Abstract
The CIR Expert Panel (Panel) assessed the safety of citric acid, 12 inorganic citrate salts, and 20 alkyl citrate esters as used in cosmetics, concluding that these ingredients are safe in the present practices of use and concentration. Citric acid is reported to function as a pH adjuster, chelating agent, or fragrance ingredient. Some of the salts are also reported to function as chelating agents, and a number of the citrates are reported to function as skin-conditioning agents but other functions are also reported. The Panel reviewed available animal and clinical data, but because citric acid, calcium citrate, ferric citrate, manganese citrate, potassium citrate, sodium citrate, diammonium citrate, isopropyl citrate, stearyl citrate, and triethyl citrate are generally recognized as safe direct food additives, dermal exposure was the focus for these ingredients in this cosmetic ingredient safety assessment.
Introduction
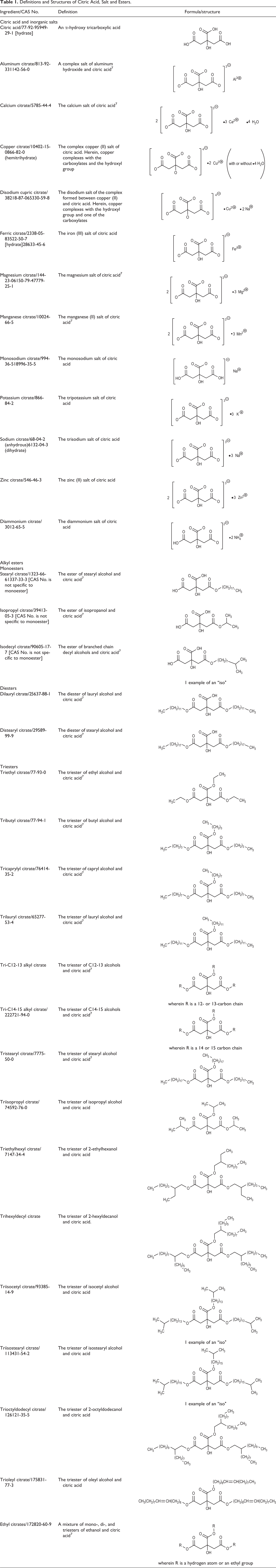
This assessment reviews the safety of citric acid, an α (and β)-hydroxytricarboxylic acid, as used in cosmetics. The following 12 inorganic citrate salts and 20 alkyl citrate esters are also included in this safety assessment, for a total of 33 ingredients: Inorganic salts aluminum citrate; calcium citrate; copper citrate; diammonium citrate; disodium cupric citrate; ferric citrate; magnesium citrate; manganese citrate; monosodium citrate; potassium citrate; sodium citrate; zinc citrate; Alkyl esters isodecyl citrate; isopropyl citrate; stearyl citrate; dilauryl citrate; distearyl citrate; tributyl citrate; tri-C12-13 alkyl citrate; tri-C14-15 alkyl citrate; tricaprylyl citrate; triethyl citrate; triethylhexyl citrate; trihexyldecyl citrate; tri-isocetyl citrate; tri-isopropyl citrate; trilauryl citrate; trioctyldodecyl citrate; trioleyl citrate; triisostearyl citrate; tristearyl citrate; ethyl citrates;
Citric acid is reported to function in cosmetics as a chelating agent, pH adjuster, or fragrance ingredient. Although some of the inorganic citrate salts are also reported to function as a pH adjuster or chelating agent, there are many other reported functions, including skin-conditioning agent, buffering agent, cosmetic astringent, oral care agent, cosmetic biocide, or pesticide. The alkyl citrate esters are reported to function primarily as skin-conditioning agents but a few have other possible functions reported, including plasticizer, solvent, and fragrance ingredient.
As listed by the Food and Drug Administration (FDA), citric acid, calcium citrate, ferric citrate, manganese citrate, potassium citrate, sodium citrate, diammonium citrate, isopropyl citrate, stearyl citrate, and triethyl citrate are generally recognized as safe (GRAS) direct food additives. Since these 10 ingredients have been shown to be safe for ingestion, this report will focus on the dermal toxicity of these ingredients. For the other ingredients, all available data will be included.
Structurally, citric acid is an α-hydroxy acid (AHA). The safety of AHAs was previously reviewed by the CIR Expert Panel (Panel).
1
In its Guidance for Industry: Labeling for Topically Applied Cosmetic Products Containing Alpha Hydroxy Acids as Ingredients,
2
the FDA specifically mentions citric acid-containing products, for which the following labeling may be warranted: Sunburn Alert: This product contains an alpha hydroxy acid (AHA) that may increase your skin's sensitivity to the sun and particularly the possibility of sunburn. Use a sunscreen, wear protective clothing, and limit sun exposure while using this product and for a week afterwards.
2
Chemistry
Definition, Structure, and Properties
Citric acid (2-hydroxy-1,2,3-propanetricarboxylic acid) is a common metabolite of plants and animals and is well known for its part in the Krebs cycle. 3 It precipitates as white, translucent crystals of monoclinic holohedra form. Citric acid is a polyprotic AHA. However, citric acid can also be classified as a β-hydroxy acid as 2 of the carboxylic acid functional groups of citric acid are 2 carbons removed from the hydroxy group (Figure 1).
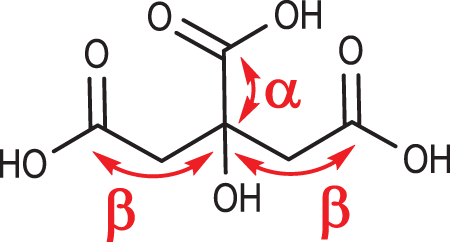
Citric acid.
Citric acid differs structurally from the AHAs reviewed previously 1 (ie, glycolic and lactic acid) having 3 carboxylic acid functional groups instead of just 1. Citric acid is therefore triprotic and thus has 3 different pKas, making it a prime buffer component. Even for the most acidic of these carboxylates, that is, the center acid functional group, is only a weak acid with a pKa of 3.1.
Citric acid is soluble in water and in some organic liquids and is very hydrophilic, with an octanol/water partition coefficient around 1. Citric acid and its salts are solids. The citrate alkyl esters, however, vary from oily liquids (for shorter chain analogs like ethyl) to powdery solids (for longer chain analogs like stearyl). Directly dependent on chain length and degree of substitution, these esters are less soluble in water and more soluble in organic liquids and are generally hydrophobic, with octanol/water partition coefficients estimated between 1 and 12.
The definitions and structures of the ingredients included in this review are provided in Table 1. The available physical and chemical property information is found in Table 2. Impurities and composition data are provided in Table 3.
Definitions and Structures of Citric Acid, Salt and Esters.
Chemical and Physical Properties.

Impurities and Composition.
Methods of Manufacture
Industrial, large-scale production of citric acid is accomplished, most commonly, via mycological fermentation of crude sugar stocks (eg, molasses), historically by strains of Aspergillus niger. 4 A common problem associated with these fermentation methods is the cosynthesis of isocitric acid (1-hydroxy-1,2,3-propanetricarboxylic acid). However, isocitric acid can be separated using a variety of crystallization techniques. Careful control of the trace element content is very important for high production. 3,5 (While citric acid can also be extracted from citrus fruits, over 99% of the world’s citric acid output is produced by microbial fermentation. 5 ) The citrate salts are produced by the same fermentation process but are simply crystallized in the presence of appropriate alkaline solutions (eg, citric acid can be crystallized with sodium hydroxide to produce sodium citrate).
Citrate alkyl esters are typically produced via the condensation of the appropriate alcohol with citric acid (eg, condensing with butyl alcohol to produce tributyl citrate). 6 Some ingredient-specific methods of manufacture are described in Table 4.
Ingredient-Specific Methods of Manufacture.

Use
Cosmetic
Citric acid is reported to function in cosmetics as a chelating agent, pH adjuster, or fragrance ingredient. 7 Some of the inorganic salts of citric acid are reported to function as a pH adjuster or chelating agent; these salts also have many other reported functions, including skin-conditioning agent, buffering agent, cosmetic astringent, oral care agent, cosmetic biocide, or pesticide. The alkyl esters are reported to function primarily as skin-conditioning agents but a few of these have other reported functions, including plasticizer, solvent, and fragrance ingredient. The various cosmetic functions of these ingredients are provided in Table 5; some ingredients have more than 1 reported function.
Reported Cosmetic Functions of Citric Acid and its Salts and Esters.a

aReference. 7
The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 20118 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 9 indicate that 22 of the 33 citrates named in this report are currently used in cosmetic formulations. Citric acid is used in almost every category of cosmetic product, with 6795 reported uses 8 at concentrations up to 4% in leave-on formulations, 10% in rinse-off formulations, and 39% in products diluted for (bath) use. 10 Sodium, tributyl, and triethyl citrate are reported to be used in 980, 331, and 244 cosmetic formulations, respectively. 8 All other in-use ingredients have less than 50 uses. The ingredient with the highest concentration of use is tri-isostearyl citrate; it is used at up to 80% in lipstick formulations. 10 Trioctyldodecyl citrate is used at up to 30% in leave-on formulations; it is used at up to 21% in products applied to the eye area and 19% in lipstick formulations. Tricaprylyl citrate is used at up to 27% in leave-on formulations. All other in-use ingredients are used at concentrations of ≤12%.
Frequency and concentration of use data are provided in Table 6. The ingredients not in use, according to the VCRP and Council survey, are listed in Table 7. Products containing citric acid and some of its salts and esters may be applied to baby skin or used near the eye area or mucous membranes. Additionally, citric acid and some of its salts and esters are used in cosmetic sprays, including hair, deodorant, body, and other propellant and pump spray products; the maximum reported concentrations of citric acid used in a spray product is 0.7% citric acid, of a salt is 0.2% sodium citrate, of an ester is 4% trioctyldodecyl citrate, and in deodorants is 2% triethyl citrate. These products could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 11 -14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 11,13 There is some evidence indicating that, generally, deodorant spray products, compared with hair sprays, can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 11 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Frequency and Concentration of Use According to Duration and Type of Exposure.
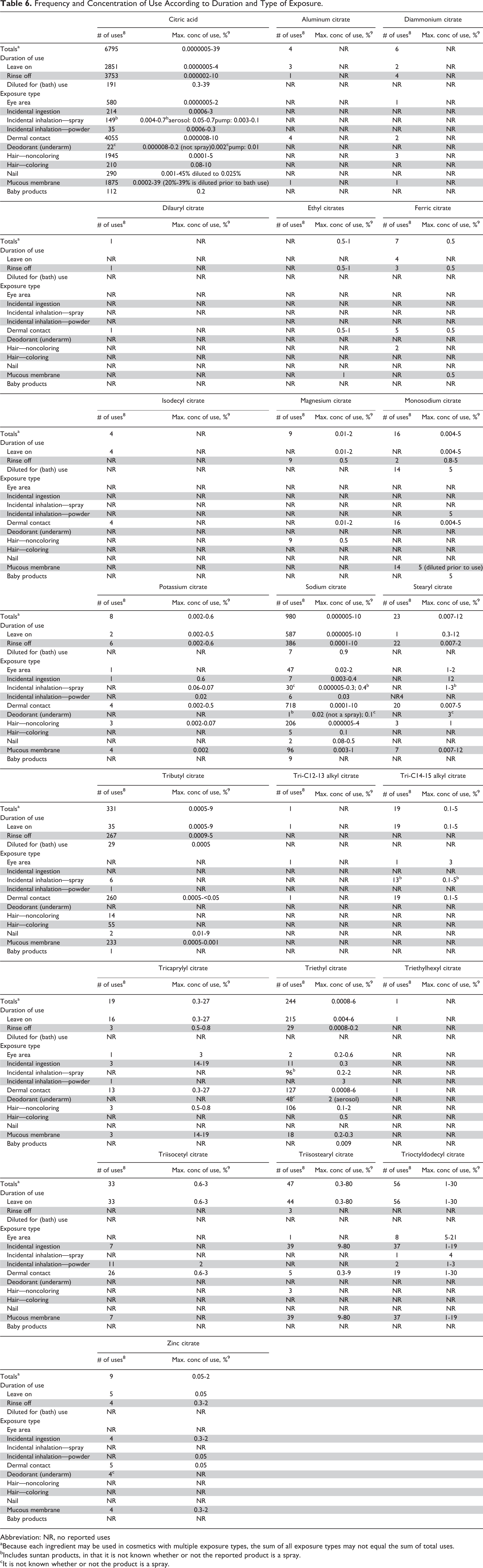







Abbreviation: NR, no reported uses
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether or not the reported product is a spray.
cIt is not known whether or not the product is a spray.
Ingredients Not Reported to be Used.

All ingredients included in this review are listed in the European Union inventory of cosmetic ingredients. 15 “Water-soluble zinc compounds” are listed in Annex III of the Cosmetic Directive, with a maximum authorized concentration in the finished cosmetic product of 1% calculated as zinc; therefore, zinc citrate has a maximum authorized concentration of use of 1%, calculated as zinc, in finished cosmetic products in the European Union. 16
Noncosmetic
The following 10 ingredients are GRAS direct food additives, restricted only by the need to follow good manufacturing practices: citric acid (21CFR184.1033), calcium citrate (21CFR184.1195), ferric citrate (21CFR184.1298), manganese citrate (21CFR184.1449), potassium citrate (21CFR184.1625), sodium citrate (defined as the trisodium salt; 21CFR184.1751), diammonium citrate (21CFR184.1140), isopropyl citrate (21CFR184.1386), stearyl citrate (21CFR184.1851), and triethyl citrate (21CFR184.1911). Additionally, the following are allowed as indirect food additives: citric acid, magnesium citrate, monosodium citrate, potassium citrate, sodium citrate, diammonium citrate, stearyl citrate, isopropyl citrate, distearyl citrate, triethyl citrate, tributyl citrate, and tristearyl citrate. 17 Citrate-containing ingredients are allowed as active ingredients, at a maximum daily dosage of 8 g, in antacid over-the-counter products (21CFR331.11). Examples of other noncosmetic uses of citric acid and some of the citrates are provided in Table 8.
Examples of Noncosmetic Uses.

Toxicokinetics
Orally administered citric acid is well absorbed and largely metabolized. 18 Exogenous and endogenous citric acid can be completely metabolized and serve as a source of energy. Citric acid is an intermediate in the Krebs (or tricarboxylic acid) cycle. 19 Citric acid completes the breakdown of pyruvate, formed from glucose through glycolysis, and it liberates carbon dioxide. Approximately 2 kg of citric acid are formed and metabolized every day in humans. Citrate is thought to be freely filterable at the glomerulus of the kidney and 65% to 90% of filtered citrate is reabsorbed in humans. 20 Filtered citrate of 10% to 35% is excreted in the urine. The normal blood citrate level in humans is approximately 25 mg/L. 21
In Vitro
Trihexyl citrate
Although trihexyl citrate is not a cosmetic ingredient, this information is presented because this chemical is structurally similar to cosmetic ingredients included in this review, supporting that citrate esters undergo hydrolysis.
Trihexyl citrate was incubated with rat serum, an intestinal cytosolic fraction, and a liver cytosolic fraction obtained from Sprague-Dawley rats to determine the hydrolysis of trihexyl citrate in each of these preparations. 22 Dimethyl sulfoxide (DMSO) was used as the vehicle; the volume of DMSO did not exceed 1% of the total volume of the incubation medium. A concentration of 50 nmol/mL was used with all 3 preparations; a concentration of 1000 nmol/mL was also used with rat serum. In rat serum, at the concentrations of 50 and 1000 nmol/mL, the half-life of trihexyl citrate hydrolysis was 4 and 90 minutes, respectively. Hexanol was produced as a product of hydrolysis. Dihexyl citrate is formed as an intermediate. Hydrolysis was concentration dependent, being faster at lower concentrations. Hydrolysis did not occur with 5 µmol/mL of serum. The half-life of hydrolysis for 50 nmol/mL trihexyl citrate in the rat liver cytosolic fraction was 1.2 minutes (The half-life was not given for the intestinal fraction.)
Aluminum citrate
The lipid bilayer permeation of neutral aluminum citrate was determined by measuring the flux across unilamellar phospholipid vesicles or liposomes, using 2 independent procedures. 23 The permeation of aluminum citrate was then compared to that of citric acid (as well as malic and lactic acids). Lipid bilayer permeation of 1.82 mmol/L aluminum citrate was slow; the permeability coefficient was, at most, 2 × 10−11 cm/s. Comparison of permeation of aluminum citrate to the acids indicated that the flux of aluminum citrate is limited by diffusion across the water–lipid interface. (The permeability coefficient for 6.0 mmol/L citric acid was 3.1 × 10−11 cm/s.)
Oral
Aluminum citrate
Eight male Sprague-Dawley rats were dosed by gavage with 100 mg aluminum/kg bw, as aluminum citrate, 6 days/wk for 4 weeks. 25 A control group was given tap water. Half of the animals were killed at the termination of dosing; the remaining animals were killed after a 5-week nontreatment period. The levels of aluminum in the cortex of the brain, the hippocampus, and the cerebellum were statistically significantly increased after 4 weeks of dosing with aluminum citrate, and there was no major difference between the animals killed at the termination of dosing and 5 weeks later. (In this study, groups of rats were also dosed with citric acid for 4 weeks or aluminum hydroxide for 9 weeks. The aluminum content in the cortex of the brain of rats dosed with citric acid was statistically significantly increased compared to controls. There were no statistically significant differences in aluminum content of the brain between control rats and those dosed with aluminum hydroxide.)
Ten female Sprague-Dawley rats were given drinking water with 80 mmol/L aluminum citrate for 8 months; a control group of 8 rats was given untreated water. 26 After 8 months of dosing, aluminum concentrations were statistically significantly increased in bone, spleen, liver, and kidneys, but not the brains, of treated animals.
Stearyl/distearyl citrate
Stearyl citrate is hydrolyzed readily to stearyl alcohol and citric acid in dogs and, to a lesser extent, in rats. 27 Stearyl citrate, predominantly as distearyl citrate, added to the feed of rats at a concentration of 2.5% to 10% was poorly absorbed (additional details were not provided). 18
Isopropyl citrate
Isopropyl citrate, mostly as the monoisopropyl ester, was administered in the diet of 6 rats in a mono- and diglycerides vehicle at concentrations of ≤10%. 18 Isopropyl citrate was nearly completely absorbed (additional details were not provided).
Effect on Transdermal Absorption
Triethyl citrate
Triethyl citrate inhibited the transdermal absorption of viprostol, a synthetic prostaglandin E2, through the skin of male hypertensive rats. 28 This effect was demonstrated by the statistically significant decrease in blood radioactivity levels following the topical application of [ 14 C]viprostol in triethyl citrate compared to those found with the use of petrolatum (pet) or silicone as the vehicle. A comparison of metabolic profiles also demonstrated slower hydrolysis of viprostol to free acid with the use of triethyl citrate as the vehicle.
Toxicological Studies
Single Dose (Acute) Toxicity
Acute toxicity studies are summarized in Table 9. Acute toxicity testing did not raise any toxicological concerns.
Acute Toxicity Studies.
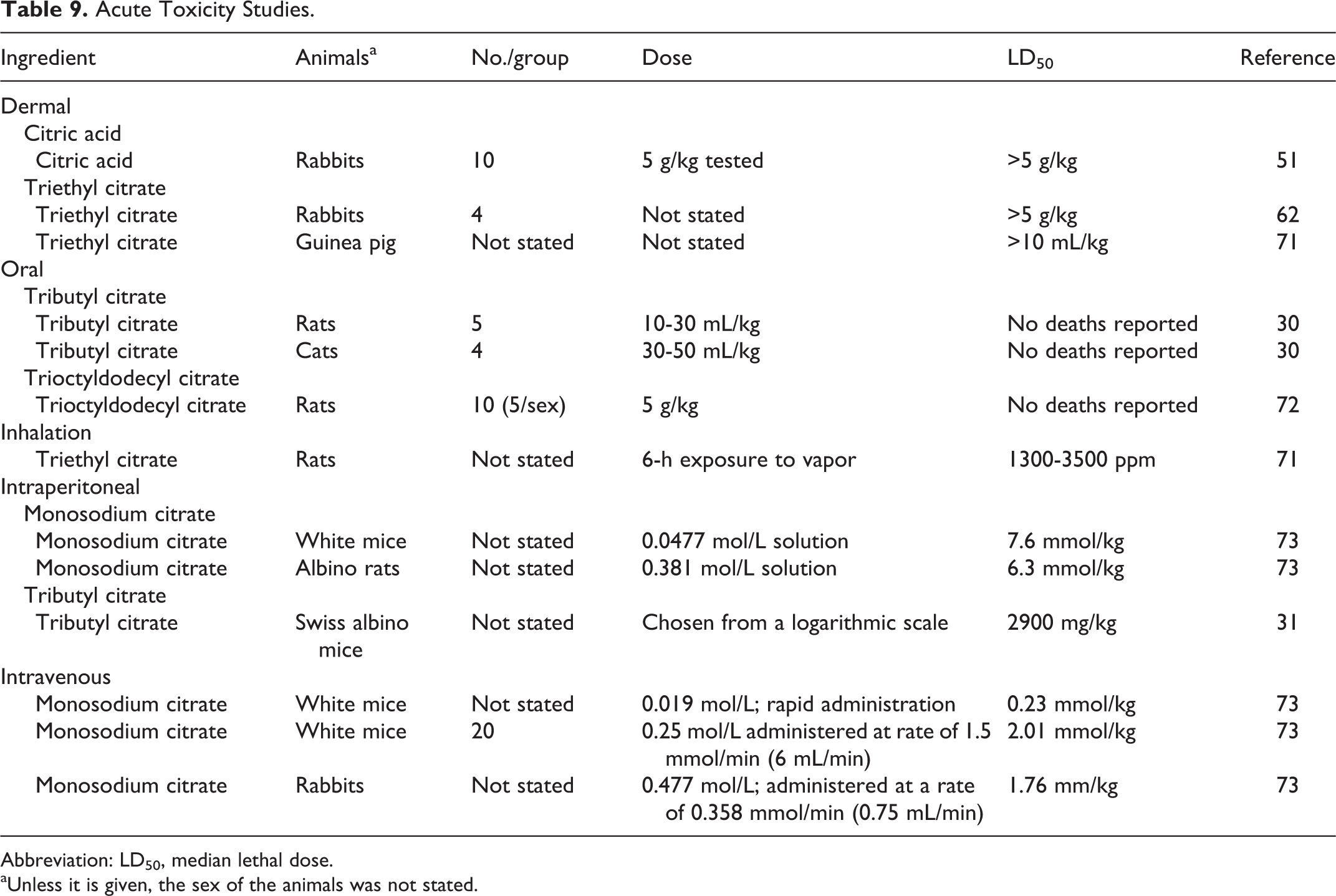
Abbreviation: LD50, median lethal dose.
aUnless it is given, the sex of the animals was not stated.
Repeated Dose Toxicity
Oral
Aluminum citrate
In a toxicokinetics study described previously, a group of 8 male Sprague-Dawley rats was dosed by gavage with 100 mg aluminum/kg bw, as aluminum citrate, 6 days/wk for 4 weeks. 25 A control group was given tap water. Half of the animals were killed at the termination of dosing; the remaining animals were killed after a 5-week nontreatment period. Body weights of test animals were similar to those of controls after 4 weeks of dosing. Body weights of treated animals decreased compared to controls during the recovery period, but the difference was not statistically significant.
In another toxicokinetics study described previously in this report, a group of 10 female Sprague-Dawley rats was given aluminum citrate in the drinking water at a concentration of 80 mmol/L for 8 months. 26 Final body weights of animals of the test group were statistically significantly decreased compared to the controls. Kidney function was not affected by dosing.
Isopropyl citrate ester mixture
A 6-week feeding study of an isopropyl citrate ester mixture consisting of 27% isopropyl citrate, 9% diisopropyl citrate, and 2% triisopropyl citrate, in a vehicle consisting of mono- and diglycerides (1:1) vegetable oil, was performed using rats. 29 Male rats had an average daily intake of 0.78 g and females 0.54 g of the citrate mixture, and no adverse effects were observed. (Additional details were not provided.)
Groups of 10 rats were fed diets containing 0%, 0.28%, 0.56%, or 2.8% of the above-mentioned isopropyl citrate ester mixture in the same vehicle (corresponding to 0%, 0.11%, 0.21%, and 1.06% isopropyl citrate ester content, respectively) for 2 years. 29 Again, no signs of toxicity were observed. Microscopic examination of select tissues did not reveal any test article-related changes.
Six-week dietary and 6-week gavage studies were performed in rabbits using the same isopropyl citrate ester mixture in the same vehicle. 29 Signs of toxicity were not observed in groups of 1 to 8 rabbits given feed containing 1.9% to 22.5% of the isopropyl citrate ester mixture or in groups of 1 to 3 rabbits dosed daily by gavage with 0%, 2.2%, 4.4%, or 9.2% of the isopropyl citrate ester mixture. Selected tissues of the 8 high-dose males used in the feeding study were examined microscopically and no abnormalities were found.
Groups of 2 cocker puppies and 2 adult mongrel dogs were also fed a diet containing the isopropyl citrate ester mixture in vehicle. 29 Adverse effects were not observed when dogs were fed a diet containing 0.06% of the test article for 12 weeks.
Distearyl citrate ester mixture
A 6-week feeding study of a distearyl citrate ester mixture consisting of 12.5% stearyl citrate, 75% distearyl citrate, and 12.5% tristearyl citrate was performed using rats. 29 Male rats had an average daily intake of 1.32 g and females 1.06 g of the mixture and no adverse effects were observed. (Additional details were not provided.)
Groups of 10 rats were fed diets containing 0%, 0.5%, 2.0%, or 10.0% of the distearyl citrate ester mixture for 2 years. 29 No signs of toxicity were observed. Microscopic examination of select tissues did not reveal any test article-related changes.
In a 6-week dietary study in rabbits with the same distearyl citrate ester mixture, 2 groups of 8 rabbits were given feed containing 2% or 10% of the mixture. 29 No signs of toxicity were observed. Select tissues of the rabbits of the 10% group, including the liver, kidneys, heart, and brain, were examined microscopically. No abnormalities were found.
Groups of 2 cocker puppies and 2 adult mongrel dogs were also fed a diet containing the distearyl citrate ester mixture. 29 Adverse effects were not observed when dogs were fed a diet containing 3.0% of the test article for 12 weeks.
Tributyl citrate
Groups of 3 or 4 rats, number per sex not specified, were fed a diet containing 0%, 5%, or 10% tributyl citrate for 6 weeks. 30 No effect on body weight gain was observed in the 5% group. Body weight gains in the 10% group were decreased; the decrease may have been attributable to frequent diarrhea. No effects on blood counts were reported and no microscopic lesions were observed.
Two cats were dosed daily by gavage with 5 mL/kg tributyl citrate daily for 2 months, and 2 cats were used as negative controls. 30 No significant effects were observed.
Intraperitoneal
Tributyl citrate
A test group of 20 mice (sex not stated) was dosed by intraperitoneal injection with 580 mg/kg tributyl citrate in 3% acacia for 14 days while a group of 20 control mice was dosed with vehicle only. 31 Two animals per group were killed at the end of the study. Body weight gains were decreased in the test animals and the decrease was significant after 7 days. No significant changes in blood counts were observed and no microscopic lesions were observed.
Reproductive and Developmental Toxicity
Oral
Aluminum citrate
A group of 20 presumed pregnant rats were dosed daily by gavage with 1064 mg/kg bw aluminum citrate and 62 mg/kg bw citric acid, concurrently, on days 6 to 15 of gestation, and a negative control group of 20 gravid rats received distilled water only. 32 All animals were killed on day 20 of gestation. The actual numbers of gravid test and control rats were 15 and 17, respectively. Administration of aluminum citrate with citric acid was not maternally toxic, embryotoxic, or fetotoxic. A statistically significant increase in the absence of xiphoids was the only skeletal variation reported. The aluminum concentration in the maternal liver, kidney, brain, bone (femur), and placenta, as well as in the whole fetus, was determined. The aluminum concentration was statistically significantly increased in the liver, bone, and placenta of the test animals compared to controls; however, no aluminum was detected (detection limit 0.05 µg/g) in the whole fetuses of control or treated animals.
Distearyl citrate ester mixture
A multigeneration study was performed in which 4 generations of rats were fed a diet containing 0%, 1.9%, or 9.5% of the distearyl citrate ester mixture that was described earlier in this report. 29 Administration of the test article did not result in any reproductive or developmental effects or any general signs of toxicity.
In Vitro
Sodium citrate
The embryotoxic potential of sodium citrate was evaluated in a whole rodent embryo culture system using 9.5-day-old embryos from female Han Wistar rats without metabolic activation. 33 The no-effect concentration for all parameters evaluated, including crown-rump length and abnormalities, was >115 µmol/L sodium citrate.
Spermicidal Effects
Citric acid
The spermicidal effect of citric acid was determined by suspending human sperm in a solution of citric acid. 34 Addition of 0.1% citric acid to human sperm reduced pH and rendered sperm immotile within 30 minutes while 1% was almost instantly spermicidal. The effect on sperm penetration of cervical mucus was also evaluated by adding the acid to human cervical mucus in capillary tubes. Addition of 0.01% citric acid reduced, and addition of 0.1% completely abolished, sperm penetration.
Genotoxicity
Genotoxicity studies are summarized in Table 10. Citric acid and its salts and esters were mostly negative in in vitro and in vivo genotoxicity tests. Exceptions were weakly positive results in host-mediated assays with citric acid, equivocal results in an Ames test with aluminum citrate, and a weak dose-related response for sodium citrate in a suspension test in Salmonella typhimurium TA1537 that was not reproducible.
Genotoxicity Studies.
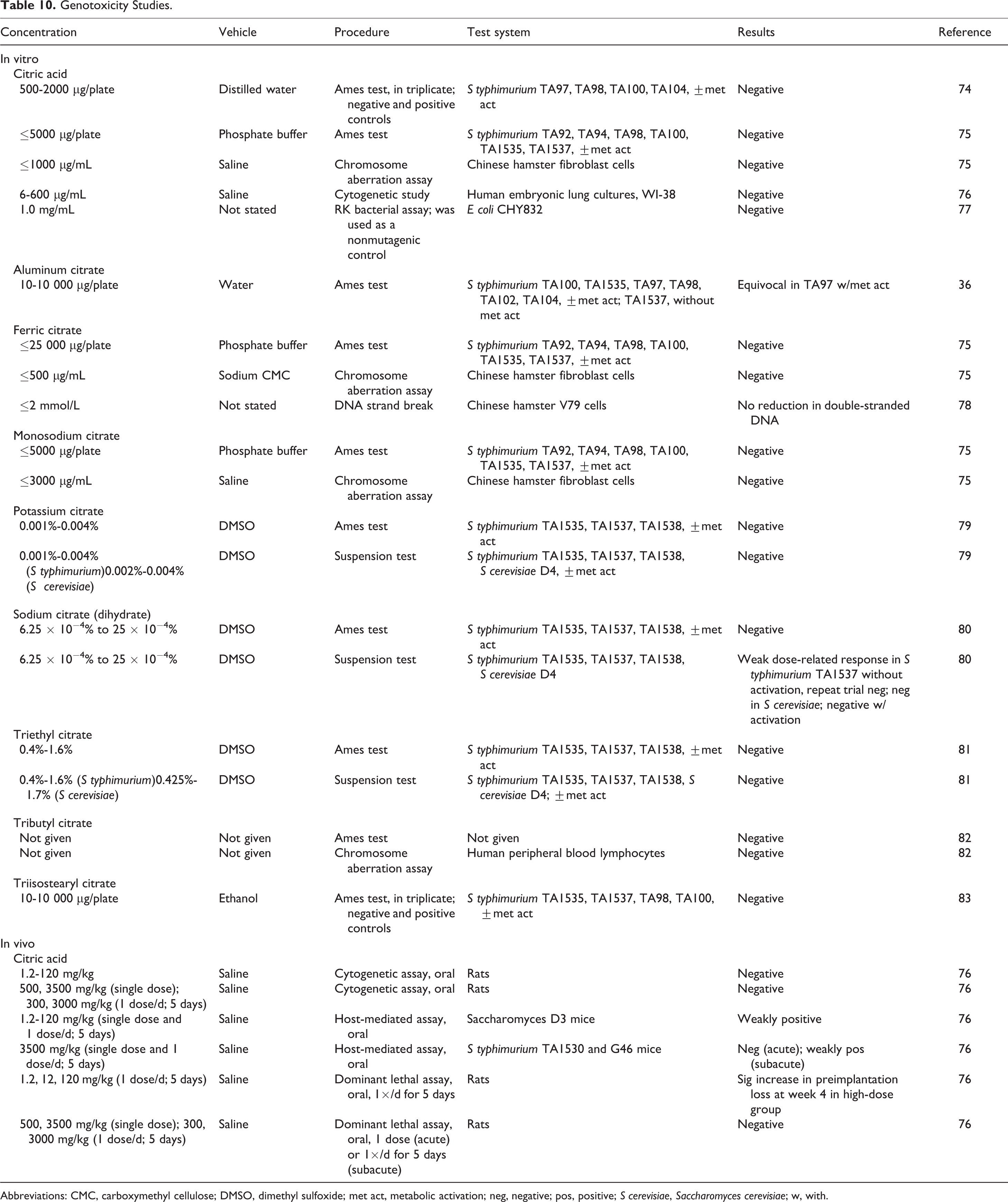
Abbreviations: CMC, carboxymethyl cellulose; DMSO, dimethyl sulfoxide; met act, metabolic activation; neg, negative; pos, positive; S cerevisiae, Saccharomyces cerevisiae; w, with.
Antimutagenic Effects
Citric acid
The antimutagenic effect of citric acid was evaluated in an Ames test, with 4-nitro-1,2-phenylenediamine and sodium azide used as mutagens. 35 Using S typhimurium strain TA97, concentrations of 1 to 1000 µg/0.1 mL/plate citric acid inhibited the mutagenicity of 20 µg/0.1 mL/plate 4-nitro-1,2-phenylenediamine by 3.54% to 67.72% without metabolic activation and by 55.34% to 71.97% with metabolic activation. Using strain TA100, concentrations of 1 to 1000 µg/0.1 mL/plate citric acid inhibited the mutagenicity of 1.5 µg/0.1 mL/plate sodium azide by 15.47% to 50.65% without metabolic activation and 37.47% to 67.10% with metabolic activation.
Carcinogenicity
Aluminum Citrate
The National Toxicology Program has planned toxicity/carcinogenicity testing for aluminum citrate. 36 The rationale for testing is that aluminum is listed by the EPA as a drinking water contaminant with a high health research priority.
Irritation and Sensitization
Skin Irritation/Sensitization
Nonhuman and human skin irritation and sensitization studies are summarized in Table 11. In irritation studies in rabbits, 30% citric acid was not a primary irritant, 60% produced some erythema and edema that subsided with time, and undiluted citric acid produced mild to severe erythema and mild to moderate edema. Triethyl citrate, at concentrations up to 100%, was not an irritant in guinea pigs or rabbits, and trioctyldodecyl citrate applied neat was not a primary skin irritant in rabbits. In human studies, citric acid was not a dermal irritant at concentrations up to 5% aqueous (aq), and 20% triethyl citrate was not irritating in humans. Sodium citrate did not produce any immediate (nonimmunologic contact urticaria) reactions. In sensitization testing, a cuticle cream containing 4% citric acid was not an irritant or a sensitizer in humans; 2.5% aq citric acid produced positive results in skin prick test in 3 of the 91 patients with urticaria or angioedema. Triethyl citrate, applied undiluted during epidermal induction, was a strong sensitizer in a guinea pig maximization test but 20% in pet was not a primary irritant or sensitizer in human studies. Trioctyldodecyl citrate was a mild sensitizer in a local lymph node assay when applied neat but the same concentration was not an irritant or sensitizer in human studies. Tributyl citrate (concentration not stated) was not a sensitizer in animal studies. In human studies, 25% tristearyl citrate and 100% triisostearyl citrate were not irritants or sensitizers in repeated insult patch tests.
Dermal Irritation and Sensitization.
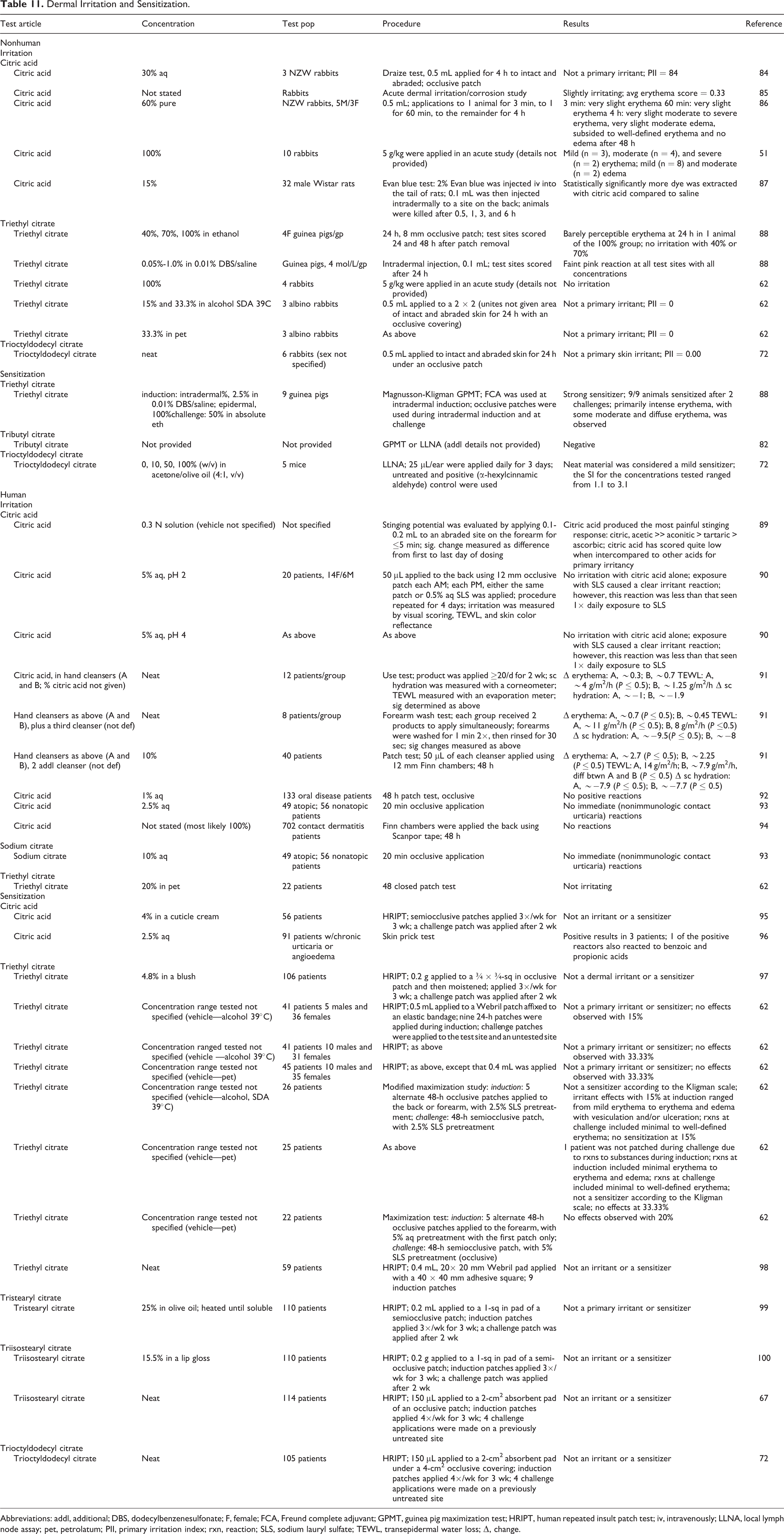
Abbreviations: addl, additional; DBS, dodecylbenzenesulfonate; F, female; FCA, Freund complete adjuvant; GPMT, guinea pig maximization test; HRIPT, human repeated insult patch test; iv, intravenously; LLNA, local lymph node assay; pet, petrolatum; PII, primary irritation index; rxn, reaction; SLS, sodium lauryl sulfate; TEWL, transepidermal water loss; Δ, change.
Ocular Irritation
Ocular irritation studies are summarized in Table 12. Citric acid was predicted to be a moderate/severe to severe/extreme ocular irritant in in vitro studies, and it was minimally irritating to rabbit eyes at a concentration of 10% and mildly irritating at a concentrations 30%. In in vitro studies, tri-isostearyl citrate was predicted to be nonirritating to eyes. Triethyl citrate, 33.3%, did produce irritation in rabbit eyes and undiluted trioctyldodecyl citrate was nonirritating.
Ocular Irritation Studies.
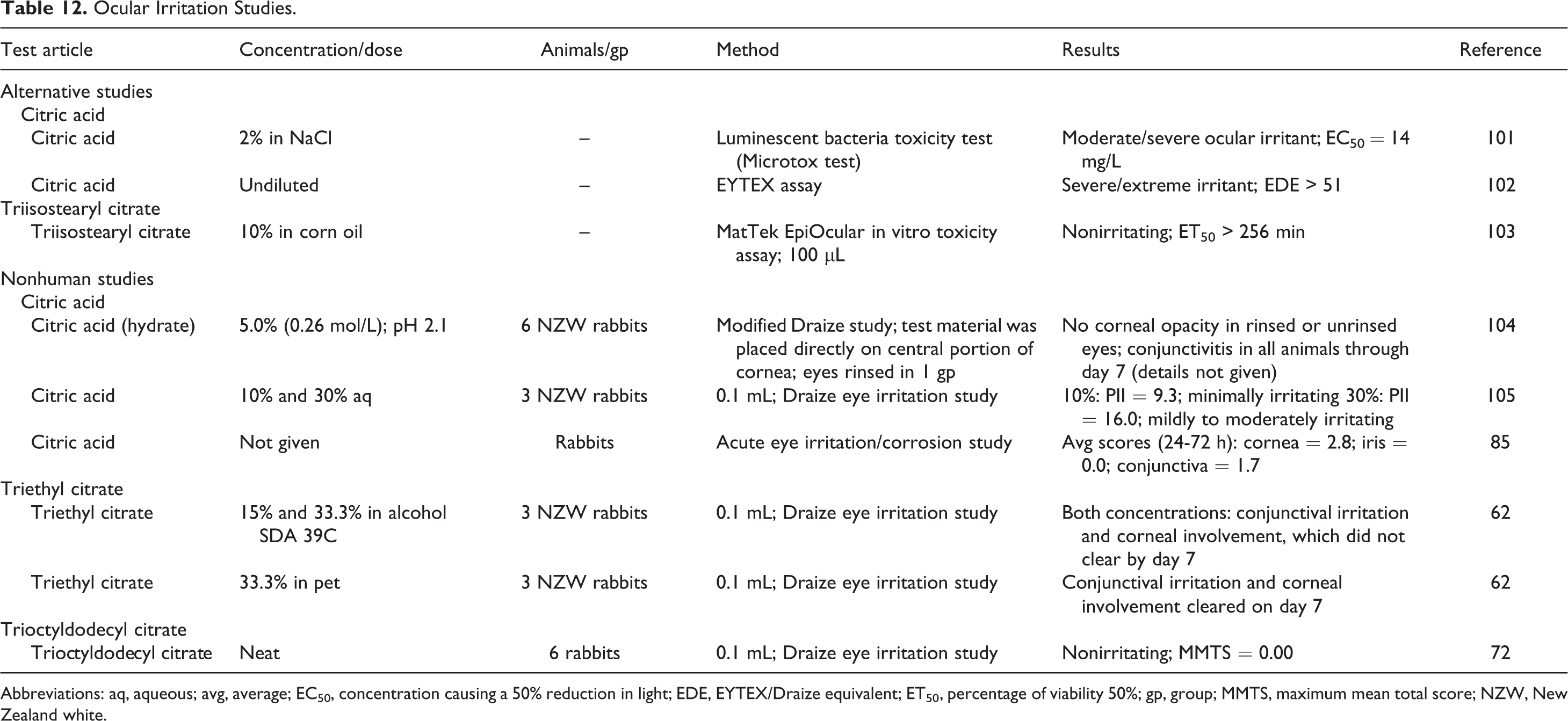
Abbreviations: aq, aqueous; avg, average; EC50, concentration causing a 50% reduction in light; EDE, EYTEX/Draize equivalent; ET50, percentage of viability 50%; gp, group; MMTS, maximum mean total score; NZW, New Zealand white.
Miscellaneous Studies
Effects in Skin
Citric acid
The effect of 1 mol/L (16%, w/w) citric acid on skin cell renewal and irritation (as stinging) was determined at a pH of 3, 5, and 7. The dansyl chloride method was used to determine skin cell renewal and irritation was evaluated subjectively as stinging in the nasal fold area; stinging was scored on a scale of 0 to 4 every minute for 15 minutes. 37 (It is not stated, but the assumed maximum score is 60.) Citric acid test product of 2 mg/cm2 was applied to the test area on the volar forearm of humans 2×/daily. The vehicle consisted of 15% ethanol (specially denatured 40), 5% ethoxydiglycol, 5% butylene glycol, and water. Cell renewal was measured in at least 8 patients; citric acid increased cell renewal by 16.1%, 12.8%, and 3% at pH 3, 5, and 7, respectively. Using a minimum of 10 patients, the irritation scores for 1 mol/L citric acid at pH 3, 5, and 7 were 38, 35.4, and 23.6, respectively.
The effect of 5% citric acid on skin cell renewal and irritation was also evaluated at the same pHs. 38 Cell renewal was greater at this concentration; 18%, 14%, and 8% increases were seen with 5% citric acid at pH 3, 5, and 7, respectively. Irritation scores (as stinging) were 2.3, 2.1, and 1.1 (on a scale of 1-5) at pH 3, 5, and 7, respectively. (Details of application were not provided.)
Five male patients participated in a 30-day study to evaluate the effects of citric acid on skin morphology. 39 Cream formulations containing 10%, 20%, or 25% citric acid were evaluated, and 0.2 mL of each cream were applied to a 2 × 2 cm2 area of the ventral forearm of each patient. A fourth site on the forearm was used as an untreated control. Occlusive patches, 3×/wk, were applied during week 1 and nonocclusive patches, 3×/wk, were applied during weeks 2 to 3. Open applications were made daily during week 4. At the end of dosing, a 3-mm punch biopsy was taken from each site. Irritation was observed with the 20% and 25% formulations. (Details as to the extent of irritation was not provided, other than it was “visible.”) Microscopically, an increase in viable epidermal thickness that increased with dose was observed at all dose levels, a “substantial” increase in Langerhans cells was observed with the 20% and 25% citric acid creams, and glycosaminoglycan (GAG) content was “markedly” increased at the sites dosed with 20% and 25% citric acid compared to that seen at the untreated and 10% citric acid sites.
A 20% citric acid lotion, pH 3.5, was applied twice daily for 3 months to photodamaged skin of the forearm of 6 female patients. 40 The lotion vehicle without citric acid was applied to the contralateral arms as a control. A 4-mm punch biopsy specimen was taken from each site after 3 months of application. Application of the lotion containing citric acid produced a statistically significant increase in skinfold thickness, with a 16.3% increase from baseline recorded. The skinfold thickness of the vehicle-treated skin decreased slightly. Viable epidermis thickness also increased in a statistically significant manner, increasing 40% when compared to untreated skin. A statistically significant increase in GAG content was evidenced by a 2.5-fold increase in epidermal hyaluronic acid staining, a 57% increase in dermal hyaluronic acid staining, and a 66% increase in dermal chondroitin sulfate staining, as compared to skin treated with vehicle only. (Although the percentage of increase in staining was greater for chondroitin sulfate, staining for hyaluronic acid was approximately double that of chondroitin sulfate in both vehicle and citric acid-treated sites.)
Seven patients with moderate to severe photoaged skin applied a lotion containing 25% citric acid, pH 3.5, to 1 forearm and a placebo lotion to the other forearm twice daily for 6 months. 41 (Similar lotions containing glycolic or lactic acid were also evaluated.) Skin thickness measurements were performed in triplicate throughout the study. The 2-skin layer thickness of the forearm treated with citric acid (and the other AHAs) increased 25% while the thickness of the control forearm decreased 2%; the difference between the citric acid and the control sites was statistically significant. (There was no statistically significant difference in skin thickness among the 3 AHAs tested.) Microscopically, the mean epidermal thickness of skin and the mean thickness of papillary dermis in samples of skin treated with the citric acid lotion were statistically significantly greater than controls. (Total number of samples examined microscopically was not given.) There was no indication of inflammation. The amount of ground substance was variably increased in the citric acid-treated samples. Collagen fibers appeared to be increased in treated skin samples, but there was not a statistically significant difference in collagen fiber density in the papillary dermis between AHA-treated and -untreated sites.
It has been hypothesized that AHAs have the following mechanism of action. 24 In the stratum corneum, a low concentration of AHAs diminish corneocyte cohesion. In keratinocytes, AHAs stimulate epidermal proliferation, possibly by improving energy and redox status of the keratinocytes. In fibroblasts, high concentrations of AHA in an appropriate vehicle are thought to induce epidermolysis and epidermal separation and impact the papillary dermis and reticular dermis, leading to dermal changes that include the synthesis of new collagen.
Case Report
Citric acid
A woman reported difficulty in breathing and severe facial pain 4 hours after a professionally administered cosmetic peel procedure with a product containing 10% citric acid (and other compounds that were not identified). 41 The facial peel was applied for 4 hours. The patient also had first- and second-degree burns to the face and anterior neck. Permanent facial and neck scars, but no airway pathology, resulted.
Cough Reflex
Citric acid
Citric acid was used as a tussive agent in cough challenge testing. 43 Ten humanswere exposed to incremental doses of citric acid (10-1000 mmol/L) using an air-driven nebulizer. Using the mean cough frequency, a statistically significant dose–response relationship was observed. Individuals had different threshold and maximum tolerable concentrations; using interpolated values, the concentration that caused 5 coughs was 141.3 mmol/L citric acid. Using 10 Dunkin Hartley guinea pigs exposed to 0.9% saline and then, 10 minutes later, a single challenge of 30 to 300 mmol/L citric acid for 2 minutes, the calculated concentration producing 5 coughs (in 10 minutes) was 74.1 mmol/L citric acid.
The cough reflex to citric acid is produced by irritation of the larynx and the trachea and thought to be mediated by receptors that are distributed mainly in the larynx and upper airways. 44 In humans, the cough reflex was decreased with higher inspiratory flow rates as opposed to lower rates. The researchers were not able to definitively state a reason the decrease was seen but did state an important factor may be laryngeal deposition of the aerosol.
The mechanism of irritant properties was examined by comparing the cough response of isotonic citric acid in saline, isotonic sodium citrate, sodium citrate in saline, isotonic
Citric acid induced airway constriction in anesthetized Hartley guinea pigs. 46 A citric acid aerosol was generated from a 0.6-mol/L citric acid solution and each animal received 50 breaths of 4 mL of the solution using a nebulizer. At 2 to 3 minutes following exposure to citric acid, the aerosol induced significant airway constriction that persisted to the end of the study (20 minutes following administration).
In another study, anesthetized guinea pigs were administered 10% weight/volume (w/v) aq aerosol citric acid for 1 minute using a nebulizer; airway resistance increased 79% and lung compliance decreased 68%. 47 In anesthetized guinea pigs in which the vagal nerve had been cut, a 5% increase in resistance and compliance was seen following exposure to citric acid. In conscious guinea pigs exposed to a 10% w/v aq aerosol of citric acid for 2 minutes using a glass nebulizer (particle size, 0.5-4 µm), the animals coughed 1 to 2 times in the first 30 seconds, and then a short period of hyperventilation was observed. The researchers theorized that the bronchoconstriction was due to an increase in airway resistance and involved parasympathetic innervation.
Anesthetic Effects
Triethyl and tributyl citrate
The corneal reflex in rabbit eyes was temporarily eliminated upon instillation of 3 drops of a 5% suspension of triethyl or tributyl citrate in 3% acacia; the number of animals used was not stated. 30 The anesthetic effect was confirmed by the intradermal administration of 0.1 mL of a 2% solution of triethyl or tributyl citrate into an area of the shaved back of guinea pigs; again, the number of animals used was not stated. Triethyl citrate resulted in insensitivity to pricking of the area lasting 12 to 20 minutes while tributyl citrate produced a “deadened area” for a period greater than 2 hours.
Summary
Citric acid is an α-hydroxytricarboxylic acid that is reported to function in cosmetics as a chelating agent, pH adjuster, or fragrance ingredient. Citric acid can also be classified as a β-hydroxy acid. The 12 inorganic salts are reported to have many diverse functions while the 20 alkyl esters are reported to function mostly as skin-conditioning agents although they can have other functions. Citric acid is used in almost every category of cosmetic product and has 6795 reported uses. Citric acid is reported to be used at concentrations up to 39% in products that are diluted for (bath) use and up to 4% in leave-on products. With the exception of sodium, tributyl, and triethyl citrate, all other in-use ingredients have less than 50 uses. Triisostearyl citrate is used at up to 80% in lipstick formulations. Trioctyldodecyl and tricaprylyl citrate are used at concentrations of 30% and 27%, respectively, in leave-on formulations; all other in-use ingredients are used at ≤12%.
Citric acid, calcium citrate, ferric citrate, manganese citrate, potassium citrate, sodium citrate, diammonium citrate, isopropyl citrate, stearyl citrate, and triethyl citrate are GRAS direct food additives. These ingredients, plus magnesium citrate, distearyl citrate, tristearyl citrate, and tributyl citrate are FDA-approved indirect food additives.
Citric acid is ubiquitously found in nature in virtually all organisms as an intermediate of the Krebs cycle. Orally administered citric acid is well absorbed and largely metabolized. Oral administration of aluminum citrate to male Sprague-Dawley rats, 6 days/wk for 4 weeks, resulted in a statistically significant increase in levels of aluminum in the brain in 1 study. In another study in which Sprague-Dawley rats were given aluminum citrate in the drinking water for 8 months, aluminum levels were increased in other parts of the body but not in the brain. Distearyl citrate, when added to the diet of rats, was poorly absorbed while nearly complete absorption was observed when isopropyl citrate was administered in the diet of rats.
The dermal median lethal dose values for citric acid and triethyl citrate were >5 g/kg in rabbits. Results of oral, inhalation, and other parenteral single-dose studies with various citrates did not indicate any notable toxic effects in mice, rats, rabbits, or dogs. Administration of 80 mmol/L aluminum citrate in water for 8 months did not affect the body weights of rats. Repeated oral dosing with an isostearyl citrate ester mixture or a distearyl citrate ester mixture did not have adverse effects on rats, rabbits, or dogs. Repeated oral dosing with tributyl citrate did not have an adverse effect on rats (10% in the diet for 6 weeks) or cats (5 mL/kg for 2 months).
Oral administration of 1064 mg/kg bw aluminum citrate concurrent with 62 mg/kg bw citric acid to rats was not maternally-, embryo-, or fetotoxic; the aluminum concentration was statistically significantly increased in the liver, bone, and placenta of the test animals but no aluminum was detected in the control or treated-group fetuses. Dietary administration of up to 9.5% of a distearyl citrate ester mixture did not produce any reproductive or developmental effects in a multigenerational study.
Citric acid and its salts and esters gave mostly negative reports in in vitro and in vivo genotoxicity tests. Exceptions were weakly positive results in in vitro and in vivo host-mediated assays with citric acid, equivocal results in an Ames test with aluminum citrate, and a weak dose-related response in a suspension test with sodium citrate in S typhimurium TA1537 that was not reproducible. Citric acid had antimutagenic effects, inhibiting the mutagenicity of 4-nitro-1,2-phenylenediamine and sodium azide.
In irritation studies in rabbits, 30% citric acid was not a primary irritant, 60% produced some erythema and edema that subsided with time, and undiluted citric acid produced mild to severe erythema and mild to moderate edema. Triethyl citrate, at concentrations up to 100%, was not an irritant in guinea pigs or rabbits, and trioctyldodecyl citrate applied neat was not a primary skin irritant in rabbits. In human studies, citric acid was not a dermal irritant at concentrations up to 5% aq and 20% triethyl citrate was not irritating in humans. Sodium citrate did not produce any immediate (nonimmunologic contact urticaria) reactions.
In sensitization testing, a cuticle cream containing 4% citric acid was not an irritant or a sensitizer in humans; 2.5% aq. citric acid produced positive results in skin prick test in 3 of the 91 patients with urticaria or anigoedema. Triethyl citrate, applied undiluted during epidermal induction, was a strong sensitizer in a guinea pig maximization test but 20% in pet was not a primary irritant or sensitizer in human studies. Trioctyldodecyl citrate was a mild sensitizer in a local lymph node assay when applied neat but the same concentration was not an irritant or sensitizer in human studies. Tributyl citrate (concentration not stated) was not a sensitizer in animal studies. In human studies, 25% tristearyl citrate and 100% triisostearyl citrate were not irritants or sensitizers in repeated insult patch tests.
Citric acid was predicted to be a moderate/severe to severe/extreme ocular irritant in in vitro studies, and it was minimally irritating to rabbit eyes at a concentration of 10% and mildly irritating at a concentration of 30%. In in vitro studies, triisostearyl citrate was predicted to be nonirritating to eyes. Triethyl citrate, 33.3%, did produce irritation in rabbit eyes, and undiluted trioctyldodecyl citrate was nonirritating.
Citric acid, ≥5%, increased cell renewal and epidermal thickness in human skin, and there appeared to be a greater increase at higher concentrations and/or lower pH of citric acid. Citric acid was a tussive agent in human inhalation challenge tests and induced airway restriction in animals. The cough reflex to citric acid is produced by irritation of the larynx and the trachea and is thought to be mediated by receptors that are distributed mainly in the larynx and upper airways. Triethyl and tributyl citrate had an anesthetic effect in rabbit eyes.
Discussion
The Panel considered that the oral safety of citric acid, calcium citrate, ferric citrate, manganese citrate, potassium citrate, sodium citrate, diammonium citrate, isopropyl citrate, stearyl citrate, and triethyl citrate has been well substantiated in that these ingredients are GRAS direct food additives. Therefore, the focus of this safety assessment was on the dermal toxicity of these ingredients. Although there are data gaps, the chemical structures, physicochemical properties, and functions and concentrations in cosmetics allow grouping these ingredients together and extending the available toxicological data to support the safety of the entire group.
Because citric acid and some of its salts and esters can be used in products that may be aerosolized, the Panel discussed the issue of incidental inhalation exposure. The limited inhalation data address the cough reflex induced by inhalation exposure to citric acid using a nebulizer so the induction of the cough reflex was not relevant to cosmetic exposure. Since inhalation data were limited, the Panel considered other available data to characterize the potential for citric acid and some of its salts and esters to cause systemic toxicity, irritation, or sensitization. They noted that as discussed earlier, many of these ingredients are GRAS ingredients and therefore oral toxicity was not a concern with these GRAS ingredients, that these ingredients gave mostly negative reports in in vitro and in vivo genotoxicity tests, and that they were not irritants or sensitizers in clinical testing.
The maximum reported concentrations of citric acid used in a spray product is 0.7%, of a salt is 0.2% sodium citrate, of an ester is 4% trioctyldodecyl citrate, and in deodorants is 2% triethyl citrate. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. Inhaled droplets/particles deposited in the nasopharyngeal and bronchial regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. Nevertheless, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
The Panel discussed whether citric acid or any of its salts or alkyl esters would be irritants. Available repeated insult patch testing at the highest leave-on concentration of 4% citric acid demonstrated an absence of both dermal irritation and sensitization, suggesting that these ingredients would not be irritants in formulation.
Although citric acid can be considered an AHA, it is also a β-hydroxy acid. Structurally, citric acid is a tricarboxylic acid, and as such, has a unique functionality and is chemically and biologically distinct from the AHAs considered in the CIR safety assessment of AHAs (ie, glycolic and lactic acid). Therefore, the concerns that stem from the mode of action of AHAs was not considered relevant to citric acid and its inorganic salts and alkyl esters.
Conclusion
The Panel concluded that citric acid and the inorganic citrate salts and alkyl citrate esters, listed subsequently, are safe in the present practices of use and concentration. Citric acid Inorganic salts: aluminum citrate; calcium citrate*; copper citrate*; diammonium citrate; disodium cupric citrate*; ferric citrate; magnesium citrate; manganese citrate*; monosodium citrate; potassium citrate; sodium citrate; zinc citrate. Alkyl esters: isodecyl citrate; isopropyl citrate*; stearyl citrate; dilauryl citrate; distearyl citrate*; tributyl citrate; tri-C12-13 alkyl citrate; tri-C14-15 alkyl citrate; tricaprylyl citrate; triethyl citrate; triethylhexyl citrate; trihexyldecyl citrate*; triisocetyl citrate; triisopropyl citrate*; trilauryl citrate*; trioctyldodecyl citrate; trioleyl citrate*; triisostearyl citrate; tristearyl citrate*; ethyl citrates.
Were ingredients in this group not in current use (as indicated by *) to be used in the future, the expectation is that they would be used at concentrations comparable to others in this group.
Footnotes
Auhtors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
