Abstract
Polycyclic aromatic hydrocarbons, such as 7,12-dimethylbenz(a)anthracene (DMBA), are environmental pollutants that exert multiple toxic and carcinogenic effects. Studies showed that these effects are mediated by activation of the aryl hydrocarbon receptor (AhR) and modulated by allelic variants of Ahr gene. Here, we investigated the effects of DMBA treatment in the inflammatory response and bone marrow (BM) hematopoietic function of maximal acute inflammatory response (AIRmax) and minimal acute inflammatory response (AIRmin) heterogeneous mouse lines selected for high and low acute inflammatory responsiveness, respectively. The phenotypic selection resulted in the segregation of the Ahrd and Ahrb1 alleles that confer low and high receptor ligand-binding affinity, respectively, in AIRmax and AIRmin mice. We observed a reduction in BM mature granulocyte population in AIRmin mice 24 hours after DMBA treatment while both blast and immature myeloid cells were increased. Proliferation and differentiation of BM myeloid cells in response to in vitro granulocyte-macrophage colony-stimulating factor stimulus were impaired in AIRmin-treated mice. These DMBA effects on myeloid BM cells (BMCs) affected the in vivo leukocyte migration to an inflammatory site induced by polyacrylamide beads (Biogel P-100, Bio-Rad, France) injection in AIRmin mice. On the other hand, these alterations were not observed in DMBA-treated AIRmax mice. These data indicate that DMBA affects myeloid cell differentiation and inflammatory response and Ahrb1 allele in the genetic background of AIRmin mice contributes to this effect.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous environmental contaminants that are produced during incomplete combustion of organic materials. Polycyclic aromatic hydrocarbons are present in cigarette smoke, grilled foods, automobile exhaust, and wood smoke. 1 –4 Most of the members of the PAH family are potent carcinogens and immunotoxicants for humans and other species. 5,6
Most PAHs such as dimethylbenz(a)anthracene (DMBA) require activation by cytochrome P450 (CYP) monooxygenases for the production of epoxy intermediates that are converted with the aid of epoxide hydrolases to diol-epoxides (DMBA-dihydrodiol-epoxide). This last metabolite is highly toxic and mutagenic and may exert deleterious biological effects such as immunosuppression, myelotoxicity, and oncogene activation. 7 –11
The induction of CYP transcription by PAHs is tissue specific and largely depends on aryl hydrocarbon receptor (AhR) activation by environmental and endogenous substances, such as indole-containing compounds, tryptophan photoproducts, bilirubin and biliverdin, and possibly arachidonic acid metabolites. 12 –14 Aryl hydrocarbon receptor mediates a broad spectrum of biological responses and modulates immune and inflammatory reactions. 15,16 Aryl hydrocarbon receptor activation effects are variable since it can provide protection against noxious agents or contribute to the pathogenesis of tissue damage. These responses are influenced by functional polymorphisms found in Ahr or in downstream Ahr-regulated enzyme genes. 11,17 In humans, studies in diverse ethnic groups point to the association between genetic variations in AHR and CYP1A1 and phenotypic differences in response to xenobiotics, ultimately leading to cancer. 18,19 In the mouse, the variations are clearly associated with the Ahr alleles coding for low (Ahrd ) and high (Ahrb1 ) affinity receptors, respectively, although the allele effects can be influenced by the genetic background of the isogenic mouse lines under study. The differential affinity of the receptors coded by each allele is due to the substitution of a valine for an alanine (A375V) residue in the AhR ligand-binding domain of the Ahrd allele. 20
An original experimental model to investigate the relationship among AhR genetic variants, inflammatory response, and cancer is provided by 2 outbred lines of mice phenotypically selected for maximal acute inflammatory response (AIRmax) or minimal acute inflammatory response (AIRmin) to a nonimmunogenic substance (Biogel P-100, Bio-Rad, France). 21 At the end of the bidirectional selective process, after about 20 generations of selective breeding, AIRmax and AIRmin mouse lines presented extreme “maximal” and “minimum” phenotypes, respectively, and, at that limit, were considered homozygous for the quantitative trait loci (QTL) that control the acute inflammatory reaction while maintaining an heterogeneous genetic background. 21,22 These lines are also strikingly divergent in resistance/susceptibility to chemically induced carcinogenesis. 23,24
It was later identified that the selective process for the derivation of the AIRmax and AIRmin mouse lines resulted in the segregation of Ahrd and Ahrb1 alleles, respectively, in each line. The Ahrd AIRmax mice are resistant while Ahrb1 AIRmin mice are susceptible to developing a contact hypersensitivity reaction and skin and lung tumors following DMBA treatment, implicating Ahr in the differential tumor susceptibility and in inflammatory response control in AIRmax and AIRmin mice. 24
The objective of the present study was to investigate the influence of Ahr alleles on the toxic effects of DMBA in the bone marrow (BM) and in the inflammatory response, in the context of AIRmax and AIRmin heterogeneous genetic backgrounds. This was studied by comparing, in the 2 mouse lines, the in vivo effect of the drug on Biogel-induced acute inflammation as well as on the structure, cell composition, and hematopoietic activity of the BM, a central organ for the production of inflammatory cells.
Materials and Methods
Reagents
7,12-Dimethylbenz(a)anthracene (DMBA; Sigma, Brazil) was dissolved in highly refined and low acidity olive oil (Sigma, CAT O1514) at a concentration of 5 mg/mL for intraperitoneal (ip) injection. RPMI 1640 medium (Gibco-BRL, Grand Island, New York) was supplemented with 10% fetal bovine serum (FBS), 2 mmol/L glutamine, 1 mmol/L sodium pyruvate, 20 µg/mL gentamicin, and 50 µmol/L 2-mercaptoethanol. Recombinant mouse granulocyte-macrophage colony-stimulating factor (GM-CSF) was purchased from Pepro Tech (PeproTech, Inc, Rocky Hill, New Jersey) and α-naphthoflavone (α-NF) was purchased from Sigma (Sigma Chemical Co, St Louis, Missouri).
Mice and Treatments
The AIRmax and AIRmin mice were obtained by bidirectional selective breeding for AIRmax and AIRmin, respectively, based on both cellular infiltrate and protein contents at the 24-hour-local inflammatory exudate produced by subcutaneous (sc) injection of polyacrylamide beads (Biogel P-100). 21,22 The selection experiment started from a foundation population constituted by the balanced intercross of the following 8 isogenic mouse lines: A/J, BALB/cL, CBA/J, C57BL/6J, DBA2/J, SJL/J, SWR/J, and P/J. After about 20 generations of selective breeding in the direction of the high or low responses, the 2 mouse lines are theoretically homozygous for the AIR-relevant genes on a heterogeneous genetic background. Mice were maintained under photoperiods of 12 hours at 21°C, standard mouse chow (Nuvital, Brazil), and water ad libitum in the animal facilities of the Immunogenetics Laboratory of the Butantan Institute. Although the formal stock designations are Ibut: AIRH and Ibut: AIRL, we refer to them in this article and in previous publications as AIRmax and AIRmin, respectively. Six to eight week-old males and females were injected with 50 mg/kg body weight (bw) DMBA ip. Control animals were injected with an equivalent volume of olive oil. This dose was chosen due to its maximal toxicity on BM cells (BMCs) from susceptible mice. 25 The AhR antagonist α-NF was injected ip (50mg/kg bw) twice daily 24 hours before and concomitantly to DMBA treatment. All animal experiments were approved by the Ethics Committee in Animal Experimentation of the Butantan Institute (Protocol no 253/06).
Histopathological Analysis
Sternums were removed and fixed in ice-cold 10% phosphate-buffered paraformaldehyde for 24 hours and decalcified for 7 days in 10% EDTA solution. After dehydration, samples were embedded in paraffin and serial 5-μm sections were performed using a microtome (Leica RM 2135, Heerbrugg, Switzerland). Sections were stained with hematoxylin and eosin (Merck, Darmstadt, Germany) for BM architecture analysis.
Bone Marrow Cell Isolation
Mice were euthanized with CO2 at different times after ip injection with DMBA or vehicle. The femurs were dissected free of muscle tissue and the femoral epiphyses were cut with a scalpel. Cells from both femurs were flushed from the bones with 2 mL of RPMI medium using a syringe equipped with a 25-gauge needle. The BMCs were homogenized by successive passages through a 22-gauge needle. Following centrifugation, red blood cells were lysed in ammonium-chloride-potassium buffer (150 mmol/L NH4Cl, 1.0 mmol/L KHCO3, and 100 mmol/L Na2EDTA, pH 7.3). Viable cells were enumerated in a hemocytometer chamber by trypan blue exclusion and identified by cytospin preparations. Cells were used for cell proliferation assays and flow cytometry.
Liquid BMC Culture
Bone marrow cells were isolated 24 hours after injection with DMBA, DMBA/α-NF, or vehicle and cultured in 24-well plates in complete RPMI-1640 medium containing 10% heat-inactivated fetal calf serum (FCS), 2 mmol/L glutamine, 20 µg/mL gentamycin, and 50 µmol/L 2-mercaptoethanol. Cells (5 × 105/mL) were cultured with 50 ng/mL GM-CSF for 5 days at 37°C, 5% CO2. The cells were then harvested, counted in hemocytometer chambers, and stained with Giemsa for morphological analysis.
Clonogenic Assay
Bone marrow cells were cultured at 2.5 × 104 in 35-mm Petri dishes in 1-mL aliquots of RPMI-1640 medium supplemented with 20% FCS, 2 mmol/L glutamine, 20 µg/mL gentamycin, 50 µmol/L 2-mercaptoethanol, and 50% methylcellulose (MethoCult3434, Stem Cell Technologies, Vancouver, Canada). Cultures were stimulated with 50 ng/mL (GM-CSF) and cell growth was evaluated after incubation (37°C, 5% CO2) for 5 days in a CO2 incubator. Colonies with at least 50 cells were enumerated and characterized according to cell morphology. A small aliquot (10 µL) was obtained from different colonies and examined microscopically for neutrophil and macrophage identification. The experiments were performed in duplicate with 6 animals/group.
Flow Cytometry
Bone marrow cell subtypes were phenotyped with antibodies to cell surface markers for myelocytes—Ly-6G/Ly-6C (GR-1, clone: RB6), CD11b (clone: Mi/70), and hematopoietic stem cells (HSCs)—c-kit (CD117, clone 2B8), Ly-6A/E (stem-cell antigen 1 [Sca-1], clone D7), and lineage (Lin; Mouse Lineage Panel; BD, San Diego, California). Freshly harvested cells from BM were plated at 1 × 106 cells/mL in 100 μL phosphate-buffered saline (PBS) wash buffer (Sigma Chemical Co, St Louis, Missouri, with 0.09% sodium azide and 1% heat inactivated FBS) and incubated for 10 minutes at 4°C in the dark with purified rat antimouse CD16/CD32 (Fcγ III/II receptor clone 2.4G2) monoclonal antibody. The cells were incubated with 20 μL of antibody cocktail containing GR-1 fluorescein isothiocyanate (FITC) + CD11b-phycoerythrin (PE) or c-kit-FITC + Sca-1-PE + APC Lineage Antibody Cocktail (Lin) for 30 minutes. Following incubation, cells were washed with 200 μL of PBS wash buffer. The pellet was suspended in 300 µL media and propidium iodide was added (2 mg/mL) to exclude dead cells from analysis. To analyze the intracellular AhR expression, cells were fixed and permeabilized with fixation and permeabilization buffers and then intracellularly stained with 1.0 µg antimouse AhR PE antibody (clone 4MEJJ; eBioscience, San Diego, California). Total viable BMCs were used for analysis and the data were expressed as the DMBA–oil median fluorescence intensity ratio. Samples were analyzed using a fluorescence-activated cell sorting (FACS) CantoII Flow Cytometer (BD Bioscience) and the data were collected from 50 000 single cell events using FlowJo software (Treestar Inc - Ashland,OR,USA).
Inflammation
Inflammation was induced as described previously. 21 Briefly, animals were shaved and 750 µL sterile 67% suspension (53 mg dry weight/mL) of Biogel P-100 (Bio-Rad) in PBS was injected sc in the back. After 24 hours, pouches were rinsed twice with 1 mL PBS + 20 U/mL heparin. Biogel beads were allowed to sediment, cells were then recovered by centrifugation, and the cell-free exudate was immediately frozen and kept at −20°C. Total cells in exudates were counted in a hemocytometer and cytospin preparations were stained with Wright-Giemsa. Cell subpopulations were differentially characterized by microscopy (cytospin) or flow cytometry.
Cytokine Quantification
The levels of interleukin (IL) 1β, IL-6, IL-10 and tumor necrosis factor α (TNF-α) cytokines were measured in the supernatant of the Biogel-induced inflammatory exudate by sandwich enzyme-linked immunosorbent assay kits (OptEIA BD Biosciences, San Diego, California), according to the manufacturer’s instructions. The plates were read in a microplate reader at 450 nm (MultisKan MS, Labsystems, Finland).
Adhesion Molecule Expression in BMCs, Peripheral Blood Cells, and Inflammatory Exudate
The expression of L-selectin (CD62L, clone MEL14), Mac-1 (CD11b, clone Mi/70), and platelet endothelial cell adhesion molecule (PECAM) 1 (CD31, clone MEC13.3) was evaluated by FACS. The BMCs were collected with 1 mL of RPMI-1640 + 10% FCS. Peripheral blood was obtained using heparinized capillary tubes and transferred to PBS–heparin. Cells were analyzed after erythrocyte lysis. Inflammatory cells were obtained in the local sc exudate 24 hours after Biogel injection.
Hydrogen Peroxide Detection
Hydrogen peroxide (H2O2) generation was assayed by the method of Pick and Mizel 26 Briefly, inflammatory cells were adjusted to 2 × 106 cells/mL, centrifuged for 10 minutes, and suspended in 1 mL of phenol red solution. The cells were plated onto 96-well plates in the presence or absence of phorbol myristate acetate (20 ng/well) and incubated in a humidified atmosphere at 37°C/5% CO2 for 1 hour. The reaction was stopped by addition of NaOH. Hydrogen peroxide-dependent phenol red oxidation was measured spectrophotometrically at 620 nm (MultisKan MS, Labsystems, Finland) and the results were compared with a standard curve generated with known amounts of H2O2.
Nitric Oxide Detection
Nitric oxide (NO) production was detected with the Griess reagent. 27 The concentration of nitrite was measured in the supernatant of inflammatory exudates. Cells were centrifuged (1200g, 10 minutes) and the supernatant was collected and mixed in 1:1 ratio with Griess reagent (1% sulfanilamide/0.1% n-(1-naphthyl) ethylenediamine dihydrochloride in 2.5% phosphoric acid). After 10 minutes of incubation, absorbance was measured at 540 nm (MultisKan MS Reader, Labsystems, Finland). Nitrite production was compared with a standard curve generated with known amounts of NaNO2.
Real-Time Polymerase Chain Reaction Assay
RNA was extracted from total BMCs isolated from femurs using TRIZOL (Invitrogen, Carlsbad, California). RNA was treated with RNAse-free-DNAse I (Roche, Basel, Switzerland) and 1 μg of total RNA was reverse transcribed using 50 μmol/L poly(dT)12-18, deoxyribonucleotide triphosphates (10 μmol/L), and SuperScript III RT (200 U/μL; Invitrogen). Messenger RNA (mRNA) levels were quantified by reverse transcription real-time polymerase chain reaction (PCR) using Platinum SYBR Green qPCR Supermix-UDG (Invitrogen) with 1 μL complementary DNA template and 0.2 μmol/L primers specific for Ahr (5′-tgcaagaagccggaaaactg-3′, 5′-agaatcccacatccgcatga-3′) gene and the reference gene Ppia (cyclophilin A, 5′-aaatgcccgcaagtcaaaag-3′, 5′-agcgttttgggtccaggaat-3′). The reactions were carried out in a Chromo4 detector (MJ Research, Waltham, Massachusetts). Relative gene expression levels were calculated by the comparative Ct method. 28 All experiments were performed in triplicate.
Statistical Analysis
Statistical analysis was performed by analysis of variance followed by Tukey multiple comparison tests. Significance was set at P ≤ 0.05 for all analyses.
Results
7,12-Dimethylbenz(a)anthracene Treatment Impairs Acute Inflammatory Response in AIRmin Mice
First, we studied the effect of DMBA, 24 hours posttreatment, on the local acute inflammation induced by sc injection of Biogel beads in both AIRmax and AIRmin mice. In DMBA-treated AIRmin mice, the number of infiltrating inflammatory cells, 24 hours after Biogel injection, was significantly reduced when compared to vehicle-treated mice whereas no changes were observed in AIRmax mice (Figure 1A). On the other hand, neither the protein and IL-1β contents in the inflammatory exudates nor the H2O2 production by inflammatory cells was affected by DMBA treatment (Figure 1B-D). Interleukin 10, TNF-α, and NO could not be detected in the control groups or at 24 hours post-DMBA treatment in the inflammatory exudates produced by Biogel (data not shown).
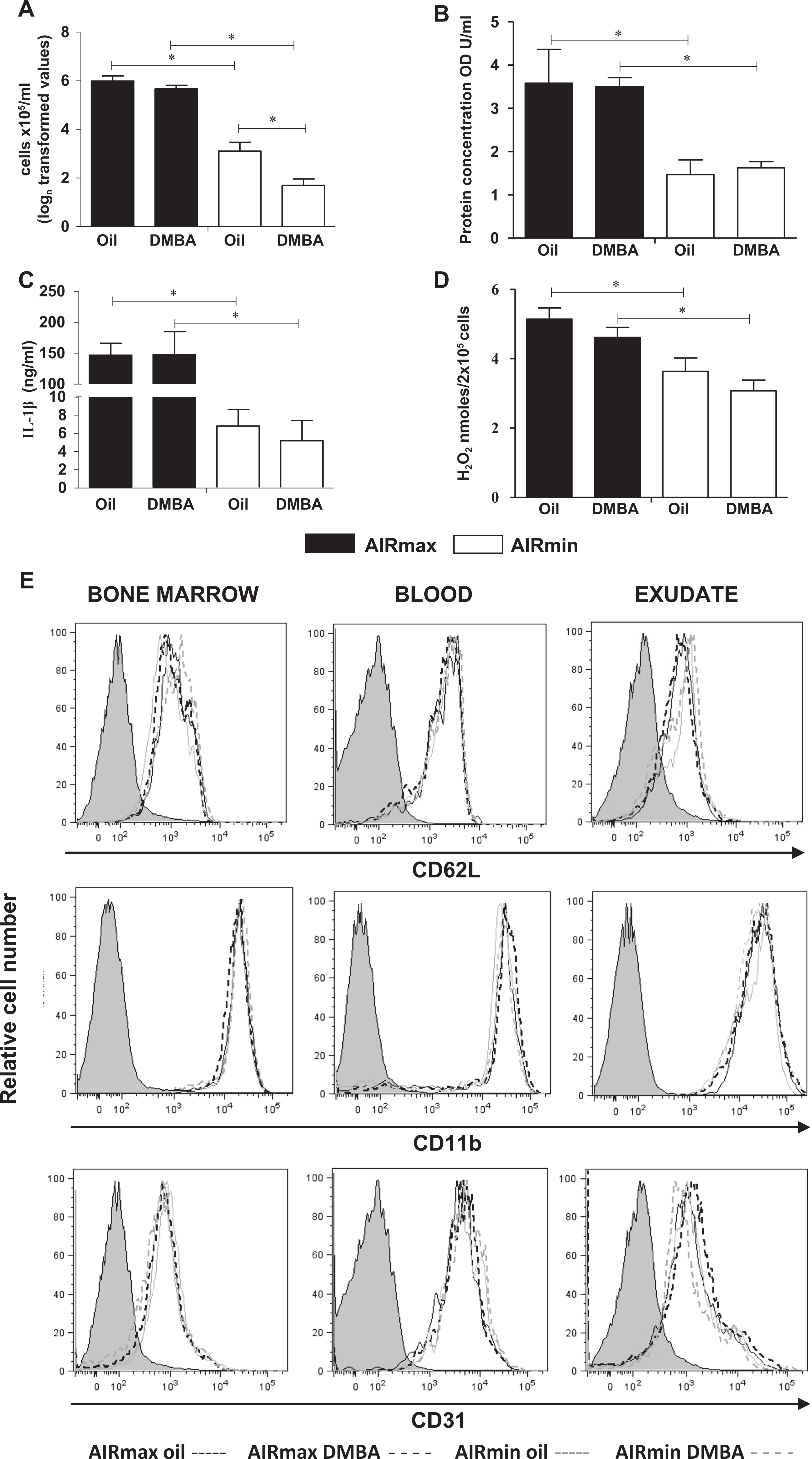
7,12-Dimethylbenz(a)anthracene treatment affects the 24-hour inflammatory reaction induced by Biogel in AIRmin mice. A, Cell number expressed in natural logarithm (logn) to improve normality and (B) protein concentration expressed in optical density units (OD U/mL) in the inflammatory site. C, IL-1β production and (D) H2O2 synthesis by freshly isolated inflammatory cells. (E) Relative expression of CD62L, CD11b, and CD31 molecules on leukocytes from BM, blood, and 24-hour inflammatory exudates AIRmax (black lines) and AIRmin (gray lines) DMBA (dashed line) or oil-treated mice (dotted line). Values represent mean ± SEM (N = 6), *P ≤ 0.05 significant difference between groups. AIRmin indicates minimal acute inflammatory response; AIRmax, maximal acute inflammatory response; IL, interleukin; BM, bone marrow; DMBA, 7,12-dimethylbenz(a)anthracene; SEM, standard error of the mean.
The AIRmin mice consistently have a lower inflammatory response to Biogel than that of AIRmax mice, regardless of treatment with DMBA. The difference in inflammatory responsiveness between AIRmax and AIRmin mice was similar for the infiltrating cell numbers, protein content, and IL-1β concentration in the 24-hour exudates in control 29,30 and DMBA-treated mice (Figure 1). Infiltrating cell phenotypes were characterized by double staining for Ly-6G (polymorphonuclear leukocytes) and CD62L (L-selectin), CD11b (β-integrin), or CD31 (PECAM). In spite of the low cell numbers in AIRmin mice exudates, we observed equivalent expression of CD62L, CD11b, and CD31 adhesion molecules in Ly-6G+ neutrophil populations from DMBA- or oil-treated mice. In both AIRmax and AIRmin mice, CD62L selectin expression decreased in a similar way on Ly6G+ cells from exudates compared to the expression in BM or blood-derived cells whereas CD11b expression remained high on cells from those 3 compartments (Figure 1E). The CD31 expression was higher in the blood than that in the BM and exudates-derived cells.
7,12-Dimethylbenz(a)anthracene Induces BM Toxicity in AIRmin Mice
Here, we examined the effects of DMBA on BMCs flushed from the femurs and on the histopathology of the sternum. We observed a reduction of around 50% in BMC numbers in AIRmin mice 24 hours after DMBA treatment. This reduction was totally reversed by pretreatment of mice with α-NF, a nontoxic AhR and CYP inhibitor. 31 The BMC numbers were significantly higher in AIRmax mice than that in AIRmin mice and were unaffected by DMBA treatment (Figure 2A). Histological analysis of the sternum revealed dilated sinusoids and disruption of stromal architecture in DMBA-treated AIRmin mice (Figure 2B) whereas the sternum structure in DMBA-treated AIRmax was similar to the control oil-treated mice. 7,12-Dimethylbenz(a)anthracene (50 mg/kg bw) caused death of 50% AIRmin mice at 5 to 10 days after ip injection whereas AIRmax mice were resistant (Figure 2C). No deaths were observed in the olive-treated mice (data not shown).
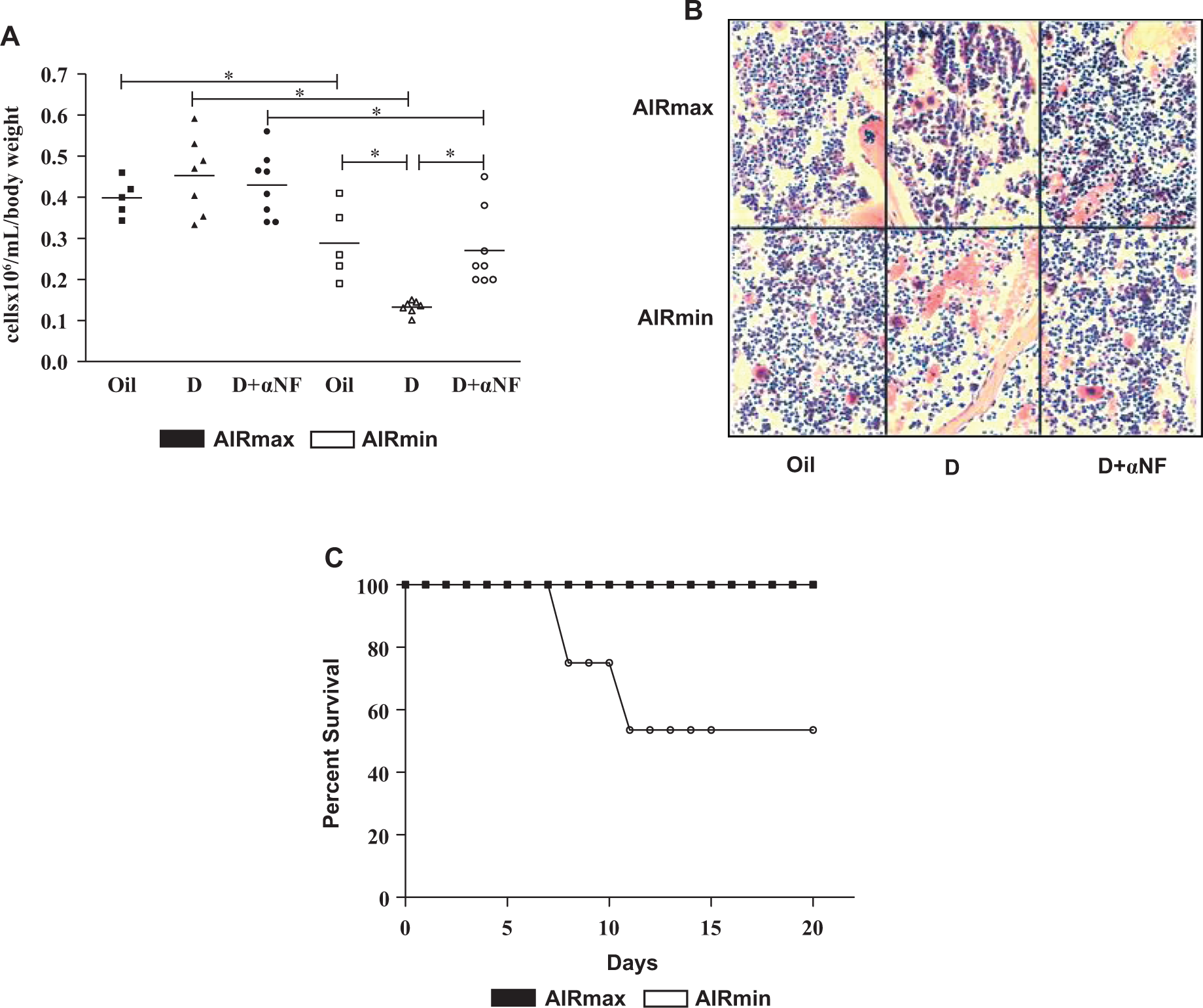
DMBA-induced BM hypocellularity in AIRmin mice. A, Comparison of bone marrow cellularity from AIRmax and AIRmin mice at 24 hours posttreatment with oil, DMBA (D) or DMBA + α-NF (D + α-NF). B, Photomicrographs of bone marrow biopsy sections from AIRmax and AIRmin sternums 24 hours after DMBA or DMBA + α-NF treatment (hematoxylin and eosin stain, original magnification ×40). C, Survival curve showing mortality of the AIRmax and AIRmin mice treated with DMBA (50 mg/kg, bw). Data are expressed as mean ± SEM of 5 to 8 mice, *P ≤ 0.05 significant difference between groups. AIRmin indicates minimal acute inflammatory response; AIRmax, maximal acute inflammatory response; BM, bone marrow; DMBA, 7,12-dimethylbenz(a)anthracene; α-NF, α-naphthoflavone; SEM, standard error of the mean.
To further characterize the changes in BMCs following DMBA treatment, we analyzed the live myeloid cell populations by flow cytometry (Figure 3A). Myeloid cells were identified by staining with anti-GR-1 and anti-CD11b where CD11b+ cells were predominantly neutrophils (GR-1hi/CD11b+) or monocytes (GR-1low/CD11b+). 29,32
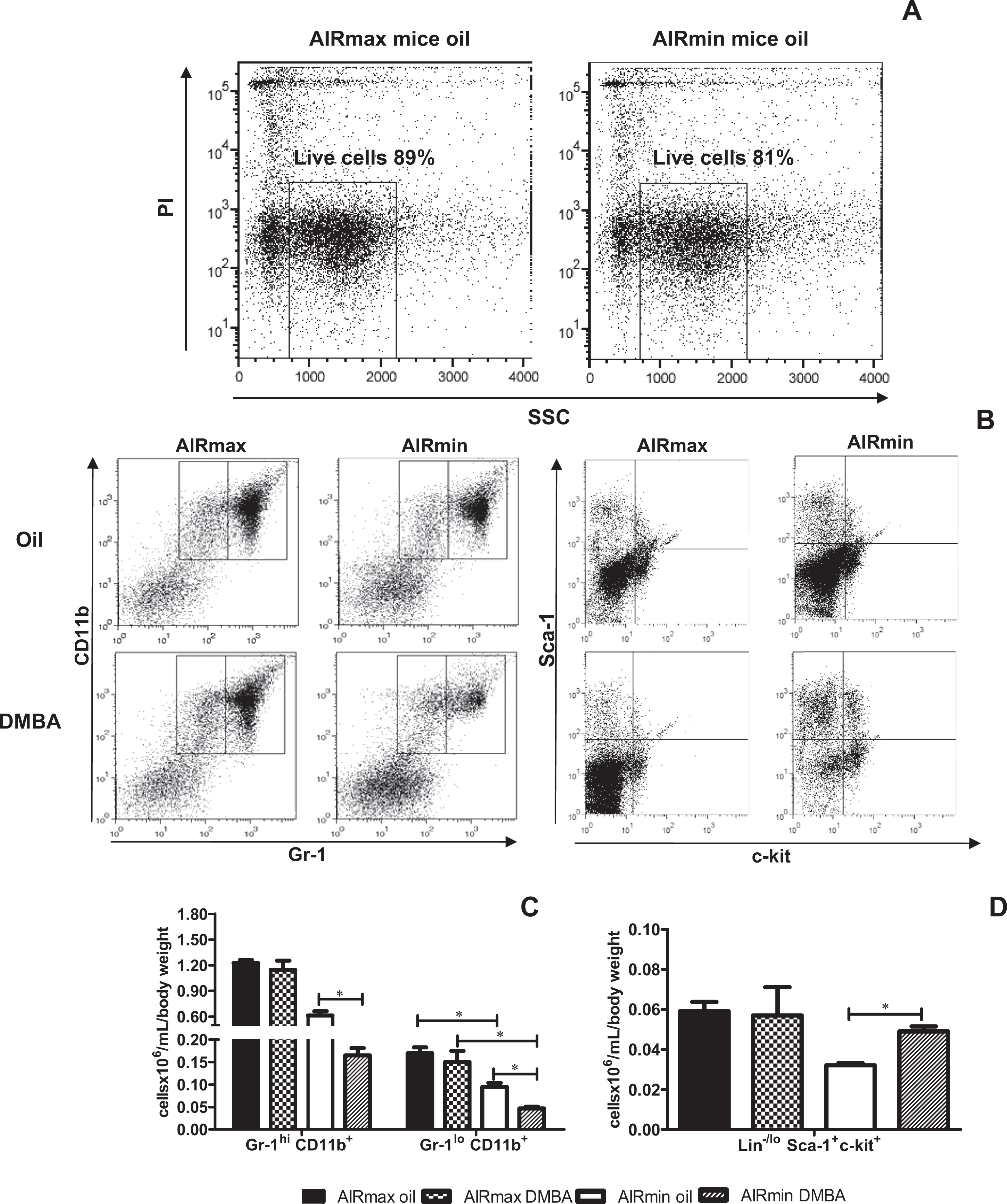
Flow cytometric analysis of freshly isolated bone marrow cells showed reduced numbers of BM-derived myeloid cells and an increase in the number of HSCs in DMBA-treated AIRmin mice. A, Representative dot-plots show live myeloid cell populations from control mice. B, Representative dot-plots for neutrophil (GR1hi/CD11b+) and monocyte (GR1lo/CD11b+) populations on the left and representative dot-plots for HSCs (Lin−/lo, c-kit+, and Sca-1+) on the right. C, Total number of neutrophil–monocytic population and (D) HSCs from AIR mice followed 24-hour DMBA treatment. Data are expressed as mean ± SEM (N = 5), *P ≤ 0.05 significant difference between groups. BM indicates bone marrow; HSCs, hematopoietic stem cells; DMBA, 7,12-dimethylbenz(a)anthracene; AIR, acute inflammatory response; AIRmin, minimal acute inflammatory response; SEM, standard error of the mean
Representative dot plots identifying granulocyte and monocyte populations are shown for control AIRmax and AIRmin mice (Figure 3B). At 24 hours after DMBA treatment, the numbers of BM-derived GR-1hi/CD11b+ myeloid cells from AIRmin mice decreased about 70%, from 0.61 to 0.16 × 106/mL/bw (Figure 3C). A similar effect was observed for the GR-1lo/CD11b+ population (Figure 3C).
This decrease could be related to a blockade of myeloid cell maturation. To investigate this hypothesis, we analyzed the HSC population. Subpopulations of Lin-negative (Lin–) and positive cell for c-kit (c-kit+) and positive cell for Sca-1 (Sca-1+) were essentially HSC. 33 We observed an increase in HSC with Lin−Sca-1+c-kit+ phenotype only in BMCs from DMBA-treated AIRmin mice when compared to the control group (Figure 3D).
Morphological analysis of BMCs from AIRmax and AIRmin mice, 24 hours after injection of DMBA, revealed variability in the cellular content. The number of both blast and immature myeloid cells was significantly increased (>2-fold) in DMBA-treated AIRmin mice (Figure 4A and B) while the neutrophil populations with ring-shaped and segmented nuclei were about one-half of the respective control vehicle-treated mice (Figure 4C and D). These cellular changes were not observed in AIRmax mice. It is important to point out that, in non-treated groups, the number of segmented neutrophils is always higher in BM from AIRmax than from AIRmin mice (Figure 4D), as reported previously. 29
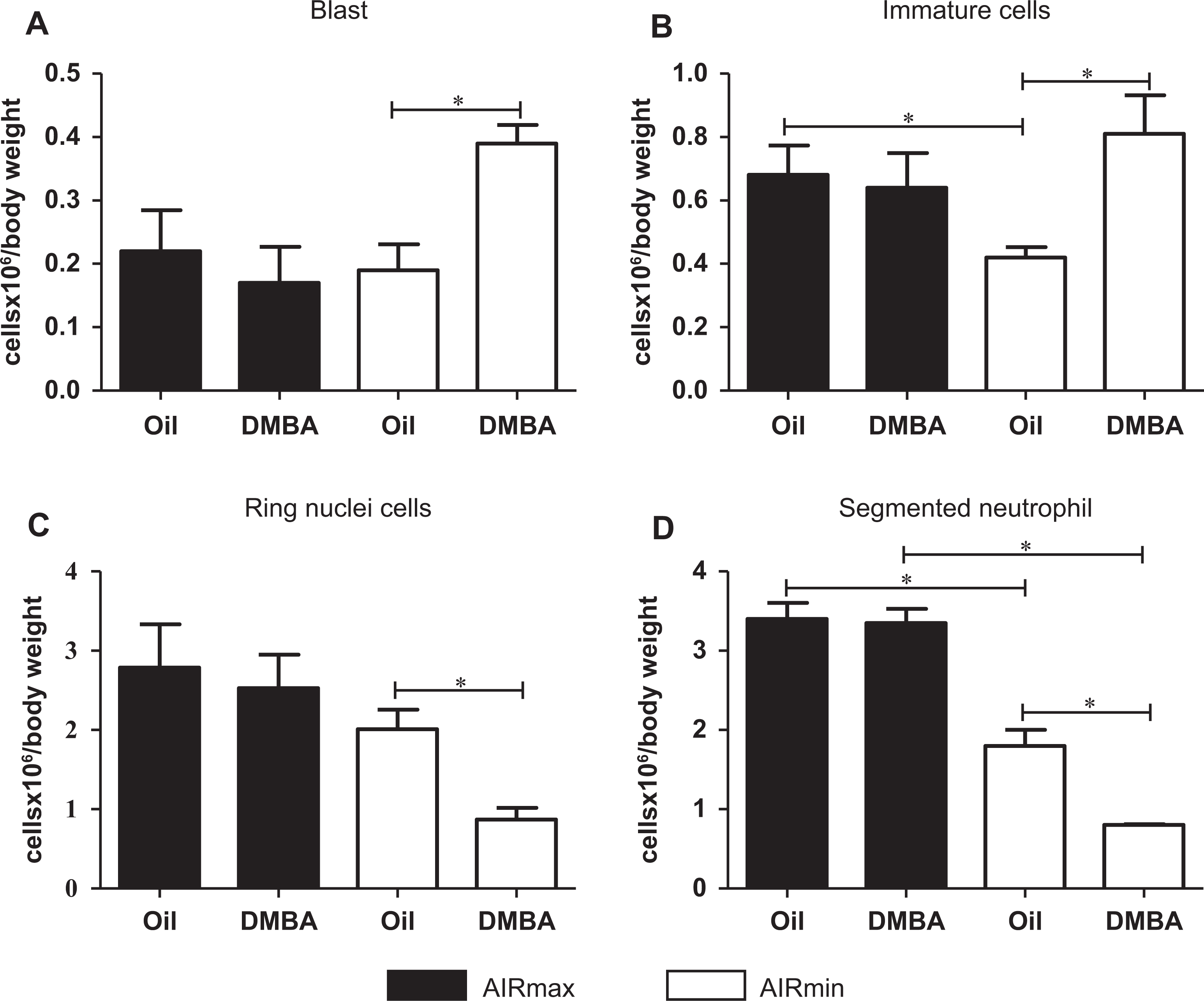
Differential bone marrow count in cytospin preparations showing decreased numbers of segmented and ring nuclei neutrophils and increased blast and immature cells in DMBA-treated AIRmin mice. Number of (A) blast cells, (B) immature cells, (C) ring nuclei neutrophils, and (D) segmented neutrophils from AIRmax and AIRmin mice 24 hours after DMBA treatment. Data are expressed as mean ± SEM (N = 9). *P ≤ 0.05 significant difference between the indicated groups. DMBA indicates 7,12-dimethylbenz(a)anthracene; AIRmin, minimal acute inflammatory response; AIRmax, maximal acute inflammatory response; SEM, standard error of the mean.
To investigate the effect of DMBA treatment on BMC functions, we compared the in vitro proliferative potential of BMCs from 24-hour DMBA-treated AIRmax and AIRmin mice that induced the hematopoietic growth factor, GM-CSF. Bone marrow cells from AIRmin-treated mice showed impaired GM-CSF-stimulated proliferation in 5 days culture whereas BMCs from AIRmax mice proliferation was unaffected by DMBA treatment (Figure 5A). Also, the clonal expansion of myeloid precursors induced by GM-CSF was inhibited in DMBA-treated AIRmin mice (Figure 5B).
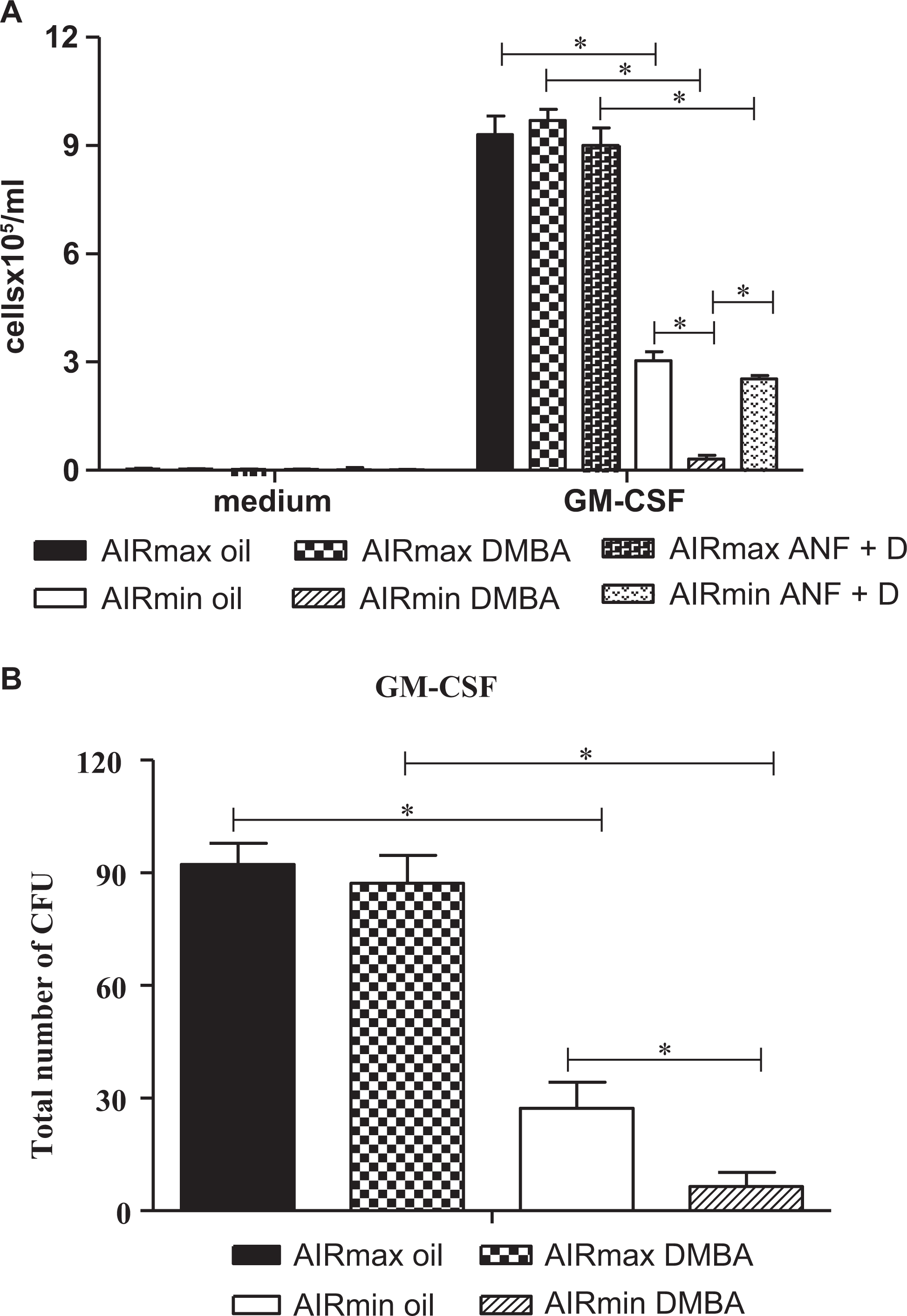
DMBA treatment impairs proliferation and differentiation capacities of bone marrow cells from AIRmin mice after in vitro stimulation with GM-CSF. A, In vitro proliferative response to GM-CSF of bone marrow cells after oil, DMBA, or DMBA + α-NF in vivo treatment. B, GM-CSF-induced BMC clonal expansion in AIRmax and AIRmin mice at 24 hours post DMBA in vivo treatment. Values represent mean ± SEM (N = 6), *P ≤ .05 significant difference between oil and DMBA-treated groups. DMBA indicates 7,12-dimethylbenz(a)anthracene; AIRmin, minimal acute inflammatory response; GM-CSF, granulocyte-macrophage colony-stimulating factor; SEM, standard error of the mean; α-NF, α-naphthoflavone; BMC, bone marrow cell.
Both the hypocellularity (Figure 2A) and the impairment of BMC proliferation (Figure 5A) observed in DMBA-treated AIRmin mice were completely abolished by prior injection of α-NF, demonstrating that toxicity in AIRmin mice is dependent on the binding of DMBA to AhR and/or on downstream components of this pathway.
Aryl Hydrocarbon Receptor mRNA and Protein Expression in BMCs
The Ahr mRNA and protein expression were studied in BMCs from AIRmax and AIRmin mice to evaluate whether there is a correlation between cell depletion or proliferation impairment and AhR expression as a result of the DMBA treatment. The Ahr mRNA expression level was significantly increased in the BMCs of AIRmin mice 12 hours after treatment with DMBA. On the contrary, the Ahr expression level in AIRmax mice decreased about 4-fold at 24 hours after treatment (Figure 6A). Immunostaining with anti-AhR antibody showed an increase in AhR intracellular expression at 24 hours in BMCs from DMBA-treated AIRmin mice than that from controls while AhR levels were unaltered in DMBA-treated AIRmax mice (Figure 6B and C).
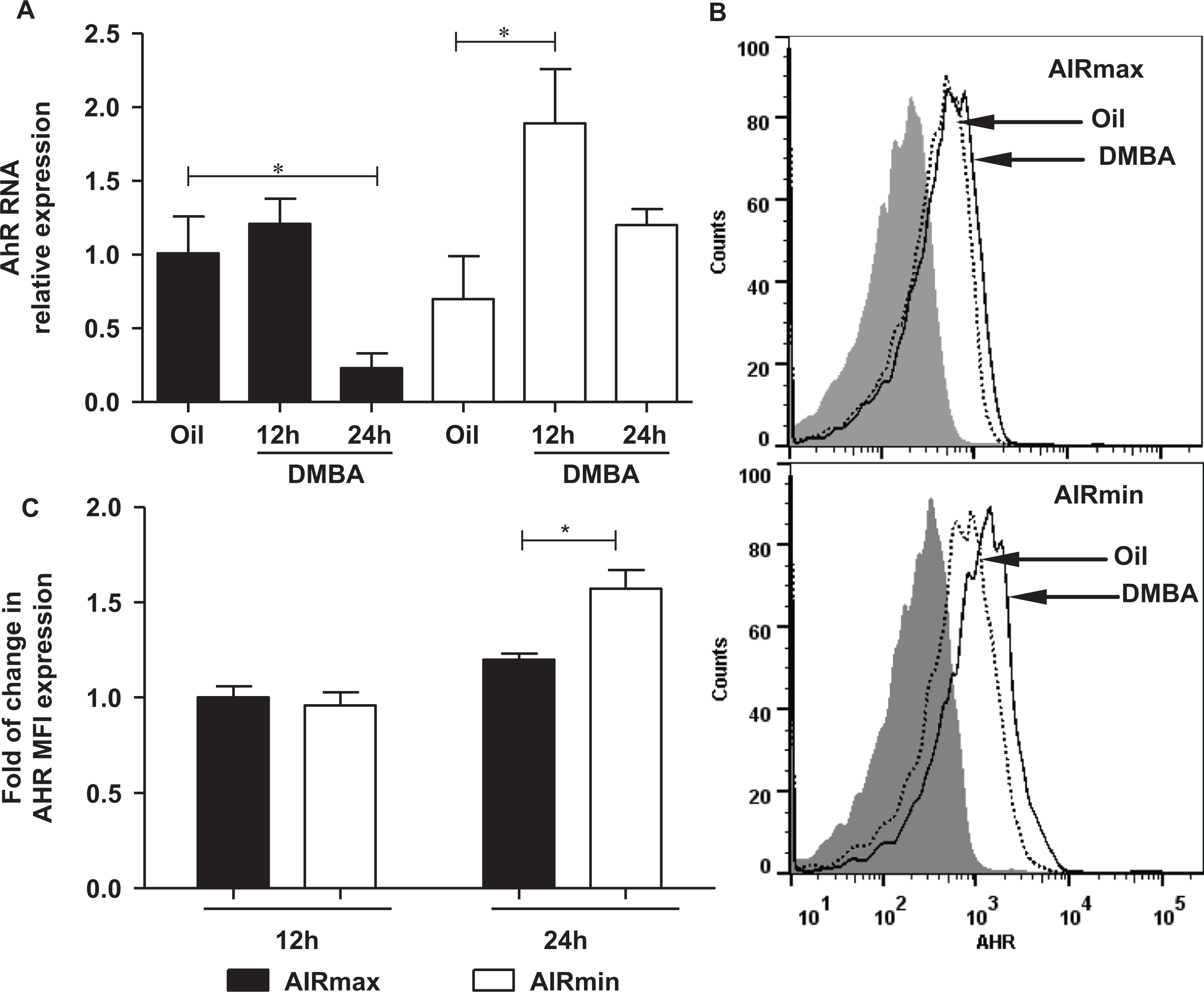
Ahr mRNA transcripts and AHR protein expression in bone marrow cells from AIRmax (black bar) and AIRmin (white bar) mice 12 and 24 hours post-DMBA or -oil treatment. A, Gene expression measured by RT-qPCR of bone marrow cells from oil or DMBA-treated AIRmax and AIRmin mice. B, Representative histogram of AhR-expressing BMCs isolated from AIRmax and AIRmin mice 24 hours after oil (dotted line) or 50 mg/kg bw DMBA (solid line) injection. Staining with isotype control IgG is also shown (gray curve). C, Relative fold change of AHR median fluorescence intensity (MFI) of total BMCs isolated from DMBA or oil-treated AIRmax and AIRmin mice. Relative fold change was calculated relative to oil-treated mice in 2 independent experiments. Values represent mean ± SEM (N = 6). *P ≤ 0.05 significant difference between groups. AHR indicates aryl hydrocarbon receptor; AIRmin, minimal acute inflammatory response; AIRmax, maximal acute inflammatory response; DMBA, 7,12-dimethylbenz(a)anthracene; IgG, immunoglobulin; bw, body weight; SEM, standard error of the mean.
Discussion
The difference between AIRmax and AIRmin mice in the susceptibility to DMBA was first demonstrated by our group in studies of skin carcinogenesis induced by epicutaneous administration of DMBA/12-O-tetradecanoylphorbol-13-acetate or by DMBA alone where AIRmax mice were more resistant than AIRmin to malignant skin tumors and lung adenocarcinoma development. The segregation of Ahrd and Ahrb1 alleles in AIRmax and AIRmin lines, respectively, was implicated in these phenotypes. 22 –24
In the present study, we analyzed the influence of receptor polymorphism on the effects of DMBA treatment on the BM and on the acute inflammatory response (AIR) of AIRmax and AIRmin mice. Intraperitoneal DMBA injection induced early hypocellularity in the BM of AIRmin mice only (Figure 2A) mainly due to a reduction in the mature neutrophil population (Gr-1hiCD11b+, Figure 3C) with ring-shaped or segmented nuclei (Figure 4C and D). On the other hand, DMBA treatment promoted an increase in the Lin–, Sca-1+, and c-kit+ (CD117) population (Lin−/lo, c-kithi, Sca-1hi) which is enriched for HSCs in mice (Figures 3C and 4A and B). 34 Accordingly, there was an impairment of the in vitro GM-CSF-induced proliferation and differentiation capacities of BMCs from DMBA-treated AIRmin mice (Figure 5) and the in vivo cell infiltration to a local inflammatory stimulus with Biogel was significantly decreased in these mice (Figure 1A). However, DMBA treatment did not affect the expression levels in granulocytes of PECAM (CD31), L-selectin (CD62L), and integrin (CD11b) adhesion molecules that are critical for their migration from the BM through the blood to the inflammatory site (Figure 1E). These results evidence that although AhR activation by DMBA results in less neutrophils in Biogel-induced inflammatory exudates, this effect is not mediated by the deregulation in levels of adhesion molecules expression on BM, blood, or exudated neutrophils.
In addition to immunosuppression, oral DMBA administration in mice causes several clinical signals such as weight loss, generalized weakness, and electrolyte imbalance with renal and cardiovascular failure leading to death in 4 weeks. 35 In our experiments, we observed some of those clinical signs in 50% of AIRmin mice (weakness and hypothermia) which died in 5 to 10 days after 50 mg/kg bw DMBA ip injection (Figure 2C). In contrast, the drug had no effect in AIRmax mice.
The persistent BM suppression induced by DMBA in AIRmin mice, in contrast with the absence of adverse effects in AIRmax mice, might contribute to the differential susceptibility of these 2 mouse lines to tumor outgrowth or metastasis development. These divergent responses might be associated with the affinity of DMBA binding to AhR, causing BM toxicity which results in impairment of both innate and adaptive immune responses. Aryl hydrocarbon receptor is expressed in HSCs 36 and, besides its role as a ligand-activated transcription factor, functions as a regulator of HSCs and of several pathways of hematopoiesis. 37 As an example, dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin [TCDD]) AhR signaling in HSCs and/or hematopoietic progenitor cells may block differentiation into the different lineages, causing their accumulation. 38 Therefore, inappropriate AhR activation may be important in malignant transformation of HSCs. 36,39,40 This idea was based on the fact that HSCs from AhR null-allele (knock-out) mice are hyperproliferative with high rates of cell division when compared to cells from wild-type mice after TCDD treatment. 41 In addition, Ahr mRNA expression was diminished in HSCs from mice treated with growth factors and cytokines, 36 indicating that Ahr gene expression is downregulated under conditions that stimulate HSC proliferation.
In our study, quantitative PCR and flow cytometric analysis showed that BMCs from AIRmax and AIRmin mice constitutively express equivalent levels of Ahr. 7,12-Dimethylbenz(a)anthracene treatment resulted in an increase in both Ahr transcript levels and AhR protein expression only in BMCs from AIRmin mice (Figure 6).
These results were not expected since several studies have demonstrated that AhR protein is rapidly downregulated following activation. Nevertheless, other reports show that AhR degradation differs among tissues and cells, the type and route of administration of AhR ligand, and the time of analysis. 42,43 According to our results, increased expression of Ahr and a putative inhibition of AhR protein degradation observed in BMC from AIRmin mice might lead to an increase in the duration and possibly magnitude of AhR-mediated DMBA toxic effects in these mice.
On the other hand, AIRmax mice have an unaltered AhR protein profile despite a significant reduction in Ahr mRNA expression 24 hours post-DMBA. The result indicates that there is a response to DMBA in AIRmax mice, even without detectable toxic effects. The effect on protein expression could be occurring at a later point (Figure 6).
There is increasing evidence suggesting a role of AhR in inflammation. Some examples are the interactions of AhR with inflammatory mediators, such as TNF-α or IL-6, prostaglandins, and leukotrienes 12,44 and the interaction of signaling pathways activated by AhR ligands and by nuclear factor-κB through inflammatory mediators. 40,45,46 This evidence, together with the previous reports of our group, suggests a link between the Ahr polymorphism present in AIRmax and AIRmin mice and the regulation of inflammatory responsiveness through neutrophil production rates in the BM. 29 In fact, a QTL has been mapped to murine chromosome 12, close to Ahr at 35 Mb, which regulates blood neutrophil numbers and the number of progenitors in the BM. 47 In our model, a genome-wide association study in combination with genetic clustering to map loci affecting inflammation phenotypes in an (AIRmax × AIRmin) F2 intercross population evidenced, among others, the rs6328018d single-nucleotide polymorphism near the Ahr locus that was found to be associated with the local infiltrating cell numbers induced by Biogel sc injection. 45 The analysis evidenced the polygenic control of inflammatory response and candidated Ahr (or nearby genes) as one of the QTL contributing to the control of the differential inflammatory responses between AIRmax and AIRmin mice.
Studies have shown that the Ahr of DBA/2 mice (Ahrd allele) shows about 10-fold lower TCDD-binding affinity than that of C57BL/6 mice (Ahrb allele). 48 Such relationship, although well established, may vary because of the interaction with the individual genetic constitution. As an example, wild-type C57BL/6J mice (Ahrb1 ) and congenic (Ahrd ) mice whose receptor is less responsive to PAHs are equally susceptible to BM depletion by DMBA treatment. 49 In our model, we demonstrated differences in DMBA susceptibility linked to Ahr polymorphism in AIRmax and AIRmin mice, 24 supporting the observation that the phenotypic expression of Ahr alleles can be influenced by the interaction with different genetic backgrounds. Variations in the phenotypic gene expression have been shown for another polymorphic gene, the solute carrier 11a1 (Slc11a1) which controls resistance to intracellular bacterial and protozoal infection in mice. 50 The phenotypic expression of the resistant and susceptibility associated alleles, evaluated by resistance to experimental Salmonella typhimurium infection, was largely modulated by AIRmax or AIRmin genetic backgrounds, since AIRmax mice homozygous for the susceptible allele were significantly more resistant to infection than AIRmin or BALB/cJ mice homozygous for the same Slc11a1 allele. 51,52 This phenomenon has also been observed in humans. Several polymorphisms in the human AHR have been found, and their effects on receptor activities are not conclusive, presumably due to the different ethnic groups’ genetic backgrounds. As an example, a polymorphism that results in an arginine to lysine substitution at the transactivation domain (codon 554) of AHR had no significant effect on CYP1A1 induction in cultured lymphocytes from Japanese individuals whereas among Caucasians, higher induced CYP1A1 activity was found in heterozygous or homozygous (Lys554/Lys554) individuals than that in homozygous Arg554/Arg554 individuals. 18
In conclusion, we have found that AIRmin mice are more susceptible than AIRmax to DMBA-induced toxicity with an increase in immature myeloid precursors and a profound decrease in mature neutrophils in the BM, affecting the acute inflammatory response. Such effects observed in AIRmin mice might be influenced by AhR activation through high affinity binding to DMBA, resulting in the imbalance of the cellular differentiation process in the BM. This might affect the supply of functional cells to the immune system hindering the immune response and underlying the high susceptibility of AIRmin mice to DMBA-induced carcinogenesis.
The AIRmax/AIRmin mouse model may contribute to the identification of genetic factors that control the intensity of AIR and their involvement in determining resistance or susceptibility to xenobiotic-induced myelotoxicity and cancer, opening new perspectives for the search of therapeutic agents.
Footnotes
Acknowledgments
The authors would like to thank Dr Jorge Ferreira for help with cell phenotyping by FACS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico [CNPq-307650/2007-3 and 305320/2010-6] and Fundação de Amparo a Pesquisa do Estado de São Paulo [FAPESP-04/11489-1].
