Abstract
Aromatic extracts (AEs; distillate AEs [DAEs] and residual AEs [RAEs]) are complex, highly viscous liquid petroleum streams with variable compositions derived by extraction of aromatic compounds from distillate and residual petroleum fractions from a vacuum distillation tower, respectively. The DAEs generally contain significant amounts of polycyclic aromatic compounds (PACs) and are carcinogenic. The RAEs typically contain lower concentrations of biologically active PACs. The PACs in refinery streams can cause effects in repeated-dose and developmental toxicity studies. In a 13-week dermal study, light paraffinic DAE had several dose-related effects involving multiple organs; no-observed-effect level was <5 mg/kg/d, with no overt toxicity. Predicted dose-responses at 10% (PDR10s), modeled doses causing a 10% effect on sensitive end points based on PAC content, ranged from 25 to 78 mg/kg/d for untested paraffinic DAEs. The no observed adverse effect level (NOAEL) for developmental toxicity for light paraffinic DAE was 5 mg/kg/d. Statistically significant developmental effects at higher doses were associated with maternal effects. The PDR10s for developmental toxicity of paraffinic DAEs ranged from 7 to >2000 mg/kg/d, reflecting differences due to variation in PACs. The NOAELs for RAEs were 500 mg/kg for 90-day studies and 2000 mg/kg for developmental toxicity. Reproductive toxicity is not considered to be a sensitive end point for AEs based on the toxicity tests with DAEs, RAEs, and other PAC-containing petroleum substances. In vivo micronucleus tests on heavy paraffinic DAE, RAEs, and a range of other petroleum substances have been negative. The exception to this general trend was a marginally positive response with light paraffinic DAE. Most DAEs are considered unlikely to produce chromosomal effects in vivo.
Introduction
In response to the High Production Volume (HPV) Challenge Program of the United States Environmental Protection Agency (US EPA), 1 approximately 400 petroleum substances were sponsored by companies belonging to the Petroleum HPV Testing Group. These substances were organized into 13 categories, one of which was aromatic extracts (AEs). This article reports previously unpublished data on toxicological hazards with repeated exposures to AEs and compares those data to published information on this category.
Aromatic extracts are complex, highly viscous liquids that are produced during the refining of crude oil to produce lubricating oil basestocks (LOBs) and waxes. In this process, the atmospheric residue (residuum) remaining from atmospheric distillation of crude oil is further distilled in a vacuum tower to produce vacuum distillate and residual fractions. These refinery streams can be further refined to make distillate LOBs and residual LOBs, respectively. In that process, undesirable components that negatively impact lubricant performance must be removed. These undesirable components include heterocyclic aromatic compounds and polycyclic aromatic compounds (PACs). One way in which these undesirable components can be removed is extraction of aromatic compounds, resulting in an AE that contains the aromatic compounds extracted from the LOB or wax. There are 2 types of AEs. The first type, distillate AEs (DAEs), is produced by extraction of distillate fractions and the second type, residual AEs (RAEs), is produced by extraction of residual fractions. A schematic of these processes is given in Figure 1. Total PACs can be of the order of 65% to 85% for DAEs and 50% to 80% for RAEs. 2 Examples of the amount of dimethyl sulfoxide (DMSO)-extractable PACs, which are only a portion of total PACs, are given in Table 1 (discussed later). This DMSO fraction is thought to contain the toxicologically active PACs. Further details of the extraction process and a description of the characterization of DAEs as either light or heavy and either paraffinic or naphthenic are given in the Appendix.
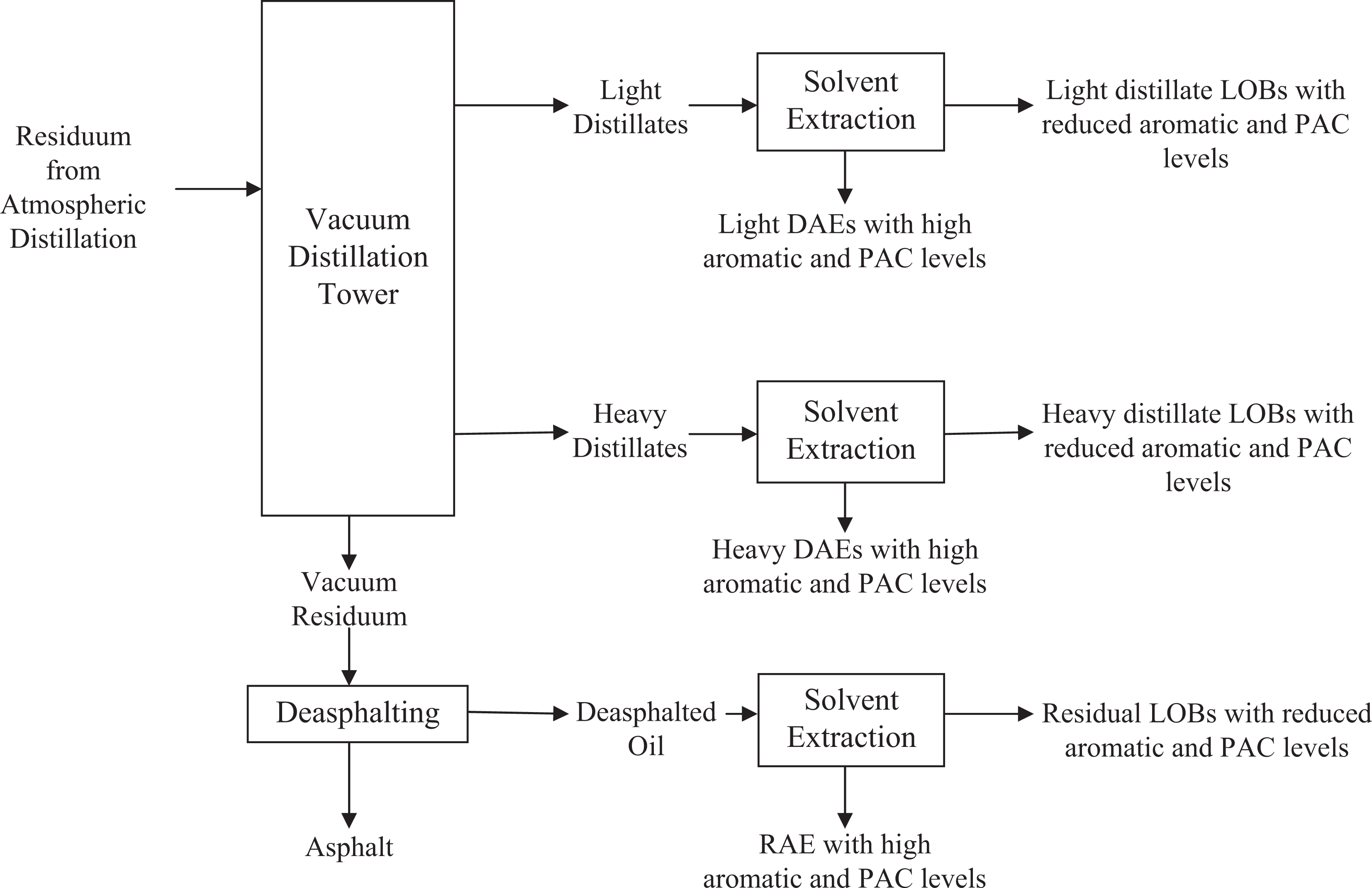
Example of general schematic of refining processes for production of distillate aromatic extracts (DAEs) and residual aromatic extracts (RAEs). The LOBs are lubricant oil basestocks (mineral oils) and PACs are polycyclic aromatic compounds.
ARC Content of Sampled Aromatic Extracts.
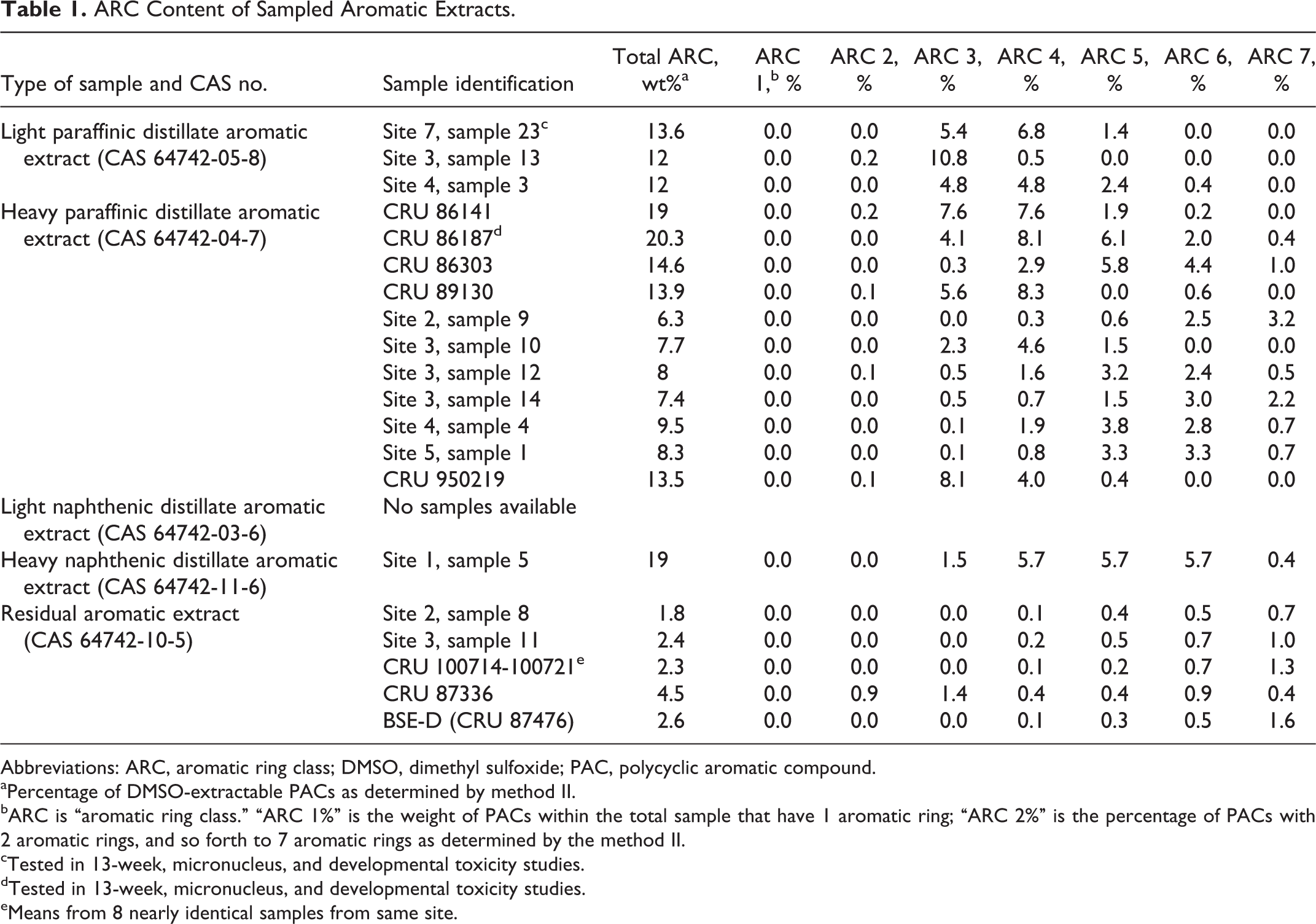
Abbreviations: ARC, aromatic ring class; DMSO, dimethyl sulfoxide; PAC, polycyclic aromatic compound.
aPercentage of DMSO-extractable PACs as determined by method II.
bARC is “aromatic ring class.” “ARC 1%” is the weight of PACs within the total sample that have 1 aromatic ring; “ARC 2%” is the percentage of PACs with 2 aromatic rings, and so forth to 7 aromatic rings as determined by the method II.
cTested in 13-week, micronucleus, and developmental toxicity studies.
dTested in 13-week, micronucleus, and developmental toxicity studies.
eMeans from 8 nearly identical samples from same site.
Toxicological research on AEs has focused on carcinogenicity. It is generally understood that the dermal carcinogenicity of many petroleum streams is related to their PAC content. Vacuum distillates can be made noncarcinogenic by extraction of aromatic components into the DAE. Therefore, DAEs are generally expected to contain relatively high amounts of PACs and to be carcinogenic. 12 -15 Vacuum residues are much less carcinogenic than distillates, 16 and RAEs are less carcinogenic than DAEs. 12 -14 The primary route of concern is dermal due to the high viscosity of AEs (limiting aerosolization) and the anticipated uses of these products in end uses in which dermal exposure is the most likely route of exposure.
Information on the nongenotoxic effects of repeated exposures to AEs has been more limited. No publications on the effects of repeated-dose or developmental toxicity studies with RAEs were located, but results from some dermal studies with DAEs have been published. More specifically, dermal dosing was used in a 13-week toxicity study on a heavy paraffinic DAE (sample 86187 in Table 1). Doses of 0, 30, 125, 500, and 1250 mg/kg/d were applied on Sprague-Dawley rats for 5 days/week. 17,18 The no-observed-effect level (NOEL) for systemic effects was <30 mg/kg/d based on the reduced body weight in females, increased relative liver weight, and decreased thymus weight at that dose. Additional effects, including lower red blood cell (RBC) number, hematocrit, and hemoglobin (Hb), were seen at doses ≥125 mg/kg/d.
In a more limited 28-day study (fewer animals dosed 3 times/week for 4 weeks), groups of New Zealand white rabbits received dermal applications of 0, 250, 500, and 1000 mg/kg of neat light paraffinic DAE. The only observed treatment-related effects were increased relative liver weights among females at all dose levels and dermal irritation. 12 The no observed adverse effect level (NOAEL) for systemic effects was 1000 mg/kg/d.
The results of 2 developmental toxicity studies with heavy paraffinic and heavy naphthenic DAEs were also available. In the first, the same undiluted heavy paraffinic DAE used in the 13-week study in rats was applied dermally to Sprague-Dawley rats. 18 Doses were 0, 8, 30, and 125 mg/kg/d on gestation days (GDs) 0 to 19, 500 mg/kg/d on GDs 0 to 16, and 1000 mg/kg/d on GDs 10 to 12. The test material was not removed between applications. Maternal effects at doses ≥125 mg/kg/d included decreases relative to controls in body weight, body weight gain, food consumption, gravid uterine weight, platelet count (500 mg/kg/d only), and thymus weight (absolute and relative), increases in relative liver weight and white blood cell (WBC) count, and dose-related changes in serum chemistry parameters. Developmental effects, again at ≥125 mg/kg/d, included reduced number of dams with viable fetuses, increased number of dams with resorptions, reduced litter size of viable fetuses, increased percentage of resorptions, and decreased fetal body weight. At 30 mg/kg/d, a 2-fold increase in percentage of resorptions was noted but was not statistically significant. No significant adverse maternal or fetal effects were seen at 8 mg/kg. Therefore, the lowest-observed-adverse-effect level (LOAEL) for statistically significant effects on fetuses was 125 mg/kg/d, with a corresponding NOAEL of 30 mg/kg/d. However, nonstatistically significant differences were seen at 30 mg/kg/d. Particularly given the gravity of a possible effect on fetal survival at 30 mg/kg/d, the LOAEL for possible biologically significant effects on fetuses is considered here to be 30 mg/kg/d, with a corresponding NOAEL of 8 mg/kg/d.
The second developmental toxicity study was a range-finding effort in which only 5 pregnant Fischer 344 rats were treated with each dose of a heavy naphthenic DAE. Doses were 0, 500, 1000, or 2000 mg/kg/d administered for 6 hours on GDs 7 to 16. The LOAEL for developmental toxicity was 2000 mg/kg/d based on the increased resorptions, a corresponding decrease in live implants, and reduced fetal and mean litter weights; the NOAEL was 1000 mg/kg/d. 19
Given that this previous work was performed with heavy DAEs (or a light paraffinic DAE in a limited study), a 13-week dermal toxicity study and a developmental toxicity study were conducted on a light paraffinic DAE in order to broaden the toxicological data available on AEs. Results of these studies are presented here. In addition to this work on DAEs, this article contains results of 13-week dermal toxicity studies on 4 RAEs and a developmental toxicity study on 1 RAE. Micronucleus assays were included as part of several of these subchronic studies to test for clastogenic activity and/or interference with spindle fibers during cell division and results of those tests are given here.
Although these data, combined with previously published results on DAEs, provide a picture of potential effects of dermal exposure to DAEs, the composition of DAEs could be variable enough that questions could arise on the possible effects of specific samples of DAE that have not been tested experimentally. For those DAEs, data on their PAC content can be used in statistical models to estimate developmental toxicity or systemic effects in repeated-dose studies. These models were based on compositional information and toxicity studies with many refinery streams and were developed after a relation between PACs and toxicity became apparent from toxicity studies on a range of heavy refinery streams. 20 -22 In essence, these models showed that the potential for systemic and developmental toxicity was associated with the aromatic ring class (ARC) profile of the types and levels of 1 to 7 ring PACs. The ARC profiles that were used for calculation of PDR10s for several samples of DAEs are in Table 1. The ARC profiles for samples of RAEs are also shown, although modeling was not done on RAEs for reasons that are discussed in the Appendix. These data came from 2 sets of analyses. The first set was obtained from company records. Those samples are identified as 5-digit “CRU” numbers, the tracking number used in the testing laboratory. The second set was comprised of samples sent to American Petroleum Institute by member companies for the purpose of providing a snapshot of AEs currently marketed in the United States. Those samples are identified by their location (site and sample numbers) or by a 6-digit CRU number. Results of modeling for DAEs are summarized in this article.
Finally, in addition to providing a more encompassing picture of the systemic and developmental toxicity of AEs, the studies presented here can be used to provide information on the potential reproductive toxicity of AEs. No published reproductive toxicity studies were identified for DAEs or RAE. However, under the HPV Challenge Program, the US EPA provided guidance on the requirements for evaluating reproductive toxicity. This EPA guidance indicates that a reproductive toxicity study may not be required for certain petroleum substances if there is (1) a 90-day repeat-dose study in which the potential for effects on reproductive organs was assessed and (2) a developmental toxicity study. 23 Such data are summarized here for light paraffinic DAE and RAE. Although results from studies on heavy paraffinic DAEs have been previously published, 18 the company that performed those studies provided final reports so that unpublished data from some aspects of the studies can be presented here.
Materials and Methods
Subchronic Dermal Studies
A 13-week dermal toxicity study was performed in Sprague-Dawley rats with light paraffinic DAE (sample identified as site# 7, sample# 23 in Table 1). The study was done in accordance with OECD TG 411, OCSPP Guideline 870.3250, and 40 CFR 798.2250. This and all other studies reported here were performed in accordance with Good Laboratory Practices. The DAE was mixed with acetone before dosing. Groups of 10 rats/sex received sham dosing (no vehicle), 1.5 mL/kg acetone (vehicle control), or diluted DAE at 5, 50, or 150 mg DAE/kg/d in a constant volume of 1.5 mL/kg. Dilution in acetone might have affected dermal absorption of PACs but absorption was not measured. Animals wore Elizabethan collars to minimize ingestion of the DAE. Doses were applied to clipped unabraded skin and spread evenly using a glass rod over ∼10% of the body surface area. Doses were given once daily on 5 days/week over 13 weeks. At 6 hours after each dose, all animals were gently wiped with a paper towel to remove unabsorbed DAE. At the end of each 5-day dosing period, residual material was removed using 1% Ivory liquid soap (Procter & Gamble, Cincinnati, Ohio) in warm tap water followed by a deionized water rinse and drying with a clean paper towel. No vehicle was applied to the sham control group, but the skins of the animals were wiped and rinsed using the same procedure as the other groups.
End points during the treatment period included daily assessment of survival and clinical signs and weekly measurement of body weight, food consumption, and dermal irritation using the Draize scale. End points at sacrifice included hematology, serum chemistry, organ weights (adrenals, brain, epididymides, heart, kidneys, liver, ovaries with oviducts, pituitary, prostate, spleen, testes, thymus, thyroid with parathyroids, and uterus), and histopathology on ∼41 organs. Each DAE-treated group was compared statistically to the vehicle control group by sex. Body weight, body weight change, food consumption, clinical pathology, and organ weight data were analyzed using a parametric 1-way analysis of variance (ANOVA) followed by Dunnett test if appropriate. Data from vehicle controls were compared to sham-dosed controls using the Student
In addition to the work on the light paraffinic DAE, 4 undiluted samples of RAE were administered dermally in Sprague-Dawley rats for 13 weeks. Samples were identified as BSE-A, BSE-B, BSE-C, and BSE-D (also identified as CRU 87476); BSE stands for bright stock extract, another name for RAE. The PAC data were available only for BSE-D (Table 1), but the other 3 BSEs can be expected to have a reasonably similar PAC content. The same procedures were used as in the published study with heavy paraffinic DAE 17,18 outlined in the introduction with the exception that doses of BSE-D were 500 and 2000 mg/kg/d while the only dose for the other 3 RAE samples was 2000 mg/kg/d. The study design was similar to OECD TG 411. Each group contained 10 rats/sex except that, because no sex difference was expected, only 10 females (no males) were treated with BSE-B and only 10 males (no females) were treated with BSE-C to reduce the total number of animals. Each RAE was applied undiluted to the shorn dorsal skin of the animals using a Tridak grease dispenser (Tridak LLC, Torrington, Connecticut) due to its high viscosity. Application was 5 days/week for 13 weeks. Treated sites were left uncovered and rats wore Elizabethan collars. On day 6 of each week, the skin of each animal was wiped to remove residual test material, and the collars were removed for the weekend. A sham-dosed control group received the same treatment except for administration of a RAE.
End points for the RAEs were the same as those with light paraffinic DAE except that food consumption was not measured, weight of pituitary and thyroid were not recorded, and there were a few differences in hematology, serum chemistry, and organs taken for histopathology. Urinalysis was performed with the RAEs. Also, the epididymides and testes from males given 0 mg/kg, 2000 mg BSE-D/kg, and 2000 mg BSE-A/kg were weighed. The testes were prepared for spermatid count and the epididymides were prepared for spermatozoa count 24 and morphological examination. 25 Quantitative data were analyzed statistically using ANOVA followed by Dunnett test or Tukey multiple range test.
Micronucleus Tests Associated With Subchronic Studies
A micronucleus assay was conducted in conjunction with the 13-week study on light paraffinic DAE and was done in accordance with OECD TG 474 and 40 CFR 798.5395. Bone marrow was collected from 5 animals/sex in each group at the time of euthanasia (approximately 24 hours after the last dose). Bone marrow was flushed from the right femur and bone marrow smears were prepared. The slides were stained with acridine orange. Three separate evaluations were made for each slide: (1) a total of 1000 erythrocytes (both polychromatic erythrocytes [PCEs, immature RBCs] and normochromatic erythrocytes [NCEs, mature RBCs]) per animal were counted and the ratio of PCE to total erythrocytes (TEs) was determined; (2) the number of micronucleated PCEs (MN PCEs) from a total of 2000 PCEs was scored per animal; and (3) the number of MN NCEs (MN NCEs) from a total of 2000 NCEs was scored per animal. A positive control group treated with a single dose of cyclophosphamide monohydrate was included. Statistical analysis of DAE-treated and vehicle controls involved a parametric 1-way ANOVA followed by Dunnett test if appropriate. In addition, the positive controls and sham-dosed controls were each separately compared to the vehicle controls using a separate parametric 1-way ANOVA.
Micronucleus assays were performed on bone marrow samples taken from femurs of 5 rats/sex in each of the groups treated for 13 weeks with RAEs and the controls at the time of sacrifice. The study design was equivalent to OECD TG 474. Three bone marrow slides were made per animal and 1000 PCEs and 1000 NCEs were scored to determine the percentage of micronucleated erythrocytes. The slides were stained with acridine orange and examined using fluorescence microscopy. Several statistical methods, including ANOVA and generalized linear model models, were used.
Results of the micronucleus test that accompanied the previously published subchronic study on heavy paraffinic DAE 18 were not previously published and therefore are included here. The study design was consistent with OECD TG 475. At the scheduled sacrifice femurs were taken from 5 rats/sex in controls and groups given 0, 30, or 125 mg/kg/d dermally. Samples were taken from females given 500 mg/kg/d dermally and from males given 125 or 500 mg/kg/d orally. The methods were the same as those used for the RAEs. Controls were sham-dosed and had Elizabethan collars; the orally dosed animals also had collars to help equalize treatment across the groups.
Developmental Toxicity With Dermal Dosing
In the dermal prenatal developmental toxicity study with light paraffinic DAE, untreated female Sprague-Dawley rats were paired with untreated males. The study was performed in accordance with OECD TG 414 and OCSPP 870.3700. The mated females (25/group) were divided into 6 treatment groups; doses administered to the groups were 0 (sham-dosed), 0 (vehicle controls), 5, 25, 150, and 450 mg/kg/d. The DAE (CAS No. 64742-05-8, identified as site #7, sample #23 in Table 1) was diluted in acetone and the resulting mixture was administered at a constant volume of 1.5 mL/kg. Doses were applied to the clipped backs of the animals once daily for ∼6 hours on GDs 0 to 19. Sites were not occluded and the animals wore Elizabethan collars. The site of application was scored daily for erythema, edema, and other dermal findings using the Draize scale.
Maternal body weights and food consumption were measured on GDs 0, 3, 6, 9, 12, 15, 18, and 20. All surviving females were euthanized on GD 20. Gravid uterine weight was measured and net body weight (body weight on GD 20 exclusive of the weight of the uterus and contents) and net body weight change (change in body weight from GDs 0 to 20 exclusive of the weight of the uterus and contents) were calculated. Specific end points during necropsy of females included the number of corpora lutea, uterine weight, number and location of all fetuses, early and late resorptions, and the total number and distribution of implantation sites. Uteri with no macroscopic evidence of implantation were opened and placed in 10% ammonium sulfide solution for detection of early implantation loss. 26 The placentae were examined. Liver, brain, and thymus were weighed and preserved together with treated skin and untreated skin.
Each viable fetus was examined externally, individually sexed, weighed, and euthanized. The external examination included the eyes, palate, and external orifices. Crown-rump length was measured. Each viable fetus was subjected to a visceral examination using a modification of the Stuckhardt and Poppe fresh dissection technique to include the heart and major blood vessels. 27 The sex of each fetus was confirmed by internal examination. Fetal kidneys were examined and graded for renal papillae development. 28 Heads from approximately one-half of the fetuses in each litter were placed in Bouin fixative for subsequent soft-tissue examination by the Wilson sectioning technique. 29 The heads from the remaining one-half of the fetuses were examined by a midcoronal slice. All carcasses were eviscerated and fixed in 100% ethyl alcohol. Following fixation in alcohol, each fetus was macerated in potassium hydroxide and stained with Alizarin Red S and Alcian Blue. External, visceral, and skeletal findings were recorded as developmental variations or malformations.
The fetal developmental findings were summarized by (1) presenting the incidence of a given finding both as the number of fetuses and the number of litters available for examination in the group and (2) considering the litter as the basic unit for comparison and calculating the number of affected fetuses in a litter on a proportional basis as follows: Summation per group (%) = Sum of viable fetuses affected per litter (%)/No. of litters per group, where viable fetuses affected per litter (%) = 100 × No. viable fetuses affected per litter/No. of viable fetuses per litter.
Maternal body weights, body weight changes, food consumption, organ weights, gravid uterine weights, fetal body weights, and numbers of corpora lutea, implantation sites, and viable fetuses were analyzed using ANOVA followed by Dunnett test or a 2-sample
The design of the developmental toxicity study on a RAE (sample BSE-D, CRU 87476) was similar to the published work on heavy paraffinic DAE 18 and did not differ substantially from that used with light paraffinic DAE although there were some differences in the methods used for fetal evaluations. The study design was similar to OECD TG 414 (Prenatal Developmental Toxicity Study) except that there were 15 animals per group rather than 20. The undiluted RAE was applied dermally to the clipped backs of pregnant Sprague-Dawley rats at doses of 0, 500, and 2000 mg/kg/d on days 0 to 19 of gestation. The RAE was heated to 35°C prior to each dosing to make it fluid enough to be drawn into a syringe, applied to the skin, and spread evenly with a spatula. The treated sites were not covered; rats wore Elizabethan collars that were replaced as necessary. The sham control animals were treated similarly, including stroking the skin with a spatula, but the RAE was not applied. In a separate group included for postnatal evaluations, the RAE was administered at 0 and 2000 mg/kg/d on GDs 0 to 19, and all females were sacrificed and examined grossly on postpartum day 4. Each presumed pregnant animal was observed at least once daily until sacrifice. Body weights of animals in the prenatal and postnatal groups were recorded on GDs 0, 3, 6, 10, 13, 16, and 20, and food consumption was determined every 3 days.
Each female rat of the prenatal group was sacrificed and necropsied on GD 20. Examinations included gravid uterine weight and the number of corpora lutea per ovary, implantations, early and late resorptions, and live and dead fetuses. Maternal blood samples were collected for measurement of alanine aminotransferase, albumin, alkaline phosphatase, aspartate aminotransferase, bilirubin, calcium, chloride, cholesterol, creatinine, glucose, iron, lactate dehydrogenase, phosphorus, potassium, sodium, sorbitol dehydrogenase, total protein, triglycerides, urea nitrogen, and uric acid. Each live fetus was gendered, weighed, and grossly examined for external abnormalities. After evaluation, approximately half the fetuses in each litter were randomly distributed into either soft tissue or skeletal groups. Those fetuses in the soft tissue group were fixed in Bouin, sectioned using a razor blade, and examined for abnormalities. 30 Fetuses assigned to the skeletal group had their soft tissues removed and their skeletons stained and evaluated for skeletal abnormalities. 31,32
In the postnatal groups, dams and offspring were weighed on days 0 and 4 after delivery but food consumption was not measured. Pups were examined for external malformations and variations on postpartum day 0. Dams and their litters were observed on postpartum days 0 through 4, including the presence of milk in pups’ stomachs. All animals were sacrificed on postpartum day 4. The thoracic and abdominal cavities of dams were examined grossly, and each uterus was examined for the total number of implantations.
Data from the maternal biophase, Cesarean sections, and fetuses in the prenatal groups and data during gestation in the postnatal groups were evaluated statistically by ANOVA followed by group comparisons using Fisher exact or Dunnett test. Data on serum chemistry were analyzed using ANOVA followed by the Student-Newman-Keul multiple comparison test. Fetal skeletal and visceral data in the prenatal groups were analyzed by ANOVA followed by Fisher exact test.
Modeled Predictions of Toxicity
The PAC constituents of the DMSO extracts of 15 DAEs and 5 RAEs were analyzed by method II as described previously. 5,33,34 Samples were extracted with DMSO, further purified, and then analyzed by gas chromatography–mass spectrometry (GC–MS). The GC/MS chromatograms of the extracts were integrated in the slice mode. Data were reported as the ARC profile, the percentage of each ring number of the PACs (1-ring, 2-ring, etc) in the original sample (Table 1).
The ARC profiles of DAEs were used to predict the doses associated with a 10% change in each of the 4 indicators of systemic effects, namely, increased liver weights, decreased thymus weights, decreased platelet counts, and decreased Hb concentration. Models were also used to predict 10% changes in developmental parameters including reductions in fetal body weight, reductions in percentage of offspring born alive, and increased percentage of resorptions. The predictions were based on a series of statistically developed empirical models described elsewhere, 21 and the indicators of effect were identified in an extensive analysis of data from several studies on petroleum streams as the most sensitive end points. 20,22 The dose associated with a 10% difference from controls for each indicator of effect was identified as the “predicted dose-response at 10%” (PDR10), which is similar in concept to the benchmark dose at 10% (BMD10) 35 in that both represent a dose that is related to a 10% difference in response from controls for a particular end point. However, the BMD10 is calculated directly from existing dose–esponse data while the PDR10 is a calculated estimation of the dose-response. The PDR10 is based on many sets of data sets. In other words, the BMD10 is a means of expressing what happened in an individual experiment and the PDR10 is an estimate of what is predicted if an experiment were conducted. General agreement between the BMD10s and PDR10s for a specific sample provides support for the recently developed models for calculating PDR10s. The lowest PDR10 in each study for each sample was identified as the “sample PDR10” for that study. The BMD10s were calculated for the indicators of effect in the 13-week and developmental toxicity studies with both tested samples of DAE (heavy paraffinic and light paraffinic DAEs). The lowest BMD10 for each study was identified as the “study BMD10.”
Reproductive Toxicity
The results of a 90-day repeated-dose study and a developmental toxicity study on heavy paraffinic DAE have been published 18 but further previously unpublished details are provided here on the weights and histopathology specifically for reproductive organs in those studies. Similar information is also given here from the studies with light paraffinic DAE and with multiple samples of RAEs.
Results
Subchronic Dermal Dosing
All animals survived to the end of the study with light paraffinic DAE except for 1 male at 150 mg/kg/d; however, the cause of death could not be determined. No differences were seen between vehicle controls and treated groups in clinical signs, dermal irritation, food consumption, macroscopic effects, or microscopic findings. Gains in body weight were lower in females treated with 150 mg/kg/d, resulting in mean final body weights that were 8.1% lower than vehicle controls (Table 2), which was not a statistically significant difference.
Selected End Points in Hematology, Clinical Chemistry, and Organ Weights for Groups Treated Dermally with Light Paraffinic DAE.a
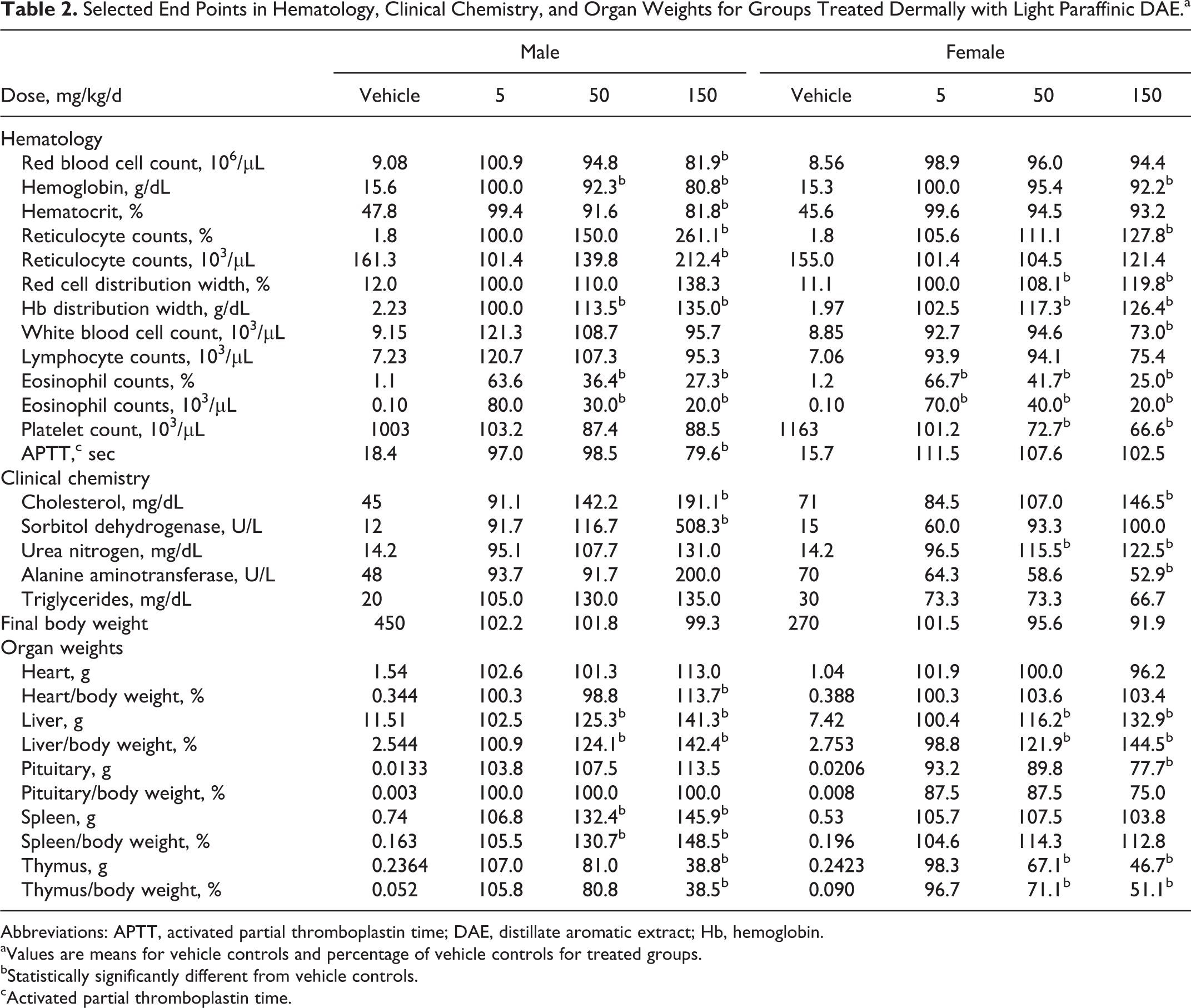
Abbreviations: APTT, activated partial thromboplastin time; DAE, distillate aromatic extract; Hb, hemoglobin.
aValues are means for vehicle controls and percentage of vehicle controls for treated groups.
bStatistically significantly different from vehicle controls.
cActivated partial thromboplastin time.
Statistically significant differences, summarized in Table 2, were seen between treated groups and the vehicle controls for some end points in hematology, serum chemistry, and organ weights. Among hematological end points, higher values were seen relative to vehicle controls in the number of reticulocytes, RBC distribution width (a measure of the variation in width of RBCs), and Hb distribution width (a measure of the variation in concentration of Hb in RBCs). Lower values were seen in RBC count, Hb, hematocrit, WBCs in females, platelet count, and activated partial thromboplastin time (males). Most notably, lower eosinophil counts (percentage and absolute numbers) were seen at all doses. The multiple changes noted in the hematogram and leukogram were investigated by cytologic evaluation of the bone marrow slides collected at necropsy. No meaningful differences in cellular morphology or granulocytic or erythroid maturation were seen in any of the examined groups. The proportion of total erythroid precursors was slightly to minimally higher and the ME ratios were correspondingly slightly to minimally lower in males administered 50 mg/kg/d of test article, and in males and females administered 150 mg/kg/d of test article compared to the sham and vehicle controls. The proportions of total granulocytic precursors were slightly lower in the 50 and 150 mg/kg/d males compared to sham and vehicle controls. The slightly higher proportion of total erythroid cells in the bone marrow may correspond to the higher percentage and absolute reticulocyte counts in the peripheral blood in the 150 mg/kg/d males and minimally higher percentage reticulocyte counts in the 150 mg/kg/d females. The statistically significant lower RBC, WBC, and eosinophil counts with no concomitant change in bone marrow cellularity suggest loss of blood cells after maturation.
Among the clinical chemistry parameters shown in Table 2, higher cholesterol and urea nitrogen were consistent in both sexes at 150 mg/kg/d. Significant differences in absolute or relative weights of the heart and pituitary were seen. However, increased weight of the liver (both sexes) and spleen (males) and decreased weight of the thymus (both sexes) appeared to have been a more consistent finding and were more clearly dose related. No treatment-related microscopic findings were noted in any of the groups examined histologically. The overall conclusion of this study was that, although overt toxicity was not seen, a NOEL could not be established due to the lower eosinophil counts at 5, 50, and 150 mg/kg/d.
In the 13-week studies with RAEs, body weight, clinical signs, urinalysis, and gross observations at necropsy were not affected by treatment. In general, the test materials did not cause skin irritation. Several small changes in serum chemistry and hematology parameters were summarized in Table 3 and included lower RBC count, Hb, and hematocrit in females and higher sorbitol dehydrogenase in both sexes relative to sham-dosed controls. Spleen weight was significantly higher with 1 RAE and liver weight was significantly higher with 2 RAEs. Among the 4 RAEs, BSE-A had the most significant differences from the control group. Gross observations at necropsy were unremarkable and no treatment-related effects were seen with microscopic examinations. Epididymal spermatozoa morphology and count and testicular spermatid counts were unaffected by treatment with BSE-D or BSE-A at 2000 mg/kg. The NOAEL was 500 mg/kg/d for BSE-D and <2000 mg/kg/d for the other 3 RAEs.
Mean Values for Selected End Points in Hematology, Clinical Chemistry, and Organ Weights for Groups Treated Dermally With RAEs, Shown as Percentage of Sham-Dosed Controls.
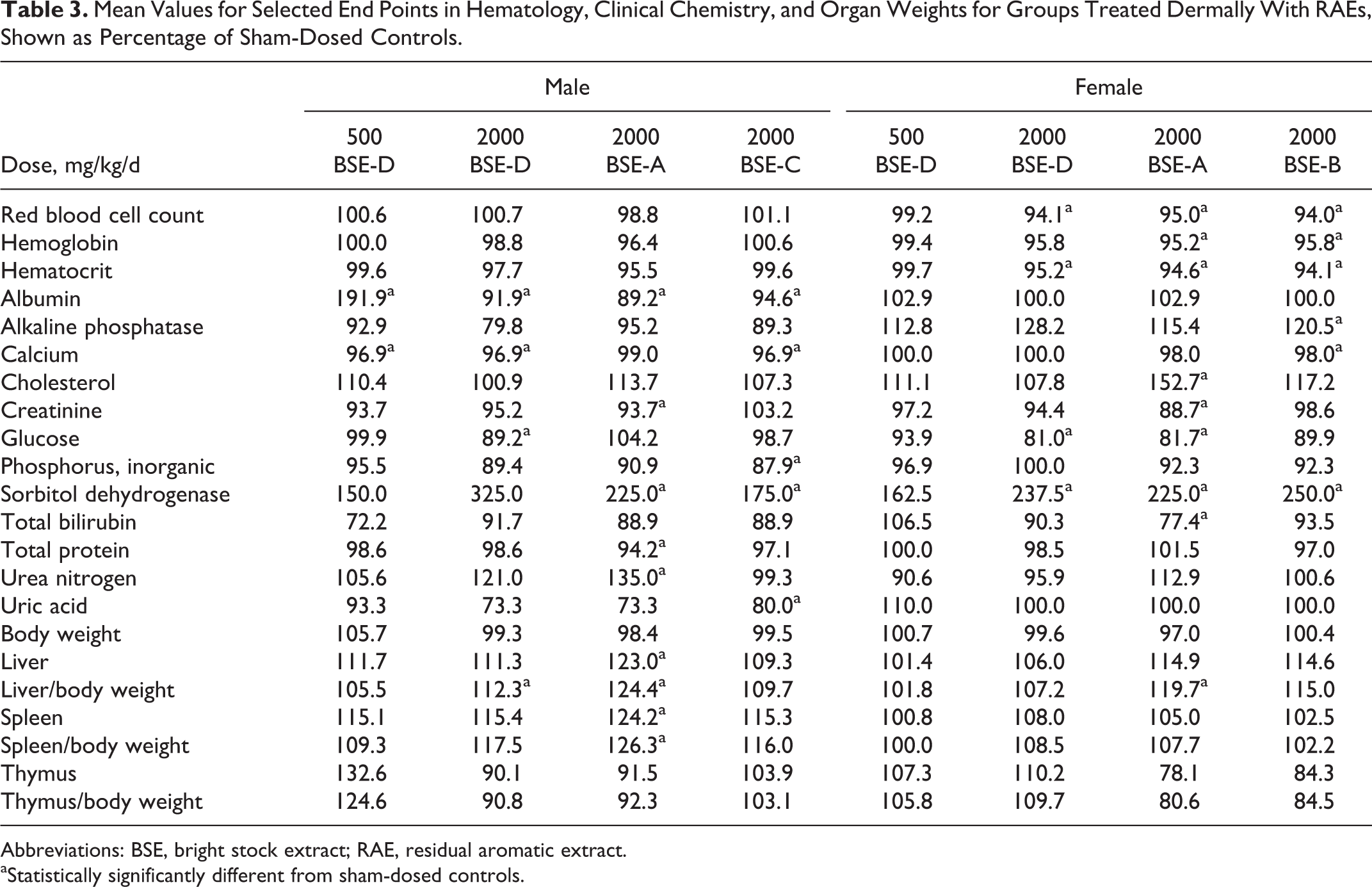
Abbreviations: BSE, bright stock extract; RAE, residual aromatic extract.
aStatistically significantly different from sham-dosed controls.
Micronucleus Tests Associated With Subchronic Studies
A statistically significant increase in the mean percentage of MN PCEs at 150 mg/kg/d in males was seen in the micronucleus test with light paraffinic DAE (Table 4) and a statistically significant increase in the mean percentage of MN PCEs was also observed in females at 50 mg/kg/d. However, a dose-response was not observed in females since an increase in mean percentage of MN PCEs was not seen at 150 mg/kg/d. Thus, the response in females was considered equivocal due to an increase at the mid dose and no effect at the high dose. The number of MN PCEs in individual animals is included in Table 4 for reference. The DAE did not produce a statistically significant increase in the mean percentage of MN NCEs or a statistically significant change in the mean ratio of polychromatic to TEs relative to the vehicle control (data not shown). The vehicle control values were within the range of historical controls for the testing laboratory and were less than 0.4% MN PCEs or MN NCEs. Overall, the light paraffinic DAE caused a positive response for induction of bone marrow micronuclei at 150 mg/kg/d for males and an equivocal response at 50 mg/kg/d for females without any meaningful differences in bone marrow cytology.
Percentage of Micronucleated Polychromatic Erythrocytes (Mean ± SD) in Micronucleus Tests With Subchronic Dermal Dosing of Light Paraffinic DAE, Heavy Paraffinic DAE, or RAEs (BSEs).a
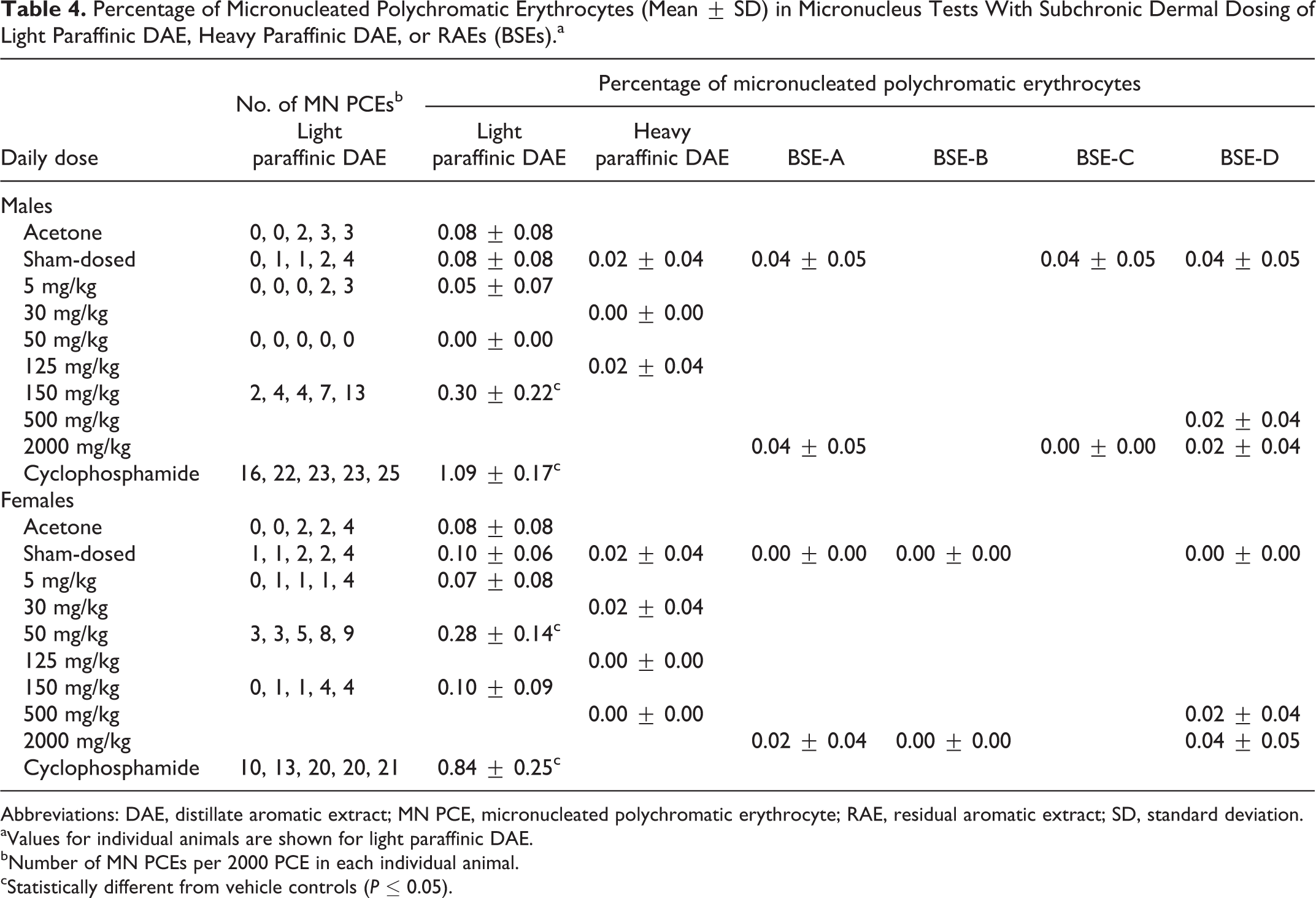
Abbreviations: DAE, distillate aromatic extract; MN PCE, micronucleated polychromatic erythrocyte; RAE, residual aromatic extract; SD, standard deviation.
aValues for individual animals are shown for light paraffinic DAE.
bNumber of MN PCEs per 2000 PCE in each individual animal.
cStatistically different from vehicle controls (
In contrast, no significant differences were seen in the percentages of MN NCEs and MN PCEs in a micronucleus test with heavy paraffinic DAE that accompanied the subchronic dermal study in this sample (Table 4). In addition, the mean percentages of MN PCEs with oral dosing of heavy paraffinic DAE in males were 0.04 ± 0.09 and 0.02 ± 0.04 for 125 and 500 mg/kg/d, respectively. These results were not significantly different from the controls (0.02 ± 0.04). The DAE was not clastogenic in this assay with dermal or oral dosing.
In micronucleus studies of RAEs, the mean ratios of PCEs to NCEs for the treated groups in the micronucleus assays were not significantly different from each other or the negative controls. Therefore, cytotoxicity was not a factor in the evaluation. No treatment-related differences were seen in the percentages of MN NCEs and MN PCEs (Table 4); the RAEs were not clastogenic in this assay.
Developmental Toxicity
Four females were euthanized in a moribund state during the dosing phase of the study with light paraffinic DAE (1 female at 150 mg/kg/d on GD 15 and 3 females at 450 mg/kg/d on GDs 14, 16, and 18). All of these females had red vaginal discharge and pale body and/or were cool to the touch up to 2 days prior to euthanasia. All other females survived to the scheduled euthanasia. Red vaginal discharge was noted in the 25, 150, and 450 mg/kg/d groups during GDs 10 to 20. In the dermal observations, higher incidences of desquamation were noted at 25, 150, and 450 mg/kg/d, generally throughout the treatment period compared to vehicle controls. No remarkable dermal effects were noted at 5 mg/kg/d.
Maternal body weight decreased during the first days of dosing with 150 and 450 mg/kg/d, and mean body weight was lower than that of vehicle controls for these 2 groups during much of gestation (Figure 2). Mean body weights in these 2 groups were significantly lower (up to 18.0% and 36.1%, respectively) than the vehicle controls during GDs 12 to 20 and 3 to 20, respectively. In addition, mean gravid uterine weights in these groups were statistically significantly lower than the vehicle controls (Table 5) and were attributed to the decreased number of viable fetuses and lower fetal weights noted at these doses (Table 6). The decreased number of viable fetuses and lower fetal weights in the 150 and 450 mg/kg/d groups also contributed to the lower body weights in these groups, especially during the latter part of gestation (Figure 2). Lower mean food consumption in these 2 groups corresponded to losses in mean body weights and lower gains in mean body weight. Other factors might have contributed to the lower maternal body weight at 150 and 450 mg/kg, as evidenced by euthanasia of 4 females and abnormal clinical signs in these groups.
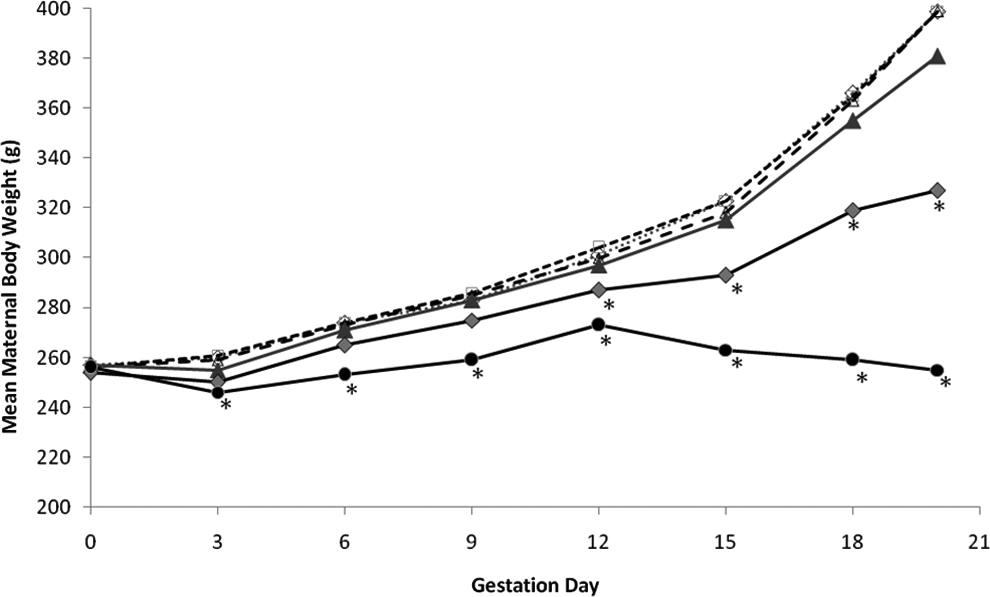
Mean maternal body weight (g) at intervals during dermal treatment with light paraffinic distillate aromatic extract (DAE). Groups were sham-exposed controls (♦) vehicle controls (□), 5 mg/kg/d (Δ), 25 mg/kg/d (▴), 150 mg/kg/d (♦), or 450 mg/kg/d (•). The asterisks denote statistically significant differences from vehicle controls (
Number of Females, Mean Maternal Body Weight, and Mean Values for Selected End Points in Rats Treated Dermally With Light Paraffinic DAE.a
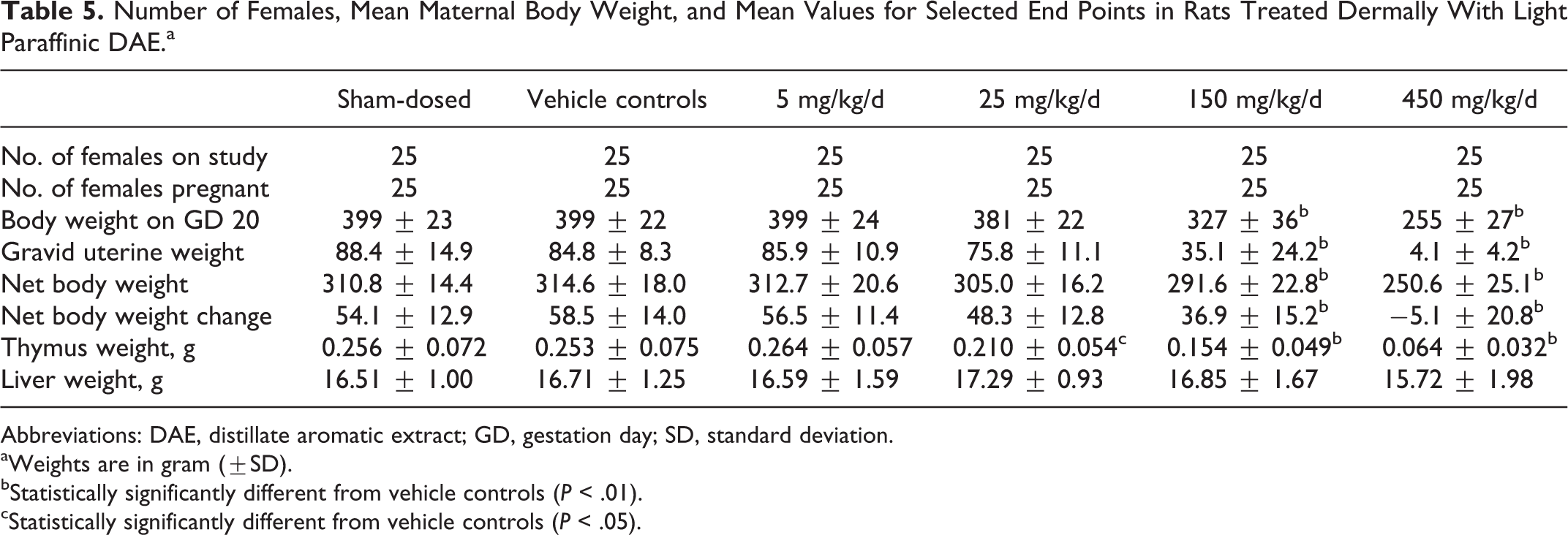
Abbreviations: DAE, distillate aromatic extract; GD, gestation day; SD, standard deviation.
aWeights are in gram (±SD).
bStatistically significantly different from vehicle controls (
cStatistically significantly different from vehicle controls (
Selected Parameters of Fetal Survival and Weight (Mean ± SD) in Rats Treated Dermally With Light Paraffinic DAE.
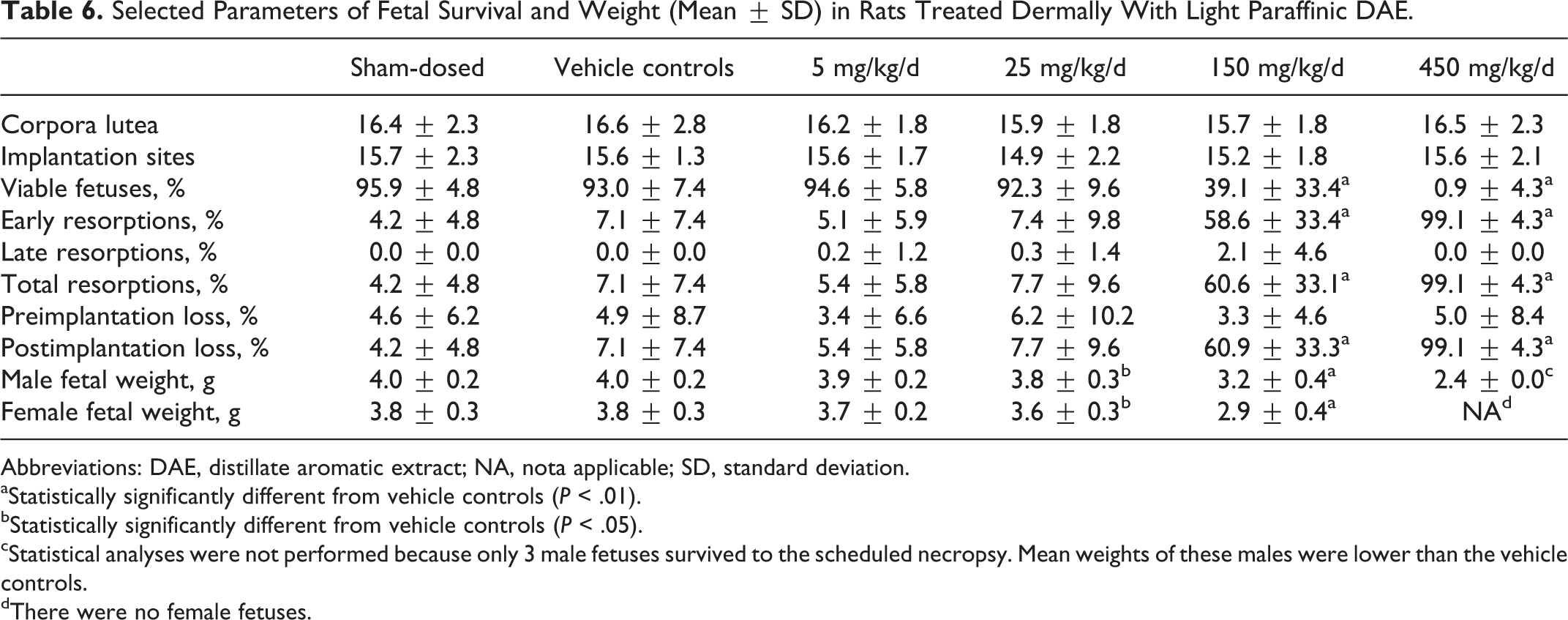
Abbreviations: DAE, distillate aromatic extract; NA, nota applicable; SD, standard deviation.
aStatistically significantly different from vehicle controls (
bStatistically significantly different from vehicle controls (
cStatistical analyses were not performed because only 3 male fetuses survived to the scheduled necropsy. Mean weights of these males were lower than the vehicle controls.
dThere were no female fetuses.
At 25 mg/kg/d, mean body weight decreased on GDs 0 to 3 (Figure 2), but mean gains in body weight were generally similar to the vehicle controls during GDs 3 to 18. However, the initial loss in weight, combined with lower gains in body weight on GDs 18 to 20 (attributed to the lower mean fetal weights), resulted in a significantly (
At the necropsy of dams on GD 20, dark red contents in the uterus, vagina, and/or cervix were noted for 5 and 4 surviving females at 150 and 450 mg/kg/d, respectively. These findings correlated with the increased number of resorptions in the surviving females in these groups. No other macroscopic findings related to DAE were observed at any dose. Thymus weight was significantly lower than vehicle controls at the highest 3 doses in a dose-related manner, but liver weight was not affected by treatment (Table 5).
Intrauterine survival was lower at 150 and 450 mg/kg/d relative to vehicle controls; the mean litter proportion of postimplantation loss (primarily early resorptions) was higher than the vehicle controls (Table 6). Correspondingly, litter proportions of viable fetuses (% viable fetuses) were significantly lower at 150 and 450 mg/kg/d. With the exception of 1 female that had 3 viable fetuses, all surviving females in the 450 mg/kg/d group had entirely resorbed litters (100% early resorptions). Two females in the 150 mg/kg/d group had 100% postimplantation loss. The numbers of viable fetuses and resorptions were not significantly affected at 5 or 25 mg/kg/d. A dose-related decrease in fetal weight in males and females was seen at the 3 highest doses (Table 6), but fetal weight was not affected at 5 mg/kg/d. The mean numbers of corpora lutea and implantation sites at 5, 25, 150, and 450 mg/kg/d were similar to the vehicle controls, and intrauterine growth and survival were similar between vehicle and sham-dosed controls.
The numbers of fetuses (litters) available for morphological evaluation were 377 (25), 361 (25), 370 (25), 329 (24), 137 (21), and 3 (1) in the sham control and vehicle control, 5, 25, 150, and 450 mg/kg/d groups, respectively. Comparative statistics were not performed on fetal morphology at 450 mg/kg/d because only 1 litter consisting of 3 fetuses survived to the scheduled necropsy. No external malformations or developmental variations were noted for any fetuses in the groups treated with the DAE. No remarkable differences in skeletal malformations or developmental variations were noted between the vehicle and the sham-dosed controls. No DAE-related visceral malformations or developmental variations were noted in fetuses and no significant differences in visceral malformations or developmental variations were noted between sham-dosed and vehicle controls. No DAE-related skeletal malformations were noted for any fetuses, but nonstatistically significant trends for increased developmental variations related to ossification occurred at 150 mg/kg/d (Table 7). Although these findings were related to dosing with the DAE, they were considered to be indicators of developmental delay and correlated to the reduced fetal weights noted at 150 and 450 mg/kg/d.
Mean Percentage Per Litter (±SD) of Fetal Skeletal Variations in Rats Treated Dermally With Light Paraffinic DAE.
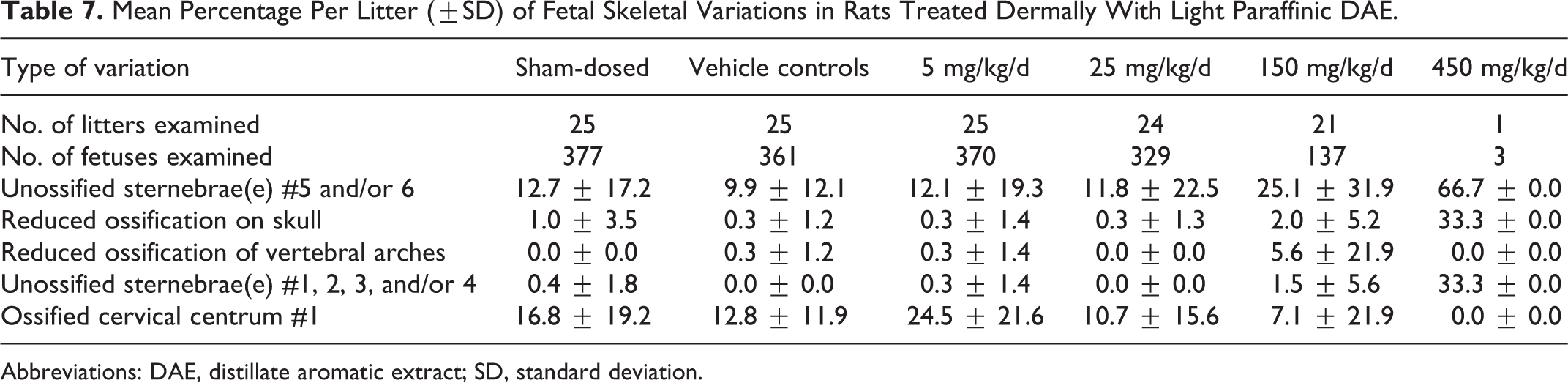
Abbreviations: DAE, distillate aromatic extract; SD, standard deviation.
Overall, developmental toxicity with light paraffinic DAE was evident in terms of decreased fetal weight at 25, 125, and 450 mg/kg/d. The lower fetal weights resulted in lower mean gravid uterine weights at these doses. Also, increased fetal resorptions occurred at 125 and 450 mg/kg/d. Indications of delayed fetal development were also seen at the 2 higher doses. Decreased maternal food consumption, net body weight, net body weight change, and thymus weight were seen at these same 2 higher doses, along with maternal moribundity. No DAE-related effects were seen at 5 mg/kg/d. Therefore, 5 mg/kg/d was considered to be the NOAEL for maternal toxicity and embryo/fetal developmental effects.
In the developmental toxicity study with RAE, no treatment-related clinical signs were seen in dams aside from slight skin irritation. Maternal body weight gains over the gestation period of the prenatal animals treated at 2000 mg/kg/d were slightly but significantly less than the controls (142 ± 19 vs 162 ± 21 g, respectively). Net body weight gain was similarly lower (67.9 ± 16.8 vs 89.6 ± 9.2 g, respectively). However, the maternal body weight gain in the treated postnatal group was similar (138 ± 16 g) to the treated prenatal group and was not significantly different from the sham-dosed postnatal controls (148 ± 16 g). Due to this variability between the 2 control groups, the biological significance of lower weight gain in the treated prenatal animals was uncertain. Food consumption in both the prenatal and the postnatal groups receiving 2000 mg/kg/d was transiently lower than their respective controls on GDs 0 to 3.
No findings attributable to treatment were seen during necropsy of prenatal dams. Furthermore, none of the reproductive parameters were affected by treatment, including number of pregnant dams, aborted fetuses, premature births, dams with viable fetuses, female mortality (%), corpora lutea, implantation sites, preimplantation loss, viable fetuses, litter size, viable male or female fetuses (%), dead fetuses, resorptions, or dams with resorptions. The only serum chemical differences recorded in prenatal dams were 20% higher aspartate aminotransferase at 2000 mg/kg/d and 3% and 4% lower serum calcium levels at 500 and 2000 mg/kg/d, respectively. Fetal weight and both skeletal and visceral examinations of the fetuses were not affected by treatment in the prenatal groups. Similarly, no treatment-related effects were noted in the postnatal group (tables are not provided). Overall, evidence of developmental toxicity was not observed in any group treated with the test substance, and the NOAEL for maternal and developmental toxicity was 2000 mg/kg/d, the highest dose evaluated.
Modeled Predictions of Toxicity
As a general comment on data for samples in Table 1, the distribution and concentration of 1- to 7-ring PACs are different for DAEs compared to RAEs. Total ARCs in each sample ranged from 6.3% to 20.3% in the DAEs compared to 1.8% to 4.5% in the RAEs. Since the DAEs in Table 1 had values well above the cutoff of 3% used with IP346 (described later), those samples can be considered potentially carcinogenic. These data also reflect the variation that can be seen among samples of petroleum streams with the same CAS number.
The value of the PAC data here was the estimation of toxicity for samples that have not been tested experimentally. The calculated PDR10s for subchronic dermal exposures are shown in Table 8. No ARC profiles were available to calculate PDR10s for light naphthenic DAE and valid PDR10s could not be calculated for the 1 sample of heavy naphthenic DAE because the ARC profile was outside the domains of the models. The sample PDR10s for 3 light paraffinic DAEs ranged from 42 to 72 mg/kg/d. Both PDR10s and BMD10s could be calculated for 1 light paraffinic DAE that was tested in a 13-week dermal study. The BMD10s ranged from 13 to 360 mg/kg/d for the 4 indicators of effect with this sample while the PDR10s ranged from 42 to 80 mg/kg/d for the same end points. Although the lowest PDR10 was ∼3 times greater than the lowest BMD10, the range of BMD10s encompassed the range of PDR10s. As mentioned previously, the fact that the PDR10s and BMD10s are in general agreement provides reassurance of the utility of the recently developed models for calculating PDR10s. The NOEL for this study (<5 mg/kg/d) was based on lower numbers of eosinophils, an end point not typically affected in studies with other heavy petroleum streams.
Repeated-Dose PDR10s and BMD10s (mg/kg/d) for DAEs by End Point, Sex, and Test Substance.a
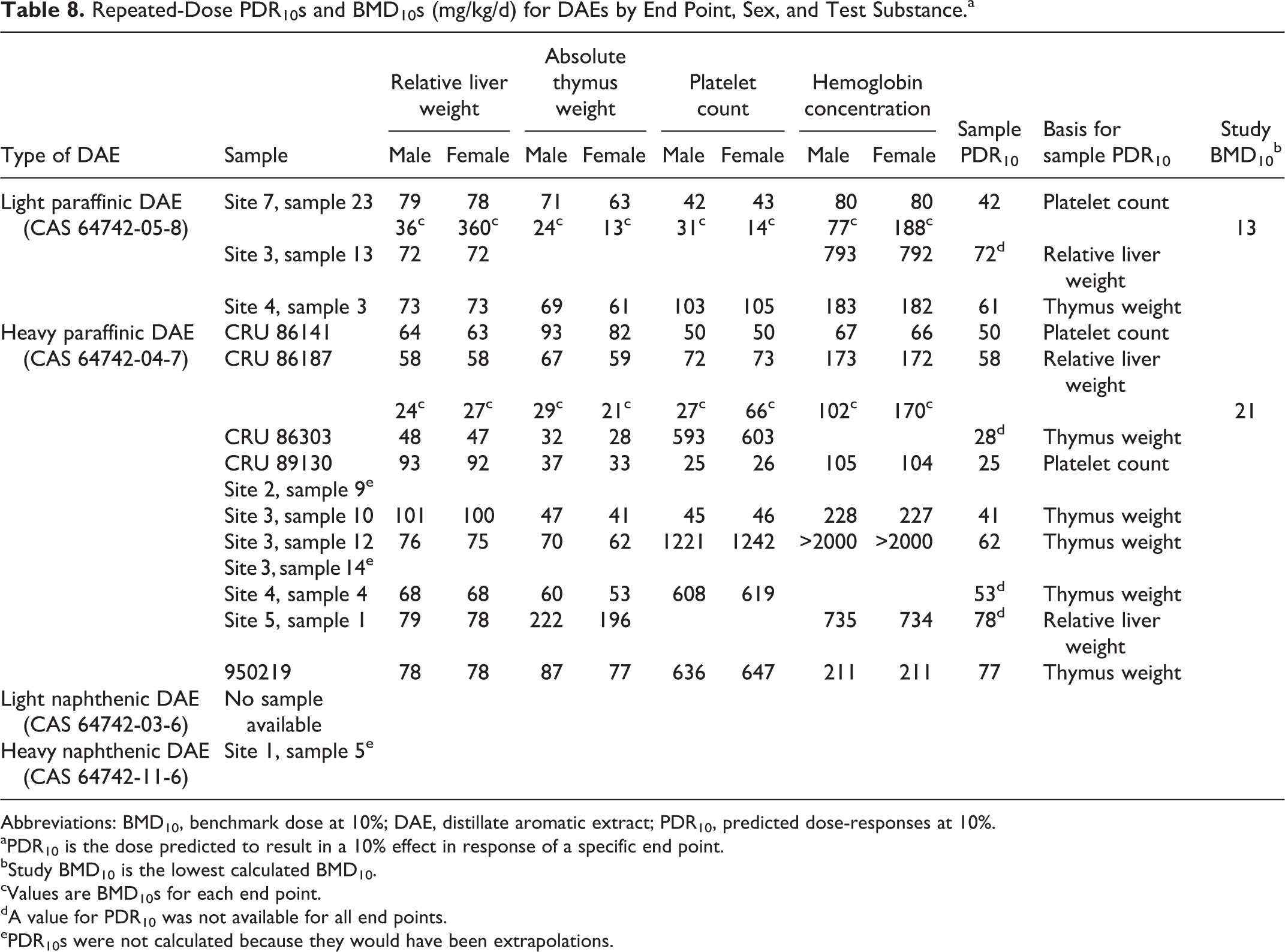
Abbreviations: BMD10, benchmark dose at 10%; DAE, distillate aromatic extract; PDR10, predicted dose-responses at 10%.
aPDR10 is the dose predicted to result in a 10% effect in response of a specific end point.
bStudy BMD10 is the lowest calculated BMD10.
cValues are BMD10s for each end point.
dA value for PDR10 was not available for all end points.
ePDR10s were not calculated because they would have been extrapolations.
A comparison of PDR10s and BMD10s was similar for the heavy paraffinic DAEs. The sample PDR10s ranged from 25 to 78 mg/kg/d for 11 samples. The BMD10s for the 1 tested sample ranged from 21 to 170 mg/kg/d and the PDR10s ranged from 58 to 173 mg/kg/d. Again the lowest PDR10 was greater than the lowest BMD10, and the range of PDR10s was virtually within the range of BMD10s. The BMD10s for thymus weight and relative liver weight agreed well with the NOEL (<30 mg/kg/d) that was based on these same end points.
Sample PDR10s for developmental toxicity (Table 9) ranged from 53 to 2000 mg/kg/d for light paraffinic DAEs and from 7 to >2000 mg/kg/d for heavy paraffinic DAEs. The sample PDR10 for the 1 tested heavy paraffinic DAE was the same as the BMD10 (15 mg/kg/d), and both values were consistent with the NOAELs for this study (between 8 and 30 mg/kg/d). In addition, the PDR10s for specific end points agreed well with the BMD10s. Agreement between PDR10s and BMD10s was not as good with the tested sample of light paraffinic DAE. The lowest BMD10 calculated from this study was 42 mg/kg/d (for living fetuses per litter). There was no PDR10 value for this end point because the modeled prediction was an extrapolation beyond the boundary of the model. The one calculated PDR10 for reduced fetal body weight (420 mg/kg/d) was greater than the BMD10 for this end point (117 mg/kg/d) and also greater than the NOAEL of 5 mg/kg/d. In this case, the model predicted the sample to be less toxic than it actually was.
Maternal and Developmental PDR10s and BMD10s (mg/kg/d) for DAEs by End Point and Sample.a
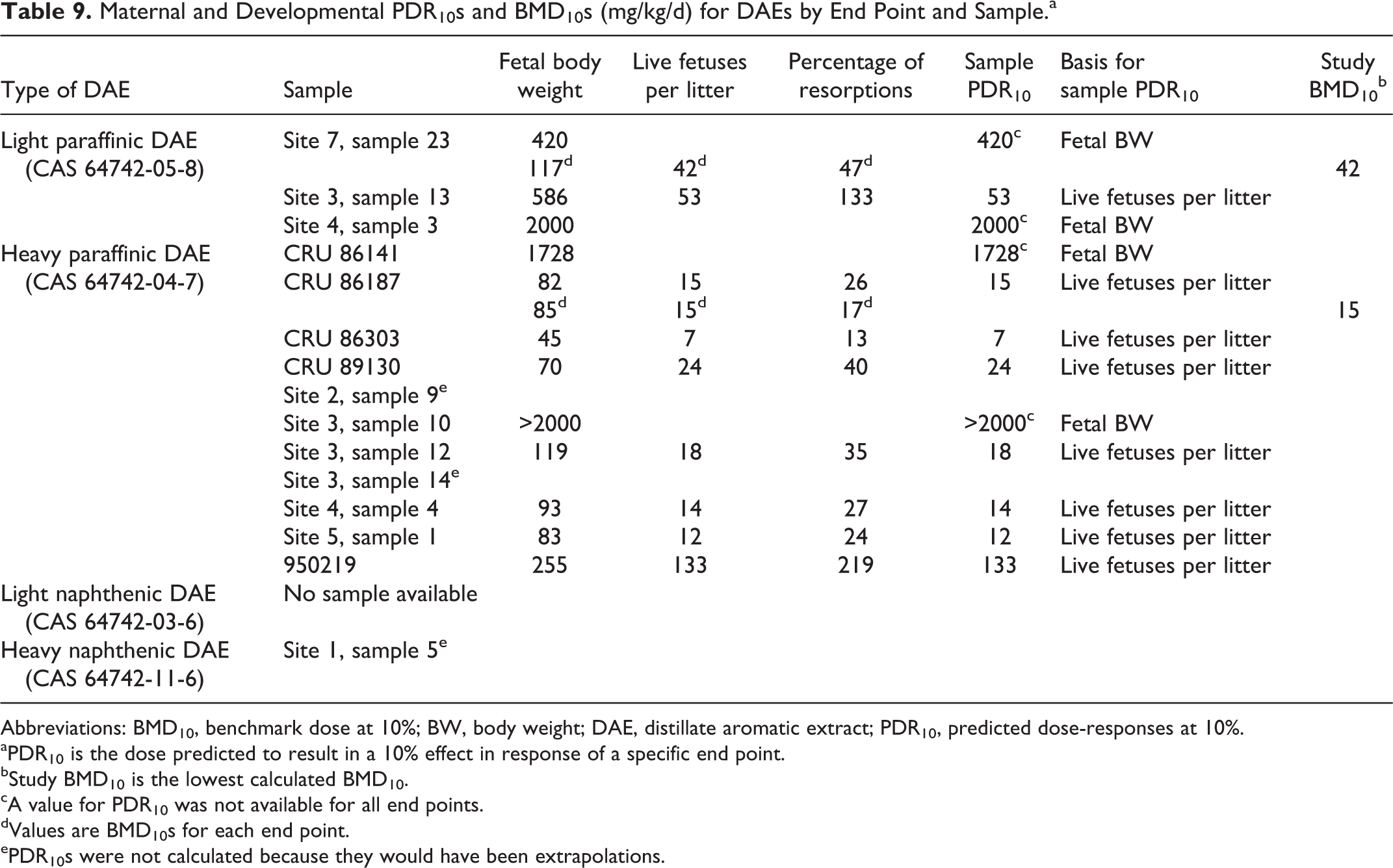
Abbreviations: BMD10, benchmark dose at 10%; BW, body weight; DAE, distillate aromatic extract; PDR10, predicted dose-responses at 10%.
aPDR10 is the dose predicted to result in a 10% effect in response of a specific end point.
bStudy BMD10 is the lowest calculated BMD10.
cA value for PDR10 was not available for all end points.
dValues are BMD10s for each end point.
ePDR10s were not calculated because they would have been extrapolations.
Reproductive Toxicity
In the 90-day study on heavy paraffinic DAE (previously published), rats were dosed dermally with 0, 30, 125, 500, and 1250 mg/kg/d. All animals receiving 1250 mg/kg, as well as all males and 3 females receiving 500 mg/kg, died or were euthanized early due to clinical signs. At the highest nonlethal dose (125 mg/kg), no effects were seen on the weights of epididymides, prostate, testes, ovaries, or uterus. Mean weights are shown in Table 10. Also no significant treatment-related changes were seen in histopathology of epididymides, ovaries, prostate, seminal vesicles, or testes. Finally, morphology of epididymal spermatozoa was evaluated in 5 controls and 5 males dermally treated with 125 mg/kg. As shown in Table 10, the measured parameters were not affected by treatment. Overall, these data showed no observed effects on the tested reproductive parameters at the highest nonlethal dermal dose.
Mean Body Weight, Organ Weights, and Parameters of Sperm Morphology in 90-Day Study With Heavy Paraffinic DAE.a
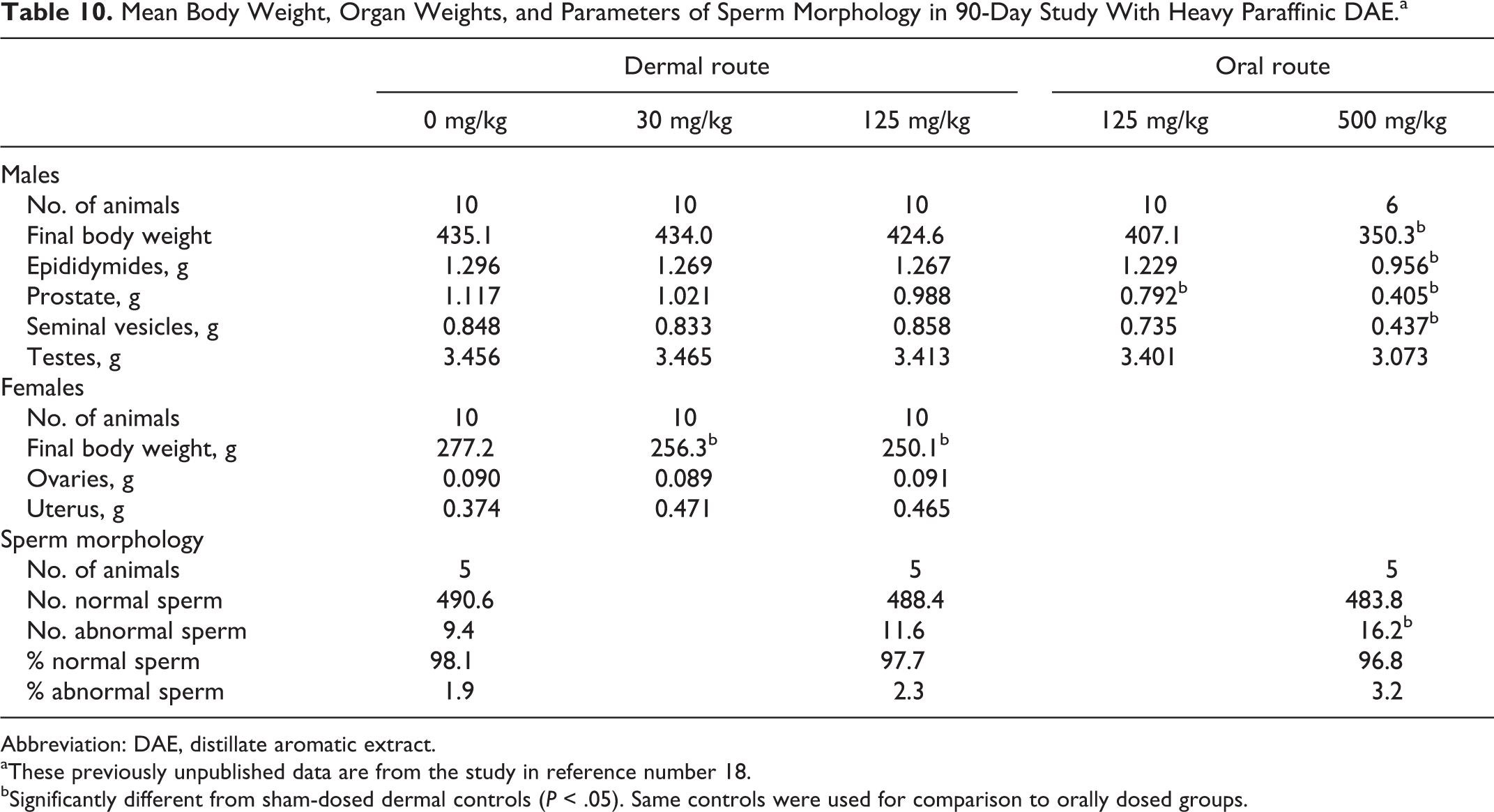
Abbreviation: DAE, distillate aromatic extract.
aThese previously unpublished data are from the study in reference number 18.
bSignificantly different from sham-dosed dermal controls (
Two additional groups of 10 males were dosed orally with 125 or 500 mg/kg/d. Four of the males dosed with 500 mg/kg died or were sacrificed in extremis. Given this mortality, other evidence of systemic toxicity, and mean body weights that were 80.5% of controls, the relevance of the significantly lower weights for epididymides, prostate, and seminal vesicles shown in Table 10 is questionable. The weight of the prostate with oral dosing at 125 mg/kg was significantly less than the weight in controls, but no significant treatment-related effects were seen in histopathology of epididymides, ovaries, prostate, seminal vesicles, or testes. In the morphological evaluation of epididymal spermatozoa, the number of abnormal sperm was increased with 500 mg/kg; similar data were not available for the nonlethal oral dose of 125 mg/kg.
No treatment-related effects on weights of reproductive organs were seen in the 13-week study with light paraffinic DAE (Table 11) and histopathological examination of the cervix, epididymides, ovaries, prostate, seminal vesicles, testes, uterus, and vagina did not reveal treatment-related changes.
Mean Body Weight and Weights of Reproductive Organs (±SD) in 90-Day Dermal Study With Light Paraffinic DAE.

Abbreviations: DAE, distillate aromatic extract; SD, standard deviation.
Similarly, no adverse effects on reproductive organs were observed with 13-week exposures to RAEs. As shown in Table 12, no treatment-related effects were seen in the following end points with dermal dosing with BSE-D or BSE-A at 2000 mg/kg: (1) weights of testes, prostate, epididymides, ovaries, and uterus, (2) histopathology of testes and ovaries, (3) weight of testicular parenchyma and cauda epididymis, (4) number of testicular sperm and number/g testis, and (5) number and morphology of epididymal sperm, as well as number/g cauda. No treatment-related effects were seen in weights of testes, prostate, and epididymides or in the histopathology of testes following dermal dosing of males with BSE-C. No treatment-related effects were seen in weights of ovaries and uterus or in the histopathology of ovaries following dermal dosing of females with BSE-B.
Mean Body Weight, Organ Weights, and Parameters of Sperm Morphology (±SD) in 90-Day Study with RAEs.
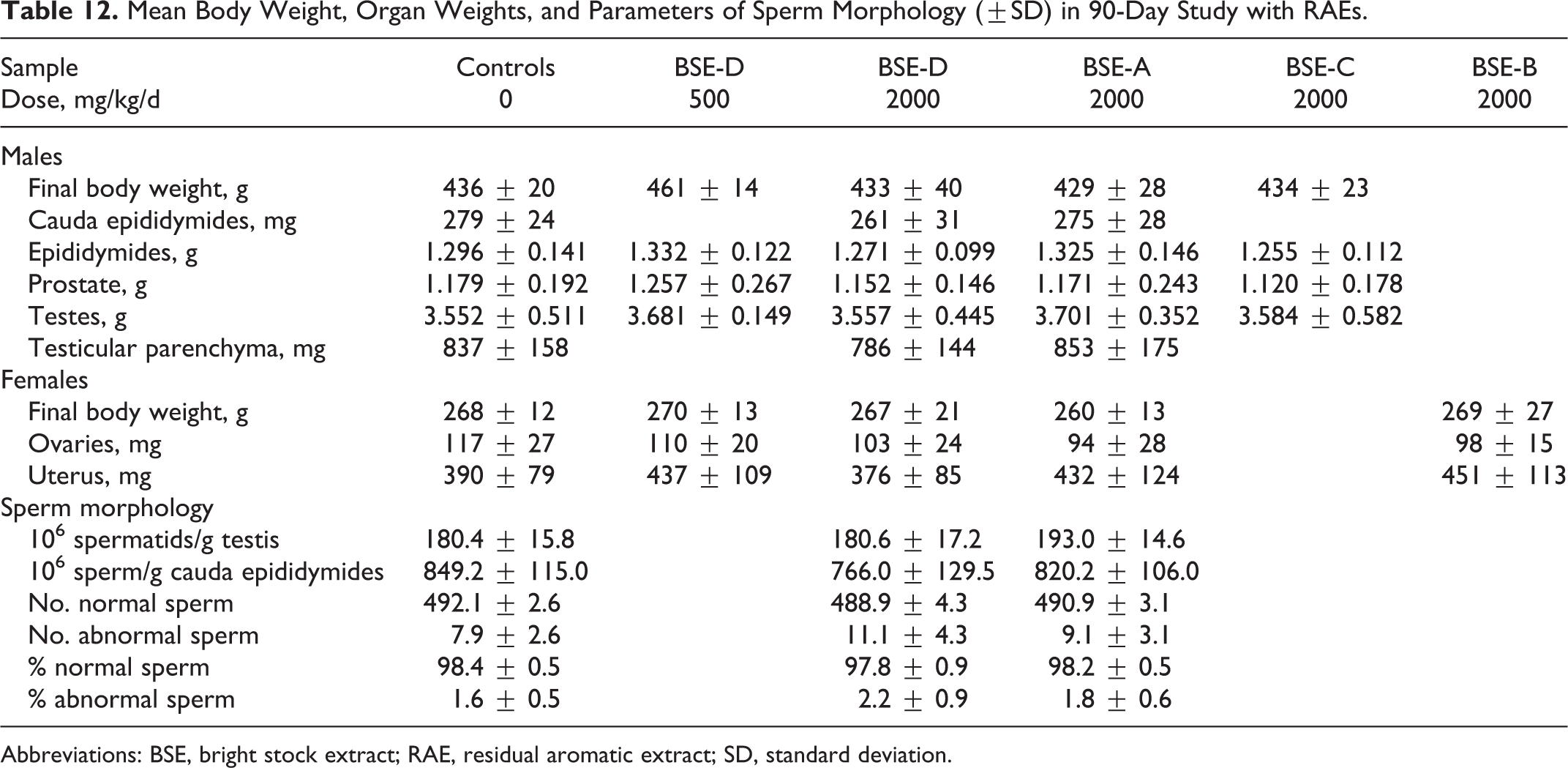
Abbreviations: BSE, bright stock extract; RAE, residual aromatic extract; SD, standard deviation.
Discussion
Systemic Toxicity and Micronucleus Tests With Repeated Dermal Exposures
The main findings in the 13-week study on light paraffinic DAE were similar in many regards to those in the previously reported 13-week study on heavy paraffinic DAE. 17,18 Lower Hb, hematocrit, RBC numbers, and thymus weight as well as higher liver weight, cholesterol, and urea nitrogen were seen with both DAEs at doses of 150 or 125 mg/kg/d, respectively. Lower body weights in females and histological effects in the liver, bone marrow, and other organs were noted with heavy paraffinic DAE but not with light paraffinic DAE. Conversely, higher numbers of reticulocytes and lower numbers of eosinophils were seen with light paraffinic DAE and not with heavy paraffinic DAE. The NOEL for each DAE was lower than the lowest dose tested (30 mg/kg/d for heavy paraffinic DAE and 5 mg/kg/d for light paraffinic DAE).
The general similarity in the effects of the 2 DAEs is consistent with the relative PAC content of each. The “total ARC wt%” was 13.6% for light paraffinic DAE (sample “site 7, sample 23”) and 20.3% for heavy paraffinic DAE (sample “CRU 86187”). Both DAEs affected those end points that had been determined to be sensitive to PACs in refinery streams. The light paraffinic DAE also affected the number of eosinophils at a dose of only 5 mg/kg/d.
The effects of 13-week dermal exposures to 4 RAEs were similar to those with the DAEs, but less pronounced, particularly given the higher doses (500 or 2000 mg/kg/d) used with the RAEs. These findings are consistent with the lower PAC content of the RAEs shown in Table 1 as well as the high viscosity of many RAEs that substantially reduces absorption. 36
The lack of clastogenic activity in the in vivo micronucleus assays that accompanied the subchronic dermal and oral dosing with heavy paraffinic DAE and dermal dosing with 4 RAEs was consistent with the general lack of genotoxic effects from other petroleum streams in vivo. Micronucleus tests with other petroleum streams that contain higher amounts of PACs have also been negative, leading to the conclusion that PAC-containing petroleum substances are unlikely to produce chromosomal effects when tested in assays performed
Developmental Toxicity
The results of the dermal developmental toxicity study with light paraffinic DAE given here paralleled those reported previously for heavy paraffinic DAE. 18 At doses of 125 to 500 mg/kg/d with either DAE, the observed effects included increased resorptions and lower body weight, body weight gain, gravid uterine weight, thymus weight, and fetal weight. No treatment-related increases in fetal skeletal or visceral abnormalities were noted except for reduced ossification with the light paraffinic DAE.
At doses of 25 to 30 mg/kg/d, marginally lower (not statistically significant) maternal body weight, thymus weight, and gravid uterine weight were seen with both DAEs, although the lower thymus weight was statistically significant with light paraffinic DAE. No effect was seen on percentage of resorptions with light paraffinic DAE, but a nonstatistically significant increase in resorption frequency with heavy paraffinic DAE was considered to be possibly biologically significant. Fetal body weight was statistically significantly lower than vehicle controls with light paraffinic DAE; the mean for both sexes combined was 3.7 ± 0.2 versus 3.9 ± 0.2 g for vehicle controls. The difference with heavy paraffinic DAE was similar (3.3 ± 0.2 vs 3.5 ± 0.2 g) but was not statistically significant using the methods in that study. No significant adverse maternal or fetal effects were seen at 8 mg/kg/d for heavy paraffinic DAE or 5 mg/kg/d for light paraffinic DAE. In both cases, significant fetal effects occurred only at doses that also caused maternal effects.
In a related effort to explore the suspected teratogenic potential of heavy paraffinic DAE, clarified slurry oil, and syntower bottoms, Feuston and Mackerer 39 conducted a developmental toxicity study in which the test materials were given by gavage on a single day of gestation. Developmental toxicity was not observed in the absence of maternal toxicity with any of these petroleum streams. Although the oral route of administration has little relevance to human occupational exposure, these samples were shown to have teratogenic potential when a large dose (2000 mg/kg) was given by gavage to pregnant rats on a single day during the critical period of gestation.
A more limited range-finding developmental toxicity study with heavy naphthenic DAE was reported in a submission to the US EPA. 19 Doses of 0 (vehicle control), 500, 1000, or 2000 mg/kg/d were administered dermally on GDs 7 to 16. All dams survived and were sacrificed on GD 20 for necropsy. All fetuses were weighed and examined externally for developmental abnormalities, and sex was determined. The LOAEL for maternal toxicity was 500 mg/kg/d based on decreases in body weight gains, matted fur, and nasal, ocular, and vaginal discharge; an NOAEL was not established. The LOAEL for developmental toxicity was 2000 mg/kg/d based on the increased resorptions, a corresponding decrease in live implants, and reduced fetal and mean litter weights; the NOAEL was 1000 mg/kg/d. These findings are in contrast to the greater toxicity seen with both light and heavy paraffinic DAEs and point to the variability that can occur with petroleum streams depending on the source of crude oil, refining conditions, and the resulting composition of the samples.
No treatment-related effects were seen in the prenatal or postnatal portions of the developmental toxicity study with RAE; the NOAEL for maternal and developmental toxicity was 2000 mg/kg/d, the highest dose evaluated. These results were consistent with the relatively lower toxicity seen in the subchronic studies with RAEs.
Modeled Predictions of Toxicity
Overall, the sample PDR10s for subchronic exposures were similar to the study BMD10s for these 2 DAEs although the PDR10s tended to be slightly higher than the BMD10s for some specific end points (Table 8). That is, the sample PDR10s were reasonable approximations of observed effects as represented by BMD10s from actual experiments. The general agreement between the PDR10s and the BMD10s lends support to the use of both approaches.
For the 2 DAEs, agreement between modeled PDR10s and actual data on developmental toxicity was mixed, with good agreement for the heavy paraffinic DAE and not good agreement with the light paraffinic DAE. In fact, the predictions from the developmental toxicity models for the light paraffinic DAE were not considered adequate. 21 Although the models did not predict false negatives, they did not provide sufficiently accurate estimates. As described in more detail elsewhere, the most likely explanation for this discrepancy is that the ARC 6 values for these new experimental samples are lower than those of the samples used to develop the model. 21 In the future, as new test data become available, they can be incorporated into the models to expand the domain of applicability and improve the accuracy of the model estimates.
The ARC profile provided a secondary measure of the samples in Table 1. The DAEs contain relatively high levels of PACs and are considered to be carcinogenic. One means of characterizing the PAC content is to measure the total amount of DMSO-extractable material using the IP346 method 40 in which substances related to the manufacture of lubricant base oil with levels >3.0 wt% of DMSO-extractable material are considered to be potentially carcinogenic. The weight percentage of DMSO-extractable material measured by method II (the “total ARC wt%”) is similar to the weight percent measured with the IP346 method. Therefore, “total ARC wt%” reported here provide some indication of the potential carcinogenicity of the sample.
Reproductive Toxicity
The results from the developmental toxicity studies on AEs provide data to address the developmental toxicity of paraffinic DAEs and RAEs in an assessment of possible reproductive toxicity. The other piece of that assessment would involve effects on reproductive organs with repeated exposures of adults. Such data come from the 13-week studies. The weights of the epididymides, ovaries, prostate, testes, and uterus were not affected at the highest nonlethal dose of heavy paraffinic DAE and no significant effects were seen histologically in the epididymides, ovaries, prostate, seminal vesicles, or testes. Morphology of epididymal spermatozoa was not affected by treatment at the highest nonlethal dermal dose. Results were similar to oral dosing. Similarly, weights of the epididymides, ovaries, prostate, testes, and uterus were not affected by dermal treatment with light paraffinic DAE and no histological changes were seen in the cervix, epididymides, ovaries, prostate, seminal vesicles, testes, uterus, or vagina. In the 13-week dermal studies with RAEs, no effects were seen in (1) weights of testes, prostate, epididymides, ovaries, and uterus, (2) histopathology of testes and ovaries, (3) weight of testicular parenchyma and cauda epididymis, (4) number of testicular sperm and number/g testis, and (5) number and morphology of epididymal sperm.
Taken together, these data from developmental and repeated-dose studies satisfy the HPV requirements for assessments of reproductive toxicity and indicate that reproductive toxicity with AEs is expected to be minimal. Also, the NOAEL for reproductive toxicity of AEs is unlikely to be lower than the NOAEL for developmental toxicity because the most sensitive end points in either developmental or reproductive toxicity studies are believed to be effects on fetal survival and growth resulting from in utero exposure. 17,20,41 Reproductive toxicity does not appear to be a sensitive end point of toxicity compared to developmental and repeat-dose toxicity. In a recent study, 41 high-boiling petroleum substances, including many with a high PAC content, have demonstrated low potential to cause male or female reproductive toxicity relative to developmental toxicity and systemic toxicity (in repeated-dose toxicity studies).
Further evidence of the low potential of AEs to cause reproductive toxicity comes from screening-level fertility studies of clarified slurry oil (CSO, CAS No. 64741-62-4, also known as catalytically cracked clarified oil, syntower bottoms, and carbon black oil). This refinery stream contains such high levels of PACs that it is believed to be “worst case” by comparison to all other refinery streams. When samples of CSO are tested in developmental toxicity studies, they typically produce developmental effects (resorptions, reduced fetal body weight, and cleft palate) at levels below 5 mg/kg/d. 42,43 In contrast, in screening studies to assess the potential for CSO to affect male and female reproductive parameters, 44 there were no effects at levels up to 250 mg/kg/d, the highest dose tested.
Conclusions
Distillate AEs are generally expected to contain significant amounts of PACs and to be carcinogenic. The RAEs also can contain significant amounts of PACs. Given that PACs in refinery streams can cause effects in repeated-dose and developmental toxicity studies, it was not surprising that rats treated dermally with light or heavy paraffinic DAE over 13 weeks had several dose-related effects involving multiple organs, resulting in NOELs <30 mg/kg/d. PDR10s based on ARC profile for untested samples of light and heavy paraffinic DAEs were similar, ranging from 25 to 78 mg/kg/d. The NOAEL for developmental toxicity for a light paraffinic DAE was 5 mg/kg/d. The NOAEL for developmental toxicity with a heavy paraffinic DAE was 8 mg/kg/d for biologically, but not statistically, significant effects. Statistically significant developmental effects at higher doses were associated with significant maternal effects. PDR10s for light and heavy paraffinic DAEs ranged from 7 to >2000 mg/kg/d, reflecting variation due to differences in the ARC profile of different samples. These findings were consistent with the premise that 1 to 7 ring PACs in AEs are largely responsible for the effects observed in these studies.
In vivo micronucleus tests on a heavy paraffinic DAE, RAEs, and a range of other petroleum substances have been negative. The exception to this general trend was a positive response in 2 of the 5 males at the highest tested dose of a light paraffinic DAE. Given the limited number of affected animals in this test, the weight of evidence from tests on several petroleum streams, and read-across from the heavy paraffinic DAE, it is concluded that most DAEs are unlikely to produce chromosomal effects under in vivo conditions.
Reproductive toxicity is not considered to be a sensitive end point for AEs based on results from subchronic and developmental toxicity tests with DAEs, RAEs, and other PAC-containing substances. The NOAELs for reproductive toxicity of DAEs would not be expected to be below NOAELs for developmental effects.
Although information on the possible effects from exposure to naphthenic DAEs is more limited, those effects are expected to result primarily from PACs in an individual sample. Since the presence of the biologically active PACs is affected greatly by the source of crude oil and refining conditions, variability can occur in levels of PACs and the resulting potential toxicity of different AEs.
Footnotes
Appendix
Acknowledgments
The authors would like to thank Chris Sexsmith for quality assurance support, Maureen Feuston (study director on 90-day study with heavy paraffinic DAE, 90-day studies with RAEs, and developmental toxicity study with RAEs), Sue Irwin (study director on micronucleus test with heavy paraffinic DAE), Stacey Kerstetter (study director on developmental toxicity study with heavy paraffinic DAE, and Frank Angelosanto (study director on micronucleus tests with RAEs).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are a mix of employees, former employees, and contractors for companies, API, and/or PHPVTG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was sponsored and funded by the Petroleum HPV Testing Group (PHPVTG), an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program.
