Abstract
The estrogenic potential of lavender oil was evaluated in a percutaneous uterotrophic bioassay in immature female rats. Four groups of 10 immature female rats each were randomly selected on postpartum day (PPD) 16. During the 3-day treatment period (PPDs 19-21), the immature rats were separated from the dams, caged in groups of 5 in a litter box for 6 hours, and administered the vehicle control article (corn oil) or lavender oil at 20 or 100 mg/kg per day. All dosages were administered as a 5 mL/kg volume in a Hilltop Chamber (25 mm diameter; absorbent material removed) placed on the shaved back of each immature rat, and secured with micropore tape and Vetrap. A positive control group was gavaged twice daily with 2.5 μg/kg per day of 17α-ethinyl estradiol. Daily observations included viability, clinical signs, body weights, and body weight gains. All rats were euthanized 24 hours after the third and final treatment, the uteri and ovaries were removed, and the paired ovaries and wet and blotted uterine weights were recorded. No unscheduled deaths occurred. No skin reactions were observed. Both dosages of lavender oil significantly reduced body weight gains after the third day of treatment, but terminal body weights and mean absolute and relative uterine weights did not differ significantly from vehicle control values. Positive controls showed significant increases in body weight and increased mean absolute and relative uterine weights as expected. Based on these data, lavender oil, at dosages of 20 or 100 mg/kg, was not active in the rat uterotrophic assay and gave no evidence of estrogenic activity.
Introduction
A report by Henley et al 1,2 suggested that lavender oil in hair- and skin-care products may be associated with abnormal development of breasts (gynecomastia) in prepubertal boys. The pediatric endocrinologist based his report on 3 boys (age 4-10) with gynecomastia who had used topical products that listed lavender oil among their ingredients. Researchers at the National Institute of Environmental Health Sciences (NIEHS) then studied these materials in an in vitro cell culture system and found that the materials turned on estrogen-regulated genes and inhibited an androgen-regulated gene. The NIEHS scientists concluded that lavender oil had weak estrogenic and antiandrogenic activities in vitro that may have contributed to an imbalance in estrogen and androgen pathway signaling. 1 This conclusion was rebutted in letters to the editor 3 –6 and by Lawrence. 7 –9 Lavender oil is a pale yellow, viscous liquid with a sweet fragrance, which is used commercially in balms, perfumes, and cosmetics since the 1920s. Lavender oil is approved by the US Food and Drug Administration (FDA) as generally recognized as safe (GRAS) in the Code of Federal Regulations Title 21 Part 182.20 under the current use conditions as a flavor. Lavender oil is produced by steam distillation of the flowering stalks of Lavandula officinalis Chaix 10 and has been extensively tested for safety in animals and humans. 11 –14
The worldwide volume of use of lavender oil as a fragrance ingredient is greater than 10 metric tons. 15 The 97.5 percentile use level in fragrance formulae in cosmetics generally has been reported to be 0.74%, 16 which would result in a maximum human skin exposure of 0.019 mg/kg per day for high-end users of multiple cosmetic products containing lavender oil (Table 1). The dermal systemic exposure for fragrance materials is based on the quantities of cosmetics used, the frequency of use, the concentration of the fragrance material in these products, and assumed 100% skin permeation. 17 How these data are obtained and exposure determined is reported by Cadby et al. 18 The major components of lavender oil reported by Lawrence 19 are linalool and linalyl acetate, and on average these 2 components add up to 63% of lavender oil. Linalool was evaluated for in vitro human skin permeation in 6 different vehicles. Under the most severe conditions (under occlusion in a 70: 30 ethanol: water vehicle) only 14.4% linalool was absorbed over a 24-hour period. 20,21 Jager et al 13 showed that linalyl acetate and linalool from lavender oil are absorbed through the skin at similar rates. As such, correcting for skin absorption the maximum daily exposure is 0.003 mg/kg per day for high-end users of multiple cosmetic products containing lavender oil.
Calculation of the Total Human Skin Exposure From the Use of 10 Cosmetic Products Containing Lavender Oil.
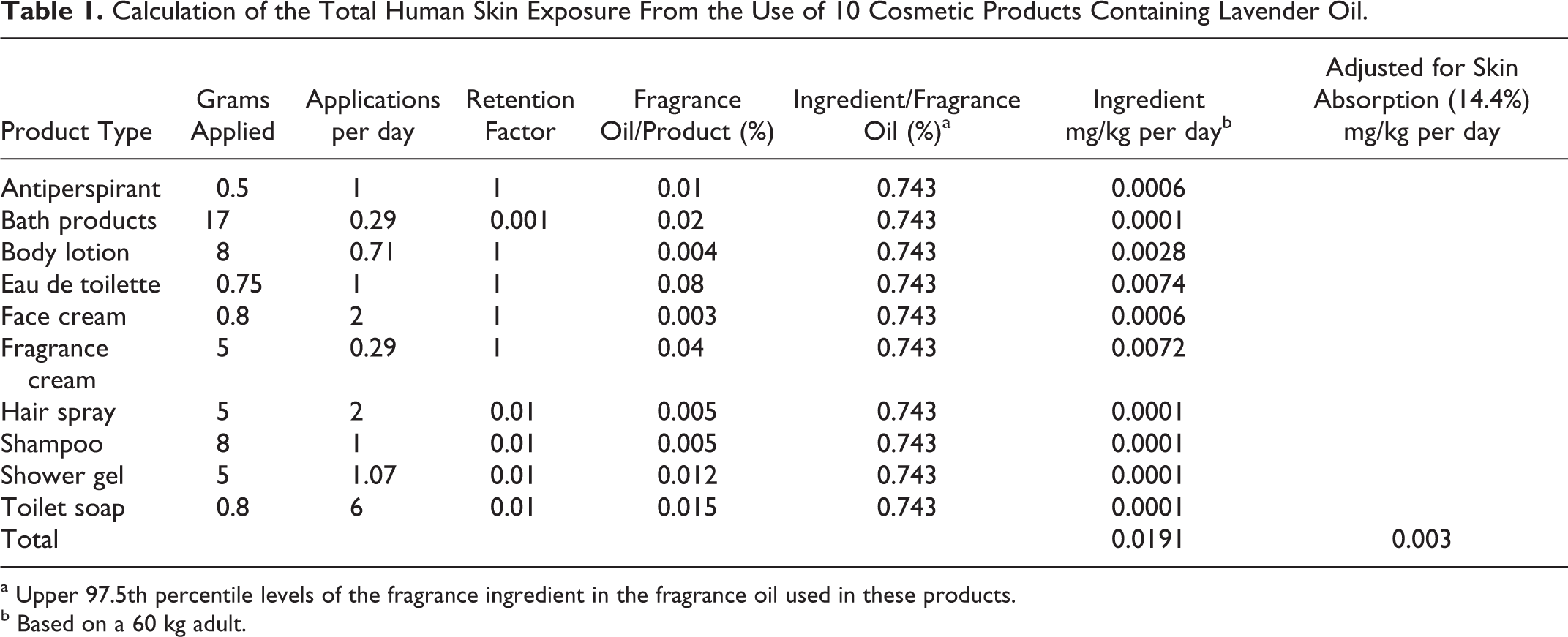
a Upper 97.5th percentile levels of the fragrance ingredient in the fragrance oil used in these products.
b Based on a 60 kg adult.
The 3 case reports 1 raise questions regarding lavender and tea tree oil as potential endocrine disruptors, and the data from the in vitro studies indicated weak estrogenic and antiandrogenic activity of the oils. However, further research is necessary before any conclusive causal relationship can be identified between the essential oils and gynecomastia. Toward this end, a uterotrophic assay of lavender oil by dermal application to rats was initiated. Dermal application was selected to mimic the exposure of the boys described in the report by Henley et al. 1 This higher level test, conducted in vivo and considered to be the benchmark animal assay for estrogenic effects, 22 is the subject of the present report.
The purpose of the study was to evaluate lavender oil, an essential oil, in a uterotrophic assay in Crl: CD(SD) immature female rats. The assay was based on methodology described in the draft guideline, the Uterotrophic Assay of the Organization for Economic Co-operation and Development 22 (Note: Organization for Economic Cooperation and Development [OECD] Testing Guideline 440 was finalized October 16, 2007), and was conducted in compliance with the Good Laboratory Practice regulations of the FDA 23 and the OECD Environment Directorate. 24
Materials and Methods
Materials
Lavender oil (CAS# 8000-28-0; 100%), a pale yellow, viscous liquid (Lot No. 731454), was supplied by Mastertaste Inc (Teterboro, New Jersey). Corn oil (Acros Organics, Fairlawn, New Jersey) was the vehicle and negative control article. The positive control article, 17α-ethinyl estradiol (CAS# 57-63-6; ≥98%), was purchased from Sigma-Aldrich, Inc, St Louis, Missouri (Lot No. 024K1196). The test and positive control articles were stored at room temperature and protected from light; corn oil was refrigerated (2°C-8°C). Dosing formulations were prepared daily from bulk materials on the 3 treatment days, and samples were analyzed on the first and last days of treatment for test article content by Charles River Laboratories Preclinical Services.
Animals
Four Crl: CD(SD) lactating dams from Charles River Laboratories, Inc (Portage, Michigan), each with 10 cross-fostered, 12- to 13-day-old female pups were used in the study (day of birth = postpartum day [PPD] 1). Each dam and its cross-fostered litter was assigned to an individual litter box and randomly designated as group I, II, III, or IV. The dams were permanently identified by Monel ear tags; tail tattoos were used to assign unique identification to the immature rats on PPD 15 (groups I, II, IV) or PPD 16 (group III). A formal randomization procedure was not used because there were only 3 litters (2 dosage groups and a positive control group) for comparison with a vehicle control group. Mean pup weights on the first day of dosage (PPD 19) were 37.5, 37.3, 38.0, and 39.8 g in groups I through IV, respectively.
All cage sizes and housing conditions were in compliance with the Guide for the Care and Use of Laboratory Animals. 25 The study room was independently supplied with at least 10 changes per hour of 100% fresh air passed through 99.97% HEPA filters. Environmental controls were set to maintain temperatures at 64°F to 79°F with relative humidity of 30% to 70%; a 12:12-hour light–dark lighting cycle was used. Certified Rodent Diet #5002 (PMI Nutrition International, St Louis, Missouri) and reverse osmosis deionized water (with chlorine added to the processed water as a bacteriostat) were provided ad libitum to the rats. A sample of the feed used in the study was analyzed for soy isoflavones (ie, phytoestrogen content; Microbac Laboratories, Inc, Wilson, North Carolina). Bed-o’cobs bedding (The Anderson Industrial Products Group, Maumee, Ohio) was used as the nesting material.
Methods
As illustrated in Table 2, a 20% solution of 100% lavender oil was used as the high dosage, and a 4% solution was used as the low dosage. On a mg/kg basis, these dosages represent greater than 30 000 and 6000 times the conservative calculation of the maximum daily human skin exposure from the use of multiple cosmetic products containing lavender oil which is calculated to be 0.003 mg/kg for a 60 kg high-end user of these products (Table 1). These dosages represent 1 000 000 to 5000 times the estimated maximum exposure from hair and skin care products used by prepubescent boys, which was calculated to be between 0.004 and 0.0001 mg/kg per day (Table 3). The dosage of the positive control article was selected on the basis of previous studies conducted by the testing facility.
Dosing Regimen Administered to Immature Female Rats. a
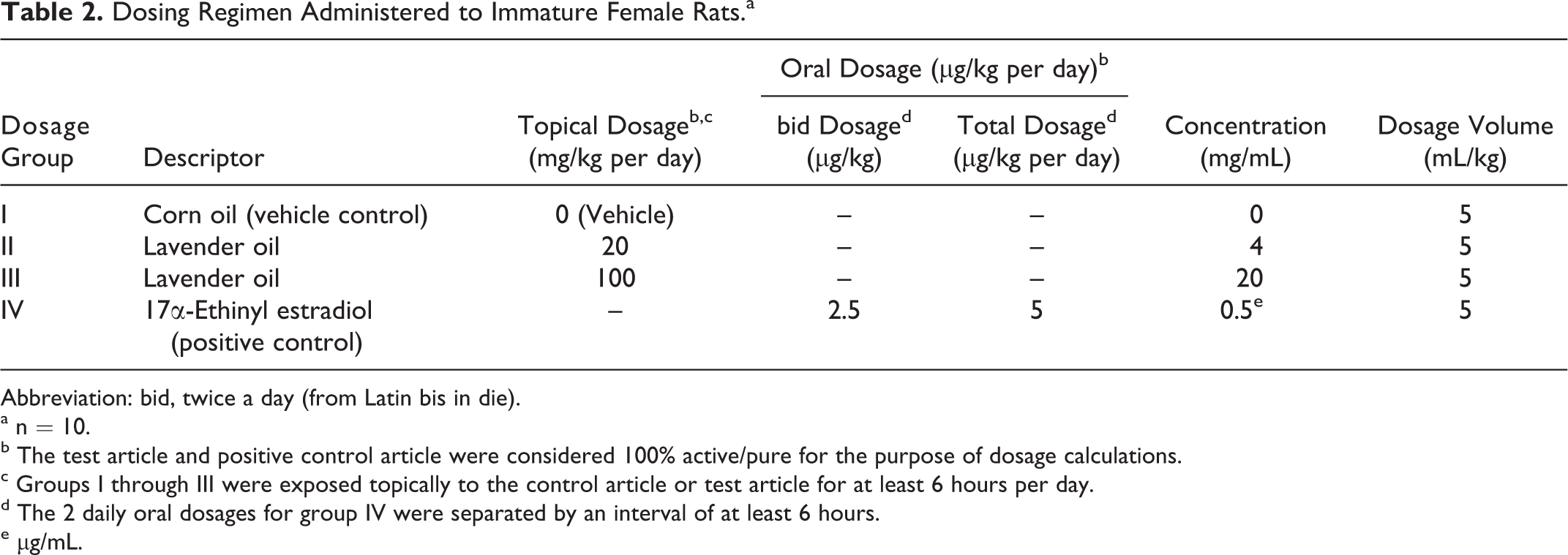
Abbreviation: bid, twice a day (from Latin bis in die).
a n = 10.
b The test article and positive control article were considered 100% active/pure for the purpose of dosage calculations.
c Groups I through III were exposed topically to the control article or test article for at least 6 hours per day.
d The 2 daily oral dosages for group IV were separated by an interval of at least 6 hours.
e μg/mL.
Estimated Skin Exposure From the Use of Cosmetic Products Containing Lavender Oil in 3 Patients.
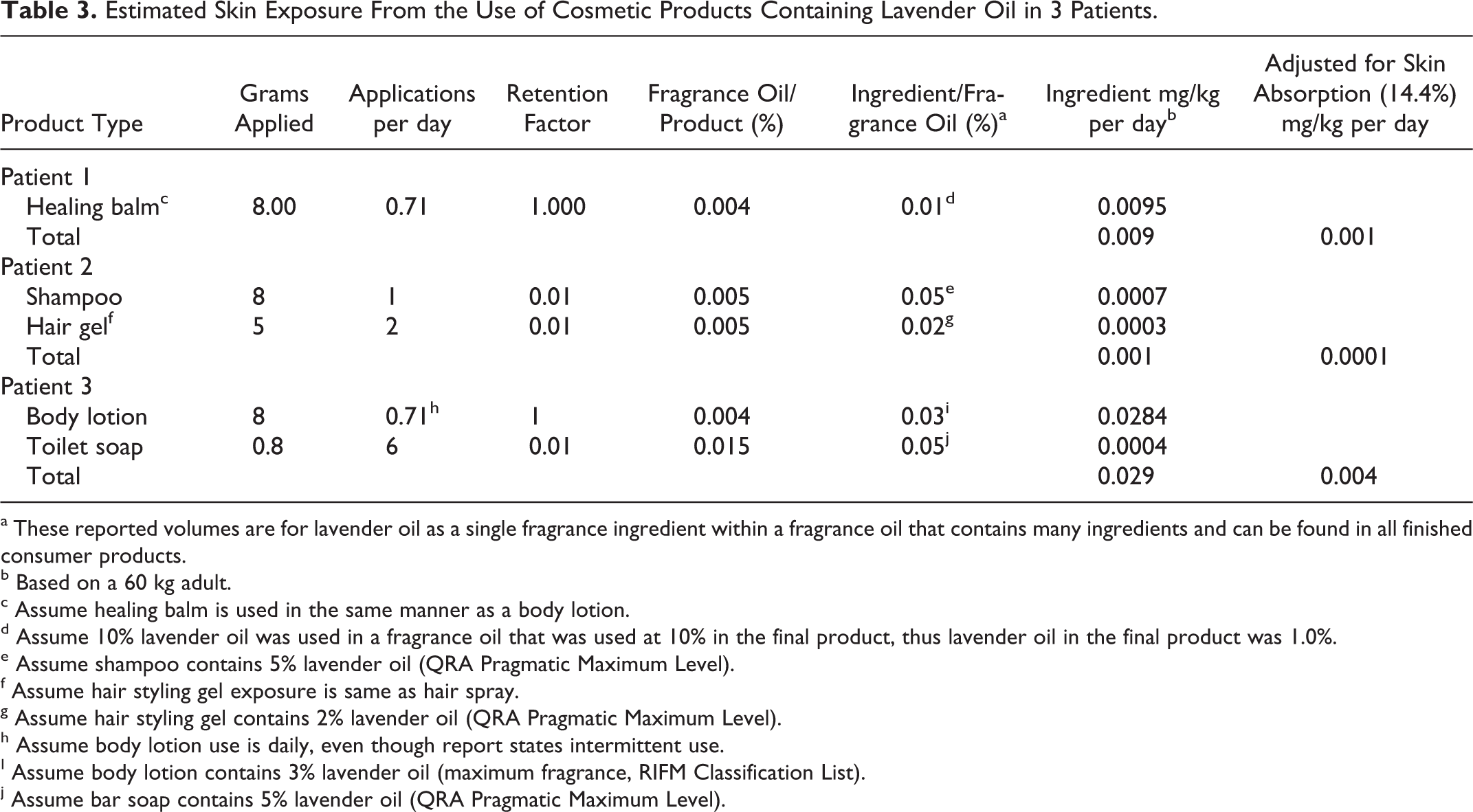
a These reported volumes are for lavender oil as a single fragrance ingredient within a fragrance oil that contains many ingredients and can be found in all finished consumer products.
b Based on a 60 kg adult.
c Assume healing balm is used in the same manner as a body lotion.
d Assume 10% lavender oil was used in a fragrance oil that was used at 10% in the final product, thus lavender oil in the final product was 1.0%.
e Assume shampoo contains 5% lavender oil (QRA Pragmatic Maximum Level).
f Assume hair styling gel exposure is same as hair spray.
g Assume hair styling gel contains 2% lavender oil (QRA Pragmatic Maximum Level).
h Assume body lotion use is daily, even though report states intermittent use.
I Assume body lotion contains 3% lavender oil (maximum fragrance, RIFM Classification List).
j Assume bar soap contains 5% lavender oil (QRA Pragmatic Maximum Level).
During exposure periods, the dams were placed in individual housing and the immature female rats were housed by dosage group in separate nesting boxes (5 rats/box). The immature female rats assigned to groups I, II, or III were percutaneously administered either the vehicle control article or 1 of the 2 dosage levels of the test article once daily on 3 consecutive days: PPD 19, 20, and 21 (PPD 1 = day of birth). On each day of treatment, the required dosage volume was inserted into a Hilltop Chamber (25 mm diameter; absorbent material removed; Hill Top Research, Cincinnati, OH), which was placed on the shaved back of the immature female rat and secured with micropore tape and Vetrap adhesive bandage (3M, St. Paul, MN) to prevent oral ingestion of the test or control article, to minimize loss of either material, and to prevent grooming among the pups. After at least 6 hours of daily exposure, the wrapping and Hilltop Chamber were removed, residual vehicle control article or lavender essential oil was gently removed by wiping the exposure area 5 times with fresh gauze pads moistened with 0.9% saline, and the rats were reunited with the dam in the home cage. All immature rats had been acclimated to the flexible adhesive bandage during the predose acclimation period.
Immature female rats assigned to group IV were given the positive control article orally via gavage twice daily (2 daily dosages separated by an interval of at least 6 hours), on PPDs 19, 20, and 21. All dosage volumes were adjusted daily for body weight changes and applied or administered at approximately the same time each day.
Immature female rats were observed daily for clinical signs during the predosage period and before and immediately after removal of the Hilltop Chambers with vehicle control or test articles, or shortly after each gavage administration of the positive control article. Individual body weights were recorded on PPDs 14 and 16 through 22; maternal body weights were recorded on PPDs 15, 19, and 22. Approximately 24 hours after the third day of treatment (PPD 22), the 4 dams and their cross-fostered immature female rats were euthanized by carbon dioxide asphyxiation. The dams were discarded without further evaluation. The vagina from each of the immature female rats was examined in situ for patency and the uterus and ovaries were removed as a unit. Any adipose tissue was removed carefully from the uterus, and the ovaries were then removed from the uterine horns and the weights of the paired ovaries and the wet as well as the blotted (luminal fluid removed) uterus were recorded to the nearest 0.001 g. The uterus, along with the ovaries and vagina, were then retained in neutral buffered 10% formalin for possible histological evaluation.
Data generated during the course of this study were recorded either by hand or by using the Argus Automated Data Collection and Management System and the Vivarium Temperature and Relative Humidity Monitoring System. All data were tabulated, summarized, and/or statistically analyzed using the above systems and Quattro Pro 8 and/or The SAS System (version 6.12). Clinical observation and other proportion data were analyzed using the variance test for homogeneity of the binomial distribution. 26 Continuous data were analyzed using Bartlett test of homogeneity of variances 27 and the analysis of variance, 28 when appropriate. Dunnett test 29 was used to identify the statistical significance of differences among individual groups. If the analysis of variance was not appropriate, the Kruskal-Wallis test 30 or Dunn method of multiple comparisons 31 was used to identify the statistical significance of differences among the individual groups. If there were greater than 75% ties, Fisher exact test 32 was used to analyze the data.
Results
Analyzed samples of group II and III first- and last-dosage formulations for lavender oil concentration were found to be within the acceptable limits (±15% of nominal concentrations). Analysis of the certified feed sample revealed the expected soy isoflavone (phytoestrogen) content of 406 μg/g of feed.
No unscheduled deaths occurred during the conduct of the study. All immature female rats, and their respective dams, survived to scheduled termination on PPD 22. No skin reactions or adverse clinical signs were observed as a result of the percutaneous exposure to corn oil or the 20 or 100 mg/kg per day dosages of lavender oil. Oral administration of the positive control article (17α-ethinyl estradiol) also did not produce any adverse clinical signs. None of the immature female rats had a patent vagina at necropsy on PPD 22.
Both dosages of lavender oil significantly reduced body weight gains on PPD 21 through 22 (after the third treatment), when compared to the vehicle control group value (Table 4). Mean body weight gain for the entire dosage period (PPDs 19-22) was unaffected in the 20 mg/kg per day dosage group (99.0% of the control group value), but was significantly reduced to 81.6% of the vehicle control group value in the 100 mg/kg per day dosage group. However, at 20 and 100 mg/kg per day, mean body weights on PPDs 19 through 22, as well as on PPD 22, were comparable to the control values (99.4% and 97.1% of the vehicle control value, respectively). In contrast, the estrogenic positive control article produced significant increases in body weight gains on the first 2 days of dosage, resulting in a significant increase (119.4%) in body weight gain for the entire dosage period (PPDs 19-22), compared to the vehicle control group values. Mean body weights were also significantly increased on PPDs 21 and 22 (109.0% of the vehicle control group value).
Effect of Lavender Oil and 17α-Ethinyl Estradiol on Immature Female Rat Body Weight and Body Weight Changes. a
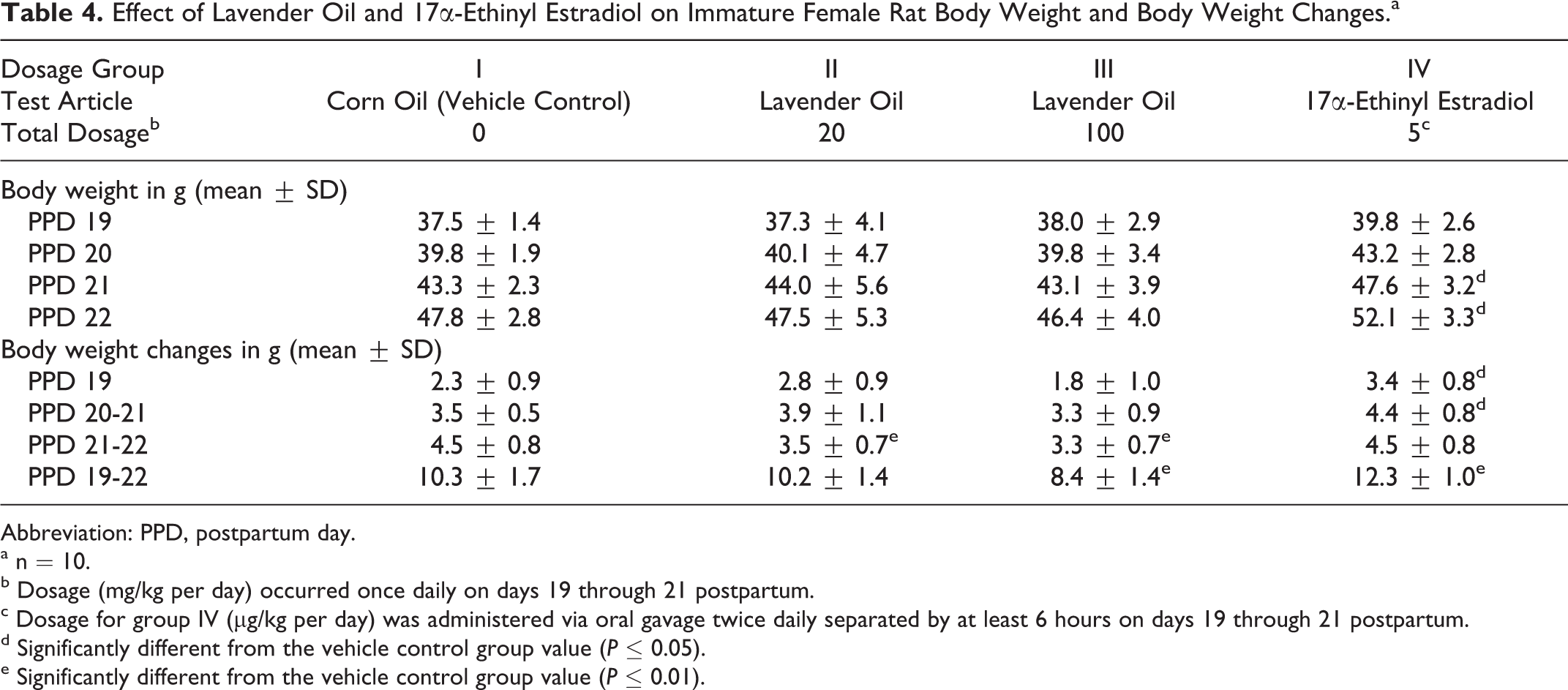
Abbreviation: PPD, postpartum day.
a n = 10.
b Dosage (mg/kg per day) occurred once daily on days 19 through 21 postpartum.
c Dosage for group IV (μg/kg per day) was administered via oral gavage twice daily separated by at least 6 hours on days 19 through 21 postpartum.
d Significantly different from the vehicle control group value (P ≤ 0.05).
e Significantly different from the vehicle control group value (P ≤ 0.01).
As shown in Table 5, mean terminal body weights, ovarian weights, wet uterine, and blotted uterine weights did not differ significantly between the vehicle control group and the groups administered 20 or 100 mg/kg per day of lavender oil. On the other hand, the estrogenic positive control article produced significant increases in mean terminal body weights and uterine weights compared to the vehicle control group values. These findings demonstrated (1) that the uterotrophic assay demonstrated appropriate response to estrogen stimulation (17α-ethinyl estradiol positive control) and (2) that topical dosages of 6000 or 30 000 times the maximum human daily exposure to lavender oil did not elicit any estrogenic stimulatory effects in this in vivo uterotrophic model.
Effect of Lavender Oil and 17α-Ethinyl Estradiol on Immature Female Rat Terminal Body Weights, Organ Weights, and Ratios (%) of Organ Weight to Terminal Body Weight. a
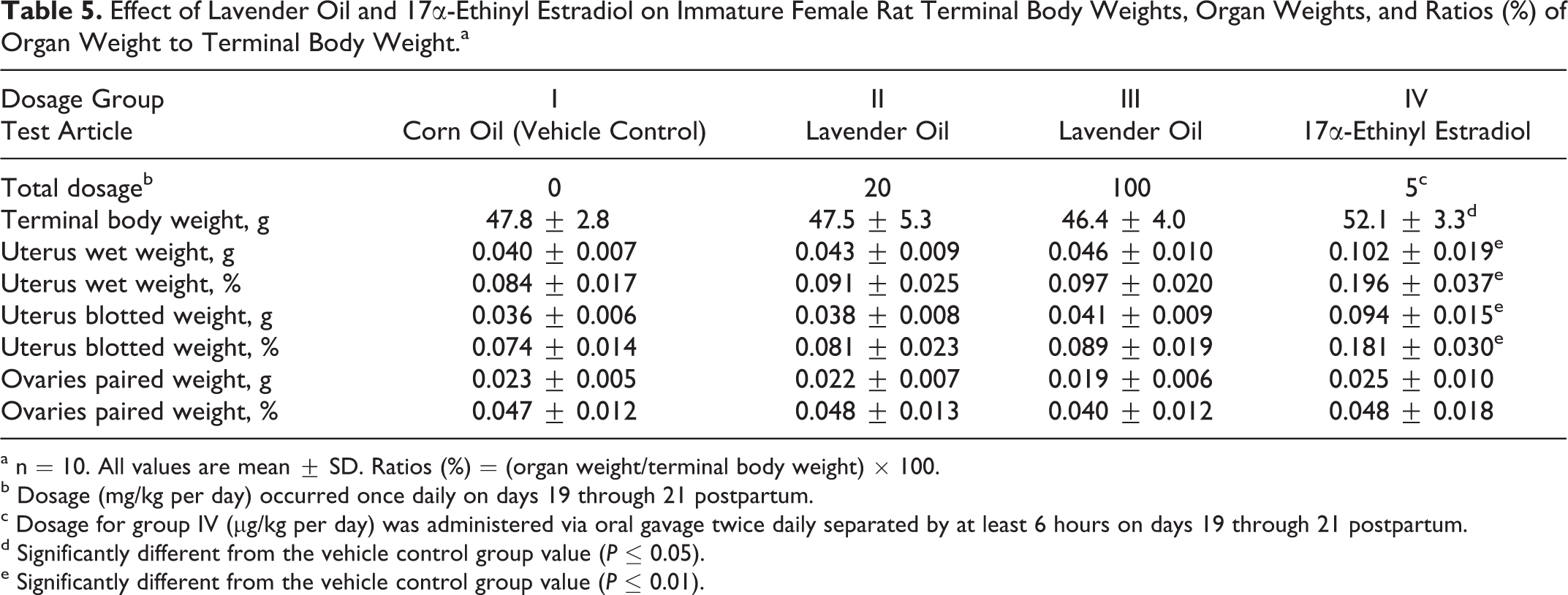
a n = 10. All values are mean ± SD. Ratios (%) = (organ weight/terminal body weight) × 100.
b Dosage (mg/kg per day) occurred once daily on days 19 through 21 postpartum.
c Dosage for group IV (μg/kg per day) was administered via oral gavage twice daily separated by at least 6 hours on days 19 through 21 postpartum.
d Significantly different from the vehicle control group value (P ≤ 0.05).
e Significantly different from the vehicle control group value (P ≤ 0.01).
Discussion
Lavender oil, a natural product from the lavender plant (L officinalis Chaix), was assayed for estrogenic activity in the in vivo uterotrophic assay, which is based on an increase in uterine weight (uterotrophic response) upon exposure to an estrogenic stimulus. The bioassay relies for its sensitivity on an animal test system in which the hypothalamic–pituitary–ovarian axis is not yet functional. Immature females after weaning and prior to puberty meet this requirement. For estrogen agonists, the mean uterine weight of the treated animal relative to the vehicle group is assessed for a statistically significant increase which indicates a positive response to this bioassay. 22
Results from the present assay indicate that the topical dosages of lavender oil equivalent to 6000 or 30 000 times the maximum daily human exposure did not elicit a positive response in this assay. Furthermore, significant decreases in mean body weight gains in both lavender-treated groups during the 3-day dosing period are in contrast to the significant gains in the positive controls and likely indicate systemic toxicity.
The negative results obtained from the present uterotrophic assay do not confirm the positive results from in vitro breast cancer cell line assays for estrogenicity and antiandrogenicity reported by Henley et al. 1 This in itself is not unusual, because there may be a large difference between the amount and specificity of a chemical that gets to a hormone receptor on a cell in a culture dish and the amount that might get to a cell’s hormone receptor in an intact living animal or human being.
Linalool and linalyl acetate are the major components of lavender oil 19 and the general toxicological characteristics of both have been previously reviewed. 33 –40 As part of its characterization of essential oils and groups of structurally related materials, the fragrance industry has conducted a developmental toxicity study with linalool in rats. 41 Linalool is one of the major components of lavender oil, accounting for as much as 57.5% of the essential oil depending on the origin. 19 No adverse effects or morphological changes (such as altered anogenital distance or genital aberrations) were observed in the 21-day old fetuses born of dams exposed to oral maternal doses of up to 1000 mg/kg per day during gestation; dams also remained unaffected except for decreased feed consumption and body weight gains at the 1000 mg/kg per day level. The developmental no observed adverse effect level (NOAEL) for linalool is ≥1000 mg/kg per day and the maternal NOAEL is 500 mg/kg per day. 41 Additionally, linalool did not show estrogenic activity in vitro in the estrogen-inducible yeast screen. 42,43 These findings support the results of the present uterotrophic study with lavender oil.
In summary, results from the present uterotrophic bioassay in rats show that percutaneous dosages of lavender oil equivalent to greater than 6000 and 30 000 times the human maximal daily exposure of 0.003 mg/kg per day (Table 1) and greater than 5000 to 1 000 000 times the estimated exposure from hair and skin care products that may be used by prepubertal boys (Table 3) did not induce a statistically significant increase in mean uterine weight. It is concluded that at the dosages tested, lavender oil is not an estrogenic agonist.
Footnotes
Authors’ Note
Parts of this work were presented at the 48th Annual Meeting of the Society of Toxicology, 2009, Baltimore, MD, USA. The study was conducted at Charles River Laboratories Preclinical Services, Horsham, PA, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Research Institute for Fragrance Materials, Inc, Woodcliff Lake, NJ, USA.
