Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 6-hydroxyindole, which functions as an oxidative hair dye ingredient. The Panel considered relevant animal and human data provided in this safety assessment and concluded that 6-hydroxyindole is safe for use in oxidative hair dye formulations.
Keywords
Introduction
This report addresses the safety of the oxidative hair dye ingredient, 6-hydroxyindole, as used in cosmetics.
Chemistry
Physical and chemical properties of 6-hydroxyindole are found in Table 1. 6-Hydroxyindole (as shown in Figure 1) is a 6-hydroxylated benzo(b)pyrrole.
Physical and Chemical Properties of 6-Hydroxyindole.
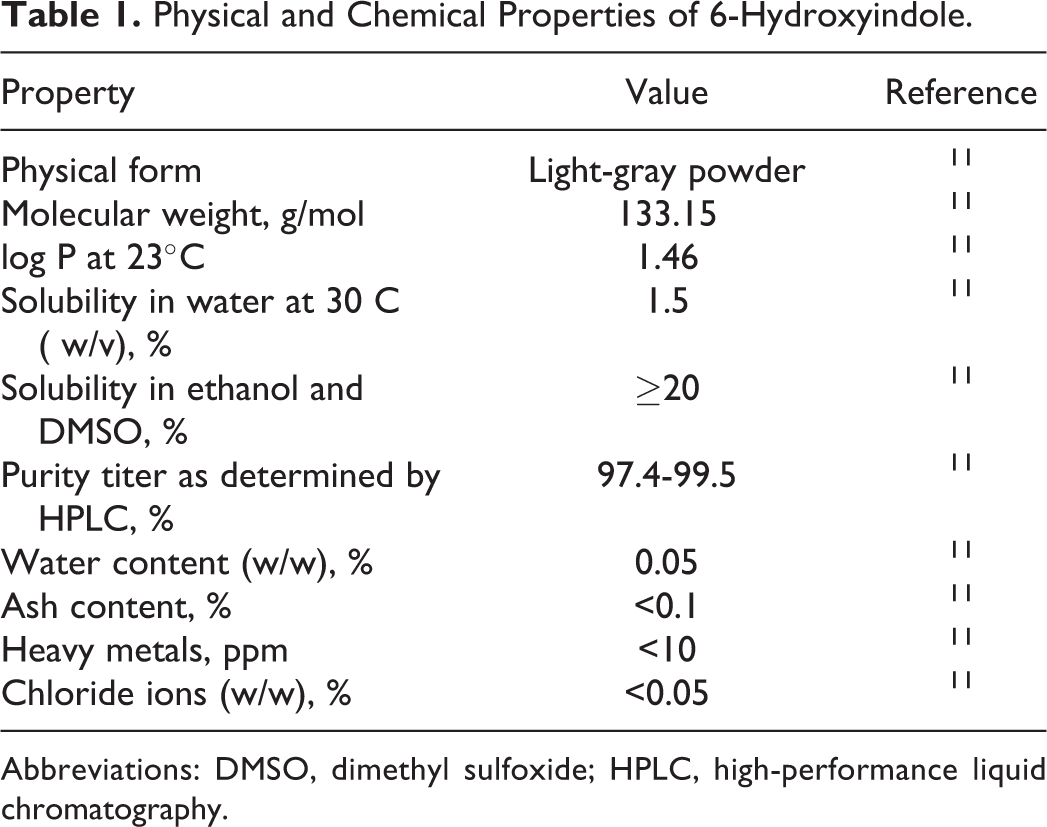
Abbreviations: DMSO, dimethyl sulfoxide; HPLC, high-performance liquid chromatography.
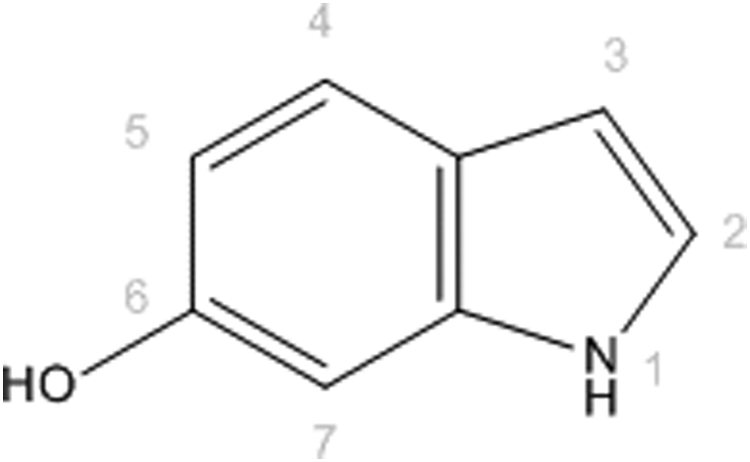
6-Hydroxyindole, a 6-hydroxylated benzo(b)pyrrole.
The synthetic process used to manufacture 6-hydroxyindole in hair dyes is the Leimgruber-Batcho indole protocol shown in Figure 2. 1,2 The commercially available 4-methyl-3-nitrophenol can be benzyl protected before condensation with pyrrolidine and N,N-dimethylformamide dimethylacetal. Reduction of this intermediate results in cyclization of the pyrrole ring and benzyl deprotection then affords 6-hydroxyindole.
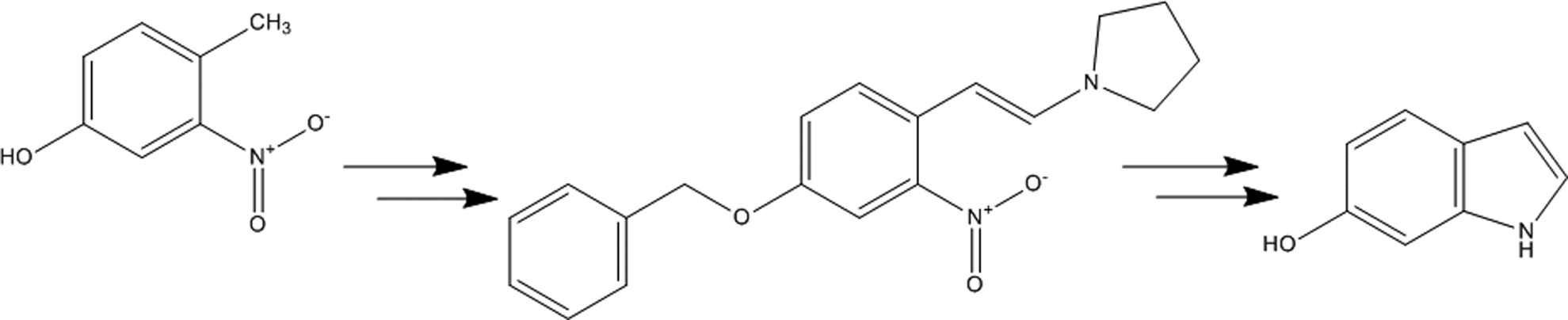
A synthetic pathway to 6-hydroxyindole.
Although nitrosamine content has not been reported, 6-hydroxyindole has a nitrogen atom in the pyrrole ring that can be potentially nitrosated. Indoles are readily nitrosated to form N-nitrosamines. 3 Of concern in cosmetics is the conversion (nitrosation) of ingredients into N-nitro compounds that may be carcinogenic. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 4 Nitrosation can occur under physiologic conditions. 5 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in the nitrosation of amines in aqueous (aq) solution. 6 Accordingly, hair dyes with 6-hydroxyindole should be formulated to avoid the formation of N-nitroso compounds.
According to a 2002 study published by COLIPA, the amount of 6-hydroxyindole used in oxidative hair-coloring products throughout the hair dye industry was 1 ton. 7 For use as a hair dye ingredient, 6-hydroxyindole is oxidized with hydrogen peroxide. 8 Oxidation purportedly leads to dimerization, trimerization, and polymerization to eumelanin-like polymers. 9,10 Linkages predominantly occur at 2-, 5-, and/or 7-positions, due to electronic effects.
Impurities
Potential impurities in 6-hydroxyindole are reagents and intermediate reaction products. These include 4-methyl-3-nitrophenylamine, 4-methyl-3-nitrophenol, 4-benzyloxy-1-methyl-2-nitrobenzene, 1-(2-[4-benzyloxy-2-nitro-phenyl]-vinyl)-pyrrolidine, and 6-benzyloxy-1 H-indole, all at levels below 500 ppm. 11
Use
Cosmetic
According to information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program, 6-hydroxyindole is used in a total of 104 hair-coloring formulations. 12 A survey of use concentrations has reported that 6-hydroxyindole is used at a maximum concentration of 0.5% in hair dyes and colors. 13
Europe’s Scientific Committee on Consumer Products (SCCP) concluded that 6-hydroxyindole would not pose a health risk to the consumer when used as an ingredient in oxidative hair dye formulations as long as the maximum concentration applied to hair does not exceed 0.5%.
11
The European Commission has added 6-hydroxyindole to Annex III List of Substances Which Cosmetic Products Must Not Contain Except Subject to the Restrictions Laid Down using the limitations suggested by the SCCP and has placed the following labeling requirements on this ingredient and all other hair dye ingredients: The mixing ratio must be printed on the label. Hair colorants can cause severe allergic reactions. Read and follow instructions. This product is not intended for use on persons under the age of 16. Temporary “black henna” tattoos may increase your risk of allergy. Do not color your hair if you have a rash on your face or sensitive, irritated and damaged scalp, you have ever experienced any reaction after coloring your hair, or you have experienced a reaction to a temporary “black henna” tattoo in the past.
14
Hair dye caution statement—FDA labeling
This ingredient is considered a coal tar hair dye for which regulations require caution statements and instructions regarding patch tests in order to be exempt from certain adulteration and color additive provisions of the United States’ Federal Food, Drug, and Cosmetic Act. In order to be exempt, the following caution statement must be displayed on all coal tar hair dye products: Caution—this product contains ingredients which may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dyeing the eyelashes or eyebrows; to do so may cause blindness.
In 2012, Thyssen et al published a report regarding such self-testing for contact sensitization to hair dyes. 15 These authors concluded that, in its present form, the hair dye self-test has severe limitations. The authors issued the warning that if the use of a hair dye self-test to predict contact sensitization becomes widespread, there is severe risk that a tool has been marketed that may cause morbidity in European consumers. In accompanying editorial, An Goossens, on behalf of the European Society of Contact Dermatitis (ESCD), asserted that industry is focussing on predicting the risks from exposure to hair dyes by having millions of European consumers perform a self-test prior to each hair dying and stated that it is the opinion of the ESCD that attention must be given to reducing the risks of serious allergic reactions by improving the safety of the products themselves. 16
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Oral exposure route
The plasma pharmacokinetics and excretion balance of 6-hydroxyindole were studied in Wistar Han Ico:WI (IOPS AF/Han) rats. 17 Twelve rats of each sex received a single 300 mg/kg body weight dose of 6-hydroxyindole(benzenering-U-14C) in a suspension of 0.5% aq carboxymethylcellulose via gavage. For the plasma pharmacokinetics portion of the study, blood samples were collected from 9 rats of each sex before dosing and at 1, 2, 4, 6, 8, 24, 48, and 72 hours after dosing. All rats were killed after the last blood sample. No necropsy was performed. In the excretion, balance portion of the study in the remaining 3 rats of each sex, urine, feces, and cage washes were collected during a 24-hour predosing period and during the periods of 0 to 6, 6 to 24 (urine), or 0 to 24 hours (feces) and 24 to 48, 48 to 72, 72 to 96, 96 to 120, 120 to 144, and 144 to 168 hours (urine and feces) after dosing. Cages were carefully rinsed after each collection of feces. All samples were frozen until analysis. All animals were killed at the end of the study and a wide range of tissues and organs were sampled. For each sample, total radioactivity was evaluated by liquid scintillation counting of plasma, urine, feces, and rinse water.
No mortalities were observed. In the first 24 hours, brown greenish urine and piloerection were observed in most animals. Other clinical signs that were observed in some animals about 45 minutes after dosing and lasting 3 to 5 hours included tremors, hypersalivation, lacrimation, lateral decubitus, jerking movements, staggering gait, half-closed eyes, and coldness to touch. These observations were more frequent in female animals. The urine remained colored for up to 3 days. Total radioactivity levels in plasma reached a maximum 1 hour after dosing with a mean Cmax value of 132 µgeq/mL (107µgeq/mL in males and 157 µgeq/mL in females). The plasma radioactivity levels decreased in a biphasic manner at a moderate rate until 24 hours postdosing, followed by a slower decline until the last sampling point (mean 0.88 µgeq/mL; 0.77 µgeq/mL in males and 0.98 µgeq/mL in females). The mean systemic exposure (area under the curve, AUC [0-∞]) was 799 µgeq-h/mL (638 µgeq-h/mL in males and 954 µgeq-h/mL in females). The mean total cumulative excretion (CE) of the radioactive dose over the 168-hour period was 95.4%, with most of the dose excreted rapidly at an average of 8.9% within 24 hours of dosing. The radioactivity was mainly excreted in the urine (mean 73.8%), and the mean CE in feces was 17.3%. No anomalies were noted in postmortem examinations. The findings of this study indicated high systemic exposure to 6-hydroxyindole after oral exposure to rats. 17
Dermal exposure route: Absorption
In an in vitro percutaneous absorption study, ( 14 C) 6-hydroxyindole was applied to human abdominal dermatomed skin in flow-through diffusion cells. 18,19 The integrity of the skin was tested by measuring transepidermal water loss prior to testing material application. The test substance was prepared at a concentration of 2% with hydrogen peroxide developer or with water to yield a final concentration of about 1%. The test substance (20 mg/cm2) was applied to the skin samples for 30 minutes and then washed off with water and 2% sodium lauryl sulfate solution. Measurements for radioactivity in the receptor fluid were made 24 hours after application. When formulated with hydrogen peroxide, the absorbed amount (epidermis + dermis + receptor fluid) was 4.70 ± 2.08 µg/cm2 (approximately 1.94%) of the applied dose. When formulated with water, the absorbed amount was 5.86 ± 2.68 µg/cm2 (approximately 2.58%).
The plasma pharmacokinetics, excretion balance, and tissue distribution of 6-hydroxyindole were studied in Wistar Han Ico:WI (IOPS AF/Han) rats.20 Twelve rats of each sex received a single dose of 25 mg/kg body weight 6-hydroxyindole(benzene ring-U-14C) in a suspension of 0.5% aq carboxymethylcellulose on clipped dorsum skin. The material was applied under a nonocclusive dressing for 30 minutes and then rinsed with water. Blood samples from designated animals were collected at 1, 2, 4, 6, 8, 24, 48, and 72 hours after dressing removal. Urine, feces, and cage washes were collected from designated animals during a 24-hour predosing period and during the periods of 0 to 6, 6 to 24 (urine), or 0 to 24 hours (feces) and 24 to 48, 48 to 72, 72 to 96, 96 to 120 h, 120 to 144 h, and 144 to 168 h (urine and feces) after dosing. Cages were carefully rinsed after each collection of feces. All samples were frozen until analysis. All animals were killed at the end of the study and a wide range of tissues and organs were sampled. For each sample, total radioactivity was evaluated by liquid scintillation counting of plasma, urine, feces, and rinse water. The minimal fraction of oral dose absorbed was derived from excretion data.
Total radioactivity levels in plasma reached a maximum of 1 or 2 hours after dosing with a mean Cmax value of 282 ± 137 ngeq/mL (233 ngeq/mL in males and 396 ngeq/mL in females). The plasma radioactivity levels decreased in a biphasic manner. By 4-hour postdosing, plasma levels were 33% of Cmax, with a slower decline until the last quantifiable time point at 24 hours postdosing (mean 35.7 ngeq/mL; 30.1 ngeq/mL in males and 39.4 ngeq/mL in females). The male rats did have a second minor peak at 6 hours. The mean systemic exposure (AUC [0-∞]) was 2809 ngeq-h/mL for the sexes combined. The mean total CE of urine, feces, and cage wash over the 168-hour period was 5.6% of the radioactive dose (sexes combined). Radioactivity was excreted equally in urine and feces with slow elimination over 96 hours (77% mean for both sexes). Most of the administered dose was recovered in the dressing (68%) and in the skin at the application site (11%). The maximal fraction of the absorbed dose was 18% in both sexes (16% in males and 19% in females). Tissue levels of radioactivity were virtually nonquantifiable. 20
Toxicological Studies
Acute Toxicity
Oral: Nonhuman
The acute oral toxicity of 6-hydroxyindole was tested in Crl:CD(Sprague Dawley [SD])BR rats. 11,21 Groups of 5 male and 5 female rats received a single dose of the test substance at 600 mg/kg body weight in a volume of 10 mL/kg. The vehicle was a 30% aq solution of polyethylene glycol (PEG) 6. The rats were observed twice daily for mortalities and daily for clinical signs of toxicity for 14 days. One male rat died immediately after dosing and a second male died the day after dosing. Clinical signs included hunched posture, hypoactivity, and prostration on the day of dosing. No clinical signs of toxicity were observed in the surviving animals after the dosing day. No abnormal weight gain was observed. At necropsy, the animals that survived to day 14 had abnormalities in the spleen of 1 male and in the uterus of 1 female. The remaining animals appeared normal. The study concluded that the maximum tolerated dose was less than 600 mg/kg body weight and the oral median lethal dose (LD50) was greater than 600 mg/kg body weight. In a preliminary range-finding study, 4 of 4 animals died at 1200 mg/kg body weight and 1 of 4 animals died at 800 mg/kg body weight.
Dermal: Nonhuman
The acute dermal toxicity of 6-hydroxyindole was tested in Crl:CD(SD)BR rats. 11,22 Groups of 5 male and 5 female rats received a single dose of the test substance at 2000 mg/kg body weight on shaved dorsal skin and occluded with a moistened porous gauze dressing. At the end of the 24-hour exposure period, remaining test substance was washed off with water. Animals were examined immediately, 30 minutes, 1-, 2-, and 4-hour posttreatment and then daily for 14 days. There were no mortalities. Brown staining of the fur at the application site was the only clinical sign. Female body weight gain was lower than normal, and the uteri of 3 of 5 females had red discoloration and were distended. These effects were discounted by the study authors. The study concluded that the maximal nonlethal dose of 6-hydroxyindole was greater than 2000 mg/kg.
A study similar to the one mentioned earlier was conducted using SD rats with the same dose of 6-hydroxyindole. 23 No mortalities, clinical signs, or cutaneous reactions were observed. At necropsy, no macroscopic abnormalities were apparent. The LD50 for 6-hydroxyindole in this acute dermal rat study was greater than 2000 mg/kg body weight.
Inhalation: Nonhuman
Groups of 5 male and 5 female Wistar rats were utilized in an acute inhalation study of 100% 6-hydroxyindole powder. 24 The animals were exposed in a nose-only chamber for 4 hours to an aerosol of the test material at 2000 mg/m3. Rats were observed for clinical signs of toxicity for 14 days and body weights were recorded daily. No mortalities or clinical signs were observed. No macroscopic or microscopic abnormalities of the lung or other tissues were observed at necropsy. The LC50 for 6-hydroxyindole was greater than 2000 mg/m3.
Repeated Dose Toxicity
Oral: Nonhuman
The potential for oral toxicity to 6-hydroxyindole was investigated in Crl:CD(SD)BR rats. 25 Dose groups were comprised of 10 animals of each sex and received 0, 30, 100, or 300 mg/kg body weight of the test material in a 30% aq PEG-6 solution. The test material was administered by oral gavage once daily for 13 weeks. Clinical signs of toxicity and mortality were observed daily, and feed consumption and body weights were recorded weekly. Ophthalmoscopic examinations were performed before and after the treatment period in the control and high-dose group animals. At week 13, complete hematology and blood chemistry investigations and urinalysis were performed. All animals were killed at the end of the treatment period. Major organs were weighed and a detailed necropsy was performed in all animals.
All animals survived until necropsy. Hair loss and scabbing were noted in some animals in all-dose groups. No clinical signs of toxicity were observed. No irregularities were observed during the ophthalmoscopic examinations. There were no treatment-related changes in body weight gains. Feed consumption was consistently higher in female rats in the high-dose group throughout the study, but this was not considered an adverse effect. The female rats in the high-dose group had minor hematological changes compared to the controls, with statistically significant dose-related increase in mean cell hemoglobin. There was a 2-fold increase in both alanine aminotransferase and aspartate aminotransferase levels in the plasma of the high-dose females. No indication of this effect was noted in the mid-dose group. A significant dose-related elevation in blood cholesterol and inorganic phosphate levels were noted in female rats, but the values were still within historical control ranges. No treatment-related effects were noted in the urinalysis.
Increased absolute and relative liver weights were observed in both sexes in the high-dose group when compared to controls (15%-18% above controls). Increased absolute and relative spleen and kidney weights were also observed in both high-dose groups (13%-24% and 11%-15% above controls, respectively). However, the changes in the spleen and kidney were within historical control ranges and there were no histopathological changes. No treatment-related macroscopic changes were noted at necropsy. Of 10 females in the high-dose group, 4 had hepatocyte hypertrophy. No other significant abnormalities were observed in the histopathological examination. The authors concluded that the no observed adverse effect level (NOAEL) was 100 mg/kg body weight/d. 25
Reproductive and Developmental Toxicity
In a teratogenicity study, mated female Crl:CD(SD)BR rats received 6-hydroxyindole by gavage on days 6 to 15 of gestation. 26 The doses used were based on the results of a range-finding study. In the main study, groups of 24 rats received 0, 50, 150, or 450 mg/kg body weight/d of the test material in a suspension of 30% aq PEG-6 solution. Body weights were recorded on gestation days 0, 6 to 15, and 20, and feed consumption was measured on gestation days 0 to 6, 6 to 9, 9 to 15, and 15 to 20. Dams were killed on gestation day 20. Complete necropsy and macroscopic examination of the organs were performed. Fetuses were removed, sexed, weighed, and examined externally. Number of corpora lutea, implantation sites, resorption sites, and fetuses were recorded. The fetuses were examined for anomalies.
Severe clinical reactions (including hypoactivity, hunched posture, and piloerection) were observed at the onset of dosing in the 450 mg/kg dose group and 14 of 24 animals were killed. The remaining 10 animals in this group and an additional 14 animals were given 300 mg/kg/d of the test material from gestation day 6. At 300 mg/kg/d, the maternal effects observed were slightly lower body weight gains and food intake from gestation days 6 to 9. Increased salivation at 300 mg/kg/d and greenish urine at 150 mg/kg/d were also observed in the dams. Body weights and feed consumption were decreased at higher doses. On gestation day 20, the dams did not have any observable treatment-related toxic effects. Preimplantation losses were similar or lower than those of the controls. Mean fetal sex ratio was similar in all groups, and mean fetal weight was statistically significantly reduced at 300 mg/kg/d. Reduced ossification in most of the fetal bones was noted in the 150 and 300 mg/kg/d dose groups. In the 50 mg/kg/d fetuses, there was a slight but nonspecific indication of reduced ossification. The maternal NOAEL was 150 mg/kg body weight and the fetal NOAEL was 50 mg/kg body weight in this rat teratology study. 26
In another teratogenicity study, mated female SD rats received 6-hydroxyindole by gavage on days 6 to 19 of gestation. 27 Groups of 24 rats received 0, 15, 60, or 240 mg/kg body weight of the test material in a suspension of 30% aq PEG-6. Maternal clinical signs were monitored daily. Body weights were recorded every 3 days and feed consumption was measured over 3-day periods. Dams were killed on gestation day 20. Organ abnormalities, pregnancy status, number of corpora lutea, implantation sites, resorption sites, and live and dead fetuses were recorded. The fetuses were weighed, sexed, and examined for soft-tissue and skeletal abnormalities.
No mortalities were observed during the study. Ptyalism in 4 rats of the 240 mg/kg/d dose group was observed. No treatment-related changes in body weight, body weight gains, or feed consumption were observed. Live fetuses were observed in 23, 24, 24, and 22 females in the 0, 15, 60, and 240 mg/kg dose groups, respectively. No treatment-related findings were observed at necropsy. In the 60 and 240 mg/kg dose groups, preimplantation loss was actually lower (statistically significant) than in the controls. The mean numbers of resorptions and dead fetuses and fetal sex ratio were comparable to controls. The mean fetal body weight was slightly lower, but not statistically significant, in the high-dose group. No treatment-related visceral anomalies were noted. Retardation of general ossification of fetuses was observed in the high-dose group, but there were no bone malformations. The maternal NOEL was 240 mg/kg body weight/d and the fetal NOEL was 60 mg/kg body weight/d in this rat teratology study. 27
Genotoxicity
In Vitro
The potential of 6-hydroxyindole to induce gene mutation was studied in Salmonella typhimurium strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 and in Escherichia coli strain WP2 uvrA using the reverse mutation assay. 28 The assay was performed with and without S9 metabolic activation at concentrations of 8, 40, 200, 1000, or 5000 μg/plate. A dose-dependent increase in revertant colony numbers was observed with and without S9 in TA 1535. It was concluded that 6-hydroxyindole was mutagenic with and without metabolic activation in S typhimurium strain TA 1535.
6-Hydroxyindole was studied for cell mutation in mouse lymphoma L5178Y TK+/− cells in 2 independent experiments. 29 Concentrations tested were 10, 40, 60, or 100 µg/mL, and the experiments were performed with and without S9 metabolic activation. In the first experiment, statistically significant positive effects were observed at 10 and 40 μg/mL with S9 activation, but there was no dose-dependent relationship. No statistically significant positive effects with or without S9 activation were observed in the second experiment. Since the results of the first experiment were not reproducible and had no trend, it was concluded that 6-hydroxyindole was not mutagenic in this assay.
The potential of 6-hydroxyindole to induce chromosomal aberrations was studied in Chinese hamster ovary (CHO) cells. 30 The study was comprised of 2 experiments, with the concentrations tested with S9 metabolic activation being 10, 50, or 100 μg/mL, and the concentrations tested without S9 being 1, 5, and 10 μg/mL for both experiments. Harvest times were 24 and 48 hours. In the first experiment, the test material induced a statistically significant increase in the number of cells with structural chromosomal aberrations at 100 μg/mL. The frequency was within historical control ranges, but the number of cells with aberrations was elevated and the types of rearrangements observed were acceptable indicators of clastogenicity. An increased frequency of aberrations was observed at 10 µg/mL without metabolic activation at 48 hours. Only 21 cells were scored, but the percentage of aberrations was very high (14%). In the second experiment, an increased number of cells with structural chromosomal aberrations were observed at the highest concentration (not described) with metabolic activation at 48 hours and without metabolic activation at 24 hours. Because the study yielded positive results at different concentrations and harvest times but clastogenic properties were observed in the cells, the authors concluded the study was equivocal.
The genotoxic potential of 6-hydroxyindole was studied in a chromosome aberration study using human peripheral blood lymphocytes. 31 In this 2-part study, the test material was tested up to 67.31 and 892.5 μg/mL, without and with S9 metabolic activation, respectively, in experiment 1, and in experiment 2, the test material was tested up to 25.03 and 900 μg/mL, without and with S9, respectively. Incubation for cells with metabolic activation was 3 and 20 hours for cells without metabolic activation. In both experiments, statistically and biologically significant increased number of cells with structural chromosomal aberrations was observed both with and without metabolic activation. Aberrations included chromatid and chromosome deletions and exchanges. Under the conditions of this study, 6-hydroxyindole was considered clastogenic.
In Vivo
In a bone marrow chromosome aberration test, groups of 5 male and 5 female SD rats received a single dose of 150, 300, or 600 mg/kg body weight 6-hydroxyindole in 0.5% aq methylcellulose solution via intragastric gavage. 32 The rats were killed 18 or 42 hours after dosing. Clinical signs of toxicity were recorded in the 600 mg/kg dose group. No statistically significant or biologically relevant increase in the incidence of cells with chromosome aberrations was observed at any dose and time point. The study concluded 6-hydroxyindole was not clastogenic.
The genotoxic potential of 6-hydroxyindole was studied in a micronucleus test using CD-1 mice. 33 Groups of 5 mice of each sex received single doses of 0, 50, 250, or 500 mg/kg body weight 6-hydroxyindole in 30% aq PEG via intragastric gavage (PEG chain length not given). Negative and positive controls were within Organization for Economic Cooperation and Development (OECD) guidelines. Bone marrow was collected at 24, 48, or 72 hours. No statistically significant or biologically relevant increase in the incidence of micronucleated polychromatic cells was observed. However, the groups of mice did not exhibit variation in the ratio of polychromatic erythrocytes (PCEs) to normochromatic erythrocytes (NCEs), which would indicate relevant exposure of the bone marrow to the test material. The Scientific Committee on Consumer Products (SCCP) determined this study was inadequate because there was no evidence that the test agent reached the target organ and because the maximum tolerated dose chosen was lower than the first set of deaths observed during the range finding study. 11
The genotoxic potential of 6-hydroxyindole was studied in a micronucleus test using SD rats. 34 Groups of 5 mice of each sex received 0, 75, 150, or 300 mg/kg body weight 6-hydroxyindole via gavage on 2 consecutive days. Control groups were within OECD guidelines. Bone marrow cells were collected at 24 hours. There was no statistically significant or biologically relevant increase in the incidence of micronucleated polychromatic cells when compared to the vehicle control values. The treated rats were also not observed to have variation in the PCE–NCE ratio; however, there was no evidence that the test material reached the bone marrow of rats. The SCCP also determined this study was inadequate. 11
The potential for 6-hydroxyindole to induce unscheduled DNA synthesis (UDS) was assessed using male Wistar HanIbm:WST(SPF) rats. 35 Groups of 3 rats received single gavage doses of 150 or 1500 mg/kg body weight 6-hydroxyindole in PEG-6. The animals were killed either 2 or 16-hour posttreatment. The UDS was analyzed by autoradiography. Positive controls were within OECD guidelines. No evidence of UDS induction by 6-hydroxyindole was observed.
Carcinogenicity
6-Hydroxyindole was assessed for carcinogenicity in a 105-week study in Wistar Han Ico:WI (IOPS AF/Han) rats. 36 The study was preceded by an 8-week toxicity study to determine the dose range. In the main experiment, groups of 50 male and 50 female rats received 0, 6, 25, or 100 mg/kg body weight/d 6-hydroxyindole in a suspension with 0.5% aq carboxymethylcellulose daily via gavage. The rats were observed daily for clinical signs of toxicity and mortality, while body weights and feed consumption were measured once a week for 13 weeks and then once every 4 weeks. Palpation for possible masses was carried out every 2 weeks from 6 weeks of treatment. Hematological investigations were performed at weeks 52 and 78 (differential white cell count in control and high-dose groups) and at study termination (red and white cell quantitative and qualitative assessment in all animals). In all groups, 8 additional animals were used for toxicokinetic investigations, with blood sample taken on day 1 and at weeks 13 and 52 for measurement of test material in plasma. The rats were killed and necropsied at the end of the treatment period.
There was a slight increase in mortality rate in the males of the 100 mg/kg dose group; however, these were due to normal causes and suggested a slight shortening of time of onset of death. This did not occur in females. Mortalities in the remaining dose groups were comparable with the control group. Body weight gains in the 6 and 25 mg/kg dose groups and the 100 mg/kg dose group females were comparable to controls. Body weight gain was slightly lower for males in the 100 mg/kg dose group. There were no significant differences in feed consumption between the treated groups and the controls. The only clinical sign of toxicity was increased salivation and/or regurgitation in all treated animals. Palpable mass incidence, time of onset, and localization in the treated groups were comparable to controls. The only hematological changes were slight decreases in main red blood cell count, packed cell volume, and plasma hemoglobin in the 100 mg/kg dose groups, indicating anemia. No quantifiable levels of the test material were observed in the plasma of the treated animals. No gross findings or neoplastic or nonneoplastic lesions attributable to treatment with 6-hydroxyindole were observed. This study concluded that the NOAEL for general toxicity was 25 mg/kg body weight/d and the NOEL for carcinogenicity was 100 mg/kg body weight/d. 36
Irritation and Sensitization
Irritation
Dermal: Nonhuman
The irritation potential of 6-hydroxyindole was assessed in 3 male New Zealand White rabbits. 37 Approximately 0.5 g of the test substance (tested as received from the test sponsor) moistened with water was applied to an intact area (6.25 cm2) of skin and then semioccluded. The patches were removed after 4 hours and remaining test substance was rinsed off. No skin reactions were observed in any of the rabbits. A slight yellow staining was observed in 2 of 3 rabbits during all observation periods. The primary irritation index was calculated to be 0.0. It was concluded that 6-hydroxyindole was nonirritating to rabbit skin.
In a 14-day irritation study, 3 male and 3 female New Zealand White rabbits received 2 mL of 6-hydroxyindole in paraffin oil daily on the left flank. 38 The test substance was initially applied at a concentration of 5%, but this was reduced to 1.5% on day 10. The right flank received paraffin oil as a control. The sites were not covered. Cutaneous reactions were evaluated in each animal immediately before each application and about 24 hours after the last application. From day 3 to the end of the study, very slight or well-defined erythema was observed on almost all of the animals on the treated flank. From days 8 to 15, moderate erythema was observed in 2 of 6 rabbits. Slight edema was noted in 2 of 6 rabbits on day 3 and slight to severe edema persisted in 1 rabbit until day 15. The maximum weekly mean irritation index was calculated to be 2.6. Very slight or well-defined erythema was noted in 3 of 6 rabbits on the control flank. These reactions were thought to be from contamination of the control with the test substance as the ears of rabbits could touch both flanks as severe erythema and crusts were observed on both ears of all animals from day 9 to end of the study. Body weight gains in the male rabbits were normal; however, body weight gains in females were decreased. The study concluded that 6-hydroxyindole was not well tolerated in rabbits.
The irritation potential of 6-hydroxyinidole was assessed in 5 male and 5 female Hartley Crl:(HA)BR guinea pigs. 39 Approximately 0.05 mL of the test substance at 5% w/w in PEG-6 at 30% in purified water was applied to the left clipped flanks of the animals once a day for 14 days. The test site was approximately 2 cm2. The test sites were neither covered nor were they rinsed. The right flanks received the vehicle as a control. Cutaneous reactions were evaluated in each animal immediately before each application and about 24 hours after the last application. No clinical signs of toxicity or mortalities were observed during the study. Body weight gains were within normal parameters. No cutaneous reactions were observed on the control flanks. Very slight erythema was observed on the treated flanks in 5 of 10 guinea pigs on days 9 and 10, very slight or well-defined erythema was observed in 7 of 10 guinea pigs on day 11, and then all animals exhibited very slight to moderate erythema from day 12 until end of the study. No edema was observed. Skin dryness was observed in 1 animal on day 11 and in 3 of 10 animals on day 12. The maximum weekly irritation index was calculated to be 1.1. It was concluded that 6-hydroxyindole was a slight to moderate skin irritant in guinea pigs.
The local tolerance of 6-hydroxyindole was tested in a 2-week guinea pig study. 40 Three male and 3 female Dunkin-Hartley guinea pigs received 0.05 mL of 20% (w/w) 6-hydroxyindole in paraffin oil on the left posterior flank daily. The test area was 2 cm2 and was not occluded. The right posterior flank received only the vehicle as a control. The concentration was lowered to 10% (w/w) on day 3 because the initial solution was poorly tolerated. The test sites were observed in each animal immediately before each treatment and about 24 hours after the last treatment. Because of skin lesions on both flanks, the animals were challenged with the test material and the vehicle after a 1-week rest period. An additional 3 untreated female animals were used as controls. The animals received 0.5 mL of the test material in paraffin oil at 10% on the anterior left flank and paraffin oil alone on the anterior right flank. The test sites were occluded for 6 hours and the skin was evaluated for reactions at 24, 48, and 72 hours after the dressing was removed.
No clinical signs of toxicity or mortalities were observed during the study. Body weight gains were within normal parameters. Moderate skin reactions were observed on the treated sites during the 2-week period with well-defined to severe erythema in all animals and slight edema in 3 of 3 males and 1 of 3 females. The reactions were first observed between days 3 and 6. Dryness of the skin and/or crusts was also noted in all animals. The weekly mean irritation indices were 3.6 for the first week and 2.3 for the second week. Cutaneous reactions, including very slight to moderate erythema in 2 of 3 males and all females, slight edema in 2 of 3 males and 1 of 3 females, and dryness and crusts, were also observed on the flank that received the vehicle only starting on day 6. No cutaneous reactions were observed in the control animals during the challenge. In the treated animals, very slight, well-defined, or moderate erythema was noted in 1 of 6, 1 of 6, and 3 of 6 animals, respectively, at 24 hours. Dryness of the skin was recorded in 1 animal. These reactions were also observed at 48 hours, except 2 of 6 animals had moderate erythema. The authors of the study did not state whether the reactions were in the same animals. Dryness of the skin was recorded in 3 animals. Moderate erythema persisted in 1 animal at 72 hours. Dryness of the skin was observed in 3 animals which was severe enough to preclude evaluation of erythema in 2 animals. Crusts were also noted in 1 animal. Microscopic examinations indicated that the lesions were due to sensitization. The study concluded that repeated topical applications of 10% and 20% 6-hydroxyindole in paraffin oil were badly tolerated in guinea pigs. 40
Ocular
The ocular irritation potential of 6-hydroxyindole was tested in a single male New Zealand White rabbit. 41 An aliquot of 0.1 g of 6-hydroxyindole (concentration not reported, used as supplied) was administered to the right eye of rabbit. Within 15 minutes, signs of irritation were observed in the treated eye. One hour after dosing, discharge, moderate hyperemia, and chemosis were observed. Opacity covered most of the cornea, making observation of the iris difficult. The test article was considered to be moderately to severely irritating to the rabbit eye in this study.
6-Hydroxyindole at 5% in 30% aq PEG-6 solution was tested for ocular irritation potential in 3 female New Zealand White rabbits. 42 The right eye of each rabbit was instilled with 0.1 mL of the test substance and not rinsed. The untreated eye served as a control. Both eyes were examined at 1, 24, 48, and 72-hour posttreatment. No adverse reactions were reported in the eye of any of the rabbits. The study determined that 5% 6-hydroxyindole in 30% PEG-6 solution was not an ocular irritant in rabbits.
Sensitization
Dermal: Nonhuman
The Buehler method was utilized to study the sensitization potential of 20% 6-hydroxyindole in paraffin oil in Dunkin-Hartley guinea pigs. 43 There were 10 guinea pigs in the control group (5 of each sex) and 20 guinea pigs in the test group (10 of each sex). During the induction period, animals in the treatment group received a 0.5 mL application of the test material on days 1, 8, and 15 on a 4 cm2 area of the anterior left flank that was occluded for 6 hours. The anterior right flank of these animals received the vehicle control. After a 2-week rest period, the guinea pigs were challenged with 0.5 mL of 20% test material on the posterior right flank and 0.5 mL of the vehicle control on the posterior left flank. The skin was assessed for cutaneous reactions 24, 48, and 72 hours after the challenge patches were removed. No clinical signs of toxicity or mortalities were observed. Very slight erythema was noted in 16 of 20 and 12 of 20 animals on days 9 and 16, respectively, of the induction period. No skin reactions were observed in the control animals in the challenge period, but the treated group had slight erythema in 3 of 20, 5 of 20, and 3 of 20 animals at 24, 48, and 72 hours, respectively. The study concluded that 20% 6-hydroxyindole in paraffin oil induced sensitization reactions in 25% of the guinea pigs.
A similar Buehler sensitization study was performed using 50% (w/w) 6-hydroxyindole in 30% PEG-6 in purified water for both the induction and the challenge phases. 44 No clinical signs or mortalities were observed during the study. A brown coloration of the skin was observed during the induction period in all animals of the treated group that may have masked erythema reactions. This was also observed at the 24-hour challenge reading. Discrete or moderate erythema was noted in 6 of 20 and 6 of 20 animals, respectively, at the 48-hour reading. Discrete, moderate, or intense erythema was observed in 4 of 20, 7 of 20, and 1 of 20 animals, respectively, at the 72-hour reading. Edema was observed in 1 animal. In both the treatment and control groups, skin dryness was observed. The study concluded that 50% 6-hydroxyindole in 30% PEG-6 induced delayed contact hypersensitivity in 55% of the guinea pigs.
In another Buehler sensitization study, 5% (w/w) 6-hydroxyindole in a solution of 30% PEG-6 in purified water was studied in guinea pigs for both the induction and the challenge phases. 45 No clinical signs or mortalities were noted during the study. Discrete erythema reactions were observed in the treated animals during the induction phase. Brown coloration of the skin was observed after the challenge application, which could have masked discrete erythema at the 24-hour reading. Discrete or moderate erythema was noted at the 48-hour reading in 6 of 20 and 6 of 20 animals, respectively. Discrete, moderate, or intense erythema was observed in 4 of 10, 7 of 20, and 1 of 20 animals, respectively, during the 72-hour reading. One animal was observed with edema. Dryness of the skin was recorded in a few animals of both groups. The study concluded that 5% 6-hydroxyindole in 30% PEG-6 induced delayed contact hypersensitivity in 30% of the guinea pigs.
The dermal sensitization potential of 6-hydroxyindole in PEG 6–water (85:10) was evaluated with a Magnusson-Kligman maximization study. 46 The test group consisted of 10 male and 10 female Dunkin Hartley guinea pigs, while the negative control group had 5 animals per sex. A concentration of 0.5% was used for the intradermal induction, 5% for topical induction, and 5% for the topical challenge patch. Dose volumes were 0.1 mL for the intradermal induction and 0.5 mL for the topical induction and challenge. Skin was examined for reactions 24 and 48 hours after removal of the challenge patch. At the 24-hour examination, 2 male and 2 female test animals had slight erythema and 3 females had well-defined erythema. Slight erythema remained in the same in 5 females at 48 hours, while the reactions had resolved in the males. No reactions were observed in control animals. The author concluded that the responses were individual irritation reactions and the substance was nonsensitizing; however, the SCCP concluded from this study that the substance caused sensitization in 40% of the test animals because there was no irritation during topical induction.
A local lymph node assay (LLNA) was performed using 6-hydroxyindole. 47 The CBA/J female mice were divided into groups of 4 and received 0.2%, 0.5%, 1%, 2%, or 5% of the test material in acetone/olive oil (4:1, v/v) on the ear surface (25 µL) once daily for 3 consecutive days. A positive control group received 25% α-hexylcinnamaldehyde and a negative control group received just the vehicle. Ear thickness was measured on days 1, 2, 3, and 6. On day 6, all animals were injected intravenously with ( 3 H) methyl thymidine and the proliferation of lymphocytes in the draining lymph nodes was measured. The stimulation indices (SIs) were calculated.
No clinical signs of toxicity or deaths occurred during the treatment period in any dose group. No cutaneous reaction or increases in ear thickness were observed in the treated animals. Significant dose-related lymphoproliferation was observed in all test groups and the positive control. The SI were 3.77, 4.63, 8.82, 8.07, and 19.17 for the 0.2%, 0.5%, 1%, 2%, and 5% dose groups, respectively. The estimated concentrations for an SI of 3 (EC3) was not calculated. The study concluded that 6-hydroxyindole induced delayed contact hypersensitivity in this LLNA. 47
Another LLNA was performed with 6-hydroxyindole using the procedures described earlier. 48 CBA/J female mice were divided into groups of 8 and received 0.02%, 0.05%, 0.1%, 0.2%, 0.5%, or 1% of the test material in acetone/olive oil (4:1, v/v). No clinical signs of toxicity or deaths occurred during the treatment period in any dose group. No cutaneous reaction or increases in ear thickness were observed in the treated animals. Significant dose-related lymphoproliferation was observed at concentrations ≥0.2% and the positive control. The SIs were 0.77, 0.98, 1.26, 6.78, 10.51, and 8.74 in 0.02%, 0.05%, 0.1%, 0.2%, 0.5%, and 1% dose groups, respectively. The EC3 was not calculated. The study concluded that 6-hydroxyindole induced delayed contact hypersensitivity at concentrations ≥0.2% in this LLNA.
Quantitative structure–activity relationship
A TOPS-MODE quantitative structure–activity relationship (QSAR) model was utilized to predict the sensitization potential of all hair dye ingredients registered in Europe (229 substances as of 2004). 7 The model predicted 6-hydroxyindole to be a moderate to strong sensitizer.
Phototoxicity
The phototoxicity and photoallergy potential of 6-hydroxyindole in paraffin oil was studied in Dunkin Hartley guinea pigs. 49 The study was consisted of 3 phases. In the first phase, groups of 5 (vehicle control irradiated animals and nonirradiated treated animals) or 10 (irradiated treated animals) received 0% or 5% of the 6-hydroxyindole in paraffin oil at a dose volume of 0.2 mL on the anterior scapula. The test sites were irradiated with 0.1 Joules/cm2 ultraviolet (UV) B followed by 9 Joules/cm2 UVA from a Toxicotronic UV lamp(312/365 nm) 30 minutes after application of the test substance. Cutaneous reactions were evaluated 1, 6, and 24 hours. Very slight cutaneous reactions were observed in most of the animals. In the second phase, the animals continued to receive dose applications of 5% for days 2 to 4; however, the concentration was lowered to 0.1% on days 7 and 8. Weak to severe erythema with edema was noted in all treated animals that were irradiated on days 5, 8, and 9 and in treated animals that were not irradiated on day 5. These reactions were considered to be due to poor local tolerance to the test material following repeated exposure. In the final phase, the animals had a 20-day rest period, after which 0% or 5% challenge applications were made to the distal part of the back. The designated groups were then irradiated 30 minutes later at suberythemal doses of UVA on the right flank or UVB on the left flank. Cutaneous reactions were evaluated at 1, 6, 24, and 48 hours. Well-defined to severe erythema was observed at 48 hours in half of the treated animals that were irradiated. No reactions were observed in the other groups. It was determined that the 5% 6-hydroxyindole in paraffin oil was a photosensitizer, but not phototoxic.
Another dermal phototoxicity and photoallergenicity study following the same procedure was performed using 2% 6-hydroxyindole in paraffin oil in the challenge phase. 50 Following the single dermal application on day 1, very slight cutaneous reactions were observed in most animals. Weak to severe erythema with edema was noted in all animals that were treated with the test substance (irradiated and nonirradiated) on days 8 and 9. Again, these reactions were considered to be due to poor local tolerance of the test material following repeated exposure. After the challenge phase, no distinct cutaneous reactions were observed in the irradiated controls and in the vehicle-treated and irradiated controls. In the treated, nonirradiated group, weak to well-defined erythema was observed in most of the animals, and in the treated, irradiated group, weak to severe erythema was observed in over half of the animals. This study concluded that 2% 6-hydroxyindole in paraffin oil was not phototoxic or photosensitizing; however, cutaneous reactions due to the poor tolerance to repeated dermal applications may have masked photosensitization reactions.
A third dermal phototoxicity and photoallergenicity study following the same procedures was performed using 2% 6-hydroxyindole in 30% aq PEG-6. 51 Following the single dermal application on day 1, very slight cutaneous reactions were observed in most animals. Weak to well-defined erythema with dryness was noted in all animals that were treated with the test substance (irradiated and nonirradiated) on days 8 and 9. After the challenge phase, no distinct cutaneous reactions were observed in the irradiated controls and in the vehicle-treated and irradiated controls. In the treated, nonirradiated group, weak erythema was noted in 1 animal at 1 and 4 hours and another animal had erythema at 24 and 48 hours. In the treated, irradiated group, questionable or weak erythema was observed in 70% of the animals and weak or well-defined erythema was observed in 1 animal at 1 and 4 hours. A weak erythema persisted in 2 animals at 24 and 48 hours. This study concluded that 2% 6-hydroxyindole in PEG-6 was not phototoxic or photosensitizing.
Epidemiology
6-Hydroxyindole is an oxidative hair dye ingredient. Although the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair dye use and disease, such studies do provide broad information. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers. A detailed summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/cir-findings.
Risk Assessment
The CIR has calculated the margin of safety for 6-hydroxyindole to be 530. To obtain this value, the systemic exposure dose (SED) of 0.094 mg/kg was determined based on the maximum absorption through the skin (8.09 μg/cm2 from an in vitro percutaneous absorption study 11 ), typical body weight (60 kg), and skin surface area (700 cm2 for scalp surface area 11 ), which was then divided into the NOAEL from an oral rat developmental toxicity study (50 mg/kg). This NOAEL was chosen because it represents the most conservative indication of a dose at which no adverse effects were seen.
Summary
6-Hydroxyindole is an oxidative hair dye ingredient with 105 uses in cosmetics reported to the FDA, with a maximum use concentration of 0.5% based on an industry survey.
The European Commission concluded that 6-hydroxyindole would not pose a health risk to the consumer when used as an ingredient in oxidative hair dye formulations as long as the maximum concentration applied to hair does not exceed 0.5%. In 2012, however, concern was expressed in Europe regarding the potential induction of sensitization that may result from the currently recommended self-test procedure for hair dyes.
A single oral dose of 300 mg/kg radiolabeled 6-hydroxyindole in a rat pharmacokinetics and excretion study resulted in a mean plasma Cmax value of 132µgeq/mL at 1 hour after dosing and the mean systemic exposure of 799 µgeq-h/mL. The mean total CE of the radioactive dose over the 168-hour period was 95.4%, with most of the dose excreted rapidly in the urine. A single dermal dose of 25 mg/kg radiolabeled 6-hydroxyindole in a similar study resulted in a mean Cmax value of 282 ± 137ngeq/mL at 1 to 2 hours after dosing and the mean systemic exposure of 2809 ngeq-h/mL. The mean total CE of urine, feces, and cage wash over the 168-hour period was 5.6% of the radioactive dose. Radioactivity was excreted equally in urine and feces with slow elimination over 96 hours. Tissue levels of radioactivity were virtually nonquantifiable.
An in vitro percutaneous absorption study in human skin samples determined the absorbed amount of radiolabeled 6-hydroxyindole was 4.70 ± 2.08 µg/cm2 of the applied dose when formulated with hydrogen peroxide and 5.86 ± 2.68 µg/cm2 when formulated with water.
The maximum tolerated dose of 6-hydroxyindole in an acute oral toxicity study in rats was less than 600 mg/kg body weight and the oral LD50 was greater than 600 mg/kg body weight. In dermal acute toxicity studies in rats, the maximal nonlethal dose of 6-hydroxyindole was greater than 2000 mg/kg. The LC50 for 6-hydroxyindole in an acute inhalation study was greater than 2000 mg/m3.
In an oral repeated dose study of 6-hydroxyindole up to 300 mg/kg/d in rats, the NOAEL was 100 mg/kg body weight/d. Female high-dose rats had a statistically significant dose-related increase in mean cell hemoglobin and increases in both alanine aminotransferase and aspartate aminotransferase levels in the plasma.
Increased absolute and relative liver weights were observed in both sexes in the high-dose group, and some females in the high-dose group had hepatocyte hypertrophy.
The maternal NOAEL was 150 mg/kg body weight and the fetal NOAEL was 50 mg/kg body weight in an oral rat teratology study of 6-hydroxyindole up to 450 mg/kg/d. High dose dams had slightly lower body weight gains and feed intake and increased salivation. Fetuses had reduced ossification in most fetal bones at 150 and 300 mg/kg/d as well as statistically significantly reduced mean fetal weight at 300 mg/kg/d. In a second oral teratology study in rats, the maternal NOEL was 240 mg/kg body weight/day 6-hydroxyindole, which was the highest dose tested. The fetal NOEL was 60 mg/kg body weight/day 6-hydroxyindole, with delayed general ossification of fetuses occurring at 240 mg/kg/d.
6-Hydroxyindole yielded positive results in a bacterial reverse mutation assay and a chromosome aberration study of human peripheral blood lymphocytes. Results were equivocal in a CHO chromosomal aberration study. 6-Hydroxyindole was not genotoxic in an in vitro mouse lymphoma cell mutation study and in an in vivo bone marrow chromosome aberration test and an in vivo UDS assay.
In a 105-week carcinogenicity study of 6-hydroxyindole in rats, the NOAEL for general toxicity was 25 mg/kg body weight/d and the NOEL for carcinogenicity was 100 mg/kg body weight/d, the highest dose tested. Decreased body weight gains, main red blood cell count, packed cell volume, and plasma were observed in the high-dose group.
6-Hydroxyindole was not irritating to rabbit skin in a single application study that tested 0.5 g of the substance neat, but it was not well tolerated in another rabbit study where it was applied daily for 2 weeks at a concentration of 1.55% to 5%. A 2-week guinea pig study concluded that 5% 6-hydroxyindole was a slight to moderate skin irritant. 6-Hydroxyindole was moderately to severely irritating to the rabbit eye when tested neat, but was not an ocular irritant when tested at 5% in an aq 30% PEG-6 solution.
Positive sensitization reactions were observed in guinea pig studies of 6-hydroxyindole at concentrations of 10% to 20% in paraffin oil, 5% to 50% in 30% PEG-6, and 5% in PEG-6/water. Two LLNA studies concluded that 6-hydroxyindole induced delayed contact hypersensitivity. A QSAR model predicted 6-hydroxyindole to be a moderate to strong sensitizer.
6-Hydroxyindole was a photosensitizer in paraffin oil at a concentration of 5% when tested in guinea pigs; however it was not a photosensitizer in paraffin oil at a concentration of 2% or in PEG-6 solution at a concentration of 2%. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers. A margin of safety for 6-hydroxyindole was calculated by CIR to be 530 based on a SED of 0.094 mg/kg, 60 kg body weight, 700 cm2 scalp surface area, and NOAEL of 50 mg/kg for oral developmental toxicity, the most sensitive toxicological end point in the database.
Discussion
The Panel was concerned with data indicating that 6-hydroxyindole caused dermal irritation and sensitization, ocular irritation, and photosensitization. Additionally, the available genotoxicity data indicated the possibility of some genotoxic potential of 6-hydroxyindole. However, no photosensitization was observed in studies up to 2%, no carcinogenicity was observed at 100 mg/kg/d in an oral study, and no ocular irritation was observed in studies of 6-hydroxyindole at 5%. The Panel determined that none of these adverse effects would be likely at the maximum use concentration of 0.5%. Furthermore, a margin of safety for 6-hydroxyindole was calculated to be 530 based on an NOAEL of 50 mg/kg for oral developmental toxicity, the most sensitive toxicological end point available.
Although nitrosamine content of these hair dyes has not been reported, the Panel noted this ingredient potentially can be nitrosated. Accordingly, hair dyes containing 6-hydroxyindole should be formulated to avoid the formation of N-nitroso compounds.
The Panel recognized that 6-hydroxyindole functions as a hair dye ingredient and that hair dyes containing this ingredient, as coal tar hair dye products, are exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act, when the label bears a caution statement and patch test instructions for determining whether the product causes skin irritation. The Panel expects that following this procedure will prospectively identify individuals who would have an irritation/sensitization reaction and allow them to avoid significant exposures. The Panel was concerned that such self-testing might induce sensitization but concluded that this alone was not a sufficient basis for changing the current caution statement and patch test instructions to consumers at this time.
In considering hair dye epidemiology data, the Panel concluded that the available epidemiology studies are insufficient to scientifically support a causal relationship between hair dye use and cancer or other toxicologic end points, based on lack of strength of the associations and inconsistency of findings.
The Panel noted that the use of oxidative hair dye formulations involves exposure to precursors and coupling agents as well as to their reaction products. Although reaction intermediates may be formed, human exposure is to the precursors and coupling agents and to reaction products, not to reaction intermediates. The exposures to the precursors and couplers are low (they are consumed in the color forming reaction), and the exposures to reaction products are even lower (they are adsorbed onto the hair shaft itself and physically retained there). Therefore, it was the consensus of the Panel that the safety assessments of oxidative hair dyes are primarily determined by the toxicological evaluation of the ingredients (ie, precursors and coupling agents), rather than the reaction intermediates or products formed during use.
Conclusion
The Panel concluded 6-hydroxyindole is safe in the present practices of use and concentration for use in oxidative hair dye formulations.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from Lillian J. Gill, Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
