Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 4 ammonium hectorite compounds used in cosmetics: disteardimonium hectorite, dihydrogenated tallow benzylmonium hectorite, stearalkonium hectorite, and quaternium-18 hectorite. These ingredients function in cosmetics mainly as nonsurfactant suspending agents. The Panel reviewed available animal and human data and concluded that these ammonium hectorite compounds were safe as cosmetic ingredients in the practices of use and concentration as given in this safety assessment.
Introduction
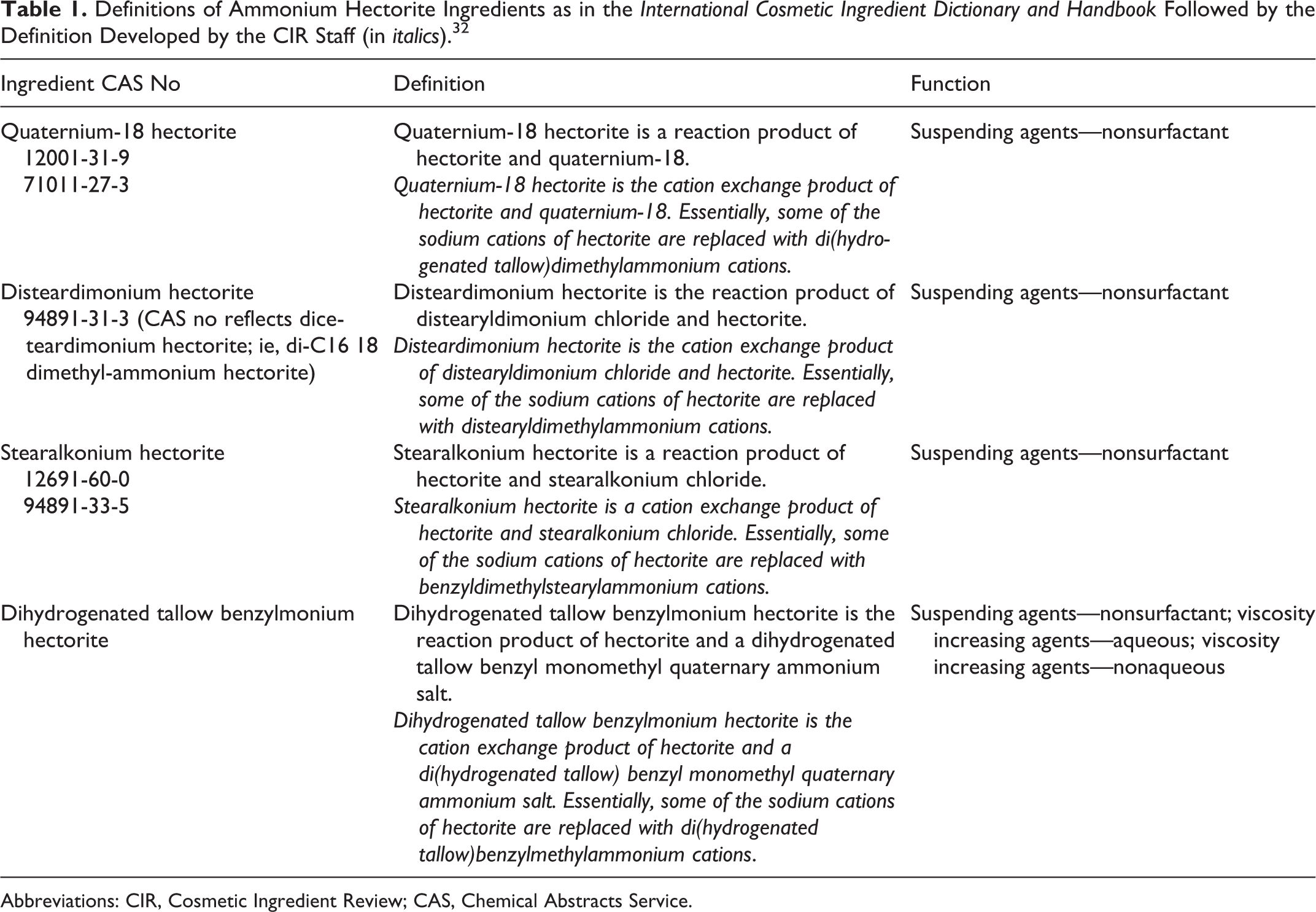
This report assesses the safety of ammonium hectorites as used in cosmetics. The ingredients in this report are disteardimonium hectorite, dihydrogenated tallow benzylmonium hectorite, stearalkonium hectorite, and quaternium-18 hectorite. These ingredients function in cosmetics mainly as nonsurfactant suspending agents (Table 1).
Definitions of Ammonium Hectorite Ingredients as in the International Cosmetic Ingredient Dictionary and Handbook Followed by the Definition Developed by the CIR Staff (in italics). 32
Abbreviations: CIR, Cosmetic Ingredient Review; CAS, Chemical Abstracts Service.
Other hectorite ingredients, stearalkonium hectorite and quaternium-18 hectorite, have been reviewed by the Cosmetic Ingredient Review Expert Panel (Panel) and were found to be safe for use in cosmetics. 1,2 Summaries of the relevant data from these reports are included in the appropriate sections.
The silicate clay, hectorite, and other clays were previously reviewed by the Panel as part of a group of aluminum silicate clays and found to be safe as used in cosmetic products. 3 Quaternium-18 was also previously reviewed and was found to be safe as a cosmetic ingredient. 1
Chemistry
Definition and Structure
Hectorite is part of a group of phyllosilicate, layered, clay-based minerals, the general term for which is smectites, the most prominent of which are montmorillonite, beidellite, nontronite, saponite, and hectorite. 4 As mentioned previously, hectorite and montmorillonite were included in the previous safety assessment of clays. 3 These various clays are differentiated by variations in chemical composition involving substitutions of aluminum for silicon in tetrahedral cation sites and aluminum, iron, magnesium, and lithium in octahedral cation sites (Figure 1).
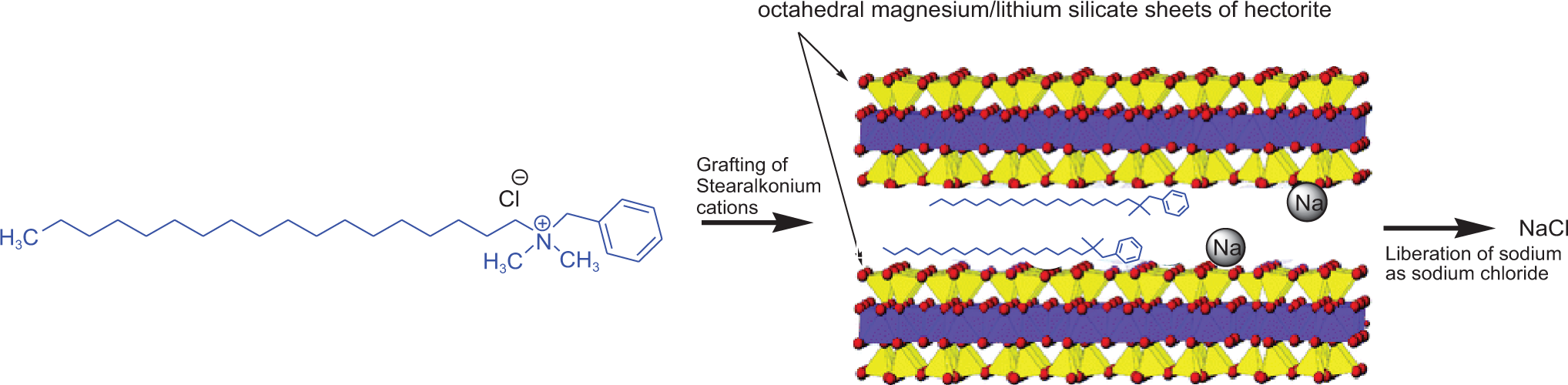
Synthesis of organoclay minerals.
Smectite minerals have a variable net negative charge, which is balanced by sodium, calcium, or magnesium ions adsorbed externally on interlamellar surfaces. The structure, chemical composition, exchangeable ion type, and small crystal size of smectite minerals are responsible for several unique properties, including a large chemically active surface area, a high cation exchange capacity, interlamellar surfaces having unusual hydration characteristics, and the ability to strongly modify the flow behavior of liquids. In the cosmetics industry, clay-based products are used to improve properties such as suspension, emulsion stability, viscosity, thermal stability, and spreadability.
Structurally, hectorite is a trioctahedral, magnesium/lithium silicate-based mineral that is amorphous and does not contain any crystalline silica.
Because of isomorphous substitution of lithium ions (a +1 charge) for magnesium ions (a +2 charge) in the octahedral sheet during hectorite formation, the surfaces of these minerals have a delocalized net negative charge in the lattice. 5 Cations located between 2 consecutive layers (octahedral sheets) contribute to compensate the structural charge and to keep the layers bound. Thus, cations like sodium are attracted to the mineral surface to counterbalance the interlayer charge. These cations can easily be exchanged, since they are retained by electrostatic attractions.
Organohectorite minerals, such as disteardimonium hectorite, stearalkonium hectorite, quaternium-18 hectorite, and dihydrogenated tallow benzylmonium hectorite, are synthesized by grafting cationic surfactants to hectorite (ie, exchanging the interlayer sodium cations with a cationic surfactant). These cationic surfactants are quaternary ammonium compounds with the template formulae [(CH3)3NR]+, [(CH3)2NRR′]+, and [CH3NRR′R′′]+, wherein R, R′, and R′′ are alkyl or aromatic hydrocarbons. For instance, in the case of disteardimonium hectorite at least some of the sodium cations of hectorite have been exchanged for the [(CH3)2NRR′]+ cation, wherein both R and R′ are octadecyl alkyl chains (ie, stearyl groups). The exchange is typically carried out by the addition of the appropriate ammonium chloride (eg, disteardimonium chloride) to an alcohol/water slurry of hectorite. 6,7 The major by-product therein is sodium chloride, which is removed during processing (Figure 2). This cation exchange shifts the nature of these minerals from hydrophilic to lipophilic. 6
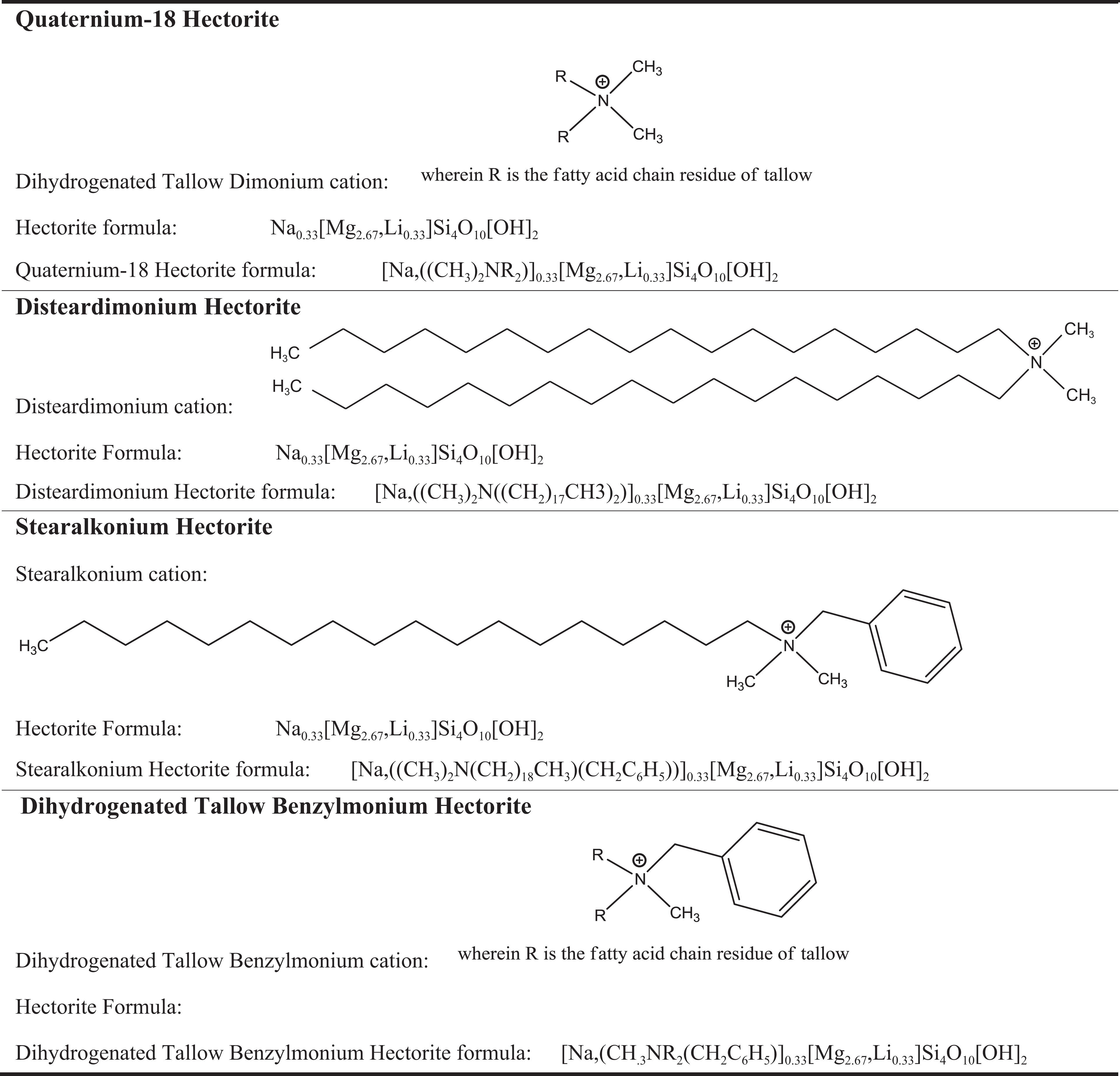
Structures/Formulas of ammonium hectorite ingredients.
Physical and Chemical Properties
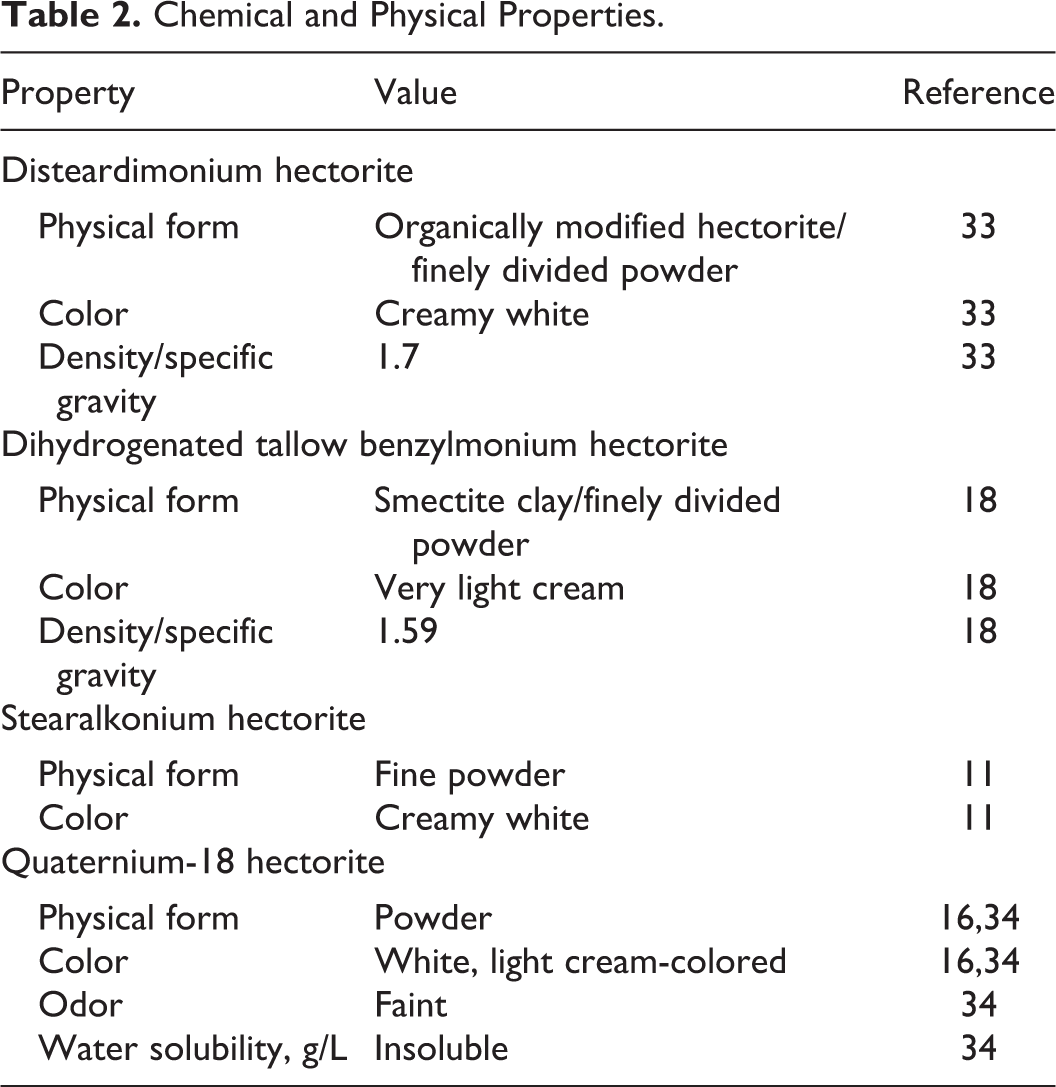
The physical and chemical properties of the ingredients in this safety assessment are provided in Table 2.
Chemical and Physical Properties.
Hectorite
The unique physicochemical properties of smectite clays, including hectorite, are the result of (1) extremely small crystal size, (2) variations in internal chemical composition, (3) structural characteristics caused by chemical factors, (4) large cation exchange capacity, (5) large surface area that is chemically active, (6) variations in types of exchangeable ions and surface charge, and (7) interactions with inorganic and organic liquids. 4
Because of aggregation, the effective particle size will be larger and the surface area will be considerably smaller than the actual particle size and aggregated surface area. During the growth of hectorites, by either transformation or neoformation, crystals become interlocked and become difficult to separate except by a strong shearing force. Differences in the effective particle size of hectorites are extremely important in the determination of properties such as ion exchange, viscosity, and fluid loss. 4,8
Quaternium-18 Hectorite
Quaternium-18 hectorite is reported to be an inert, chemically stable material. It is pH and heat stable under normal cosmetic use conditions. 9,10
Impurities
Quaternium-18 Hectorite
Methyl ditallow amine, methyl ditallow ammonium hectorite, and sodium chloride are possible impurities in quaternium-18 hectorite. 11
Stearalkonium Hectorite
Stearalkonium hectorite contains a maximum of 3 ppm and 20 ppm elemental arsenic and elemental lead, respectively. 12 Sodium chloride may be formed during the ionic exchange reaction of stearalkonium chloride and hectorite, which is washed out down to <0.5%. “Adsorbed” cations are between 3.0% and 5.0% in stearalkonium hectorite.
Use
Cosmetic
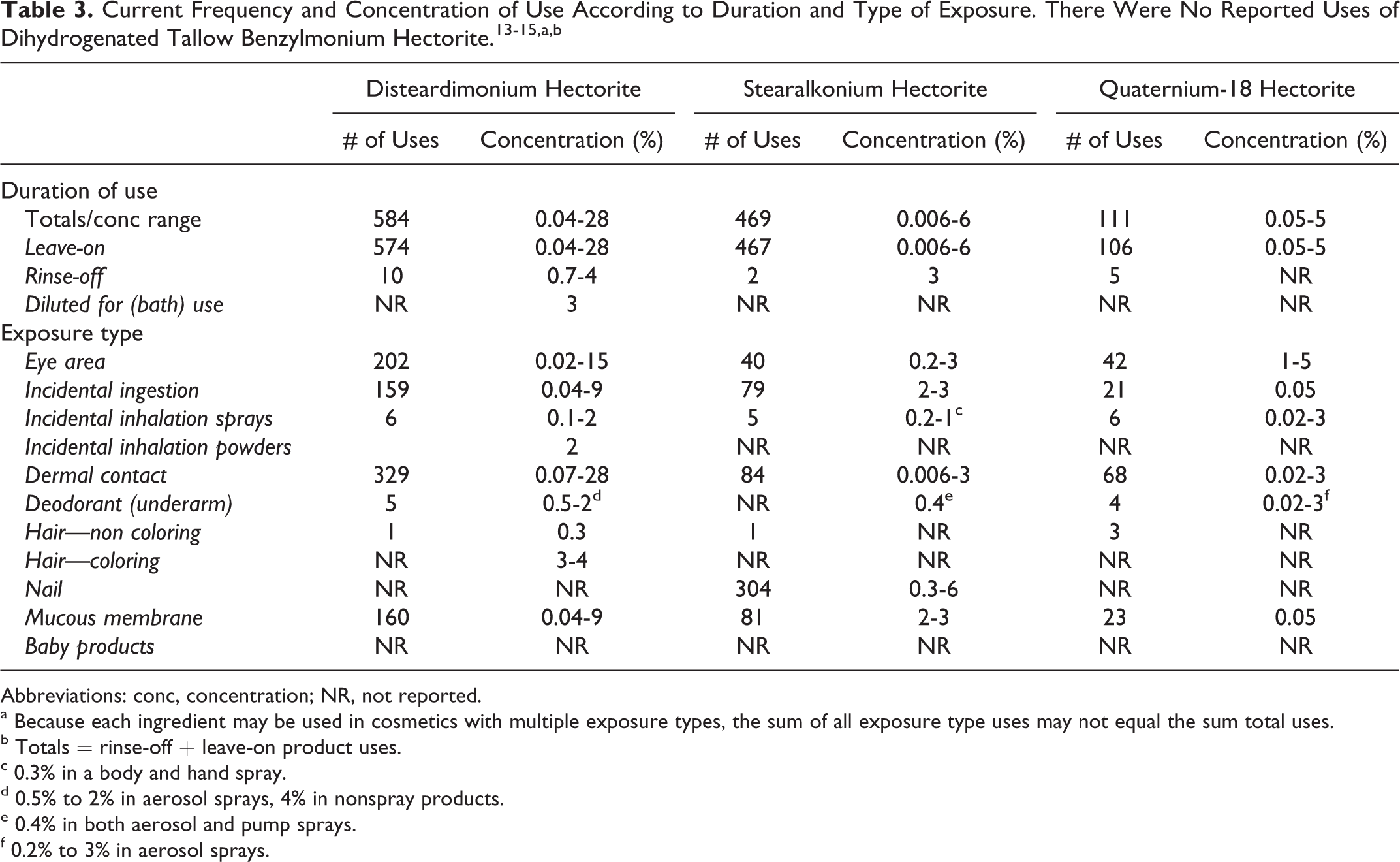
Data on ingredient usage as a function of cosmetic product type are provided to the Food and Drug Administration Voluntary Cosmetic Registration Program (VCRP), and a survey conducted by the Personal Care Products Council (Council) collected maximum use concentrations for ingredients in this group. 13 –15 These data are combined in Table 3.
Abbreviations: conc, concentration; NR, not reported.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
b Totals = rinse-off + leave-on product uses.
c 0.3% in a body and hand spray.
d 0.5% to 2% in aerosol sprays, 4% in nonspray products.
e 0.4% in both aerosol and pump sprays.
f 0.2% to 3% in aerosol sprays.
The VCRP reported that disteardimonium hectorite was used in 574 leave-on products (maximum of 28% in makeup preparations) and 10 rinse-off products (maximum of 4% in hair coloring preparations). Quaternium-18 hectorite was reported to be used in 106 leave-on products (maximum of 5% in mascara) and 5 rinse-off products (no concentrations of use for this category was reported by the Council). Stearalkonium hectorite was used in 467 leave-on products (maximum of 6% in nail polishes and enamels) and 2 rinse-off products (maximum of 3% in eye makeup remover). Of the 467 leave-on products, 277 were used in nail polish and enamels.
There were no reported uses of dihydrogenated tallow benzylmonium hectorite.
Noncosmetic
Quaternium-18 hectorite is used as a dispersant in volatile oils. 16 It has also been tested as granulators and binders in the production of tablets.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
There were no published studies on absorption, distribution, metabolism, or excretion discovered nor were unpublished data provided.
Toxicological Studies
Acute Toxicity
Dermal
There were no published acute dermal toxicity studies discovered nor were unpublished data provided.
Oral: Nonhumans
Quaternium-18 Hectorite
The oral median lethal dose (LD50) of quaternium-18 was >10 g/kg for rats. 17
Dihydrogenated Tallow Benzylmonium Hectorite
The oral LD50 for dihydrogenated tallow benzylmonium hectorite was >5.0 g/kg for Sprague-Dawley rats (n = 10). 18
Inhalation: Nonhumans
Quaterium-18 Hectorite
Aerosolized quaternium-18 hectorite (202 mg/L in isopropyl myristate) was not toxic to rats (n = 10) after 1 hour. 7,17
Dihydrogenated Tallow Benzylmonium Hectorite
The inhalation LC50 for dihydrogenated tallow benzylmonium hectorite was >5.2 mg/L for Sprague-Dawley rats (n = 5/sex) after 4 hours. 18
Repeated Dose Toxicity
Dermal: Nonhumans
Quaternium-18 Hectorite
Quaternium-18 hectorite (up to 50%) applied, unoccluded, to the exposed skin of rabbits 3 times per day for 5 days per week for 3 weeks caused no toxic effects. 7,17 There was mild drying and scaling of the upper layers of the skin during the early days of the study.
Stearalkonium Hectorite
There were no toxic effects observed when stearalkonium hectorite (12.5% to 50%) was dermally applied to rabbits twice per day, unoccluded over 3 weeks. 19
Reproductive and Developmental Toxicity
Dihydrogenated Tallow Benzylmonium Hectorite
Orally administered dihydrogenated tallow benzylmonium hectorite had a no effect level of 1000 mg/kg to Sprague-Dawley rats. 20 The test material was administered throughout the complete reproductive cycle for 1 generation. No further information was provided.
Genotoxicity
In Vitro
Stearalkonium Hectorite
Stearalkonium hectorite was not mutagenic to Salmonella typhimurium (TA98, TA100, TA1535, TA1537, and TA1538) up to 1500 µL/plate or mouse lymphoma cells up to 500 µL/plate. 21,22
Dihydrogenated Tallow Benzylmonium Hectorite
Dihydrogenated tallow benzylmonium hectorite (concentration not provided) was not mutagenic in an Ames test using Salmonella with or without metabolic activation. 20
Carcinogenicity
There were no published carcinogenicity studies discovered nor were unpublished data provided.
Irritation and Sensitization
Irritation
Dermal: Nonhumans
Quaternium-18 Hectorite
Quaternium-18 hectorite (50%) was not dermally irritating to rabbits. 17
Stearalkonium Hectorite
Stearalkonium hectorite (50% w/v in water with 2.5% polysorbate 809) did not cause erythema or edema when administered to the skin of albino rabbits. 19
Dihydrogenated Tallow Benzylmonium Hectorite
Dihydrogenated tallow benzylmonium hectorite (0.5 g in 0.5 mL saline) was not irritating when administered to the intact and abraded skin of New Zealand White rabbits (n = 6) for 24 hours. 20
Dermal: Humans
Disteardimonium Hectorite
A patch test of a cosmetic preformulation containing disteardimonium hectorite (15% with cyclomethicone and PEG-10 dimethicone [Nikkol Group, 9999 4368/id]; 23 diluted to 1.5% in mineral oil) was conducted in female subjects (n = 11) of various skin types. 24 The occlusive patches were left in place for 48 hours. The test site was examined at patch removal and 15 minutes later. There were no signs of irritation observed. A patch test of a cosmetic mixture product containing disteardimonium hectorite (15%) was conducted in subjects (n = 10). 25 The patches were left in place for 24 hours and the test sites observed at removal and after an additional 24 hours. There were no signs of irritation observed.
Stearalkonium Hectorite
Stearalkonium hectorite (50% w/v in water with 2.5% polysorbate 809; n = 50) was not irritating in a repeated insult patch test (100%) or in a facial mask containing stearalkonium hectorite (1.4%; n = 27). 26,27
Ocular
Quaternium-18 Hectorite
Quaternium-18 hectorite (50%; 0.1 mL) was not irritating to rabbits. 7 Quaternium-18 hectorite (2 mg neat; 20 g in 100 mL physiological saline or corn oil) instilled in the eyes of subjects did not cause any abnormal ocular sensations. 17 No pain was reported. A “sand-like” feeling was reported in the saline sample. No damage to the eye was observed.
Dihydrogenated Tallow Benzylmonium Hectorite
Dihydrogenated tallow benzylmonium hectorite (0.5 g in 0.5 mL saline) was practically nonirritating when administered to the eyes of New Zealand White rabbits (n = 6 rinsed after 4 seconds; n = 3 not rinsed). 20
Stearalkonium Hectorite
Stearalkonium hectorite (up to 100%; volumes not provided) was a minimal to mild ocular irritant to rabbits. 7,28 In an Eyetex in vitro test of products, an eyeliner containing stearalkonium hectorite (0.196%), a lipliner pencil (1.0%), and a face mask (5.0%) were classified as minimal to mild irritants. 29
Sensitization
Dermal: Nonhumans
Dihydrogenated Tallow Benzylmonium Hectorite
It was reported that dihydrogenated tallow benzylmonium hectorite (concentration not provided) did not cause delayed contact hypersensitivity in albino guinea pigs (n not provided). 18
Dermal: Humans
Quaternium-18 Hectorite
Quaternium-18 hectorite (100%) was not irritating or sensitizing in a repeated insult patch test (n = 50). 20 Quaternium-18 hectorite was not sensitizing in an eye shadow (n = 50), a blusher (n = 209), and 3 undisclosed products (n = 12) up to 10%. 17,30
Disteardimonium Hectorite
A human repeated insult patch test (HRIPT; n = 112) of disteardimonium hectorite (100%; 20 μg) was conducted using an occlusive Finn chamber. 31 The test material was not sensitizing.
Stearalkonium Hectorite
Stearalkonium hectorite (50% w/v in water with 2.5% polysorbate 809) was not sensitizing in a repeated insult patch test (n = 50) or in a product containing stearalkonium hectorite (1.4%). 26
Summary
Ammonium hectorites are a group of ingredients that function in cosmetics mainly as nonsurfactant suspending agents. The ingredients in this report are disteardimonium hectorite, dihydrogenated tallow benzylmonium hectorite, stearalkonium hectorite, and quaternium-18 hectorite.
Hectorite is a trioctahedral, magnesium/lithium silicate-based mineral that is amorphous and does not contain any crystalline silica. The relatively weak electrostatic interactions of thin crystalline layers can essentially be “propped open” to allow the insertion of certain molecules and atoms (ie, disteardimonium, quaternium-18).
Disteardimonium hectorite was used in 574 leave-on products up to 28% and in 10 rinse-off products up to 4%. Stearalkonium hectorite was used in 467 leave-on products up to 6% and 2 rinse-off products up to 3%. Quaternium-18 hectorite was reported to be used in 106 leave-on products up to 5% and 5 rinse-off products.
There were no published absorption, distribution, metabolism or excretion, acute dermal toxicity, or carcinogenicity studies discovered nor were unpublished data provided.
The oral LD50 for dihydrogenated tallow benzylmonium hectorite was 5.0 g/kg for rats. The oral LD50 of quaternium-18 was >10 g/kg. The inhalation LC50 for dihydrogenated tallow benzylmonium hectorite was >5.2 mg/L for rats after 4 hours. Aerosolized quaternium-18 hectorite was not toxic to rats at 202 mg/L after 1 hour.
Stearalkonium hectorite was not dermally toxic to rabbits at concentrations of 12.5% to 50% over 3 weeks. Quaternium-18 hectorite applied to the exposed skin of rabbits for 3 weeks was not toxic up to 50%.
Stearalkonium hectorite was not mutagenic to S typhimurium up to 1500 µL/plate or mouse lymphoma cells up to 500 µL/plate.
Stearalkonium hectorite did not cause erythema or edema to albino rabbits at 50% w/v. Quaternium-18 hectorite (50%) was not irritating to rabbits. Dihydrogenated tallow benzylmonium hectorite at 0.5 g in 0.5 mL saline was not irritating when administered to the intact and abraded skin of rabbits.
Disteardimonium hectorite was not irritating to humans in 2 patch tests at 15%. Stearalkonium hectorite was not irritating or sensitizing to humans at 100%. Dihydrogenated tallow benzylmonium hectorite (concentration not provided) did not cause delayed contact hypersensitivity in albino guinea pigs.
Stearalkonium hectorite was a minimal to mild ocular irritant to rabbits and humans. It was classified as a minimal to mild irritant in 3 Eyetex in vitro tests of products. Quaternium-18 hectorite was not an ocular irritant at 50% in rabbits and at 2 mg in humans. Dihydrogenated tallow benzylmonium hectorite at 0.5 g in 0.5 mL saline was practically nonirritating when administered to the eyes of rabbits.
Quaternium-18 hectorite was not irritating or sensitizing up to 100% in HRIPTs.
Discussion
Single-dose toxicity data were available for quaternium-18 hectorite and dihydrogenated tallow benzylmonium hectorite, and repeated-dose toxicity data were available for quaternium-18 hectorite and stearalkonium hectorite. Genotoxicity data were available for stearalkonium hectorite and dihydrogenated tallow benzylmonium hectorite, and reproductive and developmental toxicity data were available for dihydrogenated tallow benzylmonium hectorite. Irritation and sensitization data were available for all of these ingredients. Overall, no significant toxicity was reported, and these ingredients were not dermal irritants or sensitizers. The Panel considered that the chemical structures of these clay-based ingredients were sufficiently similar, as was the pattern of use in cosmetics, to support using data on each individual ingredient to support the safety of the entire group.
The Panel noted that the hectorites in this safety assessment have high chemical stability and are biochemically inert. The Panel considered the lithium substitution for magnesium in the magnesium/lithium silicate sheet structure but concluded lithium is tightly bound and not likely to leach. The substitution of lithium for magnesium at many sites in the lattice structure gives the material a delocalized net negative charge in the lattice. Although no data were available on dermal penetration, the Panel considered that the charge properties and the large molecular weight of these clay-like ingredients would preclude significant dermal penetration. Because they are chemically inert, no metabolites are expected that would penetrate the skin.
Conclusion
The Panel concluded that disteardimonium hectorite, dihydrogenated tallow benzylmonium hectorite, stearalkonium hectorite, and quaternium-18 hectorite are safe in the present practices of use and concentration described in this safety assessment. Were dihydrogenated tallow benzylmonium hectorite (not in current use) to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.