Abstract
Rose hip is the fruit of the rose plant, which is widely used in food, cosmetics and as a traditional medicine. Therefore, rose hip is considered safe and has a sufficient history of consumption as food. However, few studies have reported on the safety of rose hip extracts in toxicological analyses. Thus, to evaluate the safety of rosehip polyphenol MJ (RHPMJ), an aqueous ethanol extract standardized with the trans-tiliroside content, we performed genotoxicity and 90-day repeated oral dose toxicity studies in compliance with the Organisation for Economic Co-operation and Development-Good Laboratory Practice. RHPMJ did not induce gene mutations in reverse mutation tests of Salmonella typhimurium TA98, TA100, TA1535, TA1537 and Escherichia coli WP2uvrA strains and did not induce chromosomal aberrations in cultured Chinese hamster lung (CHL/IU) cells. Moreover, micronucleus tests using rat bone marrow showed RHPMJ had no micronucleus-inducing potential. Finally, 90-day repeated oral dose toxicity studies (100–1000 mg/kg) in male and female rats showed no treatment-related toxicity in rats. These data indicate that the RHPMJ had no genotoxicity and a no-observed-adverse-effect level greater than 1000 mg/kg in rats.
Introduction
The genus Rosa L. belongs to the family Rosaceae, which comprises over 100 species distributed widely in Europe, Asia, the Middle East and North America. 1 Rose hip is the fruit of the Rosa plant, and fruits from Rosa canina L. are marketed as an important food source used as a traditional medicine in numerous cultures. The fruit of R. canina is widely processed to produce jam and juice and is used as a vitamin C supplement, cosmetics component and as traditional medicine in the form of tisane. 2,3 The Complete German Commission E Monographs of Therapeutic Guide to Herbal Medicines lists rose hip with or without fruits (achenes) for the treatment of colds and as a laxative, diuretic, anti-gout and anti-rheumatism agent. 4 There have also been reports of several biological effects including anti-oxidative, 5 anti-inflammatory, 6,7 anti-glycative, 8 anti-diabetic, 9 inhibition of melanin production, 10 antibacterial, 11 and inhibition of cancer cell proliferation. 12,13 Clinical studies using standardized powders (Hyben Vital®, Hyben Vital, Langeland, Denmark or LitoZin®, Orkla Health, Oslo, Norway) containing equal amounts of seeds and shells from selected subspecies of R. canina demonstrated the effects of rose hip on osteoarthritis of the knee and hip joints, 2,14 –16 rheumatoid arthritis, 17 chronic pain 18,19 and skin ageing. 20 Moreover, Andersson et al. 21 reported the clinical effects of rose hip powder drink on risk markers for acquired type 2 diabetes, and Ginnerup-Nielsen et al. 22 demonstrated the improved biomechanical functions of knee joints following treatments with rose hip powder.
Recently, Ninomiya et al.
23
showed that 80% aqueous acetone extracts of R. canina fruits significantly suppressed body weight (BW) gain and prevented increased storage of visceral fat in non-obese mice. In addition, trans-tiliroside (
In this study, we conducted a pattern analysis to obtain information on RHPMJ ingredients other than
Materials and methods
Chemicals and materials
Trans-Tiliroside (

Chemical structures of standard compounds (
Analysis methods
The total polyphenol (expressed as catechin equivalent (CE)) in RHPMJ was determined using the Folin–Ciocalteu method at the Foundation of Food Analysis Technology Center SUNATEC (Mie, Japan). Various chemical and nutritional analyses (Online Supplementary Table S1) were contracted to SUNATEC and Japan Food Research Laboratories (Tokyo, Japan).
The pattern analysis of RHPMJ was carried out as followings. Standard solutions (10 µg/mL) and RHPMJ (5000 µg/mL) were prepared in 50% methanol and injected into the high-performance liquid chromatography (HPLC) system (LaChrom Elite®; Hitachi, Tokyo, Japan) consisting of a quaternary pump with a diode array detector, an autosampler and a column oven equipped with an InertSustain C18 column (5 μm, 4.6 × 250 mm, GL Sciences, Tokyo, Japan). The chromatographic separation was performed at 40°C with mobile phases A (water/acetonitrile/formic acid, 90:5:5, v/v/v) and B (water/acetonitrile/formic acid, 50:45:5, v/v/v). The gradient program was as follows: 0 min (A:B, 100:0, v/v) → 30 min (0:100, v/v) → 30.1 min (100:0, v/v) → 45 min (100:0, v/v). The flow rate and injection volume were 1.5 mL/min and 10 μL, respectively. The ultraviolet (UV) absorbance was monitored at a wavelength range of 190–550 nm and 280 nm was selected as the detection wavelength for comparison.
Compound
Dosing formulation for toxicity studies
The RHPMJ sample was ground in a mortar and then dissolved or suspended at specified concentrations in water for injection. Test solutions from 5 to 200 mg/mL were stable for 7 days under refrigerated conditions and 24 h at room temperature (18–25°C) in the dark. Genotoxicity studies were performed using test solutions of 50 and 5 mg/mL in the bacterial reverse mutation and chromosome aberration tests, respectively, and 50, 100 and 200 mg/mL in the micronucleus tests. Subsequent 90-day repeated dose toxicity studies were performed using test solutions of 10, 30 and 100 mg/mL.
Animals and organisms
Micronucleus tests were performed in 7-week-old male Crl: CD (Sprague-Dawley) rats (Charles River Japan, Inc., Kanagawa, Japan) that were individually housed in wire mesh cages and fed an autoclaved pellet diet (CFR-1; Oriental Yeast Co., Ltd, Tokyo, Japan) and provided with UV-irradiated tap water ad libitum. The rats were maintained at 22 ± 3°C with a 12-h light/dark cycle at 50 ± 20% humidity and a ventilation frequency of 10 to 15 air changes per hour. All animals were quarantined and acclimated for approximately 1 week, and then 30 rats were selected for the experiments.
Repeated dose toxicity studies were performed using 4-week-old male and female Crl: CD (Sprague-Dawley) rats (Charles River Japan, Inc.) that were individually housed in wire mesh cages and provided an autoclaved pellet diet (Teklad certified irradiated global 18% protein rodent diet 2918C; Harlan Laboratories, Inc., Indianapolis, Indiana, USA) and UV-irradiated tap water ad libitum. They were housed at 22 ± 3°C on a 12-h light/dark cycle at 50 ± 20% humidity with a ventilation frequency of 10 to 15 air changes per hour. All animals were quarantined and acclimated for approximately 2 weeks; 40 males and 40 females were then selected for the experiments.
Reverse mutation tests were performed in Salmonella typhimurium TA100, TA1535, TA98 and TA1537 strains from Japan Bioassay Research Center (Kanagawa, Japan) and Escherichia coli WP2uvrA from the National Institute of Technology and Evaluation Biological Resource Center (Tokyo, Japan). CHL/IU cells derived from the lungs of a female Chinese hamster were purchased from DS Pharma Biomedical Co., Ltd (Osaka, Japan) for the chromosome aberration tests.
Bacterial reverse mutation test
The reverse mutation tests were conducted in bacteria according to the pre-incubation method in the presence and absence of metabolic activation with the S9 mix. Water for injection and well-established mutagens were used as negative and positive controls, respectively (Table 1). Based on the results of dosimetric analyses, RHPMJ was used at concentrations of 5000, 2500, 1250, 625 and 313 μg/plate. The numbers of treated S. typhimurium TA100 and positive control colonies of all bacterial strains were counted using a CA-11D colony analyser (System Science Co., Ltd, Tokyo, Japan) and were counted manually on other plates. The test results were considered positive when the mean numbers of treated revertant colonies exceeded the upper limit of background data from the negative control and when significant dose-related increases in the numbers of revertant colonies were reproducibly observed.
Positive control substances (concentrations in μg/plate) for reverse mutation tests.

AF-2: 2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide; AZI: sodium azide; 9AA: 9-aminoacridine; 2AA: 2-aminoanthracene.
The statistical analyses were performed using Dunnett’s multiple comparison (one-tailed) and linear regression methods. The number of revertant colonies of each bacterial strain at each dose was compared with that of the negative control in the presence and absence of metabolic activation, and differences between groups were initially identified using multiple comparisons (p < 0.05). Dose–response relationships were then analysed using linear regression (p < 0.05).
Chromosome aberration test
RHPMJ concentrations of 500, 250 and 125 μg/mL were used based on cell growth inhibition tests (incubation with or without S9 for 6 h and without S9 for 24 h). Positive control cells were treated with cyclophosphamide (5.0 µg/mL) in assays of metabolic activation. Two hours prior to the end of the incubation, colcemid (0.2 µg/mL) was added to stop cell mitosis. Subsequently, 100 chromosomes in the metaphase were selected from each slide using a microscope (200/concentration for each treatment and group) and structurally aberrant cells with and without gaps (+gap and −gap, respectively) were separately totalled. Chromosome aberrations were determined according to the frequency of the −gap cells with structural aberrations.
Differences in frequencies of cells with structural or numerical aberrations between the negative control and treatment groups were identified at each dose using the χ 2 test with Yates’ correction and were considered significant when p < 0.05 (one-tailed). Dose dependencies were identified using Cochran–Armitage trend tests and were considered significant when p < 0.05 (one-tailed, including negative controls).
Micronucleus test
Rats were randomly assigned to the negative control, treatment (500, 1000 and 2000 mg/kg BW) and the positive control groups (six rats/group). Based on the dosimetric analyses, the high-, medium-, and low-dose levels were set at 2000, 1000 and 500 mg/kg BW, respectively. RHPMJ was then dissolved in water for injection at 50, 100 and 200 mg/mL and administered at a volume of 10 mL/kg BW. The negative control rats were treated with the vehicle (water for injection). Treatments were performed twice via oral gavage at approximately 24-h intervals, although the positive control (cyclophosphamide) was administered via single oral gavage at a dose of 20 mg/kg BW. Mortality and clinical signs were recorded once daily. BWs were measured on the dosing day and then during specimen preparation. All rats were euthanized using carbon dioxide inhalation 23–24 h after the final doses, and their femurs were then removed. Foetal bovine serum was then infused into the bone marrow of the femur, and bone marrow cells were collected. The resulting cell suspensions were centrifuged, smeared on slides and then stained with acridine orange. For each prepared slide, 1000 immature erythrocytes (IEs, 2000/animal) and the micronucleated immature erythrocytes (MNIE) were observed, and the frequency of micronucleated immature erythrocytes (%MNIE) were calculated according to the formula

The ratio of immature erythrocytes (%IE) to total mature erythrocytes (MEs) was calculated based on 500 erythrocytes/slide (1000/animal) according to the formula

Differences in the frequency of micronuclei between the negative control, test substance and positive control groups were analysed using the Kastenbaum and Bowman method. 34 Frequencies of polychromatic erythrocytes were recorded, and differences between the negative control and test substance groups were analysed using Dunnett’s test (p < 0.05).
Ninety-day repeated oral dose toxicity study
RHPMJ was orally administered to rats (10/sex per group) at doses 100, 300 and 1000 mg/kg/day of BW/10 mL water for injection for 90 days. All animals were observed for mortality, general condition and clinical signs twice daily throughout the study. BWs and food intake were recorded once weekly during the treatment period. Functional observations were performed at week 12 including responses to visual, proprioceptive, auditory and pain stimuli, as well as aerial righting reflexes and hind limb landing foot splay. Ophthalmological examinations were conducted in the control and high-dose groups at week 13. Urine samples were collected from five animals per group at week 13 and were analysed for pH, protein, glucose, occult blood, colour and turbidity within 3 h using a Combur10Test®M stick (Roche Diagnostics GmbH, Mannheim, Germany) and urine chemistry analyses (Cobas U 411, Roche, Germany). Urine samples were also collected over a 24-h period, and their volume and specific gravity (gravimeter) were recorded (Vet360; Reichert, Inc., Depew, New York, USA).
All animals were fasted for approximately 18 h prior to necropsy. The blood samples were collected from the abdominal aorta under anaesthesia with isoflurane, and the organs were harvested after euthanasia by exsanguination. The following haematological parameters were analysed using an autoanalyser (ADVIA2120i; Siemens AG, Munich, Germany): total erythrocyte counts (red blood cell, RBC), haemoglobin (HB), haematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), platelet count (PLT), total leukocyte count (white blood cell, WBC), WBC differential count (neutrophils, lymphocytes, monocytes, eosinophils and basophils) and reticulocytes (Reti). Subsequently, the blood samples were mixed with 3.2% sodium citrate and centrifuged at 3000 r/min for 10 min to obtain the plasma. The prothrombin time (PT) and activated partial thromboplastin time (APTT) were evaluated using an automatic coagulation time meter (Coapresta 2000; Sekisui Medical Co., Ltd, Tokyo, Japan). Serum was isolated from non-anticoagulated whole blood and used for the biochemical analysis. Alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), blood urea nitrogen (BUN), creatinine (Crea), total bilirubin (T-Bil), total bile acid (TBA), total protein (TP), albumin (Alb), albumin/globulin (A/G) ratio, total cholesterol (T-chol), triglycerides (TG), glucose (Glu), P, Ca, Cl, Na and K were analysed using an automatic analyser 7180 (Hitachi) and electrolyte analyser (EasyLyte; Medica Corp., Bedford, Massachusetts, USA).
Individual organ weights were obtained for the brain, thymus, heart, liver, spleen, kidneys, adrenals, testes, epididymis, ovaries and uterus. Paired organs were weighed together, and the organ/BW ratios were calculated based on the fasting BWs on the day of necropsy. In addition, the pituitary, thyroid, parathyroid, salivary gland (submandibular, sublingual and parotid glands), lungs (including the bronchi), trachea, oesophagus, stomach, duodenum, jejunum, caecum, colon, rectum, pancreas, seminal vesicle, urinary bladder, mandibular and mesenteric lymph nodes, Harderian glands, aortas, sternum and femur including bone marrow, inguinal skin and mammary glands, skeletal muscle (thigh), sciatic nerve, spinal cord (cervical, mid-thoracic and lumbar) and gross lesions were harvested. The testes, eyes and optic nerves were fixed in Davidson fixative, while the other organs were fixed in 10% neutral-buffered formalin. Subsequently, histopathological evaluations were performed on paraffin wax-embedded tissues after sectioning and staining with haematoxylin and eosin. Specimens from rats in the control and high-dose groups and rats with gross lesions in the 300 mg/kg/day group were examined.
The statistical analyses were performed using the statistical analysis software (SAS) program (version 9.3; SAS Institute Inc., Cary, North Carolina, USA). BWs, food consumption, functional observations (hindlimb landing foot splay, grip strength and motor activity), urine volume, haematology, clinical chemistry and organ weights were compared using Bartlett’s test for homogeneity of variance (p < 0.05). Normally distributed data were compared using a one-way analysis of variance and significant differences were further tested using Dunnett’s test for multiple comparisons (p < 0.05 and 0.01, two-tailed). Multiple comparisons of non-normally distributed data were performed using Kruskal–Wallis test followed by Steel test (p < 0.05 and 0.01, two-tailed).
GLP and guidelines
All the genotoxicity studies were conducted in compliance with the ‘OECD Principles of Good Laboratory Practice’ of the OECD, ENV/MC/CHEM (98)17 (revised in 1997) and the ‘Ministerial Ordinance on Good Laboratory Practice for Nonclinical Safety Studies of Drugs’ Ordinance of the Ministry of Health and Welfare No. 21 of 26 March 1997 (last amended by the Ordinance of the Ministry of Health, Labour and Welfare No. 114 of 13 June 2008). The 90-day repeated oral dose toxicity studies were performed according to the ‘OECD Principles of Good Laboratory Practice’ of the OECD, ENV/MC/CHEM (98)17 (revised in 1997) and ‘Standards and Regulations for Chemical Testing Laboratories’ Notification No. 2014-240, Ministry of Environment, Republic of Korea (30 December 2014).
All genotoxicity studies were conducted in accordance with the Guidance on Genotoxicity Testing and Data Interpretation for Pharmaceuticals Intended for Human Use (Notification No. 0920-2, dated 20 September 2012 of the Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau, the Ministry of Health, Labour and Welfare). The 90-day repeated oral dose toxicity study was conducted in accordance with the ‘OECD Guideline for the Testing of Chemicals 408, Repeated Dose 90-day Oral Toxicity Study in Rodents’ (adopted: 21 September 1998).
Animal ethics
The micronucleus tests and 90-day repeated oral dose toxicity study protocols were reviewed and approved by the Institutional Animal Care and Use Committees of CMIC Bioresearch Center Co., Ltd and Biotoxtech Co., Ltd. These facilities are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Results
Chemical analysis
The total polyphenol content values determined using the Folin–Ciocalteu method were 3.23, 3.83 and 3.65 g CEs per 100 g of RHPMJ manufactured in 2012 (lot A), 2014 (lot B) and 2016 (lot C), respectively. Other chemical and nutritional components are shown in Online Supplementary Table S1.
Chromatographic patterns of samples from the different lots were compared to confirm the potential variation in the phenolic component of RHPMJ. A typical HPLC chromatogram for RHPMJ lot B under UV (280 nm) is shown in Figure 2(a). The peaks were observed at the following retention times (t
R): 21.0 (
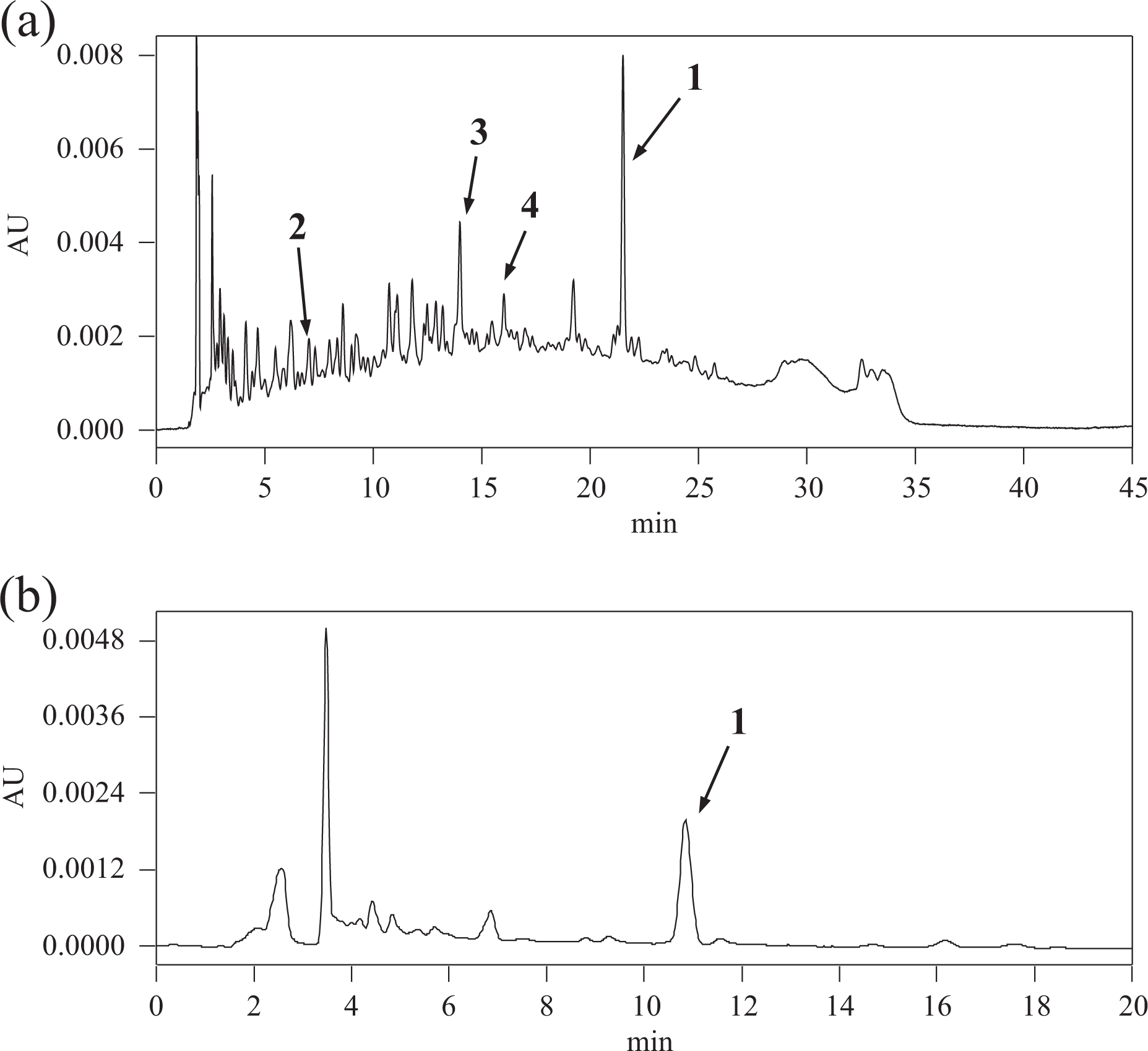
Typical HPLC chromatograms of RHPMJ lot B. (a) Pattern analysis (UV: 280 nm) and (b) quantitation (UV: 315 nm) of
As shown in Figure 2(b), the typical HPLC chromatograms for UV quantitation of
Validation parameters for quantitation of trans-tiliroside (

LOD: limit of detection; LOQ: limit of quantitation; RSD: relative standard deviation.
aIn the regression equation, x is the concentration of the analyte solution (µg/mL) and y is the peak area of the analyte.
bValues are the amount of the analyte injected into column.
cPrecision of the analytical method was tested using lot B (n = 3).
Content of trans-tiliroside (

RHPMJ: Rosehip polyphenol MJ; SD: standard deviation.
aValues are means ± SD of triplicate data.
Bacterial reverse mutation tests
The dosimetric analyses showed that the average number of revertant colonies of each tester strain in the test substance-treated groups was less than twice that of the negative control group. No dose-dependent increase in the number of revertant colonies was observed in the test substance-treated groups (data not shown). In the initial and confirmation tests (Tables 4 and 5), no significant dose responses were observed in any of the bacterial strains, regardless of the presence or absence of metabolic activation. Moreover, the average number of revertant colonies in the treatment groups was within the range of the upper limit of the negative control values. In both the main and confirmation tests, the average number of revertant colonies of each tester strain in the negative and positive control groups was within the range of the control values from background data of the testing facility (data not shown).
Reverse mutation tests without metabolic activation.a
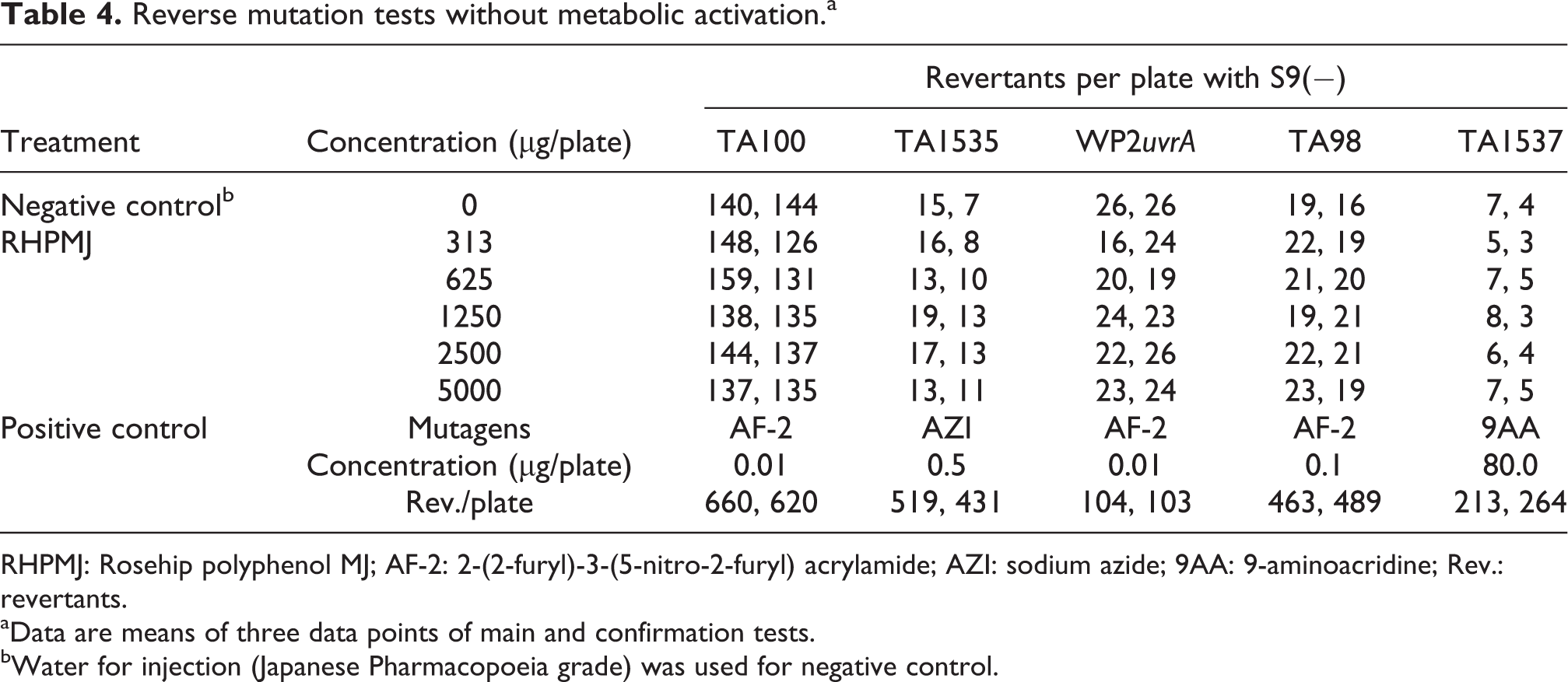
RHPMJ: Rosehip polyphenol MJ; AF-2: 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; AZI: sodium azide; 9AA: 9-aminoacridine; Rev.: revertants.
aData are means of three data points of main and confirmation tests.
bWater for injection (Japanese Pharmacopoeia grade) was used for negative control.
Reverse mutation tests with metabolic activation.a

RHPMJ: Rosehip polyphenol MJ; 2AA: 2-aminoanthracene; Rev.: revertants.
aData are means of three data points from main and confirmation tests.
bWater for injection (Japanese Pharmacopoeia grade) was used as negative control.
Chromosome aberration tests
No inhibition of cell growth or precipitation of test substances was observed. Although the number of cells with structural aberrations was dose-dependent in the 6-h treatment group without metabolic activation, this was within the range of the background data from negative controls in all the test groups. Moreover, no significant differences between the test groups and respective negative controls were observed (Table 6).
Chromosome aberration tests in 6- or 24-h treatment groups with or without metabolic activation.

CP: cyclophosphamide; RHPMJ: rosehip polyphenol MJ; RICC: relative increases in cell counts.
aWater for injection (Japanese Pharmacopoeia grade) was used as negative control.
Micronucleus test
The percentage of MNIEs (%MNIE) and IEs (%IE) did not differ significantly between the negative control and test substance groups (Table 7). Moreover, no abnormal signs were observed in the negative and positive control group rats or in those treated with the test substance at 500, 1000 or 2000 mg/kg/day, and the mean BW increases were comparable in all groups.
Micronucleus tests.

RHPMJ: rosehip polyphenol MJ; %MNIE: % micronucleated immature erythrocytes (based on 2000 IEs per animal); %IE: % immature erythrocytes (based on 1000 erythrocytes per animal); SD: standard deviation.
aValues are means ± SD.
bWater for injection (Japanese Pharmacopoeia grade) and cyclophosphamide were used as negative and positive controls, respectively.
cCyclophosphamide was administered once.
d p < 0.0l, compared with negative control.
Ninety-day repeated oral dose toxicity study
No abnormal test substance-related clinical signs or mortality were observed in the 100, 300 or 1000 mg/kg/day groups. In the 300 mg/kg/day group, loss of fur was observed in one male rat from day 66 to 90, and an open toe wound with damage to the nail, haemorrhage and crust formation was observed in one female rat from day 35 to 38. These changes were not considered to be related to the test substance. No clinical abnormalities were observed in the detailed examinations of male and female rats of the 100, 300 or 1000 mg/kg/day groups, and no significant differences in BWs or food consumption were observed between the control and treatment groups during the administration period (Figure 3(a) and (b)).

Effects of RHPMJ on BW and food consumption in male and female rats over administration period. Changes in (a) BW and (b) food consumption. Plots are means (n = 10). RHPMJ: rosehip polyphenol MJ; BW: body weight.
Similarly, no significant differences in visual, proprioceptive stimuli, auditory or pain responses or aerial righting reflexes were observed between the 100, 300 or 1000 mg/kg/day groups and the control group (data not shown). Moreover, no remarkable ophthalmological changes between animals of the control and 1000 mg/kg/day groups, and no differences in urinary parameters were observed between the treatment and control groups (data not shown).
The results of the haematological and clinical chemistry analyses are presented in Tables 8 and 9. No test substance–related changes were observed in the males or females in the 100, 300 and 1000 mg/kg/day groups, and the significant differences were small, lacked a dose–response relationship and, therefore, were not considered toxicologically significant. The absolute and relative organ weights for male and female rats are presented in Table 10 and did not differ between the 100, 300 and 1000 mg/kg/day treatment groups and the control group.
Haematological analyses of rats in 90-day repeated oral dose toxicity studies.a

RHPMJ: rosehip polyphenol MJ; RBC: red blood cell (total erythrocyte counts); HB: haemoglobin; HCT: haematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; PLT: platelet count; Reti: reticulocytes; WBC: white blood cell total leukocyte count; PT: prothrombin time; APTT: activated partial thromboplastin time; SD: standard deviation.
aAll values are means ± SD.
b p < 0.0 l compared with control group.
c p < 0.05 compared with the control group.
Serum chemistry of rats in 90-day repeated oral dose toxicity study.

RHPMJ: rosehip polyphenol MJ; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; BUN: blood urea nitrogen; Crea: creatinine; T-Bil: total bilirubin; TBA: total bile acid; TP: total protein; Alb: albumin; A/G: albumin/globulin; T-chol: total cholesterol; TG: triglycerides; Glu: glucose; SD: standard deviation.
aAll values are means ± SD.
b p < 0.05 compared with control group.
Absolute and relative organ weights of rats in 90-day repeated oral dose toxicity study.a
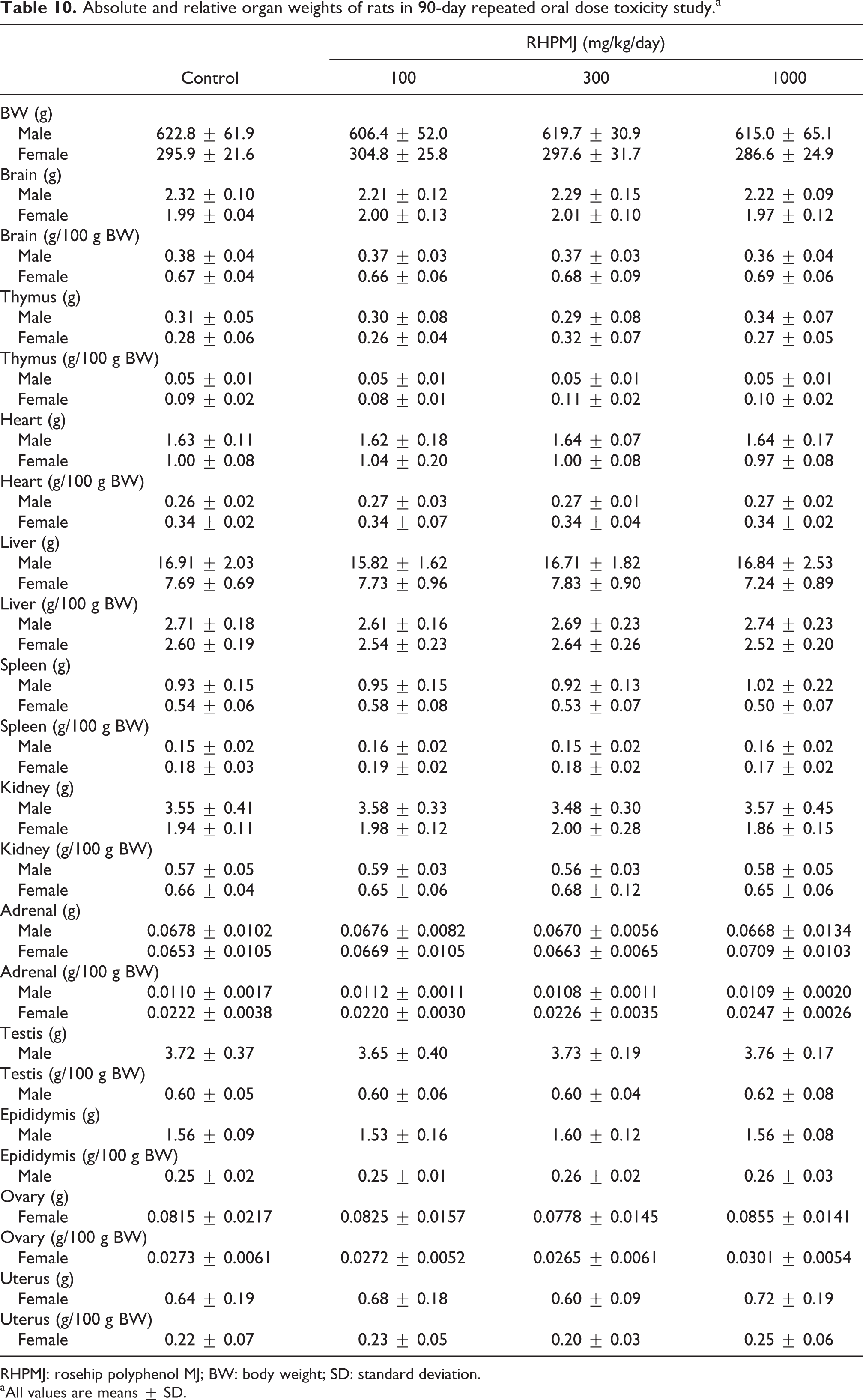
RHPMJ: rosehip polyphenol MJ; BW: body weight; SD: standard deviation.
aAll values are means ± SD.
No test substance–related macroscopic or microscopic findings were observed in animals of either sex from the 100, 300, and 1000 mg/kg/day groups. Macroscopic findings were considered incidental and unrelated to the test substance. In particular, a mammary gland adenoma was observed in one female in the 1000 mg/kg/day group. However, these have been observed previously in young Sprague-Dawley rats. 35 All microscopic observations of the organs and tissues were considered incidental and not toxicologically significant.
Discussion
Rose hip has been known for its medicinal properties since the times of Pliny the Elder (23–79 BC), and rose hip with or without the fruit (dried rose hip shells, Rosae pseudofructus cum/sine fructibus) has been used traditionally as a prophylactic and therapeutic medicine for the common cold and other infections and inflammatory diseases. Rose hip has a long history as an edible plant and, therefore, is considered highly safe. We developed RHPMJ, an aqueous ethanol extract of the fruit of R. canina, which was standardized based on the content of
The results of the polyphenol determination using the Folin–Ciocalteu method showed that the total polyphenol content in the three lots of RHPMJ was nearly constant. In addition, four known polyphenols (
The evaluation of genotoxicity potential is very important for the safety assessment of food, drugs and chemicals. Genotoxicity studies are designed to detect genetic damage, which may reflect teratogenic and tumorigenic potential. 37 In the present study, we investigated the mutagenic and clastogenic potential of RHPMJ using a battery of genotoxicity studies including bacterial reverse mutation, chromosome aberration and micronucleus tests.
The bacterial reverse mutation test is the most commonly used in vitro method for detecting mutagenic effects of genotoxicants. 38 –40 In these tests, S. typhimurium tester strains are used to detect base-pair substitutions (TA100 and TA1535) and frameshift (TA98 and TA1537) mutations. 41 In addition, base-pair substitutions are detected with a high sensitivity in the E. coli strain, WP2uvrA. 42 RHPMJ showed no mutagenicity, which was evident from the comparison of the revertant colonies/test plate and the revertant colonies/plate in the negative control. A test sample may be considered mutagenic when a twofold increase in the number of mutants is observed with at least any one of the concentrations tested. 43 For RHPMJ, the counts of the revertant colonies/plate of each concentration were not up to twofold that of the negative control. Moreover, metabolic activation by the rat liver S9 mix did not affect the number of revertant colonies in the RHPMJ-treated plate. Consequently, any mutagenic components were not produced by S9 mix, but further investigations are required to elucidate what kind of chemical reactions are occurred.
Then, we performed chromosome aberration and micronucleus tests, which have been widely used in primary screening studies of DNA damage at the chromosomal level. 44 –46 In the chromosome aberration test, dose-dependency was observed following a 6-h treatment without S9 mix group in the frequencies of cells with structural aberration. However, the frequencies were within the range of the background data of the negative control, and significant differences between the negative control and RHPMJ treated group were not observed. Since no statistically meaningful differences were observed under any test conditions and with RHPMJ treatments, the dose-dependency was assumed to be incidental. In the micronucleus test, the frequency of MNIEs and the ratio of IEs were calculated from 2000 IEs/animal and 1000 erythrocytes/animal, respectively. The RHPMJ-treated group did not show significant differences in the frequency of micronuclei compared with the negative control group. The current data of the in vitro chromosome aberration test and in vivo micronucleus test indicated that the RHPMJ had no clastogenic activity.
In a previous experiments, we assessed the acute and 28-day repeated oral dose toxicity of RHPMJ (non-GLP) and showed that the median lethal dose (LD50) and the maximum tolerated dose were greater than 5000 mg/kg in male and female animals, and no-observed-adverse-effect levels (NOAEL) were 3000 and 1500 mg/kg (male and female, respectively; unpublished data). In the present 90-day repeated oral dose toxicity study, the BWs, clinical observations and organ weights did not differ between the control and up to the 1000 mg/kg RHPMJ treatment groups, and no adverse effects or abnormalities were observed.
Previous clinical studies of the effects of rose hip on osteoarthritis, 2,14 –16 chronic pain, 18,19 skin ageing, 20 lifestyle-related diseases 21,33 and biomechanical functions of the knee joints 22 have demonstrated both efficacy and safety. Moreover, no serious adverse effects were caused by rose hip in any of these studies, although some reported mild gastrointestinal problems that may reflect the mode of administration or excess vitamin C in rose hip. Furthermore, keratitis has also been reportedly caused by rose hip intake. 47 Botanically, the seeds of rose hip are covered with stiff, sharply pointed non-barbed hairs, 48 which are highly irritating to the human skin. However, although keratitis was likely caused by these hairs, RHPMJ does not contain vitamin C or hairs, and no gastrointestinal symptoms of RHPMJ were reported in clinical studies.
In conclusion, our analysis revealed that the chromatographic pattern and content of
Supplemental material
Supplementary_Material_revised - Evaluation of genotoxicity and subchronic toxicity of standardized rose hip extract
Supplementary_Material_revised for Evaluation of genotoxicity and subchronic toxicity of standardized rose hip extract by A Nagatomo, M Oguri, N Nishida, M Ogawa, A Ichikawa, and Y Tanaka-Azuma in Human & Experimental Toxicology
Footnotes
Authors’ note
AN, MO, NN and YT-A are employees of Morishita Jintan Co., Ltd, Osaka, Japan. MO and AI are employees of CMIC Pharma Science Co., Ltd, Yamanashi, Japan.
Acknowledgements
The authors would like to thank Professor H Fukui and Dr M Ochiai at Gifu University for their efforts in identifying the varieties of rose hip from Chile and also would like to acknowledge Professor T Morikawa at Kindai University and Professor H Ito at Okayama Prefectural University for the advice on the qualitative analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All study experiments were sponsored by Morishita Jintan Co., Ltd, Osaka, Japan.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
