Abstract
Stearyl heptanoate is an ester of stearyl alcohol and heptanoic acid that functions in cosmetics as a skin conditioning agent and is in the general class of chemicals called stearyl alkanoates. Stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate are stearyl alkanoates with similar chemical structures, toxicokinetics, and functions in cosmetics. These water-insoluble stearyl alkanoates, when metabolized, yield stearyl alcohol and a corresponding fatty acid. The available information supports the safety of all of the related stearyl alkanoates. The Expert Panel concluded that stearyl heptanoate, stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate are safe in the present practices of use and concentration.
Keywords
Introduction
Stearyl heptanoate, the ester of stearyl alcohol and the fatty acid, heptanoic acid, in the general class of chemicals called stearyl alkanoates, was previously reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel. 1 The conclusion that stearyl heptanoate is safe for use in cosmetics in the present practice of use and concentration was based on the available data, including that this ingredient is an off-white waxy solid, soluble in ethanol, mineral oil, and other organic solvents but not soluble in water. This ingredient does not absorb light in the UVA or UVB region of the spectrum. Oral LD50 values of >5 g/kg were reported for male and female Sprague-Dawley rats. Because significant systemic exposure was not expected, given the chemical/physical properties, the focus of the safety assessment was on dermal exposure. Stearyl heptanoate was not a significant dermal irritant and was not a sensitizer in both animal and human studies. Formulations containing 0.7% or 1.5% stearyl heptanoate were not primary ocular irritants in animal studies. This ingredient was not genotoxic in bacterial and mammalian test systems.
No new safety test data were available for stearyl heptanoate, which supported the view that the previous conclusion was still valid for this single ingredient. In addition, however, the CIR Expert Panel determined that the available safety data described above for stearyl heptanoate can be extrapolated to support the safety of other stearyl alkanoates, including stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate. This extrapolation is predicated on similar chemical and physical properties which suggested that little dermal penetration of stearyl alkanoates would occur. Were there any systemic exposure, stearyl alkanoates can be metabolized by esterases in the body to stearyl alcohol and the related fatty acid. 2 –4
Other Relevant Safety Assessments
The CIR Expert Panel safety assessment of stearyl alcohol concluded that this alcohol is safe in the present practices of use and concentration in cosmetics. 5 That safety assessment described stearyl alcohol as a white, waxy, practically inert solid with a faint odor. Stearyl alcohol had a wide variety of uses in pharmaceutical and food industries, in addition to cosmetics. Stearyl alcohol was soluble in organic solvents but not soluble in water. Stearyl alcohol was not a significant acute oral toxicant, nor was it toxic in repeated-dose dermal toxicity studies. Stearyl alcohol was poorly absorbed from the gastrointestinal tract. In rabbit irritation tests, these alcohols produced minimal ocular irritation and minimal-to-mild primary cutaneous irritation. Tests of product formulations in humans demonstrated low potential for significant skin irritation or sensitization. Stearyl alcohol was not genotoxic in a bacterial test system, nor was it a tumor promoter when tested with DMBA.
In addition, the CIR Expert Panel assessed the safety of oleic, lauric, palmitic, myristic, and stearic acids, concluding that these ingredients were safe in present practices of use and concentration in cosmetics. 6 That safety assessment noted that these fatty acids were soluble in organic solvents but insoluble in water. Metabolism of fatty acids involved serial oxidation and reduction reactions yielding acetyl-CoA. Fatty acids were not acute toxicants. While dietary studies confirmed the correlation between dietary saturated fatty acid intake and the incidence of atherosclerosis and thrombosis, these data were not considered relevant to the safety of fatty acids in cosmetics. Dermally applied, these fatty acids were not significantly irritating in formulations, nor were they sensitizers, even in maximization studies. The weight of evidence suggested that fatty acids were not genotoxic, and they were not carcinogenic. Fatty acids were nonirritating in clinical primary and cumulative irritation studies and were nonsensitizing and nonphotosensitizing in clinical repeated insult patch tests.
Chemistry
Definition and structure
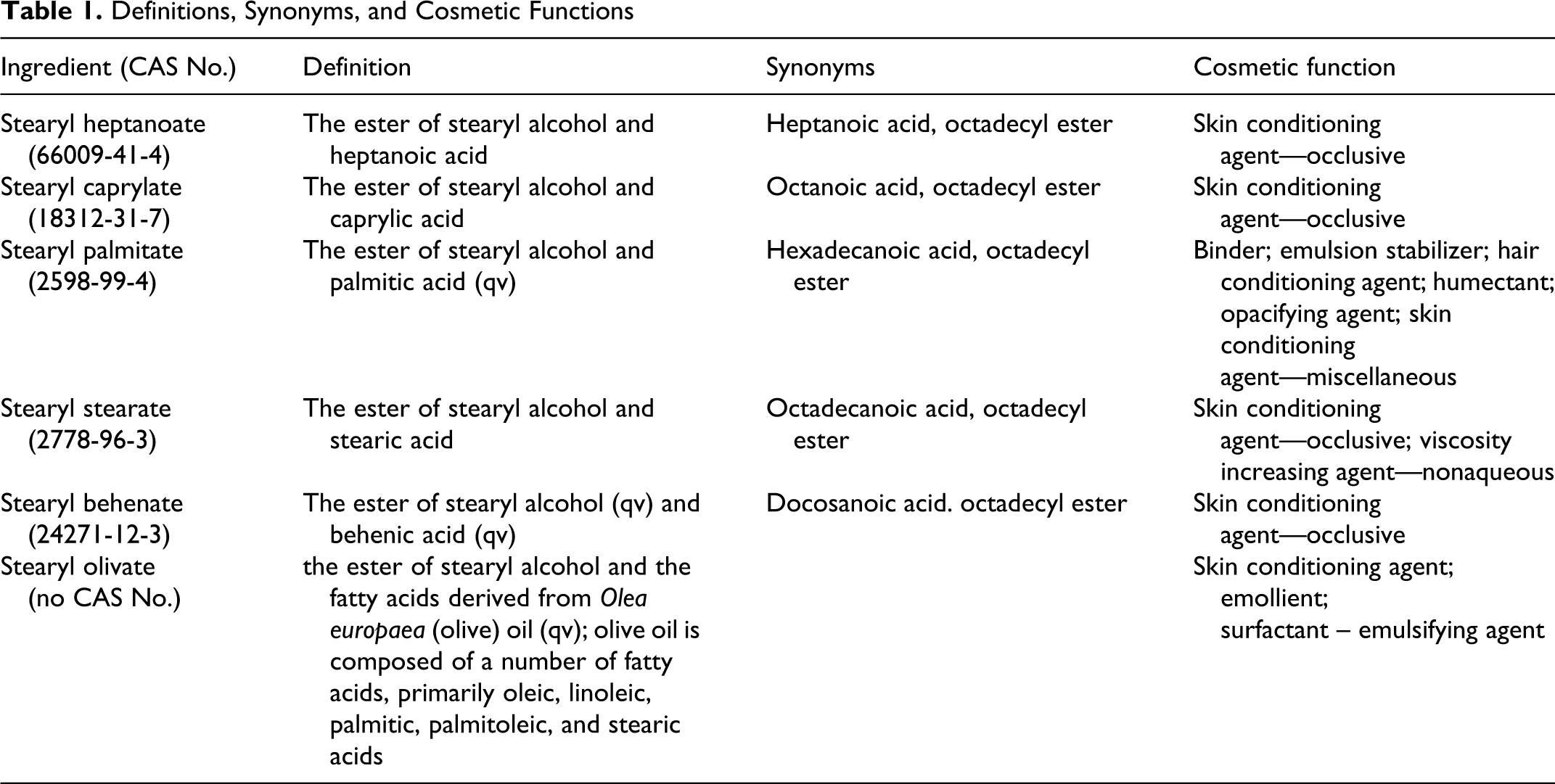
The definition, synonyms, and function in cosmetics for stearyl heptanoate, stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, 7 and stearyl olivate 8 are given in Table 1. The structural map of these chemicals is depicted in Figure 1.
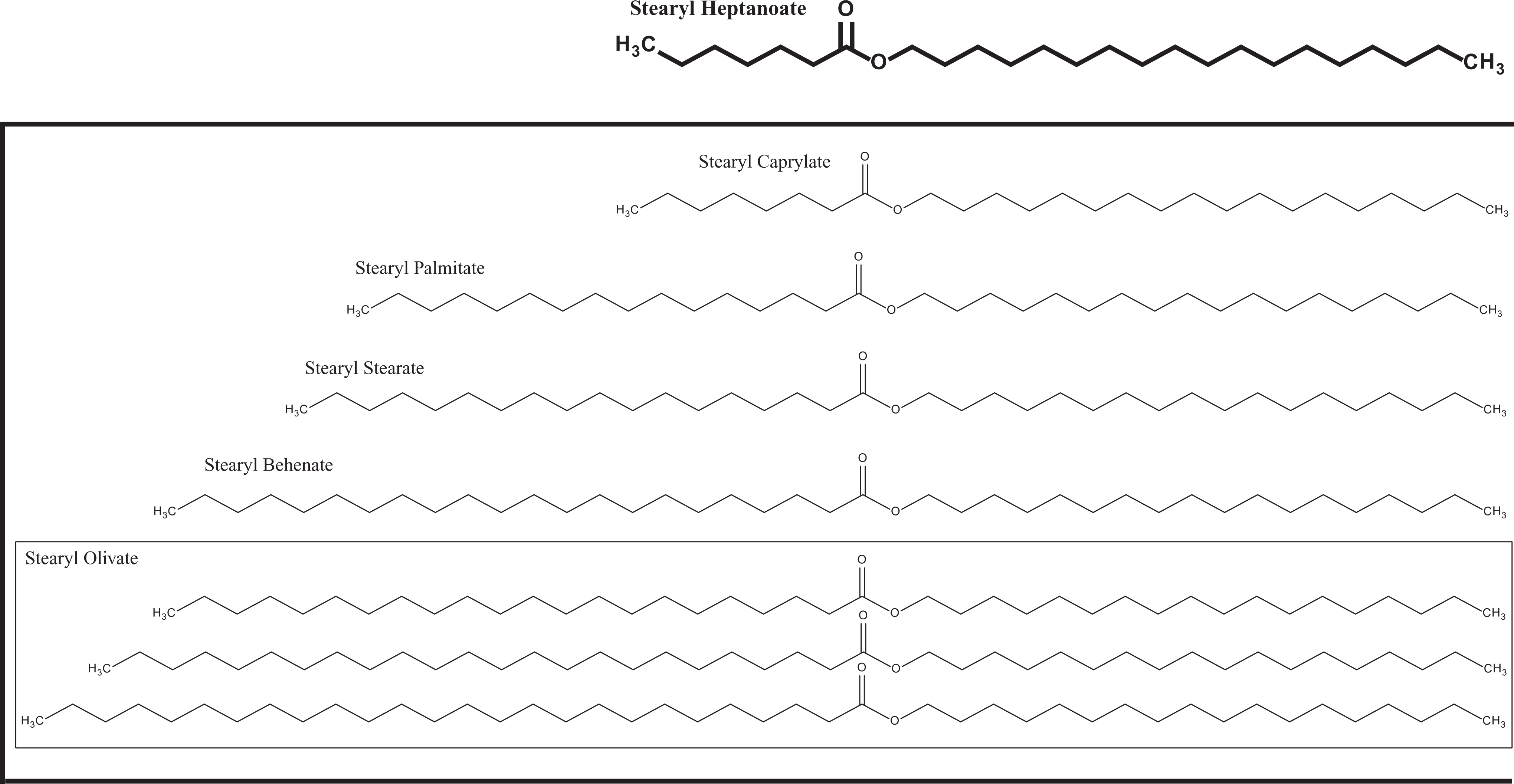
Figure map of stearyl alkanoates.
Definitions, Synonyms, and Cosmetic Functions
Use
Cosmetic
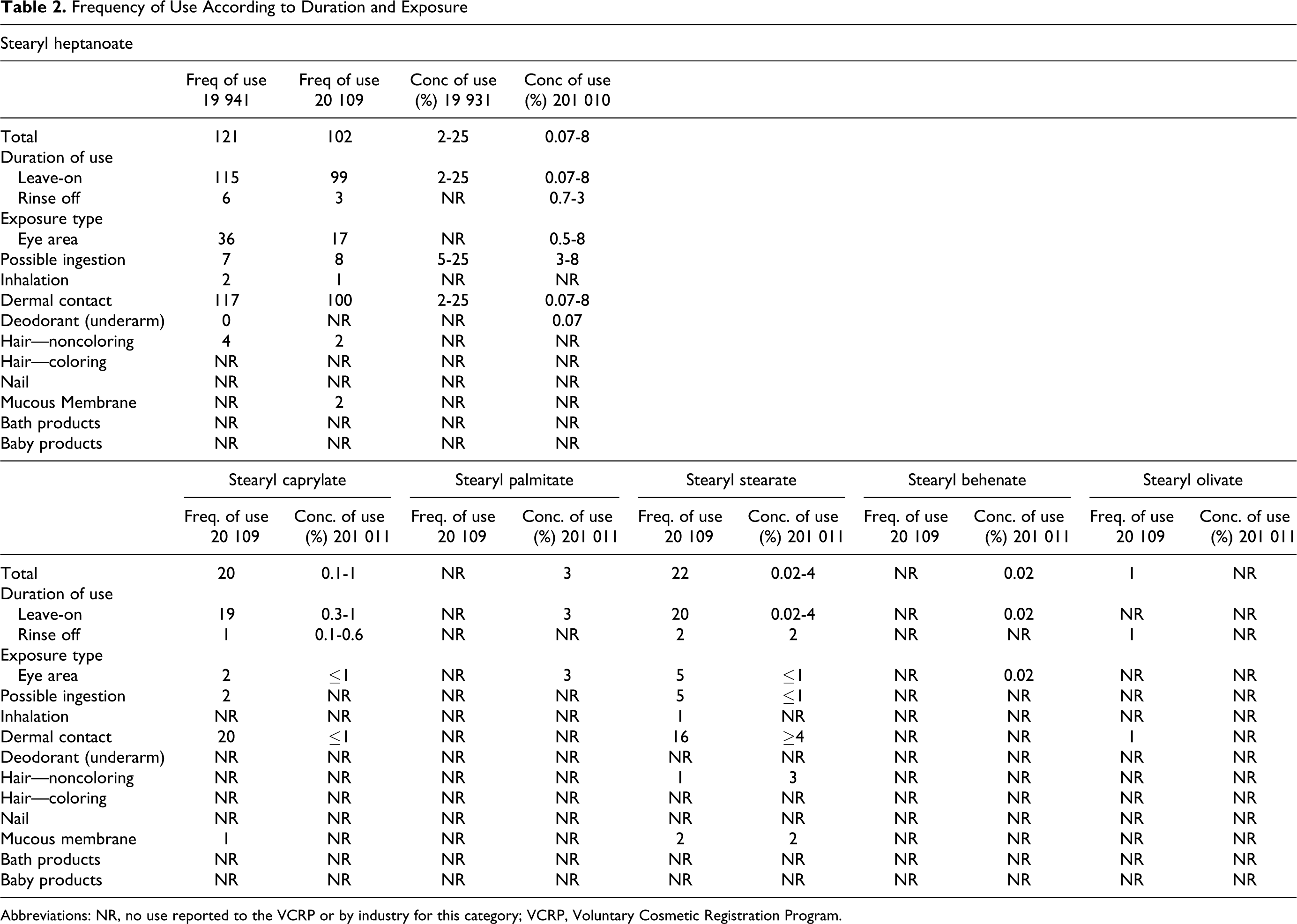
All of the stearyl alkanoates included in this report function as skin conditioning agents, with some having additional uses 7 (see Table 1). According to the information supplied to the Food and Drug Administration (FDA) as part of the Voluntary Cosmetic Registration Program (VCRP), at the time of the original assessment, stearyl heptanoate was used in 121 cosmetic formulations in 1994. 1 Concentrations of use were not reported to the FDA at the time of the original safety assessment, but unpublished data reported use at up to 25%. Since then, the frequency as well as the concentration of use has decreased. Recent VCRP data indicate that stearyl heptanoate is used in 102 cosmetic formulations, 9 and results of a survey of current use concentrations conducted by the Personal Care Products Council (Council) indicate that stearyl heptanoate is now used at concentrations of 0.07% to 8%. 10 Current and historical use data are given in Table 2.
Frequency of Use According to Duration and Exposure
Abbreviations: NR, no use reported to the VCRP or by industry for this category; VCRP, Voluntary Cosmetic Registration Program.
It was reported to the VCRP that stearyl caprylate and stearyl stearate are used in 20 and 22 cosmetic formulations, respectively, and stearyl olivate is used in 1 cosmetic formulation. 9 According to the concentration of use survey conducted by the Council, stearyl caprylate is used at concentrations of 0.1% to 1% and stearyl stearate is used at concentrations of 0.02% to 4%. 11 No uses were reported for stearyl olivate in response to the survey. Stearyl palmitate and stearyl behenate are not reported within VCRP data. 9 However, according to the survey of use concentrations conducted by the Council, stearyl palmitate and stearyl behenate are used in formulations applied to the area around the eyes at concentration of 3% and 0.02%, respectively 11 (see Table 2 for use details).
Stearyl stearate is used in products that may be inhaled, and effects on the lungs that may be induced by inhaled products containing these ingredients are of concern. The aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da , defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract. 12,13
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 14 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range, and the mean particle diameter 15 in a typical aerosol spray has been reported as ∼38 µm. Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
Stearyl heptanoate, stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate are listed in the European Union inventory. 16 Stearyl palmitate and stearyl stearate are included in the list of indirect additives used in food contact substances. 17
Summary
Stearyl heptanoate is an ester of stearyl alcohol and heptanoic acid that functions in cosmetics as a skin conditioning agent. Stearyl heptanoate is in the general class of chemicals called stearyl alkanoates. Stearyl heptanoate previously was reviewed by the CIR Expert Panel, with the conclusion that this ingredient was safe as used. A search of the literature published since that report was issued did not find any new data. The Panel agreed that stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate were similar in chemical and physical properties to stearyl heptanoate and have similar functions in cosmetics. While dermal penetration of such water-insoluble substances was unlikely, the expected esterase hydrolysis of stearyl heptanoate to stearyl alcohol and heptanoic acid, where systemic exposure to occur, was a pattern expected for all the stearyl alkanoates and prompted consideration of the previous CIR safety assessments of stearyl alcohol and fatty acids by the Panel. In 1994, it was reported to the FDA that stearyl heptanoate was used in 121 cosmetic formulations and, according to unpublished data, it was being used at up to 25%. As of 2010, the frequency of use has decreased to 102, and, according to an industry survey, use concentrations have decreased and are now ≤8%. It has been reported to the FDA that stearyl caprylate and stearyl stearate are used in 20 and 22 cosmetic formulations, respectively, and that stearyl olivate is used in 1 cosmetic formulation. According to an industry survey, stearyl caprylate is used at concentrations of 0.1% to 1%, stearyl stearate is used at 0.02% to 4%, and stearyl olivate is not reported to be used. Stearyl palmitate and stearyl behenate are not reported to be in use according to VCRP data, however, industry data reports that these ingredients are used in eye products at concentrations of 3% and 0.02%, respectively.
Discussion
The CIR Expert Panel considered that stearyl heptanoate was but one example of a broader group of stearyl alkanoates used in cosmetics. Others include stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate. Stearyl alkanoates have similar chemical and physical properties and have similar functions in cosmetics. These chemical and physical properties suggested that little dermal penetration would occur, and in case of any systemic exposure, they all would be metabolized by esterases to stearyl alcohol and the related fatty acid.
Stearyl alcohol is the esterase metabolite common to all of the stearyl alkanoates The other esterase metabolites for these ingredients would be the 5 specific fatty acids, heptanoic acid, caprylic acid, palmitic acid, stearic acid, behenic acid, and the fatty acids derived from Olea europaea (olive) oil. While these fatty acids represented various chain lengths, it was reasonable to conclude that the variation in fatty acid chain lengths would not play a role in toxicity.
Previous CIR assessments on stearyl heptanoate, stearyl alcohol, and fatty acids were considered, since new published data were not found for stearyl heptanoate or any of the other stearyl alkanoates. The information from these previous assessments did not raise any toxicological concerns. Also, these ingredients were at most mildly irritating to the skin, and they did not appear to be sensitizers. Palmitic acid and stearic acid were not phototoxic. Stearyl heptanoate produced slight-to-moderate comedogenicity in rabbit ears but was not a comedogenic for humans. At most, mild ocular irritation was seen in testing with any of these ingredients.
In the original safety assessment of stearyl heptanoate, the Panel noted that although animal irritation testing was done at 100%, sensitization testing was performed using a maximum concentration of only 1.5% stearyl heptanoate. The Panel concluded that all the available data indicated that stearyl heptanoate would not be a sensitizer. The same would apply to the stearyl alkanoates now included in this re-review. In the original safety assessment, it was also noted that mild ocular reactions were seen in ocular irritation testing with undiluted stearyl heptanoate. The Panel continued to believe that stearyl heptanoate, as used in cosmetic formulations, would not produce significant ocular irritation. The same was true for the related stearyl alkanoates.
Stearyl stearate is used in cosmetic formulations that can be inhaled. In the absence of inhalation data, the Panel considered other pertinent data indicating that incidental inhalation exposures to these ingredients in aerosolized cosmetic products would not cause adverse health effects, including data on systemic toxicity, ocular or dermal irritation or sensitization, and other effects. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Conclusion
The CIR Expert Panel concluded that stearyl heptanoate, stearyl caprylate, stearyl palmitate, stearyl stearate, stearyl behenate, and stearyl olivate are safe in the present practices of use and concentration (note 1).
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
