Abstract
Chlorogenic acid (CGA) is found in many plants that are used as medicinal substances in traditional Chinese medicine injectables (TCMIs). However, to date, there is controversy as to whether CGA is the major cause of TCMIs-related hypersensitivity administered intravenously. Therefore, the aim of this study was to evaluate the potential sensitization of CGA and structure–activity differences between its isomers using an intravenous exposure mouse model. The results showed that popliteal lymph nodes proliferation was significantly induced by CGA and its isomers. Both CGA and isochlorogenic acid A (iso-CGA A) significantly enhanced the secretion of trinitrophenyl (TNP) ovalbumin-specific immunoglobulin (Ig)G1; and iso-CGA B significantly induced TNP-specific IgG1, IgM, and IgG2b secreting. Furthermore, the results of quantitative structure–activity relationship analysis suggested that chemical structure factors, including atomic mass, electronegativity, atom shape and size, atom distribution, atomic weight, and atomic polarizabilities, the ionic currents, were significantly correlated with the potential sensitization of CGA and its isomers. In summary, when administered intravenously, the strength and type of sensitization may be correlated with structure differences in the CGA family.
Introduction
Chlorogenic acid (CGA), a natural phenolic acid which is found in many plants and is used as medicinal substances in traditional Chinese medicine injectables (TCMIs), has been widely applied in clinical practices for over 60 years. Chlorogenic acid has been demonstrated to be a potential therapeutic agent with a variety of biological activities including anti-inflammatory activity, 1 antioxidant activity, 2 antiviral activity, 3 antitumor activity, 4 and neuroprotective effects. 5
However, whether CGA has antigenic and allergenic activities that are known to be associated with systemic safety problems remains a heatedly debated topic. Some reports showed that it was an antigen found in plants that causes human atopic diseases, 6 –9 while other opposing views suggested that some other allergens, such as specific proteins, might be attributed to the cause of allergy. 10,11 In China, over the past decade, the safety of TCMIs has garnered increasing concerns, 12 for 90% of adverse reactions related to TCMIs arose from CGA-containing drugs; and most clinicians considered CGA as the cause of allergic diseases. 13 Therefore, there is a pressing need to identify the sensitization potential of CGA used intravenously for clinical application of TCMIs containing CGA.
More recent studies suggested that CGA might not represent a safety concern for humans, as it does not induce immunoreactivity following a single subcutaneous exposure in the reporter antigens popliteal lymph node assay (RA-PLNA) in BALB/c mice. 14 In RA-PLNA, levels of RA-specific antibody-forming cells (AFCs) are measured to analyze the T-cell-dependent immune activation. The activation of immune responses may be identified as markers of the induction of immunoglobulin AFCs. 15 –17 In addition, efficient immune responses to RA need cognate T-, B-cells interactions, and costimulatory adjuvant signals. 18 Therefore, potential sensitizing drugs or chemicals may induce formation of RA-specific AFCs. However, the subcutaneous administration of CGA into the footpad deviates from the normal routes of exposure, such as oral or intravenous exposure, and drug or chemical metabolism in vivo may impact its bioavailability. The critical step in the hapten hypothesis of drug hypersensitivity is the formation of adducts (drug–protein conjugates). 19 Furthermore, after CGA intravenous administration in rats, the major component in plasma was the parent compound that is highly reactive toward nucleophiles, such as glutathione, indicating that injected CGA may covalently bind to proteins and potentially lead to allergenic reactions. 20 Therefore, a proper method to evaluate the potential sensitization of CGA administrated intravenously is essential for its identification.
Therefore, by using an intravenous mouse model that we have developed in our previous study, 21 the primary issue addressed in this study was to explore the sensitization potential of CGA. Furthermore, we hypothesize that CGA or its isomers, including caffeic acid (CA), quinic acid (QA), crypto-chlorogenic acid (crypto-CGA), iso-chlorogenic acid A (iso-CGA A), and iso-chlorogenic acid B ([iso-CGA B] for structures see Figure 1), have a structure–activity relationship in immunostimulation. In these experiments, a low-molecular-weight compound (diclofenac sodium, DF) that had been demonstrated to induce immune-mediated drug hypersensitivity reaction (IDHR) in mice 16 was used as positive control compound, and phenobarbital (PB) was used as negative one. Immunoglobulin E and IgG1 antibodies in serum, PLNs weight index, and cellularity were determined in the studies, and anti-trinitrophenyl (TNP)-specific IgM, IgE, IgG1, IgG2a, and IgG2b antibody-secreting cells (AFCs) were also quantified as outcomes in tests for each groups, and quantitative structure–activity relationship (QSAR) was also analyzed in silico to find the potential structure correlation of these compounds in immune sensitization.
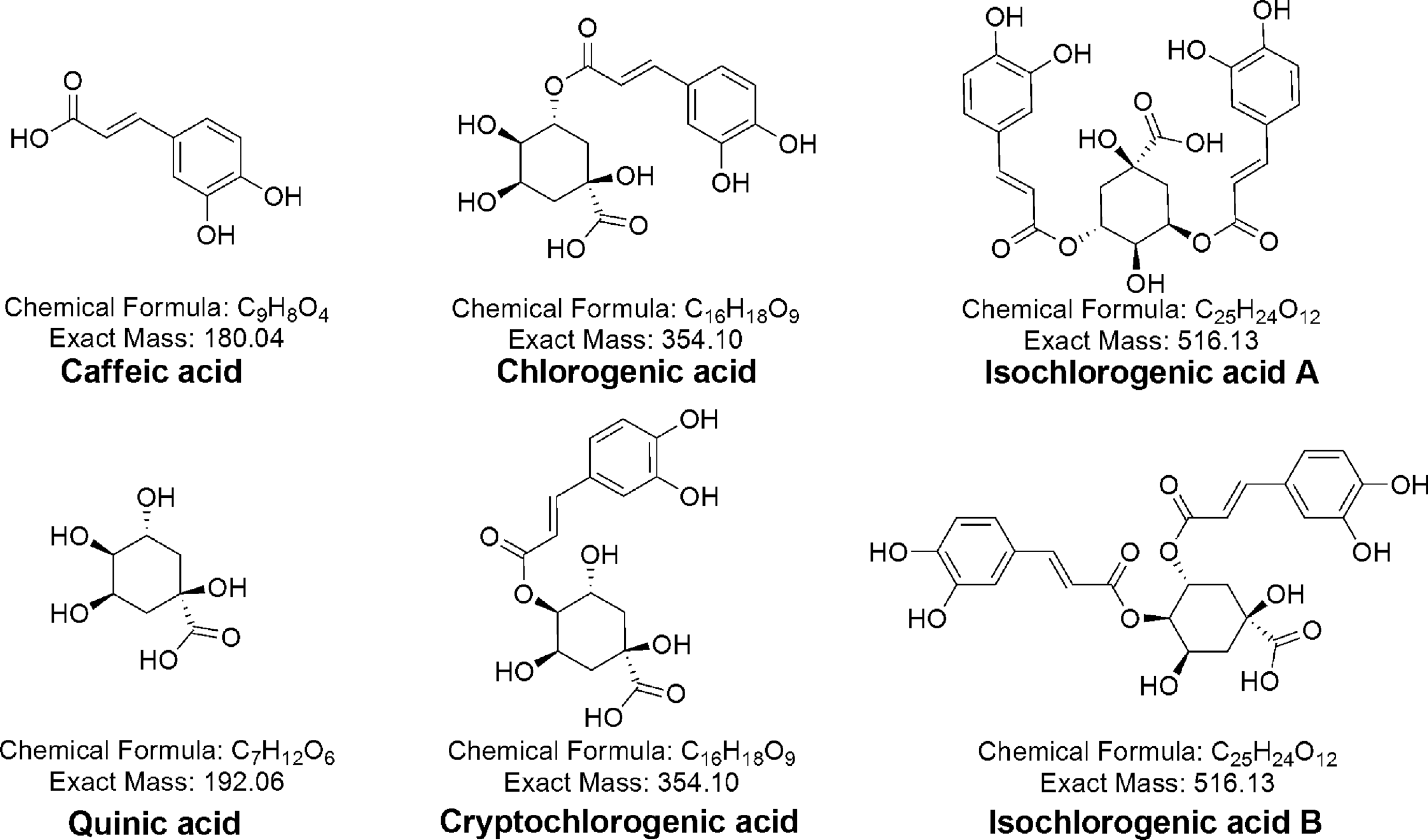
Chemical structure of chlorogenic acid (CGA) and its isomers.
Materials and Methods
Animals
Female, young adult (6- to 8-week-old, specific pathogen free) BALB/c mice (Barriered Animal Breeding Unit, Experimental Animal Department, Huazhong University of Science and Technology, Wuhan, China) were used for these studies. Food and water were available ad libitum. Special Diet Services Rat and Mouse no. 1 Maintenance Diet (Jinhaiyang Feed Co, Ltd, Wuhan, China) were used, composed primarily of cereal products. The mice were housed for 1 week in plastic cages (bisphenol A free) before testing and were maintained at 23°C ± 3°C, 50% ± 5% relative humidity with a 12-:12-hour light–dark cycle. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health, USA. The protocol was approved by the Committee on the Ethics of Animal Experiments of College of Pharmaceutical Sciences, Zhejiang University (Permit Number: 10-9771).
Chemicals and Reagents
Caffeic acid (≥97.8%), CGA (≥98.0%), crypto-CGA (≥98.0%), iso-CGA A (≥97.8%), and iso-CGA B (≥97.8%) were procured from Ronghe ScienceTechnology (Shanghai, China); DF (≥99.0%) were procured from National Institute for the Control of Pharmaceutical and Biological products (Beijing, China), PB (≥97.0%) from Toronto Research Chemicals Inc (Canada), and QA (≥95.0%) from Supelco Analytical (USA). MultiScreenHTS Filter Plates for immunoblot were purchased from Millipore Inc (Billerica, Massachusetts). Alkaline phosphatase (AP)-conjugated goat anti-mouse IgM, IgG1, IgE, IgG2a, and IgG2b were purchased from the Southern Biotech Inc (Alabama). The conjugates TNP-bovine serum albumin (TNP 26 -BSA) and TNP-ovalbumin (TNP 13 -OVA) were obtained from Biosearchtech Inc (Novato, California). The 5-bromo-4-chloro-1H-indol-3-yl) dihydrogen phosphate/nitro-blue tetrazolium (BCIP/NBT) AP substrate kit IV was purchased from VectaMount Inc (Burlingame, California).
Intravenous Exposure Mouse Model
The intravenous exposure mouse model was performed as we described previously. 21 With the exception of the negative control group (with normal sodium, NS), animals received an injection of TNP-OVA into both hind footpads on the first day. Then, each treatment group received an injection into the tail vein with the corresponding compounds (20 mg/kg) in NS on the third, fifth, and seventh day. On the ninth day, each treatment group (including TNP-OVA control) animal was challenged again with TNP-OVA injected into footpads. On the 11th day, mice were sacrificed by cervical dislocation and the draining PLNs were extracted.
Determination of PLN Weight Indexes and Cell Numbers
The weight of the PLNs was determined in each mouse, and the weight index (index = weight of PLNs/mouse body weight) was calculated. Then, the PLNs were ruptured with a 26-gauge needle and the cells were extracted by gentle agitation, washed with 1% BSA–PBS by centrifugation, and resuspended in 1% BSA–PBS and counted with a Coulter Counter. Finally, the lymph node cell numbers were presented for each PLN.
The ELISPOT Assay for TNP-Specific AFCs
Aliquots of 50 µL, from the cell suspension (1 × 106/mL), were placed in 1 well of the 96-well ImmobilonR-P bottom plate, which was precoated with 10 µg/mL TNP-BSA in PBS overnight at 4°C. 22 After being incubated at 37°C for 4 hours, the plates were washed to remove cell debris and then were incubated for 18 hours at 4°C with diluted AP-conjugated goat anti-mouse: IgM (1:5000), IgG1 (1:1000), IgE (1:500), IgG2a (1:2500), or IgG2b (1:2500) in 1% BSA–PBS. After washing with PBS and Tween-20, respectively, wells were incubated with BCIP/NBT substrate solution to visualize TNP-specific antibody spots. The membranes were dried, and the number of IgM, IgG1, IgG2b, IgG2a, or IgE AFCs were determined by Immunospot detector (Immonospot IV, C.T.L, Cleveland, OH, USA). The AFCs were reported as the number of AFCs per 106 PLN cells. Popliteal lymph node cell numbers determined previously were used to calculate the number of AFCs per 106 recovered PLN cells.
The ELISA Assay for Antibodies in Serum
At sacrifice, blood was collected from orbital venous and centrifuged at 1500
The QSAR Analysis in Silico
The QSAR models were established to find the relationships between compound structure and immune sensitization. All chemical structures studied in the work are presented in Figure 1. In QSAR models, chemical structures are presented with the numerical form of molecular descriptors. By using the software of E-DRAGON version 1.1, 23 a set of 929 molecular descriptors per compound were calculated and used to describe the chemical diversity; these descriptors encoded different aspects of molecular structures including constitutional, electronic, topological, and so on. After calculation of molecular descriptors, Fisher weights of these descriptors were obtained to select significant descriptors. Enhanced replacement method (ERM), a genetic algorithm approach of replacement method proposed by Mercader et al, 24 was used to sort out the pertinent descriptors determining the variation in the biologic activity and to construct the model equations. The prediction R2 (explained variance), root mean square error (RMS), and standard error for regressions (S) were used as model selection criterion. The predictive power of the models was evaluated using leave-one-out cross-validation method (LOOCV). By using LOOCV, at each time, only 1 sample was selected as test set, while all others were selected as training set to generate a QSAR model, and the process was repeated until every sample has been selected as test set once.
Statistical Analysis
Statistical analyses were performed with SPSS11.5 Statistical software (SPSS Inc, USA). All data were expressed as mean ± standard error of the mean and were analyzed using the Student
Results
The PLN Weight Indexes and Cell Numbers
On the 11th day, PLNs were isolated and weights were determined for weight index calculation, and cell numbers were counted. Cell proliferation in the PLNs was indicated by using PLN weight indexes (Figure 2A) and cell numbers (Figure 2B). Compared with the control, CGA and its isomers (except CA and QA) showed significant enhancement in weight indexes and cell numbers. Similar significant results were induced by the positive control compound (DF). In contrast, as a negative compound, PB failed to induce any significant increase in PLN weight indexes and cellularity.
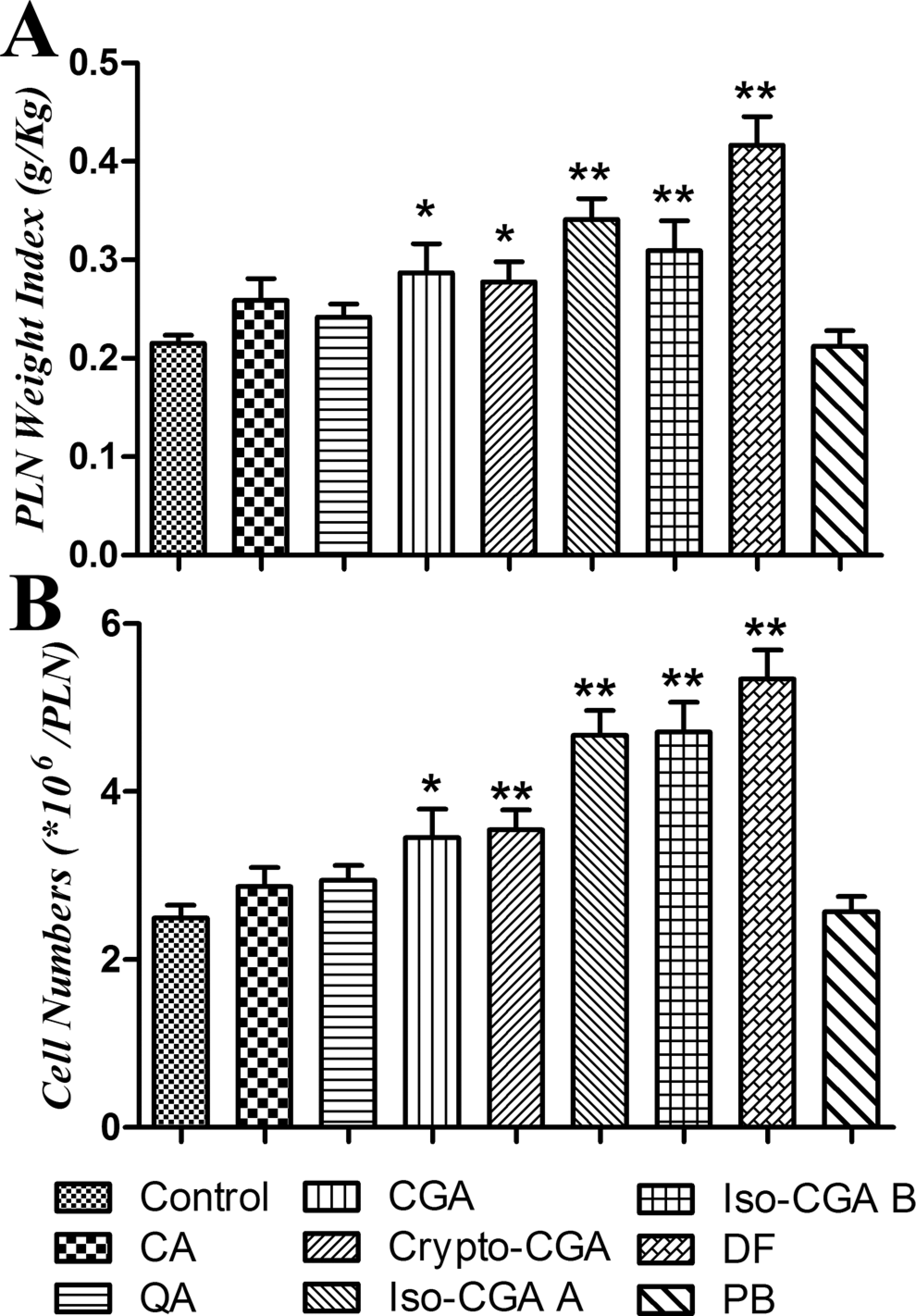
Weight indexes and cell numbers presented in popliteal lymph nodes (PLNs). Bars represent group means ± standard error of the mean (SEM) of 8 mice per group. Statistically significant differences are indicated by *
The TNP-Specific AFC Numbers in PLNs
An enzyme-linked immunosorbent spot (Elispot) assay was used to detect the TNP-specific AFCs in PLNs. Compared with the control, only iso-CGA A and iso-CGA B significantly increased the IgM AFC numbers (Figure 3A). No compounds induced significant increase in TNP-specific IgE levels (Figure 3B). The TNP-specific IgG1ASCs were significantly increased in CGA, iso-CGA A, iso-CGA B, and DF (Figure 3C). Although crypto-CGA only moderately enhanced the IgG1 AFCs, it significantly induced IgG2a production in comparison to the control (Figure 3D). In addition, only iso-CGA B was observed to induce significant increase in the IgG2b AFCs (Figure 3E). No marked reaction was observed in the negative control (PB) group.
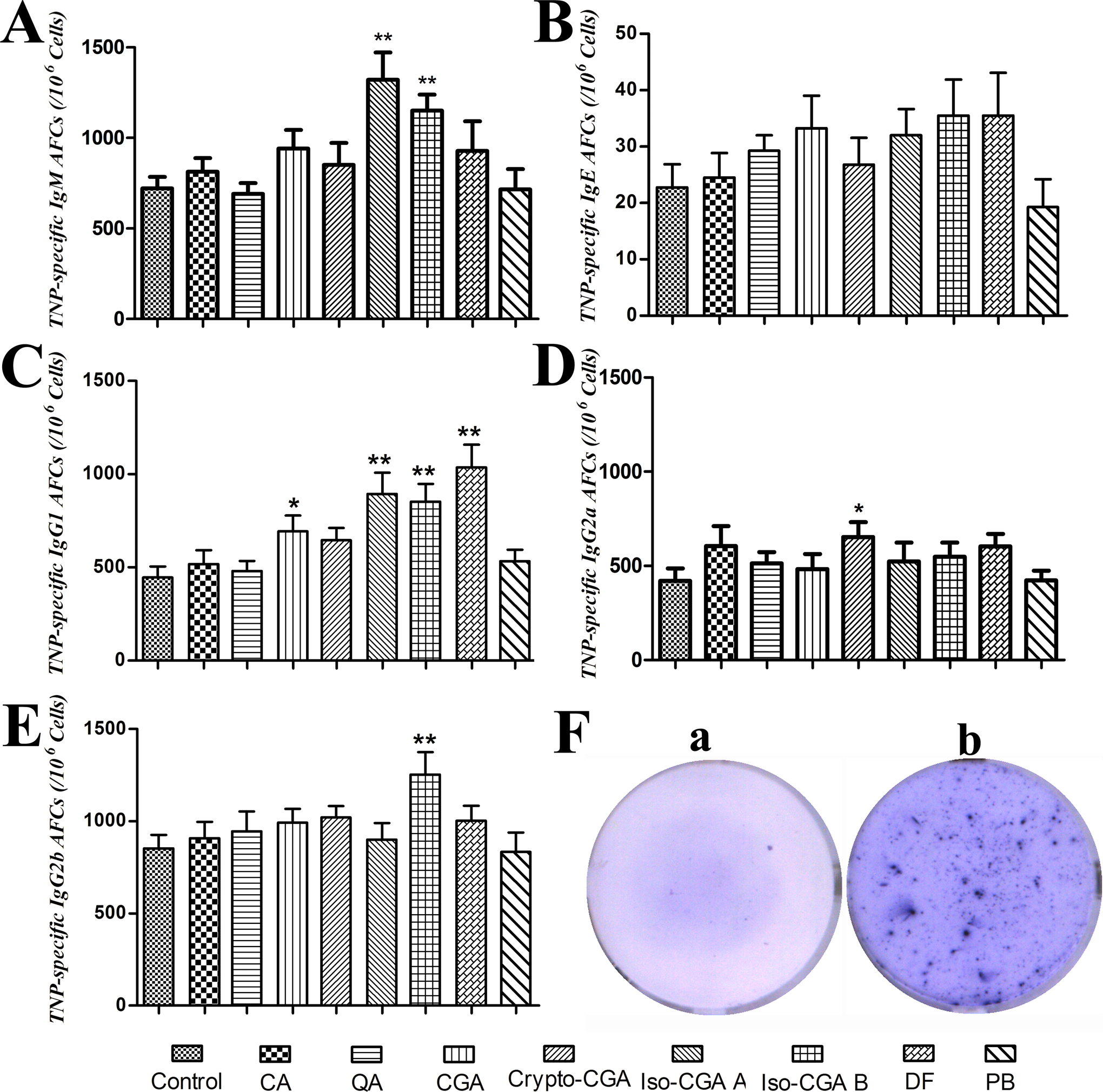
Trinitrophenyl (TNP)-specific antibody-forming cells (AFCs) in popliteal lymph nodes (PLNs) in response to specific compounds in intravenously treated mice. The TNP-specific IgM (A), IgE (B), IgG1 (C), IgG2a (D), and IgG2b (E) were measured using the Elispot assay. Bars represent AFCs/106 PLN cells (mean ± standard error of the mean [SEM]) of 8 mice per group. Typical anti-TNP-specific negative and positive immunospots appeared in F-a and F-b, respectively. *Statistically significant differences (
The TNP-Specific Antibodies in Serum
On the 11th day, mice were sacrificed and the serum was collected for the IgE (Figure 4A) and IgG1 (Figure 4B) antibodies analysis using ELISA. As shown in Figure 4B, serum IgG1 levels were significantly enhanced when compared to the control after crypto-CGA, iso-CGA A, and iso-CGA B were introduced intravenously; a similar result was observed in the positive control (DF). In addition, the IgE levels were moderately enhanced in CGA, iso-CGA A, iso-CGA B, and DF but not significantly with respect to the control. In contrast, no significant changes were observed in any of these parameters. Detail data of serum IgE and IgG1 were shown in Table 1.
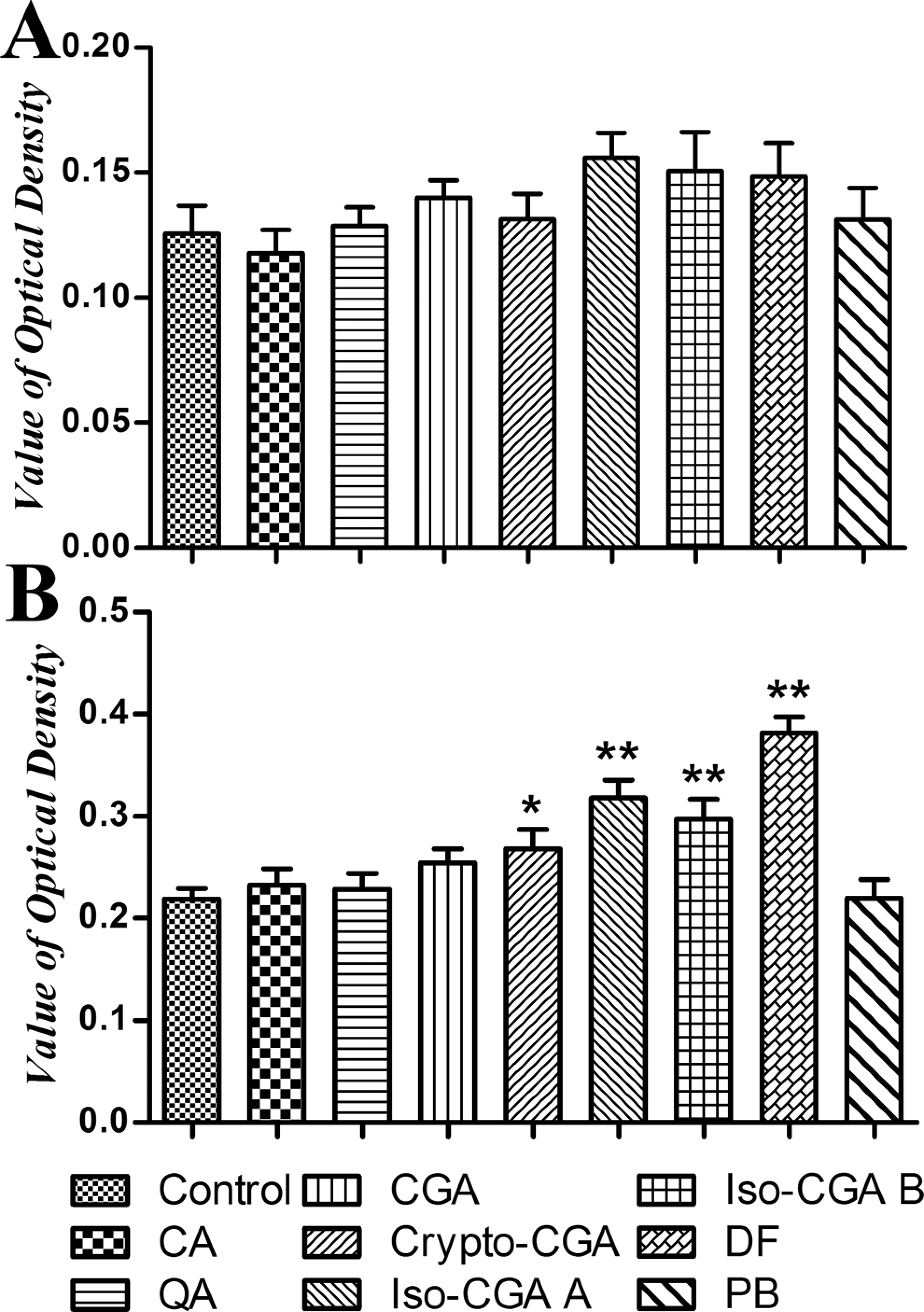
The TNP-specific antibody levels in serum after intravenous treatment. On 11th day, serum was collected, and serum IgE (A) and IgG1 (B) were determined with ELISA. The antibody levels were showed in value of optical density. Statistically significant differences are indicated by *
The Results of TNP-Specific Antibody Levels in Serum.
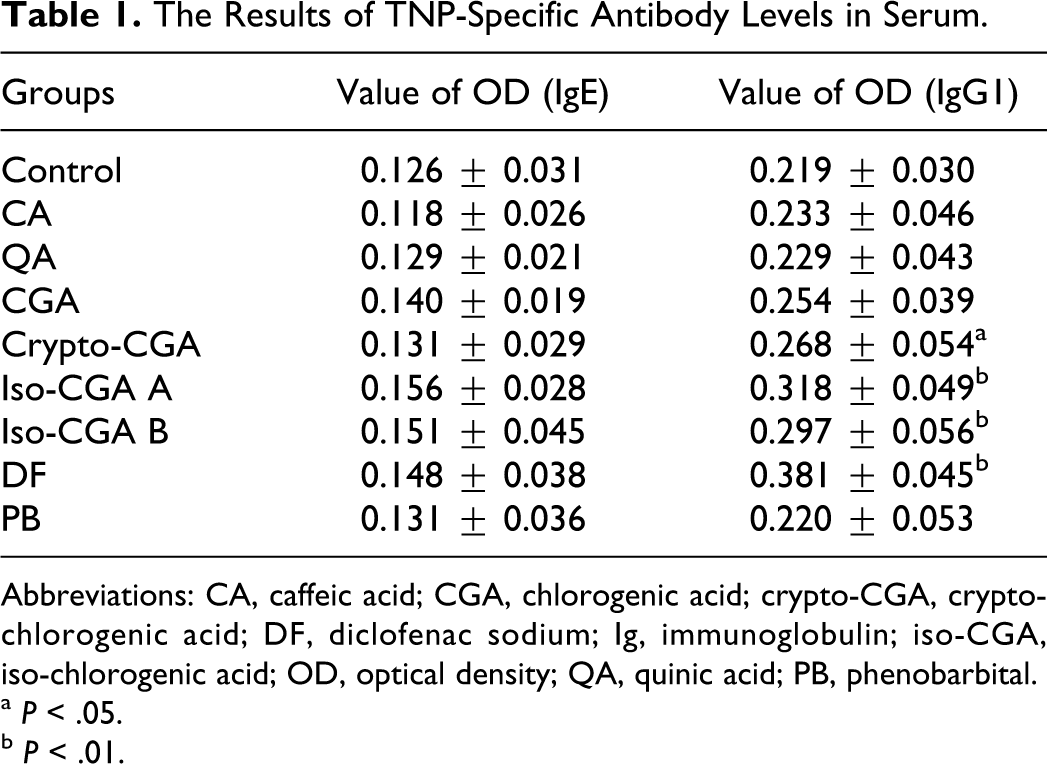
Abbreviations: CA, caffeic acid; CGA, chlorogenic acid; crypto-CGA, crypto-chlorogenic acid; DF, diclofenac sodium; Ig, immunoglobulin; iso-CGA, iso-chlorogenic acid; OD, optical density; QA, quinic acid; PB, phenobarbital.
a
b
The QSAR Analysis in Silico
Four QSAR models were established to correlate the chemical structure with IgM, IgG1, IgG2a, and IgG2b, respectively. For each model, 2 variables were selected by using ERM, validated with LOOCV method. The resulting regression equation and graphical representation of each model for the training sets (fitting) and test sets (LOOCV) were illustrated in Figure 5, with the statistical results compared and illustrated the bottom parts. In this QSAR study, methods in combination with ERM demonstrated acceptable performance in predicting IgM, IgG1, IgG2a, and IgG2b, as evidenced by the high prediction R2 and low RMS and S obtained in LOOCV validation.
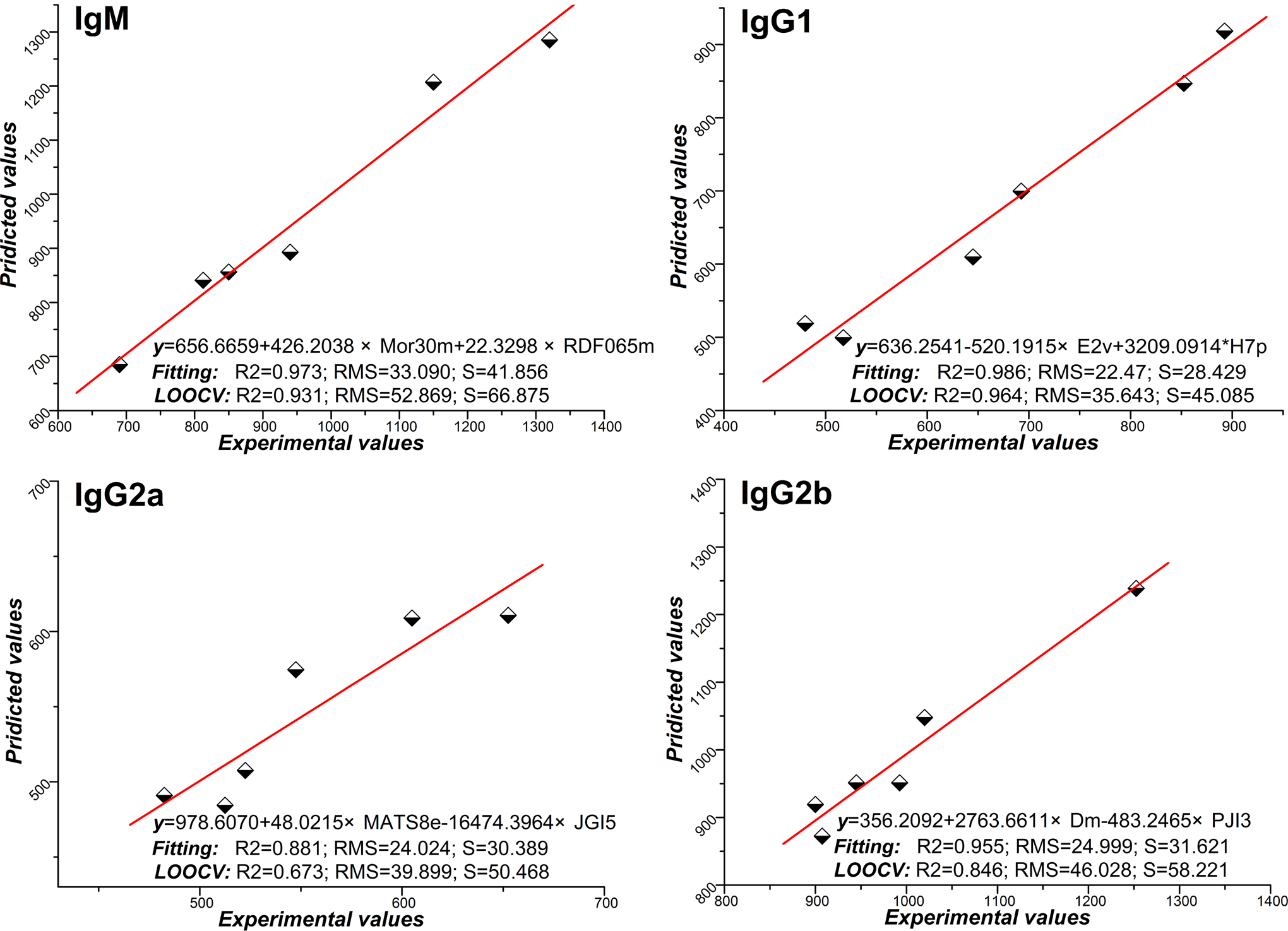
Experimental versus predicted values for enhanced replacement method (ERM), with the straight line indicating a perfect correlation of the predicted and experimental value in IgM, IgG1, IgG2a, and IgG2b. Model selection criterion
Discussion
For a long time, there have been 2 diametrically opposed views on whether CGA has antigenic and allergenic activities in IDHR. 7,10 Recently, a study suggested that CGA failed to produce any reactions in PLN in mice and that CGA may not be the major reason for IDHR caused by TCMIs. 25 However, theories of drug metabolism suggested that an immune response of small molecule drugs could initiate the formation of stable adducts with proteins. 26 Protein adducts may be formed either by direct chemical reaction or by generation of electrophilic metabolites, 27 which thus act as an antigenic signal to induce the immune response. 28 The metabolism of CGA subcutaneously injected into footpad may be different from that of intravenous injection. In circulation, with the exception of CGA parent compound, extensive metabolism of CGA occurs and is highly reactive to nucleophiles. 20 It is still unclear whether IDHR can be induced by CGA or its metabolites in circulation, which may be an allergen or hapten. Therefore, evaluating the potential sensitization of CGA when treated intravenously becomes increasingly important and necessary for CGA safety assessment, especially for TCMIs containing CGA.
Accordingly, the development of a valid model for CGA-induced hypersensitivity assessments in an intravenously injected route is critical. Based on the RA-PLNA, a particularly useful tool for the systemic sensitizing potential assessment, 29 our previous studies have demonstrated that an intravenous exposure mouse model, which was validated by 3 positive and 1 negative compounds, appears to be a potentially useful tool for drug-induced hypersensitivity screening via intravenous exposure. 21 In this study, by using the mouse model, the potential sensitization of CGA and its isomers via intravenous exposure were further determined.
Our results showed that, CGA and its isomers (except CA and QA) may significantly enhance the weight indexes and cell numbers in PLN compared with the control. The TNP-specific AFCs were significantly skewed toward the IgG1 isotype by CGA, iso-CGA A, and iso-CGA B; moreover, the increase in IgG1 production seems to be correlated with the structure differences. Thus, based on these results, CGA and its isomers (crypto-CGA, iso-CGA A, and iso-CGA B) might cause immunostimulation via intravenous treatment.
Cytokine fingerprinting has been found to be helpful in identifying a reaction and could provide the mechanistic information on drug-induced hypersensitivity. 30,31 Indirectly, the differentiation of immune responses may be analyzed by RA-specific AFCs isotypes associated with Th1 and Th2. 30,31 Therefore, RA-specific AFCs were tested for Th1/Th2 immune responses investigated in this study. In our results from the present study, after intravenous injection of CGA and iso-CGA A, the numbers of TNP-specific IgG1 were significantly increased while there were no increases in IgG2a and IgG2b AFCs, suggesting that a potential Th2-type immune response was induced. Crypto-CGA, however, was shown to induce potential Th1 response (with IgG2a increase). Furthermore, iso-CGA B seemed to induce a more mixed Th1/Th2 immune response (with IgG1, IgM, and IgG2b increase). However, further research should be carried out to support the mechanistic information of Th1/Th2 immune response with cytokine profiles.
Based on the results of cytokine fingerprinting in TNP-specific AFCs, QSAR analysis was carried out in silico to explore the structural information of CGA and its isomers correlated with sensitization. Molecular descriptors were used as the input required for training the biologic activity–predictive models. Some statistical parameters concerning the fitting and predictive performance of the models characterize the selected QSAR equations. Mor30m (signal 30/weighted by atomic masses, representing structures based on electron diffraction) 32 and RDF065m (radial distribution function −6.5/weighted by atomic masses, representing specific information in a certain 3-dimensional [3D] structure space) 32 descriptors dominate the equation of IgM AFCs formation, indicating it is directly related to the 2 descriptors. For IgG1 AFCs induction, E2v (a topological index that belongs to weight holistic invariant molecule, representing the 3D molecular structure in terms of size, shape, symmetry, and atom distribution) 33 and H7p (the molecular influence matrix H7p, belonging to H-GETAWAY descriptors, encoding information about molecular shape, size, and atom distribution) 34 descriptors were significantly correlated, which concern with the Th2-type effect in immune response. While a Th1 effect of immune response reflected by IgG2a and IgG2b seems to be related to MATS8e (Moran autocorrelation −lag 8/weighted by atomic Sanderson electronegativities, belonging to 2D autocorrelation descriptors and referring to the cationic part of the ionic currents), 24 JGI5 (mean topological charge index of order 5, belonging to Galvez topological charge indices and evaluating the charge transfers between pairs of atoms and the global charge transfers in the molecule), 35 Dm (D total accessibility index/weight by atomic masses, belonging to weight holistic invariant molecule, relevant to the distribution of the atomic masses), 36 and PJI3 (Petijean shape index, a topological anisometric descriptor, defined as where R and D are the topological radius and the topological diameter) 37 descriptors in QSAR analysis. Therefore, from these significant molecular descriptors, the structural factors, including atomic masses, electronegativity, atom shape and size, atom distribution, atomic weights, atomic polarizabilities, and the ionic currents, are significantly correlated with the potential sensitization of CGA and its isomers via intravenous exposure.
In conclusion, CGA and iso-CGA A are shown to be potential sensitizers that induce immune response via intravenous treatment. Furthermore, QSAR analysis suggests that the strength and type of these potential immune effects are correlated with the structural differences. These findings indicate that TCMIs that contain CGA and its isomers might also trigger IDHR. Collectively, the results of this study may suggest more careful applications of the CGA family and TCMIs that contain them in clinical treatments to avoid further medical complications.
Footnotes
Acknowledgments
The authors would like to thank Prof Xiongwen Wu and Dr Wei Sun (Department of Immunology, Tongji Medical College, Huazhong University of Science and Technology) for their excellent technical assistance, Nick Cheng (Georgia Institute of Technology, USA), and Don Ding (NCTR/FDA, USA) for their help during the manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National S&T Major Project (2012ZX09505001-001) and the Fundamental Research Funds for the Central Universities (2012FZA7003).
