Abstract
Introduction
First-line chemotherapy for metastatic colorectal cancer typically involves a fluoropyrimidine, like 5-fluorouracil (5-FU), along with a folate agent, levoleucovorin. However, calcium-based levoleucovorin with 5-FU necessitates sequential infusion due to incompatibility, leading to calcium carbonate precipitation and potential IV catheter occlusion. In contrast, sodium-based levoleucovorin (disodium levoleucovorin-Khapzory) exhibits higher solubility in the low pH environment of 5-FU, enabling combination within a single IV bag for simultaneous infusion. This study aims to assess the safety of combining different concentrations of disodium levoleucovorin with 5-FU to create a single IV admixture bag or single pump Y-site, without risk of precipitate formation and catheter occlusion.
Methods
Compatibility of admixture 5-FU and disodium levoleucovorin in a 0.9% sodium chloride IV bag was evaluated, focussing on clarity (suspension, precipitation, and haziness). Particulate matter analysis was conducted at 25°C/60% relative humidity, for 29 samples at five timepoints. Defined pass criteria included a minimum of 6000 particles ≥10 µm per container and 600 particles ≥25 µm.
Results
All prepared concentrations remained clear for up to 72 h with no observed suspension, precipitation or haziness at any concentration or time point.
Conclusions
Combining 5-FU and disodium levoleucovorin in admixture IV bags eliminates the risk of catheter occlusion associated with calcium-based levoleucovorin formulations. This approach offers a more favorable operational and safety profile, enhancing convenience for patients and cost-efficiency for institutions.
Introduction
Colorectal cancer (CRC) stands as the second leading cause of cancer-related death in the United States (US) with approximately 147,950 new cases and 53,200 deaths recorded in 2020. 1 The cornerstone of treatment for metastatic CRC (mCRC) often involves fluoropyrimidine-based chemotherapy combined with a folate agent. The modulation of fluoropyrimidine-based chemotherapy by folate agents has shown enhanced tumor cytotoxicity in both preclinical and clinical studies, making it a fundamental component of CRC management. 2
The commonly used combination includes 5-fluorouracil (5-FU) and leucovorin, which is a racemic mixture of dextrorotary and levorotary isomers. Levoleucovorin, the pharmacologically active levorotary isomer, has been demonstrated to be pharmacokinetically identical to leucovorin. However, in-vitro studies have highlighted the rapid conversion of the levorotary isomer to a biologically available methyl-tetrahydrofolate form. 3
Until 2018, available leucovorin or levoleucovorin products in the US were calcium salt-based, namely calcium- d,l-leucovorin or calcium levoleucovorin. However, the approval of disodium levoleucovorin (marketed as Khapzory™) in 2018 under the 505(b)(2) regulatory pathway introduced a sodium-based alternative. The formulation, devoid of calcium, comprises levoleucovorin, sodium hydroxide, and mannitol. Notably, disodium levoleucovorin is the sole sodium-based levoleucovorin approved in the US, recognized as therapeutically equivalent to other levoleucovorin products.
Clinical studies, such as the one conducted by Bleiberg et al., have compared calcium-based and sodium-based formulations, revealing potentially favorable outcomes with the latter, including higher response rates and longer overall survival durations. 4
Traditionally, the combination of levoleucovorin with 5-FU in mCRC treatment regimens required sequential infusion protocols due to the incompatibility of calcium-based levoleucovorin with 5-FU. However, the introduction of sodium-based levoleucovorin offers a solution to this issue, as its higher solubility in the low pH environment of 5-FU solutions enables simultaneous infusion or combination within the same admixture with 5-FU. This approach has demonstrated safety and efficacy in clinical settings. 5
While stability studies have previously established the viability of this combination for up to two weeks, a comprehensive analysis of the particulate matter, essential for supporting the use of admixtures in clinical practice, remains lacking. 6 Therefore, the current study aims to investigate the stability and dilution compatibility of 5-FU and disodium levoleucovorin admixtures in 0.9% sodium chloride infusion solutions stored in IV bags.
Methods
Materials and reagents
Disodium levoleucovorin (Khapzory) (50 mg/mL) was provided by Acrotech Biopharma, LLC, East Windsor, NJ, USA. 5-FU (500 mg/10 mL) was obtained from Intas Pharmaceuticals Ltd Sodium chloride for injection (saline) 0.9% used to prepare the sample mixtures, was commercially purchased from B. Braun Medical Inc. All other chemicals, reagents and laboratory equipment used were of analytical or higher grade and procured from commercial sources.
Instrumentation
High-performance liquid chromatography (HPLC) with the Waters e2695 Separations Module (Waters, USA) and either a 2998 photodiode array detector or a 2489 UV detector was utilized for sample analysis and drug concentration detection. Chromatographic data were managed using Empower Pro Chromatography software. Additionally, an inoLab pH meter was employed to determine solution pH, while particulate matter analysis was conducted using the Hiac particulate matter instrument (model 9703) with defined pass criteria, according to the European Pharmacopoeia standards. 7
Chromatographic conditions
YMC analytical columns were used with the mobile phase comprising acetonitrile and deionized water in varying ratios. The flow rate was maintained at 1.0 mL/min, and the column temperature was set at 25°C. Injection volume (20 μL/sample), sample temperature (5°C) and detection wavelength (245 nm) were also specified.
Disodium levoleucovorin, 5-FU, and buffer solution preparation
Preparation of stock solutions of disodium levoleucovorin and 5-FU, as well as the buffer solution, was conducted at Eugia Pharma Research Center (Hyderabad, Telangana) with detailed procedures provided for each. The stock solution of disodium levoleucovorin 50 mg/mL was prepared using 175 mg/mL vials and reconstituting each vial with 3.5 mL of 0.9% sodium chloride solution. Molecular formula, molecular weight (C20H21N7Na2O7, 514.41 g/mol). The 5-FU solution was prepared to 50 mg/mL by weighing and transferring 5 mg fluorouracil standard into a 50 mL volumetric flask, dissolving in a 40 mL diluent (water). Molecular formula, molecular weight (C4H3FN2O2, 130.078 g/mol). The buffer solution was prepared by dissolving 6.8 g of potassium dihydrogen phosphate in 1000 mL of water, adjusted to pH 5.7 (±0.1) with 5 M potassium hydroxide solution, and filtered through a 0.45µ membrane filter. For the mobile phase, acetonitrile and buffer were mixed in a ratio of 50: 950 (v/v). The 5 M potassium hydroxide solution was prepared by dissolving 28.0 g of potassium hydroxide pellets in 100 mL of water.
Admixture assessments
Assessments, including visual inspection, light obscuration particle count test, and chromatographic analysis, were performed at specified time intervals on prepared admixture samples. Detailed sample concentrations and preparation methods are listed in Table 1. Each admixture sample listed in Table 1 was visually inspected and then analyzed by Light Obscuration Particle Count Test using the United States Pharmacopeia (USP) 788 standards. Particulate matter analysis was performed on admixture solution at time intervals of 0, 8, 24, 48, and 72 h and samples were maintained at room temperature ranges of 25 ± 2°C and relative humidity of 60% ± 5%. Similarly, admixture samples were assessed for physical appearance, Assay (HPLC) and related substances (HPLC) for the compatibility study at 0, 8, 24, 48, and 72 h post-preparation at temperature ranges of 25 ± 2°C, and relative humidity of 60% ± 5%.
Admixture sample concentrations of levoleucovorin (Khapzory) and 5-FU included in particulate matter analysis.
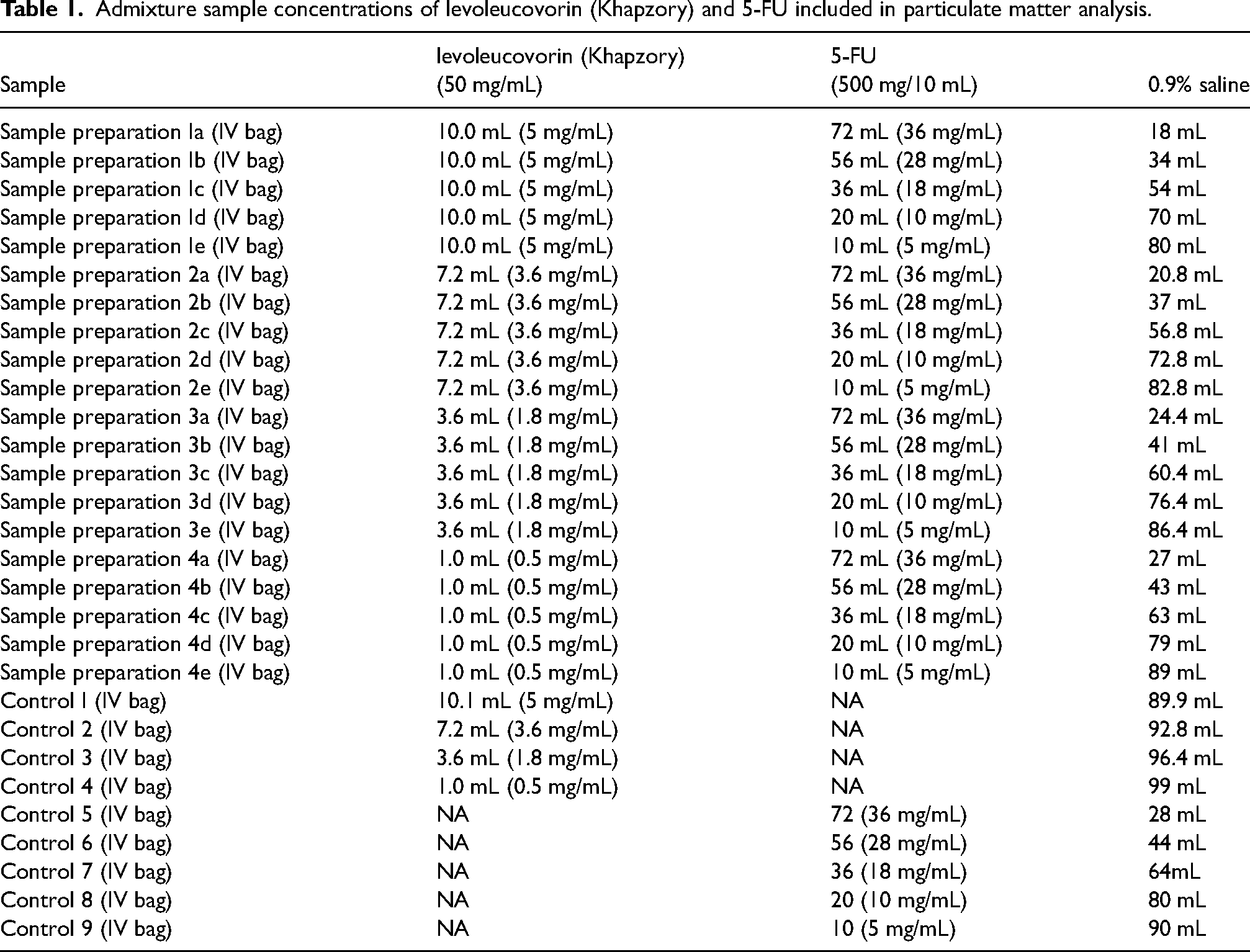
To assess dilution stability of the prepared admixtures consisting of levoleucovorin and 5-FU listed in Table 1, the following variables were assessed and documented: Physical appearance, Assay (HPLC), related substances (HPLC), fluorouracil-related compound (EP-Impurity-A and -B), Uracil (EP-Impurity-C), 5-Methoxyuracil (EP-Impurity-D), fluorouracil related compound E (EP-Impurity-E), any individual unspecified impurity and total impurities.
The validation parameters for the determination of related substances were specified, including specificity, limit of detection, linearity, precision, and system suitability.
Specified related substances included: folic acid (FA), 10-formylfolic acid (FF A), 4-aminobenzoylglutamic acid (ABGA), formyltetrahydropteroic acid (FTHPA), diformyltetrahydrofolic acid (DFTHF A), 10-formyldihydrofolic acid (FDHFA), 7, 8-dihydrofolic acid (DHFA), and methylenetetrahydrofolic acid (CH2-THF A).
The test methods described have been validated for specificity, the limit of detection, the limit of quantitation, linearity, precision (System Precision, Method Precision and Intermediate Precision (Ruggedness)), stability of solutions and system suitability. The results of these studies have been found to meet the predetermined acceptance criteria, therefore, the validated method is specific, linear and precise for the determination of related substances in fluorouracil injection USP 50 mg/mL.
Results
Admixture appearance and presence of particulate matter
All sample solutions remained clear and free of any particulates for up to 72 h across all prepared concentrations. No suspension, precipitation, or haziness was observed at any tested concentrations, or prespecified time points.
Particulate matter analysis adhered to the regulation limits of the European Pharmacopoeia, 7 (not more than 6000 ≥ 10 µm particles per container ≤ 100 mL and not more than 600 ≥ 25 µm particles per container ≤ 100 mL) with no sample nearing the specified criteria for failing the size and amount of allowable particles. The results of the particulate matter analysis, by the Hiac particulate matter instrument (model 9703), for each concentration are presented in Figure 1(a) and 1(b) and Supplemental Table S1.
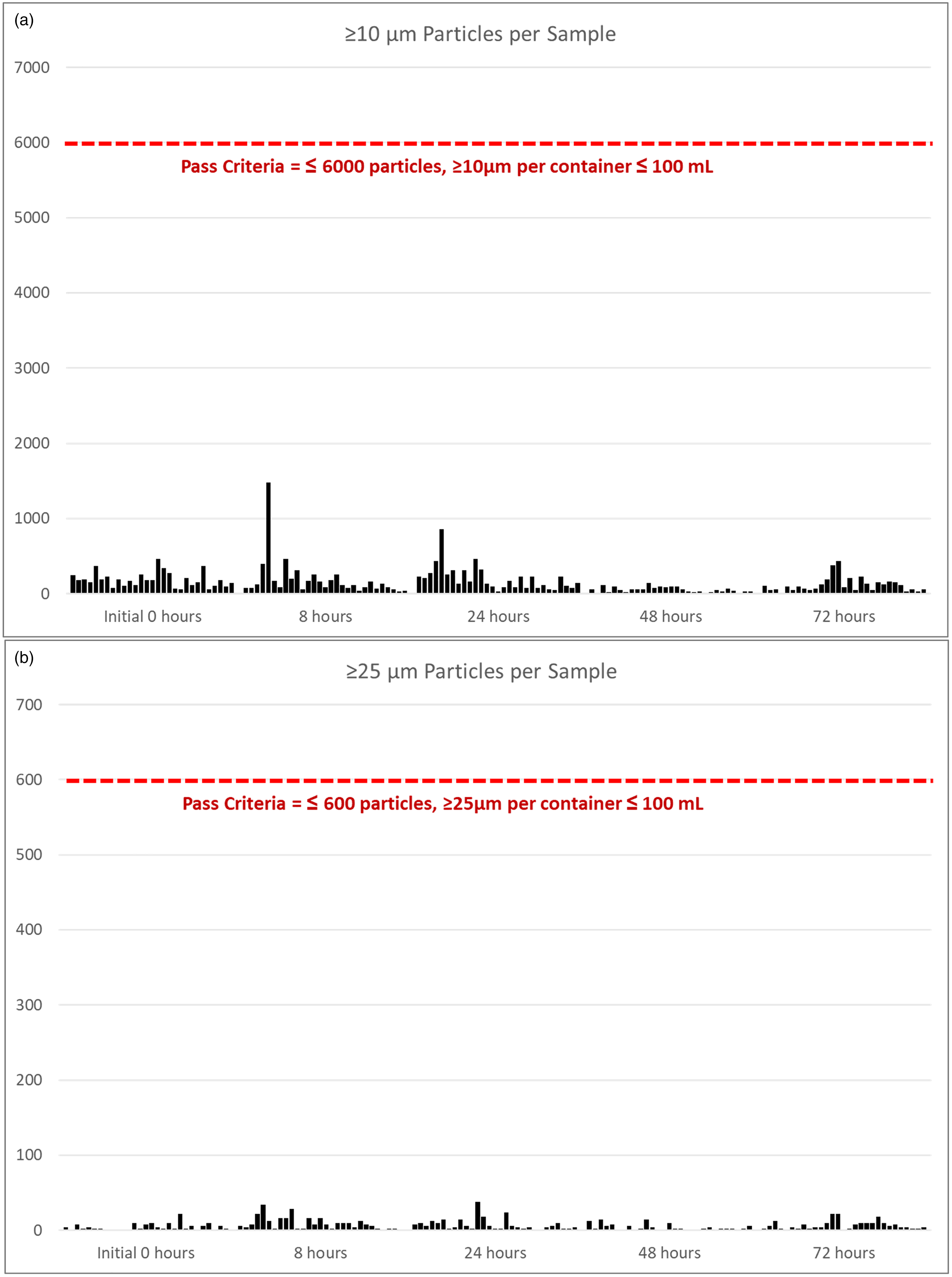
(a) Particulate matter analysis: ≥ 10 µm particles in each sample; (b) Particulate matter analysis: ≥ 25 µm particles in each sample.
Assay (%) of levoleucovorin and 5-Fu, and physical appearance
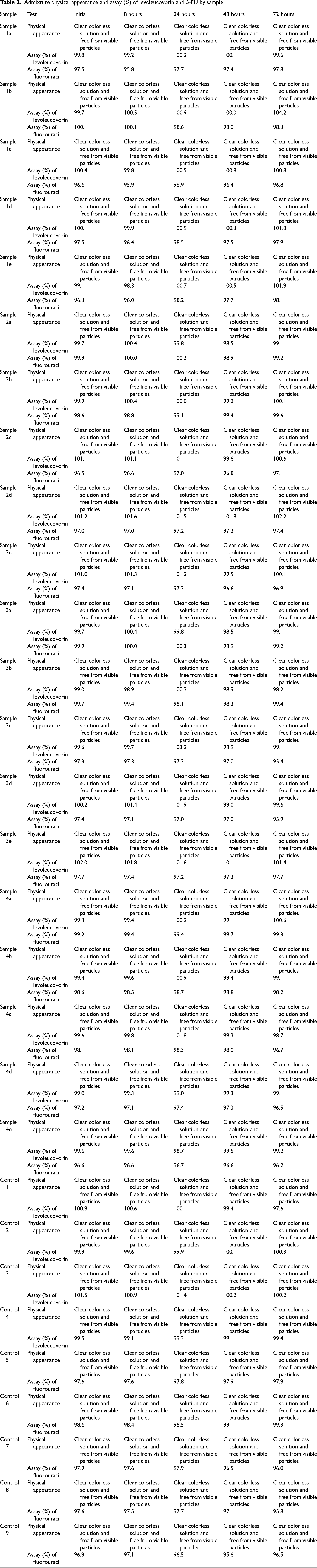
Table 2 presents the physical appearance and assay percentages of levoleucovorin and 5-FU for each sample at different time points. All samples maintained a clear, colorless appearance, free from visible particles across all timepoints throughout the evaluation period. The assay percentages of levoleucovorin and 5-FU remained within the accepted limits of 90.0 and 110.0, with values ranging between 95.8% and 102.0%, indicating the stability and quality of the admixture solutions.
Admixture physical appearance and assay (%) of levoleucovorin and 5-FU by sample.
Discussion
Currently, the foundation of treatment for mCRC includes chemotherapy and a combination of fluoropyrimidine, such as 5-FU, and a folate agent, levoleucovorin. The addition of folinic acid to 5-FU has been shown to both enhance the therapeutic effectiveness and decrease the toxicity of 5-FU in chemotherapy patients. For many years, the combination of calcium-based levoleucovorin with 5-FU has required a sequential infusion protocol due to the incompatibility of calcium-based levoleucovorin with 5-FU within the IV admixture, resulting in calcium carbonate precipitation and potential IV catheter occlusion. 8 We hereby confirm the compatibility of sodium-based levoleucovorin coadministration with 5-FU by way of clarity of the admixture and lack of particulate matter formation within the accepted standard for IV solutions.
In 2018, the sodium-based levoleucovorin agent, Khapzory (disodium levoleucovorin), was approved by the US FDA, indicated for rescue after high-dose methotrexate therapy in patients with osteosarcoma and for the treatment of patients with metastatic CRC in combination with 5-FU. Bioequivalence has been previously established for disodium levoleucovorin and calcium levoleucovorin formulations.5,9 Disodium levoleucovorin has a higher solubility in the low pH environment of 5-FU solutions, making both agents compatible and allowing for their combination in a single IV bag for simultaneous infusion. The combined, simultaneous treatment has demonstrated comparable response rates and toxicity levels with no catheter occlusion, confirming the compatibility of disodium levoleucovorin with 5-FU. 4 Additionally, simultaneous administration has the potential to improve clinical outcomes. Di Paolo et al. demonstrated the pharmacological differences between the sequential and simultaneous administration of levoleucovorin in in-vivo and in-vitro experiments, reporting that simultaneous, but not the consecutive, in-vitro and in-vivo combination of 5-FU and both folinate salt formulations enhanced the antiproliferative effects of the drugs. 9
Although this combination has been evaluated clinically and stability studies have been previously conducted, the potential precipitates within the admixture bag have not been validated by high-performance liquid chromatography analysis.
The current study confirmed the virtual absence of particulate matter of different concentrations of disodium levoleucovorin in combination with 5-FU, establishing that both can be safely mixed in one IV admixture bag for infusion, or Y-site using a single pump without the risk of precipitate formation and catheter occlusion.
The pass criteria for particulates ≥10 µm present in the admixture was ≤6000 per container, and the highest number observed was 1478 (range, 16–1478) at any time point and 366 (range, 54–366) at the time of initial preparation of the admixture IV bags of Khapzory and 5-FU, allowing us to draw the conclusion that this combination does not pose catheter occlusion risk, like calcium-based levoleucovorin formulations coadministered with 5-FU. As would be expected, no calcium carbonate precipitation was formed in the solution within the plastic IV bag, as there is no free or excess calcium present in the product. The plastic IV bag is an important consideration because when calcium-based levoleucovorin is dissolved in the low pH of 5-FU solution, it creates an environment where any CO2 that partitions through the plastic, or in catheter tubing, and combines with the free Ca++ ion will then form the insoluble calcium carbonate which precipitates out of the solution. In previous studies, higher drug concentrations have been associated with greater incompatibility than lower drug concentrations. 10 However, we did not observe any notable precipitation in any of the admixtures over the course of the experiment, including those containing the highest concentrations.
In addition to the demonstrated efficacy of this combination of 5-FU and disodium levoleucovorin, these findings have important clinical implications. The ability to coadminister levoleucovorin and 5-FU will reduce patient “chair time,” as compared to sequential infusion. This reduction in “chair time” is a recognized and validated variable of interest from an institutional operations and financial standpoint, in addition to demonstrated improvements in patient satisfaction.11,12 A better understanding of the operational benefits to institutions and characterization of the impact that coadministration may have on patient satisfaction are important areas of future study.
Conclusions
The preparation of the combination admixture IV bags of Khapzory (disodium levoleucovorin) and 5-FU does not pose a catheter occlusion risk, like calcium-based levoleucovorin formulations coadministered with 5-FU. Therefore, the advantages of coadministration of the sodium-based formulation, over a calcium-based formulation, in combination with 5-FU may be a more favorable operational profile and safety features, in addition to improved convenience for patients and cost and time-saving for institutions.
Supplemental Material
sj-docx-1-opp-10.1177_10781552241262240 - Supplemental material for In vitro compatibility of admixture solutions of 5-fluorouracil and khapzory (disodium levofolinate) in a single infusion bag
Supplemental material, sj-docx-1-opp-10.1177_10781552241262240 for In vitro compatibility of admixture solutions of 5-fluorouracil and khapzory (disodium levofolinate) in a single infusion bag by Hayder Saeed in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgments
The authors thank Benjamin J. Lee, Pharm.D. and April Ingram for their valuable editorial and writing contributions.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Acrotech Biopharma Inc.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
