Abstract
Over the last decade, electrolyzed water (EW) produced by salt and tap water has gained importance due to its antimicrobial effects. Regarding to chlorine-based compounds, EW also used in post-harvest safety of food processing and sterilization of surfaces. The latest studies suggested that EW might act as wound healing agent due to anti-infective and cell proliferative properties. In this study, we evaluated acute contact cytotoxicity in L929 mice fibroblast cells and wound healing activity of EWs in vitro. In addition, mutagenic activity was evaluated by Ames test with and without metabolic activation by S9 fraction and the stability profile of freshly prepared EWs has been followed up. According to the results, strong acid (StAEW) and mixed EW (MEW) showed dose-dependent cytotoxicity due to possible high HOCl concentration, while slightly acidic and catholyte EW (CEW) were not cytotoxic even applied directly for 30 sec. Further, StAEW and CEW showed a significant increase in L929 cell migration in scratch assay. Likewise, with/ without metabolic activation, neither of EWs had shown mutagenic profile in TA 98 and TA100 strains of Salmonella typhimurium. Follow-up of ORP (oxidation-reduction potential), pH and FCC (free chlorine concentration) showed that temperature and light were important storage conditions to maintain a stable profile particularly for ORP and FCC, which are the most important indicators for biological activity of EW. According to the present findings, it can be suggested that particularly StAEW, may represent a valuable wound healing agent with an achievable, economical and easy production system when stored under proper conditions.
Introduction
Electrolyzed, or electroactivated water (EW) might be considered as a new approach gained attention in the recent decades due to its promising antimicrobial effects, 1 which has been suggested by numerous reports. 2,3 Its high efficacy in killing bacteria, spores, viruses, molds, and fungus has been proven by studies conducted to date. 4,5 Further studies have been developed to determine the mechanism of inactivation and to define the responsible component of EW for cytotoxicity. 6,7
Different types of EW can be produced in an electrolyzer having two zones where the anode and cathode are separated by the membrane. In addition, each EW type contains different kinds of active ingredients; for instance, highly active reducing agents (eg OH−, H2, HO2 −, O2 −) are produced in the cathode chamber, while some active components (such as Cl2O, ClO2, ClO−, HClO, Cl·, O2·, O3, HO2·, OH·) can be produced in the anode chamber. Relative to these species, the oxidation-reduction potential (ORP) increases (+1000 mV or higher) and the pH of the water decreases (6.0 or lower) in the anodic chamber. For the case of the cathode chamber, there are opposite characteristics such as pH 9 or higher and minus values of ORP. The changes in pH and ORP cause to deviate the conditions for microbial growth (pH 3–10, ORP +900−400 mV). 4 The EW collected from the anodic chamber mostly serves as an antimicrobial agent since it contains chlorine-based compounds. Although HOCl is considered the dominant role in the antimicrobial effect of EW, the participation of reactive oxygen species (ROS) in the disinfection efficacy of EW has also been questioned. 4,8 In this regard, it is concluded that the OH· radical does not contribute to bacterial inactivation and the chlorine compounds are attributed to the mode of action of EW. 9 The majority of studies on EW in literature have been focused on its antimicrobial activity due to mentioned HOCl formation and chlorine compounds as well as ROS production. 3,4 Due to its antimicrobial activity, EW is frequently used in industry in the elimination of bacterial contamination in the food industry 10 –12 as well as medical 13 and dental equipment sterilization process. 14
Furthermore, EW is introduced as a product that can be used to clean certain parts of the human body, especially face and hands and formulated as a useful intravaginal antiseptic gel. 15 In addition to its wide-ranged use of area, several types of EW were suggested to have an effect on tissue epithelialization in oral mucosa in vitro. 16 Several reports have suggested that EW promoted healing of burn wounds 17,18 and full thickness cutaneous-wounds in rat model, as well. 19 Wound healing activity of EW has been generally attributed to effects of free radicals present in EW as an initiator for an early stage of wound healing and inflammation 19 or its effect on preventing infection 20 and stimulating cytokines involved in epithelialization. 18 However, the exact mechanism of EW on wound healing has not been clearly elucidated yet.
In our previous study, 21 we observed that antimicrobial EW did not exhibit irritation in vitro in EpiDerm™ and the Hen’s egg test-chorioallantoic membrane assay, for skin and eye, respectively. Also, as an indicative marker of skin irritation, IL-1α level, was not induced by exposure to produced EW, according to the maintained results. Since quality of care is a critical requirement for wound healing, topical prevention and management of microbial contamination is crucial for damaged area. Therefore, antimicrobial and anti-infective agents have a major part in wound healing are thought to be essential for wound care. 22 According to the previous reports on EW and wound healing 19,20,23 EW types might be a promising option with economic and practicable production system. Hence, in the present study, it was aimed to investigate wound healing activity as a biological function of four different types of EW on L929 mice fibroblast cells in vitro. Also, acute cytotoxicity profile of EW, a possible wound healing agent, has been elucidated in L929 cells in order to assess its direct contact with skin. Since repeated application of products and biomedical devices might represent an important safety concern in the aspect of genotoxicity, effect of EWs on mutagenicity was also studied by Ames test on Salmonella typhimurium TA98 and TA100 strains in vitro with and without metabolic activation. Besides, the stability profile of EW has been determined under different storage conditions within the aspect of future perspective for a possible conventional EW product.
Material and methods
Production and characterization of electrolyzed water
A batch-type handmade electrolyzer was used to produce EW. A detailed description of the system has been given in our previous report. 21 Briefly, the electrolyzer comprising a semipermeable diaphragm separating the rod-shaped anode and the circular cathode made of a steel sheet is connected to the DC power supply. The production of EWs with desired properties is achieved by adjusting the current, salt concentration and electrolysis time. The current density (mA/cm2) was calculated by dividing the applied current by the area of the anode surface. After the reaction, various types of EWs were collected from the split circular electrolyzer having anodic and cathodic zones.
The main factors determining the physicochemical activities of anolyte and catholyte include such factors as the formation of highly active unstable products, along with long-term quasi-stable structures. They are formed in the region of bulk charge near the surface of electrodes, in the form of both free structural complexes and hydrated shells of ions, molecules, radicals and atoms. The presence in the anolyte of a sufficient amount of strong oxidizing agents and free radicals turns it into a solution with strongly pronounced biocidal properties. The catholyte, saturated with reduction agents, acquires a high adsorption-chemical activity, as well as strong washing properties.
The EW collected from the cathodic side (CEW) had a high pH (9.36), negative ORP and no chlorine content. The properties of the EW collected from the anodic side can be changed by adjusting the electrolysis conditions. Slightly acidic EW (SAEW) can be obtained by providing one or both of the long reaction times, high salt concentration and high flow conditions. In our cases, StAEW had a low pH (2.4) and high ORP and FCC as 1140.67 mV and 32.87 mg/L, respectively. On the other hand, slightly acidic EW (e.g. pH ∼ 5.5, ORP 850–900 mV, FCC ∼ 8.7 mg/L) was produced in moderate reaction conditions. The EWs collected from the anodic and cathodic sides were mixed equally and another kind of EW was obtained with the same pH and ORP values as SAEW but with more chlorine content (∼17.8 mg/L). EWs were prepared just before the tests described below.
The characterization of EWs is maintained by measuring pH, ORP and dissolved oxygen concentration (Hach HQ40D). Free chlorine concentration (FCC) and residual chlorine concentration were measured by using the N, N-diethyl-p-phenylenediamine (DPD) colorimetric method. 24 The ·OH radical concentration was analyzed according to terephthalic acid (TA) cumulative protocol. 25 To determine ·OH radical production during electrolysis, 2 mM TA prepared in NaCl solution was in the EW system.
Cell culture and cytotoxicity assay
L929 mouse fibroblast cell line (ATCC) was used for cytotoxicity testing according to ISO 10993-5 standard 26 and maintained in DMEM (Gibco, Fountain Drive, Pailey, UK), supplemented with 10% FBS (Gibco, USA) and 1% streptomycin and penicillin (Gibco, USA) at 37°C in 5% CO2. In order to evaluate the cytotoxicity of EW in case of acute contact with skin, a 30-sec exposure procedure was followed as mentioned previously by Mokudai et al. (2015) with slight modifications. 27 Briefly, 105 cell/mL L929 cells were seeded in a 96-well plate and incubated for 24 h to form a semiconfluent layer. At the end of 24 h, freshly prepared and filtered (0.22 µm filter) EW was serially diluted (12.5–100%; v/v) with DMEM and applied directly on L929 cells and incubated 30 sec. At the end of each exposure, wells were washed with PBS (Phosphate buffer saline) (Gibco, UK). Following, cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. For this purpose, MTT was added to each well at 0.5 mg/mL of concentration and incubated for an additional 2 h at 37°C. After discarding MTT dye, 100 mL of isopropanol was added to the wells and the absorbance of the MTT-formazan was determined at 570 nm by a UV-spectrophotometric plate reader (Thermo Multiskan Spectrum, Finland). Viability was defined as the ratio (expressed as a percentage) of absorbance of the cells exposed to compounds to the cells treated with PBS. As positive control (PC), DMSO 20% (v/v) (Sigma, USA) was used. All measurements were conducted in triplicate, and results were expressed as ± SD% compared to PBS control. The cell viability ratio was determined according to our previous method. 21
Mutagenicity assay (Ames test)
Standard mutagenicity assays in plate incorporation tests were carried out following the method of Maron and Ames (1983). Mutagenicity of EW were assessed by using two of S. typhimurium strains, TA98 and TA100 (MOLTOX, USA), which was used to detect frameshift and base pair mutation, respectively. Four different types of EW were serially diluted with distilled water (25–100%; v/v) and applied as 50 µL/plate for mutagenicity assay. For testing mutagenicity, plates were co-incubated with the bacterial strains and the different concentrations of samples, inverted and placed at 37°C for 48 h in dark and revertant colonies were counted after incubation. In order to assess the impact of EW’s metabolites on mutagenicity and antimutagenicity, similar experiments were also carried out by incubating bacteria and samples with liver S9 (MOLTOX, USA) enzyme fraction. For this purpose, 50 µL distilled water was used as a negative control. 4-Nitro-o-phenylendiamine (20 µg/plate) for TA98 strain and sodium azide (1 µg/plate) for TA100 strain were used as positive control without the liver S9 fraction. In the experiment with liver S9 fraction, 2-aminofluorene (5 µg/plate) was used as positive control for both TA98 ve TA100 strains. After incubation of inverted plates at 37°C for 48 h, revertant colonies were counted. Three plates were used for each concentration tested for both with and without S9 fraction.
DPPH radical scavenging activity
1,1-Diphenyl-2-picrylhydrazine (DPPH) radical scavenging activity was performed in 96-well plates as previously described. 28 Briefly, 50 µl of each EW was mixed with 250 µl of freshly prepared 0.1 mM DPPH solution prepared in methanol. Following, the mixture was incubated in the dark for 50 min at room temperature. The absorbance was measured at 517 nm. Butylated hydroxytoluene (BHT) was used as the reference substance while PBS was used as a negative control. DPPH radical scavenging activity was calculated according to the following formula:
Abscontrol is the absorbance value of the control group, and Abssample is the absorbance of EWs.
Scratch assay
L929 mice fibroblast cells were seeded in 24-well plate and incubated for 24 hours to form a confluent layer. Following day, scratches were formed by 0.5 mm tipped scratcher (SPLScar™) and each well was washed with PBS. Scratch areas were snapped immediately (AxioCam, Leica) for time 0. After this step, 10% (v/v) of EWs diluted with DMEM were applied to each well as triplicates and incubated at 37°C in 5% CO2. Groups treated with the medium were used as the negative control. Scratches formed at time 0 (2.298 ± 0.215 μm2) followed at time 6 and 24 and migration rate was analyzed by ImageJ Software according to the following formula: 29
t 0: At time when scratch was formed
t final: The time after 24 hours of scratching
Stability assessment
Stability was assessed by measuring pH, ORP (mV) and FCC (mg/mL) of freshly prepared EWs. In order to assess stability, ORP and pH of four types of freshly prepared EWs were recorded on day 1. After the measurement, each EW was stored in 4°C or 25°C within amber-colored bottle or colorless bottle and repeated measurements were performed for a week within these conditions. To assess FCC, detectable chlorine containing StAEW, SAEW and MEW have been repetitively measured for 5 days.
Statistical analysis
GraphPad Prism Software version 6.0 (San Diego, California, USA) was used for all the statistical analyses. One-way analysis of variance followed by Dunnet’s correction was used to compare the significant differences between groups and a t-test was used when two groups are compared. p-value <0.05 was considered statistically significant for all analyses.
Results
Characteristic properties of electrolyzed water
pH and ORP are the main characteristic features of EW. The samples of anolyte and catholyte in aqueous media with different levels of mineralization are characterized by sharp changes in pH and ORP relative to the initial values. Thus, in anolyte, pH is reduced, ORP is increased to extreme positive (oxidative) values, while in catholyte, pH is increased and ORP is reduced to extreme negative (reduction) values. In multicomponent chemical and, in that number, biochemical systems, redox pairs pH/ORP are present in complex combinations and permanently changing ratios. The pH values significantly depend on the balance of oxidized and reduced forms and, in their turn, affect the ORP. Figure 1 shows the correlation between pH and ORP with the regression coefficient (R2) of 0.90. Al-Haq et al. 30 obtained a similar R2 value as 0.91 for the produced EW in their reactor. As seen in Figure 1, whereas ORP has not changed at low pH, it is more scattering in higher pH values of EW. Because pH was nearly neutral and the ORP value was 850 mV at the beginnings of the reaction. When the time increased, pH started to go acidic pH and ORP increased. Then, ORP reached its maximum value after a short time as electrolysis started. The reason for the increment is the formation of highly active components such as ozone, atomic oxygen, peroxide compounds, active chlorine compounds, including chlorine dioxide, and short-lived free radicals during electrolysis. However, the main specie in EW is hypochlorite 31 which has 1.482 eV oxidizing potential at 25°C. The reason for the decrease in pH is due to the formation of hypochlorite and Cl2 gas.

The correlation between pH and ORP (temperature 25°C) for EWs collected anodic zone.
Another characteristic behavior was evaluated in terms of dissolved oxygen, as seen in Figure 2. At the anodic side, oxygen molecules are generated during electrolysis, and some of the oxygen in gas form releases. Dissolved oxygen concentration (DOC) increased properly during reaction time, and after a while, the water was saturated with oxygen. It reached almost 20 mg/L at the end of the electrolysis time. Similarly, DOC increased to 21.3 mg/L from 11.6 mg/L during the generation of EW as reported in another paper. 32 The formation of the OH· radical, which is one of the most important ROS species, is also seen in Figure 2. Even the formation of OH· did not show a constant trend and it was detected during electrolysis all the time.

Dissolved oxygen (black line) and OH· concentrations (red line) during EW electrolysis. (at 25°C, 5 g/L of sodium chloride concentration, 15 mA/cm2 of the applied current density).
Cell viability
Cell viability of L929 cells exposed to EWs for 30 sec to assess cytotoxicity by acute contact has been shown in Figure 3. According to the results, a decline in cell viability was observed in all EW groups for a 30-sec exposure in a dose-dependent manner, which was notably in the StAEW group. The highest tested concentrations (100%) of StAEW and MEW, as well as PC, led to a significant decrease in cell viability compared to PBS control. In this test, the highest concentration of StAEW caused the most significant reduction in cell viability among the tested EWs with a 41.69 ± 9.68% viability in the L929 cell line (p < 0.01).

Relative cell viability of L929 cell line exposed to EWs for 30 sec. Statistical difference between PBS control vs groups were *p < 0.05 and **p < 0.01. CEW: catholyte EW; MEW: mixed EW; SAEW: slightly acidic EW; StAEW: strong acid EW; PC: 20% DMSO (v/v).
Mutagenicity of EWs
Mutagenicity of EWs was tested with and without metabolic activation (S9) and the test results on S. typhimurium TA98 and TA100 strains were shown in Table 1. According to the number of revertant colonies of TA98 and TA100 strains, four types of EWs did not exhibit mutagenic activity compared to revertant colony count of control group both with and without metabolic activation (p < 0.05).
Results of mutagenicity assay with EWs in S. typhimurium strains TA98 and TA100 in both with and without S9 activation.

4-nitro-o-phenylendiamine (20 µg/plate) for TA98 strain and sodium azide (1 µg/plate) for TA100 strain were used as positive control (PC) in the experiment without metabolic activation (−S9). In +S9 groups, 2-aminofluorene (5 µg/plate) was used as PC for both TA98 ve TA100 strains. CEW: catholyte EW; MEW: mixed EW; SAEW: Slightly acidic EW StAEW: strong acid EW. *p < 0.05 when compared to the negative control group.
DPPH radical scavenging activity
DPPH radical scavenging assay revealed that EW types did not exhibit radical scavenging activity compared to PBS control (results were not shown). BHT, as positive control, has shown 312 ± 9.26 μg/mL IC50 for DPPH radical scavenging activity.
Scratch assay
According to the results of the scratch assay, StAEW showed the most promising effect on L929 cell proliferation with 88.84 ± 10.37% wound healing ratio within 24 h. CEW and MEW also led to 85.12 ± 11.18% and 74.66 ± 8.81% wound healing activity, respectively. SAEW (60.35 ± 7.59%) did not show significant wound healing activity compared to the medium control group (54.26 ± 3.87%) (p < 0.05). In Figure 4, relative wound healing activity and scratch areas at time 0, 6 and 24 h were given.

In vitro wound healing activity of 10% EWs (v/v) on L929 cell. A) Wound healing was evaluated by measuring the area between the wound margins. Scratch areas at time 0, 6 and 24 h. B) Relative wound healing according to the closed gap between scratch margins within 24 h for each group. Control group was treated with DMEM. Statistical difference between PBS control vs groups were *p < 0.05 and **p < 0.01. CEW: catholyte EW; MEW: mixed EW; SAEW: slightly acidic EW StAEW: strong acid EW.
Stability of EW
According to repetitive measurements of ORP, pH and, FCC, it was seen that all variables are affected by different storage conditions. As seen in Figure 5, ORP of StAEW, SAEW and MEW decreased at the end of day 7 while ORP of CEW increased until day 7, gradually. Moreover, it was clearly observed that ORP of StAEW stored in 4°C at dark slightly changed compared to the other EWs. On the other hand, storing EW in 4°C at light led to a noteworthy change in pH, especially in StAEW and CEW, which were given in Figure 6. Similarly, chlorine concentration recorded for 5 repetitive days showed that FCC is distinctly changed in all groups. Particularly after day 3, FCC of EW stored in 25°C at light clearly decreased in all groups (Figure 7).

ORP values of EWs recorded for 7 repetitive days. CEW: catholyte EW; MEW: mixed EW; SAEW: slightly acidic EW StAEW: strong acid EW; ORP: oxidation-reduction potential.

pH of EWs recorded for 7 repetitive days. CEW: catholyte EW; MEW: mixed EW; SAEW: slightly acidic EW StAEW: strong acid EW.
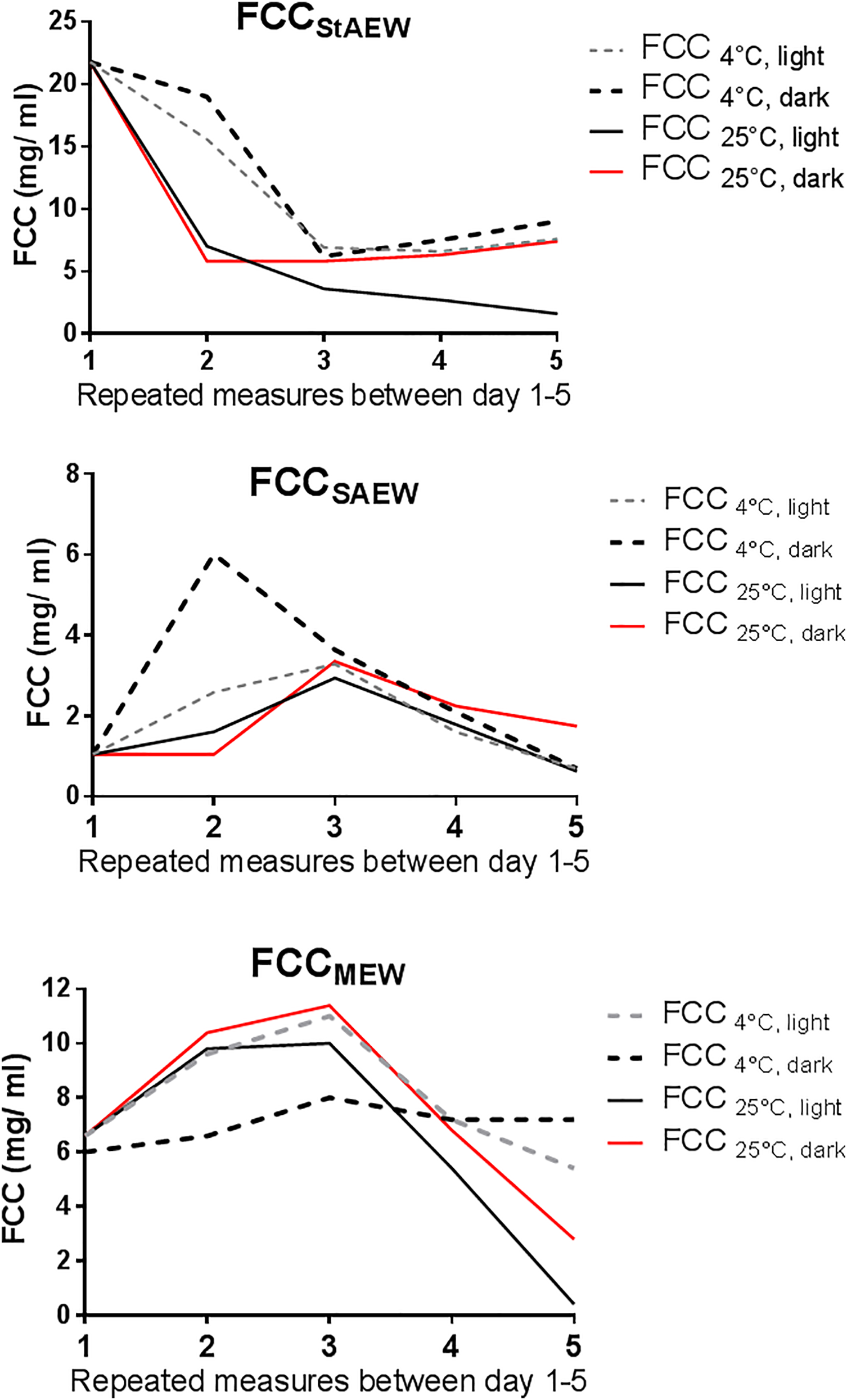
FCC of EWs recorded for 5 repetitive days. FCC: free chlorine concentration; MEW: mixed EW; SAEW: slightly acidic EW StAEW: strong acid EW.
Discussion
Regarding its achievable, economic and fast production setup, EW has become popular in recent years and acidic and alkaline types have been used for sterilization of environment, occupational equipment and food. 12,33 In our previous study, 21 we demonstrated that various types of EW with different pH and ORP have shown antimicrobial activity with a non-cytotoxic profile in L929 cells within the concept of ISO 10993-5 26 and served a non-irritant profile to the skin and mucous membrane. In the present study, we first evaluated the effects of these mentioned EWs on acute contact demonstrated with L929 cells and then wound healing activity in vitro. According to the present results, undiluted SAEW (pH 5.63 ± 0.25; ORP 865 ± 21.79) and CEW (pH 9.36 ± 0.07; ORP < −100) did not exhibit a significant cytotoxicity in L929 cells. However, one fold-diluted and undiluted StAEW (pH 2.43 ± 0.05; ORP 1140.67 ± 12.22) and undiluted MEW (pH 5.49 ± 0.10; ORP 898.33 ± 5.69) have led to a significant decline in cell viability for a 30-sec contact. However, the viability ratio seen with MEW was nearly 80%, thus considered as negligible cytotoxicity. Since StAEW has high FCC (32.87 ± 4.6 mg/L) and HOCl compared to other EWs, cytotoxicity seen with StAEW exposure might be due to a high amount of HOCl produced in the anolyte chamber during electrolysis. Similar to our hypothesis, Mokudai et al. (2015) suggested that HOCl might be the reason for cytotoxicity seen with undiluted StAEW and further, HO· radical production from HOCl that penetrated through cell membrane might be a contributing factor for the observed cytotoxicity with acute contact. 27 Therefore, in the present study, dilution rate-dependent cytotoxicity seen with StAEW and MEW might be due to HOCl content and presumed ROS production in the L929 cell line. On the other hand, HOCl has been found to be effective against wound infections. 34 In the present study, in vitro wound healing activity of 10 fold-diluted StAEW and CEW were noteworthy compared to the control group within 24 hours of exposure. Since HOCl present in EW is a well-documented microbicidal inorganic compound and part of innate immunity in the living system, several studies have evaluated its efficacy on surgical wounds 20,35 and burn-typed wounds. 17,18 As the main active ingredient of “Dakin solution,” 36 HOCl and its contribution to wound healing have been focused on its antimicrobial effect, thus it has been suggested as a preventive agent against infection. 20 On the other hand, in a report on the effect of EW on wound healing 19 it was considered that HOCl did not contribute to the process and free radicals had an effect on inflammation. However, besides its antimicrobial effect, Sakarya et al. (2007) suggested that 32 fold-diluted HOCl solution increased fibroblast and keratinocyte cell migration in vitro. Therefore, a diluted form of HOCl was suggested to promote wound healing in the proliferative phase 37 which may be responsible for the wound healing activity seen with StAEW in the present study. On the other hand, CEW showed significant activity on cell migration, which might be due to its antioxidant activity 38 or its reducing ability on intracellular ROS production. 39 In our study, we evaluated antioxidant activity of four types of EW by DPPH radical scavenging potential. However, as similarly suggested by Hamasaki et al. (2017) produced EWs, did not exhibit DPPH radical scavenging activity. 39 In contrast, it was reported that presence of acidic EW increased DPPH radical scavenger activity of mulberry leaf extract compared to the same extract prepared with tap water. 40 However, it was also suggested that the decrease in intracellular ROS by EW might be due to the activation of intracellular antioxidant systems via indirect ROS scavenging mechanism in cells. 39 Therefore, this indirect antioxidant mechanism might be involved in wound healing activity seen in the present study. In another study Lee et al. (2006) suggested that acidic EW showed antioxidant activity through xanthine–xanthine oxidase superoxide scavenging system, and increased antioxidant activity of ascorbic acid by inhibiting oxidation of ascorbic acid. 41 Hence, indirect antioxidant system might be evaluated in order to assess EWs antioxidant potential and related wound healing mechanisms in further studies.
On the other hand, chlorination of surface water might lead to formation of mutagenic or carcinogenic products derived from the reaction of chlorine with the organic compound. Therefore, it was suggested that strong disinfectants such as sodium hypochlorite, chlorine dioxide and peracetic acid might exhibit weak to moderate mutagenic effect. 42 Furthermore, these chlorinated-mutagenic compounds might have a carcinogenic effect or irritate mucous membranes in case of exposure according to a previous report. 43 Therefore, mutagenic or carcinogenic potential of EW is a critical safety concern for a disinfectant and wound healing agent designed for repeated use. Similar to previous studies, 44,45 produced EWs did not exhibit mutagenic activity on Salmonella typhimurium TA98 and T100 strains with and without metabolic activation and might be considered as safe in terms of mutagenic potential.
In addition, storage and production conditions of EWs have known as important factors on inactivation of efficacy of EWs. Recent reports have been suggested that stability of physical properties of EW might be affected by open or close storage, electrolysis duration, 46 temperature and light exposure as well. 47 According to our findings, it was seen that physical properties such as pH, ORP and FCC are affected by different storage conditions. ORP of StAEW, SAEW and MEW decreased at the end of the last day while ORP of CEW increased until day 7, gradually. In addition, it was clearly observed that ORP of StAEW stored in 4°C at dark was slightly changed compared to the other EWs and storage conditions, which might represent a proper condition for storage of StAEW. Further, chlorine concentration is distinctly changed in all groups, possibly due to the opening of the same bottle for each measurement and storing at room temperature. Since ORP and FCC are important parameters for the biological activity of EW, temperature and airtight packaging might represent important conditions for the storage of freshly prepared EW to maintain stable chlorine and ORP level.
Conclusion
EW and their diluted forms that are stored airtight containers at the dark and optimum temperature can be used as an environmentally friendly wound healing agent in addition to its well-known disinfectant action with an economical generation setup consisting of salt and tap water. Present findings might represent valuable data for further in vivo studies for developing a potential wound healing agent, especially for surgical wounds, with a non-mutagenic profile.
Footnotes
Author contributions
Conceptualization: HS, RR, OD; Data curation: RR, TK, HS, MC; Formal analysis: RR, OD, MC, HS; Investigation: RR, OD, HS; Methodology: OD, HS; Project administration: HS, OD; Resources: OD, HS; Software: RR, TK; Supervision: HS, AD, AA; Writing—original draft: RR, OD; Writing—review & editing: RR, OD, HS, MC, AD, TK, AA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by TÜBİTAK (Turkish Research and Scientific Council) 1002 program with a Grant No. 116Z169. The authors also thank to Gülenay Alyurt for her considerate effort on EW project.


