Abstract
The CIR Expert Panel assessed the safety of dicarboxylic acids and their salts and esters as used in cosmetics. Most dicarboxylic acids function in cosmetics as pH adjusters or fragrance ingredients, but the functions of most of the salts in cosmetics are not reported. Some of the esters function as skin conditioning or fragrance ingredients, plasticizers, solvents, or emollients. The Expert Panel noted gaps in the available safety data for some of the dicarboxylic acid and their salts and esters in this safety assessment. The available data on many of the ingredients are sufficient, however, and similar structural activity relationships, biologic functions, and cosmetic product usage suggest that the available data may be extrapolated to support the safety of the entire group. The Panel concluded that the ingredients named in this report are safe in the present practices of use and concentration.
Keywords
Introduction
The safety of sebacic acid and other alkyl α,ω-dicarboxylic acids, and their salts, monoesters and diesters as used in cosmetics, has been reviewed by the CIR Expert Panel (the Panel). The dicarboxylic acids are terminally functionalized straight alkyl chains characterized by a separation between the carboxylic acid functional groups of 1 to 10 carbons (1 carbon separation, 3 carbons total (C3) = malonic acid; 2 carbons separation (C4) = succinic acid; 3 carbons separation (C5) = glutaric acid; 4 carbons separation (C6) = adipic acid; 7 carbons separation (C9) = azelaic acid; 8 carbons separation (C10) = sebacic acid; and 10 carbons separation (C12) = dodecanedioic acid). The simple alkyl diesters are the result of the condensation of alkyl dicarboxylic acids and 2 equivalents of alkyl alcohols. The simple alkyl esters (mono- and di-) of these dicarboxylic acids have straight or branched side chains ranging in length from 1 to 18 carbons. Throughout this report, the data are presented by order of acid chain length (ie, beginning with malonic acid and ending with dodecanedioic acid; and beginning with dimethyl malate and ending with diisocetyl dodecanedioate).
This report presents available information in 2 groups, the 12 alkyl dicarboxylic acids/salts and the 44 corresponding (mono- and di-) esters.
The alkyl dicarboxylic acids and salts include:
malonic acid
succinic acid
sodium succinate
disodium succinate
glutaric acid
adipic acid
azelaic acid
dipotassium azelate
disodium azelate
sebacic acid
disodium sebacate
dodecanedioic acid.
The esters include:
diethyl malonate
decyl succinate
dimethyl succinate
diethyl succinate
dicapryl succinate
dicetearyl succinate
diisobutyl succinate
diethylhexyl succinate
dimethyl glutarate
diisobutyl glutarate
diisostearyl glutarate
dimethyl adipate
diethyl adipate
dipropyl adipate
dibutyl adipate
dihexyl adipate
dicapryl adipate
di-C12-15 alkyl adipate
ditridecyl adipate
dicetyl adipate
diisopropyl adipate
diisobutyl adipate
diethylhexyl adipate
diisooctyl adipate
diisononyl adipate
diisodecyl adipate
dihexyldecyl adipate
diheptylundecyl adipate
dioctyldodecyl adipate
diisocetyl adipate
diisostearyl adipate
isostearyl sebacate
diethyl sebacate
dibutyl sebacate
dicaprylyl/capryl sebacate
diisopropyl sebacate
diethylhexyl sebacate
dibutyloctyl sebacate
diisooctyl sebacate
dihexyldecyl sebacate
dioctyldodecyl sebacate
diisostearyl sebacate
dioctyldodecyl dodecanedioate
diisocetyl dodecanedioate
The acids and their salts included in this report function in cosmetics as pH adjusters, and the esters function as fragrance ingredients, plasticizers, skin-conditioning agents or solvents, and corrosion inhibitors. CAS numbers, definitions, structures and functions for the alkyl dicarboxylic acid, salt, and ester ingredients included in this report are given in Table 1.
Definitions, Functions, and Structures of Dicarboxylic Acid, Sal,t and Ester Ingredients in This Safety Assessment

A safety assessment of diethylhexyl adipate (often inaccurately named dioctyl adipate) 1 and diisopropyl adipate was published in 1984, with the conclusion that these ingredients are safe as used in cosmetics. 2 The safety of these ingredients was re-reviewed and confirmed in 20053 and 2006. 4 Additionally, dibutyl adipate was originally reviewed in 1996, and at that time the available data were found insufficient to support the safety of dibutyl adipate in cosmetic formulations. When re-reviewed in 2006, additional data were made available to address the data needs identified by the CIR Expert Panel, and an amended conclusion was issued stating that dibutyl adipate is safe for use in cosmetic formulations. 5
In order to focus on the acids and their salts separately from the dicarboxylic acid esters, this report is presented in 2 sections.
Part I: Alkyl Dicarboxylic Acids and their Salts
Chemistry
Method of Manufacture
While many of the alkyl dicarboxylic acids are present in natural sources, commercial production of these acids has historically occurred via alkali pyrolysis of lipids. 6 For example, when castor oil (a lipid which is comprised of approximately 84% ricinoleic acid side chain bearing triglycerides) is pyrolyzed with sodium hydroxide, some of the major products are sebacic acid and 2-octanol (Figure 1). 6 Sodium and potassium salts of the alkyl dicarboxylic acids are readily prepared via addition to the appropriate stoichiometric equivalent/equivalents of sodium hydroxide or potassium hydroxide, respectively.
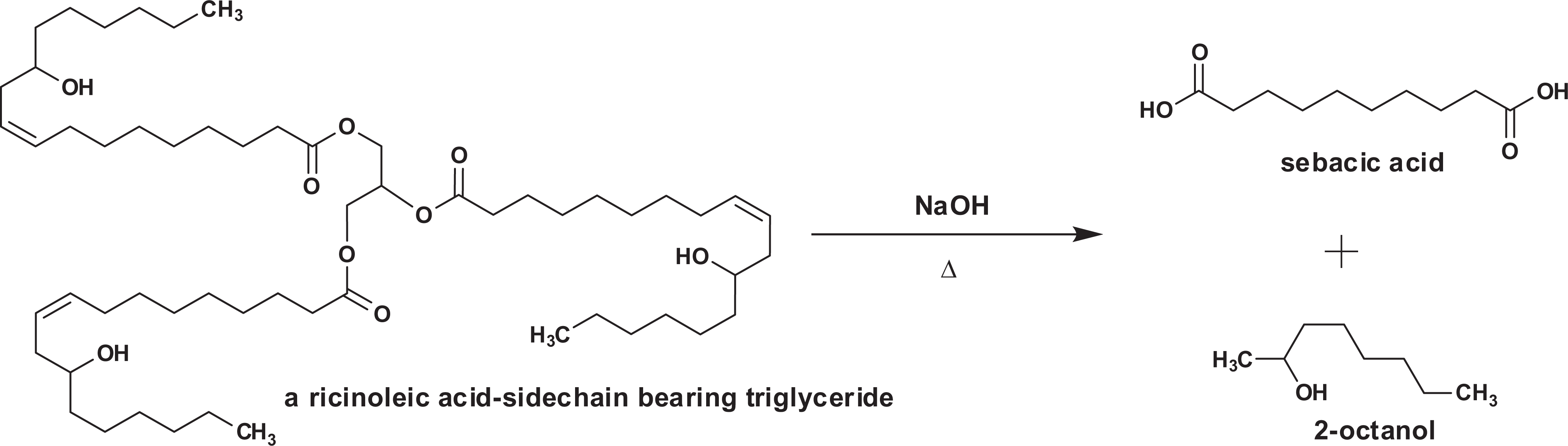
Sebacic acid synthesis from castor oil.
Some of the ingredients in this assessment are tallow derivatives. The CIR accepts the Food and Drug Administration (FDA) determination (21 CFR 700.27(a)) that tallow derivatives are not prohibited cattle materials and may be used in cosmetics.
Malonic acid (C3)
Malonic acid, first prepared by malic acid oxidation, is commonly manufactured by more recent methods including the ozonolysis of cyclopentadiene or the air oxidation of 1,3-propanediol. 7
Succinic acid (C4)
Succinic acid is an intermediate of the citric acid cycle and is found in almost all plant and animal cells, although at very low concentrations. 8 Succinic acid is commonly produced synthetically by catalytic (eg, nickel or palladium catalyst) hydrogenation of maleic anhydride.
Glutaric (C5) and adipic(C6) acids
Although glutaric acid is often encountered in nature, adipic acid is not commonly encountered in nature. Glutaric and adipic acids were first synthesized by oxidation of castor oil with nitric acid. However, adipic acid is now more commonly manufactured by the oxidation of cyclohexane, cyclohexanol, or cyclohexanone, and glutaric acid may be manufactured by ozonolysis of cyclopentene. 9
Azelaic acid (C9)
Azelaic acid, first detected in rancid fats, was originally produced via nitric acid oxidation of oleic acid. 10 Azelaic acid is a naturally occurring dicarboxylic acid that can be found in dietary sources, such as whole grains. 11 Azelaic acid is commonly manufactured by oxidative cleavage of oleic acid (obtained from grease or tallow) with chromic acid, nitric acid, or by ozonolysis.10,7
Sebacic acid (C10)
Sebacic acid was originally isolated from distillation products of beef tallow. More recently, however, sebacic acid has been manufactured via alkali pyrolysis of castor oil, as mentioned above and drawn in Figure 1, or by alkali pyrolysis of ricinoleic acid.12,7
Dodecanedioic acid (C12)
Dodecanedioic acid can be manufactured by fermentation of long-chain alkanes with a specific strain of Candida tropicalis. 13 Another method of manufacture involves the nitric acid oxidation of a mixture of cyclododecanone and cyclododecanol. 7
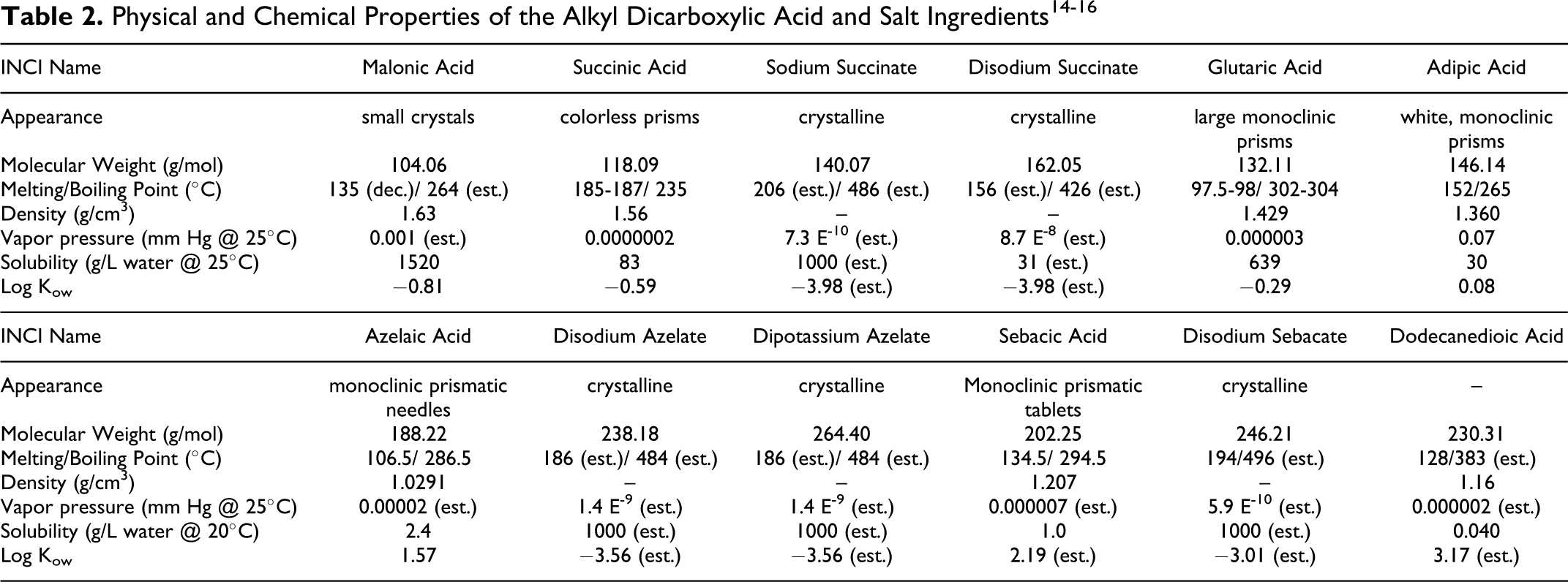
Physical and Chemical Properties
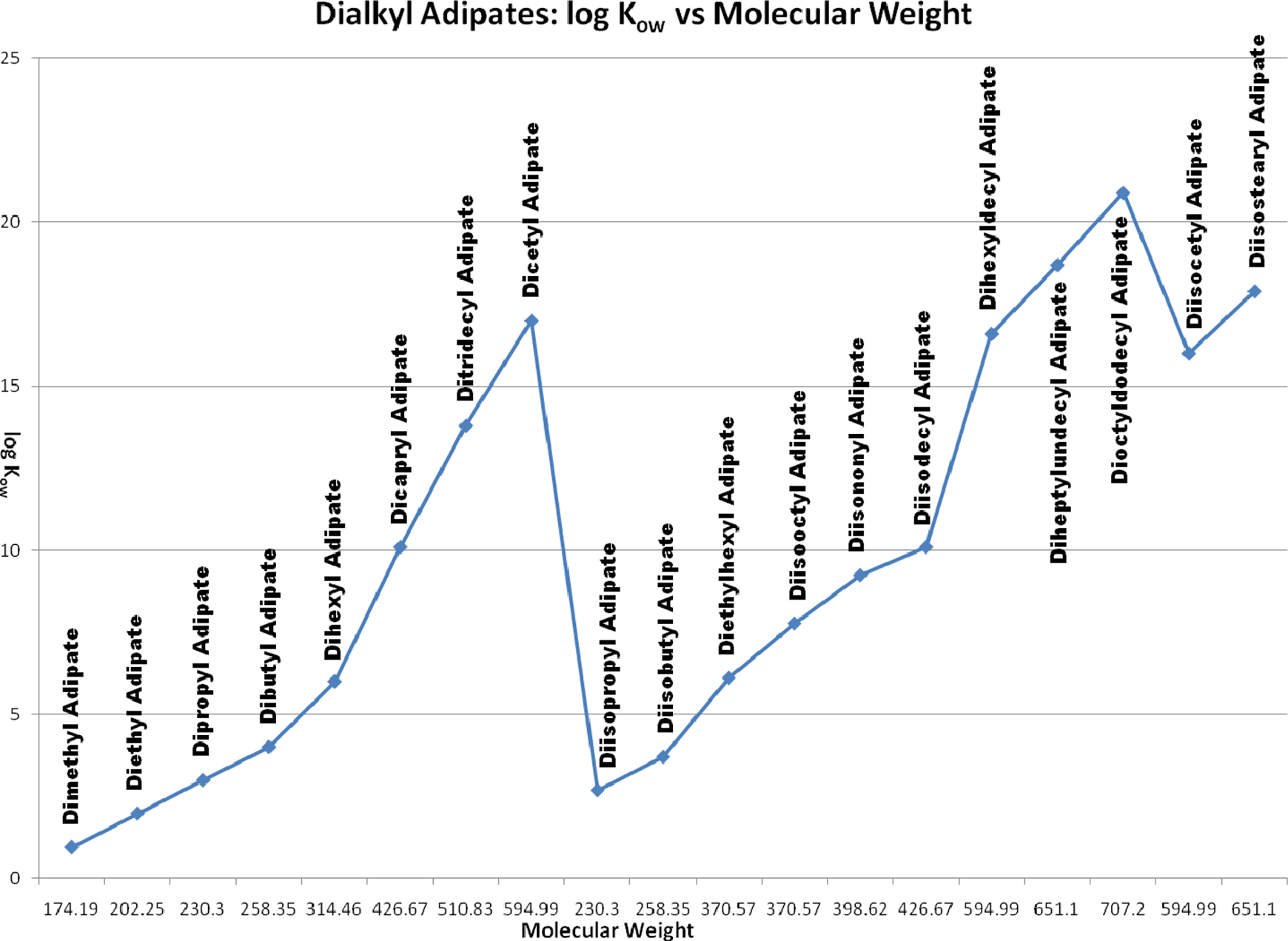
Table 2 lists the physical and chemical properties of the dicarboxylic acids and salts. Figure 2 presents the relationship between molecular weight of these ingredients and the octanol/water partition coefficient expressed as log Kow.
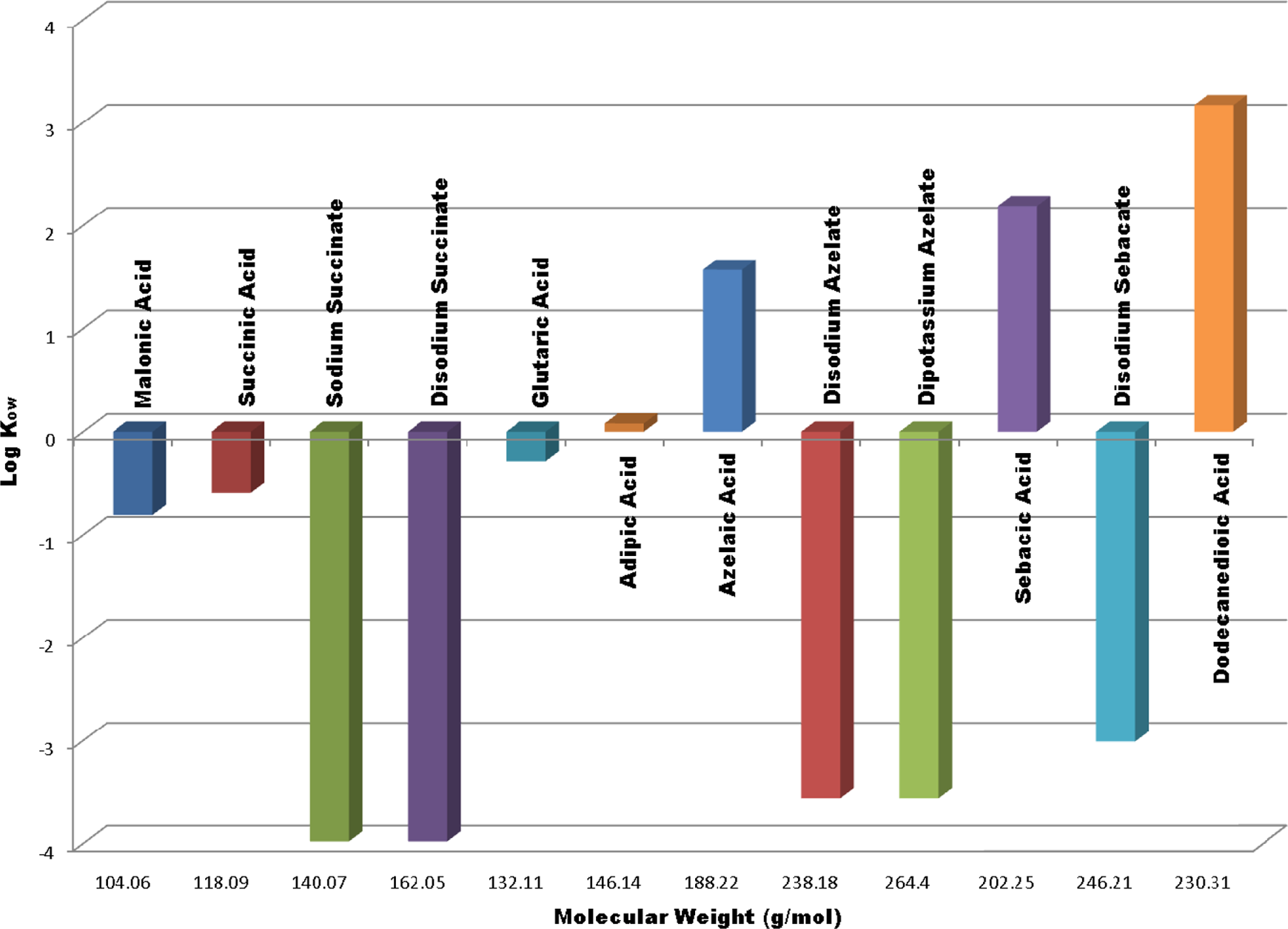
Dicarboxylic acids and their Salts; Log Kow vs molecular weight
The alkyl dicarboxylic acids vary considerably in their physical properties. The shorter chain (malonic, succinic, and glutaric) members are crystalline solids, very water soluble, and have limited solubility in organic solvents. As the chain length increases through adipic to dodecanedioic, water solubility decreases sharply (although still soluble in hot water). In other words, the water solubility of these acids is inversely proportional to their chain length. There is a marked alternation in melting point with changes in carbon number from even to odd. 7 Odd members (eg, malonic acid and glutaric acid) exhibit lower melting points and higher solubility than even carbon number alkyl dicarboxylic acids (eg, succinic acid and adipic acid). These alternating effects are believed to be the result of the inability of odd carbon number compounds to assume an in-plane orientation of both carboxyl groups with respect to the hydrocarbon chain.
Dicarboxylic acids react with Brønsted-Lowry bases (eg, sodium hydroxide) to form carboxylate salts (eg, sodium succinate or disodium succinate). Dicarboxylic acids also react with alcohols to give mono- and di-esters, such as those in this report.
Analytical Methods
Succinic acid
Methods used to analyze succinic acid include acidimetric titration for acidity; comparison with platinum–cobalt (Pt-Co) standard calibrated solutions for color; oxidation with potassium permanganate for detection of unsaturated compounds; atomic absorption or plasma spectroscopy for metals; and titration with silver nitrate or barium chloride for chloride or sulfate detection, respectively. 7 Small concentrations of succinic acid can be detected by common instrumentation such as gas–liquid chromatography (GLC) and polarography.
Adipic acid
Adipic acid can be extracted from a water sample and analyzed by gas chromatography/mass spectrometry. 17
Sebacic acid
Gas chromatography can be used to identify sebacic acid in air. 18
Diisopropyl adipate and diethylhexyl adipate
Diisopropyl adipate and diethylhexyl adipate can be identified through standard infrared (IR) spectroscopy. Gas–liquid chromatography, liquid–liquid extraction, mass spectrometry, and high-pressure liquid chromatography (HPLC) are also methods of analysis for the adipates. 2
Ultraviolet Absorption
The dicarboxylic acids and their salts included in this review would not be expected to have any meaningful ultraviolet (UV) absorption. Except for the acid functional group, these ingredients do not possess any conjugated π bonds or nonbonding electrons. The π bonds and nonbonding electrons in the acid functional group are not part of any conjugated systems. Accordingly, these ingredients are unlikely to absorb light within the UVA-UVB spectrum at a detectable molar absorptivity.
Use
Cosmetic
The ingredients included in this safety assessment have a variety of functions in cosmetics. 19 The majority of the dicarboxylic acids function in cosmetics as pH adjusters or fragrance ingredients. The functions of most of the salts are not reported, but it is stated that sodium succinate functions as a buffering agent or pH adjuster. The functions of all ingredients are listed in Table 1.
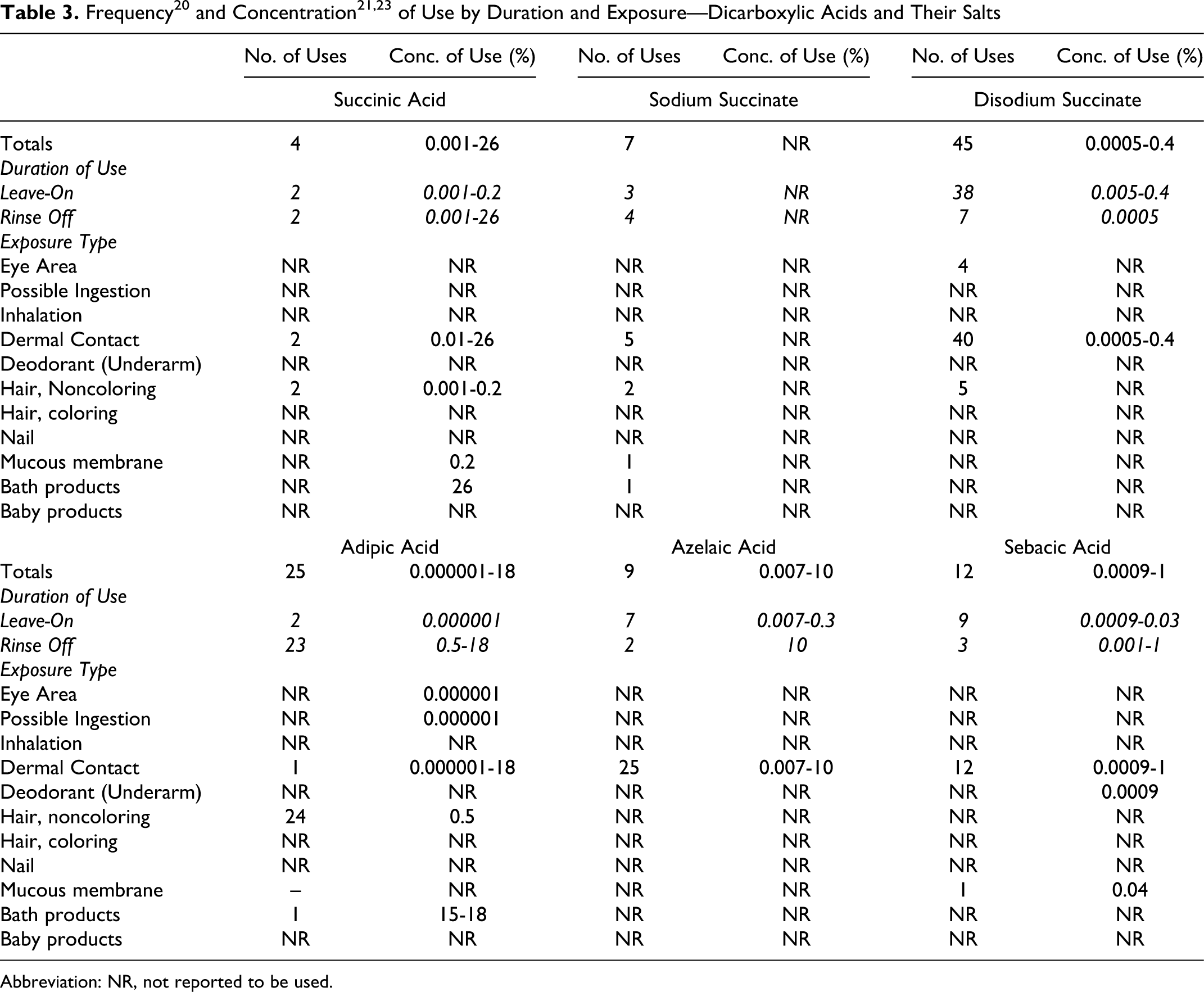
Six of the 12 dicarboxylic acids and their salts included in this safety assessment are reported to be used in cosmetic formulations. The frequency of use of the acids and salts, as supplied to the FDA by industry in 2010 as part of the Voluntary Cosmetic Registration Program (VCRP), 20 and the concentration of use, as supplied by industry in response to Personal Care Products Council (Council) surveys in 200921 and 2010, 22,23 are found in Table 3.
Abbreviation: NR, not reported to be used.
Acute Toxicity—Dicarboxylic Acids and Their Salts

For the dicarboxylic acids and their salts, disodium succinate has the greatest number of reported uses, with a total of 45. The acid with the highest concentration of use is succinic acid, with a use concentration of up to 26%; use at this concentration is in a bath product that will be diluted for use. The highest leave-on concentration is 0.4% disodium succinate, with dermal contact exposure.
Some of the ingredients are applied around the eye, can possibly be ingested, or involve mucous membrane exposure, and some are used in underarm deodorants. None are reported to be used in baby products.
The dicarboxylic acids and their salts are in the European Union (EU) inventory of cosmetic ingredients. 24
Noncosmetic
Many of the dicarboxylic acids and their salts are used in foods as direct or indirect food additives. The alkyl dicarboxylic acids are unusually versatile because of their 2 carboxyl groups. 9 This enables many additional types of useful reactions, particularly the manufacture of polymers (eg, nylon). The most common uses include functions as plasticizers, lubricants, and building blocks in the manufacture of polyesters, polyamides, and other plastics. The alkyl dicarboxylic acid salts are used to synthesize cyclic ketones, including commercially used macrocyclic musk compounds. 25
Malonic acid
Malonic acid is a useful intermediate in the manufacture of barbiturates. 14
Succinic acid
Succinic acid is listed by the FDA as a food additive that is generally recognized as safe (GRAS). 26 Succinic acid is also utilized in detergents, pigments, toners, cement additives, soldering fluxes, and as an intermediate in the synthesis of a number of pharmaceutical products. 7
Adipic acid
Adipic acid is listed as a GRAS food additive by the FDA. 27 Adipic acid has several industrial uses in the production of adhesives, plasticizers, gelatinizing agents, hydraulic fluids, lubricants, emollients, polyurethane foams, leather tanning, and urethane. 7 However, the bulk of the industrial production of adipic acid is driven by its usefulness in the manufacture of nylon-6,6 (in combination with 1,6-hexanediamine).
Azelaic acid
FDA has approved azelaic acid for use in treating acne and rosacea. A skin cream containing 20% (w/w) azelaic acid is indicated for the topical treatment of mild-to-moderate inflammatory acne vulgaris, 28 and a gel containing 15% azelaic acid is approved for treating rosacea. 29 These drugs are available by prescription only. (As a reference point, azelaic acid is reported to be used in cosmetics at 0.3% in leave-on and 10% in rinse-off formulations that have dermal exposure. 23 )
Azelaic acid is used in the manufacture of plasticizers, lubricants, and greases. Azelaic acid was identified as a molecule that accumulated at elevated levels in some parts of plants and was shown to be able to enhance the resistance of plants to infections. 30
Sebacic acid
Before 1973, sebacic acid was widely used in the United States, as an aromatic in food. 31
Sebacic acid is used in resorbable polymer systems that deliver chemotherapeutic agents (eg, cisplatin, carboplatin) that are implanted at the site of tumors to provide for sustained release of the drugs. 32 Sebacic acid and its derivatives have a variety of industrial uses as plasticizers, lubricants, diffusion pump oils, candles, and as intermediates in the synthesis of polyamides and various alkyd resins. 7
Dodecanedioic acid
Dodecanedioic acid is used in the production of nylon (nylon-6,12), polyamides, coatings, adhesives, greases, polyesters, dyestuffs, detergents, flame retardants, and fragrances. 33
Diethyl malonate
Diethyl malonate finds great utility as the starting material in malonic ester synthesis, a classic organic chemistry reaction wherein a very wide variety of esters can be synthesized. 25
Diisobutyl adipate
Diisobutyl adipate is considered by FDA to be a Prior-Sanctioned Food Ingredients, Plasticizer (21 CFR § 181.27).
Diethylhexyl adipate
Diethylhexyl adipate is used as a plasticizer for polyvinyl chloride (PVC) plastics. 34
Diethyl sebacate
Before 1973, diethyl sebacate was widely used in the United States, as an aromatic in food. 31
Dibutyl sebacate
Dibutyl sebacate is a component of PVC. 35
Toxicokinetics
Dicarboxylic acids are natural metabolic products of the ω-oxidation of monocarboxylic acids when the β-oxidation of free fatty acids is impaired. 36 Under normal physiological conditions, dicarboxylic acids are rapidly β-oxidized, resulting in very low cellular concentrations and practically nondetectable concentrations in the plasma. 37 Medium-chain dicarboxylic acids (up to 12 carbon atoms) are β-oxidized in mitochondria and peroxisomes. Oxidation of odd- and even-numbered chains proceeds to different end points. Odd-chain dicarboxylic acids are β-oxidized, giving acetyl-CoA and malonic acid (C3). Oxidation can then go no further, and malonic acid is the starter of fatty acid synthesis. Even-chain carboxylic acids are completely oxidized and produce succinyl-CoA, a gluconeogenic substrate, as an intermediate metabolite. Dicarboxylic acids are more polar than their esters, therefore they will diffuse less readily through normal cell membranes. 38
Malonic Acid
Malonic acid can be activated to malonyl-CoA and undergoes decarboxylation to acetyl-CoA by various mammalian tissues. 39
Adipic Acid Nonhuman
Adipic acid metabolism was studied using fasted male albino rats. 40 In 1 study, in which the rats were given a single oral dose, by gavage, with 50 mg radioactive adipic acid (labeled on C1 or C2), 70% of the radioactivity was exhaled as carbon dioxide in 24 hours. Adipic acid and the metabolites urea, glutamic acid, lactic acid, β-ketoadipic acid, and citric acid were recovered in the urine. Very little radioactivity was found in the tissues. Fasted male rats were also given a single dose of a solution containing 50 mg radioactive adipic acid (labeled on C1), by gavage, in conjunction with the intraperitoneal (ip) injection of 2 mL of 0.5 mol/L sodium malonate. After 24 hours, both radioactive adipic acid and succinic acid were found in the urine, which the researchers stated was an indication that adipic acid underwent β oxidation. In a study in which the rats were fed 25 mg radioactive adipic acid (labeled on C1) and 100 mg γ-phenyl-α-aminobutyric acid, followed by a 48-hour urine collection, it was determined that acetate is a metabolite of adipic acid. Finally, rats were given radioactive sodium bicarbonate with nonradioactive adipic acid. Radioactive citric acid was formed, which suggested that carbon dioxide interacted with a metabolite of adipic acid. (Details not specified.)
Two rats were dosed orally by gavage with 2.43 g/kg partially neutralized adipic acid for 28 days. In the urine, 67% of the dose was recovered unchanged. There was no change in excretion pattern over time during the study.
Rabbits were dosed orally by gavage (n = 4) or by intravenous (iv) administration (n = 2) with 2.43 g/kg partially neutralized adipic acid for 2 days. Following oral administration, 53% to 61% of the dose was recovered unchanged in the urine. With iv administration, 59% to 71% was recovered unchanged in the urine. In another study using rabbits, animals were given a subcutaneous (sc) dose of 2000 mg adipic acid; 3 rabbits were given a single dose, 1 was dosed on days 1 and 5, and 1 was dosed on days 1, 5, 9, 13, and 15. On average, 61% of the dose was recovered unchanged in the urine. There was an increase in urinary oxalic acid concentrations.
A female dog was fed either 150 mg/kg body weight (bw) adipic acid (in 2 feedings) for 5 days or 750 mg/kg bw (in 2 feedings) for 7 days. In the urine, 18% and 63.6% of the low and high doses, respectively, were recovered unchanged.
Rabbits (number not stated) were given up to 4 sc injections of ≤2000 mg sodium adipate. 41 An average of 61% of the dose was recovered unchanged in the urine. Oxalic acid was increased in the urine.
Human
In a study in which 1 participant was given 33 mg/kg bw sodium adipate, orally, for 5 days (10 g total), 6.76% of the dose was recovered in the urine. In another study in which 1 person was given 100 mg/kg bw adipic acid for 10 days (70 g total), 61% of the dose was recovered in the urine. Administration of 19.0 g adipic acid over 5 days or 23.4 g over 6 or 9 days (1 participant per dose) resulted in 53% of the administered dose recovered in the urine.
C9 to C12 Dicarboxylic Acids Nonhuman
Groups of 30 male Wistar rats were dosed orally, by gavage, with azelaic (C9), sebacic (C10), undecanedioic (C11), or dodecanedioic (C12) acid. 42 Ten rats in each group were dosed with 20, 50, or 100 mg of the respective acid. Blood, urine, and feces from the treated rats were analyzed and compared to the blank control obtained from untreated rats. (None of the C9-C12 acids were found in the blank controls.) In urine, approximately 2.5% of azelaic, 2.1% of sebacic, 1.8% of undecanedioic, and 1.6% of dodecanedioic acid was recovered after 5 days; the amount recovered was not affected by dosage. The dicarboxylic acids were not excreted in conjugated form. None of the C9-C12 dicarboxylic acids were recovered in the feces. In the plasma, dicarboxylic acid catabolites that were 2-, 4-, or 6-carbons shorter than the corresponding dicarboxylic acid were detected.
Human
Groups of 3 male and 2 female participants were also dosed with azelaic, sebacic, undecanedioic, or dodecanedioic acid orally, in gelatin capsules, once a week for 5 weeks. 42 The dose administered increased each week, from 0.5 g at week 1 to 5.0 g at week 5. None of the C9-C12 acids were found in the blank control samples of blood, urine, and feces obtained from nontreated humans. In urine, approximately 60% of azelaic, 17% of sebacic, 5% of undecanedioic, and 0.1% of dodecanedioic acid were recovered after 12 hours; the amount recovered was not affected by dosage. At 24 hours, the amounts recovered were not much increased. Initially, undecanedioic and dodecanedioic acid administration raised the urinary pH to a value of 7.4 to 8.5; the pH returned to normal within 3 to 6 hours. The dicarboxylic acids were not excreted in conjugated form. None of the C9-C12 dicarboxylic acids were recovered in the feces. In the plasma, dicarboxylic acid catabolites that were 2, 4, or 6 carbons shorter than the corresponding dicarboxylic acid were detected. Plasma levels of azelaic acid peaked at 2 hours, while the levels of the other 3 acids peaked at 3 hours. Recovery in the plasma was greatest for azelaic acid, 74.6 µg/mL with the 5 g dose, and the amount detected decreased with increasing chain length.
Azelaic Acid
Azelaic acid is a dietary constituent found in whole grain cereals and animal products. 43 It can be formed endogenously from longer chain dicarboxylic acids, metabolism of oleic acid, and Ψ-oxidation of monocarboxylic acids. 44 Endogenous plasma concentration and daily urinary excretion of azelaic acid are highly dependent on dietary intake. Azelaic acid crosses the blood–brain barrier. 45
A group of 25 male Wistar rats were dosed orally, by gavage, with 100 µCi of [1,9- 14 C]azelaic acid, and the animals were killed at various intervals 1 to 96 hours after dosing. 42 After 12 and 48 hours, 13% and 14.5% of the radioactivity was found in expired carbon dioxide, respectively. Approximately 40% of the radioactivity was recovered in the urine over 5 days. The C7 and C5 dicarboxylic acid metabolites were found in the urine up to 72 hours after dosing. Very little was recovered in the feces. Labeled dicarboxylic acids were present in the blood for up to 72 hours and consisted mainly of dicarboxylic acid metabolites. Radioactivity was found in all tissues, with the highest levels present in the liver, lungs, and kidneys after 12 hours. Tissue radioactivity levels then decreased slowly in all organs except adipose tissue, in which case increasing levels were still seen at 96 hours. Approximately 90% of the radioactivity found in the tissues was present in the lipids, and it was essentially localized in the fatty acid portion of the triglycerides and of the phospholipids. Traces of C9, C5, and C7 dicarboxylic acids were detected in the first 24 hours.
Sebacic Acid
Sebacic acid is oxidized to water and carbon dioxide, passing through acetyl-CoA and succinyl-CoA formation. 46
Disodium Sebacate Nonhuman
Disodium sebacate, 80 and 160 mg with 25 µCi of (1,10) [ 14 C]sebacic acid tracer, was administered by a single iv injection to 14 male Wistar rats, and blood samples were obtained at various intervals 5 to 320 minutes after dosing. 46 The plasma half-life of radioactive disodium sebacate was 37.86 and 39.82 minutes for the 80 and 160 mg dose groups, respectively. The apparent volume of distribution was 2.65 mL/100 g body wt.
In a second experiment, a group of 4 male Wistar rats were given a single-dose 160 mg disodium sebacate with 25 µCi sebacic acid tracer by iv injection, and expired carbon dioxide, urine, and feces were collected. The carbon dioxide half-life for radioactive sebacate was 93.64 minutes; 25% of the administered dose was expired in carbon dioxide. A total of 34.6% of sebacate was recovered in the urine in 24 hours, while 5.08% suberic acid (C8) was recovered in the same time frame. Most of the excretion occurred in the first 4 hours. Radioactivity was not found in the feces.
In the third experiment, groups of 10 male Wistar rats were also given 160 mg disodium sebacate with 25 µCi sebacic acid tracer by iv injection, and the animals were sacrificed at various intervals from 30 to 360 minutes after dosing. The amount of radioactivity in various organs was analyzed. No appreciable radioactivity was found in the body. Sebacate appeared to be in an absorption phase in fat 1 hour after dosing, but no radioactivity was found in the body after 24 hours.
The pharmacokinetics of disodium sebacate was studied in male and female Wistar rats. 47 Sebacate was administered either ip, 6 doses of 10 to 320 mg, or orally, 2 doses of 80 or 60 mg. Plasma concentrations of sebacate and urinary concentrations of sebacate and its products of β-oxidation (suberic and adipic acids) were measured using GLC/mass spectrometry. Both renal and nonrenal elimination parameters were obtained. The sebacate half-life was 31.5 minutes. The tissue elimination rate was 0.0122/min, and the overall volume of distribution was 26.817 mL/100 g. The renal clearance was 0.291 mL/min per 100 g, which was much less than the value of the glomerular filtration rate (GFR) of approximately 1 mL/min/100 g reported elsewhere, suggesting the presence of sebacate reabsorption from the ultrafiltrate. Sebacate renal clearance was found to be a concentration-independent function, suggesting the presence of a passive back diffusion. The relative bioavailability of the oral route compared to the ip route was 69.09%, showing an extensive absorption of the compound.
Human
The metabolism and excretion of disodium sebacate was studied in 7 fasting male participants that were given a continuous steady infusion of 20 g unlabeled disodium sebacate over 480 minutes. 48 At 240 minutes into the infusion, (1,10)[C 14 ]sebacic acid was infused simultaneously as a tracer (sp act 0.416 µCi/min). The was a gradual increase in the amount of radioactivity expired in carbon dioxide for the first 300 minutes; the value remained elevated for an additional 120 minutes before declining. At 24 hours, 11.38 mmol sebacate was recovered in the urine, as well as 2.04 mmol suberic acid and 1.11 mmol adipic acid, which was less than 15% of the dose administered. The serum concentration of unlabeled sebacate reached a plateau after 270 minutes of infusion. In all, 10% to 15% of serum radioactivity was found in the aqueous fraction of serum extracts. The renal clearance rate was 5.67 mL/min. The overall tissue uptake of unlabeled sebacate was 180 µmol/min, and the apparent distribution volume was 12.46 L. The percentage oxidation of sebacate was 6.14%.
The pharmacokinetic profile of disodium sebacate during a short-time infusion (5 hours at 10 g/h) was also studied in 7 male participants. 49 Sebacate in serum and urine was measured by HPLC. The apparent volume of distribution of sebacate was 8.39 L, and the plasma fractional removal rate constant was 0.0086/min.
Six male participants were given a single iv bolus of 1 g disodium sebacate, while another 6 received 10 g of sebacate in 500 mL of distilled water, iv, at a rate of 3.33 g/h over 3 hours. 50 For the group given a bolus dose, the distribution phase had a short half-life, 0.34 hours, and a rapid elimination, 2.045/h. For the group given the 3 hours infusion, 12% of the dose was excreted as sebacic acid in 24 hours; suberic acid (C8) and adipic acid were also present in the urine.
Dodecanedioic Acid
A group of 25 male Wistar rats were dosed orally, by gavage, with 100 µCi of [10,11- 3 H]dodecanedioic acid, and the animals were killed at various intervals 1 to 96 hours after dosing. 42 Approximately 50% of the radioactivity was recovered in the urine over 5 days. The C10, C8, and C6 dicarboxylic acid metabolites were found up to 72 hours after dosing. Only 2% of the radioactivity was recovered in the feces. Labeled dicarboxylic acids were present in the blood for up to 72 hours and consisted mainly of dicarboxylic acid metabolites. Radioactivity was found in all tissues, with the highest levels present in the liver, lungs, and kidneys after 24 hours. Tissue radioactivity levels then decreased slowly in all organs except adipose tissue, in which case an increase in radioactivity was still seen at 96 hours. Radioactivity levels were 20% to 40% lower in the lipid extracts of the tissues than in the residual matter. 3 H was distributed in the whole molecule, not only the fatty acid portion, of the phospholipid and triglyceride fractions. Traces of C12, C10, C8, and C6 dicarboxylic acids were detected in the first 24 hours.
Male Wistar rats were given an iv bolus of 800 µmol/kg disodium dodecanedioic acid. 51 The apparent volume of distribution was 0.248 L/kg, and the plasma half-life was 12.47 minutes. The renal clearance was 0.00051 L/kg/min, while systemic clearance was 0.0138 L/kg/min. Only 3% to 5% of the dose was recovered in the urine.
Percutaneous Absorption
Azelaic acid
The in vitro percutaneous absorption of a 15% azelaic acid gel through human skin, prior to or after the application of 3 different moisturizer formulations, was determined. 52 All doses were applied as 5 µL/cm2. The second dose was applied 15 minutes after the first. [ 14 C]Azelaic acid had a finite dose absorption profile, with a rise to peak penetration followed by a slow but steady decline. In vitro, 70% of the azelaic acid diffused into the reservoir solution over 48 hours. The application of a moisturizer, and whether it was applied prior to or following azelaic acid administration, did not have a statistically significant effect on the penetration of azelaic acid. However, there was a trend toward greater percutaneous penetration and mass distribution with the application of a moisturizer lotion prior to the azelaic acid gel.
The percutaneous absorption of azelaic acid was determined using 6 male participants. A total of 5 g of a cream containing 20% azelaic acid was applied to the face (1 g), chest (2 g), and upper back (2 g) of each participant, giving an area dose of approximate 5 mg cream/cm2 skin. The test areas were covered 1 hour after dosing with cotton tissues and washed 24 hours after dosing. After 1 week, 100 mL of an aqueous microcrystalline suspension containing 1 g azelaic acid was given orally to each participant. Urinary excretion of unchanged azelaic acid was measured after each dose. Following dermal application, 1.29% of the dose was recovered unchanged in the urine in 24 hours, and a total of 2.2% was recovered by day 3. Following oral administration, 61.2% of the dose was recovered within 4 hours; excretion was complete at this point. Assuming similar rates and pathways in biotransformation following both routes of exposure, percutaneous absorption of azelaic acid was determined to be 3.6% of the dermally applied dose. 53
Toxicological Studies
Single Dose (Acute) Toxicity
The acute oral, dermal, inhalation, and parenteral toxicity of the dicarboxylic acids and some of the salts are summarized in Table 4. 40,54 –59 The oral LD50 values of the dicarboxylic acids had a wide range, for example, adipic acid had values for rats ranging from 940 mg/kg to greater than the highest dose tested (11 000 mg/kg). Most reported values for the acids were >200 mg/kg. The reported dermal LD50 values ranged from >6000 mg/kg dodecanedioic acid to >10 000 mg/kg glutaric acid.
Repeat Dose Toxicity
Cellular effects
Dicarboxylic acids have a cytotoxic effect on the abnormally hyperactive and malignant epidermal melanocytes. Dicarboxylic acids, C8 to C13, have been shown to inhibit mitochondrial oxidoreductases, 62 and they have been shown to reversibly inhibit microsomal NADPH and cytochrome P450 reductase. 63 Medium chain length dicarboxylic acids are also competitive inhibitors of tyrosinase in vitro.
Adipic acid
The effect of adipic acid on primary keratinocyte cultures was evaluated using epidermal cells from neonatal NMRI mice. 64 Concentrations of ≤30 mmol/L did not inhibit 3 H-thymidine incorporation or affect DNA synthesis, while 40 and 50 mmol/L inhibited both of these parameters. No effect on labeling indices was observed with 1 to 30 mmol/L adipic acid.
Azelaic acid
Azelaic acid, a naturally occurring competitive inhibitor of tyrosinase, has a cytotoxic effect on malignant melanocytes. 65 Azelaic acid is also a competitive inhibitor of a number of oxidoreductive enzymes, enzymes involved in DNA synthesis, and of oxidoreductases of the respiratory chain. 66 It has been reported that, in vitro, azelaic acid has time- and dose-dependent, reversible, and antiproliferative and cytotoxic effects on a number of tumoral cell lines. Azelaic acid had no effect on normal cell lines.
Disodium azelate
Disodium azelate inhibited cell proliferation and affected viability of Cloudman and Harding-Passey murine melanomata at concentrations ≥10−2 mol/L when incubated over a 3-day period. 62 The mitochondria were the prime target of action.
The effect of disodium azelate on primary keratinocyte cultures was evaluated using epidermal cells from neonatal NMRI mice. 64 A dose-dependent inhibition of 3 H-thymidine incorporation into DNA, ranging from 50% inhibition with 20 mmol/L to 90% inhibition with 50 mmol/L disodium azelate, was observed following a 12-hour incubation period. Concentrations of 1 and 10 mmol/L did not affect DNA synthesis, but a marked reduction was seen with 20 to 50 mmol/L. The effects on DNA synthesis were time dependent, with the maximum inhibitory effect observed at 4 hours; this effect was reversible. RNA and protein synthesis were also inhibited during the first 4 hours of incubation with 50 mmol/L disodium azelate. Cellular structure was altered upon incubation with disodium azelate, primarily affecting mitochondria and the rough endoplasmic reticulum. These effects were also reversible.
Dodecanedioic acid
The disodium salt of dodecanedioic acid inhibited cell proliferation and affected viability of Cloudman and Harding-Passey murine melanomata at concentrations ≥10−2 mol/L, when incubated over a 3-day period. 62 The mitochondria were the prime target of action.
Animal studies
Most of the available animal studies were oral exposures, although limited inhalation studies were available for adipic acid.
Adipic acid Oral
Groups of 6 male Sprague-Dawley rats were dosed orally (method not specified) with 3600 to 5600 mg/kg bw adipic acid as an 18.6% to 24.9% solution in saline for 14 days. 40 Three animals of the 3600 mg/kg bw group, 5 of the 4000 mg/kg bw group, and all of the 4500 to 5600 mg/kg bw groups died prior to study termination. Signs of toxicity included depressed activity, labored respiration, ataxia, and convulsions. No gross findings were noted at necropsy at study termination.
Groups of 5 rats were dosed with 0 or 3000 mg/kg bw of a neutralized 20% adipic acid solution orally, by gavage, for 4 weeks. 49 A nonsignificant decrease in bw gain was observed. In a 4-week study in which a group of 3 rats was dosed orally, by gavage, with 2400 mg/kg bw adipic acid, no significant toxicological effects were noted.
In a 4-week dietary study in which groups of 17 to 20 female rats were fed 0 to 40 mg/d (0-435 mg/kg bw per d) adipic acid, no effects were reported. 49 The no-observable adverse effect level (NOAEL) was >435 mg/kg bw per d. In a 5-week dietary study in which groups of 15 to 18 male rats were fed 0 to 800 mg/d (0-13 333 mg/kg bw/d) decreased bw gains, an unkempt appearance, and diarrhea were observed for the animals fed 800 mg/d the first 3 weeks. In another 5-week dietary study in which groups of 4 rats, gender not specified, were fed 100 or 200 mg/d (310-922 mg/kg bw/d) of a 20% adipic acid solution in ethanol, 5 days/week, no signs of toxicity were observed.
Ten rats were dosed orally, method not specified, with 199 mg/d (638-1332 mg/kg bw/d) sodium adipate, 5 days/week for 9 weeks. 49 No toxicological effects were observed.
A group of 5 guinea pigs, gender not specified, were dosed orally using capsules with 400 mg/d (682-942 mg/kg bw/d) adipic acid for 5 days, followed by dosing with 600 mg/d (1032-1739 mg/kg bw/d), 5 days/week for 5 weeks. 49 No signs of toxicity were observed.
No toxicity was observed in a study in which pigs were fed 1% adipic acid in the diet for 7 days. 49
Groups of 8 to 10 male rats were given 0, 420, 840, 1700, or 3400 mg/kg bw/d sodium adipate for 19 weeks in a protein deficient diet. 57 Animals were killed after either 7 weeks or at study termination. For unexplained reasons, only 5 to 7 animals/group survived until study termination. Rats of the 3400 mg/kg bw/d group had decreased bw gains and decreased bws. (Statistical significance not stated.) Slight effects were seen in the liver, and the NOAEL was 3333 mg/kg bw.
Groups of 13 to 15 male and female rats were fed a diet containing 0, 1600, or 3200 mg/kg bw/d adipic acid for 33 weeks. 40 Rats were killed at various intervals throughout the study. Ten of 14 rats fed 3200 mg/kg bw/d died during weeks 0 to 4; surviving rats had decreased weight gains during this time. However, at study termination, bws were for surviving animals of this group were similar to controls. Slight effects were seen in the liver. (Statistical significance not stated.)
In a 2-year study, groups of 20 male rats were fed a diet containing 0%, 0.1%, 1%, 3%, and 5% adipic acid (equiv. to 0, 75, 750, 2250, and 3750 mg/kg bw/d), and groups of 10 and 19 females were fed 0% and 1% adipic acid, respectively. 49 Weight gains of male rats fed 3% and 5% adipic acid were significantly less than controls. There were no significant toxicological findings upon gross or microscopic observation. The NOAEL was 1% adipic acid for male and female rats.
The effect of adipic acid on hepatic peroxisome proliferation was evaluated in an in vivo study in which 4 male F344 rats were fed chow containing 2% adipic acid dissolved in alcohol. 67 After 3 weeks of dosing, the animals were killed. Adipic acid did not induce peroxisome proliferation and did not affect relative liver to bws.
Inhalation
Mice were exposed to 460 mg/m3 adipic acid dust for 1.5 mos, or to 13 or 129 mg/m3 adipic acid for 4 mos (details not given). 40 Decreased weight gain, altered oxidase activity, and upper respiratory tract, liver, kidney, and central nervous system effects were observed.
Two male and 2 female rats were exposed to 126 mg/m3 adipic acid dust for 15 days, 6 h/d. 49 No signs of toxicity were observed, and no gross or microscopic findings were noted at necropsy.
Sodium succinate
The oral toxicity of sodium succinate was evaluated using F344 rats. 58 Groups of 10 males and 10 females were given 0, 0.3, 0.6, 1.25, 2.5, 5 or 10% sodium succinate in the drinking water for 13 weeks. All animals were killed at the termination of dosing. Body weight gains of animals of the 10% group were significantly decreased, and all animals of this group died by week 4. These animals were extremely emaciated; however, no compound-related microscopic lesions were found. Body weight gains were decreased in animals given ≥2.5% sodium succinate, as compared to controls. No toxicological treatment-related effects were observed.
Glutaric acid
Groups of 15 male and 15 female Sprague Dawley rats were fed a diet containing 0% to 2% glutaric acid for 90 days. 55 Body weight gains were decreased for males and statistically significantly decreased for females of the 2% group. No differences were noted between test and control animals in hematology, clinical chemistry, or urinalysis. There were no microscopic findings or organ weight changes attributable to the test substance. There was no treatment-related mortality. The NOAEL was ≥1%, and the LOAEL was 2% glutaric acid.
Four male and 4 female Beagle dogs were fed a diet containing 0% to 5% glutaric acid for 90 days. 68 Decreased bws, accompanied by reduced feed consumption, were observed for the males and females of the 5% group and females of the 3% group. No other treatment-related effects were observed. The NOAEL was ≥2% and the LOAEL was 3%.
Adipic/glutaric/succinic acid mixture
Groups of 15 male and 15 female rats were dosed orally, by gavage, for 90 days with 0% to 30% of a mixture that contained 4% adipic, 16% glutaric, and 5% succinic acid. 55 The vehicle was deionized water, and the dosing volume was 10 mL/kg. Two males and 1 female of the 30% group died, and the deaths were considered dose-related. Also in this group, bws were reduced for males and females, and feed consumption was statistically significantly reduced in males. An increased incidence of labored breathing and rales was noted. The urine pH was statistically significantly reduced in both males and females dosed with 30% of the mixture. In the 10% group, bw gains were slightly, but not statistically significantly, reduced in females and feed consumption was statistically significantly reduced in males. The NOAEL was 3% and the LOAEL was 10%.
Azelaic acid
Groups of 15 male and 15 female Wistar rats were fed a diet containing 140 or 280 mg/kg bw azelaic acid for 180 days, and a control group of 10 males and 10 females was given untreated feed. 59 No significant toxicological effects were observed. Growth was similar between test and control groups, as were the microscopic examinations and clinical chemistry parameters. The researchers found similar, negative, results when groups of 10 male and 10 female New Zealand rabbits were fed diets containing 0, 200, or 400 mg/kg bw azelaic acid for 180 days.
Disodium sebacate
Groups of 10 male and 10 female Wistar rats were fed a diet containing 0, 500, or 1000 mg/kg bw disodium sebacate for 6 mos, after which time they were killed and necropsied. 54 Growth was similar between test and control groups, as were the microscopic examinations and clinical chemistry parameters. The researchers found similar, negative, results when groups of 10 male and 10 female New Zealand rabbits were fed diets containing 0, 750, or 1000 mg/kg bw disodium sebacate for 6 mos.
Ocular Irritation
Ocular irritation studies are summarized in Table 5.
Ocular Irritation—Dicarboxylic Acids
Succinic acid
The ocular irritation potential of succinic acid was evaluated using albino rabbits. 55 Undiluted test material, 0.005 mL, was applied to the center of the cornea. The eyes were not rinsed. Succinic acid was a severe eye irritant, with necrosis visible upon staining. The score for ocular irritation, on a scale of 1 to 10, was 8.
Glutaric acid
A Draize ocular irritation study was performed in which 100 mg of glutaric acid was instilled in the eyes of 3 rabbits and the eyes were rinsed 24 hours after application. 55 Glutaric acid was irritating to rabbit eyes, with a primary irritation index (PII) of 35.2/110. Mild erythema, slight edema, and slight dullness were still present after 7 days.
Adipic acid
The ocular irritation of adipic acid was evaluated using groups of 2 albino rabbits. 55 Ten or 57.1 mg of adipic acid was placed in the eye of each rabbit, and the eye of 1 animal in each group was rinsed. With 10 mg followed by rinsing, mild conjunctival irritation was observed; and the eye was normal within 3 days. In the unrinsed eye, mild conjunctival irritation and a minimal iritic effect were observed; minimal conjunctival irritation was still observed after 7 days and the eye was normal after 14 days. With instillation of 57.1 mg adipic acid followed by rinsing, moderate to mild conjunctival irritation and transient mild opacity were observed; the eye was normal in 3 days. In the unrinsed eye, moderate to mild conjunctival irritation, mild opacity of the cornea, and a minimal iritic effect were observed; the eye was normal at day 7. However, other studies have reported that adipic acid produced severe irritation in rabbit eyes, and the signs of irritation were still present after 8 days. 40
Adipic/glutaric/succinic acid mixture
The ocular irritation potential of a mixture of adipic, glutaric, and succinic acid, percentages not specified, was evaluated using 2 male albino rabbits. 55 One-tenth milliliter of the test substance was instilled in the conjunctival sac of each animal, and the eye of 1 animal, but not the other, was rinsed. The contralateral eye served as the negative control. Mild to severe conjunctivitis was observed in on both the rinsed and unrinsed rabbit eyes. Both eyes were normal within 21 days.
Dodecanedioic acid
In studies using rabbits that evaluated the ocular irritation of dodecanedioic acid, slight irritation was reported in 1 study, with a PII of 11.96/110, and small areas of corneal opacity and mild conjunctival irritation were seen in the other study. 56 Details were not provided.
Dermal Irritation/Sensitization
Most of the available dermal irritation and sensitization data regarding alkyl dicarboxylic acids and their salts were from animal studies. These animal data are summarized in Table 6. Human data were available for adipic and azelaic acids only.
Dermal Irritation and Sensitization—Dicarboxylic Acids

Succinic acid
Succinic acid was a slight irritant to rabbit skin. 55 Details were not provided.
Glutaric acid
The dermal irritation potential of glutaric acid was determined using 2 male and 4 female New Zealand white rabbits. 53 A 0.5 g aliquot of glutaric acid was applied to the clipped skin on the back of the rabbits. The test site was scored for irritation after 3 minutes, and the site was then washed. The test material was then applied to 2 other test sites, which were covered with a rubber wrap. The sites were examined at 1 hour and 4 hours, and the site was washed after both examinations. The sites were then evaluated at 24 and 48 hours after application. Slight erythema was seen in 1 rabbit throughout the study. Irritation was not observed in the other rabbits.
Adipic Acid Nonhuman
A dermal irritation study was performed in which 500 mg of 50% aqueous adipic acid was applied under an occlusive patch to a 5 cm x 5 cm area of intact and abraded skin of 6 rabbits for 24 hours. 38 With intact skin, an erythema score of 2 to 3/4 was reported, with clearing by day 3. With abraded skin, mild to severe erythema and edema were reported, which cleared by day 7.
Adipic acid, undiluted or as an 80% aqueous paste, was applied occlusively to the backs of ears of rabbits for 24 hours. Two rabbits were used per group. No irritation was observed on the backs of animals. Erythema was observed on the ear, with clearing by 72 hours. In another study in which adipic acid was applied occlusively for 24 hours, irritation was not observed. Details were not provided.
A semi-occlusive application of 500 mg of a paste of 50% adipic acid in propylene glycol to 6 rabbits produced slight to mild irritation in 3 of the rabbits. A semi-occlusive application of undiluted adipic acid was not corrosive. Adipic acid, 50% in propylene glycol, was not irritating to a group of 10 guinea pigs.
The sensitization potential of adipic acid was evaluated using groups of 10 guinea pigs. For induction, 0.1 mL of 1% aqueous adipic acid was given as a sacral intradermal injection, once a week for 4 weeks. After a 2-week nontreatment period, the dermal challenge was performed with 0.05 mL of 50 and 25% adipic acid in propylene glycol. Adipic acid produced very mild or no irritation and it was not a sensitizer.
Human
In 2 case reports involving occupational exposure to adipic acid, positive sensitization reactions were reported with follow-up testing. 38
Adipic/glutaric/succinic acid mixture
A mixture of adipic, glutaric, and succinic acid (percentages not specified) was evaluated for irritation and for sensitization using groups of 10 male guinea pigs. 53 The primary irritation potential was evaluated by applying 0.05 mL of an 8 or 80% suspension in dimethyl phthalate to the shaved, intact skin on the shoulder of the animals. The sensitization potential was also evaluated, using 4 sacral intradermal injections of 0.1 mL of a 1% suspension for induction. After a 13-day nontreatment period, a dermal challenge was performed with 0.05 mL of an 8% and 80% suspension of the mixture. Ten previously untreated guinea pigs were exposed to the same challenge applications as the test animals. In the test for primary irritation, the 8% suspension produced no irritation, and no to mild irritation was observed 24 hours after exposure to the 80% suspension. No sensitization was observed at either dose.
Azelaic acid
The cumulative irritation potential of a 15% azelaic acid gel (prescription formulation; vehicle not identified) was determined in a study using 31 female and 2 male participants. 64 (During the study, 1 participant withdrew for personal reasons.) White petrolatum was used as a negative control. Azelaic acid and petrolatum, 0.2 g of each, were applied under occlusion to 2 cm x 2 cm sites on the back of each participant 3 times per week for 3 weeks. Weekday patches were removed after 24 hours, while the patches applied on Fridays were removed after 72 hours. The test sites were evaluated 15 to 30 minutes after removal of the patch, and then a new patch was applied. Application was discontinued if severe irritation, which was designated by a maximum erythema score of 3, was observed. A 15% azelaic acid gel was statistically significantly more irritating than the negative control, with a mean cumulative irritancy index of 1.05/3. Individual reaction scores for the test article ranged from 0 to 3, and 5 participants discontinued patching with azelaic acid due to an irritation score ≥3. Cumulative irritancy increased with successive patching. The researchers noted that since the vehicle used for azelaic acid was not tested, there was uncertainty as to whether the vehicle components affected the irritation scores.
Twice daily application of a cream containing 20% azelaic acid has been reported to cause erythema, irritation, pruritus, dryness, scaling, and burning. 65
Dodecanedioic acid
Dodecanedioic acid was not an irritant to rabbit skin in a 4-hour exposure study or upon application of 0.5 g. 54 In a maximization study using female guinea pigs, 0.5% dodecanedioic acid was injected intracutaneously at induction and 25 and 50% was used for the dermal challenge. Dodecanedioic acid was not a sensitizer.
Mucosal Irritation
Succinic acid
Succinic acid has been considered to be an exacerbating factor in ulcerative colitis, therefore its influence on rat colonic mucosa in terms of mucosal blood flow and superoxide generation was investigated. 66 The left side of the colon of 5 male and 5 female rats was exposed, and 0.9% to 5% succinic acid in physiological saline was instilled into the colonic lumen. A segment of the colon was then ligated as to not include the mesenteric blood vessel. Mucosal blood flow decreased with all dose levels. Microscopically, the higher the concentration of succinic acid, the greater was the erosion formation in the colonic mucosa. Significant polymorphonuclear cell infiltration superoxide generation from colon tissue was observed with 0.01% succinic acid, as compared to higher or lower concentrations. Succinic acid, at fecal concentrations found in active stage ulcerative colitis, appears to be implicated in mucosal injury, mediated by a decrease in colonic mucosal blood flow and infiltration of superoxide-generating polymorphonuclear cells into the mucosa.
Reproductive and Developmental Toxicity
Malonic Acid
Malonic acid, 0.1%, reduced the pH of sperm suspensions from 7.5 to 4.5 to 5.5 and it rendered human spermatozoa immotile within 30 minutes. 67 A concentration of 1.0% reduced the pH to 1.5 to 3.0 and was almost instantaneously spermicidal.
Succinic Acid
Thirty ovariectomized female rats were given daily sc injections of 5.0 mg/d succinic acid for 3 weeks. 53 Ten females were used as controls. Daily vaginal smears were similar for test and control animals. Microscopically, no changes were seen in the uterine horn, cervix, or vagina of the animals.
Glutaric Acid
The reproductive toxicity of glutaric acid was evaluated using groups of 25 female rats. 53 The animals were dosed orally, by gavage, with 0, 125, 400, or 1300 mg/kg glutaric acid on days 6 to 15 of gestation, and the animals were killed on day 20 of gestation. No toxicological or reproductive effects were observed for the 125 mg/kg group. In the 400 mg/kg group, salivation, rales, and nasal discharge were observed. One dam of the 1300 mg/kg group died on day 10 of gestation, and 1 was killed due to moribund condition on day 13 of gestation. Mean bw gains were decreased in the 1300 mg/kg group during dosing, but bw gains in this group were normal post-dosing. Clinical signs of toxicity in the 1300 mg/kg group included salivation, rales, nasal discharge, and staining around the mouth, nares, and anogenital area. No adverse effects on pregnancy and no teratogenic effects were reported at any of the dose levels. There was a significant increase in resorptions in the 1300 mg/kg group compared to controls, but the value was within normal expected limits and, therefore, not considered biologically meaningful.
Groups of 18 gravid female New Zealand white rabbits were dosed orally, by gavage, on days 6 to 18 of gestation with 0, 50, 160, or 500 mg/kg glutaric acid, and the animals were killed on day 29 of gestation. 53 No test-article related mortality occurred. There were no clinical signs of toxicity, and bws were not affected. No embryotoxic, teratogenic, or adverse reproductive effects were reported.
Adipic Acid
Groups of 20 to 24 gravid albino CD-1 mice were dosed orally, by gavage, with 0, 2.6, 12, 56, or 263 mg/kg bw adipic acid on days 6 to 15 of gestation. 38 All animals were killed on day 17 of gestation. No reproductive, developmental, or maternal effects were observed, and the NOAEL for maternal and developmental toxicity was 263 mg/kg bw. Similar results were obtained in a study in which gravid Wistar rats were dosed orally, by gavage, with 0, 2.9, 13, 62, or 288 mg/kg bw adipic acid on days 6 to 15 of gestation. The NOAEL for maternal and developmental toxicity was 288 mg/kg bw. 38
Groups of 21 to 24 gravid hamsters were dosed orally, by gavage, with 0, 2.9, 5, 44, or 205 mg/kg bw adipic acid on days 6 to 10 of gestation. A significant increase in resorption per implant site was observed with 205 mg/kg bw adipic acid, resulting in a decreased number of live fetuses. (This decrease was not evaluated statistically.) No other effects were reported. 38
Groups of 10 to 14 gravid Dutch-belted rabbits were dosed by oral intubation with 0, 2.5, 12, 54, or 250 mg/kg bw adipic acid on days 6 to 18 of gestation. No reproductive, developmental, or maternal effects were observed. The NOAEL for maternal toxicity was ≥250 mg/kg bw and for developmental toxicity was 250 mg/kg bw. 38
Azelaic Acid
Reproductive and teratogenic effects of azelaic acid were evaluated using Wistar rats and New Zealand rabbits. 57 A group of 20 gravid rats was fed a diet containing 140 mg/kg bw/d azelaic acid, and a control group of 10 gravid rats was given untreated feed. Half of each group was killed and necropsied on day 19 of gestation, and the remaining animals continued dosing for 3 mos. The day of gestation that dosing started is not clear. No gross or microscopic lesions were observed for the uteri, placentas, or ovaries. There were no differences in reproductive, teratogenic, or developmental effects between treated and control groups, nor were there any differences in fetal weights of the live fetuses. Similar results were seen using groups of 20 gravid rabbits fed 200 mg/kg bw/d azelaic acid; 10 untreated gravid rabbits were used as a negative control group.
Embryotoxic effects were observed in oral studies with rats receiving 2500 mg/kg bw/d of azelaic acid. 42 Similar effects were observed in studies in rabbits given 150 to 500 mg/kg bw/d and in monkeys given 500 mg/kg bw/d. The doses at which these effects were noted were all within toxic dose ranges for the dams. No teratogenic effects were observed. (Details were not provided.)
Disodium Sebacate
Reproductive, teratogenic, and developmental effects of disodium sebacate were evaluated using Wistar rats and New Zealand rabbits. 57 Groups of 20 gravid rats were fed a diet containing 0 or 500 mg/kg bw/d disodium sebacate, and groups of 20 gravid rabbits were fed 0 or 1000 mg/kg bw. Half of each group was killed and necropsied on day 19 of gestation, and the remaining animals continued dosing for 3 mos. The day of gestation that dosing started is not clear. No gross or microscopic lesions were observed for the uteri, placentas, or ovaries. There were no differences in reproductive or developmental effects between treated and control groups, nor were there any differences in fetal weights of the live fetuses.
Dodecanedioic Acid
The reproductive toxicity of 0 to 1000 mg/kg bw dodecanedioic acid was evaluated in an OECD combined repeated doe and reproductive/developmental toxicity screening test using male and female Crl:CD:BR rats. 54 The no-observable effect level (NOEL) for reproductive and developmental toxicity was 1000 mg/kg bw.
Sodium Salt of Adipic, Azelaic, Sebacic, and Dodecanedioic Acids
The influence of the sodium salt of some dicarboxylic acids (adipic acid, azelaic acid, sebacic acid, dodecanedioic acid) on both spontaneous and evoked muscle activity of the uterine horns of 35 female Wistar rats (250-300 g) has been studied in vitro. 68 Spontaneous activity of uterine muscle was inhibited by dicarboxylic salts causing the total abolition of mechanical events at concentrations of 24, 32, 40, and 64 × 10−3mol/L. Dicarboxylic salts antagonized the maximal isometric contraction of the uterine horn induced by administration of acetylcholine, oxytocin or prostaglandins (PGF2-α). The amount of antagonism was dependent upon the concentration of dicarboxylic salt used. Dicarboxylic salts had an specific inhibitory effect on the uterine horn which progressively increased with their chain length. The results suggested that the inhibitory effects of dicarboxylic salts on smooth muscle could be due to a cellular membrane hyperpolarization.
Genotoxicity
Available genotoxicity studies are summarized in Table 7.
Genotoxicity Studies—Dicarboxylic Acids
In Vitro
Malonic acid
Malonic acid, 3333 µg/plate, was not mutagenic in a National Toxicology Program (NTP) preincubation assay, with or without metabolic activation. 70
Succinic acid
The genotoxic potential of succinic acid was evaluated in an Ames test and in a chromosomal aberration study using a Chinese hamster fibroblast cell line. 71 Succinic acid, at a concentration of ≤5.0 mg/plate in phosphate buffer, was not mutagenic in the Ames test. (Whether metabolic activation was used is not stated.) Concentrations of ≤1.0 mg/mL in saline were not genotoxic in the chromosomal aberration assay.
Disodium succinate
The genotoxic potential of disodium succinate was evaluated in an Ames test and in a chromosomal aberration study using a Chinese hamster fibroblast cell line. 71 In the Ames test, disodium succinate was not mutagenic at concentration up to 5.0 mg/plate in phosphate buffer. (Whether metabolic activation was used is not stated.) Equivocal genotoxic results were obtained in the chromosome aberration assay of ≤15.0 mg/mL disodium succinate in saline using Chinese hamster fibroblast cells.
Disodium succinate, ≤10 mg/plate, was negative in another Ames test, with and without metabolic activation. 77
Glutaric acid
Glutaric acid was evaluated in vitro in a standard Ames assay, the L5178Y/TK ± mouse lymphoma assay with and without metabolic activation, and the mammalian in vitro Balb/c-3T3 cell transformation assay with and without metabolic activation. 78 The Ames tests were negative. However, the cell transformation assay was positive both in the presence and absence of metabolic activation and the results in the mouse lymphoma assay were dependent upon pH of the culture medium. The researchers stated that the variable response in the mouse lymphoma assay and the positive effect in the cell transformation assay may have been an indirect effect of other factors (such as the pH or osmolarity of the media in which the cells were exposed), rather than a direct effect of glutaric acid.
Adipic acid
Adipic acid was evaluated in a number of Ames assays using Salmonella typhimurium and Escherichia coli; results were negative, with or without metabolic activation, at concentrations as high as 10 000 mg/plate. 40,74,75 Negative results were also obtained in an Ames test with 0 to 200 mg/L adipic acid using S typhimurium TA1530 and G-46 without metabolic activation. 40 Results were negative in a yeast gene mutation assay using Saccharomyces cerevisiae without metabolic activation at concentrations ≤200 mg/L. A mouse lymphoma assay using L5178Y/TK ± cells was negative with and without metabolic activation at concentrations of ≤2000 µg/plate, 75 as was a cytogenetic assay using human embryonic lung fibroblast cells with ≤200 mg/L adipic acid. 40 In a viral enhanced cell transformation assay using Syrian hamster embryo cells at doses of 62 to 1000 µg/mL adipic acid, results were negative.
Adipic/glutaric/succinic acid mixture
A mixture of adipic, glutaric, and succinic acid, percentages not specified, tested as a 50% aqueous solution, was not mutagenic in an Ames assay using S typhimurium, with or without metabolic activation, at concentrations of ≤300 µg/plate. 55 Negative results were also obtained in an unscheduled DNA synthesis assay at concentrations of ≤5000 µg/plate using rat hepatocytes and in an HGPRT assay at concentrations of ≤2500 µg/plate, without, and of ≤3500 µg/plate, with, metabolic activation. In an invitro transformation assay using Chinese hamster ovary (CHO) cells at concentrations of ≤1500 µg/mL without and ≤2500 µg/mL with metabolic activation, positive results were obtained with, but not without, metabolic activation at 2000 µg/plate.
Azelaic acid
Azelaic acid, 20%, was not mutagenic or genotoxic in an Ames assay, HGPRT test in CHO cells, or human lymphocyte test. 44 Details were not provided.
Dodecanedioic acid
Dodecanedioic acid was not mutagenic in an Ames assay at concentrations of ≤5000 µg/plate, with and without metabolic activation. 56 Toxicity occurred at ≥500 µg/plate.
In Vivo
Glutaric acid
Glutaric acid was evaluated in a mammalian micronucleus cytogenetic assay in mice. 78 Glutaric acid was not genotoxic in this assay. (Details not specified.)
Adipic acid
Adipic acid was not genotoxic in in vivo cytogenetic assays using chromosomes from rats dosed orally, by gavage, with a single dose of 5000 mg/kg bw or daily for 5 days with 2500 mg/kg bw. 40 Adipic acid was also not genotoxic in dominant lethal studies with doses up to 5000 mg/kg bw.
Adipic/glutaric/succinic acid mixture
A mixture of adipic, glutaric, and succinic acid, percentages to specified, was not genotoxic in vivo using male and female Sprague Dawley rats dosed orally by gavage with 2750 and 1375 mg/kg of the mixture, respectively. 55
Azelaic acid
Azelaic acid was not genotoxic in a dominant lethal assay in mice. 44 (Details not specified.)
Dodecanedioic acid
Dodecanedioic acid, ≤5000 mg/kg bw, was not mutagenic in a micronucleus assay using mice. 56
Carcinogenicity
Sodium Succinate
Groups of 50 male and 50 female F344 rats were given drinking water containing 0, 1, or 2% sodium succinate for 2 years, and the carcinogenic potential was determined. 58 Dosing was discontinued after 104 weeks, and, after a 9-week recovery period, the rats were killed. Body weights of the high-dose animals were decreased by 10% as compared to controls. There were no statistically significant differences in overall tumor incidence or mean survival time between treated and control animals. An increase in the incidence of C-cell adenoma/carcinoma of the thyroid in females of the 2% group, and a positive trend in the occurrence of this tumor, was considered a function of experimental variability and not related to dosing. Sodium succinate was not toxic or carcinogenic to male or female F344 rats when given in the drinking water for 2 years.
Adipic Acid
Adipic acid was not carcinogenic in the 2-year chronic oral toxicity study (described previously) in which groups of 20 male rats were fed diets containing 0%, 0.1%, 1%, 3%, and 5% adipic acid, and groups of 10 and 19 females were fed 0% and 1% adipic acid, respectively. 57
Tumor Promotion
Succinic acid, sodium succinate, disodium succinate
The promotion of urinary bladder carcinogenesis by sodium succinate was evaluated using male F344 rats. 79 Groups of 16 male F344 rats were given 5% succinic acid, sodium succinate, or disodium succinate with 0.05% N-butyl-N- (4-hydroxybutyl)nitrosamine (BBN) in the drinking water for 4 weeks, followed by dietary administration of 5% of the respective test article without BBN for 32 weeks. Negative controls were given water with BBN only and untreated feed. Groups of 8 male F344 rats followed the same protocol without the addition of BBN to the drinking water, as did a group of non-BBN-treated negative controls. The animals were killed at week 37.
In the BBN-pretreated groups, many rats given sodium or disodium succinate developed hematuria towards the end of the study. There were no statistically significant differences in body or organ weights between the control and test groups. (Information on organ and bws was not provided for the non-BBN groups.) Large tumors were found on the urinary bladders of the BBN-pretreated animals given sodium and disodium succinate; tiny lesions were found in the control or succinic acid BBN-pretreated animals. The incidence and number of urinary bladder carcinomas and papillomas and of papillary or nodular hyperplasia (preneoplastic lesions) were statistically significantly increased in the sodium and disodium succinate BBN-pretreated groups as compared to the succinic acid and control BBN-pretreated groups. The incidence and numbers observed in the sodium and disodium succinate groups were not statistically significantly different from each other. An association between tumor area and sodium intake was noted. Urinary bladder lesions were not observed in any of the animals that were not pretreated with BBN. Urinary pH and electrolyte concentrations were affected by dosing with sodium or disodium succinate with BBN, as compared to the control and succinic acid groups, and statistically significant differences between these 2 groups were observed as well.
The researchers also evaluated cell proliferation and DNA synthesis in the urinary bladder epithelium. Groups of 20 male F344 rats were given 5% succinic acid, sodium succinate, or disodium succinate in the feed, without BBN pretreatment for 8 weeks. Negative controls were given basal diet. Five rats per group were given an ip injection of 50 mg/kg bw 5-bromo-2’-deoxyuridine (BrdU) 1 hour prior to being killed. Compared to control values, BrdU uptake was statistically significantly increased by increased disodium succinate and was increased, but not in a statistically significant manner, by sodium succinate. Succinic acid did not have any effect on DNA synthesis. Microscopically, simple hyperplasia was observed in the urinary bladders of animals given sodium and disodium succinate. The appearance of the urinary bladder epithelial surface was altered by sodium and disodium succinate. Spermidine/spermine N 1-acetyltransferase activity in the urinary bladder epithelium was increased for disodium succinate, but not sodium succinate, when compared to controls. Urinary pH and electrolyte concentrations were affected as described previously.
Part II: Esters of alkyl Dicarboxylic Acids Chemistry
Method of Manufacture
Alkyl dicarboxylic acids are easily esterified with the appropriate alcohol, with our without acid or metal catalyst (Fischer esterification). 9 For example, diethylhexyl adipate can be manufactured from adipic acid and ethylhexanol with an acid catalyst (Figure 3).
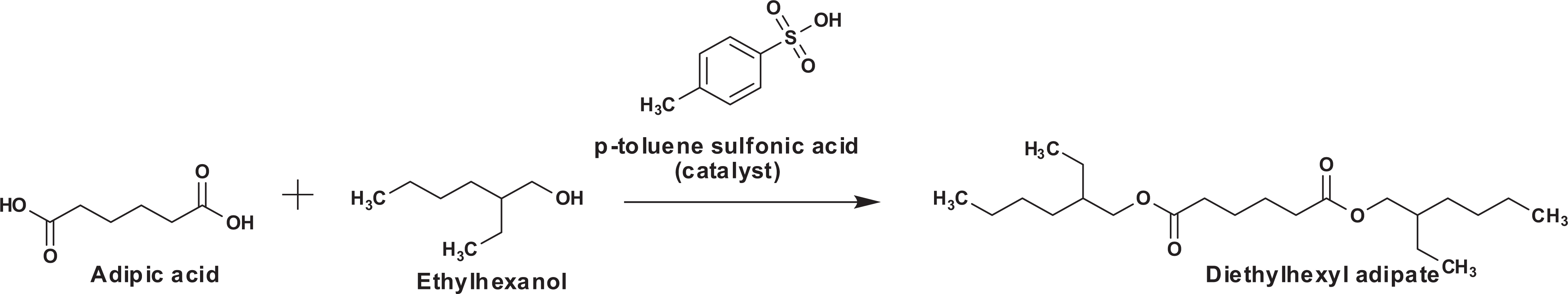
Diethylhexyl adipate synthesis from adipic acid.
Diethyl malonate
Malonic acid esters can be produced either by cobalt-catalyzed alkoxycarbonylation of chloroacetates with carbon monoxide in the presence of the appropriate alcohol, or by hydrolysis of cyanoacetic acid followed by esterification with the respective alcohol. 39 Diethyl malonate is prepared from chloroacetic acid and sodium cyanide followed by esterification with ethanol and sulfuric acid. 80
Diisopropyl adipate
Diisopropyl adipate is produced by esterification of adipic acid with an excess of isopropanol. The excess alcohol is removed by vacuum stripping and the ester is then alkali-refined and filtered. 2
Dibutyl adipate
Adipic acid is esterified with butyl alcohol by a continuous distillation process. 81
Diethylhexyl adipate
Diethylhexyl adipate can be prepared by the reaction of adipic acid and 2-ethylhexanol in the presence of an esterification catalyst such as sulfuric acid or para-toluenesulfonic acid (Figure 2). 17 Purification of the reaction product includes removal of the catalyst, alkali refining, and stripping. 2
Alkyl succinates
Succinic anhydride reacts readily with alcohols to give monoesters of succinic acid (eg, decyl succinate from decanol), which are readily further esterified to the diesters by Fischer methods. 7 Dimethyl succinate can be produced from methanol and succinic anhydride or succinic acid, or by hydrogenation of dimethyl maleate. Diethyl succinate can be prepared by the same methods (from ethanol or diethyl maleate).
Chemical and Physical Properties
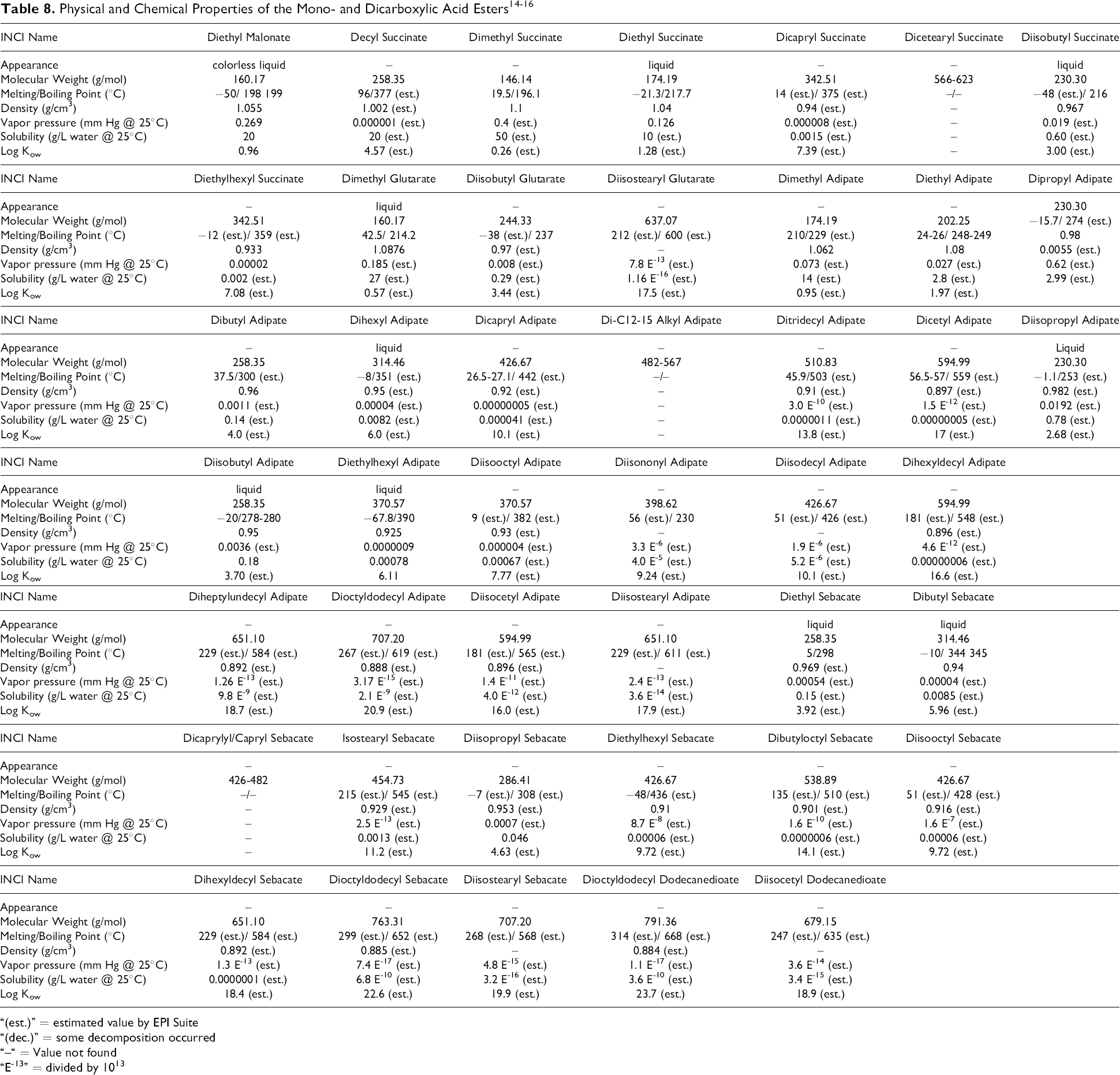
Table 8 lists the properties of the alkyl dicarboxylic acid esters. Figure 4 shows the relationship between molecular weight and the octanol water partitioning coefficient, expressed as log Kow, for these ingredients.
Example of the effects of chain length and branching on solubility. Log Kow vs molecular weight
“(est.)” = estimated value by EPI Suite
“(dec.)” = some decomposition occurred
“–“ = Value not found
“E-13” = divided by 1013
The diesters, in contrast to the free acids, are much more lipid soluble and more difficult to dissolve in water. The mono-esters, by definition, are hybrids of the acids and diesters, but their physical properties are much more closely related to the diesters.
Short-chain alkyl (ie, methyl, isopropyl, and butyl) mono- and diesters are more soluble in water, less lipophilic, and relatively more volatile than the corresponding longer chain alkyl (ie, C8-C13 alcohol) esters. 21 Most esters with molecular weights greater than 340 have boiling points greater than 300°C and are relatively nonvolatile and lipophilic (log Kow >7).
Impurities
Diethyl malonate
Diethyl malonate is a colorless organic liquid with an ester like odor. 39 The purity is typically > 99 %. Impurities from the production process include ethanol (ca. 0.1 % w/w), ethyl acetate (ca. 0.05 % w/w), and ethyl methyl malonate (ca. 0.05 % w/w).
Dibutyl adipate
Impurities are generally not found due to the manufacturing process, but available data demonstrate that arsenic levels are below a detection limit of 1 ppm, heavy metals (as lead) are below a detection limit of 10 ppm, and sulfated ash is below a detection limit of 0.1%. 81
Diisopropyl adipate and diethylhexyl adipate
Diisopropyl adipate and diethylhexyl adipate are considered stable; however, hydrolysis of the ester groupings may occur in the presence of aqueous acids or bases. No known impurities occur in either diisopropyl adipate or diethylhexyl adipate, although the acid values imply the presence of adipic acid or of the monoester in both. 2
Diethylhexyl adipate is commercially available with the following specifications: purity—99% to 99.9%; acidity—0.25 µg/100g max; moisture—0.05% to 0.10% max. 17
Diisopropyl sebacate
A supplier reported that the expected impurities in diisopropyl sebacate are the starting material sebacic acid, <0.3%, and isopropyl alcohol, <0.2%. 82
Ultraviolet Absorption
The alkyl dicarboxylic acid esters included in this review would not be expected to have any meaningful UV absorption. Except for the acid and ester functional groups, these ingredients do not possess any conjugated π bonds or nonbonding electrons. The π bonds and nonbonding electrons in the acid and ester functional groups are not part of any conjugated systems. Accordingly, these ingredients are unlikely to absorb light within the UVA-UVB spectrum at a detectable molar absorptivity.
Use
Cosmetic
The ingredients included in this safety assessment have a variety of functions in cosmetics. 19 For the esters, some of the common functions include skin conditioning agents, fragrance ingredients, plasticizers, solvents, and emollients. The functions of all ingredients are listed in Table 1.
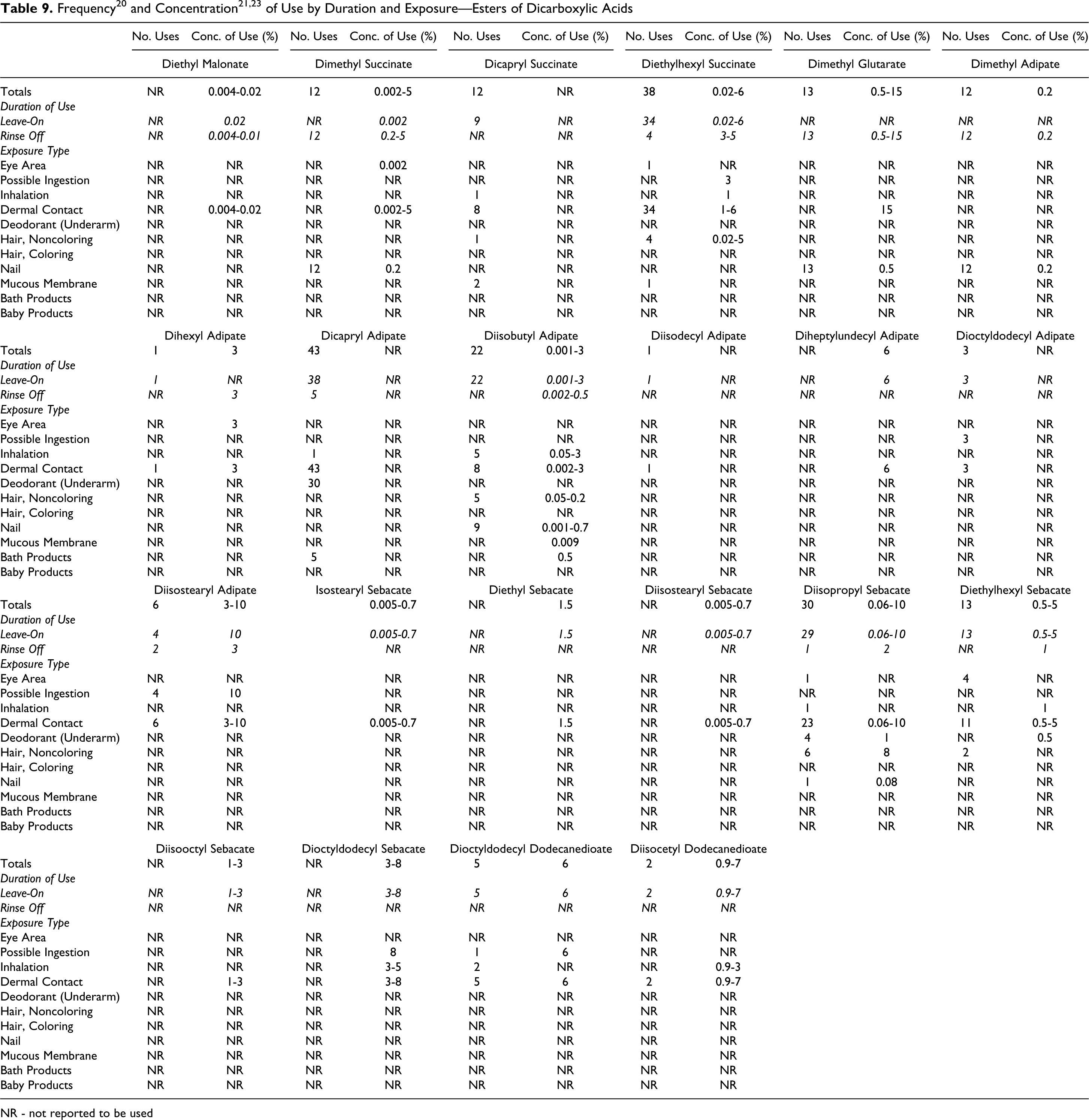
A total of 24 of the 44 esters included in this safety assessment are reported to be used in cosmetic formulations. The frequency of use of the esters, with the exception of dibutyl, diisopropyl, and diethylhexyl adipate, which have previously been reviewed, as supplied to the FDA by industry in 2010 as part of the Voluntary Cosmetic Registration Program (VCRP), 20 and the concentration of use, as supplied by industry in response to Personal Care Products Council (Council) surveys in 2009 21 and 2010, 22,23 are found in Table 9. The 2010 and historical use data for the 3 previously reviewed esters are found in Table 10. The 20 esters not currently reported to be used are listed in Table 11.
NR - not reported to be used
Current and Historical Frequency and Concentration of Use According to Duration and Type of Exposure—Previously Reviewed Esters

NR - not reported to be used
Ingredients Not Reported to Be Used

Induction of Peroxisome Proliferation—Esters of Dicarboxylic Acids

Diisopropyl adipate has the greatest number of current uses, with 70 reported. The highest concentration of use is for dimethyl glutarate, 15% in a dermal rinse-off product. The ingredients with the highest leave-on use concentrations, which are all dermal contact exposures, are diethylhexyl adipate, 14%, diisostearyl adipate, 10%, and diisopropyl sebacate, 10%.
Some of the alkyl dicarboxylic acid ester ingredients are applied around the eye, can possibly be ingested, or involve mucous membrane exposure, and some are used in underarm deodorants. None are reported to be used in baby products.
Dicapryl and diethylhexyl succinate, dibutyl, dicapryl, diisopropyl, diisobutyl, and diethylhexyl adipate, diisopropyl, diethylhexyl, and dioctyldodecyl sebacate, and dioctyldodecyl and diisocetyl dodecanedioate are used in hair sprays, and effects on the lungs that may be induced by aerosolized products containing this ingredient, are of concern.
The aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da, defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤ 10µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract. 86,87
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 88 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ~38 µm. 89 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable. Alkyl dicarboxylic acids esters are in the European Union (EU) inventory of cosmetic ingredients. 24
Noncosmetic
Many of the dicarboxylic acids esters are used in foods as direct or indirect food additives. 9 The diesters have widespread use as lubricants, plasticizers, and solvents. 86
Toxicokinetics
The simple alkyl di-esters are the result of the condensation of alkyl dicarboxylic acids and 2 equivalents of alkyl alcohols. These ingredients can be metabolized via hydrolysis back to the parent alcohol, the monoester, and the parent dicarboxylic acid (Figure 5). Previous safety assessments conducted by the Panel have addressed the safety of cetyl, methyl, isostearyl, myristyl, and behenyl alcohol.90,200
Metabolism of diesters in animals is expected to occur, initially, via enzymatic hydrolysis, leading to the corresponding dicarboxylic acids and the corresponding linear or branched alcohol. 87 These dicarboxylic acids and alcohols can be further metabolized or conjugated to polar products that are excreted in urine. However, other studies have shown that enzymatic hydrolysis of at least some diesters may be incomplete and result, instead, in the production of monoesters. 91
Diethyl malonate
Diethyl malonate is hydrolyzed via a 2-step reaction to malonic acid and the corresponding alcohol, ethanol. 39
Dimethyl malonate, which is not listed in the International Cosmetic Ingredient Dictionary, has similar physico-chemical properties and hydrolyzes in the same manner to malonic acid and methanol. Because of this similarity, data on dimethyl malonate are included in subsequent sections of part II to provide read-across data.
Distribution of diethyl malonate (and dimethyl malonate) is likely to occur in the water compartments, and accumulation in fat is unlikely based on physical and chemical properties. Both esters are likely to be metabolized by unspecific (serine-) esterases of different tissues, in particular, in the liver to the mono- esters and then to malonic acid and ethanol (or methanol). The hydrolysis product is likely to be metabolized via physiological pathways, such as the tricarboxylic acid cycle, as they are part of the normal intermediate metabolism. Both are assumed to readily absorb via mucous membranes.
In Vitro—Nonhuman
The percutaneous absorption of radiolabeled diethyl malonate was determined in vitro using skin from Yorkshire pigs. 39 [2- 14 C]Diethyl malonate was applied either undiluted (100 μg/cm2) or diluted in ethanol at 12.5 mg/mL with an applied dose of 100 μg/cm2 or as 0.5 mg/mL with an applied dose of 4 μg/cm2. At 50 hours, with undiluted diethyl malonate, 8.8% of the radioactivity was found in the skin and 3% was in the receptor fluid. With 100 µg in ethanol, 13% of the radioactivity was found in the skin and 6% in the receptor fluid and with 4 µg in ethanol, 30% was found in the skin and 10% in the receptor fluid. Absorption appeared to be enhanced with ethanol.
The percutaneous absorption of 1 mg/cm2 [2- 14 C]diethyl malonate in 10 µl acetone was determined in vitro also using skin from Yorkshire pigs. At 24 hours, 0.2% to 1.6% of the diethyl malonate was found in the receptor fluid, 0.2% to 0.9% was found in the skin, and 0.6% to 0.7% was found on the skin surface. Skin mediated hydrolysis amounted to 15% to 35% of the applied dose. In the receptor fluid, 20% to 21% of the applied dose was present as hydrolysis products. In the skin and on the skin surface, 3% to 5% and 2% to 4%, respectively, of the applied dose was present as hydrolysis products.
In Vivo—Nonhuman
The percutaneous penetration of radiolabeled diethyl malonate was studied in vivo in the following animal models: athymic nude mouse, human, and pig skin grafted to athymic nude mice, in weanling pigs, and in hairless dogs. 39 [ 2 –14 C]Diethyl malonate was applied at a dose of 0.1 mg/cm2 for 24 hours to a 1.27 cm2 area of mouse skin, or for 48 hours to a 25 cm2 area of pigs and hairless dogs using nonoccluded applications. According to the authors, the percutaneous absorption, was estimated from the recovery of radioactivity in urine and feces and corrected for the recovery observed after parenteral (sc) administration. Absorption was 15% in nude mice, 4 % in human skin grafted to nude mice, 6% in pig skin grafted to nude mice, 2.5% in pigs, and 4% in dogs.
In Vitro - Human
An in vitro skin absorption study was performed using diethyl malonate, no vehicle given. 39 Human cadaver split thickness skin was used in flow through cells. Diethyl malonate (4 μl) was applied to the skin samples. After 24 hours, 16% of the applied dose had penetrated through the skin. The maximum flux rate was reached after 5 hours and amounted to 280 μg/h (350 μg/cm2/h); the mean penetration rate was 99 μg/h (120 μg/cm2/h). Much of the test substance, 45 to 50%, evaporated from the skin, and 34 to 39% remained on the skin.
Ditridecyl adipate
The percutaneous absorption of [ 14 C]ditridecyl adipate was determined using groups of 10 male and 10 female Sprague-Dawley rats that were untreated or that had previously been exposed to unoccluded dermal applications of 0 or 2000 mg/kg bw ditridecyl adipate, 5 days/week for 13 weeks. 92 (This study is described in the section on ‘Subchronic Dermal Toxicity’). A single 58 µl dose of 2000 mg/kg bw [ 14 C]ditridecyl adipate was applied topically (size of test site not specified), and urine and feces were collected for 4 days. In the previously untreated rats, a total of 11.6 and 10.6% of the [ 14 C] solution was absorbed by male and female rats, respectively, over 4 days. Approximately 63 and 52% of the absorbed dose (7.4 and 5.5% of the applied dose, respectively) was found in the tissues of males and females, respectively. A total of 3.5% to 4.7% of the applied dose was recovered in the urine and 0.4% to 0.7% in the feces of previously untreated rats. The values for the animals previously dosed with 2000 mg/kg bw ditridecyl adipate were not statistically significantly different from the controls. In the previously dosed animals, a total of 10.8 and 9.1% of the dose was absorbed by males and females, respectively, over the 4 days, with approximately 87 and 81% of the absorbed dose (9.4 and 7.4% of the applied dose, respectively) found in the tissues of the male and female rats, respectively. A total of 0.7% to 1.3% of the [ 14 C] was recovered in the urine and 0.4% to 0.6% in the feces. Based on the radioactivity recovered in the urine, the bioavailability of ditridecyl adipate was 2% to 6%, and previous dosing did not significantly affect absorption.
Diethylhexyl Adipate In Vitro
The in vitro hydrolysis of diethylhexyl adipate (and mono-(2-ethylheyxl) adipate [MEHA]) using tissue preparations from the liver, pancreas, and small intestine of 2 rats was examined, as were the effects of diethylhexyl adipate on serum and hepatic enzymatic activities in vitro. 93 Diethylhexyl adipate was readily hydrolyzed to MEHA or adipic acid by each tissue preparation. The formation of adipic acid was rapid and approximately the same for all 3 tissues, while the formation of MEHA was rapid only in pancreatic tissue and was negligible in the intestine. The rate of hydrolysis from MEHA to adipic acid was greater that than from diethylhexyl adipate and the highest activity was found in intestinal tissue. In examining the effects on serum and hepatic enzymes, only N-demethylase activity was considerably inhibited by diethylhexyl adipate.
In Vivo—Nonhuman
The elimination, distribution, and metabolism of diethylhexyl adipate was investigated using male Wistar rats. 93 In these studies, diethylhexyl adipate was labeled at the carbonyl carbon. In elimination studies, 2 rats were dosed by gavage with 500 mg/kg bw [ 14 C]diethylhexyl adipate (1.26 µCi/rat) as a saturated solution in dimethyl sulfoxide (DMSO), and respired carbon dioxide, urine, and feces were collected for 2 days. At 24 hours after dosing, 86% of the administered dose was excreted, and at 48 hours, more than 98% of the dose was excreted. In 1 animal, 44.8% of the dose was excreted in expired carbon dioxide and 33.9% in the urine at the 48 hours measurement, while in the other rat, 21.1% and of the dose was excreted in expired carbon dioxide and 52.2% in the urine. Little (1.4 or 5% of the dose) was excreted in the feces.
In the distribution study, 3 rats per group were given a single dose as described above. The animals were killed at various intervals, and blood, organ, and tissue samples were collected. Not taking into account the stomach and intestines, the greatest levels of radioactivity, as a percent of dose administered, were found in the liver, kidney, blood, muscle, and adipose tissue. These values ranged from 0.34% to 8.21% at 6 hours, with the greatest percentage found in the adipose tissue, and from 0.54% to 3.44% at 12 hours, with the greatest percentage found in the muscle. In most tissues, the amount of residual radioactivity reached a peak by 6 hours, except for the liver, kidneys, testicles, and muscle, which reached a peak at 12 hours. The researchers stated that the elimination of radioactivity from the tissues and organs was very rapid, and there was no specific organ affinity.
The metabolism of diethylhexyl adipate was examined in rats dosed orally, by gavage, with 100 mg of nonlabeled diethylhexyl adipate as a 5% solution in DMSO. A control group was dosed with vehicle only. The rats were killed 1, 3, or 6 hours after dosing. The metabolites were determined using GLC. Diethylhexyl adipate was rapidly hydrolyzed to adipic acid, the main intermediate metabolite, and MEHA. In the urine, adipic acid was detected at 1 hours, and excretion as adipic acid in the urine reached 20% to 30% at 6 hours. Diethylhexyl adipate and MEHA were not detected in the urine. Adipic acid only also was detected in the blood and the liver, with constant excretion of 0.5% to 0.7% of the dose in the blood and excretion in the liver increasing with time, with 2% to 3.3% excreted in the liver at 6 hours. In the stomach, diethylhexyl adipate, adipic acid, and MEHA were found. The concentrations of diethylhexyl adipate declined rapidly, while the levels of adipic acid (9% to 10%) and MEHA (6% to 11.5%) peaked at 3 hours. Adipic acid, but not MEHA, was found in the intestine and increased with time, reaching 19% at 6 hours.
The absorption, distribution, and elimination of diethylhexyl adipate was examined using radioactive labeling on the acid [carbonyl- 14 C] (specific activity 39.5 mCi/mmol) or the alcohol [2-ethylhexyl-1- 14 C] (44.1 mCi/mmol). 34 The researchers used both DMSO and commercial corn oil as vehicles for all tests, since the were of the opinion that DMSO is an active penetrant and carrier of other substances through tissue membranes. It was also their opinion that a fat-soluble substance, such as diethylhexyl adipate, is more realistically studied dissolved in corn oil. The following groups of animals were dosed with 84.3 µg (9 µCi) [carbonyl 14 C]diethylhexyl adipate or 84.3 µg (10 µCi) [2-ethylhexyl-1- 14 C]diethylhexyl adipate in both vehicles: 12 male NMRI mice were dosed iv and killed at intervals from 5 minutes to 4 days after dosing; 10 male NMRI mice were dosed intragastrically (i.g.) and killed at intervals from 20 minutes to 4 days after dosing; 12 gravid NMRI mice were dosed iv or i.g. on day 17 of gestation and killed at intervals from 20 minutes to 24 hours. Six male rats were dosed i.g. with 843 µg (90 µCi) [carbonyl 14 C]diethylhexyl adipate or 843 µg (100 µCi) [2-ethylhexyl-1- 14 C]diethylhexyl adipate and killed at intervals from 20 minutes to 4 hours. Whole body autoradiography was used to determine tissue distribution.
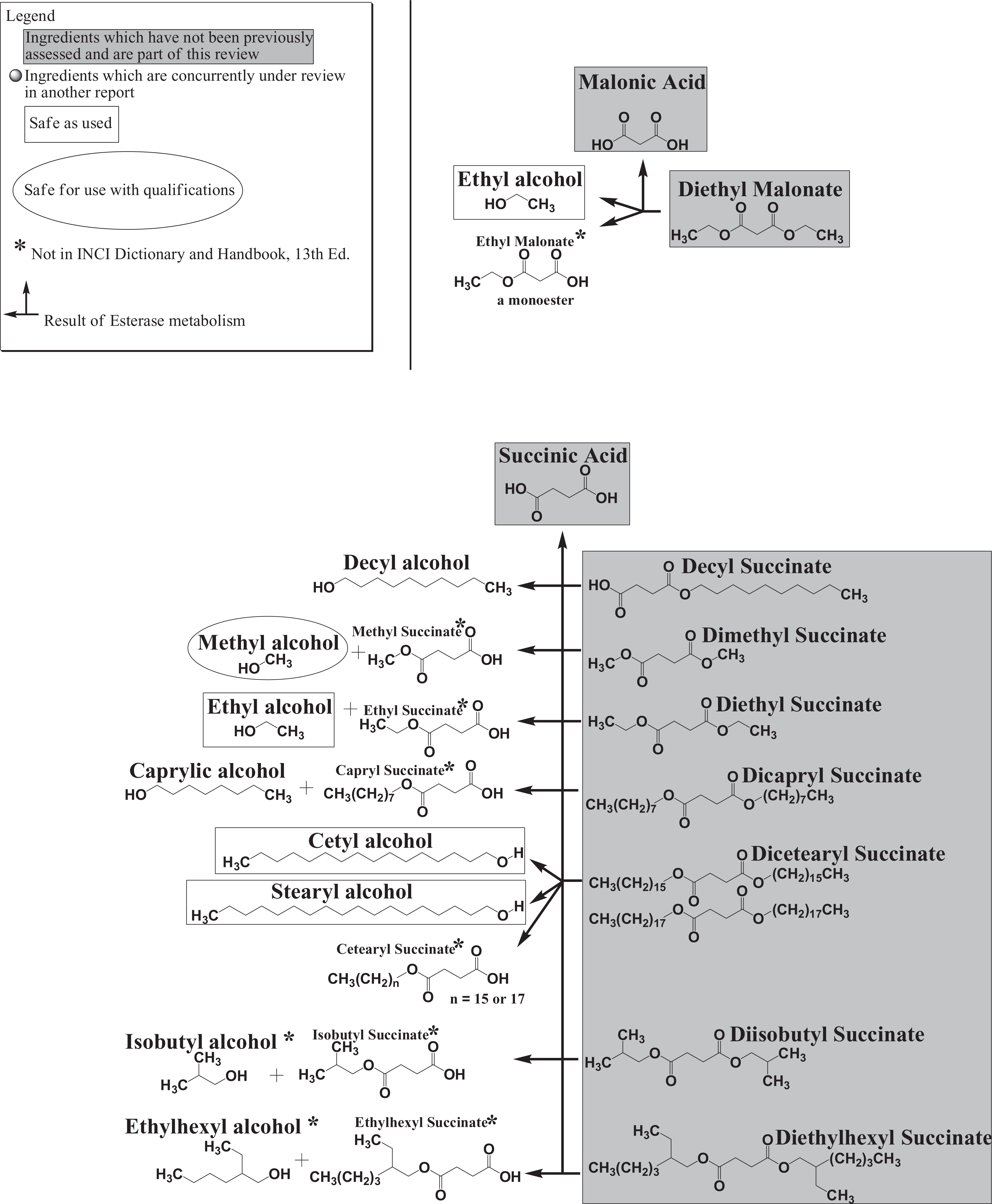
Map of the malonic and succinic ester ingredients in this assessment and associated esterase metabolites.
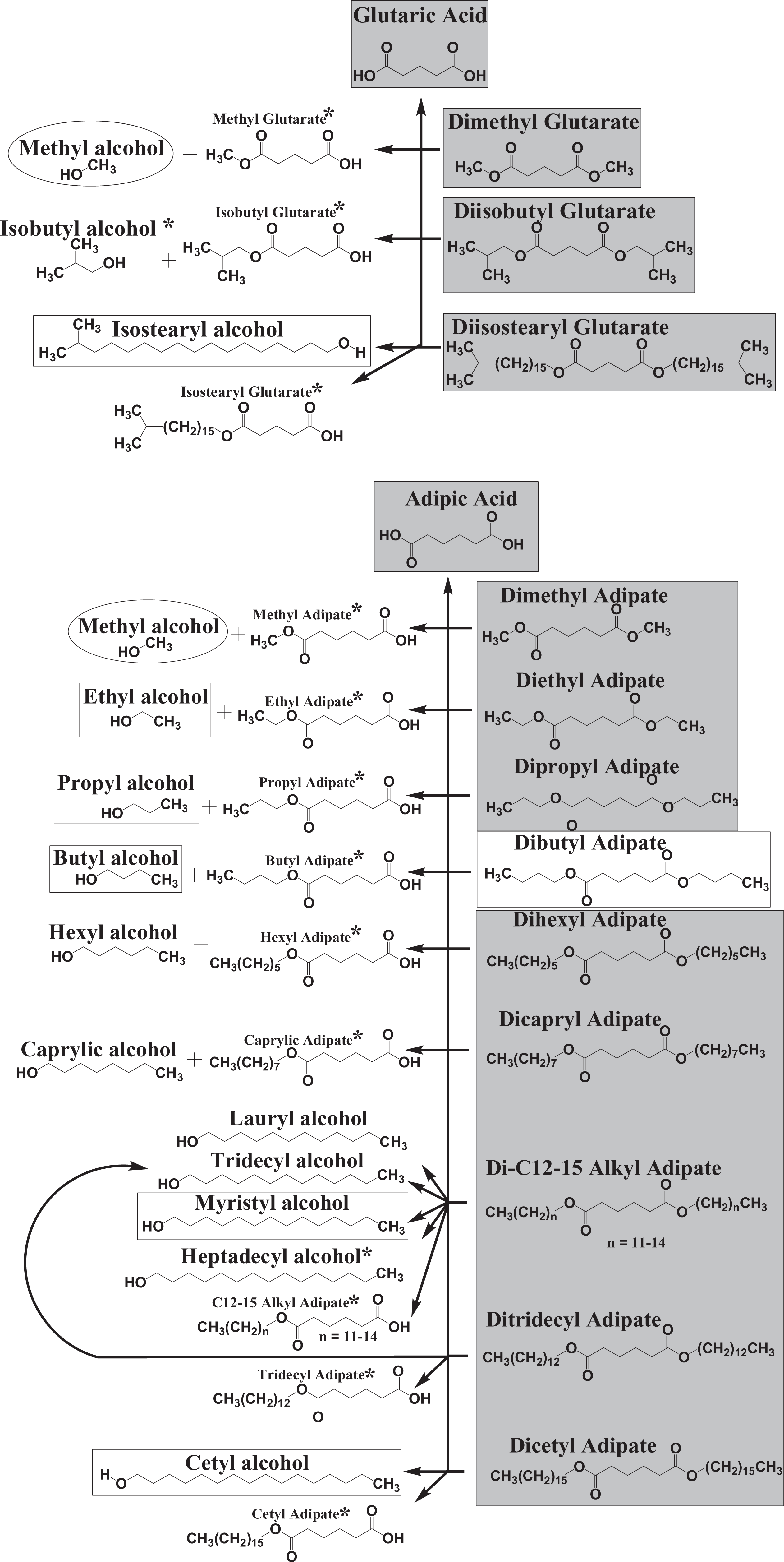
Map of the glutaric and straight-chain adipic ester ingredients in this assessment and associated esterase metabolites.
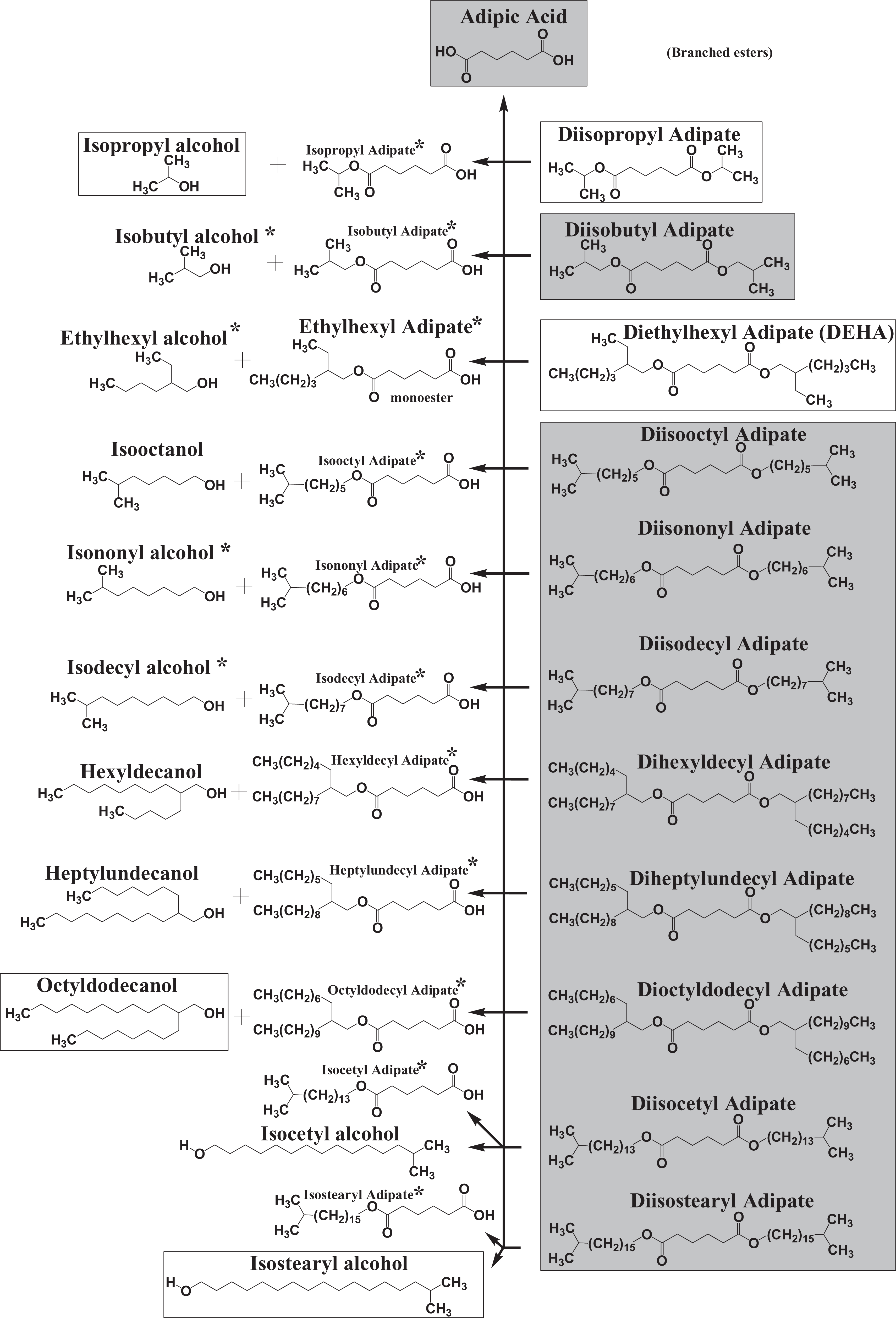
Map of the branched chain adipic ester ingredients in this assessment and associated esterase metabolites.

Map of the sebacic and dodecanedioic ester ingredients in this assessment and associated esterase metabolites.
Following dosing with [carbonyl- 14 C]diethylhexyl adipate, distribution was similar in male mice, male rats, and gravid mice. The amount of radioactivity in the tissues peaked at a later time following i.g. dosing as compared to iv dosing. The presence of radioactivity in the gastrointestinal tract following i.v dosing indicated biliary excretion. Four hours following both iv and i.g. dosing, the greatest uptake of radioactivity was found in the liver, bone marrow, brown fat, adrenal cortex, kidneys, and a few other tissues. At 24 hours after i.g. dosing, significant levels of radioactivity remained in several tissues, including the liver, of both rats and mice. In gravid mice, a “remarkable strong uptake” of radioactivity in the corpora lutea of the ovary was observed at all time intervals with both iv and i.g. dosing, and some radioactivity was found in the fetal intestine, liver, and bone marrow.
Similar distribution patterns were seen following dosing with [2-ethylhexyl-1- 14 C]diethylhexyl adipate as were seen with [carbonyl- 14 C]diethylhexyl adipate. Following i.g. dosing, the appearance of radioactivity was lessened and not as great as it was with iv dosing. Very high radioactivity levels were seen in the liver and kidney at 5 minutes to 1 hour after iv dosing and at 20 minutes to 4 hours after i.g. dosing. The radioactivity in the liver was still high at 24 hours after i.g. dosing in mice and rats. Radioactivity was also seen in the intestinal contents at 1 to 4 hours after iv dosing, again indicating biliary excretion. At longer intervals after iv injection, 4 hours-4 days, radioactivity was detected in the bronchi of mice. While radioactivity was observed in the ovaries of gravid mice and some fetal tissues following dosing with [carbonyl- 14 C]diethylhexyl adipate, none was detected in the ovaries of gravid mice after dosing with [2-ethylhexyl-1- 14 C]diethylhexyl adipate, and very little radioactivity was seen in some fetal tissues.
The effect of vehicle on the absorption and biliary and urinary excretion of diethylhexyl adipate was also examined using rats in a gavage study with [ 14 C]diethylhexyl adipate. Radioactivity was measured every 30 minutes for 7.5 hours. The times and extent of absorption were different for all 4 preparations of [ 14 C]diethylhexyl adipate. Radioactivity levels in the blood increased faster and were greater with DMSO as the vehicle, as compared to corn oil. The highest blood radioactivity levels were found with [carbonyl- 14 C]diethylhexyl adipate in DMSO. Biliary excretion of [ 14 C]diethylhexyl adipate was greatly affected by vehicle; with DMSO, 41% of the dose was detected in the bile, while only 10% of the dose was found with the corn oil vehicle. This difference was not seen with [carbonyl- 14 C]diethylhexyl adipate. Finally, vehicle did not have much influence on urinary excretion. However, unlike the results reported by the previous researchers, little radioactivity was excreted in the urine. The researchers hypothesized that since the study duration was only 7.5 hours, urinary excretion may not have been complete.
The metabolism of diethylhexyl adipate was examined in vivo using male Wistar rats and compared to in vitro metabolism using hepatocytes. 94 In vivo, rats were dosed with 0.665 or 1.5 g/kg diethylhexyl adipate in corn oil by gavage for 5 days, and the controls were given vehicle only. Urine was collected daily. Diethylhexyl adipate was not recovered in the urine after 24 hours. Adipic acid was the main metabolite of diethylhexyl adipate. In vitro, the first hydrolysis of diethylhexyl adipate appears to be a rate-limiting step. In vivo, it was thought that this hydrolysis probably occurs in the gastrointestinal tract. Metabolic pathways (ω and ω-1oxidations, glucuronidation) seemed to prove that transformations of diethylhexyl adipate are localized mainly in the liver.
Oral administration of diethylhexyl adipate to cynomolgus monkeys results in rapid elimination, with 47% to 57% of the dose excreted in the urine. 17 Unchanged diethylhexyl adipate is absorbed from the gastrointestinal tract, and the glucuronide of MEHA and traces of unchanged diethylhexyl adipate were found in the urine. (Details were not provided).
In Vivo—Human
The pharmacokinetics of [ 2 H10]diethylhexyl adipate, labeled on the ethyl side-chains, were examined using 6 male participants. 95 A dose of 46 mg [ 2 H10]diethylhexyl adipate in corn oil, for a total volume of 0.5 cm3, was administered orally in a gelatin capsule. Blood samples were taken for up to 31 hours after dosing, and urine samples were taken at intervals for up to 96 hours after dosing. In the plasma, unconjugated [ 2 H5]2-ethylhexanoic acid was the only measurable diethylhexyl adipate-related compound. This compound appeared rapidly in the plasma, and the peak concentrations (1.6 ±0.5 µg/cm3) occurred between 1 and 2 hours. [ 2 H5]2-Ethylhexanol was detected, but it was below the limit of quantification. The rate of metabolite formation was calculated, since there was no evidence of diethylhexyl adipate absorption, as 1.63 ± 1.19 hr-1. The rate of elimination from the plasma was also rapid and estimated to be 0.42 ± 0.15/h, which corresponded to an elimination half-life of 1.65 hours. Although there were inter-individual differences in the rate and extent of [ 2 H5]2-ethylhexanoic acid formation, it was below the limit of detection in all participants by 31 hours.
In the urine, [ 2 H5]2-ethylhexanoic acid was again the principal metabolite, and it was probably eliminated as a conjugated product. This conjugated form, most likely the glucuronide, accounted for up to 99% of the total [ 2 H5]2 ethylhexanoic acid measured. Conjugation of the other urinary metabolites was minimal. Peak urinary elimination of the measured metabolites occurred within 8 hours of dosing, and no metabolites were detected in the urine after 36 hours. The rates of elimination were similar for all metabolites, with a mean elimination half-life of 1.5 hours. The measured urinary metabolites accounted for 12.1% of the dose, with the majority being eliminated in 24 hours. Fecal analysis determined that a minor portion of the dose was present as diethylhexyl adipate (0.43%) and [ 2 H5]MEHA (0.27%). The researchers noted that recovery of the administered dose was incomplete and hypothesized that it was most probably due to further systemic metabolism.
Diethylhexyl sebacate
Diethylhexyl sebacate is not readily absorbed through the skin of guinea pigs (no further details were provided). 1 It was noted that the metabolism of diethylhexyl sebacate in rodents and humans may follow partially common pathways.
Toxicologic studies
Peroxisome Proliferation
Diethylhexyl adipate is a peroxisome proliferator requiring extensive phase I metabolism to produce the proximate peroxisome proliferator, which in both mice and rats, appears to be 2-ethylhexanoic acid. 96,97 Diethylhexyl adipate has been studied because it is structurally related to diethylhexyl phthalate, although diethylhexyl adipate is not as potent a proliferator as diethylhexyl phthalate. 98,99
Peroxisome proliferation causes an increase in liver weights and can induce hepatocarcinogenicity in rats and mice. Peroxisome proliferation is not believed to pose the risk of inducing hepatocarcinogenesis in humans, as a species difference in response to peroxisome proliferators exists. 100 In vitro and in vivo studies examining the induction of peroxisome proliferation by diethylhexyl adipate and diethylhexyl sebacate are summarized in Table 12. 67,96 –99,101,102 While proliferation was observed, these ingredients have much weaker activity than diethylhexyl phthalate and ciprofibrate, which are very effective peroxisome proliferators.
Humans do not react to peroxisome proliferators in the manner that rodents do. 100 There is no effect on organelle proliferation and induction of peroxisomal and microsomal fatty acid-oxidizing enzymes in species other than rats and mice, including humans. Consequently, these results have no relevance to humans.
Single Dose (Acute) Toxicity—Esters of Dicarboxylic Acids
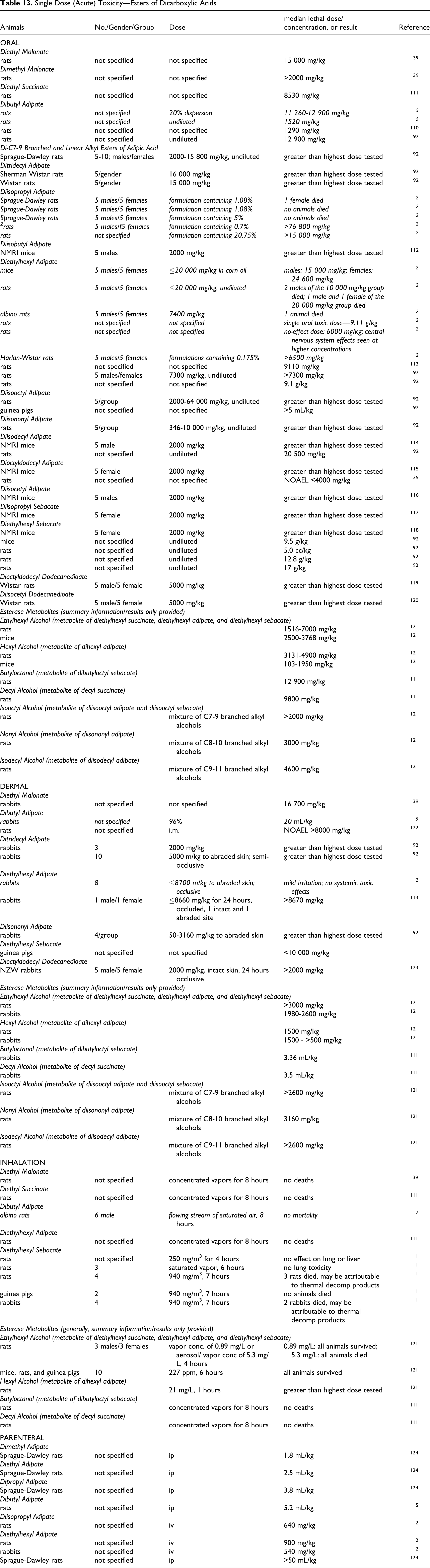
DNA Binding/DNA Synthesis
Diethylhexyl adipate
The potential of diethylhexyl adipate to bind to liver DNA of female NMRI mice was evaluated by administering a solution of 119 mg diethylhexyl adipate/mL with 3.85 mCi/mL of [ 14 C]diethylhexyl adipate (labelled at C1 of the alcohol moiety) and 27.7 mCi/mL of [ 3 H]diethylhexyl adipate (tritiated at positions 2 and 3 of the alcohol moiety) in olive oil. 103 The animals were dosed by gavage, and the livers were excised 16 hours after dosing. Some animals were pretreated with 10 g/kg of unlabeled dietary diethylhexyl adipate for 4 weeks. Diethylhexyl adipate did not covalently bind to hepatic DNA in mice. Pretreatment with diethylhexyl adipate caused an increase in liver weight, but no increase in DNA binding. The researchers stated that tumorigenicity of diethylhexyl adipate must be due to an activity other than DNA binding.
The ability of diethylhexyl adipate to stimulate liver DNA synthesis in male F344 rats was investigated using radiolabeled thymidine. 104 Contrary to expected results, diethylhexyl adipate did stimulate DNA synthesis. The stimulation factor, which is indicated by the ratio of the thymidine incorporation in treated animals compared to controls, was 10.5 and the doubling dose, which is the dose that produced a doubling of the control level DNA synthesis, was 0.7 mmol/kg.
The effect of dosing with diethylhexyl adipate on 8-hydroxydeoxyguanosine (8-OH-dG) in liver and kidney DNA of rats was examined. 105 Groups of 10 male F344 rats were fed a diet containing 0 or 2.5 diethylhexyl adipate. Five animals per group were killed after 1 week, and the other 5 after 2 weeks of dosing. Relative liver to bws were statistically significantly increased after 1 and 2 weeks of dosing, and the relative kidney to bws were statistically significantly increased only after 2 weeks. A statistically significant increase in 8-OH-dG was observed in the liver DNA, but not the kidney DNA, at week 1 and 2.
The IARC remarked that the weight of evidence for diethylhexyl adipate, and other rodent peroxisome proliferators in general, demonstrated that rodent peroxisome proliferators do not act as direct DNA-damaging agents. 17
Hepatic Lipid Metabolism
Diethylhexyl adipate
Dietary administration of diethylhexyl adipate affects hepatic lipid metabolism. 17 Hepatic fatty acid-binding protein and microsomal stearoyl-CoA desaturation were increased in Wistar rats fed 2% diethylhexyl adipate for 7 days. 106,107 When fed to rats for 14 days, an increase in hepatic phospholipid levels and a decrease in phosphatidylcholine:phosphatidylethanolamine ratio was reported. 108 In male NZB mice fed 2% diethylhexyl adipate for 5 days, an induction of fatty acid translocase, fatty acid transporter protein, and fatty acid binding protein in the liver was reported. 109
Cellular Effects
Dibutyl adipate
Dibutyl adipate was tested for cytotoxicity in the metabolic inhibition test. A dilution series of dibutyl adipate was suspended in HeLa cells. Dibutyl adipate had no acute toxicity to the cells, which was attributed to its insolubility in water. 5
Single Dose (Acute) Toxicity
Acute toxicity data on esters of dicarboxylic acids esterase metabolites are presented in Table 13.
Ocular Irritation—Esters of Dicarboxylic Acids a
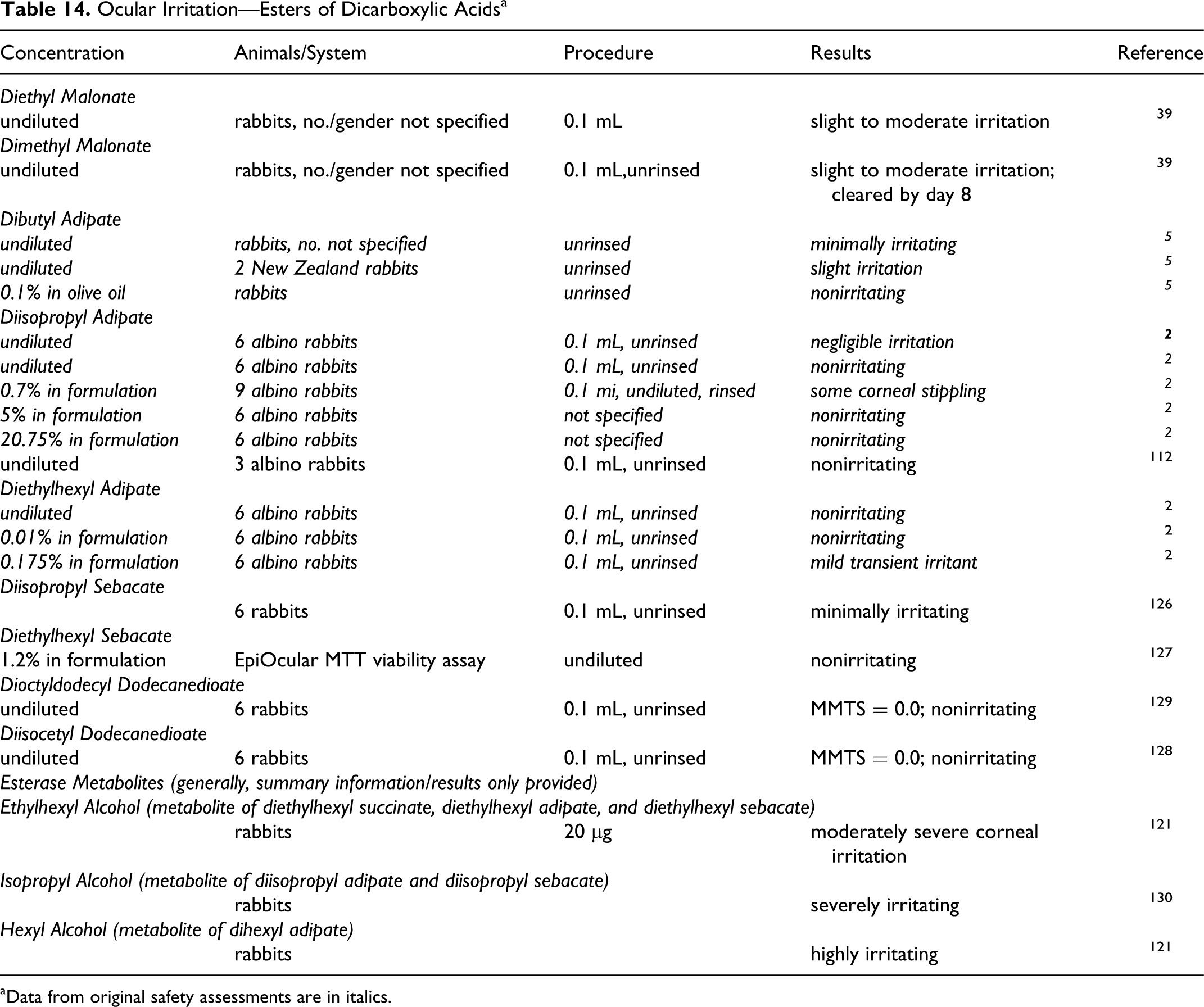
aData from original safety assessments are in italics.
Repeated Dose Toxicity
Dibutyl Adipate
Oral
Male and female Crj:CD(SD) rats, number per group not specified, were dosed orally, by gavage, with 0, 20, 140, or 1000 mg/kg bw dibutyl adipate in olive oil daily for 28 days. 110 No clinical, hematological, or microscopic test-article related changes were observed.
Dermal
Groups of 10 rabbits were dosed dermally with 0.5 or 1.0 mL/kg/d of a 20% dispersion of dibutyl adipate, 5x/week for 6 weeks. A significant reduction in bw gain was seen for animals of the 1.0 mL/kg/d group, and renal lesions were seen in 1 animal of each group. 5
No adverse effects were reported in a study using 4 dogs in which entire-body applications of an emulsion con×tain×ing 6.25% dibutyl adipate were made 2x/week for 3 mos. 5
Diethylhexyl Adipate
Oral
In a 14-day dietary study, groups of 5 male rats and mice were given ≤50 000 ppm and groups of 5 female rats and mice were given ≤100,000 ppm diethylhexyl adipate. Male rats and mice fed 50 000 ppm and female rats and mice fed ≥25,000 ppm had decreased weight gains or weight loss. (It is not specified whether the results were statistically significant.) One female rat and all female mice of the 100 000 ppm group died. 2
In a 14-day study in which 5 male and 5 female Wistar and F344 rats and Swiss and B6C3F1 mice were dosed with 0 to 2.5 g/kg diethylhexyl adipate in corn oil for 14 days, diethylhexyl adipate was toxic to female B6C3F1 mice, causing mortality, at a dose level of 2.5 g/kg. 97 The toxicity of 2 metabolites of diethylhexyl adipate, 2-ethylhexanol and 2-ethylhexanoic acid, was also examined using Wistar rats and Swiss mice. 2-Ethylhexanol was toxic to male and female rats, with mortality reported at doses >1.05 g/kg in male and female rats. 2-Ethylhexanoic acid was toxic to female rats, with mortality reported at doses ≥1.9 g/kg; mortality was not reported for male rats. These effects were not reported in mice.
In a 1 and 4-week dietary study in which groups of 5 to 8 rats and mice were fed diets containing 0% to 4.0% and 0% to 2.5% diethylhexyl adipate, respectively, feed consumption by rats was decreased in the 4.0% group at 1 week and in the 2.5 and 4.0% dose groups at 4 weeks. 101 Body weights were significantly decreased in these groups. Feed consumption by mice was not affected, but a significant decrease in bws was seen in the 1.2 and 2.5% dose groups at 4 weeks.
Toxicity was evaluated in a study in which groups of 10 female Crl:CD(SD) rats were dosed, by gavage, with 5 mL/kg of 0, 200, 1000, or 2000 mg/kg bw diethylhexyl adipate in corn oil for 2 or 4 weeks. 125 All animals survived until study termination. In the 2-week study, no statistically significant findings were observed for the animals dosed with 200 mg/kg bw, and the only statistically significant finding in the 1000 mg/kg bw dose group was an increase in relative liver to bw. In the 2000 mg/kg bw dose group, there was staining around the perineum, statistically significant increases in relative liver and kidney to bws, and a statistically significant decrease in the relative weight of the left ovary. Microscopically, abnormal findings were reported for both the ovary and kidney. In the ovary, an increase in atresia of the large follicle and a decrease in currently formed corpora lutea were seen in animals dosed with 1000 and 2000 mg/kg bw, and in the 2000 mg/kg bw group, an increase in follicular cysts was observed. In the kidney, an eosinophilic change of the proximal tubule was observed for the 2000 mg/kg bw dose group. The NOAEL was 200 mg/kg bw.
In the rats dosed for 4 weeks, similar observations were made. There was staining around the perineum of animals dosed with 1000 and 2000 mg/kg bw diethylhexyl adipate, and final bws of animals dosed with 2000 mg/kg bw were statistically significantly decreased. The relative kidney to bws were statistically significantly increased in animals at all dose levels, and liver weights were statistically significantly increased in animals of the 1000 and 2000 mg/kg bw dose groups. The mean estrous cycle length was statistically significantly decreased in the 200 mg/kg bw dose group, but this was not considered treatment-related since a dose-response was not seen. The same microscopic abnormalities reported in the 2 weeks study were seen in the ovaries and kidneys of the animals dosed with 1000 and 2000 mg/kg bw in the 4-week study. As in the 2-week study, the NOAEL for ovarian toxicity was 200 mg/kg bw.
In a 13-week dietary study, groups of 10 rats and 10 mice were fed ≤25 000 ppm diethylhexyl adipate. With the exception of decreased weight gain for some of the groups, no compound-related toxicologic effects were observed. 2
In a 90-day dietary study, groups of 10 rats per group were fed 0 to 4740 mg/kg bw diethylhexyl adipate for 90 days. 113 Mortality occurred in the 4740 mg/kg bw group, but the number of deaths was not specified. Decreased growth and feed consumption was reported for animals fed 2920 mg/kg bw. Changes in kidney and liver weights were noted, but no details were given. The NOEL was 610 mg/kg bw, and the LOEL was 2920 mg/kg bw diethylhexyl adipate.
Groups of 15 male and 15 female Sprague Dawley rats were fed 0 or 2.5% diethylhexyl adipate for 90 days. 92 At study termination, all animals were killed for necropsy. Body weight gains were statistically significantly decreased for treated males and females, and relative kidney and liver to bws were statistically significantly increased for treated females, when compared to controls.
In a 13-week dietary study in which groups of 5 to 8 rats and mice were fed diets containing 0% to 4.0% and 0% to 2.5% diethylhexyl adipate, feed consumption by rats was decreased in the 2.5 and 4.0% dose groups, and bws were significantly reduced in these groups. 101 Feed consumption by mice was not affected, but a significant decrease in bws was seen in the 1.2 and 2.5% dose groups.
Intragastric doses of ≤2.0 g/kg diethylhexyl adipate to rats (number not stated) for 6 mos produced no enzymatic changes, but levels of sulphydryl compounds in the blood were increased. Hepatic detoxification appeared depressed at the onset of the study, but it was accelerated after 6 mos. Administration of 0.1 g/kg for 10 mos decreased central nervous system excitability. 2
Dermal
Diethylhexyl adipate, 410 or 2060 mg/kg bw, was applied to the shaved abdomen of male rabbits, 4 per group, 5 days per week for 2 weeks. 113 Mineral oil was applied in the same manner to a group of 4 rabbits as a negative control. A collar was used to restrict ingestion. One animal in the 410 mg/kg bw group died during week 2 of the study. All other animals in this group appeared normal. Slight to moderate erythema was observed at the test site. No animals of the 2060 mg/kg bw group died, but 3 of the 4 did not gain weight, and they had labored breathing and were lethargic during week 2. Moderate erythema was observed in this group. Microscopically, 1 animal of the 2060 mg/kg bw group had altered cytology of the liver parenchymal cells. No other microscopic lesions were noted.
Diisopropyl adipate
An immersion study was performed using guinea pigs in which a product containing 20.75% diisopropyl adipate was diluted, giving an actual adipate concentration of 0.10%. The animals were immersed 4 h/d for 3 days. There were no signs of systemic toxicity, and the degree of dermal irritation was considered minimal. 2
Diethylhexyl sebacate
No deaths occurred when 4 rats, 2 guinea pigs, 2 rabbits and 1 cat were exposed to 400 mg diethylhexyl sebacate/m3, 7 h/d, for 10 days. 1 Details were not provided.
Groups of 12 F344 rats, gender not specified, were exposed 4 hours/d, 5 days/week, to 25 or 250 mg/m3 diethylhexyl sebacate for ≤13 weeks. 92 No adverse systemic or lung effects were observed.
Diethyl malonate
Groups of 10 to 16 male and female CD rats were fed diets containing either 0, 36 (males) or 41 mg/kg bw/d (females) diethyl malonate for 90 days. 39 No treatment related effects were observed.
Di-C7-9 branched and linear alkyl esters of adipic acid
Groups of 15 male and 15 female Sprague-Dawley rats were fed a diet containing 0, 0.1, 0.5, or 2.5% di-C7-9 branched and linear alkyl esters of adipic acid for 90 days, corresponding to approximately 1500 mg/kg bw/d for high-dose males and 1900 mg/kg bw/d for high-dose females. 92 All rats were killed for necropsy at study termination. No systemic toxicity was reported. Small, but significant, increases in absolute and relative kidney to bws reported for females of the 2.5% dose group were not considered treatment-related. The NOAELs for male and female rats were 1500 and 1950 mg/kg bw/d, respectively.
Diisononyl adipate
Groups of 10 male and 10 female rats were fed 0, 50, 150, or 500 mg/kg bw diisononyl adipate for 13 weeks. 92 A statistically significant increase in relative kidney to bws was reported for males and females given 500 mg/kg bw, but absolute kidney weights were not affected and no significant microscopic effects were seen. Microscopic changes in any of the organs, including the testes and epididymis of males and ovaries of females, were not observed. There were no significant toxicological findings, and the NOAEL was 500 mg/kg bw/d.
In another 13-week study, groups of 4 male and 4 female Beagle dogs were fed 0, 0.3, 1.0, or 3.0% diisononyl adipate; the high dose was increased to 6% during weeks 9 to 13. 92 No significant findings were reported for the 0.3 or 1.0% groups. In the high-dose group, decreased bws, testes weight, and feed consumption, increased liver weights, elevated enzyme levels, liver and kidney discoloration, and microscopic changes in the liver, testes, spleen, and kidneys were reported. The dietary NOAEL for diisononyl adipate was 1.0%.
Ditridecyl adipate
Ditridecyl adipate, 0, 800, or 2000 mg/kg bw, was applied to the backs of groups of 10 male and 10 female Sprague-Dawley rats, 5 days/week for 13 weeks. 92 The test sites were not occluded, but the animals wore Elizabethan collars. Slight erythema and flaking of the skin was observed in the treated groups, with hyperplasia of the sebaceous glands in the dermis, but otherwise no significant differences were observed between test and control animals. Differences in relative organ to bws were not statistically significant, and ditridecyl adipate did not appear to cause systemic toxicity.
Dibutyl sebacate
Groups of 5 male and 5 female Sprague-Dawley rats were fed a diet containing 0, 0.01, 0.05, 0.25, or 1.25% dibutyl sebacate for 1 year. 122 Necropsies were performed whenever rats exhibited significant weight losses or other evidences of severe concurrent infection. Dibutyl sebacate had no effect on growth or well-being.
The researches then fed groups of 16 male Sprague Dawley rats a diet containing 0.01, 0.05, 0.25, 1.25, or 6.25% dibutyl sebacate for 2 years. 122 Two control groups were given untreated feed. Necropsies were performed on 3 rats from each group after 1yr, and the experiment was terminated at the end of the 2-year feeding period. Interim, animals were killed whenever they became moribund. In such instances the rats usually had incapacitating tumors or severe intercurrent infections. Dibutyl sebacate did not adversely affect growth or survival, and it did not produce significant hematological changes in peripheral blood. As the rats increased in age, slight changes in distribution of leukocytes were found, but these trends occurred in both the control and treatment groups.
Ocular Irritation
Ocular irritation data on esters of dicarboxylic acids and esterase metabolites are presented in Table 14.
Dermal Irritation and Sensitization—Esters of Dicarboxylic Acids a
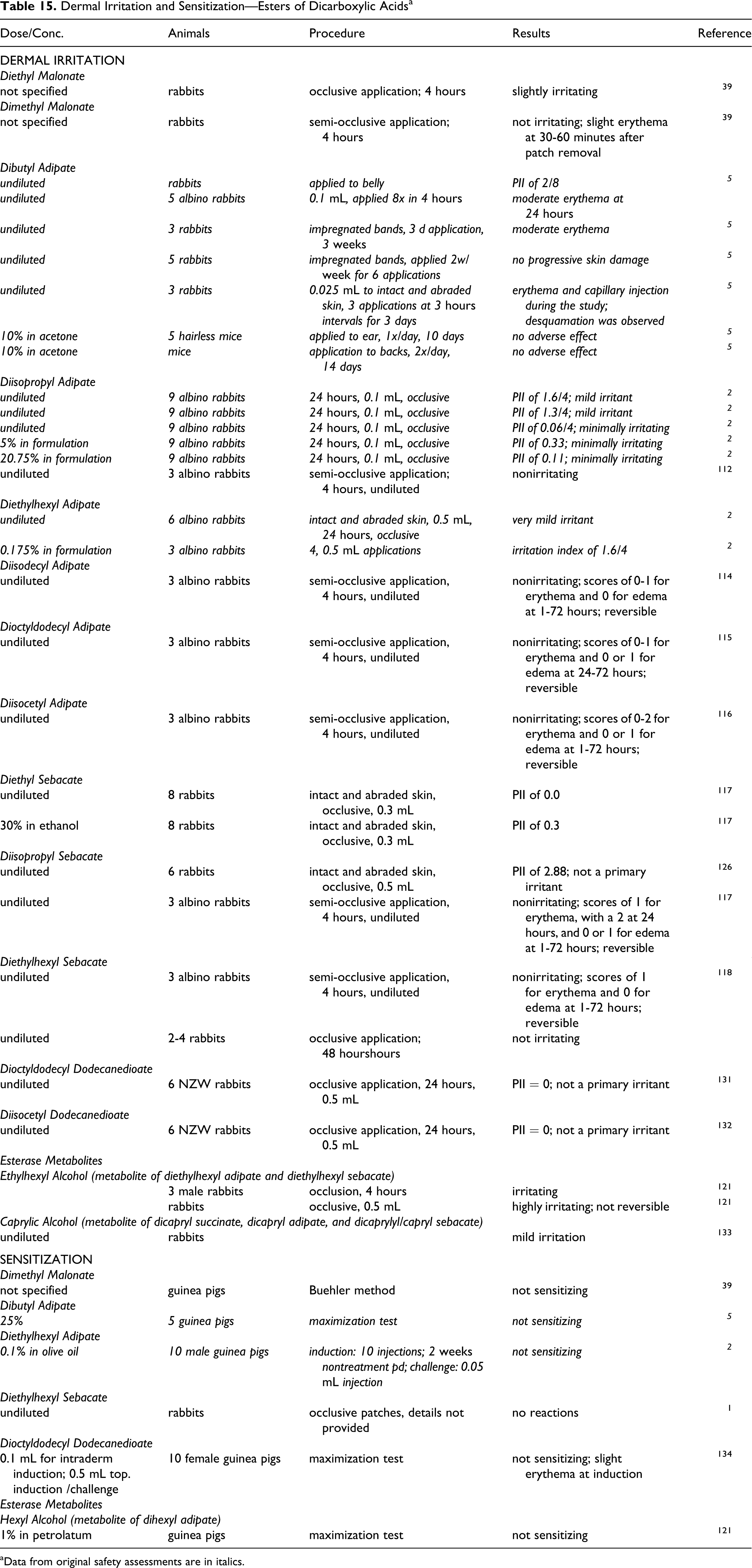
aData from original safety assessments are in italics.
Diethyl malonate
The ocular irritation potential of diethyl malonate was evaluated using rabbits, number and gender not specified. 39 A volume of 0.1 mL was instilled into the conjunctival sac of 1 eye, which was not rinsed, and the contralateral eye was untreated and served as the negative control. Diethyl malonate produced slight to moderate irritation.
In a similar study, undiluted dimethyl malonate produced slight to moderate irritation in rabbit eyes. 39 All signs of irritation were cleared by day 8.
Dibutyl Adipate Nonhuman
Undiluted dibutyl adipate was minimally irritating to the eyes of rabbits, and 0.1% in olive oil was nonirritating. 5
Human
Dibutyl adipate, 0.1% in paraffin oil, was not an ocular irritant in 2 participants. 5
Diisopropyl Adipate
The ocular irritation potential of 2 lots of undiluted diisopropyl adipate was evaluated using rabbits. One caused negligible irritation, while the other was nonirritating. A formulation containing 0.7% diisopropyl adipate produced some corneal stippling in rabbit eyes, while a formulation containing 5.0% and 1 containing 20.75% were nonirritating to rabbit eyes. 2
The ocular irritation of undiluted diisopropyl adipate was evaluated using 3 albino rabbits. 112 A volume of 0.1 mL was instilled into the conjunctival sac of 1 eye, which was not rinsed. The contralateral eye was untreated and served as the negative control. Diisopropyl adipate was not irritating.
Diethylhexyl adipate
Undiluted diethylhexyl adipate was nonirritating to rabbit eyes and a formulation containing 0.0175% was, at most, a mild transient irritant. 2
Diisopropyl Sebacate
A primary ocular irritation study was performed using 6 New Zealand white rabbits to determine the ocular irritation potential of diisopropyl sebacate. 126 A volume of 0.1 mL was instilled into 1 eye of each animal, which was not rinsed, and the contralateral eye of each animal served as the control. The average Draize scores were 2.0 at 24 and 48 hours, 0.3 at 72 hours, and 0.0 at 4 days. Diisopropyl sebacate was a minimal ocular irritant.
Diethylhexyl sebacate
The ocular irritation of a cream containing 1.2% diethylhexyl sebacate was evaluated using the in vitro EpiOcular MTT viability assay. 127 The tissue samples were exposed to undiluted test material for 64 minutes, 256 minutes, or 1200 minutes. Following treatment, the viability of those tissues were calculated. The ET50 (time for tissue viability to be reduced by 50%) was 484.9 minutes, and diethylhexyl sebacate was considered to be nonirritating.
Dioctyldodecyl dodecanedioate
The primary eye irritation of dioctyldodecyl dodecanedioate was evaluated using 6 albino rabbits. 119 A volume of 0.1 mL was instilled into 1 eye of each animal, which was not rinsed. and the contralateral eye served as a negative control. They eyes were evaluated at 24, 48, and 72 hours. At 24 hours, the maximum mean total score (MMTS) was 0.00, and dioctyldodecyl dodecanedioate was considered not irritating.
Diisocetyl dodecanedioate
The primary eye irritation of diisocetyl dodecanedioate was evaluated using the procedure described above. 128 The MMTS was 0.00, and diisocetyl dodecanedioate was considered not irritating to the eyes of rabbits.
Comedogenicity
Dibutyl adipate
Dibutyl adipate, 10% to 100% (vehicle not stated), was not comedogenic in clinical testing. 5
Dermal Irritation/Sensitization
Nonhuman dermal irritation and sensitization data on esters of dicarboxylic acids and esterase metabolites are presented in Table 15. Human dermal irritation and sensitization data on esters of dicarboxylic acids and esterase metabolites are presented in Table 16.
Clinical Dermal Irritation and Sensitization—Esters of Dicarboxylic Acids a
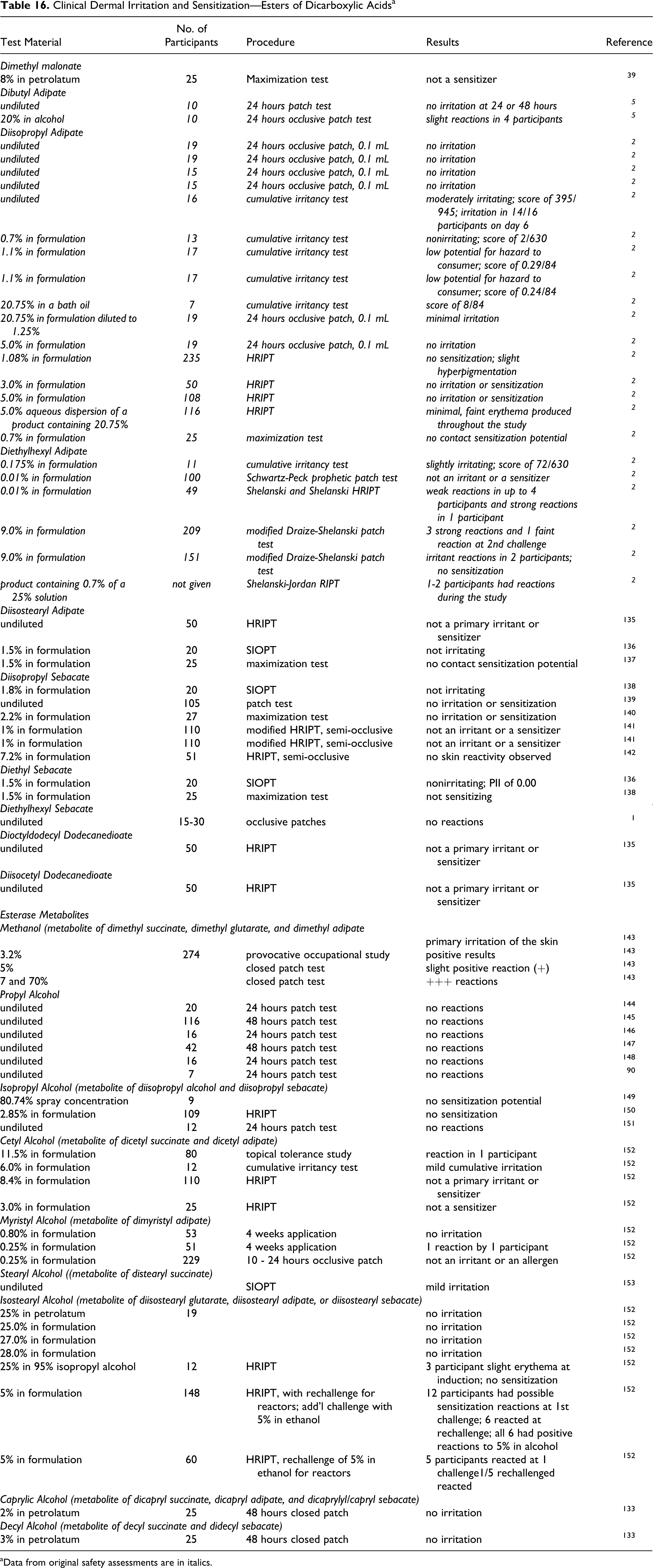
aData from original safety assessments are in italics.
Genotoxicity Studies—Esters of Dicarboxylic Acids a
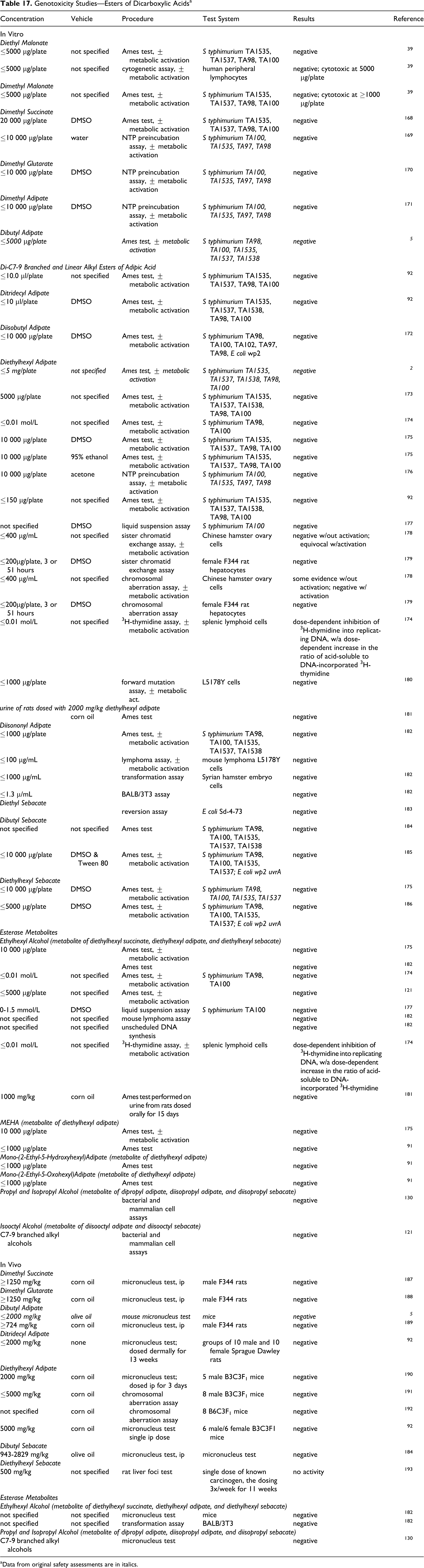
aData from original safety assessments are in italics.
Dimethyl Malonate Nonhuman
Dimethyl malonate was applied undiluted to rabbit skin for 4 hours under a semi-occlusive patch. 39 Slight erythema was observed only at 30 to 60 minutes after patch removal, and dimethyl malonate was considered nonirritating to rabbit skin.
Dimethyl malonate was not a sensitizer in a Buehler guinea pig sensitization test according to OECD TG 406. 39
Human
The sensitization potential of 8% dimethyl malonate in petrolatum was evaluated in a maximization test using 25 participants. 39 Dimethyl malonate was not a sensitizer.
Diethyl Malonate
The dermal irritation potential of diethyl malonate was evaluated using a 24 hours occlusive application. 39 Diethyl malonate was slightly irritating to rabbit skin.
Dibutyl Adipate Nonhuman
Application of undiluted butyl adipate to rabbit skin resulted in a primary irritation score of 2/8. Undiluted dibutyl adipate caused moderate erythema in rabbits following repeated dermal exposure. However, material impregnated with dibutyl adipate was not irritating to the skin of rabbits. Application of dibutyl adipate at 10% in acetone produced no observable adverse effect when applied to rabbit ears, and no dermal reaction was observed following twice daily application for 14 days to the backs of hairless mice. Two perfume formulations containing 1.1% diisopropyl adipate were not primary dermal irritants using rabbits 5
Dibutyl adipate was not a dermal sensitizer in guinea pigs when tested at 25% in a maximization test. 5
Human
Undiluted dibutyl adipate was not irritating in a 24-hr clinical patch test with 10 participants. Slight reactions (not defined) were reported for 4 of 18 participants in a 24-hour patch test with dibutyl adipate, 20% in alcohol.
Diisopropyl Adipate Nonhuman
Draize tests of undiluted diisopropyl adipate resulted in, at most, mild irritation of rabbit skin. In Draize tests with formulations containing 5.0% or 20.75% diisopropyl adipate, minimal irritation was reported with both formulations. 2
Human
The dermal irritation and sensitization of diisopropyl adipate was evaluated in a number of studies. Undiluted diisopropyl adipate produced no irritation in 4 hours patch tests, but was moderately irritating in a 21-day cumulative irritancy test. Formulations containing 0.26% to 20.75% diisopropyl adipate caused minimal to mild irritation, but no sensitization.
Diethylhexyl Adipate Nonhuman
Undiluted diethylhexyl adipate was a very mild irritant when applied under occlusion to intact and abraded rabbit skin. A formulation containing 0.175% diethylhexyl adipate had an irritation index of 1.6/4. 2
Diethylhexyl adipate, 0.1%, was not a sensitizer in a maximization study using guinea pigs. 2
Human
The dermal irritation and sensitization of diethylhexyl adipate was evaluated in a number of studies with formulations containing 0.01% to 9% diethylhexyl adipate. Mild reactions were seen with a formulations containing 0.01%. Using a formulation containing 0.7%, on participant reacted strongly following the second challenge, with erythema and papules observed. Strong reactions were seen for 3 participants in a patch test of a formulation containing 9.0% diethylhexyl adipate. 2
Diisodecyl Adipate, Dioctyldodecyl Adipate, Diisocetyl Adipate
The dermal irritation potential of diisodecyl adipate, dioctyldodecyl adipate and diisocetyl adipate was determined using 3 albino rabbits. 114 –116 Undiluted test material was applied to the skin for 4 hours under a semi-occlusive patch. The erythema scores for each of the 3 materials were 0 to 1 during 1 to 72 hours, and the edema scores were 0. Diisodecyl adipate, dioctyldodecyl adipate and diisocetyl adipate were considered nonirritating to rabbit skin.
Diisostearyl Adipate
A human repeat insult patch test (HRIPT) using 50 participants was used to evaluate the irritation and sensitization potential of diisostearyl adipate. 135 Two-tenths mL was applied neat to the back of each participant under an occlusive patch for 24 hours, after which time the participant removed the patch. This procedure was performed 3 times per week for 3 weeks, for a total of 9 induction patches. Following a 10 to 14 day nontreatment period, a 24-hour challenge patch was applied to a previously untreated site, and reactions were scored at 24 and 48 hours. No adverse reactions were observed, and diisostearyl adipate was not a primary irritant or a sensitizer.
Diethyl Sebacate Nonhuman
Undiluted diethyl sebacate and 30% diethyl sebacate in ethanol were tested on 8 male Japanese White strain rabbits (gender not specified). 117 The flank of the animals was clipped free of hair 1 day prior to application of test substance. The skin of 4 animals was abraded. The test substance, 0.3 mL, was applied occlusively to the back of all animals for 24 hours. The skin reactions were evaluated at 24 hours and 72 hours. The primary irritation score was 0.0 (none to weak irritant) in undiluted diethyl sebacate and 0.3 (none to weak irritant) in 30% diethyl sebacate. These results suggest that 100% diethyl sebacate has no primary skin irritation under these test conditions.
Human
A single insult occlusive patch test (SIOPT) was performed using 20 participants to determine the irritation potential of a body cream containing 1.5% diethyl sebacate. 136 The test patch was applied for 24 hours. The PII was 0.00, and the body cream containing 1.5% diethyl sebacate was nonirritating.
The sensitization potential of a body cream containing 1.5% diethyl sebacate was evaluated in a maximization study. 137 During induction, 0.05 mL of 0.25% aqueous sodium lauryl sulfate (SLS) was applied under an occlusive patch for 24 hours. At that time, the patch was removed and 0.05 mL of the test material was applied to the same site under an occlusive patch for 48 to 72 hours. If no irritation was present at the test site upon patch removal, an occlusive patch with 0.25% aqueous SLS was applied for 24 hours, followed by a patch of the test material. This sequence was used for 5 induction patches. If irritation developed during induction, the SLS patch was eliminated. After a 10-day nontreatment period, a challenge was performed at a previously untreated site. The challenge site was pretreated with 0.05 mL of 5.0% aqueous SLS under an occlusive patch for 1 hour, followed by an occlusive patch of the test material for 48 hours. Twenty-five participants completed the study. No reactions were seen at challenge, and a body cream containing 1.5% diethyl sebacate did not have contact-sensitizing potential.
A number of investigators have reported cases of allergic contact dermatitis in response to diethyl sebacate-containing products, and have demonstrated diethyl sebacate to be the substance, or 1 of several substances in the products, eliciting the dermatitis. 31,154 –158
Diisopropyl Sebacate Nonhuman
A primary dermal irritation study on diisopropyl sebacate was performed using 6 New Zealand white rabbits. 126 A dermal application of 0.5 mL of undiluted test material was applied to an abraded and an intact site on each animal. The test sites were occluded for 24 hours and observed individually for erythema, edema, and other effects 24 and 72 hours after application. Mean scores from the 24 and 72 hours reading were averaged to give a primary irritation index (PII) of 2.88. Diisopropyl sebacate was not considered a primary dermal irritant.
The dermal irritation potential of diisopropyl sebacate was determined using 3 albino rabbits. 117 Undiluted test material was applied to the skin for 4 hours under a semi-occlusive patch. The erythema scores were 1 during 1 to 72 hours, and the edema scores were 0 to 1. Diisopropyl sebacate was considered nonirritating to rabbit skin.
Human
An SIOPT was performed using 20 participants to determine the irritation potential of a foundation containing 1.8% diisopropyl sebacate. 139 The patch was applied for 24 hour. The foundation containing 1.8% diisopropyl sebacate was not irritating.
The irritation and sensitization potential of diisopropyl sebacate was evaluated in a patch test that consisted of four 24-hour applications of diisopropyl sebacate as supplied (approximately 100%) during weeks 1, 2, 3, and 6 on a 2 cm x2 cm area of skin on the right upper arm of each participant. 139 Examinations were performed immediately after patch removal. The induction phase was performed during weeks 1 to 4 using 107 participants. No clinically significant effects were detected on any of the participants during this phase. During week 6, the challenge phase was conducted on 105 participants. No clinically significant effects were noted in any of the participants during this phase. Diisopropyl sebacate was not observed to have any significant skin-irritating or sensitizing activity under the conditions of this study.
A maximization assay was performed, using a modified protocol of the maximization assay procedure described earlier, to determine the contact-sensitization potential of a foundation containing 2.2% diisopropyl sebacate. 140 In this study, the test material was allowed to volatilize for 30 minutes before the occlusive patch was applied. Twenty-five participants completed the study. No reactions were seen at challenge, and a foundation containing 2.2% diisopropyl sebacate did not have contact-sensitizing potential.
Two heat protection hair spray products containing 1% diisopropyl sebacate were tested using a modified Draize HRIPT procedure to determine the potential of those products to induce irritation and contact sensitization. 141 The products were tested neat and allowed to volatilize prior to patch application. Samples were patched under semi-occlusive conditions. Approximately 0.2ml was used in each patch. One hundred ten participants completed the study. Generally transient, barely perceptible (0.5-level) to mild (1-level) patch test responses on 22 test participants for 1 formulation and only barely perceptible (0.5-level) patch test response on 15 test participants with the other formulation during the induction and/or challenge phases of the study were reported. Both products were considered to be nonirritating and nonsensitizing.
A heat protection hair spray product containing 7.2% diisopropyl sebacate was tested using an HRIPT to determine the potential of this product to induce irritation and contact sensitization. 142 The product was tested neat under semi-occlusive conditions. Approximately 0.2 mL sample was used in each patch. Fifty-one participants completed the study. No skin reactivity was observed in any of the test participants during the course of the study.
Two case studies were reported of allergic reactions to lotion containing diisopropyl sebacate.159,160
Diethylhexyl Sebacate Nonhuman
The dermal irritation potential of diethylhexyl sebacate was evaluated using the same procedure. 118 The erythema scores were 1 during 1 to 72 hours, and the edema scores were 0. Diethylhexyl sebacate was considered nonirritating to rabbit skin.
Patch tests with diethylhexyl sebacate (neat; 48-hr occluded) did not irritate the skin of 2 to 4 rabbits. 1 It was also reported that diethylhexyl sebacate was nonirritating to the skin of guinea pigs. No further study details were provided.
A limited attempt was made to sensitize a group of 2 to 4 rabbits by applying diethylhexyl sebacate using occlusive patches. 1 No reactions were seen in an occlusive challenge with the undiluted test article 2 weeks later. Details were not provided.
Human
Diethylhexyl sebacate was applied neat using occlusive patches to the skin of 15 to 30 participants (sex not specified) for 48-hour. 1 No local reactions were observed in the challenge phase (48-hour covered contact with neat liquid) that was carried out 2 weeks later, presumably due to limited induction.
In a case study where 1 patient was sensitized to other sebacate esters, a patch test with diethylhexyl sebacate did not elicit a response. 159
Dioctyldodecyl Dodecanedioate Nonhuman
A maximization test was performed to evaluate the sensitization potential of dioctyldodecyl dodecanedioate. 134 Ten female guinea pigs were used. The dose used at intradermal injection was 0.1 mL, and 0.5 mL was used for the topical challenge. Slight erythema was observed at induction, but a sensitization reaction was not observed.
Human
Dioctyldodecyl Dodecanedioate
An HRIPT with 50 participants was performed to evaluate the irritation and sensitization potential of dioctyldodecyl dodecanedioate. 135 Two-tenths milliliter of the test material, neat, was applied to the back of each participant under an occlusive patch for 24 hours, after which time the participant removed the patch. This procedure was performed 3 times per week for 3 weeks, for a total of 9 induction patches. Following a 10- to 14-day nontreatment period, a 24 hours challenge patch was applied to a previously untreated site, and reactions were scored at 24 and 48 hours. No adverse reactions were observed, and dioctyldodecyl dodecanedioate was not a primary irritant or a sensitizer.
Diisocetyl dodecanedioate
An HRIPT with 50 participants was performed as described above to evaluate the irritation and sensitization potential of diisocetyl dodecanedioate. 135 No adverse reactions were observed, and diisocetyl dodecanedioate was not a primary irritant or a sensitizer.
Phototoxicity
Dibutyl adipate
Dibutyl adipate, as a 10% dilution in liquid paraffin, was not phototoxic in a clinical phototoxicity study using 30 participants. 5
Diethylhexyl adipate
In a photopatch test on 9.0% diethylhexyl adipate using 25 participants, no phototoxic or photoallergic reactions were observed.
Diisopropyl Adipate
Nonhuman
Two perfume formulations containing 1.1% diisopropyl adipate were not phototoxic in rabbits. 2
Human
In photopatch test studies using 49 to 98 participants, formulations containing 0.7% to 17.0% diisopropyl adipate were not phototoxic, primary irritants, or sensitizers.
Mucous Membrane Irritation
Diethylhexyl adipate
A product containing 0.175% diethylhexyl adipate did not produce irritation of the genital mucosa in rabbits. 2
Reproductive and Developmental Toxicity
Dimethyl Malonate
Groups of 10 male and 10 female Wistar rats were dosed with 0, 100, 300, or 1000 mg/kg bw dimethyl malonate orally, by gavage. 39 Males were dosed for 2 weeks prior to mating, during mating, and 2 weeks after mating, for a total of 39 doses. Females were dosed 2 weeks prior to mating, during mating, and through day 4 of lactation. A recovery group of 5 male and 5 female high-dose animals were observed for 14 days after the termination of dosing. Microscopically, the incidence of treatment-related hepatocellular hypertrophy of the liver was observed for males and females given 1000 mg/kg bw dimethyl malonate. This effect was not observed in the recovery animals or in the other test groups. No other significant toxicological effects were observed. Performance in a functional observation battery was similar for test and control animals. There was no effect on fertility. In the 100 mg/kg bw group, a statistically significant decrease in the number of live pups was due to an increase in post-implantation loss. This effect was not considered treatment related, and no developmental toxicity was reported. The NOAEL was 300 mg/kg bw for repeated doses and maternal toxicity and 1000 mg/kg bw for fertility and developmental toxicity.
Dimethyl Adipate
Groups of 5 gravid Sprague Dawley rats were dosed ip with 0.0603 to 0.6028 mL/kg dimethyl adipate (1/30, 1/10, 1/5, and 1/3 of the ip LD50 value) on days 5, 10, and 15 of gestation. 124 A pooled volume control consisted of animals dosed with 10 mL/kg distilled water, saline, or cottonseed oil. A positive control group was not used. All animals were killed and examined on day 20 of gestation. The mean fetal weights and the numbers of live fetuses were not statistically significantly different between treated and blunt-needle control groups. Resorptions in animals dosed with 0.1809 mL/kg were statistically significantly increased when compared to the pooled controls, but not the blunt-needle controls. Gross and skeletal abnormalities, but not visceral, were statistically significantly increased in fetuses of the 0.3617 and 0.6028 mL/kg groups. The NOEL was 0.0603 mL/kg dimethyl adipate.
Diethyl Adipate
Following the same procedure described above, rats were dosed ip with 0.0837 to 0.8373 mL/kg diethyl adipate. 124 The mean fetal weight and the number of live fetuses were not statistically significantly different between treated and blunt-needle control groups, and the number of resorptions was similar between treated animals and both the blunt needle and pooled controls. There were no differences in the incidences of gross, skeletal, or visceral abnormalities in fetuses of the treated groups compared to pooled controls.
Dipropyl Adipate
Following the same procedure described above, rats were dosed ip with 0.1262 to 1.2619 mL/kg dipropyl adipate. 124 The numbers of live and dead fetuses were not statistically significantly different between treated and blunt-needle control groups, but there was a statistically significant decrease in the mean fetal weight of the 0.7572 mL/kg group. Resorptions in animals dosed with 1.2619 mL/kg were statistically significantly increased when compared to the pooled controls, but not the blunt-needle controls. Gross abnormalities, but not skeletal or visceral, were statistically significantly increased in fetuses of the 1.2619 mL/kg group. The NOEL was 0.1262 mL/kg dipropyl adipate.
Dibutyl Adipate
Groups of 5 gravid Sprague Dawley rats were dosed ip with 0.1748 to 1.7480 mL/kg dibutyl adipate on days 5, 10, and 15 of gestation. The incidence of gross abnormalities was only statistically significantly increased in the high-dose group when compared to pooled controls. 5
Dibutyl adipate was evaluated in a study Sprague-Dawley rats. 110 Groups of 13 male and 13 female rats were dosed with 0, 100, 300, or 1000 mg/kg bw dibutyl adipate orally, by gavage, for 14 days prior to mating through parturition; males were dosed for a total of 42 days and female dams were dosed until day 3 of lactation. The test article had no effect on fertility. Body weight gains of males of the 1000 mg/kg bw group were slightly decreased. Kidney weights of the high-dose males and females sere increased compared to controls. No gross or microscopic effects were noted at necropsy, and the internal genitalia were normal. Dosing with dibutyl adipate did not produce any reproductive effects. The only effect on the offspring was a decrease in pup weight on post-natal days 0 and 4 and in viability on post-natal day 4 in the 1000 mg/kg group. The NOEL for parental and offspring toxicity was 300 mg/kg bw/d. The reproductive NOEL was 1000 mg/kg bw/d.
Di-C7-9 Branched and Linear Alkyl Esters of Adipic Acid
Groups of 24 gravid Sprague Dawley rats were dosed orally by gavage with 0, 1000, 4000, or 7000 mg/kg bw/d di-C7-9 branched and linear alkyl esters of adipic acid on days 6 to 19 of gestation, and all animals were killed and examined on day 20. 92 All dams survived until study termination. Body weights were significantly decreased for dams of the 7000 mg/kg bw group. Weights of male and female fetuses of the 7000 mg/kg bw group were slightly, but not statistically significantly, decreased compared to the other groups. A greater incidence of rudimentary structures was observed for high-dose fetuses as compared to the other groups in this study, but the incidence was within the range of historical controls. There was no evidence of developmental toxicity at any dose tested.
Ditridecyl Adipate
Groups of 15 mated female Sprague-Dawley rats were given doses of 0, 800, and 2000 mg/kg bw of ditridecyl adipate applied dermally without occlusion on days 0 to 19 of gestation, and the dams were killed on day 20. 92 . Mild skin irritation consisting of erythema and flaking were observed at the test sites of the treated animals. No maternal mortality was reported. Weight gains were statistically significantly decreased for the 2000 mg/kg bw group during days 0 to 3 and 16 to 20 of gestation. Weight gains were statistically significantly decreased in the 800 mg/kg bw group during days 0 to 3 of gestation. No differences in skeletal anomalies were observed, but there were some differences in visceral anomalies, including increased incidence of levocardia at 2000 mg/kg bw. These anomalies were not considered treatment-related. The NOAEL for maternal toxicity was 2000 mg/kg bw/d, and for developmental and reproductive effects it was 800 mg/kg bw/d.
Groups of 25 mated female rats were dosed dermally with 0 and 2000 mg/kg bw ditridecyl adipate following the same study protocol as above. Again, there were no signs of maternal toxicity. No developmental toxicity was reported, and there were no visceral anomalies or levocardia.
Tridecyl adipate, 2000 mg/kg bw, was applied, unoccluded, to groups of 10 male Sprague-Dawley rats, 5 days/week for 13 weeks, and the effect on sperm morphology was evaluated. 92 (The ‘Subchronic Dermal Toxicity’ study was described earlier.) No differences in sperm morphology were observed between control and test animals.
Diisobutyl Adipate
Diisobutyl adipate was evaluated following the procedure described earlier in the ip study. 124 These rats were dosed ip with 0.1983 to 1.9833 mL/kg diisobutyl adipate. The numbers of live and dead fetuses were not statistically significantly different between treated and blunt-needle control groups, but there was a statistically significant decrease in the mean fetal weight of the 1.1900 and 0.9833 mL/kg dose groups. The number of resorptions was similar between treated animals and both the blunt needle and pooled controls. Gross abnormalities, but not skeletal or visceral, were statistically significantly increased in fetuses of the 0.5950 and 1.9833 mL/kg groups.
Diethylhexyl Adipate
Groups of 10 male Swiss mice were dosed ip with ≤9.3 g/kg diethylhexyl adipate and then mated with undosed females. A reduction in the number of gravid females was considered an anti-fertility effect, and the dominant lethal mutation was determined directly from the dose-dependent increase in the number of early fetal deaths and indirectly from the dose- and time-dependent decrease in implantations. There were no test article-related changes in the incidence of late fetal deaths. It was noted that the experimental design and interpretation have been questioned by some. Diethylhexyl adipate, ≤9.3 g/kg, was administered by ip injection to groups of 5 gravid Sprague Dawley rats on day 5, 10, and 15 of gestation. Resorption rates were similar to controls. A decrease in the mean fetal bw and a significant increase in gross fetal abnormalities at the high dose were observed when compared to pooled control values. However teratogenic effects were not observed when compared to concurrent controls. It was stated that the lack of historical and positive controls affected the validity of the results. 2
Groups of 15 male and 30 female Wistar rats were fed a diet containing 0, 0.03, 0.18, or 1.2% diethylhexyl adipate (calculated as 28, 170, or 1080 mg/kg bw/d) for 10 weeks prior to mating. 92 Dosing was terminated, and the animals were mated. (A different source indicated that dosing continued throughout the study). 161 A reduction in bw gain was reported during gestation for the dams of the 1.2% group. No test article-related effects on fertility were observed. Fetal weight, total litter weight, and litter size were reduced in the 1.2% group, but the number of pups born live, or their survival, was not affected. The NOAEL was 170 mg/kg bw/d and the LOAEL was 1080 mg/kg bw/d.
In another study in which gravid females were fed the same doses as above on days 1 to 22 of gestation, maternal bw and feed consumption were statistically decreased in the 1.2% group. No significant effects on fetal weight or litter size were reported. Animals of the 0.18 and 1.2% groups had slightly increased incidences of minor skeletal abnormalities; this increase was attributed to fetotoxicity. The NOEL for maternal toxicity was 170 mg/kg bw/d. The NOAELs for developmental toxicity and fetotoxicity were 170 and 28 mg/kg bw/d, respectively. The LOAEL was 1080 mg/kg bw day.
A dose-range finding study was performed using groups of 8 gravid Wistar rats that were dosed by gavage with 2 mL/kg of 0, 800, or 1200 mg/d diethylhexyl adipate, in peanut oil, from day 7 of gestation until day 17 after parturition. 162 No signs of toxicity were reported in any of the groups. In the 800 mg/kg bw group, the only statistically significant observation made was decreased bws of male and female pups on day 3. In the 1200 mg/kg bw group, statistically significant effects were observed for a number of parameters, including decreased maternal weight gain during days 7 to 21 of gestation, increased length of gestation (by 1 day), decreased pup bws at birth and day 3, and an increase in perinatal loss per litter. (Perinatal loss was 42% in the 1200 mg/kg bw groups, as compared to 4.6% in controls.)
Based on the results of the dose-range finding study, groups of 20 gravid Wistar rats were dosed with 2 mL/kg of 0, 200, 400, or 800 mg/kg bw diethylhexyl adipate, in peanut oil, from day 7 of gestation until post-natal day 17. At postnatal day 21, all dams and pups were killed, with the exception that 1 male and 1 female pup per litter was kept for further evaluation. No signs of toxicity were reported in any of the groups. No significant effects were observed in the 200 mg/kg bw group. In the 400 mg/kg bw dose groups, the number of postnatal deaths per number of pups was statistically significant increased. In the 800 mg/kg bw group, statistically significant effects were observed for a number of parameters, including increased length of gestation (by 1 day), decreased pup bws at birth and days 3 and 13, increased mean number of postnatal deaths, and an increase in postnatal death per number of pups. The percentage of perinatal loss per litter was twice as high in the 400 and 800 mg/kg bw groups (23%) as compared to controls (11%), but the change was not statistically significant. Testicular testosterone levels were unaffected in any of the pups that were killed on postnatal day 21 or the adult male offspring, and serum luteinizing hormone and prolactin levels were similar to controls. None of the sperm parameters that were evaluated were affected by dosing. The only statistically significant effects, noted in the 800 mg/kg bw group, were increased relative liver to bws in male pups on day 21 and increased bws and decreased adrenal weights in adult male offspring. Diethylhexyl adipate did not produce any antiandrogenic effects in the study. Fetal steroidogenesis was not evaluated. NOAEL was 200 mg/kg bw.
Groups of 10 female Crl:CD(SD) rats were dosed with 5 mL/kg, by gavage, of 0, 200, 1000, or 2000 mg/kg bw diethylhexyl adipate in corn oil for 2 weeks prior to mating with undosed males, throughout mating, and until day 7 of gestation. 125 The dams were killed on day 14 of gestation. All animals survived until study termination. Body weights and body weight gains were significantly decreased in the 2000 mg/kg bw dose group prior to mating. Staining around the perineum was observed in the 1000 and 2000 mg/kg bw dose groups. No statistically significant differences were observed for the 200 mg/kg bw group compared to controls. The mean estrous cycle length was statistically significantly increased in the 1000 and 2000 mg/kg bw groups, and the post-implantation loss rate was also statistically significantly increased in these groups. Additionally, in the 2000 mg/kg bw group, there was a significant decrease in implantation rate, and the number of live embryos was statistically significantly decreased and the pre-implantation loss rate statistically significantly increased. The researchers stated that the effects observed in this fertility study, in conjunction with the ovarian effects described earlier in the repeated dose study, suggest that diethylhexyl adipate disturbed ovulation. This correlated with the effect on estrous cycle length.
The testicular toxicity of diethylhexyl adipate was examined using male F344 rats. 163 Groups of 6 rats were fed a diet containing 6000 or 25 000 ppm diethylhexyl adipate for 4 weeks, and the controls were given untreated feed. Some groups were dosed ip with 200 mg/kg bw thioacetamide, 3x/week for 4 weeks, and prior to dosing with diethylhexyl adipate to evaluate whether liver disease enhanced testicular effects. (There was a 1-week rest period prior to dosing with diethylhexyl adipate.) The final bws of animals given 25 000 ppm diethylhexyl adipate, with and without prior administration of thioacetamide, were statistically significantly decreased compared to their respective controls. The relative liver to bws of these animals were statistically significantly increased. No significant effect on the relative weights of the testes or epididymis was seen for any of the test groups. Diethylhexyl adipate did not have any testicular toxic effects, with or without the induction of hepatic damage.
Diisononyl Adipate
In a subchronic dietary study described earlier, groups of male and female Beagle dogs were fed 0, 0.3, 1.0, or 3.0% (weeks 1-8) and 6.0% (weeks 9-13) diisononyl adipate for 13 weeks. 92 Reproductive tissues were evaluated. No significant findings were reported for the 0.3% and 1.0% groups. In the high-dose group, testes weight was decreased. At microscopic examination, it was found that the epididymal ducts were devoid of spermatozoa, the seminiferous tubules were composed of Sertoli cells and spermatogonia, spermatocytes and spermatids were not evident, and there was almost total aspermatogenesis. Ovaries were not weighed at necropsy. There were no gross or microscopic changes in the ovaries of the high-dose group compared to controls.
Dibutyl Sebacate
A test group of 20 male and 20 female Sprague-Dawley rats was fed a diet containing 6.25% dibutyl sebacate for 10 weeks, while a control group of 12 male and 12 female rats were fed the basal diet, and then animals of each group were then mated. 122 The dams were allowed to deliver their litters, and at weaning, 24 male and 24 female offspring were randomly chosen, fed the test diet for 21 days, and then killed for necropsy. The study results indicated that ingestion of a diet containing 6.25% dibutyl sebacate had no adverse effect on fertility, litter size, or survival of offspring. Growth was decreased during the pre-weaning and post-weaning periods. However, no gross pathological changes were found among young rats killed at the end of the 21-day post-weaning period.
Diethylhexyl Sebacate
Reproduction, suckling and growth were normal in a 4-generation study of rats fed a diet containing 200 ppm diethylhexyl sebacate (~10 mg/kg bw/d). 1 No reproductive or developmental toxicity was observed.
Dimethyl Glutarate/Dimethyl Succinate/Dimethyl Adipate Mixture
The developmental toxicity produced by the inhalation of dibasic esters (mixture of 65.1% dimethyl glutarate, 17.8% dimethyl succinate, and 16.8% dimethyl adipate) was evaluated in rats. 164 Groups of 24 gravid Crl:CD rats were exposed for 6 hours/d to 0, 0.16, 0.4, or 1.0 mg/L dibasic esters, by whole body inhalation, on days 7 to 16 of gestation. The aerosol particle size in the 1.0 mg/L chamber was 5.3 to 5.4 µm, with 72% to 74% of the aerosol <10 µm. The animals were killed on day 21 of gestation. All animals survived until study termination. Body weight gains were statistically significantly decreased in the 0.4 and 1.0 mg/L groups. Feed consumption by these groups was reduced during the first 6 exposures; statistical significance was not given. Statistically significant differences in absolute and relative liver to bws were not observed, but there was a significant trend of decreased absolute, but not relative, liver weights. The only significant clinical signs observed were perinasal staining and wet fur of rats in the 1.0 mg/L group. Reproductive and developmental effects were not observed, and the dibasic esters mixture was not a developmental toxicant in rats following inhalation of ≤1.0 mg/L.
Groups of 20 Crl:CD(SD)BR rats/gender were exposed for 6 hours/d, 5 days/week, to 0, 0.16, 0.40, or 1.0 mg/L dibasic esters by whole body inhalation for 14 weeks prior to mating, and then 7 days/week for 8 weeks of mating, gestation, and lactation. 165 The mean aerosol particle size in the 1.0 mg/L chamber was 6.2 µm, with 69% of the aerosol <10 µm. Exposure was discontinued from day 19 of gestation through day 3 post-partum. All parental rats and 10 pups/gender were killed and necropsied on day 21 post-partum. The remaining pups were not necropsied. Maternal bws in the 0.40 mg/L group were decreased during the last week of the study, while bws of male and female rats of the 1.0 mg/L group were slightly decreased from week 7 on. Relative liver to bws were slightly, but not significantly, decreased in the 0.4 and 1.0 mg/L groups. Other differences in organ weights were not considered dose-related. With the exception of a statistically significant decrease in pup bws at birth and day 21, no reproductive or developmental effects were observed. The only microscopic findings were squamous metaplasia in the olfactory epithelium of all treated parental rats. This effect was minimal in the 0.16 mg/L group and mild to moderate in the 0.4 and 1.0 mg/mL groups. The NOEL for reproductive parameters was 1.0 mg/L.
Endocrine Disruption
Diethylhexyl adipate
A 28-day repeated-dose toxicity study was performed based on the Enhanced OEDC Test Guideline no. 407 (enhanced TG 407) to determine whether diethylhexyl adipate has endocrine-mediated activities. 166 Groups of 10 male and 10 female Crj:CD (SD) rats were dosed orally by gavage with 0, 40, 200, or 1000 mg/kg bw diethylhexyl adipate in corn oil, at a volume of 10 mL/kg, for a minimum of 28 days. In addition to clinical observations, a functional observation battery was performed during week 4, estrous cycling was assessed from day 22, hormone analysis was measured at the end of the test period, and sperm morphology and sperm count were examined. Male animals were killed and necropsied on day 29, while females were killed and necropsied on days 30 to 34 when in diestrous. Signs of toxicity were not observed, and no clinical chemistry or hematological findings were recorded. Hormonal and spermatological analyses were normal. Statistically significant increases were seen in relative kidney to bws in males of the 200 and 1000 mg/kg bw groups, relative liver to bws of males in the 1000 mg/kg bw group, and in relative liver, kidney, and adrenal to bws in females of the 1000 mg/kg bw group. Microscopically, increased eosinophilic bodies and hyaline droplets were seen in the kidneys of male rats of the 1000 mg/kg bw group. Ovarian follicle atresia was observed in 4 females of the 1000 mg/kg bw group, accompanied by a prolonged estrous cycle in 2 of these rats. A change in the estrous cycle is an important endpoint for determination of endocrine-mediated effects in the enhanced TG 407 assay. The researchers stated that this effect, in conjunction with the microscopic findings, appears to be related to endocrine-mediated effects of diethylhexyl adipate. However, it was also stated that these findings may be attributable to the disturbance of ovarian function according to the hypothalamic-pituitary-gonad axis. The changes in relative kidney to bws and liver to bws, and accompanying histopathological changes, were considered toxic effects, and these findings indicated that the NOEL was 40 mg/kg bw/d.
The effect diethylhexyl adipate. at concentrations of 1 × 10−10 to 5 × 10−5mol/L, on estrogen receptor and thyroid hormone (TH) functions was also examined. 167 The TH-like activity was assessed using the rat pituitary tumor cell line GH3 expressing intracellular TH and estrogen receptors and responding to physiological concentration of TH by proliferation. At “low potency”, diethylhexyl adipate stimulated the TH- dependent rat pituitary GH3 cell proliferation in a concentration-dependent manner. (The lowest tested concentration at which a statistically significant effect was detected was 10−5 mol/L.) Cotreatment of GH3 cells with diethylhexyl adipate potentiated the L-3,5,3′-triiodothyronine (T3)-EC50 potentiated the T3-induced GH3 cell proliferation.
Genotoxicity
Details of the genotoxicity studies on esters of dicarboxylic acids and esterase metabolites are described in Table 17.
Diethyl Malonate
Diethyl malonate was not mutagenic in an Ames test or a cytogenetic assay using human peripheral lymphocytes at concentrations ≤5000 μg/plate. 39
Dimethyl Malonate
Dimethyl malonate was not mutagenic in an Ames test at concentrations ≤5000 μg/plate. 39
Dimethyl Succinate
Dimethyl succinate was not mutagenic in an Ames tests with concentrations of ≤20 000 µg/plate 168 or in a preincubation assay with concentrations of ≤10 000 μg/plate. 169
Dimethyl Glutarate
Dimethyl glutarate was not mutagenic in a preincubation assay with concentrations of ≤10 000 μg/plate. 170
Dimethyl Adipate
Dimethyl adipate was not mutagenic in a preincubation assay with concentrations of ≤10 000 μg/plate. 171
Dibutyl Adipate
Dibutyl adipate was mutagenic in an Ames test at concentrations of ≤5000 µg/plate. It was not genotoxic in an in vivo mouse micronucleus assay in which the animals were dosed with ≤2000 mg/kg bw.
Di-C7-9 Branched and Linear Alkyl Esters of Adipic Acid
Di-C7-9 branched and linear alkyl esters of adipic acid were not mutagenic in an Ames test at concentrations of ≤10.0 µl/plate. 92
Ditridecyl Adipate
Ditridecyl adipate was not mutagenic in an Ames test at concentrations of 0 to 10 µL/plate, and it was not clastogenic in an in vivo micronucleus assay using rats dosed dermally with 0, 800, or 2000 mg/kg bw ditridecyl adipate. 92
Diethylhexyl Adipate
Diethylhexyl adipate was not mutagenic in an Ames (concentrations tested were not provided) test.
Diethylhexyl adipate was not mutagenic in a number of genotoxicity studies. In vitro, negative results were reported in Ames tests at concentrations ranging from ≤150 to 10 000 µg/plate,92,173–175 in an NTP preincubation assay, 176 in a liquid suspension assay, 177 and in a forward mutation assay using L5178Y cells at concentrations ≤1000 µg/mL. 180 In an assay for sister chromatid exchanges and chromosomal aberrations using concentrations of ≤200 μg/plate, results were negative, 179 while in another assay with ≤400 μl/plate, results were negative without, but equivocal with, metabolic activation in the sister chromatid exchange assay and there was some evidence of genotoxicity without, but none with, metabolic activation in the chromosomal aberration assay. 178 In a 3 H-thymidine assay, there was a dose-dependent inhibition of 3 H-thymidine incorporation into replicating DNA, with a dose-dependent increase in the ratio of acid-soluble DNA-incorporated 3 H-thymidine. 174 In vivo, results were negative in micronucleus tests92,190 and chromosomal aberration assays.191,192
An Ames test was performed on urine of rats dosed with diethylhexyl adipate to assess whether mutagenic substances occur in the urine following diethylhexyl adipate adminstration. 181 Groups of ≥6 male Sprague-Dawley rats were dosed orally by gavage with 0 or 2000 mg/kg bw diethylhexyl adipate in corn oil for 15 days. Urine was collected daily. The urine was not mutagenic in the Ames test, indicating that diethylhexyl adipate is not converted to mutagenic urinary metabolites. The urine of rats dosed with 1000 mg/kg bw 2-ethylhexanol by gavage for 15 days was also tested in an Ames assay. The urine of these rats also was not mutagenic. Urine from rats that were dosed with a known mutagen gave a positive response in an Ames test.
Diisononyl Adipate
Diisononyl adipate was not mutagenic in an Ames assay at ≤1000 μg/plate, and it was not genotoxic in a mouse lymphoma assay, a transformation assay, or a BALB/3t3 assay at concentrations of ≤100, 1000, or 1.3 μg/mL, respectively. 182
Diethyl Sebacate
Diethyl sebacate was nonmutagenic in an Escherichia coli Sd-4-73 reversion (streptomycin dependence to independence) assay. 183
Dibutyl Sebacate
Dibutyl sebacate, ≤10 000 μg/plate, was not mutagenic in the Ames assay.184,185
Diethylhexyl Sebacate
Diethylhexyl sebacate was not mutagenic in an Ames assay at concentrations of ≤10 000 µg/plate.175,186 In the rat liver foci test, diethylhexyl sebacate demonstrated no evidence of promotion activity when administered orally at 500 mg/kg bw 3x/week for 11 weeks, following a single oral treatment with a known carcinogen. 193
Carcinogenicity
Diethylhexyl Adipate
In an NTP carcinogenicity study, administration of ≤25 000 ppm diethylhexyl adipate to rats in the diet for 103 weeks did not produce carcinogenic effects. However, mice fed the same amount for 103 weeks had dose-related bw reductions and a higher incidence of hepatocellular adenoma and carcinoma than the controls. In another study in which rats were fed ≤2.5% diethylhexyl adipate for 2 years, tumor incidence for the test animals was similar to that of controls. The same researchers found no tumors in dogs fed up to 0.2% diethylhexyl adipate for 1 year. A single 10 mg dose of diethylhexyl adipate given by sc injection was not carcinogenic in mice. In a lifetime study, diethylhexyl adipate caused no skin tumors when 10 mg was applied weekly to the back skin of mice.
Research has shown that other compounds with a 2-ethylhexyl group that have been evaluated for carcinogenicity had some evidence of hepatocarcinogenicity, ranging from very strong to equivocal, in rodents. 194
In an evaluation of the carcinogenic risk of diethylhexyl adipate, the IARC stated that there was limited evidence in experimental animals for the carcinogenicity of diethylhexyl adipate. 17 Therefore, the overall evaluation of diethylhexyl adipate was not classifiable as to its carcinogenicity to humans (Group 3).
Diethylhexyl Sebacate
No evidence of carcinogenicity was observed in an unspecified number of rats fed a diet providing about 10 mg diethylhexyl sebacate/kg/d for up to 19 months. 1 No further study details were provided.
Tumor Promotion
Diethylhexyl adipate
A group of 14 male F344 rats were used to assess the carcinogenic potential of diethylhexyl adipate in a medium-term liver bioassay. 195 The rats were given a single ip dose of diethylnitrosamine, and 2 weeks later they were given 20 000 ppm diethylhexyl adipate in the diet. At week 3, a partial hepatectomy was performed. Positive results for carcinogenic potential were indicated by a significant increase in GST-P positive foci. Diethylhexyl adipate did not have an enhancing effect on the development of GST-P-positive foci.
Risk Assessment
Diethylhexyl adipate
According to the Integrated Risk Information System of the EPA, the weight-of-evidence classification for diethylhexyl adipate was “possible human carcinogen”. 161 The classification was based on an absence of human data and increased liver tumors in female mice. The only genotoxic effect was a positive dominant lethal assay. It was noted that diethylhexyl adipate exhibits structural relationships to other nongenotoxic compounds that are classified as probable and possible carcinogens.
Summary
This safety assessment includes sebacic acid and other alkyl α,ω-dicarboxylic acids, salts, monoesters and diesters, for a total of 56 ingredients. The dicarboxylic acids are terminally functionalized straight alkyl chains characterized by a separation between the acid functional groups of 1 to 10 carbons. The simple alkyl di-esters are the result of the condensation of alkyl dicarboxylic acids and 2 equivalents of alkyl alcohols. These ingredients can be metabolized via hydrolysis back to the parent alcohol, the mono-ester, and the parent dicarboxylic acid. The simple alkyl esters (mono- and di-) of these dicarboxylic acids have straight or branched side chains ranging in length from 1 to 18 carbons. This safety assessment is divided into 2 parts—(1) 12 dicarboxylic acids and their salts and (2) 44 esters of dicarboxylic acids.
A safety assessment of diethylhexyl adipate (called dioctyl adipate at the time of that assessment) and diisopropyl adipate was published in 1984 with the conclusion that these ingredients are safe as used in cosmetics. This conclusion was reaffirmed in 2006. Additionally, dibutyl adipate was previously reviewed in 1996 and the available data were found insufficient to support the safety of dibutyl adipate in cosmetic formulations. When re-reviewed in 2006, additional data were made available to address the needs identified by the CIR Expert Panel, and an amended conclusion was issued stating that dibutyl adipate is safe for use in cosmetic formulations.
While many of the alkyl dicarboxylic acids occur in natural products, commercial production of these acids has historically occurred via alkali pyrolysis of lipids.
A relationship exists between the molecular weight and the log octanol—water partioning coefficient. Physical properties change as chain length increases, and the water solubility of these acids is inversely proportional to their chain length. Odd versus even chain length also plays a role. The alternating effects are believed to be the result of the inability of odd carbon number compounds to assume an in-plane orientation of both carboxyl groups with respect to the hydrocarbon chain. The diesters, in contrast, are much more lipid soluble and more difficult to dissolve in water. The short-chain alkyl mono- and diesters are more soluble in water, less lipophilic, and relatively more volatile than the corresponding longer chain alkyl esters.
The ingredients included in this review would not be expected to have any meaningful UV absorption.
The ingredients in this report function in cosmetics as pH-adjusters, fragrance ingredients, plasticizers, skin-conditioning agents and/or solvents and corrosion inhibitors. The majority of the dicarboxylic acids function in cosmetics as pH adjusters or fragrance ingredients. Six of the 12 dicarboxylic acids and their salts and 24 of the 44 esters included in this safety assessment are reported to be used in cosmetic formulations. For the dicarboxylic acids and their salts, disodium succinate has the greatest number of reported uses, with a total of 45. The acid with the greatest concentration of use is succinic acid, 26%; use at this concentration is in a bath product that will be diluted during use. The greatest leave-on concentration is 0.4%, disodium succinate, with dermal contact exposure. For the esters, diisopropyl adipate has the greatest number of uses, with 70 reported. The concentration of use is greatest for dimethyl glutarate, 15% in a dermal rinse-off product. The ingredients with the greatest leave-on use concentrations, which are all dermal contact exposures, are diethylhexyl adipate, 14%, diisostearyl adipate, 10%, and diisopropyl sebacate, 10%.
Dicarboxylic Acids and Their Salts
Dicarboxylic acids are endogenous metabolic products of the ω-oxidation of monocarboxylic acids when the β-oxidation of free fatty acids is impaired. Under normal physiological conditions, dicarboxylic acids are rapidly β-oxidized, resulting in very low cellular concentrations and practically nondetectable concentrations in the plasma. Oxidation of odd- and even-numbered chains proceeds to different end points; even chains are completely, while odd-number chains are not completely, oxidized.
Unchanged dicarboxylic acid was found in the urine of rats. With oral dosing, approximately 53% to 67% adipic acid, 40% azelaic acid, and 50% dodecanedioate was recovered with the respective acid. With iv dosing, 59% to 71% adipic acid and 35% sebacate was recovered. In humans, 6.76 to 61 adipic acid, and 61% azelaic acid were found in the urine after dosing with the respective acid. With azelaic acid and dodecanedioic acid, radioactivity was found in all tissues, and it decreased after 24 hours in all tissues except adipose tissue. Radioactivity was found in expired carbon dioxide at 24 hours after dosing adipic acid (70%), azelaic acid (14.5%), and disodium sebacate (25%). For rats dosed orally with azelaic, sebacic, undecanedioic, and dodecanedioic acid, 2.5, 2.1, 1.8, and 1.6% of the respective acid was found in the urine unchanged. The amount recovered decreased with increasing chain length. After oral dosing, 60, 17, 5, and 0.1% of azelaic, sebacic, decanedioic, and undecanedioic acids, respectively, were recovered unchanged in the urine. In the plasma of both animals and humans, dicarboxylic acid catabolites that were 2-, 4-, or 6- carbons shorter than the corresponding dicarboxylic acid were found.
Adipic acid did not induce peroxisome proliferation. Dicarboxylic acids did have some cellular effects and inhibited mitochondrial oxidoreductases, reversibly inhibited microsomal NADPH and cytochrome P450 reductase, and competitively inhibited tyrosinase in vitro.
The oral LD50 values of the dicarboxylic acids had a wide range; for example, adipic acid had values in rats ranging from 0.94 g/kg to greater than the highest dose tested (11 g/kg). Most reported values for the acids were >2 g/kg. The reported dermal LD50 values ranged from >6 g/kg dodecanedioic acid to >10 g/kg glutaric acid.
In short-term oral toxicity studies, ≤3000 mg/kg bw/d adipic acid did not produce significant toxicological effects in rats. Signs of toxicity were seen at >3600 mg/kg bw/d. No toxicity was observed with guinea pigs fed 400 to 600 mg/d adipic acid. Short-term inhalation exposure to 126 mg/m3 adipic acid to rats did not produce signs of toxicity, but exposure of mice to 460 mg/m3 resulted in decreased weight gain and produced effects in the upper respiratory tract, liver, kidneys, and central nervous system.
In a subchronic oral study, 10 male and 10 female rats exposed to 10% sodium succinate in the drinking water died, but no compound-related lesions were found. Body weights were decreased in rats given ≥2.5% sodium succinate for 13 weeks, but toxicological treatment-related changes were not observed. Glutaric acid had a low degree of toxicity to rats (at 2%) and dogs (concentration not specified) when given in the feed. Dietary administration of ≤3400 mg/kg bw/d adipic acid for 19 weeks produced slight effects in the liver of male rats; the NOAEL was 3333 mg/kg bw. A mixture of adipic, glutaric, and succinic acids had a low degree of toxicity in rats when tested at 3% for 90-days. Signs of toxicity were reported in a subchronic inhalation study in which mice were exposed to 13 or 120 mg/m3 adipic acid.
Slight effects were seen in the livers of rats fed ≤3200 mg/kg bw/d adipic acid for 33 weeks, and the NOAEL for rats fed a diet containing adipic acid for 2 years was 1%; no significant toxicological effects were seen at concentrations of ≤5%. No significant toxicological effects were observed for mice fed ≤280 mg/kg bw or rabbits fed ≤400 mg/kg bw azelaic acid for 180 days. Disodium sebacate was not toxic to rats or rabbits fed up to 1000 mg/kg bw for 6 mos.
For the dicarboxylic acids, the severity of ocular irritation seems to decrease with increasing carbon number. Succinic acid was a severe ocular irritant, glutaric acid was moderately irritating, and dodecanedioic acid was a slight irritant. Ocular irritation produced by adipic acid was dose-dependent. Slight to mild dermal irritation was observed in rabbits for succinic, glutaric, and adipic acid, while dodecanedioic acid was not an irritant in rabbits. Using guinea pigs, adipic acid, dodecanedioic acid, and a mixture of succinic, glutaric, and adipic acids are not sensitizers.
Reproductive and developmental effects were not seen upon oral dosing with the dicarboxylic acids or disodium sebacate. Malonic acid, at 0.1% in vitro, has a spermicidal effect on human spermatozoa. Glutaric acid was tested at doses of ≤1300 mg/kg bw in rats and 500 mg/kg bw in rabbits, adipic acid at doses of ≤263 mg/kg bw in mice, 288 mg/kg bw in rats, 205 mg/kg bw in hamsters, or 250 mg/kg bw in rabbits, azelaic acid at doses of ≤140 mg/kg bw in rats and 200 mg/kg bw in rabbits, disodium sebacate at 500 mg/kg bw in rats and 1000 mg/kg bw in rabbits, and dodecanedioic acid was tested at ≤1000 mg/kg bw using rats. Embryotoxic effects were reported in a reproductive study of 2500 mg/kg bw/d azelaic acid using rats and in reproductive studies with ≤500 mg/kg bw/d azelaic acid using rabbits and monkey. In vitro, sodium salts of some dicarboxylic acid had a specific inhibitory effect on muscle activity of the uterine horn, and this effect progressively increased with chain length.
The dicarboxylic acids are not genotoxic, and consistently were not mutagenic in Ames tests. Positive results were seen in a transformation assay on glutaric acid using Balb/c-3T3 cells, both with and without metabolic activation. The results of a mouse lymphoma assay, with and without metabolic activation, on glutaric acid were negative in a neutral pH range. Equivocal results were obtained in an in vitro chromosomal aberration assay of ≤15 mg/mL disodium succinate using Chinese hamster fibroblast cells. The dicarboxylic acids were not genotoxic in in vivo assays.
Carcinogenicity was not seen in rats given up to 2% sodium succinate in the drinking water or 5% adipic acid in feed for 2 years. An increase in the incidence of C-cell adenoma/carcinoma of the thyroid in females given 2% sodium succinate, and a positive trend in the occurrence of this tumor, was considered a function of experimental variability and not related to dosing. Adipic acid was not carcinogenic when given orally to rats at up to 5% in the diet.
In a cumulative irritancy test, the cumulative irritation of a 15% azelaic acid gel increased with successive patching. It is not known if the vehicle played a role in the irritation scores. Daily application of a 20% azelaic cream causes erythema and irritation.
Esters of Dicarboxylic Acids
The metabolism of diesters in animals is expected to occur, initially, via enzymatic hydrolysis, leading to the corresponding dicarboxylic acids and the corresponding linear or branched alcohol. These dicarboxylic acids and alcohols can be further metabolized or conjugated to polar products that are excreted in urine, or, the enzymatic hydrolysis may be incomplete and result, at least for some diesters, in the production of monoesters.
In in vitro absorption studies using pig skin, 8.8% and 3% of undiluted diethyl malonate were found in the skin and receptor fluid, respectively, after 50 hours. Absorption was enhanced when diethyl malonate was diluted with ethanol and reduced when diluted in acetone. Using human skin, 16% of the applied diethyl malonate penetrated in 24 hours. In vivo, absorption of diethyl malonate, estimated from urinary and fecal recovery, was 15% in nude mice, 4% in human skin grafted to nude mice, 6% in pig skin grafted to nude mice, 2.5% in pigs, and 4% in dogs.
Approximately 11% of ditridecyl adipate was absorbed through the skin of rats; 5.5% to 7.4% of the applied dose was found in the tissues, 3.5% to 4.7%was found in the urine, and 0.4% to 0.7% was found in the feces after 4 days. Prior dosing with ditridecyl adipate did not significantly affect absorption.
In vitro, diethylhexyl adipate was readily hydrolyzed to mono-(2-ethylhexyl) adipate (MEHA) or adipic acid in rat liver, pancreas, and small intestine tissue preparations. In rats, diethylhexyl adipate is hydrolyzed to adipic acid and 2-ethylhexanol or MEHA. 2-Ethylhexanol is converted to 2-ethylhexanoic acid, which may form a glucuronide conjugate or may be subjected to ω- and (ω-1)-oxidation and further metabolism. More than 98% of diethylhexyl adipate administered orally to rats was excreted in 48 hours; 21% to 45% of the radioactivity was expired in carbon dioxide and 34% to 52% was excreted in the urine. Diethylhexyl adipate and MEHA are not found in the blood or urine; diethylhexyl adipate or the metabolites are recovered in the tissues. Metabolism studies have shown that excretion in the urine is not as unchanged diethylhexyl adipate; mostly adipic acid is found. In humans, peak urinary elimination of all metabolites occurred within 8 hours of dosing.
Diethylhexyl sebacate is not readily absorbed through the skin of guinea pigs. Metabolism in rodents and humans may follow partially common pathways, producing 2-ethylhexanol as an intermediary metabolite.
Diethylhexyl adipate is a peroxisome proliferator requiring extensive phase I metabolism to produce the proximate peroxisome proliferator, which in both mice and rats appears to be 2-ethylhexanoic acid. Diethylhexyl adipate is not as potent a proliferator as diethylhexyl phthalate. Peroxisome proliferation causes an increase in liver weights and can induce hepatocarcinogenicity in rats and mice. Peroxisome proliferation is not believed to pose the risk of inducing hepatocarcinogenesis in humans, as a species difference in response to peroxisome proliferators exists.
Diethylhexyl adipate did not bind covalently to hepatic DNA in mice. It did stimulate DNA synthesis in livers of rats. In another study, a statistically significant increase in 8-OH-dG occurred in the liver DNA, but not the kidney DNA, at week 1 and 2. The IARC remarked that the weight of evidence for diethylhexyl adipate demonstrated that rodent peroxisome proliferators do not act as direct DNA-damaging agents.
The oral and dermal LD50 values are greater than 2 g/kg. No mortality occurred in rats exposed to concentrated vapors of diethyl malonate diethyl succinate, dibutyl adipate, or diethylhexyl adipate for 8 hours. Some deaths, possibly due to thermal decomposition were seen in rats and rabbits exposed to 940 mg/m3 for 7 hours. In a 4-hr inhalation toxicity study, a mixture of dimethyl glutarate, dimethyl succinate, and methyl adipate, the anterior and posterior nasal passageways were affected.
Oral administration of ≤1000 mg/kg bw dibutyl adipate for 28 days did not produce toxic effects in rats. In short-term oral dosing with diethylhexyl adipate, decreased weight gain was reported for rats and mice. The NOELs for rats and mice were 2 and 0.63%, respectively, in feed; 5/5 female mice fed 10% dibutyl adipate in feed died. In 2- and 4-week studies of diethylhexyl adipate, the oral NOAEL for ovarian toxicity was 200 mg/kg bw in rats; an increase in atresia of the large follicle and a decrease in currently formed corpora lutea were seen in females dosed with 1000 and 2000 mg/kg bw diethylhexyl adipate.
In a short-term dermal study in which 10 rabbits were dosed dermally with 0.5 or 1.0 mL/kg of a 20% dispersion of dibutyl adipate for 6 weeks, there was a significant decrease in bws in the high-dose group, and renal lesions in 1 animal of each group. There were no signs of toxicity in guinea pigs in an immersion study with 20.75% diisopropyl adipate, diluted to an actual concentration of 0.10% adipate. Dermal administration of diethylhexyl adipate to rabbits for 2 weeks resulted in slight to moderate erythema at the test site, but toxic effects were not reported for most of the animals.
In a 90-day oral toxicity study in which rats were fed 36 to 41 mg/kg bw diethyl malonate, no treatment-related effects were observed. Dietary administration of ≤2.5% di-C7-9 branched and linear alkyl esters of adipic (approx. 1500 and 1900 mg/kg bw/d for males and females, respectively) for 90 days did not result in systemic toxicity. The NOAELS for male and female rats were 1500 and 1950 mg/kg bw/d, respectively. Subchronic oral administration of diethylhexyl adipate to rats caused significant decreases in bw gains and increases in liver and kidney weights. The dietary NOEL for rats in a 90-day study was 610 mg/kg bw. A decrease in bws was seen in mice fed a diet with 1.2 and 2.5% diethylhexyl adipate. For diisononyl adipate, dietary administration of up to 500 mg/kg bw to rats for 13 weeks resulted in a statistically significant increase in relative kidney weights, but there were no toxicological findings. With dogs, 3.0% dietary diisononyl adipate resulted in a decrease in bws, testes weight, and feed consumption, increased liver weight, elevated enzyme levels, liver and kidney discoloration, and microscopic changes in the liver, testes, spleen, and kidneys.
No adverse effects were reported with whole-body application of a 6.25% emulsion of dibutyl adipate to dogs 2x/week for 3 mos. Unoccluded dermal application of up to 2000 mg/kg bw ditridecyl adipate for 13 weeks to rats produced slight erythema, but no systemic toxicity.
In a 6-month study in which rats were dosed intragastrically with diethylhexyl adipate, hepatic detoxification appeared depressed at the beginning of the study, while in a 10-mos study, a decrease in central nervous system excitability was noted. Dietary administration of ≤1.25% dibutyl sebacate for 1 year or ≤6.25% for 2 years did not have an effect on growth
Ocular irritation appeared to lessen in severity as chain length of the dicarboxylic acid esters increased. Undiluted diethyl malonate was slightly to moderately irritating to rabbit eyes. Dibutyl, diisopropyl, and diethylhexyl adipate, at concentrations ranging from 0.1% to 100%, were non- or minimal ocular irritants. Diisopropyl sebacate was minimally irritating. Diethylhexyl sebacate was nonirritating in an MTT viability assay. Undiluted dioctyldodecyl and diisocetyl dodecanedioate were not irritating to rabbit eyes.
The esters of dicarboxylic acids were mostly non- or mildly irritating to rabbit skin. Some minimal irritation was seen with diisopropyl adipate, undiluted or at 5 to 20; 75% in formulation, and moderate erythema was reported with undiluted dibutyl adipate. Dimethyl malonate, dibutyl and diethylhexyl adipate, diethylhexyl sebacate, and dioctyldodecyl dodecanedioate were not sensitizers in guinea pigs or rabbits. Perfume formulations containing 1.1% diisopropyl adipate were not phototoxic in rabbits.
Oral administration of up to 1000 mg/kg bw dimethyl malonate to Wistar rats did not have an effect on fertility, and no developmental toxicity was reported. The NOAEL was 300 mg/kg bw for repeated dose and maternal toxicity and 1000 mg/kg bw for fertility and developmental toxicity. Oral administration of up to 100 mg/kg bw dibutyl adipate to Sprague-Dawley rats did not cause any reproductive effects, and the NOEL for parental and offspring toxicity was 300 mg/kg bw/d and for reproductive toxicity was 100 mg/kg bw/d. Oral administration of ≤7000 mg/kg bw di-C7-9 branched and linear alkyl esters of adipic acid to Sprague-Dawley rats did not result in developmental toxicity. Dietary administration of up to 1.2% diethylhexyl adipate did not affect fertility when fed to rats prior to mating. Fetal weight, total litter weight, and litter size were reduced with 1.2% diethylhexyl adipate. In a study in which gravid rats were fed the same doses during gestation, no significant effects on fetal weight or litter size were reported. An increased incidence of minor skeletal abnormalities was attributed to fetotoxicity. In a study in which diethylhexyl adipate was given orally to rats from day 7 of gestation until post-natal day 17, antiandrogenic effects were not observed, although some increase in post-natal death was observed. Administration of up to 2000 mg/kg bw diethylhexyl adipate prior to dosing and through day 7 of gestation did have an effect on the mean estrous cycle length at a dose of 1000 and 2000 mg/kg bw, and did appear to disturb ovulation. Significant decreases were also seen in implantation rate and number of live embryos, as well as an increase in pre-implantation loss. Diethylhexyl adipate did not produce testicular toxic effects in male F344 rats when fed at up to 25 000 ppm in the diet for 4 weeks. Dietary administration of 6.25% dibutyl sebacate to male and female Sprague-Dawley rats for 10 weeks prior to mating had no adverse effects on fertility, litter size, or survival of offspring. Diethylhexyl sebacate, 200 ppm in the diet, did not produce reproductive or developmental effects in rats.
Dermal applications of 2000 mg/kg bw ditridecyl adipate did not have an effect on sperm morphology. Some visceral anomalies were reported. The NOAELs for maternal toxicity and developmental and reproductive effects were 2000 and 800 mg/kg bw/d, respectively.
Dimethyl, diethyl, dipropyl, dibutyl, diisobutyl, and diethylhexyl adipate were evaluated for fetotoxic and teratogenic effects in rats when administered ip at 1/3 to 1/30 of the ip LD50 values. Some effect on resorptions and abnormalities were seen with all but diethyl adipate.
Inhalation by rats of ≤1.0 mg/L of a mixture of dimethyl glutarate, dimethyl succinate, and dimethyl adipate on days 7 to 16 of gestation or for 14 days prior to mating, during mating and gestation, and lactation, no adverse developmental or reproductive effects were observed. The only exception was a statistically significant decrease in pup weight at birth and day 21.
Diethylhexyl adipate appeared to have endocrine-mediated effects in Crj:CD (SD) rats in a 28-day oral study; however, it was stated that the findings may be attributable to the disturbance in ovarian function according to the hypothalamic-pituitary-gonad axis. Diethylhexyl adipate simulated thyroid hormone-dependent rat pituitary GH3 cell proliferation in a concentration-dependent manner.
The esters of dicarboxylic acids were not mutagenic or genotoxic in a battery of in vitro and in vivo tests. The only nonnegative results reported were equivocal results in a sister chromatid exchange assay with ≤400 µg/mL diethylhexyl adipate in the presence of metabolic activation and a dose-dependent inhibition of 3 H-thymidine into replicating DNA, with a dose-dependent increase in the ratio of acid-incorporated 3 H-thymidine with ≤0.01 mol/L diethylhexyl adipate. (The same effect was seen in the 3 H-thymidine assay with 2-ethylhexanol.)
In an NTP 2-year dietary study, ≤25 000 ppm diethylhexyl adipate did not produce tumors in male or female rats, but it did increase the incidence of hepatocellular adenoma and carcinoma in male and female mice. Diethylhexyl adipate did not cause skin tumors with weekly application of 10 mg to the back of mice in a lifetime study. Other compounds with a 2-ethylhexyl group that have been evaluated for carcinogenicity had some evidence of hepatocarcinogenicity, ranging from very strong to equivocal, in rodents. Feeding of diethylhexyl sebacate to rats for 19 mos did not result in carcinogenic effects.
In a number of clinical irritation and sensitization studies, the diesters of dicarboxylic acids are not irritants or sensitizers. The only exception noted was that undiluted diisopropyl adipate was moderately irritating in 1 cumulative irritancy test, and some slight irritation was seen with formulations containing diethylhexyl adipate. A 10% dilution of dibutyl adipate tested on 30 participants and formulations containing 0.7% to 17% diisopropyl adipate, tested on 49 to 98 participants, and 9% diethylhexyl adipate, tested on 25 participants, were not phototoxic.
Cases of allergic contact dermatitis in response to diethyl sebacate-containing products have been reported, and it has been demonstrated that diethyl sebacate was the substance, or one of several substances in the products, eliciting the dermatitis. Two case studies were reported of allergic reactions to lotion containing diisopropyl sebacate.
According to the Integrated Risk Information System of the EPA, the weight-of-evidence classification for diethylhexyl adipate was “possible human carcinogen”. The classification was based on an absence of human data and increased liver tumors in female mice. The IARC has stated that diethylhexyl adipate is not classifiable as to its carcinogenicity in humans.
Discussion
The Expert Panel reviewed the available data on dicarboxylic acids and their salts, and the data on the esters of dicarboxylic acids, and determined that these ingredients are safe as used in cosmetics. In reaching this conclusion, the Expert Panel considered a number of issues.
The Expert Panel noted gaps in the available safety data for some of the dicarboxylic acid and salts and esters of dicarboxylic acids in this safety assessment. The available data on many of the ingredients are sufficient, and similar structural activity relationships, biologic functions, and cosmetic product usage, suggest that the available data may be extrapolated to support the safety of the entire group. For example, a concern was expressed regarding the extent of dermal absorption for certain long-chain branched diesters. The Panel inferred that since dermal penetration of long chain alcohols is likely to be low and the dermal penetration for the diesters is likely to be even lower, toxicity characteristics from ingredients where toxicity data were available was appropriate.
The CIR Expert Panel considered the dangers inherent in using animal-derived ingredients, namely the transmission of infectious agents. While tallow may be used in the manufacture of some ingredients in this safety assessment and is clearly animal-derived, the Expert Panel notes that tallow is highly processed and tallow derivatives even more so. The Panel agrees with determinations by the U.S. FDA that tallow derivatives are not risk materials for transmission of infectious agents.
The Panel noted that the only significant toxic effect of the dicarboxylic acids was irritation to the skin and eyes, which would be expected for acids. Dicarboxylic acids reviewed in this safety assessment are not expected to be appreciably absorbed from cosmetic formulations, exhibit low single-dose or repeated-dose toxicity in animal studies, and are not genotoxic or carcinogenic in animal studies. Since a use of these acids in cosmetics is as a pH adjuster, the irritating property of these acids would be lost. The highest use of an acid in leave-on formulations is 0.3% azelaic acid, of a salt is 0.4% disodium succinate, and of an ester is 14% diethylhexyl adipate. Although bath products can contain higher concentrations of these acids, salts, or esters, contact time is short and the product will be diluted as it is being rinsed.
Case studies have reported reactions to products containing diethyl sebacate. Follow-up patch tests performed with ≥5% diethyl sebacate, which is greater than the reported use concentration, had positive results. Diethyl sebacate is reported to be used in cosmetics at 1.5%, and no irritation or sensitization was reported in clinical studies of a formulation containing 1.5% diethyl sebacate.
The Expert Panel also noted that esters of dicarboxylic acids, in particular diethylhexyl adipate, have the potential to induce peroxisome proliferation. This effect has been examined because ethylhexyl adipate is structurally related to a notable peroxisome proliferator, diethylhexyl phthalate. Diethylhexyl adipate is not as potent a peroxisome proliferator as diethylhexyl phthalate, and, while peroxisome proliferation is toxicologically well-studied, this is an effect observed only in rodents and is not relevant to humans. Accordingly, the hepatocarcinogenic effects observed in rodents are related to this effect and not believed to pose the risk of inducing hepatocarcinogenesis in humans.
The reproductive and developmental toxicity of the dicarboxylic acids and their esters were generally well studied. The results of these studies did not cause any concern for the Panel.
The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration and the duration of the exposure and their site of deposition within the respiratory system. In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ~38 µm. Particles with an aerodynamic diameter of ≤ 10µm are respirable. In the absences of inhalation toxicity data, the panel determined that dicarboxylic acids and their salts and the esters of dicarboxylic acids can be used safely in hair sprays, because the product size is not respirable.
Conclusion
The CIR Expert Panel concluded that dicarboxylic acids and their salts, and the esters of dicarboxylic acids, as listed below, are safe in the present practices of use and concentration. Were ingredients in these groups not in current use (as indicated by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in these groups.
Acids and salts:
malonic acid*
succinic acid
sodium succinate
disodium succinate
glutaric acid*
adipic acid
azelaic acid
dipotassium azelate*
disodium azelate*
sebacic acid
disodium sebacate*
dodecanedioic acid*
Esters:
diethyl malonate
decyl succinate*
dimethyl succinate
diethyl succinate*
dicapryl succinate
dicetearyl succinate*
diisobutyl succinate*
diethylhexyl succinate
dimethyl glutarate
diisobutyl glutarate*
diisostearyl glutarate
dimethyl adipate
diethyl adipate*
dipropyl adipate*
dibutyl adipate
dihexyl adipate
dicapryl adipate
di-C12-15 alkyl adipate*
ditridecyl adipate*
dicetyl adipate*
diisopropyl adipate
diisobutyl adipate
diethylhexyl adipate
diisooctyl adipate*
diisononyl adipate*
diisodecyl adipate
dihexyldecyl adipate*
diheptylundecyl adipate
dioctyldodecyl adipate
diisocetyl adipate*
diisostearyl adipate
isostearyl sebacate
diethyl sebacate
dibutyl sebacate*
dicaprylyl/capryl sebacate*
diisopropyl sebacate
diethylhexyl sebacate
dibutyloctyl sebacate*
diisooctyl sebacate
dihexyldecyl sebacate*
dioctyldodecyl sebacate
diisostearyl sebacate*
dioctyldodecyl dodecanedioate
diisocetyl dodecanedioate
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.