ethylene glycol.
Clinical Use
Polyethylene Terephthalate
Polyethylene terephthalate sutures were used in 2 studies of penetrating keratoplasty surgery (n = 20 and 45) comparing PET and nylon sutures and style of suture using these materials. 34 All complications were mechanical or technique related and not toxicological.
Polyethylene terephthalate sutures were used in penetrating keratoplasty for keratoconus surgery (n = 14). 35 The individuals were followed for 22 to 48 months. There was no vascularization of the stitches, cheesewiring, or graft rejections. There were 4 cases of stitch removal, 3 for mechanical reasons and 1 for an ulceration (which resolved when the stitch was removed). The author concluded that the problems had to do with technique and not toxicity.
In an evaluation of the use of a mesh made of PET for use in brow suspension ptosis surgery, a survey of 5 case reports and other cases in the literature were reviewed. 36 There were reported cases of extrusion and granuloma formation 1 month to 1 year after implantation characterized as foreign body reactions. There were also cases followed for up to 45 months with no complications. The authors concluded that technique (for example, knot size) and placement were the reasons for the problems and not toxicity.
Summary
The safety of modified terephthalate polymers as used in cosmetics were assessed. The 6 ingredients in this safety assessment mostly function as exfoliants, bulking agents, hair fixatives, and viscosity-increasing agents—nonaqueous.
Cosmetic Ingredient Review considers that the modified terephthalate polymers produced for cosmetics are chemically equivalent to the polymers in surgical sutures and other commercial medical devices made of terephthalate polymers. The safety information for those medical devices was provided to the FDA in medical device applications including acute and long-term biocompatibility testing for cytotoxicity, irritation or intracutaneous reactivity, sensitization, systemic toxicity, implantation effects, and hemocompatibility. The FDA found those data to be adequate and determined that PETs were safe and effective for use in surgical sutures, esophageal dilators, and surgical mesh. The PET-related ingredients in this safety assessment are considered by the Panel to be equivalent to those used in medical devices.
An analysis of the impurities of a sample of PET glitter showed the following: arsenic, < 0.05; antimony, 169.0; lead, 2.0; cadmium, <0.5; mercury, <0.1; nickel, <1; and chromium, <1 mg/kg. The analysis of the migration of heavy metals showed that arsenic, antimony, lead, cadmium, mercury, chromium, barium, and selenium were below the levels of detection. Free formaldehyde was below the limits of detection. Diisononylphthalate, diethylhexylphthalate, DBP, diisodecylphthalate, di-n-octylphthalate, BBP, DiisoBP, DMP, DEP, dibutylsebacate, di(2-ethylthexyl)adipate, and tributylacetaylcitrate were below the levels of detection.
Polyethylene terephthalate was reported to be used in 634 cosmetic products at concentrations up to 100%, polybutylene terephthalate in 34 products up to 12%, polyethylene isoterephthalate in 2 products, and polypropylene terephthalate in23 products up to 0.5%. Many of these uses were reported to be in flake, powder, and fiber form. Polypropylene terephthalate was reported to be used in 13 leave-on and 10 rinse-off products. There were no reported uses for adipic acid/1,4-butanediol/terephthalate copolymer and polypentaerythrityl terephthalate.
Polyethylene terephthalate is safe for use in food packaging that may be stored, heated, or microwaved.
In use tests of several different eye products containing PET up to 46.272%, the products were found to be nonirritating and nonsensitizing including in individual wearing contact lenses.
An eyeliner containing PET (1.5%) was not irritating or sensitizing in a repeated insult patch test.
In several tests of water stored in PET, the water was not mutagenic to S typhimurium except for one test, where the water was mutagenic after storage for 1 month but not at 3 and 6 months.
Complications from the use of PET sutures were attributed to mechanical or technique issues and not toxicological issues.
Discussion
The Panel concluded that, since the PET polymers used in cosmetics are considered to be chemically equivalent to PET used in medical devices (ie, surgical sutures, esophageal dilators, and surgical mesh), the safety review by the FDA can be used as a basis for the assessment of the safety of PET polymers in cosmetics. Although the terminology might suggest it, terephthalates do not contain phthalates. Thus, there is no concern regarding estrogenic activity of terephthalates.
Although there are data gaps, the similar chemical structures, functions, concentrations in cosmetics, and expected physicochemical properties allow grouping these ingredients together and interpolating the available toxicological data to support the safety of the entire group. All of these ingredients are large polymers and their surfaces would have no chemical reactivity. It was the consensus that these long-chain polymers would not penetrate the skin.
The Panel considered the issue of incidental inhalation exposure from body and hand sprays, powders, and fragrance preparations. There were no inhalation toxicity data available. The Expert Panel believes that the sizes of a substantial majority of these ingredients, as manufactured, are outside the respirable range. These ingredients are reportedly used at concentrations up to 8% in cosmetic products that may be aerosolized and up to 99.6% in face powders that may become airborne. Some products contain these ingredients in the form of flakes, fibers, or powders that would be expected to have aerodynamic diameters greater than 10 µm, although the dimensions and size distributions of the fibers and powders have not been reported.
The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount.
Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Since PET polymers are used in implanted medical devices approved by the FDA, the Panel was satisfied that the modified terephthalate polymers in this safety assessment are chemically inert under physiological conditions and conditions of use, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel addressed the potential concern regarding ocular injury by some glitter products composed of these ingredients. Usage studies of eye products failed to demonstrate that the glitter products entered the eye under normal conditions. These data combined with the lack of case reports and no ongoing FDA regulatory actions suggested to the Panel that ocular injury is not likely.
Conclusion
The Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment: Adipic acid/1,4-butanediol/terephthalate copolymer*, polybutylene terephthalate, polyethylene isoterephthalate, polyethylene terephthalate, polypentaerythrityl terephthalate*, polypropylene terephthalate.
The * indicates not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from Lillian J. Gill, Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.