Abstract
2-Amino-4-hydroxyethylaminoanisole and its salt, 2-amino-4-hydroxyethylaminoanisole sulfate, are used as coupling agents in oxidative hair dyes. The Cosmetic Ingredient Review Expert Panel reviewed relevant animal and human data related to the ingredient. The Expert Panel concluded that 2-amino-4-hydroxyethylaminoanisole and 2-amino-4-hydroxyethylaminoanisole sulfate are safe for use in oxidative hair dye formulations. The Expert Panel cautioned that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
Introduction
This report addresses the safety of 2-amino-4-hydroxyethy-laminoanisole and its salt, 2-amino-4-hydroxyethylamin-oanisole sulfate. Both of these cosmetic ingredients function as coupling agents in oxidative hair dyes.
Chemistry
Definition and Structure
The definitions and structures of these 2 ingredients are presented in Table 1.
Names, CAS Registry Numbers, Definitions, and Structures of the Diaminoanisole Ingredients.

2-Amino-4-hydroxyethylaminoanisole and 2-amino-4-hydroxyethylaminoanisole sulfate are commonly used as components of oxidative hair dyes. 1 These ingredients act as “couplers” and react with “precursors.” In a typical formulation, a precursor, such as p-phenylenediamine, is activated via an oxidant, such as hydrogen peroxide. The resultant activated imino-iminium precursor can then proceed to couple with 2-amino-4-hydroxyethylaminoanisole or 2-amino-4-hydroxyethylaminoanisole sulfate to form a new compound (Figure 1). This in situ coupled product is purported to be the actual dye that colors the hair in these types of oxidative hair dyes.
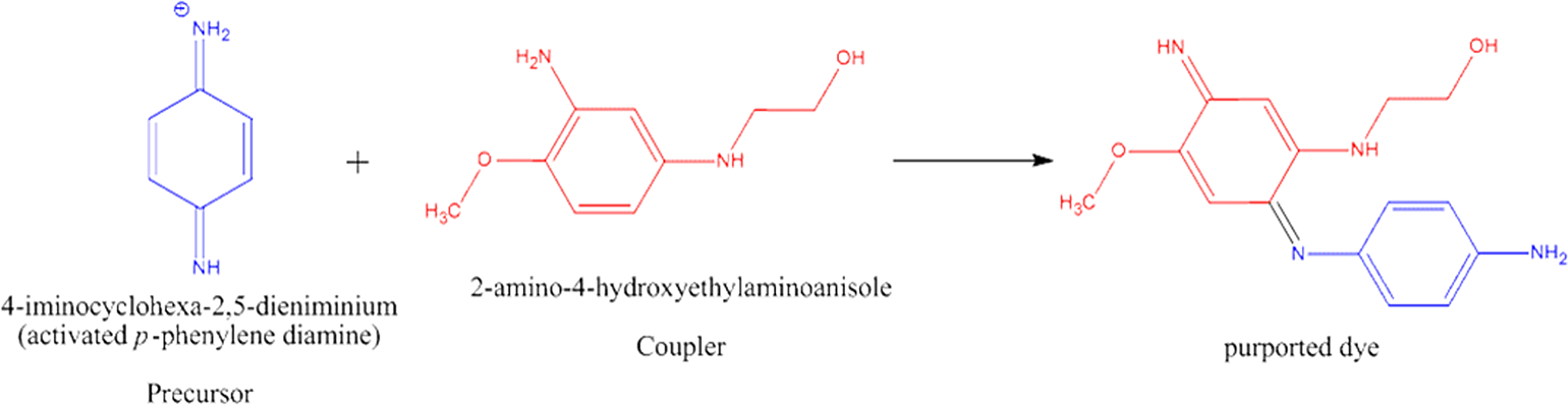
In situ colorant formation. 1
N-Nitrosation and Safety Issues
Although nitrosamine content has not been reported, 2-amino-4-hydroxyethylamino-anisole is a secondary amine and potentially can be nitrosated. Of concern in cosmetics is the conversion (nitrosation) of secondary amines (R1-NH-R2), such as 2-amino-4-hydroxyethylamino-anisole (wherein R1 and R2 are ethanol and methoxyaniline), into N-nitrosamines that may be carcinogenic. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 2 Nitrosation can occur under physiologic conditions. 3 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in the nitrosation of amines in aqueous solution. 4 Accordingly, 2-amino-4-hydroxyethylamino-anisole and 2-amino-4-hydroxyethylaminoanisole sulfate should be formulated to avoid the formation of nitrosamines.
Physical and Chemical Properties
The available information on the physical and chemical properties of 2-amino-4-hydroxyethylaminoanisole and its sulfate salt is presented in Table 2.
Chemical Properties.
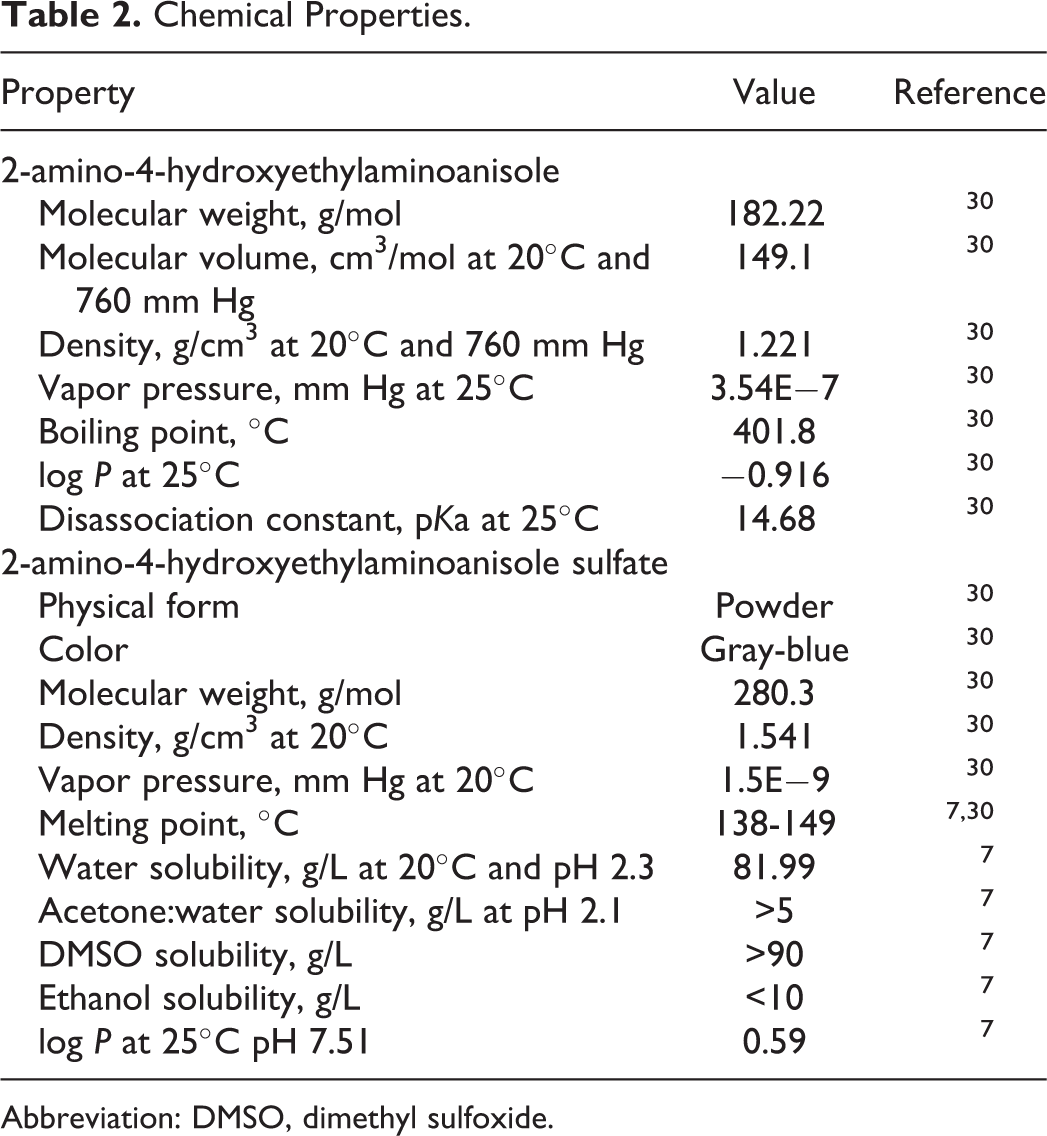
Abbreviation: DMSO, dimethyl sulfoxide.
Method of Manufacture
The manufacture of 2-amino-4-hydroxyethylaminoanisole sulfate can be accomplished via a 6-step synthesis from commercially available 4-methoxyaniline (Figure 2). 5,6 In the first step, 4-methoxyaniline is acetylated with acetic anhydride. The resultant acetamide is then nitrated with nitric and sulfuric acids. Then, N-(4-methoxy-3-nitrophenyl)acetamide is reduced from the amide to the amine, via reflux in hydrochloric acid. Next, the amine is oxidized to a carbamate using β-chloroethyl chloroformate. Then, the resulting chloroethylcarbamate is heated with potassium hydroxide to produce the alcohol. Finally, the nitro group is reduced to the primary amine over Raney nickel and treated with sulfuric acid to produce the salt, 2-amino-4-hydroxyethylaminoanisole sulfate. The free base, 2-amino-4-hydroxyethylaminoanisole, can be easily prepared by neutralizing this salt.
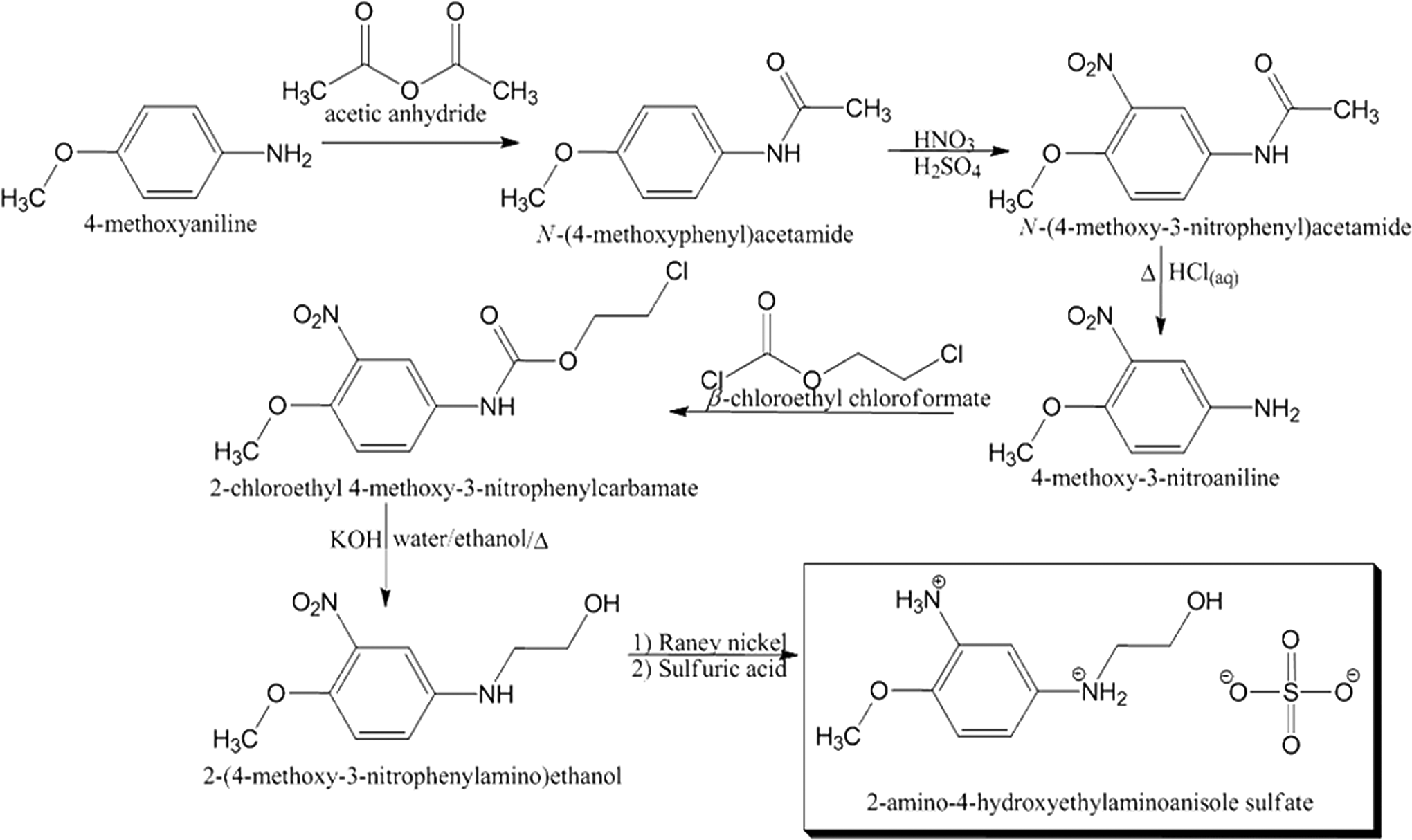
A method of synthesis for 2-amino-4-hydroxyethylaminoanisole sulfate.5,6
Impurities
Purity of 2-amino-4-hydroxyethylaminoanisole sulfate was reported to be 99.3% to 100% with high-performance liquid chromatography (HPLC) at 210 to 304 nm. 7 The impurities 4-methoxyaniline (a starting material), 4-methoxy-3-nitroaniline (a synthesis intermediate), and 2-methoxy-5-nitroaniline (a by-product) were reported to be below the detection limit of 10 ppm; however, 2,4-diaminoanisole (a by-product) was reported at concentrations of 120 to 600 ppm in 4 different test batches. Methanol, ethanol, isopropanol, acetone, ethyl acetate, cyclohexane, methyl ethyl ketone, and monochlorobenzene (solvents used for the synthesis) were not detected at 100 ppm (detection limit).
Use
Cosmetic
According to the information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program, 2-amino-4-hydroxyethylaminoanisole sulfate is used in a total of 94 hair coloring formulations. 8 In a survey of use concentrations conducted by the Personal Care Products Council, 2-amino-4-hydroxyethylaminoanisole sulfate is used at a concentration range of 0.008% to 1.5% (maximum 3% before dilution) in hair coloring products. 9 No uses or use concentrations were reported for 2-amino-4-hydroxyethylaminoanisole.
According to the information provided by the Hair Coloring Technical Committee of the Personal Care Products Council, 2-amino-4-hydroxyethylaminoanisole sulfate is used as an oxidative hair coloring agent. 10 The intended maximum use (“on-head”) concentration is 1.5%. This ingredient and a developer would be mixed at ratios between 1:1 and 1:3 (g dye:g hydrogen peroxide) during the hair dying process. It is general practice to apply 100 g of finished mixed hair dye product for approximately 30 minutes before rinsing off with water and shampoo, and this process may be repeated on a monthly basis.
The Scientific Committee on Consumer Safety (SCCS) for the European Commission concluded that 2-amino-4-hydroxyethylaminoanisole sulfate would not pose a health risk to the consumer when used as an ingredient in oxidative hair dye formulations at a maximum concentration of 1.5%. 7 The SCCS could not exclude the possibility that this ingredient may be sensitizing. The Committee also determined that because 2-amino-4-hyroxyethylaminoanisole sulfate is a secondary amine, it should not be used with nitrosating substances, and the nitrosamine content in the ingredient should be <50 ppb.
Hair Dye Caution Statement—FDA Labeling
2-Amino-4-hydroxyethylaminoanisole and its sulfate salt are considered coal tar hair dyes for which regulations require caution statements and instructions regarding patch tests in order to be exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act. In order to be exempt, the following caution statement must be displayed on all coal tar hair dye products: Caution – this product contains ingredients which may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dyeing the eyelashes or eyebrows; to do so may cause blindness.
Product labels shall also bear a caution statement and patch test instructions for determining whether the product causes skin irritation. The Cosmetic Ingredient Review (CIR) Expert Panel recommends that an open patch test be applied and evaluated by the beautician and/or consumer for sensitization 48 hours after application of the test material and prior to the use of a hair dye formulation.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
In vitro Dermal
In an in vitro study, [14C] 2-amino-4-hydroxyethylaminoanisole sulfate at 1.5% in the presence of hydrogen peroxide, and a reaction partner was applied to excised, dermatomed pig skin. 11 The integrity of the skin was tested by measuring transepidermal water loss prior to test material application. The test substance (100 μg/cm2) was applied to the skin samples for 30 minutes and then washed off with water and shampoo. Measurements for radioactivity in the receptor fluid were made at 16, 24, 40, 48, 64, and 72 hours after application. The upper skin was mechanically separated from the lower. Most of the test material was found in the rinsing solution (mean value ± standard deviation [SD] = 1340.92 ± 50.57 μg/cm2; 92.69%). The test material was also found in the upper skin (2.352 ± 0.824 μg/cm2; 0.162%), the lower skin (0.303 ± 0.219 μg/cm2; 0.021%), and the fractions of receptor fluid were collected over 72 hours (0.409 ± 0.223 μg/cm2; 0.028%). Total recovery of the radiolabeled 2-amino-4-hydroxyethylaminoanisole sulfate was 1446.71 ± 7.86 μg/cm2 (96.59%). The total amount of radiolabeled 2-amino-4-hydroxyethylaminoanisole sulfate that was biologically available was 3.064 ± 0.88 μg/cm2 (0.211%). In a separate analysis of these data, the SCCS found that this study did not follow its requirements and performed a worst case scenario calculation that incorporated 2 SDs for dermal absorption of 2-amino-4-hydroxyethylaminoanisole sulfate, which yielded a value of 4.82 μg/cm2. 7
In Vivo Dermal Non-Human
In an in vivo dermal absorption study, [14C] 2-amino-4-hydroxyethylamino-anisole dihydrochloride was applied to the skin of Sprague-Dawley rats. 12 Groups of 3 male and 3 female rats received the test material at 0.75% in a commercial hair dye with hydrogen peroxide, 0.75% in a commercial hair dye without hydrogen peroxide, or in a 3.47% aqueous solution on a 9 cm2 area. The mean dose for all 3 applications was 0.83 mg/cm2 free base. Application time was 30 minutes, after which the formulations were scraped off, and the test sites were rinsed with approximately 100 mL of 3% shampoo solution and water. The areas were then covered to prevent the animals from licking the test sites during the 72 hours observation period. Urine and feces were collected daily, and the rats were killed 72 hours after application. The application sites, blood, and several organs were collected and analyzed for radioactivity. After complete removal of the skin, the radioactivity in the carcass was measured.
The mean recovery rates for the free base at 0.75% in a commercial hair dye with hydrogen peroxide, 0.75% in a commercial hair dye without hydrogen peroxide, and 3.47% in a aqueous solution were 97.7%, 95.1%, and 99.9%, respectively. Most of the applied dose (93.6%-98.9%) was recovered in the rinse solutions. At the cutaneous application site, 1.51%, 0.57%, and 0.75% of the applied formulation with hydrogen peroxide, without hydrogen peroxide, and in aqueous solution were recovered, respectively. Absorbed material was mainly excreted via the urine at 0.02%, 0.097%, and 0.180% of the applied formulation with hydrogen peroxide, without hydrogen peroxide, and in aqueous solution, respectively, and a very small amount was excreted in feces: 0.008%, 0.027%, and 0.047%, respectively. The amount of test material in the carcasses and organs was close to or below the limit of detection for all the 3 dose groups (0.005%-0.008%). From these observations, it was determined that the absorption rates were 1.54% (12.8 μg/cm2), 0.70% (5.8 μg/cm2), and 0.99% (8.2 μg/cm2) for the formulations with hydrogen peroxide, without hydrogen peroxide, and in aqueous solution. Using the highest absorption rate (12.8 μg/cm2 as free base), the equivalent penetration rate for 2-amino-4-hydroxyethylaminoanisole sulfate was 19.7 μg/cm2. 12 This study was reported by the SCCS not to be good laboratory practice (GLP) compliant. 7
In Vivo Dermal-Human
To the limit of detection (1.6 μg/cm2), no parent compounds or metabolites were found in the blood of 5 female volunteers after application of 2-amino-4-hydroxyethylamino-anisole sulfate plus hydrogen peroxide oxidizing agent, containing 2.2% of the dye to the hair by a professional hair dresser for 15 minutes. 7
Other In Vitro Dose Administration
In an in vitro study, human intestinal epithelial (TC-7) cells were used to determine the bioavailability of 2-amino-4-hydroxyaminoanisole sulfate across the intestinal barrier. 13 Analysis of the donor (apical) and receiver (basolateral) samples was done using HPLC-mass spectrometry (MS)/MS, and the apparent permeability coefficient (P app) was calculated for 2 independent experiments. 14 C-mannitol (∼4 µmol/L) was used to demonstrate the integrity of the cell monolayer. Only monolayers revealing a permeability of < 2.5 × 10−6 cm/sec were used. According to the laboratory’s classification system, a P app < 2 × 10−6 cm/sec indicates low permeability. Ranitidine, which has a 50% absorption in humans, was used as a low permeability reference compound, and it and another reference compound, propranolol, were well within the acceptance range and validated the study. The P app for 2-amino-4-hydroxyethylaminoanisole sulfate was 73.3 × 10−6 cm/sec, which equates to a high permeability classification (P app ≥ 20 × 10−6 cm/sec) by this laboratory’s classification system. It was concluded that 2-amino-4-hydroxyethylaminoanisole is readily absorbed in the gastrointestinal tract after oral administration. 7
Toxicological Studies
Acute Toxicity
Oral—Non-Human
In an acute oral toxicity study, doses of 2-amino-4-hydroxyethylaminoanisole sulfate were admin-istered to Wistar rats and CF-1 mice. 14 The dose groups for the rats (5 rats per sex per dose group) and mice (10 females per dose group) were 250, 375, 500, 625, and 750 mg/kg body weight. There was an additional dose group of 875 mg/kg body weight in the mice. Doses were established after a range finding study in mice found the median lethal dose (LD50) to be less than 875 mg/kg body weight. The test material was administered once by oral gavage. Mortality and clinical signs of toxicity were recorded during the 14-day observation period. Body weights were recorded weekly. All animals were necropsied. Clinical signs of toxicity observed after dosing included tonic spasm, piloerection, and higher respiratory rate in both rats and mice. In all, 3 male and 2 female rats and 2 mice in the 375 mg/kg, 4 male and 2 female rats and 6 mice in the 500 mg/kg, all male and 4 female rats and 5 mice in the 625 mg/kg, and all male and female rats and 9 mice in the 750 mg/kg dose groups died between the first 24 hours and 6 days after dosing. Additionally, all mice in the 875 mg/kg dose group died. No macroscopic changes were noted at necropsy. No data were provided regarding control groups. The LD50 of 2-amino-4-hydroxyethylaminoanisole sulfate in male rats, female rats, and female mice was calculated to be 475, 588, and 538 mg/kg body weight, respectively. This study was reported by the SCCS not to be GLP compliant. 7
In another acute oral toxicity study, groups of 5 male and 5 female Naval Medical Research Institute (NMRI) white mice received 125, 250, 500, 750, or 1000 mg/kg 2-amino-4-hydroxyethylaminoanisole sulfate by gavage. 15 Doses were established after a range finding study in mice found the LD50 to be less than 2000 mg/kg body weight. The test material was administered as a 1.25% to 10% dilution in deionized water. Mortality and clinical signs of toxicity were recorded during the 14-day observation period. Clinical signs of toxicity included those related to the central nervous system, coordination, reflexes, and autonomic functions with dose-dependent severity up to 72 hours after administration. Weight gains were reduced in all surviving animals. One male and 1 female in the 125 mg/kg, 2 males and 1 female in the 250 mg/kg, 4 males and 3 females in the 500 mg/kg, 3 males and all females in the 750 mg/kg, and all males and 4 females in the 1000 mg/kg dose groups died within 24 to 72 hours of dosing. No macroscopic changes were noted at necropsy. No data were provided regarding control groups. The calculated LD50 of 2-amino-4-hydroxyethylaminoanisole sulfate in male mice was 333 mg/kg body weight and in female mice was 351 mg/kg body weight. An LD50 for male and female mice combined was calculated to be 327 mg/kg body weight.
Repeated Dose Toxicity
Dermal—Non-Human
The dermal toxicity of 2-amino-4-hydroxyethylaminoanisole sulfate was evaluated in a 28-day study in specific pathogen free (SPF) Pirbright White guinea pigs. 16 Dose groups of 5 animals of each sex received 0, 50, 150, or 300 mg/kg body weight of the test material in tap water at a dose volume of 1 mL/kg body weight. The test material was applied once daily to a 3 × 4 cm2 area on clipped dorsal skin. The animals were checked twice daily for mortality. Clinical signs of toxicity were recorded daily, and body weights were recorded weekly. Complete hematology and blood chemistry investigations and urinalysis were performed on day 0 and day 28. All animals were killed at the end of the treatment period. Selected organs were weighed, and a detailed necropsy was performed in all animals. Hearts and kidneys of the control and high-dose group animals were studied histopathologically. Additionally, all gross lesions observed in the liver and skin of all dose groups were examined microscopically.
No deaths occurred and no relevant clinical signs were observed. Body weight gains were comparable to the control group. No treatment-related changes were observed in hematology, blood chemistry parameters, or urinalysis. No gross lesions were noted at necropsy, and organ weights were comparable to the control group. The NOAEL for this 28-day dermal study of 2-amino-4-hydroxyethylaminoanisole sulfate was 300 mg/kg body weight. 16
Oral—Non-Human
The oral toxicity of 2-amino-4-hydroxyethylaminoanisole sulfate was investigated in Wistar HanBrl: WIST (SPF) rats. 17 Dose groups were comprised of 15 animals of each sex and received 0, 15, 50, or 200 mg/kg body weight of the test material in distilled water at a dose volume of 10 mL/kg body weight. The test material was administered by oral gavage once daily for 108/109 days. Clinical signs of toxicity, feed consumption, and body weights were recorded weekly. Ophthalmoscopic examinations were performed before and after the treatment period. At week 15, functional observational battery, locomotor activity, and grip strength were tested, as were thyroid hormone levels (5 rats/sex/dose). Complete hematology and blood chemistry investigations and urinalysis were performed at the completion of the dosing period. All animals were killed at the end of the treatment period. Selected organs were weighed, and a detailed necropsy was performed in all animals. Histological examinations were performed on organs and tissues of the control and high-dose group animals and on all gross lesions from all animals. Additionally, thyroids, spleens, kidneys, and pituitary glands were examined in the mid- and low-dose groups.
All animals survived until necropsy. Blue discoloration of the urine was noted in the female rats of the 15 mg/kg dose group as well as all animals in the 50 and 200 mg/kg dose groups. No clinical signs of toxicity were observed. No irregularities were observed during the functional observational battery or ophthalmoscopic examinations. There were no treatment-related changes in feed consumption or body weight gains. Thyroid hormone levels were comparable to the control group.
In the 200 mg/kg body weight dose group, both sexes had slight anemia with compensatory reticulocytosis that presented as decreased red blood cell counts, decreased hemoglobin, increased methemoglobin, decreased hematocrit levels, and increased reticulocyte counts and maturity indices. Decreased creatinine, increased triglyceride sodium and chloride concentrations, slight proteinuria, and increased bilirubin and nitrite were observed in both the sexes of this dose group as well. Marginally increased thyroid-to-brain weight ratios were observed in both the sexes, and in females, increased mean absolute and relative liver, kidney, and spleen weights were noted. Follicular cell enlargement of the thyroid gland was observed in both the sexes of the 200 mg/kg dose group. Additionally, pigment storage and tubular swelling with necrosis of tubular cells and basal membrane thickening were observed in the kidneys, and increased mean grade of extra medullary hemopoiesis was observed in the spleen. Slight hypertrophy of chromophobic cells of the pituitary gland was seen in males of the 200 mg/kg dose group. Similar adverse effects were observed in the 50 mg/kg body weight dose group but occurred mostly in the female rats. No adverse effects were noted in hematology, blood chemistry, or gross necropsy of the 15 mg/kg body weight dose group. Due to the slight anemia and morphological and histological changes in the thyroid gland, kidneys, and pituitary gland in the 50 mg/kg dose group, the NOAEL was determined to be 15 mg/kg body weight of 2-amino-4-hydroxyethylaminoanisole sulfate. 17
Reproductive and Developmental Toxicity
Mated female Wistar HanBrl: WIST (SPF) rats received 2-amino-4-hydroxyethylaminoanisole sulfate by gavage on days 6 to 20 of gestation. 18 The doses used were based on the results of a range finding study. In the main study, groups of 22 rats received 0, 10, 30, or 150 mg/kg body weight of the test material in bidistilled water. Maternal clinical signs were monitored twice daily. Body weights were recorded daily, and food consumption was measured over 3-day periods. Dams were killed on gestation day 21. Complete necropsy and macroscopic examination of the organs were performed. Gravid uterus weights were determined, and fetuses were removed, sexed, weighed, and examined externally. Implantation sites, resorption sites, and live and dead fetuses were recorded. Half of the fetuses were examined for soft-tissue abnormalities and half for skeletal abnormalities.
One death in the high-dose group was considered the result of a dosing error. No other treatment-related clinical signs of toxicity were observed. Urine of the 30 and 150 mg/kg dose groups was darkly discolored. A slight decrease in mean food consumption was observed for the entire treatment period in the 150 mg/kg dose group. This group also had slightly reduced body weight gain up to gestation day 16. No maternal treatment-related effects were observed at gross necropsy. There were also no treatment-related effects observed viscerally or skeletally in the fetuses. Uterus and placenta weights, number of corpora lutea, and implantations were similar to controls in all dose groups. Litter size, fetal mortality, fetal body weight, and sex ratio were also comparable to the controls. The maternal NOAEL was 30 mg/kg body weight, and the fetal NOAEL was 150 mg/kg body weight. 18
Genotoxicity
In Vitro
The mutagenic potential of 2-amino-4-hydroxyethylaminoanisole sulfate was studied in an Ames test using Salmonella typhimurium strains TA98, TA100, TA102, TA1535, and TA1537, with and without S9 metabolic activation. 19 The test concentrations were 33, 100, 333, 1000, 2500, or 5000 μg/plate. The positive controls were 4-nitro-o-phenylenediamine, sodium azide, methyl methane sulfonate, and 2-aminoanthracene. Decreased background growth was observed at 2500 and 5000 μg/plate. No biologically relevant increases in revertant colony numbers were observed in any test strain at any dose level, with or without metabolic activation. Controls yielded expected results. It was concluded that 2-amino-4-hydroxyethylaminoanisole sulfate was not mutagenic in this assay.
The mutagenic activity of 2-amino-4-hydroxyethylaminoanisole sulfate was studied in mouse lymphoma L5178Y TK+/− cells at the tk locus. 20 The concentrations ranged from 0.5 to 100 μg/mL without S9 metabolic activation (precipitation was observed without S9 at ≥ 50 μg/mL) and 1.0 to 500 μg/mL (with S9 metabolic activation). The vehicle control was culture medium. The positive controls were benzo[α]pyrene with S9 and ethylmethanesulfonate without S9. The cultures were incubated with the test material for 4 hours. Mutation frequency and cell survival were determined as was as the size/optical density of colonies and the ratio of small versus large colonies. A biologically significant increase in the number of mutant colonies was observed with and without S9. A biologically relevant shift toward small colonies indicating a clastogenic effect was observed following treatment with the test material, with and without metabolic activation. The controls yielded expected results. It was concluded that 2-amino-4-hydroxyethylaminoanisole sulfate was mutagenic in this mouse lymphoma assay.
The genotoxic potential of 2-amino-4-hydroxyethylaminoanisole sulfate was studied in a micronucleus test using human peripheral blood lymphocytes. 21 The test material was tested at concentrations of 25, 100, and 150 µg/mL with S9 metabolic activation and at concentrations of 3, 5, and 8 µg/mL without S9 metabolic activation. The positive controls were cyclophosphamide with S9 and 4-nitroquinoline-1-oxide and vinblastine without S9. Cells were incubated with the test material 24 hours after mitogen stimulation with phytohemagglutinin. Incubation for cells with metabolic activation was 3 and 20 hours for cells without metabolic activation. Cells were harvested 72 hours after mitogen stimulation. The replication index (RI) was calculated from the proportions of mononucleate, binucleate, and multinucleate cells in 500 cells per replicate. One thousand binucleate cells from each culture were analyzed for the occurrence of micronuclei.
The RI at the highest concentration tested with and without metabolic activation were 63% and 68%, respectively. With S9, a concentration-related increase in the frequency of micronucleated binucleate (MNBN) cells was observed with statistical significance at 100 and 150 µg/mL. The frequency of MNBN also exceeded historical control range in single cultures in the same concentrations. Without S9, the frequencies of MNBN were similar to concurrent controls at all concentrations and within the historical range for vehicle controls. A small, borderline increase in the frequency of MNBN at 8 µg/mL was observed, which exceeded historical vehicle controls in one culture only. The effect was considered equivocal due to the high level of cytotoxicity (68%) at this concentration. In this study, 2-amino-4-hydroxyethylaminoanisole sulfate was considered to be genotoxic. 21
In Vivo
The genotoxic potential of 2-amino-4-hydroxyethylaminoanisole sulfate was studied in a micronucleus test using NMRI mice. 22 A dose range finding experiment preceded the main study. In the main study, groups of 5 mice of each sex received single intraperitoneal injections of 0, 20, 100, or 200 mg/kg body weight of 2-amino-4-hydroxyethylaminoanisole sulfate in distilled water, with an additional group of mice receiving 200 mg/kg dose. Control groups received distilled water or 40 mg/kg body weight cyclophophamide in 0.9% NaCl. Bone marrow cells were collected at 24 or 48 hours (for the 200 mg/kg dose group). At least 2000 polychromatic erythrocytes per animal were analyzed, and the ratio between polychromatic and total erythrocytes per animal was determined by counting at least 200 immature polychromatic erythrocytes per animal. In the dose range finding study, toxic effects observed at 200 mg/kg included palpebral closure and lethargy within the first hour of treatment. Also in the range finding study, a dose of 400 mg/kg caused mortality, and signs of systemic toxicity were observed in both 400 mg/kg and 200 mg/kg. In the main study, no treatment-related mortalities or clinical signs of toxicity were observed. There was no statistically significant increase in micronuclei in the treatment groups when compared to the controls. The authors concluded that up to 200 mg/kg body weight of 2-amino-4-hydroxyethylaminoanisole sulfate was not genotoxic in this micronucleus assay.
The potential for 2-amino-4-hydroxyethylaminoanisole sulfate to induce unscheduled DNA synthesis (UDS) was assessed using male Wistar rats. 23 A dose range finding experiment preceded the main study. In the main study, groups of 5 rats received single oral doses of 75 or 750 mg/kg body weight 2-amino-4-hydroxyethylaminoanisole sulfate in bidistilled water. Control groups received 10 mL/kg body weight bidistilled water or 100 mg/kg body weight of 2-acetylaminofluorene. Rats were killed either 4 hours after treatment (one group, 750 mg/kg dose group) or 16 hours after treatment (75 mg/kg dose group and additional 750 mg/kg dose group). Liver perfusion was performed. At least 5 primary hepatocyte cultures were made from each animal and exposed for 4 hours to 3H-thymidine. The dye-exclusion method was utilized to determine whether any liver cell toxicity occurred. After the radiolabel exposure, the cells were washed, and the slides were prepared. At least 2 slides per animal were evaluated from the occurrence of UDS for 3 animals per dose group, which equated to 100 cells/animal. Heavily labeled S-phase cells were excluded from counting, and background grains were subtracted from the grains observed from the nucleus to obtain relevant net nuclear grains.
In the range finding study, mortality occurred at 1000 mg/kg within 24 hours of dosing. Survivors in this group had reduced spontaneous activity, eyelid closure, and piloerection. At 750 mg/kg, there was no mortality. The kidneys, urine, and liver of the rats showed dark discoloration. In the main study, no mortality or clinical signs of toxicity were observed. No UDS induction was observed in the hepatocytes of the treated animals at any dose level or time period when compared to controls. There was no increase in the number of nuclear grains or the resulting net grains at either the dose or the time period. The authors concluded that 2-amino-4-hydroxyethylaminoanisole sulfate did not induce DNA damage in this UDS assay. 23
Irritation and sensitization
Irritation
Dermal—Non-Human
In a dermal irritation study, 0.5 g of a commercial hair dye formulation that contained 3% 2-amino-4-hydroxyethylaminoanisole sulfate was applied to shaved skin (∼6 cm2) of 3 New Zealand White rabbits. 24 The test sites were then semioccluded for 4 hours, after which the test material was washed off with water. The skin was evaluated for reactions at 30 minutes, 1, 24, 48 and 72 hours, and then daily up to 14 days after the material was removed. Slight erythema (scores of 1 and 2) and very slight edema (score of 1) were recorded at several observation periods. These effects had completely disappeared within 7 days. In each rabbit, the mean 24-, 48-, 72-hour scores for erythema and edema were 1.33 and 1.0, 1.67 and 1.0, and 1.33 and 1.0, respectively. The authors concluded that 2-amino-4-hydroxyethylaminoanisole sulfate was a mild transient skin irritant when tested at 3%.
In another dermal irritation study, 1% 2-amino-4-hydroxyethylaminoanisole sulfate suspended in gum Arabic was applied to 10 female SPF white guinea pigs. 25 The test material was applied with a brush 3 times a day for 20-minute durations on 2 consecutive days on the left and right clipped flanks of the animals. The treatment period was followed by a 3-day observation period. Any effects were scored according to the Draize method. Very slight erythema was observed in 2 of the animals on the second treatment day. No edema was observed. It was concluded that 2-amino-4-hydroxyethylaminoanisole sulfate at 1% was essentially nonirritating. This study was reported not to be GLP compliant. 7
Ocular
A hair dye formulation containing 3% 2-amino-4-hydroxyethylaminoanisole sulfate was tested for ocular irritation potential in 3 New Zealand White rabbits. 26 The conjunctival sac of one eye of each rabbit was instilled with 0.1 mL of the test substance and not rinsed. The untreated eye served as a control. Both eyes were examined at 1, 24, 48, and 72 hours posttreatment according to the Draize method. No cornea or iris effects were noted at any observation time. Slight conjunctival redness (score of 1) was noted at 24 hours in all animals, but these effects were gone within 3 days of treatment. In each rabbit, the mean 24-, 48-, and 72-hour scores for conjunctival erythema were 0.67, 0.33, and 0.67, respectively. The study concluded that 3% 2-amino-4-hydroxyethylaminoanisole sulfate was a transient and mild irritant in the eyes of rabbits.
In another ocular irritation study, a 1% aqueous solution of 2-amino-4-hydroxyethylaminoanisole sulfate was tested in 10 female Pirbright White (SPF) guinea pigs. 27 The conjunctival sac of the right eye of the guinea pigs was instilled with 0.1 mL of the test substance and not rinsed. The left eye of the animals was left untreated and served as a control. At 24 hours after application, the eyes were washed with fluorescein-sodium solution. Redness and discharge were observed in 5 of the guinea pigs 30 minutes after treatment. Redness was also observed in 2 animals at 7 hours posttreatment, but this effect cleared at 24 hours. No other ocular effects were observed. It was concluded that 1% 2-amino-4-hydroxyethylaminoanisole sulfate caused transient conjunctival irritation in this study. It should be noted that this study was reported not to be GLP compliant. 7
Sensitization
Dermal—Non-Human
A local lymph node assay (LLNA) was performed using 2-amino-4-hydroxyethylaminoanisole sulfate dissolved in dimethyl sulfoxide (DMSO). 28 The CBA/Ca female mice were divided into groups of 5 and received 0.25%, 0.5%, 1%, or 2% of the test material on the ear surface (25 µL) once daily for 3 consecutive days. After each application, the ears were dried with a hair dryer for ∼5 minutes. A positive control group received 0.25%, 0.5%, 1%, or 2% p-phenylenediamine in DMSO. Five days after the initial topical treatment, all animals were intravenously injected with 250 µL of phosphate-buffered saline containing 20 µCi of [3H] methyl thymidine. Approximately 5 hours after injection, the animals were killed, and the auricular lymph nodes were excised. Single-cell suspensions were prepared from pooled lymph nodes, with the cells precipitated by trichloroacetic acid, and radioactivity was measured by liquid scintillation. The stimulation indices (SI) were calculated.
No clinical signs of toxicity or deaths occurred during the treatment period in any dose group. The SI were1.29, 1.03, 1.12, and 1.42 for the 0.25%, 0.5%, 1%, and 2% dose groups, respectively. The estimated concentrations for SI of 3 (EC3) could not be calculated. The positive control group produced expected results and validated the study. The authors concluded that 2-amino-4-hydroxyethylaminoanisole sulfate tested up to 2% in DMSO was not a skin sensitizer. 28
Quantitative Structure–Activity Relationship
A topological structural molecular design (TOPS-MODE) quantitative structure–activity relationship (QSAR) model was utilized to predict the sensitization potential of all hair dye ingredients registered in Europe (229 substances as of 2004). 29 The model predicted 2-amino-4-hydroxyethylaminoanisole to be a weak sensitizer. The sensitization potential of the sulfate salt was not evaluated.
Clinical Use
Epidemiology
2-Amino-4-hydroxyethylaminoanisole and its sulfate salt are oxidative hair dye ingredients. Although the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair dye use and disease, such studies do provide broad information. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers.
A detailed summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/findings.shtml.
Risk Assessment
The SCCS calculated the margin of safety for 2-amino-4-hydroxyethylaminoanisole sulfate to be 267. 7 To obtain this value, the systemic exposure dose of 0.056 mg/kg was determined based on the maximum absorption through the skin (4.82 μg/cm2 = the mean ± 2SD from an in vitro percutaneous absorption study), typical body weight (60 kg) and skin surface area (700 cm2 for scalp surface area), and dermal absorption per treatment (3.374 mg, calculated using the values for maximum absorption and average body weight and skin surface area assumptions), which was then divided into the NOAEL of the 15-week rat (repeated dose oral toxicity) study (15 mg/kg).
Summary
The cosmetic ingredients 2-amino-4-hydroxyethylaminoanisole and its salt, 2-amino-4-hydroxyethylaminoanisole sulfate, function as coupling agents in oxidative hair dyes. The free base currently has no reported uses by the FDA or the cosmetics industry. The sulfate salt is used in a total of 94 hair coloring formulations at a concentration range of 0.008% to 1.5%. 2-amino-4-hydroxyethylaminoanisole is not in current use.
In an in vitro percutaneous absorption study in dermatomed pig skin, 3.064 ± 0.88 μg/cm2 (the mean ± SD; 0.211%) of 1.5% radiolabeled 2-amino-4-hydroxyethylaminoanisole sulfate in an oxidative hair dye formulation with a reaction partner was found to be biologically available. An in vivo study in rats found the equivalent penetration rate of radiolabeled 2-amino-4-hydroxyethylaminoanisole sulfate, which was calculated from the measured penetration rate of the free base, to be 19.7 μg/cm2. A bioavailability study of 2-amino-4-hydroxyethylaminoanisole sulfate in human intestinal epithelial cells concluded that this chemical was readily absorbed in the gastrointestinal tract after oral administration.
An acute oral toxicity study of 2-amino-4-hydroxyethylaminoanisole sulfate determined LD50 values of 475 and 588 mg/kg for male and female rats, respectively. An LD50 of 538 mg/kg was calculated in female mice. Clinical signs of toxicity observed after dosing included tonic spasm, piloerection, and higher respiratory rate in both rats and mice. In another acute oral toxicity study, the calculated total LD50 of 2-amino-4-hydroxyethylaminoanisole sulfate in male and female mice combined was 327 mg/kg.
The NOAEL for a 28-day dermal study of 2-amino-4-hydroxyethylaminoanisole sulfate in guinea pigs was 300 mg/kg body weight, which was the highest dose tested. In a 15-week oral study in rats, the NOAEL was determined to be 15 mg/kg body weight of 2-amino-4-hydroxyethylaminoanisole sulfate. Slight anemia and morphological and histological changes in the thyroid gland, kidneys, and pituitary gland were observed at higher dose levels.
In a teratogenicity study where female rats received 2-amino-4-hydroxyethylaminoanisole sulfate by gavage at doses of 0, 10, 30, or 150 mg/kg body weight of the test material in bidistilled water, the maternal NOAEL was 30 mg/kg body weight, and the fetal NOAEL was 150 mg/kg body weight. Dams in the high-dose group experienced effects on food consumption and body weight gains.
The ingredient 2-amino-4-hydroxyethylaminoanisole sulfate was not mutagenic in an Ames assay but was found to be mutagenic in an in vitro mouse lymphoma assay and in a micronucleus study using human peripheral blood lymphocytes. In in vivo studies, 2-amino-4-hydroxyethylaminoanisole sulfate was not genotoxic in a mouse micronucleus test, and it did not induce DNA damage in a UDS assay in rats.
In dermal irritation studies, 2-amino-4-hydroxyethylaminoanisole sulfate at 1% was essentially nonirritating to guinea pig skin; however, mild transient skin irritation was observed at 3% in rabbits. At these concentrations, 2-amino-4-hydroxyethylaminoanisole sulfate was a transient and mild irritant in the eyes of rabbits and guinea pigs. Using a LLNA, 2-amino-4-hydroxyethylaminoanisole sulfate tested up to 2% in DMSO was not a skin sensitizer.
A QSAR model predicted 2-amino-4-hydroxyethylaminoanisole to be a weak sensitizer.
The most recent CIR review of available epidemiology studies concluded that there is insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers.
A margin of safety calculation for 2-amino-4-hydroxyethylaminoanisole sulfate by the European Union’s SCCS yielded a value of 267 compared to the NOAEL for repeated dose oral toxicity in rats.
Discussion
The available data on 2-amino-4-hydroxyethylaminoanisole sulfate are sufficient; however, the Expert Panel noted gaps in the available safety data for the free base, 2-amino-4-hydroxyethylaminoanisole, in this safety assessment. The toxicological profile for the sulfate salt may be extended to the free base; and therefore, the data for the sulfate salt can be used to support the safety of both the ingredients.
The Expert Panel recognized that 2-amino-4-hydroxyethylaminoanisole and its sulfate salt function as hair dye ingredients, and that limited irritation and sensitization data are available. However, hair dyes containing these ingredients, as coal tar hair dye products, are exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act, when the label bears a caution statement and patch test instructions for determining whether the product causes skin irritation. The Expert Panel expects that following this procedure will identify prospective individuals who would have an irritation/sensitization reaction and allow them to avoid significant exposures.
Although nitrosamine content of these hair dyes has not been reported, the Expert Panel noted these ingredients are secondary amines and can be potentially nitrosated. Accordingly, their use should be restricted to hair dye formulations to avoid the formation of N-nitroso compounds.
In considering hair dye epidemiology data, the CIR Expert Panel concluded that the available epidemiology studies are insufficient to conclude there is a causal relationship between hair dye use and cancer or other toxicologic end points, based on the lack of strength of the associations and inconsistency of findings.
The CIR Expert Panel noted that the use of oxidative hair dye formulations involves exposure to precursors and coupling agents as well as to their reaction products. Specifically, 2-amino-4-hydroxyethylaminoanisole sulfate is a coupler that reacted with a precursor in the presence of an oxidizing agent to produce the final dye product. Although reaction intermediates may be formed, human exposure is to the precursors and coupling agents and to reaction products and not to the reaction intermediates. The exposures to the precursors and couplers are low (they are consumed in the color forming reaction), and the exposures to reaction products are even lower (they are adsorbed into the hair shaft itself and physically retained there). Therefore, safety assessments of oxidative hair dyes are driven by the toxicological evaluation of the ingredients (ie, precursors and coupling agents), more than by the reaction products formed during use and not at all by reaction intermediates.
Conclusion
The CIR Expert Panel concluded that 2-amino-4-hydroxyethylaminoanisole and 2-amino-4-hydroxyethylaminoanisole sulfate are safe for use in oxidative hair dye formulations. Were the free base (2-amino-4-hydroxyethylaminoanisole) to be used in the future, the expectation is that it would be used at concentrations similar to the sulfate salt. The Expert Panel cautions that these ingredients should not be used in hair dye products in which N-nitroso compounds may be formed.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
