Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) has issued an amended safety assessment of 7 Hypericum perforatum-derived ingredients as used in cosmetics. A common name for this plant is St John wort. These ingredients function in cosmetics as skin-conditioning agents—miscellaneous and antimicrobial agents. The Panel reviewed relevant animal and human data related to the H perforatum-derived ingredients. Because formulators may use more than 1 botanical ingredient in a formulation, caution was urged to avoid levels of toxicological concern for constituent chemicals and impurities. The Panel concluded that H perforatum-derived ingredients were safe as cosmetic ingredients in the practices of use and concentration as described in this safety assessment.
Introduction
The Definitions and Functions of the Hypericum Perforatum-Derived Cosmetic Ingredients.
H perforatum extract;
H perforatum flower extract;
H perforatum flower/leaf extract;
H perforatum flower/leaf/stem extract;
H perforatum flower/twig extract;
H perforatum leaf extract;
H perforatum oil.
In 2001, the Cosmetic Ingredient Review (CIR) published a safety assessment of H perforatum extract and H perforatum oil as used in cosmetics,
2
finding insufficient data to determine that these ingredients were safe for use in cosmetics. Additional data needs were identified: current concentration of use data; function in cosmetics; photosensitization and phototoxicity data using visible light (550-610 nm; 5-10 J); gross pathology and histopathology in skin and other major organ systems associated with repeated dermal exposures; dermal reproductive/developmental toxicity data; skin irritation/sensitization data in humans on H perforatum oil; and ocular irritation data, if available.
Additional data have been submitted and are summarized subsequently along with new data discovered in the literature. Data on the major constituents of H perforatum are also included.
Since the original report was published, the name of H perforatum extract was changed to H perforatum flower/leaf/stem extract. 3 Since then, another ingredient named H perforatum extract, defined as an extract of the whole plant, has been added to the International Cosmetic Ingredient Dictionary and Handbook. 1
Original Safety Assessment
This is a summary of the data in the original safety assessment.
Hypericum perforatum extract is an extract of the capsules, flowers, leaves, and stem heads of the Hypericum, H perforatum. In 1998, it was reported to the Food and Drug Administration (FDA) that H perforatum extract and H perforatum oil were used in 64 and 11 cosmetic formulations, respectively. 2 One manufacturer reported that H perforatum extract is used at concentrations of ≤5%, and it was reported by another supplier that a mixture of H perforatum extract and propylene glycol is used at concentrations of 1% to 10%. In 1984, H perforatum extract and H perforatum oil were reported to be used at concentrations of ≤5% and unknown concentrations, respectively.
In male subjects, a single oral administration of Hypericum extract resulted in a nonlinear increase, with increasing dose in the amount of hypericin or pseudohypericin appearing in the plasma, and the increase was statistically significant for hypericin. With long-term dosing of Hypericum extract, steady state occurred after 14 days. The polyphenol fraction of H perforatum had immunostimulating activity on the mononuclear phagocyte system and cellular and humoral immunity, and the lipophilic portion had immunosuppressive activity on cellular and humoral immune responses.
The oral median lethal dose (LD50) values for rats and mice of mixtures containing H perforatum extract were >20 mL/kg. The minimum lethal subcutaneous dose of H perforatum using guinea pigs was 0.1 mL. The intraperitoneal LD50 values of the polyphenol, lipophile, and water soluble fractions of H perforatum were 780, 4300, and 2800 mg/kg, respectively. Signs of toxicity were observed in Awasi sheep fed H perforatum flowers (4 g/kg) for 14 days. In a chronic study in which Long-Evans rats were fed H perforatum (5%), average daily weight gain was statistically significantly decreased when compared to control animals. Mixtures containing H perforatum extract and H perforatum oil were not irritants (up to 5%) or sensitizers (up to 5%) in animals. Hypericum perforatum is a primary photosensitizer in animals because of the pigment hypericin that causes photoactivated damage by absorbing visible light. A mixture containing H perforatum oil, butylene glycol, and water was not phototoxic. Mixtures containing H perforatum extract (0.5%) and H perforatum oil (0.1%) were non to slightly irritating, respectively, in rabbit eyes.
In an Ames test, a tincture of Hypericum had mutagenic effects at 20 mg/100 µL suspension, which the researchers attributed to flavonols. However, the origin of the plant and the mode of preparation of the tincture were considered to play a role in the mutagenic potential. In another Ames test, H perforatum (10 µL) had mutagenic activity; in testing fractions of 3 extracts, the mutagenic potential was found exclusively in quercetin, and hypericin was not mutagenic. Hypericum extract (500 µL) and hypericin were not genotoxic in unscheduled DNA synthesis assays using primary rat hepatocytes. Hypericum extract (4.00 µL/mg) was not mutagenic in a cell transformation assay using Syrian golden hamster embryo cells, and it was not genotoxic in a mouse fur spot test or in a chromosome aberration test.
A mixture of H perforatum oil, butylene glycol, and water was not irritating in clinical studies. In human testing, Hypericum extract did not appear to be toxic, although some undesirable drug interactions were observed.
Chemistry
Definition
The definitions and functions of these H perforatum-derived ingredients are provided in Table 1.
Constituents
Constituents of H perforatum are listed in Table 2.
Constituents Found in Hypericum Perforatum. 60

Hypericum perforatum flower contains not less than 0.08% of total hypericins expressed as hypericin calculated with reference to the dried drug.
4
–6
Constituents of H perforatum include: phloroglucinol derivatives: 0.2% to 4%, depending on the age of the herbal drug, mainly hyperforin and its homolog adhyperforin and furanohyperforin; naphthodianthrone: 0.06% to 0.4%, mainly pseudohypericin and hypericin, protohypericin, protopseudohypericin, cyclopseudohypericin, and skyrin derivatives; the amount of pseudohypericin is about 2 to 4 times higher than that of hypericin; flavonoids: 2% to 4%, mainly glycosides of the flavonol quercetin: hyperoside, rutin, isoquercitrin, quercitrin; also biflavones (I3, II8-biapigenin, amentoflavone); procyanidines: for example, procyanidine B2, tannins with catechin skeletal (6%-15%); xanthones: in trace amounts; essential oil: 0.1% to 0.25%; the essential oil of dried flowering tops contains as main compounds 2-methyloctane (16%) and α-pinene (10.6%). In the essential oil of leaves of Indian origin, 58 components were identified, α-pinene (67%) being dominant; the other components included caryophyllene, geranyl acetate, and nonane (each about 5%); other constituents: include small amounts of chlorogenic acid and other caffeoylquinic and p-coumaroylquinic acids and also free amino acids.
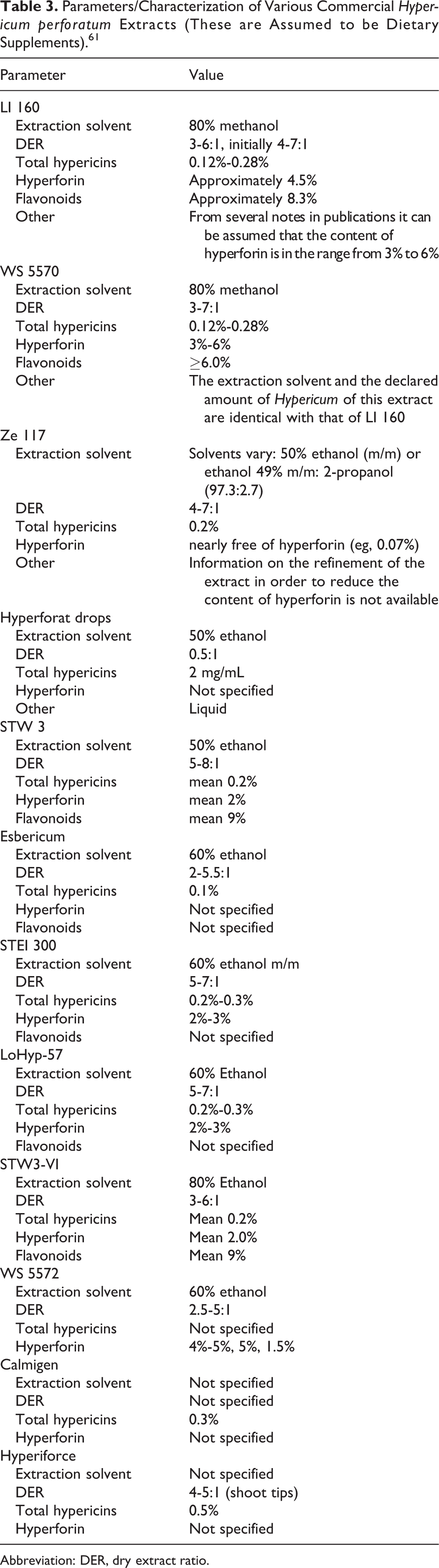
Information on the characterization of different commercial H perforatum extracts with regard to hypericins, hyperforin, and flavonoids are provided in Table 3.
Parameters/Characterization of Various Commercial Hypericum perforatum Extracts (These are Assumed to be Dietary Supplements). 61
Abbreviation: DER, dry extract ratio.
In a batch of St. John wort extract capsules, the label stated that they contained 300 mg of extract and 900 μg of hypericin. 7 Analysis found that the contents actually weighed 444 ± 20 mg and contained 840 ± 56 μg of hypericin and 11 ± 0.63 mg of hyperforin.
Method of Manufacture
It was reported that cosmetic grade H perforatum flower/leaf/stem extract is mostly extracted from the dried plant but may occasionally be extracted from fresh material. 3 Extraction solvents include water/propylene glycol, propylene glycol, 86% ethanol, 50% butylene glycol, water, sunflower oil, olive oil, caprylic/capric triglycerides, or glycerin. Solids in these extracts measure 0.1% to 5%. The hypericin content from an 86% ethanol (3% solids) extract of fresh plant materials was reported to be 60 to 65 μg/mL, and the hyperforin content was 240 to 900 μg/mL.
Use
Cosmetic
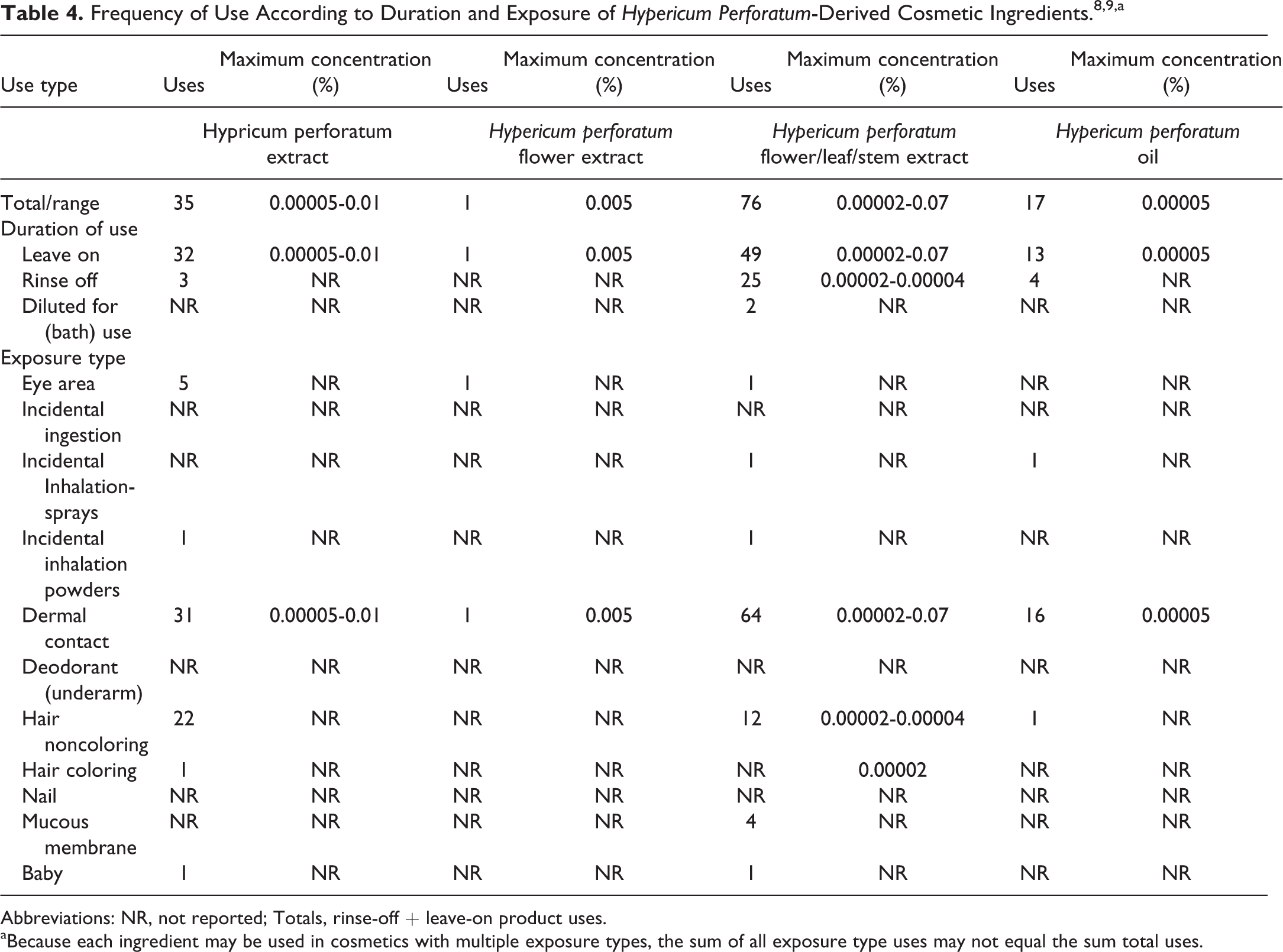
Data on ingredient usage are provided to the FDA Voluntary Cosmetic Registration Program (VCRP; Table 4). 8 A survey was conducted by the Personal Care Products Council (Council) of the maximum use concentrations for ingredients in this group. 9
Abbreviations: NR, not reported; Totals, rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Hypericum perforatum extract was reported to be used in 32 leave-on products (up to 0.01%), 3 rinse-off products (no use concentration reported), and 1 baby product (no use concentration reported). Hypericum perforatum flower was reported to be used in 1 leave-on product; maximum concentration of use was reported to be 0.005% in face and neck creams, lotions, and powders. Hypericum perforatum flower/leaf/stem extract is reported to be used in 49 leave-on products (up to 0.07% in body and hand creams, lotions, and powders) and in 25 rinse-off products (up to 0.00004% in shampoos and rinses), mostly in skin care products. The VCRP reports that it is also used in 2 products that are diluted for bath (no use concentration reported). There is 1 reported use in baby lotions, powders, and creams. Hypericum perforatum oil is reported to be used in 13 leave-on products and in 4 rinse-off products. Use concentration was only reported for skin preparations up to 0.00005%.
There were no reported uses or concentration of use for H perforatum flower/leaf extract, H perforatum flower/twig extract, and H perforatum leaf extract. Hypericum perforatum flower and Hypericum flower/leaf/stem extract are used at concentrations up to 0.07% in cosmetic products that may include loose powders of which airborne particles may be inhaled. The size distribution of the particles in cosmetic powders has not been reported. However, particles incidentally inhaled from cosmetic aerosols would likely be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 10 –15
Noncosmetic
Oral therapeutic use H perforatum was reported to be safe up to 900 mg/d (∼13 mg/kg/d) for humans. 16
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Dermal/percutaneous
Hypericin
Hypericin is absorbed through the intestinal epithelium by passive transcellular diffusion. 17 There was no hypericin detected in the plasma of Balb/c mice after administration to the ear (0.1%-1% in Beeler base) for 24 hours. 18 The distribution of hypericin-related fluorescence in the skin after dermal administration (1%) was concentrated in the stratum corneum and epidermis with only faint fluorescence in the dermis observed. At lower concentrations (0.1% and 0.01%), the fluorescence was concentrated only in the stratum corneum and was faint in the epidermis.
Oral
Hypericum perforatum extract
After a single oral dose of H perforatum extract (300 mg; tablet form; 900 μg hypericin + pseudohypericin), the mean serum level in subjects (n = 12) of total hypericin + pseudohypericin was 43 ng/mL and the mean skin blister fluid level was 5.3 ng/mL at 6 hours. 19 After steady state administration (1 tablet, 3×/d for 7 days), the mean serum level of total hypericin + pseudohypericin was 12.5 ng/mL and the mean skin blister fluid level was 2.8 ng/mL. The authors stated that these skin levels are far below hypericin skin levels that are estimated to be phototoxic (>100 ng/mL).
After a single oral dose of a H perforatum extract (1600 mg/kg in agarose gel; 1.35% isoquercitrin, 0.38% quercitrin, 3.26% rutin, 1.83% hyperoside) administered to male Sprague Dawley rats (n = 30; control n = 6), the quercetin plasma level increased rapidly and reached the maximum of about 700 ng/mL after 4 hours. 20 After 24 hours, 50% of the Cmax was still measurable. In contrast, the concentration level of isorhamnetin/tamarixetin increased much slower; the maximum was reached after 24 hours with a Cmax of 903 ng/mL. Repeated doses of H perforatum extract (1600 mg/kg/d for 8 days) caused a continuous increase in the plasma levels of quercetin and isorhamnetin for 5 days; after that time the concentration remained constant.
Short-term oral administration of H perforatum extract (300 mg, 3×/d) to humans resulted in a selective induction of CYP3A activity in the intestinal wall. 7 Hypericum perforatum did not alter the activities of CYP2C9, CYPlA2, or CYP2D6 after 2 weeks.
In 36 samples of breast milk from mothers (n = 5) who were taking H perforatum extract (300 mg times/d), hyperforin was present in the milk at 0.9% to 2.5% (infant hyperforin dose/kg body weight expressed as a percentage of the maternal hyperforin dose/kg body weight). 21 The plasma from 2 of the infants contained low levels of hyperforin (0.1 ng/mL).
Hyperforin was detected in the breast milk of a mother who took 3 H perforatum extract pills (3 × 300 mg/d; 0.12%-0.28% hypericins, ∼4.5% hyperforin). 22 Hyperforin and hypericin were below the limits of detection in the infant’s plasma.
Constituents
The half-lives for hypericin, pseudohypericin, hyperforin quercetin, and isorhamnetin were similar whether H perforatum extract (612 mg) was administered to subjects (n = 18) in 1 dose or daily for 14 days. 23
The Cmax of hyperforin was ∼370 ng/mL (∼690 nmol/L) at ∼3 hours after oral administration of an ethanol/water extract of H perforatum (0, 300 mg/kg; 5% hyperforin) to Sprague-Dawley rats (n = 5 for each sampling interval). 24 Blood samples were taken at 15 and 30 minutes and 1, 2, 4, 6, 8, and 24 hours.
In humans, the maximum plasma levels of ∼150 ng/mL hyperforin (∼280 nmol/L) were reached 3.5 hours after oral administration of a H perforatum ethanol/water extract. 24 In an open, single-dose, 4-way crossover study, the same H perforatum extract (300, 600, 1200 mg; in pill form) or a second extract (0.5% hyperforin) was orally administered to subjects (n = 6) for 8 days. Blood samples were taken at 0, 15, 30, and 45 minutes and 1, 1.5, 2.5, 3, 4, 6, 8, 10, 12, and 24 hours on days 1 and 8. Washout period was 3 days.
In a second human double-blind, placebo-controlled, parallel-group, 8-day study of H perforatum extract (300, 600, and 1200 mg; in pill form) or a second extract (0.5% hyperforin), the half-life and mean residence time were 9 and 12 hours, respectively. Hyperforin pharmacokinetics were linear up to the 600 mg dose. Increasing the doses to 900 or 1200 mg resulted in lower Cmax and area under the curve values than those expected from linear extrapolation of data from lower doses. Plasma concentration curves in volunteers fitted well in an open 2-compartment model. In the repeated-dose study, there was no accumulation of hyperforin in the plasma. The estimated steady state of hyperforin in plasma was ∼100 ng/mL (∼180 nmol/L).
Intravenous
Hypercicin
Intravenous administration of hypericin (2 mg/kg in 2% benzyl alcohol and saline) to rhesus monkeys (Maccaca mulatta; n = 3) resulted in a mean peak plasma concentration of 142 ± 45 μmol/L; elimination was biexponential with an average α half-life of 2.8 ± 0.3 hours and terminal half-life of 26 ± 14 hours. 25 Hypericin was not detected in the cerebrospinal fluid of any animal.
In vitro
Using human colonic Caco-2 cells as a model for human intestinal absorption, porcine capillary endothelial cells for the blood–brain barrier, and plexus choriodei epithelial cells for the blood–cerebrospinal fluid barrier, it was shown that orally ingested miquelianin (quercetin 3-O-beta-
Anti-Inflammatory Activity
Hypericum perforatum flower extract
Hypericum perforatum flower extracts (a hydroalcoholic extract, a lipophilic extract, and an ethylacetic fraction) provoked a dose-dependent reduction in Croton oil-induced ear edema in mice. 27 Inflammation was induced in the right ear of male albino Swiss mice (n = 10) by applying Croton oil, 80 mg dissolved in 15 mL vehicle with and without the test substances. The following vehicles were used: acetone for extracts, the ethylacetic fraction, hypericin, hyperforin dicyclohexylammonium salt, dicyclohexylamine and the relevant controls, ethanol:acetone (3:1, v/v) for hyperoside and its controls, ethanol:acetone (1:1, v/v) for adhyperforin, amentoflavone, isoquercitrin, and the relevant controls. The left ear remained untreated. Control animals were treated only with Croton oil.
The doses that inhibited by 50% (ID50) the Croton oil-induced ear edema in mice had the following order of activity: lipophilic extract (ID50 = 220 mg/cm2) > ethylacetic fraction (ID50 = 267 mg/cm2) > hydroalcoholic extract (ID50 >1000 mg/cm2). Amentoflavone (ID50 = 0.16 mmol/L/cm2), hypericin (ID50 = 0.25 mmol/L/cm2), hyperforin DHCA salt (ID50 = 0.25 mmol/L/cm2), and adhyperforin (ID50 = 0.30 mmol/L/cm2) had anti-inflammatory activity that was more potent or comparable to that of indomethacin (ID50 = 0.26 mmol/L/cm2), whereas isoquercitrin and hyperoside were less active (ID50 ∼ 1 mmol/L/cm2). As dicyclohexylamine alone was inactive, the effect of hyperforin DHCA salt can be attributed completely to the phloroglucinol moiety. The pharmacological activity and phytochemical profile of the tested extracts and fractions suggest that different constituents are involved in the topical antiphlogistic property of H perforatum in vivo.
Drug Interactions
Hypericin
In an open-label, fixed schedule study, subjects (n = 12) were administered tolbutamide (CYP2C9), caffeine (CYPlA2), dextromethorphan (CYP2D6), oral midazolam (intestinal wall and hepatic CYP3A), and intravenous midazolam (hepatic CYP3A). 7 Blood and urine samples were taken before and during treatment. Subjects continued to take the H perforatum extract for 14 days. There were no serious adverse events but some cases of hypoglycemia occurred during the study. The bioavailability of midazolam was reduced to 55% of the control value after 2 weeks of treatment. The authors conclude that H perforatum reduced the therapeutic efficacy of drugs metabolized by CYP3A, and this effect should be anticipated during long-term administration.
Toxicological Studies
Acute Toxicity
Intravenous
Hypericin
Intravenous administration of hypericin (2 mg/kg in 2% benzyl alcohol and saline) was well tolerated by rhesus monkeys (n = 3). 25 At a dose of 5 mg/kg, a transient severe photosensitivity rash was observed at 12 hours that resolved within 12 days. Edema and a pruritic erythematous rash with evolution to eschar were observed on the face and light-exposed skin. Mild anorexia and transient elevation in hepatic transaminases were observed.
Repeated-Dose Toxicity
Oral: nonhuman
Hypericum perforatum extract
Hypericum perforatum extract (900 and 2700 mg/kg) was orally administered to rats and dogs daily for 26 weeks. 16 Decreased body weight, slight changes in the hematological parameters, and changes in the clinical chemistry parameters, which indicate a slight load damage to the liver and kidneys, were observed in both dose groups. A mild hypertrophy of the zona glomerulosa of the adrenals was observed.
Oral: human
Hypericum perforatum extract
In a randomized, double-blind, crossover study, H perforatum extract (255-285 mg; 900 μg hypericin content) orally administered to healthy male subjects (n = 12) 3 times/day for 13 days had no effect on vasoconstrictor responses (VRs) of cutaneous blood flow or skin conductance response (SR). 28 The VR and SR were measured before treatment and at 0.5, 3, and 5 hours after the last dose was given. Systolic and diastolic blood pressures were monitored before the start of medication as well as on treatment days 11 and 14. Hypericum perforatum extract and the controls (25 mg amitriptyline, and placebo) were administered to the subjects with at least a 14-day wash out period between treatments.
Reproductive and Developmental Toxicity
Animal
Hypericum perforatum extract
There were no reproductive or developmental effects observed in a 2-generational study of H perforatum extract using CD-1 mice (n = 20). 29 The female mice were administered H perforatum (180 mg/kg in feed) for 2 weeks prior to mating through gestation. Males were not treated. Increases in body weight, body length, and head circumference (measurements taken from postnatal day 3 through adulthood) were similar between the 2 groups of offspring, regardless of gender. No differences in reaching physical milestones (ie, teeth eruptions, eye opening, external genitalia) were noted between the 2 groups. Reproductive capability, perinatal outcomes, and growth and development of the second-generation offspring were unaffected by parental exposure to H perforatum extract.
There were no clinical signs of maternal or developmental toxicity when pregnant Wistar rats (n = 15) were orally administered H perforatum extract (36 mg/kg/d in saline; 0.4% hypericin) during gestation days 9 to 15. 30 Maternal toxicity was evaluated through water and food intake, body weight gain, piloerection, locomotor activity, diarrhea, and mortality. Animals were killed on day 21 of gestation and necropsied. The indices of implantation and resorption were calculated.
Examination of the liver, kidney, heart, lungs, brain, and small intestine of the pups of Wistar rats (n = 6) orally treated with H perforatum extract (methanol extraction solution containing 0.3% hypericin; 0, 100, 1000 mg/kg/d) showed severe damage to the liver and kidneys of animals killed postnatally on days 0 and 21. 31 Three dams were treated starting 2 weeks prior to mating through 21 days of lactating. The other 3 were treated from delivery through 21 days of lactation. Maternal body weights, gestation time, number of live pups, and weight of pups at birth were similar between the groups. The livers of newborn pups of dams in the low-dose group treated before and during pregnancy showed focal hepatocyte damage was apparent, with vacuolization of cells. In the high-dose group, these lesions were much more evident, with hepatocyte hyaline degeneration, lobular fibrosis, and disorganization of hepatocyte arrays. In the low-dose group, the kidneys showed a decrease in glomerular size with decrease in Bowman space and hyaline tubular degeneration, and in the high-dose group, these lesions were more severe. The same lesions, but much more diffuse and serious, were observed in pups killed after 21 days of ingesting milk from dams that were exposed to the test material throughout pregnancy and lactation. The same lesions were evident also in pups that were exposed to the substance only through nursing.
There were no effects on maternal weight gain or gestation length nor any effect on offspring body weights (up to postnatal day 56) observed behavior or whole and regional brain weights in Sprague-Dawley rats (n = 35) fed diets containing H perforatum extract (0, 180, 900, 1800, 4500 ppm; 0, 0.18, 0.90, 1.80, 4.50 g/kg; 0.3% hypericin) from gestation day 3 to postnatal day 21. 32 Offspring body weights in the treated groups were lower than tin controls at post natal days 56 (180, 900, 1800 ppm groups) and 78 (180, 1800 ppm groups). Offspring were tested using the open field test, acoustic startle response test, complex maze test, Morris water maze test, and the elevated plus maze activity test.
There were no behavioral effects to the offspring of CD-1 mice (n = 45) orally administered H perforatum extract (0.75 mg/g/d in feed; 0.3% hypericin) for 2 weeks before and through gestation. 29 There were also no effects on reproductive behavior or success in the next 3 generations of offspring. In the male pups, the treatment group weighed less than the controls. The offspring were tested with homing, locomotor activity, exploratory, forced swim, and anxiety tests.
Hypericum perforatum flower extract
In an in vitro study, the contractility of the vas deferens of Wistar rats exposed to the hydromethanolic extract of the flowering tops of H perforatum (1-300 μg/mL; 0.3% hypericin) and hyperforin (10−8 to 10−4mol/L was inhibited in a concentration-dependent manner. 33 Stimulation for the contractions was through electrical field stimulation or exposure to α,β-methylene adenosine triphosphate. Hypericin, quercitrin rutin, and kaempferol did not inhibit phenylephrine-induced contractions.
Hypericin
Sprague-Dawley rat embryos explanted into a culture of hypericin (0-142 ng/mL) for 2 days exhibited morphological changes when compared to controls starting at 71.0 ng/mL. 34 Embryos were explanted at gestational day 9.5 and were examined on day 11.5. The embryos exposed to high concentrations of hypericin (71.0 and 142.0 ng/mL) had lower total morphological score and number of somites compared with the control group. There was a negative linear trend in total morphological score, yolk sac diameter, and number of somites, indicating a progressive reduction in these parameters with increasing concentration of hypericin. There were no differences detected in crown-rump length. There were no adverse effects up 28.4 ng/mL.
Human
The frequency of live births and premature births in women in Canada who were taking St John wort (H perforatum; n = 54; average age = 32.6 ± 5.3) during their pregnancy were similar to those with no exposure (n = 108; average age = 32.5 ± 4.9). 35 Women were interviewed during pregnancy and followed for 5 to 7 years after birth. Hypericum perforatum was consumed by 76% of the pregnant women during the first trimester, 5.5% during the first and second trimester, 7.3% during the entire pregnancy, and 9.1% during some combination of the second and third trimester. Their average daily dose as reported by the subjects was 615 mg among those using tablets. The dose could not be estimated for a few of the subjects because they took H perforatum in the form of teas (3), tincture (1), or granules (1).
There were no differences in milk production, maternal adverse events, and infant weight over the first year of life observed when breastfeeding women (n = 33) were orally administered H perforatum extract (704.9 ± 463.6 mg/day, no further characterization) compared to disease-matched controls (n = 101) and age- and parity-matched nondisease controls (n = 33). 36
In 36 samples of breast milk from mothers (n = 5) who were taking H perforatum extract (300 mg times/d), hyperforin was present in the milk at 0.9% to 2.5%. 21 The plasma from 2 of the infants contained low levels of hyperforin (0.1 ng/mL). No side effects were seen in the mothers or infants. The authors concluded that these results add to the evidence of the relative safety of St John wort while breastfeeding. Hyperforin was detected in the breast milk of a mother took 3 Hypericum extract pills (3 × 300 mg/d; 0.12%-0.28% Hypericum, ∼4.5% hyperforin). 22 No clinical effects were observed in the mother and infant.
Hypericum perforatum flower extract
The above-mentioned contractility experiment was repeated with segments (3-4 cm) of the epididymal part of the vas deferens taken from subjects (n = 15) who underwent prostatectomy (9 who were 60-72 years old) or orchiectomy (3 who were 28-35 years old). Hypericum perforatum flower extract and hyperforin inhibited contractions stimulated by phenylephrine (3 × 10−6 mol/L). 33 The IC50 values were 13.9 ± 2.0 and 0.45 ± 0.04 μmol/L, respectively.
Genotoxicity
There were no new published genotoxicity studies discovered and no additional data were provided.
Irritation and Sensitization
Irritation
Dermal: human
Hypericum perforatum extract
In an irritation test (n = 18), a bath oil containing H perforatum extract (concentration not provided; 50 μL) did not cause irritation and was similar to the control of distilled water. 37 The test material was administered to the volar surface of the arm under occlusion for 24 hours. After an hour, the test areas were evaluated and the test substance readministered for another 24 hours and evaluated again. The evaluations were transepidermal water loss, photometric measurements of skin erythema, and visual scoring.
Sensitization
No dermal sensitization studies were discovered or submitted.
Phototoxicity
Dermal administration
Hypericum perforatum extract
A product containing H perforatum extract (1.1%) was not photosensitizing to the backs of guinea pigs when applied to tape-stripped skin. 38 The backs of the guinea pigs were irradiated (320-400 nm; 10.2 J/cm2) for 5 consecutive days after the product (1, 5, 10, and 20% in distilled water; 0.011%, 0.055%, 0.11%, 0.22%) was administered. Two weeks later, the product (0.1% and 1%) was applied and the skin irradiated. The test sites were observed at 24 and 48 hours.
Incubation in methanolic extract of H perforatum (> 50 μg/mL; 0.3% hypericin-like derivatives) was phototoxic to human keratinocyte HaCaT cells in ultraviolet A (UVA) light. 39 The cells were incubated for 4 hours then irradiated (1 J/cm2 UVA or 150 mJ/cm2 ultraviolet B [UVB]) for 3 hours. The test substance was not phototoxic in UVB light.
Hypericum perforatum oil
Hypericum perforatum oil (110 μg/mL) and an ointment containing Hypericum oil (30 μg/mL) were not phototoxic when administered to subjects (n = 8) with skin types II and III and no history of skin disease or photosensitivity. 40 There was no change in the minimal erythema dose (MED) after administration of the test materials. There was an increase in the erythema index after treatment with H perforatum oil using a more sensitive photometric measurement. The light doses were 24, 48, 96, and 144 J/cm2 (290-2500 nm), and the treated area was observed at treatment and after 24 and 48 hours.
Hypericin
Dermal administration of hypericin (n = 5-10; 0.1%-1%) resulted in minimal photosensitization to the ears of Balb/c mice at the highest concentration. 18 Hypericin acetate (n = 5-10; 0.015%-1.5%) induced more severe and prolonged response after irradiation characterized by intense erythema and ear swelling at all concentrations; skin damage was healed in 14 days with no scar formation. Residual photosensitization effects declined to almost nondetectable at day 7. Radiation exposure (586 and 589 nm) was performed 24 hours after administration of the test material.
Oral administration
Hypericum perforatum extract
In an oral study of 2 different H perforatum extracts (STW3, 80% ethanol extract, 612 mg, 1.4 mg hypericin; STW3-VI, 50% ethanol extract, 900 mg, 1.75 hypericin), male subjects (n = 20) had no change in minimum erythema dose of irradiation after administration of the test substances for 2 weeks. 41 Plasma steady state of hypericin/pseudohypericin was obtained before day 14 of treatment. The UV dose was adjusted for skin type. Two adverse events were reported, both described as hypersensitivity to light in mild intensity
In the presence of a stable plasma concentration of hypericin (6.72 ng/mL) the MED values did not differ from controls. 42 Hypericum perforatum extract (three 60 mg capsules) was orally administered twice daily for 2 weeks. Photosensitivity was tested before and after administration of the test material.
Oral administration of H perforatum extract in a single dose (5400 and 10 800 μg hypericin; n = 12) or over 7 days (5400 μg initial dose, 2700 μg /d; n = 24) did not increase dermal erythema or pigmentation when subjects were exposed to UVB, UVA, visible light, or solar simulated radiation. 43 There was no evidence of phototoxicity. Phototesting was performed prior to first dose and 6 hours after last administration of hypericin tablets. The postadministration erythema index and melanin index were similar to preadministration measurements in all cases except for visible light where there was an increase in the erythema index in the single dose study at both dose levels.
The single dose (5400 and 10 800 μg hypericin; n = 48) and steady state (5400 μg initial dose, 2700 μg /d hypericin; n = 24) studies were repeated with similar results. 44
In vitro
Hypericum perforatum extract, hypericin, quercetin, and pseudohypericin
Hypericum perforatum extracts (0, 30, 40, 50, 60, 70, 90, 100 μg/mL) from 3 different sources and hypericin (0, 0.1, 0.3 μg/mL) were cytotoxic to human keratinocyte cells (HaCaT cells) after incubation and exposure to UVA radiation (250-700 mJ/cm2) in a concentration- and UVA dose-dependent manner. 45 The cells were incubated in the test substances for 24 hours, irradiated, and then tested for viability using a neutral red assay. As for other constituents, quercetin was cytotoxic without radiation, rutin was phototoxic, and quercitrin had antiphototoxic properties. The UVA irradiation by itself was not cytotoxic up to 1000 mJ/cm2, where it was mildly cytotoxic.
Hypericin combined with H perforatum extracts (plant parts not specified) or constituents exerted less phototoxicity than pure hypericin to HaCaT keratinocytes. 46 The keratinocytes were exposed to 2 H perforatum extracts, (1) an ethanol reextraction of residue following a chloroform extraction (3.35 μmol/L hypericin and 124.0 μmol/L total flavonoids) and (2) a chloroform extract (hypericin and flavonoids not detected) supplemented with hypericin (20 μmol/L) and hypericin (20 μmol/L). Each plate was exposed to ambient light provided by fluorescent light bulbs that supplied 5.2 ± 5% J/cm2 after 30 minutes of exposure to the test materials at room temperature. The extracts showed 24% and 40% less phototoxicity to the keratinocytes, respectively, than to those exposed to hypericin.
In a neutral red uptake assay of HaCAT keratinocytes exposed to UVA light (320-400 nm) after incubation in hypericin (0.1, 0.5, and 1 μmol/L) for up to 60 minutes, there was a dose-dependent increase in DNA damage as irradiation dose increased. 47 However, the authors stated that although the results show that the combination of hypericin and UVA light increased the genotoxic burden, when all factors are taken into account, the risk of significant photogenotoxic damage incurred by the combination of H perforatum extracts and UVA phototherapy may be low in the majority of individuals.
Treatment with both photoactivated hypericin and pseudohypericin resulted in a dose-dependent inhibition of proliferation of human acute T leukemic lymphoma cells; nonphotoactivated plant pigments had no effect on cell proliferation. 48 The IC50 of irradiated hypericin was 100 and 200 ng/mL for pseudohypericin.
In a test of the protective effect of quercetin, a natural antioxidant compound, on hypericin-induced cytotoxicity under light conditions using human promyelocytic leukemia cells (HL-60), hypericin (10−5 mol/L) alone decreased cell survival to 21%. 49 The combination of quercetin (10−5 mol/L) increased survival to 46%. Lower concentrations of quercetin had no protective effect. The authors suggested that these results indicate that oxygen radicals can play a role in hypericin-induced phototoxic effects.
Ocular
Hypericin
Human lens epithelial cells incubated in hypericin (0.1-10 μmol/L) and irradiated (4 J/cm2 UVA or 0.9 J/cm2 visible light) had increased necrosis and apoptosis. 50 Neither hypericin exposure alone nor light exposure alone reduced cell viability. The addition of the ocular antioxidants lutein and N-acetyl cysteine did not prevent the damage. The authors concluded that ingested H perforatum extract is potentially phototoxic to the eye and could contribute to early cataractogenesis.
Photosensitized photopolymerization was induced in lens alpha-crystalline, isolated from calf lenses, after irradiation (>300 nm, 24 mW/cm2) in the presence of hypericin (5 × 10−5 mol/L in 10 mmol/L ammonium bicarbonate; pH 7.0). 51 Further analysis of the oxidative changes using mass spectrometry showed specific oxidation of methionine, tryptophan, and histidine residues, which increased with time of irradiation. Hypericin did not damage the lens protein without irradiation. Damage to alpha-crystalline could undermine the integrity of the lens directly by protein denaturation and indirectly by disturbing chaperone function. The authors suggest that in the presence of light, hypericin can induce changes in lens protein that could lead to the formation of cataracts.
Human retinal pigment epithelial (hRPE) cells exposed to hypericin (10−7 to 10−5 mol/L) and irradiated (0.72 J/cm2) reduced cell viability compared to untreated cells and cells that were either just exposed to the test material or irradiated. 52 Viability was measured by (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt) and lactate dehydrogenase assays after 1.5 hours incubation in hypericin and irradiated for 1, 3, 5, and 10 minutes. The presence of hypericin in irradiated hRPE cells significantly changed the redox equilibrium of glutathione and a decrease in the activity of glutathione reductase. Increased lipid peroxidation as measured by the 2-thiobarbituric acid reactive substances assay correlated with hypericin concentration in hRPE cells and visible light radiation.
The UVB irradiation of bovine lenses exposed to hypericin (10−6 mol/L) caused an increase in focal length variability and protein leakage compared to lenses that were only UVB irradiated. 53 The lenses were placed in tissue culture wells and irradiated (0.2 J/cm2) then followed for 7 days. Lenses treated with hypericin and irradiated had an increase in focal length variability as compared with the lenses that were only UVB irradiated. Lenses without UVB irradiation had lower focal length variability than irradiated lenses. For nonhypericin-treated lenses, UVB-irradiated lenses had a larger variability (4.58 mm) than the unirradiated lenses (1.78 mm). The lenses incubated in elevated glucose concentrations had a focal length variability (3.23 mm) equivalent to that of the unirradiated hypericin-treated lenses (3.54 mm). The authors concluded that photooxidative damage by hypericin results in changes in the optical properties of the lens, protein leakage, and finally cataract formation. This is evidence that people should protect their eyes from intense sunlight when taking H perforatum-derived substances.
Using the data collected in questionnaires by the National Center for Complementary and Alternative Medicine and Alternative Health/Complementary and Alternative Medicine Supplement (ALT; a total of 120 142 753 responses), an association between the oral use of H perforatum among person 40 years of age and older and the presence of cataracts was reported to have an odds ratio of 1.59 (05% confidence interval 1.02-2.46) or that persons with cataracts are 59% more likely to report St. John’s wort use. 54 The authors stated that Hypericum perforatum may increase the risk of cataracts but the mechanism is not established.
Clinical Use
Oral
There are many clinical studies of the oral use of H perforatum extracts for effectiveness as an antidepressant and for safety. Table 5 is a summary of adverse effects that have been reported with the oral administration of H perforatum extracts. Adverse events included nausea, headache, dizziness abdominal pain, insomnia/sleep disturbance, cold symptoms, and diarrhea. Except for sleep disturbance, and to a lesser extent headache, the adverse events were reported in low percentages of the subjects.
Reported Adverse Events in Oral Clinical Trials.

Abbreviations: AE, adverse event; GI, gastrointestinal.
a See Table 3 for parameters/characterizations of these extracts.
Dermal
In a half-side comparison study of a cream with and without H perforatum extract (1.5% hyperforin), there were 4 reported adverse events in 3 subjects that were classified as not serious but resulted in not finishing the study. 55 One subject developed contact eczema to the vehicle. In the subjects, all with atopic dermatitis, that finished the 4-week study (n = 18), both sides of the skin lesions improved, with fewer skin colonies of Staphylococcus aureus on the H. perforatum extract side on days 7, 14, and 28.
Case Studies
Hypericum perforatum extract
A 45-year-old female subject developed large blisters that resolved with some hyperpigmentation after laser treatment at 532 nm at 1.5 J/cm2. 56 She had received a previous treatment with no ill effects. It was discovered that the subject had started taking medication that contained St John wort (H perforatum). Another treatment a month after stopping the medication resulted in no ill effects.
A case of an overdose of H perforatum extract in a suicidal attempt of a 16-year-old girl resulted in seizures and confusion that resolved after 6 days. 57 It has been reported that the girl had taken up to fifteen 300-μg tablets/day for 2 weeks and 50 tablets just before hospitalization. After 6 days, the electroencephalogram was normal and no further seizures occurred in the following 6 months.
A case of acute neuropathy was reported in a woman after taking powdered H perforatum extract (500 mg/d) and exposure to sunlight. 58 The pain started after 4 weeks of use and increased over time and after sunbathing. Symptoms decreased with discontinuation of use after 3 weeks and disappeared after 2 months.
Two pregnant women taking Hypericum extract (not characterized as to plant part, 900 mg/day) had no signs of toxicity or other harmful effects. 59 The authors stated concern about the use of H perforatum instead of an established effective treatment because safety of H perforatum in pregnancy and lactation has not been established.
Summary
Hypericum perforatum (aka St. John’s wort)-derived ingredients function in cosmetics as skin-conditioning agents—miscellaneous, skin-conditioning agents—humectants; skin protectants; antioxidants, hai-conditioning agents; and antimicrobial agents. New information has been submitted to meet the data needs that were identified because of the insufficient conclusion of the previous report.
Since the original report was published, the name of H perforatum extract was changed to H perforatum flower/leaf/stem extract and H perforatum extract is now defined as an extract of the whole plant.
Hypericum perforatum extract was reported to be used in 32 leave-on products, 3 rinse-off products, and 1 baby product in concentrations of up to 0.003%. Hypericum perforatum flower was reported to be used in 1 leave-on product; maximum concentration of use was reported to be 0.005%. Hypericum perforatum flower/leaf/stem extract is reported to be used in 49 leave-on products and in 25 rinse-off products, mostly in skin care products and 2 products that are diluted for bath up to 0.07%. Hypericum perforatum oil is reported to be used in 13 leave-on products and in 4 rinse-off products. Use concentration was only reported for skin fresheners up to 0.00005%.
Hypericin, the most active constituent of H perforatum, penetrated the stratum corneum and epidermis of mouse ear skin, with little evidence of penetration into the dermis at 1%, with less penetration into the skin at 0.1% and 0.01%. Hypericin, pseudohypericin, hyperforin quercetin, and isorhamnetin were observed in the plasma after oral administration of H perforatum extract. Hyperforin was detected in human breast milk but not in the feeding infant’s plasma in mothers that ingested H perforatum extract.
Orally administered H perforatum extract at 900 and 2700 mg/kg to rats and dogs resulted in signs of load damage to the liver and kidneys at the high doses. Orally administered H perforatum extract at 255 to 285 mg to healthy male subjects 3 times/day for 13 days had no effect on vasoconstrictor responses of cutaneous blood flow or SR.
There was liver damage to the pups of rats orally treated with H perforatum extract at 100 and 1000 mg/kg/d. Lower doses had no effects on rat and mice dams or pups and had no effect on the cognitive abilities of pups. Rat embryos incubated in hypericin at 71.0 and 142 ng/mL had a negative linear trend in total morphological score, yolk sac diameter, and number of somites.
No effects were reported or observed in women who ingested H perforatum during pregnancy nor any effects to their infants. No effects were observed in breast feeding infants of mothers who took H perforatum.
There was inhibited contractile response in rat and human vas deferens exposed to H perforatum up to 300 μg/mL. Human sperm had DNA denaturation when exposed to H perforatum extract.
Hypericin demonstrated antiviral, anti-inflammatory, and antitumor effects to human leukocytes.
A bath oil with an unknown concentration of H perforatum extracts was nonirritating to humans.
Dermal administration of H perforatum extract was not photosensitizing to the backs of guinea pigs at 1.1%. Hypericum perforatum oil in a product was not phototoxic to humans at 110 μg/mL. Hypericin at 0.1% and hypericin acetate at 0.015% caused more severe and prolonged dermal response when mouse skin was irradiated. Single dose and short-term oral administration of H perforatum extract did not increase photosensitization in humans. Human keratinocyte cells incubated in H perforatum extracts and constituents demonstrated increased cytotoxic and photogenotoxic effects when exposed to UVA. Human and bovine ocular cells/lens epitheliums had increased apoptosis and reduced cell viability after incubation in hypericin and exposure to UVA. A survey showed a connection between H perforatum use and the development of cataracts.
Adverse events in oral efficacy clinical trials included nausea, headache, dizziness abdominal pain, insomnia/sleep disturbance, cold symptoms, and diarrhea.
Discussion
Although an earlier safety assessment of H perforatum extract and oil found the available data insufficient to support safety, additional data were submitted addressing the concentration of use and function in cosmetics and providing photosensitization/phototoxicity, reproductive/developmental toxicity, irritation/sensitization, and ocular irritation data.
Although there are data gaps in this report, the relatedness of constituents, physicochemical properties, functions, and concentrations in cosmetics allowed grouping these ingredients together and extrapolating the available toxicological data to support the safety of the entire group.
The Cosmetic Ingredient Review Expert Panel (Panel) expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
A cosmetic formulation may contain multiple botanical ingredients, each of which can contribute to the total concentration of constituents of concern in the botanical ingredients. For example, the Panel noted that 1 constituent of Hypericin perforatum-derived ingredients is hypericin. Hypericin has been shown to be a photosensitizer in visible light and to have possible teratogenic effects in an in vitro study in rats. Hypericin was reported to be present in the various plant parts at 5 to 18,000 ppm. Another constituent is quercetin. Quercetin may be genotoxic and is reported to be in H perforatum plant parts at 1000 to 20 000 ppm. Because the maximum concentration of use in cosmetics that contain these H perforatum extracts was reported to be 0.07%, the Panel concluded that the amount of exposure to these constituents would be below the threshold of toxicological concern.
The Panel also noted that the use of other botanical ingredients that may contain hypericin and/or quercetin, in combination with H perforatum-derived ingredients in a single formulation, could result in exposures that exceed levels of toxicological concern. Potential sensitizers are also constituents of concern that may be present. Thus, cosmetic products containing multiple botanical ingredients should be formulated to ensure that total exposures to such constituents remain below levels of toxicological concern when used as intended.
The Panel discussed the issue of incidental inhalation exposure from face and neck powders. There were no inhalation toxicity data available. The sizes of a substantial majority of the particles of these ingredients, as manufactured, would be expected to be larger than the respirable range (ie, aerodynamic equivalent diameters > 10 µm) and to aggregate and agglomerate to form much larger particles in formulation and would not be respirable to any appreciable amount. Furthermore, particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used (at concentrations up to 0.07% in cosmetic products that may become airborne), the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
The Panel considered other data available to characterize the potential for H perforatum-derived ingredients to cause irritation and sensitization, systemic toxicity, and reproductive/developmental toxicity. They noted the lack of systemic toxicity at doses much higher than any cosmetic exposure in acute and subchronic oral exposure studies. There was also little or no irritation or sensitization in multiple tests of dermal and ocular exposure. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following H perforatum-derived ingredients were found safe in the present practices of use and concentration in cosmetics:
H perforatum extract;
Hperforatum flower extract;
H perforatum flower/leaf extract*;
H perforatum flower/leaf/stem extract;
H perforatum flower/twig extract*;
H perforatum leaf extract*;
H perforatum oil.
indicates not in current use. Were the ingredients not in current use to be used in the future, the expectation is that they would be used in products categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from Lillian J. Gill, Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.