Abstract
Quaternary ammonium salts, including alkyl chain, alkanol, and polymer derivatives (trimoniums) are used in cosmetics mainly as surfactant-cleansing agents, hair-conditioning agents, and antistatic agents. The Cosmetic Ingredient Review Expert Panel reviewed the relevant animal and human data and noted gaps in the available safety data for some of the trimomiums. The available data on many of the trimoniums are sufficient, however, and similar structural activity relationships, functions in cosmetics, and cosmetic product usage supported extending these data to the entire group. These ingredients were determined to be safe in the present practices of use and concentration when formulated to be nonirritating.
Keywords
Introduction
Three quaternary ammonium salts used as cosmetic ingredients, cetrimonium chloride, cetrimonium bromide, and steartrimonium chloride, were found to be safe for use in rinse-off products and safe for use at concentrations of up to 0.25% in leave-on products by the Cosmetic Ingredient Review (CIR) Expert Panel in 1997. 1 These trimethyl ammonium ingredients are part of a larger group of quaternary ammonium salts used in cosmetics. Because the available data on these 3 ingredients are considered relevant to the safety of other quaternary ammonium salts, the safety assessment of cetrimonium chloride, cetrimonium bromide, and steartrimonium chloride was expanded to consider the safety of a larger group of structurally similar ingredients.
The International Cosmetic Ingredient Dictionary and Handbook defines many cosmetic ingredients that are structurally similar to cetrimonium chloride, cetrimonium bromide, and steartrimonium chloride, and these additional ingredients have been included in this amended safety assessment. 2 Each of these additional cosmetic ingredients is also a quaternary ammonium salt wherein 3 of the 4 substituents on the nitrogen atom that comprise the quaternary ammonium moiety are methyl groups. This gives rise to the “trimonium” naming convention (a combination of trimethyl and ammonium) used for many of the cosmetic ingredients addressed in this safety assessment. Accordingly, the overall grouping has been given the designation trimoniums and includes 52 ingredients (see Tables 1–3; Figure 1).
Continued

Continued
Continued
Continued
A, Structure map of the straight chain alkyl ingredients in this assessment (including original 3 assessed ingredients: cetrimonium chloride, cetrimonium bromide, and steartrimonium bromide, in boldface).
Straight Chain Alkyl, Trimonium Compound and Their Salts in This Safety Assessment With CAS Numbers, Functions in Cosmetics, Technical Names, and Trade Names.
Alkanol Trimonium Ingredients and Related Ethers/Esters/Acids in This Safety Assessment With CAS Numbers, Functions in Cosmetics, Technical Names, and Trade Names.

Polymers Containing Trimonium in This Safety Assessment With CAS Numbers, Functions in Cosmetics, Technical Names, and Trade Names.
The fourth substituent on the nitrogen atom differentiates each of the trimoniums and may be a straight, branched, substituted, or unsubstituted alkyl chain. Having 4 nonhydrogen substituents on the nitrogen atom results in a positively charged ion (cation) which is not pH dependent. The charged nature of these ingredients has a significant, but predictable, impact on their chemical and physical properties.
The ingredients in this safety assessment are segregated according to the function of the fourth substituent into 1 of 3 groups: (1) unsubstituted straight or branched alkyl chain trimoniums; (2) alcohol bearing alkyl chain (alkanol) trimoniums; or (3) an alkyl chain trimonium incorporated into a polymer backbone (Tables 1-3; Figure 1). These trimonium ingredients mostly function as antistatic agents, hair-conditioning agents, cleansing agents, surfactants, and film formers.
While many of these ingredients vary only by hydrocarbon chain length, there are also branched ingredients, alcohols, esters, ethers, and polymers. Nonetheless, these ingredients do comprise a family of chemicals with similar functions in cosmetics. It is expected that the toxicity of alkanol trimonium compounds would be similar to that of alkyl trimonium compounds and the data on one ingredient may be extrapolated to the other ingredients in this safety assessment—a process usually termed as read-across. To test the read-across reliability, similar toxicity end points were considered across the fourth substituent. Where data were available, the findings were similar. For example, trimoniums dissociate into their ionic components in aqueous cosmetic formulations. Upon incorporation of trimoniums into cosmetic formulations, the cationic chains are attracted to anionic charges in the protein structure of skin and hair, resulting in a conditioning effect. These charged ingredients are unlikely to cross the lipid bilayer but tend to be irritating to the skin and eyes. 3 The lowest molecular weight, and shortest chain, alkyl trimonium in this assessment, laurtrimonium bromide, exhibited percutaneous absorption of <0.1% over 48 hours in rats with no measurable presence in the blood.4,5 Higher molecular weight, longer chain trimonium ingredients would likely be similarly or less absorbable. Studies of the percutaneous absorption of a trimonium decorated polymer resulted in no evidence of dermal penetration in rats. 5
The polymers represent very large molecular entities, wherein the trimonium groups function to bring a certain charge density to the molecule. Copolymerization or polymerization with nonionic comonomers serves to decrease this charge density to a desired level for applications such as hair and skin conditioning. 6
Polyquaternium 10, a trimonium functionalized cellulose, is similar to the polymers in this assessment and was previously assessed by the CIR Expert Panel in 1988. It was concluded that “. . . [p]olyquaternium-10 is safe as a cosmetic ingredient in the present practices of use.” 7 This conclusion was confirmed in 2005. 8 While the structure of polyquaternium 10 did not make it a candidate to add to this safety assessment, summaries of the data in that assessment will be included in the appropriate sections here because those data do support the safety of trimonium polymers.
The data from the original safety assessment of cetrimonium chloride, cetrimonium bromide, and steartrimonium chloride are summarized in the appropriate sections in this safety assessment.
Chemistry
Straight and Branched Chain Alkyl Trimonium Ingredients
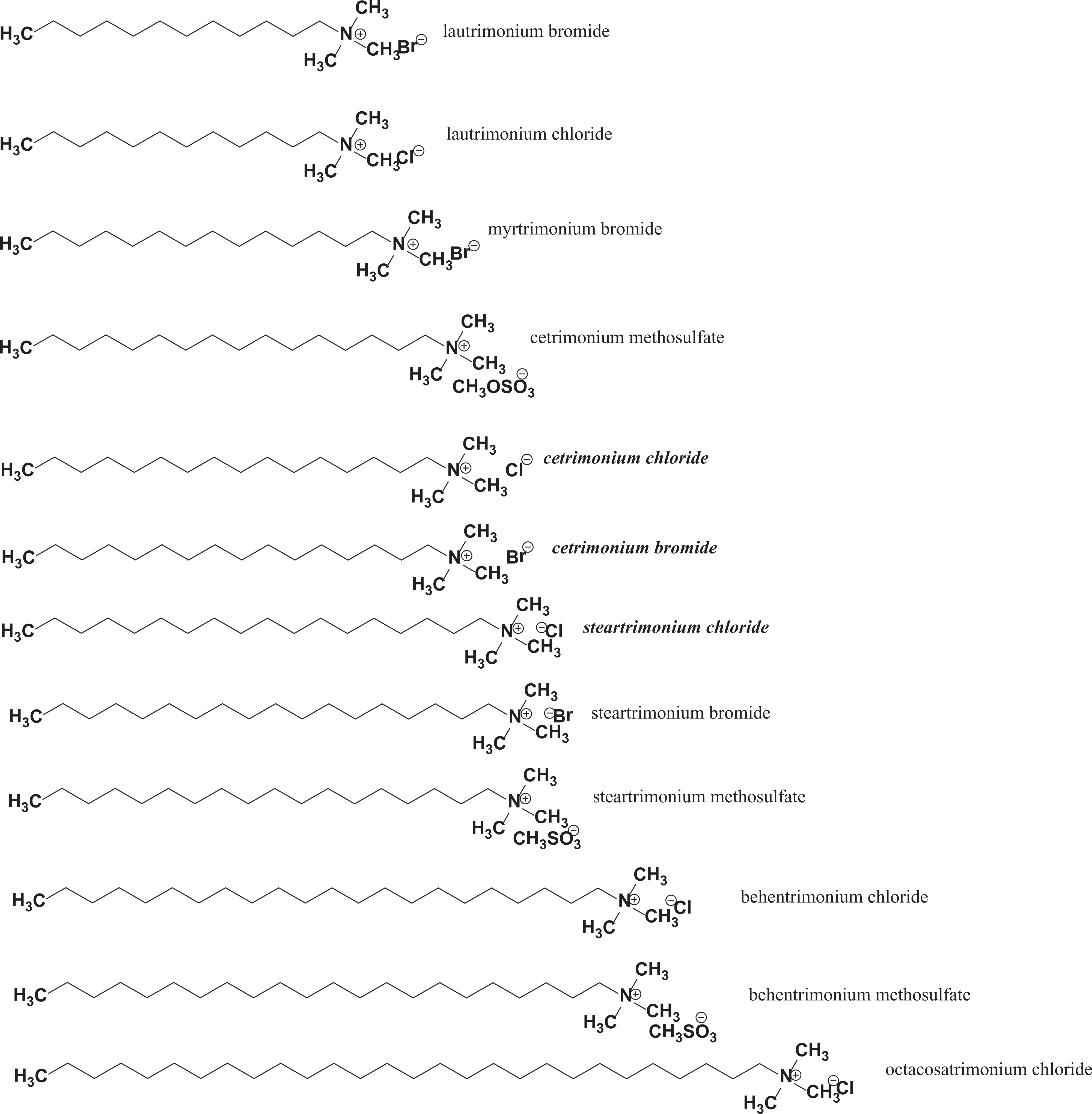
Cetrimonium bromide, cetrimonium chloride, and steartrimonium chloride and the other unsubstituted alkyl trimoniums vary by alkyl chain residue length, the degree of unsaturation, the existence of chain branching, or a combination of these variations. Ingredients in this group are shown in Table 1 with CAS Registry numbers, function in cosmetics, technical names, and trade names. These ingredients are:
Laurtrimonium bromide
Laurtrimonium chloride
Myrtrimonium bromide
Cetrimonium chloride
Cetrimonium bromide
Cetrimonium methosulfate
Steartrimonium chloride
Steartrimonium bromide
Steartrimonium methosulfate
Behentrimonium chloride
Behentrimonium methosulfate
Octacosatrimonium chloride
Ceteartrimonium chloride
Hydrogenated tallowtrimonium chloride
Hydrogenated palmtrimonium chloride
Soytrimonium chloride
Tallowtrimonium chloride
Cocotrimonium chloride
Cocotrimonium methosulfate
Octyldodecyltrimonium chloride and
Dodecylhexadecyltrimonium chloride
Some of the alkyl residues in this group are derived from botanical sources and are actually mixtures of different chain length salts (eg, soytrimonium chloride). Furthermore, this first group also varies in the identity of the corresponding anions (eg, bromide, chloride, and methosulfate). A mapping of how these molecules vary in length is shown in Figure 1A.
Alkanol Trimonium Ingredients
The alkanol trimonium ingredients consist of various alcohol-substituted trimoniums and related ethers, esters, and acids. These ingredients can be subdivided into those with a choline function and those with a carnitine function. These ionic compounds also vary by chain length. In one instance, the alkyl residue is derived from botanical ingredients and results in a mixture of salts (cocoylcholine methosulfate). The CAS Registry numbers, functions in cosmetics, technical names, and trade names of the ingredients in this group are shown in Table 2 and the structures are presented in Figure 1B. These ingredients are:
Choline chloride
Stearoxypropyltrimonium chloride
Lauroyl ethyltrimonium methosulfate
Myristoyl ethyltrimonium methosulfate
Palmitoyl ethyltrimonium methosulfate
Stearoyl ethyltrimonium methosulfate
Cocoylcholine methosulfate
Carnitine
Carnitine HCl
Carnitine hydroxycitrate
Carnitine PCA
Palmitoyl carnitine
Acetyl carnitine and
Acetyl carnitine HCl
Polymeric Trimonium Ingredients
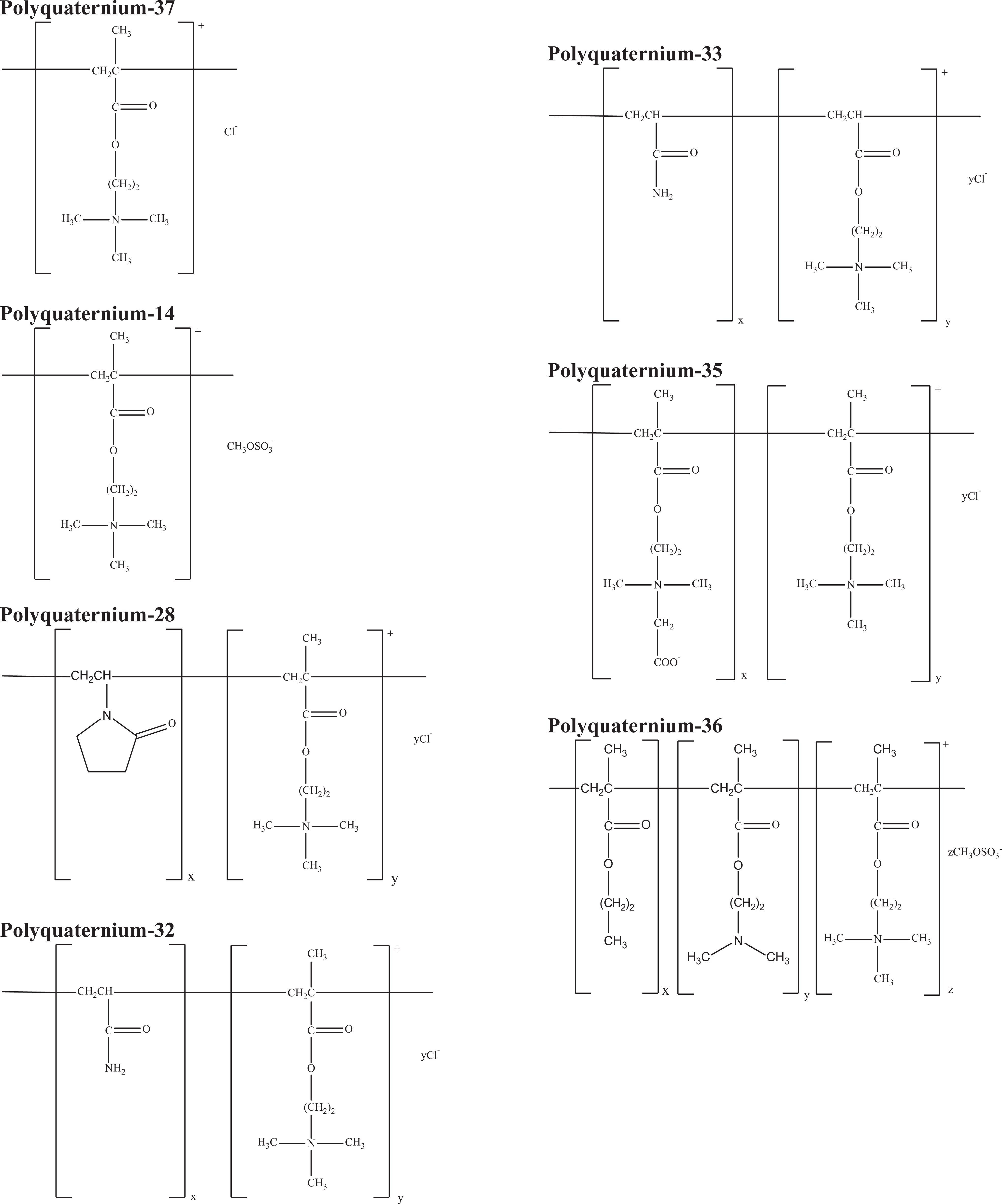
The final group of ingredients consists of various quaternized polymers. These ingredients consist of homo- and co-polymers with at least 1 trimonium-containing repeat unit, and range in molecular weight from ∼400 000 to 7 000 000. 6 Ingredients in this group are shown in Table 3 with CAS Registry numbers, functions in cosmetics, technical names, and trade names. Idealized structures are provided in Figure 1C. These ingredients are:
Polyquaternium-37
Polyquaternium-14
Polyquaternium-28
Polyquaternium-32
Polyquaternium-33
Polyquaternium-35
Polyquaternium-36
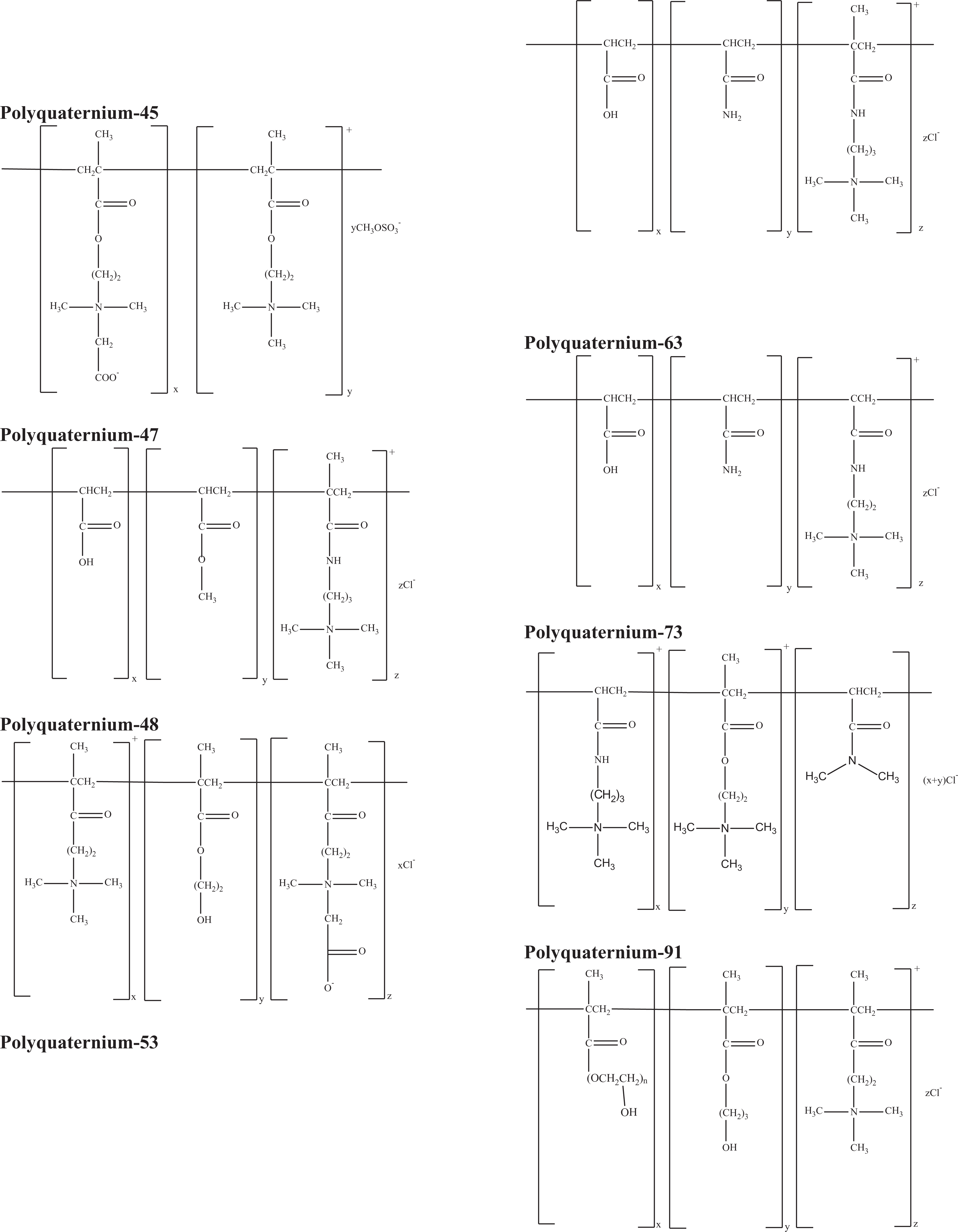
Polyquaternium-45
Polyquaternium-47
Polyquaternium-48
Polyquaternium-53
Polyquaternium-63
Polyquaternium-73
Polyquaternium-91
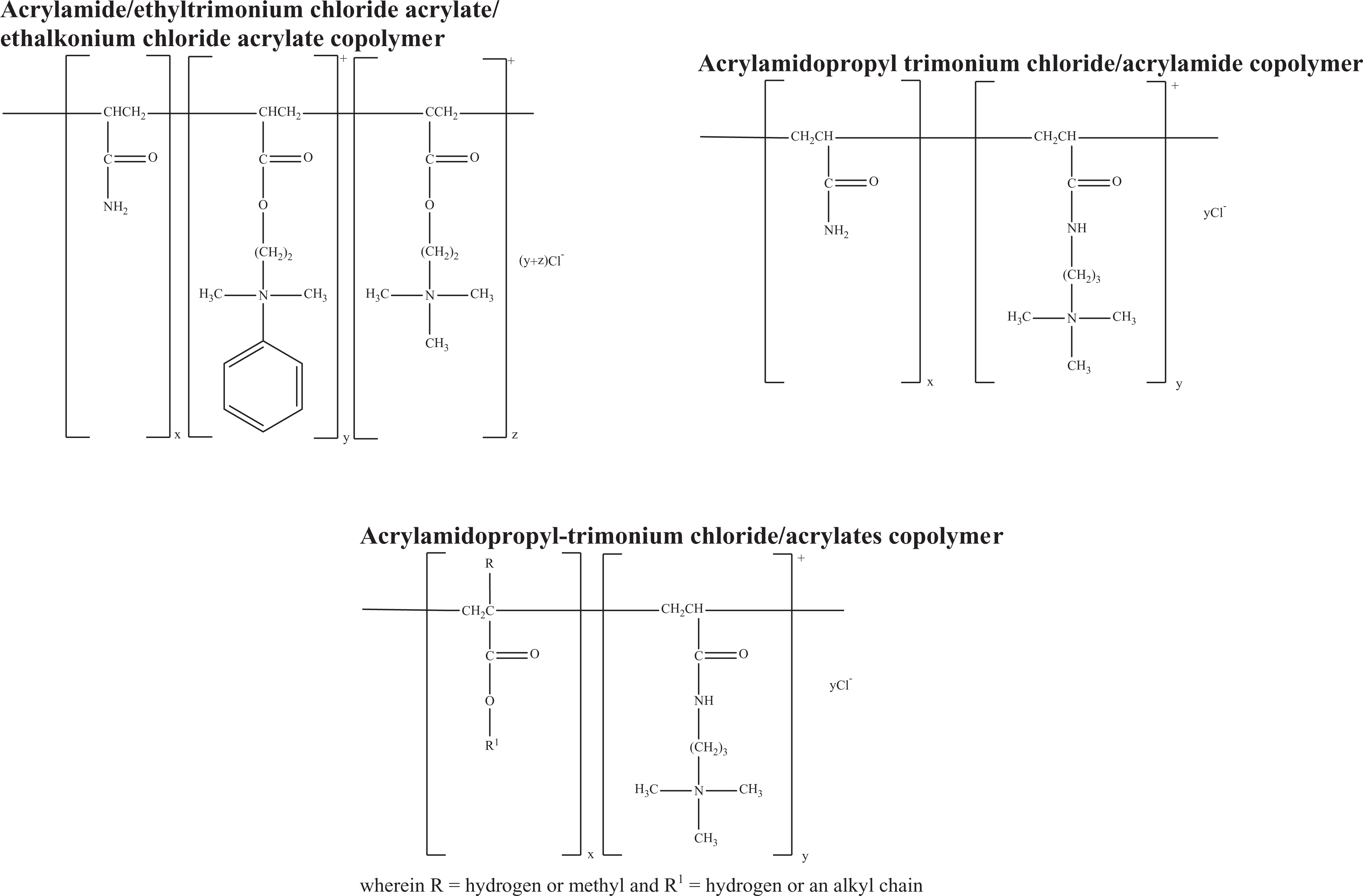
Acrylamide/ethyltrimonium chloride acrylate/ethalkonium chloride acrylate copolymer
Acrylamidopropyl trimonium chloride/acrylamide copolymer and
Acrylamidopropyl-trimonium chloride/acrylates copolymer
Physical and Chemical Properties
The calculated and available experimental chemical properties for the included ingredients are given in Table 4. Surfactants, or surface-active agents, are by definition amphipathic. 9 Typically, one portion of the surfactant will be hydrophilic, while the other end is hydrophobic. One particular category of surfactants is the cationic surfactants. While these ion pairs (or salts) have negatively charged counter ions (anions; eg chloride or bromide), the positively charged cations impart the majority of the surfactant character. The trimoniums are a subcategory of the quaternary ammonium surfactants, wherein 3 of the 4 substituents on the nitrogen atom, comprising the quaternary ammonium, are each methyl group.
Physical and chemical properties.



a Estimation Programs Interface Suite T. 111
Typically, the manufacture of trimoniums occurs by the reaction of a tertiary amine (ie, a nitrogen atom with 2 methyl groups and 1 alkyl group bonded to it) with a classic alkylating agent, such as a methyl halide or dimethyl sulfate. 10 For example, behentrimonium methosulfate can be prepared by the addition of N, N-dimethyldocosan-1-amine to dimethyl sulfate (Figure 2). Mixtures, like ceteartrimonium chloride (a mixture of cetrimonium chloride and steartrimonium chloride), are often synthesized by the addition of various haloalkanes (ie, a cetyl halide and a stearyl halide) to trimethylamine. 11
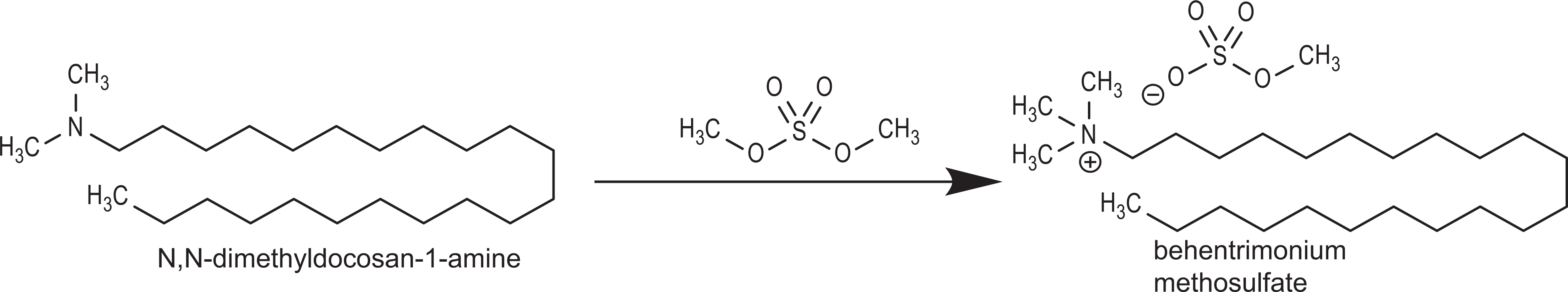
Example synthesis of alkyl trimoniums (behentrimnonium methosulfate).
Straight and Branched Chain Alkyl Trimonium Ingredients
The first group of trimoniums in this safety assessment (Table 1) consists of cations, each comprising a nitrogen atom bonded to 3 methyl groups and a simple alkyl chain, which can vary in length from 12 (eg, laurtrimonium bromide) to 28 carbons in length (eg, octacosatrimonium chloride; Figure 1A). All of these straight chain trimoniums are waxy-solids at human physiological temperatures. The branched chain trimoniums, however, are liquids at the same temperatures. Water solubility and volatility decrease and melting/boiling points increase as chain length is increased. For instance, straight chain trimoniums up to 16 carbons in length (eg, lautrimoniums [C12], myrtrimonium bromide [C14], and cetrimoniums [C16] are water soluble, but longer chain trimoniums (eg, steartrimoniums [C18], behentrimoniums [C22], and octacosatrimonium chloride [C28]) are not water soluble. 6
For example, behentrimonium chloride (Table 1) is a waxy solid and is dispersible in water. Behentrimonium chloride is stable but is incompatible with strong oxidizing agents. 12 When mixed with cetearyl alcohol (concentration not provided) behentrimonium chloride is a white solid with a specific gravity < 1. 13
Behentrimonium chloride may originate from plant sources that one manufacturer states do not contain any genetically modified organisms. 14 It is made through the process of quaternization of behenyl dimethylamine with methyl chloride, in 30% dipropylene glycol.
Alkanol Trimonium Ingredients
The alkanol trimonium ingredients in this safety assessment (Table 2) differ from the alkyl trimonium ingredients by the addition of an ethoxy functional group attached to the trimonium core nitrogen (Figure 1B). The simplest of these, and a major metabolite of all of the other members in the group, is choline. Choline chloride was reported to not be volatile at 21ºC and is stable under ordinary conditions of use and storage. 15 Choline chloride may produce carbon monoxide, carbon dioxide, nitrogen oxides, and hydrogen chloride when heated to decomposition. Choline chloride was reported to be incompatible with strong oxidizers.
Included within this group are the choline ethers and esters, with various chain lengths, which are crystalline solids. Also within this group are the carnitine acids and esters. The carnitines, which are also solids, differ from the cholines by the attachment of an acid or ester functional group at the hydroxyl-bearing carbon of the choline core.
Carnitine is an amino acid derivative found in high energy demanding tissues and was reported to be an essential cofactor for the transport of long-chain fatty acids across the inner mitochondrial membrane into the mitochondrial matrix. 16 Carnitine was reported to be stable under normal temperatures and pressures. 17 It was also reported to be incompatible with strong oxidants and exposure to moist air or water. Carnitine decomposes into nitrogen oxides, carbon monoxide, carbon dioxide, and nitrogen.
Polymeric Trimonium Ingredients
The third group of trimoniums in this safety assessment (Table 3 and Figure 1C) differ from the other groups by their incorporation into a polymer backbone (ie, these are trimonium functionalized/decorated polymers). The molecular weights and various physical properties of the trimonium polymers can vary greatly based on the polymerization conditions utilized in their synthesis. Accordingly, molecular weights of the individual monomer units are provided instead. Typical molecular weights of these polymers, however, are usually above 400 000 g/mol. 6
One polymer, polyquaternium-28, was reported to be 100% ionized in water and has an infinitely small pK. 18 Polyquaternium-28 is a copolymer comprised of a trimonium monomer and a lactam (ie, a heterocycle containing an amide in the ring structure) monomer. The amide group from the lactam monomer behaves as a weak base. Polyquaternium-28 was reported to be incompatible with strong oxidizing agents and reducing agents. This polymer was reported to be used in an aqueous solution at 20%.
Polyquaternium-10 differs structurally from all of the other trimonium polymers in this safety assessment by having a cellulose backbone. Otherwise, polyquaternium-10 is very closely related to these trimonium polymer ingredients. This large chemical entity (molecular weight 250 000-600 000 g/mol) is substituted with the same trimonium bearing side chains as the other trimonium polymers and provides the same type of cationizing or conditioning effect.
Other
Physical properties were not found for: cocotrimonium methosulfate, cocoylcholine methosulfate, hydrogenated palmtrimonium chloride, and hydrogenated tallowtrimonium chloride and were not calculable since these are mixtures of different chain length trimoniums with unknown ratios.
Methods of Manufacture
The synthesis of behentrimonium methosulfate is representative of ingredients in the alkyl trimonium group and is shown in Figure 2.
The synthesis of polyquaternium-37 is representative of the ingredients in the trimonium polymer group and is shown in Figure 3. The typical synthesis of polyquaternium-10 is via an epoxide and is shown in Figure 4.
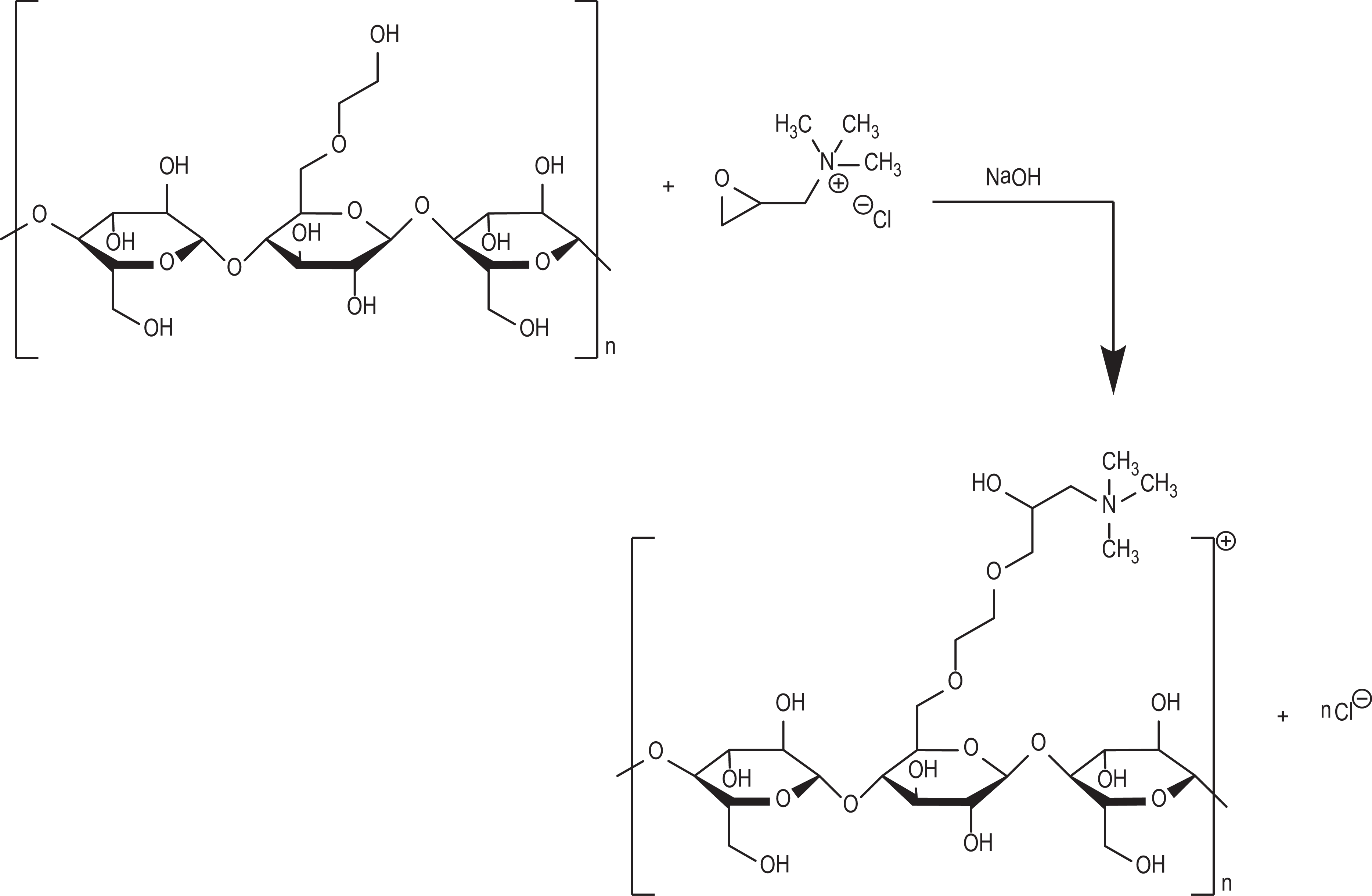
Synthesis of cellulose backbone trimonium decorated polymers (Polyquaternium-10).
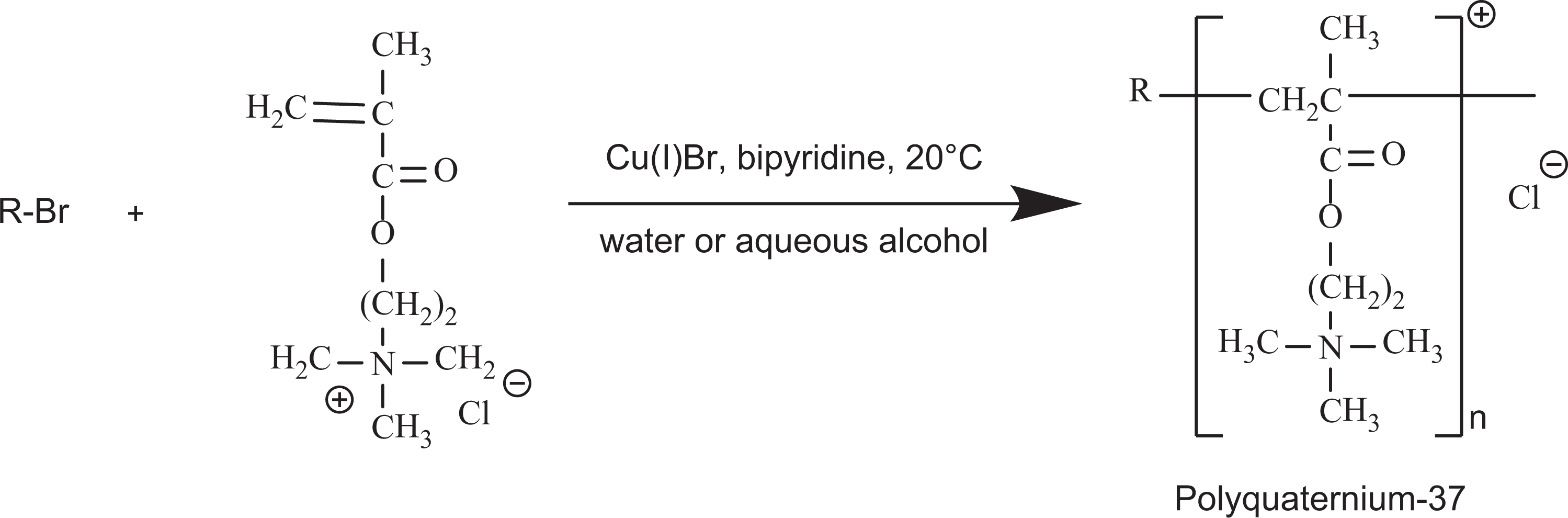
Example synthesis of trimonium decorated polymers (Polyquaternium-37).
Impurities
Straight and Branched Chain Alkyl Trimonium Ingredients
Cetrimonium bromide and myrtrimonium bromide are reported to be 99% pure. 19
Alkanol Trimonium Ingredients
One supplier reports that the organic impurities of choline chloride to be trimethylamine (max 500ppm), ethylene glycol (500 ppm), organic impurities (ie, trimonium methosulfate, glycol, chloroethanol; max 1500 ppm), color (max 50 hazen), and heavy metals (ie, lead; 20 pm). 20
Available sources of carnitine are reported to be > 99% pure. 17
One available source of acetyl carnitine HCl was reported to be 98.5% to 100.0% pure. 21 Lead was reported to be present at <20 ppm and sulfated ash at ≤0.30%.
Polymeric Trimonium Ingredients
Polyquaternium-28 was reported to be >99% pure by one supplier. 18 The maximum content of the residual monomer was reported to be <1%.
Use
Cosmetic
According to information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), cetrimonium bromide was used in a total of 37 cosmetic formulations at the time of the first safety assessment in 1997 (Table 5). 1 No use concentrations were reported at that time. Currently, VCRP data indicated that cetrimonium bromide was reported to be used in 68 cosmetic products of multiple types. 22 A survey of current use concentrations conducted by the Personal Care Products Council (Council) reported a range from 0.1% to 3%. 23
Cetrimonium chloride was used in a total of 162 cosmetic formulations at the time of the first safety assessment. 1 Currently, VCRP data indicated that cetrimonium chloride was reported to be used in 959 cosmetic products. 22 A survey of current use concentrations reported a range from 0.0008% to 10%. 23
Steartrimonium chloride was used in a total of 6 cosmetic formulations at the time of the first safety assessment. 1 Currently, VCRP data indicated that cetrimonium chloride was reported to be used in 40 cosmetic products. 22 A survey of current use concentrations reported a range from 0.06% to 4%. 23
The other straight chain alkyl trimonium ingredients (behentrimonium chloride, behentrimonium methosulfate, cetrimonium methosulfate, cocotrimonium chloride, cocotrimonium methosulfate, laurtrimonium chloride, myrtrimonium bromide, soytrimonium chloride, and tallowtrimonium chloride) have reported uses mostly in hair care, skin care, and makeup products at 0.0005% to 10% in rinse-off and at 0.001% to 4% in leave-on products (Table 6).22,23 These ingredients function as cosmetic biocides, hair-conditioning agents, antistatic agents, emulsifying agents, and surfactant-emulsifying agents. No uses or concentrations of use were reported for ceteartrimonium chloride, cocotrimonium methosulfate, dodecylhexadecyltrimonium chloride, hydrogenated palmtrimonium chloride, hydrogenated tallowtrimonium chloride, laurtrimonium bromide, octacosatrimonium chloride, octyldodecyltrimonium chloride, steartrimonium bromide, and steartrimonium methosulfate.
a 0.4% after dilution.
b 3% after dilution.
c 1% after dilution.
The alkanol trimonium ingredients and related ethers/esters/acids (acetyl carnitine HCl, carnitine, carnitine HCl, carnitine hydroxycitrate, palmitoyl carnitine) are mostly used in skin care and make-up products at concentrations between 0.001% and 1% in both rinse-off products and leave-on products (Table 7).22,24 These ingredients function as skin-conditioning agents, antistatic agents, hair-conditioning agents, surfactant-cleaning agents, surfactant-emulsifying agents, surfactants-foam boosters, viscosity increasing agents, humectants, and exfoliants. No uses or concentrations of use were reported for choline chloride, stearoxypropyltrimonium chloride, lauroyl ethyltrimonium methosulfate, myristoyl ethyltrimonium methosulfate, palmitoyl ethyltrimonium methosulfate, stearoyl ethyltrimonium methosulfate, and acetyl carnitine.
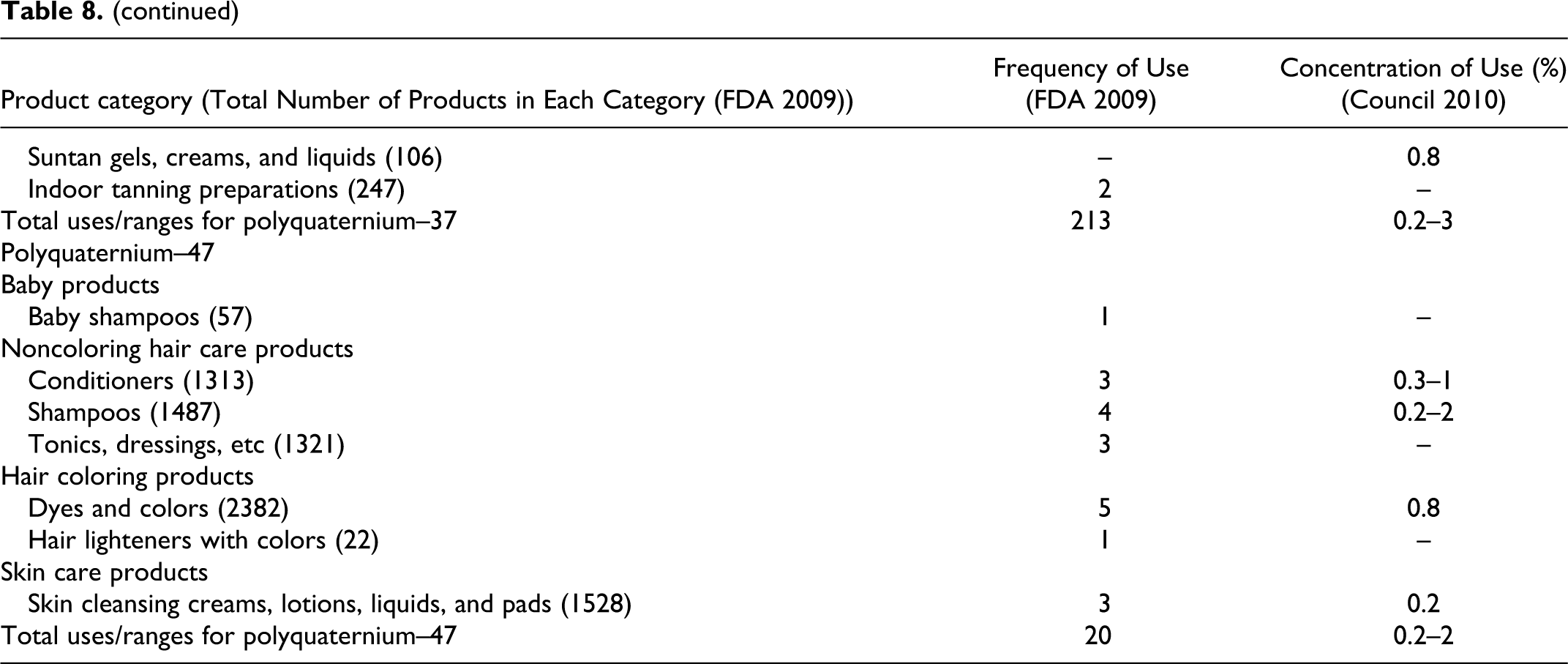
Trimonium polymers (acrylamidopropyltrimonium chloride/acrylamide copolymer, polyquaternium-28, polyquaternium-32, polyquaternium-35, polyquaternium-37, and polyquaternium-47) are mostly used in hair care and skin care products at concentrations between 0.04% to 10% in rinse-off products and 0.2% and 3% in leave-on products (Table 8). These ingredients function as antistatic agents, hair fixatives, film formers, skin-conditioning agents–miscellaneous, hair-conditioning agents, slip modifiers, and surface modifiers. No uses or concentrations of use were reported for polyquaternium-14, polyquaternium-33, polyquaternium-36, polyquaternium-45, polyquaternium-48, polyquaternium-53, polyquaternium-63, polyquaternium-73, polyquaternium-91, acrylamide/ethyltrimonium chloride acrylate/ethalkonium chloride acrylate copolymer, and acrylamidopropyl-trimonium chloride/acrylates copolymer.
For ingredients used in cosmetic sprays and aerosols, it is important to consider inhalation safety. Safety of inhaled aerosols depends on the ingredient, the concentration, the duration of the exposure and where they are deposited within the respiratory system. 25 The site of deposition is associated most with the particle size and density of the particle being inhaled.
Absorption of gases and vapors by inhalation is determined by the partitioning of the compound between the blood and the gas phase along with its solubility and tissue reactivity. The important characteristics that affect absorption after exposure to aerosols are the aerosol size and water solubility of any chemical present in the aerosol. In general, the smaller the particle, the further into the respiratory tree the particle will deposit and the greater the impact on the respiratory system.
The parameter most closely associated with this regional deposition is the aerodynamic diameter, da, defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract.26,27 Nanoparticles have the potential to deliver high amounts of particulates to the lung. 28
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 29 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. 30 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
The Panel has discussed this issue and has decided that in the absence of inhalation toxicity data on a specific ingredient, they will consider the aerosol and spray particle sizes in determining its inhalation safety.
In the European Union (EU), behentrimonium chloride, ceteartrimonium chloride, cetrimonium bromide, cetrimonium chloride, cocotrimonium chloride, hydrogenated palmtrimonium chloride, hydrogenated tallowtrimonium chloride, laurtrimonium bromide, laurtrimonium chloride, myrtrimonium bromide, soytrimonium chloride, steartrimonium bromide, steartrimonium chloride, and tallowtrimonium chloride were restricted to 1% when used as a preservative. 31
The EU has limited the following ingredients to 1 mg/kg in leave-on body care products and 0.5 mg/kg in other products: polyquatermium-32, polyquaternium -47, polyquaternium-53, acrylamide/ethyltrimonium chloride acrylate/ethalkonium chloride acrylate copolymer, and acrylamidopropyl trimonium chloride/acrylamide copolymer. 32
Choline chloride is an Annex II substance in the EU and is not to be used in cosmetics in Europe. 33 The Scientific Committee on Cosmetic Products (SCCP) 34 concluded that the available data on choline chloride was not sufficient to address concerns about mucous membrane irritation when used in cosmetics.
Noncosmetic
Straight and branched chain alkyl trimonium ingredients
Laurtrimonium bromide was reported to be used for the separation and purification of DNA fragments. 35
A commercial product containing cetrimonium bromide (and other quaternary ammonium salts) was reported to be used as a topical antiseptic. 1
Alkanol trimonium ingredients
Choline chloride was reported to be considered generally recognized as safe (GRAS) as a nutrient by the FDA. 36
Choline and carnitine are reported to be used as dietary supplements.37,38
Absorption, Distribution, Metabolism, and Excretion
Oral
Straight and branched chain alkyl trimonium ingredients
The original safety assessment reported that orally administered cetrimonium bromide (0.8 mg/kg) was poorly absorbed by the intestinal tract of rats. 1 Greater than 90% of the cetrimonium bromide was recovered 3 days after oral administration in the feces.
Alkanol trimonium ingredients
Choline consumed in the diet is important to the structural integrity of cell membranes, methylation metabolism, cholinergic neurotransmission, transmembrane signaling, and lipid and cholesterol transport and metabolism. 20 Choline was obligatory to human cell survival. It was available in the diet as free choline and as phosphatidylcholines, such as lecithin in egg yolks, vegetables, and animal fat.
Choline consumed in the diet by mammals was reported to be absorbed from the lumen of the small intestine using transporter proteins in the enterocyte.39–42 In the gut, choline can be liberated from phosphatidylcholine by pancreatic enzymes. 43 Some metabolism by bacteria to form betaine and methylamines was reported to be required before choline can be absorbed from the gut. 44 The free choline enters the portal circulation of the liver, whereas phosphatidylcholine may enter via lymph in chylomicrons. 45
Choline functions as a precursor for acetylcholine, phospholipids, and the methyl donor betaine.46,47
All tissues accumulate choline by diffusion and mediated transport. 48 A specific carrier mechanism transports free choline across the blood–brain barrier at a rate that was reported to be proportional to the serum choline concentration. In the neonates, this choline transporter has a high capacity. 49 The rate at which the liver takes up choline was reported to be sufficient to explain the rapid disappearance of choline injected systemically. 50 The kidney also accumulates choline. 51 Some choline from the kidney appears in the urine unchanged but most was reported to be oxidized within the kidney to form betaine. 52 A significant portion of choline was reported to be oxidized to form betaine in the liver and kidney.53,54
Choline was reported to be phosphorylated, converted to cytidine diphosphocholine, and then converted to phosphatidylcholine in the predominant pathway for phosphatidylcholine biosynthesis.55,56 In an alternative pathway, phosphatidylethanolamine was reported to be sequentially methylated to form phosphatidylcholine by the enzyme phosphatidylethanolamine-N-methyltransferase using S-adenosylmethionine as the methyl donor.57,58 This was reported to be the major, and possibly only, pathway for synthesis of the choline moiety in adult mammals. It was reported to be most active in the liver but has been identified in many other tissues.59–61 There are no estimates available as to the relative extent of choline obtained from cell turnover.
Acetyl
Polymeric trimonium ingredients
No information was discovered on the oral absorption, distribution, metabolism, and excretion of the trimonium polymers.
Dermal
Straight and branched chain alkyl trimonium ingredients
The percutaneous absorptions of [ 14 C] laurtrimonium bromide (0.5%) through nonoccluded rat skin and the resulting blood levels were studied. 4 Percutaneous absorption was low. Application in a cream hair-rinse preparation under user conditions (10 cm2 area, lathered for 3 minutes, left for 15 minutes, rinsed, dabbed dry) resulted in the absorption of ∼0.1% of the administered radioactivity in 48 hours. No measurable radioactivity was present in the blood. Application of the surfactant at a higher concentration (3%) in an aqueous solution resulted in a higher absorption of 0.6% in 72 hours. Some radioactivity, equivalent to < 100 ng of unchanged [ 14 C] laurtrimonium bromide, was measured in the blood after administration to the skin without subsequent rinsing.
The original safety assessment reported that a percutaneous absorption study showed that 0.59% of 1% cetrimonium bromide penetrated rat skin after 15 minutes; 0.93% of 0.5% cetrimonium bromide in a hair rinse formulation penetrated after 5 min of exposure followed by rinsing; and 3.15% of 3.0% cetrimonium bromide in water penetrated after 15 minutes of exposure. 1
Dehyquart A-CA (25% cetrimonium chloride in a 3.5% emulsion; 25 mg/cm2 100 mg on 4 cm2) was applied to the dermatomed, thawed, full thickness skin (1000 µm; n = 6) of the back and flank of castrated male pigs in a diffusion chamber for 30 minutes. 63 The test substance was then washed off and the receptor fluid sampled up to 72 hours. At 72 hours, the amount of test substance was measured by high-pressure liquid chromatography/electron spectroscopic imaging/mass spectrometry (HPLC/ESI/MS) and was below detection limits in the receptor fluid, 0.7 ± 0.6% in the stratum corneum, 0.3% ± 0.3% in the dermis, 1.0% ± 0.9% total in the skin, and 90.2% ± 4.5% in the rinsing solution. The authors concluded that cetrimonium chloride may not be systemically available by a dermal route.
Alkanol trimonium ingredients
Full thickness human skin (n = 12) was used in Franz cells to test the penetration of radiolabeled choline chloride (5%) with and without occlusion. 64 Samples were taken up to 24 hours. At 24 hours, 0.457 µg/cm2 choline had penetrated into the receptor chamber for occluded skin and 0.383 µg/cm2 for unoccluded skin, 0.127% and 0.110% of the applied dose, respectively. There was no statistical difference in these results. Total absorption (epidermis, dermis, and receptor fluid) was 7.42 µg/cm2 (1.9%) and 13.86 µg/cm2 (3.43%) for occluded and unoccluded skin. Most of the choline remained in the epidermis and dermis. The authors concluded that choline chloride has a low potential for percutaneous absorption.
Polymeric trimonium ingredients
Less than 1.5% of polyquaternium-10 (100%; 4.0 mL/kg) penetrated the skin of Fisher 344 rats (n = 12) over 24 h. 7
Intravenous, Subcutaneous, and Intraperitoneal
Straight and branched chain alkyl trimonium ingredients
In the original safety assessment, cetrimonium bromide was rapidly excreted in the urine and feces of rats after subcutaneous and intervenous administration. Greater than 90% of the cetrimonium bromide was recovered 2 days after subcutaneous administration. There were only traces of cetrimonium bromide in the body 24 hours after intravenous (iv) injection. 1
Alkanol trimonium ingredients
[ 14 C]Carnitine (100 µCi/kg) was injected intraperitoneally (ip) into pregnant CD-1 mice on day 17 of pregnancy. 65 At 1, 2, 3, and 6 h after the injection, the mice were killed and a full body x-ray was performed. The highest concentrations of carnitine were found in the liver, placenta, kidney, myocardium and choroid plexus. No carnitine was observed in the brains of the fetuses or the dam. There was carnitine in the fetuses but less than that in the dam; the distribution was similar and increased with time. Carnitine crossed the placental barrier.
Hepatic Metabolism
Palmitoyl-
Penetration Enhancement and Inhibition
Straight and branched chain alkyl trimonium ingredients
Cetrimonium bromide (in a mineral oil-water emulsion) enhanced penetration of phenylazoaniline (pH 7.0), benzocaine (pH 7.0), and benzoic acid (pH 7.0) up to 0.5% in a Franz cell using dialysis or hydrophobic polydimethylsiloxane membrane membranes. 67 Concentrations above 0.5% were less effective until penetration was inhibited at ≥1.0%. Cetrimonium bromide inhibited penetration of benzoic acid (pH 3.0) and the penetration of phenol (pH 7.0) was not affected.
Alkanol trimonium ingredients
Palmitoyl carnitine (0.2 mmol/L) was reported to enhance the penetration of Lucifer yellow and Ruthenium red across Caco-2 monolayers by ∼10-fold and ∼20-fold, respectively. 68 Acetyl carnitine (with a shorter alkyl chain) did not enhance penetration using Caco-2 cell monolayers. Palmitoyl carnitine did not enhance the penetration of PEG 4000. The authors suggest that since there was no damage to the epithelium observed and there was rapid reversal of the effects with the removal of palmitoyl carnitine, the penetration enhancement was not due to cell lysis.
Nasal administration of palmitoyl carnitine (20%) simultaneously with human growth hormone increased the peak penetration by 260%, area under the absorption curve by 64%, and bioavailability by 12.2% in male Wistar rats (n = 5). 69
Cytotoxicity
Straight and branched chain alkyl trimonium ingredients
The lethal concentration of laurtrimonium chloride for rat primary hepatocytes was >0.048 µL/ mL. 70
Human K562 erythroleukemic cells were incubated with cetrimonium bromide (0.1 to 10 μmol/L) for 5 days (n = 3). 71 Cell growth decreased in a dose-dependent manner compared to controls. When in a liposome suspension or micellar solution, cell growth decreased in a dose-dependent manner; the IC50s (concentration that inhibits 50% of growth) were 0.88 and 0.62 mmol/L, respectively.
Human keratinocytes from foreskins were incubated in cetrimonium chloride for 3 days. 72 Metabolism was then measured using tetrazolium dye. Concentrations >3 µg/mL completely inhibited dye reduction. At 0.8 µg/mL, there was no inhibition of dye reduction. Concentrations between 0.01 and 0.1 µg/mL enhanced dye reduction. Measurement of DNA content revealed that at these low concentrations, cetrimonium chloride had a stimulatory effect on cell proliferation.
The minimum inhibitory concentration (MIC) of cetrimonium bromide was 16 mg/mL for Escherichia coli and 128 µg/mL for Pseudomonas aeruginosa. 73 The authors suggested that cetrimonium bromide was able to disrupt membrane function of gram-negative bacteria. In addition to and simultaneously with this disruption, this ingredient can also chelate K+ ions from the bathing medium.
The EC50 (effective concentration) values for cetrimonium bromide and laurtrimonium bromide for Salmonella typhimurium were 4.88 ± 0.08 × 10−6 mol/L and 75.00 ± 3.90 × 10−6 mol/L, respectively. 74 The MICs were 1.65 × 10−4 and 4.86 × 10−4 M, respectively.
Toxicology
Acute Oral Toxicity
Straight and branched chain alkyl trimonium ingredients
In 2 separate studies, the oral LD50 of laurtrimonium chloride in Sprague-Dawley CD rats (n = 5) was 490 mg/kg (confidence interval [CI] 420 to 570 mg/kg) and 560 mg/kg (500 to 630 mg/kg). 70
The original safety assessment reported that the oral LD50 of 40% w/v cetrimonium chloride was 1000 mg/kg for rats. 1
The LD50 for cetrimonium chloride ranged from ∼1550 to 2970 mg/kg for rats in unpublished studies of acute oral toxicity that were reported by the European Union’s Committee on Consumer Products (SCCP). 63 The LD50 for steartrimonium chloride was ∼700 mg/kg. A mixture of cetrimonium and steartrimonium chloride had an LD50 of >2000 mg/kg in rats.
A material safety data sheet (MSDS) reported the LD50 for behentrimonium chloride for rats to be >4 g/kg. No further information was provided. 13
The combined LD50 for tallowtrimonium chloride for Sprague-Dawley CFY rats was 1260 (CI 1061-1496) mg/kg, 1289 (CI 1145-1444) mg/kg for males, and between 1000 and 2000 mg/kg for females. 70 In Swiss-Webster mice, the oral LD50 of cetrimonium chloride was between 400 and 600 mg/kg. 70 The oral LD50 of steartrimonium chloride was 633 mg/kg for male mice and 536 mg/kg for female mice.
Alkanol trimonium ingredients
The oral LD50 was reported to be between 3150 and ≥ 5000 mg/kg for choline chloride in rats. 20 Clinical signs after ingestion were restlessness, increased respiration, hypoactivity, convulsions, ruffled coat, staggered gait, and dyspnea. There was some diarrhea. At necropsy, 3 of 10 rats had inflamed lungs.
The oral LD50 of choline chloride in mice was reported to be in the range of 3900 and 6000 mg/kg. 20
The oral LD50 of choline chloride was 340 mg/kg for ICR mice and >400 mg/kg for Sprague-Dawley rats. 75
The oral LD50 of choline HCl in Swiss CD-1 holoxenic mice was 3900 mg/kg. 76 All animals showed salivation, lacrimation, respiratory depression, and convulsions prior to death. The maximum cholinergic effects were observed within the first hour, and all the animals died during the first 24 hours of observation. The LD0 was 2000 mg/kg.
Polymeric trimonium ingredients
The oral LD50 of polyquaternium-28 for rats was reported to be >5 g/kg. 18
Polyquaternium-47 (<30% aqueous; 5000 mg/kg) orally administered to Wistar rats caused no mortality, clinical signs, or body weight changes. 77
The oral LD50 of polyquaternium-10 was >16 g/kg for rats. 7
Acute Dermal Toxicity
Straight and branched chain alkyl trimonium ingredients
In an acute dermal test of cetrimonium chloride (100%; 4.3 mL/kg) using New Zealand White rabbits (n = 6; 3/sex), 50% of the rabbits died at the only dose level administered. 70 The test substance was applied under occlusion to intact or abraded skin for 24 hours then washed off. All rabbits exhibited normal behavior until day 3 when the rabbits became lethargic, had depressed reflexes, and were cold to the touch. They defecated little or none and had clear fluid coming from their noses and mouths. There was reddening of the nictitating membranes and eyelids. There was substantial weight loss. Skin irritation was noted after 24 hours of exposure, including slight to severe erythema, moderate or severe edema, and whitening of the skin. On day 3, there was moderate or severe atonia and moderate or marked coriaceous skin from day 2. Fissuring was observed in 3 rabbits and desquamation in 1. Necropsy revealed brown, liquid fecal matter; lungs adhered to the chest wall and filled with white granular pockets; enlarged gall bladder; and brownish or clear fluid around the nose and mouth. No visible lesions were observed in the rabbits that survived the 14-day observation period.
Tallowtrimonium chloride (100%; 4.0 mL/kg) applied to the intact and abraded skin of New Zealand White rabbits (3/sex) for 24 hours resulted in 100% mortality between days 3 and 8 of observation. 70 Clinical signs included drooping head and ears, increased respiration rate, increased heart rate, ataxia, depression, excessive salivation, reduced motor reflexes, reduced or lack of feed consumption and defecation. Moderate to severe skin irritation was observed. There was dilation of dermal blood vessels, gastrointestinal tract, and the brain surface. There was enlarged renal blood vessels and posterior vena cava. The pituitary was dark red to purple.
Three of 6 New Zealand White rabbits (3/sex) died during the 24-hour dermal administration of tallowtrimonium chloride (4.7 mg/kg). 70 Two more died during the 14-day observation period. There were no signs of systemic toxicity during exposure. After exposure, the rabbits had depressed reflexes; cold, drooping ears, and intermittent tremors. The 1 surviving rabbit began eating, defecating, and exhibiting normal behavior after 5 days. Slight to moderate erythema, edema, and atonia were observed at the removal of occlusion until death or end of observation period. Necropsy revealed gas-filled, distended large intestines, a thin stomach wall, and red lungs that were adhered to the chest wall.
Polymeric trimonium ingredients
Polyquaternium-10 (4.0 g/kg) was not toxic to rabbits (n = 5). 7 The dermal LD50 of a formulation containing polyquaternium-10 (0.5%) was greater than 2 g/kg on the clipped and abraded skin of rabbits.
Acute Intravenous Toxicity
Straight and branched chain alkyl trimonium ingredients
Intravenous (through tail vein) LD50 values for female SPF-bred NMRI mice and female Sprague-Dawley rats for various alkyltrimethylammonium bromides (C10, 12, 14, 16, and 20) ranged from 2.8 to 20 mg/kg for mice and 5.5 to 44 mg/kg in rats. 78 The LD50 values increased proportionally with the length of the alkyl group. Most animals that died did so within 1 minute from apparent respiratory failure. The rest died within 20 minutes and had tail necrosis.
Alkanol trimonium ingredients
No data were available on alkanol trimonium ingredients themselves. However, data were found for choline HCl. The LD50 of choline HCl in Swiss CD-1 holoxenic mice was 53 mg/kg iv 76 All animals showed salivation, lacrimation, respiratory depression, and convulsions prior to death. The maximum cholinergic effects were observed within the first hour, and all the animals died during the first 24 hours of observation. The LD0 was 21.5 mg/kg iv
Acute Intraperitoneal Toxicity
Alkanol trimonium ingredients
The ip LD50 of choline chloride (50% powder containing 29% colloidal silicic acid and 21% water) was reported to be 225 mg/kg in male and female mice (calculated for pure choline chloride). 64 Mice died within 2 minutes at 1600 mg/kg, at 1 hour at 640 and 800 mg/kg, and at 1 day at 500 mg/kg. Clinical signs were abdominal position, increased respiration rate, convulsions, dyspnea, exophthalmus, and cyanosis immediately after administration. Occasional adhesions in the liver were observed at necropsy.
Male albino rats (n = 14) injected with choline at 45 mg/kg had 29% mortality; guinea pigs (n = 45) had 20% mortality. 79 When injected with 60 mg/kg, rats (n = 20) had 60% mortality and the guinea pigs (n = 39) had 74% mortality. Animals that lived longer than 30 minutes survived in most cases.
Acute Inhalation Toxicity
Polymeric trimonium ingredients
Rats (n = 6) exposed to polyquaternium-10 dust (50 g on a tray stirred up by an intermittent fan) was not toxic over 8 hours. 7
Short-Term Oral Toxicity
Straight and branched chain alkyl trimonium ingredients
Sprague-Dawley CD rats (n = 10/sex plus 5/sex in high dose group for recovery study) were administered cetrimonium chloride (24% to 26%; 0, 30, 100, and 300 mg/kg in distilled water) by gavage 5 days/week for a total of 23 or 24 applications. 63 The recovery group was allowed to recover for 27 days. There were no effects on survival, feed consumption, or body weight. The males in the high-dose group had increased water consumption compared to controls. No treatment effects were observed through ophthalmological and hematological examination. Clinical chemistry parameters were unaffected by treatment except a small increase (but within the range of historical controls) in serum alanine aminotransferase (ALT) in both sexes in the high-dose group.
There was a slight increase in absolute and relative adrenal weights and a decrease in absolute and relative spleen weights in males. Necropsy revealed a thickening of the forestomach mucosa, associated with edema and sporadic ulceration in males and females in the high-dose group. Inflammatory edema in the forestomach mucosa, sporadic ulceration, and acanthosis up to papillomatous hyperplasia were observed in both sexes in the high-dose group at microscopic examination. No histopathological or microscopic alterations were observed in the mid- and low-dose groups. All treatment-related effects were reversed following the recovery period. The authors concluded that the oral no observed adverse effect level (NOAEL) was 100 mg/kg.
Alkanol trimonium ingredients
Choline (200 mg/kg) was orally administered to Balb/c mice of both sexes (n = 10) daily for 28 days. 47 Saline served as the control. No effects to body weights, organ weights, hematological parameters, splenic cell counts, pathology of the organs, and clinical biochemistry were reported.
Wistar rats were administered
Short-Term Dermal Toxicity
Straight and branched chain alkyl trimonium ingredients
A 28-day dermal toxicity test using rabbits of 0.5% cetrimonium chloride dose group showed mild, transient dermal irritation. 81
Cetrimonium chloride (54.5% in aqueous isopropanol) was dermally administered (0 and 0.5%; 0 and 10 mg/kg) to the clipped skin of New Zealand White rabbits (n = 10; 5/sex) for 5 days/week for 4 weeks. 63 Skin was abraded with a clipper head prior to each application. Treated skin was cleaned with water after 6.5 to 7 hours. Two control rabbits died during the study. There were no treatment-related effects to body weight, hematology, organ weight, gross necropsy findings, or histopathology except that treated areas of the skin had mild to marked acanthosis with active mitosis, hyperkeratosis, and necrosis of the epidermis and hair follicles, with some encrustation and exudates. Slight to moderate erythema was observed in all treated rabbits from days 4 to 8; it disappeared in 4 rabbits by day 17. Very slight to slight edema was observed from days 6 to 12 in 4 rabbits; it subsided by day 17. There was intermittent slight edema during week 4 in 2 rabbits and 1 rabbit developed edema on day 20. There was no desquamation or coriaceousness observed. Three rabbits had slight atonia up to week 4. Slight skin fissuring was observed in most of the treated rabbits that typically disappeared by the end of the study. The SCCP concluded that the skin changes were due to local irritation and not evidence of systemic toxicity. The dermal no observed effects level (NOEL) was 10 mg/kg/d.
Polymeric trimonium ingredients
A 21-day dermal toxicity test of a conditioner containing polyquaternium-10 (1%) demonstrated no toxicity to rabbits (n = 10). 7
Short-Term Intraparenteneal Toxicity
Alkanol trimonium ingredients
Choline (200 mg/kg) was administered ip to Balb/c mice of both sexes (n = 10) every other day for 28 days. 47 Saline served as the control. No effects on body weights, organ weights, hematological parameters, splenic cell counts, pathology of the organs, and clinical biochemistry, except for increased creatinine levels, were reported.
Male guinea pigs (n = 10) were administered choline (50 mg/kg/d) ip for 5 days/week for 8 weeks (40 doses). 82 The controls (n = 5) were untreated. They were then killed and necropsied. The treated guinea pigs developed lung lesions consisting of peripheral nodules of small cells, neoplastic bronchiolar epithelium, carcinomatous lesions, and changes in the pleural surface.
Short-Term Inhalation Toxicity
Alkanol trimonium ingredients
Choline (200 mg/kg) was administered by inhalation (described as nasally; no further information was provided) under light anesthesia to Balb/c mice of both sexes (n = 10) every other day for 28 days. 47 Saline served as the control. No effects on body weights, organ weights, hematological parameters, splenic cell counts, pathology of the organs, and clinical biochemistry were observed.
Subchronic Intraperitoneal Toxicity
Alkanol trimonium ingredients
Male albino rats (n = 25) were administered choline chloride (0, 45, 148.5, 225 mg/kg; 0, 0.1, 0.33 or 0.5 × LD50 of 450 mg/kg) ip for up to 8 months. 83 The rats were killed at 1, 3, or 8 months and necropsied. After the injections, the rats were initially excited and active and then became dull and sluggish. Weight gains were similar between groups except for the mid dose at 3 months, which was greater. There was a dose-dependent decrease in relative lung weight at 1 month and an increase in the high-dose group at 3 months. There was a decrease in relative weight of the liver and thymus at 1 month, which, in the thymus, continued to 8 months. In the high-dose group, relative weights of the peripheral lymph nodes were increased. There was a dose-dependent decrease in thymocytes at 8 months. Peripheral lymph nodes had increased cell counts in all doses at 1 month and decreased until 8 months. Regional lymph nodes had reduced cell counts at 8 months in the mid- and high-dose groups. At 3 months in the mid-dose group, cuboidal bronchiolar epithelium, collections of lymphoid cells around blood vessels and bronchiols were observed. The high-dose group had collections of plasma cells and lymphocytes around bronchiovascular structures.
Chronic Oral Toxicity
Straight and branched chain alkyl trimonium ingredients
In the original safety assessment, rats administered cetrimonium bromide at 10, 20, and 45 mg/kg/d in their drinking water for 1 year exhibited decreased body weight gain in the high-dose group. 1
Alkanol trimonium ingredients
Fischer 344 rats (n not provided) were administered choline chloride (500 mg/kg/d) in feed for 72 weeks followed by 30 weeks of observation. 84 Necropsy was performed at 103 weeks. Survival rates, body weights, and relative liver weights were not affected by treatment. The NOAEL was ≥500 mg/kg/d.
Male CD1 mice were fed choline-rich (1.6%; n = 6), choline-deficient (0%; n = 7), or normal choline (0.36%; n = 9) diets for 20 to 24 months. 85 There were no differences in body weight throughout the experiment. There were no differences in mortalities, and these were similar to historical survival for this strain of mice. There were no differences in dendritic spine densities.
CD-1 mice were orally administered choline (0 [n = 23] or 1.6% [n = 17]) in feed for 20 to 24 months. 86 The mice were killed and the brains examined. There were no differences in body weights over the course of the experiment. There were no differences in the concentrations of choline in the striatum, hippocampus, or the cortex between groups.
Baboons (n = 24; Papio hamadryas) were orally administered normal or increased levels of choline (100 or 500 mg/1000 calories) in their feed for 3 to 4 years. 87 Weights were steady or slightly increased throughout the study. Serum transaminases and glutamate dehydrogenase activity were increased in the high-dose group. Bilirubin was increased, albumin was decreased, and total protein was similar in the high-dose group.
Chronic Intraperitoneal Toxicity
Alkanol trimonium ingredients
Male albino rats (n = 10) were administered choline chloride (25 mg in distilled water) ip for 5 d/week. 88 Two of the rats were then killed and the lungs necropsied at 90, 180, and 330 days. Controls (n = 5) were handled differently and administered saline intratracheally. Three rats died in the treatment group, none in the control group. At necropsy, the lungs were light pink in color at 90 and 180 days. At 330 days, small white patches appeared on the lobes and the cut surface also revealed clear white mass. At 90 days, sections of lung showed cuboidal type of bronchiolar epithelium along with prominent musculature of the bronchioles and blood vessels. Heavy collections of lymphoid cells around bronchioles were observed together with dilation of lymphatic vessels. A collection of macrophages with pigments as inclusions were observed at the pleural surfaces. Few giant cells were observed in the parenchyrna along with adenomatoid changes. At 180 days, the lungs had hyperreactive ciliated bronchiolar epithelium and mucus adhering on the top of the epithelial lining. The muscles around bronchioles and blood vessels were hypertrophied and found in patches. Heavy collections of plasma cells and lymphocytes were around bronchioles and larger blood vessels. The alveolar macrophages were very prominent and laden with yellowish black pigment. Sporadically, adenomatoid changes of bronchiolar epithelium were found, which were more prominent at 330 days. Examination of the musculature of the bronchioles revealed prominent eosinophilic characteristics and lumen filled with cell debris with slight thickening of pleura.
Ocular Irritation
In the original safety assessment, cetrimonium chloride was classified as a severe ocular irritant as demonstrated by in vitro and in vitro studies. 1 In the original safety assessment, steartrimonium chloride was found to be severely irritating to the eyes of rabbits and guinea pigs. 1
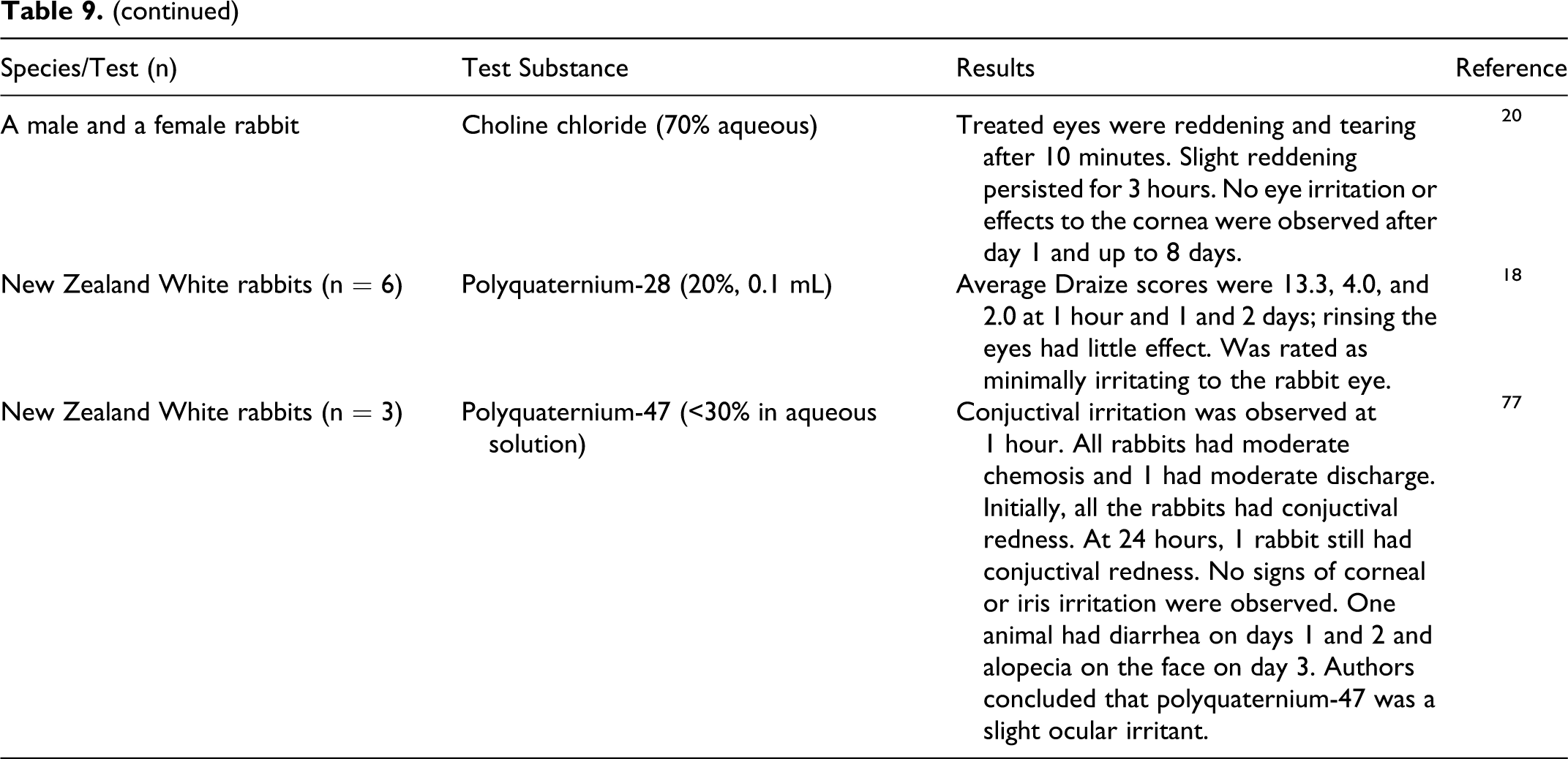
In in vivo tests, cetrimonium bromide (25%) and cetrimonium chloride (10%) were severe ocular irritants that caused irreversible damage in rabbits and rats (Table 9). Steartrimonium chloride was rated an irritant in several studies. In in vitro tests, laurtrimonium bromide, cetrimonium chloride, steartrimonium chloride, and behentrimonium chloride were rated mild to severe ocular irritants. Choline chloride was not an irritant. Polyquaternium-28 was rated a minimal irritant. Choline chloride, carnitine, and carnitine HCl may be ocular irritants as demonstrated by in vitro tests. Polyquaternium-33 was reported to be an ocular irritant and polyquaternium-47 was a slight ocular irritant. Polyquaternium-10 was not an ocular irritant to rabbits up to 5% and produced trace irritation in one eye at 10%.
In Vitro and In Vivo Ocular Irritation Tests.


Dermal Irritation
Straight and branched chain alkyl trimonium ingredients—in vitro
A dermal irritation prediction test of cetrimonium bromide using 3T6 mouse fibroblast cells and NCTC 2544 human keratinocyte cells in neutral red uptake (NRU) assay and 3-(4,5-dimethylthiazol-2-Yl)-2,5-diphenyltetrazolium bromide (MTT) assays was conducted. 89 Cetrimonium bromide had IC50’s of 165.66 ± 19.75 and 102.6 ± 3.96 µg/mL for the 3T6 fibroblasts and 203.23 ± 16.23 and 117.87 ± 13.70 µg/mL for the NCTC 2544 keratinocytes, respectively. The authors concluded that cetrimonium bromide was more irritating than synthetic lysine-derived anionic surfactants.
The relative irritancy potential of cetrimonium bromide (100%) was 518 by the cell suspension agar diffusion test. The relative irritation potential of sodium lauryl sulfate (> 99%) was 995. 90 In the original safety assessment, cetrimonium chloride (100%) was classified as a skin irritant in an in vitro study using rat skin. 1 Behentrimonium chloride mixed with isopropanol (concentration not provided) was reported in an MSDS to be nonirritating in vitro. 12
Straight and branched chain alkyl trimonium ingredients—in vivo
Cetrimonium chloride (as Genamin CTAC; 29%; 0.5 mL) was tested on New Zealand albino rabbits (n = 3) under occlusion for 4 hours. 63 There were no mortalities or systemic effects observed. There were slight erythema and edema at 30 minutes. Grades 2 to 3 erythema and 1 to 2 edema were observed up to 72 hours. Dry and brownish patch skin was observed at 48 and 72 hours and at 7 days. There was hardened skin at 7 days, ablation of large scales at 7 and 14 days, and shiny skin at 14 days. Grade 2 erythema was observed at 7 and 14 days. Skin reactions had resolved at day 21. The authors concluded that the test substance was irritating to skin at 29%.
Cetrimonium chloride (as Quartanim 60W25; 25%; 0.5 mL) was tested on New Zealand albino rabbits (n = 3; male). 63 The test substance was placed on shaved skin under semiocclusive dressing for 4 hours. There were no mortalities or systemic effects observed. Grades 2 and 3 erythema were observed up to 14 days. Grades 1 and 2 edema was observed between 60 minutes and 7 days. At day 14, no edema was observed in 2 rabbits and grade 2 edema was observed in the third rabbit. Skin dryness was observed from 24 hours to 14 days. The authors concluded that cetrimonium chloride was irritating to skin at 25%.
In the original safety assessment, steartrimonium chloride (0.75%) was positive in an in vivo skin irritation study using guinea pigs (n = 20). 1
A single application of steartrimonium chloride (as Genamin STAC; 79.2%; 0.5 g) was tested on New Zealand albino rabbits for 3 minutes (n = 3) and 1 hour (n = 1). 63 The test substance was applied to shaved skin under semiocclusion. There were no mortalities or systemic effects observed. The rabbit exposed for 1 hour had grade 2 erythema up to day 22 and grade 1 edema up to 7 days. At day 22, pink new skin and a scar were noted. No erythema or edema was observed for the rabbits exposed for 3 minutes. The authors concluded that steartrimonium chloride at 79.2% was irritating at 1 hour of exposure but not at 3 minutes.
Steartrimonium chloride and cetrimonium chloride (as Quartamin 86W; 80:20; 28%; 2% and 20%; 0.5 mL) was applied to the shaved skin New Zealand albino rabbits (n = 3) under a semiocclusive patch for 4 hours. 63 There were no mortalities or observed clinical effects. The high-dose group had grade 2 erythema up to 72 hours and followed by crust formation at day 7. Grade 1 edema was observed up to 72 hours. The low dose had grade 1 erythema at 1, 24, and 48 hours in 1 rabbit but no edema was observed. The authors concluded that this mixture of steartrimonium chloride and cetrimonium chloride was irritating to the skin at 20% (4.08% active ingredients) and nonirritating at 2% (0.408% active ingredients).
Behentrimonium chloride (5% in a solution with 0.5% methylcellulose in purified water; 0.5 mL) was applied on the shaved skin of New Zealand albino rabbits (n = 3) under semiocclusion for 3 minutes. 63 There were no mortalities or systemic effects observed. There were no erythema or edema up to 72 hours. The authors concluded that a 5% solution of the test substance was nonirritating after 3 minutes of exposure.
Behentrimonium chloride/cetearyl alcohol mixture (proportion not provided) was reported to be nonirritating to rabbits at 25%. 13 No other information was provided.
Behentrimonium chloride (7.7%-8.3% in 5% methylcellulose in purified water; 0.5 mL) was placed on the shaved skin of New Zealand albino rabbits (n = 3) using a gauze pad for 3 minutes. 91 There were no clinical signs over 5 days. At 1 hour, erythema was observed on all treated rabbits, which resolved by 72 hours. This experiment was repeated. 91 No erythema or edema was observed at any time period up to 72 hours.
Alkanol trimonium ingredients
According to an MSDS, choline chloride may cause irritation with redness and pain. 15 According to an MSDS, carnitine and carnitine HCl may cause skin irritation. 17
Polymeric trimonium ingredients. According to an MSDS, polyquaternium-33 may be a dermal irritant. 92 Polyquaternium-47 (<30% in aqueous solution; 0.5 mL) was applied to 2 separate patch sites of New Zealand White rabbits (n = 6) for 24 hours. 77 There was no edema during the 48-hour observation period. One rabbit had moderate erythema 30 to 60 minutes after patch removal. All treated rabbits had slight erythema under at least one patch during observation. After 24 hours, 4 rabbits had slight erythema. One rabbit had mucoid diarrhea during most of the observation time. All responses were resolved at 72 hours. The authors concluded that Polyquaterium-47 at <30% was a slight dermal irritant.
Polyquaternium-10 (4 g/kg) caused slight erythema to rabbits. 7 In another experiment, there was no irritation at 2% and there was moderate erythema at 5.0% and 10%. A shampoo containing polyquatermiun-10 (0.5%) was a severe skin irritant. A conditioner containing polyquaterium-10 (1%) was not a primary dermal irritant to rabbits on intact and abraded skin.
Dermal Sensitization
Straight and branched chain alkyl trimonium ingredients
A guinea pig maximization test was performed on cetrimonium chloride (as Quartamin 60W25; 25% cetrimonium chloride) using Dunkin-Hartley albino guinea pigs (n = 10/sex; control group n = 5/sex). 63 Induction on day 0 was by intradermal injections of 0.125% test substance in saline and/or Freud’s complete adjuvant and on day 7 at 3% (0.75% active). Challenge was a topical application of the test substance at 0.5% (0.125% active ingredient; 0.5 mL) on day 21 and day 28. There were no deaths or clinical signs in the test group. At the first challenge, 3 males of the treatment group and 1 female in the control group had mild erythema. Three males and 1 female in the control group also had mild erythema on the vehicle-treated side. At 48 hours, no skin reactions were observed. Histopathology on all treated skin showed erythema. Reactivity to the vehicle was similar in both groups. SCCP concluded that the results were unclear.
A Buehler test on cetrimonium chloride (as Genamin CTAC; 30% cetrimonium chloride) using Pirbright white guinea pigs (n = 6) was conducted. 63 Dermal induction was at 4% (1.27% active) in distilled water; the challenge was at 1% (0.3% active ingredient). There was slight to well-defined erythema and very slight edema at the treatment sites during induction. There were no skin reactions in the treatment and controls groups at challenge. The authors concluded that the test substance was not a sensitizer under these conditions.
A guinea pig maximization test was performed using Dunkin Hartley albino guinea pigs (n = 10/sex) to test cetrimonium chloride (as Quartanim 60W25; 24%-26% cetrimonium chloride). 91 Induction was intradermal injections of Qartanim 60W25 (0.125%) and the challenge was topical administration of Quartamin 60W25 (0.5%) on 8 cm2 under occlusion for 24 hours. There were no deaths, clinical signs, or changes in body weights. Skin reactions (erythema) were similar between the treatment and control groups.
A Buehler test was performed on steartrimonium chloride (as Genamin STAC; 79.8% steartrimonium chloride) using Pirbright white guinea pigs (n = 20; n = 10 in control group). 63 Induction was at 4% (3.192% active) in ethanol:water (80:20) under an occlusive patch on clipped skin for 6 hours. Induction was repeated on days 8 and 15. The challenge was on day 29 at 1% (0.798%) in isopropanol (instead of ethanol:water) on a naive site. The patch was left on for 6 hours. No clinical signs were observed. During induction, the treated group had slight, well-defined to severe erythema and very slight to well-defined edema at the treatment area. No skin reactions were observed after the challenge. The authors concluded that the test substance was not a sensitizer under these test conditions.
A guinea pig maximization test using Dunkin-Hartley guinea pigs (n = 10; n = 5 in control group) was performed on steartrimonium chloride: cetrimonium chloride (80:20; as Quartamin 86W; 28% active [20.47%]) in distilled water or Freund complete adjuvant (FCA; 50%) and water. 63 Induction was 0.1% v/v (0.0204%) by intradermal injection then a topical patch at 5% (1.20%) on shaved skin for 48 hours. Challenge was at day 21 with a patch at 10% (2.4%) on the right shoulder and 5% (1.2%) on the left for 24 hours. There was well-defined or moderate to severe erythema at the induction sites in the test group at 24 and 48 hours. Slight erythema was noted at the induction sites of the control group at 24 hours and at 48 hours in 1 guinea pig. After topical induction, slight to well-defined erythema was noted at 24 hours in the test group; no reaction was noted in the control group. No skin reactions were observed at the challenge sites. The authors concluded that the test substance was not a sensitizer under these conditions.
Hartley guinea pigs (n = 10/sex in treated group; n = 5/sex in control group) were treated with a 10% solution of behentrimonium chloride (vehicle not stated) on a 8-cm2 filter paper patch applied to clipped skin on the left flank for 6 hours, with occlusion on days 1, 8, and 15. 63 During the induction phase, the skin sites were examined for local effects 24 hours after each treatment. The challenge exposure consisted of a 0.5% solution of the test substance loaded onto a Finn chamber and applied to clipped skin on the right flank for 6 hours, with occlusion on day 29. A second challenge was performed on day 43 due to equivocal cutaneous reactions from the first challenge. One male animal from the treated group died on day 14, but this was considered by the investigators to be unrelated to treatment. A few treated animals (number not specified) showed slight to well-defined erythema during the induction phase. After the challenge, grade 1 erythema was observed in 3 of 19 animals at 24 hours and 5 of 19 animals at 48 hours. A grade 1 erythema was observed in 1 of 19 and a grade 2 erythema in 2 of 19 animals at 72 hours. No reactions were observed in control animals, after the challenge. The authors concluded that behentrimonium chloride was a sensitizer at 10%.
A Buehler test for sensitization was performed on behentrimonium chloride (as Genamin KDMP; 77% to 83% diluted to a 20% solution in ethanol:water [80:20]; 16%). 63 Female Pirbright white guinea pigs (n = 20, control n = 10) were treated with a 20% solution of the test substance on a 2 × 2 cm cellulose patch, applied to clipped skin on the left flank for 6 hours, with occlusion on days 1, 8, and 15. During the induction phase, the skin sites were examined for local effects 24 hours after each treatment. The challenge exposure consisted of a 0.8% solution of the test substance in isopropanol on a 2 × 2 cm occluded patch, applied to clipped skin on the right flank for 6 hours, on day 29. Skin reactions were scored 24 and 48 hours after patch removal. Animals were observed daily for signs of systemic toxicity and body weights were recorded on days 1 and 31. Treated animals showed slight to well-defined erythema and very slight edema at the site of treatment during the induction phase. No skin reactions were observed in the treated or control animals after challenge.
In a Buehler test using Dunkin Hartley guinea pigs (n = 10/sex), behentrimonium chloride (as Genamin KDMP; 77%-83%) was used at induction at 10% Genamin KDMP (7.7%-8.3%) and challenge at 0.5% (3.5%-4.25%) in Finn chambers. 91 No clinical signs were observed and weights were similar between groups. There was some well-defined erythema in some of the treatment groups. There were 3 reactions observed after challenge and no reactions at rechallenge.
Reproductive and Developmental Toxicity
Straight and Branched Chain Alkyl Trimonium Ingredients
In a range-finding study of laurtrimonium chloride (20, 50, 100, 200, 400 mg/kg/d), pregnant New Zealand White rabbits (n = 3) were orally administered the test substance from day 6 through 18 of pregnancy. 70 At 25 and 50 mg/kg, 1 in 3 rabbits died; at 100 mg/kg, 2 rabbits died; and at 400 mg/kg, all 3 rabbits died. There were embryonic effects (not defined) observed at 50 mg/kg.
In the main study, laurtrimonium chloride (2, 8, 24 mg/kg) was orally administered to pregnant New Zealand White rabbits (n = 13-14) from day 6 to 18 of pregnancy. The dams were killed on day 19 and necropsied. There were no adverse effects reported for the dams and no developmental or teratogenic effects observed.
The original safety assessment reported that cetrimonium bromide (25 mg/kg/d) administered on days 5 to 14 of gestation was not teratogenic in an oral study using rats. 1 Mild embryonic effects were observed at 50 mg/kg/d, but these were attributed to maternal toxicity rather than a teratogenic effect. There were no embryotoxic or teratogenic effects at lower doses. In an intraperitoneal study, cetrimonium bromide interfered with the embryonic development of mice at 10 mg/kg on day 8, 10, 12, or 14 of gestation and was lethal to developing embryos at 35.0 mg/kg. Teratogenic effects were observed in both treatment groups. There was no evidence of teratogenicity by 2.0% cetrimonium chloride administered days 7 to 18 of gestation in a dermal study using rabbits. The only adverse effect observed was dermal irritation at the application sites. When tested in a dermal teratogenicity study, 2.5% steartrimonium chloride administered on days 6 to 15 of gestation was not maternally toxic, embryotoxic, or teratogenic.
Alkanol Trimonium Ingredients
Male rats were administered choline chloride (80 mg/kg/d) ip for 12 or 24 days. 93 Another group was administered choline chloride (10 to 12 mg/kg/d) in feed. The rats were necropsied at 2, 5, 8, and 12 days after the treatment period. There were no effects on body weight gain or weights of testes, epididymides, liver, kidney, and adrenals. After 12 days of treatment (one cycle of the seminiferous epithelium), epithelial vacuoles, spermatogonia with pyknotic nuclei and cellular debris were observed 2 days after the termination of treatment. Five days after termination of treatment, the seminiferous tubules were normal. After 24 days of treatment, a few tubules of stages I to IV were observed on day 2 after treatment termination. Most spermatocytes were normal with some necrotic pachytene stages with an essential restoration to normal after 12 days.
Male rats (n = 25; strain not provided) were administered choline (25 mg/kg/d) ip for 12 or 24 days. 93 At 12 days, spermatogenesis was not changed. At 24 days, pachytene spermatocytes were decreased until day 5 posttreatment. Slight proliferation of spermatogonia was observed from day 5 posttreatment onward. By day 12 posttreatment, tubules showed almost normal cellular associations. The authors suggest that prolonged administration of excess choline may be toxic to male reproduction.
Pregnant mice (n = 7 – 16) were administered choline chloride (1250 to 20,000 mg/kg/d) in feed on gestations days 1 to 18. 20 There were no untreated controls. All groups except the lowest dose group had reduced maternal body weight gain. All fetuses were resorbed in the highest dose group but no resorptions were observed in the lowest dose group. At 4160 and 10 800 mg/kg/d, there was 35% and 69% embryonic/fetal lethality. Developmental toxicity was observed in all but the lowest dose group. There were no increases in malformations observed. An NOAEL was not determined for teratogenicity due to the lack of pups.
Genotoxicity
Straight and Branched Chain Alkyl Trimonium Ingredients
In a reverse mutation assay using S typhimurium, laurtrimonium chloride (0.004 to 0.4 µl/plate) was not cytotoxic or genotoxic, with or without metabolic activation. 70 In a forward mutation assay using L5178Y/TK+/− mouse lymphoma cells, laurtrimonium chloride (0.0038-0.050 µl/mL without metabolic activation and 0.012-0.16 µl/mL with metabolic activation) was not mutagenic. The results were negative in an unscheduled DNA synthesis assay of laurtrimonium chloride (0.004-0.1 µl/mL) using rat primary hepatocytes. In a bone marrow cytogentic assay, laurtrimonium chloride (16, 53.3, and 160 mg/kg) orally administered to male and female Sprague-Dawley rats for 5 days did not induce an increase in chromosomal aberrations.
In the original safety assessment, cetrimonium chloride was negative both with and without metabolic activation in an bacterial test up to 625 µg/plate, in forward-mutation and reverse-mutation tests at concentrations up to 50 µg/mL, and in a cell transformation assay up to 1.0 µg/mL. 1 Negative results were also obtained in a chromosome aberration test at concentrations up to 3.0 µg/mL, without metabolic activation and 10.0 µg/mL in tests with exogenous metabolic activation. Cetrimonium chloride (62.5 µg/plate) and cetrimonium bromide (no concentration provided) were both negative in bacterial tests. Cetrimonium chloride (1.0 µg/mL) was also negative in an in vitro cell transformation assay.
Cetrimonium chloride and steartrimonium chloride were not mutagenic in a reverse mutation assay using S typhimurium (TA98 and TA100). 70
In an in vitro chromosome aberration test, V79 Chinese hamster cells were incubated with cetrimonium chloride (24%-26% cetrimonium chloride; 0.1-6.0 µg/mL without metabolic activation; 0.1-10.0 µg/mL with metabolic activation). 63 There were no increases in the number of cells with structural aberrations at any concentration with or without metabolic activation.
Salmonella typhimurium (TA98, TA100, TA1535, T1537) were incubated in steartrimonium chloride (as Genamin STAC; 79.8%; 4, 20, 100, 500, 2500, or 5000 µg/plate in ethanol) with and without metabolic activation. 63 This was repeated with lower concentrations of test material (0.8, 4, 20, 100, 500, and 2500 µg/plate). The test compound was toxic at 100 µg/plate, with and without metabolic activation. There was increase in the number of revertant colonies compared to controls with or without metabolic activation.
Salmonella typhimurium (TA98, TA100, TA1535, TA1537, TA1538) were incubated in steartrimonium chloride:cetrimonium chloride (80:20; as Quartamin 86 W; 28%; 0.15 to 50 µg/plate in acetone with metabolic activation; 1.5 to 500 µg/plate without metabolic activation). 63 The experiment was repeated with lower concentrations of test material (0.5 to 50 µg and 0.5 to 150 µg, respectively). There were incomplete bacterial lawns at 15 µg/plate, without activation, and 150 µg with activation. The test substance was toxic at 50 µg/plate, with and without activation. The test substance did not increase the number of revertant colonies compared to controls.
Behentrimonium chloride (as Genamin KDMP; 77% to 83%) was found not to be mutagenic, with or without metabolic activation, in a bacterial assay when tested at concentrations of 4 to 5000 µg active substance/plate in S typhimurium (TA98, TA100, TA1535, and TA1537). 63 Cytotoxicity occurred at 500 µg/plate without metabolic activation and at 2500 µg/plate with metabolic activation.
Tallowtrimonium chloride was not mutagenic in a reverse mutation assay using S typhimurium (TA98, TA100, TA1535, TA1537, and TA1538), with or without metabolic activation. 70 In another reverse mutation assay using S typhimurium (TA98, TA100, TA1535, TA1537, and TA1538), tallowtrimonium chloride was cytotoxic at 500 µg/plate and was genotoxic at 50 µg/plate with TA1538, with or without metabolic activation. The results were negative for the other strains. The study authors suggest that the positive result may be due to impurities.
Alkanol Trimonium Ingredients
Choline chloride did not produce any gene mutations, clastogenicity, or DNA damage when tested in vitro in studies reported by the United Nations Environment Programme (UNEP; Table 10). 20
Genotoxicity Tests of Choline Chloride. 20
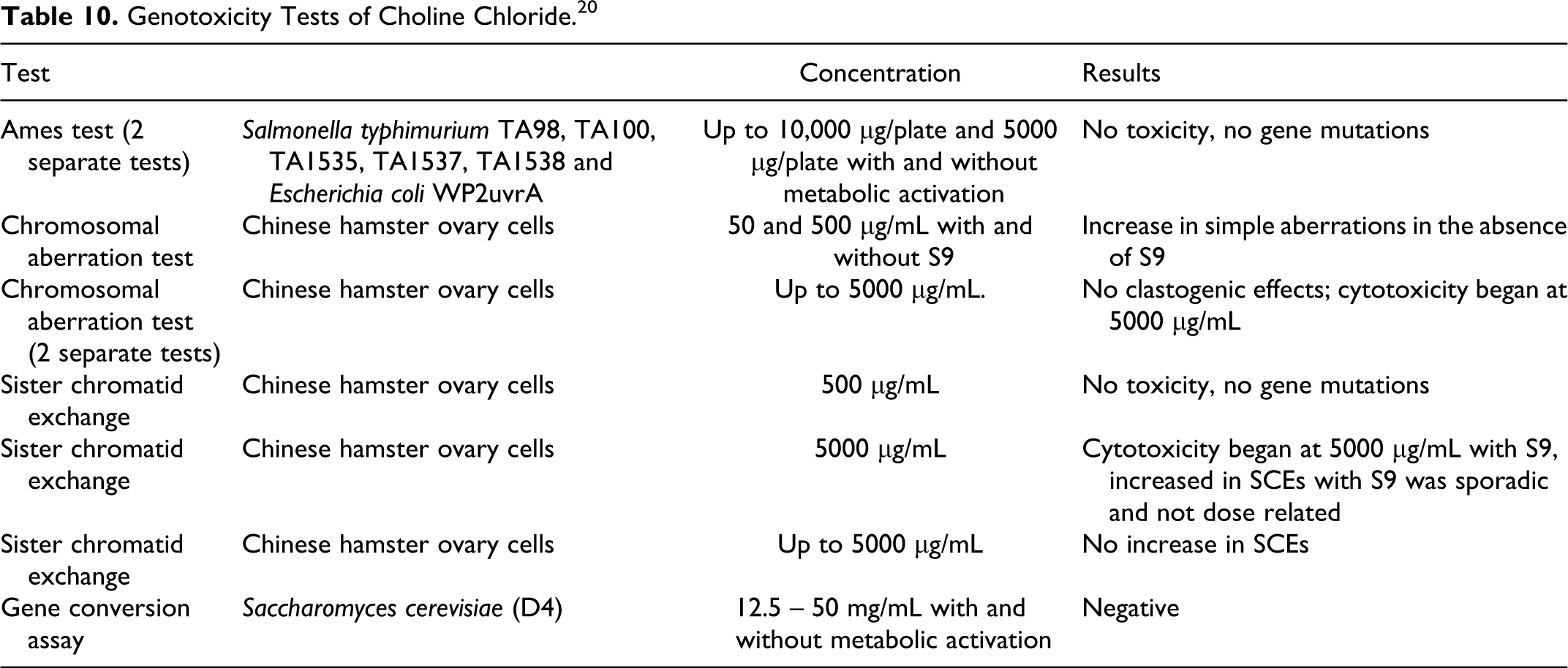
Polymeric Trimonium Ingredients
Polyquaternium-28 (20%) was negative in a reverse mutation assay using S typhimurium (TA1535, TA1538, TA98, and TA100), with or without metabolic activation. 18 In a micronucleus assay using the bone marrow cells of mice, polyquaternium-28 did not induce an increase in bone marrow polychromatic erythrocytes.
Polyquaternium-47 (< 30% in aqueous solution) was negative in a reverse mutation assay using S typhimurium (TA 1535, TA1537, TA100, TA98) and E coli (WP2 uvrA) with or without metabolic activation up to 5000 µg/plate. 77
Polyquaternium-10 was not mutagentic in Ames test using S typhimurium (TA98, TA100, TA1535, Ta1537, TA1538) with or without metabolic activation. 7 It was also not mutagenic to Chinese hamster ovary cells up to 0.285% with or without metabolic activation. There was no increase in micronucleated polychromatophilic erythrocytes when Swiss mice were treated with polyquaternium-10 up to 0.4 g/kg. 7
Carcinogenicity
Alkanol Trimonium Ingredients
Fischer 344 rats (n not provided) were administered choline chloride (500 mg/kg/d) in feed for 72 weeks followed by 30 weeks of observation. 84 Necropsy was performed at 103 weeks. There was no increase in the number of liver nodules, hepatocellular carcinomas, lung tumors, leukemia, or other tumors in the treated rats compared to controls.
Clinical Assessment of Safety
Absorption, Distribution, Metabolism, and Excretion
Alkanol Trimonium Ingredients
Patients with dyskinesia and cerebellar ataxia were orally treated with choline chloride (150 and 220 mg/kg/d [10 and 16 g/d, respectively] for 2-6 weeks). 94 The patients developed fishy body odor, vomiting, salivation, sweating, and gastrointestinal effects.44,95
Fasting levels of choline in the plasma were reported to be around 10 µmol/L, ranging from 9 to 20 µmol/L. 96 A fishy odor from ingested choline has been reported, possibly due to excessive amounts of trimethylamine, a metabolite produced by bacteriological action and formation of methylamines.44,95
Total body carnitine was reported to be mostly contained in skeletal muscle carnitine (∼20 g of carnitine in a 70-kg man, of which more than 19 g was reported to be in skeletal muscle). 97 The bioavailability of orally administered carnitine was reported to be ∼16% to 18% at doses of 1 to 2 g and may be even lower at higher doses.98,99 Once synthesized or absorbed, carnitine was reported to be eliminated from the body only via the urine as carnitine or acylcarnitines. Renal tubules contain a saturable carnitine transport system that conserves most filtered carnitine but leads to large carnitine losses if the plasma concentration exceeds 60 to 90 μmol/L. Carnitine was reported to be absorbed at ∼25% when consumed orally. 100
Suspected and known coronary artery disease patients had blood analyzed after an overnight fast. 101 The average free carnitine was 41.9 ± 8.9 µmol/L; short-chain acylcarnitine, 10.5 ± 5.3 µmol/L; total acid-soluble carnitine, 52.4 ± 9.4 µmol/L, long-chain acylcarnitine, 2.8 ± .7 µmol/L, and total carnitine, 55.2 ± 9.9 µmol/L.
Participants (n = 8) were orally administered acetyl-
Clinical Testing
Alkanol trimonium ingredients
Participants with hyperthyroidism-related symptoms (n = 5) were orally administered
Participants (n = 7) with dementia of the Alzheimer type were orally administered acetyl-
Patients with Alzheimer (n = 7) were orally administered choline chloride (10 g/d, 7.5 g of choline). 105 The patients developed nausea, diarrhea, and slight hypotension.
Participants, with and without cirrhosis, have been orally administered choline (6 g/d; form not stated) for 4 weeks with no liver toxicity. 106 Oral ingestion of choline (20 g/d) for 3 to 4 weeks have been associated with depression. 107 Mild, transient signs of Parkinson disease have been observed in patients with tardive dyskinesia after oral administration of choline (12.7 g/d). 108 Patients with tardive dyskinesia and Huntington disease who were orally administered choline (20 g/d) for 4 weeks had no adverse effects. 107
The critical adverse effect from high intake of choline was reported to be hypotension, with corroborative evidence on cholinergic side effects (eg, sweating and diarrhea) and fishy body odor. The Tolerable Upper Intake Level (UL) for adults was reported to be 3.5 g/d.
Dermal Irritation
Straight and branched chain alkyl trimonium ingredients
Laurtrimonium bromide (7.5%) was applied to the volar surface of the arms of 11 white, healthy participants for 20 minutes for 8 consecutive days (excluding weekends, days 5 and 6). 109 An untreated site and a site treated with water served as controls. The amount of irritation increased with time. The transepidermal water loss (TEWL) measurement did not decrease after the weekend break and did not return to baseline for 20 days after treatment ended. Erythema was still present at 23 days.
In the original safety assessment, slight dermal irritation was observed during the induction phases of multiple human repeated insult patch tests (HRIPT) experiments of cetrimonium chloride (0.25%). 1
Several studies of dermal irritation of products containing cetrimonium chloride, behentrimonium chloride, and cocotrimonium methosulfate are reported in Table 11. Cetrimonium chloride was not an irritant up to 1% and mixed results were reported above 1%. Behentrimonium chloride was not an irritant at 5.0% and cocotrimonium methosulfate at 0.42%.
Human Dermal Irritation Studies of Cetrimonium Chloride.
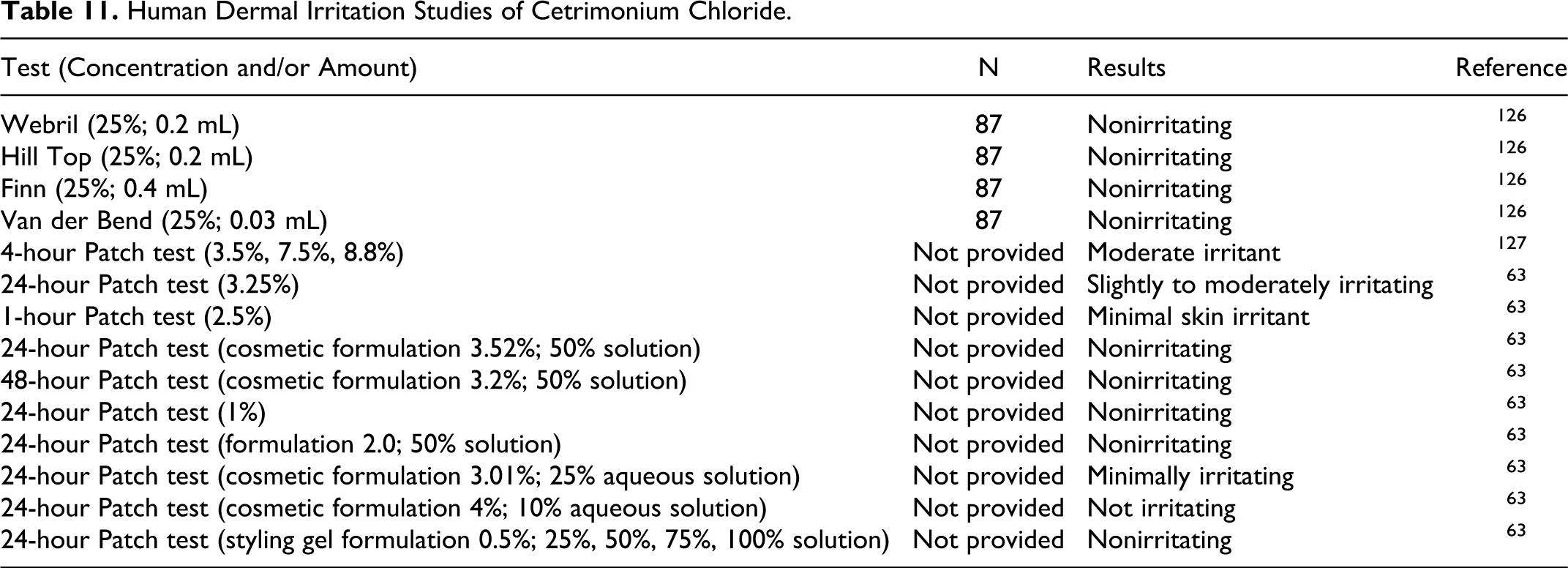
The irritancy potential of a formulation containing behentrimonium chloride (5.0%, vehicle not provided) was tested in participants (n = 51; 5 male and 46 female participants) using a Finn chamber applied to the participants’ backs, with occlusion, for 24 hours. 63 Deionized water was used as the negative control substance. No difference in irritant reactions between treatment and control sites was reported.
Alkanol trimonium ingredients
Bar soap and liquid body soap, with and without choline chloride (0.5%), were used by people (n = 25) with self-perceived sensitivity to choline chloride (0.5%) for 21 days. 20 Compared to controls, there was no difference in cumulative irritancy.
Polymeric trimonium ingredients
Polyquaternium-10 at 5% in water was not a dermal irritant to participants for 21 days of application. 7
Dermal Sensitization
Straight and branched chain alkyl trimonium ingredients. In the original safety assessment, cetrimonium chloride was negative in 4 human repeated insult patch tests (HRIPT; n = 101-5202) at concentrations up to 0.25% for 100% active solutions and up to 0.4% for 25% active solutions. 1 HRIPTs of cetrimonium chloride and behentrimonium chloride are presented in Table 12. Cetrimonium chloride at 0.25% and behentrimonium chloride at 2.4% were not sensitizing in humans.
HRIPTs of Cetrimonium Chloride, Behentrimonium Chloride, Choline Chloride, and Polyquaternium-28.

Alkanol trimonium ingredients
An HRIPT of choline chloride showed that it was not sensitizing at 0.5% (Table 12).
Polymeric trimonium ingredients
HIRPTs of polyquaternium-28 and polyquaternium-47 are presented in Table 12. Polyquaternium-28 and polyquaternium-47 were not sensitizing at 5%. Polyquaternium-10 was not sensitizing at up to 2%. 7
Phototoxicity
Polymeric trimonium ingredients
Polyquaternium-28 (2%; 0.2 mL; vehicle not provided) was placed on patches and applied to the volar surfaces of the arms of participants (n = 10). 18 After 24 hours, 1 arm was exposed to UV-A for 15 minutes. The unirradiated arm served as the control. There were no reactions observed at 24 and 48 hours.
Photoallergy
Polymeric trimonium ingredients
Polyquaternium-28 (5%; 0.2 mL; vehicle not provided) was placed on patches and applied to the volar surfaces of the arms of participants (n = 28) 6 times over 3 weeks. 18 One arm was exposed to UV-A (3.33 j/cm2) and UV-B (90-126 mj) irradiation for 75 to 105seconds (depending on skin type) 24 hours after each application of the test substance. The unirradiated patches and untreated areas served as the controls. Two weeks after the completion of induction phase, a single application of the test substance (2%, 0.2 mL) followed by irradiation was administered to a naive site.
Minimal erythema or erythema and/or slight edema were observed in 26 of the irradiated sites during induction which was similar to untreated irradiated sites in 6 participants. In the challenge phase, minimal erythema was observed in 4 participants on treated irradiated sites and in 1 participant on a treated, nonirradiated site. No reactions were observed on untreated irradiated sites. Overall, slight irritation was observed on treated skin exposed to UV-A and UV-B irradiation, but polyquaternium-28 did not induce contact photoallergy in humans under these conditions.
Case Reports
One woman who had acute contact dermatitis and worked in a garden center patch tested positive for choline chloride (1% in water and petrolatum). 110 Ten control patients were negative.
Summary
The safety assessment for cetrimonium chloride, cetrimonium bromide and steartrimonium chloride was reopened to add structurally similar ingredients that include:
straight and branched chain alkyl trimonium ingredients,
alkanol trimonium ingredients, and
polymeric trimonium ingredients.
Many of these ingredients vary only by the nontrimonium hydrocarbon chain length. Others also vary by branching and further functionalization by alcohols, esters, ethers, and polymers. It is expected that the toxicity of alkanol trimonium compounds would be similar to that of alkyl trimonium compounds and the available data may be extrapolated to the other ingredients in this safety assessment—a process usually termed read-across. To test the read-across reliability, similar toxicity end points were considered across the fourth substituent. Where data were available, the findings were similar.
Straight and branched chain alkyl trimonium ingredients in this safety assessment consist of cations each comprising a nitrogen atom bonded to 3 methyl groups and a simple alkyl chain, which can vary in length from 12 carbons to 28 carbons in length. These straight chain trimoniums are waxy solids at human physiological temperatures. Water solubility and volatility decrease and melting/boiling points increase as chain length increases. Cetrimonium bromide and myrtrimonium bromide are reported to be 99% pure.
The alkanol trimonium ingredients in this safety assessment differ from the alkyl trimonium ingredients by the addition of an ethoxy functional group attached to the trimonium core nitrogen. The simplest of these, and a major metabolite of all of the other members in the group, is choline.
The third group of trimoniums in this safety assessment differs from the other groups by their incorporation into a polymer backbone. Typical molecular weights of these polymers are usually above 400 000 g/mol. The residual monomer of polyquaternium-28 was <1%.
Trimoniums function mostly as hair-conditioning agents, antistatic agents, and surfactants. Several of these ingredients function as cosmetic biocides. Impurities for alkanol trimonium compounds include trimethylamine, ethylene glycol, organic impurities, color, heavy metals. The maximum content of the residual monomer for polyquaternium-28 was reported to be <1%.
The straight chain alkyl trimonium ingredients are reported to be used at 0.0005% to 10% in rinse-off products and 0.001% to 4% in leave-on products. The alkanol trimonium ingredients and related ethers/esters/acids are mostly used in skin care and makeup products. Dihydropropyltrimonium chloride was reported to be used in leave-on products at 0.05%. Choline chloride is considered GRAS as a nutrient by the FDA.
Straight and Branched Chain Alkyl Trimonium Ingredients
Orally administered cetrimonium bromide was poorly absorbed by the intestinal tract and was rapidly excreted in the urine and feces. Cetrimonium bromide and cetrimonium chloride did not penetrate the skin well. Cetrimonium bromide enhanced penetration (phenylazoaniline, benzocaine, and benzoic acid) at concentrations below 0.5% and inhibited penetration above 1.0% in artificial membranes. The oral LD50 of straight and branched chain alkyl trimoniums ranged from 490 to 2970 mg/kg for rats and 400 to 633 mg/kg for mice.
Tallowtrimonium chloride (4.0 mL/kg) applied to the intact and abraded skin of rabbits resulted in 100% mortality.
The short-term oral NOAEL for 5 weeks was 100 mg/kg for cetrimonium chloride for rats. Cetrimonium chloride at 0.5% produced mild, transient dermal irritation in rabbits. The short-term dermal NOEL for cetrimonium chloride was 10 mg/kg/d for rabbits. Chronic oral administration of cetrimonium bromide at 10 mg/kg/d for 72 weeks resulted in decreased body weights in rats.
Cetrimonium bromide and cetrimonium chloride were severe ocular irritants that caused irreversible damage at 25% and 10%, respectively, in in vitro studies. Steartrimonium chloride was rated an ocular irritant in several studies. Behentrimonium chloride caused irreversible ocular damage at 10%. In in vitro tests, laurtrimonium bromide, cetrimonium chloride, steartrimonium chloride, and behentrimonium chloride were rated mild to severe ocular irritants.
Cetrimonium chloride was reported to be a skin irritant at 25%, steartrimonium chloride at 20%, and behentrimonium was not an irritant at 25% in animals. Laurtrimonium bromide was dermally irritating in humans at 7.5%. Cetrimonium bromide was not an irritant at 1%. Behentrimonium chloride was not irritating at 5.0%. Cetrimonium chloride at 3% and steartrimonium chloride at 3.192% were not sensitizers in guinea pigs. Behentrimonium chloride was a sensitizer at 16% in guinea pigs. Cetrimonium chloride at 0.25% and behentrimonium chloride at 2.4% were not sensitizing in humans. Laurtrimonium chloride (0.4 µg/plate), cetrimonium chloride (625 µg/plate), steartrimonium chloride (5000 µg/plate), and behentrimonium chloride (500 µg/plate) were not found to be genotoxic in bacterial tests. Tallowtrimonium chloride was genotoxic at 50 µg/plate in 1 S typhimurium strain. Laurtrimonium chloride and cetrimonium bromide produced embryonic effects at 50 mg/kg in rabbits when administered orally. There was no evidence of teratogenicity by 2.0% cetrimonium chloride in a dermal study using rabbits.
Alkanol Trimonium Ingredients
Orally administered choline was poorly absorbed by the intestinal tract and was recovered in the feces. Acetyl
After carnitine was injected into pregnant mice; of the amount that remained in the bodies, the highest concentrations of carnitine were in the liver, placenta, kidney, myocardium, and choroid plexus in the dam. Carnitine crossed the placental barrier and was present in the same proportions, but lower concentrations, in the fetuses.
Palmitoyl carnitine enhanced dermal penetration of Lucifer yellow and Ruthenium red across caco-2 monolayers; acetyl carnitine (with a shorter alkyl chain) did not.
Oral LD50 was reported to be between 3150 and ≥5000 mg/kg for choline chloride in rats and 3900 and 6000 mg/kg in mice.
No effects were observed in mice orally administered choline at 200 mg/kg/d for 28 days and rats administered 1.2 mmol/kg/k carnitine for 7 days.
Short-term ip administration of choline at 200 mg/kg for 28 days to mice had no effects on body weights, organ weights, hematological parameters, splenic cell counts, pathology of the organs, and clinical biochemistry. Short-term administration ip of choline at 50 mg/kg/d to guinea pigs for 8 weeks resulted in lung lesions.
Short-term nasally administered choline at 200 mg/kg to Balb/c mice for 10 days had no effects on body weights, organ weights, hematological parameters, splenic cell counts, pathology of the organs, and clinical biochemistry. Subchronic ip administration of choline chloride to rats resulted in changes in the relative weights and/or cells of the liver, bronchiolar epithelium, thymus, and peripheral lymph nodes.
Chronic oral administration of choline had no adverse effects on rats (72 weeks), mice (20-24 months), and baboons (3-4 years). Chronic ip administration of choline to rats at 25 mg/d for 5 days/week for 10 weeks resulted in adverse effects to the lungs.
Choline chloride was not a dermal irritant to rabbits at 70%. Choline chloride was not sensitizing at 0.5% in humans. Choline chloride, carnitine, and carnitine HCl may be ocular irritants. Choline chloride was not found to be genotoxic. There was no increase in the number of liver nodules, hepatocellular carcinomas, lung tumors, leukemia, or other tumors in rats treated with 500 mg/kg/d of choline chloride.
Choline at 25 mg/kg/d ip was toxic to male rat reproduction after 24 days. In mice, there was no developmental toxicity at 1250 mg/kg. There were maternal and developmentat toxicity above this dose.
In humans, choline levels in plasma range from 9 to 20 µmol/L. Carnitine is only eliminated from the body via the urine as carnitine or acylcarnitines.
Carnitine at 2 mg/kg/d and acetyl-
Polymeric Trimonium Ingredients
The oral LD50 of quaternium-28 and quaternium-47 for rats was reported to be >5 g/kg.
In rabbits, polyquaternium-28 was rated a minimal ocular irritant at 20% and polyquaternium-47 was a slight ocular irritant. Polyquaternium-33 was listed as an ocular irritant.
Polyquaternium-47 was a slight dermal irritant at <30% in rabbits. Polyquaternium-28 and polyquaternium-47 were not sensitizing at 5% in humans.
Polyquaternium-28 and polyquaternium-47 were not genotoxic in reverse mutation assays. Polyquaternium-28 was not found to be genotoxic.
Polyquaternium-28 was not phototoxic or photoallergic in humans.
Discussion
The Expert Panel noted gaps in the available safety data for some of the trimoniums in this safety assessment. The available data on many of the trimoniums are sufficient, however, and similarity between structural activity relationships, biologic functions, and cosmetic product usage suggests that the available data may be extrapolated to support the safety of the entire group.
For example, these trimoniums vary by chain length; presence of branching; further functionalization by an alcohol, ester, or ether; or polymerization. Where available, the information for multiple ingredients is consistent; for example the smallest of the alkyl substituted ingredients, laurtrimonium bromide, has been shown to not penetrate skin after 2 days of application. The larger trimonium ingredients have similar results, suggesting that the various length ingredients in this assessment will also not penetrate the skin. The Panel noted that despite the structural diversity in the ingredients group, the dominant feature of the group is its trimonium moiety. The similarity in their physical properties and uses justify their consideration together in this assessment.
The Expert Panel noted that most of genotoxicity data were on choline chloride and data on the other ingredients was lacking. The lack of dermal penetration, combined with the negative results of the bacterial genotoxicity assays, supports the absence of any genotoxic risk from the use of these ingredients in cosmetics.
The Expert Panel noted the new uses of trimoniums in baby products. These new uses are reported to be at very low concentrations (<1%) and are at a level lower than that which caused irritation. Coupled with the lack of dermal penetration, the low concentration of use levels provides assurance that the use of these ingredients in baby products would not present any risk of systemic or dermal adverse effects. It was also noted that the literature lacks clinical reports of irritation and/or sensitization by the trimonium compounds, including the ingredients that are used in hundreds of products (ie, cetrimonium chloride and behentrimonium chloride).
Ocular irritation was moderate at 2.0% for cetrimonium chloride and mild for behentrimonium chloride at 3.0%. All the uses of eye-related products that contain trimonium ingredients were reported to be at 0.3% or less, well below these concentrations. The other straight and branched chain alkyl trimonium ingredients for which there is data (laurtrimonium chloride, steartrimonium chloride, behentrimonium chloride) were irritating at higher concentrations; choline chloride was minimally irritating at 70%, and polyquaternium-28 was minimally irritating at 20%. While there are uses in lipstick and eye care that would involve exposure of mucous membranes, the concentrations of trimonium in such products is very low (< 1%). Adequate data available to support that such levels in these product types would not result in toxic systematic exposure.
The Expert Panel recognized that these ingredients (ie, cetrimonium bromide and palmitoyl carnitine) can enhance the penetration of other ingredients through the skin (eg, phenylazoaniline, benzocaine, and benzoic acid). The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their low dermal absorption, or where dermal absorption was a concern.
The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration, the duration of the exposure, and the site of deposition within the respiratory system. In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. Particles with an aerodynamic diameter of ≤10 µm are respirable. In the absence of inhalation toxicity data, the Panel determined that trimoniums can be used safely in hair sprays, because aerosol particle size is not respirable.
The Expert Panel was not concerned about the plant and animal derived components of ingredients in this group. The extensive amount of processing to extract the alcohol moieties removed any concern about residual pesticides, heavy metals, or prions.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment (ingredients not in current use identified with an *), when formulated to be nonirritating:
laurtrimonium bromide*
laurtrimonium chloride
myrtrimonium bromide
cetrimonium chloride
cetrimonium bromide
cetrimonium methosulfate
steartrimonium bromide*
steartrimonium chloride
steartrimonium methosulfate*
behentrimonium chloride
behentrimonium methosulfate
octacosatrimonium chloride*
ceteartrimonium chloride*
hydrogenated tallowtrimonium chloride*
hydrogenated palmtrimonium chloride*
soytrimonium chloride
tallowtrimonium chloride
cocotrimonium chloride
cocotrimonium methosulfate
octyldodecyltrimonium chloride*
dodecylhexadecyl-trimonium chloride*
choline chloride*
stearoxypropyltrimonium chloride*
lauroyl ethyltrimonium methosulfate*
myristoyl ethyltrimonium methosulfate*
palmitoyl ethyltrimonium methosulfate*
stearoyl ethyltrimonium methosulfate*
cocoylcholine methosulfate*
carnitine
carnitine HCl
carnitine hydroxycitrate
carnitine PCA*
acetyl carnitine*
acetyl carnitine HCl
palmitoyl carnitine
polyquaternium-37
polyquaternium-14*
polyquaternium-28
polyquaternium-32
polyquaternium-33*
polyquaternium-35
polyquaternium-36*
polyquaternium-45*
polyquaternium-47
polyquaternium-48*
polyquaternium-53*
polyquaternium-63*
polyquaternium-73*
polyquaternium-91*
acrylamide/ethyltrimonium chloride acrylate/ethalkonium chloride acrylate copolymer*
acrylamidopropyl trimonium chloride/acrylamide copolymer and
acrylamidopropyltrimonium chloride/acrylates copolymer.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.