Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of calcium sodium borosilicate, calcium aluminum borosilicate, calcium titanium borosilicate, silver borosilicate, and zinc borosilicate as used in cosmetics. These borosilicate glasses function mostly as bulking agents. Available animal and human data were considered along with data from a previous safety assessment of magnesium silicates. The similar structure, properties, functions, and uses of these ingredients enabled grouping them and using the available toxicological data to assess the safety of the entire group. Data submitted on calcium borosilicate, which is not a cosmetic ingredient, are also included as additional support for the safety of borosilicate glass ingredients. The Panel concluded that borosilicate glasses are safe as cosmetic ingredients in the practices of use and concentration as given in this safety assessment.
Introduction
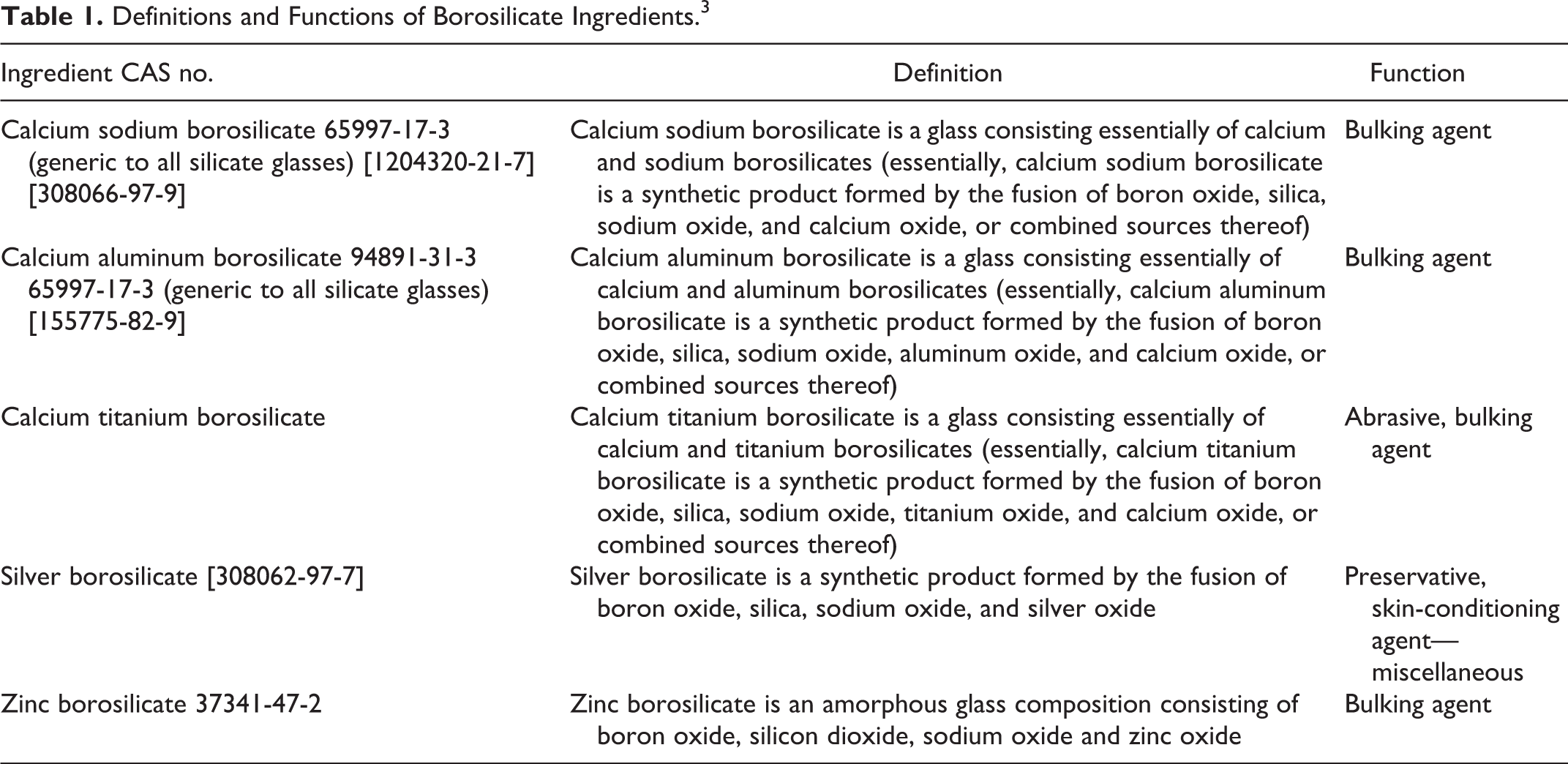
The borosilicate glass ingredients used in cosmetics include:
calcium sodium borosilicate,
calcium aluminum borosilicate,
calcium titanium borosilicate,
silver borosilicate, and
zinc borosilicate.
These ingredients function in cosmetics mostly as bulking agents and are also used as abrasives, preservatives, and skin-conditioning agents—miscellaneous.
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) previously reviewed the related ingredients silica, alumina magnesium metasilicate, aluminum calcium sodium silicate, aluminum iron silicates, hydrated silica, and sodium potassium aluminum silicate. 1 These ingredients were determined to be safe in the present practices of use and concentration. Data supporting the safety of silica and related silicate salts as used in cosmetics included single-dose toxicity, where, for example, the acute dermal no observed effect level (NOEL) for silica was >2 g/kg for rabbits, repeated-dose studies, genotoxicity studies, reproductive and developmental toxicity studies, and clinical and animal irritation and sensitization studies. Data were available regarding silicosis, but these data were not relevant to the amorphous silica ingredients under review.
The Panel also reviewed the safety of various silicates and silicate clays used in cosmetics. 2 These ingredients are not significantly toxic in single-dose or repeated-dose studies except that inhalation toxicity is readily demonstrated. Skin irritation and sensitization studies supported the safety of these ingredients as did ocular irritation studies. The risk of pulmonary damage was clearly affected by particle size, fiber length, and concentration. The Panel considered that any spray containing these solids should be formulated to minimize their inhalation. With this caveat to the cosmetics industry, the Panel concluded that these ingredients are safe as currently used in cosmetic formulations.
Data on the related compound calcium borosilicate, which is not an ingredient in the International Cosmetic Ingredient Dictionary and Handbook, 3 are included since this chemical is similar to the ingredients under review, and the data supplement the few toxicity data available on the ingredients covered in this report.
Chemistry
Definition
The
Definitions and Functions of Borosilicate Ingredients. 3
Borosilicate glass is frequently referred to as vitreous silica and consists of a network of SiO4 tetrahedra and metal cations. 4 Borosilicates display heterogeneity of structure, with submicroscopically small volumes of ordered structure but a lack of long-range order.
Physical and Chemical Properties
Physical and chemical properties are provided in Table 2.
Chemical and Physical Properties.
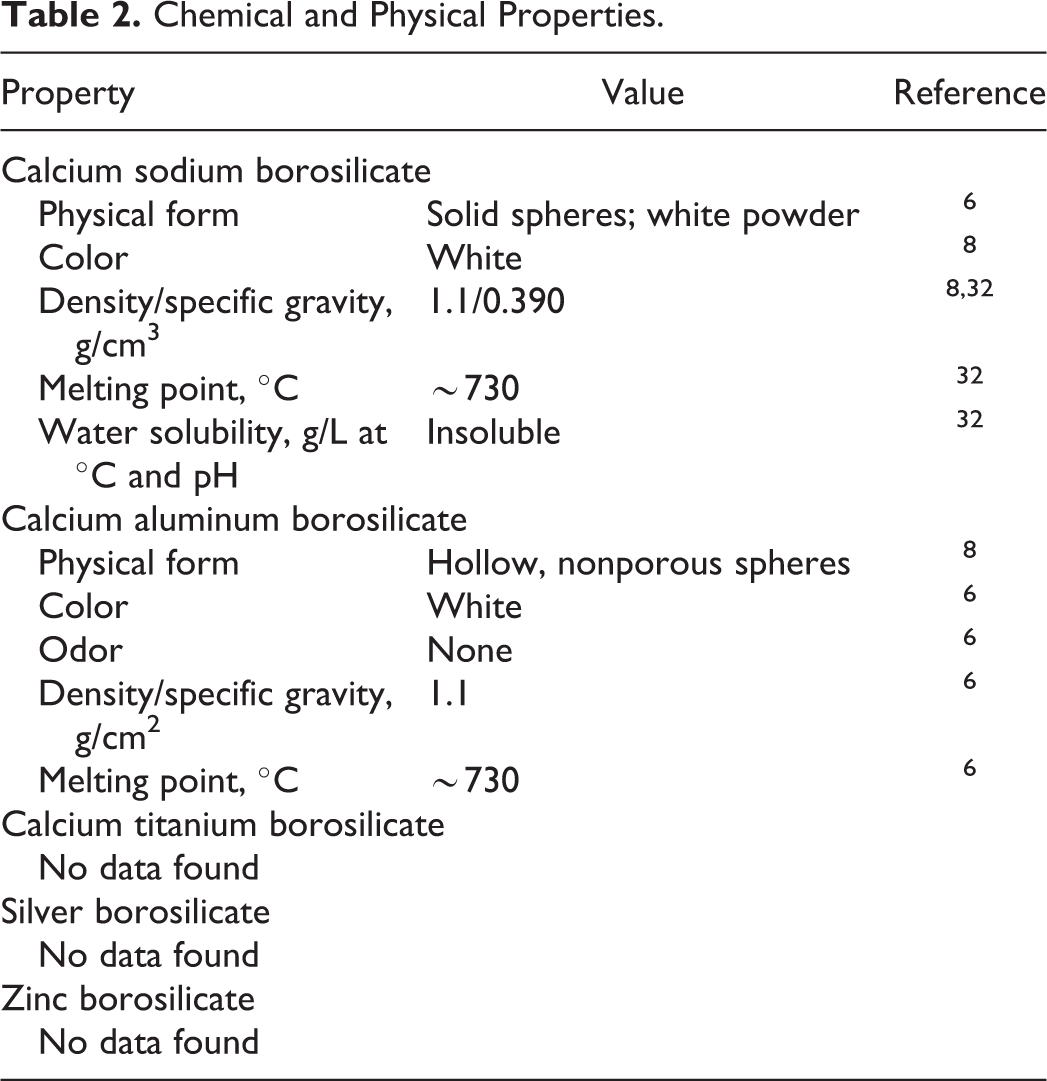
One manufacturer reported that calcium aluminum borosilicate, calcium sodium borosilicate, or calcium titanium borosilicate were all sold under the same product names, further designated as low alkali or as extra corrosion resistant. 5 The glass flakes were highly planar platelets with very smooth surfaces and were transparent. The “apparent” density of the “low-alkali” borosilicate glass was reported to be 0.09 g/cm3, and real density was reported to be 2.6 g/cm3. The melt temperature for both types of borosilicates was reported to be 930°C to 1020°C. The glass composition (Table 3) may vary from batch to batch. These products are reported to be stable.
Chemical Analysis Calcium Sodium Borosilicate Glasses. 5
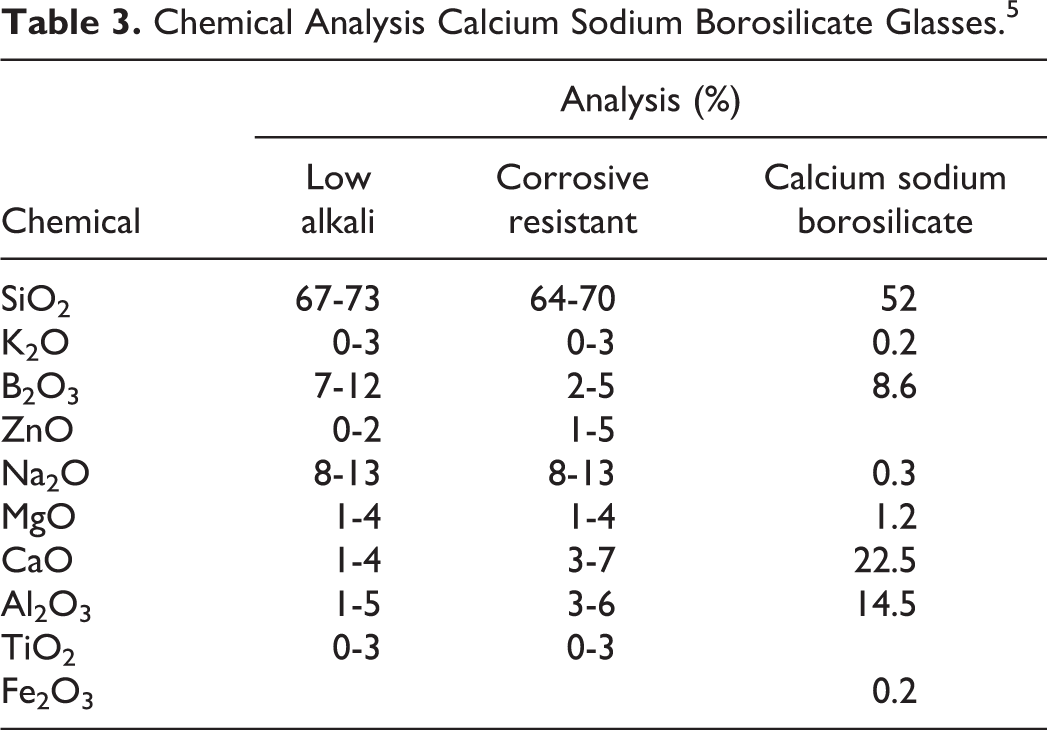
The chemical composition of calcium sodium borosilicate was reported to be: SiO2, 52%; Na2O, 0.3%; CaO, 22.5%; MgO, 1.2%; Al2O3, 14.5%; Fe2O3, 0.2%; K2O, 0.2%, and B2O3, 8.6% (Table 3). 6
The borosilicate in a cosmetic was described in a patent document as comprising a cosmetic grade flaky glass constituted of components comprising, by weight, at least 52% alkali metal oxide. 7 The flaky glass may be unmodified, surface coated (silicone), or complexed (titania and rare metal, ultramarine, or other pigments).
One manufacturer reported that its calcium aluminum borosilicate product consists of microspheres instead of flakes. 6,8
The coordination of the boron network cations readily varied from 3 to 4 depending on composition and temperature in turn varying the properties. 4 There were also large differences caused by varying the size and charge of the “network modifier” cations (eg, Na+ vs Ca2+), B–Si ratio, and Al content.
Borosilicates are chemically unreactive and of high-dielectric strength. 4 Chemical analyses of samples of borosilicates of different compositions showed that B2O3 evaporation was negligible.
Calcium Borosilicate
The melting point of calcium borosilicate is >1540°C, its specific gravity is 2.65, and it has a water solubility of 0.035 g/L. 9 It is reported to be 100% pure, chemically stable, and unreactive.
Particle Size
A manufacturer reported that their borosilicate glass particles are flakes that are 50 nm to 5 μm in thickness with a particle size of 15 to 350 μm. 5 For the low-alkali borosilicate glass, the particle size distribution is stated as 1000 to 300 μm, 10% or less; 300 to 50 μm, 65% or more; and <50 μm, 25% or less. The product may also be milled to have a specific size range of 4 to 65 μm. For the low-corrosion-resistant borosilicate glass, the particle size distribution is specified as being the same as the low-alkali borosilicate.
In a product designed for cosmetic formulations, the calcium sodium borosilicate flakes had a geometric particle size of 9 to 13 μm. 8 The size distribution is 3 to 6 μm 10%; 8 to 12 μm, 50%; and 16 to 23 μm, 90%.
In cosmetic applications, calcium aluminum borosilicate is used as a substrate for metal oxide colors. 10 When coated with the colors, the particle size is reported to be 20 to 200 μm.
The borosilicate in a cosmetic was reported to have an average particle diameter of 1 to100 µm and an aspect ratio (average thickness dividing the average particle diameter) of ≥10 (in comparison to a value of 1 for a sphere). 7
Manufacturers report supplying calcium aluminum borosilicate in sphere form with a mean diameter of 11.7 μm. 6,8
Calcium Borosilicate
Calcium borosilicate was reported to be a fine, white, odorless powder with a particle size of the flakes ranging from 3.4 to 4.2 µm. 9
Method of Manufacture
Borosilicate powder is prepared by a so-called rotary method. The first step is melting glass in a furnace. 5 Once molten, the glass is poured into a spinning cup. Centrifugal force causes the glass to leave the cup as a thin film. This film is broken into flakes. These flakes are then collected downstream if the product is classified as unmilled (large particle size). In further downstream processing, the flake is passed through a hammer mill if the product is to be classified as milled (smaller particle size). For micronized glass flakes, an opposed jet mill (high-velocity air jets through a series of nozzles accelerating the particles to produce collisions to further reduce the particle size) is used.
Borosilicate glasses may be prepared by the wet-chemical method. 4 Reagent-grade metal salts (except for sodium and calcium) are introduced as nitrates, boron as boric acid, and silicon as a colloidal solution of SiO2.
Borosilicate glasses may also be prepared by the sol–gel method, which results in reproducible properties. 4 In this method, HBO3 is dissolved in the colloidal SiO2 sol. A second solution of all the other glass constituents, such as nitrates, is then added. After partial evaporation, the sol sets to a gel, and the gel is dried at 200°C for 16 hours, which usually results in a friable product that is ball milled. This results in a fine powder in which all elements are mixed. This powder is melted to a bubble-free glass in a furnace for 2 hours at temperatures varying from 800 to 1400°C (depending on the composition of the glass).
Calcium aluminum borosilicate is manufactured by placing finely divided sodium tetrahydrate pentahydrate (80 g), quartz (100 g), kaolin (180 g), and wollastonite (40 g) as a dry mix into a small mixer then stirred at low speed. 11 Water is added slowly until the mixture forms a crumb. The crumb was heated at 1000°C until completely dry. The product is placed in a furnace and heated at a rate of 100°C/min up to a maximum of 1500°C, where the temperature is held for 90 minutes. The product is then allowed to cool to ambient temperature in the furnace, removed, and broken up with a hammer. The resulting composition is reportedly opaque with a creamy-white color and particulate form.
Silver borosilicate is prepared through introduction of Ag2O during the melting process. 12
Impurities
One manufacturer reported heavy metal impurities in calcium sodium borosilicate which included lead (<10 ppm), arsenic (<2 ppm), and mercury (<1 ppm). 8
It was noted in a safety data sheet that calcium aluminum borosilicate has no free SiO2; all components are amorphous/noncrystalline. 6
Another manufacturer reported that a sample of calcium aluminum borosilicate contained lead (21.8 mg/kg) and barium (63.5 mg/kg). 5 Arsenic, antimony, mercury, cadmium, chromium, or nickel were below the detection limit of 2 mg/kg.
Use
Cosmetic
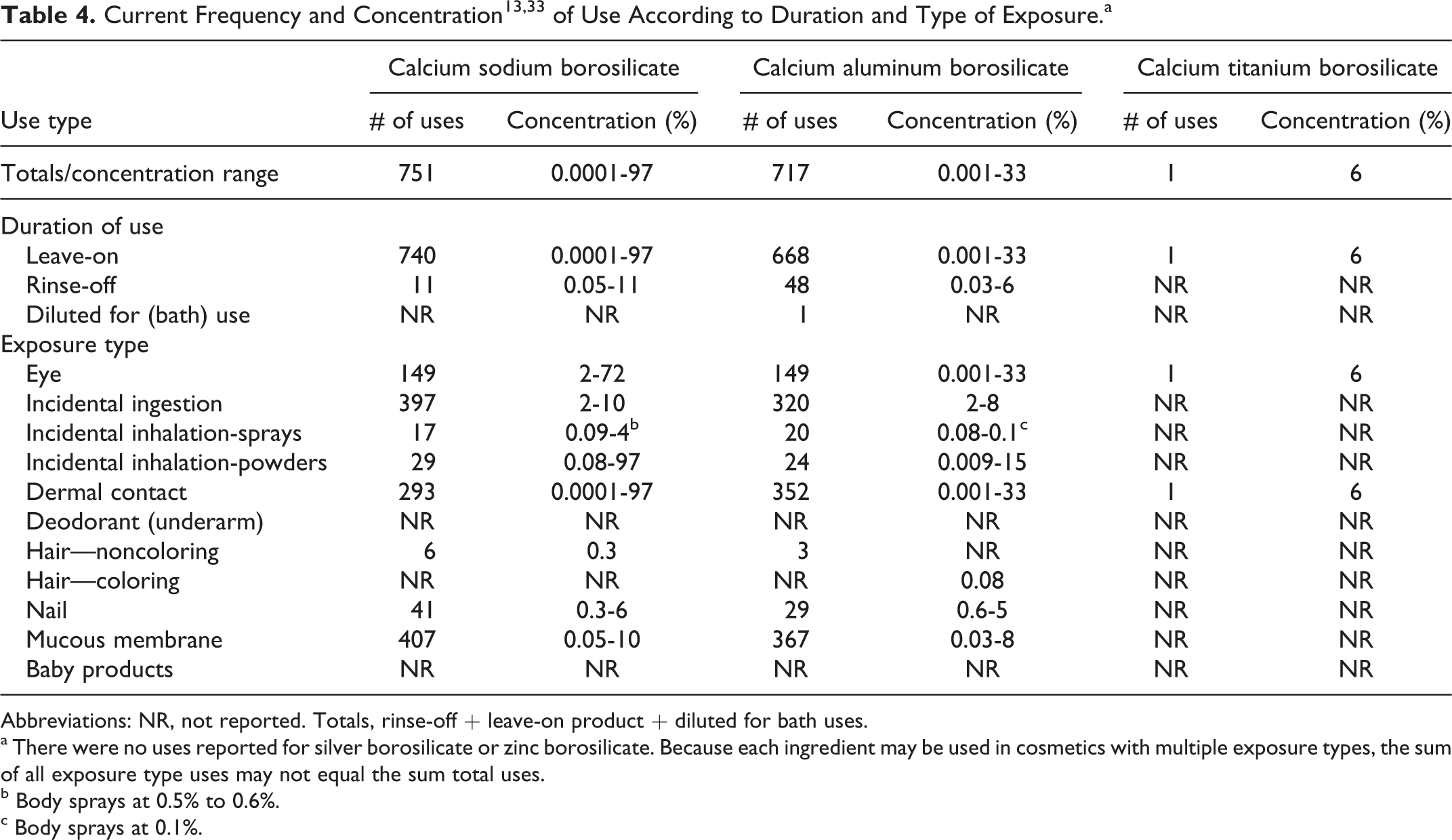
Data on ingredient usage as a function of cosmetic product type are provided by industry to the Food and Drug Administration Voluntary Cosmetic Registration Program (VCRP) and a survey conducted by the Personal Care Products Council (Council) collected maximum use concentrations for ingredients in this group. 13,14 These data are combined in Table 4.
Abbreviations: NR, not reported. Totals, rinse-off + leave-on product + diluted for bath uses.
a There were no uses reported for silver borosilicate or zinc borosilicate. Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
b Body sprays at 0.5% to 0.6%.
c Body sprays at 0.1%.
Calcium sodium borosilicate was reported to be used in 751 cosmetic products with a range of maximum concentrations of 0.0001% to 97%. Of these, 740 products were leave-on products (including 397 lipsticks with a maximum concentration range of 2%-10%; body sprays at 0.5%-0.6%; and face powders at 4%-97%). There were 11 rinse-off products with a maximum concentration range of 0.05%-11%. Calcium aluminum borosilicate was used in 717 cosmetic products with a maximum range of 0.001%-33%. Of these, 668 were leave-on products (including 320 lipsticks at 2%-8%; eye shadow at 0.001%-33%; body sprays at 0.1%; and aerosol hair color sprays at 0.08%), 48 rinse-off products with a maximum range of 0.03%-6%; and 1 diluted for bath product (no concentration of use reported). Calcium titanium borosilicate was reported to be used in 1 eye shadow at a maximum concentration of 6%.
There were no reported uses in the VCRP or use concentrations in the Council survey for silver borosilicate or zinc borosilicate. 13,14
Calcium sodium borosilicate and calcium aluminum borosilicate were reported to be used in body sprays and aerosol hair color sprays up to 0.8% and face powders up to 97%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 15,16
Noncosmetic
Borosilicates are used for laboratory glassware, household cooking ware, industrial piping, bulbs for hot lamps, and electronic tubes of high wattage such as x-ray tubes. 4 Multicomponent borosilicate glasses have many applications, such as fiber reinforcements in composites, in flat-panel display screens for computers and cell phones and in nuclear waste sequestration. 17 Borosilicate glass impregnated with silver has antibacterial and antialgal uses. 18,19
Toxicokinetics
There were no published toxicokinetics data available for the cosmetic ingredients in this report nor were unpublished data provided.
Toxicological Studies
Acute Toxicity
Dermal—Nonhuman
Calcium Borosilicate
The dermal lethal dose, 50% (LD50) of calcium borosilicate (moistened with distilled water and applied to ∼20% of the body under occlusion; clipped and intact or abraded skin) was reported to be >2000 mg/kg (the highest amount tested) for New Zealand White rabbits (n = 5/sex). 9,20 There were no deaths. Two animals showed decreased activity and 1 fecal staining of the anogenital area within 24 hours. Some animals showed discharges from the nose or eyes in the second week after dosing. Examination at necropsy showed no changes considered related to the test substance.
Oral—Nonhuman
Calcium Borosilicate
The oral LD50 of calcium borosilicate (administered as a 30% solution in 1% carboxymethylcellulose) was reported to be >5000 mg/kg for Sprague Dawley CD rats (n = 5/sex). 9,21 One female rat showed slight weight loss at day 14 and 1 male had a soft stool 2 hours after dosing.
Reproductive and Developmental Toxicity
There were no published reproductive or developmental toxicity studies available for the cosmetic ingredients in this report nor were unpublished data provided.
Genotoxicity
In Vitro
Calcium Borosilicate
An Ames test using Salmonella typhimurium (TA100, TA98, TA1535 and TA1537) of the supernatant of calcium borosilicate in dimethyl sulfoxide (DMSO) was conducted. Calcium borosilicate (1 g/mL) was steeped in DMSO overnight, and the supernatant was used for the assay. The supernatant was not mutagenic with or without metabolic activation. Controls were sodium azide, 2-nitrofluorene, and 9-aminoacridine which elicited the expected results. 9,22
Carcinogenicity
There were no published carcinogenicity studies available for the cosmetic ingredients in this report nor were unpublished data provided.
Irritation and Sensitization
Irritation
Dermal—Nonhuman
Calcium borosilicate
Calcium borosilicate was moistened with distilled water and applied to ∼20% of the body under occlusion to clipped skin (intact or abraded) of New Zealand White rabbits (n = 5/sex). 23 Mild erythema was observed in 2 of 5 animals in which the test material was applied to abraded skin and in of 3 of 5 animals that received the test material applied to intact skin.
Calcium borosilicate (0.5 g in 0.5 mL saline) was administered to the abraded and nonabraded skin of New Zealand White Rabbits (n = 3/sex) under occlusion for 24 hours. 9,20 At 30 minutes and 48 hours after removal, there was no difference between the response of intact and abraded skin. At removal, all treatment sites showed erythema ranging from very slight to moderate to severe. Edema was present at abraded and nonabraded sites in 2 animals, being barely perceptible in one and slight in the other. At 72 hours, the irritation was much decreased. Calcium borosilicate was reported to be slightly irritating to rabbit skin.
Ocular
Calcium sodium borosilicate
In a use test (n = 31), an eye shadow containing calcium sodium borosilicate (53%) was used daily for 4 weeks. 24 Ophthalmological examinations were conducted before and after the test period. Of the subjects, 21 were self-perceived as “having sensitive eyes.” There were no adverse effects reported, and all ophthalmologic examinations were normal.
In a use test, (n = 41), several eye shadows containing calcium sodium borosilicate (94%-99%; 81%-97%; 74%-93%; 60%-97%; 50%-80%) were used daily for 4 weeks. 25 Ophthalmological examinations were conducted before and after the test period. No adverse effects were observed during the treatment.
Calcium aluminum borosilicate
An Epiocular human cell assay was conducted on a powder eye shadow containing calcium aluminum borosilicate (31%). 26 Test results showed no expectation of ocular irritation.
Mucosal
Calcium sodium borosilicate
In a use test (n = 41), several lipsticks containing calcium sodium borosilicate (94%-99%; 81%-97%; 92%-97%; 74%-93%; 60%-97%; 50%-80%) were used daily for 4 weeks. 27 No adverse effects were observed during treatment, and there were no reports of irritation.
Sensitization
Human
Calcium sodium borosilciate
In an human repeat insult patch test (HRIPT; n = 98) of a powder eye shadow containing calcium sodium borosilicate (53%; 0.2 g) under semiocclusion, there were no signs of irritation or sensitization observed. 28
In an HRIPT (n = 104) of a cosmetic product containing calcium sodium borosilicate (60%-97%; 0.2 g applied) under semiocclusion, there were no signs of irritation or sensitization observed. 29
In an HRIPT (n = 103) of a cosmetic product containing calcium sodium borosilicate (53.5%; 0.2 g applied) under semiocclusion, there were no signs of irritation or sensitization observed. 30
Calcium aluminum borosilicate
In an HRIPT (n = 105) of a powder eye shadow containing calcium aluminum borosilicate (31%; 0.2 g applied), there were no adverse effects observed, and there was no evidence of sensitization. 31
Summary
Borosilicate glasses, including calcium sodium borosilicate, calcium aluminum borosilicate, calcium titanium borosilicate, silver borosilicate, and zinc borosilicate, are described as cosmetic ingredients although not all are reported to be in current use. These ingredients function mostly as bulking agents. Borosilicate glasses are chemically unreactive.
A previous safety assessment of silica and related silicate salts included single-dose toxicity, where, for example, the acute dermal NOEL for silica was >2 g/kg for rabbits, repeated-dose studies, genotoxicity studies, reproductive and developmental toxicity studies, and clinical and animal irritation and sensitization studies. Data were available regarding silicosis, which is related to crystalline silica. These data were not relevant to the amorphous silica ingredients under review. A previous safety assessment of various silicates and silicate clays used in cosmetics noted that these ingredients are not significantly toxic in single-dose or repeated-dose studies, except that inhalation toxicity is readily demonstrated. Skin irritation and sensitization studies supported the safety of these ingredients as did ocular irritation studies. The risk of pulmonary damage was clearly affected by particle size, fiber length, and concentration.
Potential heavy metal impurities of calcium sodium borosilicate are reported to be <10 ppm lead, <2 ppm arsenic, and <1 ppm mercury. Calcium aluminum borosilicate has no free SiO2; all components are amorphous/noncrystalline.
Calcium sodium borosilicate was reported to be used in 681 cosmetic products with a range of maximum concentrations from 0.0001% to 97%. Calcium aluminum borosilicate was used in 555 cosmetic products with a maximum range from 0.001% to 33%. Calcium titanium borosilicate was reported to be used at a maximum concentration of 6% in eye shadows. There were no reported uses for silver borosilicate or zinc borosilicate.
No toxicokinetics, reproductive or developmental toxicity, or carcinogenicity studies were available for the cosmetic ingredients in this report.
No acute or repeated-dose toxicity data were found for the cosmetic ingredients in this report, but, calcium borosilicate, a related chemical, had a dermal LD50 of >2000 mg/kg for rabbits and an oral LD50 of >5000 mg/kg for rats.
The supernatant of calcium borosilicate steeped in DMSO was not genotoxic in an Ames test.
An eye shadow containing calcium sodium borosilicate was not irritating or sensitizing nor did it cause eye irritation when used daily for 4 weeks in clinical tests. Calcium aluminum borosilicate was not predicted to be an ocular irritant in an Epiocular assay. Calcium borosilicate was a slight to mild dermal irritant and a moderate ocular irritant to rabbits.
Lipsticks containing calcium sodium borosilicate up to 97% were not irritating in 4-week use studies.
Products containing calcium sodium borosilicate at ∼53% and calcium aluminum borosilicate at 31% were not sensitizing in HRIPTs.
Discussion
Although there were data gaps for borosilicate glasses such as reproductive and developmental toxicity, these ingredients have similar chemical structures, physicochemical properties, functions, and concentrations in cosmetics compared to silicate salts and calcium borosilicate. These similarities allow grouping the ingredients together and extending the available toxicology data for the silicate salts and calcium borosilicate to support the safety of the entire group.
The Panel noted that these borosilicate ingredients are insoluble, inert, and will not significantly penetrate the skin. The zinc and silver are secure in the molecules and will not be bioavailable. Therefore, there would not be any systemic toxicity expected from dermal application. These ingredients are not dermal irritants or sensitizers. Since these ingredients were not orally toxic in acute tests, the Panel was not concerned about their use in lipsticks.
The Panel discussed the issue of incidental inhalation exposure from body and hand sprays, hair color sprays, and fragrance preparations. There were no inhalation toxicity data available. However, the particle size of borosilicate glasses was reported to range from 50 nm to 1000 μm with the largest portion being in the 50 to 300 μm range. The Panel expects that the sizes of a substantial majority of the particles of these ingredients, as manufactured, be larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. These ingredients are reportedly used at concentrations up to 4% in cosmetic products that may be aerosolized and up to 97% in products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the short exposure time, this information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects. The Panel considered other data available to characterize the potential for borosilicate glasses to cause irritation and sensitization. They noted the lack of irritation or sensitization in tests of dermal exposure, no systemic toxicity at 5000 mg/kg, and the absence of genotoxicity in an Ames test for a supernatant of the related chemical calcium borosilicate. Borosilicate glasses are chemically inert and thus not systemically toxic. A detailed discussion of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that borosilicate glasses are safe in the present practices of use and concentration as described in this assessment. The ingredients in this safety assessment include:
calcium sodium borosilicate,
calcium aluminum borosilicate,
calcium titanium borosilicate,
silver borosilicate*, and
zinc borosilicate*.
The * indicates not reported in use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.