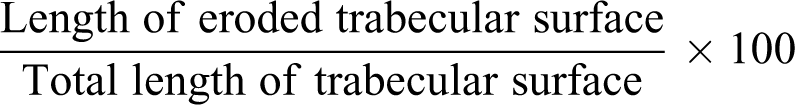Abstract
The present study aimed to evaluate the role of rivastigmine against the effect of a single unilateral botulinum toxin-A (BTX-A) injection on the bone and bone marrow of adult albino rats 4 weeks after injection. Twenty-four Wistar albino rats were divided into four equal groups: group I, which received distilled water; group II, which received rivastigmine (0.3 mg/kg daily, intraperitoneally for 4 weeks); group III, which received BTX-A (4 IU in 0.2 mL physiological saline) single dose, intramuscularly; and group IV, which received BTX-A + rivastigmine. The results revealed that BTX-A induced a significant decrease in the calcium level with a significant increase in the phosphorus, alkaline phosphatase, C-reactive protein, and tumor necrosis factor α levels in serum. Furthermore, a significant increase in malondialdehyde with a significant decrease in reduced glutathione activities in both bone and bone marrow. Histologically, a distortion and thinning out of the compact bone and trabeculae of cancellous bone of the rat femur in the BTX-A group with an increase in adipocytes in adjacent bone marrow were detected. Immunohistochemically, Cluster of Differentiation 68 (CD68) showed a significant increase in both osteoclasts and bone marrow macrophage. Rivastigmine treatment could relieve the toxic effects induced by BTX-A. In conclusion, rivastigmine has a protective effect against the hazardous effects of BTX-A on bone and bone marrow.
Introduction
Osteoporosis is a systemic skeletal disease where reduction of bone mass and bone mineral density leads to an increase in the risk of bone fracture. 1 It is common among the old especially women after menopause due to the reduction of estrogen level. Osteoporosis can be caused by a number of medications such as chemotherapy, glucocorticoids, and antiepileptic drugs. 2 In addition, some diseases such as kidney diseases, endocrinal diseases, and anorexia may result in osteoporosis. 3,4
Clostridium botulinum bacteria is the source of production of botulinum toxin (BTX). It has seven toxin subtypes (A, B, C1, D, E, F, and G). BTX-A and BTX-B are commonly used clinically nowadays. However, BTX-A is preferred due to its longer duration of action. 5
The mechanism of action of the BTX is to stop the release of acetylcholine (ACh) neurotransmitter at the axon end. The toxin degrades the Synaptosomal-associated protein 25 (SNAP-25) protein (soluble N-ethylmaleimide-sensitive factor (NSF)-attachment protein receptors) which is essential to the ACh vesicle fusion at the presynaptic membrane of the neuromuscular junctions. 6 Therefore, the action potentials of the motor nerve cannot be transmitted to the muscle. This leads to transient selective paralysis of the muscle at the site of injection, which disappears after 3–6 months. 7,8
BTX-A has become the most commonly used nonsurgical cosmetic product. 9 It is used to reduce facial wrinkles, as it relaxes the overactive facial muscles and enhances the cosmetic appearance. 10 Frequent injections of BTX are required in many conditions that may cause some reversible side effects such as pain, bruising, and edema at the site of injection. 11,12 However, undesirable effects have been reported such as paralysis of distant muscle groups and hyposthenia. 13
It has been reported that bone cells have acetylcholine receptor (AChR) subunits and the acetylcholinesterase (AChE) enzyme is highly expressed on them, particularly at the stage of osteoblast differentiation. 14 On osteoblasts, AChR and cholinergic components have an important role in controlling alkaline phosphatase (ALP) activity. 15 The low level of ACh is a common cause of bone loss. 16
The acetylcholinesterase inhibitor drugs (AChEIs) that prevent the action of AChE and increase the levels of ACh in the synaptic space lead to stimulation of AChR. 17 Rivastigmine—one of the AChEIs—has been extensively used in the treatment of dementias particularly Alzheimer’s disease since the mid-1990s. 18 The present study aimed to evaluate the role of rivastigmine against the effect of a single unilateral BTX injection on the bone and bone marrow of adult albino rats 4 weeks after injection.
Materials and methods
Chemicals
BTX-A (KC Pharmaceuticals, Inc. CA,) stored at −20 to −5°C, rivastigmine (Novartis Co. Cairo, Egypt), reduced glutathione (GSH) (Biodiagnostic, Egypt), rabbit monoclonal CD68 antibody (Abcam, Cambridge, MA, USA), tumor necrosis factor (TNF) α ELISA kit (IDlabsT- Minc. Biotechnology, Canada), C-reactive protein (CRP) (Agappe Diagnostic LTD, Mumbai, India), ALP, calcium (Ca), and phosphorus (P) (BioMed, Egypt) were used for analyses. All other chemicals were of analytical grade and were obtained from commercial sources.
Animals
A total of 24 Wistar albino adult healthy rats weighing 180–210 g were used in this study. Animals were harbored on a constant temperature (24°C ± 1°C) with 12-h light/dark cycle (lights on from 08:00 a.m.) and humidity. Water was available ad libitum with normal rat chow. Our study was approved by the Institutional Review Board and followed the guidelines for animal welfare of Faculty of Medicine, Minia University.
Experimental protocol
Four equal groups of rats were studied. Group I (control group) that received only distilled water; group II (rivastigmine group) that received rivastigmine (0.3 mg/kg daily, intraperitoneally for 4 weeks) 19 ; group III (toxic group) that received BTX-A (4 IU BTX dissolved in 0.2 mL physiological saline) single dose, intramuscular injection, distributed in the quadriceps (2 IU), hamstrings (1 IU), and posterior calf muscles (1 IU) of the right hind limb 20,21 ; and group IV (treated group) that received BTX-A + rivastigmine starting at the day of BTX injection (with the same previously described doses).
Collection of blood and tissue samples
At the end of the experiment, blood samples were collected from the abdominal aorta after anesthetizing the rats with ether. The blood samples were centrifuged (Centrifuge Janetzki, T30, Germany) at 5000 r/min for 10 min for serum collection, and then the sera were separated and stored in aliquots at −80°C until used for the estimation of Ca, P, ALP, TNF-α, and CRP. The right femur of each rat was cleaned from the adhering muscles. Moreover, the bone marrow was collected from the right femur of each rat by aspiration using Salah’s marrow puncture needle. The bone and bone marrow were kept at −80°C until biochemical analysis. Histopathological studies of the bone and bone marrow as well as an immunohistochemical study of the bone were done.
Preparation of tissue homogenates
The bones were perfused with phosphate-buffered saline with a pH of 7.4 to remove any blood cells or clots. Then, 0.25 g of bone was weighed and crushed using mortar and pestle on ice. The bone tissue was transferred to a 10 mL tube containing 2 mL of 20 mM HEPES buffer (20 mM HEPES buffer with a pH of 7.2, containing 1 mM EGTA, 210 mM Mannitol, and 70 mM sucrose per gram tissue). After that, the tissue was placed on ice and homogenized using polytron homogenizer (Tri-R Stir-R homogenizer, Tri-R Instruments, Inc., Rockville Centre, New York, USA.). Next, the homogenized mixture was centrifuged at 5000 r/min for 10 min at 4°C. The supernatant was kept at −80°C until the assessment of malondialdehyde (MDA) and GSH activities.
The bone marrow was homogenized in approximately 2 mL of ice-cold saline and 10 volumes of ice-cold phosphate buffer (prepared by dissolving 8.01 g of NaCl, 0.20 g of KCl, 1.78 g of Na2HPO4.2H2O, and 0.27 g of KH2PO4 in 1 l of distilled water and the pH was adjusted to 7.4), respectively, using a polytron homogenizer (Tri-R Stir-R homogenizer, Tri-R Instruments, Inc., Rockville Centre, New York, USA.). Aliquots were prepared and stored at −80°C until the determination of the MDA and GSH activities.
Ca, P, ALP, GSH, TNF-α, and CRP were detected according to the manufacturers’ guidelines using commercially available kits. The bone and bone marrow MDA, the index of lipid peroxidation, was determined using 1,1,3,3-tetramethoxypropane as standard. 22
Histological and immunohistochemical study
The excised parts of the left femurs were cut transversely at their upper ends (to check cancellous bone condition) and their shafts (to check compact bone condition). Fresh small specimens were fixed in 10% neutral formal saline for 48 h and then decalcified in daily exchanges using the chelating agent formalin-EDTA (disodium salt) for 4 weeks. An ample volume of decalcifying solution should be used, at least 30–50 times the volume of tissue. 23 The decalcified specimens were dehydrated in ascending grades of alcohol, cleared in xylene, and impregnated in paraplast carefully for 3 h in an oven at 58°C. Serial sections of the bone tissue were cut at a thickness of 6–8 μm and stained with hematoxylin and eosin (H&E) for studying the general histological structure. Additional slides were preceded for an immunohistochemical study. Immunohistochemical staining was performed using CD68 monoclonal rabbit antibodies (Lab Vision Laboratories, USA) according to the manufacturer’s protocol. The slides were then counterstained, dehydrated, and mounted. 24 The positive control for the anti-CD68 antibody was human tonsil. For the negative control slides, the same steps were followed, but without using the primary antibody.
After the bone marrow aspiration, smears were prepared as soon as possible, collected on a glass slide, quickly dried, and were stained with Leishman stain.
Photography
Slides were photographed using an Olympus digital camera connected to an Olympus, made in China (U.TV0.5XC-3) light microscope at the Histology Department, Faculty of Medicine, Minia University. Images were processed using Adobe Photoshop 7.
Morphometrical study
Quantitative data were collected for five parameters:
Parameters were measured in five fields from three serial sections of each rat. Measurements were taken using the image analyzer system in the Faculty of Dentistry, Minia University. Image J program analysis software (version 1.5) was used to measure cortical bone thickness in μm, which was measured by drawing a vertical line from just beneath the periosteum to the endosteum; trabecular bone thickness in μm, bone trabeculae were measured at their midpoint away from their branching areas; mean diameter of Haversian canals from each H&E section at magnification ×400; mean number of osteoclast and bone marrow macrophage CD68 immunoreactive cells at magnification ×400; and eroded surface percentage, which represented the percentage of eroded parts of the trabecular surface by the following equation
Statistical analysis
Results were expressed as means ± standard error of mean. One-way analysis of variance (ANOVA) followed by the Tukey’s post analysis test was used to analyze the results for a statistically significant difference. p Values less than 0.05 were considered significant. GraphPad Prism was used for statistical calculations (version 5 for Windows, Graphpad Software, San Diego, California, USA, http://www.graphpad.com).
Results
Effect of BTX and rivastigmine on biochemical parameters
As shown in Table 1, there was a significant decrease in serum Ca level and a significant increase in the serum levels of P and ALP in BTX intoxicated group in comparison with normal control and rivastigmine groups. Meanwhile, the combination of BTX and rivastigmine treated rats had a significantly increased serum level of Ca and decreased serum levels of P and ALP when compared to BTX intoxicated group.
Effect of BTX-A and rivastigmine on Ca, P, and ALP serum levels in different rat groups.a

Ca: calcium; P: phosphorus; ALP: alkaline phosphatase; and BTX-A: botulinum toxin-A.
Group I: normal control group; group II: rivastigmine group; group III: toxic group; group IV: treated group.
a Results presented as mean ± SE (n = 6).
b Significant (p < 0.05) difference from normal control group.
c Significant (p < 0.05) difference from the treated group.
d Significant (p < 0.05) difference from the toxic group.
BTX caused a significant increase in the levels of MDA and a significant decrease in the levels of GSH in the bone and bone marrow as compared to the control and rivastigmine groups. On the other hand, a significant decrease in the levels of MDA with a significant increase in the levels of GSH in the bone and bone marrow were recorded in the treated group when compared to the toxic group (Table 2).
Effect of BTX-A and rivastigmine on bone and bone marrow MDA and GSH levels in different rat groups.

MDA: malondialdehyde; GSH: reduced glutathione; and BTX-A: botulinum toxin-A.
Group I: normal control group; group II: rivastigmine group; group III: toxic group; group IV: treated group.
a Results presented as mean ± SE (n = 6).
b Significant (p < 0.05) difference from normal control group.
c Significant (p < 0.05) difference from the treated group.
d Significant (p < 0.05) difference from the toxic group.
As shown in Table 3, there was a significant increase in the serum levels of CRP and TNF-α in the toxic group when compared to the control and rivastigmine groups. Combination of BTX and rivastigmine treated rats had a significantly decreased serum levels of CRP and TNF-α when compared to BTX-intoxicated group.
Effect of BTX and rivastigmine on CRP and TNF-α in different rat groups.a

CRP: C-reactive protein; TNF-α: tumor necrosis factor α; and BTX: botulinum toxin.
Group I: normal control group; group II: rivastigmine group; group III: toxic group; group IV: treated group.
a Results presented as mean ± SE (n = 6).
b Significant (p < 0.05) difference from normal control group.
c Significant (p < 0.05) difference from the treated group.
d Significant (p < 0.05) difference from the toxic group.
Effect of BTX and rivastigmine on histopathological changes
Histological study of femur rat diaphysis using H&E
The compact bone of group I and group II, respectively, was covered by periosteum and lined by endosteum. The compact bone was formed of Haversian canals arranged in regular concentric lamella with interfering osteocytes lying inside their lacunae. Nuclei of osteocytes were observed as densely stained basophilic dots. Regular continuous basophilic lines (cement lines) were clearly noticed (Figure 1). Group III recorded an apparent increase in periosteum thickness as compared with that of group I. In most sections, distorted (Haversian canal and bone lamellae) and ill-defined cement lines were seen. Furthermore, multiple scattered less acidophilic staining areas were clearly noticed in the bone matrix. Multiple eroded irregular areas were frequently seen on the endosteal surface and in the bone matrix. Regarding the bone cells, osteocytes were completely absent in some sections and others were degenerated, which led to empty lacunae, while others appeared with dark nuclei. Osteoclasts were frequently seen housed within erosion cavities which and appeared as large irregular cells with eosinophilic cytoplasm (Figure 2). In group IV, the compact bone had restored the normal arrangement of the Haversian system. Less apparent distortion and less eroded cavities were observed. In most sections, the bone matrix appeared homogenously eosinophilic. The outer border of the cortical bone showed many cement lines. Numerous regularly arranged osteocytes were seen within their lacunae. Little osteocytes appeared with a dark nucleus. The periosteum still showed some degree of thickening (Figure 3).

Photomicrographs of the transverse sections of rat femoral diaphysis of group I (a and c) and group II (b and d) stained by H&E showing the same histological structure of the compact bone with regularly arranged multiple bone lamellae arranged in a regular concentric manner. The bone matrix appears homogenously eosinophilic. The outer and inner bone surfaces are covered with periosteum (P) and endosteum (E), respectively. Notice the Haversian system (H) containing blood vessels (V) and multiple distinct cement lines (L). The osteocytes (black arrows) settle inside their lacunae. (Magnification for (a) and (b) is ×100 and for (c) and (d) ×400). H&E: hematoxylin and eosin.

Photomicrographs of the transverse sections of rat femoral diaphysis of group III stained by H&E showing (a) thickened periosteal layer (P). Notice the massive distortion (D) and faintly stained bone matrix (blue arrows). (b) Distorted Haversian system (H). Some osteocytes appear with dark nuclei (dark arrows). (c) Eroded irregular endosteal bone surface (E) and cement lines (L) can be hardly seen. Most osteocytes are degenerated leaving wide empty lacunae (U). (d) Resorption cavities (C) and multiple vacuoles (v) and within the bone matrix. The inset showing higher magnification of erosion cavity (at the endosteal surface of the cortical bone. These cavities lined with abundant osteoclast cells (blue arrow). (Magnification for (a) ×100 and (b and c) ×400 and for insets ×1000). H&E: hematoxylin and eosin.

Photomicrographs of the transverse sections of rat femoral diaphysis of group III stained by H&E showing (a) relatively thick periosteal layer (P) but with less apparent distortion of bone matrix (D). Notice the cement line (white arrow) under the periosteum. Notice the homogenously eosinophilic bone matrix. (b) Most osteocytes lie inside their intact lacuna (dash black arrows). Few osteocytes appear with dark nuclei (dark arrow). Less eroded irregular endosteal bone surface (E) is observed. The Haversian system (H) and multiple cement lines start to be more distinct (L). Less apparent resorption cavities (C) lying within the bone matrix can be noticed. (Magnification for (a) ×100 and (b) ×400). H&E: hematoxylin and eosin.
Histological study of rat femur metaphysis using H&E
The cancellous bone of group I and group II revealed multiple networks of thick anastomosing bone trabeculae enclosing red bone marrow cavities of variable sizes containing a wide variety of bone marrow cells. The bone matrix appeared homogenously eosinophilic. Group III showed evident histopathological changes: the bone trabeculae was arranged sparsely and disorderly and became fewer, thinner, and discontinuous when compared with that of group I and group II. Multiple endosteal eroded surfaces could be seen. Additionally, a generalized faintly stained bone trabecula was also observed. The intertrabecular spaces were obviously wide and were filled with necrotic hypocellular bone marrow containing numerous adipocytes. In group IV, the trabecular connections were clearly enhanced and apparently seen thicker and arranged more regularly. But, few trabeculae still showed interrupted areas. Less widening of bone marrow with more obvious bone marrow cells could be seen (Figure 4).
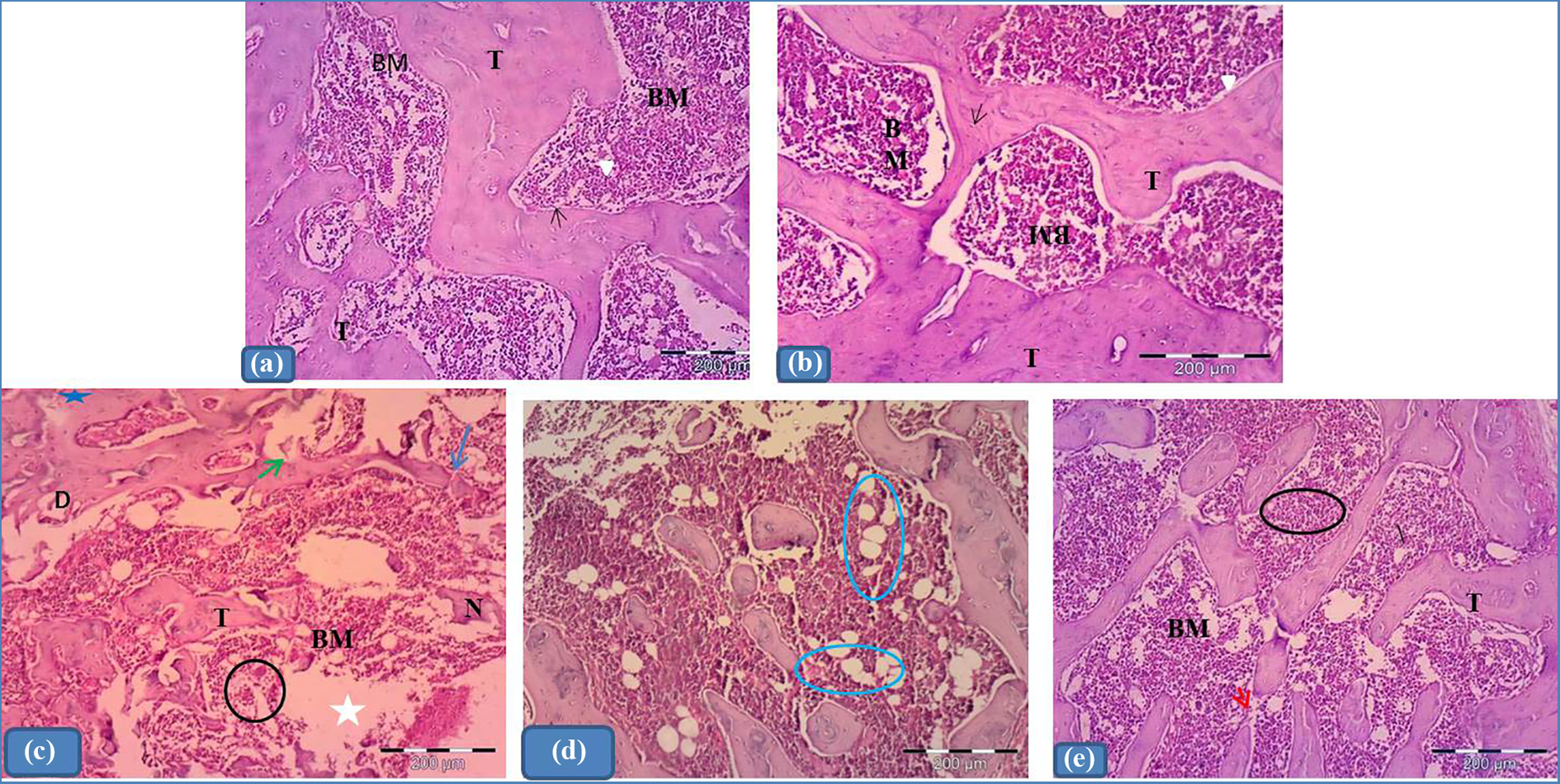
Photomicrographs of transverse sections of rat’s femoral metaphysis stained by H&E. (a and b) Groups I and II respectively showing cancellous bone with multiple network of thick branching and anastomosing bone trabeculae (T) enclosing red bone marrow cavities (BM) of variable sizes. Bone matrix appears homogenously eosinophilic. Bone trabeculae (T) appear with smooth inner surfaces. Many osteocytes reside in their lacunae (black arrow) within the trabeculae. The endosteal surface is smooth (arrow head). (c) Group III revealing distorted (D) cancellous bone with localized faintly stained areas (blue star). Notice the apparent fewer, thinner (T), and interrupted (blue arrow) bone trabeculae with apparent few osteocytes. Notice also the eroded surface of bone trabeculae (green arrow). Apparent hypocellular (black circle), wide (BM), focal loss and necrotic (white star) bone marrow are shown. (d) More obvious fat cells replacing bone marrow hematopoietic cells are clearly noticed (circles). (e) Group IV showing apparent less distortion of bone trabeculae. These trabeculae more or less regain their original morphology: they became apparently more thick and more continuous than those of group III (T). Less widening of the bone marrow can also be seen (BM) with more obvious bone marrow cells (circles). Few trabeculae still showed an interrupted area (red arrow). (Magnification ×100).
Effect of BTX and rivastigmine on immunohistochemical study
Immunohistochemical study of the rat femoral diaphysis and bone marrow using anti-CD68 antibody
CD68 was used as a marker of macrophage and osteoclastic activity. Positive control slides were prepared from human tonsil. The positive reaction of CD68 appeared in the form of cytoplasmic brown staining in tonsillar macrophages. For the negative control, slides appeared with a negative reaction. Groups I and II showed cytoplasmic expression in the osteoclast cluster cells and in the bone marrow macrophages. In group III, the bone sections showed an apparent intense positive cytoplasmic expression in more numerous enlarged osteoclast and bone marrow macrophages when compared to groups I and II. Group IV showed an apparent decrease in the CD68 cytoplasmic expression in less numerous osteoclast and bone marrow macrophages when compared with group III. (Figures 5 and 6).

(a) Positive control tonsillar tissue showing positive immune reaction for CD68 (arrows). (b) Negative reaction in a negative control slide for CD68. (c) to (f) Photomicrographs of transverse sections of rat femoral diaphysis for CD68 reactivity. (c) and (d) Groups I and II respectively showing faint ostoclastic cytoplasmic immunoreactivity. (e) Group III showing high (arrows) positive cytoplasmic expression apparent in more numerous and more enlarged macrophages related to the bone cavities. (f) Group IV showing apparent decrease in the CD68 cytoplasmic expression with less numerous osteoclast cells (arrows). (Immunohistochemistry for CD68 counter stain H ×400).

Photomicrographs of bone marrow of rat femoral metaphysic for the CD68 reactivity. (a) and (b) Groups I and II respectively showing cytoplasmic immunoreactivity in bone marrow macrophage. (c) Group III showing intensive (arrows) positive cytoplasmic expression apparent in diffused distributed macrophages. (d) Group IV showing low cytoplasmic expression in the apparent fewer bone marrow macrophages (arrows). (Magnification ×400).
Bone marrow smears of rat femoral diaphysis stained by Leishman stain
Groups I and II showed different bone marrow cells with fat cells. Group III showed an apparent increase in the fat cells to become more apparent than the bone marrow cells when compared with groups I and II. Group IV showed apparently less proportion of fat cells and more bone marrow cells when compared with group III (Figure 7). Morphmetrical and statistical results: Group III showed a significant decrease in Mean (outer cortical & trabecular bone thickness) while, there was a significant increase in other parameters if compared to group I, II and IV. (Significant P value < 0.05) (Table 4, Figures 8 and 9).

Photomicrographs of bone marrow smear of rat femoral diaphysis stained by Leishman stain. (a) and (b) Groups I and II respectively showing different bone marrow cells (circle) with fat cells. (c) Group III showing apparent increase in fat cells (F) with apparent decrease in hemopiotic cells (circle). (d) Group IV showing more obvious hemopiotic cells (circle) with less fat cells (F) than group III. (Magnification ×400).
Morphometric results of the femur bone in the studied groups.a

Group I: normal control group; group II: rivastigmine group; group III: toxic group; group IV: treated group.
a Results presented as mean ± SE (n = 6).
b Significant (p < 0.05) difference from normal control group.
c Significant (p < 0.05) difference from the treated group.
d Significant (p < 0.05) difference from the toxic group.
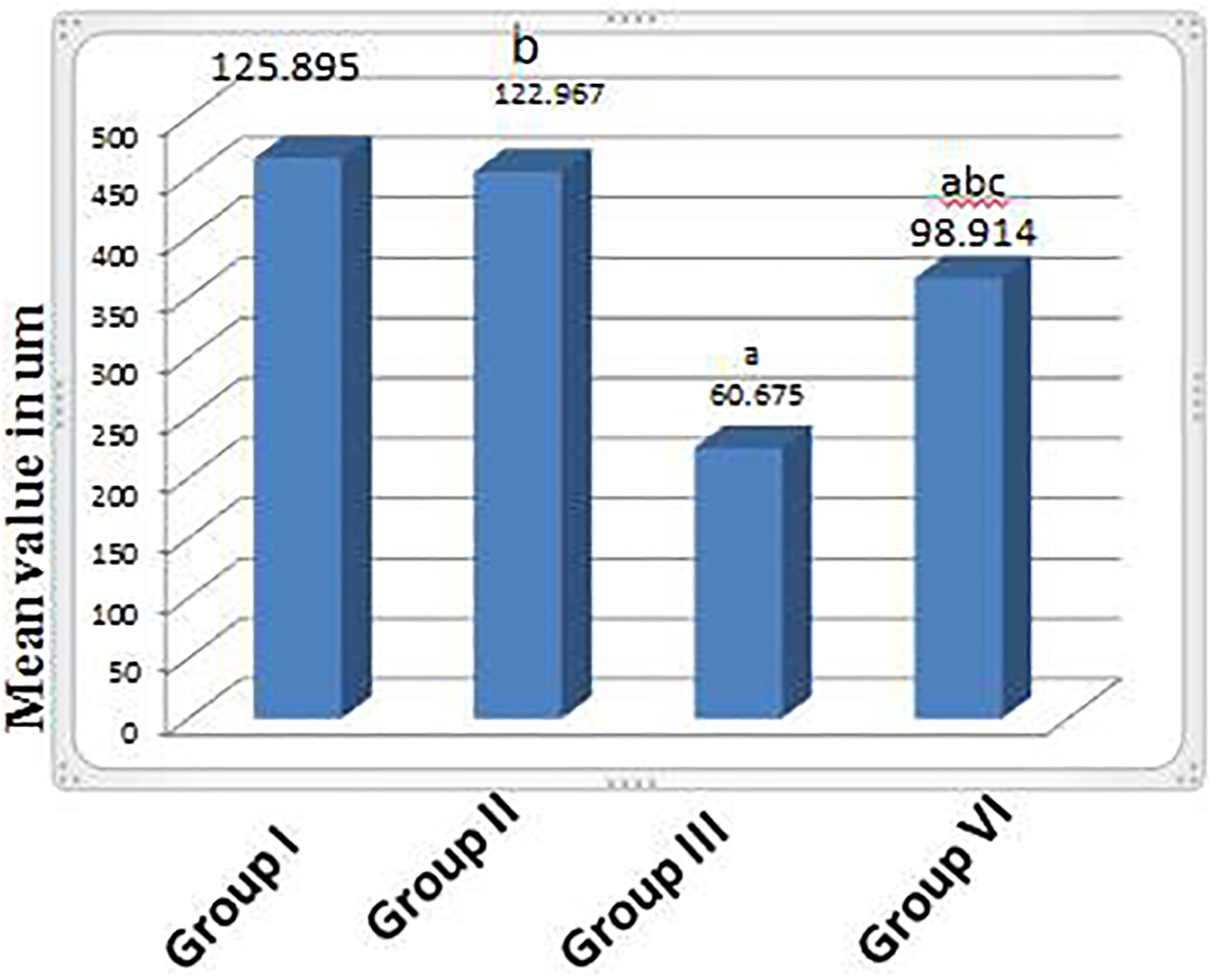
Histogram showing the mean cortical bone thickness in the different groups. Results represent the mean ± SE (n = 6). aSignificant (p < 0.05) difference from the control group, bsignificant (p < 0.05) difference from the toxic group, and csignificant (p < 0.05) difference from the treated group.

Histogram showing the mean trabecular bone thickness in the different groups. Results represent the mean ± SE (n = 6). aSignificant (p < 0.05) difference from the control group, bsignificant (p < 0.05) difference from the toxic group, and csignificant (p < 0.05) difference from the treated group.
Discussion
The use of BTX-A for therapeutic and plastic indications has been broadly accepted by physicians due to its safety and efficacy. 25 The present study was designed to determine the effect of BTX-A on the femoral bone and bone marrow of the rats. In this study, the biochemical analysis revealed that BTX-A reduced the serum Ca level and significantly increased the serum P level in the toxic group when compared to the control group. Ca plays an important role in the pathogenesis of osteoporosis and bone turnover. Reduction in the Ca level results in impaired bone mineralization. 26
A significant increase in the serum level of ALP was noticed as well in the toxic group when compared to the control rats. ALP is the bone turnover marker. It has been reported that BTX-induced immobilization led to the loss of bone strength in experimental animals. 27 Our results are in accordance with Unis et al., 28 who revealed that an increase in bone resorption more than bone formation was reflected by the increase in serum ALP. However, this is in contrast to Brüel et al., 27 who revealed an insignificant effect of BTX on serum ALP. On the other hand, Thomsen et al. 21 showed a significant reduction in the serum ALP level in the first week, which then returned to the control level after 4 weeks. However, their results were not reliable because a part of the rats did not undergo complete immobilization that led to dilution of the immobilization effect on the biochemical marker.
In addition, our results showed a significant increase in the MDA level and a significant decrease in the GSH level in the bone and bone marrow when compared to the normal control group. Our data suggested that BTX-A induced an oxidative stress which has a vital role in the pathogenesis of osteoporosis because the high level of osteoclastic activity and the reduction of osteoblastic activity are related to a disturbance between the oxidant and antioxidant levels. 29 Furthermore, previous studies revealed that reactive oxygen species increase the osteoclasts production and activity leading to higher bone resorption. 30,31 Moreover, it has been reported that MDA has an osteoclastic activity and is a lipid peroxidation indicator. 32
These results are in agreement with Unis et al. 28 ; however, Breikaa et al. 13 disagree with our data: they showed no change in the MDA level and a reduction in the GSH level, while the total antioxidant capacity showed insignificant effect of the BTX injection in rat testis. This may be attributed to the usage of different doses and the longer duration after the last injection (5 weeks).
In the present study, there was a significant increase in the serum levels of the inflammatory markers CRP and TNF-α in the toxic group compared to the control group which indicated that BTX provoked the inflammatory response. Similar results were reported by Breikaa et al. 13
The histopathological results corroborated all the previous biochemical parameters. This study showed various morphological changes in the bone tissue (compact and spongy) after the BTX-A injection. There was a significant increase in periosteum thickness; this might be explained as a compensatory mechanism to maintain the bone strength in case of bone loss. 33 Evidence of scattered irregular bone resorption cavities containing multiple osteoclasts and a remarkable empty lacunae were noticed clearly in most sections. Tsazawa et al. 34 and Warnera et al. 35 speculated that to be an evidence of osteocyte lysis. It is also in line with Takata and Yasui 36 who suggested that BTX disuse was associated with an unbalanced bone remodeling due to an increase in the bone resorption and a decrease in the bone formation. In this study, a significant widening of the Haversian canal was clearly detected, and it is in the same line with Dutra et al., 37 who suggested that the injection of BTX in the masseter muscle leads to decreased mineralization and matrix deposition with a decrease in the osteoclastic activity.
The assessment of multiple histological bone marrow sections provides great information on the tissue architecture and hematopoietic status that is relevant to the detection of direct or indirect responses to BTX exposure. Our results showed an apparent decrease in the bone marrow cellularity with an apparent increase in adiposity. This was explained by the switch in the differentiation of stromal cells from osteoblast cells to adipocytes with BTX injection, resulting in decreased bone formation. 38 Other investigators believed that it was the result of the lack of markers necessary for osteoblastic differentiation, leading to adipogenic differentiation in the case of BTX abuse. 39
In the present study, the toxic group showed an intense positive cytoplasmic expression in osteoclast and bone marrow macrophage when compared with the control and rivastigmine groups. It is in accordance with Tang et al. 40 who stated that osteoclast became more activated in osteoporosis and its activity clearly exceeded the osteoblast activity.
Rivastigmine was found to recover and alleviate the osteoporotic process induced by BTX-A in our study. In the group treated with rivastigmine, there was a significant improvement in the serum levels of Ca, P, ALP, and inflammatory markers (CRP and TNF-α) when compared to the toxic group. Moreover, rivastigmine significantly improved the oxidative stress caused by BTX-A in the bone and bone marrow.
Schiavo et al. 41 and Wilkinson 42 suggested that BTX inhibits the release of ACh in response to nerve impulses. Therefore, rivastigmine, which maintains a high level of ACh, can contribute to bone protection against the BTX-induced osteoporotic changes. The osteoprotective effect of rivastigmine was confirmed in many previous researches. 16,43
Histologically, rivastigmine administration, in group IV, ameliorated most of the osteoporotic changes induced by the BTX injection. The compact and trabecular bone almost regained their uniform thickness and diameter. This was confirmed by the histomorphometric study, which showed a significant increase in both cortical and trabecular bone thickness when compared to the toxic group. Rivastigmine treatment following BTX also significantly ameliorated bone microstructure, where the examined bones almost retained their normal architecture except for a few small resorption cavities. This highlights the great protective role of rivastigmine.
There is a clear evidence that a high level of ACh with the use of rivastigmine would positively modulate the glutamatergic function in animal models. 44 Furthermore, Szczesniak et al. 45 suggested that glutamate receptor activation regulates osteoblast and osteoclast differentiation and activity.
Administration of rivastigmine apparently decreased the immunoactivity for CD68 both in osteoclast and bone marrow macrophage; which is a macrophage marker. This might be explained to be an indicator of a decrease in osteoclastic activity with a consequent decrease in bone eating that preserves the normal level of bone remodeling with normal balance between bone eating and bone formation. This finding is in line with Tamimi et al., 43 who reported that using rivastigmine could enhance the activity of bone-regulating neurons that have a good beneficial effect on bone strength with a decreased risk of hip fracture in Alzheimer’s patients. Inderjeeth et al. 16 revealed that AChEI, such as rivastigmine, bound to muscarinic as well as nicotinic receptors may have an extra advantage of decreasing osteoclastogenesis.
Conclusion
The present study revealed that rivastigmine treatment had a protective effect against a single dose of the BTX-A-induced osteoporosis and bone marrow toxicity in the examined rats 4 weeks after injection and relieved the oxidative stress and inflammatory response. Hence, many patients who are in need of frequent BTX injection may benefit from the treatment with rivastigmine.
Footnotes
Acknowledgements
The authors would like to thank Dr Saher Sayed Ali Mohammed, Lecturer Oral Biology, Oral Biology Department, Faculty of Dentistry, Minia University, Egypt, for her kind help and support in the statistical analysis of bone morphometric measurements. The authors also thank Dr Rehab Ahmed Rifaai, Assistant Professor, Histology Department, Faculty of Medicine, Minia University, Egypt, for her efficient assistance and great effort throughout the processing stages of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.