Abstract
The Cosmetic Ingredient Review Expert Panel assessed the safety of lauriminodipropionic acid, sodium lauriminodipropionate, and disodium lauriminodipropionate as used in cosmetics. These ingredients function in cosmetics as hair-conditioning agents and surfactant-cleansing agents. The Panel reviewed relevant animal and human data related to the safety of these ingredients in cosmetics. The Panel concluded that lauriminodipropionic acid, sodium lauriminodipropionate, and disodium lauriminodipropionate are safe as cosmetic ingredients in the present practices of use and concentration.
Introduction
Sodium lauriminodipropionate is the sodium salt of a substituted propionic acid that is used as a surfactant, a hair-conditioning agent, and an antistatic agent in cosmetic formulations. In a safety assessment for sodium lauriminodipropionate and sodium lauraminopropionate published by the Cosmetic Ingredient Review (CIR) in 1997, 1 the CIR Expert Panel concluded that the available data were insufficient to support safety of both the ingredients for use in cosmetics.
Now, data that support the safety of sodium lauriminodipropionate as used in cosmetics are available. Because of the chemical similarities to sodium lauriminodipropionate, the CIR Expert Panel determined that the newly available data may be extrapolated to the parent compound, lauriminodipropionic acid, and to the disodium salt.
No new safety test data were available for sodium lauraminopropionate; therefore, CIR Expert Panel did not reopen the final report to include this ingredient and reaffirmed the conclusion of insufficient data for sodium lauraminopropionate.
Chemistry
Definition and Structure
The definitions and structures of these substituted tertiary amines are presented in Table 1.
Names, CAS Registry Numbers, Definitions, and Structures of Dipropanoic Acid Ingredients.

Abbreviation: CAS, Chemical Abstracts Service.
Physical and Chemical Properties
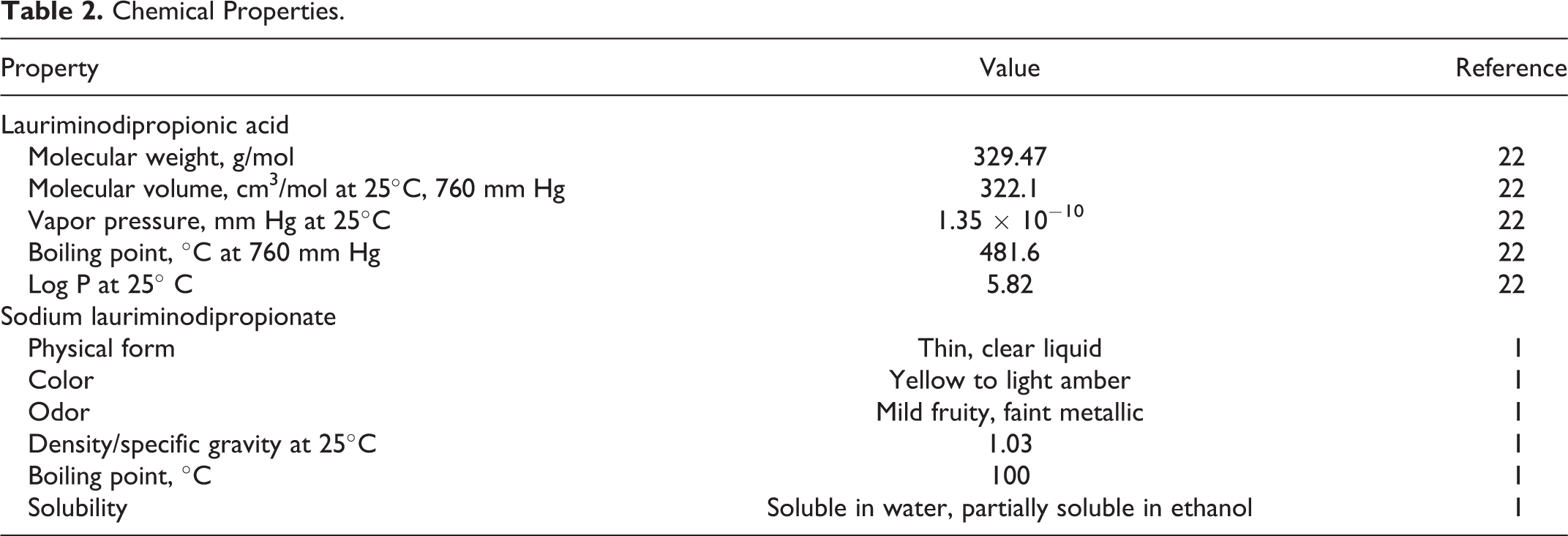
Lauriminodipropionic acid can exist in both anionic and cationic forms, depending on pH. 2 Additional physical and chemical properties of lauriminodipropionic acid and its sodium salts are summarized in Table 2.
Chemical Properties.
Method of Manufacture
The salts of lauriminodipropionic acid are produced in a 2-step synthesis. 2-4 First, dodecylamine (laurylamine) undergoes a Michael addition to acrylic acid or methyl acrylate, in methanol, to yield monopropionic acid methyl ester and dipropionic acid methyl ester. These esters can be separated by flash column chromatography. 3 The esters are then saponified with sodium hydroxide or sodium trimethylsilanolate to yield either the monosodium or the disodium salts. 2-4 Either of these salts can be acidified to the free acid under the appropriate pH.
Impurities
Sodium Lauriminodipropionate
The main impurity in the manufacture of sodium lauriminodipropionate is sodium acrylate. 4 Trace amounts of dodecyl-chain compounds associated with the starting material, dodecylamine, may also be present. Total impurities are typically found at levels less than 2% in the product as sold. Dodecylamine, polyacrylic acid, or sodium polyacrylate has not been detected in the batch samples of the commercial product. In the production of sodium lauriminodipropionate, trace levels of sodium lauraminopropionate may occur.
Use
Cosmetic
Lauriminodipropionic acid, sodium lauriminodipropionate, and disodium lauriminodipropionate are used as hair-conditioning agents and surfactant-cleansing agents in cosmetic formulations. 5 The monosodium and disodium salts are also used as antistatic agents, and the monosodium salt is used as a surfactant-foam booster.
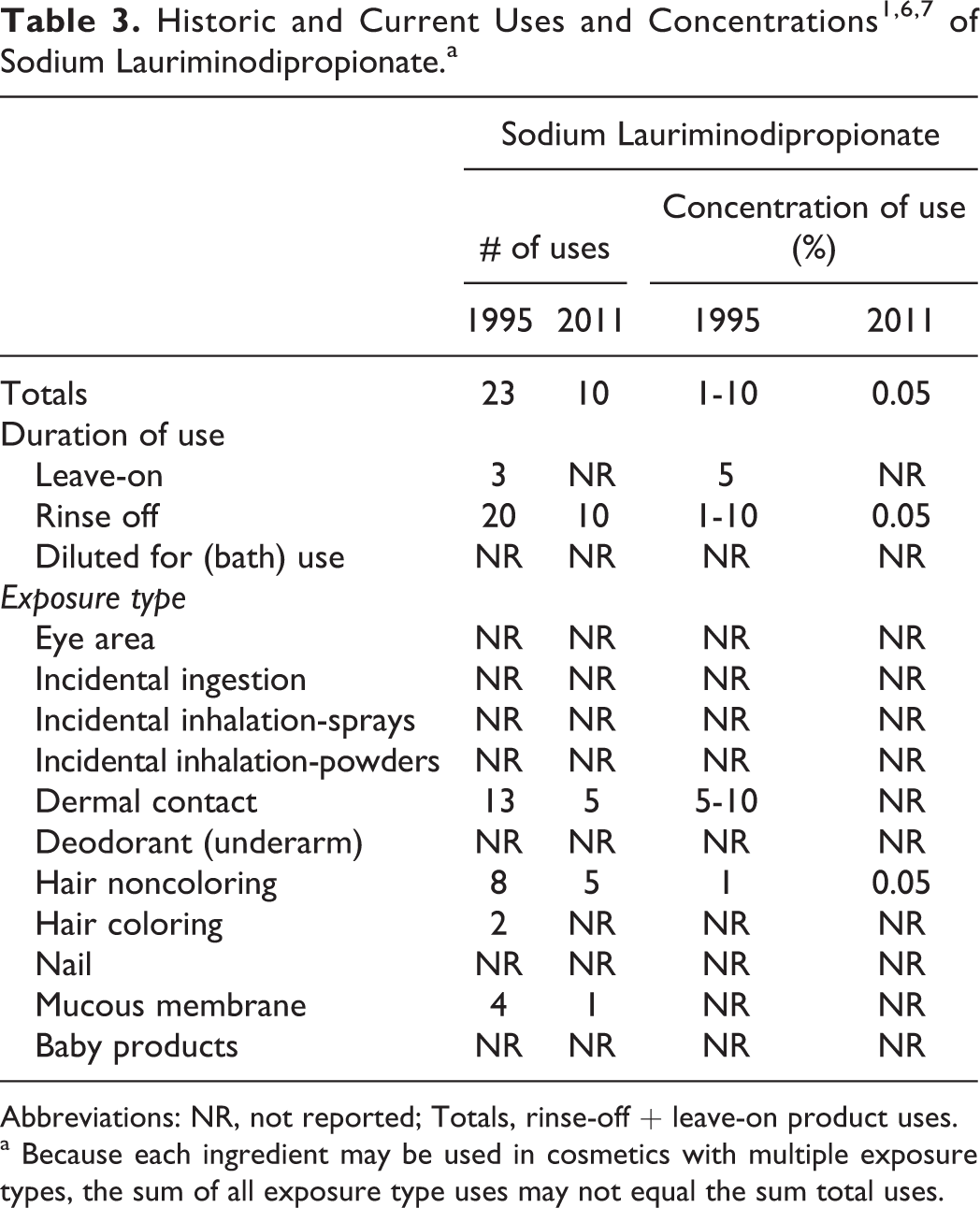
Table 3 presents the product formulation data for sodium lauriminodipropionate. A previous CIR safety assessment showed that, according to information supplied to the Food and Drug Administration (FDA) by the industry as part of the Voluntary Cosmetic Registration Program (VCRP), sodium lauriminodipropionate was used in 23 cosmetic formulations. 1 An industry survey reported the use of concentration range of 1% to 10%. 1 Current VCRP data indicate that sodium lauriminodipropionate is used in 10 cosmetic formulations, half of which are used in hair conditioners. 6 In a survey of current use concentrations conducted by the Personal Care Products Council, sodium lauriminodipropionate is used at a concentration of 0.05% in hair conditioners. 7
Abbreviations: NR, not reported; Totals, rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Currently, the VCRP data indicate that disodium lauriminodipropionate is used in 2 cosmetic formulations, both of which are face and neck preparations. 6 No uses are reported for lauriminodipropionic acid. 6 No use concentrations were reported for the acid or the disodium salt. 7
Noncosmetic
Sodium lauriminodipropionate has applications in heavy-duty alkaline cleaners, corrosion inhibitors, leather cleaners, and acid cleaners.
The Environmental Protection Agency (EPA) has ruled that the sodium and potassium salts of N-alkyl (C8-C18)-β-iminodipropionic acid, such as lauriminodipropionate, do not need a maximum permissible level when they are used as inert ingredients in pesticide formulations for pre- and postharvest applications in food crops (40 CFR §180).
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Sodium Lauriminodipropionate
Although no specific information was found on the absorption, distribution, metabolism, and excretion of sodium lauriminodipropionate, a possible metabolic scheme was described based on the features of a structure–activity relationship (SAR) assessment (see Figure 1). 8 According to the assessment, sodium lauriminodipropionate is likely to be metabolized by glucuronidation and/or N-dealkylation. Glucuronide products are known to be rapidly excreted and not likely to be biologically active. Although this reaction is conceivable, it is rare for carboxylic acids to be glucuronidated.
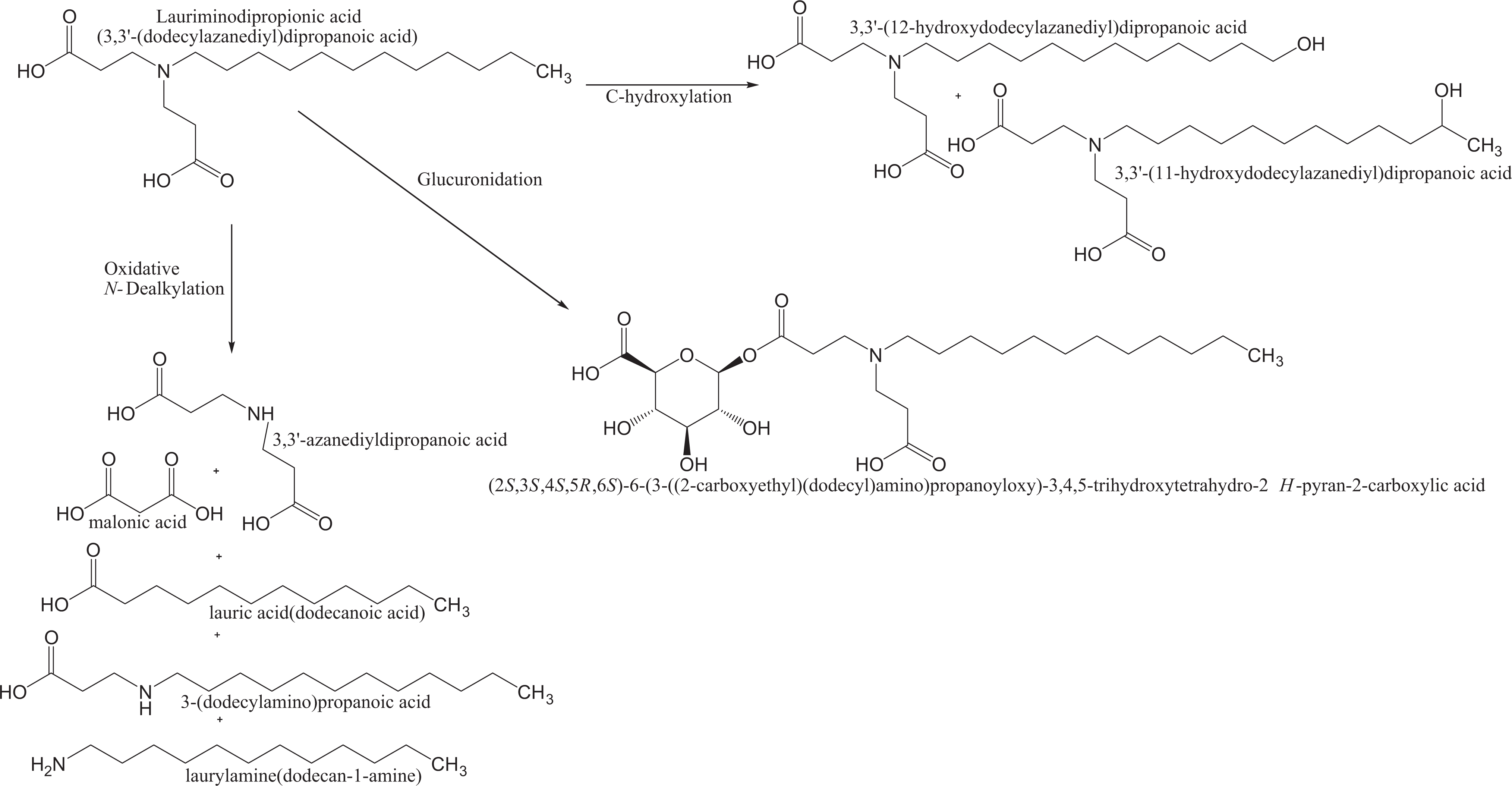
Predicted metabolic pathways of lauriminodipropionic acid, sodium lauriminodipropionate, and disodium lauriminodipropionate. 8
N-Dealkylation products are usually dietary chemicals, such as straight chain fatty acids, that undergo intermediary metabolism. Lauric acid and other straight chain fatty acids and related ingredients were found safe for use in cosmetic products. 9–11 In this scheme, the tertiary amine group of sodium lauriminodipropionate may undergo oxidative N-dealkylation to form 3,3 ′-azanediyldipropanoic acid, lauric acid, and/or 3-(dodecylamino)propanoic acid. Other N-dealkylation products formed may be laurylamine and/or malonic acid. This reaction is nearly always catalyzed by cytochrome P450s. The mechanism involves hydrogen abstraction and hydroxylation at a carbon atom α to the nitrogen atom.
In this scheme, sodium lauriminodipropionate may also undergo C-hydroxylation reactions on the alkyl chain. The products of this reaction may be 3,3’-(11-hydroxydodecylazanediyl) dipropanoic acid and 3,3’-(12-hydroxydodecylazanediyl) dipropanoic acid. On the longer alkyl chain, hydroxylation at the methylene group and hydroxylation at the terminal methyl group may be favorable. 8
Toxicological Studies
Acute Toxicity
Dermal—Nonhumans
Sodium Lauriminodipropionate
In acute dermal studies, no deaths or adverse reactions were observed in rabbits treated with 6.8 g/kg or 10.2 g/kg sodium lauriminodipropionate (10% active). 1
Oral—Nonhumans
Sodium Lauriminodipropionate
The oral median lethal dose (LD50) in albino mice treated with sodium lauriminodipropionate, 16% solids and pH 7.0, was estimated to be 17.8 mL/kg. 1
Repeated Dose Toxicity
Dermal—Nonhumans
Sodium Lauriminodipropionate
The percutaneous toxicity potential of sodium lauriminodipropionate was evaluated in a 91-day study on New Zealand White rabbits. 12 Male and female rabbits (5 each) received 2 mL/kg/d of 20% (w/w) solution in distilled water of 10.5% sodium lauriminodipropionate (35% of a 30% solution) in a foaming face wash. A control group of 5 male and 5 female rabbits received distilled water. The rabbits received the treatment or the control solutions daily, 5 days/week, to clipped, intact dorsal skin. Body weights were measured prior to study commencement, once a week during treatment, at 28 days, and at study termination. Hematology parameters were measured prior to study commencement and at study termination. All animals were killed at the end of the treatment period and underwent macroscopic and microscopic examination. Absolute and relative weights of the liver and kidney were determined.
Dermal irritation indicated by light to moderate erythema (starting on day 7), slight to moderate edema (starting on day 7), and slight to marked desquamation (days 7-14) was observed in the test group. No signs of systemic toxicity were observed. All animals survived until study termination. Body weights of the treated animals were comparable to the control animals. There were no treatment-related changes in hematology parameters or differences in organ weights when compared to controls. No macroscopic or microscopic changes that suggested systemic toxicity were observed at necropsy in any of the animals. This study concluded that although sodium lauriminodipropionate did not cause dermal toxicity, it was a dermal irritant. 12
Disodium Lauriminodipropionate
In a study of a hair dye formulation containing 1.5% disodium lauriminodipropionate, no systemic effects were observed in New Zealand White rabbits after topical application twice weekly for 13 weeks. 13
Reproductive and Developmental Toxicity
Sodium Lauriminodipropionate
Although there is no information available in the literature on the reproductive and developmental toxicity of sodium lauriminodipropionate, an SAR assessment was conducted which considered the potential metabolites of this ingredient (see Figure 1). 8 Sodium lauriminodipropionate is likely to be metabolized by glucuronidation and/or N-dealkylation. The glucuronide product for sodium lauriminodipropionate has not been assessed for reproductive or developmental toxicity; however, glucuronides in general have not been classified as developmental or reproductive hazards.
Among the products of N-dealkylation, straight-chain fatty acids, such as lauric acid, have been evaluated for reproductive and developmental hazards, and no adverse effects have been reported. 8,14 The CIR Expert Panel has reviewed several straight-chain fatty acids, including lauric acid and related ingredients, including plant-derived fatty acid oils, and has found these ingredients to be safe for use in cosmetic products. 9-11
Although the possible N-dealkylation product laurylamine has not been tested for reproductive toxicity, a similar compound, oleylamine, has been assessed and found not to affect the development in rats and rabbits, even at maternally toxic oral doses. 8 The developmental no-observed-adverse effect-level (NOAEL) was 80 mg/kg/d for rats and 30 mg/kg/d for rabbits in these studies. A mixture of tallow alkyl amines, another possible N-dealkylation product, did produce a decrease in offspring weight and a decrease in fertility at severely toxic levels that can be attributed to being secondary to the toxicity. In a previous assessment by the CIR Expert Panel, malonic acid, a potential product of N-dealkylation, was evaluated for developmental toxicity in rats assessed and determined to be safe for use in cosmetic products. 15 At concentration of 9% or 12% malonate in feed (approximate dosage for 9% malonate was 4.5 g/kg/d), low toxicity was observed in dams and fetuses. 16 A few malformations were reported for 4.5 g/kg/d, but these incidences were within historical control ranges, and this dose was considered the NOAEL for developmental toxicity.
Disodium Lauriminodipropionate
In a study of a hair dye formulation containing 1.5% disodium lauriminodipropionate, no embryotoxic or teratogenic effects were observed in Charles River CD rats that were exposed to the formulation on days 1, 4, 7, 10, 13, 16, and 19 of gestation. 13
Genotoxicity
In Vitro
Sodium Lauriminodipropionate
The mutagenicity potential of 30% sodium lauriminodipropionate was assessed in an Ames test with Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538. 17 The assay was performed with and without S9 metabolic activation. The concentration ranges tested were 0.003 to 10 μL/plate with S9 and 0.01 to 1.0 μL without S9. Because of lack of toxicity in test strains TA98, TA1535, and TA1538 without S9, additional assays were run using concentration ranges of 0.33 to 20 μL/plate. No positive responses were observed in any of the test strains, with or without S9. The vehicle control (water) and the positive controls 2-nitrofluorene, 2-aminoanthracene, sodium azide, and ICR-191 yielded expected results. It was concluded that sodium lauriminodipropionate was not mutagenic in this Ames test.
A Chinese hamster ovary (CHO) cell assay was performed to assess the potential of 30% sodium lauriminodipropionate to induce chromosome aberrations. 18 Following a range finding study, the concentration ranges for the 8-hour incubation study were 0.21, 0.28, and 0.38 μL/mL without S9 and 0.48, 0.63, and 0.84 μL/mL with S9. In the 12-hour incubation study, the concentration ranges were 0.28, 0.38, and 0.50 μL/mL without S9 and 0.63, 0.84, and 1.13 μL/mL with S9. Relative cloning efficiencies at the highest doses in the 8-hour study were 34% and 70% without and with S9, respectively, while in the 12-hour study, the values were 1% and 38% without and with S9, respectively. The controls, which were water, untreated cells, cyclophosphamide (CP), and triethylenemelamine (TEM), yielded expected results. The test material did not induce significant chromosome aberrations in either incubation period, with or without S9 metabolic activation. It was concluded that sodium lauriminodipropionate was not clastogenic.
Carcinogenicity
Published carcinogenicity data were not found nor were unpublished data provided.
Irritation and Sensitization
Irritation
Dermal—Nonhumans
Sodium Lauriminodipropionate
Sodium lauriminodipropionate, 10% solids, had a mean primary irritation index (PII) of 3.04/8 and was considered corrosive to the skin of rabbits due to eschar formation. In another study, sodium lauriminodipropionate, 16% solids at pH 7.0, was a moderate irritant to rabbit skin, with a PII of 2.17. 1
Ocular—Nonhumans
Sodium Lauriminodipropionate
As reported in a previous assessment, a solution of sodium lauriminodipropionate, 10% solids, was classified as mildly irritating to the eyes of rabbits without rinsing and practically nonirritating with rinsing. Another solution of sodium lauriminodipropionate, 16% solids at pH 7.0, was a moderate irritant. 1
Ocular—Humans
Sodium Lauriminodipropionate
The 2 shampoo formulations containing 3.56% active sodium lauriminodipropionate (11.86% of a 30% solution) were tested for ocular tolerance in 102 human participants. 19 In Phase I, the test materials were instilled into 1 eye of each participant at single rising concentrations (5% or 10%). The other eye received sterile water. In Phase II, the test material was instilled into 1 eye at a concentration determined to be likely tolerable from Phase I (10%), while the other eye received sterile water. Eyes were evaluated before instillation, and objective and subjective tolerance was measured immediately after instillation, between 30 and 60 seconds after instillation, 15 minutes, and 1 hour after instillation. Objective evaluations were measured with slit lamp examination, bulbar and conjunctival irritation, and assessment of lacrimation. Albumin levels in tears before and after application of the test material were evaluated by the turbidimetric method in Phase II only. Subjective evaluations included reports by the participants of stinging and itching sensation and epiphora. No ophthalmological lesions were observed with any of the test materials. The test materials also did not induce a significant increase in albumin levels in tears. Minimal observations of stinging or dryness sensations were made in both the phases of the study.
Sensitization
Nonhuman
Sodium Lauriminodipropionate
No sensitization was observed in guinea pigs injected with 0.1% sodium lauriminodipropionate intracutaneously in a previous assessment. 1
Human
Sodium Lauriminodipropionate
Sodium lauriminodipropionate was reported to be “practically nontoxic” to the skin and “minimally irritating” upon skin contact. It was also reported to be “minimally irritating” to the eye. Sodium lauriminodipropionate was “practically nontoxic” upon ingestion. 1
A human repeat insult patch test (HRIPT) was conducted (104 participants) to determine the potential of 2.2% sodium lauriminodipropionate (7.34% of a 30% solution) to induce contact sensitization. 20 The participants received 0.2-mL applications of a 2.0% foaming face wash solution in distilled water. Induction applications were made to the infrascapular region of the back with a 2-cm2 Webril pad portion of an adhesive dressing. The test sites were occluded. The patches were removed after 24 hours, and there were 9 consecutive applications. Following 2-week nontreatment period, the challenge application was applied to a previously untreated site for 24 hours, and the site was scored 24 and 48 hours after patch removal. During the induction and the challenge phases, responses were doubtful and barely perceptible erythema was noted. There were no other responses during the induction and challenge phases. The study concluded that there was no evidence of sensitization to a foaming face wash formulation containing 2.2% sodium lauriminodipropionate.
In another HRIPT, the potential of 3.5% active sodium lauriminodipropionate (11.67% of a 30% solution) to induce contact sensitization was studied in 116 participants. 21 The participants received 0.5-mL applications of 2.5% shampoo formulation solution in distilled water. One participant had mild to moderate erythema during the induction and had a positive response at challenge in the original test site. In a subsequently rechallenge patching with the test material, with a 2.5% w/v aqueous solution of another shampoo formulations containing 3.5% sodium lauriminodipropionate and a 0.4% w/v solution of the fragrance used in the original test formulation in mineral oil, mild erythema was observed similar to the shampoo formulation at the 48-hour scoring, which resolved completely at the 96-hour scoring. No other responses were observed in this participant. Mild erythematous reactions were reported in several other participants during the induction phase as well as during challenge. These reactions, and the ones observed in the participant that underwent rechallenge, were determined to be irritant responses consistent with the surfactant use. The study concluded that there was no evidence that the shampoo formulation containing 3.5% sodium lauriminodipropionate caused delayed contact hypersensitivity.
Summary
Lauriminodipropionic acid and its sodium salts are reported to function as surfactants, hair-conditioning agents, and antistatic agents in cosmetic formulations. A possible metabolic scheme based on features of a SAR assessment suggested that sodium lauriminodipropionate is likely to be metabolized by glucuronidation and/or N-dealkylation.
Current FDA VCRP data indicate that sodium lauriminodipropionate is used in 10 cosmetic formulations, mainly in hair conditioners. The current concentration of use for sodium lauriminodipropionate is 0.05% in hair conditioners. Disodium lauriminodipropionate has been reported to have uses in 2 face and neck preparations. No uses are reported for lauriminodipropionic acid. No use concentrations were reported for the disodium salt or the acid.
Sodium lauriminodipropionate is used in heavy-duty alkaline cleaners, corrosion inhibitors, leather cleaners, and acid cleaners. The EPA has ruled that the sodium and potassium salts of N-alkyl (C8-C18)-β-iminodipropionic acid do not need a maximum permissible level when they are used as inert ingredients in pesticide formulations for pre- and postharvest applications in food crops.
In studies of 10% active solutions of sodium lauriminodipropionate, the oral LD50 for rats was 31.3 g/kg, and the dermal LD50 was greater than 10.2 g/kg; the oral LD50 for mice of a 16% solids solution was estimated to be 17.8 mL/kg.
A 91-day study in rabbits that received 20% (w/w) solution in distilled water of 10.5% sodium lauriminodipropionate (35% of a 30% solution) concluded that although sodium lauriminodipropionate did not cause dermal or systemic toxicity, it was a dermal irritant. No systemic effects were observed in rabbits that received topical applications of a hair dye formulation containing 1.5% disodium lauriminodipropionate for 13 weeks.
A SAR assessment conducted for potential metabolites of sodium lauriminodipropionate concluded that the metabolites were not developmental or reproductive toxicants. No embryotoxic or teratogenic effects were observed in a developmental study in rats on a hair dye formulation containing 1.5% disodium lauriminodipropionate. Sodium lauriminodipropionate was not mutagenic in an Ames test, nor was it clastogenic in a Chinese hamster ovary cell assay.
Sodium lauriminodipropionate at 10% active solution was severely irritating to the skin of rabbits. Sodium lauriminodipropionate at 16% solids was a moderate irritant to rabbit skin. Sodium lauriminodipropionate was irritating to the eyes of rabbits. Two shampoo formulations containing 3.56% active sodium lauriminodipropionate did not cause ocular lesions or induce a significant increase in albumin levels in tears in humans. Minimal observations of stinging or dryness sensations were made in both phases of the study.
There was no evidence of sensitization to sodium lauriminodipropionate in studies with guinea pigs. A foaming face wash containing 2.2% active sodium lauriminodipropionate and a shampoo formulation containing 3.5% active sodium lauriminodipropionate did not cause delayed contact hypersensitivity in HRIPT studies.
Discussion
A safety assessment for sodium lauriminodipropionate and sodium lauraminopropionate published by CIR in 1997 concluded that the available data were insufficient to support their safety in cosmetics. The Expert Panel reopened the final report on sodium lauriminodipropionate based on new data and determined that the report should also address the safety of lauriminopropionic acid and disodium lauriminodipropionate.
The Panel reaffirmed the conclusion of insufficient data to determine the safe use of sodium lauraminopropionate in cosmetics.
The CIR Expert Panel considered that the available single-dose and repeated-dose animal studies, including reproductive and developmental toxicity studies, were supportive of the safety of sodium lauriminodipropionate. The Expert Panel noted the absence of carcinogenicity data but considered the data demonstrating that sodium lauriminodipropionate was not mutagenic or clastogenic in in vitro genotoxicity studies adequate to support the safety of these ingredients.
The Expert Panel noted gaps in the available safety data for lauriminodipropionic acid and its disodium salt. The available data on the monosodium salt were sufficient, however, and because any of the molecules would assume the same form once in solution, based on pH, any toxicological effects would be the same for any of the 3. The Panel also noted that there is potential for the aminopropionate moiety to be present as a trace impurity in the iminodipropionate ingredient. Because these iminodipropionate ingredients are used only in rinse-off products, such trace levels did not raise any safety issues.
Conclusion
The CIR Expert Panel concluded that lauriminodipropionic acid, sodium lauriminodipropionate, and disodium lauriminodipropionate are safe as cosmetic ingredients in the present practices of use and concentration. Were the acid and the disodium salt to be used in the future, the expectation is that they would be used at concentrations similar to the monosodium salt.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.