Abstract
Alpha-iso-methylionone (AIM), a fragrance ingredient, was evaluated for systemic toxicity in rats. Male and female Sprague Dawley rats were administered 0, 5, 30, or 500 mg/kg/d AIM via gavage for 90 days. Statistically significant changes in blood chemistry parameters (reduced aspartate aminotransferase [AST], and increased cholesterol, creatinine, and total protein) were observed in both sexes at 500 mg/kg/d. There were statistically significant increases in liver and kidney weights in both sexes and in spleen weights in males at 500 mg/kg/d. Adaptive hepatocyte enlargement was observed in both sexes at 500 mg/kg/d. Globular accumulations of eosinophilic material were observed in the renal tubular epithelium in males at ≥30 mg/kg/d. Thyroid and bone marrow histopathological changes were observed in males at 500 mg/kg/d. The no-observed-effect level was 5 mg/kg/d for males and 30 mg/kg/d for females. Based on histopathological changes in the kidney in males, the no-observed-adverse-effect level was 30 mg/kg/d.
Introduction
Alpha-iso-methylionone (AIM, Figure 1), also known as 3-butene-2-one, 3-methyl-4-(2,6,6-trimethyl-2-cyclohexen-1-yl), is a clear, pale yellow oily liquid with an extremely versatile odor, ranging from sweet floral to tobacco-like or woody. 1 Alpha-iso-methylionone is found in the fragrances used in a variety of cosmetics and toiletry products as well as in noncosmetic products such as household cleaners and detergents. Worldwide, its use in fragrances is greater than 100 metric tons per annum. 2 Due to its presence in a variety of consumer products, humans have the potential to be exposed to low but continuous levels of AIM. Potential consumer exposure to fragrance materials occurs through the dermal and inhalation routes of exposure. Worst-case scenario calculations indicate that depositions on the surface of the skin following the use of cosmetics represents the major route of exposure to fragrance ingredients when conservative estimates for evaporation, rinsing, and other forms or product removal are employed. 3 Therefore, the dermal route is the major route in assessing the safety of AIM.
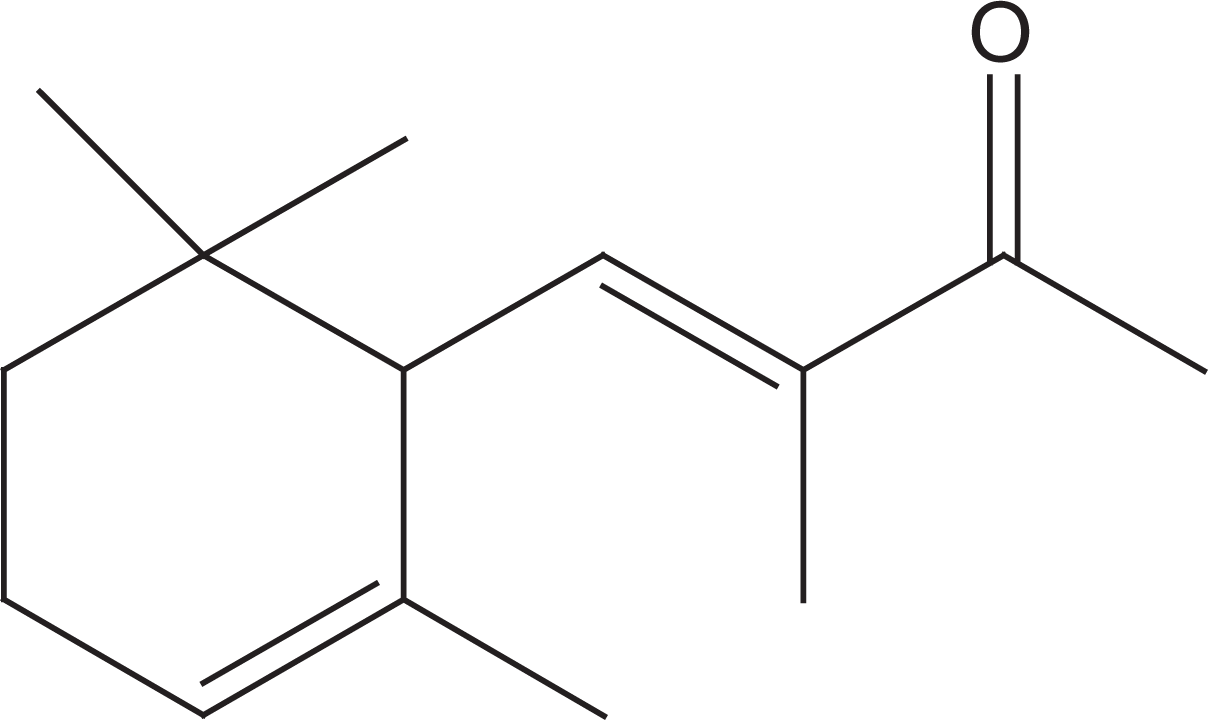
Structure of alpha-iso-methylionone (AIM).
The systemic exposure (total human skin exposure) for fragrance materials from cosmetic products is determined from the quantities used, the frequency of use, the concentration of the fragrance material in the products, and skin permeation. 4 The total maximum exposure to AIM was calculated from 10 types of the most frequently used personal care and cosmetic products containing AIM using these factors. These 10 product categories included antiperspirant, bath products, body lotion, eau de toilette, face cream, fragrance cream, hair spray, shampoo, shower gel, and toilet soap. The 97.5 percentile use level in fragrance formulae for use in cosmetics has been reported to be 13% according to data provided by industry. 5 This upper 97.5 percentile concentration was then used for all 10 consumer products. These concentrations were multiplied by the amount of product applied, the number of applications per day for each product type, and a “retention factor” to account for the length of time a product may remain on the skin and/or the likelihood of the fragrance ingredient being removed by washing. The resultant calculation represents the total consumer exposure (mg/kg/d). 3,4 Thus, in a high-end user of multiple AIM-containing cosmetic products, the maximal daily systemic exposure would be 0.3 mg/kg/d AIM. 5 In view of all of the above assumptions, the total calculated consumer exposure is conservative; it is unlikely that a consumer will consistently use a number of different consumer products which are all perfumed with the upper 97.5 percentile level of the fragrance ingredient from a fine fragrance type product. In the absence of skin absorption data, it is assumed that 100% of the applied dose of AIM is absorbed through the skin.
The general toxicological characteristics of AIM have been previously reviewed. 6,7 To summarize, the mammalian oral and dermal LD50 values were determined to be >5 g/kg. 8 –10 Alpha-iso-methylionone was found to be negative in an Ames assay. 11 While there are no further genotoxicity studies on AIM, several studies have been conducted on related isomers of methyl ionone, concluding that methyl ionone does not possess significant genotoxic potential. 12 –14 A developmental toxicity study conducted at 0, 3, 10, or 30 mg/kg/d AIM in rats found maternal and developmental no-observed-adverse-effect levels (NOAELs) of greater than or equal to 30 mg/kg/d. 15
In a dermal 90-day toxicity study in rats, severe AIM-related dermal irritant effects were observed at all dose levels of 50, 170, 580 and 2000 mg/kg/d. 16 A second dermal 90-day toxicity study was conducted in rats with 1 mL/kg of a 0% or 1% solution of AIM in phenethyl alcohol in which the back was divided up into a 7-area grid so a separate area was treated each day in the week. The no-observed-effect Level (NOEL) was determined to be 1%. 17 The existing oral 90-day toxicity studies on AIM provide insufficient details. Rats fed AIM in the diet at 3.7 mg/kg/d for 90 days had no adverse effects on body weight gain, feed consumption, clinical pathology parameters, or on the tissues (including bone marrow, liver, kidney, and thyroid) examined histologically. 18 Daily gastric intubation of 3.4 mg/kg/d AIM for 12 weeks in rats was reported to have no effect; no further details were provided. 19
Due to the high volume of use of AIM 2 and lack of an adequate subchronic toxicity study available in scientific literature, the Research Institute for Fragrance Materials (RIFM) contracted with SafePharm Laboratories Limited for a 90-day toxicity study to evaluate the continued safe use of AIM in fragrances. The study complied with the requirements of the Organization for Economic Cooperation and Development Guidelines for Testing of Chemicals No. 408. 20 The purpose of the study was to determine whether exposure to AIM for 90 days could produce any systemic adverse effects in rats and to determine the NOEL in Sprague-Dawley rats. All facets of the study were conducted in compliance with the Good Laboratory Practice regulations of the United Kingdom 21 and the OECD. 22
Methods
Materials
Alpha-iso-methylionone (CAS# 127-51-5, batch 929473) was supplied by International Flavors and Fragrances, Inc (Union Beach, New Jersey). The sample was comprised of AIM (60%) and its isomers (39.9%), for a purity of 99.9%. The vehicle and control article was corn oil. Formulations were stored at 4°C in the dark and were tested for stability, homogeneity, and concentration of AIM. The formulations were found to be stable for at least 14 days and to be within acceptable concentration limits (mean concentrations found to be nominal ±3%).
Animals
Male and female Sprague-Dawley Crl:CD IGS BR strain rats were obtained from Charles River (UK) Ltd. (Margate, Kent). This study was conducted in accordance with the Safepharm policy on animal welfare and the requirements of the United Kingdom’s Animals (Scientific Procedures) Act 1986. After a 6-day acclimatization period, the rats were randomly allocated to treatment groups using a total randomization procedure and the group mean bodyweights were then determined to ensure similarity between the treatment groups. Rats were housed by sex in 2 groups of 3 and 1 group of 4. At the beginning of treatment males weighed 131 to 172 g, females weighed 122 to 155 g, and were 6 to 8 weeks old.
The single study room was supplied with at least 15 air changes per hour, and the temperature and relative humidity controls met the target values of 21°C ± 2°C and 55% ± 15%, respectively. A 12:12-hour light–dark cycle was followed with light beginning at 0600 hours (0700 hours after daylight savings time ended). The rats were allowed free access to certified Rodent 5LF2 Diet (BCM IPS Ltd, London, UK) and local water. Wooden chew blocks (B&K Universal Ltd, Hull, UK) and cardboard fun tunnels (Datesand Ltd, Cheshire, UK) were provided for environmental enrichment.
Dose Range Finding
A 14-day range-finding study was conducted to determine the maximum tolerated dose level following repeated oral administration and to provide information for selection of dose levels for use in the definitive study. Doses of 0 or 1000 mg/kg/d AIM in corn oil were administered daily to 3 rats/sex/dose for 14 days via gavage. There were no unscheduled deaths. Rats of both sexes treated with 1000 mg/kg/d AIM showed increased salivation following dosing. Body weights were slightly reduced for males treated with 1000 mg/kg/d AIM on day 4 only. At necropsy, 2 males and 1 female treated with 1000 mg/kg/d showed pale kidneys. Based on these data, doses of 0, 5, 30, and 500 mg/kg/d AIM were selected for the definitive 90-day toxicity study.
Parameters Investigated
Alpha-iso-methylionone at doses of 0, 5, 30 or 500 mg/kg/d in a corn oil vehicle was administered orally by gavage for 90 consecutive days to groups of 10 rats/sex/dose. The dose volume was 4 mL/kg/d, adjusted for body weight which was measured weekly.
All rats were observed twice daily for overt signs of toxicity, ill health, or behavioral change. Observations were made immediately before dosing and 5 hours after dosing during the work week, or immediately before dosing and 1 hour after dosing on weekends. Bodyweights were recorded on day 1 and at weekly intervals for the remainder of the study. Food consumption was recorded for each cage group at weekly intervals. Water consumption was observed daily for each cage group by visual inspection for overt changes.
Prior to the start of treatment and at weekly intervals throughout the study, rats were individually observed for signs of functional/behavioral toxicity using a purpose-built open arena. The parameters the rats were observed for included gait, tremors, twitches, convulsions, bizarre/abnormal/stereotypic behavior, salivation, piloerection, exophthalmia, lachrymation, hyperthermia/hypothermia, skin color, respiration, palpebral closure, urination, defecation, transfer arousal, and tail elevation. During week 12, functional performance tests including motor activity, grip strength, and sensory reactivity were performed on all rats. Motor activity, the percentage of time active and mobile for the entire period and for the final 20% of the period, was measured using a 44 infrared beam automated activity monitor. Each rat was evaluated for 1 hour. An automated grip strength meter was used to evaluate forelimb and hindlimb grip strength for each rat over 3 consecutive trials. Sensory reactivity to auditory, visual, and proprioceptive stimuli was assessed for the grasp response, vocalization, toe pinch, tail pinch, finger approach, touch escape, pupil reflex, startle reflex, and blink reflex.
Prior to the start of treatment and during week 12, the eyes of all control and 500 mg/kg/d AIM treated rats were examined for observations of the anterior structures of the eye, papillary, and corneal blink reflex, and following dilation, the internal structure of the eye.
On day 90, blood samples from all rats were taken from the lateral tail vein for hematological and blood chemical analyses. Hematological parameters measured included hemoglobin, erythrocyte count, hematocrit, erythrocyte indices (mean corpuscular hemoglobin, mean corpuscular volume, and mean corpuscular hemoglobin concentration), total leukocyte count, differential leukocyte count (neutrophils, lymphocytes, monocytes, eosinophils, and basophils), platelet count, reticulocyte count, prothrombin time, and activated partial thromboplastin time. Blood chemistry parameters measured included urea, glucose, total protein, albumin, albumin/globulin ratio, sodium, potassium, chloride, calcium, inorganic phosphorous, aspartate aminotransferase (AST), alanine aminotransferase, alkaline phosphatase, creatinine, total cholesterol, and total bilirubin.
On day 91, rats were euthanized by intravenous overdose of sodium pentobarbitone followed by exsanguination. A full external and internal examination was performed. The following organs were excised, dissected of fat, and weighed: adrenals, brain, epididymides, heart, kidneys, liver, ovaries, spleen, testes, thymus, and uterus. Samples of the following tissues were removed and fixed in buffered 10% formalin or Davidson's fluid: adrenals, thoracic aorta, bone and bone marrow (femur including stifle joint and sternum), brain (cerebrum, cerebellum, and pons), cecum, colon, duodenum, epididymides, eyes, gross lesions, heart, ileum (including Peyer patches), jejunum, kidneys, liver, lungs (with bronchi), lymph nodes (cervical and mesenteric), mammary glands, muscle (skeletal), esophagus, ovaries, pancreas, pituitary, prostate, rectum, salivary glands (submaxillary), sciatic nerve, seminal vesicles, skin (hind limb), spinal cord (cervical, midthoracic, and lumbar), spleen, stomach, testes, thymus, thyroid/parathyroid, tongue, trachea, urinary bladder, and uterus. All tissues from control and 500 mg/kg/d AIM dose groups were examined microscopically. Changes were observed in the liver, kidneys, thyroid, and bone marrow, thus these tissues were subsequently examined microscopically in the 5 and 30 mg/kg/d AIM dose groups.
Statistical Analysis
Quantitative data were analyzed by the Provantis Tables and Statistic Module. The homogeneity of means was assessed using analysis of variance (ANOVA) or analysis of covariance (ANCOVA) and Bartlett test. Transformed data were assessed using the Williams test (parametric) or the Shirley test (nonparametric). If no dose response was found, but the data showed nonhomogeneity of means, analysis by a stepwise Dunnett (parametric) or Steel (nonparametric) test was conducted to determine significance. If needed, pairwise tests were performed using the Student t test (parametric) or the Mann-Whitney U test (nonparametric). Histopathology data were analyzed by the chi-square analysis for differences in lesions with a frequency of 1 or greater and the Kruskal-Wallis 1-way nonparametric analysis if variance for the comparison of severity grades for the more frequently observed graded conditions.
Results
There were no unscheduled deaths. Instances of increased salivation and noisy respiration were observed in treated rats as well as hunched posture and tiptoe gait in 500 mg/kg/d rats. Such changes are often observed at the testing facility following oral administration of an unpalatable or slightly irritant test article formulation and are not considered to be indicative of systemic toxicity. One female each from the 500 and 30 mg/kg/d dose groups showed isolated episodes of tail elevation with the latter female also showing ataxia; due to the sporadic nature of the findings and the absence of any supporting behavioral assessment to suggest neurotoxicity, they were considered to be of no toxicological importance. Isolated instances of generalized fur loss, scab formation, and generalized red/brown stained fur were observed in a number of control and treated animals and were considered to be incidental and unrelated to treatment. There were no treatment-related effects observed on the behavioral parameters, functional performance parameters, or sensory reactivity assessments.
There were no adverse effects on body weight development. Female rats treated with 500 mg/kg/d AIM had statistically significantly reduced body weight gains during week 7 and statistically significantly increased body weight gains during weeks 9 and 13. At termination, body weights were 99% of the control group, thus the changes were determined to reflect normal biological variation and to be of no toxicological significance. There were no adverse effects on food consumption, food efficiency, or water consumption during the study period.
There were no treatment-related changes in the ophthalmoscopic examination or the hematological parameters measured. Blood chemistry evaluation revealed a statistically significant increase in plasma creatinine, total protein, and cholesterol in rats treated with 500 mg/kg/d when compared to controls (Table 1). Males from this treatment group also showed a statistically significant increase in plasma albumin. Rats of both sexes treated with 500 mg/kg/d showed a statistically significant reduction in AST, and females from this group also showed a statistically significant reduction in alkaline phosphatase. These changes were considered to be of no toxicological importance. Males treated with 500 mg/kg/d AIM showed a statistically significant reduction in plasma chloride concentration; however, all individual values were within the normal range for rats of the strain and age used, and the intergroup difference was considered not to be toxicologically significant. Females from all treatment groups showed a statistically significant reduction in plasma bilirubin. The majority of the individual bilirubin values were within the normal range for rats of the strain and age used, and in the absence of a dose-related response the intergroup differences were considered to be of no toxicological importance.
Summary of Blood Chemical Changes Following Oral Administration of AIM.a
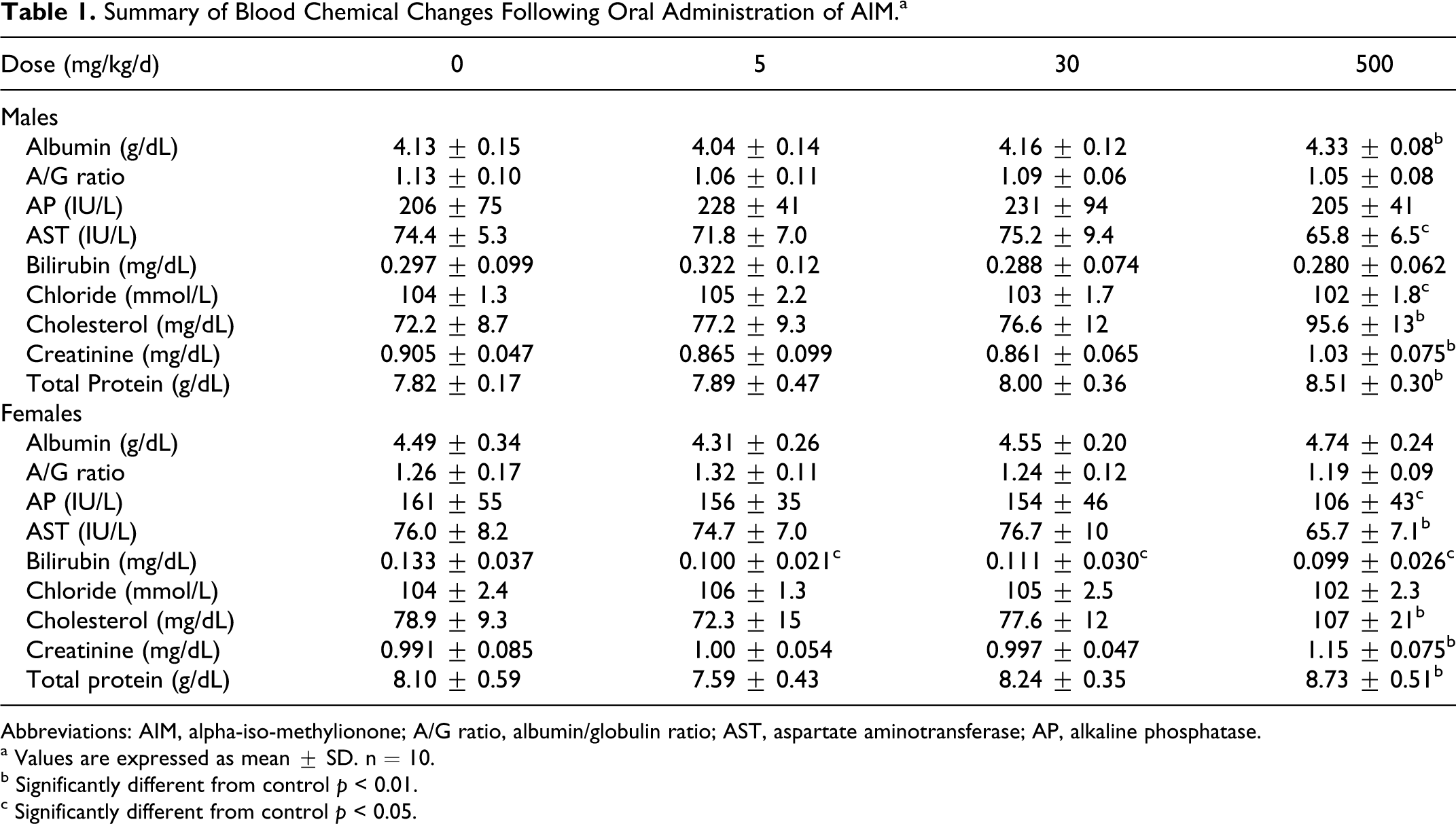
Abbreviations: AIM, alpha-iso-methylionone; A/G ratio, albumin/globulin ratio; AST, aspartate aminotransferase; AP, alkaline phosphatase.
a Values are expressed as mean ± SD. n = 10.
b Significantly different from control p < 0.01.
c Significantly different from control p < 0.05.
No treatment-related macroscopic abnormalities were detected at necropsy. Rats treated with 500 mg/kg/d showed a statistically significant increase in liver and kidney weights, both absolute and relative to terminal body weight (Table 2). Males from this group also showed statistically significant increases in absolute and relative spleen weight. Rats treated with 30 mg/kg/d showed a statistically significant reduction in absolute and relative heart weight; however, all individual values were within the normal range for rats of the strain and age used and in the absence of a dose-related response the differences were considered to be of no toxicological importance. Females treated with 30 mg/kg/d showed a statistically significant increase in absolute and relative liver weight. In the absence of any histological correlates the difference was considered not to be toxicologically significant. Males treated with 5 mg/kg/d showed a statistically significant increase in absolute and relative adrenal weight. This observation was also considered not to be toxicologically significant in the absence of a dose-related response or any histological correlates.
Summary of Relative Organ Weight Changes Following Oral Administration of Alpha-Iso-Methylionone.a
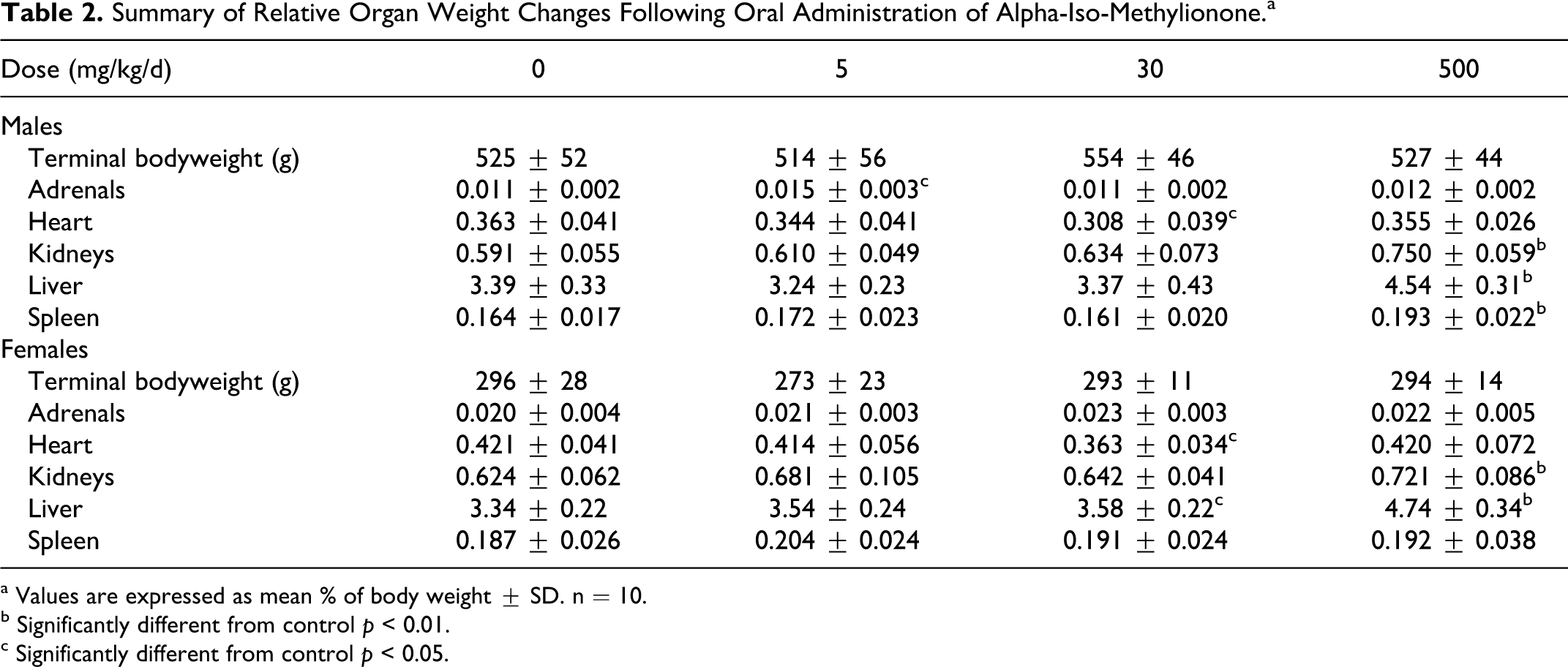
a Values are expressed as mean % of body weight ± SD. n = 10.
b Significantly different from control p < 0.01.
c Significantly different from control p < 0.05.
Histopathological examination revealed statistically significant treatment-related changes in the liver, kidney, thyroid, and bone marrow (Table 3). Liver changes observed included hepatocyte enlargement (centrilobular or generalized) in 4 of 10 males and 9 of 10 females treated with 500 mg/kg/d AIM. This observation is commonly seen following the administration of xenobiotics and is generally regarded as adaptive in nature in the absence of associated degenerative changes. A greater incidence of higher severity grades of globular accumulations of eosinophilic material was observed in the tubular epithelium in kidneys of males treated with 30 and 500 mg/kg/d. This finding may indicate the presence of hydrocarbon nephropathy, which results from the excessive accumulation of α2-microglobulin in renal proximal tubular epithelial cells. α2-Microglobulin is found only in the proximal tubular epithelium of adult male rat kidneys. 23 –25 In the thyroid, a higher incidence of follicular cell hypertrophy was observed in males treated with 500 mg/kg/d. A higher incidence of lower grades of severity of adipose infiltration of the bone marrow, indicative of marrow hyperplasia, was observed in males treated with 500 mg/kg/d. The observation of higher grades of tubular basophilia in 1 of 10 and 2 of 10 males treated with 30 and 500 mg/kg/d, respectively, was not statistically significant.
Summary of Histopathological Findings Following Oral Administration of Alpha-Iso-Methylionone.a
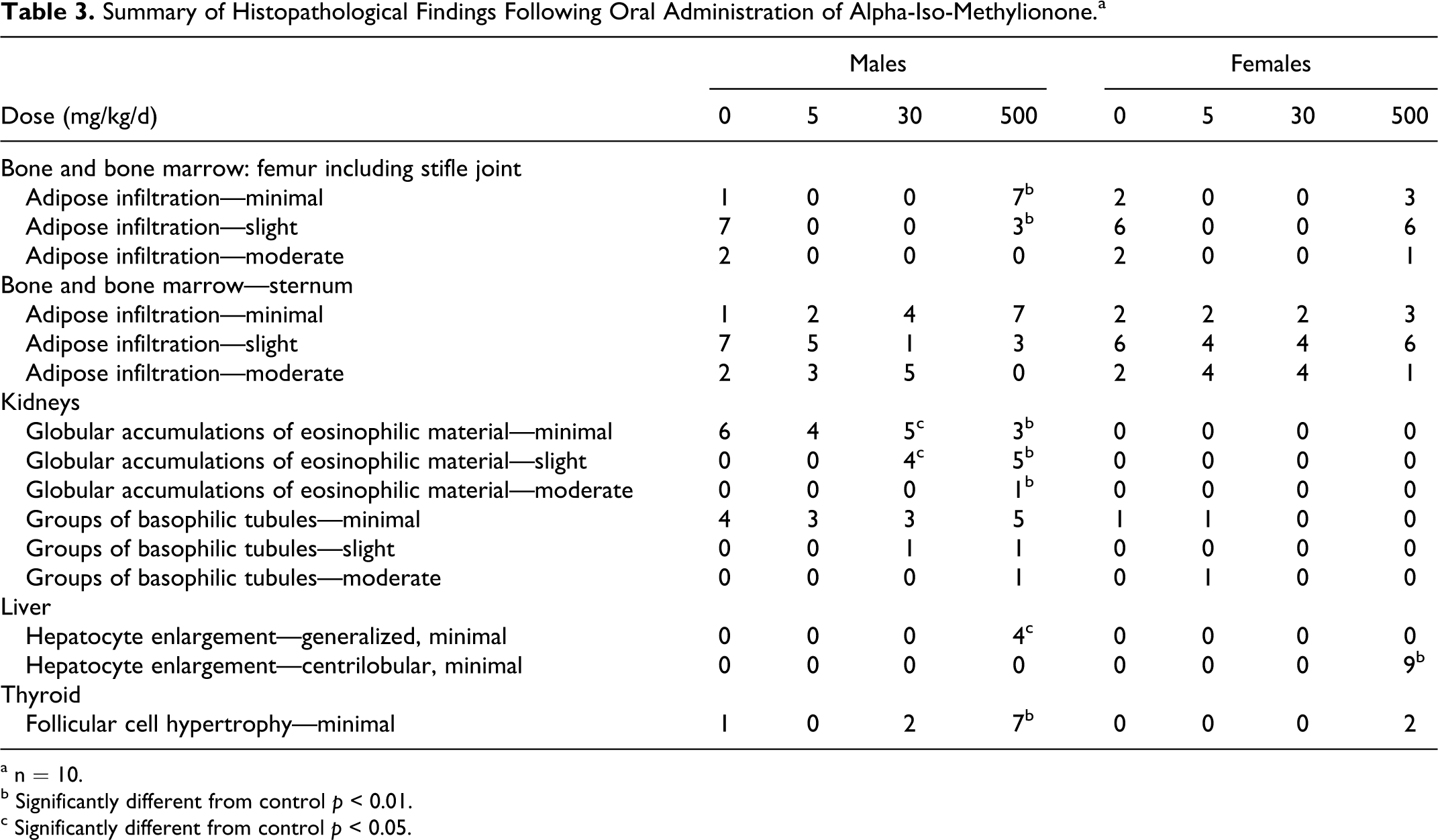
a n = 10.
b Significantly different from control p < 0.01.
c Significantly different from control p < 0.05.
Discussion
The purpose of this study was to determine the systemic NOEL following daily oral (gavage) administration of AIM to male and female rats. While exposure to AIM in fragrances is primarily topical, due to the severe dermal irritation and damage in rats administered AIM topically if given in large enough doses required to test systemic toxicity, 16 gavage administration was selected for this study. The oral administration of AIM to male and female rats for a period of 90 consecutive days at dose levels of 0, 5, 30, or 500 mg/kg/d resulted in treatment-related effects in males at 30 and 500 mg/kg/d and in females at 500 mg/kg/d.
Microscopic examinations of liver sections revealed generalized or centrilobular hepatocytes enlargement at the 500 mg/kg/d dose level in both male and female rats. Increased hepatic activity of this type and in the absence of associated inflammatory or degenerative changes is considered a normal adaptive biological response. It is usually the result of detoxification mechanisms involving hepatic enzyme induction. Organ weight data supported this finding with increased absolute and relative liver weights detected in the 500 mg/kg/d dose group. A higher incidence of minimal follicular cell hypertrophy was seen in the thyroids of 7 of 10 male rats treated with 500 mg/kg/d AIM. Thyroxine is ultimately excreted via the bile, having first been conjugated in the liver. It is conceivable that the conjugating hepatic enzymes may have been induced in response to the test material. This would stimulate the thyroid to compensate by increasing the output of thyroid-stimulating hormone and thyroxine production, which might result in the microscopic changes identified. There was an increase in absolute and relative spleen weight for male rats in the 500 mg/kg/d dose group. The absence of pathological changes identified in the spleen minimizes the toxicological significance of this finding. However, microscopic examinations of the bone marrow did reveal a higher incidence of lower grades of severity of adipose infiltration in these animals. This could suggest a subtle effect on hematopoiesis. The significance of these effects is lessened by the absence of changes in hematological parameters.
Based on the findings in this study, the NOELs are considered to be 5 mg/kg/d for males and 30 mg/kg/d for females. However, the kidney changes identified histopathologically in male rats are consistent with well-documented changes that are peculiar to the male rat in response to treatment with some hydrocarbons, and therefore, not indicative of a hazard to human health. As a result, a NOAEL for males is established as 30 mg/kg/d. A margin of safety of 100 exists between the maximum daily human exposure to AIM from dermally applied fragranced cosmetic products (0.3 mg/kg) 5 and the NOAEL of 30 mg/kg/d.
Footnotes
Authors' Note
Portions of this work were presented at the 46th Annual Meeting of the Society of Toxicology, 2007, Charlotte, North Carolina, USA. The study was conducted at SafePharm Laboratories, Shardlow, UK, and funded by the Research Institute for Fragrance Materials, Inc, Woodcliff Lake, New Jersey, USA.
Declaration of Conflicting Interests
The authors are employees of the Research Institute for Fragrance Materials, an independent research institute supported by the manufacturers of fragrances and consumer products containing fragrances.
Funding
This research was supported by the Research Institute for Fragrance Materials, an independent research institute that is funded by the manufacturers of fragrances and consumer products containing fragrances.
