Abstract
The fragrance ingredient 4-tert-butylcyclohexyl acetate (4-tBCHA) was evaluated for potential developmental toxicity in pregnant rats at oral dosages of 0, 40, 160, or 640 mg/kg per d in corn oil on gestational days 7 to 20. Increased salivation was observed at 160 and 640 mg/kg per d. The 640 mg/kg per d dosage was associated with the presence of a red perioral substance, ungroomed, sparse hair coat on the limbs, localized alopecia, reduced feed and body weight gains, or body weight losses, and mortality. Fetal body weights also were reduced at 640 mg/kg per d. This effect was associated with transient delays in fetal development, including significant increases in fetal incidences of moderate enlargement of the renal pelvis and reversible delays in ossification of the caudal vertebrae, fore and hind limb phalanges, and hind limb metatarsals. Maternal and developmental no observable adverse effect levels (NOAELs) were 160 mg/kg per d. It was concluded that 4-tBCHA is not a developmental toxicant in rats.
Introduction
The fragrance ingredient 4-tert-butylcyclohexyl acetate (4-tBCHA), CAS# 32210-23-4, is a clear, colorless-to-pale-yellow liquid with a sweet, rich, woody odor. Its molecular formula is C12H22O2 and its molecular weight is 198.31. 1 The molecular structure is presented in Figure 1.
Structure of 4-tert-butylcyclohexyl acetate.
4-tert-BCHA is a fragrance ingredient used in many fragrance compounds. It may be found in decorative cosmetics, fine fragrances, shampoos, and toilet soaps, as well as in household cleaners and detergents. Worldwide use exceeds 1000 metric tons/annum. 2 The 97.5 percentile use level in formulations for use in cosmetics has been reported to be 7.69%, which would result in a maximum daily exposure on the skin of 0.2 mg/kg for high end users. 3 Determination of systemic exposure for fragrance materials is based on the quantities of cosmetic used, the frequency of their use, the concentration of the material in the product, and assumed 100% skin permeation. 4 Cadby et al 5 have described how these data are obtained and how exposure is determined.
The safety profile of 4-tBCHA has been extensively investigated in mice, rats, guinea pigs, rabbits, and humans. A review was compiled by Bhatia et al 1 that contains a detailed summary of all available toxicology and dermatology articles that are related to this individual fragrance ingredient but has not been peer reviewed or intended as a stand-alone document. The studies, including acute oral and dermal toxicity, dermal and eye irritation, phototoxicity, mutagenicity, and developmental toxicity, all established that the use of 4-tBCHA as a fragrance ingredient was safe in humans. Results from these studies have been summarily reviewed by Belsito et al, 6 including the present developmental toxicity study. This article presents the previously reported developmental toxicity data as a stand-alone safety report.
The lack of developmental toxicity studies on cyclic acetates, of which 4-tBCHA is one, prompted the Research Institute for Fragrance Materials, Inc to conduct such a study. The study was designed to meet the requirements of the Food and Drug Administration 7 for evaluation of International Conference on Harmonization Harmonized Tripartite Guideline stages C and D of the reproductive process and to determine the maternal and developmental no observable adverse effect levels (NOAELs) in Sprague-Dawley rats. All procedures were conducted in compliance with the Good Laboratory Practice regulations of the Food and Drug Administration, 8 the Japanese Ministry of Health and Welfare, 9 and the Organization for Economic Cooperation and Development. 10
Methods
Materials
4-tert-Butylcyclohexyl acetate (lot No. 1003915) was supplied and manufactured by International Flavors and Fragrances, Inc (Union Beach, New Jersey). The fragrance material was 99.3% pure and met all the analytical standards set by International Flavors and Fragrances, Inc (Union Beach, New Jersey). Corn oil (Sigma-Aldrich Inc, St Louis, Missouri) was the vehicle and control article. Both 4-tBCHA and corn oil were stored at room temperature and protected from light. Dosage formulations were prepared weekly from bulk materials and stirred continuously during use.
Samples from each dosing suspension concentration (first and last dosage days) were analyzed for 4-tBCHA content by Charles River Laboratories Preclinical Services. The dose formulation samples were quantitated using gas chromatography flame ionization detector with 6 matrix-matched calibration standards; a process validated in Charles River Laboratories Preclinical Services. Formulation concentrations were evaluated also for stability at room temperature.
Animals
Male and female Crl:CD (Sprague-Dawley) rats (Charles River Laboratories, Inc, Raleigh, North Carolina), approximately 65 days of age on arrival, were used in the study. After a short period of acclimatization, the rats were assigned to individual housing on the basis of computer-generated random units, except during the 5-day mating period, when each pair of male and female rats was housed in the male rat’s cage. The healthy, mated female rats, weighing 221 to 257 g, were assigned to 4 dosage groups, 25 rats/group, using a computer-generated (weight-ordered) randomization procedure based on body weights recorded on the day when sperm was found in the vaginal smear or a copulatory plug was found in the vagina (gestational day 0 [GD 0]).
All cage sizes and housing conditions were in compliance with the Guide for the Care and Use of Laboratory Animals. 11 The study room was independently supplied with at least 10 changes per hour of 100% fresh air passed through 99.97% high-efficiency particulate air filters. Environmental controls were set to maintain temperatures of 64°F to 79°F with a relative humidity of 30% to 70%; a 12:12-hour light–dark lighting cycle was used. Certified Rodent Diet #5002 (PMI Nutrition International, St Louis, Missouri) and reverse osmosis deionized water (with chlorine added to the processed water as a bacteriostat) were provided ad libitum to the rats.
Dosage Determination
Dosages of 4-tBCHA were selected on the basis of a range finding study, in which 0 (corn oil), 37.5, 50, 150, or 300 mg/kg per d were gavaged daily to 8 rats/group on GDs 7 through 20. No mortality occurred at dosages as high as 300 mg/kg per d. Slight excess salivation was noted at ≥50 mg/kg per d, but these dosages were not associated with any significant reductions in maternal body weight, body weight gains, or feed consumption during the entire dosing period, although some reductions occurred during the first 2 days of dosage. No Caesarean sectioning, litter, or fetal gross effects were observed.
Based on the dose-range finding data, dosages of 0, 40, 160, or 640 mg/kg per d were administered via gavage to 4 groups of 25 presumed pregnant rats on GDs 7 to 20. The dosage volume of 10 mL/kg was adjusted daily according to individual body weights recorded directly before gavage and was administered the same time each day.
Parameters Investigated
Animals were observed for viability and examined for abnormal clinical signs twice daily: before dosage administration and approximately 1 hour later. Body weights were recorded prior to the start of the study, on GD 0, and daily during the dosage and postdosage periods. Feed consumption was recorded on GDs 0, 7, 10, 12, 15, 18, and 21. On GD 21, all rats were euthanized by the inhalation of carbon dioxide, Caesarean sectioned, and a gross necropsy of the thoracic, abdominal, and pelvic viscera was performed. Uteri of the nonpregnant rats were examined while pressed between glass plates to confirm the absence of implantation sites. The uteri from pregnant rats were excised and examined for numbers and distribution of implantations, live and dead fetuses, and resorptions. The number of corpora lutea in each ovary also was recorded.
Fetuses were removed from the uterus, weighed, and examined for gender and gross external alterations. Live fetuses then were euthanized by a 0.02 mL intraperitoneal injection of sodium pentobarbital (65 mg/mL) before undergoing further examination. Approximately half of the fetuses in each litter were fixed in Bouin's solution and examined for soft tissue alterations, using a variation of Wilson's sectioning technique. 12 The remaining fetuses in each litter were eviscerated, cleared, stained with alizarin red S, 13 and examined for skeletal alterations.
Statistical Analysis
Data generated during the course of the study were recorded either by hand or by using the Argus Automated Data Collection and Management System and the Vivarium Temperature and Relative Humidity Monitoring System. All the data were tabulated, summarized, and/or statistically analyzed using the above systems in conjunction with Microsoft Excel (Microsoft Office 97/2000/XP), Quattro Pro 8, and/or The SAS System (version 6.12).
Clinical observations and other proportional data were analyzed using the variance test for homogeneity of the binomial distribution. 14 Continuous data were analyzed using Bartlett's Test of Homogeneity of Variances 15 and the analysis of variance. 16 Dunnett's test 17 was used to identify statistical significance of individual groups. If the analysis of variance was not appropriate, the Kruskal-Wallis test 18 or Dunn's method of multiple comparisons 19 was used to identify statistical significance of the individual groups. If there were greater than 75% ties, Fisher's exact test 20 was used to analyze the data.
Results
Results of all 4-tBCHA concentration and homogeneity analyses were within ±15% of calculated concentrations and ≤5% relative standard deviation, respectively. The concentrations of the formulations used were found to be stable at room temperature for a period of 14 days.
All rats in this study survived to scheduled sacrifice on GD 21, except for 1 animal in the high-dosage group. This rat was euthanized on GD 20 because on 2 previous days it was anorexic and had lost 26 g of body weight, was observed to have decreased motor activity and ptosis, was cold to the touch, had traces of a red perioral substance along with extreme salivation, an ungroomed coat, scant liquid feces, and apparent dehydration. Necropsy revealed distention of the stomach with gas and yellow fluid, and the uterus contained a litter of 17 dead fetuses. The conceptuses were examined to the extent possible, using the same methods described for term fetuses and all appeared normal upon gross external and skeletal examination. At soft tissue examination, 3 fetuses had moderate dilation of the pelvis of the left kidney, 2 fetuses had moderate dilation of the pelvis of both kidneys, and the other 3 fetuses appeared normal for their developmental age.
Clinical signs of excessive salivation were significantly increased in rats 1 to 2 hours after gavage with 160 or 640 mg/kg per d. Effects were minimal at 160 mg/kg per d but occurred with increasing frequency and severity in the animals given high dosage. The 640 mg/kg per d dosage was also associated with the presence of a red perioral substance, ungroomed appearance, sparse hair coat on the limbs, localized alopecia, significantly reduced feed and body weight gains, or body weight losses (Table 1), and mortality (euthanasia of the rat described above).
Effect of Oral Administration of 4-tBCHA on Maternal Absolute Feed Consumption and Body Weight Gains
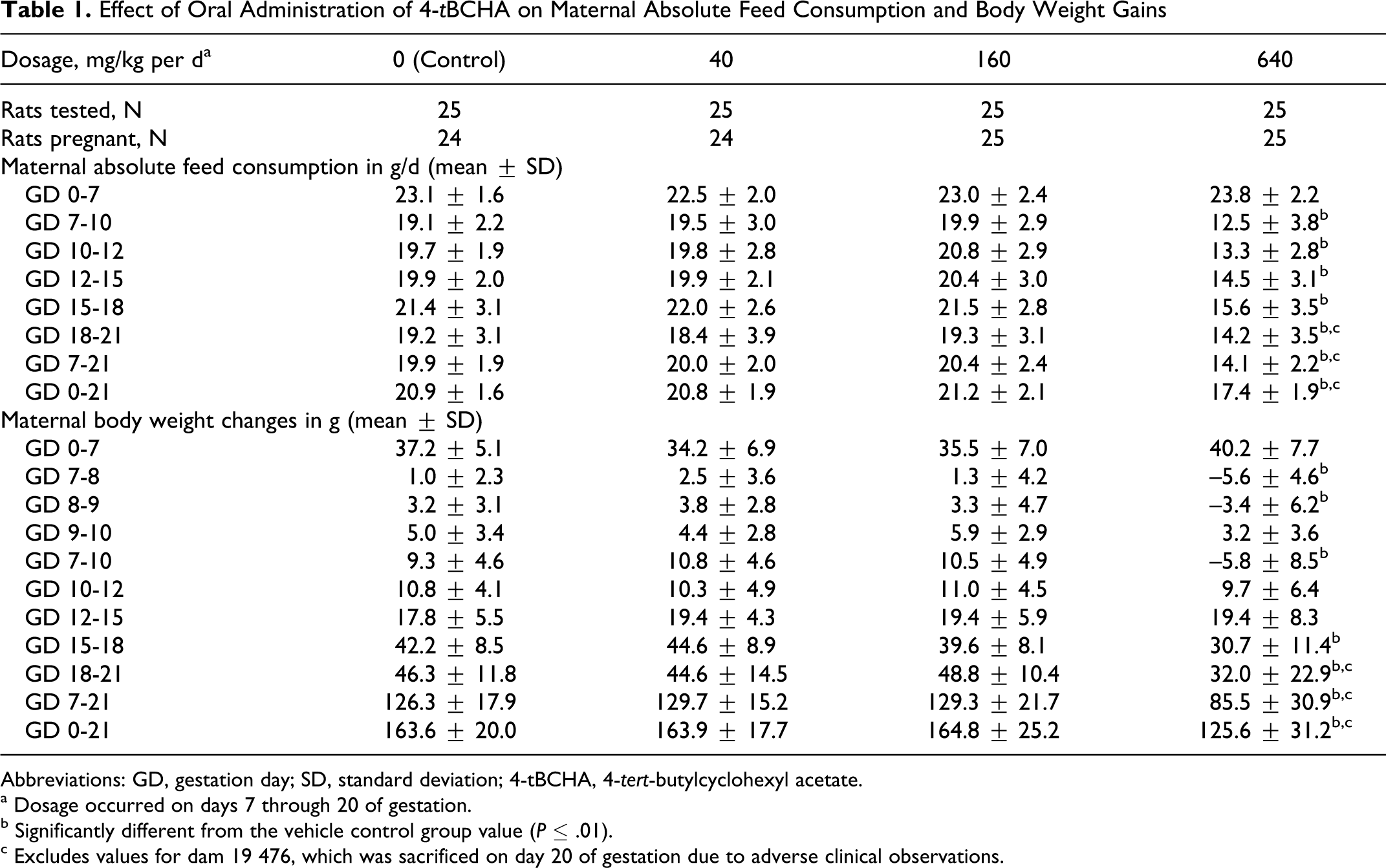
Abbreviations: GD, gestation day; SD, standard deviation; 4-tBCHA, 4-tert-butylcyclohexyl acetate.
a Dosage occurred on days 7 through 20 of gestation.
b Significantly different from the vehicle control group value (P ≤ .01).
c Excludes values for dam 19 476, which was sacrificed on day 20 of gestation due to adverse clinical observations.
Pregnancy occurred in 24 to 25 rats in each dosage group. Fetal body weights were significantly reduced in the 640 mg/kg per d dosage group when compared with vehicle control group values. The mean value for the combined male and female fetal body weights in the 640 mg/kg per d dosage group was approximately 11% less than that of the vehicle control group (Table 2).
Effect of Maternal Administration of 4-tBCHA on Caesarean Section and Litter Data
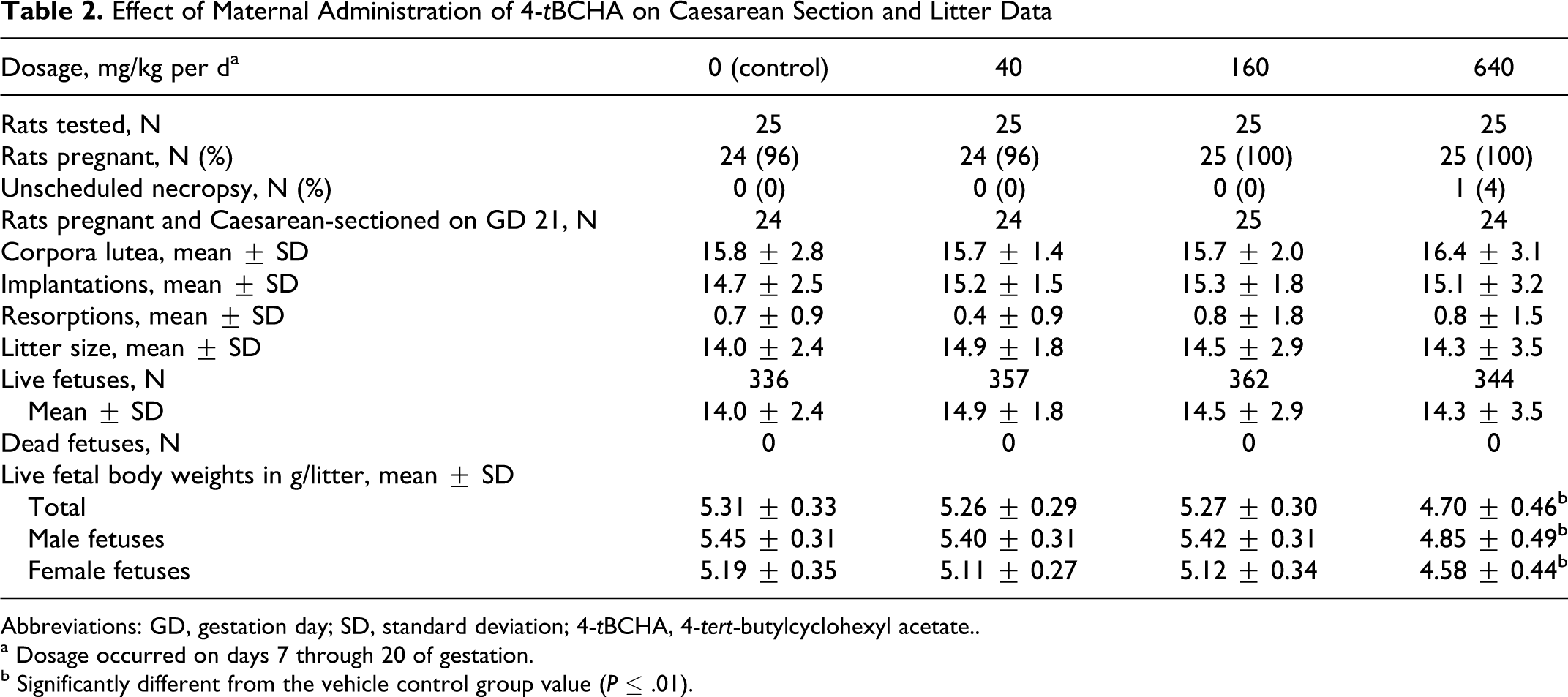
Abbreviations: GD, gestation day; SD, standard deviation; 4-tBCHA, 4-tert-butylcyclohexyl acetate..
a Dosage occurred on days 7 through 20 of gestation.
b Significantly different from the vehicle control group value (P ≤ .01).
No other Caesarean section or litter parameters were affected by 4-tBCHA dosages as high as 640 mg/kg per d. Litter averages for corpora lutea, implantations, litter sizes, live fetuses, and resorptions were comparable among the 4 dosage groups and did not significantly differ (Table 2). No dam had a litter that consisted only of resorbed conceptuses, and there were no dead fetuses. All placentae appeared normal.
Fetal evaluations were based on 336, 357, 362, and 344 live Caesarean-delivered fetuses in 24, 24, 25, and 24 litters in the 0, 40, 160, and 640 mg/kg per d dosage groups, respectively. Each of these fetuses was examined for gross external alterations, and 161, 170, 174, and 166 fetuses were examined for soft tissue alterations, while 175, 187, 188, and 178 were examined for skeletal alterations and fetal ossification site averages. Fetal alterations were defined as (1) malformations (irreversible changes that occur at low incidences in this species and strain) or (2) variations (common findings in this species and strain and reversible delays or accelerations in development).
The 640 mg/kg per d dosage of 4-tBCHA was associated with transient delays in fetal development, which included significant increases in fetal (but not litter) incidences of moderate enlargement of the renal pelvis of 1 or both kidneys, and reversible delays in ossification of the caudal vertebrae, fore limb and hind limb phalanges, and hind limb metatarsals. Two litters resulted in 7 (41.2%) of 17 and 1 (6.2%) of 16 alterations within fetuses showing slight-to-moderate enlargement of the renal pelvis. One litter resulted in 1 (5.6%) fetus out of 18 showing a folded right retina. Four litters resulted in 4 (25%) of 16, 3 (21.4%) of 14, 1 (6.7%) of 15, and 1 (6.2%) of 16 reversible skeletal alterations. The remaining 17 litters showed no other fetal variations.
There were no fetal malformations, and gross external alterations were limited to a single occurrence of an absent tail in 1 fetus in the 160-mg/kg per d group. Skeletal examination confirmed the absence of the tail and the presence of fewer than normal ossified lumbar, sacral, and caudal vertebrae (5, 1, and 0, rather than the normal 6, 3, and 7, respectively). This alteration was considered spontaneous in origin and unrelated to 4-tBCHA because it was a single event and not dosage related. No other gross external, soft tissue, or fetal skeletal alterations (either malformations or variations) were associated with 4-tBCHA dosages as high as 640 mg/kg per d (Table 3).
Fetal Alterations Observed (Gross External, Soft Tissue or Skeletal Variations, or Malformations) Following Oral Administration of 4-tBCHA to Pregnant Rats
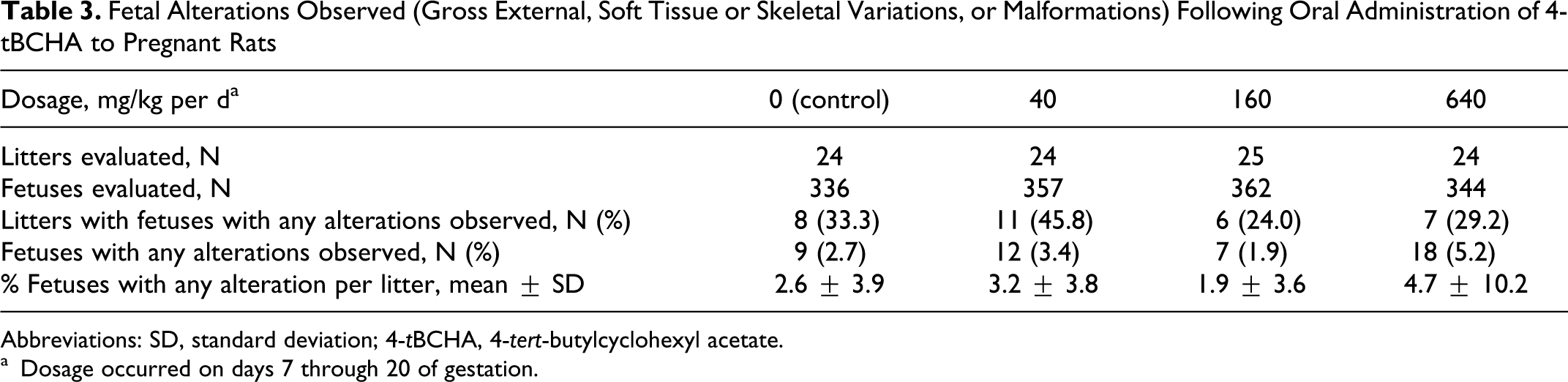
Abbreviations: SD, standard deviation; 4-tBCHA, 4-tert-butylcyclohexyl acetate.
a Dosage occurred on days 7 through 20 of gestation.
Discussion
The primary purpose of this study was to (1) determine whether daily systemic exposure to 4-tBCHA during gestation could produce adverse effects in pregnant rats or in the developing embryo-fetus and (2) determine the oral maternal and developmental NOAELs. The oral route was used in this study, even though 4-tBCHA use in fragrances is topical, because large topical doses in animals are difficult to interpret, due to confounding factors. These factors include licking of application sites, irritation when the site is occluded, and variations in skin absorption, metabolism, and macromolecules. 21
Cyclic acetates, of which 4-tBCHA is one, are known to be slight or moderate skin irritants when topically applied undiluted. For example, a 4-hour topical application of 0.5 mL neat 4-tBCHA under a semiocclusive dressing caused skin irritation in rabbits. 22 However, human studies show no evidence of irritation at current levels of cosmetic use at concentrations of 2% to 20%. 6 In the present study, the most sensitive clinical sign of a test article–related effect was excessive salivation. This observation appeared predominantly at the high dose of 640 mg/kg per d and is reportedly caused by local irritation due to the release of the acetate moiety from the 4-tBCHA molecule. 6 Irritation of the oral cavity may also have decreased maternal feed intake and consequent body weight gains, which in turn resulted in the observed reduced mean fetal body weights and delayed development.
Delays in fetal development that are associated with significant reductions in fetal body weight are not an uncommon finding. 23 The significant increase in slight or moderate dilation of the renal pelvises, a normal aspect of renal development in rats during late gestation, 24 is also a manifestation of this delayed fetal development and occurred in 1, 2, 0, and 8 fetuses of the control, low-, mid-, and high-dosage groups, respectively. The reversible delays in ossification 23 of the caudal vertebrae, fore limb and hind limb phalanges, and hind limb metatarsals likewise reflect the reduced fetal body weights in the high-dosage (640 mg/kg per d) group.
Conclusions
Adverse maternal and fetal effects were limited to the 640 mg/kg per d high-dosage group, thus 160 mg/kg per d was established as the maternal and the developmental NOAEL in this study. Based on a maximum human dermal exposure of 0.2 mg/kg per d for high end users and 100% dermal absorption, a margin safety of at least 800 exists between the maximum daily human dermal exposure and possible adverse fetal effects. It is concluded that 4-tBCHA is not a developmental toxicant in rats.
Footnotes
Authors’ Note
Portions of this work were presented at the 47th Annual Meeting of the Society of Toxicology, 2008, Seattle, Washington, USA. This study was conducted at Charles River Laboratories Preclinical Services, Horsham, Pennsylvania, USA, and funded by the Research Institute for Fragrance Materials, Inc, Woodcliff Lake, New Jersey, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
