Abstract
A recombinant vaccine (rF1V) is being developed to protect adults 18 to 55 years of age from fatal pneumonic plague caused by aerosolized Yersinia pestis. A comprehensive series of studies was conducted to evaluate the general toxicity and local reactogenicity of the rF1V vaccine prior to first use in humans. Toxicity was evaluated in CD-1 mice vaccinated with control material and three dosage concentrations of rF1V with or without Alhydrogel® by intramuscular (IM) injection on Study Days 1, 29, 57 and 71 in a volume of 0.1 mL. Total immunizing protein given in each dose was 0, 20 or 60 μg/animal. Local reactogenicity was evaluated in mice at the dosages given and in New Zealand white (NZW) rabbits using the same injection volume and formulations (40, 80, 160 and 320 μg/mL total antigen and 0.3% (w/v) Alhydrogel®) intended for human use (0.5 mL). The rF1V vaccine produced no apparent systemic toxicity and only transient edema and erythema at the injection site. Together these results indicated a favorable safety profile for rF1V and supported its use in a Phase 1 clinical trial.
Keywords
Introduction
Plague is caused by the gram-negative bacterium
Currently, no licensed plague vaccines are available for human use in the United States. The previously available US Pharmacopeia vaccine was a killed, whole-cell vaccine that provided protection against bubonic plague but not pneumonic plague. 6 –9 Significant local and systemic reactions were reported following administration of that vaccine. 8,9 Live-attenuated vaccines have been used in several countries; however, due to unacceptable reactogenicity associated with these vaccines, they have not been licensed for use in the United States. 10
Efforts to develop new plague vaccines have focused primarily on the proteins that play an important role in
A recombinant plague vaccine (rF1V) composed of the F1 capsular protein and the V virulence protein fused into a single protein and formulated with Alhydrogel is being developed by the US Department of Defense to protect military personnel aged 18 to 55 years against battlefield exposures to aerosolized
Typically, only a single species is required for vaccine safety evaluations. Mice were chosen as a model for evaluation, since the pathology of experimental plague in mice is similar to that in humans and since the immunogenicity of recombinant plague vaccines in mice has been demonstrated. 3,7 Evaluation of the response to a full human dose volume of 0.5 mL could not be conducted in the mouse; therefore, a separate study in rabbits was performed to evaluate local reactogenicity to the full human dose volume. The overall safety assessment included the evaluation of general toxicity following repeat-dose administration in mice and local reactogenicity following administration of a full human dose of vaccine in rabbits. The results of these assessments showed that the rF1V vaccine produced no apparent systemic toxicity and only mild transient redness and inflammation at the injection site.
Methods
All studies were conducted at Gene Logic (Gaithersburg, Maryland) in compliance with Good Laboratory Practices. The Gene Logic Institutional Animal Care and Use Committee and the United States Army Medical Research and Materiel Command Animal Care and Use Review Office approved the use of mice and rabbits in these studies.
Animals
Male and female CD-1 mice were purchased from Harlan Sprague Dawley Inc (Indianapolis, Indiana) and were used for the evaluation of the general toxicity and immunogenicity of the rF1V vaccine candidate. Mice were approximately 8 weeks of age and weighed from 23 to 39 g (male) and 20 to 32 g (female) at the time of first dosing. Mice were housed individually in polycarbonate cages with Sanichip-certified hardwood bedding and provided with Certified Global Harlan Teklad Laboratory 2018 Diet and fresh tap water, ad libitum, throughout the study.
Male New Zealand White rabbits were purchased from Harland Sprague Dawley Inc (Oxford, Michigan) and were used to evaluate local reactogenicity of the full human vaccine dose volume (0.5 mL). Rabbits were approximately 13 weeks of age and weighed between 2.4 and 3.0 kg at the time of first dosing. Rabbits were housed individually in stainless steel cages and provided with Certified Global Harlan Teklad Laboratory 2030 Diet and fresh tap water, ad libitum, throughout the study.
General procedures for animal care and housing were in accordance with the
Test and Control Articles
For the repeat-dose toxicity study in mice, the rF1V test articles were supplied formulated at concentrations of 200 and 600 µg/mL in phosphate-buffered saline (PBS; 5 mmol/L phosphate and 150 mmol/L sodium chloride buffer) with and without 0.3% (w/v) Alhydrogel. The placebo control was sterile PBS with and without 0.3% (w/v) Alhydrogel.
For the reactogenicity study in rabbits, phase 1 clinical rF1V lots formulated at 40, 80, 160, and 320 µg/mL in PBS with 0.3% (w/v) Alhydrogel, pH 7.2 to 8.0, were used as the test articles. The 600 µg/mL rF1V without Alhydrogel formulation was diluted to 320 µg/mL in PBS and was used as an rF1V-only control. The placebo control was PBS with 0.3% (w/v) Alhydrogel. The rF1V-only formulation and the placebo control were prepared fresh on each day of dosing.
All materials were maintained at 2°C to 8°C prior to and throughout the study. The vaccine antigens, adjuvant, and PBS used to formulate the rF1V test articles were manufactured according to Current Good Manufacturing Practices. All materials were maintained on ice throughout the dose administration.
Vaccination
Mice in each of the 6 study groups (Table 1) administered the appropriate the vaccine (20 or 60 µg) or placebo control by intramuscular (IM) injection on days 1, 29, 57, and 71 in a volume of 0.1 mL. To minimize tissue damage, injections were alternated between 4 different anatomic sites, right quadriceps muscle, left quadriceps muscle, right shoulder area, and left shoulder area.
Repeat-Dose Study Groups.
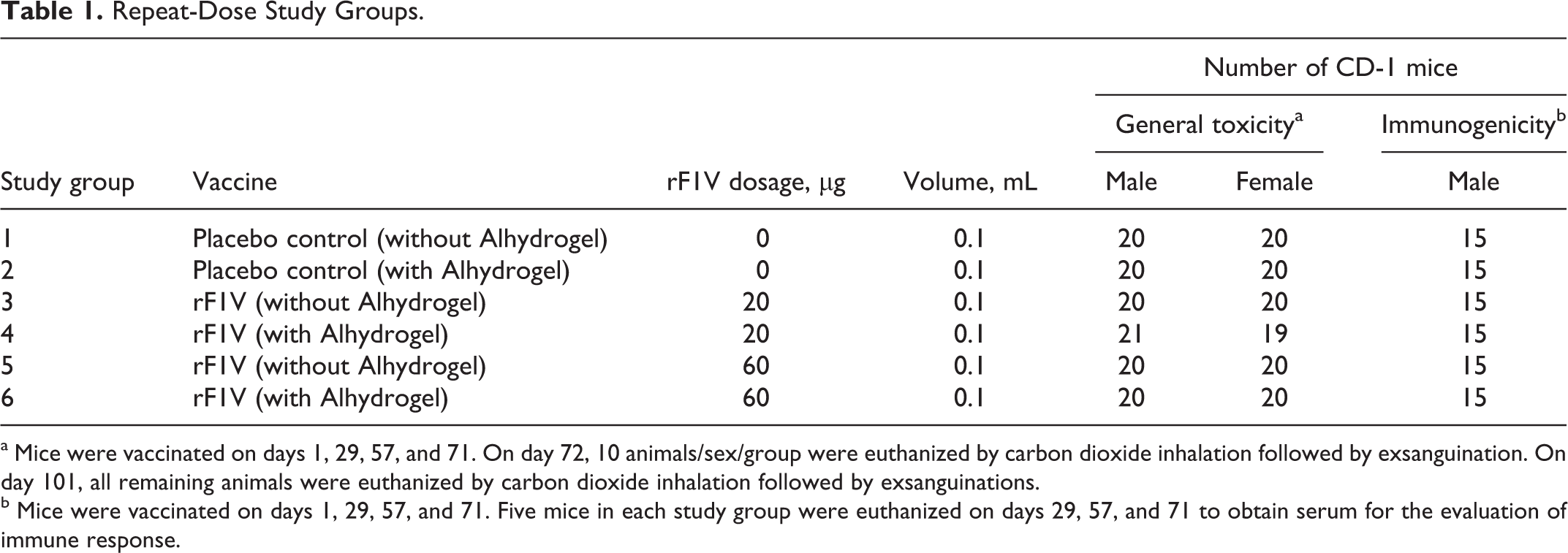
a Mice were vaccinated on days 1, 29, 57, and 71. On day 72, 10 animals/sex/group were euthanized by carbon dioxide inhalation followed by exsanguination. On day 101, all remaining animals were euthanized by carbon dioxide inhalation followed by exsanguinations.
b Mice were vaccinated on days 1, 29, 57, and 71. Five mice in each study group were euthanized on days 29, 57, and 71 to obtain serum for the evaluation of immune response.
Rabbits were divided into 6 study groups (Table 2) and administered 20, 40, 80, or 160 µg of the vaccine with Alhydrogel, 160 µg of the vaccine without Alhydrogel or placebo control by IM injection on day 1 or days 1 and 15 in a volume of 0.5 mL. Vaccinations were administered in the right (day 1) or left (day 15) quadriceps muscle.
Rabbit Local Reactogenicity Study Groups.
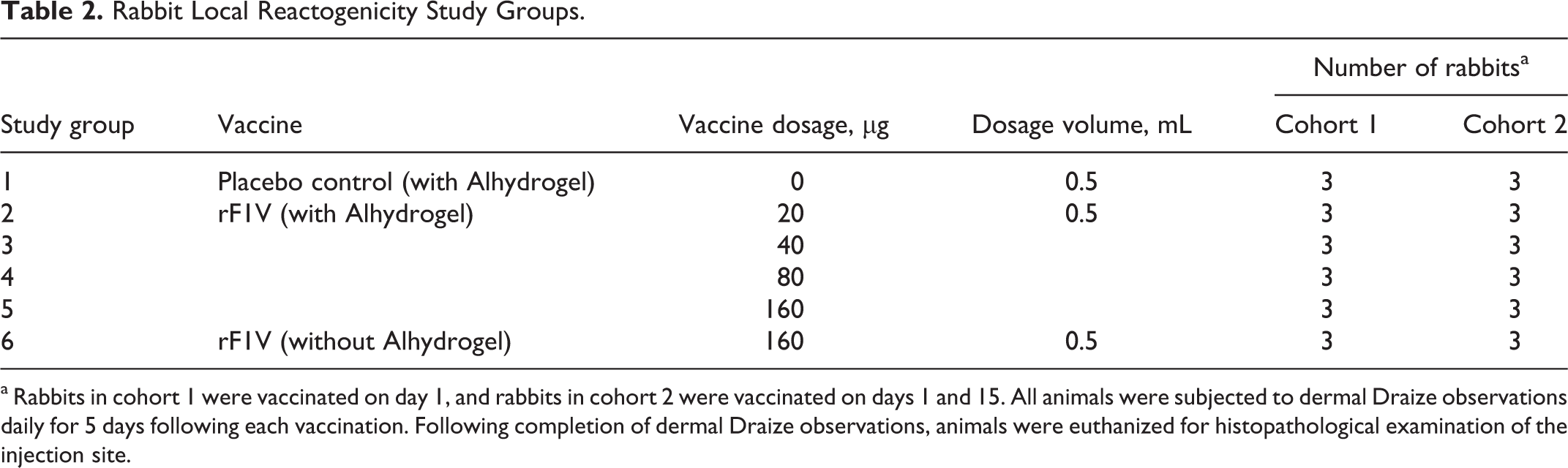
a Rabbits in cohort 1 were vaccinated on day 1, and rabbits in cohort 2 were vaccinated on days 1 and 15. All animals were subjected to dermal Draize observations daily for 5 days following each vaccination. Following completion of dermal Draize observations, animals were euthanized for histopathological examination of the injection site.
Evaluation of Repeat-Dose Toxicity in Mice
Cageside observations were performed twice daily and included evaluation of mortality, moribundity, general health, and signs of toxicity. Clinical observations were performed prior to initial dose, twice weekly throughout the study, and at termination. Observations included skin and fur, eye and mucous membranes, respiratory, circulatory, autonomic and central nervous systems characteristics, and somatomotor and behavior patterns. Body weight and food consumption were determined weekly. Reactogenicity of immunization sites was scored for edema and erythema using a scale from 0 for no symptoms to 4 for severe symptoms (Table 3). Scoring was performed prior to dosing on day 1 and then daily starting on day 2.
Dermal Draize Observations.

One day after administration of the last dose of either rF1V or placebo control (day 72), 10 animals/sex/study group were euthanized for interim necropsy. Thirty days after administration of the last vaccination (day 101), all the remaining animals groups were euthanized for recovery necropsy.
Hematology (leukocyte count, erythrocyte count, hemoglobin, platelet count, hematocrit, leukocyte differential, cellular morphology, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, mean corpuscular volume, and mean platelet volume) and serum chemistry (alanine aminotransferase, albumin, globulin, total protein, albumin/globulin ratio [calculated], alkaline phosphatase, creatinine, total bilirubin, sodium, potassium, chloride, calcium, cholesterol, glucose, inorganic phosphorus, and triglycerides) were performed on blood samples collected from all the animals at termination on days 72 and 101.
All animals euthanized on days 72 and 101 were subjected to a full gross necropsy, which included examination of the external surface of the body, the injection sites, all orifices, and the cranial, thoracic, and abdominal cavities and their contents. Weights were obtained for the following organs: adrenal glands, brain, heart, kidney, liver, lungs, spleen, testes/ovaries, and thymus. Paired organs were weighed together. The following organs were collected and preserved in 10% neutral-buffered formalin: adrenal glands, aorta, bone marrow (sternum), brain, esophagus, eyes, heart, large intestine, kidneys, liver, lungs with mainstem bronchi, lymph nodes (mesenteric and popliteal), mammary glands, optic nerves, ovaries (females), pancreas, pituitary, prostate (males), salivary glands, sciatic nerve, skeletal muscle (injection site and non-injected), skin-covering immunization sites, small intestine, spinal cord, spleen, stomach, testes with epididymides (males), thymus, thyroid and parathyroid, trachea, urinary bladder, uterus with cervix (females), and abnormalities or gross lesions. Tissues were examined microscopically for all animals in the control and high-dose groups at the terminal necropsy (groups 1, 2, 5, and 6).
Evaluation of rF1V Immunogenicity in Mice
Parallel groups of mice were required to assess the immunogenicity of the rF1V vaccine since at terminal euthanasia; the volume of blood available from each study animal was insufficient for concurrent clinical pathology (hematology and chemistry) and analysis of humoral immune response by enzyme-linked immunosorbent assay (ELISA). Six groups given dosages identical to those used in the repeat-dose toxicity study were evaluated (Table 1). Five mice in each study group were euthanized to obtain serum for evaluation of the immune response at the time points (days 29, 57, and 71) selected to coincide with the assessment time points in the repeat-dose study. Resultant serum samples were stored at −20°C until analysis.
Immunogenicity of rF1V was assessed using a DVC-developed Bridge ELISA. 21 This assay measures antibodies against recombinant F1 protein, recombinant V protein, and recombinant F1V fusion protein in mouse serum. Ninety-six-well microtiter plates are coated with the antigen, blocked, and stored at 2°C to 8°C until use. After dilution, calibrator standards, controls, and samples are pipetted into the microtiter plate well and incubated with agitation for approximately 1 hour at room temperature. The plate is washed to remove any unbound antibodies. Biotin-labeled antigen is then added to the wells and incubated for approximately 1 hour at room temperature. The plate is washed to remove any unbound antigen. Horseradish peroxidase-conjugated streptavidin is added to the wells and incubated with agitation for approximately 30 minutes. Following a wash to remove any unbound conjugate, a substrate, tetramethylbenzidine is added to the wells. The enzyme substrate reaction is halted, and the absorbance is measured at 450 nm in a microtiter plate reader. Results are reported as U/mL.
Evaluation of Local Reactogenicity in Rabbits
A local reactogenicity study in rabbits was performed, because it was not feasible to administer the full human dose volume (0.5 mL) to mice. Rabbits were grouped into 2 separate cohorts with 3 rabbits per cohort (Table 2). Rabbits in cohort 1 received 0.5 mL of formulated vaccine, rF1V-only vaccine, or placebo control with Alhydrogel on day 1. Rabbits in cohort 2 received 0.5 mL of formulated vaccine, rF1V-only, or placebo control with Alhydrogel on days 1 and 15. Vaccination sites were evaluated for reactogenicity prior to each vaccination and daily for 5 days following each dose administration. Reactogenicity of immunization sites was scored for edema and erythema using a scale from 0 to 4 based on the severity (Table 3). Rabbits were monitored for morbidity and mortality throughout the course of the study. Following the final reactogenicity observation in each cohort, the animals were euthanized, and tissue (skin and muscle) was collected from each vaccination site for histopathological examination. The presence of a humoral immune response was confirmed on animals in cohort 2 on day 21 by ELISA.
Statistical Analysis
Quantitative parameters were analyzed using the Kolmogorov-Smirnov test for normality, the Levene median test for equal variance, and the 1-way analysis of variance (ANOVA). If either the normality or the equal variance test failed, then the analysis was continued using the nonparametric Kruskal-Wallis ANOVA on rank-transformed data. For parametric data, if the ANOVA indicated statistical significance among experimental groups, then the Dunnett
Results
Repeat-Dose Toxicity
A summary of the in-life findings from the repeat-dose toxicity study is shown in Table 4.
Summary of Mouse Repeat-Dose Toxicity In-life Findings.
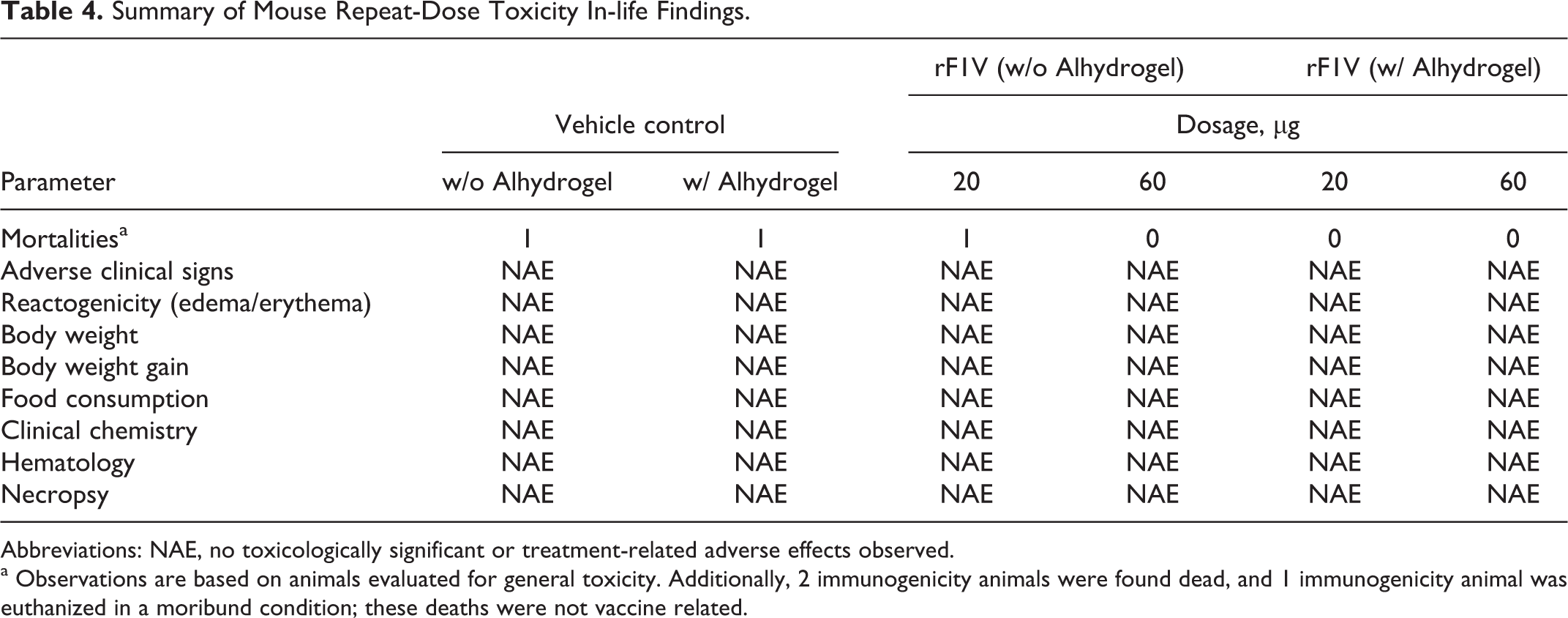
Abbreviations: NAE, no toxicologically significant or treatment-related adverse effects observed.
a Observations are based on animals evaluated for general toxicity. Additionally, 2 immunogenicity animals were found dead, and 1 immunogenicity animal was euthanized in a moribund condition; these deaths were not vaccine related.
Immunogenicity
Antibodies to rF1, rV, and rF1V were below the limit of quantitation (<10 µg/mL) in animals that received the placebo control. Mice vaccinated with rF1V (with or without Alhydrogel) generally presented a dose-dependent increase in antibody titer. The response to all vaccine antigens increased vaccination. However, a reduced immune response at both dose levels was observed in animals receiving rF1V only when compared to rF1V with Alhydrogel (Figure 1).
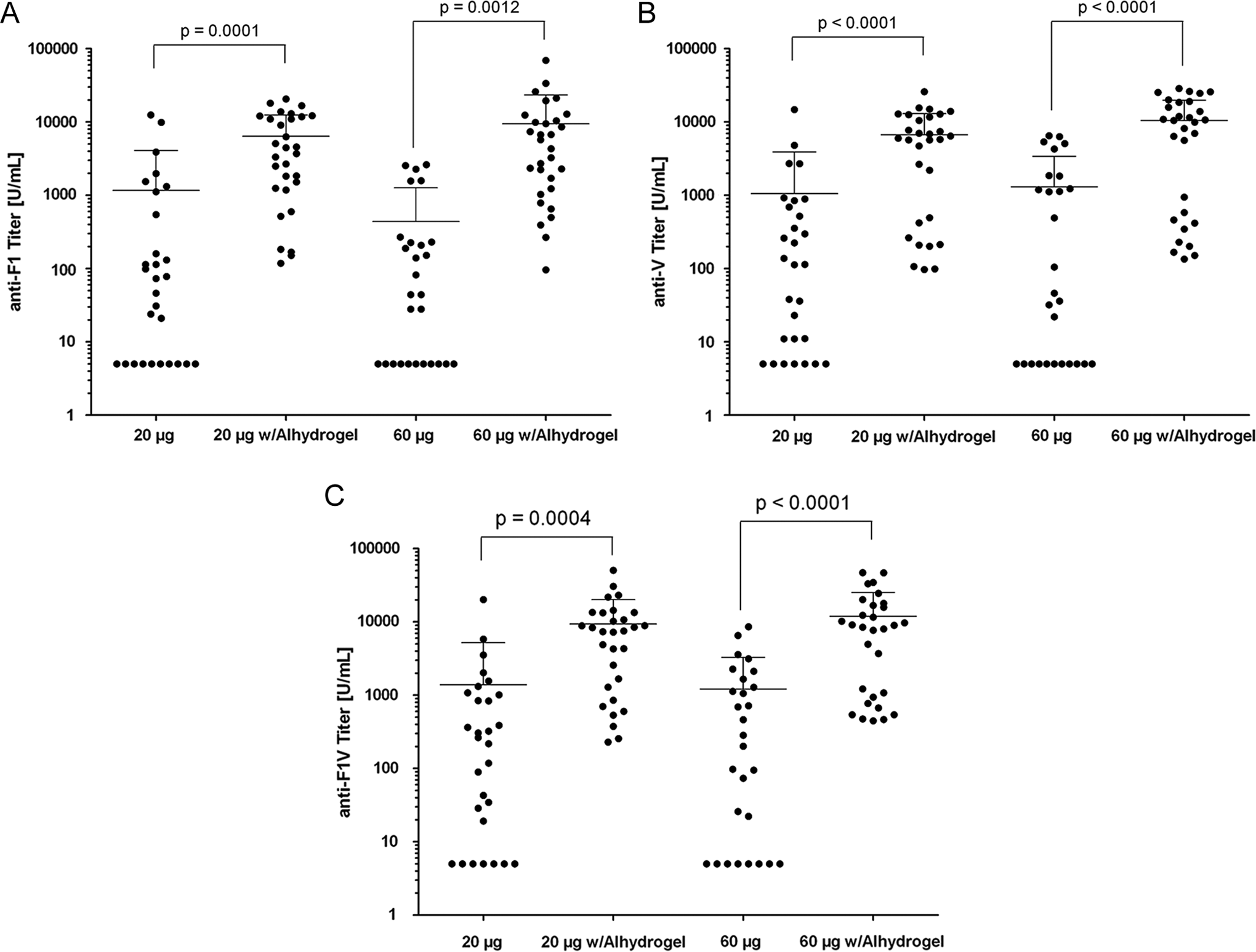
Mean mouse antibody concentrations following vaccination with rF1V. Groups of mice were vaccinated with 20 or 60 µg of rF1V with or without 0.3% (w/v) Alhydrogel by IM injection on days 1, 29, and 57. Samples were collected on day 72 for analysis of circulating antibody titers (U/mL) to rF1 (A), rV (B), and rF1V (C). All animals vaccinated with the placebo control presented titers below the limit of quantitation for the assay (10 U/mL). IM indicates intramuscular.
Mortality
A total of 5 males did not survive until the scheduled termination; however, their deaths were not considered to be related to rF1V vaccination, since there was no obvious dosage or time-related pattern. One mouse in the 60 µg rF1V-only immunogenicity group was found dead on day 7. No abnormal observations were noted for this animal during the study. One mouse in the 20 µg rF1V-only immunogenicity group was found dead on day 54. This animal was observed as thin on day 22 and with a hunched posture and rough hair coat on days 22 and 26 but was considered to be normal on days 29 through 50. Three animals were euthanized as moribund during the study. One placebo control animal and 1 placebo control with Alhydrogel animal were euthanized on days 53 and 60, respectively. Both animals had observations of hunched posture, thin appearance, and rough hair coat. Finally, one 20 µg rF1V-only immunogenicity group animal was euthanized on day 22 due to head tilt, convulsions, squinting, and languid behavior; the animal had appeared normal until this time. All other animals survived until the scheduled study termination. As early deaths occurred in both treated and control groups, they were not considered test article related.
Clinical Observations
Observations of rough hair coat, abrasions, thin appearance, hunched posture, and alopecia were noted. These observations were made throughout the study in all groups and across both sexes and were not considered to be related to the test article.
Reactogenicity
Treatment with rF1V both with and without Alhydrogel produced several transient observations of minimal-to-moderate edema and erythema in mice (Figure 2). Overall, a trend toward a dose-dependent increase in the frequency of observations and time to recover was observed compared to the control groups, although the severity of observations for all the groups of animals was not greater than “moderate” (score of 2/4). Those groups containing Alhydrogel in the formulation generally tended to have a higher incidence and intensity of observations as well as a longer time to recovery than their comparable rF1V-only counterparts. All observations resolved prior to each subsequent vaccination time point and all injection sites appeared normal at the recovery necropsy (day 101).
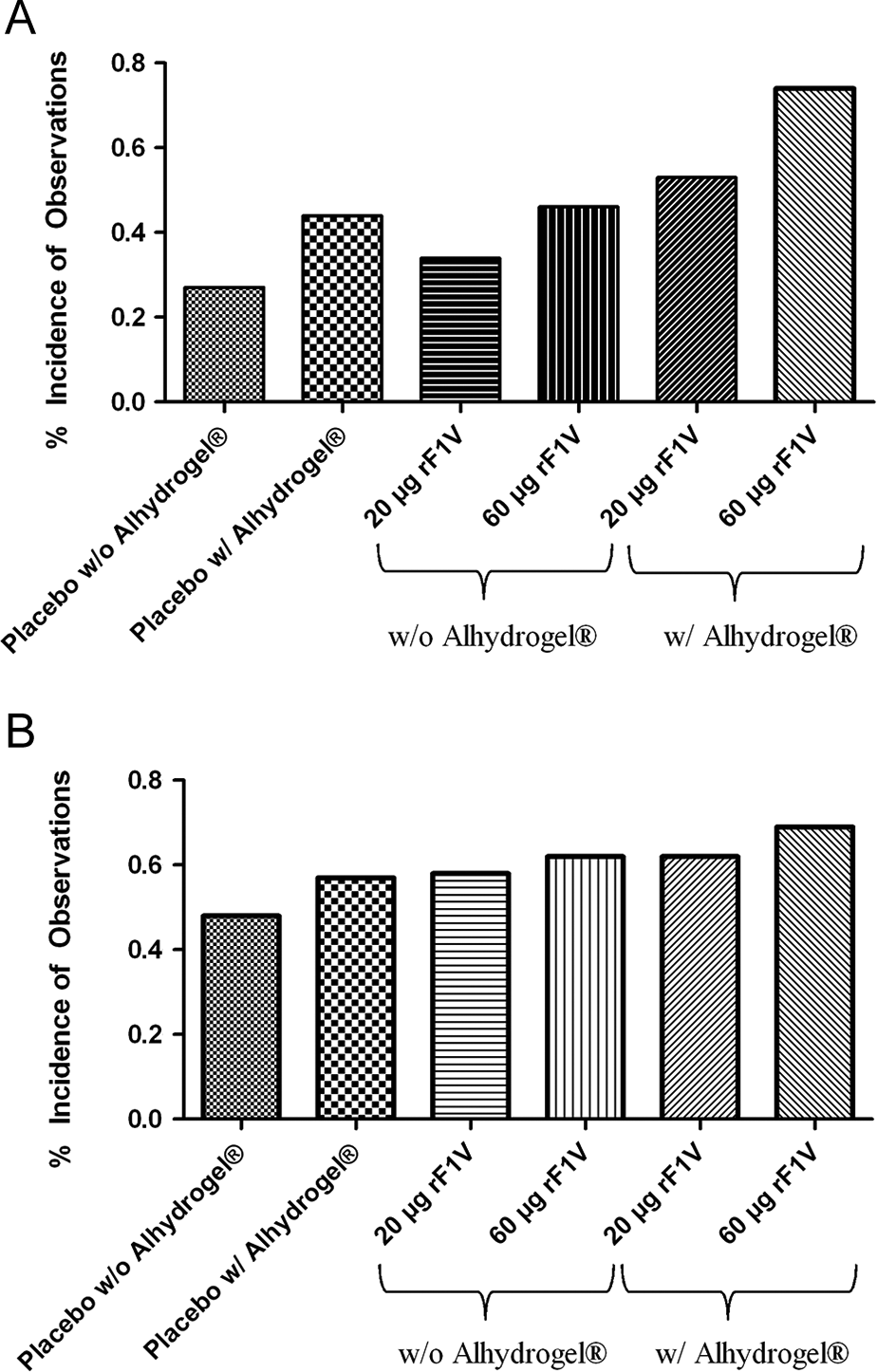
Edema (A) and erythema (B) were observed in mice following rF1V vaccination administration. The presence of Alhydrogel in both the placebo control and the formulated vaccine indicated a trend toward an increased incidence of both edema and erythema. However, the severity of observations for all the groups of animals was not greater than “moderate” (score of 2/4). Incidence of edema and erythema in mice following vaccination with rF1V.
Body Weight, Body Weight Change, and Food Consumption
No significant effects on body weight, body weight change, or food consumption were observed during the study that could be attributed to rF1V administration. Any statistically significant changes in body weight observed were considered incidental due to lack of correlating changes between the main study and the immunogenicity subsets and lack of simultaneous change in food consumption for the same observation interval. Therefore, the test article is not considered causative for these statistical findings. Overall, most animals gained weight throughout the study. Observed statistically significant changes in food consumption were transient and were not observed in the opposite sex for the same observation interval and therefore were not considered to be treatment related.
Clinical Pathology
No toxicologically significant effects on hematology and clinical chemistry were observed in any groups during the study that could be attributed to rF1V administration either with or without Alhydrogel. All observed statistical differences in hematology and clinical chemistry were attributed to individual animal variation and were within the laboratory’s historical range for normal values.
Anatomical Pathology
Observations of gross lesions were infrequent. Gross pathology observations included white masses at the injection site, reduction in thymus size, pale liver, enlarged mandibular or iliac lymph nodes, discoloration of the lungs, ovarian cysts, stomach nodule, and distended uterus. These observations were infrequent, occurred in the control group, and/or presented no dose relationship and were therefore considered unrelated to treatment. Statistically significant differences in organ weights were observed but without a clear correlation with the dosage level. These changes in organ weights were attributable to individual animal variation and were not considered treatment related, since they were not correlated during gross or microscopic evaluation and did not follow a dose-dependent response.
Histopathology
Microscopic examination of the collected tissues was performed on mice in the placebo control and 60 µg rF1V groups both with and without Alhydrogel.
The primary microscopic finding was localized inflammatory lesions at the injections sites, especially in both the placebo control and the rF1V with Alhydrogel groups. These lesions consisted of a focal area in the subcutaneous tissue or muscle that represented the inoculated material and appeared as a finely granular, lightly basophilic material with inflammatory cells in the peripheral area. The injection sites of mice in the placebo control and rF1V groups without Alhydrogel had very few lesions. Injection site 4 (day 71) had the highest incidence of lesions throughout all the groups and consisted of hemorrhage and chronic/active inflammation in the subcutaneous tissue. The microscopic inflammation observed was considered to be a typical foreign body host response and not related to rF1V administration. Several mice had inflammation along the sciatic nerve and changes in the iliac lymph nodes which were considered to be a response to the distal inflammatory process.
Rabbit Local Reactogenicity Study
A summary of the findings of the rabbit reactogenicity study is presented in Table 5. All animals survived until scheduled termination, and all gained weight throughout the study. All rabbits appeared normal upon gross necropsy. Transient dermal Draize score findings at the injection sites (day 1: right quadriceps muscle and day 15: left quadriceps muscle) were limited to minimal edema and/or erythema, and a total of 4 rabbits developed a local reaction. Observations were noted in 1 rabbit in the placebo control with Alhydrogel group on days 16 and 17; 2 rabbits in the 20-μg rF1V with Alhydrogel group, 1 on days 2 to 4 and 1 on day 16; 1 rabbit in the 40-μg rF1V with Alhydrogel group on day 2; and 1 rabbit in the 80-μg rF1V with Alhydrogel group on days 2 to 4. All other animals were considered to be normal “no swelling; normal color.”
Summary of Rabbit In-Life Findings.

Abbreviation: NAE, No toxicologically significant or treatment-related observed effects in either sex.
a Number of animals exhibiting the reactogenicity score at some point during the study.
Histopathology observations consisted of minor IM histiocytic or pyogranulomatous infiltrates at the injection sites. The infiltrates were not observed in a dose-dependent manner or in all rabbits. Further, they were not observed in any of the rabbits given 160 μg of rF1V without Alhydrogel. This suggests that the infiltrates may be due to the Alhydrogel and not the rF1V. Following the first vaccination, 1 rabbit in each of the groups receiving 80 and 160 μg with Alhydrogel was observed with histiocytic infiltrates at the injection site. After the second vaccination, 1 rabbit in each of the groups receiving 20, 40, or 160 μg with Alhydrogel had pyogranulomatous infiltrates at injection site 1; 1 animal in the 80 μg with Alhydrogel group had the same observation at injection site 2. Additionally, after the second vaccination, 1 animal in each of the groups receiving 80 or 160 μg with Alhydrogel was observed with histiocytic infiltrates at injection site 2. Finally, 1 placebo control with Alhydrogel animal had observations of histiocytic infiltrates at both injection sites. All rF1V-vaccinated animals in cohort 2 were observed with a quantifiable ELISA titer to rF1V on day 21; control animals were all below the limit of quantitation for the assay.
Discussion
As with many vaccines, the target population for the rF1V vaccine will be primarily healthy individuals; therefore, the tolerance for any potential toxicity and local reactogenicity is low. Preclinical safety studies are not intended to (or expected to) identify rare toxicities or effects in subpopulations, which are generally addressable only in human studies, since safety evaluation in animal models does not consistently predict toxicity in humans. The safety assessment of preventive vaccines is a continuous process that begins with the development of a vaccine candidate at the pre-IND stage and continues throughout the life cycle of clinical development and postlicensure use. 17 The approach reported in this article is similar to a successful study design used previously by our group to evaluate the potential toxicity of a recombinant botulinum neurotoxin vaccine candidate. 22
When normalized by body weight, the 60 μg/20 g mouse dosage evaluated in this study was approximately 1500 times greater than the maximum 160 μg/70-kg dosage intended for human use. Repeated administration of rF1V to mice had no toxicologically significant effect on mortality, clinical appearance, body weight, or food consumption. Mice gained weight and had normal food consumption during the study. Additionally, there was no apparent effect on measured clinical pathology parameters due to multiple injections of rF1V. Transient edema and erythema were observed at the vaccination site in some mice; however, all observations resolved prior to the final necropsy. Organ weights were within normal limits, and there were no obvious abnormalities on gross and histopathological examination of tissues related to rF1V administration. Microscopic findings of infiltrating cells or chronic active inflammation observed at the injection sites were consistent with the known responses to vaccine–Alhydrogel combination. 23,24
Inclusion of an immunogenicity arm in the studies was extremely important for establishing a relationship between a toxic effect and the magnitude of the immune response to the vaccine. Although not statistically significant, a dose-dependent increase in rF1, rV, and rF1V antibody concentrations was observed. Thus, the design allowed assessment of toxicity associated with an active immune response to vaccination. Evaluation of immunogenicity with and without adjuvant in the repeat-dose toxicity study also provided a risk–benefit assessment supporting the inclusion of adjuvant in the rF1V formulation. Alhydrogel was required for induction of a robust immune response; however, its inclusion in the formulation did not lead to overt safety concerns.
The rabbit local reactogenicity study showed that the full human 0.5 mL dose volume did not result in any significant changes in dermal Draize observations, gross or microscopic pathology at the injection site. Overall, no vaccine-related adverse findings were observed in the repeat-dose toxicity or local reactogenicity studies.
Footnotes
Acknowledgments
The authors would like to thank Drs Shannon Martin, Doris Snow, and Mary Kate Hart and Ms Patricia Fellows for critical review of the manuscript.
Authors’ Note
Information contained in this manuscript does not represent official DoD positions, policies, or decisions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Chemical Biological Medical System-Joint Vaccine Acquisition Program (CBMS-JVAP), US Department of Defense (DoD) Contract DAMD 17-98-C-8024.
