Abstract
The Cosmetic Ingredient Review Expert Panel assessed the safety of bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 as used in cosmetics, finding that these ingredients are safe in cosmetic formulations in the present practices of use and concentration. Both ingredients are lanolin substitutes and are reported to function in cosmetics as skin-conditioning agents—emollients. The Panel reviewed available animal and clinical data in making its determination of safety.
Introduction
Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl poly-acyladipate-1are cosmetic ingredients used in over 500 cosmetic formulations as skin-conditioning agents—emollients.
Chemistry
Definitions
Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are mixed fatty acid esters. Bis-diglyceryl polyacyladipate-1 (CAS No. 135229-94-6) is the adipic acid diester of a mixed diglyceryl ester of caprylic, capric, isostearic, and hydroxystearic acids; bis-diglyceryl polyacyladipate-2 (CAS No. 82249-33-0) is a similar adipic acid diester of a mixed diglyceryl ester including those same acids and stearic acid. 1
There are multiple structural configurations within each of these ingredients. This is the case because there are 3 different isomers (alpha,alpha-, beta,beta-, and alpha,beta-) present in diglycerin, the 2 different connectivities (alpha- and beta-) in which diglycerin can be attached to adipic acid, and the variety of fatty acid esters (e.g., caprylate, caprate, stearate, isostearate, and hydroxystearate) to be formed with the reactive hydroxyls of bis-diglyceryl adipate. Figure 1 depicts the structures of the mixture of chemicals present in the bis-diglyceryl polyacyladipate ingredients.
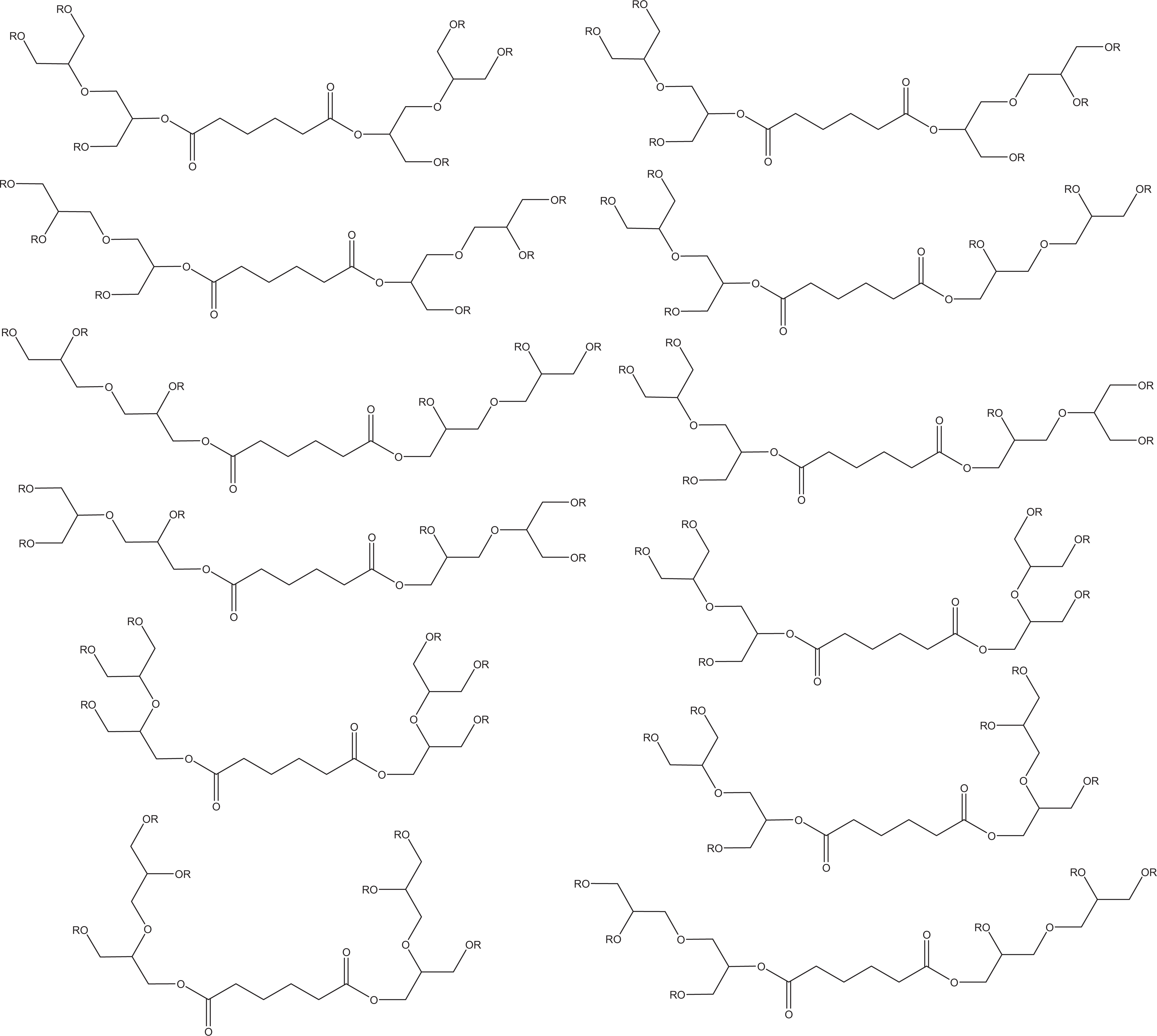
The structures represent the mixture of chemicals in bis-diglyceryl polyacyladipate ingredients. O-R represents the ester of caprylic, capric, stearic, isostearic, or hydroxystearic acid, in the case of bis-diglyceryl polyacyladipate-2; but in the case of bis-diglyceryl polyacyladipate-1 O-R represents the ester of caprylic, capric, isostearic, or hydroxystearic acid. The structures of caprylic, capric, stearic, isostearic, and hydroxystearic acids are depicted. (Figure continued on next page.)
Physical and Chemical Properties
Available physical and chemical properties’ data are provided in Table 1.
Chemical and Physical Properties.

Abbreviations: KOH, potassium hydroxide; Max, maximium.
Method of Manufacture
Both bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are manufactured in 1-pot reactions with heat, catalysts, and reduced pressure. 2 The raw materials used in the production of both of these esters are diglycerin, adipic acid, isostearic acid, 12-hydroxystearic acid, n-octanoic acid, and n-decanoic acid. Stearic acid is also a raw material for the production of bis-diglyceryl polyacyladipate-2.
Impurities
Acid number and hydroxyl number indicate that bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are essentially free of starting raw materials. 2 A manufacturer also reports that the following parameters are checked regularly in bis-diglyceryl polyacyladipate-2, with typical findings given as aflatoxin B1, B2, G1, and G2, <0.10 ppb each; heavy metals, that is, As, Cd, Cr, Ni, Pb, Hg, Cu, Sn, <1 parts peer million (ppm) each; pesticides—organochlorine, organocholorophosphorous, and pyrethroids, <0.01 ppm each class; various PCBs <0.01 ppm each class; microbiology: pathogens absent, total viable count <10 colony forming unit (CFU)/g, and yeast/molds <10 CFU/g.
Use
Cosmetic
Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are suitable as lanolin substitutes3,4 and are reported to function as skin-conditioning agents—emollients. 1
The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2012 indicate that bis-diglyceryl polyacyladipate-2 is used in 521 cosmetic formulations and that bis-diglyceryl polyacyladipate-1 is used in 6 cosmetic formulations. 5
Responses to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) report that bis-diglyceryl polyacyladipate-2 is used in leave-on products at concentrations up to 36% (reported for lipsticks) and in rinse-off products at concentrations up to 21% (reported for eye makeup remover). 6 Bis-diglyceryl polyacyladipate-1 is used in leave-on products at concentrations up to 10% (reported for tonics, dressings, and other hair grooming aids) and in rinse-off products at a concentration of 4% (reported for eye makeup remover). Frequency and concentration of use data categorized by exposure and duration of use are provided in Table 2.
Frequency and Concentration of Use According to Duration and Type of Exposure.
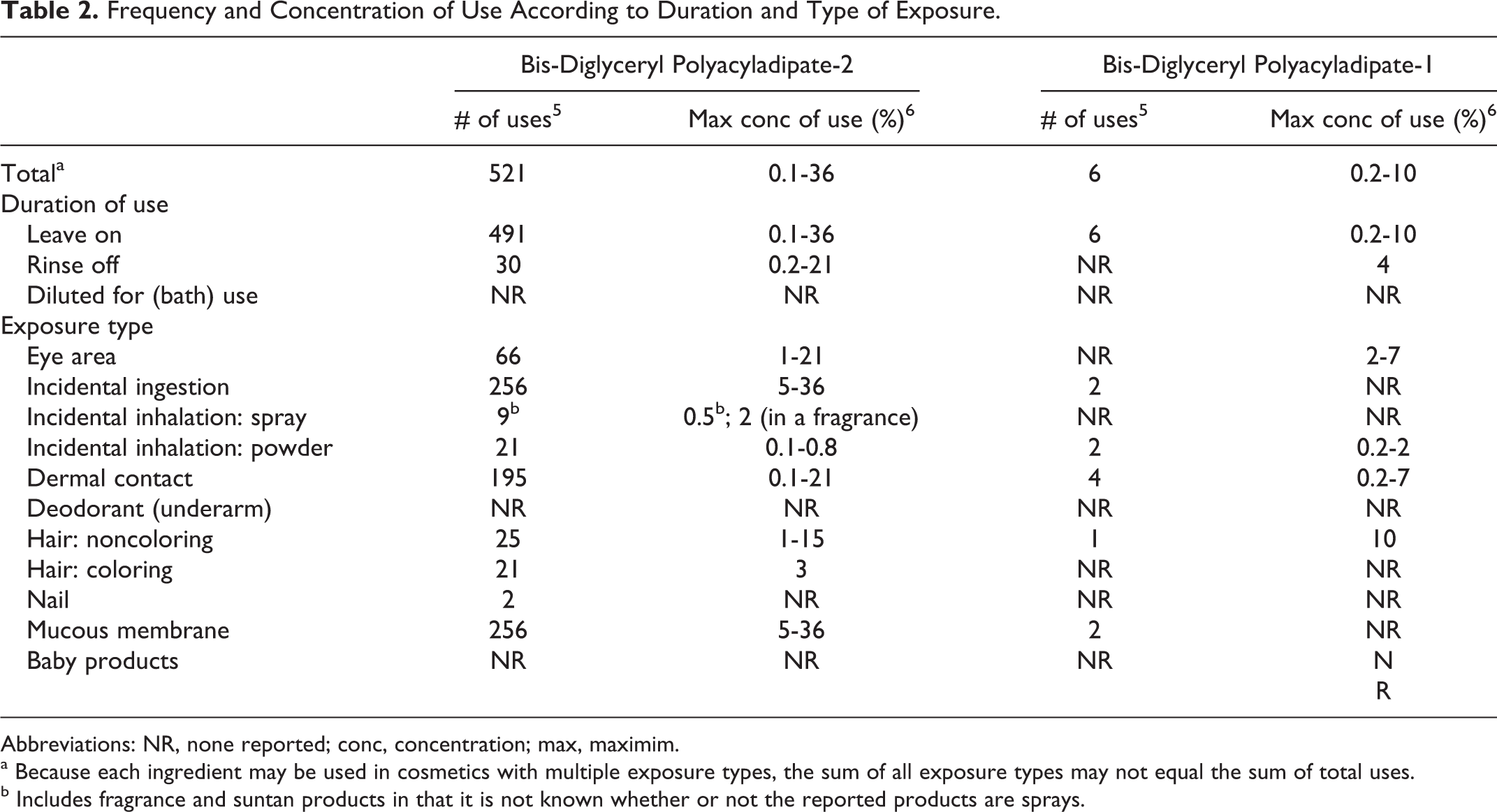
Abbreviations: NR, none reported; conc, concentration; max, maximim.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b Includes fragrance and suntan products in that it is not known whether or not the reported products are sprays.
Products containing bis-diglyceryl polyacyladipate-2 may be used near the eye area and in products in which incidental ingestion may occur. Additionally, bis-diglyceryl polyacyladipate-2 is used in face powders at up to 0.8% and in “other” fragrance preparations at 2%; it is possible that this fragrance preparation is sprayed. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm.7–10 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.7,9 Both bis-diglyceryl polyacyladipate-2 and polyacyladipate-1 are listed in the European Union inventory of cosmetic ingredients. 11
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Published toxicokinetic data were not found and unpublished data were not provided.
Toxicological Studies
Single-Dose (Acute) Toxicity
Dermal
Bis-Diglyceryl Polyacyladipate-2
Groups of 5 male and 5 female Wistar rats were dosed with a single 24-hour application of 2 g/kg body weight (bw) undiluted bis-diglyceryl polyacyladipate-2. 12 The exposure site was a shaved 5 × 10 cm area on the back. None of the animals died, and no signs of dermal irritation were observed. Body weight gains were normal in males but reduced in females. The dermal LD50 of bis-diglyceryl polyacyladipate-2 is >2 g/kg bw.
Bis-Diglyceryl Polyacyladipate-1
Groups of 5 male and 5 female Sprague-Dawley rats were exposed to a single 24-hour semiocclusive application of 2 g/kg bw undiluted bis-diglyceryl polyacyladipate-1. 13 The exposure site was a shaved 5 × 4 cm area on the back. None of the animals died, and no signs of toxicity or dermal irritation were observed. The dermal LD50 of bis-diglyceryl polyacyladipate-1 is >2 g/kg bw.
Oral
Bis-Diglyceryl Polyacyladipate-2
Groups of 5 male and 5 female Wistar rats were dosed once by gavage with 2 g/kg bw bis-diglyceryl polyacyladipate-2 in corn oil. 14 None of the animals died and the oral LD50 of bis-diglyceryl polyacyladipate-2 is >2 g/kg bw.
Bis-Diglyceryl Polyacyladipate-1
Groups of 5 male and 5 female Sprague-Dawley rats were dosed once by gavage with 5 g/kg bw bis-diglyceryl polyacyladipate-1 in arachis oil, with a dose volume of 10 mL/kg. 15 None of the animals died, and no signs of systemic toxicity were reported. The oral LD50 of bis-diglyceryl polyacyladipate-1 is >5 g/kg bw.
Repeated-Dose Toxicity
Oral
Bis-Diglyceryl Polyacyladipate-2
Groups of 5 male and 5 female Wistar rats were dosed by gavage with 0, 180, 1800, or 4500 mg/kg bw (corresponding to 0, 0.2, 2.0, or 5.0 mL/kg bw, respectively) bis-diglyceryl polyacyladipate-2 in corn oil once daily for 28 days. 16 The test volume was 10 mL/kg bw. The animals were killed at the termination of dosing, and gross and microscopic examinations were performed. The only observations made were in the high-dose group males. The slight but significant reduction in total bilirubin content was not considered biologically relevant and was attributed to coincidence, and an increase in prostate weight may be considered either a coincidental finding or a result of a test article-induced load reaction without histopathological manifestation. The no-observable adverse effect level (NOAEL) was 1800 mg/kg bw (2 mL/kg bw) bis-diglyceryl polyacyladipate-2.
Reproductive and Developmental Toxicity
Bis-Diglyceryl Polyacyladipate-2
The reproductive toxicity potential of bis-diglyceryl polyacyladipate-2 was determined in a 1-generation study using groups of 24 male and 24 female Sprague-Dawley rats. 17 The male animals were dosed daily by gavage with 0 or 1000 mg/kg bw bis-diglyceryl polyacyladipate-2 in corn oil from 10 weeks prior to mating until the day before being killed (day 99), and the female rats were dosed by gavage with the same doses from 2 weeks prior to mating until weaning and were killed on day 21 following delivery. The dose volume was 10 mL/kg bw. The litters were culled on day 4, and the remaining 8 pups/litter were killed on day 21. Bis-diglyceryl polyacyladipate-2 had no effects on reproduction, fertility, or development, and no signs of general toxicity were observed.
Genotoxicity
In Vitro
Bis-Diglyceryl Polyacyladipate-2
The mutagenic potential of bis-diglyceryl polyacyladipate-2 was evaluated in an Ames test performed using Salmonella typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100, with and without metabolic activation. 18 The researcher stated that the test material was insoluble in all solvents specified for the Ames test, and for this reason, a spot test was performed and the product was tested directly and undiluted. Bis-diglyceryl polyacyladipate-2 was not mutagenic with or without metabolic activation. Appropriate negative and positive control results were valid.
A chromosomal aberration assay was performed using Chinese hamster lung fibroblasts (V79 cells) with and without metabolic activation for 40 to 400 µg/mL bis-diglyceryl polyacyladipate-2. 19 Ethanol was the solvent. Bis-diglyceryl polyacyladipate-2 did not induce a significant increase in the incidence of chromosomal aberrations and was not clastogenic. Appropriate negative and positive control results were valid.
In Vivo
Bis-Diglyceryl Polyacyladipate-2
A micronucleus test was performed in mice to evaluate the genotoxic potential of bis-diglyceryl polyacyladipate-2. 20 Three groups of 10 NMRI mice, 5 males and 5 females per group, were given a single oral dose of 15 000 mg/kg bw in corn oil, and the groups were killed 24, 48, or 72 hours after dosing. The dose volume was 30 mL/kg bw. In males, differences in the number of polychromatic erythrocytes compared to normochromatic erythrocytes (an increase in the 48 h group and decrease in the 72 h group) were observed; this observation might be explained by an increased neoformation of polychromatic erythrocytes in male mice after administration of the test article first, resulting in an increase in polychromatic erythrocytes after 48 hours. After 72 hours, the number of normochromatic erythrocytes, which arise from polychromatic erythrocytes, increased, resulting in a decrease in polychromatic erythrocytes. This was not considered a genotoxic effect. Vehicle and appropriate positive control results were valid.
Carcinogenicity
Published carcinogenicity studies were not found and unpublished data were not provided.
Irritation and Sensitization
Skin Irritation
Nonhuman
Bis-Diglyceryl Polyacyladipate-2
Bis-diglyceryl polyacyladipate-2 was nonirritating to rabbit skin after a single 4-hour semiocclusive application when evaluated in an acute dermal irritation/corrosion test in 3 rabbits. 21 A dose of 2000 mg/kg was applied neat to a shaved 8 × 15 cm area on the back of each animal under a semiocclusive covering for 4 hours. The test site was examined for signs of irritation at various intervals for 0.5 to 72 hours after patch removal. No erythema or edema was observed.
Bis-diglyceryl polyacyladipate-2, tested at 5% and 40% in petrolatum, was a weak irritant in guinea pig skin. 22 The cumulative irritation potential was evaluated in groups of 3 guinea pigs. The test material was applied to a shaved area of the flank of each animal once daily for 3 consecutive days, and the test sites were scored 24 hours after each application. The test volume was not stated. The cumulative irritation index was 1.2/4 with 5% and 1.3/4 with 40% bis-diglyceryl polyacyladipate-2.
Human
Bis-Diglyceryl Polyacyladipate-2
A single 24-hour application of 5% bis-diglyceryl polyacyladipate-2 in petrolatum was not irritating to human skin. 22 The dermal irritation potential was evaluated in 44 subjects, and the test material was applied to the intact skin of the forearm for 24 hours under an occlusive patch. The test volume was not stated. No reactions were observed after 24 hours.
Undiluted bis-diglyceryl polyacyladipate-2 had no irritating potential in human skin. 23 A total of 15 male and 35 female participants were used to evaluate the dermal irritation potential of undiluted bis-diglyceryl polyacyladipate-2. In all, 12 participants were classified as atopic and 7 as dermal sensitive. An occlusive patch (defined as a commercial plaster) containing 2 mg/cm2 of the test article was applied to the back of each participant for 48 hours; the size of the application area was not specified. The test site was scored for irritation upon patch removal and 24 hours later. Well-defined erythema was observed in 1 participant upon patch removal but not 24 hours later; the researcher determined this reaction to be toxic irritative.
Skin Sensitization
Nonhuman
Bis-Diglyceryl Polyacyladipate-2
Bis-diglyceryl polyacyladipate-2 did not induce any allergic response and was classified as nonsensitizing in a guinea pig maximization test (GPMT). 24 Groups of 10 male and 10 female Pirbright white guinea pigs were used. A test concentration of 5% was used during intradermal induction. The topical induction concentration was 25% in petrolatum (w/v), and the test article was applied for 48 hours under an occlusive patch to a shaved 4 × 6 cm area on the shoulder of each animal; the test area was pretreated with 10% sodium lauryl sulfate (SLS) 24 hours prior to patching. The challenge was performed 14 days after induction by applying a 24-hour occlusive patch with 25% bis-diglyceryl polyacyladipate-2 in petrolatum to a shaved 5 × 5 cm area on the flank of each animal. Vehicle controls (20 animals) were used, and 2,4-dinitrochlorobenzene was used as a positive control.
Bis-Diglyceryl Polyacyladipate-1
Bis-diglyceryl polyacyladipate-1 did not produce any sensitization reactions in a GPMT performed in Dunkin-Hartley guinea pigs. 25 The test concentration for intradermal induction was 25% (w/v), and the topical induction was 0.2 to 0.3 mL undiluted test article applied with a 2 × 4 cm filter paper without SLS pretreatment. The challenge application was 0.1 to 0.2 mL undiluted test article applied with a 2 × 2 cm filter paper. Ten vehicle control animals were used, and the positive control was formaldehyde.
Human
Bis-Diglyceryl Polyacyladipate-2
A lipstick formulation containing 36% bis-diglyceryl polyacyladipate was not an irritant or a sensitizer in a human repeated insult patch test (HRIPT). 26 The HRIPT was initiated in 114 participants and completed in 102 participants. None of the participants discontinued testing because of test-article-related effects. Approximately 0.1 mL of neat test material was applied to a 2 × 2 cm occlusive patch, and the patches were applied for 48 to 72 hours 3 times per week for 3 consecutive weeks during the induction phase of the study. The 48-hour challenge patches were applied to a treated and naive site after a 10- to 15-day nontreatment period. One participant had a reaction score of 2 (moderate erythema) at induction readings 4 to 7; there were no other reactions recorded for this subject during induction or at challenge.
Comedogenicity
Bis-Diglyceryl Polyacyladipate-2
Bis-diglyceryl polyacyladipate-2 was noncomedogenic in rabbits. 27 A volume of 0.5 mL of the test article was applied neat once daily, 5 days/week for 4 weeks, to the right ears of 4 male New Zealand White (NZW) rabbits. The contralateral ears served as untreated controls. Increasing visible hyperkeratosis extending to possible comedones (score 1 of 3) was observed in all 4 test ears following the initial application. However, the scores were 0 of 3 for all remaining test days, and the overall comedogenic score was 0 of 3 for all 4 rabbits. Redness of the treated ears was observed throughout the study.
Bis-Diglyceryl Polyacyladipate-1
Bis-diglyceryl polyacyladipate-1 was noncomedogenic in NZW rabbit. 28 An unspecified volume of the test article was applied neat once daily, 5 days/week for 3 weeks, to the left ears of 3 female rabbits. The right ears served as untreated controls. Gross examination reported slight transient hyperkeratosis on the control and/or treated ears of 2 rabbits. No hyperkeratosis or comedones were found upon microscopic examination.
Ocular Irritation
Bis-Diglyceryl Polyacyladipate-2
Bis-diglyceryl polyacyladipate-2 was classified as nonirritating in an acute eye irritation/corrosion test using NZW rabbits. 29 Undiluted test substance, 0.1 mL, was instilled into the conjunctival sac of 1 eye of each of 3 rabbits, and the contralateral eye served as a negative control. Eyes were examined for up to 72 hours postinstillation. Some mild irritation of the conjunctivae was observed (a single report of a score of 2 of 4); all effects were reversible after 5 days.
Summary
Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipdate-1 are mixed fatty acid esters reported to function in cosmetics as skin-conditioning agents —emollients. Bis-diglyceryl polyacyladipate-1 is the adipic acid diester of a mixed diglyceryl ester of caprylic, capric, isostearic and hydroxystearic acids; bis-diglyceryl polyacyladipate-2 is a similar adipic acid diester of a mixed diglyceryl ester including those same acids and stearic acid. Structurally, there are a multitude of configurations possible within each bis-diglyceryl polyacyladipate ingredient.
Bis-diglyceryl polyacyladipate-2 is used in 521 cosmetic formulations; it is used in leave-on products at concentrations of up to 36% (reported for lipsticks) and in rinse-off products at concentrations of up to 21% (reported for eye makeup remover). Bis-diglyceryl polyacyladipate-1 is used in 6 cosmetic formulations; it is used in leave-on products at concentrations of up to 10% (reported for tonics, dressings, and other hair grooming aids) and in rinse-off products at a concentration of 4% (reported for eye makeup remover).
Single doses of dermally applied undiluted bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipdate-1 were not irritating to rabbit skin. None of the animals died during the studies, and the dermal LD50 was >2000 mg/kg bis-diglyceryl polyacyladipate-2 and for bis-diglyceryl polyacyladipdate-1.
Oral single-dose, repeated-dose, and reproductive studies were performed using rats. No mortality was observed following a single dose of 2000 mg/kg bis-diglyceryl polyacyladipate-2 and of 5000 mg/kg bis-diglyceryl polyacyladipate-1. In a 28-day oral toxicity study with bis-diglyceryl polyacyladipate-2, the NOAEL was 1800 mg/kg bw (2 mL/kg bw). In a 1-generation reproduction study, oral administration of 1000 mg/kg bis-diglyceryl polyacyladipate-2 had no effects on reproduction, fertility, or development, and no signs of general toxicity were observed during the study.
Undiluted bis-diglyceryl polyacyladipate-2 was not mutagenic in a spot test in S typhimurium with or without metabolic activation, and 40 to 400 µg/mL bis-diglyceryl polyacyladipate-2 was not clastogenic in a chromosomal aberration assay using Chinese hamster lung fibroblasts. Bis-diglyceryl polyacyladipate-2 was not genotoxic in a micronucleus test in which male and female NMRI mice were given a single oral dose of 15 000 mg/kg bw in corn oil and each killed 24, 48, or 72 hours after dosing.
A single 24-hour semiocclusive application of 2000 mg/kg bis-diglyceryl polyacyladipate-2, applied neat, was not irritating in rabbit skin, but 5% to 40% bis-diglyceryl polyacyladipate-2 in petrolatum was a weak irritant in guinea pig skin in a 3-day cumulative irritation study. Bis-diglyceryl polyacyladipate-2 patched occlusively at 5% for 24 hours or undiluted for 48 hours was not irritating to human skin. Neither bis-diglyceryl polyacyladipate-2 nor bis-diglyceryl polyacyladipate-1 was a sensitizer in the GPMT. For bis-diglyceryl polyacyladipate-2, the intradermal induction concentration was 5% with SLS pretreatment, the topical induction concentration was 25%, and the challenge concentration was 25% with SLS pretreatment. With bis-diglyceryl polyacyladipate-1, 25% without SLS was used for intradermal induction and undiluted test article was used for topical induction and for the challenge. A lipstick containing 36% bis-diglyceryl polyacyladipate was not an irritant or a sensitizer in an HRIPT. Neither ingredient was comedogenic in rabbit ears. Undiluted bis-diglyceryl polyacyladipate-2 was not an ocular irritant in rabbit eyes.
Discussion
Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are not acute or reproductive toxicants, they are not genotoxic, and they are not dermal irritants/sensitizers. The Cosmetic Ingredient Review (CIR) Expert Panel recognized that data gaps remained regarding toxicokinetics and carcinogenicity data. Bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are used mostly in leave-on dermal applications, and both ingredients are large, highly lipid-soluble compounds that are not expected to efficiently pass through the stratum corneum of the skin. As noted, even if these ingredients were to penetrate the skin, the constituent compounds do not present a toxicological concern based on the available toxicity data. Although many of the available data are only for bis-diglyceryl polyadipate-2, bis-diglyceryl polyacyladipate-1 was considered to be entirely analogous from a toxicological perspective. Therefore, the Expert Panel concluded that the data were adequate to assess the safety of both ingredients.
The Panel discussed the issue of incidental inhalation exposure to bis-diglyceryl polyacyladipate-2 from face powders and “other” fragrance preparations that may be aerosolized. There were no inhalation toxicity data available. This ingredient is reportedly used at concentrations up to 2% in cosmetic products that may be aerosolized and up to 0.8% in other products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered the data available to characterize the potential for bis-diglyceryl polyacyladipate-2 to cause systemic toxicity, irritation, sensitization, or other effects. They noted that bis-diglyceryl polyacyladipate-2 did not produce systemic toxicity at high doses in single-dose oral and dermal studies or in a 28-day oral repeated-dose study; was not a reproductive or developmental toxicant; was not genotoxic in an Ames test or in a chromosomal aberration assay with Chinese hamster lung fibroblasts; and did not cause irritation or sensitization in humans at concentrations up to 36%. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that bis-diglyceryl polyacyladipate-2 and bis-diglyceryl polyacyladipate-1 are in the present practices of use and concentration in cosmetics.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
