Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 62 dimethicone crosspolymer ingredients as used in cosmetics. These ingredients function mostly as absorbents, bulking agents, film formers, hair-conditioning agents, emollient skin-conditioning agents, slip modifiers, surface modifiers, and nonaqueous viscosity-increasing agents. The Panel reviewed available animal and human data related to these polymers and addressed the issue of residual monomers. The Panel concluded that these dimethicone crosspolymer ingredients are safe in the practices of use and concentration as given in this safety assessment.
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook,
1
these 62 dimethicone crosspolymers mostly function as absorbents, bulking agents, film formers, hair-conditioning agents, emollient skin-conditioning agents, slip modifiers, surface modifiers, and nonaqueous viscosity-increasing agents (Table 1). The ingredients included in this report are: acrylates/bis-hydroxypropyl dimethicone crosspolymer; behenyl dimethicone/bis-vinyldimethicone crosspolymer; bis-phenylisopropyl phenylisopropyl dimethicone/vinyl dimethicone crosspolymer; bis-vinyldimethicone/bis-isobutyl PPG-20 crosspolymer; bis-vinyldimethicone crosspolymer; bis-vinyldimethicone/PEG-10 dimethicone crosspolymer; bis-vinyldimethicone/PPG-20 crosspolymer; butyldimethicone methacrylate/methyl methacrylate crosspolymer; C30-45 alkyl cetearyl dimethicone crosspolymer; C4-24 alkyl dimethicone/divinyldimethicone crosspolymer; C30-45 alkyl dimethicone/polycyclohexene oxide crosspolymer; cetearyl dimethicone crosspolymer; cetearyl dimethicone/vinyl dimethicone crosspolymer; cetyl dimethicone/bis-vinyldimethicone crosspolymer; cetyl hexacosyl dimethicone/bis-vinyldimethicone crosspolymer; crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer; dimethicone/bis-isobutyl PPG-20 crosspolymer; dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer; dimethicone crosspolymer; dimethicone crosspolymer-3; dimethicone/divinyldimethicone/silsesquioxane crosspolymer; dimethicone/lauryl dimethicone/bis-vinyldimethicone crosspolymer; dimethicone/PEG-10 crosspolymer; dimethicone/PEG-10/15 crosspolymer; dimethicone/PEG-15 crosspolymer; dimethicone/phenyl vinyl dimethicone crosspolymer; dimethicone/polyglycerin-3 crosspolymer; dimethicone/PPG-20 crosspolymer; dimethicone/titanate crosspolymer; dimethicone/vinyl dimethicone crosspolymer; dimethicone/vinyltrimethylsiloxysilicate crosspolymer; diphenyl dimethicone crosspolymer; diphenyl dimethicone/vinyl diphenyl dimethicone/silsesquioxane crosspolymer; divinyldimethicone/dimethicone crosspolymer; hydroxypropyl dimethicone/polysorbate 20 crosspolymer; isopropyl titanium triisostearate/triethoxysilylethyl polydimethylsiloxyethyl dimethicone crosspolymer; lauryl dimethicone PEG-15 crosspolymer; lauryl dimethicone/polyglycerin-3 crosspolymer; lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer; PEG-10 dimethicone crosspolymer; PEG-12 dimethicone crosspolymer; PEG-8 dimethicone/polysorbate 20 crosspolymer; PEG-12 dimethicone/bis-isobutyl PPG-20 crosspolymer; PEG-12 dimethicone/PPG-20 crosspolymer; PEG-10 dimethicone/vinyl dimethicone crosspolymer; PEG-10/lauryl dimethicone crosspolymer; PEG-15/lauryl dimethicone crosspolymer; PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer; perfluorononyl dimethicone/methicone/amodimethicone crosspolymer; polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer; polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer; silicone quaternium-16/glycidoxy dimethicone crosspolymer; styrene/acrylates/dimethicone acrylate crosspolymer; trifluoropropyl dimethicone/PEG-10 crosspolymer; trifluoropropyl dimethicone/trifluoropropyl divinyldimethicone crosspolymer; trifluoropropyl dimethicone/vinyl trifluoropropyl dimethicone/silsesquioxane crosspolymer; trimethylsiloxysilicate/dimethicone crosspolymer; vinyl dimethicone/lauryl/behenyl dimethicone crosspolymer; vinyl dimethicone/lauryl dimethicone crosspolymer; vinyl dimethicone/methicone silsesquioxane crosspolymer; vinyldimethyl/trimethylsiloxysilicate/dimethicone crosspolymer; vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer.
Definitions and Functions of the Ingredients in This Safety assessment. 1,a

aThe italicized text represents additions made by CIR staff.
Several of the components of these ingredients have been previously reviewed by the Panel including dimethicone, which was found to be safe as a cosmetic ingredient (Table 2). 2
Component Ingredients Previously Reviewed by CIR.

Chemistry
Overview and Method of Manufacture
Definitions, functions in cosmetics, and CAS numbers for the ingredients addressed in this safety assessment are provided in Table 1.
These cosmetic ingredients are silicone elastomers comprising dimethicone copolymers cross-linked with a bifunctional agent. Idealized structures are shown in Figure 1.
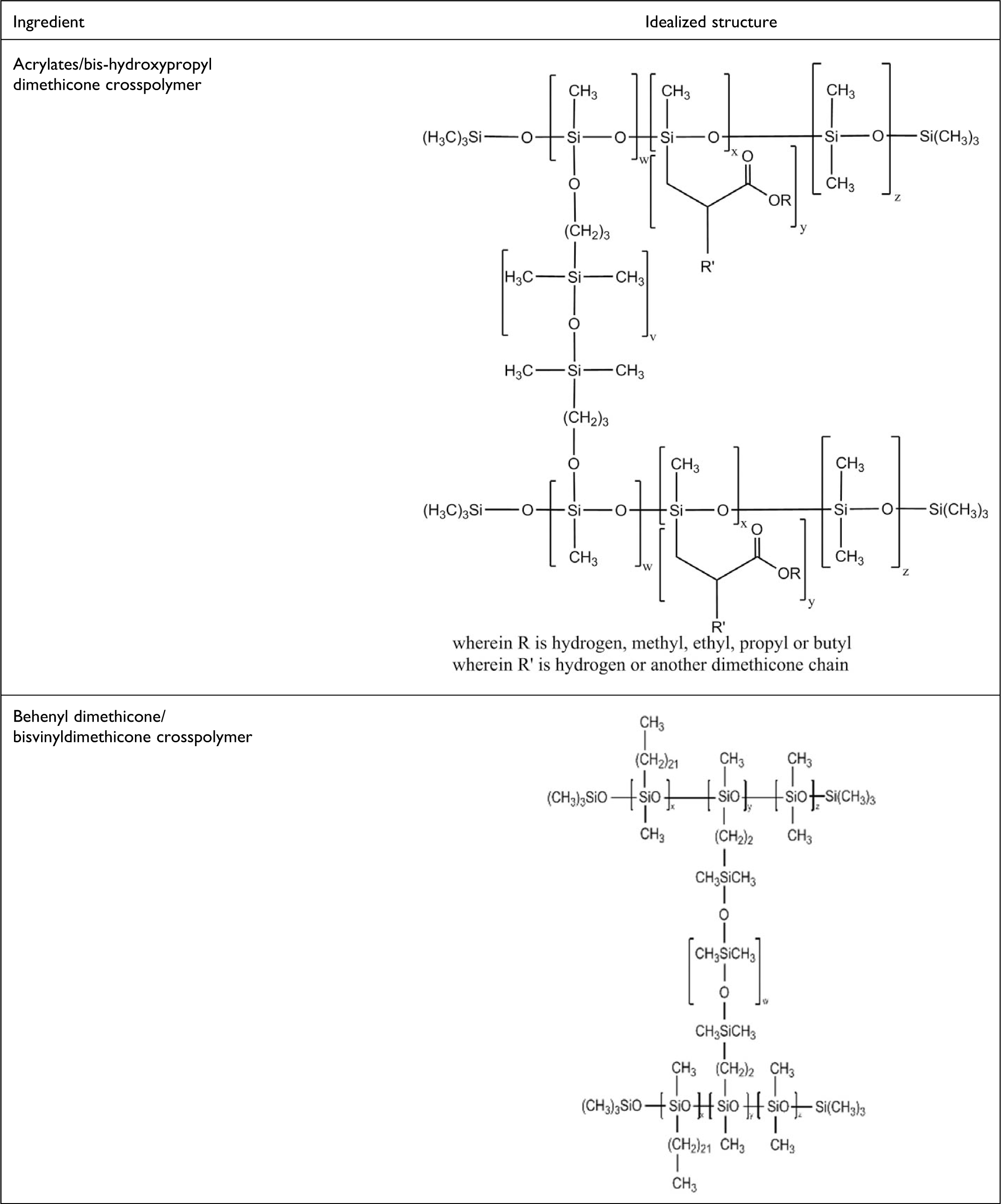
Idealized structures of the dimethicone crosspolymers ingredients in this safety assessment. These idealized structures are merely generalized, 2-dimensional depictions of the true 3-dimensional frameworks that comprise these polymers. Although monomer units are drawn sequentially, by necessity, this by no means implies that these are block-type polymers. Instead, these structures are meant to represent only 1 example of the multitude of potentially produced connectivities found within these macromolecules.
For use in cosmetics, these crosspolymers are typically supplied to finishing houses as swollen gels (ie, trade name mixtures) that contain various oils (eg, silicone oils such as dimethicone). 3 The addition of hydrophilic components (eg, addition of polyethylene glycol [PEG] chains to produce dimethicone/PEG-10 crosspolymer) or hydrophobic components (eg, addition of long alkyl chains to produce behenyl dimethicone/bis-vinyldimethicone crosspolymer) affects both the chemical and the rheological properties of the resultant ingredient. Accordingly, dimethicone crosspolymers represent a wide variety of materials ranging from liquids to elastomeric solids.
The majority of the ingredients in this review are produced by cross-linking dimethicone polymeric chains via a hydrosilation reaction. 3 This reaction consists of the addition of silicon hydride bonds (SiH) within the dimethicone polymer backbones across vinyl bonds within the selected cross-linking agents (Figure 2). These reactions usually require a catalyst, such as platinum. The reactions are rapid and produce chemically stable products. Since these reactions are net additions across a double bond, the only expected by-products are the starting materials, particularly the catalysts.
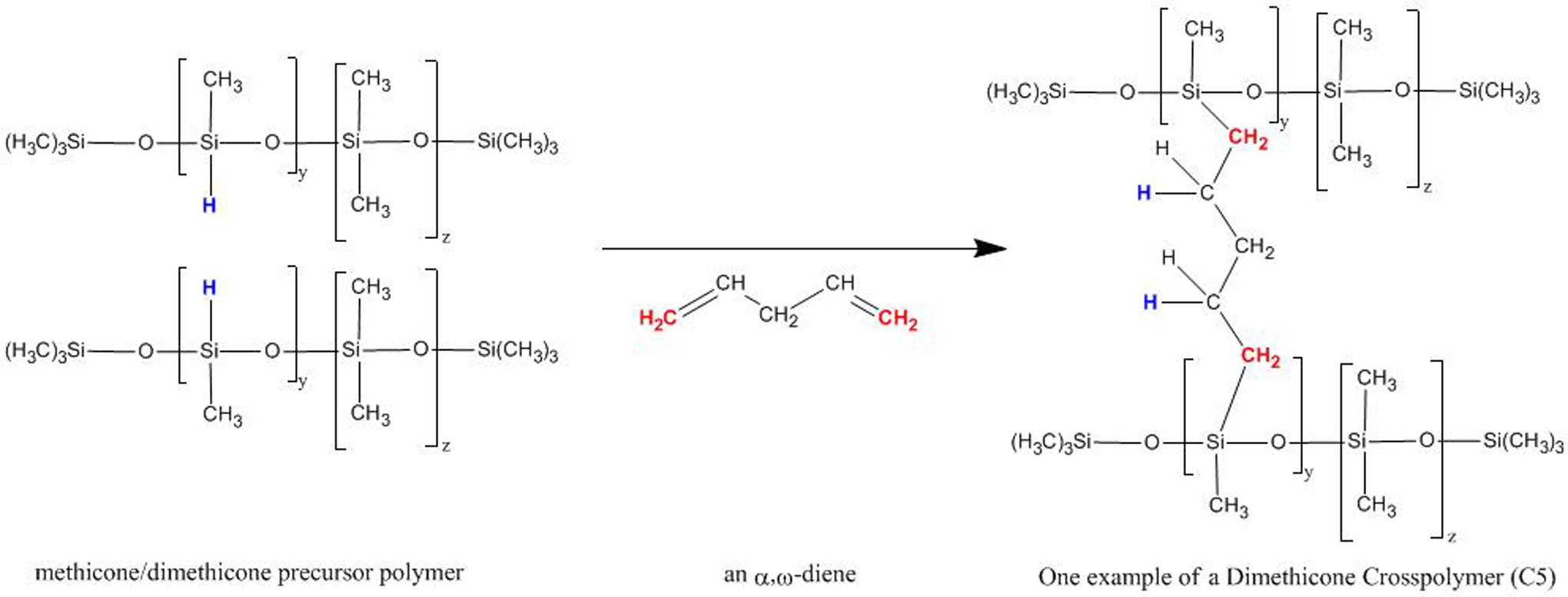
Example of the hydrosilation cross-linking of a dimethicone precursor polymer.
In some silicone polymers such as dimethicone, which has no silicon–hydrogen bonds, some amount of silicon hydride may exist. However, a silicone precursor polymer is made in order to add the silicon hydride groups that are utilized for the cross-linking process. 3 For example, a dimethicone precursor polymer is made by the copolymerization of dimethyl siloxane units with methylhydrogen siloxane units. Accordingly, although we define dimethicone crosspolymer as “a polymer of dimethicone cross-linked with a C3 to C20 alkyl group,” it is more likely that dimethicone crosspolymer is a methicone/dimethicone copolymer (methicone has 1 methyl and 1 hydrogen on each silicon in the polymer backbone. This contrasts with dimethicone, which has 2 methyl groups on each silicon in the polymer backbone) cross-linked with an α,ω-diene (ie, the double bonds are at the ends of the chain) that is 3 to 20 carbons long.
Physical and Chemical Properties
Available information on the physical and chemical properties is provided in Table 3. Notable among these data are that these crosspolymers are not water soluble. Other data are provided subsequently.
Chemical and Physical Properties of Dimethicone Crosspolymers.

Crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer
Crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer is stable at <20°C in a sealed container protected from light for at least 12 months. 4
Dimethicone crosspolymer
In a product mixture containing dimethicone crosspolymer (12% in cyclomethicone), the crosspolymer has a molecular weight of >15 500 to 1 000 000. 5 The product is a clear/slightly translucent paste.
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer is stable at room temperature for 36 months. 6
Dimethicone/vinyltrimethylsiloxysilicate crosspolymer
Dimethicone/vinyltrimethylsiloxysilicate crosspolymer is provided by a manufacturer as a mixture with cyclopentasiloxane that creates a semitransparent gel with thixotropic properties. 7
PEG-12 dimethicone crosspolymer
PEG-12 dimethicone is an amphiphilic molecule; the PEG-12 moieties are hydrophilic whereas the dimethicone backbone is lipophilic. 8
Vinyl dimethicone/methicone silsequioxane crosspolymer
Vinyl dimethicone/methicone silsequioxane crosspolymer products were reported to have specific gravity ranging from 0.98 to 1.11. 9 These products were reported to be white, spherical powders.
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer (20% in isododecane) is stable for at least 1 year with no special storage requirements. 10 The mixture with isododecane reportedly creates a semitransparent gel with thixotropic properties.
Particle Size
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer was reported in a patent to be spherical-shaped particles with diameters ranging from 2 to 10 μm. 11 In finished products, even in those that are powders, these particles generally aggregate stably to produce much larger particles.
A manufacturer’s product information sheet reported that vinyl dimethicone/methicone silsesquioxane crosspolymer had an average particle size range of 2 to 30 μm, depending on the product. 9 Diphenyl dimethicone/vinyl diephenyl dimethicone/silsequioxane crosspolymer has an average particle size of 5 μm.
Impurities
Crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer
Crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer is reported to not contain any heavy metals, polycyclic aromatic hydrocarbons, organohalogens, or nitrosamines. 4 Residuals from manufacturing included tert-butanol (<100 ppm), isododecane (<1000 ppm), vinyl acetate (≤100 ppm), vinyl tert-decanoate (≤2000 ppm), crotonic acid (≤200 ppm), and trace amounts of isopropanol and ethyl acetate.
Dimethicone crosspolymer
A manufacturer’s product containing dimethicone crosspolymer was reported to have no hazardous impurities. 5
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer was reported to be 100% pure by a manufacturer. 6 The same manufacture reported the content of heavy metals to be <20 ppm and arsenic <2 ppm. 12
Dimethicone/vinyltrimethylsiloxysilicate crosspolymer
Dimethicone/vinyltrimethylsiloxysilicate dimethicone crosspolymer (20% in cyclopentasiloxane) was reported to not contain any heavy metals, polycyclic aromatic hydrocarbons, organohalogen compounds, or nitrosamines. 7 Residuals from manufacturing included platinum (catalyst, <25 ppm) and cyclotetrasiloxane (maximum 0.1%).
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer (20% in isododecane) was reported to not contain any heavy metals, polycyclic aromatic hydrocarbons, organohalogen compounds, or nitrosamines. 10 Residuals from manufacturing included platinum (catalyst, <25 ppm) and cyclotetrasiloxane (maximum < 1%).
Product mixtures
A manufacturer’s product sheet reported that product mixtures containing dimethicone/vinyl dimethicone crosspolymer (4%-30%), dimethicone/phenyl vinyl dimethicone crosspolymer (10%-20%), vinyl dimethicone/lauryl dimethicone crosspolymer (20%-35%), dimethicone/PEG-10/15 crosspolymer (15%-30%), PEG-15/lauryl dimethicone crosspolymer (15%-35%), or dimethicone/polyglycerin-3 crosspolymer (20%-35%) had <20 ppm heavy metal and <2 ppm arsenic. 9
Use
Cosmetic
Data on the usage of ingredients are provided by the manufacturers to the Food and Drug Administration’s Voluntary Cosmetic Registration Program (VCRP) and a survey conducted by the Personal Care Products Council (Council) collected use concentrations for ingredients in this group (Table 4). 13,14 Dimethicone/vinyl dimethicone crosspolymer and dimethicone crosspolymer have the greatest number of uses at 457 and 442, respectively.
Abbreviations: NR, not reported; totals, rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
The VCRP and Council data were available for: Behenyl dimethicone/bis-vinyldimethicone crosspolymer was used in 6 leave-on products at concentrations up to 10% (eye liners at 2%-10%, lipstick 0005%-2%, and foundation 0.001%). C30-45 alkyl cetearyl dimethicone crosspolymer was reported to be used in 25 leave-on products (up to 4%; including 5 eye products) and 2 rinse-off products. C4-24 alkyl dimethicone/divinyldimethicone crosspolymer was reported to be used in 1 leave-on product (a moisturizer) and in foundations up to 2%. Cetearyl dimethicone crosspolymer was reported to be used in 20 leave-on products (0.002%-23%) in 1 rinse-off product (0.2%) and in products diluted for bath use (0.002%). Dimethicone/bis-isobutyl PPG-20 crosspolymer was reported to be used in 12 leave-on products (0.1%-2%; 1 lipstick). Dimethicone crosspolymer was reported to be used in 430 leave-on products (0.02%-25%; including 40 eye products, 9 lipsticks, and 11 deodorants; body paint sprays up to 0.3%) and in 12 rinse-off products (0.007%-5%). Dimethicone crosspolymer 3 was reported to be used in 52 leave-on products (0.02%-2%; including 13 eye products) and in rinse-off products (0.2%). Dimethicone/divinyldimethicone/silsesquioxane crosspolymer was reported to be used in 14 leave-on products (0.5%-5%). Dimethicone/PEG-10/15 crosspolymer was reported to be used in 51 leave-on products (0.03%-3%) and in a hair conditioner (0.8%). Dimethicone/phenyl vinyl dimethicone crosspolymer was reported to be used in 10 leave-on products (0.8%-2%). Dimethicone/vinyl dimethicone crosspolymer was reported to be used in 444 leave-on products (0.003%-46%; including 1 baby product, 59 eye products, 9 lipsticks, and 47 products that may be inhaled) and 13 rinse-off products (0.06%-37%). Dimethicone/vinyltrimethylsiloxysilicate crosspolymer was reported to be used in 14 leave-on products (0.04%-6%; including eye products). Diphenyl dimethicone/vinyl diphenyl dimethicone/silsesquioxane crosspolymer was reported to be used in 13 leave-on products (0.1%-7%; up to 7% in face powders). Divinyldimethicone/dimethicone crosspolymer was reported to be used in 4 leave-on products (0.007%) and up to 0.7% in rinse-off products. Lauryl dimethicone/polyglycerin 3 crosspolymer was reported to be used in 3 rinse-off products (2%). PEG-10 dimethicone crosspolymer was reported to be used in 15 leave-on products (0.6%-2%). PEG-12 dimethicone crosspolymer was reported to be used in 28 leave-on products (0.3%-2%; 17 deodorants) and 3 rinse-off products (0.3%). PEG-15/lauryl dimethicone crosspolymer was reported to be used in 4 leave-on products (up to 2%) and 3 rinse-off products. Silicone quaternium-16/glycidoxy dimethicone crosspolymer was reported to be used in 2 leave-on products (0.003%) and 4 rinse-off products (1%-3%). Vinyl dimethicone/lauryl dimethicone crosspolymer was reported to be used in 3 leave-on products (0.3%-2% including lipstick) and in rinse-off products up to 0.09%. Vinyl dimethicone/methicone silsesquioxane crosspolymer was reported to be used in 104 leave-on products (0.1%-20%; mostly in make-up products) and 1 rinse-off product (0.5%-0.6%).
VCRP
15
data were only available for: C30-45 alkyl dimethicone/polycyclohexene oxide crosspolymer was reported to be used in 2 dermal products. Dimethicone/polyglycerin-3 crosspolymer was reported to be used in 7 leave-on products. Isopropyl titanium triisostearate/triethoxysilylethyl polydimethylsiloxyethyl dimethicone crosspolymer was reported to be used in 5 leave-on products. PEG-10 dimethicone/vinyl dimethicone crosspolymer was reported to be used in 7 leave-on products. Styrene/acrylates/dimethicone acrylate crosspolymer was reported to be used in 1 nail product.
Council
16
data were only available for: Cetyl dimethicone/vinyldimethicone crosspolymer was reported to be used in leave-on and rinse-off products up to 0.005% including eye shadow, bath soap and detergents, and shaving cream. Dimethicone/PEG-10 crosspolymer was reported to be used in leave-on products (0.5%; foundations). Dimethicone/PPG-20 crosspolymer was reported to be used in skin fresheners (0.2%). PEG-10/lauryl dimethicone crosspolymer was reported to be used in leave-on products (0.5%-0.7%) and rinse-off products (0.6%). Perfluorononyl dimethicone/methicone/amodimethicone crosspolymer was reported to be used in lipstick (0.7%).
There were no reported uses in either the VCRP or the Council survey for: acrylates/bis-hydroxypropyl dimethicone crosspolymer; bis-phenylisopropyl phenylisopropyl dimethicone/vinyl dimethicone crosspolymer; bis-vinyldimethicone/bis-isobutyl PPG-20 crosspolymer; bis-vinyldimethicone crosspolymer; bis-vinyldimethicone/PEG-10 dimethicone crosspolymer; bis-vinyldimethicone/PPG-20 crosspolymer; butyldimethicone methacrylate/methyl methacrylate crosspolymer; cetearyl dimethicone/vinyl dimethicone crosspolymer; cetyl hexacosyl dimethicone/bis-vinyldimethicone crosspolymer; crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis- vinyldimethicone crosspolymer; dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer; dimethicone/lauryl dimethicone/bis-vinyldimethicone crosspolymer; dimethicone/PEG-15 crosspolymer; dimethicone/titanate crosspolymer; diphenyl dimethicone crosspolymer; hydroxypropyl dimethicone/polysorbate 20 crosspolymer; lauryl dimethicone PEG-15 crosspolymer; lauryl polydimethylsiloxyethyl dimethicone/bis- vinyldimethicone crosspolymer; PEG-8 dimethicone/polysorbate 20 crosspolymer; PEG-12 dimethicone/bis-isobutyl PPG-20 crosspolymer; PEG-12 dimethicone/PPG-20 crosspolymer; polydimethylsiloxyethyl dimethicone/bis- vinyldimethicone crosspolymer; PEG-15/lauryl polydimethylsiloxyethyl dimethicone; polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer; trifluoropropyl dimethicone/PEG-10 crosspolymer; trifluoropropyl dimethicone/trifluoropropyl divinyldimethicone crosspolymer; trifluoropropyl dimethicone/vinyl trifluoropropyl dimethicone/silsesquioxane crosspolymer; trimethylsiloxysilicate/dimethicone crosspolymer; vinyl dimethicone/lauryl/behenyl dimethicone crosspolymer; vinyldimethyl/trimethylsiloxysilicate/dimethicone crosspolymer; vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer.
Dimethicone crosspolymer and dimethicone/vinyl dimethicone crosspolymer are used in cosmetic products that may be sprays, including hair and body paint products, and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm compared with pump sprays. 17,18 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal region and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 19,20 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 21 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays compared to other cosmetic sprays. None of the deodorants containing these ingredients were reported to be sprays.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
No published toxicokinetics data were discovered and no unpublished data were submitted.
Toxicological Studies
Acute Toxicity
Dermal—nonhuman
Dimethicone crosspolymer
Dimethicone crosspolymer (12% in cyclomethicone) is reported to have a dermal median lethal dose (LD50) of >2000 mg/kg in rabbits (n = 5/sex).5 There were no deaths or clinical signs.
Oral—nonhuman
Dimethicone crosspolymer
The oral LD50 of dimethicone crosspolymer (12% in cyclomethicone) was reported to be >2000 mg/kg for rats (n = 5/sex).5 There were no deaths or clinical signs of toxicity.
Inhalation—nonhuman
Crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer
The acute inhalation lethal concentration, 50% (LC50) of crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer (10% in ethanol/water, 4 hours) for rats was >5.29 mg/L.4
In vitro
Dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer
In an agar diffusion cytotoxicity test, dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer (concentration not provided, 100% assumed) was not cytotoxic to mammalian cell cultures (type of cell not provided). 22
Repeated Dose Toxicity
No published repeated dose dermal or inhalation toxicity studies were discovered and no unpublished data were submitted.
Oral—nonhuman
Dimethicone/bis-isobutyl PPG-20 crosspolymer
Dimethicone/bis-isobutyl PPG-20 crosspolymer (0, 100, 300, and 1000 mg/kg/d) was orally administered to Crl: CD(SD) rats (n = 5/sex) for 14 consecutive days. All rats survived. There were no effects on body weight or food consumption. Macroscopic findings at necropsy were unremarkable. The mean absolute liver and relative liver weights in all test article-treated female groups was increased in a dose-dependent manner. However, only the high-dose group values were statistically significant. The authors concluded that oral administration of dimethicone/bis-isobutyl PPG-20 crosspolymer to rats for 14 consecutive days was well tolerated at all doses. 23
Reproductive and Developmental Toxicity
No published reproductive or developmental toxicity studies were discovered and no unpublished data were submitted.
Genotoxicity
In Vitro
Dimethicone/bis-isobutyl PPG-20 crosspolymer
Dimethicone/bis-isobutyl PPG-20 crosspolymer (0-5000 μg/plate) was not mutagenic to Salmonella typhimurium (strains TA98, TA100, TA1535, and TA1537) and Escherichia coli (WP2uvrA [pKM101] and WP2 [pKM101]) with or without metabolic activation. 23
Dimethicone crosspolymer
Dimethicone crosspolymer (0-1000 μg/plate; dissolved in tetrahydofuran) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (WP2uvrA [pKM101] and WP2 [pKM101]) with or without metabolic activation. 5
Dimethicone/PEG-10/15 crosspolymer
A product mixture containing dimethicone/PEG-10/15 crosspolymer (∼24%) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 24
In a chromosomal aberration assay using cultured mammalian cells (CHL/IU), a product mixture containing dimethicone/PEG-10/15 crosspolymer (∼24%; 1250, 2500, and 5000 μg/mL) did not produce any chromosomal aberrations. 24
Dimethicone/phenyl vinyl dimethicone crosspolymer
Dimethicone/phenyl vinyl dimethicone crosspolymer (∼16%) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 25
Dimethicone/polyglycerin-3 crosspolymer
A product containing dimethicone/polyglycerin-3 crosspolymer (∼40% in dimethicone; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 26
Dimethicone/vinyl dimethicone crosspolymer
A product containing dimethicone/vinyl dimethicone crosspolymer (∼24% in dimethicone; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 27
Diphenyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer
Dipheynyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer (100%; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 28
Lauryl dimethicone/polyglycerin-3 crosspolymer
Lauryl dimethicone/polyglycerin-3 crosspolymer (40% in mineral oil; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 29
Lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer
Lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer (100%; 312.5 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E. coli (strain WP3uvrA) with or without metabolic activation. 30
PEG-15 lauryl dimethicone crosspolymer
PEG-15 lauryl dimethicone crosspolymer (100%; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 31
PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer
PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (100%; 5000 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 32
Polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer
Polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (100%; 2500 μg/plate) was not mutagenic to S typhimurium (strains TA98, TA100, TA1535, and TA1537) and E coli (strain WP3uvrA) with or without metabolic activation. 33
Carcinogenicity
No published carcinogenicity studies were discovered and no unpublished data were submitted.
Irritation and Sensitization
Irritation
Dermal—nonhuman
Dimethicone crosspolymer
Dimethicone crosspolymer (100%; 0.5 mL) was not dermally irritating when administered to female New Zealand White rabbits (n = 3) under semiocclusion for 4 hours. 5
Dimethicone/PEG-10/15 crosspolymer
A product mixture containing dimethicone/PEG-10/15 crosspolymer (∼24%; 0.5 mL) had a Producer Price Index (PPI) of 1.20 when administered under occlusion to the intact and abraded skin of New Zealand White rabbits (n = 3). 24 The authors concluded that the test substance was nonirritating.
Dimethicone/phenyl vinyl dimethicone crosspolymer
A product mixture containing dimethicone/vinyl dimethicone crosspolymer (∼16% in diphenylsiloxy phenyl trimethicone; 0.5 mL) had a PPI of 2.38 when administered under occlusion to the intact and abraded skin of New Zealand White rabbits (n = 3). 25 The authors concluded that the test substance was a moderate irritant.
Dimethicone/polyglycerin-3 crosspolymer
A product containing dimethicone/polyglycerin-3 crosspolymer (∼40% in dimethicone; 0.5 mL) had a PPI of 1.30 when administered under occlusion to the intact and abraded skin of New Zealand White rabbits (n = 6). 26 The authors concluded that the test substance was nonirritating.
Dimethicone/vinyl dimethicone crosspolymer
A product containing dimethicone/vinyl dimethicone crosspolymer (∼24% in dimethicone) had a PPI of 1.42 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 27 The authors concluded that the test article was a mild irritant.
Diphenyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer
Diphenyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer (100%) had a PPI of 0.10 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 28 The authors concluded that the test article was nonirritating.
Lauryl dimethicone/polyglycerin-3 crosspolymer
Lauryl dimethicone/polyglycerini-3 crosspolymer (40% in triethylhexanoin; 0.5 mL) had a PPI of 1.50 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 28 The authors concluded that the test article was not a primary irritant.
Lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer
Lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer (100%; 0.5 g) had a PPI of 0.98 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 30 The authors concluded that the test article was not a primary irritant.
PEG-10/lauryl diemthicone crosspolymer and PEG-15 lauryl dimethicone crosspolymer
A mixture of PEG-10/lauryl dimethicone crosspolymer and PEG-15 lauryl dimethicone crosspolymer (100%; 50/50 mix assumed) had a PPI of 0.25 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 34 The authors concluded that the test article was not a primary irritant.
PEG-15 lauryl dimethicone crosspolymer
PEG-15 lauryl dimethicone crosspolymer (100%) had a PPI of 0.10 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 31 The authors concluded that the test article was not a primary irritant.
PEG-15/lauryl polydimethylsiloxyethyl dimethicone
PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (100%; 0.5 g) had a PPI of 1.05 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 32 The authors concluded that the test article was not a primary irritant.
Polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer
Polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (100%; 0.5 g) had a PPI of 0.33 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 33 The authors concluded that the test article was not a primary irritant.
Vinyl dimethicone/lauryl dimethicone crosspolymer
Vinyl dimethicone/lauryl dimethicone crosspolymer (100%; 0.5 g) had a PPI of 0.33 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 35 The authors concluded that the test article was not a primary irritant.
Vinyl dimethicone/methicone silsesquioxane crosspolymer
Vinyl dimethicone/lauryl dimethicone crosspolymer (100%; 0.5 mL) had a PPI of 0.25 when administered under occlusion to intact and abraded skin of New Zealand White rabbits (n = 6). 36 The authors concluded that the test article was not a primary irritant.
Dermal—human
Dimethicone/bis-isobutyl PPG-20 crosspolymer
Dimethicone/bis-isobutyl PPG-20 crosspolymer (10%, 40%, 70%, and 100% in isodecyl neopentanoate [IDNP]) was not irritating in an irritation test (n = 28). Erythema was observed in 0 to 6 patients at evaluation on days 1, 3, and 5. 23
Dimethicone crosspolymer
In a cumulative irritation test (n = 27), dimethicone crosspolymer (100%; 0.2 mL) was applied 10 times (with the patches remaining over the weekend) over 2 weeks. The authors concluded that dimethicone crosspolymer was dermally nonirritating to humans. 23
Ocular
Dimethicone crosspolymer
Dimethicone crosspolymer (12% in cyclomethicone; 0.1 mL) was not an ocular irritant to male New Zealand White rabbits (n = 3). 5 There were no effects to the iris or corneal observed.
Dimethicone crosspolymer (100%; 0.1 mL) produced a mild, transient ocular irritant to male New Zealand White rabbits (n = 3). 23
Dimethicone/phenyl vinyl dimethicone crosspolymer
In a Skin ZK-1200 (tissue equivalent) ocular assay, dimethicone/phenyl vinyl dimethicone crosspolymer (25 μL) was not predicted to be an ocular irritant after 30 minutes of exposure. 37
Sensitization
Dermal—nonhuman
Dimethicone crosspolymer
Dimethicone crosspolymer (12% in cyclomethicone) was not sensitizing to the clipped backs of Hartley guinea pigs (n = 10/sex). 5 There was no difference between the treatment and control groups.
Dimethicone crosspolymer (100%; 0.3 mL) was not sensitizing to guinea pigs (n = 10/sex). There was no dermal responses in the treatment group. 23
Dimethicone/PEG-10/15 crosspolymer
A product mixture containing dimethicone/PEG-10/15 crosspolymer (∼24%) was not sensitizing when administered by intradermal injection to albino Hartley-strain guinea pigs (n = 5). 24
Dimethicone/polyglycerin-3 crosspolymer
A product containing dimethicone/polyglycerin-3 crosspolymer (∼40% in dimethicone; 0.4 g) was not sensitizing to guinea pigs (n = 6/sex). 26
Dimethicone/phenyl vinyl dimethicone crosspolymer
In a dermal sensitization assay using albino Hartley-derived guinea pigs (n = 5/sex), a product containing dimethicone/phenyl vinyl dimethicone crosspolymers (∼16%) was not sensitizing. 25
Dimethicone/vinyl dimethicone crosspolymer
In a dermal sensitization assay using albino Hartley-derived guinea pigs (n = 5/sex), a product containing dimethicone/vinyl dimethicone crosspolymers (∼24%) was not sensitizing. 27
Diphenyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer
In a dermal sensitization assay using albino Hartley-derived guinea pigs (n = 6/sex), diphenyl dimethicone/vinyl diphenyl dimethicone silsesquioxane crosspolymer (100%) was not sensitizing. 28
Lauryl dimethicone/polyglycerin-3 crosspolymer
In a dermal sensitization assay using albino Hartley-derived guinea pigs (n = 6/sex), lauryl dimethicone/polyglycerin-3 crosspolymer (40% in mineral oil) was not sensitizing. 29
Lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer
In a local lymph node assay, lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer (1.5%, 3%, and 7.5%, w/v) was not a sensitizer. 30
PEG-15/laurel polydimethylsiloxyethy dimethicone crosspolymer
In a local lymph node assay using mice, PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (1.5%, 3%, and 7.5%, w/v) was not a sensitizer. 32
Polyglyceryl-3/lauryl polydimethylsiloxyethy dimethicone crosspolymer
In a local lymph node assay using mice, polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer (1.5%, 3%, and 7.5%, w/v) was not a sensitizer. 33
Vinyl dimethicone/methicone silsesquioxane crosspolymer
In a dermal sensitization assay using albino Hartley-derived guinea pigs (n = 10), lauryl dimethicone/polyglycerin-3 crosspolymer (50% in vaseline; 0.1 g) was not sensitizing. 36
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer
Vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer (20% in isododecane) was not sensitizing to guinea pigs. 10
Dermal—human
Dimethicone/bis-isobutyl PPG-20 crosspolymer
In a human repeated insult patch test (HRIPT; n = 100), dimethicone/bis-isobutyl PPG-20 crosspolymer (70% in IDNP) was not sensitizing. There were no reactions during the challenge phase. 23
Dimethicone crosspolymer
In an HRIPT (n = 101) of dimethicone crosspolymer (100%; 0.2 g), there were no adverse reactions of any kind during the course of this study. Dimethicone crosspolymer was not sensitizing. 23
Dimethicone/divinyldimethicone/silsesquioxane crosspolymer
In an HRIPT (n = 55) of dimethicone/divinyldimethicone/silsesquioxane crosspolymer (30% in corn oil), there were no adverse reactions of any kind during the course of this study. 38
Dimethicone/vinyl dimethicone crosspolymer
In 2 modified HRIPTs (n = 107), a facial lotion containing dimethicone/vinyl dimethicone crosspolymer (1%) was not sensitizing. 39
Summary
Dimethicone crosspolymers function in cosmetics as absorbents, bulking agents, film formers, hair-conditioning agents, skin-conditioning agents—emollient, slip modifiers, surface modifiers, and viscosity-increasing agents—nonaqueous. The 62 dimethicone crosspolymer ingredients in this report are silicone elastomers comprising dimethicone copolymers that are cross-linked with a bifunctional agent.
These crosspolymer ingredients are typically supplied as swollen gels that contain various oils (eg, silicone oils such as dimethicone). They are not soluble in water. Dimethicone/divinyldimethicone/silsesquioxane crosspolymer has a spherical shape with a particle diameter ranging from 2 to 10 μm.
Heavy metals, polycyclic aromatic hydrocarbons, organohalogen compounds, or nitrosamines were not detected in several dimethicone crosspolymers. Residuals from manufacturing of crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer included low levels of platinum tert-butanol, iso-dodecane, vinyl acetate, vinyl tert-decanoate, crotonic acid, isopropanol, and ethyl acetate.
Dimethicone/vinyl dimethicone crosspolymer and dimethicone crosspolymer have the greatest number of uses at 457 and 442, respectively.
Reported use and use concentration data were available for: behenyl dimethicone/bis-vinyldimethicone crosspolymer; C30-45 alkyl cetearyl dimethicone crosspolymer; C4-24 alkyl dimethicone/divinyldimethicone crosspolymer; cetearyl dimethicone crosspolymer; dimethicone/bis-isobutyl PPG-20 crosspolymer; dimethicone crosspolymer; dimethicone crosspolymer-3; dimethicone/divinyldimethicone/silsesquioxane crosspolymer; dimethicone/PEG-10/15 crosspolymer; dimethicone/phenyl vinyl dimethicone crosspolymer; dimethicone/vinyl dimethicone crosspolymer; dimethicone/vinyltrimethylsiloxysilicate crosspolymer; diphenyl dimethicone/vinyl diphenyl dimethicone/silsesquioxane crosspolymer; divinyldimethicone/dimethicone crosspolymer; lauryl dimethicone/polyglycerin-3 crosspolymer; PEG-10 dimethicone crosspolymer; PEG-12 dimethicone crosspolymer; PEG-15/lauryl dimethicone crosspolymer; silicone quaternium-16/glycidoxy dimethicone crosspolymer; vinyl dimethicone/lauryl dimethicone crosspolymer; vinyl dimethicone/methicone silsesquioxane crosspolymer.
Reported uses, but not use concentration data, were available for: C30-45 alkyl dimethicone/polycyclohexene oxide crosspolymer; Dimethicone/polyglycerin-3 crosspolymer; isopropyl titanium triisostearate/triethoxysilylethyl polydimethylsiloxyethyl dimethicone crosspolymer; PEG-10 dimethicone/vinyl dimethicone crosspolymer was reported to be used in 7 leave-on products; styrene/acrylates/dimethicone acrylate crosspolymer.
Use concentration data were only available for: cetyl dimethicone/bis-vinyldimethicone crosspolymer; dimethicone/PEG-10 crosspolymer; dimethicone/PPG-20 crosspolymer; PEG-10/lauryl dimethicone crosspolymer; perfluorononyl dimethicone/methicone/amodimethicone crosspolymer.
There were no reported uses or use concentrations for: acrylates/bis-hydroxypropyl dimethicone crosspolymer; bis-phenylisopropyl phenylisopropyl dimethicone/vinyl dimethicone crosspolymer; bis-vinyldimethicone/bis-isobutyl PPG-20 crosspolymer; bis-vinyldimethicone crosspolymer; bis-vinyldimethicone/PEG-10 dimethicone crosspolymer; bis-vinyldimethicone/PPG-20 crosspolymer; butyldimethicone methacrylate/methyl methacrylate crosspolymer; cetyl dimethicone/bis-vinyldimethicone crosspolymer; cetyl hexacosyl dimethicone/bis-vinyldimethicone crosspolymer; crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer; dimethicone/divinyldimethicone/silsesquioxane crosspolymer; dimethicone/lauryl dimethicone/bis-vinyldimethicone crosspolymer; dimethicone/PEG-15 crosspolymer; dimethicone/titanate crosspolymer; diphenyl dimethicone crosspolymer; hydroxypropyl dimethicone/polysorbate 20 crosspolymer; lauryl dimethicone PEG-15 crosspolymer; lauryl dimethicone/polyglycerin-3 crosspolymer; lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer; PEG-8 dimethicone/polysorbate 20 crosspolymer; PEG-12 dimethicone/bis-isobutyl PPG-20 crosspolymer; PEG-12 dimethicone/PPG-20 crosspolymer; PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer; polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer; polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer; trifluoropropyl dimethicone/PEG-10 crosspolymer; trifluoropropyl dimethicone/trifluoropropyl divinyldimethicone crosspolymer; trifluoropropyl dimethicone/vinyl trifluoropropyl dimethicone/silsesquioxane crosspolymer; trimethylsiloxysilicate/dimethicone crosspolymer; vinyl dimethicone/lauryl/behenyl dimethicone crosspolymer; vinyldimethyl/trimethylsiloxysilicate/dimethicone crosspolymer; vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer.
Dimethicone crosspolymer had a dermal LD50 of >2000 mg/kg in rabbits. The oral LD50 of dimethicone crosspolymer was >2000 mg/kg for rats. The acute inhalation LC50 of crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer at 10% over 4 hours for rats was >5.29 mg/L.
Dimethicone/bis-isobutyl PPG-20 crosspolymer at 1000 mg/kg/d was not toxic when orally administered to rats for 14 days.
In an agar diffusion cytotoxicity test, dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer was not cytotoxic to mammal cell cultures.
Dimethicone crosspolymer and several other ingredients were not mutagenic to S typhimurium and E coli with or without metabolic activation up to 312.5 to 5000 μg/plate.
Several of the dimethicone crosspolymers were not dermally irritating when administered to rabbits up to 100%. However, dimethicone/vinyl dimethicone crosspolymer at ∼16% and dimethicone/vinyl dimethicone crosspolymer at ∼24% were mild irritants to rabbits.
Dimethicone crosspolymer was not an ocular irritant to rabbits at 100%. In a Skin ZK-1200 ocular assay, dimethicone/phenyl vinyl dimethicone crosspolymer was not predicted to be an ocular irritant. Dimethicone/vinyltrimethylsiloxysilicate crosspolymer was not an ocular irritant to rabbits.
Dimethicone crosspolymer was not sensitizing to guinea pigs at 100%.
Several dimethicone crosspolymer were not sensitizing to guinea pigs up to 12% to 100%.
A product containing dimethicone/vinyl dimethicone crosspolymer at 1% was not sensitizing in an HRIPT. In an HRIPT of dimethicone/divinyldimethicone/silsesquioxane crosspolymer at 30%, there were no adverse reactions of any kind during the course of this study.
No published studies regarding toxicokinetics, repeated dose toxicity, reproductive or developmental toxicity, or carcinogenicity were discovered and no unpublished data for these end points were provided.
Discussion
The Panel determined that the available data on acute toxicity, genotoxicity, irritation, and sensitization were adequate for assessing the safety of these ingredients.
There was a lack of toxicokinetics, repeated dose toxicity, carcinogenicity, and reproductive/developmental toxicology data for the dimethicone crosspolymers in this safety assessment. The Panel was not concerned about these gaps in information because these ingredients are large polymers that will not penetrate the skin. Also, the silicone backbone is stable under anticipated conditions of use and these ingredients do not contain monomers above the levels of toxicological concern. There are multiple animal irritation and sensitization studies as well as 2 HRIPT studies that were negative for effects. Ames tests were negative for 3 of these ingredients.
The Panel did express concern over the absence of information on the levels of residual monomers and catalysts. The monomers of concern include: α-Methylstyrene; bis-vinyldimethicone; methyl methacrylate; butyldimethylsilylmethacrylate; vinyl cyclohexene oxide; vinyl acetate; α,ω-Divinyl alkenes (C4-20); bis-vinyl phenylmethyldimethicone; allyl alcohol; isobutanol; titanium species used in cross-linking [(0-IPr)4 or (0-iPr)2Cl2]; unidentified diamine cross-linking agent in silicone quaternium-16/glycidoxy dimethicone crosspolymer; styrene; divinylbenzene; allyl polyglyceryl-3; bisvinyl trifluoroproyl methicone (if n ∼ ≤8).
In those cases where data were available, the monomer levels were low (eg, vinyl acetate < 100 ppm in crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer) or below the limits of detection. In those cases where data were not available, the Panel determined that monomer levels would be very low or undetectable because any residual monomers/catalyst are likely entrapped in the silicone backbone of these crosspolymers. Any monomers not so trapped are likely to disappear quickly because of their high volatility. This would be true because in general, based on Panel members experience, these volatile monomers have a distinctive odor that would render crosspolymer ingredients problematic for use in cosmetics. For all of these reasons, the Panel determined that the current methods of manufacture are adequate to assure monomer levels are as low a reasonably achievable but urged ingredient suppliers to continue to take steps to ensure that residual monomers and catalysts remain below any level of toxicological concern and as low at reasonably achievable.
The Panel discussed the issue of incidental inhalation exposure from face powders, foot powders, and sprays, perfumes, and hair sprays. The data available from 1 acute inhalation exposure study indicated that the LC50 for crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone is greater than 5.29 mg/L. The Panel concluded that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. These ingredients are reportedly used at concentrations up to 20% in spray and up to 46% in powder cosmetic products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, these ingredients are not likely to cause direct toxic effect in the upper respiratory tract, based on the chemical and biological properties of the dimethicone crosspolymers. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for dimethicone crosspolymers to cause genotoxicity, irritation, and sensitization. They noted the lack of systemic toxicity in acute oral exposure studies, little or no irritation or sensitization in multiple tests of dermal and ocular exposure, and the absence of genotoxicity in multiple Ames tests. In addition, these ingredients are large macromolecules, insoluble in water, and chemically inert under physiological conditions or conditions of use, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. A detailed discussion of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment: acrylates/bis-hydroxypropyl dimethicone crosspolymer*; behenyl dimethicone/bis-vinyldimethicone crosspolymer; bis-phenylisopropyl phenylisopropyl dimethicone/vinyl dimethicone crosspolymer*; bis-vinyldimethicone/bis-isobutyl PPG-20 crosspolymer*; bis-vinyldimethicone crosspolymer*; bis-vinyldimethicone/PEG-10 dimethicone crosspolymer*; bis-vinyldimethicone/PPG-20 crosspolymer*; butyldimethicone methacrylate/methyl methacrylate crosspolymer*; C30-45 alkyl cetearyl dimethicone crosspolymer; C4-24 alkyl dimethicone/divinyldimethicone crosspolymer; C30-45 alkyl dimethicone/polycyclohexene oxide crosspolymer; cetearyl dimethicone crosspolymer; cetearyl dimethicone/vinyl dimethicone crosspolymer; cetyl dimethicone/bis-vinyldimethicone crosspolymer*; cetyl hexacosyl dimethicone/bis-vinyldimethicone crosspolymer*; crotonic acid/vinyl C8-12 isoalkyl esters/VA/bis-vinyldimethicone crosspolymer*; dimethicone/bis-isobutyl PPG-20 crosspolymer; dimethicone/bis-vinyldimethicone/silsesquioxane crosspolymer*; dimethicone crosspolymer; dimethicone crosspolymer-3; dimethicone/divinyldimethicone/silsesquioxane crosspolymer; dimethicone/lauryl dimethicone/bis-vinyldimethicone crosspolymer*; dimethicone/PEG-10 crosspolymer; dimethicone/PEG-10/15 crosspolymer; dimethicone/PEG-15 crosspolymer*; dimethicone/phenyl vinyl dimethicone crosspolymer; dimethicone/polyglycerin-3 crosspolymer; dimethicone/PPG-20 crosspolymer; dimethicone/titanate crosspolymer*; dimethicone/vinyl dimethicone crosspolymer; dimethicone/vinyltrimethylsiloxysilicate crosspolymer; diphenyl dimethicone crosspolymer*; diphenyl dimethicone/vinyl diphenyl dimethicone/silsesquioxane crosspolymer; divinyldimethicone/dimethicone crosspolymer; hydroxypropyl dimethicone/polysorbate 20 crosspolymer*; isopropyl titanium triisostearate/triethoxysilylethyl polydimethylsiloxyethyl dimethicone crosspolymer; lauryl dimethicone PEG-15 crosspolymer*; lauryl dimethicone/polyglycerin-3 crosspolymer*; lauryl polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer*; PEG-10 dimethicone crosspolymer; PEG-12 dimethicone crosspolymer; PEG-8 dimethicone/polysorbate 20 crosspolymer*; PEG-12 dimethicone/bis-isobutyl PPG-20 crosspolymer*; PEG-12 dimethicone/PPG-20 crosspolymer*; PEG-10 dimethicone/vinyl dimethicone crosspolymer; PEG-10/lauryl dimethicone crosspolymer; PEG-15/lauryl dimethicone crosspolymer; PEG-15/lauryl polydimethylsiloxyethyl dimethicone crosspolymer*; perfluorononyl dimethicone/methicone/amodimethicone crosspolymer; polydimethylsiloxyethyl dimethicone/bis-vinyldimethicone crosspolymer*; polyglyceryl-3/lauryl polydimethylsiloxyethyl dimethicone crosspolymer*; silicone quaternium-16/glycidoxy dimethicone crosspolymer; styrene/acrylates/dimethicone acrylate crosspolymer; trifluoropropyl dimethicone/PEG-10 crosspolymer*; trifluoropropyl dimethicone/trifluoropropyl divinyldimethicone crosspolymer*; trifluoropropyl dimethicone/vinyl trifluoropropyl dimethicone/silsesquioxane crosspolymer*; trimethylsiloxysilicate/dimethicone crosspolymer*; vinyl dimethicone/lauryl/behenyl dimethicone crosspolymer*; vinyl dimethicone/lauryl dimethicone crosspolymer; vinyl dimethicone/methicone silsesquioxane crosspolymer; vinyldimethyl/trimethylsiloxysilicate/dimethicone crosspolymer*; vinyldimethyl/trimethylsiloxysilicate stearyl dimethicone crosspolymer*.
*Not reported in use. If ingredients in this group to be in current use are to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L. St, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.