Abstract
Sustainable use of military training ranges requires the development of compounds that have a minimal impact to the environment when used in a weapon system. Guanidinium 3,4-dinitropyrazolate (GDNP) is a novel explosive compound of interest for application in some weapon systems. Little is known of its toxicologic properties. To ensure the health of potentially exposed personnel and the environment, initial toxicity investigations were conducted and the results were compared with another widely used energetic (hexahydro-1,3,5-trinitro-1,3,5-triazine [RDX]). In a microplate Ames assay, GDNP was not cytotoxic to bacterial tester strains at concentrations less than 100 μg/mL. However, GDNP was mutagenic to 4 of 5 bacterial strains with and without S9 metabolic incubation at concentrations as low as 0.7 μg/mL. Unlike RDX, GDNP did not have an affinity for the γ-aminobutyric acid(A) receptor convulsant site and was predicted to not induce seizure. After acute oral dosing in female rats, the median lethal dose in female rats of GDNP in tap water solution was determined to be 720 mg/kg. Daily oral exposure to 500 mg/kg per d of GDNP for 14 days caused weight loss, increased liver and spleen weights, and adverse histopathologic events in kidney and spleen. These adverse events were not observed in animals receiving lower doses of GDNP. In this study, the lowest-observed-adverse-effect-level from oral exposure to GDNP for 14 days was 500 mg/kg per d and the no-observable-adverse-effect-level was 152 mg/kg per d.
Introduction
Current land use patterns and expanding suburban populations present encroachment issues for many military installations. Although there have been efforts to close and realign many installations, wise stewardship of remaining active training sites is a priority for the US Army. Therefore, early in the weapon system development process, environmental and occupational toxicity data for new compounds are collected so that decisions can be made regarding further testing and implementation. 1 The US Army is engaged in a major effort as part of the Ordinance Environmental Program to produce munitions and propellant formulations that have reduced environment, safety and occupational health (ESOH) impacts compared to compounds now widely in use but known to have ESOH issues.
One novel compound, guanidinium 3,4-dinitropyrazolate (GDNP) is being developed for use as an energetic. The molecular structure of GDNP is shown in Figure 1. It is a salt developed by Physical Sciences Inc (Andover, Massachusetts). Although there is considerable toxicological information on guanidinium and its hydrochloride and nitrate salts, 2 –8 there is little information on the toxicity of nitropyrazolates and nothing is known about GDNP. Studies were initiated to evaluate the toxicity of this compound. These studies included the determination of GDNP mutagenicity using a microplate Ames assay; evaluation of its in vitro cytotoxicity in bacteria by measuring adenosine triphosphate (ATP) luminescence; and evaluation of its in vivo acute and subacute oral toxicity in female, Sprague-Dawley rats.
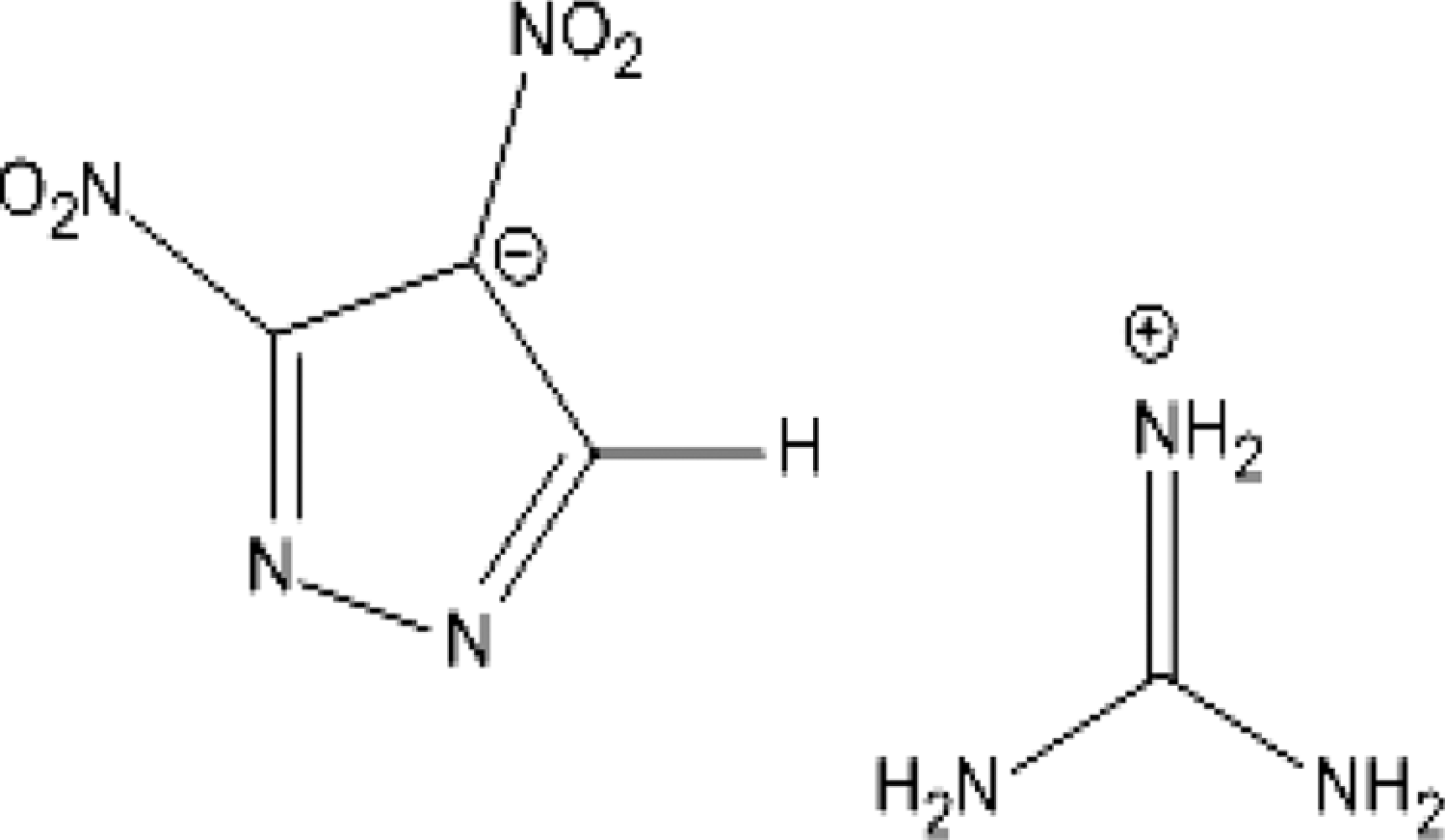
Molecular structure of guanidinium 3,4-dinitropyrazole (GDNP).
Materials and Methods
Test Compound
The GDNP is a light yellow, medium to fine grain powder. The material was developed by Physical Sciences Inc (SBIR Contract No W911QX-06-C-0138) and supplied by BAE Ordinance Systems (Kingsport, Tennessee); Batch # 1083-97. The compound was certified to be >99% pure as measured by differential scanning calorimetry and 100% pure by nuclear magnetic resonance spectral analysis. Analytical validation of GDNP purity was accomplished using high-performance liquid chromatography (HPLC, CAD Method 98.2) by the Chromatographic Analysis Division (Explosives Team), Laboratory Sciences Portfolio of the Army Institute of Public Health.
Preliminary studies indicated that GDNP dissolves slowly in water at concentrations up to 100 mg/mL with stirring. However, for the acute study (described below), dry crystalline GDNP was weighed out in plastic pans on a per-animal basis. At the higher doses of 1 and 2 g/kg, the compound did not dissolve readily in water in the weigh pan. A suspension of the 1 g/kg dose could be pulled into the dosing syringe and administered to the rat. However, the 2 g/kg dose was too viscous and could not be administered. In stage 2 of the acute study, prolonged water exposure of the compound weighed per animal was attempted to improve the solubility. However, upon standing in water, the crystal structure of the solid appeared to change, becoming a smooth, hard, insoluble mass. For stage 3, the GDNP was suspended in corn oil; suspension was difficult due to the apparent insolubility of GDNP in corn oil and necessity for extensive grinding to get the GDNP into small enough granules to be pulled through the gavage needle for dosing. For stage 4, a 100 mg/mL stock of GDNP was prepared in tap water and animals were dosed with this solution.
For stability tests, a solution of 25 mg/mL was prepared in tap water and stored at room temperature. Aliquots taken up to 18 days after preparation of the solution were analyzed by HPLC; the results indicated no loss of compound under these conditions, that is, GDNP is stable at room temperature up to 18 days. For the subacute study, half-log serial dilutions were prepared from a stock solution with a nominal concentration of approximately 50 mg/mL. The concentration of the lowest dilution was validated by HPLC for GLP compliance and the concentrations of the higher dosing solutions determined by back calculation. On day 8 of the subacute study, a rust-colored flocculent precipitate was observed in the highest concentration dosing solution. To make sure that the dosing concentration was not affected by the precipitate, a fresh stock was prepared and validated by HPLC. The volume of solution administered to each rat was adjusted, based on animal weight, so that the rats continued to receive the intended dose.
γ-Aminobutyric Acid(A) Receptor Binding Assay
The GDNP was tested at a single concentration of 33 μmol/L for affinity to the γ-aminobutyric Acid(A) (GABAA) receptor convulsant site. The [ 35 S]t-butylbicyclophosphorothionate ([ 35 S]-TBPS) convulsant site assay based on the method of Maksay 9 was performed by Ricerca Biosciences (Bothell, West Virginia), using 200 μmol/L of picrotoxin as a standard positive control.
In Vitro Microplate Ames Assay
A microplate Ames assay (Xenometrics MPF Ames Assay, AG, Switzerland) was used that provided a convenient, high throughput capability for mutagenicity testing (Xenometrix MPF is a trademark of Xenometrix AG, Switzerland). The test uses the Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strains WP2 uvrA and WP2 [pKM101]. 10,11 The assays were conducted in a manner consistent with the standards found in Title 40 Code of Federal Regulations (CFR), Part 792, Good Laboratory Practices.
The GDNP was weighed and dissolved in sterile distilled water to a nominal concentration of 50 mg/mL, that is, the 25× starting stock solution of GDNP, immediately prior to serial dilution in water and final dilution in Ames exposure medium. 12 The HPLC verified that the actual concentration of the 25× starting stock solution was within 95% of this intended concentration. Due to cytotoxicity observed at high exposure concentrations in the initial assay (see Results section), the starting 25× stock solution used in later experiments was reduced to 2.5 mg/mL.
Bacterial suspensions were diluted with incubation medium and aliquoted to tissue culture wells containing serial dilutions of the 25× stock solution of GDNP. The bacterial suspensions were incubated with shaking for 90 minutes at 37°C with and without the inclusion of Aroclor-induced, S9 liver extract (±S9; the S9 extract was prepared from male rats and had a final protein concentration of 5.5 mg/mL). 12 At the end of the incubation, each well was diluted 11-fold with purple indicator medium and 50 μL aliquots were distributed into 384-well plates. These plates were incubated anaerobically for 2 days at 37°C and then scored for reversion, that is, evidence of a positive mutagenic event. The colorimetric indicator reacts to changes in pH resulting from metabolism by living, mutated bacteria. Positive wells change from purple to yellow and are scored as a mutagenic event.
A positive control appropriate for each strain was run coincidently with GDNP to assure the assay was valid for each trial: 2 μg/mL 2-nitrofluorene for TA98 − S9; 0.1 μg/mL 4- nitroquinoline-N-oxide for TA100 − S9; 100 μg/mL N4-aminocytidine for TA1535 − S9; 15 μg/mL 9-aminoacridine for TA1537 − S9; 1 μg/mL 4-nitroquinoline-N-oxide for E coli–S9; 5 μg/mL 2-aminoanthracene for TA98, TA100, TA1535, and TA1537 + S9; and 50 μg/mL 2-aminoanthracene for E coli + S9. The assay as a whole was determined to be valid if the number of control background reversions and the number of positive control reversions are within prescribed limits. A positive result was indicated if the number of reversions induced by the test compound was at least 3-fold above the background control, and there was a concentration-related increase over the range tested. 12
In Vitro Cytotoxicity Assays
ATP assay
Coincident with the Ames assay, a duplicate plate was prepared for the determination of GDNP cytotoxicity using ATP luminescence. After the 90-minute incubation at 37°C, samples from the incubation plate were aliquoted to a 96-well plate. An equal volume of luminescent reagent was added to each well according the method described for the BacTiter-Glo microbial cell viability assay (BacTiter-Glo is a trademark of Promega Corporation Madison, Wisconsin). Luminescence was measured using a Synergy HT Multi-Detection Microplate Reader (Model SIAFRTD) and Gen5 software (BioTek Instruments Inc, Winooski, Vermont). Cytotoxicity of GDNP in the BacTiter-Glo assay was indicated when the luminosity of ATP in compound-treated cultures was decreased below the levels in vehicle-treated cultures; the level of ATP-generated luminosity correlates with the number of living bacteria. Data are expressed as a percentage of the level of luminosity generated by the control and vehicle-treated bacteria. Cytotoxicity of GDNP in cultures incubated with S9 was not evaluated.
Animals
Animal use procedures were approved by the Animal Care and Use Committee at the US Army Public Health Command (USAPHC). Animal care and use was conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals and in accordance with all applicable Federal and Department of Defense (DOD) regulations. The USAPHC animal care program is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. The study was conducted in a manner consistent with the standards found in Title 40 CFR, Part 792, Good Laboratory Practices.
Oral toxicity studies were conducted using young adult female Sprague-Dawley CD-1 rats obtained from Charles River Laboratories (Wilmington, Massachusetts). Rats were 8 and 6 weeks old for the acute and 14-day subacute toxicity studies, respectively. The attending veterinarian examined the animals and found them to be in acceptable health. The animals were quarantined for a minimum of 5 days after arrival. All rats were maintained in a temperature-, relative humidity-, and light-controlled room. The conditions were 64°F to 79°F, 30% to 70% relative humidity with a 12-hour light/dark cycle. A certified pesticide-free rodent chow (Harlan Teklad, 8728C Certified Rodent Diet) and drinking quality water were available ad libitum (Teklad Certified Rat Diet is a registered trademark of Harlan, Teklad, Madison, Wisconsin). Rats were housed individually in suspended polycarbonate boxes with Harlan Sani-Chip bedding (Harlan Sani-Chip is a registered trademark with P. J. Murphy Forest Products Corporation, Montville, New Jersey). Each rat was uniquely identified by number using cage cards only for the acute study and both cage cards and tail tattoos for the 14-day study.
Acute Study (Sequential Stage-Wise Probit [SSWP])
The objective of this study was to determine the acute oral median lethal dose (LD50) of GDNP in the female Sprague-Dawley rat using the SSWP 13,14 and to guide oral exposures for the subacute (14 days) study. The general procedures of this acute study followed the US Environmental Protection Agency (USEPA) Health Effects Test Guidelines for Acute Oral Toxicity (OPPTS 870.1100). 15 Tests were performed using 4 separate stages of dosing.
All animals were fasted overnight prior to dosing and for up to 4 hours postdosing. Doses for the first stage of the acute tests were 125, 250, 500, and 1000 mg/kg. All doses were calculated based on body weights taken immediately prior to dosing. The amount of GDNP appropriate for each rat was weighed individually in a weigh pan. Tap water was added up to the maximum allowed (10 mL/kg); the GDNP in solution/suspension was pulled into the gavage syringe and administered by oral gavage using a 16 gauge × 2-inch stainless steel gavage needle. 16
In the second stage, there were difficulties encountered due to apparent insolubility of GDNP at higher intended doses of 500 to 1000 mg/kg (see Methods section). For stage 3, suspensions of GDNP were attempted using corn oil as the vehicle. Gavage dosing with this vehicle was also challenging due to density of insoluble granules at the higher doses; 3 water vehicle groups were also included. The final stage 4 dosing used tap water solutions of GDNP. A solution of approximately 100 mg/mL was prepared; the concentration was verified by HPLC to be 94 mg/mL. In this final stage, 3 rats each were given a single dose of 620, 714, or 818 mg/kg.
Following administration of the test compound for each phase of the acute test, the rats were observed for 14 days. All clinical signs or incidences of death were recorded on a daily basis. Individual body weights were recorded daily (5 days a week) throughout the 14-day observation period to determine recovery.
Surviving animals were euthanized on day 14 and submitted for gross pathological examination. The data from the SSWP were analyzed according to the methods of Feder et al 17,18 in order to obtain an estimated LD50 value, 95% confidence interval, and slope.
14-Day Oral Repeated Dose Toxicity Study
Seventy female Sprague-Dawley rats were randomly distributed into 7 treatment groups consisting of 10 rats each. The animals were then divided into 3 evenly distributed experimental groups; the start dates for each group were staggered over a period of 3 days to facilitate scheduling of necropsies. On the morning of each day, each rat received either 0 (tap water vehicle control), 1.3, 4.2, 13.9, 45.9, 151.5, or 500 mg GDNP/kg body weight-day (mg/kg per d) via oral gavage using a 16 gauge × 2-inch stainless steel gavage needle.
Using filtered tap water from the animal room as the vehicle, the 6 dosing solutions were prepared by making 1:3.3 (approximately half-log) serial dilutions beginning from a concentrated stock of 50 mg/mL. The final dilution, 0.13 mg/mL was verified by HPLC. The doses were administered daily, 7 days per week for a total of 14 doses.
On day 8 of the subacute study, a rust-colored flocculent precipitate was observed in the most concentrated dosing solution. To make sure that the dosing concentration was not affected by the precipitate, a fresh stock was prepared and validated by HPLC. The volume of solution administered to each rat was adjusted, based on animal weight, so that the rats continued to receive the intended dose.
Body weights were recorded on days −1, 0, 1, 3, 7, and 14. Food consumption based on change in feeder weights was monitored weekly. Animals were observed daily for toxic signs and morbidity. Water consumption was not monitored during this study. All data were recorded onto hardcopy spreadsheets and transcribed to an EXCEL spreadsheet for computer analysis.
At the end of the 14-day study period, the rats were anesthetized with carbon dioxide gas, and blood was collected by intracardiac puncture. The animals were then euthanized using carbon dioxide. Clinical chemistry and hematology values were determined from all useable samples. The brain, heart, kidneys, liver, ovaries, spleen, and uterus were removed and weighed. Gross necropsies were completed on all terminal animals. The following parameters, by test group, were analyzed and compared to the controls: (a) body weights, (b) weight gains, (c) food consumption, (d) absolute organ weights, (e) organ-to-body weight ratios, and (f) organ-to-brain weight ratios.
Hematology (Cell-Dyn 3700 Hematology Analyzer, Abbott Laboratories, Abbott Park, Illinois) variables included: white blood cell (WBC) count, WBC differential, % neutrophils (NEU, %N), % lymphocytes (LYM, %L), % monocytes (MONO, %M), % eosinophils (EOS, %E), % basophils (BASO, %B), red blood cell (RBC) count, hemoglobin (Hb), hematocrit (HCT), mean cell volume, mean cell Hb, mean cell Hb concentration, RBC distribution width, platelets (PLT), and mean PLT volume (MPV).
Clinical Chemistry (VetTest 8008 Chemistry Analyzer and VetLyte Na, K, Cl Analyzer, IDEXX Laboratories, Inc, One IDEXX Drive, Westbrook, Maine) variables included: albumin (ALB), alkaline phosphatase, alanine aminotransferase (ALT), blood urea nitrogen (BUN), calcium (Ca), cholesterol (CHOL), creatinine (CREA), glucose (nonfasting), globulin, lactate dehydrogenase, phosphorus, total bilirubin, total protein (TP), sodium (Na), potassium (K), and chlorine (Cl).
Statistical Analyses
Statistical analysis was done using Prism Version 4.03 (GraphPad Software, Inc, La Jolla, CA). For variables that were measured only at the end of the study, the dosage groups were compared using a 1-factor analysis of variance (ANOVA) assuming a normal distribution and equivalent variance between treatments. Organ-to-brain and organ-to-body-weight ratios were calculated and analyzed similarly to the other parameters measured at the end of the study with the initial ratio then multiplied by factors of 10 so that the resulting number was greater than 1, that is, normalization. If the ANOVA was significant, Dunnett multiple comparison test was used to determine the statistical significance (P ≤ .05) of individual GDNP dosage groups to the control group.
Histopathology
Liver, spleen, kidney, ovary, uterus, and other tissues noted to demonstrate gross abnormalities were collected and preserved in 10% buffered formalin, selectively trimmed and placed in cassettes labeled with protocol number, animal identification number, and laboratory-assigned accession number. Tissues were processed for embedding in paraffin, microtomed at 5 μm thickness, automatically stained with hematoxylin and eosin, and coverslipped. The pathologist examined slides for compound-induced histopathologic changes via light microscopy. Prevalence and severity of findings were graded in comparison to controls. Findings are classified as none, minimal, mild, moderate, or severe.
Results
GABAA Receptor Binding Assay
Picrotoxin at 200 μmol/L was included in the screen as a positive control; it inhibited [ 35 S]-TBPS binding by 90% (half maximal inhibitory concentration [IC50] = 0.182 μmol/L). The GDNP was screened at a single concentration of 33 μmol/L for affinity to the GABAA (GABA receptor classes) convulsant site. At this concentration GDNP inhibited [ 35 S]-TBPS binding by −2%. Biologically relevant binding is significant when [ 35 S]-TBPS binding is inhibited by ≥50% of maximum. For comparison, 33 μmol/L hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) inhibited [ 35 S]-TBPS binding by 78% (IC50 = 20 μmol/L). 19
In Vitro Cytotoxicity of GDNP
Figure 2 illustrates the toxicity demonstrated by GDNP. Evidence of cytotoxicity (a decrease in ATP luminescence) was observed consistently in all bacterial strains except the E coli combination culture at concentrations ≥100 μg/mL; GDNP was not cytotoxic to E coli up to 100 μg/mL. The cytotoxicity of GDNP metabolites, that is, bacterial viability after S9 incubation, was not tested.
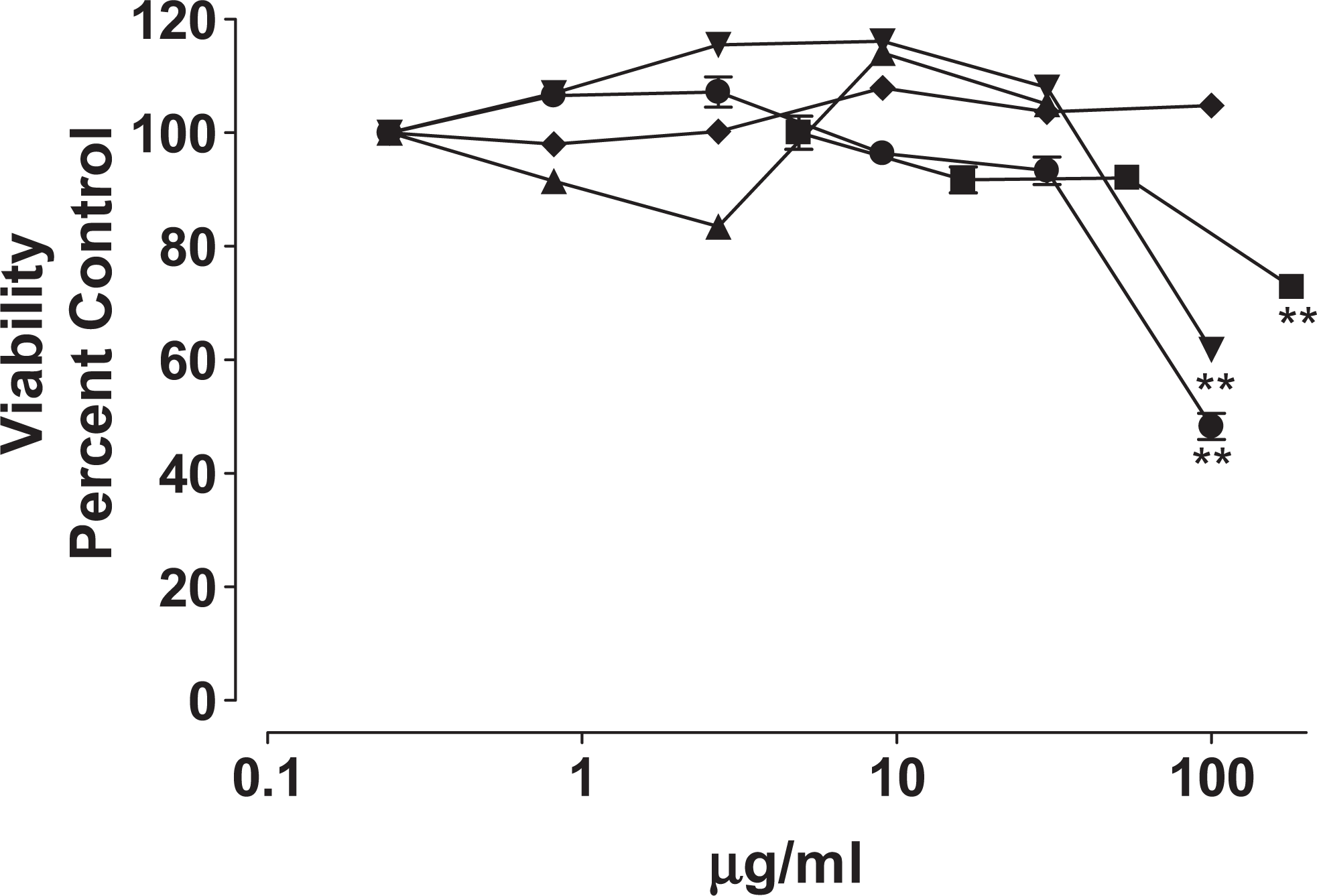
Cytotoxicity of GDNP—ATP luminescence is expressed as percentage change compared with control. (▪) TA98, (•) TA 100, (▴) TA1535, (▾) TA1537, and (♦) Escherichia coli. **P ≤ .01. ATP indicates adenosine triphosphate; GDNP, guanidinium 3,4-dinitropyrazole.
Mutagenicity of GDNP
The results are illustrated in Figure 3 and Table 1. The GDNP was mutagenic in all strains both with and without S9 metabolic incubation except for the TA1537 strain. The lowest concentration at which GDNP induced mutation was strain-dependent and ranged from 0.7 to 48 μg/mL.
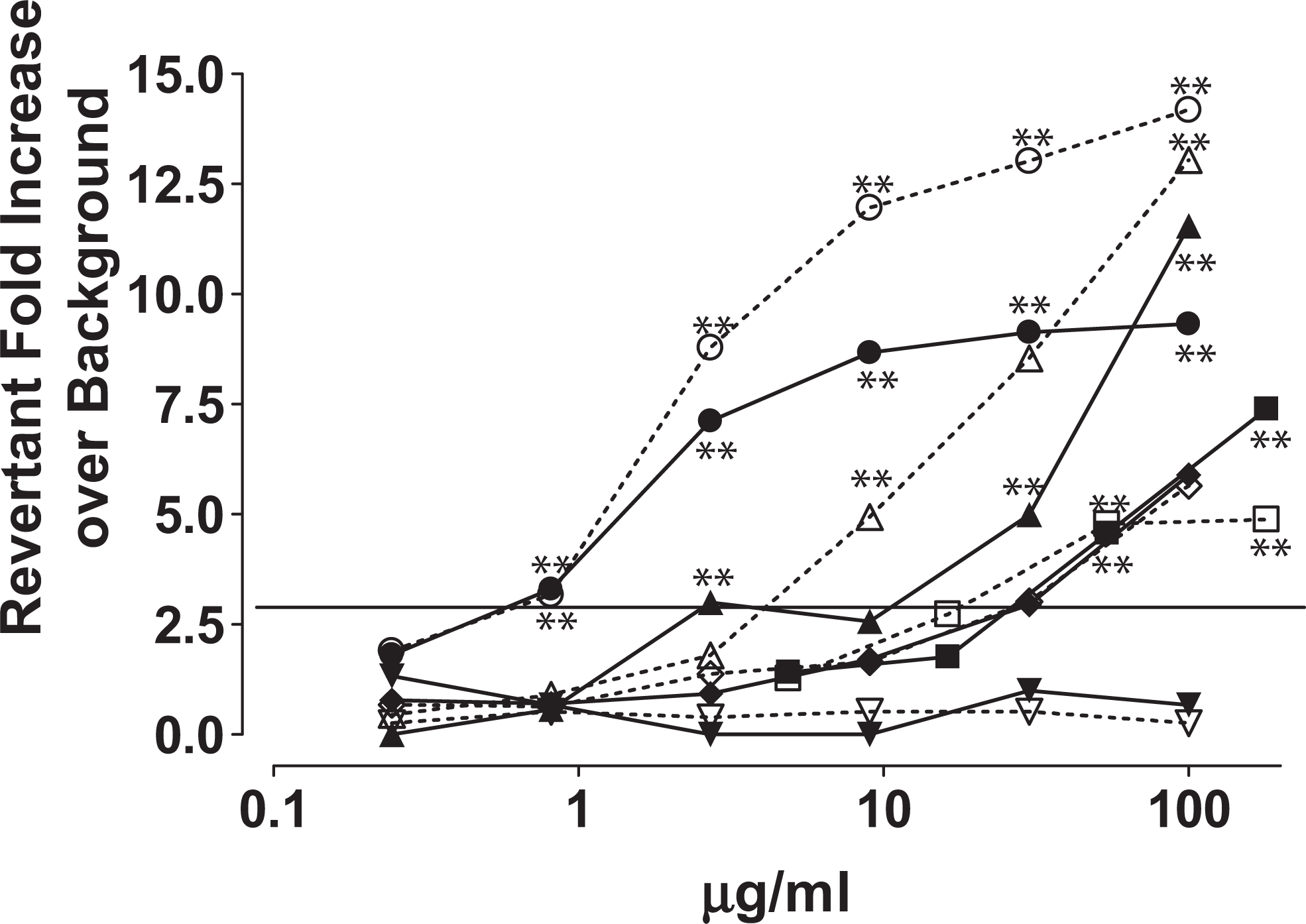
Mutagenicity of GDNP—significant increases in revertants, greater than 3-fold over background, occurred in all bacterial strains with and without S9 metabolism with exception of TA1537. (▪) TA98 − S9, ( □ ) TA98 + S9, (•) TA 100 − S9, (^) TA 100 + S9, (▴) TA1535 − S9, (▵) TA1535 + S9, (▾) TA1537 − S9, (▿) TA1537 + S9, (♦) Escherichia coli. − S9, and (♦) E coli. + S9. **P ≤ .01. GDNP indicates guanidinium 3,4-dinitropyrazole.
Results of GDNP Ames Assay
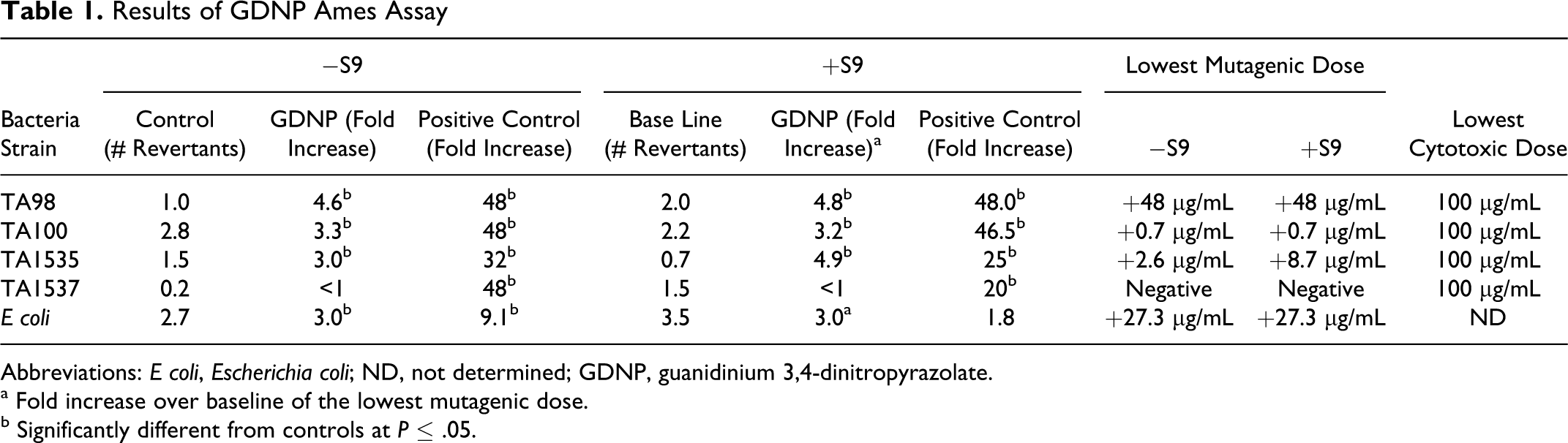
Abbreviations: E coli, Escherichia coli; ND, not determined; GDNP, guanidinium 3,4-dinitropyrazolate.
a Fold increase over baseline of the lowest mutagenic dose.
b Significantly different from controls at ≤ .05.
Acute In Vivo Toxicity
The first stage of the SSWP indicated an LD50 above 500 mg/kg. The affected animals quickly (within 20 minutes) became sluggish and progressively sedated in a dose-dependent manner. One animal showed an indication of diarrhea. Most of the affected animals became inactive on the cage floor, exhibited 2 to 3 brief convulsions and died, becoming immediately tetanic, that is, clear stiffening of core body and extremity musculature. The results from the water vehicle dosings for all stages of the SSWP are presented in Table 2; calculations using these data indicated the LD50 of GDNP in tap water to be 720 mg/kg. The slope of the dose–response curve is very steep, but confidence intervals could not be derived due to the small sample size of the final dosing groups using the GDNP tap water solution. Gross pathology observations in these animals at necropsy were unremarkable.
Data Used for the Determination of LD50 Using SSWPa
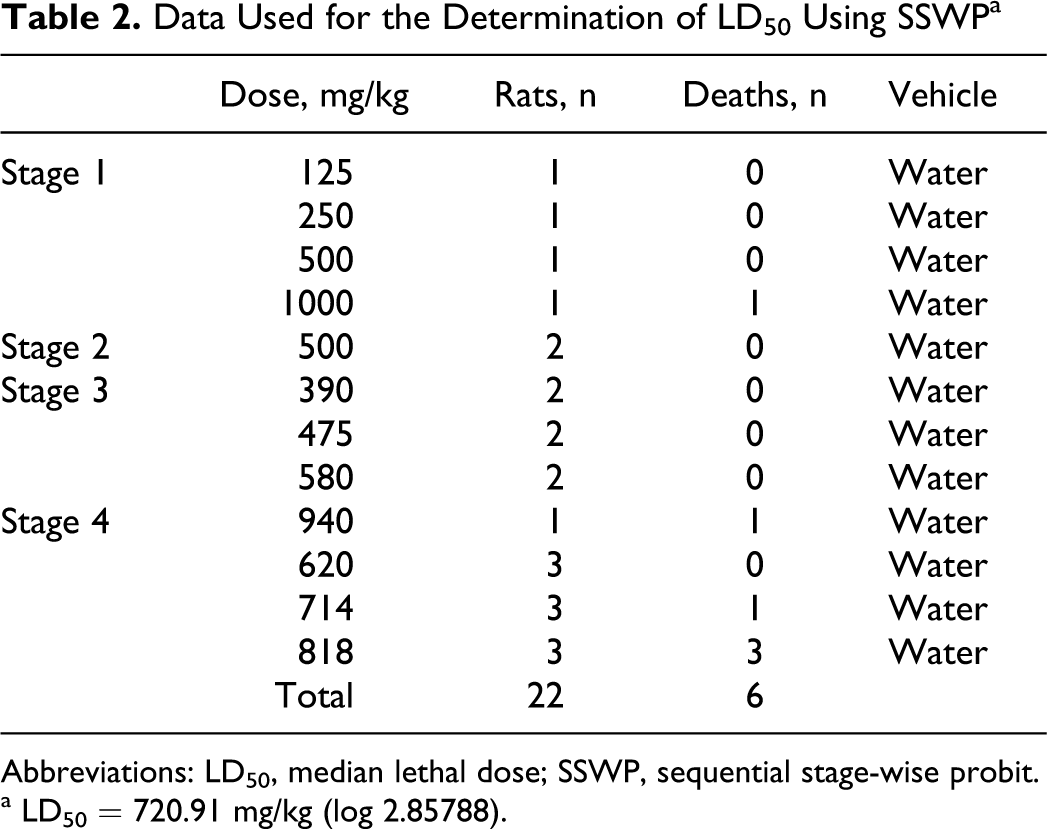
Abbreviations: LD50, median lethal dose; SSWP, sequential stage-wise probit.
a LD50 = 720.91 mg/kg (log 2.85788).
14-Day Oral Repeated Dose Toxicity Study
On the first days of dosing, lethargy was observed within the first 30 minutes after dosing in about half of the animals in the 500 mg/kg per d dosing group; sedation and death were not observed. After about 4 days of dosing, the effect of high doses of GDNP on activity—compound-induced lethargy—was less apparent and was not observed during the second week of dosing. Lethargy was not observed at any time at dosages less than 500 mg/kg per d. No other clinical signs were observed in any of the dosing groups.
The body weight of the animals increased similarly with time for all dosage groups except for the 500 mg/kg per d dosage group. Weight gain was reduced in this group for the first week of dosing (Figure 4). Over the second week of dosing, the 500 mg/kg per d group gained weight faster than the other groups including the control group so that by 13 days, the average weight of the 500 mg/kg per d group was not significantly different from control. The effect of high doses of GDNP on body weight was reflected in food consumption rates (data not shown). The amount of food eaten by the 500 mg/kg per d group (82.3 ± 17.7 g, standard deviation [SD]; P ≤ .01) during the first week was less than the control group (136.8 ± 6.5 g). The amount of food eaten by the high-dose group during the second week was similar to the other groups, including the control group. However, the total food consumption over the 2 weeks of dosing was still less for rats in the 500 mg/kg per d dose group (108.2 ± 17.8 g; P ≤ .05) than in the control group (125.3 ± 7.0 g).
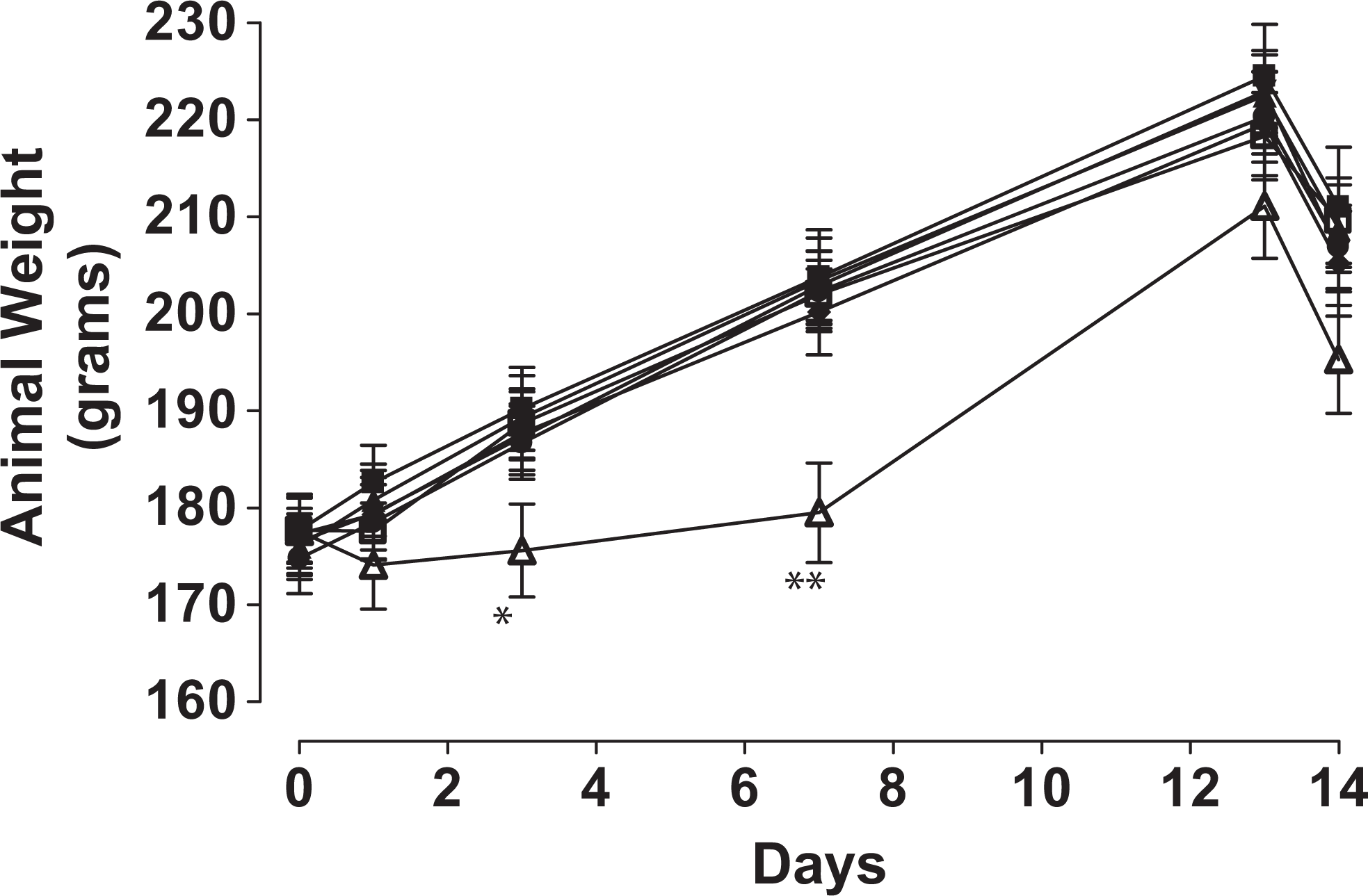
Body weight changes during 14-day GDNP dosing study. The net body weight of the animals increased similarly with time for all dose groups except for the 500 mg/kg per d dose group. Weight gain was reduced in these groups for week 1. (▪) Vehicle, (▴) 1.3 mg/kg per d, (▾) 4.2 mg/kg per d, (♦) 13.9 mg/kg per d, (•) 45.9 mg/kg per d, (□ ) 151.5 mg/kg per d, and (^) 500 mg/kg per d. *P ≤ .05. GDNP indicates guanidinium 3,4-dinitropyrazole.
At necropsy, observed gross lesions included dark red spleen, enlarged spleen or liver, pale liver or kidneys, dilated uterus (hydrometra), enlarged mesenteric lymph nodes, and prominent Peyer patches of the small intestine. Enlarged spleen, enlarged liver, and pale kidneys were found in 2 of 10, 2 of 10, and 2 of 10 animals in the 500 mg/kg per d dose group, respectively; pale kidneys were noted in 1 of 10 controls and in 1 of 10 rats in the 14 mg/kg per d dose group. None of these gross findings were noted in the 152, 45.9, 4.2, and 1.3 mg/kg per d dose groups. All gross observations of hydrometra correlated with histologic uterine dilatation. However, hydrometra was not considered an adverse finding but was equated with distension associated with proestrus. 20
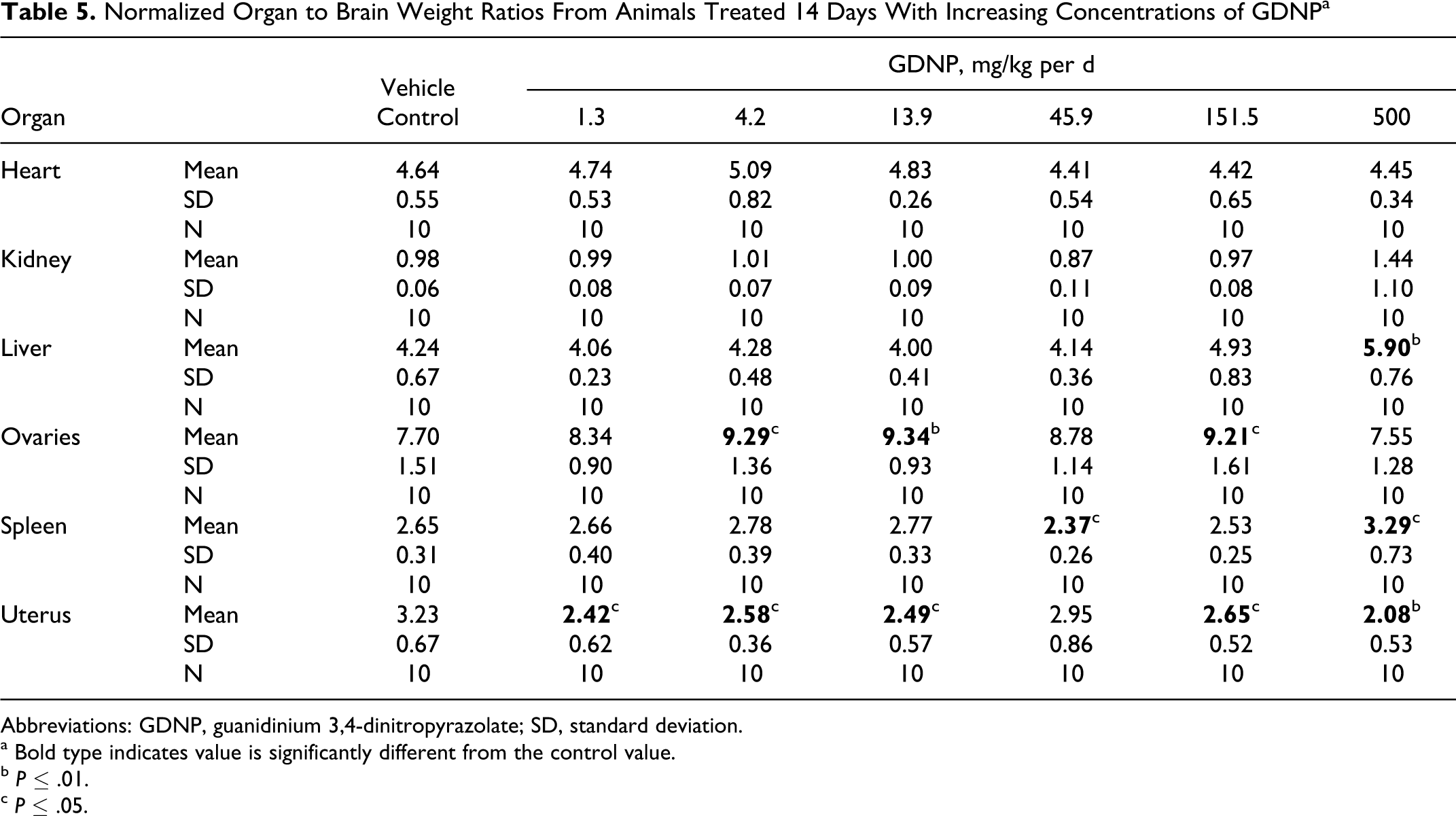
Differences were observed between the dose groups and the control group in mean organ weights, and organ-to-body and brain weight ratios for the liver, spleen, uterus, and ovaries (Tables 3- 5). The differences were similar in all 3 methods of analysis. There was a distinct increase in liver (38%-45%) and spleen (23%-47%) weights in rats from the 500 mg/kg per d dose group. Although statistical evaluations indicated differences in uterus and ovary weights, the impact of dose was inconsistent. There was a decrease (30%-60%) in uterine weight consistently in the 500 mg/kg per d dose group. There was an increase (16%-20%) in ovary weight compared to controls at the intermediate dosages of 13.9 to 151.5 mg/kg per d, but no impact at the 500 mg/kg per d dosage, that is, ovary weight within this high-dose group was not statistically different from the controls.
Absolute Weights of Organs From Animals Treated 14 Days With Increasing Concentrations of GDNPa
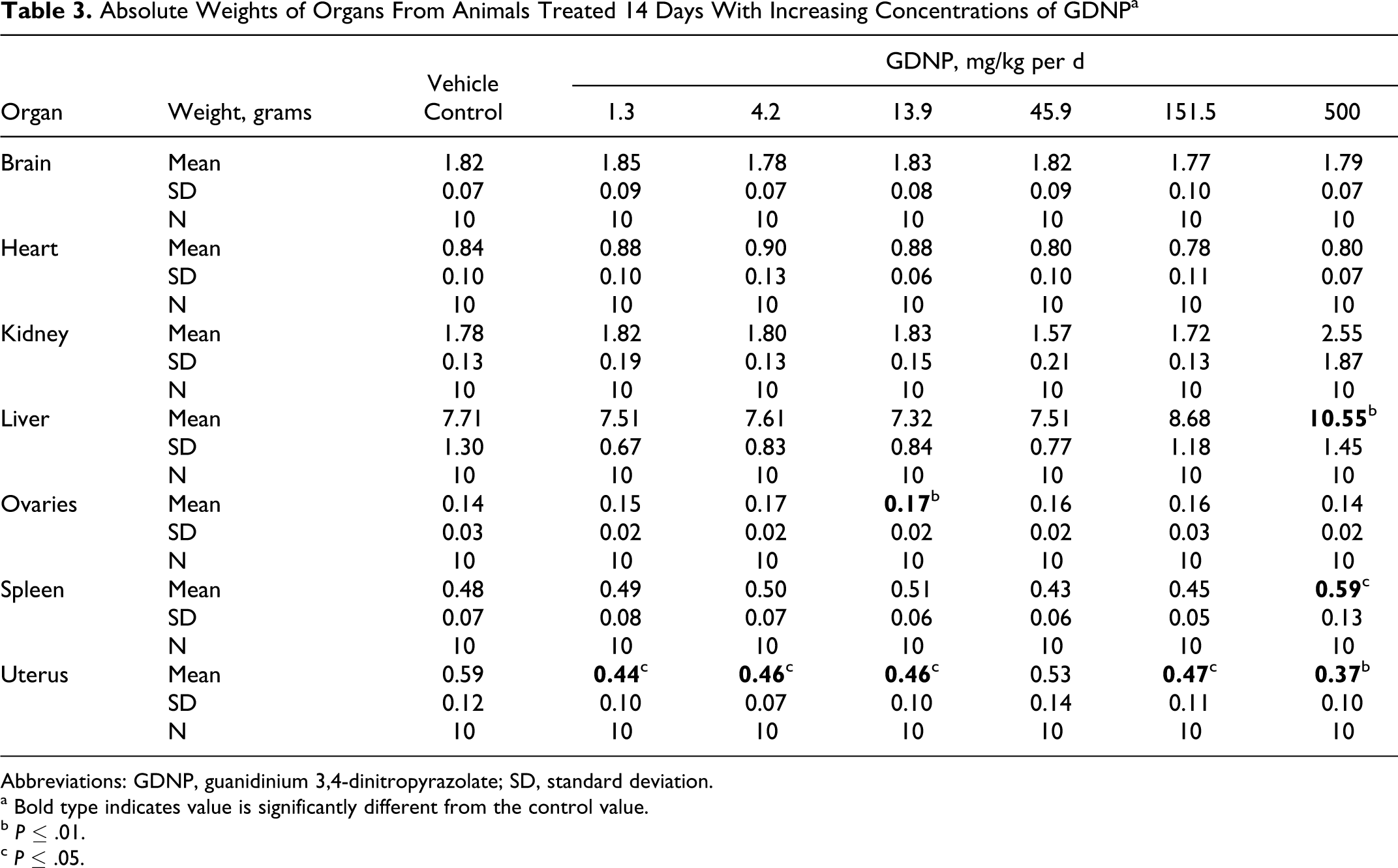
Abbreviations: GDNP, guanidinium 3,4-dinitropyrazolate; SD, standard deviation.
a Bold type indicates value is significantly different from the control value.
b P ≤ .01.
c P ≤ .05.
Normalized Organ to Body Weight Ratios From Animals Treated 14 Days With Increasing Concentrations of GDNPa
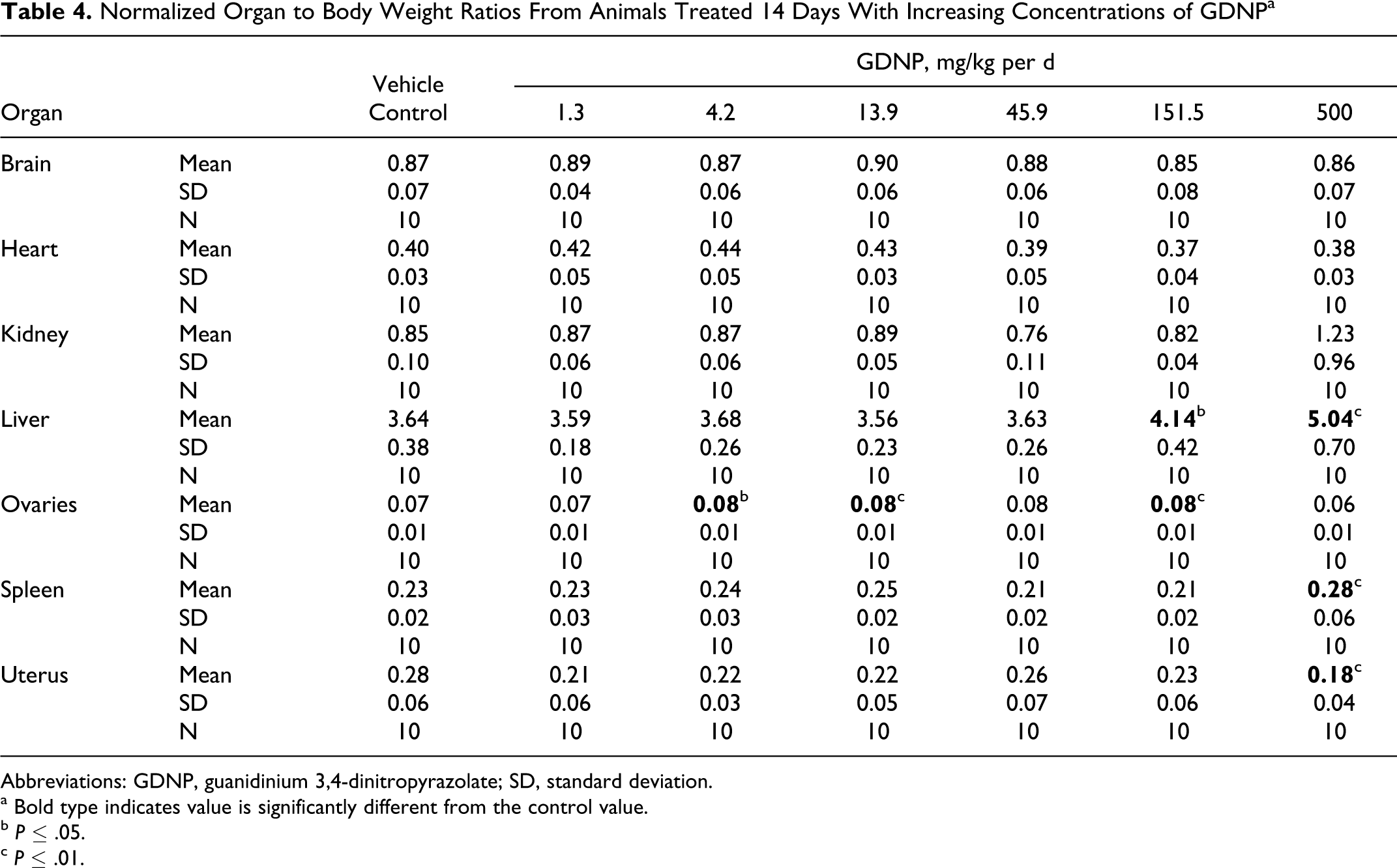
Abbreviations: GDNP, guanidinium 3,4-dinitropyrazolate; SD, standard deviation.
a Bold type indicates value is significantly different from the control value.
b P ≤ .05.
c P ≤ .01.
Normalized Organ to Brain Weight Ratios From Animals Treated 14 Days With Increasing Concentrations of GDNPa
Abbreviations: GDNP, guanidinium 3,4-dinitropyrazolate; SD, standard deviation.
a Bold type indicates value is significantly different from the control value.
b P ≤ .01.
c P ≤ .05.
Histopathology revealed several abnormalities (Figure 5). Based on incidence and severity, lesions associated with repeated dose exposure to GDNP were noted in a dose-dependent manner in the kidney and spleen. Extramedullary hematopoiesis (EMH, increased presence of hematological precursors in the splenic red pulp) was noted to be increased compared to vehicle controls in the 500 mg/kg per d group (6 of 10 moderate, 3 of 10 mild, and 1 of 10 normal; Figure 5A). Only 2 other groups demonstrated this finding, 14 mg/kg per d group as 1 of 10 mild and 4.2 mg/kg per d groups as 1 of 10 minimal. In the kidney, basophilic tubules, indicating tubular regeneration with or without tubular degeneration and luminal debris, were noted in the 500 mg/kg per d group, compared to vehicle controls (3 of 10 minimal, 5 of 10 mild, and 2 of 10 moderate; Figure 5B). Minimal numbers of basophilic tubules were noted in 1/0, 3/10, 3/10, 1/10, 2/10, and 1/10 of the control, 152, 46, 14, 4.2, and 1.3 mg/kg per d groups, respectively.
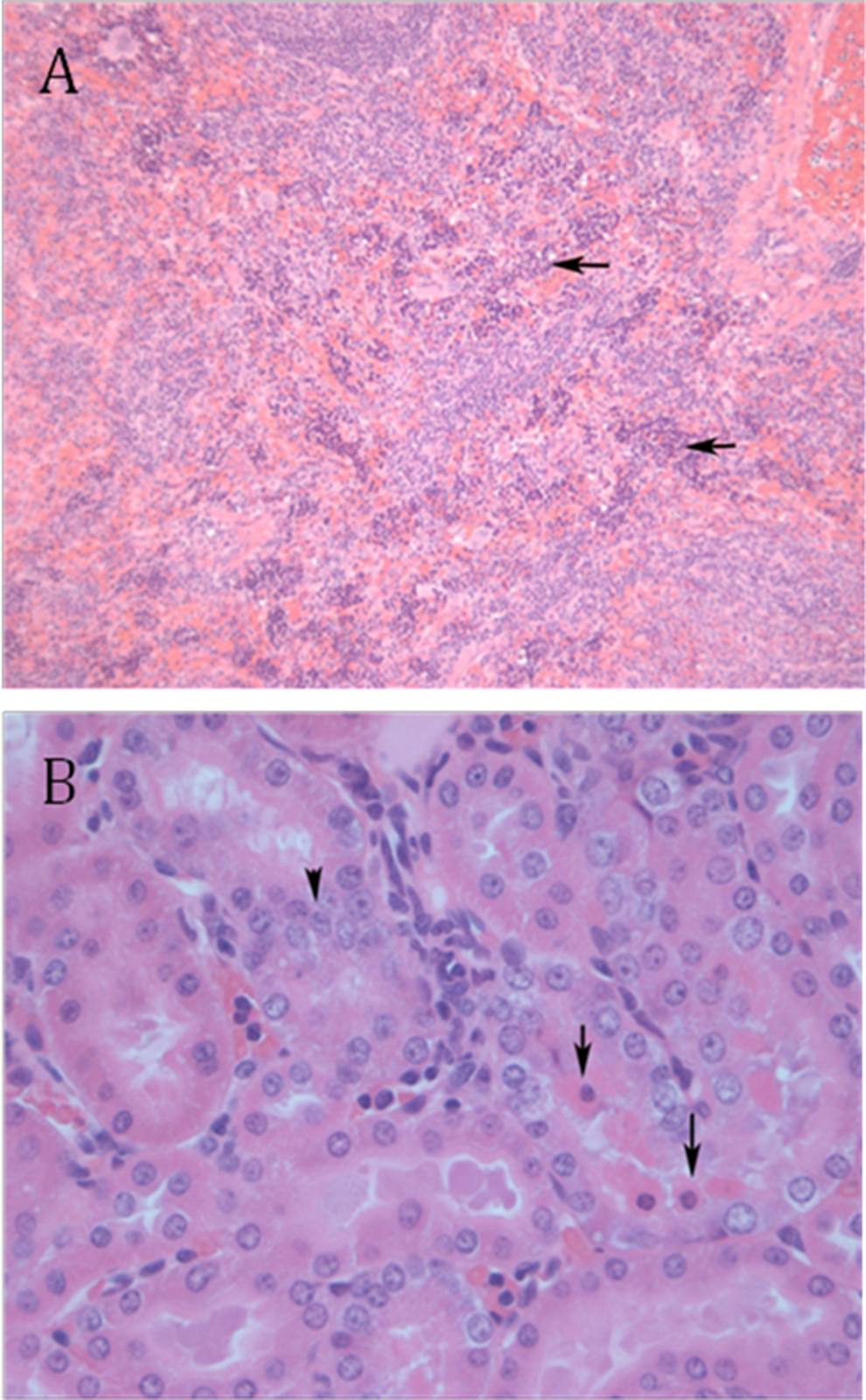
Subacute histopathology. Extramedullary hematopoeisis was increased in the 500 mg/kg per d group (A, 10×). Throughout the red pulp, there are numerous erythropoetic precursor cells, evidenced by round dark nuclei with indiscernible cytoplasm (arrows). In the kidney, necrotic tubular cells, hypereosinophilic cells with dark, shrunken nuclei, are noted within tubular lumen (long arrows). Basophilic, regenerative cells are identified by large, “open” nuclei with epithelium often stacked upon each other (arrowhead; B, 40×).
Basophilic tubules noted were often small, had an increased fraction of epithelial cells, a distinct blue cytoplasm, occasional mitotic figures and large “open” nuclei. Occasionally, there was tubular epithelial degeneration and luminal eosinophilic debris, indicating recent degeneration and necrosis of epithelial cells. These lesions were often observed along the proximal tubule. Luminal debris was only noted in the 500 mg/kg per d group. This is consistent with repeated injury of different tubules from treatment. Additionally, the severity of tubular damage and regeneration were increased in the 500 mg/kg per d group. It is likely related to repetitive dosing, damage, and repair as opposed to isolated tubular death and turnover in organ maintenance.
Analysis of the clinical chemistry results revealed differences for TP, ALB, ALT, BUN, CREA, CHOL, and Ca compared to the vehicle control group (Table 6). Statistically significant increases in TP, 13% to 20% were observed in dose groups of 45.9 mg/kg per d and above. The ALB concentration was increased 15% and 21% in dose groups 45.9 and 151.5 mg/kg per d, respectively, but was not increased in the 500 mg/kg per d dose group. The ALT was significantly increased 70% and CREA was increased 83% in the 500 mg/kg per d dose group. The CHOL was increased significantly, albeit only 6% in the 45.9 mg/kg per d and above. The BUN level exhibited significant 22% decreases in the 45.9 and 151.5 mg/kg per d dosages but was elevated 7% above control levels in the 500 mg/kg per d dose group. The increased BUN and CREA levels are consistent with the kidney dysfunction as also evidenced by increased kidney weight and histopathology of basophilic tubules. The largest increases were found in blood Ca concentrations; Ca was increased 52% and 62% in dose groups 45.9 and 151.5 mg/kg per d, respectively, and increased 24% in the 500 mg/kg per d dose group.
Clinical Plasma Chemistry Results for Female Rats Exposure Orally to GDNP for 14 Daysa
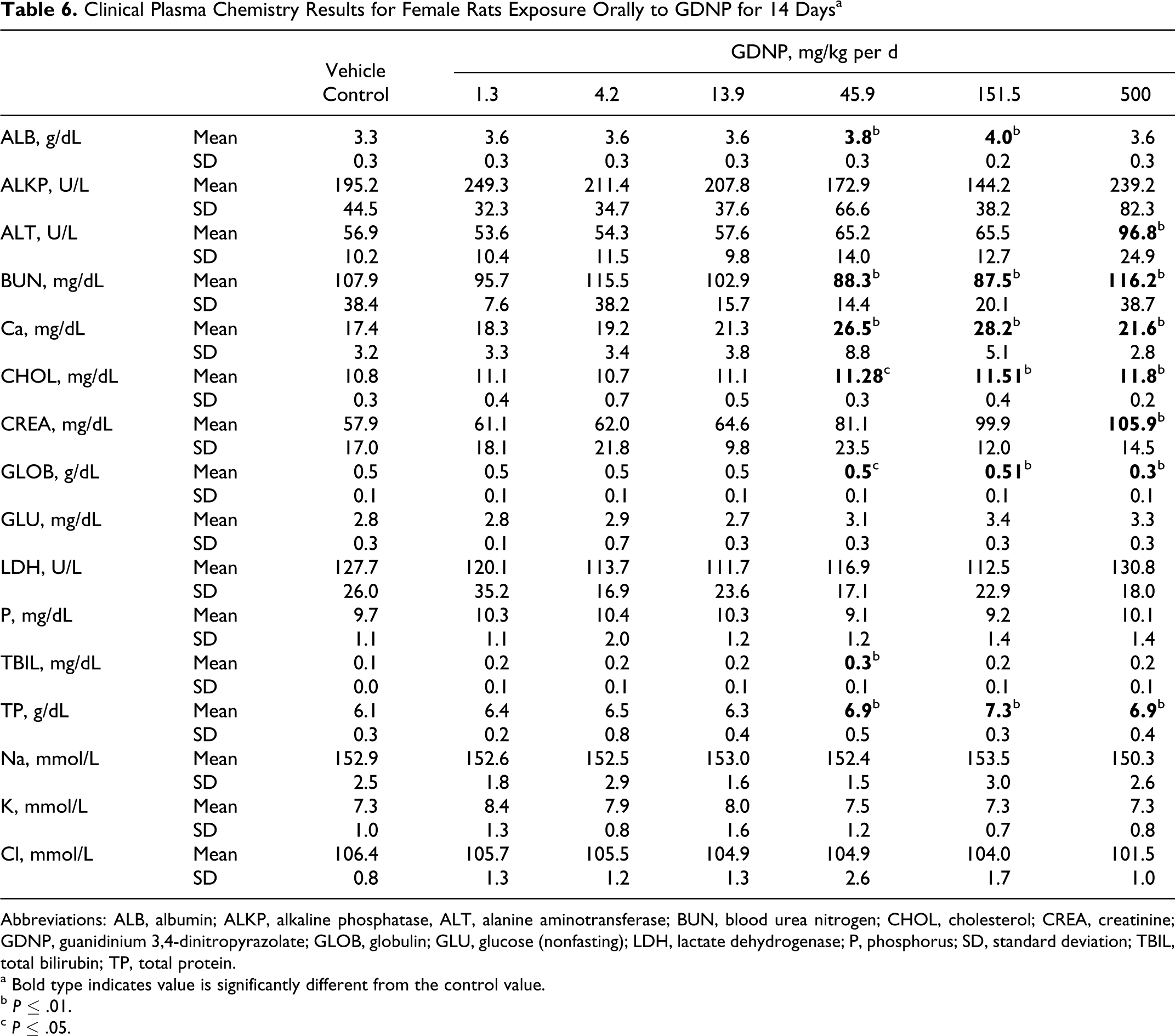
Abbreviations: ALB, albumin; ALKP, alkaline phosphatase, ALT, alanine aminotransferase; BUN, blood urea nitrogen; CHOL, cholesterol; CREA, creatinine; GDNP, guanidinium 3,4-dinitropyrazolate; GLOB, globulin; GLU, glucose (nonfasting); LDH, lactate dehydrogenase; P, phosphorus; SD, standard deviation; TBIL, total bilirubin; TP, total protein.
a Bold type indicates value is significantly different from the control value.
b P ≤ .01.
c P ≤ .05.
Statistical analysis of the hematology results revealed a dose-related increase in the concentrations of WBC (K/μL) reaching a 1.9-fold increase in the 500 mg/kg per d dose group (Table 7). Similarly, there were significant increases in the absolute concentrations of NEU, BASO, EOS, and LYM in the 500 mg/kg per d dose group when compared with the data of the control group. The increases were predominantly neutrophils and basophils (4-5-fold increase) with corresponding increases in the percent contribution to the total WBC population. Interestingly, although there was a 1.5-fold increase in the absolute concentration of lymphocytes, the contribution to the total WBC pollution was decreased by 15%. There was no change in the absolute density of RBCs, but a slight decrease (6%) in HCT and Hb with a 13% increase in RDW was observed.
Changes in Red Blood Cell Count, Hematocrit, Hemoglobin, White Blood Cell Count, and Proportion of White Blood Cell Types as a Function of Oral GDNP Exposure in Female Ratsa
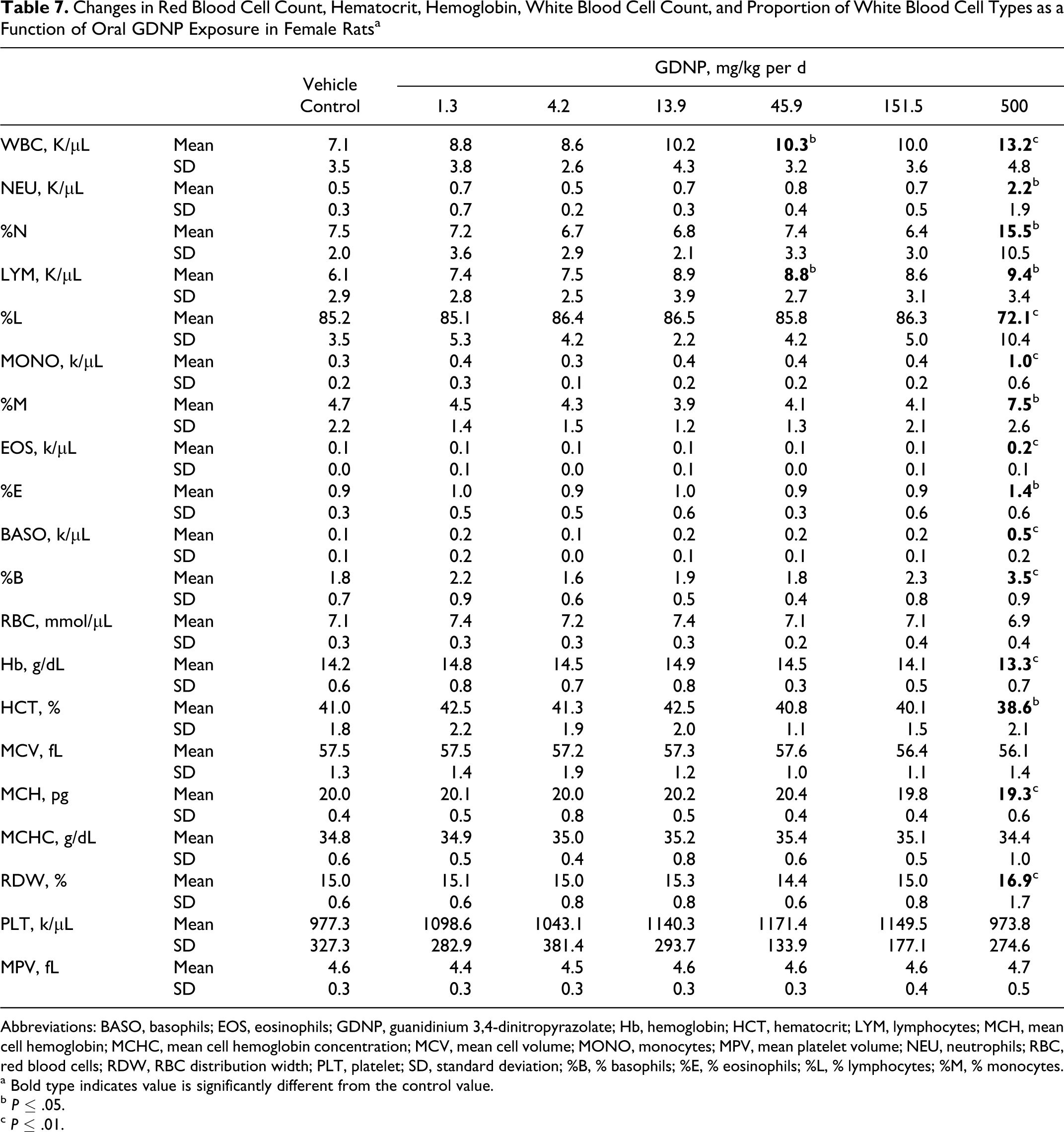
Abbreviations: BASO, basophils; EOS, eosinophils; GDNP, guanidinium 3,4-dinitropyrazolate; Hb, hemoglobin; HCT, hematocrit; LYM, lymphocytes; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; MCV, mean cell volume; MONO, monocytes; MPV, mean platelet volume; NEU, neutrophils; RBC, red blood cells; RDW, RBC distribution width; PLT, platelet; SD, standard deviation; %B, % basophils; %E, % eosinophils; %L, % lymphocytes; %M, % monocytes.
a Bold type indicates value is significantly different from the control value.
b P ≤ .05.
c P ≤ .01.
Discussion
Testing new compounds requires an integrative, holistic approach that is phased proportional with the level of investment or value of the new substance and how it will be used. 1 Many assays are available that provide value in assessing the risk from substance exposure. However, all assays have drawbacks, shortcomings, and uncertainties, when applying assay results in a risk assessment paradigm. Therefore, the results of many tests can help provide a more complete evaluation of relative impact and serve to reduce the levels of certainty while maintaining costs consistent with the level of effort devoted to the system or platform. Here, we compare the results of these tests on GDNP relative to a compound with a similar function, that is, RDX.
Guanidine and nitroguanidine are reported to be nonmutagenic in traditional agar plate Ames assays. 2,3 Nitroguanidine is also reported to be nongenotoxic. 4,5 The mutagenicity of 3,4-dinitropyrazole has not been previously reported. However, 4-nitropyrazole is a reported mutagen. 21 Thus, the mutagenicity of GDNP is likely due entirely to the 3,4-dinitropyrazole component.
The GDNP is not cytotoxic to bacterial tester strains at concentrations less than 100 μg/mL. However, GDNP is mutagenic to 4 of 5 bacterial strains with and without S9 metabolic incubation at concentrations as low as 0.7 μg/mL. Thus, by USEPA guidelines, GDNP is mutagenic under the conditions of this assay.
The GDNP did not have affinity for the GABAA receptor in the in vitro binding assay, that is, did not inhibit [ 35 S]-TBPS when screened at 33 μm. This result predicted that GDNP would probably not induce seizures like RDX, 19 when administered to rats. However, doses of GDNP above the lethal dose quickly caused progressive lethargy, sedation, and mild convulsions before cessation of breathing and death. These convulsions were not like the severe seizures induced by RDX. The mechanism of GDNP-induced convulsions is not known.
The LD50 of guanidine (free base), the uncharged form of the guanidinium cation of GDNP, is 475 mg/kg in female rats. 22 The hydrochloride and nitrate salts of guanidinium have LD50 in female rats of 774 and 729 mg/kg, respectively. 6,7 Clinical signs were observed in both the gastrointestinal (GI) tract and the central nervous system–neuromuscular junction (CNS-NM). Symptoms referable to the GI tract included increased salivation, hunched posture, and diarrhea. The CNS-NM signs included disorientation, increased startle reflex, hyperactivity, jumping behavior, presence of tremors/twitching, and depression of grasping and righting reflexes. Interestingly, nitroguanidine is not particularly toxic after oral administration either acutely (LD50 is >2 g/kg in rats) 23,24 or subchronically at doses as high as 1000 mg/kg per d. 25
In the present study, the LD50 of GDNP in tap water solution in female rats as determined by the SSWP procedure is 720 mg/kg. Progressive disorientation and lethargy were consistently observed at doses approaching this LD50. This is similar to the LD50 reported for guanidine nitrate. 7
In the subacute study, the highest dosage used was 500 mg/kg per d. This dose was just below the LD50 and was hypothesized to have a cumulative effect over 14 days that would result in mortality. Animals in this dose group exhibited moderate lethargy during the first 30 minutes after dosing, but none of them lost consciousness. Over the course of the 14 days of dosing, the lethargic response to dosing became less apparent. Similarly, the effect of high-dose GDNP impacted food intake and body weight gain during the first week of dosing. However, this effect was not observed during the second week of dosing, where the animals recovered from the weight loss and gained weight faster than the other dose groups. These observations suggest that rather than GDNP having a cumulative toxic effect, the animals appear to become tolerant to GDNP and less responsive to the adverse effects of the compound.
Despite evidence of compound tolerance in clinical signs, food intake, and weight gain, consistent effects were observed on WBC concentrations, blood chemistries, and organ weights. The WBC counts were increased almost 2-fold above control, due predominantly to the 4-fold increase in neutrophils. Interestingly, although the absolute density of lymphocytes increased by 50%, this increase was overshadowed by the larger increases in other WBCs, predominantly NEU, so that their contribution to the total WBC population as a percentage total decreased.
An increase in serum ALT activity in the range of 2 to 4× or higher in individual or group mean data when compared with concurrent controls should raise concern as an indicator of potential hepatic injury unless a clear alternative explanation is found. 26 A mild elevation of ALT was noted in the 500 mg/kg per d group but there was no supporting histopathological evidence. The mild increase may be due to concurrent hepatic microsomal induction. 26
Rats in this study exhibited some microscopic and hematologic changes similar to rats orally exposed to other explosives, such as 2,4,6-trinitrotoluene (TNT). With TNT, dose-dependent anemia was seen in treated rats with reductions in Hb, HCT, and RBC counts. 27 The 500 mg/kg per d group demonstrated reductions in all the 3 blood values. Additionally, EMH of the spleen was most evident in this group; EMH can occur under conditions of demand such as anemia.
Hepatocellular centrilobular hypertrophy is an enlargement of hepatocellular cytoplasm in response to enzyme induction and is considered an adaptive response to chemical stress. 28 In the 500 mg/kg per d group, 7 of the 10 rats demonstrated a minimal-to-mild response. This lesion was treatment related but was considered adaptive and not adverse. Excessive hypertrophy can result in hepatocellular degeneration and necrosis.
Nephropathy (chronic progressive nephropathy—CPN) is a spontaneous disease of the F344 strain of rat, with minimal lesions of scattered foci of tubular regeneration, occurring as early as 5 months of age. 29 Exacerbation of lesions of CPN represents the most commonly reported minimal expression of nephrotoxicity of the rat. Most nephrotoxicants, at doses less than those that induce overt evidence of nephron or tubular injury, cause or exacerbate injury morphologically the same as CPN. 30 Tubular basophilia, that is, tubular regeneration, was minimally evident in all dosage groups. The 500 mg/kg per d group noted tubular regeneration in the minimal, mild, and moderate ranges with 5 of the 10 rats demonstrating tubular debris and/or tubular epithelial degeneration. The renal lesions in the 500 mg/kg per d appear to be treatment related and may be an exacerbation of underlying early CPN, due to the minimal presence of tubular basophilia in all groups, including the vehicle control.
The current USEPA regulatory values for RDX are based on prostatic inflammation in rodents as a result of chronic oral exposures 31 and hepatocellular carcinomas only in female mice. 32 –34 The RDX has relatively low water solubility (60 mg/L) and is not mutagenic in conventional bacterial strains used for the Ames assay. 19 However, the primary toxicity mechanism of RDX is its induction of epileptiform seizure following oral ingestion. 35 –37 The mechanism of seizure induction has recently been shown to involve blockage of chloride flux through the GABAA receptor ligand-gated channel as a result of RDX binding to the convulsant site of the receptor. 19 The LD50 of RDX in rat is 60 mg/kg, with death always following induction of seizure. 38 The LD50 of GDNP in female rats is 720 mg/kg. Thus, the acute oral toxicity of GDNP is an order of magnitude less than RDX. The water solubility of GDNP is 100 g/L and GDNP is clearly a mutagen.
Daily oral exposure to 500 mg/kg per d of GDNP for 14 days causes weight loss, increased liver and spleen organ weights, and adverse histopathologic events in kidney and spleen including increased EMH in the splenic red pulp and basophilic tubules in the kidneys. These adverse events were not observed in animals receiving lower doses of GDNP. The 14-day subacute no-observable-adverse-effect-level (NOAEL) of RDX in rat is 15 mg/kg per d, with death always following induction of seizure. 38
The lowest-observed-adverse-effect-level from oral exposure to GDNP for 14 days, as determined from this study, is 500 mg/kg per d based on significant adverse event of increased spleen and liver weight ratios and adverse histological alterations in the spleen and kidney. The NOAEL is, therefore, determined to be 152 mg/kg per d where no adverse events were found.
Footnotes
Acknowledgments
The authors would like to acknowledge the support and encouragement of Erik Hangeland of the US Army Research, Development and Engineering Command, Environmental Acquisition and Logistics Sustainment Program. We also thank Wilfred McCain, PhD, Lee Crouse, Matthew A. Bazar, Rebecca Kilby, Theresa Hanna, Robert Sunderland, Major Dawn Fitzhugh, DVM, Pat Beal, Mark Way, Jason Williams, Karen Deaver, Shane Hall, Michael Kefauver, and Martha Thompson for providing technical support. We also thank Desmond Bannon, PhD, DABT, and William Eck, PhD, for their critical review of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the US Army Environmental Quality Technology Program through the US Army Research, Development and Engineering Command, Environmental Acquisition and Logistics Sustainment Program.
