Abstract
Phenylethyl alcohol (PEA) was tested for developmental toxicity. Pregnant rats were fed 0, 83, 266, or 799 mg/kg/d PEA on gestation days (GDs) 6 to 15; only minimal, nonsignificant effects were observed. In dermal studies, PEA (neat) was applied to the skin on GDs 6 to 15 at dosages of 0, 140, 430, or 1400 mg/kg/d and at 0, 70, 140, 280, 430, or 700 mg/kg/d in a corroborative study. Observations included maternal and embryo-fetal toxicity/abnormalities at 1400 mg/kg/d, increased incidences of rudimentary cervical ribs at ≥430 mg/kg/d, and reduced fetal body weights at ≥140 mg/kg/d. Dermal maternal and developmental no-observed-adverse-effect levels are 70 mg/kg/d, based on dermal irritation and reductions (nonsignificant) in fetal body weights. Human exposure from fragrances is 0.02 mg/kg/d, resulting in a margin of safety >2600, when marked differences in dermal absorption between rats and humans are considered. Under normal fragrance use conditions, PEA is not a developmental toxicity hazard for humans.
Introduction
Phenylethyl alcohol (PEA) is a natural fragrance material that is widely used in perfumes, soaps, and lotions and is the major ingredient of natural rose oil. It is also found in other natural products, such as foods, spices, tobacco, and wine.1,2 Phenylethyl alcohol is a colorless, slightly viscous liquid with a specific gravity of 1.02. The acute oral LD50 for rats was 2.5 mL/kg3 (2500 mg/kg). Diets containing PEA at concentrations of 0.06 to 2 g/kg provided to rats for 14 days produced no adverse effects. 4 However, open topical applications of 0.25 to 2 mL PEA/kg administered to rats for 90 days produced significant decreases in body weight at 1 and 2 mL/kg and significant decreases in hemoglobin in male rats at 2 mL/kg. The no effect level was 0.5 mL/kg/d (∼500 mg/kg/d). 4 The dermal LD50 of PEA in rabbits and guinea pigs was 790 mg/kg and 5 g/kg, respectively. 5 Phenylethyl alcohol (32% in acetone) was moderately irritating to the skin of rabbits and guinea pigs but not to the skin of miniature swine. 6
A comprehensive summary of PEA safety studies has been published by the Cosmetic Ingredient Review Expert Panel.1,2 Phenylethyl alcohol was given generally recognized as safe (GRAS) status by the Flavoring Extract Manufacturers’ Association and is also approved by the Food and Drug Administration (FDA) for food use. 7 Therefore, when Mankes et al 8 reported that developmental effects (fetal death, malformation, and growth retardation) had been caused by gavaged dosages of 4.3, 43, and 432 mg/kg of PEA administered to Long-Evans pregnant female rats during organogenesis (gestation days 6 through 15), it seemed imperative that additional oral and topical developmental toxicity studies be conducted to establish the safe use of PEA.
Relevant animal studies were sponsored by the Research Institute for Fragrance Materials (RIFM) and conducted to extend the Mankes et al 8 oral developmental toxicity study in rats and to compare the pharmacokinetics of PEA in animals and humans, simulating human dermal exposure and determining, if possible, a realistic margin of safety. Results from these studies have been previously reviewed by the Cosmetic Ingredient Review Expert Panel,1,2 have been presented in a poster format at the Society of Toxicology,9–11 and as proceedings from scientific meetings,12,13 but they have not been published as peer-reviewed articles. This article presents the previously generated developmental toxicity data as a stand-alone report for future reference. The pharmacokinetic/metabolism studies are reported in a separate publication. 14
Methods
All studies were sponsored by the RIFM and conducted in compliance with appropriate Good Laboratory Practices (Organization for Economic Cooperation and Development [OECD] 1981; US FDA 1978, 1987).15–17 All studies were conducted at Huntingdon Research Center, Ltd., Huntingdon, Cambridgeshire, UK, except for the corroborative dermal developmental toxicity study in rats (conducted at the Charles River Research Laboratories, Horsham, Pennsylvania), which was used to assess the validity of the earlier observations.
Oral Developmental Toxicity Study of PEA in Rats
The oral study was designed to determine the effect of PEA on pregnant dams and their conceptuses when provided with diets given to the dams during the period of organogenesis. Nine-week-old healthy, time-mated, Crl:COBSCD(SD)BR female rats were obtained from Charles River UK Ltd. (Margate, Kent). After acclimation, 112 pregnant rats were assigned to 4 groups of 28 rats each, using a computerized stratified randomization for body weight. Ad libitum access to feed and water was provided, and the rats were housed individually in suspended galvanized wire mesh cages in an animal room maintained at 20°C ± 2°C with 53% ± 11% humidity and a 12-hour light/dark cycle. Ground laboratory diet (Labsure Laboratory Diet No.2) containing 0, 1000, 3000, or 10 000 ppm of PEA (calculated intake: 0, 83, 266, or 799 mg/kg/d) was provided on days 6 through 15 of gestation (GDs 6 through 15; sperm/sperm plug = GD 0). The PEA was microencapsulated in spray-dried gum arabic and then incorporated into the diet. The concentrations provided remained unchanged during the course of the study and were selected on the basis of results from a previous pilot study in which dietary inclusion of up to 9000 ppm PEA was well tolerated by pregnant rats and appeared to maintain diet palatability. High-performance liquid chromatographic (HPLC) analyses were used to validate the concentration, homogeneity, and stability of the PEA used throughout the study.
All animals were monitored daily for signs of toxicity. Feed consumption was measured daily, and body weights of the dams were taken on GDs 1, 3, 6, and then on alternate days until necropsy. The rats were sacrificed on GD 20, and in utero development of the conceptuses was assessed by determination of litter values and examination of the fetuses for external, soft tissue, and skeletal alterations (malformations and variations). Half of each litter was sectioned according to the method of Wilson, 18 and evaluated for visceral changes, while the other half was evaluated for skeletal changes after clearing and staining with alizarin red. 19
Dermal Developmental Toxicity Study of PEA in Rats
The dermal study consisted of 4 groups of 25 to 35 per group time-mated female rats. The rats were of the same strain, age, and source as described for the oral study, and animal handling and housing conditions were also the same. Phenylethyl alcohol (neat) was applied to a 7 × 5 cm shaved (by electric clipper), interscapular area of the skin on GDs 6 through 15 at dosages of 140, 430, or 1400 mg/kg/d. A control group was administered applications of water at the same mL/kg volume as the high-dosage group (1.40 mL/kg). The 430 mg/kg/d dosage was selected because it was approximately equivalent to the oral high dosage used in the Mankes et al 8 study. The highest dosage, 1400 mg/kg/d, was used to extend the range of dosages tested, in case there was differential absorption by the dermal route. To minimize possible oral ingestion of the control or test material, the dermal applications were occluded with an aluminum foil patch that was held in place by a porous medical adhesive bandage. The applied daily dosages were kept on the animals for 24 hours. A total of 3 satellite animals (plus a spare) in the low- and high-dosage groups were administered radiolabeled PEA on GD 15, after which urine was collected for 24 hours to determine absorption.
The following parameters were evaluated in the study: signs of maternal toxicity; mortality; body weight and feed consumption on GDs 1, 3, 6, and on alternate days until necropsy on GD 20; liver and kidney weights at necropsy and hematology and serum chemistry parameters in 15 control and 13 high-dosage group rats on GD 20; uterine contents (absence of pregnancy was confirmed by uterine staining with ammonium sulfate 20 ), including the number, sex, and weights of live fetuses; number of resorptions and fetuses with external malformations; determination of skeletal and soft tissue changes. Half of each litter was sectioned according to the method of Wilson (1965), and evaluated for visceral changes, while the other half was evaluated for skeletal changes after clearing and staining with alizarin red. 19
Corroborative Dermal Developmental Toxicity Study of PEA in Rats
The object of this second topical study was to corroborate the finding of dose-dependent increases in fetal and litter incidences of cervical ribs at 140, 430, and 1400 mg/kg/d found in the first topical study and to define more accurately the dosages that may have produced the reported fetal changes. The methodology and animals used in this study closely replicated those used in the first study.
Differences in procedures included: The Crl: COBS®CD®(SD)BR Charles River rats were obtained from Kingston, NY and housed individually in stainless steel cages, except during cohabitation, in an animal room independently supplied with a minimum of 10 changes per hour of 100% fresh air that had been passed through 99.97% HEPA-filters; breeding occurred at the testing facility in the room in which the study was conducted (no male rat impregnated more than one female rat); 2 randomizations were performed, 1 at receipt and another (weight-ordered randomization) based on GD 0 body weights; 10 rats were assigned to each dosage group and 5, rather than 3 dosages of PEA were tested (dosages of 0 [deionized water], 70, 140, 280, 430, and 700 mg/kg/d); feed and body weights of the dams were recorded daily (dosages were adjusted daily on the basis of maternal body weight); no clinical pathology or pharmacokinetic parameters were investigated.
In addition, all fetuses (rather than half, as in the previous dermal study) were stained by alizarin red-S (method of Staples and Schnell 21 ) and examined for skeletal defects, while fewer fetuses per litter (1/3, rather than 1/2) were examined for visceral alterations, by using a modification of Staples’ technique. 22 These modifications were made to allow examination of all fetuses for cervical ribs, a skeletal variation that had an increased incidence in the original topical study.
Statistical analyses of clinical observation and other proportion data were performed using the variance test for homogeneity of the binomial distribution. 23 Continuous data (eg, body weights and body weight changes) were analyzed using Bartlett test of homogeneity of variances 24 and the analysis of variance (ANOVA), 25 when appropriate (ie, Bartlett test was not significant, P > .001). If the ANOVA was significant (P ≤ .05), Dunnett test 26 was used to identify the statistical significance of the individual groups. If the ANOVA was not appropriate (ie, Bartlett's test was significant, P ≤ .001), the Kruskal-Wallis test 27 was used, when less than or equal to 75% ties were present. In cases where the Kruskal-Wallis test was statistically significant (P ≤ .05), Dunn method of multiple comparisons 28 was used to identify the statistical significance of the individual groups. If there were greater than 75% ties, Fisher exact test 29 was used to analyze the data.
Results
Oral Developmental Toxicity Study in Rats
Analytical chemistry studies that were conducted before and during the study indicated that the mean concentrations of PEA in the test diets were within ± 4% of theoretical values. Stability of the microencapsulated PEA in the diet was also confirmed.
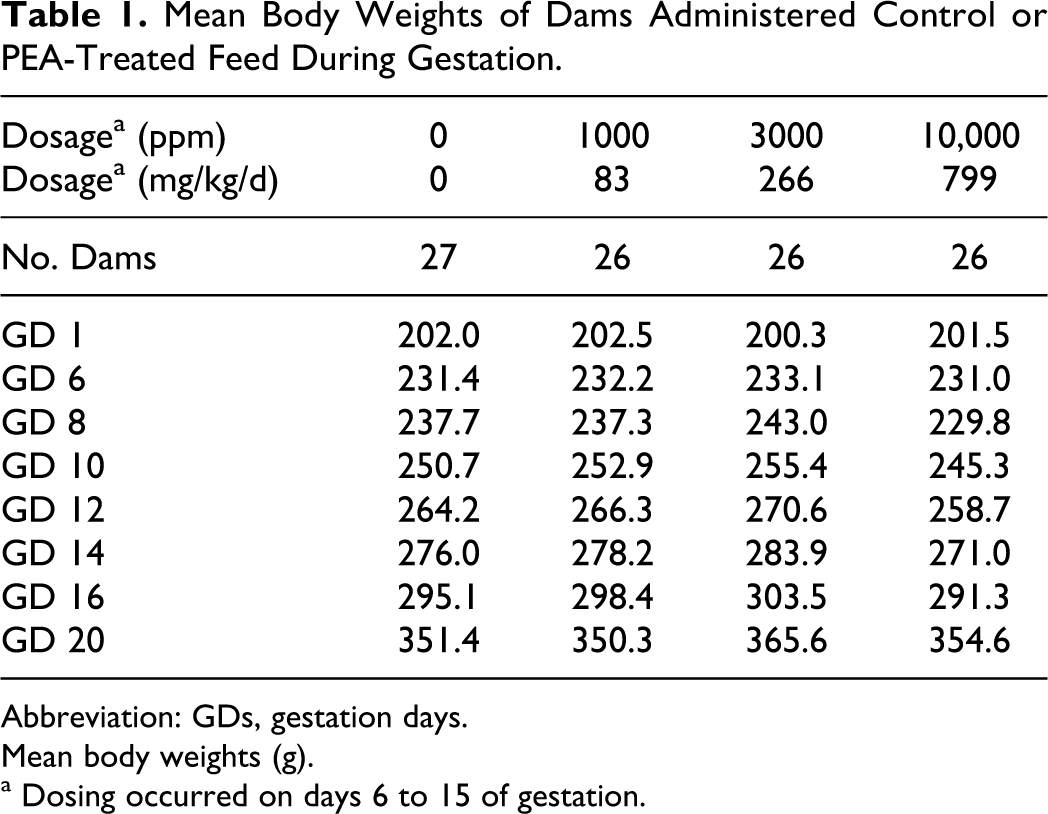
There were no clinical signs of toxicity or mortality at any dosage level. Calculation of feed consumption indicated that the mean group intake of PEA was 0, 83, 266, or 799 mg/kg/d for the groups administered 0, 1000, 3000, or 10 000 ppm of PEA in the diet on GDs 6 through 15. Feed consumption and body weight gains (Table 1) were reduced predominantly on the first 2 days of treatment at the highest dosage level (results were not statistically different). No other maternal effects were observed during the study.
Mean Body Weights of Dams Administered Control or PEA-Treated Feed During Gestation.
Abbreviation: GDs, gestation days.
Mean body weights (g).
a Dosing occurred on days 6 to 15 of gestation.
On GD 20, necropsy revealed that 27 of 28 control rats and 26 of 28 rats in each of the low, mid, and -dosage groups were pregnant with live fetuses. Litter parameters, as assessed by mean values for embryo-fetal loss, litter sizes, sex ratios, and litter and mean fetal weights, were equivalent in control and PEA-exposed groups. The incidence, type, and distribution of fetal malformations, anomalies, and skeletal variants were statistically unaffected by maternal exposure to PEA at concentrations as high as 10 000 ppm (799 mg/kg/d). However, at the highest dosage level, a marginal delay in fetal ossification might have occurred, as a slightly higher incidence of fetuses with incomplete ossification of some skeletal elements, particularly the sacrocaudal vertebral arches, was observed (Table 2).
Skeletal Effects in Gestation Day 20 Fetuses After Dietary Administration of Phenylethyl Alcohol to Dams.
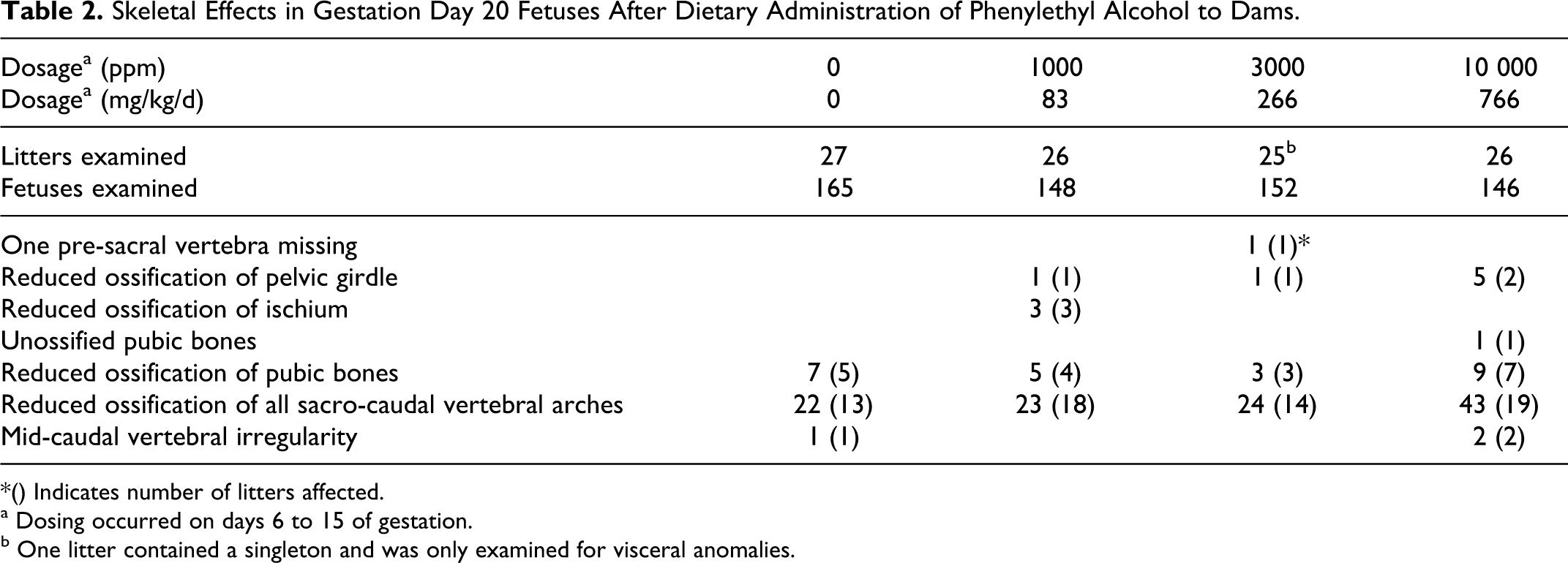
*() Indicates number of litters affected.
a Dosing occurred on days 6 to 15 of gestation.
b One litter contained a singleton and was only examined for visceral anomalies.
Dermal Developmental Toxicity Study in Rats
In the first dermal study, pregnant dams were treated with 0, 140, 430, or 1400 mg PEA/kg/d on GDs 6 through 15. Local irritation at the application site was observed in some rats early in the treatment period, especially in the high-dosage group. Signs of toxicity included: irritability, hunched posture, walking on toes, pilo erection, and periorbital staining (these effects were restricted almost entirely to dams in the high-dosage group). Also, in the high-dose group, 1 moribund euthanasia was required on GD 11, and 2 deaths occurred on GD 13 (Table 3). The toxic signs were first observed around GD 12 and progressively worsened during the remainder of the treatment period. After cessation of treatment (the last dosage was administered on GD 15), the rats recovered; most appeared normal by termination (GD 20).
Mean Body Weights and Disposition of Dams given Dermal Applications of Phenylethyl Alcohol During Gestation.
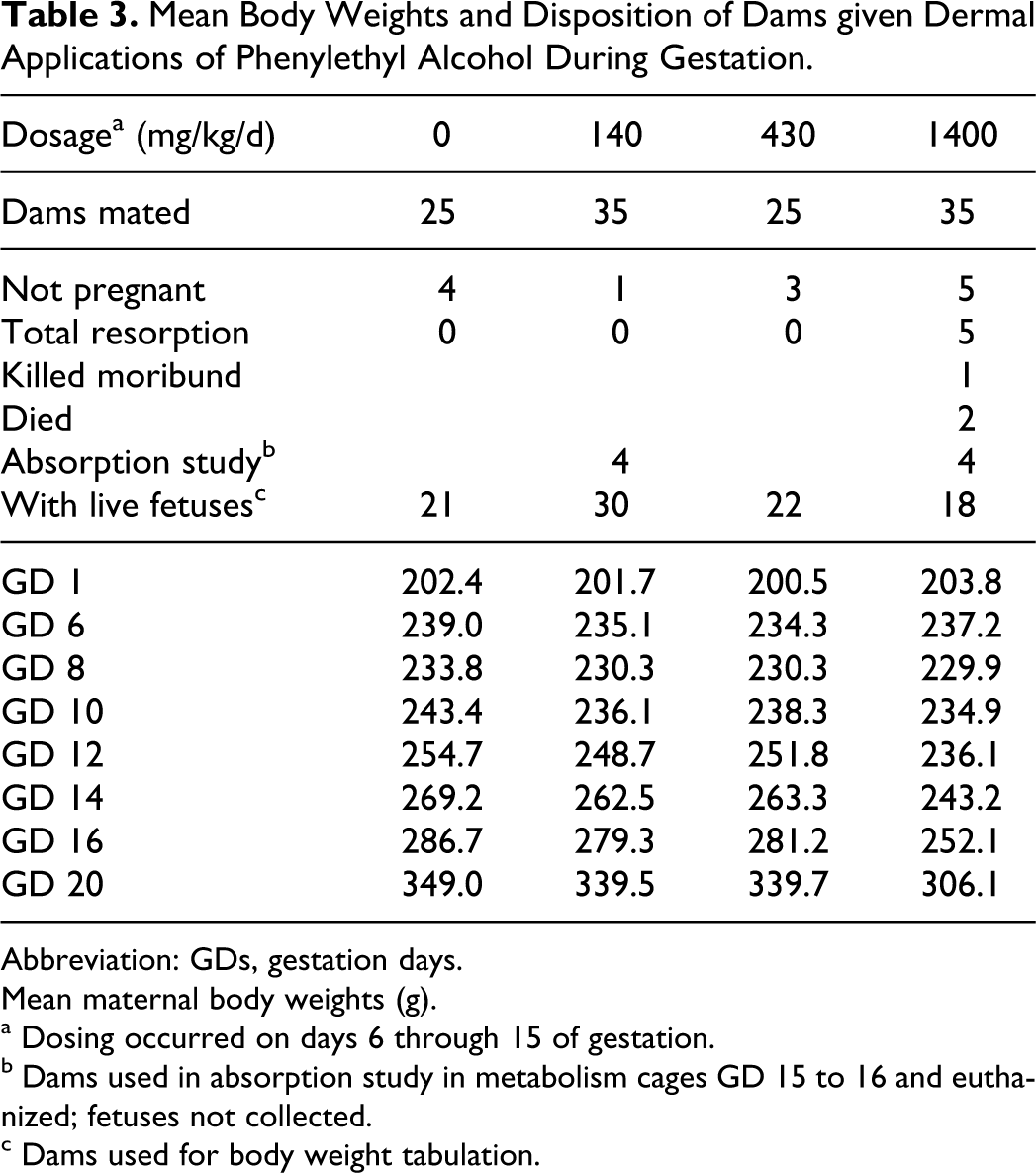
Abbreviation: GDs, gestation days.
Mean maternal body weights (g).
a Dosing occurred on days 6 through 15 of gestation.
b Dams used in absorption study in metabolism cages GD 15 to 16 and euthanized; fetuses not collected.
c Dams used for body weight tabulation.
Mean feed consumption and body weight gains were markedly reduced in the high-dosage group during the treatment period. Reversal of these effects occurred upon cessation of treatment, but mean body weights were still statistically depressed at termination (Table 3). There were no significant differences between control and test groups in mean liver or kidney weights, pregnancy rates, mean preimplantation losses, or gross changes at necropsy on GD 20. Clinical pathology determinations conducted in control and high-dosage dams revealed no adverse hematological or serum chemistry findings.
Systemic absorption of topical PEA was estimated by measurement of radioactivity in 24-hour urine samples from the 3 “marker” rats in the low- and high-dosage groups. An application of 14 C-phenylethanol was made on the final day of treatment. The results indicated that 63% to 66% of the dosage had been absorbed. Absorption was rapid and extensive, but not dose dependent.
In the high-dose group, embryo-fetal toxicity was manifested as statistically significant increases in post-implantation loss (litter means for early and total [early + late] resorptions; Table 3). The changes were accompanied by a statistically significant reduction in mean litter size (7 or 9 pups versus 11 in the control group and 12 in both the 140 and 430 mg/kg/d group), and a marked depression of mean fetal body weight (2.08 g versus 3.26 g in control group, 3.33 g in the 140 mg/kg/d group, and 3.18 g in the 430 mg/kg/d group). None of these changes occurred at the 2 lower dosages.
When fetuses were examined for soft tissue and skeletal abnormalities, a dose-related increase in defects was observed. Statistical analysis of incidences of abnormalities was considered unnecessary given the results obtained. At the highest dosage (1400 mg/kg), morphological changes were present in 160 of the 161 fetuses; the variety, incidence, and degree of the skeletal and soft tissue changes varied considerably among individual fetuses. However, high incidences of the following specific types of defects were noted in more than 40% of the fetuses and 70% of the litters at the high dosage (1400 mg/kg/d): anophthalmia or microphthalmia, ventricular septal defects, defects or irregularities affecting thoracic, lumbar, and sacro-caudal vertebrae (associated with short or kinky tail), defects of the thoracic ribs, and occurrence of rudimentary cervical ribs (RCR). At the mid-dosage (430 mg/kg/d), occasional fetuses displayed the specific changes observed at 1400 mg/kg/d, and the number of fetuses with RCR, thoracic changes, or moderate degrees of reduced ossification exceeded concurrent controls. At the low dosage (140 mg/kg/d), findings were equivocal; incidences of RCR and thoracic vertebral irregularities were only marginally higher than those in the controls.
Corroborative Dermal Developmental Toxicity Study in Rats
In the corroborative study, the previously tested topical dosages of 140 and 430 mg/kg/d were repeated and additional dosages (70, 280, and 700 mg/kg/d) were tested to better characterize the dose response curve for RCR. No deaths occurred in the study, but all dosages of PEA (70, 140, 280, 430, or 700 mg/kg/d) produced localized, low levels of erythema and/or desquamation at the intrascapular application sites. The severity of these reactions was dose related.
Additional signs of toxicity included ptosis and/or urine-stained abdominal fur, noted only in dams at the 700 mg/kg/d dosage level. Classic signs of maternal toxicity, that is, reduced feed intake and body weight gains, did not occur in a consistent manner, which is often the case when the primary effects are associated with local irritation. Dosages as high as 700 mg/kg/d did not adversely affect average litter sizes, numbers of implantations, live fetuses, or post-implantation loss (resorptions).
Fetal effects (Table 4) included dose-dependent reductions of the litter mean for live fetal body weights. The reductions were statistically significant at ≥ 140 mg/kg/d but not at the lowest dosage of 70 mg/kg/d. A significantly increased incidence of RCR occurred at 700 mg/kg/d; and minimal increases of reversible delays in ossification33–32 were observed at ≥70 mg/kg/d. The incidences of the reversible delays in ossification were within the historical ranges of the Testing Facility. 34 Specific delays included: delayed sternal and/or pelvic ossification (all dosage levels); incompletely ossified (hypoplastic) ribs and/or wavy ribs at 70 and at ≥430 mg/kg/d; and delays in ossification of the metacarpals and hindpaw phalanges (≥140 mg/kg/d), caudal vertebrae (≥280 mg/kg/d), forepaw phalanges (≥430 mg/kg/d), and sternal centers (700 mg/kg/d).
Corroborative Study: Summary of Mean Fetal Body Weights and Statistically Significant Fetal Skeletal Alterations in Gestation Day 20 Fetuses from Dams Given Dermal Applications of Phenylethyl Alcohol During Gestation.
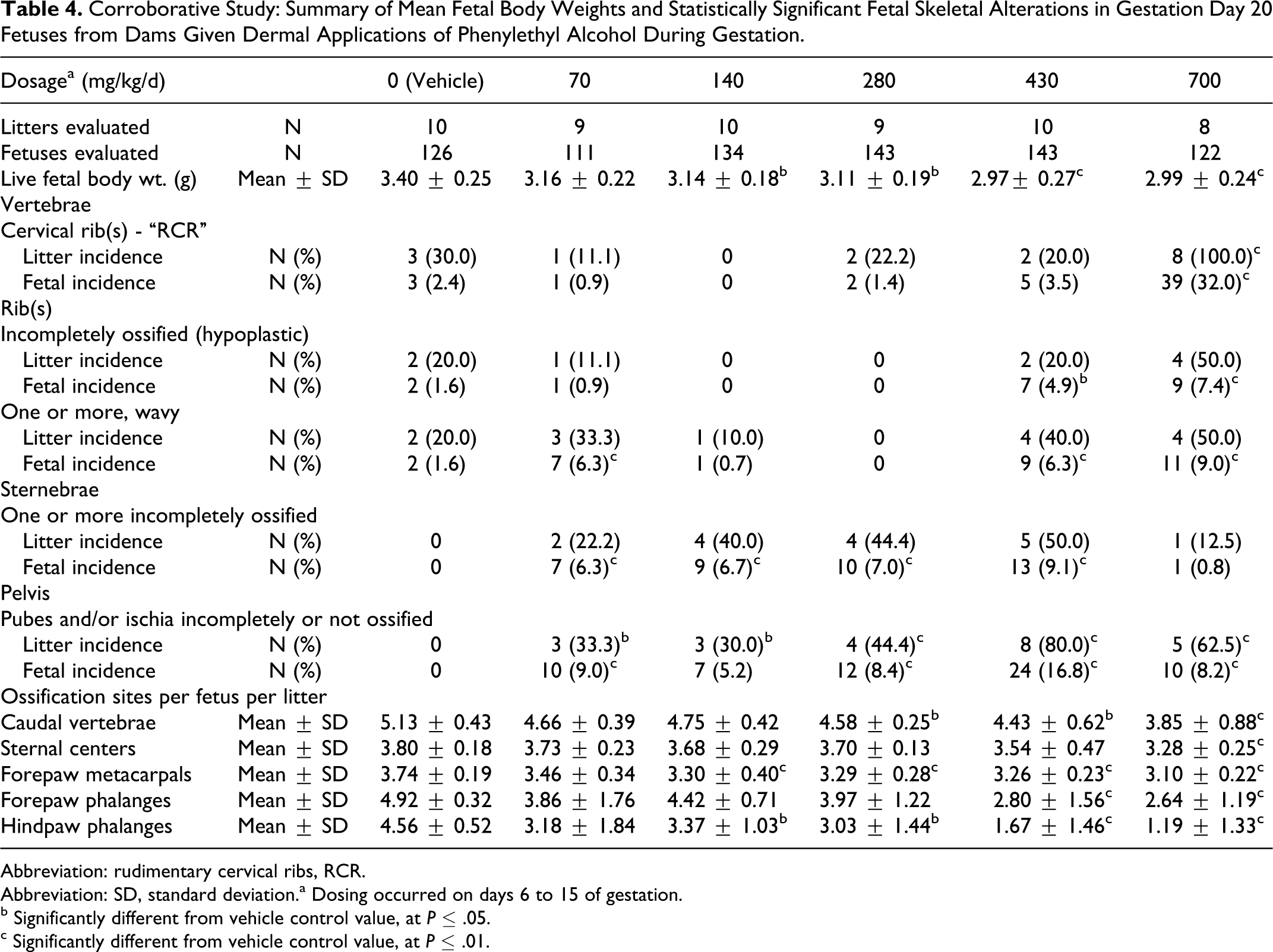
Abbreviation: rudimentary cervical ribs, RCR.
Abbreviation: SD, standard deviation.a Dosing occurred on days 6 to 15 of gestation.
b Significantly different from vehicle control value, at P ≤ .05.
c Significantly different from vehicle control value, at P ≤ .01.
Discussion and Conclusions
Phenylethyl alcohol is used in many fragrance compounds in a variety of cosmetic and household products. Establishing the human exposure level of PEA when it is used as a fragrance ingredient has been done by a survey of marketed hydroalcoholic products 35 using the method described by Cadby et al. 36 Using the 97.5 percentile use level of PEA in formulae for use in fragranced cosmetics in general results in a conservative calculated maximum daily exposure on the skin of 0.3 mg/kg/d for a 60 kg adult.
When dietary concentrations of 1000 or 3000 ppm of PEA (calculated intakes of 83 or 266 mg/kg/d, respectively) were administered to pregnant rats during the period of organogenesis (GDs 6 through 15), no deleterious effects were produced in the dams, and embryo-fetal viability, development, and morphology were unaffected. Even at the highest dosage of 10 000 ppm (799 mg/kg/d), adverse effects were minimal: transient decrease in maternal feed consumption and body weight gains on GDs 6 and 7 and a slightly higher incidence of delayed ossification in some fetal skeletal elements, such as the sacro-caudal vertebral arches. Based on these results, the maternal and developmental no-observed-adverse-effect levels (NOAELs) for dietary administration were 266 mg/kg/d, resulting in a theoretical margin of safety of greater than 850 times the near-maximal (0.3 mg/kg/d) fragrance use level in humans.
These results differ remarkably from those of Mankes et al. 8 These investigators reported that oral gavaged dosages of 4.3, 43, and 432 mg/kg/d produced fetal death, malformations, and growth retardation. The difference can probably be attributed to the single daily bolus dosages that likely produced spikes of high blood serum levels that could not be detoxified in a timely manner. Adverse developmental effects to the fetus are often considered to be due to excessively high peak exposures of noxious agents. 37 Furthermore, although gavaged dosages can be accurately defined and have fewer confounding factors than topical application, they do not mimic the human use of fragrances, which tend to be applied (and absorbed) several times a day. Such applications produce lower but more prolonged serum levels that resemble those seen after dietary intake, rather than bolus administration. Finally, there was a difference in the strain of rat studied (Long-Evans rats were used by Mankes et al 8 ).
However, neither of the oral studies addressed possible differences caused by the route of administration of the PEA. Therefore, 2 developmental toxicity studies were performed to characterize and define the effects of PEA when applied topically to the skin of pregnant rats on GDs 6 through 15. The corroborative, second study was conducted with fewer animals and was designed to define more accurately the dose-response curves achieved in the first study. The combined results of these two studies demonstrated that dosages of ≥ 70 mg/kg/d produced dose-dependent local irritation at the application site, a common confounding factor in these types of studies. 38 Decreases in fetal body weights and consequent reversible delays in ossification at 140 mg/kg/d and higher were probably associated with maternal stress39,40 produced by the localized dermal irritation at the application site. Delays in ossification are considered fetal variations (reversible delays in development) in rodents that are usually related to reduced fetal body weight and, in the absence of fetal malformations, are not considered a significant hazard to humans. 39 Maternal and developmental (NOAELs) for dermal application were considered to be 70 mg/kg/d, based on minimal dermal irritation in the dams and small (not statistically significant) reductions in fetal body weights at this dosage level. Such NOAELs suggest the presence of a theoretical margin of safety greater than 230 times the near-maximal human fragrance use level of 0.3 mg/kg/d.
However, species differences in absorption, metabolism, and excretion are major factors in calculating a realistic margin of safety for PEA. Therefore, the data from one dermal absorption and disposition study in rats, rabbits, and humans, as well as determination of plasma and urine concentrations and pharmacokinetics in rats reported by Politano et al, 14 are highly relevant. Results from those studies indicated that biotransformation and excretion of PEA were similar in rats and humans. The major metabolite is phenylacetic acid and the maximum concentration in blood occurred 1.5 hours after dermal application in humans. Systemic absorption from topical application was much greater in rats than in humans. Based on urinary excretion of radioactivity, rats absorbed 77% of a single topical dosage of 14 C-PEA, while only 7.6% was absorbed by humans. The results of the present study provide a similar extent of dermal absorption in rats. About 65% of the applied dose was recovered in the urine by 24 hours. Thus, systemic exposure to PEA (and the metabolite phenylacetic acid) is markedly less in humans than in rats. This exposure to PEA from fragrances, 0.3 mg/kg/d can then be adjusted based on human skin absorption of 7.6% of the applied dose, resulting in a realistic human exposure level of 0.02 mg/kg/d. Utilizing the observed absorption differences resulting in human exposure of 0.02 mg/kg/d from multiple PEA-containing cosmetic products, Politano et al 14 calculated a human margin of safety greater than 2600. Based on this more realistic exposure-derived margin of safety, it is concluded that PEA, under normal fragrance use conditions, is not a developmental toxicity hazard for humans.
Footnotes
Declaration of Conflicting Interests
Valerie T. Politano, Gretchen Ritacco and Anne Marie Api are employees of the Research Institute for Fragrance Materials, an independent research institute supported by the manufacturers of fragrances and consumer products containing fragrances.
Funding
This research was supported by the Research Institute for Fragrance Materials, an independent research institute that is funded by the manufacturers of fragrances and consumer products containing fragrances.
