Abstract
A recombinant ricin toxin A-chain 1-33/44-198 vaccine (RVEc) was administered to male and female New Zealand white (NZW) rabbits (10/sex/group) in a repeat-dose toxicity study. The RVEc vaccine was administered on study days 1, 29, 57, and 85 via intramuscular (IM) injection (0, 100, or 200 μg/dose). All study animals were observed throughout treatment until euthanized and submitted for necropsy on study day 88 or 99 (recovery period). There were no treatment-related or toxicologically significant effects observed. There were no statistically significant differences noted in the antibody titers and/or concentrations in 100 μg RVEc-treated animals when compared to 200 μg RVEc-treated animals, suggesting that both doses produced comparable antibody titers/concentrations during the study. The highest immune response was observed on study day 99 (ie, 2 weeks after the last dose). The immune response observed demonstrated that RVEc is biologically active in the rabbit model, with no apparent marked sex differences.
Introduction
The armed forces and civilian personnel need protection against aerosolized ricin toxin, a potential biological threat agent. Ricin is an extremely potent toxin extracted from the castor bean, Ricinus communis, family Euphorbiaceae. 1 –3 It was reportedly first identified by Stillmark, with an estimated lethal dose in humans of 1 to 10 μg/kg. 4,5 Because of its relatively high toxicity and extreme ease of production, ricin toxin (code-named Compound W) was considered for weaponization during the US offensive Biological Warfare Program. 6,7 In recent years, ricin toxin has become a favorite tool of extremists who seek to harm others. 5 Ricin toxin was used in the highly publicized assassination of Bulgarian defector Georgi Markov. 8
Ricin belongs to a family of plant toxins, including abrin, modeccin, and viscumin, which have similar structure and mechanism of action. 9 The toxin is synthesized as a single polypeptide in maturing castor beans where it accumulates in the storage granules of the seeds. Posttranslational processing converts the single chain into 2 dissimilar polypeptide chains (ricin toxin A [RTA] and ricin toxin B [RTB]) which are held together by a single disulfide bond at residues Cys259 in RTA with Cys4 in RTB. 10,11 The RTA subunit (32 000 Da) is a potent inhibitor of protein synthesis and the RTB (34 000 Da) is a galactose- or an N-acetylgalactosamine-binding lectin. 12
The toxic consequences of ricin are attributed to the biologic activity of the A-chain, whereas the B-chain function is to bind the toxin to cell surface receptors thereby inducing internalization. The B-chain binds reversibly to galactose moieties presented by cell surface glycopeptides or glycolipids and results in endocytosis of the toxin. 13,14 The cell-surface binding domain on the B-chain has a translocation domain to promote transfer of the A-chain across the cell membrane. After endocytosis, the ricin holotoxin travels retrograde through the Golgi to the endoplasmic reticulum where the disulfide bond between the A and B chains is reduced. Subsequently, some of the A-chain molecules transfer to the cytosol where they can attack the ribosomes.
The A-chain is one of a family of ribosome-inactivating proteins (RIPs). Ribosome-inactivating proteins damage single-stranded RNA by removing a protein-specific set of adenines and cleavage at the resulting base-free sites. Once in the cytoplasm of a eukaryotic cell, the A-chain enzymatically attacks the 28S ribosomal subunit. 5 This results in an inhibition of protein synthesis. 12 The A-chain may also induce apoptotic changes in cells. Nearly all animal cells are subject to the lethal effects of ricin. 15 However, when the A-chain of ricin is separated from the B-chain and administered parenterally to animals, it has little or no toxicity. 12,16
The development of vaccines to protect against this lethal toxin has been a focus of the US Army Medical Research Institute of Infectious Diseases (USAMRIID) laboratories. To that end, a recombinant ricin A-chain (RTA) with the deletion of residues 34 to 43 and 199 to 267 (rRTA 1-33/44-198; Escherichia coli optimized gene) protein (RVEc) was recently selected for clinical investigation as a vaccine candidate. The current preclinical study was therefore conducted to evaluate the potential toxicity and immunogenicity of this vaccine candidate (RVEc) in preparation for human clinical trials.
Materials and Methods
Production of RVEc
The current RVEc was engineered to significantly mitigate or eliminate the N-glycosidase activity by the disruption of the ribotoxic site (deletion of residues 199 through 267) and to have increased stability while removing a potential protease-sensitive site on the protein surface (removal of loop, residues 34-43). 17 For the current study, a RVEc master cell bank (MCB) and a working cell bank (WCB) were manufactured by Charles River Laboratories, Inc (Malvern, Pennsylvania). Bulk drug substance was manufactured at the University of Nebraska–Lincoln Biological Process Development Facility (UNL-BPDF) using a process that optimized the purity and yield of rRTA 1-33/44-198 (drug substance). RVEc was formulated and filled at the Walter Reed Army Institute of Research (WRAIR) Pilot Bioproduction Facility (PBF, Silver Spring, Maryland). Vaccine diluent was also manufactured at UNL-BPDF and formulated and filled at the WRAIR PBF.
The drug substance, rRTA 1-33/44-198, was manufactured from an E coli codon-optimized gene, purified, and supplied as a sterile bulk in pillow bags at a concentration of 1.07 mg/mL in 20 mmol/L sodium succinate, 100 mmol/L NaCl, pH 6.5, with 0.1% to 0.2% Tween-20, and shipped frozen to WRAIR for the aseptic fill/finish (formulation). The drug product, RVEc, was formulated as 0.2 mg of recombinant protein in 20 mmol/L sodium succinate, 100 mmol/L NaCl, pH 6.5, adsorbed to 0.2% Alhydrogel at a final volume of 1 mL per single-use vial. RVEc contains 0.02% to 0.04% Tween-20 as a carryover from the bulk manufacturing. The diluent (used as a control injection in the rabbit study) was formulated to contain 20 mmol/L sodium succinate, 100 mmol/L NaCl, pH 6.5, and 0.2% Alhydrogel at a final volume of 2 mL per single-use vial.
Experimental Design
Before the initiation of in vivo work, the study protocol involving experimental animals was reviewed and approved by the IIT Research Institute Animal Care and Use Committee. New Zealand white (NZW) rabbits (30/sex, 4 months old, 2.5-3 kg, uniquely identified) were received from Harlan (Oxford, Michigan) and housed individually in suspended stainless steel cages under conventional environmental conditions in accordance with the Guide for the Care and Use of Laboratory Animals. 18 Certified Rabbit High Fiber Diet #2031 (Harlan Teklad, Madison, Wisconsin) and coarse-filtered city water were available ad libitum. Animals deemed to be healthy after quarantine were randomized into experimental groups based on body weight. Animals were administered test or control article by intramuscular (IM) injection in the relatively large muscle mass on the posterior aspect of the hind limb once on study days 1, 29, 57, and 85. At each dosing day, the inoculation site alternated between hind limbs, with the left hind limb receiving the first dose on study day 1. At each dosing day for groups 1 and 3, diluent or vaccine was given as a single, 1.0-mL injection. At each dosing day for group 2, vaccine was given as a single, 0.5-mL injection. From each group, 10 rabbits (5/sex/group) were euthanized and submitted for necropsy on study day 88 (3 days after the last dose). The remaining rabbits (5/sex/group) were euthanized and submitted for necropsy on study day 99 (2 weeks after the last dose). The study design is summarized in Table 1 .
Study Design
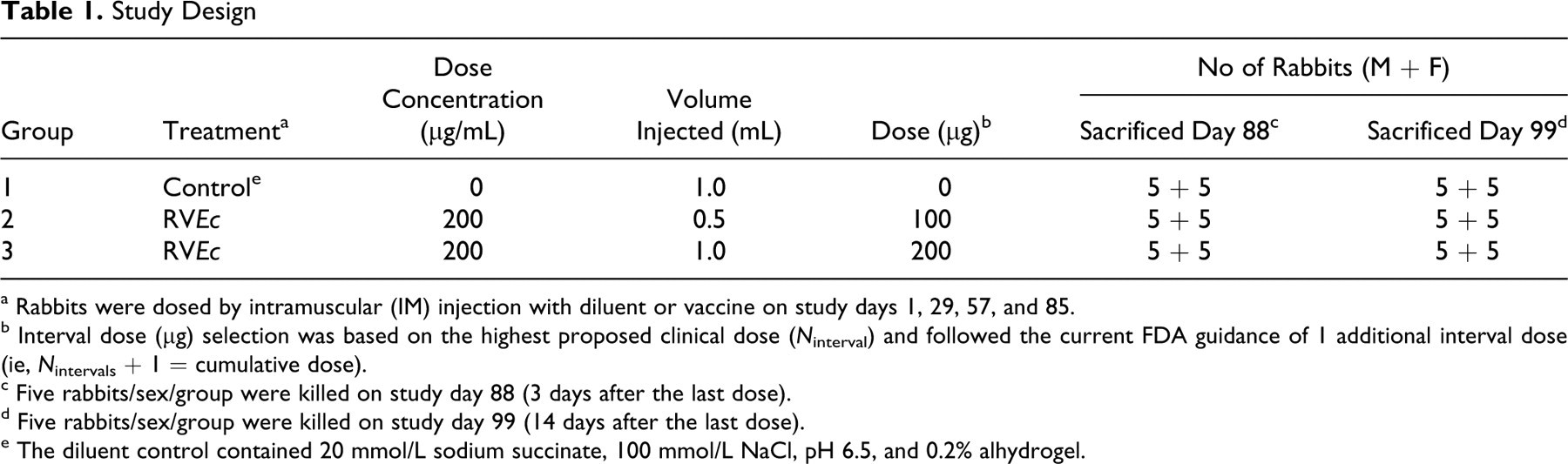
a Rabbits were dosed by intramuscular (IM) injection with diluent or vaccine on study days 1, 29, 57, and 85.
b Interval dose (μg) selection was based on the highest proposed clinical dose (N interval) and followed the current FDA guidance of 1 additional interval dose (ie, N intervals + 1 = cumulative dose).
c Five rabbits/sex/group were killed on study day 88 (3 days after the last dose).
d Five rabbits/sex/group were killed on study day 99 (14 days after the last dose).
e The diluent control contained 20 mmol/L sodium succinate, 100 mmol/L NaCl, pH 6.5, and 0.2% alhydrogel.
Antemortem Evaluations
Moribundity/mortality and clinical observations
Rabbits were observed for moribundity and mortality at least once daily during the quarantine period and at least twice daily during the treatment and observation periods. A detailed handheld clinical and physical observation was performed once during the quarantine (randomization) and at least once weekly during the treatment and observation periods. Detailed handheld clinical and physical observations were also performed before and after each dose. Cage-side observations were also performed daily after dosing initiation to assess their general health.
Inoculation site reactogenicity
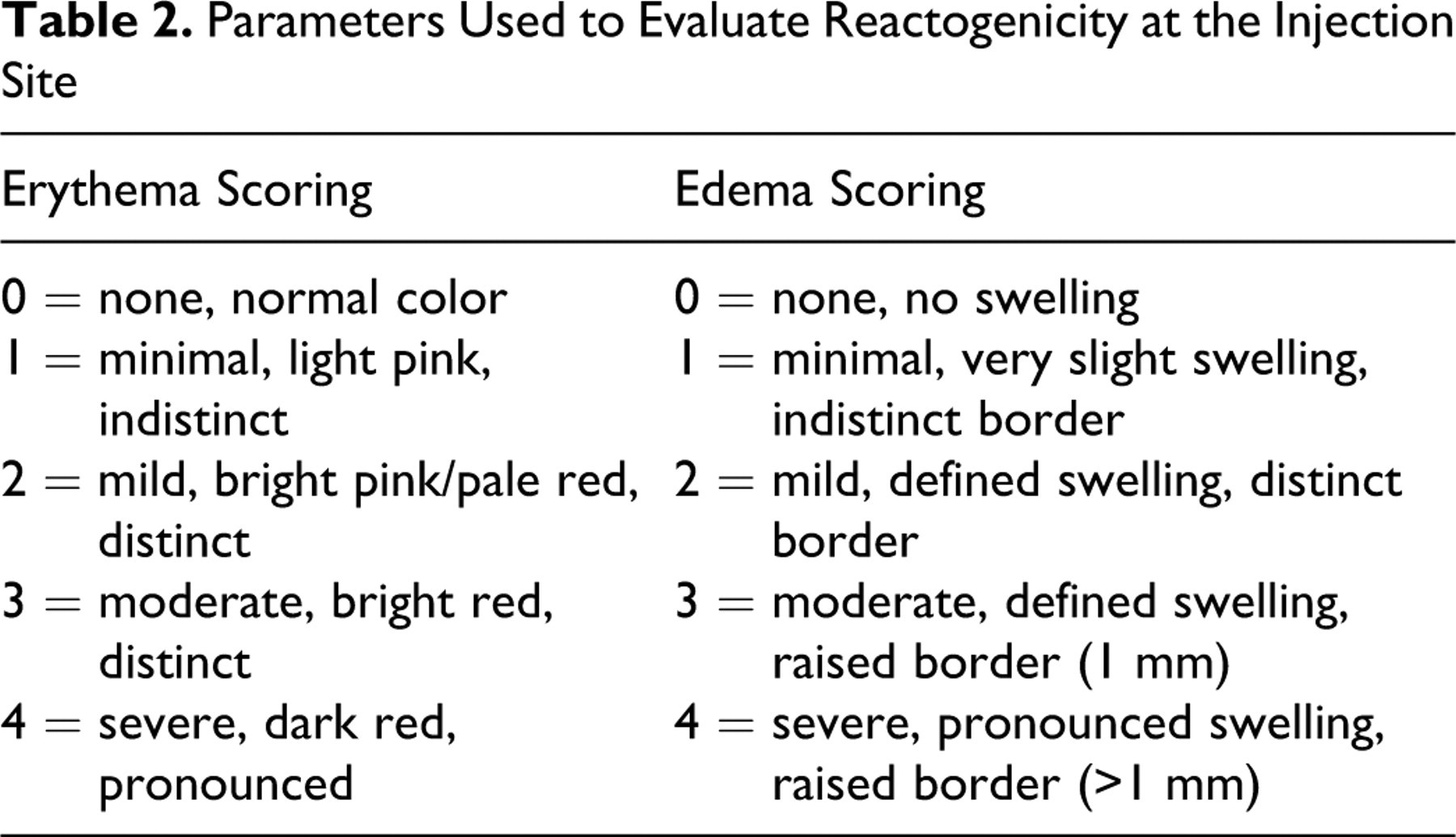
Inoculation sites were evaluated before each dosing on study days 1, 29, 57, and 85, and then at approximately 1 hour postdose, and once daily for 6 days after dosing for indications of reactogenicity (erythema and edema). Inoculation site reactogenicity was also evaluated at least once weekly during the remainder of the study and on the day of scheduled necropsy. Reactogenicity was graded using a scale of 0 (no reaction) to 4 (severe reaction) as indicated in Table 2 , using a scale similar to that described by Draize. 19
Parameters Used to Evaluate Reactogenicity at the Injection Site
Body weights, body weight gains, and food consumption
Body weights were measured 1 day after animal receipt and once during quarantine (randomization). Animals were weighed before inoculation, 1 and 2 days after each dosing, at least once weekly thereafter during the remainder of the study, and once before scheduled necropsy (fasted body weight). Total body weight gains were also calculated for the animals surviving to the end of the study antemortem phase (study day 99). Food consumption was measured daily and summarized weekly during the treatment and observation periods, except during overnight fasting periods before clinical pathology blood collections and necropsies.
Body temperature and ophthalmic examinations
Rectal body temperatures were measured before dosing on study days 1, 29, 57, and 85, and then at 6 hours (± 15 minutes) and 24 hours (± 30 minutes) after each dose. Indirect and slit lamp ophthalmoscopy was performed during pretest and before the study day 88 necropsy. Before the examinations, each eye was dilated with 1% tropicamide ophthalmic solution (Baush & Lomb, Tampa, Florida).
Clinical Pathology
After overnight fasting, blood samples for clinical pathology were collected from all surviving study animals once during quarantine (pretest), on study day 4 (3 days after the first dose), before dosing on study days 29 and 57, and before scheduled necropsy on study days 88 and 99 (ie, 3 and 14 days after the last injection, respectively). A full battery of clinical pathology tests included analysis of blood samples for clinical chemistry (Synchron LX20 Clinical System, Beckman Coulter, Inc, Fullerton, California), hematology (ADVIA 120 Hematology System Analyzer, Siemens Healthcare Diagnostics, Tarrytown, New York), and coagulation (STA Compact CT Coagulation Analyzer, Diagnostica Stago, Parsippany, New Jersey) parameters. Blood samples for C-reactive protein (CRP) analysis (Synchron LX20 Clinical System) were also collected from all surviving study animals during pretest, approximately 24 hours (± 1 hour) after dosing on study days 2, 30, 58, and 86 and then 7 days after each dose on study days 8, 36, 64, and 92. Urine samples were collected at each scheduled necropsy (study days 88 and 99) by aspiration directly from the urinary bladder and analyzed using a Chemstrip Urine Analyzer (Boehringer Mannheim Corp, Indianapolis, Indiana), evaluated using a refractometer, or graded visually.
Because slight increases in the group means for CRP were noted on study day 86 in the vaccine-treated groups, serum protein electrophoresis analyses were also performed to determine the serum protein fractions using blood samples collected during this time point and during pretest (8-10 rabbits/sex/group/time point) by Ani Lytics, Inc (Gaithersburg, Maryland).
Necropsy and Microscopy Evaluation
All study animals received a complete necropsy. Necropsy included examination of the external surface of the body, all orifices, and the cranial, thoracic, and peritoneal cavities and their contents. Rabbits scheduled for necropsy were anesthetized with sodium pentobarbital and euthanized by exsanguination. Tissues as specified in the Organisation for Economic Cooperation and Development (OECD) 411 and ANSI/AAMI/ISO10993-1 were collected at necropsy and fixed in 10% neutral-buffered formalin, with the exception of the eyes with optic nerves which were fixed in Davidson solution and the testes and epididymides which were fixed in Bouin fixative. 20,21 Bone marrow smears were prepared from the sternum and were fixed in methanol. The adrenals, brain, gallbladder, heart, kidneys, liver, lungs, ovaries, pituitary, spleen, testes, thymus, thyroids (with parathyroids), and uterus were weighed at necropsy and organ-to-body weight ratios were calculated. Tissues collected from all study animals in groups 1 (control) and 3 (high dose) killed on study days 88 and 99 (except bone marrow smears) were embedded in paraffin, processed routinely, and sections were stained with hematoxylin and eosin before evaluation by a board-certified veterinary pathologist. No target tissues were identified in any animals in the high-dose group; therefore, no tissues from animals in group 2 (low dose) were examined microscopically.
Immunogenicity Assessment
Blood was collected from all surviving study animals during quarantine (pretest), on study day 4 (3 days after the first dose); before dosing on study days 29 and 57, and before each scheduled necropsy on study days 88 and 99 (ie, 3 and 14 days after the last injection, respectively) for immunogenicity analyses.
Determination of relative serum neutralization antibody concentration and titer
The relative neutralizing anti-ricin antibody concentration and endpoint titer in rabbit serum was determined by a colorimetric cell proliferation toxin neutralization assay (TNA). The assay was run on 96-well, flat-bottom microplates. Each unknown rabbit serum was diluted in a house-made incomplete cell culture medium using Dulbecco's Modified Eagle’s Medium (D-MEM, Gibco, Langley, OK) to seven 2-fold serial dilutions (usually started at 1:200 dilution) and was tested in duplicate (50 μL/well). The plate format was designed to accommodate 5 unknown serum samples, 7 standards (polyclonal anti-ricin immunoglobulin G [IgG], purified from mouse serum, Perimmune, Inc, Rockville, Maryland) in duplicate, a negative control (pooled normal rabbit serum, prepared in house), a toxin control (R communis agglutinin II, Vector Laboratories, Inc, Burlingame, California), a cell control (EL4 cells, American Type Culture Collection, Manassas, Virginia), and a medium control (as blank). Each control was run in triplicate. Ricin challenge solution (2.5 ng/mL, 50 μL/well) was added to all standards, controls, and sample dilutions and was then neutralized by the anti-ricin antibody that possibly existed in the mixed solutions, by incubation at 37°C for 30 minutes. Then 50 μL of EL4 cells (9.5 × 10 5 cells/mL) was added to each well except the medium control wells. After incubating overnight (>18 hours), 30 μL/well of CellTiter 96 aqueous solution, a tetrazolium salt, 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) solution (Promega Co, Madison, Wisconsin), was added as the reporter or signal system. Sample-mediated neutralization of ricin toxin was manifested as a suppression of cytotoxicity and hence the preservation of cell viability. The toxin-mediated cytotoxicity was measured by determining cell viability colorimetrically by measuring the optical density (OD) at wavelength 490 nm with a microplate reader (BioTek Instruments, Inc, Winooski, Vermont). The relative rabbit neutralizing anti-ricin antibody concentration (μg/mL) of unknown rabbit serum was calculated automatically by Bio-Tek Gen5 Analytical software based on the mouse anti-ricin IgG standard curve run on each plate, which fit a 4-parameter logistic (4PL) equation, (a − d)/[1 + (x/c)b] + d. The rabbit neutralizing anti-ricin antibody endpoint titer was reported as the dilution factor of the highest dilution of a test sample that resulted in a positive reaction. The mean concentration and geometric mean titers (GMTs) of each test group were calculated with Microsoft Excel software.
Determination of total anti-ricin IgG antibody titers
The total anti-ricin IgG antibody titers of all rabbit serum samples were determined by an enzyme-linked immunosorbent assay (ELISA;) modified for analyzing rabbit sera. 22 Briefly, 96-well, U-bottom polyvinyl chloride (PVC) microtiter plates were coated with ricin toxin (5 μg/mL, 100 μL/well) as capture antigen. Normal rabbit serum (1:2000) was used as a negative control and a positive rabbit serum (1:20 000) was used as a positive control to monitor the assay performance. All samples were tested in duplicate at a starting dilution of 1:2000 and followed by 6 additional 3-fold serial dilutions (1:2000-1:1 458 000, 100 μL/well). Affinity-purified peroxidase-conjugated goat anti-rabbit IgG (H + L; KPL, Kirkegaard & Perry Laboratories, Gaithersburg, Maryland) was used as the detector antibody (0.2 μg/mL, 100 μL/well). After adding 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) peroxidase substrate (KPL) and then ABTS peroxidase stop solution, the OD was measured at a wavelength of 405 nm. The total anti-ricin IgG ELISA titer of each serum was reported as the dilution factor of the highest dilution of the serum that resulted in a mean OD value 0.07 higher than the cutoff point OD value.
Statistical Analysis
All statistical analyses were performed and reported by the Department of Math, Statistics, and Computer Science, University of Illinois at Chicago (Chicago, Illinois) using programs available through SAS OnlineDoc Version 9 (SAS Institute Inc, Cary, North Carolina). Data on immunogenicity, serum protein electrophoresis, clinical chemistry, hematology, coagulation, and physiologic observations were analyzed by repeated measures analysis of covariance (RMANCOVA). The significance level of α = .01 was used in the study.
The SAS procedure MIXED was used, and treatment group (control, RVEc low, and RVEc high) comparisons were performed through the ESTIMATE statements. Treatment group comparisons were RVEc low versus control, RVEc high versus Control, and RVEc low versus RVEc high. With the exception of organ weights and urinalysis, analyses were performed by intervals because treatment effect, if any, would be expected to differ across intervals. Distribution of each variable was assessed through graphic display for checking the normality assumption, and a transformation was applied when necessary, prior to performing analysis for a better approximation to normality. Outliers were scanned through graphic display. When a log transformation was used, a statistical comparison was expressed as a geometric ratio (anti-log of the difference) instead of a difference, and the standard error was expressed as the anti-log of the standard error of the log-transformed comparison (geometric standard error).
For the majority of comparisons, pretreatment values were used as a covariable and factors in the model were treatment (and day, when appropriate), sex, and treatment by sex interaction, with a significance level of α = .01. Analyses were performed and reported by sex and combining sex. When the day by treatment by sex interaction was significant (P < .01), group comparisons were done by sex. Otherwise, group comparisons were done by combining sex.
For the analysis of covariance on body weight, body weight gain, and food consumption, 4 intervals were created for the analysis and data were averaged per interval before performing the analysis. For the analysis of covariance on inoculation site reactogenicity, factors in the model were treatment, sex, and treatment by sex interaction, with a significance level of α = .01. Analyses were performed and reported by each time point because the treatment effect would not be expected to be similar across time points. The pretreatment scores were not analyzed, nor treated as covariables, because they were all zero values.
Descriptive statistics, antibody concentrations, and GMTs for the immunogenicity data, were calculated with Excel software and analyzed at the University of Illinois at Chicago (Chicago, Illinois), as described previously.
Results
Antemortem Evaluations
No animals died during the study and no treatment-related clinical signs were observed. Statistical analyses of the reactogenicity scores, body weight, and body weight gain interval data, and food consumption interval data did not reveal any noteworthy changes in the test article-treated animals as compared to the respective control group (Table 3 ). A statistically significant increase in body temperature was observed on study day 29 (~6 hours postdose) in group 3 (200 μg RVEc, males and females combined) when compared to the group 1 control group (Table 4 ). However, this was considered an incidental finding as the percentage change from the control was less than 1% and was not noted at the other time points. No other noteworthy or statistically significant findings were observed.
Summary of In-Life Findings
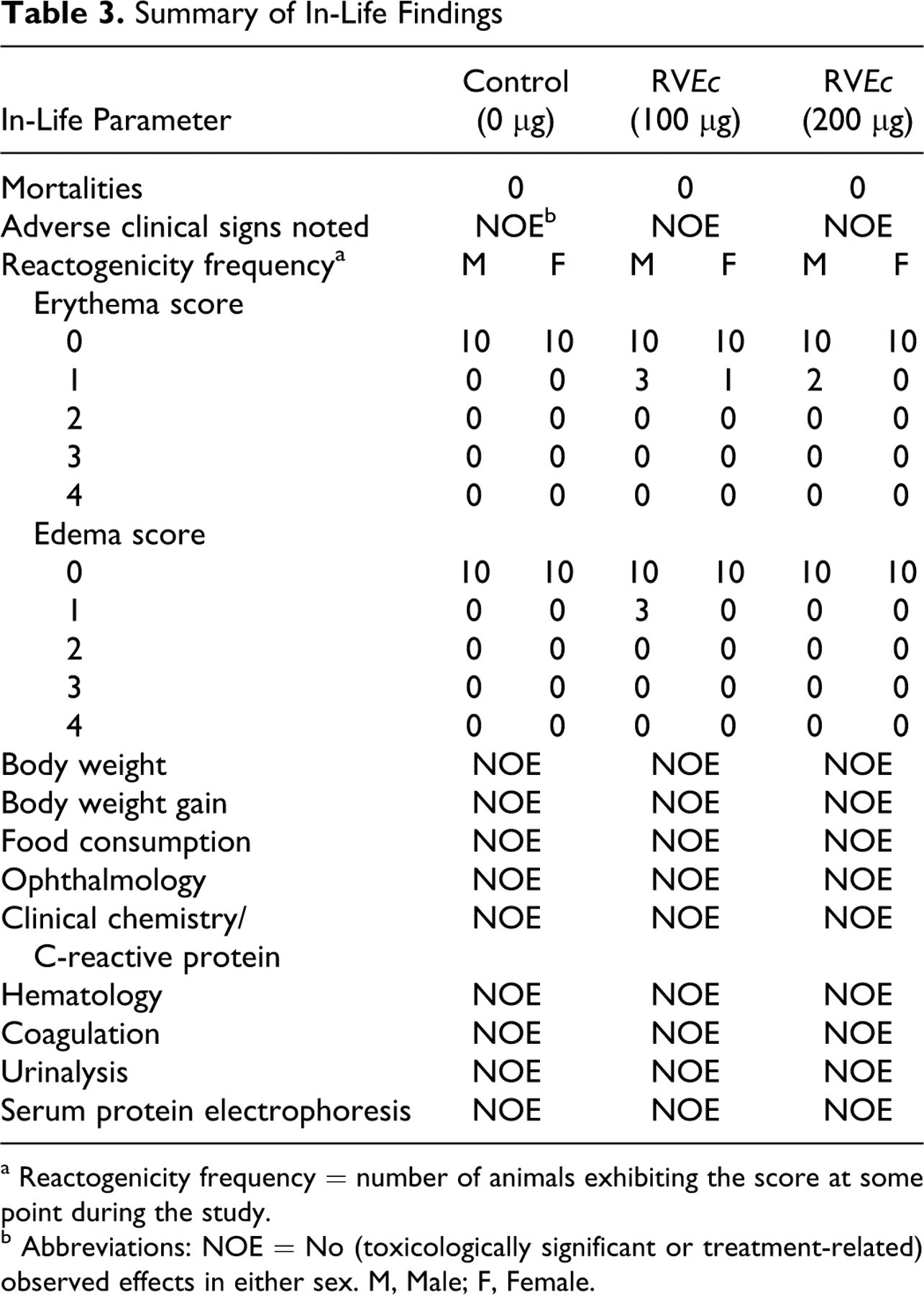
a Reactogenicity frequency = number of animals exhibiting the score at some point during the study.
b Abbreviations: NOE = No (toxicologically significant or treatment-related) observed effects in either sex. M, Male; F, Female.
Study Summary of Statistically Significant Findings
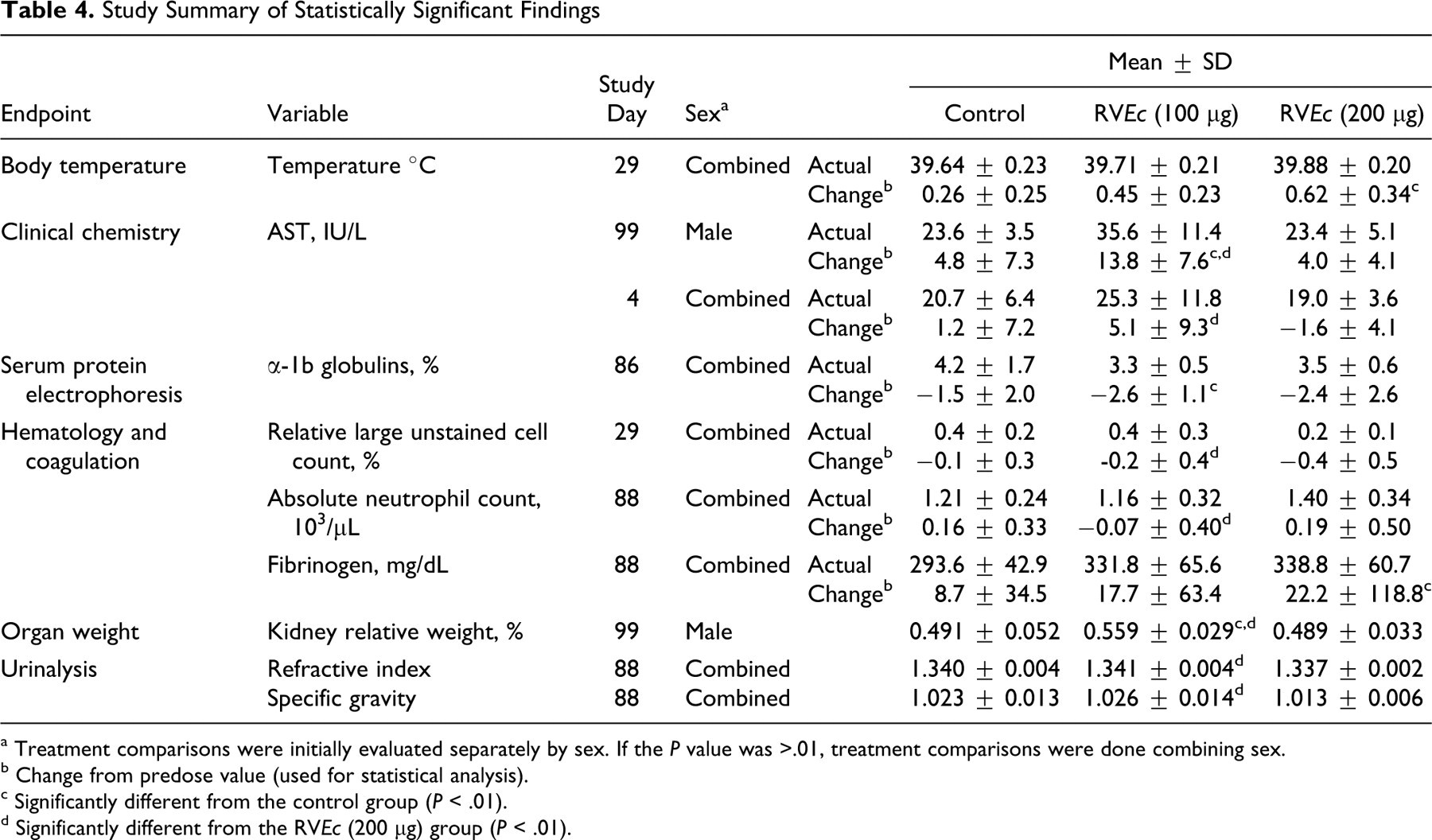
a Treatment comparisons were initially evaluated separately by sex. If the P value was >.01, treatment comparisons were done combining sex.
b Change from predose value (used for statistical analysis).
c Significantly different from the control group (P < .01).
d Significantly different from the RVEc (200 μg) group (P < .01).
Clinical chemistry and CRP
No treatment-related or toxicologically significant findings were observed for any clinical chemistry parameter throughout the study (Table 3). Aspartate aminotransferase (AST; IU/L) for group 2 (100 μg RVEc) males was greater than group 1 and group 3 males on day 99, but no dose response was apparent. Similarly, AST (IU/L) for group 3 was less than group 2 when males and females were combined on day 4, but neither group was different from controls (Table 4).
Hematology
No treatment-related or toxicologically significant findings were observed for any hematology parameter throughout the study (Table 3). Relative large unstained cell count (%) for group 3 was less than group 2 when males and females were combined on day 29, but neither group was different from controls. Similarly, absolute neutrophil count (10 3 /μL) for group 3 was greater than group 2 when males and females were combined on day 88, but neither group was different from controls (Table 4).
Coagulation
No treatment-related or toxicologically significant findings were observed for any coagulation parameter throughout the study (Table 3). Fibrinogen (mg/dL) for group 3 was greater than group 1 when males and females were combined on day 88, but the response was considered to be of minimal toxicological significance since this increase was no longer observed on study day 99 (Table 4).
Urinalysis
No treatment-related or toxicologically significant findings were observed for refractive index, specific gravity, and pH on study days 88 and 99. Visual examination of the remaining urinalysis data did not reveal any noteworthy changes in the test article-treated animals as compared to the respective control group (Table 3). Refractive index and specific gravity for group 3 was less than group 2 on day 88 when males and females were combined, but neither group was different from controls (Table 4).
Serum protein electrophoresis
No treatment-related or toxicologically significant findings were observed for any serum protein electrophoresis parameter on study day 86 (Table 3). Alpha-1b globulins (%) for group 2 was less than controls on day 86 when males and females were combined, but the effect was not considered biologically significant (Table 4).
Immunogenicity
Study rabbits inoculated on study days 1, 29, 57, and 85 with 100 μg (group 2) or 200 μg (group 3) RVEc stimulated an immune response. Blood was collected at various study time points and analyzed by TNA and ELISA. Table 5 summarizes the relative anti-ricin neutralizing antibody GMT and mean relative anti-ricin neutralizing antibody concentration determined by TNA and total anti-ricin IgG GMT determined by ELISA in rabbit sera of group 2 and group 3 (males and females combined). Four weeks after the second inoculation (day 57), both study groups produced considerably high antibody levels. In both test article-treated groups, the highest immune response was observed on study day 99 (ie, 2 weeks after the last dose). The relative neutralizing GMT and ELISA GMT were 16 000 and 939 526, respectively, for both groups. The relative mean neutralizing antibody concentration was 7416 μg/mL for group 2 and 7614 μg/mL for group 3. There were no statistically significant differences noted in the antibody titers and/or concentrations in group 2 animals (males and females combined) when compared to group 3 animals (males and females combined), suggesting that both doses produced comparable antibody titers/concentrations during the study. The control group showed no detectable antibodies during the entire study (data not shown). Immune response curves by group and sex are shown in Figures 1, 2, and 3. The immune response observed demonstrates that RVEc was biologically active in the rabbit model, with no apparent marked sex differences.
Summary of Immune Response in Rabbits Vaccinated With RVEc at Doses of 100 or 200 μg
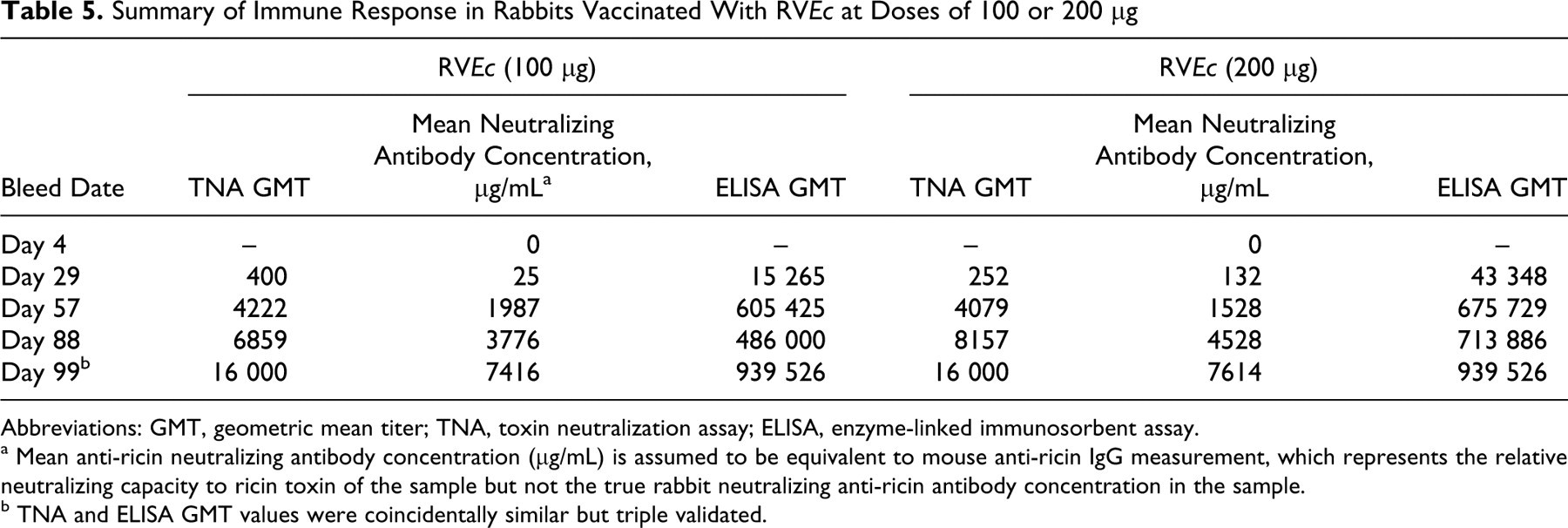
Abbreviations: GMT, geometric mean titer; TNA, toxin neutralization assay; ELISA, enzyme-linked immunosorbent assay.
a Mean anti-ricin neutralizing antibody concentration (μg/mL) is assumed to be equivalent to mouse anti-ricin IgG measurement, which represents the relative neutralizing capacity to ricin toxin of the sample but not the true rabbit neutralizing anti-ricin antibody concentration in the sample.
b TNA and ELISA GMT values were coincidentally similar but triple validated.
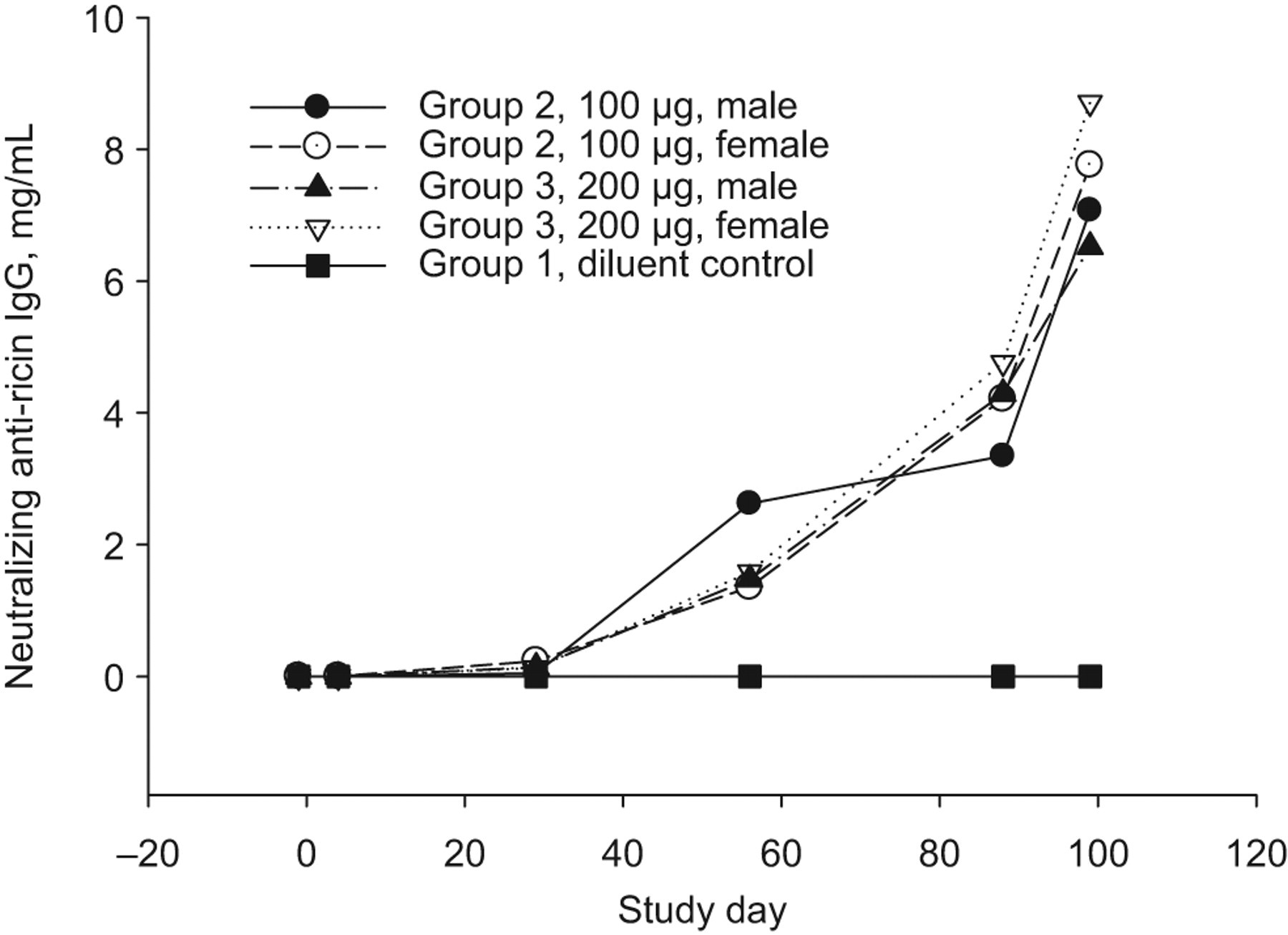
Mean relative neutralizing anti-ricin IgG concentration in rabbit sera collected during pretest and on study days 4, 29, 57, 88, and 99 in rabbits receiving 0 (diluent control), 100, or 200 μg of RVEc, determined with colorimetric toxin neutralization assay (TNA) using mouse polyclonal anti-ricin IgG as the standard. IgG indicates immunoglobulin G.
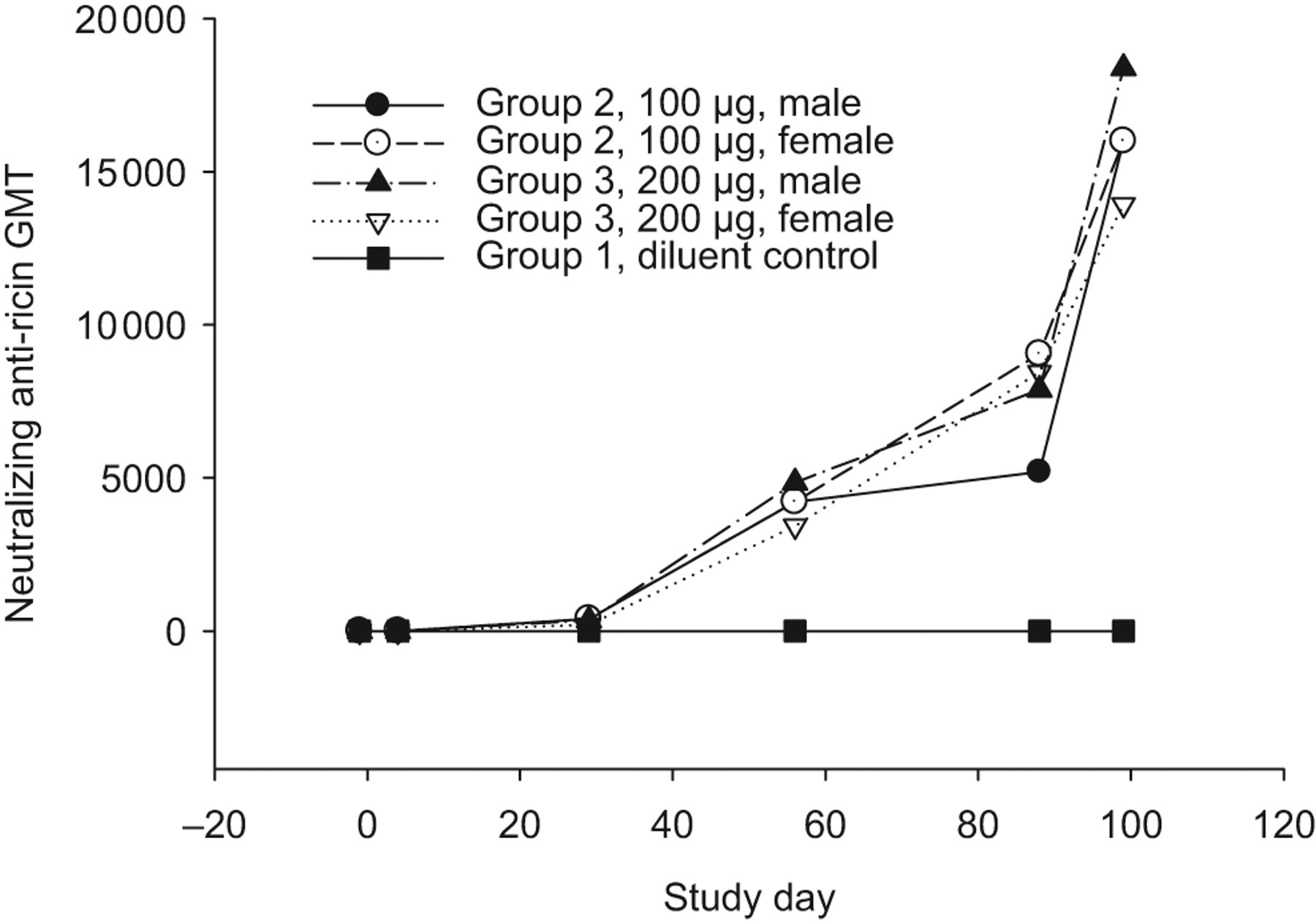
Neutralizing anti-ricin antibody geometric mean titer (GMT) in rabbit sera collected during pretest and on study days 4, 29, 57, 88, and 99 in rabbits receiving 0 (diluent control), 100, or 200 μg of RVEc, determined by colorimetric toxin neutralization assay (TNA).
Enzyme-linked immunosorbent assay (ELISA) anti-ricin IgG geometric mean titer (GMT) in rabbit sera collected during pretest and on study days 4, 29, 57, 88, and 99 in rabbits receiving 0 (diluent control), 100, or 200 μg of RVEc.
Postmortem Evaluations
Organ weights
A statistically significant increase in the relative kidney weights was observed on study day 99 in group 2 males when compared to the male control group and group 3 males. However, this increase was considered an incidental finding as the increase was not dose-related. No other noteworthy or statistically significant findings were observed (Table 4).
Necropsy
There were no gross lesions noted at necropsy on day 88. The few gross lesions noted at day 99 (recovery; eg, pale liver in a group 3 male; cyst on the left kidney and black foci on the left medial lobe of the liver in a control female) were not considered related to treatment with the test article.
Histopathology
Mixed cell infiltrates in the fascia and adipose around the sciatic nerves were observed, graded minimal to mild, but present only in animals (2 males and 4 females) at study day 88, which received 200 μg of RVEc. This finding was minimal in the perineural tissues of 1 group 3 (200 μg RVEc)-treated male at study day 99, indicating almost complete recovery after an additional 11 days without treatment. This change was considered an expected test article-related finding. Microscopic changes in the areas of the injection sites of group 3 animals administered 200 μg of RVEc vaccine were also noted at low incidences and were considered an expected response to the deposition of a vaccine substance. Other nontest article-related microscopy findings observed were considered incidental, were of the nature observed in this strain and age of rabbits, and/or were of similar incidence and severity in control and treated animals and therefore were considered unrelated to administration of RVEc.
Discussion
Justification and Risk of Animal Testing
The risk of RIP activity or symptoms from RIP activity was felt to be unlikely based on the fact that the toxic activity from ricin is attributed to the A-chain. The A-chain alone without the B-chain of ricin, which is required for binding to the cell surface receptors, has been demonstrated to have decreased toxicity (RTA is 1000-fold less toxic than natural ricin), resulting in no or minimal symptoms when low doses are administered to animals. Additionally, a cell-free translation assay (a microtiter assay developed to measure the biological activity of ricin or other RIPs, which uses nuclease-treated rabbit reticulocyte lysate containing luciferase mRNA) detected no effect of the vaccine on translation or inhibition of protein synthesis.
The portion of RTA containing amino acid residues leucine 74, aspartic acid 75, and valine 76 has been reported to be involved in its ability to induce vascular leak. 23 While the RVEc has the reported vascular leak syndrome (VLS) sequence, common clinical indicators of vascular leak (weight gain, hypoalbuminemia, edema) were not observed in pilot testing with animals or in an in vitro cell assay with human umbilical vein and human bronchioalveolar cell assays that may be more sensitive in detecting VLS. 24 The absence of VLS with RVEc may be explained by the elimination of the RIP activity and/or by the relatively small amounts and rapid clearance of RTA immunogens compared with RTA-antibody conjugates. Biological activity of the buried “vascular leak peptide” (VLP) also may require RTA to partly unfold or undergo other structural reorganization dependent upon the C-terminal domain that is lacking in the rRTA 1-33/44-198 vaccine. 17
Toxicity Study
Toxicity studies in rodents and nonhuman primates (NHPs) have been conducted with ricin utilizing different routes of exposure. In animal models, the histopathology of ricin aerosol exposure is characterized by necrotizing airway lesions causing tracheitis, bronchitis, broncheolitis, and interstitial pneumonia with perivascular and alveolar edema. 25 In contrast, lesions induced by lethal parenteral doses of ricin include lymphocytic necrosis of lymphoid organs; hemorrhages, necrosis, hyaline droplets, and fibrin thrombi in the liver; hemorrhages and necrosis in the adrenal glands; tubular hyaline changes in the kidney; degeneration of the heart muscle; and congestion and hemorrhages of the gastrointestinal tract. 26 –30 The onset of illness, morbidity, and death is rapid in animal models; dose-related time to death has been reported to be 29 to 32 hours in rats, 36 to 40 hours in rhesus monkeys, and 20 to 72 hours in African green monkeys postexposure to inhaled ricin. 31 The biological toxicity of the RVEc protein was evaluated by a cell-free translation assay, with more current results demonstrating that the RVEc protein lacked RIP biological toxicity. 32
In the current preclinical study, male and female NZW rabbits were administered RVEc vaccine at 100 or 200 μg by IM injection in a repeat-dose toxicity study. Control animals received the vaccine diluent. Study animals were dosed once on study days 1, 29, 57, and 85. The rabbits were observed after dosing to evaluate the potential toxicity of the vaccine until scheduled necropsy on study day 88 or 99.
All study rabbits survived to the scheduled necropsy dates. No treatment-related clinical signs were observed. No treatment-related or toxicologically significant effects were observed for inoculation site reactogenicity, body weights, body weight gains, food consumption, body temperatures, ophthalmology, clinical pathology, serum protein electrophoresis, and organ weights. There was no effect on serum albumin as has been reported with VLS. 33
No treatment-related, gross findings were noted at the terminal (study day 88) and recovery (study day 99) necropsy. Mixed cell infiltrate in the perineurium around the sciatic nerves was noted on study day 88 in animals inoculated with 200 μg of RVEc vaccine; however, recovery from this finding was nearly complete on study day 99. This finding along with a low incidence of microscopic changes in the areas of the injection sites following treatment with 200 μg RVEc were considered expected findings after injection with an immunogenic substance. All other microscopy findings were considered incidental and unrelated to treatment with the test article. There was no microscopic evidence of pulmonary edema or peripheral edema as has been reported in VLS. 33
In the immunogenicity analytical study, a mouse polyclonal anti-ricin IgG was used as the standard to determine rabbit-neutralizing anti-ricin antibody concentration serologically. Therefore, the neutralizing anti-ricin antibody concentrations calculated based on the mouse IgG standard curves are equivalent to the mouse anti-ricin IgG measurement, which represented the relative neutralizing capacity to ricin toxin of the samples but not the true rabbit neutralizing anti-ricin antibody concentrations in the rabbit sera. The immunogenicity analytical results showed that the RVEc stimulated very high immune response in rabbits receiving 4 IM injections of RVEc at 100 or 200 μg. The relative mean neutralization antibody concentration in the rabbit sera collected on study day 99 from these 2 vaccinated groups was >7 mg/mL. There were no nonresponders and no antibody was determined from the diluent control group. The TNA and ELISA analytical results obtained from this immunogenicity study were compatible. The high TNA and ELISA titers were well correlated to survival in challenge studies, showing that there existed a high correlation between protection and serum titers. The TNA measured the neutralizing capacity of anti-ricin antibodies against the ricin toxin. The ELISA measured the total neutralizing and nonneutralizing antibodies to ricin but did not specifically measure neutralizing antibodies. Statistical data (Wannemacher, unpublished results, 2004) showed that anti-ricin IgG concentrations and titers measured by ELISA correlated with the concentrations and titers measured by a radioactive TNA. The correlation coefficient 22 , r, was >.9. Previous studies showed that the improved formulation of rRTA 1-33/44-198 (RVEc) increased both stability and immunogenicity. 34
The high immune response data and preclinical toxicity study results strongly demonstrated that RVEc is safe in rabbits at the doses evaluated. In addition to other standard nonclinical studies, this information may be used to support a clinical evaluation of safety.
Conclusion
Repeated administration of RVEc vaccine by IM injection was well tolerated as there were no toxicologically significant or treatment-related findings noted throughout the study, and appropriate immunogenic/neutralizing antibody titers after repeated administration were observed. Based on the data, the no observed adverse effect level for this study was ≥200 μg of RVEc/dose when administered by repeat IM injection to male and female rabbits. These data provide evidence that the rabbit model is an appropriate model for preclinical safety and immunogenicity evaluation of the RVEc vaccine in preparation for human studies.
Footnotes
Acknowledgments
The authors thank Nahid Torabazari and Rene Aguirre for their technical support and Dr Samad Hedayat (University of Illinois at Chicago) for the statistics support. The views and opinions expressed in this article are those of the author(s) and do not reflect official policy or position of the Department of the Army, Department of Defense, or the US Government.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: the Joint Science and Technology Office for Chemical-Biological defense, Defense Threat Reduction Agency.
