Abstract
Formaldehyde and methylene glycol may be used safely in cosmetics if established limits are not exceeded and are safe for use in nail hardeners in the present practices of use and concentration, which include instructions to avoid skin contact. In hair-smoothing products, however, in the present practices of use and concentration, formaldehyde and methylene glycol are unsafe. Methylene glycol is continuously converted to formaldehyde, and vice versa, even at equilibrium, which can be easily shifted by heating, drying, and other conditions to increase the amount of formaldehyde. This rapid, reversible formaldehyde/methylene glycol equilibrium is distinguished from the slow, irreversible release of formaldehyde resulting from the so-called formaldehyde releaser preservatives, which are not addressed in this safety assessment (formaldehyde releasers may continue to be safely used in cosmetics at the levels established in their individual Cosmetic Ingredient Review safety assessments).
Keywords
Introduction
In 1984, Cosmetic Ingredient Review (CIR) Expert Panel (Panel) published its original safety assessment of formaldehyde, 1 concluding that this ingredient is safe for use in cosmetics applied to the skin if free formaldehyde was minimized, but in no case >0.2%. This conclusion was based on the data from numerous human skin irritation and sensitization tests (number of patients ranging from 8 to 204) of cosmetic products (skin cleansers and moisturizers and a hair rinse) containing 0.2% formalin (37%, w/w aqueous formaldehyde solution). Except for a few mild, equivocal, or inconsistent reactions, the results of these tests showed that such products have little potential to irritate or sensitize the skin. The Panel also determined that it cannot be concluded that formaldehyde is safe in cosmetic products intended to be aerosolized.
The Panel rereviewed the safety assessment of formaldehyde and affirmed the original conclusion in 2003. 2
Since that rereview, methylene glycol has been listed as a cosmetic ingredient, and CIR has become aware of increasing uses of formaldehyde/methylene glycol in hair-smoothing products intended to be heated. In addition to the issues related to increasing uses and identification of methylene glycol as a cosmetic ingredient, the US Environmental Protection Agency (EPA) National Center for Environmental Assessment (NCEA) released a draft toxicological review of formaldehyde for external review on June 2, 2010, including interagency comments on an earlier draft of the document. 3 The NCEA risk assessment provides a comprehensive summary of the toxicological literature, including both human and animal studies and all the major exposure routes of concern (inhalation, ingestion, and skin contact). The US National Research Council (NRC) has released their review of the draft assessment. 4 Much of the significant new toxicology data are related to genotoxicity, carcinogenicity, and reproductive and developmental toxicity.
Data and analysis were provided by the Nail Manufacturer’s Council (NMC), the Professional Keratin Smoothing Council (PKSC), the Personal Care Products Council, and the American Chemistry Council. Additional data from the US Food and Drug Administration’s (FDA’s) adverse event reporting system and results of FDA laboratory product analyses are included.
In consideration of these additional data, the Panel has issued this amended safety assessment.
Chemistry
Formaldehyde–Formalin–Methylene Glycol
Formaldehyde, a gas, is not used in cosmetics in its pure, anhydrous form but is instead most commonly produced as an aqueous solution called formalin. 5 Formalin is industrially produced from methanol. First, a mixture of vaporized methanol and steam is passed over a catalyst bed, where the methanol is oxidized to formaldehyde gas. Since this reaction is highly exothermic, the gas stream is cooled directly after passing over the catalyst to prevent thermal decomposition. Next, the formaldehyde reacts with water in an absorption column, because formaldehyde in its pure, gaseous form is highly unstable. Formaldehyde quickly reacts with water to produce methylene glycol and, without a polymerization inhibitor (eg, methanol), polymethylene glycols via a series of reversible reactions (Scheme 1). In the absence of methanol, these reactions proceed to form a mixture of long-chain polymethylene glycols, which are referred to as paraformaldehyde.
Methylene glycol, as a pure and separate substance, is not commercially available but is instead produced as an aqueous solution called formalin, as previously denoted for formaldehyde. Methylene glycol is a geminal (gem) diol or a diol with both hydroxyl groups on the same carbon. Gem diols are typically unstable compounds. Indeed, methylene glycol exists only in aqueous solution, where it is stabilized by hydrogen bonding with water molecules. Thus, the high solubility of formaldehyde in water is due to the rapid hydration of formaldehyde to methylene glycol and the capacity of the aqueous solution to stabilize methylene glycol and small polymethylene glycols (ie, 2-10 methylene glycol units long). 6 The rate of the hydration reaction is very fast (the half-life of formaldehyde in water is 70 milliseconds), and the equilibrium between methylene glycol and formaldehyde strongly favors methylene glycol at room temperature and neutral pH. 7 The equilibrium is dependent on temperature, solution density, pH, and the presence of other solutes. Increased temperature favors formation of formaldehyde. Although the concentration of methylene glycol in formalin is much greater than formaldehyde, at room temperature, neutral pH stasis, this says nothing about the reversibility of this equilibrium shift or about the rate of dehydration when this stasis is disrupted (eg, formalin is exposed to air or a formulation containing formalin is heated). This reaction is reversible. The dehydration of methylene glycol to formaldehyde happens rapidly and can be catalyzed by lower pH. 8
The formation of the higher polymethylene glycols is much slower than the rates of hydration and dehydration and can be inhibited by methanol. Accordingly, a typical solution of formalin consists of water (∼40%-60%), methylene glycol (∼40%), methanol (∼1%-10%), small methylene glycols (eg, dimers and trimers; ∼1%), and a very small amount of formaldehyde (∼0.02%-0.1%). The multiple equilibria between these components favor methylene glycol at room temperature. 9 However, removal of water, increase in solution density, heating, reduction in pH, and/or the reaction of the small amount of free formaldehyde in the solution will drive the equilibrium back toward formaldehyde. 10 Moreover, a product formulated with either of the ingredients methylene glycol or formaldehyde actually contains an equilibrium mixture of the components: methylene glycol, polymethylene glycols, and formaldehyde. Although it can be pointed out that formaldehyde and methylene glycol are different and distinct molecules, the ever present equilibrium between the 2 makes this distinction of virtually no relevance to ingredient safety. 11 Due to the equilibria demonstrated previously, any aqueous formulation that reportedly contains formalin, formaldehyde, or methylene glycol actually contains both formaldehyde and methylene glycol. Accordingly, the ingredients formaldehyde and methylene glycol can be referred to as formaldehyde equivalents.
Under any normal conditions of cosmetic use, including at room temperature and above, methylene glycol is not stable in the gas phase and very rapidly dehydrates to formaldehyde and water. 12 Accordingly, heating a formulation containing formaldehyde or methylene glycol will primarily off-gas formaldehyde. For this reason, the hazards of formaldehyde equivalents in a heated solution are the same as the hazards of gaseous formaldehyde, since the solution so readily releases gaseous formaldehyde.
Formaldehyde Equivalents
Formalin, as previously described, is an aqueous solution of formaldehyde, methylene glycol, and polymethylene glycols, all in equilibria and often stabilized with methanol. Formalin, per se, is not listed as an ingredient in the International Cosmetic Ingredient Dictionary and Handbook (INCI Dictionary) but is often the material tested in safety studies (therefore representing formaldehyde/methylene glycol). Of special importance is an understanding of the meaning of percent formalin. “100% formalin” means an aqueous solution wherein formaldehyde has been added to water to the saturation point of these equilibria, which is typically 37% (by weight) formaldehyde equivalents in water. Accordingly, a 10% formalin solution contains approximately 3.7% formaldehyde equivalents. More specifically, an aqueous solution which is 3.7% of formaldehyde (by weight) relates directly to a solution which is 5.9% methylene glycol (because the molecular weight of formaldehyde is 30 g/mol and the molecular weight of methylene glycol is 48 g/mol).
All toxicity studies that are relied upon determining the current 0.2% limitation in cosmetic products are based on the idea of “free formaldehyde,” what we are now calling formaldehyde equivalents. However, it seems quite probable that this number actually meant 0.2% formalin. Accordingly, based on the average formalin solution being 37% formaldehyde equivalents, this represents a true limit of 0.074% formaldehyde equivalents.
The reader is reminded that the ingredients in this review are not to be confused with “formaldehyde releasers,” which are not analogous to formaldehyde or methylene glycol but release small amounts of formaldehyde over considerable intervals (eg, diazolidinyl urea), acting as preservatives.
Analytical Methods
Most commonly used analytical methods for qualitative and quantitative detection of formaldehyde are nonspecific to nonhydrated formaldehyde but can accurately describe formaldehyde equivalent presence and quantity. A typical method, for example, the method used by the Oregon Occupational Safety and Health Administration (OSHA) Laboratory, can detect formaldehyde equivalents present in a formulation, or released into the air, via a 2-stage processes: (1) derivatization of a sample with a hydrazine (which reacts with formaldehyde or methylene glycol, in a formulation sample or in an air sample) and (2) detection of the resultant hydrazone (ie, the reaction product of the hydrazine and formaldehyde) with a diode array, after separation on a column (eg, high-performance liquid chromatography [HPLC] separation followed by ultraviolet/visible [UV/Vis] light detection). 11 Accordingly, published values for “formaldehyde” levels should be taken to mean formaldehyde equivalents.
Although other formaldehyde/methylene glycol detection techniques are known, the methods used by OSHA are the most common methods and are what current regulations, globally, have been based on. These techniques would find that a typical formalin solution contains approximately 37% formaldehyde equivalents. Some may argue that using nuclear magnetic resonance (NMR) spectrometry techniques would demonstrate that this same formalin solution is only 0.037% formaldehyde. 13 This is a technically correct interpretation of the amount of nonhydrated formaldehyde molecules present in the static environment of an NMR sample tube. This scenario, however, exists only in the highly controlled experimental system where the conditions (room temperature, neutral pH, and closed NMR tube) maintain an artificially constant level of nonhydrated formaldehyde. This does not represent the conditions under which formaldehyde or methylene glycol are used in hair-smoothing products and as such drastically underestimates the exposure risk. In use, hair-smoothing treatments containing formaldehyde or methylene glycol involve elevated temperatures (eg, 450°F) and reduced pH formulations (eg, as low as pH = 4). 13 Further, the solutions are used in a system where the bottle is opened, the solution is poured, applied, and allowed to partially evaporate/off-gas. Focusing on the equilibrium between formaldehyde and methylene glycol in a closed system that artificially favors a liquid state is not representative of the conditions of use of these ingredients in hair-smoothing products.
An alternative technique has also been proposed for specifically addressing the vapor/gas present in the headspace above an aqueous formaldehyde/methylene glycol solution, which involves trimethylsilyl derivatization of those moieties present, followed by detection of the resultant derivatives. 13 However, the chemical specificity of this method is not conclusively defined. The resultant derivatives detected could have arisen from a variety of constituents present in the headspace. Furthermore, no standards were found, which validate the ability of this method to detect nonhydrated formaldehyde.
Cosmetic Use
As given in the INCI Dictionary, 14 formaldehyde functions in cosmetic products as a cosmetic biocide, denaturant, and preservative. According to the 2010 13th Edition of the INCI Dictionary, methylene glycol is reported to function as an artificial nail hardener. 14
In the FDA’s Voluntary Cosmetic Registration Program (VCRP), 15 there are 77 uses of formaldehyde and formaldehyde solution (formalin) reported. Since all these are probably the same ingredient as added to cosmetics, they are combined in Table 1. 2,15,16 Industry surveys of formaldehyde use concentrations and FDA reports yielded data are shown in Table 1. 16 –19 No uses of methylene glycol are currently reported to the VCRP, but the industry survey of use concentration included reports of methylene glycol in nail hardeners at concentrations ranging from 0.8% to 3.5% (corresponding to 0.5%-2.2% calculated as formaldehyde). 16 –19
Frequency and Concentration of Use of Formaldehyde, Formalin, and Methylene Glycol.
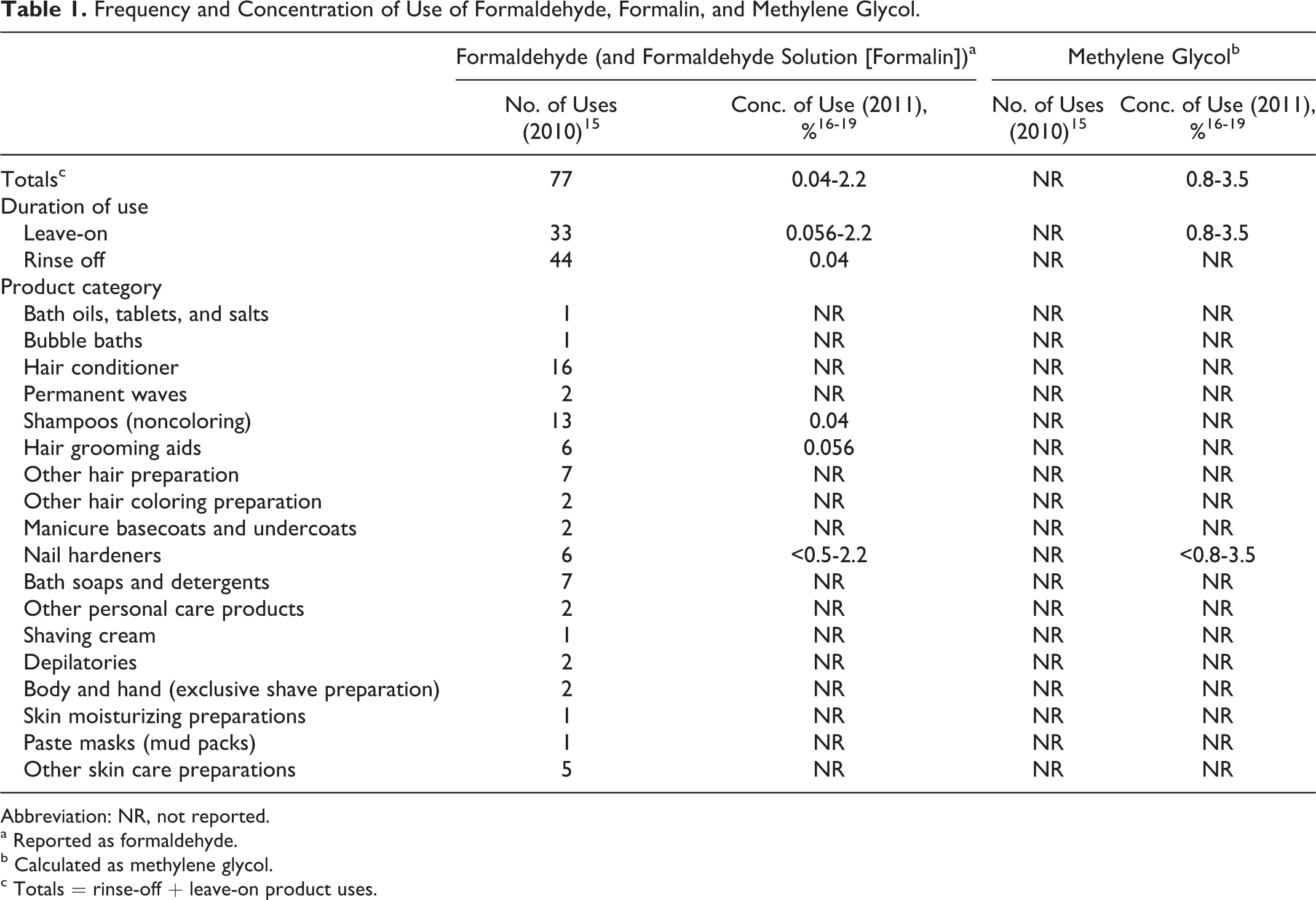
Abbreviation: NR, not reported.
a Reported as formaldehyde.
b Calculated as methylene glycol.
c Totals = rinse-off + leave-on product uses.
The Material Safety Data Sheet (MSDS) provided by Brazilian Blowout for their salon product also included methylene glycol. 20 The list of ingredients provided by the manufacturer is shown in Table 2, with methylene glycol listed at <5.0%.
List of Ingredients in Brazilian Blowout From the Brazilian Blowout MSDS Dated October 26, 2010.
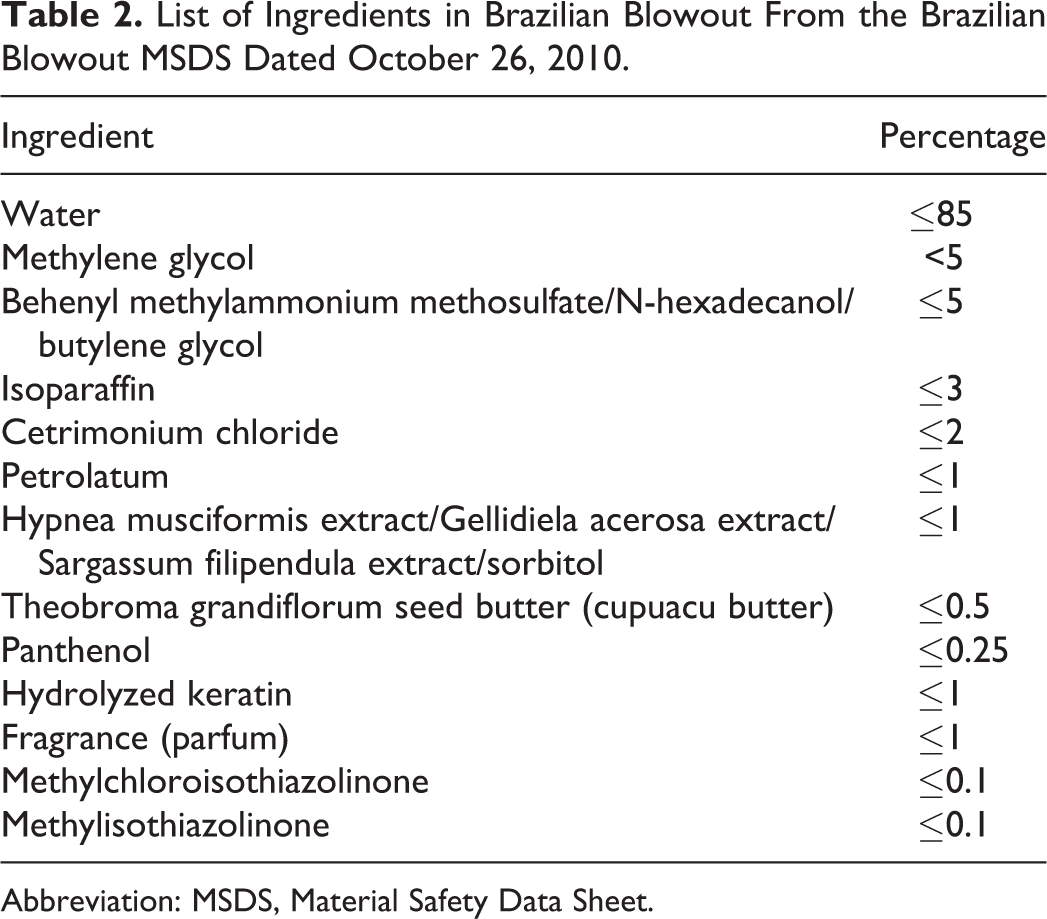
Abbreviation: MSDS, Material Safety Data Sheet.
From a high of 805 reported uses of formaldehyde/formalin in 1984, VCRP data from 2001/2002, 2006/2007, and 2009/2010 show that uses have decreased to less than 100 uses, as shown in Figure 1. The VCRP, however, does not include reporting of ingredients used in cosmetics labeled “for professional use.”
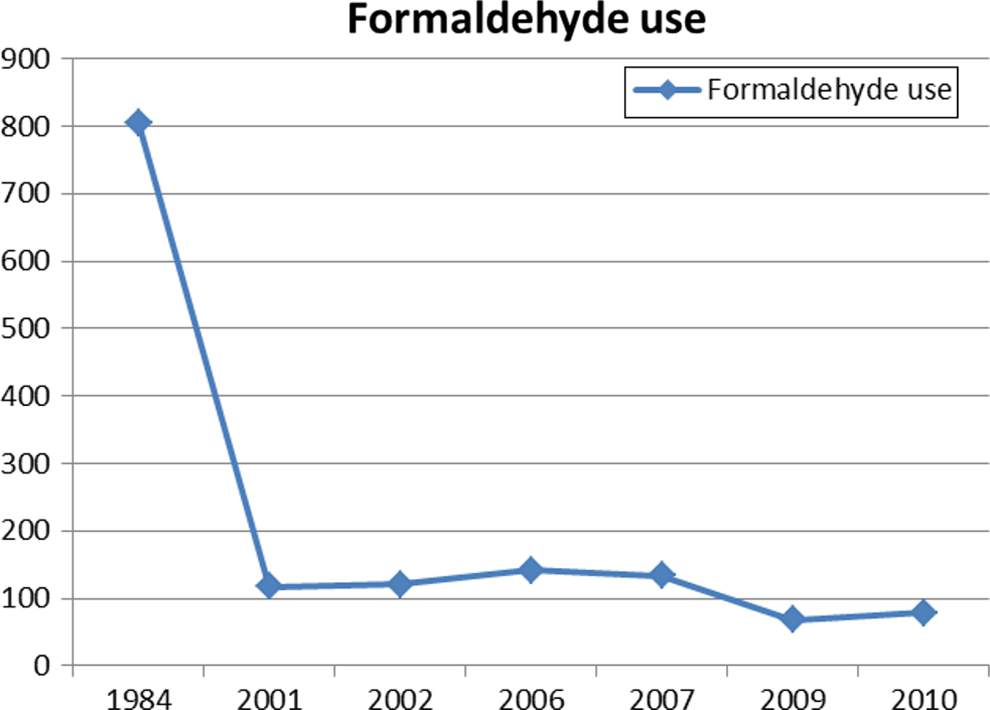
Declining use of formaldehyde in cosmetic products as reported to the Food and Drug Administration’s Voluntary Cosmetic Registration Program (FDA VCRP).
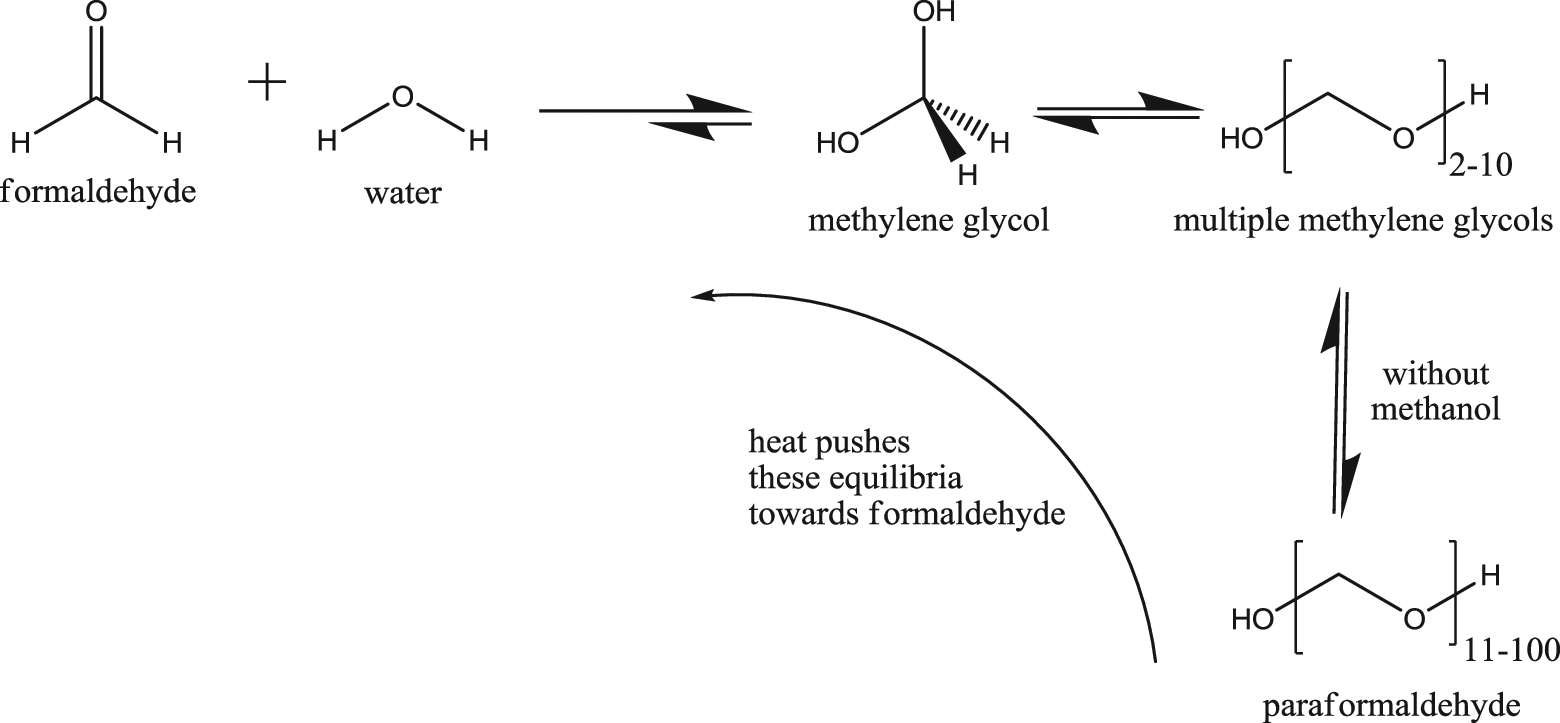
Equilibria in aqueous formaldehyde solutions such as formalin.
In Europe, formaldehyde is also permitted for use in cosmetics at concentrations ≤0.2% (the limit for oral hygiene products is ≤0.1%). 21 Products containing >0.05% formaldehyde must be labeled “contains formaldehyde.” The maximum authorized concentration in finished nail hardeners is 5%, provided that the product is labeled “Protect cuticles with grease or oil. Contains formaldehyde.” These limits are expressed as “free formaldehyde” or “calculated as formaldehyde.” Formaldehyde is prohibited for use in aerosol dispensers. Canada, Australia, China, and Association of Southeast Asian Nations have regulatory limits very similar to those of the European Union. 22 –27
Use of Formaldehyde/Methylene Glycol in Nail-Hardening Products
The FDA Guide to Inspections of Cosmetic Product Manufacturers 28 stated that nail hardeners often contain formaldehyde as the active ingredient and that the agency has not objected to its use as an ingredient of nail hardeners if the product (1) contained not more than 5% formaldehyde, (2) provided the user with nail shields that restrict application to the nail tip (and not the nail bed or fold), (3) furnished adequate directions for safe use, and (4) warned consumers about the consequences of misuse and potential for causing allergic reactions in sensitized users. Based on the comments given at the June 27 to 28, 2011 CIR Expert Panel meeting, it appears that nail shields are no longer supplied with nail hardeners in the United States because consumers did not use the shields.
As previously noted, in Europe, formaldehyde is permitted for use in nail hardeners at concentrations ≤5% “calculated as formaldehyde,” and the product label must instruct the user to protect cuticles with grease or oil. 29 If the formaldehyde concentration in the product exceeds 0.05%, the label must also state “contains formaldehyde.”
In the earlier CIR safety assessment of formaldehyde, 1 the CIR Expert Panel acknowledged reports of the use of formaldehyde in nail hardeners at a concentration of 4.5%. It now appears that methylene glycol is considered to be the appropriate ingredient name to use to describe formaldehyde/methylene glycol in nail hardeners. 14
Recent data provided by the NMC 30 indicated that to make a nail hardener nominally “1% formaldehyde”—which should be considered a typical marketplace level—a formulator would add 2.703% formalin (2.703% × 37% = 1%). Because of the well-recognized equilibrium relationship between formaldehyde and methylene glycol, the formaldehyde converts to methylene glycol. Therefore, a product with 2.703% formalin would contain 1.60% methylene glycol (2.703% × 59.2% = 1.60%). A recent survey of the US marketers conducted by the NMC indicated that formaldehyde/methylene glycol is not used in all brands of nail hardeners. 18 The survey results indicated that brands using methylene glycol/formaldehyde contain 0.7% to 1.85%, calculated as formaldehyde. Analyses of 2 finished nail hardener products (brand/origin not identified) indicated that they contained 1.9% and 2% formaldehyde equivalents, expressed as formaldehyde. 19
Food and Drug Administration recently reported finding 2.2% formaldehyde/methylene glycol in a nail-hardening product that was cited often in a compilation of customer self-reports from the Internet sites indicating adverse effects including skin irritation, burning sensation of nail beds and exposed skin, and pain 17,31 and 2 cases of eyelid dermatitis reported by a member of the CIR Expert Panel. The cases reported by the Panel member patched tested negative for 1% formaldehyde equivalents (calculated as formaldehyde) in water; higher concentrations (eg, 2%) were not tested.
Use of Formaldehyde/Methylene Glycol in Hair-Smoothing Products
The use of formaldehyde/methylene glycol containing hair-smoothing products largely appears to take place in salons, but home use is not precluded. Workplace surveys conducted by the Oregon OSHA uncovered a wide variety of ventilation approaches, including simply having a building HVAC system, propping the business’s doors open, or operating ceiling fans. 11
Although the purpose and mechanism of action of formaldehyde/methylene glycol in hair relaxers/straighteners is not well documented, formaldehyde (as part of a formalin solution) is known to induce a fixative action on proteins (eg, keratin). 32 This is at least in accord with formaldehyde’s function as a denaturant, in the classic sense of the term (ie, reacting with biological molecules, such as disrupting the tertiary structure of proteins, not just making liquids nonpotable). Purportedly, formaldehyde/methylene glycol hair-straightening formulations, such as Brazilian-style or keratin-based straightening products, maintain straightened hair by altering protein structures via amino acid cross-linking reactions, which form cross-links between hair keratins and with added keratin from the formulation. 33
One proposed reaction scheme involves (1) hemiacetal formation between a keratin hydroxyl group and formaldehyde, (2) reaction of 2 such hemiacetals, in a dehydration step, to form a methylene ether cross-link, and (3) formaldehyde elimination to finalize the new methylene cross-link. 34 Stoichiometrically, this proposed scheme purports that some of the formaldehyde that initially reacts with keratin is eventually released as formaldehyde during the hair-straightening process. Formaldehyde can react with multiple protein residue side chains, although the principal reactions are with the epsilon amino groups of lysine residues. 35 Besides proteins, formaldehyde is known to react with other biological molecules such as nucleic acids and polysaccharides. 36 The action of formaldehyde in intramolecular and intermolecular cross-linking of macromolecules can considerably alter the physical characteristics of the substrates.
The US OSHA has issued a hazard alert concerning hair-smoothing products that could release formaldehyde into the air. 37 The alert stated that OSHA investigations uncovered formaldehyde concentrations greater than OSHA’s limits of exposure. 38 One investigation reported such levels of formaldehyde even though the product was labeled “formaldehyde-free.” The hazard alert stated that formaldehyde gas presents a health hazard if workers are exposed, described the other chemical names to look for on the label that would signal reason for concern, and told businesses what to do to reduce exposure when using formaldehyde-releasing hair-smoothing products.
Canada issued health advisories informing consumers of the risks associated with hair-smoothing products containing excessive levels of formaldehyde and has recalled several such products. 39 –42 Hair-smoothing products with formaldehyde at levels >0.2% are not permitted for sale in Canada. 41
France’s health authority warned consumers and hairdressers against using hair-straightening treatments that contain high levels of formaldehyde and has removed a number of such products from the market. 43 Germany's Federal Institute for Risk Assessment advised against the use of hair-straightening products that contain formaldehyde in high concentrations. 44 The Irish Medicines Board, which is the competent authority in Ireland for cosmetics, took action to remove hair-smoothing products from the market if they contain greater than 0.2%, the level established by the European Commission. 45
Toxicokinetics
Formaldehyde is a highly water-soluble, reactive, rapidly metabolized chemical with a relatively short biological half-life. Inhaled formaldehyde is absorbed primarily in the respiratory epithelium lining the upper airways, where it undergoes extensive local metabolism and reactions with macromolecules. Based on the weight of the evidence, the NRC concluded that formaldehyde does not penetrate beyond the superficial layer of the nasopharyngeal epithelium and is unlikely to appear in the blood as an intact molecule, except possibly at concentrations high enough to overwhelm the metabolic capacity of the epithelium. 4 The NRC concluded that formaldehyde is not available systemically in any reactive form, and systemic effects are unlikely from the direct delivery of formaldehyde or methylene glycol to distal sites, except possibly in highly exposed people.
Toxicology
Previous CIR Safety Reports on Formaldehyde—Summary
In low amounts, formaldehyde is generated and present in the body as a normal metabolite, and as such or when taken into the body, it is rapidly metabolized by several pathways to yield carbon dioxide. It is a very reactive chemical. Not surprisingly, formaldehyde is an irritant at low concentrations, especially to the eyes and the respiratory tract. Formaldehyde exposure can result in a sensitization reaction. Under experimental conditions formaldehyde is teratogenic, mutagenic and can induce neoplasms. Perhaps the single most important attribute common to these toxic effects of formaldehyde is that they are all concentration/time dependent. A higher concentration or duration of exposure than that which produces irritation, for example, induces degenerative changes in the tissues exposed to it. There was no evidence that formaldehyde can induce neoplasia at concentration/time relationships that do not damage normal structure and function of tissues, even under laboratory conditions. From the Final Report on the Safety Assessment of Formal-dehyde
1
New clinical studies reviewed in 2003 confirmed that formaldehyde can be a skin irritant and sensitizer, but at levels higher than the 0.2% free formaldehyde upper limit established by the CIR Expert Panel. The developmental toxicity, genotoxicity, and carcinogenicity of high doses of formaldehyde were also confirmed in the new studies (published between 1984 and 2003). These studies demonstrated that there is a threshold effect; that is, high doses are required before any effect is seen. From the Published Re-Review of Formaldehyde
2
New Data on Safety of Formaldehyde
The US EPA NCEA released a 4-volume draft toxicological review of formaldehyde for external review on June 2, 2010, including interagency comments on an earlier draft of the document. 3 The US EPA is conducting this assessment to support the development of new chronic inhalation toxicity values for formaldehyde. Ultimately, the final versions of these values will be incorporated into the US EPA Integrated Risk Information System (IRIS).
The NRC recently released their review of US EPA’s draft assessment, 4 and their findings are also summarized subsequently, where appropriate. The NRC noted that the systemic delivery of formaldehyde may not be required for some of the systemic effects attributed to formaldehyde inhalation (eg, lymphohematopoietic [LHP] cancers and reproductive toxicity). Instead, systemic effects could be secondary, indirect effects of the local effects of exposure, including local irritation and inflammation, and stress.
This article provides a summary of the toxicological literature, including both human and animal studies and all the major exposure routes of concern (inhalation, ingestion, and skin contact). Much of the significant new toxicology data are related to genotoxicity, carcinogenicity, and reproductive and developmental toxicity. A comprehensive summary of the findings is presented in Tables 3 to 11.
Skin Irritancy/Sensitization Studies of Formaldehyde/Methylene Glycol in Test Animals.

Abbreviation: Ig, immunoglobulin.
Genotoxicity Inhalation Studies of Formaldehyde/Methylene Glycol in Test Animals.

Genotoxicity Inhalation Studies of Formaldehyde/Methylene Glycol in Human.

Nasal Tissue Studies of Formaldehyde/Methylene Glycol in Test Animals.

Abbreviation: NPC, nasopharyngeal cancer.
Epidemiological Studies of Formaldehyde/Methylene Glycol and Nasopharyngeal Cancers.
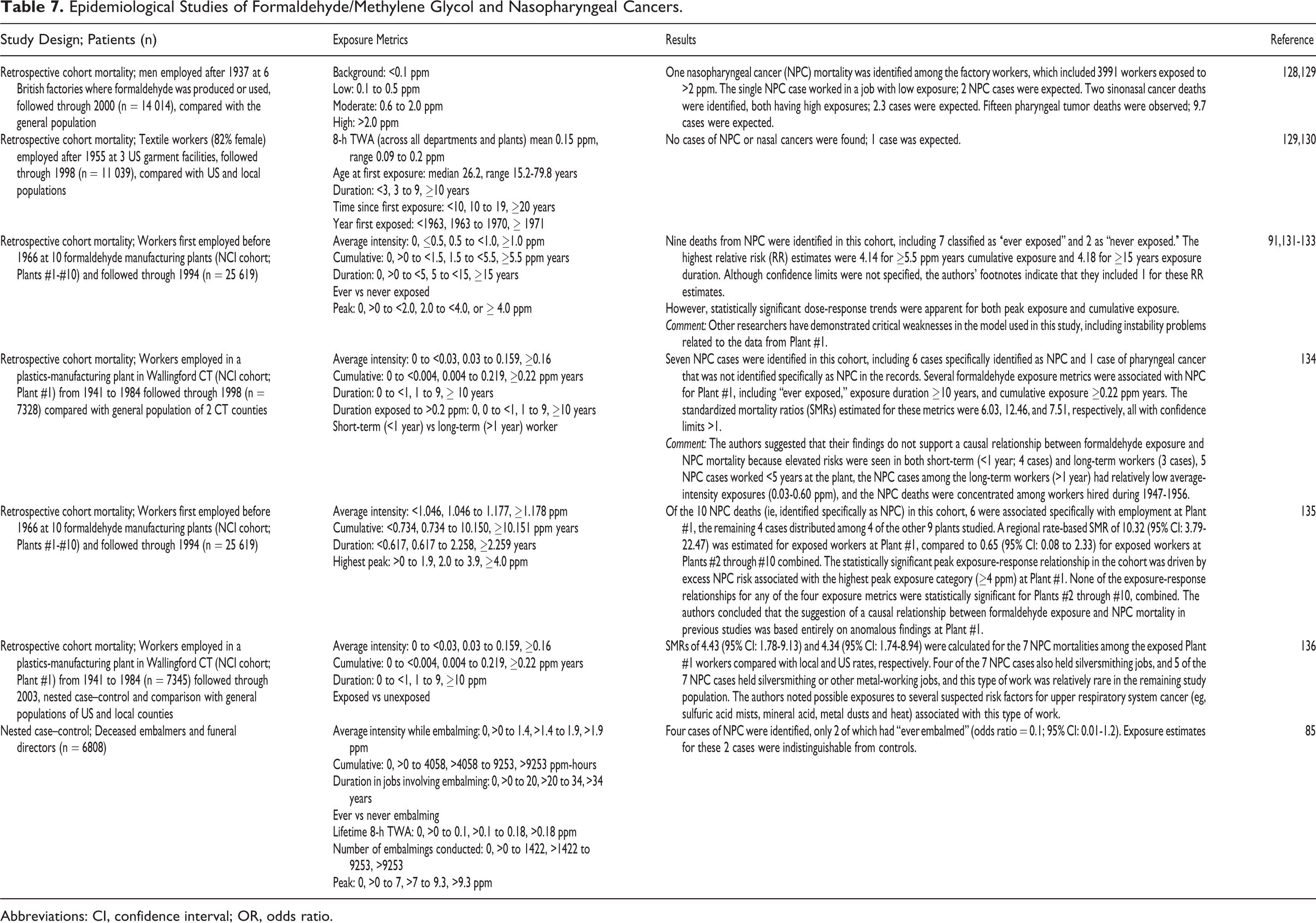
Abbreviations: CI, confidence interval; OR, odds ratio.
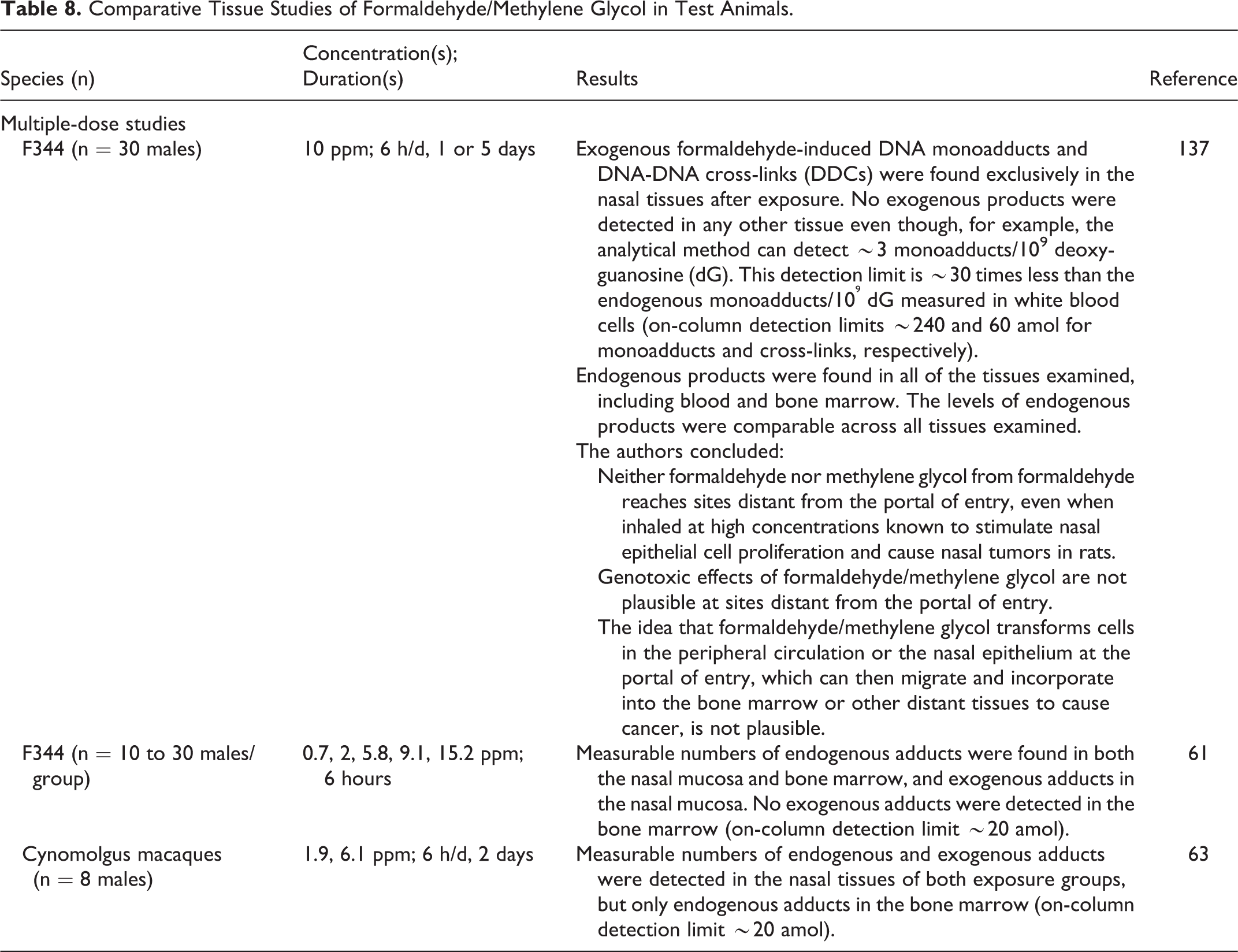
Comparative Tissue Studies of Formaldehyde/Methylene Glycol in Test Animals.
Epidemiological Studies of Formaldehyde/Methylene Glycol and Lymphohematopoietic Cancers.
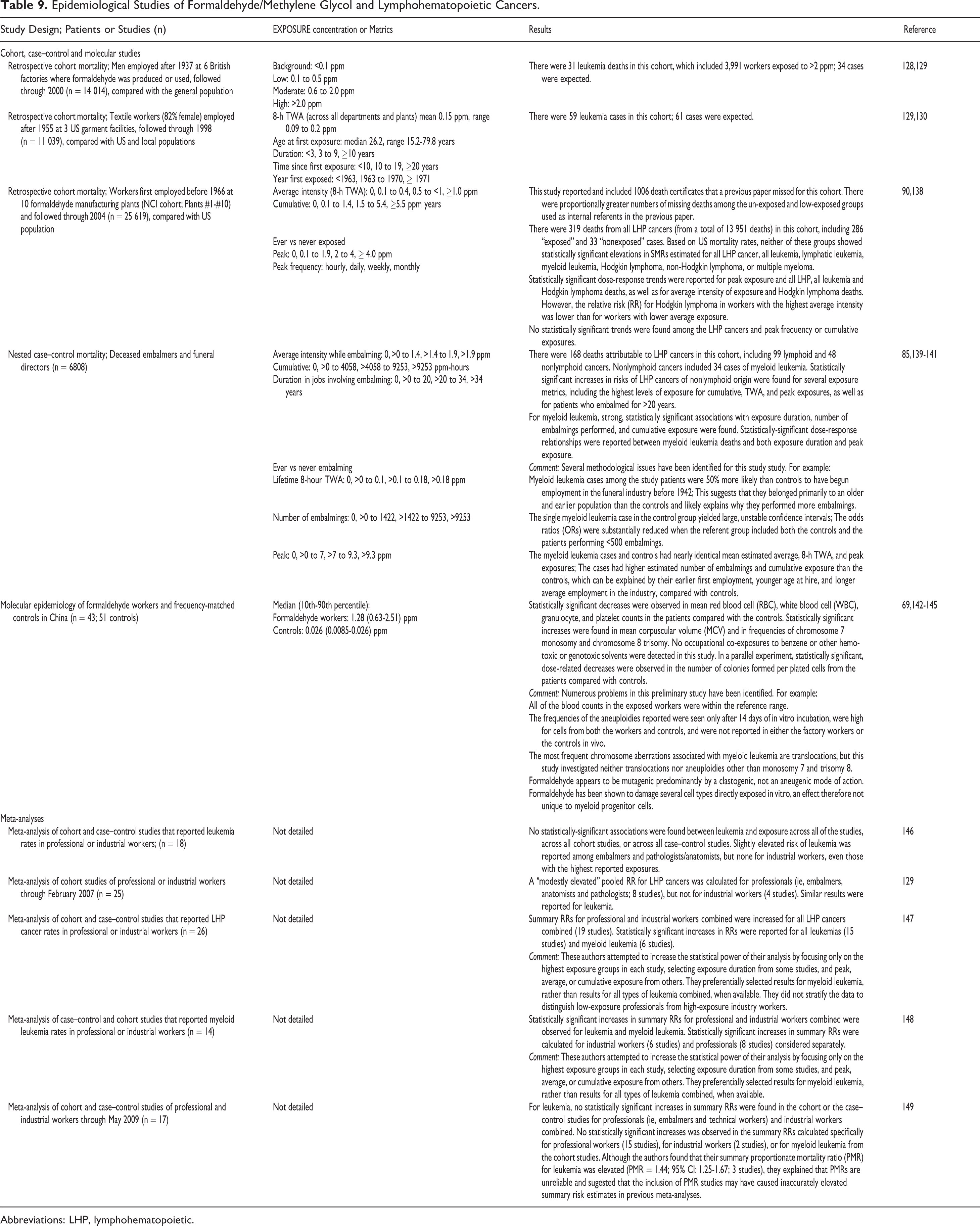
Abbreviations: LHP, lymphohematopoietic.
Reproductive and Developmental Toxicity Studies of Formaldehyde/Methylene Glycol in Test Animals.

Epidemiological Studies of Formaldehyde/Methylene Glycol and Reproductive Effects.

Abbreviation: CI, confidence interval.
Reproductive and Developmental Toxicity
Several potential modes of action of formaldehyde for reproductive and developmental outcomes have been suggested by animal studies, including endocrine disruption, genotoxic effects on gametes, and oxidative stress or damage. 46,47 However, the evidence for causality is weak. In addition, it is not clear that inhaled formaldehyde or its metabolites can penetrate fast the portal of entry or cross the placenta, blood–testis barrier, or blood–brain barrier.
The findings of studies on male reproduction generally used concentrations that result in significant weight loss and overt toxicity. There are no multigenerational tests for reproductive function. 3 These deficiencies, particularly for male reproductive effects, represent important data gaps in the assessment of risks of reproductive and developmental toxicity associated with inhalation exposures to formaldehyde. 4
The NRC noted that a small number of epidemiological studies 48 –51 suggest an association between occupational exposure to formaldehyde and adverse reproductive outcomes in women. 4
Genotoxicity
Clear evidence of systemic mutagenicity does not emerge from animal inhalation bioassays, despite the reactivity and mutagenicity demonstrated in isolated mammalian cells. 52 –54
Similarly, the evidence that inhaled formaldehyde may be directly genotoxic to humans and is systemically inconsistent and contradictory. 55 –60
Carcinogenicity
Nasopharyngeal Cancers
The NRC agreed with EPA that there is sufficient evidence from the combined weight of epidemiologic findings, results of animal studies, and mechanistic data of a causal association between the inhalation of formaldehyde and cancers of the nose, nasal cavity, and nasopharnyx. 4 Formaldehyde is highly reactive, readily forms DNA and protein adducts and cross-links, and is a direct-acting genotoxicant. Among the potential modes of action that have been considered for the development of nasopharyngeal cancers (NPCs) through the inhalation of formaldehyde in animal studies include direct mutagenesis of cells at the site of first contact and cytotoxicity-induced cell proliferation (CICP), which correlates with tumor incidence. 61 –68
The subchronic or chronic inhalation of formaldehyde at high concentrations (eg, ≥6 ppm) can clearly cause NPCs in mice and rats. However, there is still debate in the scientific community about whether this effect should be considered to be a nonthreshold effect or a threshold effect in cancer risk assessments.
The NRC concluded that these 2 primary modes of action contribute to formaldehyde-induced carcinogenicity in nasal tissues, including mutagenicity and CICP. 4 A mutagenic mode of action is generally the reason for adopting the default low-dose linear extrapolation methods in a quantitative cancer risk assessment. However, the NRC noted that formaldehyde is endogenous, that nasal tumors are rare in both rats and humans, and that no increases in tumor frequency are also observed in animal studies at formaldehyde concentrations that do not cause cytotoxicity. Further, the animal studies reveal a substantial nonlinearity in dose–response relationships among formaldehyde uptake, cytotoxicity, cell proliferation, and tumor formation.
Thus, the NRC recommended that the quantitative assessment of the risks of formaldehyde-induced NPCs incorporate the nonlinear phenomenon of CICP as well as the mutagenicity of formaldehyde. 4
Lymphohematopoietic Cancers
The 3 proposed modes of action by which formaldehyde exposure may cause leukemia include
69
: Transport of formaldehyde/methylene glycol from the portal of entry through the blood to the bone marrow, followed by direct toxic action to hematopoietic stem cells in the marrow. Direct toxic action of formaldehyde/methylene glycol on circulating blood stem cells and progenitors at the portal of entry, followed by return of the damaged cells to bone marrow. Direct toxic action of formaldehyde/methylene glycol on primitive pluripotent stem cells at the portal of entry, followed by migration of damaged cells to bone marrow.
Similarly, direct toxic action of formaldehyde/methylene glycol on lymphocytes in mucosa-associated lymphoid tissues at the portal of entry may cause lymphoid cancers. 3
Remarkably little evidence from animal studies indicates that formaldehyde exposure can cause LHP cancer.
Studies have consistently failed to find elevated levels of free formaldehyde or methylene glycol in the blood of exposed humans and animals, or DNA-protein cross-links (DPCs) in the bone marrow of exposed animals. 70 Further, formaldehyde is a highly reactive, rapidly metabolized chemical yielding short-lived DPCs and DNA-adducts that are amenable to rapid reversal and repair. 71,72 These observations are consistent with conventional wisdom, which has been that the expected sites of action of formaldehyde are limited to portals of entry (eg, nasal epithelium), and would not likely include distal sites, such as the bone marrow, where leukemias originate. 70,73 –75 Although several possible modes of action have been postulated to explain associations between LHP cancers and formaldehyde exposure in epidemiological studies, little scientific evidence supports these hypotheses, and there is some recent evidence against them. Thus, these proposals remain speculative and continue to represent a highly controversial topic in the scientific community.
The NRC noted that little is known about the potential modes of action by which formaldehyde might cause LHP cancers, other than mutagenicity. 4 A mechanism that would explain the occurrence of LHP cancers has not been established, the epidemiological data are inconsistent, the animal data are weak, and there is a growing body of evidence that formaldehyde is not available systemically in any reactive form. Further, the lack of consistency in exposure–response relationships between several exposure metrics and the LHP cancers in the epidemiological data could reflect the absence of causal mechanisms associating these cancers with formaldehyde exposure.
Irritation and Sensitization
As noted in the original safety assessment of formadehyde, 1 aqueous formaldehyde/formalin solutions can irritate the skin and cause contact urticaria and allergic sensitization in both occupationally and nonoccupationally exposed persons. The North American Contact Dermatitis Group reported a 9% incidence of skin sensitization among 4454 patients exposed to formaldehyde in aqueous solution. 76 Aqueous formaldehyde solutions as low as 0.01% can elicit skin responses in some sensitized persons under occlusive conditions. Most sensitized individuals can tolerate repeated topical axillary application of products containing up to 0.003% aqueous formaldehyde solution on normal skin. 77 Cosmetic products containing 0.0005% to 0.25% formalin (0.000185%-0.0925% calculated as formaldehyde) were essentially nonirritating and nonsensitizing in 1527 patients in 18 studies summarized in Table 5 of the original safety assessment. 1
Recent reviews addressing the human irritation and sensitization potential for aqueous formaldehyde/formalin solutions are consistent with the observations reported in the original assessment. 78,79
Healthy volunteers (n = 30; ≥18 years old) of either sex were exposed to 11 personal care products and 2 controls (ie, deionized water and 0.3% sodium lauryl sulfate) using an occlusive patch-testing protocol. 80 The products included 3 keratin hair straighteners containing methylene glycol (concentration not reported). All of the products were diluted to 8%, presumably with deionized water, before applying 0.2 mL of the diluted product to Webril disks. Note that, based on the manufacturer’s directions, hair straighteners are applied undiluted to the hair. The patches were applied to the skin of the upper arms of each patient and left in place for 23 hours, and removed and examined during the 24th hour, for 4 consecutive days. Each patient was exposed to each of the 11 products and 2 controls on patches applied to the same site of the skin each day. The specific site of application for each product/control varied from patient to patient, depending on the random assignment of each patient to 1 of the 5 groups. None of the diluted products or the negative control elicited any more than minimal erythema throughout the study. In contrast, the positive control elicited substantial erythema.
Clinical Use
Adverse Event Reporting
Nail-Hardening Products
A compilation of 33 customer self-reports from Internet sites and blogs of nail-hardening products indicated adverse effects including skin irritation, burning sensation of nail beds and exposed skin, severe finger pain, scabbing under the nails, and drying, flaking, splitting, crumbling, or peeling of the nails. 31 Two additional reports noted that the product contained formaldehyde and has a strong odor, without noting any other adverse effects. Three reports indicated that the product contained 4% to 4.5% formaldehyde.
Hair-Smoothing Products
Canada
Some 50 to 60 individuals have reported adverse reactions to Health Canada resulting from use of hair-smoothing products containing formaldehyde. These reports concerned burning eyes, nose, throat, and breathing difficulties, with 1 report of hair loss, 41 but additional reports also were received of headache, arthritis, dizziness, epistaxis, swollen glands, and numb tongue (Health Canada, personal communication).
United States
The Center for Research in Occupational and Environmental Toxicology (CROET) at the Oregon Health Sciences University (OHSU) has received numerous phone calls and e-mails from stylists from around the United States since first posting an alert on a hair product on September 16, 2011. 11 Many of the stylists reported health symptoms associated with the use of this product at work. The health symptoms reported include the following: burning of eyes and throat, watering of eyes, dry mouth, loss of smell, headache and a feeling of “grogginess,” malaise, shortness of breath and breathing problems, a diagnosis of epiglottitis attributed by the stylist to their use of the product, fingertip numbness, and dermatitis. Some of these effects were also reported to have been experienced by the stylists’ clients. Center for Research in Occupational and Environmental Toxicology also received e-mails from persons who report hair loss after having the treatment. Oregon OSHA has received similar, although generally less detailed, reports from individuals who have contacted the agency as a result of recent media coverage.
The US OSHA recently issued a Hazard Alert and identified safeguards that should be in place to keep formaldehyde concentrations below the US OSHA occupational exposure limits. 37
The FDA has been notified by some state and local organizations of reports from salons about problems associated with the use of Brazilian Blowout, a product used to straighten hair. 81 Complaints include eye irritation, breathing problems, and headaches. State and local organizations with authority over the operation of salons are currently investigating these reports.
The FDA adverse reporting system includes 33 adverse event reports from use of hair-smoothing and -straightening products from hair stylists, their customers, and individual users from September 29, 2008 through March 1, 2011. 82 The results clearly link the use of formaldehyde/methylene glycol-containing hair-smoothing products to clinical signs and symptoms that would be expected from the vaporization and inhalation of toxic levels of this ingredient. These reported effects include irritation of the eyes, nose and throat, nasal discharge, nose bleeds, congested sinuses, hoarseness, persistent coughing, bronchitis, difficulty breathing, feeling of pressure, tightness, or pain in chest. Two reports note inhalation pneumonitis in a professional hair stylist. Other complaints include headache, dizziness, fainting, and vomiting. Reported effects potentially attributable to direct contact with these products include irritation, inflammation, or blistering of the skin, especially on the scalp and hair loss. In addition to these 33 reports, there were 7 reports of hair loss that did not indicate whether other possible adverse effects also occurred.
Risk Assessments
Carcinogenicity
In 2006, the International Agency for Research on Cancer (IARC) 83 concluded that there was sufficient epidemiological evidence that formaldehyde causes NPC in humans and strong but not sufficient evidence for a causal association between leukemia and occupational exposure to formaldehyde. They also elevated their evaluation of formaldehyde from probably carcinogenic to humans (Group 2A) to carcinogenic to humans (Group 1).
In 2009, IARC
84
updated their evaluation to conclude that there is sufficient evidence for a causal association between leukemia, particularly myeloid leukemia, and occupational exposure to formaldehyde. This conclusion was based primarily on: The statistically significant association between embalming and myeloid leukemia, including statistically significant trends for cumulative years embalming and peak formaldehyde exposure.85 The levels of chromosome 7 monosomy and chromosome 8 trisomy in myeloid progenitor cells and hematological changes in formaldehyde exposed workers.69
The IARC Working Group was almost evenly split on the prevailing view that the evidence was sufficient for formaldehyde causing leukemia in humans. 84
The US National Toxicology Program (US NTP) concluded that formaldehyde is known to be a human carcinogen based on epidemiological reports indicating that exposures are associated with nasopharyngeal, sinonasal, and LHP cancers and data on mechanisms of carcinogenicity from laboratory studies. 86 –88
In 1991, US EPA classified formaldehyde as a B1 carcinogen (ie, a probable human carcinogen), based on limited evidence in humans, and sufficient evidence in animals. 89 They estimated an upper bound inhalation cancer unit risk of 1.6 × 10−2 per ppm (1.3 × 10−5 per µg/m3), using a linearized multistage, additional risk procedure to extrapolate dose–response data from a chronic bioassay on male F344 rats. An upper bound 10−6 human cancer risk would be associated with continuous inhalation of 0.06 parts per billion (ppb; 63 ppt) formaldehyde over a lifetime, based on this unit risk.
Recently, the US EPA proposed to identify formaldehyde as carcinogenic to humans. 3 They proposed an upper bound inhalation cancer unit risk of NPC, Hodgkin lymphoma, and leukemia, combined, using log-linear modeling and extra risk procedures to extrapolate cumulative exposure estimates from the epidemiological studies. 90 The NRC agreed that the Hauptmann et al’s study 91 of the NCI cohort is the most appropriate for deriving cancer unit risk estimates for respiratory cancers and other solid tumors but noted that this study is being updated. 4 The update will likely address the deaths reported to be missing from this study. 90 However, the NRC explicitly did not recommend that US EPA wait until the release of the update to complete its assessment.
Noncancer Effects
In 1990, US EPA published a chronic reference dose of 0.2 mg/kg/d for oral exposure to formaldehyde, based on the results of a 2-year bioassay in rats. 89,92 Formaldehyde (methylene glycol/formaldehyde) was administered to Wistar rats (70/sex/dose) in drinking water, yielding mean doses of 0, 1.2, 15, or 82 mg/kg/d for males and 0, 1.8, 21, or 109 mg/kg/d for females. Severe damage to the gastric mucosa was observed at 82 and 109 mg/kg/d in males and females, respectively, but no tumors were found. In this study, the no observed adverse effect level (NOAEL) was 15 mg/kg/d.
The US EPA released a draft risk assessment for formaldehyde for public comment and review by the NRC. 3 They proposed a chronic reference concentration for formaldehyde exposure by inhalation, based on 3 “cocritical” epidemiological studies. These studies reported associations between formaldehyde exposure and increased physician-diagnosed asthma, atopy, 93 and respiratory symptoms 94 and decreased pulmonary peak expiratory flow rate 95 in residential populations, including children. The NRC agreed with US EPA’s assessment of a causal relationship between formaldehyde and respiratory effects, except for incident asthma based on one of the “cocritical” studies. 4,93
Exposure Assessments
Formaldehyde is ubiquitous in both indoor and outdoor air. Substantial sources of airborne formaldehyde include both natural and anthropogenic sources. Formaldehyde concentrations are generally greater in urban air than in agricultural areas and greater in indoor air than in outdoor air. 3,4,83,96,97 It is estimated that the general population is exposed to an average of 0.016 to 0.032 ppm formaldehyde in indoor air. 98 In addition, formaldehyde is a natural metabolic intermediate in humans and other animals and is, thus, normally present in all tissues, cells, and bodily fluids. 96 The concentration of endogenous formaldehyde in the blood of rats, monkeys, and humans is about 0.1 mmol/L. 99,100 Endogenous tissue formaldehyde concentrations are similar to genotoxic and cytotoxic concentrations observed in vitro. 70 In addition, formaldehyde is likely present normally in exhaled breath at concentrations of a few ppb. 4
Standards and Guidance for Formaldehyde Inhalation Exposures
US OSHA Enforceable Standards
38
8-hour threshold time-weighted average (Threshold-TWA) for Hazard Communication Requirements 0.1 ppm 8 hour action level (AL-TWA) 0.5 ppm 8-hour permissible exposure limit (PEL-TWA) 0.75 ppm 15-minute short-term exposure limit (STEL-TWA) 2 ppm
The 8-hour Threshold-TWA is the time-weighted average concentration (0.1 ppm) above which employers are required to meet the US OSHA’s hazard communication requirements. 38
US National Institute of Occupational Health Recommended Exposure Limits
10-hour recommended exposure limit (REL-TWA) 0.016 ppm 15-minute Recommended STEL (REL-STEL-TWA) 0.1 ppm
The US National Advisory Committee for Acute Exposure Guideline Levels Committee
Acute Exposure Guideline Level 1 (AEGL-1) 0.9 ppm The AEGL-1 was based on the NOAEL for eye irritation in a study in which 5 to 28 healthy participants previously shown to be sensitive to 1.3 or 2.2 ppm formaldehyde were exposed eye-only for 6 minutes to 0, 0.35, 0.56, 0.7, 0.9, or 1.0 ppm.
103
Subjective eye irritation responses ranged from none to slight at 0, 0.35, 0.56, 0.7 and 0.9 ppm. The 0.9 ppm AEGL-1 was applied across all acute exposure durations (10 minutes to 8 hours) because several studies show that there is adaptation to irritation at such concentrations and because in the absence of exercise, there are no decrements in pulmonary function parameters in healthy or asthmatic patients inhaling 3 ppm for 3 hours.
104
–106
American Conference of Governmental Industrial Hygienists
Threshold Limit Value-Ceiling (TLV-C) 0.3 ppm.
World Health Organization
30-minute average indoor air guideline 0.08 ppm
Formaldehyde Exposures During use of Nail Products
Time-weighted average formaldehyde exposures of nail technicians and customers were measured simultaneously, during normal operations at 30 nail salons throughout California in winter and summer. 109,110 Nail hardeners containing formaldehyde were used in some of these salons and other products containing formaldehyde resins were used in most, if not all, of the salons during the study. 109 2,4-Dinitrophenylhydrazine (DNPH)-treated silica gel absorption tubes and high-flow pumps were used to collect the samples. One sample inlet tube was placed close to the technician’s breathing zone, and another close to the customer’s breathing zone during the application of the nail products. A third sampler was placed in the salon about 10 feet from the work station to collect “area samples” to measure concentrations in the salon during the application of the nail products. A fourth sampler was placed inside the salon early in the morning before the salon opened, inside during the first 2 hours the salon was open, or outside the salon while the salon was open, to provide background data. Preliminary air samples were collected from 2 office buildings for comparison.
Most of the air samples were collected for approximately 4 hours, and some for about 2 or 8 hours. 109 The samples were analyzed using HPLC, in accordance with US EPA method TO-11. 110 The measured concentrations were used to calculate 8-hour TWAs.
The authors reported 8-hour TWA formaldehyde concentrations in the breathing zones ranging from 0.0032 to 0.065 ppm (median = 0.01 ppm; mean = 0.0187 ppm; standard deviation [SD] = 0.0187 ppm) during the application of the nail products. 110 The corresponding area concentrations ranged from 0.0038 to 0.06 ppm (median = 0.01 ppm; mean = 0.0196 ppm; SD = 0.0195 ppm). The background concentrations, pooled, ranged from 0.0023 to 0.12 ppm (0.021-0.12 ppm early morning before opening; 0.014-0.081 ppm during first 2 hours after opening; 0.0023-0.013 ppm outside; overall: median = 0.014 ppm; mean = 0.033 ppm; SD = 0.038 ppm). The concentrations ranged from 0.015 to 0.021 ppm (mean = 0.018 ppm) in 1 office building and was 0.043 ppm in the other office building. The authors did not determine the sources of the formaldehyde measured in the background samples.
Thus, the reported 8-hour TWA formaldehyde concentrations in the breathing zones during the application of the products appear to be indistinguishable from the salon area concentrations and comparable to the background concentrations. In addition, the reported concentrations measured in the breathing zone, area, and outside background locations were uniformly lower than standards for formaldehyde, including the US OSHA PEL-TWA (0.75 ppm), AL-TWA (0.5 ppm), and Threshold-TWA (0.1 ppm).
Of the 7 remaining inside background concentrations (collected during the first to hours after opening), 1 exceeded the Threshold-TWA, and none exceeded the PEL-TWA, AL-TWA, or AEGL-1.
In another study, aluminum foil over a wooden support was used as the substrate for a nail-hardening product in a chamber (1.43 m3) under 2 conditions: “Typical:” 70ºF, 1 air change/h; “Elevated:” 80ºF, 0.3 air changes/h. 111 Formaldehyde concentrations were measured at 5-minute intervals in the chamber air over a 10.5-hour period. The nail hardener (15 mg/cm2) was painted on 70 cm2 of the surface of the substrate (>7 times the total surface of nails on the on a person’s 10 fingers, assuming ∼1 cm2/nail). The peak chamber air concentrations (5-minute samples) were 0.15 to 0.6 ppm under the “Typical” conditions and 0.2 to 0.24 ppm under the “Elevated” conditions. The peak concentrations measured in the chamber in this study are not directly comparable to the OSHA/ACGIH/WHO standards and guidelines, because they are not estimates of the concentrations of formaldehyde in the breathing zones of a customer or manicurist over relevant exposure durations. In any case, the 5-minute peak concentrations in the chamber were all about an order of magnitude less than the 15-minute STEL-TWA of 2 ppm.
Formaldehyde Exposure During Use of Hair-Smoothing Products
Air samples during use of hair-smoothing products were measured in 6 separate studies. The results are summarized below and in Table 12.
Measured Formaldehyde (Form) Levels During Use of Hair-Smoothing Products.
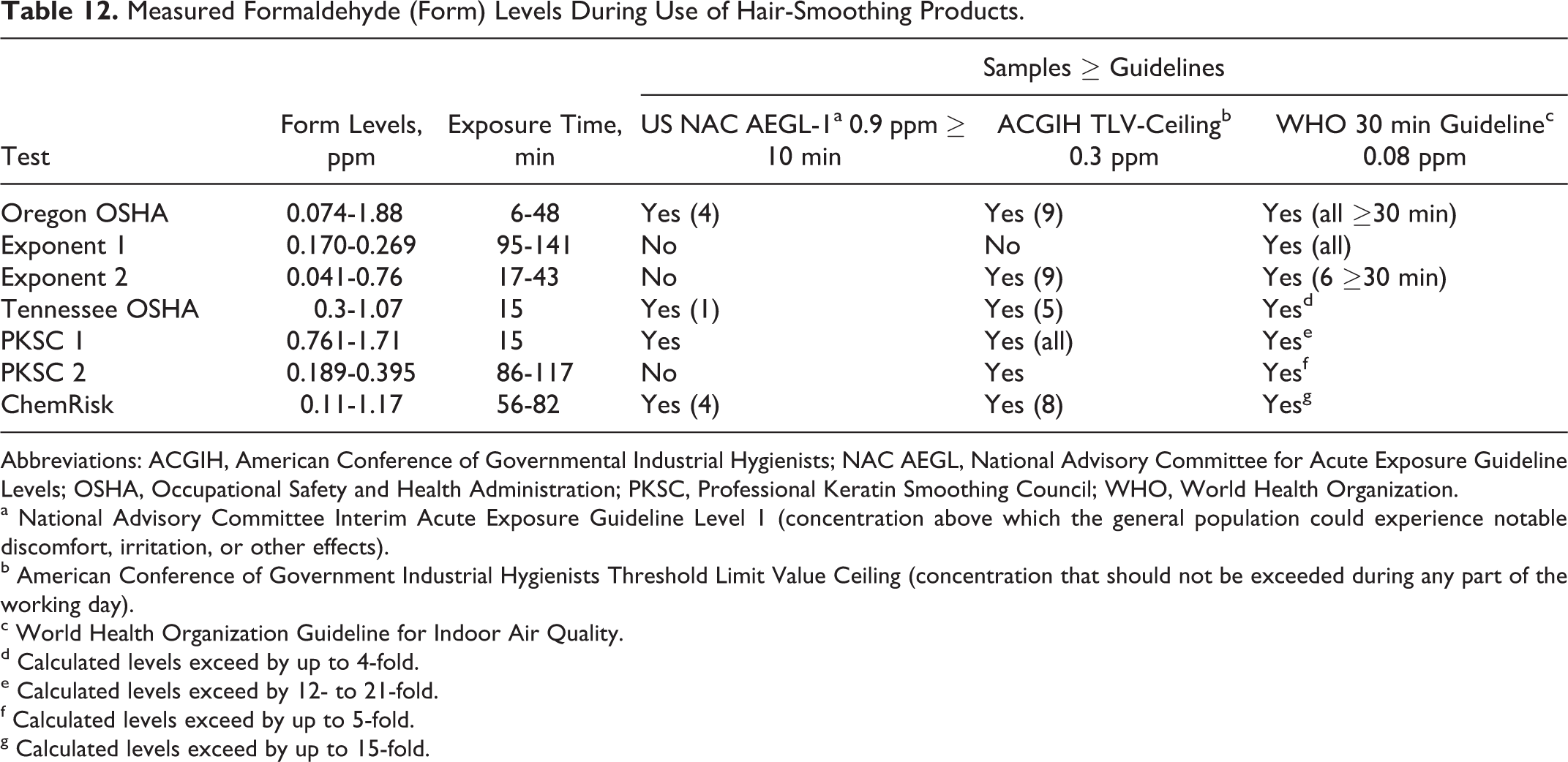
Abbreviations: ACGIH, American Conference of Governmental Industrial Hygienists; NAC AEGL, National Advisory Committee for Acute Exposure Guideline Levels; OSHA, Occupational Safety and Health Administration; PKSC, Professional Keratin Smoothing Council; WHO, World Health Organization.
a National Advisory Committee Interim Acute Exposure Guideline Level 1 (concentration above which the general population could experience notable discomfort, irritation, or other effects).
b American Conference of Government Industrial Hygienists Threshold Limit Value Ceiling (concentration that should not be exceeded during any part of the working day).
c World Health Organization Guideline for Indoor Air Quality.
d Calculated levels exceed by up to 4-fold.
e Calculated levels exceed by 12- to 21-fold.
f Calculated levels exceed by up to 5-fold.
g Calculated levels exceed by up to 15-fold.
Oregon OSHA and CROET collected 15 air samples from 7 beauty salons during the use of a “formaldehyde-free” hair-smoothing product.
11
They used DNPH-treated silica gel absorption tubes (SKC 226-119) and high-flow pumps and analyzed the samples using NIOSH method 2016, which is comparable to US EPA method TO-11. The concentrations of formaldehyde at the stylists’ workstations ranged from 0.074 to 1.88 ppm (median = 0.34 ppm; mean = 0.62 ppm; SD = 0.59 ppm) during sampling/exposure periods ranging from 6 to 48 minutes (median = 19 minutes; mean = 23 minutes; SD = 12 minutes): 4 samples (ranging from 1.26 ppm for 34 minutes to 1.88 ppm for 26 minutes) exceeded the US NAC AEGL-1 (0.9 ppm for ≥10 min).102
9 samples (0.303-1.88 ppm) exceeded the ACGIH TLV-Ceiling (0.3 ppm).107
All 3 samples collected for ≥30 minutes (1.26 ppm for 34 minutes, 0.34 ppm for 47 minutes, and 1.35 ppm for 48 minutes) exceeded the WHO 30-minute guideline (0.08 ppm).108
Further, 2 of the 24 area samples collected during the procedures (0.319 and 0.471 ppm) exceeded the TLV-C, and 10 of the 12 area samples collected for ∼30 minutes or more (eg, 0.226 ppm for 26 minutes and 0.255 ppm for 97 minutes) exceeded the WHO guideline.
Exponent collected two 30-minute background air samples in a salon before the use of a hair-smoothing product, and duplicate samples in the stylist’s breathing zone, the customer’s breathing zone, and within 3 feet of the customer’s location during the application of the product. 112 They used US EPA method TO-11 to collect and analyze the samples. The background formaldehyde concentrations were 0.024 and 0.025 ppm. The concentrations in the samples collected during the procedure ranged from 0.170 ppm for 141 minutes to 0.269 ppm for 95 minutes. All of these concentrations were from 57% to 90% of the ACGIH TLV-C (0.3 ppm), and all exceeded the WHO 30-minute guideline (0.08 ppm).
The Tennessee OSHA conducted an inspection of a salon, including the collection and analysis of air samples. 113 They used DNPH-treated silica gel absorption tubes (XAD-2) and high-flow pumps (SKC AirChek 2000) to collect, apparently, 1 air sample every 15 minutes for 75 minutes during the use of the product. The analytical method was not specified. The 15-minute concentrations ranged from 0.3 to 1.07 ppm. One of these values is equal to the TLV-C (0.3 ppm), and the 4 others exceeded the TLV-C (0.3 ppm) by up to nearly 4-fold. The highest value (1.07 ppm) exceeds the US NAC AEGL-1 (0.9 ppm). In addition, the 75-minute TWA calculated from the reported series of 15-minute concentrations is 0.558 ppm, which is approximately 7-times greater than the WHO 30-minute guideline (0.08 ppm).
The PKSC submitted the results of the analysis of 15-minute air samples collected during the blow drying or flat ironing steps of 4 hair-smoothing treatments. 13,114 They used Sep-Pak DNPH-Silica Cartridges to collect the samples. No further details were provided about the methodology. Formaldehyde was not detected (reporting limit 0.0082 ppm) in one of the samples collected during blow drying, and was not included in the PKSC summary table, presumably because of technical difficulties encountered with this sample. The 15-minute concentrations in the 7 remaining samples ranged from 0.761 to 1.71 ppm. None of these samples exceeded the 15-minute STEL-TWA. However, all of the samples exceeded the ACGIH TLV-C (0.3 ppm) by 2.5- to 5.7-fold, and all but one of them exceeded the US NAC AEGL-1 (0.9 ppm) by 1.3- to 1.9-fold. The TWAs (30 minute) calculated from each complete 15-minute sample pairs (ie, blow drying plus flat ironing) ranged from 0.996 to 1.69 ppm, exceeding the WHO 30-minute guideline (0.08 ppm) by 12 to 21 times.
The PKSC submitted the results of air samples collected to estimate the stylist’s and customer’s inhalation exposures in a beauty salon during hair-smoothing treatments conducted on 2 separate occasions. 13,115 They used Sep-Pak DNPH-Silica Cartridges to collect the samples. No further details were provided. The results ranged from 0.189 ppm for 117 minutes to 0.395 ppm for 86 minutes. The concentrations in 2 of the samples (customer exposure to 0.355 ppm for 117 minutes; stylist exposure to 0.395 ppm for 86 minutes) exceeded the ACGIH TLV-C (0.3 ppm). All of the air samples exceeded the WHO 30-minute guideline (0.08 ppm) by 2.4 to 5 times.
In another study, Exponent collected 63 air samples at 6 salons where hair-smoothing treatments were performed.
116,117
These included 6 area (background) samples collected before any hair-smoothing procedures were conducted and 35 samples collected in the stylists’ breathing zones during a total of 9 treatments. An additional 22 area samples were collected in the salons within 5 feet of the stylists during and after the procedures. They used DNPH-treated silica gel absorption tubes (SKC 226-119) and followed NIOSH method 2016 to collect and analyze the samples. Following is a summary of the results: Concentrations in the 6 background samples ranged from 0.0068 to 0.032 ppm. Concentrations in the other 22 area samples ranged from <0.005 ppm for 45 minutes to 0.14 ppm for 73 minutes. The 3 highest area concentrations (ranging from 0.084 ppm for 69 minutes to 0.14 ppm for 73 minutes) were collected during the treatments and exceeded the WHO 30-minute guideline (0.08 ppm). Calculated 8-hour TWAs ranged from 0.02 to 0.08 ppm. The highest of these is equal to the WHO 30-minute guideline. Concentrations in 9 samples collected in the breathing zones during the procedures (including application of the product, blow drying and flat ironing) ranged from 0.11 ppm for 63 minutes to 0.33 ppm for 73 minutes. The highest concentration (0.33 ppm) exceeded the ACGIH TLV-C (0.3 ppm), and all of them exceeded the WHO 30-minute guideline (0.08 ppm) by up to 4-fold. Concentrations in the 26 samples collected in the breathing zones during each of the separate steps the procedures ranged from 0.041 ppm for 43 minutes (during flat ironing) to 0.76 ppm for 17 minutes (during blow drying). The 4 highest concentrations (ranging from 0.66 for 20 minutes to 0.76 ppm for 17 minutes) were 73% to 84% of the US NAC AEGL-1 (0.9 ppm). Concentrations in 9 of the 26 samples (ranging from 0.31 ppm for 32 minutes to 0.76 for 17 minutes) exceeded the ACGIH TLV-C (0.3 ppm) by up to 2.5-fold. Concentrations in 6 of the 10 samples collected for 30 minutes or more during each step of the treatments (ranging from 0.084 ppm for 31 minutes to 0.31 ppm for 32 minutes) exceeded the WHO 30-minute guideline (0.08 ppm) by up to 4 times.
ChemRisk collected air samples at a salon during 4 consecutive keratin hair-smoothing treatments performed by a licensed cosmetologist (stylist) on 4 separate human hair wigs mounted on mannequin heads over a 6-hour period.
118
Four different hair-smoothing products were used, in random order, during this 1-day study. The mean aqueous formaldehyde concentration was below the limit of detection (LOD <5 × 10−7%, w/w) in 1 product and 3%, 8.3%, and 11.5% (w/w) in the others, as measured using a modified NIOSH 3500 method. Background air samples were collected in the stylist’s breathing zone immediately before each treatment. Treatment-duration and task-duration samples were collected in the stylist’s and mannequin’s breathing zones, in areas representing the breathing zones of potential bystanders, and in the salon’s reception area. The samples were collected on DNPH-treated silica gel absorption tubes (SKC 226-119) using sample pumps (SKC AirChek 52) with low-flow adaptors. All of the samples were analyzed using a modified NIOSH 2016 method coupled with HPLC and UV detection. Following is a summary of the results: The concentrations of formaldehyde in the air samples collected during the treatments were directly related to the concentrations measured in the bulk samples. The mean concentrations in the treatment duration breathing zone samples for the 3 products containing measurable concentrations of aqueous formaldehyde ranged from 0.11 ppm for 82 to 84 minutes to 1.17 for 56 to 57 minutes. The concentrations in 4 of these 16 samples (ranging from 1.13 to 1.21 ppm) exceeded the US NAC AEGL-1 (0.9 ppm), and the concentrations in 8 of them (ranging from 0.58 to 1.21 ppm) exceeded the ACGIH TLV-C (0.3 ppm) by up to 4-fold. The concentrations in all 16 of these samples (ranging from 0.09 to 1.21 ppm) exceeded the WHO 30-minute guideline (0.08 ppm) by up to 15 times. The highest mean concentrations in the treatment-duration samples collected 6 to 10 m from the stylist were 0.37 ppm for 51 minutes and 0.52 ppm for 56 minutes. These values exceed both the ACGIH TLV-C (0.3 ppm) and the WHO 30-minute guideline (0.08 ppm). The highest mean concentrations in duplicate samples collected in the breathing zones during the blow drying step (task) of the treatments were 2.35 and 3.47 ppm for 10 minutes. The corresponding TWAs of the mean concentrations reported for the blow drying and flat ironing steps, combined, approached the OSHA 15-minute STEL-TWA (2 ppm) in the stylist’s breathing zone (1.65 ppm for 23 minutes) and exceeded this standard in the mannequin’s breathing zone (2.1 ppm for 23 minutes). ChemRisk estimated 8-hour TWA concentrations over all 4 treatments conducted sequentially over the 6-hour period. The 8-hour TWAs ranged from 0.25 ppm 6 to 10 m from the stylist to 0.46 ppm in the stylist’s breathing zone. None of the 8-hour TWAs exceeded the OSHA PEL-TWA (0.75 ppm). However, they approached the OSHA AL-TWA (0.5 ppm) by up to 92%, and they all exceeded the OSHA Threshold-TWA (0.1 ppm).
Simulated Use: Calculated Formaldehyde Levels
Berkeley Analytical placed 0.0946 g of a hair-smoothing product in a glass Petri dish, placed the dish in a small-scale, ventilated environmental chamber (0.067 m3), and followed ASTM D 5116 procedures for measuring organic emissions from indoor materials and products. 119,120 They collected 3 consecutive 1-hour air samples from the chamber (1 air change/h), at room temperature (73.4°F), using Sep-Pak XPoSure samplers. They reported emissions factors for formaldehyde ranging from 1020 µg/g h for the first hour to 1670 µg/g h for the third hour. Indoor Environmental Engineering calculated formaldehyde concentrations in a hypothetical hair salon (240 ft2; 8-ft ceiling) from single 90-minute emissions of formaldehyde from the hair-smoothing product. They conservatively assumed a 1020 µg/g h emission rate at room temperature, likely underestimating the emissions during actual use. 34 The emission rates are most probably much higher when the product is heated (eg, during blow-drying and flat-ironing). They modeled TWA exposure concentrations for the customer (110 minutes) and the stylist (8 hours), assuming 3 outdoor air ventilation rates (0.13-0.6 ft3/min-ft2) and 3 different amounts of the product applied the customer’s hair (12.6-37.8 g). The amounts were selected from recommendations provided in the manufacturer’s training video for using the product on short, medium, and long hair.
The 110-minute formaldehyde concentrations ranged from 0.033 ppm (12.6 g product; 0.6 ft3/min-ft2) to 0.269 ppm (37.8 g product; 0.6 ft3/min-ft2). Of the three 110-minute estimates, 2 assuming 25.2 g of product (0.096-0.18 ppm at 0.38 and 0.13 ft3/min-ft2, respectively) and all of the estimates assuming 37.8 g (0.098-0.269 ppm), exceeded the WHO 30-minute guideline (0.08 ppm). The highest estimate (0.269 ppm) was about 90% of the ACGIH TLV-C (0.3 ppm). In addition, the highest estimated 8-hour TWA was 0.108 ppm (37.8 g; 0.13 ft3/min-ft2), which exceeds the US OSHA 8-hour Threshold-TWA (0.1 ppm).
Discussion
Based on the available data, the Panel considered that formaldehyde and methylene glycol are safe for use in cosmetics when formulated to ensure use at the minimal effective concentration, but in no case should the formalin (note 1) concentration exceed 0.2% (w/w), which would be 0.074% (w/w) calculated as formaldehyde or 0.118% (w/w) calculated as methylene glycol. Additionally, formaldehyde and methylene glycol are safe in the present practices of use and concentration in nail-hardening products. However, formaldehyde and methylene glycol are unsafe in the present practices of use and concentration in hair-smoothing products.
The Panel emphasized that a large body of data has demonstrated that formaldehyde gas exposure can cause NPCs. Although debate is ongoing regarding the dose–response relationship for the induction of NPCs, the Panel maintained its view that formaldehyde gas can produce such cancers at high doses.
Epidemiology studies have suggested a weak association between exposure to formaldehyde and LHP cancers. The reported association of formaldehyde exposure with LHP cancers is just that, an association, and the Panel is not aware of a plausible mechanism by which formaldehyde exposure could be causally linked to LHP tumors. Based on the testicular effects observed in rats exposed to formaldehyde, the Panel acknowledged that a mechanism of action by which formaldehyde might cause the testicular effects is not known and these effects may be secondary to local effects, such as irritation and inflammation, and stress at high doses.
The NMC, the PKSC, the American Chemistry Council, the Personal Care Products Council, and 1 individual provided new data and comments. After reviewing the comments and additional data, the Panel determined that the data were sufficient to support the safety of formaldehyde/methylene glycol in nail hardeners.
The additional data confirmed the current use concentration of formaldehyde/methylene glycol in the 1% to 2% range in nail hardeners (1 product tested had a value of 2.2%). Given the rapid reaction on the nail surface and the use of nail hardeners at room temperature, the Panel did not consider that formaldehyde/methylene glycol at 1% to 2% in nail hardeners would present a risk of sensory irritation to the eyes, nose, or throat of users. The Panel noted that the present practices of use of nail hardeners include instructions that cautioned users to limit application of the material to the top surface of the nail only, to allow it to dry fully, and to not get the material on the skin.
The Panel noted that the OSHA occupational safety limits include a TWA permissible exposure level of 0.75 ppm for a work day and a short-term exposure limit of 2 ppm. Air monitoring and medical examinations are triggered when formaldehyde concentrations in workplace air exceed 0.5 ppm averaged over an 8-hour shift, and ventilation and training when concentrations exceed 0.75 ppm averaged over 8 hours or 2 ppm averaged over 15 minutes. Formaldehyde must be listed in a company’s Material Safety Data Sheet if formaldehyde is present at 0.1% or more, or if the product releases formaldehyde gas above 0.1 ppm.
Although such requirements are mandated by OSHA, the Panel remained concerned about adverse reports of sensory irritation consistent with measured air levels of formaldehyde in salons using hair-smoothing products (also known as hair-straightening products) containing formaldehyde/methylene glycol. Because the use of these products involves the application of heat, the Panel remained concerned about the amounts of formaldehyde vapor that can be released. The reported levels of formaldehyde gas measured in the air around salon work stations can be below occupational exposure standards and guidelinesbut also may be at or only marginally below occupational exposure standards and above indoor air quality guidelines. The Panel noted that the PKSC suggested that these products are manufactured with the expectation that adequate ventilation would be provided during use; ie, safe use requires adequate ventilation. Occupational Safety and Health Administration and other inspections, however, reported a range of ventilation controls, many of which were inadequate.
Additional use studies were done on behalf of the PKSC to demonstrate that exposure to formaldehyde could be minimized with proper procedures and use of personal ventilation devices. The Panel acknowledged that formaldehyde levels in air samples were lower in the most recent data compared to data submitted earlier, but proper safety procedures, including positioning of personal ventilation devices, were not uniformly followed.
In concept, therefore, limits on the concentration of formaldehyde/methylene glycol in hair-smoothing products, control of the amount of product applied, use of lower temperatures, and approaches to mandate adequate ventilation, are among the steps that could be taken to ensure that these products would be used safely in the future. However, in the present practices of use and concentration (on the order of 10% formaldehyde/methylene glycol, blow drying and heating up to 450°F with a flat iron, inadequate ventilation, resulting in many reports of adverse effects), hair-smoothing products containing formaldehyde and methylene glycol are unsafe.
The Panel adopted a suggestion to include limits for formalin concentration because formalin is what formulators actually add to cosmetic products. Formalin is an aqueous solution typically containing 37% (w/w) formaldehyde. Formalin contains both formaldehyde and methylene glycol because of the equilibrium between formaldehyde and methylene glycol in aqueous solution.
While retaining the concept that formaldehyde and methylene glycol should be used only at the minimal effective concentration, the Panel stated that in no case should the formalin concentration exceed 0.2% (w/w), which would be 0.074% (w/w) calculated as formaldehyde or 0.118% (w/w) calculated as methylene glycol. Although these numbers appear to be disparate, they are not. The value of 0.074% (w/w) of formaldehyde simply reflects that formalin typically contains 37% formaldehyde (0.2% (w/w) formalin multiplied by 0.37 = 0.074%, w/w formaldehyde). The value of 0.118% (w/w) for methylene glycol simply reflects the difference in molecular weight between formaldehyde and methylene glycol.
The Panel recognized that the most commonly used analytical methods for the detection and measurement of formaldehyde are not specific for nonhydrated formaldehyde but can accurately indicate the presence and quantity of formaldehyde equivalents. A typical method, for example, can detect formaldehyde equivalents in a formulation, or released into the air, via a 2-stage process (1) derivatization of a sample with a hydrazine (which reacts with formaldehyde or methylene glycol, in a formulation sample or in an air sample) and (2) detection and measurement of the resultant hydrazone (ie, the reaction product of the hydrazine and formaldehyde) with a diode array, after separation on a column (eg, HPLC separation followed by UV/Vis light detection).
While other formaldehyde/methylene analytical techniques are known, such as NMR spectrometry, the Panel found that the methodology used by OSHA and FDA produces consistent results that are directly and meaningfully comparable to regulatory standards and guidelines. As the conditions under which formaldehyde is measured in products can affect the results, the method used to measure formaldehyde in products should be appropriate for the conditions, such as temperature and pH, under which the product is used.
The Panel reasoned that the term “formaldehyde equivalents” best captures the idea that methylene glycol is continuously converted to formaldehyde, and vice versa, even at equilibrium, which can be easily shifted by heating, drying, and other conditions to increase the amount of formaldehyde. Any other term would not distinguish the rapid, reversible formaldehyde/methylene glycol equilibrium from the slow, irreversible release of formaldehyde resulting from the so-called formaldehyde releaser preservatives (eg, diazolidinyl urea). Formaldehyde releaser preservatives are not addressed in this safety assessment. The formaldehyde releasers may continue to be safely used in cosmetics at the levels established in their individual CIR safety assessments.
Conclusion
The CIR Expert Panel concluded that formaldehyde and methylene glycol are safe for use in cosmetics when formulated to ensure use at the minimal effective concentration, but in no case should the formalin (note 1) concentration exceed 0.2% (w/w), which would be 0.074% (w/w) calculated as formaldehyde or 0.118% (w/w) calculated as methylene glycol. Additionally, formaldehyde and methylene glycol are safe in the present practices of use and concentration in nail-hardening products. However, formaldehyde and methylene glycol are unsafe in the present practices of use and concentration in hair-smoothing products (also known as hair-straightening products).
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
