Abstract
Uva-ursi folium (bearberry leaf) has been traditionally used to treat symptoms of lower urinary tract infections. The most representative constituent of this herbal drug is arbutin that is rapidly absorbed in the small intestine and undergoes hepatic conjugation to form hydroquinone (HQ) conjugates. As free HQ is crucial for the safety of the herbal preparation, we reviewed published and unpublished experimental and human studies to clarify some outdated assumptions and to support the safety of therapeutic daily doses of Uva-ursi folium extract. Specifically, data on pharmacokinetics and the human exposure of arbutin and HQ were reviewed. A therapeutic recommended human daily dose of bearberry leaf extract (420 mg hydroquinone derivatives calculated as anhydrous arbutin) liberates free HQ in urine at a maximum exposure level of 11 µg/kg body weight (bw)/d. By means of an experimental no observed effect level value, a permitted daily exposure dose below which there is a negligible risk to human health was estimated for free HQ (100 µg/kg bw/d). Dietary sources of arbutin/HQ that are regularly consumed long term by humans generate comparable free HQ exposure levels. There is no direct evidence, regarding human data, supporting the fact that free HQ causes convulsion, hepatotoxicity, nephrotoxicity, or promotion of tumors in humans. Free HQ had no activity promoting pancreatic, bladder, stomach, or liver carcinogenesis. In conclusion, under the recommended use conditions Uva-ursi folium is a safe therapeutic option for treating lower urinary tract infections.
Introduction
Arctostaphylos Uva-ursi folium (bearberry leaf) has traditionally been used for medicinal purposes in Europe and America, especially in the treatment of symptoms of lower urinary tract infections. A monograph of the Herbal Medicinal Products Committee (HMPC) 1 has recently confirmed this traditional use.
The most representative constituents of dried leaf of Uva-ursi are the phenolic glycoside arbutin and the derivate hydroquinone (HQ; Figure 1). The content of arbutin in leaves depends on the season as well as on the geographical distribution and varies from 5% to 15% and up to 10% according to several authors. 1 –3 Herbal medicinal preparations of bearberry leaves have been standardized or quantified to the content of arbutin. 1 –4
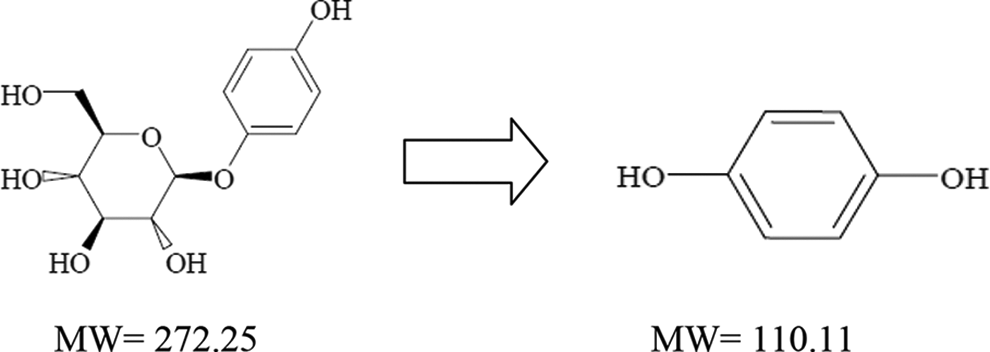
Chemical structure of arbutin (A) and hydroquinone (B).
Although arbutin is the major pharmacological active constituent of the extract of bearberry leaves, experimental studies revealed that the whole extract is responsible for the global pharmacological action. Uva-ursi leaves also contain small amounts of free HQ (<0.3%), another HQ derivative—the glycoside methylarbutin (up to 4%), 2,3 and flavonoids such as hyperoside (0.8%-1.5%).
Because arbutin is broken down to yield free HQ and glucose, concerns regarding the safety of free HQ have raised questions about the safety of herbal preparations of bearberry leaves’. 1,5 –8 It was primarily suspected to induce hepatotoxicity, nephrotoxicity, and other organ toxicity as well as genotoxicity.
In this review, we searched international published data as well as unpublished data of a manufacturer of herbal preparations containing bearberry leaf to clarify this concern. Special reference is made to the pharmacokinetics and metabolism of arbutin and whether its minor metabolite, mainly free HQ, has nephrotoxic, hepatotoxic, or carcinogenic potential.
The following databases were searched for pharmacology and toxicology data: DIMDI, Medline, TOXLINE, ToxNet, HSDB, GENETOX, CCRIS, CHEMIDplus, CAS, and RTECS.
Information was also obtained from the National toxicology Program (NTP) technical report series, C International Agency for Research on Cancer (IAR), International Program on Chemical safety (IPCS), and Organization for Economic Cooperation and Developments / Screening Information Data (OECD/SIDS).
Special reference was made to the European Scientific Cooperative on Phytotherapy (ESCOP), World Health Organization (WHO), and HMPC monographs on Arctostaphylos Uva-ursi folium.
Pharmacokinetics of Arbutin and HQ
In the past, intestinal bacteria have been considered responsible for the major arbutin breakdown, producing free HQ directly in the intestinal lumen. After absorption, the free HQ is metabolized in the liver forming HQ conjugates, which were further eliminated in the urine (Figure 2B). However, the contribution of bacteria to arbutin hydrolysis in the intestine is very small—since bacterial density in the small human intestine is very low, it would have to reach the more distal part of the intestinal tract, the colon, to find a higher density of bacteria. 10 In addition, HQ was found only in traces in fecal slurries in animal experiments. 11 Therefore, intestinal bacteria cannot be responsible for the major arbutin metabolism.
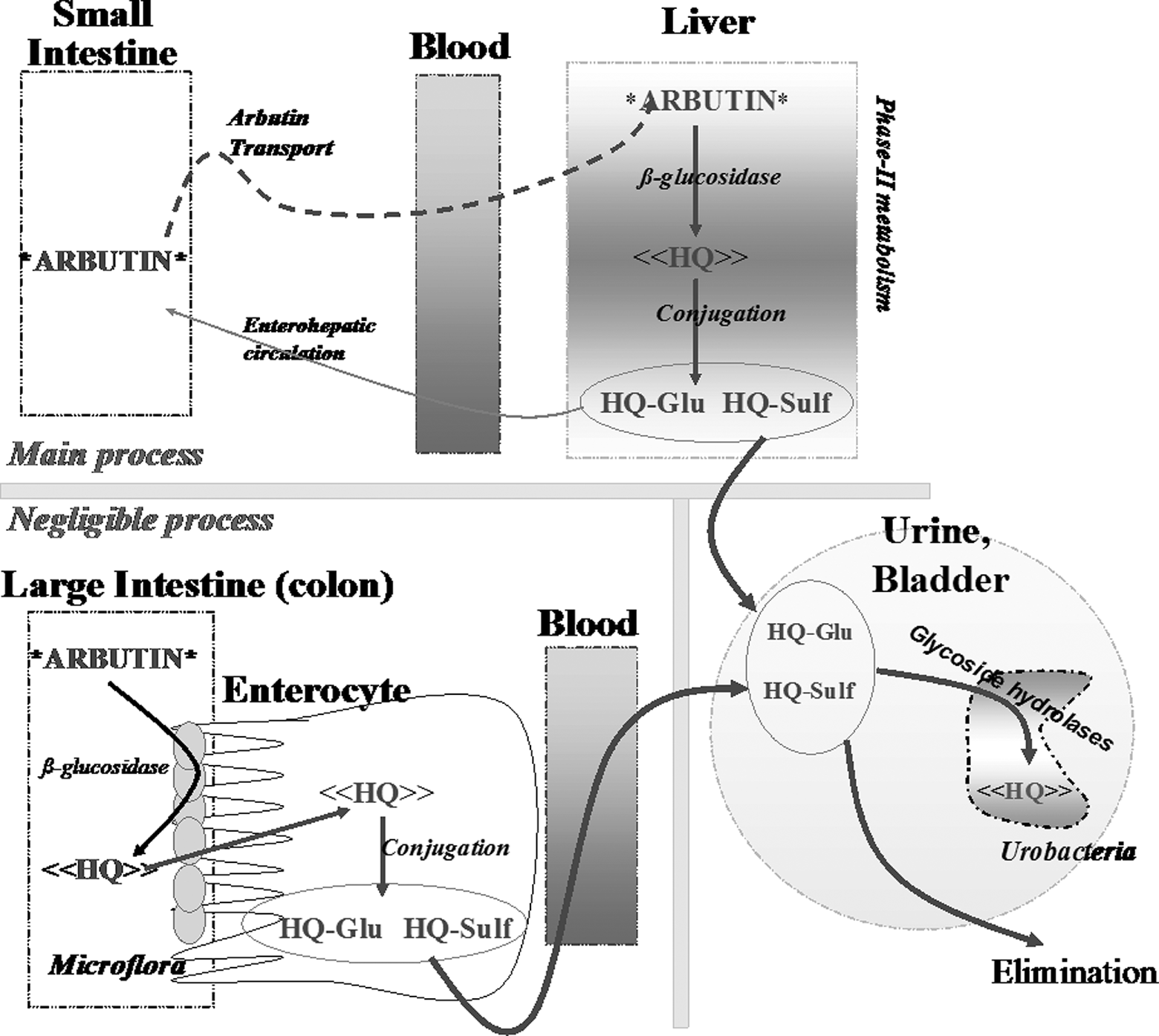
Schematic representation of the pharmacokinetic profile for arbutin and hydroquinone (HQ). HQ-Glu indicates HQ-glucuronide; HQ-Sulf, HQ-sulfate. Both HQ-conjugates were found in high concentration in urine and free HQ only in very small concentration or in traces. 9
Arbutin does not undergo hydrolysis in the acidic environment of the stomach and, therefore, in vivo would reach the small intestine intact. Experimental in vitro data as well as in vivo studies in rats and mice indicate that arbutin is absorbed via the Na+/glucose carrier in the small intestine, as it does in humans. 9,13,14
Arbutin (Figure 1A) in a human organism is rapidly absorbed in the small intestine and later metabolized in the liver (Figure 2). Thus, arbutin is first deglycosylated in the liver and immediately metabolized by conjugation with glucuronic acid or sulfate. Total arbutin metabolites (free HQ or HQ conjugated) are eliminated via both kidneys and urine. 15 –19 Up to 85% of the arbutin is eliminated in urine as HQ metabolite (HQ glucuronide and HQ sulfate) and only up to 0.6% of the dose was eliminated as free HQ. In humans, most healthy volunteers tested for arbutin derivative did not eliminate a detectable amount of free HQ in urine. 15 –19 Arbutin itself however has not been identified in human urine. 12 Experimental 11 and clinical 12 studies as well as the International Programme on Chemical Safety 20,21 and the toxicological evaluation done by NTP for free HQ 22 confirmed that HQ is devoid of potential for accumulation. The amount of free HQ detected in the tissues, organs, bones, and the like was less than 2% of the total arbutin/HQ dose administered. Extensive conjugation and rapid excretion, primarily via the urine, suggests that free HQ is effectively detoxified. 1,12,20
Therefore, from the entire daily dose of Uva-ursi extract ingested, only the portion of free HQ formed after liver metabolism and once eliminated by urine should be considered for safety concerns, as only the portion of non-metabolized HQ is regarded to be free for reacting with the human organism. The conjugates HQ glucuronide and sulfate, the major arbutin metabolites formed after oral human administration of Uva ursi extract of leaves, are not toxic. 1
Human Exposure to HQ and Arbutin
Herbal Medicinal Products
Herbal preparations of Uva-ursi folium for medicinal use are currently quantified to 23.5% to 29.3% of HQ derivatives calculated as anhydrous arbutin. 1 The recommended adult daily dose varies from 200 to 840 mg of HQ derivatives daily calculated as anhydrous arbutin. Commission E, WHO, and ESCOP monographs claim a daily dose ranging from 400 to 800 mg of arbutin per day divided into 2, 3, or 4 doses during a maximum period of treatment of 1 or 2 weeks, respectively. 2 –4 The WHO monograph also agrees with this dose although does not specify the duration of the treatment. 2
A pharmacokinetic study tested an HMPC-compliant Uva-ursi extract in 12 healthy volunteers at the daily dose of 420 mg HQ derivatives calculated as anhydrous arbutin. 16 This study reported that 70% (n = 11) of the arbutin ingested was eliminated after 36 hours in the form of glucuronide and sulfate conjugates via the urine. An average of 0.99 mg free HQ was recovered in the urine collected after 36 hours. 16 Of the 11 volunteers, 6 did not eliminate free HQ in the urine. Considering that the average of urine volume was up to 1500 mL/36 hours in this study, the exposure level of free HQ to liver, kidney, bladder, and other organs was estimated as approximately 0.66 µg/mL urine. Accordingly, from the therapeutic dose of 420 mg of HQ derivatives, calculated as anhydrous arbutin, only 0.99 mg of free HQ would be circulating and present in the urine and thus in contact with the human organism. This quantity of free HQ corresponds to an exposure level 11 µg/kg body weight (bw)/d free HQ (considering a mean human bw of 60 kg).
Food Sources
An analysis of food sources of HQ revealed significant amounts of arbutin as well as free HQ in numerous products humans consume. 12 The highest level of free HQ (0.02-0.05 µg/g) was found in pears. The most frequent exposure to free HQ from dietary intake occurs from consumption of wheat-derived foods (0.2 µg/g) that are present in the majority of meals.
One glass (200-300 mL) of coffee and tea contains 20 to 90 µg and 1 glass of red wine (250 mL) contains 562 µg of free HQ. 12,21 Considering that 2 to 3 glasses of coffee/tea and 1 glass of red wine are consumed in average for a 60-kg bw person per day, the level of exposure to free HQ would vary from 0.66 to 4.5 µg/kg bw/d to 9.3 µg free HQ/kg bw/d, respectively.
Smoke from 1 nonfiltered cigarette contains 110 to 300 µg free HQ. 22 Considering that the average of number of cigarettes smoked per day in Europe is 18, adults would be exposed to levels of free HQ ranging from 33 to 90 µg/kg bw/d.
The elimination of HQ products via urine after consuming a low-HQ or a high-HQ containing diet (4 mg/kg dose of free HQ) has been evaluated in a clinical study with 2 groups of 4 healthy volunteers. A greater rate of urinary excretion of HQ products was detected following the high-HQ diet, with a range of 2.5 to 5.5 mg of total HQ being excreted in the 8 hours following the ingestion of a high-HQ meal. Therefore, the human organs of this group were exposed to levels ranging from 125 to 275 µg/kg bw/d. No adverse reactions were observed in all the volunteers. 12
It should be noted that these foods are consumed several times a day and often for a lifetime without causing safety concerns nor requiring any special control or regulation.
Safe Human Exposure to Free HQ Regarding Permitted Daily Exposure and Thresholds of Toxicological Concern
A permitted daily exposure (PDE) estimates the dose below which there is a negligible risk to human health. This value is more accurate for estimating the risk. Several no-observed-effect levels (NOELs) from repeated free HQ administration in animal experiments are determined in subacute, chronic, and other toxicity studies and are used for the PDE calculation (Table 1).
Abbreviations: bw, body weight; LD50, lethal dose 50%; NOEL, no observed effect level.
Based on the NOEL as determined in a 13-week oral chronic toxicity study in rats (<25 mg/kg bw/d) 21,22 and according to the European Medicines Agency guideline for human PDE estimation, 25 the modifying factors of 5 for extrapolation from rats to humans, 10 for variability between individuals, and 5 for a 3-month toxicity study in rodents, a PDE for free HQ ([25 mg/kg bw/d x 50 kg] / [5 × 10 × 5]) of 5 mg/d for a person of 50 kg or 100 µg/kg bw/d can be estimated. This value is 9 times higher than in the maximum exposure level to free HQ in a worst-case scenario (11 µg/kg bw/d) after ingestion of a therapeutic human daily dose (HDD; 420 mg of arbutin) of a herbal medicinal product containing Uva-ursi folium.
Accordingly, the level of free HQ produced by the administration of a recommended daily dose (RDD) of Uva-ursi folium has negligible probability of any toxicological risk to human health.
Supporting this low human exposure level, the HMPC on Uva-ursi folium concluded that concentrations reached in the human body by monographed Uva-ursi folium products are below the most conservative thresholds of toxicological concern (TTC) value of 1 mg/mL. 1
In agreement with the above-mentioned statement, the benefit–risk ratio of the medicinal use of Uva-ursi folium extract has been considered favorable for the claimed indication and dosage by the German Commission E, the HMPC, ESCOP, and WHO monographs on Uva-ursi folium. 1 –4
In the United States, bearberry leaf extract is used as a urinary antiseptic and diuretic in a wide range of dietary supplements. 26,27 However, in the United States the data are not sufficiently robust to be approved for the suggested use. 28
Safety Pharmacology of Free HQ
To date, there are several human studies involving more than a thousand volunteers who have been administrated herbal products containing Uva-ursi folium extracts for 3, 4, 6, or 12 months without reporting any adverse event (AE) regarding hepatotoxicity, nephrotoxicity, or others (Table 2). These studies did not conform to International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), used a different dosage, and had longer periods of treatment than those recommended by the HMPC monograph. 1 Nevertheless, these studies provide information about human safety (Table 2).
Safety Data on Uva-ursi folium Preparations, Alone (Extract) or in Combination (comb), in Different Urological Indications.
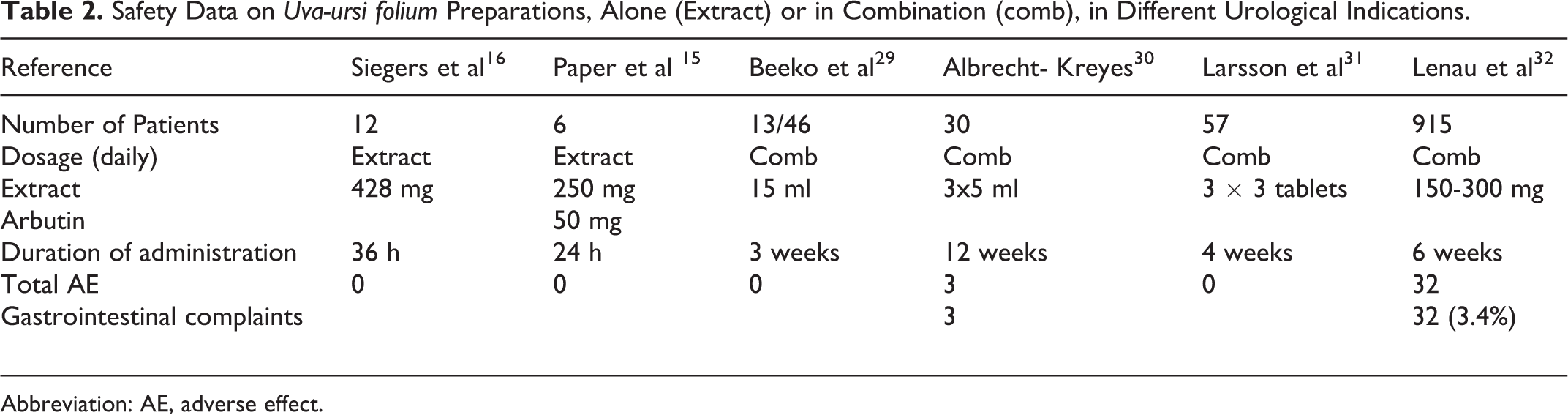
Abbreviation: AE, adverse effect.
During a 12-week therapy, 3 of 30 patients developed light gastrointestinal complaints, which did not force the patient to give up the therapy. 30 In all, 46 patients having urinary tract infections received a combination of herbal products including a bearberry leaf extract for 3 weeks, and only 1 patient had gastrointestinal problems. 29 Therefore, only gastrointestinal complaints have been reported from studies in humans, and there are no data available from studies with humans supporting the occurrence of other AEs. 33
Lenau et al 32 reported about 924 patients having an overactive bladder (with or without urge incontinence) and treated them for 1 and 5 weeks with 300 and 150 mg Uva-ursi folium extract, respectively. Only 3.4% of the patients reported gastrointestinal complaints (32 AE).
The current assessment report accompanying the HMPC monograph 1 reported that although several books described the safety concerns of long-term unsupervised use of bearberry leaf extract, 7,34,35 there are no case reports supporting these concerns. In animals, HQ toxicity has only been shown at very high doses.
Effect of Free HQ on Liver
No AEs were observed in dogs that received oral doses of 16 mg/kg for 26 weeks, 40 mg/kg bw for 49 weeks, or 16 mg/kg bw for 80 weeks. 36
Rats exposed to 25 and 100 mg/kg bw free HQ per day for 13 weeks did not show hepatotoxicity. 23 The International Program on Chemical safety (IPCS) 20 did not report experimental or clinical data supporting an effect of HQ in the liver. The HQ has been considered to be a relatively weak inducer of enzyme-altered foci in rat liver when tested for tumor-promoting activity in a liver focus test. 20
With the exception of ocular and dermal effects, there are no clearly demonstrated cases of prolonged HQ exposure in humans. 37 No hematological or urinary changes were observed in 2 male volunteers ingesting daily doses of 500 mg/d HQ for 2 months or in 17 male and female volunteers ingesting 300 mg/d HQ for 5 months. 22,37
Isolated case reports have postulated the occurrence of health effects without any specific evidence. For example, a report from Nowak 5 described hepatotoxicity in a dark room worker and attributed this finding to inhalation of HQ “fumes” or “dust.” Two letters to the editor criticized this assumption and clarified that free HQ was barely if at all present, is not volatile, and that no data were recorded to implicate HQ, other than its presence in the developing fluid with more organic solvents. 38,39 Thus, HQ could not be involved in the reported hepatotoxicity.
A cohort study of 858 men and 21 women involved in the manufacture and use of HQ was conducted to evaluate total mortality and cancer-related deaths. These individuals worked for a mean of 13.7 years in jobs that involved the manufacturing or use of HQ. No deaths from liver cancer or leukemia occurred during this time. 40
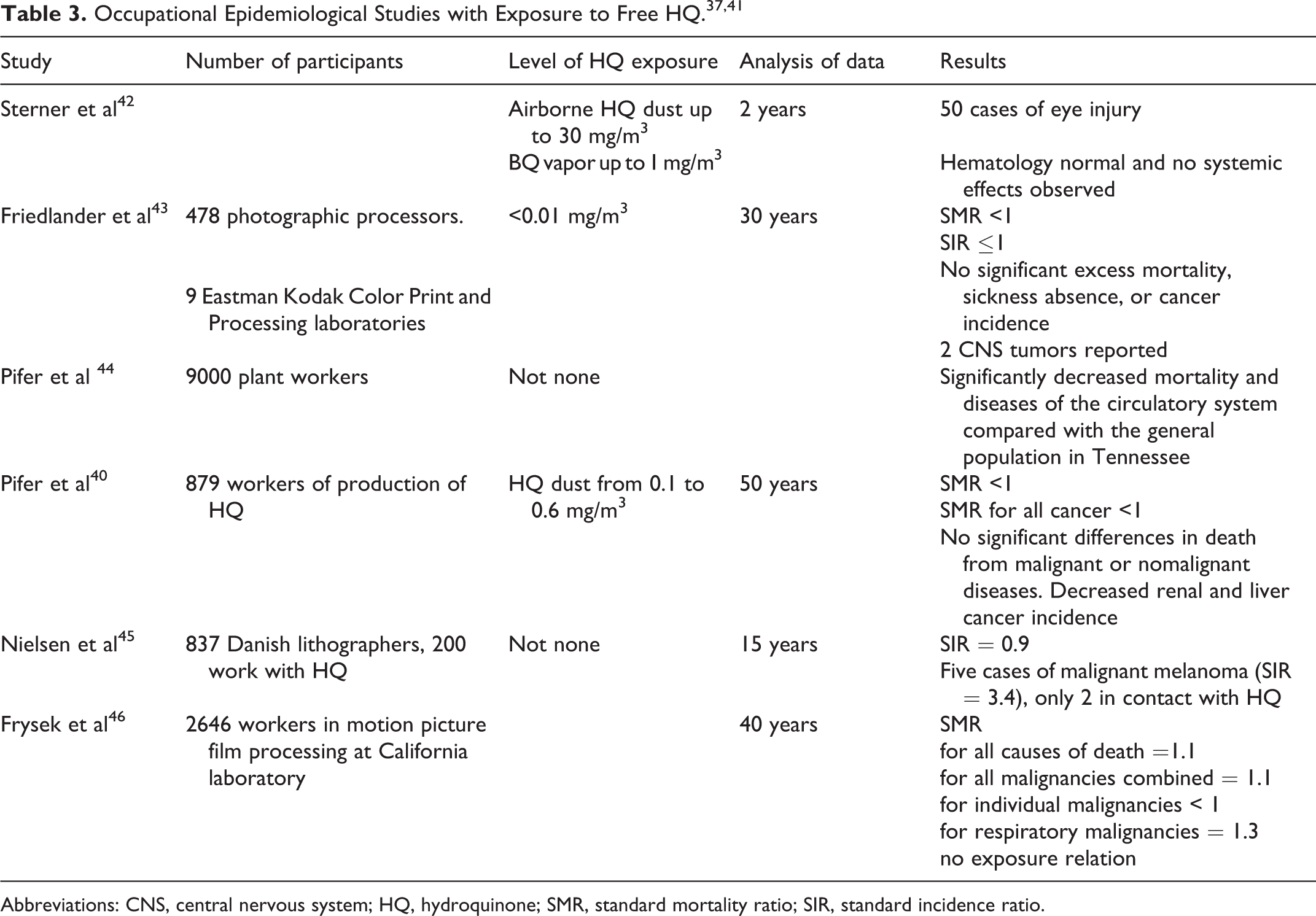
Several occupational epidemiological studies on workers exposed to HQ, via inhalation or dermal contact, provided no evidence for systemic toxic effects including hepatotoxicity, from the long-term occupational exposure to HQ 22,37,41 (Table 3).
Abbreviations: CNS, central nervous system; HQ, hydroquinone; SMR, standard mortality ratio; SIR, standard incidence ratio.
Therefore, there appears to be no clinical data that associate hepatotoxicity with arbutin or free HQ from dietary or herbal product consumption. Most of the publications related to hepatotoxicity of HQ are old, outdated, or misinterpreted. 1,22,37,47
Effect of Free HQ on Central Nervous System
Effects on the central nervous system (CNS) have only been seen in cases of acute human poisoning. 20,21 Symptoms that are associated with acute toxic exposure include tremor, convulsions, hyperexcitability, and dyspnea.
Death in humans has been reported after ingestion of 3 to 12 g of photographic-developing agents containing HQ at concentrations ranging from 0.5% to 10% combined with other organic toxic solvents. 21,24,37 In contrast, a controlled study on human volunteers reported that ingestion of 300 to 500 mg HQ daily (5 to 8.3 mg/kg bw/d) for 3 to 5 months did not produce any observable pathological changes in blood and urine. 20,21 This dose is 500-fold higher than the exposure level to HQ reached after a therapeutic HDD of a medical product containing Uva-ursi.
In experimental studies, CNS symptoms were observed at HQ oral doses close to lethal dose of 50% (LD50). Repeat dosing in rat and mouse studies caused tremors and reduced activity at doses ≥64 mg/kg and convulsions at doses ≥400 mg/kg. 22,27,41,50 These effects were reversible when exposure was discontinued. Topping et al 48 found that tremors occurred within 1 hour following dosing of 64 to 200 mg/kg bw/d for 13 weeks without neuropathological changes.
An NOEL for all CNS effects was experimentally estimated at 20 mg/kg bw/d. 20,21,24
Supporting the low human exposure level to free HQ, the HMPC monograph on Uva-ursi folium concluded that concentrations reached in the human body by monographed Uva-ursi folium-herbal medicinal products are below the most conservative TTC value of 1 mg/mL. 1
Effect of Free HQ on Renal Apparatus
Hydroquinone nephrotoxicity has been linked to the presence of HQ-glutathione conjugates, 47 which have been detected in animals after intraperitoneal (ip) /subcutaneous (sc) route of free HQ and in much lower degree (4% of de given dose) after oral administration. To date, however, the presence of HQ-glutathione conjugate in plasma/urine of humans has not been reported after oral administration. 1,11,22,37,47
DeCaprio 37 confirmed that after increasing the dose of ingested free HQ, the rates of urinary HQ metabolite excretion were greater than expected. In addition, HQ levels in human plasma and urine were unaffected by the ingestion of 1000 mg of acetaminophen, confirming the important detoxification process of HQ derivates in humans. In the control, nonexposed participants, the mean urinary rate was 115.4 µg/h and the background of total HQ concentration was reported in other studies as1.2 mg/kg, which is similar to values such as 0.6 µg/mL or 4.2 µg/mL. 37
Peters et al 49 provides data from an in vitro system supporting the toxicity of glutathione conjugates of tert-butyl-hydroquinone to the kidney and bladder but he does not provide evidence for the presence of HQ-glutathione conjugates in human urine or plasma after oral arbutin administration nor for the toxicity risk of free HQ in humans.
Although the use of bearberry extracts extends over several decades, no case reports of nephrotoxicity have been reported. 22,37,41,47
In experimental studies, after oral dosing of free HQ, the toxicity of HQ in rat kidneys has been concentrated in the outer stripe of the outer medulla (OSOM), particularly in the renal proximal tubules. 22,50,51 Expert literature reviews have reevaluated the focus of nephrotoxicity in this area of the kidney and concluded that HQ exacerbates chronic progressive nephropathy (CPN), a spontaneous, age-related disease occurring primarily in male rats with no known counterpart in humans. 22,37,41,47
Supporting this hypothesis, a reevaluation of histological data from the 1989 NTP 50 found that the degree of CPN in male F344 rats increased with HQ treatment. 22 Atypical tubule hyperplasias or adenomas were found only in areas of severe or end-stage CPN, and their association with the degree of CPN was statistically significant. 22
In addition, a 2-year feeding study (0.8% hydroquinone in diet) also found renal tubular hyperplasia and adenomas associated with CPN in male F344 rats. 52 Severity of CPN increased only slightly in female rats.
Hydroquinone administered via gavage for 6 weeks at 50 mg/kg bw to male F344 rats caused proximal tubular damage, as supported by increases in the rate of excretion of renal injury-specific enzymes. 53 Such renal toxicity was not observed in female rats. In this study, no nephrotoxicity was observed in Sprague-Dawley male and female rats.
Topping et al 48 confirmed the strain differences in susceptibility to HQ renal toxicity after repeated oral administration by gavage for up to 6 weeks at dose levels up to 50 mg/kg/d. Male F344 showed modestly increased indicators for renal toxicity, while female F344 and Sprague-Dawley rats did not show evidence of nephrotoxicity nor histological signals at dose level up to 200 mg/kg/d. 48
Expert evaluations 22,37,41,47 concluded that several glutathione compounds target a very specific region of the kidney—the outer stripe of the outer medulla. This suggests that a nongenotoxic mode of action that involves exacerbation of a spontaneously occurring rodent renal disease, CPN, seems to be the most valid reason.
This disease is particularly prominent in male rats, thus, species specific, and the evidence is consistent with an absence of any human cases. Therefore, the increased incidence of renal tubule adenomas in HQ-dosed male rats is not relevant to humans.
Carcinogenic Potential of Free HQ
Data From Experimental Studies
In a 2-year gavage study in which free HQ was administered in water to F344/N rats (25 or 50 mg/kg) and to B6C3F1 mice (50 or 100 mg/kg), survival rates were lower than that of the controls, and some evidence of carcinogenic activity was observed in all groups except the male mice. 50,51 Male rats developed renal tubular cell adenomas when given 25 mg/kg (4 of 55) and 50 mg/kg (8 of 55); female rats developed mononuclear cell leukemia when given 25 mg/kg (15 of 55), 50 mg/kg (22 of 55), and controls (9 of 55); female mice developed hepatocellular adenomas or carcinomas when given 50 mg/kg (16 of 55), 100 mg/kg (13 of 55) and the controls (3 of 55). 51
In 1995, Whysner et al 54 analyzed this published data regarding HQ’s carcinogenic potential including the analysis of renal adenomas in male F344 rats and its relation to areas of spontaneous progressive nephropathy. The authors concluded that although HQ is a metabolic product of benzene, several lines of evidence suggest that the effects of HQ oral exposure are significantly different from those of benzene. Based upon the plausible mechanisms by which HQ may produce kidney tumors in male rats, they considered that occupational exposure levels of HQ are not expected to be a cancer risk for humans.
Hard et al 55 reevaluated kidney sections from the 1989 NTP 50 study 51 and determined that free HQ did not directly cause renal tumor development but rather exacerbated existing CPN in the high-dose male rats and stimulated proliferation of renal tubular cells in the advanced stages of CPN. 55 In their evaluation, the authors found that 3 of 55 and 7 of 55 male rats given 25 and 50 mg/kg free HQ, respectively, had developed benign renal adenomas. In addition, the kidneys of 2 of 55 and 11 of 55 male rats (receiving 25 and 50 mg/kg, respectively) had 1 or 2 foci of atypical tubule hyperplasia. 55 The authors noted that the low-dose male rats exhibited some atypical hyperplasia and benign adenomas, which were not associated with an increase in CPN. 55
Because CPN is a spontaneous, age-related renal disease that affects various strains of rats but has no counterpart in humans, the authors suggest that this mechanism of HQ toxicity has little relevance to humans. 55,56 Regarding the potential for HQ carcinogenicity and risk to humans, Whysner 54 and McGregor 47 concluded that HQ is carcinogenic only in the context of end-stage CPN, which is irrelevant to humans. 47,54 Shibata et al 52 exposed male and female B6C3F1 mice to HQ via diet at concentrations of 0% to 0.8% for 96 weeks. The daily doses were 10-fold and 15-fold higher than that used by Kari et al. 51 The incidence of hepatocellular adenoma was increased from 21% in control males to 47% in exposed males (P < .05), whereas the incidence of hepatocellular carcinomas was unaffected by treatment. In female mice, zero incidences of hepatocellular adenoma and carcinoma in the control group were unaffected by treatment.
The mutagenic and genotoxicity capacity of free HQ is directly related to the exposure route. 22 Although most in vitro studies were mutagenic positive, in vivo studies are generally negative, mostly due to rapid detoxification, except when parenteral administration is used. A number of studies have shown in vitro formation of DNA adducts following exposure to free HQ. Similar adduction does not occur in vivo and is not likely to be involved in HQ toxicity. 22 According to the NTP, ip or sc routes of HQ exposure are considered inappropriate. 22
Five studies used an oral route for HQ administration: a mouse bone marrow cell micronucleus test in which a weak, marginally positive response was obtained following a single oral dose of 80 mg/kg body weight. The remaining oral route studies did not show a significant effect. They included a mouse bone marrow cell micronucleus test in which there was no genotoxic activity after exposure to a diet containing 0.8% free HQ for 6 days; 2 (32)P-post-labeling assays, one with targets of Zymbal gland, liver, and spleen in Sprague-Dawley rats and the other with the kidney as the target in F344 rats; and the last oral assay was for 8-hydroxydeoxyguanosine adducts in F344 rat kidney DNA. Thus, the evidence for any genotoxic effect in vivo is sparse and none has been observed in the kidney. 47
The potential of HQ to induce covalent DNA adducts in the kidney was investigated in male and female F344 rats after administration of 0, 2.5, 25, or 50 mg/kg HQ by gavage for 6 weeks.
53
The nuclear DNA isolated from kidneys was analyzed by the 32P-postlabeling assay. At 50 mg/kg, males, but not females, showed an increase in the rate of excretion of N-acetyl-β-
In conclusion, several tumors have been reported at a higher incidence in long-term HQ oral exposure experiments with rodents. Only rat renal tubule cell adenomas are clearly increased as a response to treatment. Therefore, it is proposed that: HQ produced renal adenomas in male F344 rats and these tumors arise from areas of spontaneous CPN; the nephropathy itself has been found to be increased by HQ. Other kinds of tumors were not confirmed to be causally related to HQ among the reported bioassays. In the male F344 rat, HQ administered alone was not DNA reactive. The HQ produced enhanced proliferation of renal tubular epithelium, presumably through toxicity involving glutathione conjugate formation. Glutathione conjugates could be responsible for the tumor induction, and careful histology shows that of the most actively toxic of several glutathione compounds tested, 2,3,5-triglutathion-S-yl hydroquinone targets a very specific region of the kidney, the outer stripe of the outer medulla, whereas HQ-associated adenomas are more randomly distributed and occur in the cortex as well as the medulla. The nongenotoxic carcinogenic mode of action involves exacerbation of a spontaneously occurring rodent renal disease, CPN: this disease is particularly prominent in male rats and the evidence is consistent with an absence of any human comparison. Although HQ is a metabolic product of benzene, several lines of evidence suggest that the effects of HQ oral exposure are significantly different from those of benzene. Based upon the plausible mechanisms by which HQ may produce kidney tumors in male rats, all reviews and assessment reports have concluded that occupational exposure levels of HQ are not predicted to be a cancer risk for humans.
Thus, this mode of action of HQ carcinogenesis has little relevance to humans. 55,56 The HQ is carcinogenic only in the context of end-stage CPN in rats, which is not relevant to humans. 22,47 Therefore, the increased incidence of renal tubule adenomas in HQ-dosed male rats is not applicable to human health. 22,47,54
Data from Human Studies
The evaluation on HQ carcinogenic risk made by IARC 41 concluded that HQ is not classifiable as to its carcinogenicity to humans (group 3).
It has been considered that there is inadequate evidence in humans and limited evidence in experimental animals regarding carcinogenicity of HQ. Thus, a discussion and some specialized reviews were required. 47
The epidemiological information comes from 4 cohort studies involving occupational exposures (Table 3). 40,44 –46 A cohort of lithographers, 200 of whom had worked with HQ, had an excess of malignant melanoma based on 5 cases, but only 2 of them had reported exposure to HQ. 45
In a study of photographic processors, the number of exposed individuals was uncertain and the numbers of cases of individual cancer sites were small. In view of the statistical limitations of these studies for individual diagnostic categories of cancers, they are not considered to be evidence with regard to the carcinogenicity of HQ. A cohort of workers with confirmed and lengthy exposure to HQ, during either its manufacture or its use, had low cancer rates compared with 2 comparison populations. The reason for the lower than expected rates is unclear. 40 In a motion picture film-processing group, there were significant excess malignancies of the respiratory system among workers engaged in developing, where there was exposure to HQ as well as other chemicals. 46
Studies in Table 3, involving more than 10 000 humans did not provide evidence of an increased risk of having cancer because of the exposure to free HQ. 40,42 –46 All of the standard relative risk values were below 1 or at worst = 1. This means that cancer is less likely to occur in the population exposed to HQ than in the nonexposed, or in the worst possible outcome, the risk is the same in both the groups. 40,42 –46
Free HQ Exerts Protective Effects Against Known Carcinogens
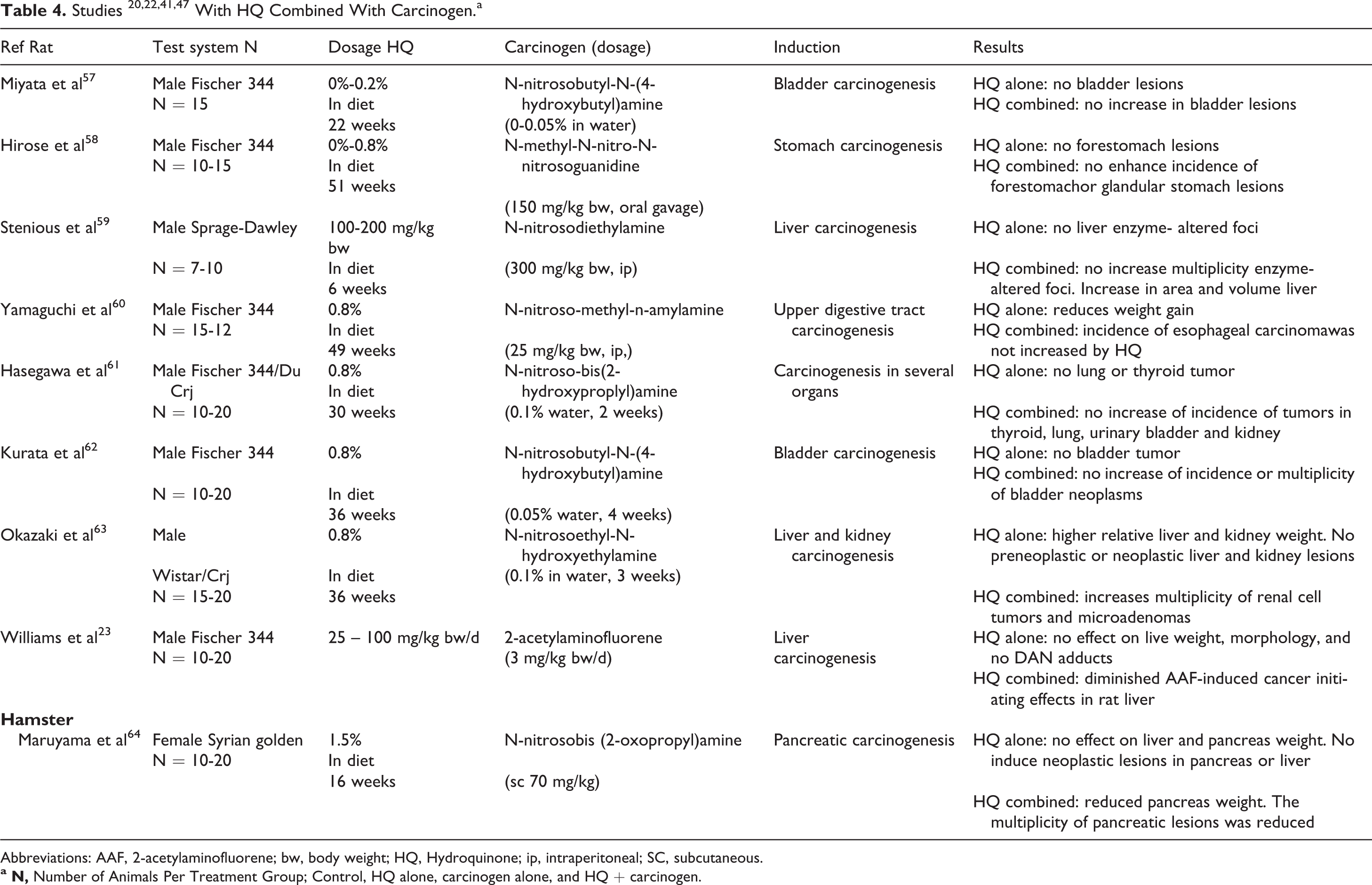
Nine experimental studies have investigated the carcinogenic potential of HQ alone or in combination with known carcinogens where tumors were the end point of the experiment (Table 4). In these studies, HQ was administered for a period varying from 6 to 51 weeks. The number of animals (rats or hamsters) per treatment group varies from 10 to 20.
Abbreviations: AAF, 2-acetylaminofluorene; bw, body weight; HQ, Hydroquinone; ip, intraperitoneal; SC, subcutaneous.
Hydroquinone alone did not increase the incidence of tumors in those organs examined (Table 4). Shibata et al 65 also supported the premise that HQ does not induce gastric tumors.
Hydroquinone given after an initial dose did not increase the incidence of urinary bladder tumors in rats induced by N-nitrosobutyl-N-(-4hydroxybutyl)amine 57,62 ; thyroid, lung, kidney, or urinary bladder tumors in rats induced by N-nitroso-bis(2-hydroxyproplyl)amine 61 ; forestomach tumors in rats induced by N-methyl-N-nitro-N-nitrosoguanidine 58 ; or pancreatic tumors in Syrian hamsters induced by N-nitrosobis(2-oxopropyl)amine. 64
The effects of HQ have also been evaluated on altered hepatocytes. 59 The HQ administered alone had no effect on γ-glutamyltranspeptidase foci in the liver of partially heapatectomized rats. In combination with N-nitrosodiethylamine, the multiplying of foci was not increased, although there were increases in area and volume. Thus, HQ showed a protective effect on the induction of liver-altered foci following diethynitrosamine exposure coupled with partial hepatotectomy. 59
Williams et al 23 confirmed the protective effect of HQ on the initiating effects in the rat liver of the DNA-reactive carcinogen 2-acetylaminofluorene (AAF) at doses varying from 25 and 100 mg/kg bw/d.
Male Fischer 344 rats were treated with AAF and fed daily with approximately 25 and 100 mg/kg bw/d HQ ad libitum for 13 weeks, starting 1 week before AAF administration commenced. The rats were observed daily and bws were taken before initial dosing and at weekly intervals thereafter. Body weight gain over time, bw, and absolute (mg) and relative liver weights were measured. At the end of the 13 weeks, DNA adducts ([32]P-postlabeling), cell proliferation (proliferating cell nuclear antigen immunohistochemistry), and preneoplastic hepatocellular altered foci (HAF; glutathione S-transferase-placental type immunohistochemistry) were measured. No significant differences were observed in bw gains or liver weights. The AAF produced liver DNA adducts, and at low dose of HQ adduct levels were 90% of that for AAF alone, whereas at high dose adducts were reduced by 33% (P < .05). The AAF exposure yielded about a 50% increase in hepatocellular proliferation and both HQ doses reduced the AAF-induced increases in proliferation by about 25%. Likewise, the AAF-induced Glutathione S-transferase-placental form (GST-P)-positive hepatocellular altered foci (HAF) per cm2 of liver tissue were decreased by about 50% both doses of HQ. Thus, under the conditions of this experiment, HQ at both 25 and 100 mg/kg bw/d in the diet diminished AAF-induced cancer-initiating effects in the rat’s liver. 23
Additionally, in vitro studies have also provided information about the lack of carcinogenetic potential of HQ and the antiproliferative effects of arbutin. 66 In this case, experimental work tested the effects of arbutin on TCCSUP human bladder carcinoma cell proliferation. Arbutin did not exhibit any cytotoxic effects in TCCSUP cells at concentrations of ≤500 µg/mL. In addition, arbutin significantly decreased TCCSUP cell proliferation in a concentration- and time-dependent manner (0-500 µg/mL). Furthermore, cell cycle analysis revealed that arbutin strongly disrupted the cell cycle in a time-dependent manner. Western blot analysis demonstrated that arbutin led to the inactivation of extracellular signal-regulated kinase (ERK), which is known to critically regulate cell proliferation. In addition, arbutin markedly increased the expression of p21WAF1/CIP1 (p21), which is known to be highly involved in cell cycle regulation. Therefore, this study suggests that arbutin inhibits TCCSUP cell proliferation via ERK inactivation and p21 upregulation. 66
The effect of arbutin on tumorigenesis and on the gene expression level of human skin with A375 human malignant melanoma cells has also recently been evaluated. 67 The DNA microarray analysis provided the differential gene expression pattern of arbutin-treated A375 cells with the significant changes in 324 differentially expressed genes, containing 88 upregulated genes and 236 downregulated genes. The gene ontology of differentially expressed genes was classified as belonging to cellular component, molecular function, and biological process. In addition, 4 downregulated genes of AKT1, CLECSF7, FGFR3, and LRP6 served as candidate genes and correlated to suppress the biological processes in the cell cycle of cancer progression and in the downstream signaling pathways of malignancy of melanocytic tumorigenesis. 67
In summary, HQ had no promoting activity on pancreatic, bladder, stomach, and liver carcinogenesis in most of the studies. In none of the studies with HQ, an increased incidence of malignant neoplasm related to treatment was detected. The only consistent neoplasm has been observed in renal tubule in cell adenoma in male F344 rats. The mechanism of this tumor induction has no consequences for humans.
Conclusions: Evidence-Based Safety of Therapeutical HDD of Uva-ursi Folium Extract, Implications for Human Health
There is no evidence that free HQ liberated from a therapeutic exposure level of Uva-ursi folium extracts could exert hepatotoxicity, carcinogenesis, or induce convulsions in humans. Safety issues regarding free HQ exposure have repeatedly been linked to the therapeutic use of herbal medicinal products containing arburtin, which is not justified by the current scientific knowledge.
Ingested arbutin as well as free HQ are extensively metabolized and only a very low percentage is eliminated as free HQ. Therefore, only this free HQ amount should be considered for discussion of safety concerns, since only the not-metabolized and reactive part of the total arbutin could interact with the liver, kidney, bladder, and other human organs. The HQ conjugate is not reactive, not toxic, and is eliminated in urine without interacting with any organ or tissue.
Dietary sources of HQ/arbutin as well as other products such as cigarettes consumed daily by many humans have shown to generate comparable or even higher exposure levels to free HQ than to the HDD of Uva-ursi folium extract recommended by the HMPC monograph. It should be noted that these products are consumed many times a day over the course of a lifetime, by all segments of the overall population.
The level of free HQ produced by the administration of a therapeutic HDD was estimated to be in the order of 11 µg/kg bw/d. This exposure level represents 11% of the PDE dose below which there is a negligible risk to human health.
Central nervous system stimulation has only been shown at doses close to lethal or poisoning doses. No case reports have been found with human data.
Toxicity on renal apparatus, nephrotoxicity, has only been reported in rats, and several evaluations of those studies concluded that the nephrotoxicity shown in rats is not relevant to humans.
Hepatotoxicity has been linked to free HQ in outdated publications, which have been later actively criticized because of their lack of evidence. No reports regarding hepatotoxicity in humans have been found to date. Several occupational epidemiological studies including more than 10 000 workers with exposure to free HQ and other organic solvents for more than 15 years did not show any incidence of an increase in nephrotoxicity or hepatotoxicity. These studies did not provide any evidence of an increased risk of having cancer due to the exposure to free HQ, providing standard relative risk values of less than 1 or in the worst case equal to 1. Accordingly, cancer is less likely to occur in the population exposed to HQ than those not exposed, or in the worst-case scenario, the risk is the same in both the groups.
Nine experimental studies gave evidence that free HQ alone did not increase the incidence of tumors in the urinary bladder, thyroid, lung, liver, kidney, forestomach, and pancreas even after an initiating dose of a known carcinogen. In some cases, it was observed to have a significant protective effect.
As is the case with other medicinal herbs, it is recommended that bearberry extracts should comply with the quality requirements and dosage guidelines of international experts.
In summary, under the recommended use conditions, extracts of Uva-ursi folium are a safe therapeutic option for the treatment of lower urinary tract infections. The use of Uva-ursi folium in herbal medicinal products has a historic tradition. Despite the long-term experience with Uva-ursi folium extracts, no case report has been published to date regarding toxicity in either the liver or the kidneys, nor has it been linked to tumors in humans. In addition, HQ showed no promoting activity on pancreatic, bladder, stomach, or liver carcinogenesis. In none of the experimental studies with free HQ, an increased incidence of malignant neoplasm related to treatment was detected.
Nevertheless, the most relevant monographs consider reserving a level of safety concerns, since the whole toxicological profile of free HQ is still not completely known. For this reason, further research in this area is warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: the authors work for the pharmaceutical company Schaper and Brümmer GmbH & Co KG.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.