Abstract
Brassinosteroids (BRs) are close analogues of animal cholesterol. Brassinosteroids have shown their great value as yield promoters of a variety of plants. In view of its steroidal moiety and recent use in agriculture in many countries, the teratogenic potential of homobrassinolide (HBR) was evaluated in Wistar rats. Homobrassinolide was administered by oral gavage at doses 0, 100, and 1000 mg/kg body weight in water during gestation days (GD) 6 to 15 in groups of 20 mated females. Maternal and embryo-fetal toxicity was analyzed by studying the effects such as clinical signs, mortality/morbidity, abortions, body weight, feed consumption, and pregnancy data, gravid uterine weights, implantation losses, litter size, external, visceral, and skeletal malformations. No treatment-related effect was observed on any of the maternal/fetal end points in any dose group. From the results, it can be concluded that HBR is nonteratogenic at doses as high as up to 1000 mg/kg body weight in Wistar rats.
Introduction
Brassinolide (BL), a polyhydroxy steroidal lactone, was identified as a plant growth promoter in 1979. Since then, many related chemicals known as brassinosteroids (BRs) were developed, among which 28-homobrassinolide (HBR) and 24-epibrassinolide (EP) are the most biologically active. 1,2 Homobrassinolide was first synthesized in 1982. 3 Use of BRs as yield promoters is well established in Japan, China, USSR, and India. 4 –6 In China, HBR was registered to increase the yield of tobacco, sugarcane, tea, and rapeseed. 7 In India and USSR, HBR was introduced as plant growth regulator (PGR) in the early 1990s for a diverse variety of crops such as grapes, vegetables, cotton, rice, tea, oilseeds and tomato, potato, pepper, cucumber, and barley.
Brassinosteroids are ubiquitous in the plant kingdom, and they are derived from phytosterol (PS) in plants. Phytosterols are structurally similar to cholesterol and found in a wide range of plants, including dicots, monocots, gymnosperms, and algae 8,9 and in various plant parts such as pollen, leaves, flowers, roots, seeds, shoots, galls, and stems. 10,11 Their exogenous application enhances potential yield of crops by active action on proton pump resulting in cell elongation. 12 –14 BRs were considered the sixth phytohormone at the 16th International conference on plant growth substances (Makuhari, Japan) because of their specific activities. 15
Brassinosteroids influence developmental processes such as growth, seed germination, rhizogenesis, flowering, senescence, abscission and maturation, metabolic processes like photosynthesis, 16,17 protein synthesis, vascular differentiation, activation of CO2/nitrogen fixation, induction of ethylene biosynthesis, xylem differentiation, and the regulation of gene expression. 18 –21 They are also known to ameliorate various biotic and abiotic stresses. 22 –24 A survey of literature revealed considerable animal toxicity for few PGRs/phytohormones at high doses for example, gibberellic acid (GBA), indole-3-acetic acid (IAA), and sitosterol.
Gibberellic acid, an extended biosynthetic product of cholesterol metabolism in plant, is reported to be carcinogenic and genotoxic in mice, hepatotoxic and estrogenic in rats. 25 Another plant hormone/an auxin, indole-3-acetic acid, is a well-known teratogen, inducing cleft palate in both mice and rats at a dose of 500 mg/kg body weight/d. 26 Sitosterol, a closely related PS, was found to be estrogenic in fish 27,28 and affected testis and sperm in rats. 29,30 Homobrassinolide has been found to decrease lactate dehydrogenase (LDH) activity in the brain and liver and to increase LDH activity in kidneys and testis in rats. 31 Homobrassinolide also changed the hematology profile and was associated with decreased blood glucose and altered the total cholesterol level. 31 Interestingly, among many effects, PSs interfere with the absorption of β-carotenoids, which are vitamin A precursors. 32
Because the chemistry of BRs indicates a steroidal moiety, and because of their extensive use in agriculture, it is expected that a large fraction of population may be exposed to the action of BRs in general and HBR in particular. Furthermore, daily consumption of PS is almost equivalent to that of cholesterols, that is, 150 to 400 mg. 33 Hence, this study was conducted to examine the possible teratogenic potential of HBR in rat, using the applicable Indian test guideline 34 (Gaitonde Committee Guideline) for registration of crop protection/growth promoter products.
Materials and Methods
Animals and Condition
Healthy adult nulliparous female Wistar rats weighing 200 to 225g were procured from the animal house facility of Fredrick Institute of Plant Protection and Toxicology (now IIBAT), Padappai, India. The current investigation was duly approved by the Institutional Animal Ethics Committee. Animals were acclimatized for 7 days in the test room before the conduct of the experiment. Standard animal house conditions (temperature: 22°C ± 3°C, relative humidity: 30%-70%, photoperiod: 12-hour light/12-hour dark) were maintained throughout the experiment. A total of 60 mated females were randomized in 3 groups, each containing 20 animals. Animals were housed individually in standard polypropylene rat cages (Messer’s Vishnu Traders, Roorkee, Uttarakhand, India) with cleaned and sieved paddy husk (procured from local vendor) as the bedding material. The cages were autoclaved regularly. Standard rat pellet feed (Messer’s Amrut Laboratory Animals Feed, Maharashtra (Pune), India) and filtered water (Aquaguard, Eureka Forbes, Ltd, Karnataka, India) was provided to animals ad libitum. Ear tags were used for identification of animals.
Test Chemical
Homobrassinolide (Trade name Combine, purity 86.3%) was supplied by Messer’s Godrej Agrovet Ltd, Mumbai, India. It was kept in a cool and dry place as recommended. Distilled water was selected as the vehicle, and the administered dose volume (10ml/kg body weight) was adjusted based on weekly body weight taken during the treatment period. The test substance was weighed and mixed with distilled water to prepare desired dose levels. All other chemicals used were of laboratory grade.
Mating
Animals were tested for their phase in the estrous cycle and those animals in the pro-estrous phase were selected for mating. Mating was done in a ratio of 1:1. Vaginal plug was used to confirm mating and was designated as gestation day 0 (GD 0).
Study Design
Study design was per the national guideline for registration of crop protection/growth promoter product—Gaitonde Committee guideline. 34 A preliminary study was conducted with 3 groups of pregnant rats each containing 3 animals. They were treated with 1000, 2000, and 3000 mg/kg body weight of Combine (HBR), respectively, on day 6 to day 15 of the gestation period. Test substance was administered orally using gavage intubation needle. Signs of toxicity, body weight, feed consumption, gross pathology, and external examination of fetuses were monitored. Animals treated with 3000 mg/kg body weight of Combine (HBR) were sacrificed humanely on GD 14 and 15 because of moribund condition (2/3). Rats treated with 2000 mg/kg body weight showed a very high decrease in body weight and feed consumption, and exhibited signs of toxicity like abortion (1/3), dullness, lethargy, and anorexia during GD 8 to GD 17. Animals treated with 1000 mg/kg body weight exhibited dullness and lethargy during GD 8 to GD 10, which resolved subsequently.
Based on preliminary study results, 3 groups were maintained in the main study. Mated females were allocated to one of these 3 groups (20/group) by stratified randomization; mean body weight was restricted not to exceed by 20% between the groups. Group 1 served as the control receiving distilled water alone, groups 2 and 3 received 100 mg/kg body weight and 1000 mg/kg body weight of HBR, respectively. Dosing was done from GD 6 to GD 15.
Maternal End Points
Body weights of all dams were recorded on GD 0, 7, 14, and 20. Feed consumption was recorded daily and reported as an average on GD 0, 7, 14, and 20. Animals were observed for mortality, morbidity, and any overt clinical signs of toxicity during dosing, postdosing, and at sacrifice. On GD 20, all animals were sacrificed by cervical dislocation. The abdominal cavity was opened to expose the reproductive system and the pregnancy status was determined. Uterine components were examined for the number of implantations, implantation defects, resorptions (early and late), dead/live fetuses, and number of corpora lutea. Uteri/unilateral uterine horns with no visible implantation sites were stained with 10% ammonium sulfide. Dams were exhaustively examined at necropsy; cervical, thoracic, and abdominal viscera were inspected for abnormalities.
Fetal End Points
Fetuses and placentas were examined in situ and carefully separated from the dams. All fetuses were weighed, sexed, and observed for morphological abnormalities. Fetuses were sacrificed by intraperitoneal administration of thiopental sodium and preserved in appropriate fixatives. About two thirds of the fetuses were preserved in 70% propanol and analyzed for skeletal defects. After 15 to 20 days of preservation, the fetuses were skinned and eviscerated, macerated in 1% KOH followed by staining with Alizarin Red S 35 and carefully examined for any skeletal abnormalities. The remaining one third of the fetuses was preserved in Bouin fixative and sectioned serially 36 for detection of visceral abnormality, if any.
Statistical Analysis
The statistical significance of differences between the means of body weight, feed consumption, resorptions, fetal weights, total number of live fetuses, and number of male and female fetuses was assessed by 1-way analysis of variance (ANOVA). Values were expressed as mean ± SD. One-way ANOVA was used to compare the values between different groups and P value less than .05 was considered statistically significant.
Results
Maternal End Points
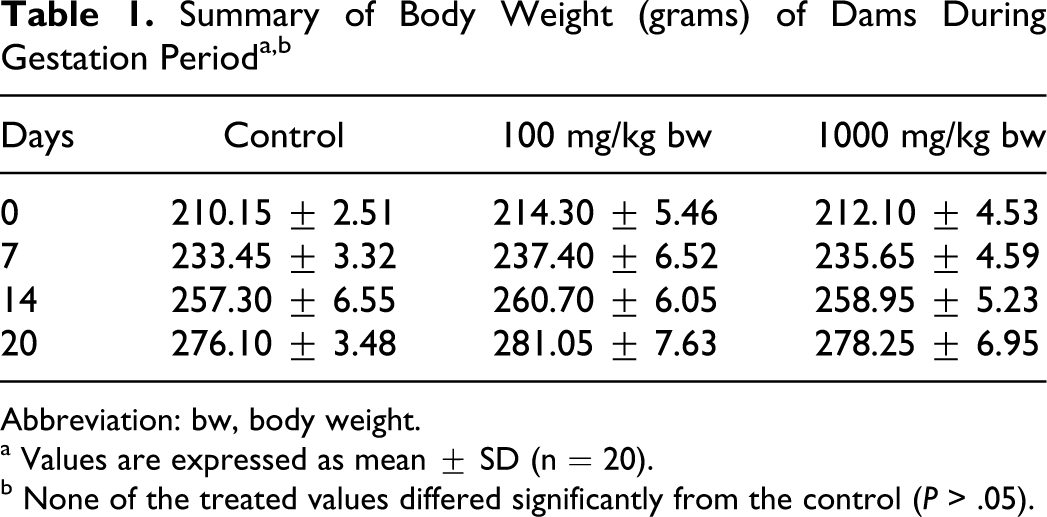
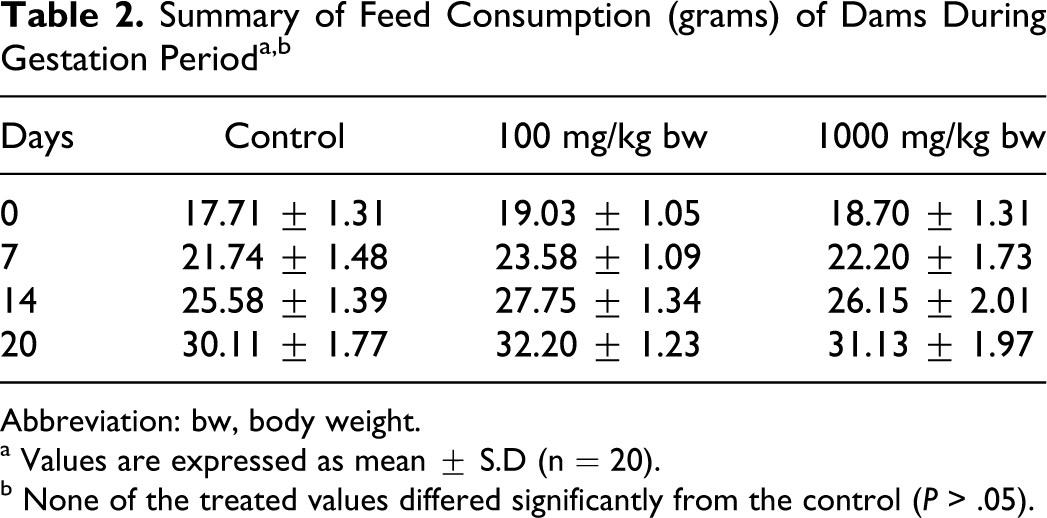
There was no morbidity or mortality at any dose tested. Clinical signs of toxicity were unremarkable. No significant changes to body weights and feed consumption were recorded in females from the treated group when compared with the control (Tables 1 and 2). There was no incidence of spontaneous abortion or premature delivery in any of the groups. No remarkable gross pathological changes were observed in dams of any group including the control (data not shown). No changes in the weights of gravid uterus in any of the treated groups were recorded when compared with the control.
Abbreviation: bw, body weight.
a Values are expressed as mean ± SD (n = 20).
b None of the treated values differed significantly from the control (P > .05).
Abbreviation: bw, body weight.
a Values are expressed as mean ± S.D (n = 20).
b None of the treated values differed significantly from the control (P > .05).
Table 3 represents the mean number of corpora lutea and the implantation sites among the various treatment groups. Early and late resorptions, mean litter size, and viable fetuses were unanimous among all the groups. No statistically significant changes were observed.
Summary of Uterine and Fetal Data
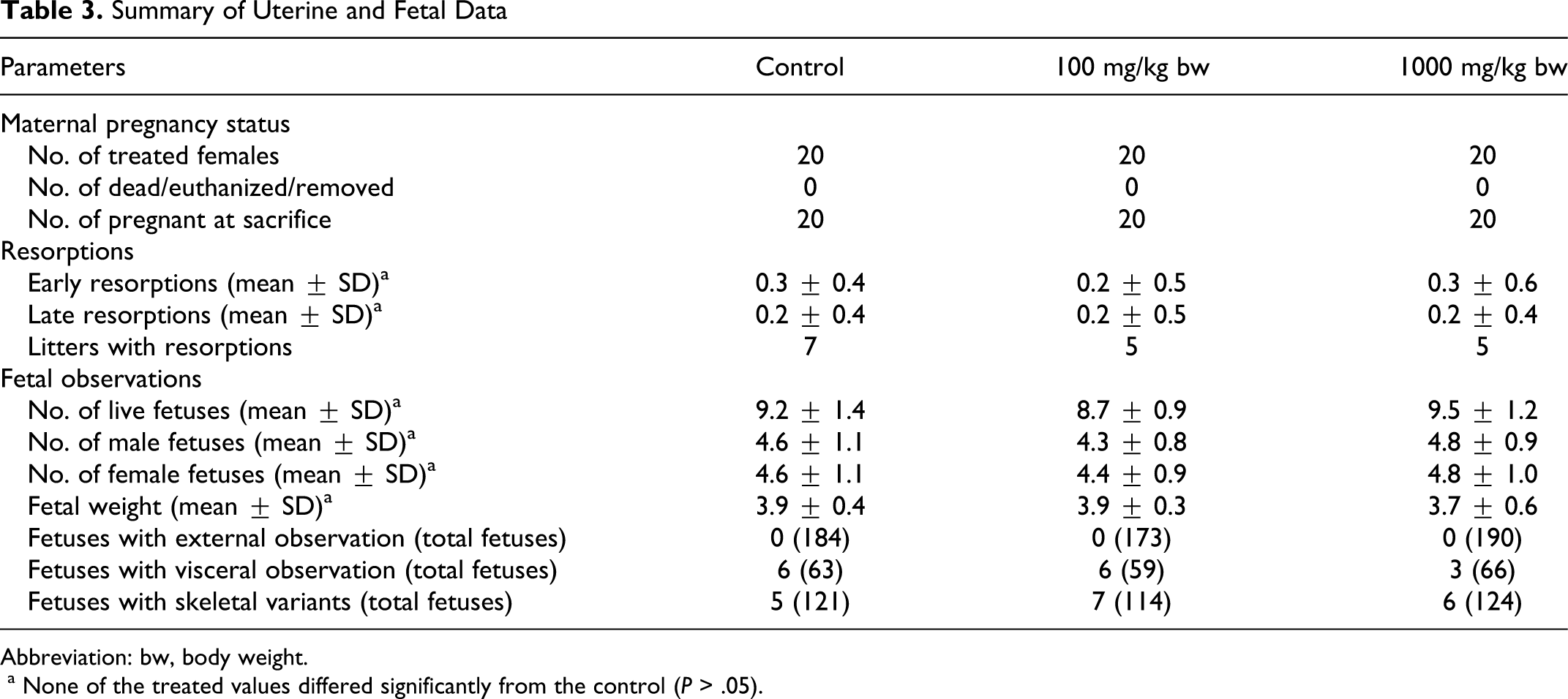
Abbreviation: bw, body weight.
a None of the treated values differed significantly from the control (P > .05).
Fetal End Points
Mean fetal weights and sex ratio were undisputedly similar among the groups. No fetal external malformations were observed. Treatment-related visceral malformations were not observed in any treated group. External hydrocephalus and hemoperitoneum (each 1 of 66 fetus) was observed at the 1000 mg/kg body weight dose, and dilated renal pelvis was observed (2 of 59) in 100 mg/kg body weight group along with other commonly occurring spontaneous findings which were scattered among all the groups (Tables 3 and 4). No skeletal malformations were observed in any of the group that could be attributed to test substance. However, routinely recorded skeletal variations such as supernumerary, rudimentary ribs, incomplete ossification of ribs, and bi-lobed/doubled centrum were found in all the groups (Tables 3 and 4).
Fetal Visceral and Skeletal Observation a
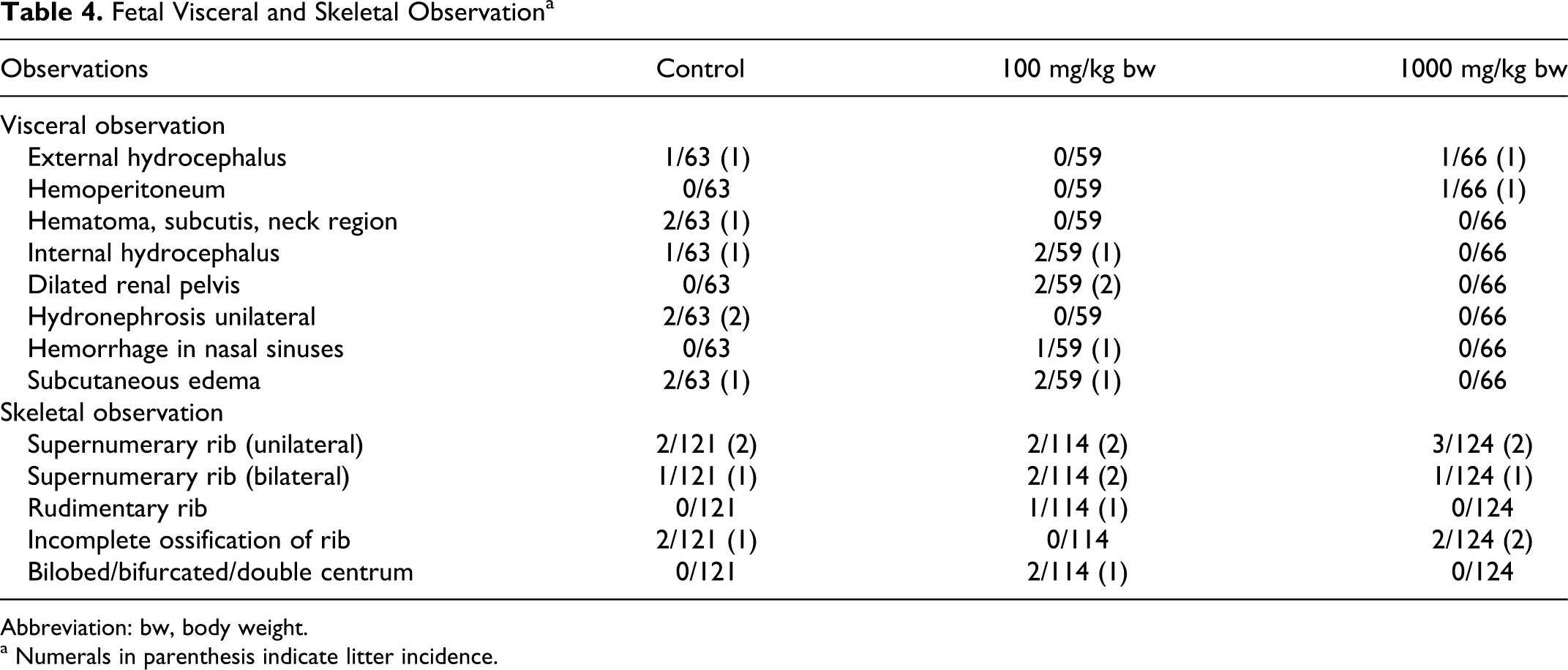
Abbreviation: bw, body weight.
a Numerals in parenthesis indicate litter incidence.
Discussion
Homobrassinolide is derived from campesterol in plants through the extension of cholesterol biosynthetic pathway, 31 whereas stigmasterol was considered a superior source for the synthesis of HBR. 37 Phytosterols are close analogues of animal cholesterol. Campesterol and sitosterol are formed by adding a methyl or ethyl group at carbon 24 of the cholesterol side chain. The dehydrogenation of the carbon 22 to 23 bond yields stigmasterol. Hydrogenation leads to the formation of campestanol and sitostanol. Daily consumption of PS is almost equivalent to that of cholesterol, that is, 150 to 400 mg, wherein the absorption was found to be slightly higher in women than men. 33,38 Phytosterol consumption is reported to alter cholesterol levels in humans and impair cholesterol metabolism, which is known to cause birth defects. 39 Plant sterols and stanols are also known to deter the absorption of β-carotenoids. 32 Phytosterol accumulates in steroid-secreting tissues (liver, adrenals, and gonads) of rats 40 –42 and can function as precursors of cortisol and sex steroids in humans and rats. 43,44
Few natural PGRs, PS, and phytohormones are reported to produce various toxic effects. Gibberellic acid has been shown to cause breast and lung adenocarcinomas in mice, chromosomal aberrations in human lymphocytes and mice, and estrogenic activity in rat. 25 Gibberellic acid also has been shown to alter rat liver enzymatic activities at 24 ppm when administered for 3 weeks. 25 Indole-3-acetic acid is a well-known teratogen, inducing cleft palate in both mice and rats at 500 mg/kg per day. 26 Policosanol did not cause teratogenic effects in either rats or rabbits at dosages up to 500 and 1000 mg/kg per day, respectively, nor did it induce reproductive toxicity in rats at dosages up to 500 mg/kg per day. 45 Sitosterol, a closely related PS, showed clear estrogenic effects in fish 27,28 and significant decrease in testicular weight and sperm count after long-term treatment in albino male rats. 29 In another study, Sitosterol caused an increase in absolute and relative testicular and epididymal weights as well as an increased number of implantation loss in rats. 30 Uterine glucose-6-phosphate dehydrogenase activity and uterine wet weight was increased in female rats due to β-sitosterol. 46,47
Homobrassinolide administered to rats at 10 and 50 μg intradermally decreased LDH activity in the brain and liver but elevated it in the kidneys and testes. Lactate dehydrogenase M deficiency has been reported to cause dystocia due to decreased adenosine triphosphate production. Frequent pain with increased serum pyruvate levels were observed during late pregnancies. 48 Changes in red blood corpuscles (RBC) and white blood corpuscles (WBC) counts, increases in hemoglobin and decreases in blood glucose, and changes in total cholesterol levels have been recorded as well. 31 Homobrassinolide as a PS is routinely consumed by animals and humans. It is estimated that, like other phytohormones, which in general are potent physiological mediators, HBR might be acting at multiple points and may have influence on metabolic events in animals. 31
The current investigation was carried out to determine the effect of “Combine,” a commercial HBR formulation, on the embryonic and fetal development of rats when administrated during organogenesis. All the parameters measured were found to be normal and no untoward embryonic or fetal effects were recorded. No maternal toxicity like body weight and feed consumption changes, toxicity signs, mortality, morbidity, and/or abortion was observed at either of the doses employed. Pre- and postimplantation losses, litter size, and number of live fetuses in the treated group were comparable with the concurrent control group. Mean fetal body weight and sex ratio showed no differences among groups. Few skeletal findings were recorded, but there were variants found scattered among all groups and hence not attributed to HBR treatment. Visceral examination revealed dilated renal pelvis in the 100 mg/kg body weight group (2 of 59) but not at 1000 mg/kg body weight, whereas hemoperitoneum was observed in 1 fetus at 1000 mg/kg body weight. Because there was a very low incidence of fetal findings along with almost insignificant litter incidence, these findings were not attributed to HBR treatment.
Conclusion
Considering the lack of maternal and embryo-fetal toxicity, it can be concluded that HBR is nonteratogenic at doses as high as up to 1000 mg/kg body weight in Wistar rats when dosed orally during GD 6 to 15.
Footnotes
Acknowledgments
We thank Dr B. N. Vyas (Godrej Agrovet Ltd) for his useful suggestions during preparation of this article. We also thank Dr P. V. Ramachandran, Ms T. N. Sathya, and N. Subashini for their help in the preparation of this article.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This study was sponsored by Godrej Agrovet Ltd as a part of preparing the regulatory dossier of this compound.